Home » Posts tagged 'Stakel'
Tag Archives: Stakel
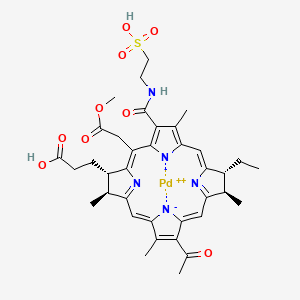
PADELIPORFIN
PADELIPORFIN
759457-82-4; 457P824,
RN: 759457-82-4
UNII: EEO29FZT86
3-[(2S,3S,12R,13R)-8-acetyl-13-ethyl-20-(2-methoxy-2-oxoethyl)-3,7,12,17-tetramethyl-18-(2-sulfoethylcarbamoyl)-2,3,12,13-tetrahydroporphyrin-22,24-diid-2-yl]propanoic acid;palladium(2+)
(SP-4-2)-[(7S,8S,17R,18R)-13-acetyl-18-ethyl-5-(2-methoxy-2-oxoethyl)-2,8,12,17-tetramethyl-3-[[(2-sulfoethyl)amino]carbonyl]-21H,23H-porphine-7-propanoato (4-)-kN21,kN22,kN23,kN24] palladate(2-)
Other Names
- (SP-4-2)-[(7S,8S,17R,18R)-13-Acetyl-18-ethyl-7,8-dihydro-5-(2-methoxy-2-oxoethyl)-2,8,12,17-tetramethyl-3-[[(2-sulfoethyl)amino]carbonyl]-21H,23H-porphine-7-propanoato(4-)-κN21,κN22,κN23,κN24]palladate(2-)
| Molecular Formula: | C37H43N5O9PdS |
|---|---|
| Molecular Weight: | 840.257 g/mol |

Chemical Formula: C37H41K2N5O9PdS
Molecular Weight: 916.43
cas 698393-30-5
WST11; WST-11; WST 11; Stakel; padeliporfin; palladiumbacteriopheophorbide monolysine taurine.
Palladate(2-), [(7S,8S,17R,18R)-13-acetyl-18-ethyl-7,8-dihydro-5-(2-methoxy-2-oxoethyl)-2,8,12,17-tetramethyl-3-[[(2-sulfoethyl)amino]carbonyl]-21H,23H-porphine-7-propanoato(4-)-κN21,κN22,κN23,κN24]-, potassium (1:2), (SP-4-2)-
Tookad : EPAR -Product Information
| Tookad : EPAR – Summary for the public | (English only) | 29/11/2017 |
Product details
Pharmacotherapeutic group
Antineoplastic agents
Therapeutic indication
Tookad is indicated as monotherapy for adult patients with previously untreated, unilateral, low risk, adenocarcinoma of the prostate with a life expectancy ≥ 10 years and:
- Clinical stage T1c or T2a;
- Gleason Score ≤ 6, based on high-resolution biopsy strategies;
- PSA ≤ 10 ng/mL;
- 3 positive cancer cores with a maximum cancer core length of 5 mm in any one core or 1-2 positive cancer cores with ≥ 50 % cancer involvement in any one core or a PSA density ≥ 0.15 ng/mL/cm³.
| Name | Tookad |
|---|---|
| Agency product number | EMEA/H/C/004182 |
| Active substance | padeliporfin di-potassium |
| International non-proprietary name(INN) or common name | padeliporfin |
| Therapeutic area | Prostatic Neoplasms |
| Anatomical therapeutic chemical (ATC) code | L01XD07 |
| Additional monitoring | This medicine is under additional monitoring. This means that it is being monitored even more intensively than other medicines. For more information, see medicines under additional monitoring. |
| Marketing-authorisation holder | STEBA Biotech S.A |
|---|---|
| Revision | 0 |
| Date of issue of marketing authorisation valid throughout the European Union | 10/11/2017 |
Contact address:
STEBA Biotech S.A
7 place du theatre
L-2613 Luxembourg
Luxembourg
Padeliporfin is a vascular-acting photosensitizer consisting of a water-soluble, palladium-substituted bacteriochlorophyll derivative with potential antineoplastic activity. Upon administration, paldeliporfin is activated locally when the tumor bed is exposed to low-power laser light; reactive oxygen species (ROS) are formed upon activation and ROS-mediated necrosis may occur at the site of interaction between the photosensitizer, light and oxygen. Vascular-targeted photodynamic therapy (VTP) with padeliporfin may allow tumor-site specific cytotoxicity while sparing adjacent normal tissues.
WST-11 (Stakel) is a water-soluble bacteriochlorophyll (chemical structure shown below) derivative coordinated with palldium, which has maximum absorption wavelength in the near infrared (753 nm) and rapid clearance from the body ( t 1/2 = 0.37 hour for a 10-mg/kg drug dose in the rat and t 1/2 = 0.51 hour, 1 hour, and 2.65 hours for 1.25-, 2.5-, and 5-mg/kg drug doses, respectively. It binds to serum albumin and has potent antivascular activity through the generation of hydroxyl radicals when stimulated by the proper light wavelength.

Photodynamic therapy (PDT) is a non-surgical treatment of tumors in which non-toxic drugs and non-hazardous photosensitizing irradiation are combined to generate cytotoxic reactive oxygen species in situ. This technique is more selective than the commonly used tumor chemotherapy and radiotherapy. To date, porphyrins have been employed as the primary photosensitizing agents in clinics. However, current sensitizers suffer from several deficiencies that limit their application, including mainly: (1) relatively weak absorption in the visible spectral range which limits the treatment to shallow tumors; (2) accumulation and long retention of the sensitizer in the patient skin, leading to prolonged (days to months) skin phototoxicity; and (3) small or even no differentiation between the PDT effect on illuminated tumor and non-tumor tissues. The drawbacks of current drugs inspired an extensive search for long wavelength absorbing second-generation sensitizers that exhibit better differentiation between their retention in tumor cells and skin or other normal tissues.
In order to optimize the performance of the porphyrin drugs in therapeutics and diagnostics, several porphyrin derivatives have been proposed in which, for example, there is a central metal atom (other than Mg) complexed to the four pyrrole rings, and/or the peripheral substituents of the pyrrole rings are modified and/or the macrocycle is dihydrogenated to chlorophyll derivatives (chlorins) or tetrahydrogenated to bacteriochlorophyll derivatives (bacteriochlorins).
Due to their intense absorption in favorable spectral regions (650-850 nm) and their ready degradation after treatment, chlorophyll and bacteriochlorophyll derivatives have been identified as excellent sensitizers for PDT of tumors and to have superior properties in comparison to porphyrins, but they are less readily available and more difficult to handle.
Bacteriochlorophylls are of potential advantage compared to the chlorophylls because they show intense near-infrared bands, i.e. at considerably longer wavelengths than chlorophyll derivatives.
The spectra, photophysics, and photochemistry of native bacteriochlorophylls (Bchls) have made them optimal light-harvesting molecules with clear advantages over other sensitizers presently used in PDT. In particular, these molecules have a very high extinction coefficient at long wavelengths (λmax=760-780 nm, ε=(4-10)xl04 M-1cm-1), where light penetrates deeply into tissues. They also generate reactive oxygen species (ROS) at a high quantum yield (depending on the central metal).
Under normal delivery conditions, i.e. in the presence of oxygen at room temperature and under normal light conditions, the BChl moieties are labile and have somewhat lower quantum yields for triplet state formation, when compared with, e.g., hematoporphyrin derivative (HPD). However, their possible initiation of biological redox reactions, favorable spectral characteristics and their ready degradation in vivo result in the potential superiority of bacteriochlorophylls over other compounds, e.g. porphyrins and chlorophylls, for PDT therapy and diagnostics and for killing of cells, viruses and bacteria in samples and in living tissue. Chemical modification of bacteriochlorophylls is expected to further improve their properties, but this has been very limited due to lack of suitable methods for the preparation of such modified bacteriochlorophylls .
The biological uptake and PDT efficacy of metal-free derivatives of Bchl have been studied with the objective to manipulate the affinity of the sensitizers to the tumor cellular compartment. Cardinal to this approach is the use of highly lipophilic drugs that may increase the accumulation of the drug in the tumor cells, but also renders its delivery difficult. In addition, the reported biodistribution shows significant phototoxic drug levels in non-tumor tissues over prolonged periods (at least days) after administering the drug.
In applicant’s previous Israel Patent No. 102645 and corresponding EP 0584552, US 5,726,169, US 5,726,169, US 5,955,585 and US 6,147,195, a different approach was taken by the inventors. Highly efficient anti- vascular sensitizers that do not extravasate from the circulation after administration and have short lifetime in the blood were studied. It was expected that the inherent difference between vessels of normal and abnormal tissues such as tumors or other tissues that rely on neovessels, would enable relatively selective destruction of the abnormal tissue. Hence, it was aimed to synthesize Bchl derivatives that are more polar and, hence, have better chance to stay in the vascular compartment, where they convey the primary photodynamic effect. To this end, the geranylgeranyl residue at the C-17 position of Bchl a (Compound 1, depicted in Scheme 1 herein) has been replaced by various residues such as amino acids, peptides, or proteins, which enhance the sensitizer hydrophilicity. One particular derivative, Bchl-Ser (Scheme 1, Compound 1, wherein R is seryl), was found to be water-soluble and highly phototoxic in cell cultures. Following infraperitoneal injection, the Bchl-Ser cleared from the mouse blood and tissues bi-exponentially in a relatively short time (t1/2~2 and 16 h, respectively). Clearance from the circulation was even faster following intravenous injection. Under the selected treatment protocol (light application within minutes after drug injection), phototoxicity was predominantly conferred to the tumor vasculature (Rosenbach-
Belkin et al., 1996; Zilberstein et al., 2001 and 1997). However, unfortunately, like native Bchl, the Bchl-Ser derivative undergoes rapid photo-oxidation, forming the corresponding 2-desvinyl-2-acetyl-chlorophyllide ester and other products.
To increase the stability of the Bchl derivatives, the central Mg atom was replaced by Pd in the later applicant’s PCT Publication WO 00/33833 and US 6,569,846. This heavy atom was previously shown to markedly increase the oxidation potential of the Bchl macrocycle and, at the same time, to greatly enhance the intersystem-crossing (ISC) rate of the molecule to its triplet state. The metal replacement was performed by direct incorporation of Pd2+ ion into a Bpheid molecule, as described in WO 00/33833. Based on the pigment biodistribution and pharmacokinetics, it was assumed that the derivative Pd-Bpheid remained in the circulation for a very short time with practically no extravasation to other tissues, and is therefore a good candidate for vascular-targeting PDT that avoids skin phototoxicity. The treatment effect on the blood vessels was demonstrated by intravital microscopy of treated blood vessels and staining with Evans-Blue. Using a treatment protocol with a minimal drug-to-light interval, Pd-Bpheid (also designated Tookad) was found to be effective in the eradication of different tumors in mice, rats and other animal models and is presently entering Phase I/II clinical trials in patients with prostate cancer that failed radiation therapy (Chen et al, 2002; Schreiber et al., 2002; Koudinova et al., 2003).
Because of its low solubility in aqueous solutions, the clinical use of Pd-Bpheid requires the use of solubilizing agents such as Cremophor that may cause side effects at high doses. It would be highly desirable to render the Pd-Bpheid water-soluble while retaining its physico-chemical properties. Alternatively, it would be desirable to prepare Bchl derivatives that are cytophototoxic and, at the same time, more water-soluble than Pd-Bpheid itself. Such water solubility is expected to further enhance the drug retention in the circulation and, thereby, the aforementioned selectivity. In addition, having no need to use carriers such as detergents or lyposomes, may prevent side effects.
SYNTHESIS
START FROM CAS 17499-98-8, Phorbine, magnesium deriv., Bacteriochlorophyll aP
PADELIPORFIN
Paper
Novel water-soluble bacteriochlorophyll derivatives for vascular-targeted photodynamic therapy: Synthesis, solubility, phototoxicity and the effect of serum proteins
Photochemistry and Photobiology (2005), 81, (July/Aug.), 983-993
PAPER
Journal of Medicinal Chemistry (2014), 57(1), 223-237

With the knowledge that the dominant photodynamic therapy (PDT) mechanism of 1a (WST09) switched from type 2 to type 1 for 1b (WST11) upon taurine-driven E-ring opening, we hypothesized that taurine-driven E-ring opening of bacteriochlorophyll derivatives and net-charge variations would modulate reactive oxygen species (ROS) photogeneration. Eight bacteriochlorophyll a derivatives were synthesized with varying charges that either contained the E ring (2a–5a) or were synthesized by taurine-driven E-ring opening (2b–5b). Time-dependent density functional theory (TDDFT) modeling showed that all derivatives would be type 2 PDT-active, and ROS-activated fluorescent probes were used to investigate the photogeneration of a combination of type 1 and type 2 PDT ROS in organic- and aqueous-based solutions. These investigations validated our predictive modeling calculations and showed that taurine-driven E-ring opening and increasing negative charge generally enhanced ROS photogeneration in aqueous solutions. We propose that these structure–activity relationships may provide simple strategies for designing bacteriochlorins that efficiently generate ROS upon photoirradiation.
Modulation of Reactive Oxygen Species Photogeneration of Bacteriopheophorbide a Derivatives by Exocyclic E-Ring Opening and Charge Modifications
CHEMICAL EXAMPLES
Example 1. Palladium 31-oxo-15-methoxycarbonylmethyl-Rhodobacteriochlorin 131-(2-sulfoethyl)amide dipotassium salt ( Compound 4)
Nine hundred and thirty five (935) mg of Pd-Bpheid (3) were dissolved in a 1 L round bottom flask with 120 ml of DMSO while stirring under Argon (bubbled in the solution). Taurine (1288 mg) was dissolved in 40 ml of 1M K2HPO4 buffer, and the pH of the solution was adjusted to 8.2 (with HCl ). This aqueous solution was added into the DMSO solution while stirring, and the Argon was bubbled in the solution for another 20 minutes. Then the reaction mixture was evaporated at 30°C for 3.5 hours under ~2 mbar and then for another 2 hours at 37°C to a complete dryness. The dry solids were dissolved in 300 ml of MeOH and the colored solution was filtered through cotton wool to get rid of buffer salts and taurine excess.
The progress of the reaction was determined by TLC (Rf of unreacted Pd- Bpheid is 0.8-0.85 and of the reaction (aminolysis) product is 0.08-0.1) and by following the optical absorption spectrum of the reaction mixture after liophylization and resolubihzation in MeOH. The absorption spectrum was characterized by a Qytransition shift from 756 nm (for Pd-Bpheid) to 747 nm (for the product 4) and by Qx shift from 534 nm of Pd-Bpheid to 519 nm (of the product 4). The MeOH was evaporated and the product 4 was purified by HPLC with ODS-A 250X20 S10P μm column (YMC, Japan). Solvent A: 95% 0.005 M phosphate buffer, pH 8.0 and 5% MeOH. Solvent B: 100% MeOH. The dry solid was dissolved in 42 ml of distilled water and injected in portions of 1.5 ml each .
The elution profile is described in Table 1. The product 4_(Scheme 1, see below) was eluted and collected at ~ 9-11 minutes. The main impurities, collected after at 4-7 min (ca 5-10%), corresponded to byproduct(s) with the proposed structure 7. Peaks at 22-25 min (ca 2-5%) possibly corresponded to the iso-form of the main product 4 and untreated Pd-Bpheid residues.

The solvent (aqueous methanol) was evaporated under reduced pressure. Then, the purified product 4 ]was re-dissolved in ~150 ml MeOH and filtered through cotton wool. The solvent was evaporated again and the solid pigment 4 was stored under Ar in the dark at -20°C. The reaction yield: ~90% (by weight, relative to 3).
The structure of product 4 was confirmed by electrospray mass spectroscopy. (ESI-MS, negative mode, Fig.2), (peaks at 875 (M–K-H), 859 (M–2K-H+Na), 837 (M–2K), 805 (M2K-H-OMe), 719) and 1H-NMR spectrum (Fig. 4 in MeOH-d4). Table 4 provides the shifts (in ppm units) of the major NMR peaks.
Optical absorption (UN-VIS) spectrum (MeOH): λ, 747 (1.00), 516 (0.13), 384 (0.41), 330 (0.50); ε747 (MeOH) is 1.2 x 105 mol-1 cm _1.
ΝMR (MeOH-d4): 9.38 (5-H, s), 8.78 (10-H, s), 8.59 (20-H, s), 5.31 and 4.95 (151-CH2, dd), 4.2-4.4 (7,8,17,18-H, m), 3.88 (153-Me, s), 3.52 (21-Me, s), 3.19 (121 -Me, s), 3.09 (32-Me, s), 1.92-2.41, 1.60-1.75 (171, 172-CH2, m), 2.19 (81-CH2, m), 1.93 (71-Me, d), 1.61 (181-Me, d), 1.09 (82-Me, t), 3.62, 3.05 (CH2‘s of taurine).
Octanol/water partition ratio is 40:60.
Example 2. Preparation of 31-oxo-15-methoxycarbonylmethyl- Rhodobacteriochlorin 131-(2-sulfoethyl)amide dipotassium salt (Compound 5) One hundred and sixty (160) mg of taurine were dissolved in 5 ml of 1M
K2HPO4 buffer, and the pH of the solution was adjusted to 8.2. This solution was added to 120 mg of compound 2 dissolved in 15 ml of DMSO, and the reaction and following purification were analogous to those described in previous Example.
Absorption spectrum (MeOH): λ, 750 (1.00), 519 (0.30), 354 (1.18) nm.
ESI-MS (-): 734 (M–2K).
ΝMR (MeOH-d4): 9.31 (5-H, s), 8.88 (10-H, s), 8.69 (20-H, s), 5.45 and 5.25 (151-CH2, dd), 4.35 (7,18-H, m), 4.06 (8,17-H, m), 4.20 and 3.61 (2-CH2, m of taurine), 3.83 (153-Me, s), 3.63 (21-Me, s), 3.52 (3-CH2, m oftaurine), 3.33 (121-Me, s), 3.23 (32-Me, s), 2.47 and 2.16 (171-CH2, m), 2.32 and 2.16 (81-CH2, m), 2.12 and 1.65 (172-CH2, m), 1.91 (71-Me, d), 1.66 (181– Me, d), 1.07 (82-Me, t).
Octanol/water partition ratio is 60:40.
Example 3. Preparation of copper(II) 31-oxo-15-methoxycarbonylmethyl- Rhodobacteriochlorin 131-(2-sulfoethyl)amide dipotassium salt (Compound 10)
Fifty (50) mg of compound 5 of Example 2 and 35 mg of copper (II) acetate were dissolved in 40 ml of methanol, and argon was bubbled into solution for 10 minutes. Then 500 mg of palmitoyl ascorbate was added, and the solution was stirred for 30 min. The absorption spectrum was characterized by a Qy transition shift from 750 nm (for 5) to 768 nm (for the product 10) and by Qx shift from 519 nm of 5 to 537 nm (of the product 10). Then the reaction mixture was evaporated, re-dissolved in acetone and filtered through cotton wool to get rid of acetate salt excess. The acetone was evaporated and the product was additionally purified by HPLC at the conditions mentioned above with the elution profile, described in Table 2.
The solvent (aqueous methanol) was evaporated under reduced pressure. Then, the purified pigment 10 was re-dissolved in methanol and filtered through cotton wool. The solvent was evaporated again and the solid pigment 10 was stored under Ar in the dark at -20°C. Reaction yield: -90%.

Absorption spectrum (MeOH): λ, 768 (1.00), 537 (0.22), 387 (0.71) and 342 (0.79) nm.
ESI-MS (-): 795 (M–2K).
Octanol/water partition ratio is 40:60.
Example 4. Preparation of zinc 31-oxo-15-methoxycarbonylmethyl-Rhodobacteriochlorin 131-(2-sulfoethyl)amide dipotassium salt (Compound 11)
Zn insertion into compound 5 was carried out with Zn acetate in acetic acid as previously described (US Patent No. 5,726,169). Final purification was carried out by HPLC in the same conditions as for compound 5 in Example 2 above.
Absorption spectrum (MeOH): λ, 762 (1.00), 558 (0.26), 390 (0.62) and 355 (0.84) nm.
Octanol/water partition ratio is 50:50.
Example 5. Preparation of manganese(III) 31-oxo-15-methoxycarbonylmethyl-Rhodobacteriochlorin 131-(2-sulfoethyl)amide dipotassium salt (Compound 12)
Mn insertion into compound 5 was carried out with Zn acetate in acetic acid as previously described (WO 97/19081; US 6,333,319) with some modifications. Thus, fifty (50) mg of compound 5 in 10 ml of DMF were stirred with 220 mg of cadmium acetate and heated under argon atmosphere at 110°C about 15 min (Cd-complex formation is monitored by shifting Qx transition absorption band from 519 to 585 nm in acetone). Then the reaction mixture was cooled and evaporated. The dry residue was re-dissolved in 15 ml of acetone and stirred with manganese (II) chloride to form the Mn(III)-product 12. The product formation is monitored by shifting Qx transition band from 585 to 600 nm and Qy transition band from 768 to 828 nm in acetone. The acetone was evaporated and the product 12 was additionally purified by HPLC in the conditions mentioned in Example 2 above with the elution profile described in Table 3 below where the] solvent system consists of: A – 5% aqueous methanol, B -methanol.

The solvent (aqueous methanol) was evaporated under reduced pressure and the solid pigment 12 was stored under Ar in the dark at -20°C.
Absorption spectrum (MeOH): λ, 828 (1.00), 588 (0.32) and 372 (0.80) nm. Octanol/water partition ratio is 5:95.
Example 6. Preparation of palladium bacteriopheophorbide a 17 -(3-sulfo-1-oxy- succinimide)ester sodium salt (Compound 6)
Fifty (50) mg of Pd-Bpheid (compound 2), 80 mg of N-hydroxy- sulfosuccinimide (sulfoNHS) and 65 mg of 1-(3-dimethylaminopropyl)-3- ethylcarbodiimide (EDC) were mixed in 7 ml of dry DMSO for overnight at room temperature. Then the solvent was evacuated under reduced pressure. The dry residue was re-dissolved in chloroform (ca. 50 ml), filtered from insoluble material, and evaporated. The conversion was ab. 95%) (TLC). The product 6 was used later on without further chromatographic purification. ESI-MS (-): 890 (M–Na).
NMR (CDCl3): 9.19 (5-H, s), 8.49 (10-H, s), 8.46 (20-H, s), 5.82 (132-H, s), 4.04- 4.38 (7,8,17,18-H, m), 3.85 (134-Me, s), 3.47 (21-Me, s), 3.37 (^-Me, s), 3.09 (32– Me, s), 1.77 (71-Me, d), 1.70 (lδ’-Me, d), 1.10 (82-Me, t), 4.05 (CH2 of sNHS), 3.45 (CH ofs NHS).
Example 7. Preparation of palladium bacteriopheophorbide a 173-(3-sulfopropyl) amide potassium salt (Compound 7)
Ten (10) mg of compound 6 in 1 ml of DMSO was mixed with 20 mg of homotaurine (3-amino-1-propane-sulfonic acid) in 1 ml of 0.1 M K-phosphate buffer, pH 8.0 for overnight. Then the reaction mixture was partitioned in chloroform/water. The organic layer was dried over anhydrous sodium sulfate and evaporated. The dry residue was re-dissolved in chloroform-methanol (19:1) and applied to a chromatographic column with silica. The product 7 was obtained with chloroform-methanol (4:1) elution. The yield was about 80-90%.
ESI-MS (-): 834 (M-K) m/z.
NMR (MeOH-d4): 9.16 (5-H, s), 8.71 (10-H, s), 8.60 (20-H, s), 6.05 (132-H, s), 4.51, 4.39, 4.11, 3.98 (7,8,17,18-H, all m), 3.92 (134-Me, s), 3.48 (21-Me, s), 3.36 (121-Me, s), 3.09 (32-Me, s), 2.02-2.42 (171 arid 172-CH2, m), 2.15 ( 81-CH2, q), 1.81 (71-Me, d), 1.72 (181-Me, d), 1.05 (82-Me, t), 3.04, 2.68, and 2.32 (CH2‘s of homotaurine, m).
Example 8. Preparation of palladium 31-oxo-15-methoxycarbonylmethyl-Rhodo-bacteriochlorin 13 ,17 -di(3-sulfopropyl)amide dipotassium salt (Compound 8)
Ten (10) mg of compound 6 or 7 were dissolved in 3 ml of DMSO, mixed with 100 mg of homotaurine in 1 ml of 0.5 M K-phosphate buffer, pH 8.2, and incubated overnight at room temperature. The solvent was then evacuated under reduced pressure as described above, and the product 8 was purified on HPLC. Yield: 83%.
Absorption spectrum (MeOH): 747 (1.00), 516 (0.13), 384 (0.41), 330 (0.50), ε747 =1.3×105 modern-1.
ESI-MS(-):1011 (M–K), 994 (M–2K+Na),972 (M–2K), 775 (M–2K-CO2Me-homotaurineNHCH2CH2CH2SO3), 486 ([M-2K]/2)
NMR (MeOH-d4): 9.35 (5-H, s), 8.75 (10-H, s), 8.60 (20-H, s), 5.28 and 4.98 (15-1-CH2, dd), 4.38, 4.32, 4.22, 4.15 (7,8,17,18-H, all m), 3.85 (15~3-Me, s), 3.51 (21-Me, s), 3.18 (121-Me, s), 3.10 (32-Me, s 2.12-2.41 (171-CH2, m), 2.15-2.34 (81-CR2, m), 1.76-2.02 (172-CH2, m), 1.89 (71-Me, d), 1.61 (lδ^Me, d), 1.07 (82-Me, t). 3.82, 3.70,
3.20, 3.10, 2.78, 2.32, 1.90 (CH2‘s of homotaurine at C-131 and C-173)
Example 9. Palladium 31-(3-sulfopropylimino)-15-methoxycarbonylmethyl-Rhodo-bacteriochlorin 131,173-di(3-sulfopropyl)amide tripotassium salt (Compound 9)
Compound 9 was obtained from HPLC as a minor product during synthesis of 8.
Absorption spectrum (MeOH): 729 (1.00), 502 (0.10), 380 (0.69), 328 (0.57).
ESI-MS (30.4.2000): 1171 (M-K+H), 1153 (M–2K-H+Na), 1131 (M-2K), 566 ([M-K]/2), 364 ([M-3K]/3).
NMR (MeOH-d4): 8.71 (1H), 8.63 (1.5H), 8.23 (0.5H) (5-, 10- and 20-H, all-m), 5.30 and 4.88 (151-CH2, dd), 4.43 and 4.25 (7,8,17,18-H, m), 3.85 (15~3-Me, s), 3.31 (21-Me, s), 3.22 (121-Me, s), 3.17 (32-Me, m), 1.89-2.44 (171 and 172-CH2, m), 2.25 (81-CH2, m), 1.91 (71-Me, s), 1.64 (181– Me, s), 1.08 (82-Me, t), 4.12, 3.56, 3.22, 3.16, 2.80 and 2.68 (CH2‘s of homotaurine).
Example 10. Palladium 31-oxo-15-methoxycarbonylmethyl-Rhodobacteriochlorin 131-(2-sulfoethyl)amide, 173-(N-immunoglobulin G)amide potassium salt (Compound 13)
Ten (10) mg of compound 4 were reacted with 20 mg of sulfo-NHS and 15 mg of EDC in 1 ml of dry DMSO for 1 hour at room temperature, then rabbit IgG (0.6 mg) in PBS (2.5 ml) was added, and the mixture was further incubated overnight at room temperature. The mixture was evaporated to dryness, then re-dissolved in 1 ml of PBS and loaded on Sephadex G-25 column equilibrated with PBS. A colored band was eluted with 4-5 ml of PBS. The pigment/protein ratio in the obtained conjugate 13 was determined by optical density at 753 and 280 mn, respectively, and varied between 0.5/1 to 1/1 of pigment 13/protein.
Example 11. Preparation of palladium 31-oxo-15-methoxycarbonylmethyl-Rhodobacteriochlorin 131-(2-carboxyethyl)amide dipotassium salt (Compound
M)
The preparation and purification of the title compound 14 were carried out as described in Example 2, by reaction of compound 2 with 3-aminopropionic acid (β-alanine) (150 mg) instead of taurine. Yield: 85%.
Example 12. Preparation of palladium 31-oxo-15-methoxycarbonylmethyl-Rhodobacteriochlorin 131-(3-phosphopropyl)amide tripotassium salt (Compound
15)
The preparation and purification of the title compound 15 were carried out as described in Example 2, by reaction of compound 2 with 3 -amino- 1-propanephosphonic acid (180 mg) instead of taurine. Yield: 68%.
Example 13. Palladium 31-(3-sulfopropylamino)-15-methoxycarbonylmethyl-Rhodobacteriochlorin 131,173-di(3-sulfopropyl)amide tripotassium salt (Compound 16)
For reduction of the imine group in 31-(3-sulfopropylimino) to the correspondent 31-(3-sulfopropylamino) group, compound 9 (8 mg) was reacted by stirring with sodium cyanoborohydride (15 mg) in 5 ml of methanol overnight at room temperature. Then the reaction mixture was treated with 0.05 M HCl (5 ml), neutralized with 0.01 M KOH, and evaporated. The title product 16 was purified using HPLC conditions as described in Example 2. Yield: 80-90%).
REFERENCES
1: Kessel D, Price M. Evaluation of DADB as a Probe for Singlet Oxygen Formation during Photodynamic Therapy. Photochem Photobiol. 2012 Feb 2. doi: 10.1111/j.1751-1097.2012.01106.x. [Epub ahead of print] PubMed PMID: 22296586.
2: Betrouni N, Lopes R, Puech P, Colin P, Mordon S. A model to estimate the outcome of prostate cancer photodynamic therapy with TOOKAD Soluble WST11. Phys Med Biol. 2011 Aug 7;56(15):4771-83. Epub 2011 Jul 13. PubMed PMID: 21753234.
3: Chevalier S, Anidjar M, Scarlata E, Hamel L, Scherz A, Ficheux H, Borenstein N, Fiette L, Elhilali M. Preclinical study of the novel vascular occluding agent, WST11, for photodynamic therapy of the canine prostate. J Urol. 2011 Jul;186(1):302-9. Epub 2011 May 20. PubMed PMID: 21600602.
4: Dandler J, Wilhelm B, Scheer H. Photochemistry of bacteriochlorophylls in human blood plasma: 1. Pigment stability and light-induced modifications of lipoproteins. Photochem Photobiol. 2010 Mar-Apr;86(2):331-41. Epub 2009 Nov 23. PubMed PMID: 19947966.
5: Dandler J, Scheer H. Inhibition of aggregation of [Pd]-bacteriochlorophyllides in mesoporous silica. Langmuir. 2009 Oct 20;25(20):11988-92. PubMed PMID: 19772311.
6: Ashur I, Goldschmidt R, Pinkas I, Salomon Y, Szewczyk G, Sarna T, Scherz A. Photocatalytic generation of oxygen radicals by the water-soluble bacteriochlorophyll derivative WST11, noncovalently bound to serum albumin. J Phys Chem A. 2009 Jul 16;113(28):8027-37. PubMed PMID: 19545111.
7: Moore CM, Pendse D, Emberton M. Photodynamic therapy for prostate cancer–a review of current status and future promise. Nat Clin Pract Urol. 2009 Jan;6(1):18-30. Review. PubMed PMID: 19132003.
8: Preise D, Oren R, Glinert I, Kalchenko V, Jung S, Scherz A, Salomon Y. Systemic antitumor protection by vascular-targeted photodynamic therapy involves cellular and humoral immunity. Cancer Immunol Immunother. 2009 Jan;58(1):71-84. Epub 2008 May 17. PubMed PMID: 18488222.
9: Fleshker S, Preise D, Kalchenko V, Scherz A, Salomon Y. Prompt assessment of WST11-VTP outcome using luciferase transfected tumors enables second treatment and increase in overall therapeutic rate. Photochem Photobiol. 2008 Sep-Oct;84(5):1231-7. Epub 2008 Apr 8. PubMed PMID: 18399928.
10: Berdugo M, Bejjani RA, Valamanesh F, Savoldelli M, Jeanny JC, Blanc D, Ficheux H, Scherz A, Salomon Y, BenEzra D, Behar-Cohen F. Evaluation of the new photosensitizer Stakel (WST-11) for photodynamic choroidal vessel occlusion in rabbit and rat eyes. Invest Ophthalmol Vis Sci. 2008 Apr;49(4):1633-44. PubMed PMID: 18385085.
11: Fabre MA, Fuseau E, Ficheux H. Selection of dosing regimen with WST11 by Monte Carlo simulations, using PK data collected after single IV administration in healthy subjects and population PK modeling. J Pharm Sci. 2007 Dec;96(12):3444-56. PubMed PMID: 17854075.
12: Brandis A, Mazor O, Neumark E, Rosenbach-Belkin V, Salomon Y, Scherz A. Novel water-soluble bacteriochlorophyll derivatives for vascular-targeted photodynamic therapy: synthesis, solubility, phototoxicity and the effect of serum proteins. Photochem Photobiol. 2005 Jul-Aug;81(4):983-93. PubMed PMID: 15839743.
13: Mazor O, Brandis A, Plaks V, Neumark E, Rosenbach-Belkin V, Salomon Y, Scherz A. WST11, a novel water-soluble bacteriochlorophyll derivative; cellular uptake, pharmacokinetics, biodistribution and vascular-targeted photodynamic activity using melanoma tumors as a model. Photochem Photobiol. 2005 Mar-Apr;81(2):342-51. PubMed PMID: 15623318.
14: Plaks V, Posen Y, Mazor O, Brandis A, Scherz A, Salomon Y. Homologous adaptation to oxidative stress induced by the photosensitized Pd-bacteriochlorophyll derivative (WST11) in cultured endothelial cells. J Biol Chem. 2004 Oct 29;279(44):45713-20. Epub 2004 Aug 31. PubMed PMID: 15339936.
////////PADELIPORFIN, WST11, WST-11, WST 11, Stakel, padeliporfin, palladiumbacteriopheophorbide monolysine taurine, EU 2017, EMA 2017
CCC1C(C2=NC1=CC3=C(C(=C([N-]3)C(=C4C(C(C(=N4)C=C5C(=C(C(=C2)[N-]5)C(=O)C)C)C)CCC(=O)O)CC(=O)OC)C(=O)NCCS(=O)(=O)O)C)C.[Pd+2]

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....