Home » Uncategorized (Page 111)
Category Archives: Uncategorized
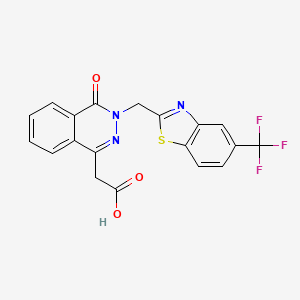
Zopolrestat




http://pubs.acs.org/doi/abs/10.1021/jm00105a018
……………………………………
Mylari, Banavara L.; Zembrowski, William J.; Beyer, Thomas A.; Aldinger, Charles E.; Siegel, Todd W.
Journal of Medicinal Chemistry, 1992 , vol. 35, 12 p. 2155 – 2162
………………………………..
Mylari; Beyer; Scott; Aldinger; Dee; Siegel; Zembrowski
Journal of Medicinal Chemistry, 1992 , vol. 35, 3 p. 457 – 465
…………………………….
Drugs Fut 1995, 20(1): 33
J Label Compd Radiopharm 1991, 29(2): 143
|
3-19-1992
|
HETEROCYCLIC OXOPHTHALAZINYL ACETIC ACIDS
|
|
|
3-6-1992
|
3-(5-TRIFLUOROMETHYLBENZOTHIAZOL-2-YLMETHYL)-4-OXO-3H-PHYTHALAZIN-1-YLACETIC ACID MONOHYDRATE
|
|
|
7-4-1990
|
Heterocyclic oxophthalazinyl acetic acids
|
|
3-24-2006
|
Medical devices to treat or inhibit restenosis
|
|
|
12-30-2004
|
N-[(SUBSTITUTED FIVE-MEMBERED DI- OR TRIAZA DIUNSATURATED RING)CARBONYL]GUANIDINE DERIVATIVES FOR THE TREATMENT OF ISCHEMIA
|
|
|
10-7-2004
|
COMBINATION OF AN ALDOSE REDUCTASE INHIBITOR AND A GLYCOGEN PHOSPHORYLASE INHIBITOR COMBINATION OF AN ALDOSE REDUCTASE INHIBITOR AND A GLYCOGEN PHOSPHORYLASE INHIBITOR
|
|
|
9-30-2004
|
Aldose reductase inhibition in preventing or reversing diabetic cardiomyopathy
|
|
|
5-27-2004
|
SUBSTITUTED FUSED HETEROCYCLIC COMPOUNDS
|
|
|
4-15-2004
|
Compounds for treating and preventing diabetic complications
|
|
|
3-32-2004
|
IMPROVED MUTANTS OF (2,5-DKG) REDUCTASE A
|
|
|
12-18-2003
|
Pharmaceutical composition for use in treatment of diabetes
|
|
|
11-14-2003
|
Salts of zopolrestat
|
|
|
4-18-2002
|
Use of an aldose reductase inhibitor for reducing non-cardiac tissue damage
|
Кальцитонин, Calcitonin

Molecular formula of calcitonin is C145H241N43O49S2
• Molecular weight is 3434.8 g/mol
| Calcitonin-related polypeptide alpha | |||
|---|---|---|---|
 NMR solution structure of salmon calcitonin in SDS micelles.[1] |
The structural formula
Calcitonin (also known as thyrocalcitonin) is a 32-amino acid linear polypeptide hormone that is produced in humansprimarily by the parafollicular cells (also known as C-cells) of the thyroid, and in many other animals in the ultimobranchial body.[2] It acts to reduce blood calcium (Ca2+), opposing the effects of parathyroid hormone (PTH).[3]
Calcitonin has been found in fish, reptiles, birds, and mammals. Its importance in humans has not been as well established as its importance in other animals, as its function is usually not significant in the regulation of normal calcium homeostasis.[4] It belongs to the calcitonin-like protein family.
UV – range
IR – spectrum
Links
-
UV and IR Spectra. H.-W. Dibbern, R.M. Muller, E. Wirbitzki, 2002 ECV
-
NIST/EPA/NIH Mass Spectral Library 2008
-
Handbook of Organic Compounds. NIR, IR, Raman, and UV-Vis Spectra Featuring Polymers and Surfactants, Jr., Jerry Workman. Academic Press, 2000.
-
Handbook of ultraviolet and visible absorption spectra of organic compounds, K. Hirayama. Plenum Press Data Division, 1967.
| Calcitonin-related polypeptide alpha | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
 NMR solution structure of salmon calcitonin in SDS micelles.[1] |
|||||||||||
|
|||||||||||
| Identifiers | |||||||||||
| Symbols | CALCA ; CALC1; CGRP; CGRP-I; CGRP1; CT; KC | ||||||||||
| External IDs | OMIM: 114130 MGI: 2151253HomoloGene: 88401 ChEMBL: 5293GeneCards: CALCA Gene | ||||||||||
|
|||||||||||
| RNA expression pattern | |||||||||||
 |
|||||||||||
 |
|||||||||||
 |
|||||||||||
| More reference expression data | |||||||||||
| Orthologs | |||||||||||
| Species | Human | Mouse | |||||||||
| Entrez | 796 | 12310 | |||||||||
| Ensembl | ENSG00000110680 | ENSMUSG00000030669 | |||||||||
| UniProt | P01258 | P70160 | |||||||||
| RefSeq (mRNA) | NM_001033952 | NM_001033954 | |||||||||
| RefSeq (protein) | NP_001029124 | NP_001029126 | |||||||||
| Location (UCSC) | Chr 11: 14.99 – 14.99 Mb |
Chr 7: 114.63 – 114.64 Mb |
|||||||||
| PubMedsearch | [1] | [2] | |||||||||
Biosynthesis and regulation
Calcitonin is formed by the proteolytic cleavage of a larger prepropeptide, which is the product of the CALC1 gene (CALCA). The CALC1 gene belongs to a superfamily of related protein hormone precursors including islet amyloid precursor protein, calcitonin gene-related peptide, and the precursor of adrenomedullin.
Secretion of calcitonin is stimulated by:
-
- an increase in serum [Ca2+][5]
- gastrin and pentagastrin.[6]
Effects
The hormone participates in calcium (Ca2+) and phosphorus metabolism. In many ways, calcitonin counteracts parathyroid hormone (PTH).
More specifically, calcitonin lowers blood Ca2+ levels in three ways:
-
- Inhibits Ca2+ absorption by the intestines[7]
- Inhibits osteoclast activity in bones[8]
- Stimulates osteoblastic activity in bones. [8]
- Inhibits renal tubular cell reabsorption of Ca2+ allowing it to be excreted in the urine[9][10]
However, effects of calcitonin that mirror those of PTH include the following:
In its skeleton-preserving actions, calcitonin protects against calcium loss from skeleton during periods of calcium mobilization, such as pregnancy and, especially, lactation.
Other effects are in preventing postprandial hypercalcemia resulting from absorption of Ca2+. Also, calcitonin inhibits food intake in rats and monkeys, and may have CNS action involving the regulation of feeding and appetite.
Receptor
The calcitonin receptor, found on osteoclasts,[12] and in kidney and regions of the brain, is a G protein-coupled receptor, which is coupled by Gs to adenylate cyclase and thereby to the generation of cAMP in target cells. It may also affect the ovaries in women and the testes in men.
Discovery
Calcitonin was purified in 1962 by Copp and Cheney.[13] While it was initially considered a secretion of the parathyroid glands, it was later identified as the secretion of the C-cellsof the thyroid gland.[14]
Pharmacology
Salmon calcitonin is used for the treatment of:
It has been investigated as a possible non-operative treatment for spinal stenosis.[16]
The following information is from the UK Electronic Medicines Compendium[17]
General characteristics of the active substance
Salmon calcitonin is rapidly absorbed and eliminated. Peak plasma concentrations are attained within the first hour of administration.
Animal studies have shown that calcitonin is primarily metabolised via proteolysis in the kidney following parenteral administration. The metabolites lack the specific biological activity of calcitonin. Bioavailability following subcutaneous and intramuscular injection in humans is high and similar for the two routes of administration (71% and 66%, respectively).
Calcitonin has short absorption and elimination half-lives of 10–15 minutes and 50–80 minutes, respectively. Salmon calcitonin is primarily and almost exclusively degraded in the kidneys, forming pharmacologically inactive fragments of the molecule. Therefore, the metabolic clearance is much lower in patients with end-stage renal failure than in healthy subjects. However, the clinical relevance of this finding is not known. Plasma protein binding is 30% to 40%.
Characteristics in patients
There is a relationship between the subcutaneous dose of calcitonin and peak plasma concentrations. Following parenteral administration of 100 IU calcitonin, peak plasma concentration lies between about 200 and 400 pg/ml. Higher blood levels may be associated with increased incidence of nausea, vomiting, and secretory diarrhea.
Preclinical safety data
Conventional long-term toxicity, reproduction, mutagenicity, and carcinogenicity studies have been performed in laboratory animals. Salmon calcitonin is devoid of embryotoxic, teratogenic, and mutagenic potential.
An increased incidence of pituitary adenomas has been reported in rats given synthetic salmon calcitonin for 1 year. This is considered a species-specific effect and of no clinical relevance. Salmon calcitonin does not cross the placental barrier.
In lactating animals given calcitonin, suppression of milk production has been observed. Calcitonin is secreted into the milk.
Pharmaceutical manufacture
Calcitonin was extracted from the ultimobranchial glands (thyroid-like glands) of fish, particularly salmon. Salmon calcitonin resembles human calcitonin, but is more active. At present, it is produced either by recombinant DNA technology or by chemical peptide synthesis. The pharmacological properties of the synthetic and recombinant peptides have been demonstrated to be qualitatively and quantitatively equivalent.[17]
Uses of calcitonin
Treatments
Calcitonin can be used therapeutically for the treatment of hypercalcemia or osteoporosis.
Oral calcitonin may have a chondroprotective role in osteoarthritis (OA), according to data in rats presented in December, 2005, at the 10th World Congress of the Osteoarthritis Research Society International (OARSI) in Boston, Massachusetts. Although calcitonin is a known antiresorptive agent, its disease-modifying effects on chondrocytes and cartilage metabolisms have not been well established until now.
This new study, however, may help to explain how calcitonin affects osteoarthritis. “Calcitonin acts both directly on osteoclasts, resulting in inhibition of bone resorption and following attenuation of subchondral bone turnover, and directly on chondrocytes, attenuating cartilage degradation and stimulating cartilage formation,” says researcher Morten Karsdal, MSC, PhD, of the department of pharmacology at Nordic Bioscience in Herlev, Denmark. “Therefore, calcitonin may be a future efficacious drug for OA.”[18]
Subcutaneous injections of calcitonin in patients suffering from mania resulted in significant decreases in irritability, euphoria and hyperactivity and hence calcitonin holds promise for treating bipolar disorder.[19] However no further work on this potential application of calcitonin has been reported.
Diagnostics
It may be used diagnostically as a tumor marker for medullary thyroid cancer, in which high calcitonin levels may be present and elevated levels after surgery may indicate recurrence. It may even be used on biopsy samples from suspicious lesions (e.g., lymph nodes that are swollen) to establish whether they are metastasis of the original cancer.
Cutoffs for calcitonin to distinguish cases with medullary thyroid cancer have been suggested to be as follows, with a higher value increasing the suspicion of medullary thyroid cancer:[20]
- females: 5 ng/L or pg/mL
- males: 12 ng/L or pg/mL
- children under 6 months of age: 40 ng/L or pg/mL
- children between 6 months and 3 years of age: 15 ng/L or pg/mL
When over 3 years of age, adult cutoffs may be used
Increased levels of calcitonin have also been reported for various other conditions. They include: C-cell hyperplasia, Nonthyroidal oat cell carcinoma, Nonthyroidal small cell carcinoma and other nonthyroidal malignancies, acute and chronic renal failure, hypercalcemia, hypergastrinemia and other gastrointestinal disorders, and pulmonary disease.[21]
Structure
Calcitonin is a polypeptide hormone of 32 amino acids, with a molecular weight of 3454.93 daltons. Its structure comprises a single alpha helix.[1] Alternative splicing of the gene coding for calcitonin produces a distantly related peptide of 37 amino acids, called calcitonin gene-related peptide (CGRP), beta type.[22]
The following are the amino acid sequences of salmon and human calcitonin:[23]
- salmon:
Cys-Ser-Asn-Leu-Ser-Thr-Cys-Val-Leu-Gly-Lys-Leu-Ser-Gln-Glu-Leu-His-Lys-Leu-Gln-Thr-Tyr-Pro-Arg-Thr-Asn-Thr-Gly-Ser-Gly-Thr-Pro
- human:
Cys-Gly-Asn-Leu-Ser-Thr-Cys-Met-Leu-Gly-Thr-Tyr-Thr-Gln-Asp-Phe-Asn-Lys-Phe-His-Thr-Phe-Pro-Gln-Thr-Ala-Ile-Gly-Val-Gly-Ala-Pro
Compared to salmon calcitonin, human calcitonin differs at 16 residues.
Description: Cellular and molecular coordination of tissues which secrete chemical compounds to regulate growth, reproduction, metabolism, and ion homeostasis.
References
- ^ Jump up to:a b PDB 2glhAndreotti G, Méndez BL, Amodeo P, Morelli MA, Nakamuta H, Motta A (August 2006). “Structural determinants of salmon calcitonin bioactivity: the role of the Leu-based amphipathic alpha-helix”. J. Biol. Chem. 281 (34): 24193–203.doi:10.1074/jbc.M603528200. PMID 16766525.
- Jump up^ Costoff A. “Sect. 5, Ch. 6: Anatomy, Structure, and Synthesis of Calcitonin (CT)”.Endocrinology: hormonal control of calcium and phosphate. Medical College of Georgia. Retrieved 2008-08-07.
- Boron WF, Boulpaep EL (2004). “Endocrine system chapter”. Medical Physiology: A Cellular And Molecular Approach. Elsevier/Saunders. ISBN 1-4160-2328-3.
- Jump up^ Costoff A. “Sect. 5, Ch. 6: Biological Actions of CT”. Medical College of Georgia. Retrieved 2008-08-07.
- Costanzo, Linda S. (2007). BRS Physiology. Lippincott, Williams, & Wilkins. p. 263.ISBN 978-0-7817-7311-9.
- Jump up^ Erdogan MF, Gursoy A, Kulaksizoglu M (October 2006). “Long-term effects of elevated gastrin levels on calcitonin secretion”. J Endocrinol Invest. 29 (9): 771–775.PMID 17114906.
- Costoff A. “Sect. 5, Ch. 6: Effects of CT on the Small Intestine”. Medical College of Georgia. Retrieved 2008-08-07.
- Costoff A. “Sect. 5, Ch. 6: Effects of CT on Bone”. Medical College of Georgia. Retrieved 2008-08-07.
- Jump up^ Potts, John; Jüppner, Harald (2008). “Chapter 353. Disorders of the Parathyroid Gland and Calcium Homeostasis”. In Dan L. Longo, Dennis L. Kasper, J. Larry Jameson, Anthony S. Fauci, Stephen L. Hauser, and Joseph Loscalzo. Harrison’s Principles of Internal Medicine (18 ed.). McGraw-Hill.
- Rhoades, Rodney (2009). Medical Physiology: Principles for Clinical Medicine. Philadelphia: Lippincott Williams & Wilkins. ISBN 978-0-7817-6852-8.
- Jump up^ Carney SL (1997). “Calcitonin and human renal calcium and electrolyte transport”.Miner Electrolyte Metab 23 (1): 43–7. PMID 9058369.
- Jump up^ Nicholson GC, Moseley JM, Sexton PM, et al (1986). “Abundant calcitonin receptors in isolated rat osteoclasts. Biochemical and autoradiographic characterization”. J Clin Invest 78 (2): 355–60. doi:10.1172/JCI112584. PMC 423551. PMID 3016026.
- Jump up^ Copp DH, Cheney B (January 1962). “Calcitonin-a hormone from the parathyroid which lowers the calcium-level of the blood”. Nature 193 (4813): 381–2.doi:10.1038/193381a0. PMID 13881213.
- Jump up^ Hirsch PF, Gauthier GF, Munson PL (August 1963). “Thyroid hypocalcemic principle and recurrent laryngeal nerve injury as factors affecting the response to parathyroidectomy in rats”. Endocrinology 73 (2): 244–252. doi:10.1210/endo-73-2-244.PMID 14076205.
- Jump up^ Wall GC, Heyneman CA (April 1999). “Calcitonin in phantom limb pain”. Ann Pharmacother 33 (4): 499–501. doi:10.1345/aph.18204. PMID 10332543.
- Jump up^ Tran de QH, Duong S, Finlayson RJ (July 2010). “Lumbar spinal stenosis: a brief review of the nonsurgical management”. Can J Anaesth 57 (7): 694–703. doi:10.1007/s12630-010-9315-3. PMID 20428988.
- ^ Jump up to:a b “Electronic Medicines Compendium”. Retrieved 2008-08-07.
- Jump up^ Kleinman DM (2006-01-04). “Oral Calcitonin May Delay Onset of Joint Disease and Relieve Pain of OA”. Musculoskeletal Report. Musculoskeletal Report, LLC. Retrieved 2008-08-07.
- Jump up^ Vik A, Yatham LN (March 1998). “Calcitonin and bipolar disorder: a hypothesis revisited”. J Psychiatry Neurosci 23 (2): 109–17. PMC 1188909. PMID 9549251.
- Jump up^ Basuyau, J. -P.; Mallet, E.; Leroy, M.; Brunelle, P. (2004). “Reference Intervals for Serum Calcitonin in Men, Women, and Children”. Clinical Chemistry 50 (10): 1828–1830.doi:10.1373/clinchem.2003.026963. PMID 15388660. edit
- Jump up^ Burtis CA, Ashwood ER, Bruns DE. Tietz Textbook of Clinical Chemistry and Molecular Diagnostics, 5th edition. Elsevier Saunders. p. 1774. ISBN 978-1-4160-6164-9.
- Jump up^ “calcitonin domain annotation”. SMART (a Simple Modular Architecture Research Tool). embl-heidelberg.de. Retrieved 2009-02-22.
- Jump up^ http://www.newworldencyclopedia.org/entry/Calcitonin
Further reading
- MacIntyre I, Alevizaki M, Bevis PJ, Zaidi M (1987). “Calcitonin and the peptides from the calcitonin gene”. Clin. Orthop. Relat. Res. &na; (217): 45–55. doi:10.1097/00003086-198704000-00007. PMID 3549095.
- Di Angelantonio S, Giniatullin R, Costa V, et al. (2004). “Modulation of neuronal nicotinic receptor function by the neuropeptides CGRP and substance P on autonomic nerve cells”.Br. J. Pharmacol. 139 (6): 1061–73. doi:10.1038/sj.bjp.0705337. PMC 1573932.PMID 12871824.
- Findlay DM, Sexton PM (2005). “Calcitonin”. Growth Factors 22 (4): 217–24.doi:10.1080/08977190410001728033. PMID 15621724.
- Sponholz C, Sakr Y, Reinhart K, Brunkhorst F (2007). “Diagnostic value and prognostic implications of serum procalcitonin after cardiac surgery: a systematic review of the literature”. Critical care (London, England) 10 (5): R145. doi:10.1186/cc5067.PMC 1751067. PMID 17038199.
- Schneider HG, Lam QT (2007). “Procalcitonin for the clinical laboratory: a review”. Pathology39 (4): 383–90. doi:10.1080/00313020701444564. PMID 17676478.
- Grani, G; Nesca, A; Del Sordo, M; Calvanese, A; Carbotta, G; Bianchini, M; Fumarola, A (Jun 2012). “Interpretation of serum calcitonin in patients with chronic autoimmune thyroiditis.”. Endocrine-related cancer 19 (3): 345–9. doi:10.1530/ERC-12-0013.PMID 22399011.
External links
- The Calcitonin Protein
- Calcitonin at the US National Library of Medicine Medical Subject Headings (MeSH)

 cas 57014-02-5
cas 57014-02-5Directing Venom To Fight Cancer ACS Meeting News: Encapsulated venom peptide can skip healthy cells


Venom from scorpions or honeybees sounds like it wouldn’t do a person much good. But by directing a modified component just to tumors, researchers might leverage it into a drug.
Peptides in some venoms bind to cancer cells and block tumor growth and spread. But they have not yet been developed successfully as anticancer agents because they attack healthy cells too.
Bioengineer Dipanjan Pan and coworkers at the University of Illinois, Urbana-Champaign, are now using polymeric nanoparticles to deliver venom toxin directly to cancer cells.
read at
http://cen.acs.org/articles/92/i33/Directing-Venom-Fight-Cancer.html


WHO issues draft proposal for biosimilar naming
DRUG REGULATORY AFFAIRS INTERNATIONAL

A long debate is ongoing about biosimilar naming around the world. Although EU accepted the same INN system years ago, with the latest developments around the world, biosimilar naming uncertainty is still ongoing.
View original post 74 more words
Neuroprotective effects of Asiaticoside – a Saponin of Centella asiatica
PUBLIC RELEASE DATE:
10-Aug-2014
In the central nervous system, Asiaticoside has been shown to attenuate in vitro neuronal damage caused by exposure to β-amyloid. However, its potential neuroprotective properties in glutamate-induced excitotoxicity have not been fully studied. Researchers from Fourth Military Medical University of Chinese PLA, China reported that pretreatment with Asiaticoside decreased neuronal cell loss in a concentration-dependent manner and restored changes in expression of apoptotic-related proteins Bcl-2 and Bax. Asiaticoside pretreatment also attenuated the upregulation of NR2B expression, a subunit of N-methyl-D-aspartate receptors, but did not affect expression of NR2A subunits. Additionally, in cultured neurons, Asiaticoside significantly inhibited Ca2+ influx induced by N-methyl-D-aspartate. Their results provide a new insight into the neuroprotective effects of Asiaticoside. The relevant study has been published in the Neural Regeneration Research (Vol. 9, No. 13, 2014).
View original post 85 more words
Lipid Metabolism
Leaders in Pharmaceutical Business Intelligence Group, LLC, Doing Business As LPBI Group, Newton, MA
Lipid metabolism
Larry H. Bernstein, MD, FCAP, Reporter and Curator
Leaders in Pharmaceutical Intelligence
http://pharmaceuticalintelligence.com/8-10-2014/Lipid_metabolism
This is fourth of a series of articles, lipid metabolism, that began with signaling and signaling pathways. These discussion lay the groundwork to proceed in later discussions that will take on a somewhat different approach. These are critical to develop a more complete point of view of life processes. I have indicated that many of the protein-protein interactions or protein-membrane interactions and associated regulatory features have been referred to previously, but the focus of the discussion or points made were different. The role of lipids in circulating plasma proteins as biomarkers for coronary vascular disease can be traced to the early work of Frederickson and the classification of lipid disorders. The very critical role of lipids in membrane structure in health and disease has had much less attention, despite the enormous importance, especially in…
View original post 10,510 more words
Macrocycles in new drug discovery

Summary | Full Text | PDF (3354 KB) | PDF Plus (3440 KB) | Add to Favorites | Related
http://www.future-science.com/doi/full/10.4155/fmc.12.93?src=recsys
The use of drug-like macrocycles is emerging as an exciting area of medicinal chemistry, with several recent examples highlighting the favorable changes in biological and physicochemical properties that macrocyclization can afford. Natural product macrocycles and their synthetic derivatives have long been clinically useful and attention is now being focused on the wider use of macrocyclic scaffolds in medicinal chemistry in the search for new drugs for increasingly challenging targets. With the increasing awareness of concepts of drug-likeness and the dangers of ‘molecular obesity’, functionalized macrocyclic scaffolds could provide a way to generate ligand-efficient molecules with enhanced properties. In this review we will separately discuss the effects of macrocyclization upon potency, selectivity and physicochemical properties, concentrating on recent case histories in oncology drug discovery. Additionally, we will highlight selected advances in the synthesis of macrocycles and provide an outlook on the future use of macrocyclic scaffolds in medicinal chemistry.
Carbohydrate Metabolism
Leaders in Pharmaceutical Business Intelligence Group, LLC, Doing Business As LPBI Group, Newton, MA
Carbohydrate Metabolism
Larry H. Bernstein, MD, FCAP, Reporter and Curator
Leaders in Pharmaceutical Intelligence
http://pharmaceuticalintelligence.com/8-9-2014/Carbohydrate_Metabolism
This is the third portion of the discussion in a series of articles that began with signaling and signaling pathways. There are fine features on the functioning of enzymes and proteins, on sequential changes in a chain reaction, and on conformational changes that we shall return to. These are critical to developing a more complete understanding of life processes. I have indicated that many of the protein-protein interactions or protein-membrane interactions and associated regulatory features have been referred to previously, but the focus of the discussion or points made were different. Even though I considered placing this after the discussion of proteins and how they play out their essential role, I needed to lay out the scope of metabolic reactions and pathways, and their complementary changes. These may not appear to be adaptive, if the…
View original post 6,261 more words
Valdecoxib


Valdecoxib is a non-steroidal anti-inflammatory drug (NSAID) used in the treatment of osteoarthritis, rheumatoid arthritis, and painfulmenstruation and menstrual symptoms. It is a cyclooxygenase-2 selective inhibitor.
Valdecoxib was manufactured and marketed under the brand name Bextra by G. D. Searle & Company. It was approved by the United States Food and Drug Administration on November 20, 2001,[1] and was available by prescription in tablet form until 2005, when it was removed from the market due to concerns about possible increased risk of heart attack and stroke. The prodrugparecoxib is available in many countries.
Uses until 2005
In the United States, the Food and Drug Administration (FDA) approved valdecoxib for the treatment of osteoarthritis, adultrheumatoid arthritis, and primary dysmenorrhea.[2]
Valdecoxib was also used off-label for controlling acute pain and various types of surgical pain.[2]
Side-effects and withdrawal from market
On April 7, 2005, Pfizer withdrew Bextra from the U.S. market on recommendation by the FDA, citing an increased risk of heart attackand stroke and also the risk of a serious, sometimes fatal, skin reaction. This was a result of recent attention to prescription NSAIDs, such as Merck’s Vioxx. Other reported side-effects were angina and Stevens–Johnson syndrome.
Pfizer first acknowledged cardiovascular risks associated with Bextra in October 2004. The American Heart Association soon after was presented with a report indicating patients using Bextra while recovering from heart surgery were 2.19 times more likely to suffer a stroke or heart attack than those taking placebos.
In a large study published in JAMA 2006, valdecoxib appeared less adverse for renal (kidney) disease and heart arrhythmia compared to Vioxx, however elevated renal risks were slightly suggested.[3]
2009 settlement for off-label uses promotions
|
|
An editor has expressed a concern that this section lends undue weight to certain ideas relative to the article as a whole. Please help to discuss and resolve the dispute before removing this message. (December 2012) |
On September 2, 2009, the United States Department of Justice fined Pfizer $2.3 billion after one of its subsidiaries, Pharmacia & UpJohn Company, pled guilty to marketing four drugs including Bextra “with the intent to defraud or mislead.”[4] Pharmacia & UpJohn admitted to criminal conduct in the promotion of Bextra, and agreed to pay the largest criminal fine ever imposed in the United States for any matter, $1.195 billion.[5] A former Pfizer district sales manager was indicted and sentenced to home confinement for destroying documents regarding the illegal promotion of Bextra.[6][7] In addition, a Regional Manager pled guilty to distribution of a mis-branded product, and was fined $75,000 and twenty-four months on probation.[8]
The remaining $1 billion of the fine was paid to resolve allegations under the civil False Claims Act case and is the largest civil fraud settlement against a pharmaceutical company. Six whistle-blowers were awarded more than $102 million for their role in the investigation.[9] Former Pfizer sales representative John Kopchinski acted as a qui tam relator and filed a complaint in 2004 outlining the illegal conduct in the marketing of Bextra.[10] Kopchinski was awarded $51.5 million for his role in the case because the improper marketing of Bextra was the largest piece of the settlement at $1.8 billion.[11]
Assay of Valdecoxib[13]
Several HPLC-UV methods have been reported for valdecoxib estimation in biological samples like human urine,[14] plasma,.[15][16] Valdecoxib has analytical methods for bioequivalence studies,[17][18] metabolite determination,[19][20][21] and estimation of formulation,[22] HPTLC method for simultaneous estimation in tablet dosage form.[23]
Brief background information
| Salt | ATC | Formula | MM | CAS |
|---|---|---|---|---|
| – | M01AH03 | C 16 H 14 N 2 O 3 S | 314.37 g / mol | 181695-72-7 |
| Systematic (IUPAC) name | |
|---|---|
| 4-(5-methyl-3-phenylisoxazol-4-yl)benzenesulfonamide | |
| Clinical data | |
| Trade names | Bextra |
| Pregnancy cat. | C (AU) May cause premature closure of the ductus arteriosus |
| Legal status | Prescription Only (S4) (AU)Withdrawn in U.S., EU, Canada& parts of Asia |
| Routes | Oral |
| Pharmacokinetic data | |
| Bioavailability | 83% |
| Protein binding | 98% |
| Metabolism | Hepatic (CYP3A4 and 2C9involved) |
| Half-life | 8 to 11 hours |
| Excretion | Renal |
| Identifiers | |
| CAS number | 181695-72-7 |
| ATC code | M01AH03 |
| PubChem | CID 119607 |
| DrugBank | DB00580 |
| ChemSpider | 106796 |
| UNII | 2919279Q3W |
| KEGG | D02709 |
| ChEBI | CHEBI:63634 |
| ChEMBL | CHEMBL865 |
| Chemical data | |
| Formula | C16H14N2O3S |
| Mol. mass | 314.364 g/mol |
Using
-
anti-inflammatory
-
antirheumatic
-
COX-2 inhibitor
Classes of substances
-
Benzenesulfonamide (s -imidy), as well as their derivatives
-
Isoxazoles
-
Synthesis pathway
| Synthesis a) |
|---|
 |
Synthesis
Source:[12]



Deoxybenzoin (I) is converted to the corresponding oxime (II) by treatment with NH2OH稨Cl under basic conditions either with sodium acetate in aqueous ethanol or in toluene in presence of potassium hydroxide in absolute ethanol. Deprotonation of the oxime under nitrogen with 2eq of butyllithium in THF followed by cyclization in ethyl acetate or acetic anhydride affords isoxazoline (III). Finally, treatment of (III) with cold chlorosulfonic acid followed by reaction of the intermediate sulfonyl chloride with aqueous ammonia affords the desired product.
J Med Chem2000,43,(5):775
Trade Names
| Country | Trade name | Manufacturer |
|---|---|---|
| Germany | Bextra | Pharmacia |
| USA | – “- | – “- |
| Ukraine | No | No |
Formulations
-
Tablets of 10 mg, 20 mg
Valdecoxib is chemically designated as 4-(5-methyl-3-phenyl-4-isoxazolyl) benzenesulfonamide and is a diaryl substituted isoxazole.
The empirical formula for valdecoxib is C16H14N2O3S, and the molecular weight is 314.36. Valdecoxib is a white crystalline powder that is relatively insoluble in water (10 µg/mL) at 25° C and pH 7.0, soluble in methanol and ethanol, and freely soluble in organic solvents and alkaline (pH=12) aqueous solutions.
BEXTRA (valdecoxib) Tablets for oral administration contain either 10 mg or 20 mg of valdecoxib. Inactive ingredients include lactose monohydrate, microcrystalline cellulose, pregelatinized starch, croscarmellose sodium,magnesium stearate, hydroxypropyl methylcellulose, polyethylene glycol, polysorbate 80, and titanium dioxide
………………………
NMR

Links
-
Talley, JJ et al .: J. Med. Chem. (JMCMAR) 43, 775-777 (2000).
-
US 5,859,257 (GD Searle; 12.1.1999; USA-prior. 13.2.1995).
Literature References:
Selective cyclooxygenase-2 (COX-2) inhibitor. Active metabolite of parecoxib, q.v. Prepn: J. J. Talley et al., WO 9625405 (1996 to Searle); eidem, US 5633272 (1997); and activity: eidem, J. Med. Chem. 43, 775 (2000).
Chromatographic determn of purity: D. A. Roston et al., J. Pharm. Biomed. Anal. 26, 339 (2001).
Gastrointestinal tolerability study: G. M. Eisen et al., Aliment. Pharmacol. Ther. 21, 591 (2005).
Clinical trial in hip arthroplasty: F. Camu et al., Am. J. Ther.9, 43 (2002).
Clinical comparison with oxycodone/acetominophen in dental pain: S. E. Daniels et al., J. Am. Dent. Assoc. 133, 611 (2002).
Clinical trial in migraine: D. Kudrow et al., Headache 45, 1151 (2005).
Review of clinical experience: M. Goldman, S. Schutzer, Formulary 37, 68-77 (2002); of clinical efficacy and safety: G. P. Joshi, Expert Rev. Neurother. 5, 11-24 (2005).
References
- Jump up^ Thomson Micromedex. “Valdecoxib. U.S. FDA Drug Approval.” Last accessed June 8, 2007.
- ^ Jump up to:a b “Pfizer to pay $2.3 billion to resolve criminal and civil health care liability relating to fraudulent marketing and the payment of kickbacks”. Stop Medicare Fraud, US Dept of Health & Human Svc, and of Justice. Retrieved 2012-07-04.
- Jump up^ “Adverse Effects of Cyclooxygenase-2 Inhibitors on Renal and Arrhythmia Events: Meta-Analysis of Randomized Trials”, (JAMA 2006, by Zhang JJ, Ding EL, Song Y.).
- Jump up^ http://news.bbc.co.uk/2/hi/business/8234533.stm Pfizer agrees record fraud fine
- Jump up^ http://www.usdoj.gov/usao/ma/Press%20Office%20-%20Press%20Release%20Files/Sept2009/PharmaciaPlea.html
- Jump up^ http://www.usdoj.gov/usao/ma/Press%20Office%20-%20Press%20Release%20Files/Mar2009/FarinaconvictionPR.html
- Jump up^ http://industry.bnet.com/pharma/10002882/pfizers-off-label-bextra-team-were-called-the-highlanders/
- Jump up^ http://www.usdoj.gov/usao/ma/Press%20Office%20-%20Press%20Release%20Files/June2009/HollowayMarySentencingPR.html
- Jump up^ http://www.fbi.gov/pressrel/pressrel09/justice_090209.htm
- Jump up^ http://www.phillipsandcohen.com/CM/NewsSettlements/NewsSettlements536.asp
- Jump up^ http://www.phillipsandcohen.com/CM/NewsSettlements/NewsSettlements531.asp
- Jump up^ Talley, J. J.; Brown, D. L.; Carter, J. S.; Graneto, M. J.; Koboldt, C. M.; Masferrer, J. L.; Perkins, W. E.; Rogers, R. S.; Shaffer, A. F.; Zhang, Y. Y.; Zweifel, B. S.; Seibert, K. (2000). “4-[5-Methyl-3-phenylisoxazol-4-yl]- benzenesulfonamide, Valdecoxib: A Potent and Selective Inhibitor of COX-2”. Journal of Medicinal Chemistry 43 (5): 775–777. doi:10.1021/jm990577v.PMID 10715145.
- Jump up^ Prafulla Kumar Sahu and M. Mathrusri Annapurna, Analytical method development by liquid chromatography, LAP Lambert Academic Publisher, Germany, 2011 ISBN 3-8443-2869-6.
- Jump up^ Zhang J Y, Fast D M and Breau A P, J Chromatogr B Analyt Technol Biomed Life Sci., 2003, 785(1), 123-134
- Jump up^ Ramakrishna N V S, Vishwottam K N; Wishu S and Koteshwara M, J Chromatogr B Analyt Technol Biomed Life Sci., 2004, 802(2), 271.
- Jump up^ Sane R T, Menon S, Deshpande A Y and Jain A, Chromatogr., 2005, 61(3-4), 137-141.
- Jump up^ Prafulla Kumar Sahu*, K. Ravi Sankar and M. Mathrusri Annapurna, Determination of Valdecoxib in human plasma using Reverse Phase HPLC”, E-Journal of Chemistry, 2011, 8(2), 875-881.
- Jump up^ Mandal U, Jayakumar M, Ganesan M, Nandi S, Pal T K, Chakraborty M K, Roy Chowdhary A. and Chattoraj T K, Indian Drugs, 2004, 41, 59.
- Zhang J.Y, Fast D.M and Breau, A.P, J Pharm Biomed Anal., 2003, 33, 61.
- Werner U, Werner D, Hinz B, Lanbrecht C and Brune K, J Biomed Chromatogr., 2004, 19, 113.
- Zhang J V, Fast D M and Breau A P, J Chromatogr B Anal Technol Biomed Life Sci., 2003, 785, 123.
- Sutariya V B, Rajashree M, Sankalia M G. and Priti P, Indian J Pharm Sci., 2004, 93, 112.
- J Gandhimathi M, Ravi T K, Shukla Nilima and Sowmiya G, Indian J Pharm Sci., 2007, 69(1), 145-147.
External links[edit]
- FDA Alert on Bextra withdrawal
- Large systematic review of adverse renal and arrhythmia risk of valdcoxib and other COX-2 inhibitors, JAMA 2006

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....