Daprodustat, GSK1278863
ダプロデュスタット
CAS 960539-70-2
GSK1278863; GSK 1278863; GSK-1278863; Daprodustat
C19H27N3O6
Exact Mass: 393.18999
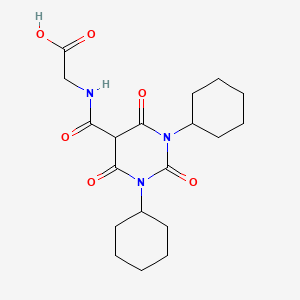
(1,3-dicyclohexyl-2,4,6-trioxohexahydropyrimidine-5-carbonyl)glycine
N-[(l,3-dicyclohexyl-6-hydroxy-2,4-dioxo-l,2,3,4- tetrahydro-5-pyrimidinyl)carbonyl]glycine
2-(1,3-dicyclohexyl-2,4,6-triohexahydropyrimidine-5-carboxamide acetic acid
Mechanism of Action: HIF-prolyl hydroxylase inhibitor
Indication: anemia, diabetic wounds, and reduction of ischemic complications
Development Stage: Phase II
Developer:GlaxoSmithKline
UNII:JVR38ZM64B
ダプロデュスタット
Daprodustat

C19H27N3O6 : 393.43
[960539-70-2]
Daprodustat , also known as GSK1278863, is a novel HIF-prolyl hydroxylase inhibitor. Hypoxia inducible factor (HIF) stabilization by HIF-prolyl hydroxylase (PHD) inhibitors may improve ischemic conditions such as peripheral artery disease (PAD). Short-term treatment with a novel HIF-prolyl hydroxylase inhibitor (GSK1278863) failed to improve measures of performance in subjects with claudication-limited peripheral artery disease
- Originator GlaxoSmithKline
- Class Antianaemics; Pyrimidines; Small molecules
- Mechanism of ActionErythropoiesis stimulants; Prolyl hydroxylase inhibitors
- Phase II Anaemia; Perioperative ischaemia
- Phase I Diabetic foot ulcer; Tendon injuries
- DiscontinuedPeripheral arterial disorders
Most Recent Events
- 27 Jul 2015No recent reports of development identified – Phase-II for Anaemia in India and New Zealand (PO)
- 27 Jul 2015Daprodustat is still in phase II trials for Anaemia in the USA, Australia, Canada, Czech Republic, Denmark, France, Germany, Hungary, Japan, Poland, Russia, Spain, South Korea, and United Kingdom
- 01 Jun 2015GlaxoSmithKline completes a phase I trial in Tendon injuries (In volunteers) in USA (PO) (NCT02231190)
| WHO ATC code: | B03 (Antianemic Preparations)C (Cardiovascular System)
C01 (Cardiac Therapy) D03 (Preparations for Treatment of Wounds and Ulcers) M09A-X (Other drugs for disorders of the musculo-skeletal system) |
| EPhMRA code: | B3 (Anti-Anaemic Preparations)C1 (Cardiac Therapy)
C6A (Other Cardiovascular Products) D3A (Wound Healing Agents) M5X (All Other Musculoskeletal Products) |
Daprodustat (INN) (GSK1278863) is a drug which acts as a HIF prolyl-hydroxylase inhibitor and thereby increases endogenous production of erythropoietin, which stimulates production of hemoglobin and red blood cells. It is in Phase III clinical trials for the treatment of anemia secondary to chronic kidney disease.[1][2] Due to its potential applications in athletic doping, it has also been incorporated into screens for performance-enhancing drugs.[3]
SYN 1

SYN 2

PATENT
WO 2007150011
https://www.google.com.ar/patents/WO2007150011A2
Illustrated Methods of preparation
Scheme 1
a) 1. NaH, THF, rt 2. R1NCO, 60 0C; b) 1. NaH, THF or dioxane, rt 2. R4NCX, heat; c) H2NCH2CO2H, DBU, EtOH, 1600C, microwave.
Scheme 2
a) R1NH2, CH2Cl2 or R1NH2-HCl, base, CH2Cl2; b) CH2(C(O)Cl)2, CH2Cl2, reflux or CH2(CO2Et)2, NaOEt, MeO(CH2)2OH, reflux or 1. EtO2CCH2COCl, CHCl3, 70 0C 2.
DBU, CHCl3, 70 0C; c) 1. YCNCH2CO2Et,, EtPr’2N, CHCl3 or CH2Cl2 2. aq NaOH, EtOH, rt. Scheme 3 (for R1 = R4)
a) CDI,
DMF, 70 0C or , EtOAc, rt
Scheme 4
a) OCNCH2CO2Et, EtPr’2N, CHCl3 or CH2Cl2; b) 1. R1HaI, Na/K2CO3, DMF or DMA, 100 0C or R1HaI, pol-BEMP, DMF, 120 0C, microwave 2. aq NaOH, MeOH or EtOH, rt.
Scheme 5
a) 1. CH2(CO2H)2, THF, O 0C – rt 2. EtOH, reflux; b) 1. OCNCH2CO2Et, EtPr’2N, CH2Cl2 2. aq NaOH, EtOH, rt.
Scheme 6
a) 1. Phthalimide, DIAD, PPh3, THF 2. (NH2)2, EtOH, reflux.
Scheme 7
a) Ac2O, AcOH, 130 0C.
Example 18
N-T(1 ,3-Dicvclohexyl-6-hydroxy-2,4-dioxo- 1 ,2,3,4-tetrahvdro-5-pyrimidinyl)carbonyl1grycine Method 1
18.1a) h3-Dicvclohexyl-2A6(lH,3H,5H)-pyrimidinetrione. Dicyclohexylurea (3.0 g, 13.39 mmoles) was stirred in chloroform (80 mL) and treated with a solution of malonyl dichloride (1.3 mL, 13.39 mmoles) in chloroform (20 mL), added dropwise under argon. The mixture was heated at 500C for 4 hours, wasahed with 1 molar hydrochloric acid and evaporated onto silica gel. Flash chromatography (10-30% ethyl acetate in hexane) to give the title compound (2.13 g, 55%). 1Η NMR (400 MHz, OMSO-d6) δ ppm 4.46 (tt, J=12.13, 3.54 Hz, 2 H), 3.69 (s, 2 H), 2.15 (qd, J=12.46, 3.28 Hz, 4 H), 1.77 (d, J=13.14 Hz, 4 H), 1.59 (t, J=12.76 Hz, 6 H), 1.26 (q, J=12.97 Hz, 4 H), 1.04 – 1.16 (m, 2 H)
18.1b) N-r(1.3-Dicvclohexyl-6-hvdroxy-2.4-dioxo-1.2.3.4-tetrahvdro-5- pyrimidinvDcarbonyll glycine. Ethyl isocyanatoacetate (802 uL, 7.15 mmoles) was added to a mixture of l,3-dicyclohexyl-2,4,6(lH,3H,5H)-pyrimidinetrione (2.1 g, 7.15 mmoles) and diisopropylethylamine (2.47 mL, 14.3 mmoles) in dichloromethane (100 mL) and stirred overnight. The reaction mixture was washed with 1 molar hydrochloric acid (x2) and evaporated. The residue was dissolved in ethanol (10 mL) and treated with 1.0 molar sodium hydroxide (5 mL). The mixture was stirred for 72 hours, acidified and extracted into ethyl acetate. Some ester remained, therefore the solution was evaporated and ther residue was dissolved in 1 molar soldium hydroxide solution with warming and strred for 2 hours. The mixture was acidified with IM HCl and extracted with ethyl acetate (x2). The combined extracts were washed with 1 molar hydrochloric acid , dried and evaporated to a solid which was slurried in a mixture of diethyl ether and hexane, collected, washed with the same solvent mixture and dried to give the title compound (1.86 g, 66%). IH NMR (400 MHz, DMSO-^6) δ ppm 13.07 (br. s., 1 H), 10.19 (t, J=5.31 Hz, 1 H), 4.63 (t, J=10.99 Hz, 2 H), 4.12 (d, J=5.56 Hz, 2 H), 2.27 (q, J=I 1.71 Hz, 4 H), 1.79 (d, J=12.88 Hz, 4 H), 1.50 – 1.69 (m, 6 H), 1.28 (q, J=12.97 Hz, 4 H), 1.12 (q, J=12.72 Hz, 2 H)
Method 2
18.2a) 1.3-Dicvclohexyl-2.4.6πH.3H.5H)-pyrimidinetrione. A solution of N5N- dicyclohexylcarbodiimide (254 g; 1.23 mol.) in anhydrous TΗF (700 mL) was added dropwise to a cold (0 0C) solution of malonic acid (64.1 g; 0.616 mol.) in anhydrous TΗF (300 mL) over a period of- 30 minutes. The mixture was stirred and allowed to warm to room temperature over 2 h. (After 1 h, the mixture became very thick with precipitate so further anhydrous TΗF (500 mL) was added to facilitate agitation.). The mixture was filtered and the filtrate evaporated to afford a yellow solid which was immediately slurried in ethanol (1 L) and heated to reflux temperature. The mixture was then allowed to cool to room temperature then filtered and the solid washed with cold ethanol (250 mL) to afford the title compound (129.4 g; 72%) as a colorless solid. 1Η NMR (400 MHz, DMSO-(Z6) δ ppm 1.03 – 1.18 (m, 2 H) 1.18 – 1.34 (m, 4 H) 1.59 (t, J=13.14 Hz, 6 H) 1.76 (d, J=12.88 Hz, 4 H) 2.04 – 2.24 (m, 4 H) 3.69 (s, 2 H) 4.35 – 4.54 (m, 2 H).
18.2b) Ethyl N-[(l .3-dicvclohexyl-6-hvdroxy-2.4-dioxo- 1.2.3.4-tetrahydro-5- pyrimidinyPcarbonyll glycinate. A solution of l,3-dicyclohexyl-2,4,6(lH,3H,5H)-pyrimidinetrione (120.0 g; 0.41 mol.) and diisopropylethylamine (105.8 g; 0.82 mol.) in dichloromethane (1 L) was stirred and treated dropwise with a solution of ethyl isocyanatoacetate (53.0 g; 0.41 mol.) in dichloromethane (500 mL) and the mixture was then stirred at room temperature overnight. The mixture was then treated dropwise with 6M aq. hydrochloric acid (500 mL) and the separated organic layer was dried and evaporated. The resulting solid was slurried in hexanes (500 mL) and heated to reflux temperature. The mixture was then allowed to cool and filtered to afford ethyl N- [(1 ,3-dicyclohexyl-6-hydroxy-2,4-dioxo- 1 ,2,3,4-tetrahydro-5-pyrimidinyl)carbonyl]glycinate (159.1 g; 92%) as a cream powder. IH NMR (400 MHz, CHLOROFORM-,/) δ ppm 1.24 (s, 2 H) 1.37 (s, 7 H) 1.52 – 1.76 (m, 6 H) 1.78 – 1.94 (m, 4 H) 2.25 – 2.48 (m, 4 H) 4.17 (d, J=5.81 Hz, 2 H) 4.28 (q, J=7.24 Hz, 2 H) 4.74 (s, 2 H) 10.37 (t, J=4.67 Hz, 1 H). 18.2c)
N-rπ^-Dicyclohexyl-ό-hydroxy^^-dioxo-l^J^-tetralivdro-S- pyrimidinyDcarbonyll glycine. A stirred suspension of ethyl Ν-[(l,3-dicyclohexyl-6-hydroxy-2,4- dioxo-l,2,3,4-tetrahydro-5-pyrimidinyl)carbonyl]glycinate (159.0 g; 0.377 mol.) in ethanol (1.5 L) was treated dropwise with 6M aq. Sodium hydroxide (250 mL) and stirred at room temperature for 3 h. The solution was then acidified by the dropwise addition of 6M aq. hydrochloric acid (300 mL), diluted with water (IL) and then filtered. The crude solid was slurried in water (2 L) then stirred vigorously and heated at 35 0C for 1 h and filtered and dried. The solid material (~ 138 g) was then crystallized from glacial acetic acid (1.5 L) (with hot filtration to remove a small amount of insoluble material). The solid, which crystallized upon cooling, was collected and washed with cold glacial acetic acid (3 x 100 mL) to afford N-[(l,3-dicyclohexyl-6-hydroxy-2,4-dioxo-l,2,3,4- tetrahydro-5-pyrimidinyl)carbonyl]glycine (116.2 g; 78%) as a colorless solid.
IH NMR (400 MHz, DMSO-(Z6) δ ppm 1.11 (d, J=12.88 Hz, 2 H) 1.27 (q, J=12.80 Hz, 4 H) 1.62 (s, 6 H) 1.70 – 1.90 (m, J=12.88 Hz, 4 H) 2.11 – 2.44 (m, 4 H) 4.11 (d, J=5.81 Hz, 2 H) 4.45 – 4.77 (m, 2 H) 10.19 (t, J=5.81 Hz, 1 H) 13.08 (s, 1 H).
References
- Jump up^ Schmid H, Jelkmann W. Investigational therapies for renal disease-induced anemia. Expert Opin Investig Drugs. 2016 Aug;25(8):901-16. . doi:10.1080/13543784.2016.1182981. PMID 27122198. Missing or empty
|title=(help) - Jump up^ Ariazi JL, Duffy KJ, Adams DF, Fitch DM, Luo L, Pappalardi M, Biju M, DiFilippo EH, Shaw T, Wiggall K, Erickson-Miller C. Discovery and Preclinical Characterization of GSK1278863 (Daprodustat), a Small Molecule Hypoxia Inducible Factor-Prolyl Hydroxylase Inhibitor for Anemia. J Pharmacol Exp Ther. 2017 Dec;363(3):336-347. . doi:10.1124/jpet.117.242503. PMID 28928122. Missing or empty
|title=(help) - Jump up^ Thevis M, Milosovich S, Licea-Perez H, Knecht D, Cavalier T, Schänzer W. Mass spectrometric characterization of a prolyl hydroxylase inhibitor GSK1278863, its bishydroxylated metabolite, and its implementation into routine doping controls. Drug Test Anal. 2016 Aug;8(8):858-63. . doi:10.1002/dta.1870. PMID 26361079. Missing or empty
|title=(help)
 |
|
| Clinical data | |
|---|---|
| Synonyms | GSK1278863 |
| ATC code |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| Chemical and physical data | |
| Formula | C19H27N3O6 |
| Molar mass | 393.44 g/mol |
| 3D model (JSmol) | |
//////////////Daprodustat, GSK1278863, ダプロデュスタット , HIF-prolyl hydroxylase inhibitor, anemia, diabetic wounds, reduction of ischemic complications, Phase II, GlaxoSmithKline
- Daprodustat
- 960539-70-2
- GSK1278863
- UNII-JVR38ZM64B
- GSK-1278863
- JVR38ZM64B
- N-((1,3-Dicyclohexylhexahydro-2,4,6-trioxopyrimidin-5-yl)carbonyl)glycine
- Daprodustat [USAN:INN]
- GSK 1278863
- D0F6JC
- Daprodustat(GSK1278863)
- Daprodustat; GSK1278863
- Daprodustat (JAN/USAN/INN)
- GTPL8455
- Daprodustat (GSK1278863)
- CHEMBL3544988
- BCP16766
- EX-A1121
- KS-00000M8Z
- s8171
C1CCC(CC1)N2C(=O)C(C(=O)N(C2=O)C3CCCCC3)C(=O)NCC(=O)O

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....