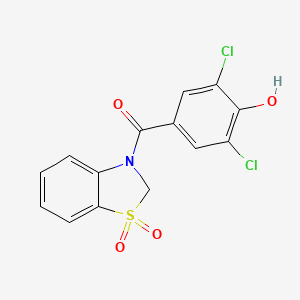
Dotinurad
ドチヌラド
(3,5-dichloro-4-hydroxyphenyl)-(1,1-dioxo-2H-1,3-benzothiazol-3-yl)methanone
| Formula |
C14H9Cl2NO4S
|
|---|---|
| CAS |
1285572-51-1
|
| Mol weight |
358.1966
|
PMDA, Urece, APROVED JAPAN 2020/1/23, Antihyperuricemic
305EB53128UNII-305EB53128
1285572-51-1,
Dotinurad is a urate transporter inhibitor.
Patents
WO 2011040449

https://patents.google.com/patent/WO2011040449A1/en
Uric acid is produced by metabolizing a purine produced by the degradation of a nucleic acid in the body and adenosine triphosphate (ATP), which is an energy source of the living body, to xanthine, and further undergoes oxidation by xanthine oxidase or xanthine dehydrogenase. In humans, uric acid (dissociation constant pKa = 5.75) is the final metabolite of purines and exists in the body as free forms or salts.
Uric acid is normally excreted in the urine, but when uric acid production exceeds excretion and blood uric acid increases, hyperuricemia occurs. If a state in which the blood level of uric acid exceeds the upper limit of solubility (about 7 mg / dL) continues for a long period of time, crystals of urate (usually sodium salt) precipitate.
In the blood, the precipitated crystals deposit on cartilage tissue and joints, form precipitates and become gouty nodules, causing acute gouty arthritis, and then transition to chronic gouty arthritis.
When uric acid crystals are precipitated in urine, renal disorders such as interstitial nephritis (gouty kidney), urinary calculi, and the like are caused. After the seizures of acute gouty arthritis have subsided, drug therapy is given along with lifestyle improvement guidance to correct hyperuricemia.
Correcting hyperuricemia and appropriately managing uric acid levels are also important in preventing acute gouty arthritis, gouty kidneys, urinary tract stones, and the like.
Hyperuricemia is considered to be associated with a high rate of lifestyle-related diseases such as obesity, hyperlipidemia, impaired glucose tolerance, and hypertension (see Non-Patent Document 1 (pp7-9)). Increased serum uric acid levels are positively related to cardiovascular mortality, and higher serum uric acid levels increase mortality due to ischemic heart disease. It has been suggested that it is associated with the risk of death from disease (see Non-Patent Document 2).
Furthermore, serum uric acid levels have also been shown to be a powerful risk factor for myocardial infarction and stroke (see Non-Patent Document 3). To date, hyperuricemia is obesity, hyperlipidemia, dyslipidemia, impaired glucose tolerance, diabetes, metabolic syndrome, kidney disease (eg, renal failure, urine protein, end-stage renal disease (ESRD), etc.), heart It is known to be associated with vascular diseases (for example, hypertension, coronary artery disease, carotid artery disease, vascular endothelial disorder, arteriosclerosis, cardiac hypertrophy, cerebrovascular disease, etc.) or risk factors of these diseases (Non-Patent Documents 2 to 11) reference). In cerebrovascular dementia, it has also been reported that the concentration of uric acid in the cerebrospinal cord is increased (see Non-Patent Document 12).
Under such circumstances, it has been suggested that the treatment for lowering the blood uric acid level may delay the progression of kidney disease and reduce the risk of cardiovascular disease (Non-Patent Documents 5, 8, 13, 14), it has been reported that it should also be applied to asymptomatic hyperuricemia (see Non-Patent Document 14).
Therefore, reducing the blood uric acid level in the above-mentioned diseases is effective for the treatment or prevention of these diseases, and is considered to be important in terms of preventing recurrence of cardiovascular accidents and maintaining renal function.
The main factors that increase blood uric acid levels include excessive uric acid production and decreased uric acid excretion. Therefore, as a method for lowering blood uric acid level, it is conceivable to suppress the production of uric acid or promote the excretion of uric acid, and allopurinol is a drug having the former mechanism of action (uric acid production inhibitor). Benzbromarone, probenecid, JP-A 2006-176505 (Patent Document 1) and the like are known as drugs having the latter mechanism of action (uric acid excretion promoters).
According to the Japanese guidelines for treatment of hyperuricemia and gout, in principle, uric acid excretion-promoting agents are applied to hyperuricemia-reducing types and uric acid production-inhibiting agents are applied to excessive uric acid production types, respectively. (See Non-Patent Document 1 (pp31-32)).
In Japan, it is said that about 60% of hyperuricemia patients have a reduced uric acid excretion type, and about 25% are a mixed type of reduced uric acid excretion type and excessive uric acid production type (Non-patent Document 15). About 85% of the patients showed a decrease in uric acid excretion, and the average value of uric acid clearance was significantly lower than that of healthy individuals even in patients with excessive uric acid production, and the decrease in uric acid excretion was fundamental in all gout patients. Is also reported (Non-Patent Document 16).
Therefore, in hyperuricemia (especially gout), treatment for patients with reduced uric acid excretion is considered to be important, and the existence significance of uric acid excretion promoters is extremely large.
Among the major uric acid excretion promoters, probenecid is weakly used and is rarely used because of its gastrointestinal tract disorders and interactions with other drugs. On the other hand, severe liver damage has been reported for benzbromarone, which has a strong uric acid excretion promoting action and is widely used in Japan as a uric acid excretion promoting drug (see Non-Patent Document 17).
Benzbromarone or its analogs inhibit mitochondrial respiratory chain enzyme complex activity, uncoupling action, respiration inhibition, fatty acid β oxidation inhibition, mitochondrial membrane potential reduction, apoptosis, generation of reactive oxygen species, etc. Has been suggested to be involved in the development of liver damage (see Non-Patent Documents 18 and 19). Hexahydrate, which is the active body of benzbromarone, is also toxic to mitochondria.
Furthermore, benzbromarone has an inhibitory action on cytochrome P450 (CYP), which is a drug metabolizing enzyme. In particular, the inhibition against CYP2C9 is very strong, suggesting the possibility of causing a pharmacokinetic drug interaction (non-) (See Patent Documents 20 and 21).
Furthermore, although a nitrogen-containing fused ring compound having a URAT1 inhibitory action, which is a kind of uric acid transporter, and having a structure similar to that of the compound of the present invention is described in JP-A-2006-176505 (Patent Document 1), the effect is sufficient. In addition, no practical uric acid excretion promoter has been developed yet.
Recently, it has been found that the uric acid excretion promoting action depends on the urinary concentration of a drug having the same action, that is, the uric acid excretion promoting drug is excreted in the urine and exhibits a medicinal effect (Patent Document 2). Non-Patent Documents 22 and 23).
Therefore, a stronger pharmacological effect is expected if it is a uric acid excretion promoter that is excreted more in the urine, but the above existing uric acid excretion promoters have a very low concentration in urine, and a satisfactory activity can be obtained sufficiently. I can’t say that.
Regarding the urinary excretion of drugs, it is assumed that the administered drug is excreted as it is as an unchanged form or converted into an active metabolite and excreted. In the latter case, the active metabolite is produced. There is a risk that the individual difference in the amount becomes large, and in order to obtain stable drug efficacy and safety, a drug excreted as an unchanged substance is more desirable.
As described above, there is a demand for the development of a highly safe pharmaceutical having a high unchanged body urine concentration and a remarkable uric acid excretion promoting action as compared with existing uric acid excretion promoting drugs.
JP 2006-176505 A WO2005 / 121112
Treatment Guidelines for Hyperuricemia and Gout (1st Edition) pp7-9 and pp31-32, Gout and Nucleic Acid Metabolism, Volume 26, Supplement 1, 2002 Japan Gout and Nucleic Acid Metabolism Society JAMA 283: 2404-2410 (2000) Stroke 37: 1503-1507 (2006) Nephrology 9: 394-399 (2004) Semin. Nephrol. 25: 43-49 (2005)J. Clin. Hypertens. 8: 510-518 (2006) J. Hypertens. 17: 869-872 (1999) Curr. Med. Res. Opin. 20: 369-379 (2004) Curr. Pharm. Des. 11: 4139-4143 (2005)Hypertension 45: 991-996 (2005) Arch. Intern. Med. 169: 342-350 (2009) J. Neural. Transm. Park Dis. Dement. Sect. 6: 119-126 (1993) Am. J. Kidney Dis. 47: 51-59 (2006) Hyperuricemia and gout 9: 61-65 (2001) Japanese clinical trials 54: 3230-3236 (1996) Japanese clinical trial 54: 3248-3255 (1996) J. Hepatol. 20: 376-379 (1994) J. Hepatol. 35: 628-636 (2001) Hepatology 41: 925-935 (2005) Saitama Medical University Journal (J. Saitama. Med. School) 30: 187-194 (2004) Drug Metab. Dispos. 31: 967-971 (2003) 42nd Annual Meeting of the Japanese Gout and Nucleic Acid Metabolism General Assembly Program / Abstracts, p59 (2009) ACR 2008 Annual Scientific Meeting, No. 28




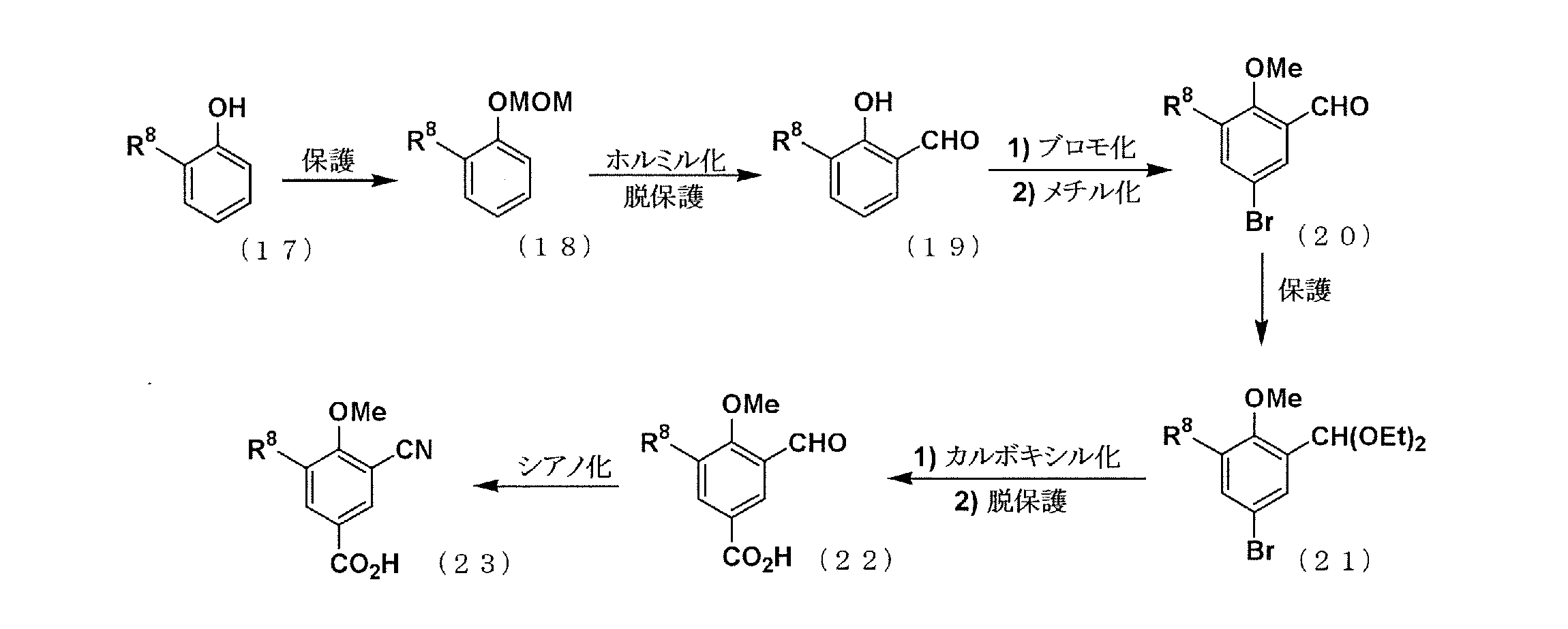

PATENT
JP 2011074017
PATENT
WO 2018199277
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2018199277
//////////Dotinurad, Antihyperuricemic, JAPAN 2020, 2020 APPROVALS , ドチヌラド , VOFLAIHEELWYGO-UHFFFAOYSA-N, HY-109031, CS-0030545
C1N(C2=CC=CC=C2S1(=O)=O)C(=O)C3=CC(=C(C(=C3)Cl)O)Cl

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....