Home » Posts tagged 'searle'
Tag Archives: searle
I did not run away from a NaCN Exotherm
DELTAMETHRIN
DID NOT RUN AWAY FROM NACN ie sodium cyanide EXOTHERM
ALMOST VIRTUAL ACCIDENT AT ISAGRO.RPG LIFESCIENCES (SEARLE) PANOLI GUJARAT INDIA 1999-2000
DELTAMETHRIN PROJECT, 1999-2000 Panoli Gujarat India
ww were trying to add acid chloride into an aldehyde at zero degrees cent using PTC conditions and one of ingredient was sodium cyanide, cooling was done by brine
We Did not run away when instead of adding acid chloride in 2 hrs the operator added it on 10 min…………..I waited at the reactor and controlled an exotherm in plant by switching off brine supply to other reactors,
The reaction got controlled at 59 deg cent and luckily was ok…………the exotherm was fearful. This extreme tension is experienced in a lifetime when everyone runs away and no one to help, FEARFUL, In this case the ‘I’ worked not the ‘we’
Despite all odds God saves us
 MAKE IN INDIA
MAKE IN INDIA
http://makeinindia.com/
http://makeinindia.com/sector/pharmaceuticals/

THANKS AND REGARD’S
DR ANTHONY MELVIN CRASTO Ph.D
web link

New Drug Approvals, ALL ABOUT DRUGS, WORLD DRUG TRACKER
MEDICINAL CHEM INTERNATIONAL, DRUG SYN INTERNATIONAL
SCALEUP OF DRUGS, ALL FOR DRUGS ON WEB,
MY CHINA, VIETNAM AND JAPAN BLOGS
ICELAND, RUSSIA, ARAB
BOBRDOBR, BLAND ICELAND, 100zakladok, adfty
GROUPS
you can post articles and will be administered by me on the google group which is very popular across the world
OPD GROUPSPACES, SCOOP OCI, organic-process-development GOOGLE, TVINX, MENDELEY WDT, SCIPEOPLE OPD, EPERNICUS OPD, SYNTHETIC ORGANIC CHEMISTRYLinkedIn group,DIIGO OPD, LINKEDIN OPD, WDT LINKEDIN, WDTI ZING

![]()


 I was paralysed in dec2007, Posts dedicated to my family, my organisation Glenmark, Your readership keeps me going and brings smiles to my family
I was paralysed in dec2007, Posts dedicated to my family, my organisation Glenmark, Your readership keeps me going and brings smiles to my familyDrug spotlight, Celecoxib from G. D. Searle Company


CELECOXIB
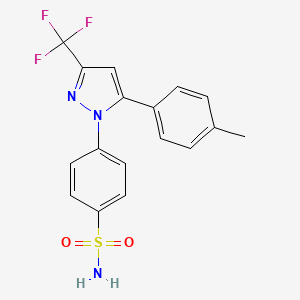
4-[5-(4- methylphenyl)-3-(trifluoromethyl)-lH-pyrazol-l-yl]benzenesulfonamide
169590-42-5
mp…157-159 deg C
Celecoxib is a sulfonamide non-steroidal anti-inflammatory drug (NSAID) and selective COX-2 inhibitor used in the treatment of osteoarthritis, rheumatoid arthritis, acute pain, painful menstruation and menstrual symptoms, and to reduce numbers of colon and rectum polyps in patients with familial adenomatous polyposis. It is marketed by Pfizer. It is known under the brand name Celebrex or Celebra for arthritis and Onsenal for polyps. Celecoxib is available by prescription in capsule form.
Celecoxib was discovered developed by G. D. Searle & Company and was approved by the FDA on December 31, 1998. It was co-promoted by Monsanto Company (parent company of Searle) and Pfizer under the brand name Celebrex. Monsanto merged with Pharmacia, from which the Medical Research Division was acquired by Pfizer, giving Pfizer ownership of Celebrex. The drug was at the core of a major patent dispute that was resolved in Searle’s favor (later Pfizer) in 2004. In University of Rochester v. G.D. Searle & Co., 358 F.3d 916 (Fed. Cir. 2004), the University of Rochester claimed that United States Pat. No. 6,048,850 (which claimed a method of inhibiting COX-2 in humans using a compound, without actually disclosing what that compound might be) covered drugs such as celecoxib. The court ruled in favor of Searle, holding in essence that the University had claimed a method requiring, yet provided no written description of, a compound that could inhibit COX-2 and therefore the patent was invalid.

After the withdrawal of rofecoxib (Vioxx) from the market in September 2004, Celebrex enjoyed a robust increase in sales. However, the results of the APC trial in December of that year raised concerns that Celebrex might carry risks similar to those of Vioxx, and Pfizer announced a moratorium on direct-to-consumer advertising of Celebrex soon afterwards. After a significant drop, sales of Celebrex have recovered, and reached $2 billion in 2006.[6] Pfizer resumed advertising Celebrex in magazines in 2006, and resumed television advertising in April 2007 with an unorthodox, 2 1⁄2-minute advertisement which extensively discussed the adverse effects of Celebrex in comparison with other anti-inflammatory drugs. The ad drew criticism from the consumer advocacy group Public Citizen, which called the ad’s comparisons misleading. Pfizer has responded to Public Citizen’s concerns with assurances that they are truthfully advertising the risk and benefits of Celebrex as set forth by the FDA.
In late 2007, Pfizer released another U.S. television ad for Celebrex, which also discussed celecoxib’s adverse effects in comparison with those of other anti-inflammatory drugs.
Daniel L. Simmons of Brigham Young University, who discovered the COX-2 enzyme, is suing Pfizer to be credited with discovery of the technique in 1989 that eventually led to the drug, and for $1 billion USD. The company has made about $30 billion from the drug as of 2006. A settlement was finally reached in April 2012.
Celecoxib is licensed for use in osteoarthritis, rheumatoid arthritis, acute pain, painful menstruation and menstrual symptoms, ankylosing spondylitis and to reduce the number of colon and rectal polyps in patients with familial adenomatous polyposis. It was originally intended to relieve pain while minimizing the gastrointestinal adverse effects usually seen with conventional NSAIDs. In practice, its primary indication is in patients who need regular and long term pain relief; there is probably no advantage to using celecoxib for short term or acute pain relief over conventional NSAIDs, except in the situation where non-selective NSAIDs or aspirin cause cutaneous reactions (urticaria or “hives”). In addition, the pain relief offered by celecoxib is similar to that offered by paracetamol (acetaminophen).
Synthesis
https://www.google.com/patents/WO2010095024A2?cl=en
US 5,466,823, also discloses a process for the preparation of Celecoxib, which comprises reacting 4-methylacetophenone (II) with 1-ethyltrifluoroacetate (III) in the presence of methyl t-butyl ether and sodium methoxide, followed by recrystallisation from isooctane to produce l-(4-methylphenyl)-4,4,4-trifluorobutane-l ,3-dione (IV), which is further condensed with 4-hydrazinophenylsulfonamide hydrochloride (V) in the presence of ethanol to produce crude Celecoxib, which is recrystallised from ethyl acetate and isooctane to give Celecoxib (I),
The process is as shown in Scheme -I below:
HI rv
The synthesis of celecoxib was first described in 1997 by a team of researchers at Searle Research and Development. Celecoxib is synthesized by a Claisen condensation reaction of an acetophenone with N-(trifluoroacetyl)imidazole catalyzed by the strong base, sodium bis(trimethylsilyl)amide to produce a 1,3-dicarbonyl adduct. Condensation of the diketone with (4-sulfamoylaphenyl)hydrazine produces the 1,5-diarylpyrazole drug moiety.
Scheme-I The above process involves isolation of the intermediate l-(4-methylphenyl)-4,4,4- tiϊfluorobutane-l ,3-dione (IV) by crystallization, before condensing with 4- sulphonamido-phenylhydrazine, which adds to the cost and complexity of the synthesis.
Further, the above process proceeds with less selectivity to Celecoxib, which is having about 4 wt. % of regioisomer (VI) by-product under commercial conditions.
US 6, 150,534 discloses a process for the preparation of Celecoxib, which comprises, condensing l-(4-methylphenyl)-4,4,4-trifluorobutane-l ,3-dione (IV) with 4- sulphonamido-phenylhydrazine in presence of an amide solvent at controlled temperature to produce amide solvate of Celecoxib, which is further desolvated by recrystallization from isopropanol and water.
The above process also involves isolation of the intermediate l-(4-methylphenyl)- 4,4,4-trifluorobutane-l ,3-dione (IV) by crystallization, before condensing with 4- sulphonamido-phenylhydrazine,
US 5,892,053 discloses a process for the preparation of Celecoxib by condensing 4- methylacetophenone (II) with 1-ethyltrifluoro acetate (III) to produce l-(4- methylphenyl)-4,4,4-trifluoiObutane-l ,3-dione (IV), which is further reacted with 4- hydrazinophenylsulfonamide (V) in presence of aqueous mixture of alcohol and acid to produce Celecoxib.
US 6,579,988 discloses a preparation of Celecoxib via novel intermediate compound of formula VII. Formula VII
US 2007/0004924 Al discloses a process for the preparation of Celecoxib by condensing l-(4-methylphenyl)-4,4,4-trifluorobutane-1.3-dione (IV) with 4- hydrazinophenylsulfonamide (V) in presence of a solvent system containing an organic solvent, the salt of the 4-sulphonamidophenylhydrazine having a solubility in the organic solvent at least 0.05 M. ‘
US 2008/0234491 Al discloses the condensation of l-(4-methylphenyl)-4,4,4- trifluorobutane-l,3-dione (IV) with 4-hydrazinophenylsulfonamide (V) or its acid addition salts in the presence of a solvent medium comprising an alkyl ester, water or mixtures thereof to produce Celecoxib. Further, crystallization of crude Celecoxib is carried out in toluene alone.
l-(4-Methylphenyl)-4,4,4-trifluorobutane-l,3-dione (IV) is condensed with 4- hydrazinophenylsulfonamide (V) or its acid addition salt in a solvent selected from water, inert organic solvent to produce 4-[5-(4-methylphenyl)-3-(trifluoiOmethyl)-lH- pyrazol-l-yl]benzenesulfonamide (Celecoxib) of Formula I. The acid addition salts of compound of the formula IV includes, but are not limited to, hydrochloride, hydrobromide, sulfate, nitrate, oxalate, mesylate, methane sulfonate, and tartrate, preferably, hydrochloride salt. The suitable inert organic solvents for the above reaction include but are not limited to ketone solvents, such as acetone, methyl ethyl ketone, methyl isobutyl ketone, n-butanone, and tertiary-butyl ketone; nitrile solvents, such as acetonitrile. and propionitrile; halogenated solvents, such as dichloromethane, ethylene dichloride, and chloroform; esters, such as ethyl acetate, n-propylacetate, isopropyl acetate, and tertiary-butyl acetate; aprotic polar solvents, such as N,N- dimethylformamide, dimethylsulfoxide, and N,N-dimethylacetamide; ethers, such as diisopropyl ether, tetrahydrofuran and 1,4-dioxane; hydrocarbon solvents, such as cyclohexane, toluene and xylene; and mixtures thereof. The preferred solvent is water. The reaction may be performed at a temperature ranging from about 25°C to about reflux temperature of the solvent or mixture of solvents used for the reaction. The above reaction is conducted in presence of an acid selected from aqueous hydrochloric acid, aqueous sulfuric acid, p-toluene sulfonic acid, trifluoroacetic acid, and acetic acid to maintain the pH of the reaction mixture is below 7. More preferably, aqueous HCl is added. Crude Celecoxib (I) produced may be isolated by precipitation of compound from the reaction mixture, which may be performed by cooling the reaction mixture, followed by addition of an organic solvent selected from alcohols such as methanol, ethanol, isopropanol or aromatic hydrocarbons such as toluene, xylene, ethyl benzene and mixtures thereof solvents. The preferred solvent is mixture of methanol and toluene.
It has been observed that preparation of Celecoxib (I) using above reaction conditions results in regioisomer of compound (VI) to less than 2.5% by HPLC analysis.
EXAMPLE 1
Stage-1:
Preparation of l-(4-methylphenyl)-4,4,4-trifluorobutane-l,3-dione (IV)
4-Methylacetophenone (50 g, 0.373 mol) was dissolved in toluene (250 ml) and 30% methanolic sodium methoxide solution (80.6 g, 0.447 mol), followed by 1- ethyltrifluoro acetate (63.58 g, 0.447 mol) were added at 25-3O0C. Temperature of the reaction mass was raised to 55-600C and stirred ~ 4 hr to complete the reaction. The reaction mass was cooled to 20-250C and washed with 10% aqueous hydrochloric acid (200 ml). The layers were separated and concentrated the organic layer at 50-550C under reduced pressure to produce 80 g of l-(4-methylphenyl)-4,4,4-trifluoiObutane- 1,3-dione (IV) as an oily mass.
Stage-2:
Preparation of 4-[5-(4-methylphenyl)-3-(trifluorornethyl)-lh-pyrazol-l- yljbenzenesulfonamide (Celecoxib) (I) l-(4-Methylphenyl)-4,4,4-trifluorobutane-l,3-dione (IV) (80 g, 0.348 mol), 4- hydrazinophenylsulfonamide (V) (77.74 g, 0.348 mol) and concentrated hydrochloric acid (18.6 g) were added to DM water (500 ml) and heated to 98-1000C. The mass was stirred for 4 hr to complete the reaction. The reaction mass was cooled to 70-75 C and a mixture of toluene (600 ml) and methanol (10 ml) was added to the reaction mass. After 1 hr stirring at 70-750C, the reaction mass was cooled to 20-250C, the product was filtered and λvashed with toluene (100 ml) followed by DM water (200 ml). The product obtained was dried at 55-600C under reduced pressure to produce 1 15 g of Celecoxib crude. Chromatographic purity: 99%(by PTPLC, by area normalization)
……………………..
The synthesis of celecoxib was first described in 1997 by researchers at Searle Research and Development. It is synthesized by a Claisen condensation reaction of an acetophenone with N-(trifluoroacetyl)imidazole catalyzed by the strong base, sodium bis(trimethylsilyl)amide to produce a 1,3-dicarbonyl adduct. Condensation of the diketone with (4-sulfamoylaphenyl)hydrazine produces the 1,5-diarylpyrazole drug moiety.
Penning TD, Talley JJ, Bertenshaw SR, Carter JS, Collins PW, Docter S, Graneto MJ, Lee LF, Malecha JW, Miyashiro JM, Rogers RS, Rogier DJ, Yu SS, Burton EG, Cogburn JN, Gregory SA, Koboldt CM, Perkins WE, Seibert K, Veenhuizen AW, Zhang YY, Isakson PC (1997). “Synthesis and Biological Evaluation of the 1.5 Diarylpyrazole Class of Cyclooxygenase-2 Inhibitors: Identification of 4-[5-(4-Methylphenyl)-3-(trifluoromethyl)-1H-pyrazole-1-yl]benzenesulfonamide (SC-58634, Celecoxib)”. Journal of Medicinal Chemistry40 (9): 1347–1365. doi:10.1021/jm960803q. PMID 9135032.

……………………………….


- The condensation of 4-methylacetophenone (I) with ethyl trifluoroacetate (II) by means of NaOMe in refluxing methanol gives 4,4,4-trifluoro-1-(4-methylphenyl)butane-1,3-dione, which is cyclized with 4-hydrazinophenylsulfonamide (III) in refluxing ethanol.
…………………
http://www.google.com/patents/US7759497
In U.S. Pat. Nos. 5,892,053 and 5,910,597, Zhi et al. describe a scalable two step process for the preparation of pyrazoles from the condensation of diketones and hydrazines. In the first step, a diketone is formed by the treatment of a ketone with base and ester in a suitable solvent. In the second step, the diketone is solubilized in an aqueous alcohol and condensed with a hydrazine to form the pyrazole product. This two step process has been used on a commercial scale for the preparation of celecoxib (4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyrazole-1-yl]benzenesulfonamide) sold under the trademark CELEBREX® by Pharmacia Corporation as shown in the following reaction:
While this synthetic approach proceeds with high selectivity to celecoxib, about 2-5 wt. % of regioisomer and hydroxyregioisomer by-products are formed under commercial conditions.
The regioisomer and hydroxyregioisomer by-products must be separated from celecoxib in a purification step to enable the celecoxib to meet purity requirements. The separation is typically done through a crystallization step in which celecoxib preferentially crystallizes while the regioisomer and hydroxyregioisomer by-products predominantly remain in solution. The celecoxib crystals are then removed from the resultant slurry and separated from impurities by solid-liquid separation techniques known to those skilled in the art, such as centrifugation or filtration.
Under commercial conditions used to date, of the two by-products, regioisomer is selectively formed over hydroxyregioisomer. This is problematic, however, since the regioisomer is generally more difficult to separate through crystallization from celecoxib than is the hydroxyregioisomer, and regioisomer concentrations of greater than about 1% typically require two crystallizations to achieve desired celecoxib purity. The second crystallization adds time to the manufacturing process and thus negatively impacts product throughput. Additionally, a second crystallization reduces yield as some celecoxib remains uncrystallized and is not recovered from the liquid phase.

Example 7Preparation of Celecoxib with Hydrazine Reactant Containing Water
To a 250 mL reactor which had been purged with nitrogen and which had been fitted with a mechanical stirrer and a chilled condenser was charged while stirring, isopropyl alcohol (50.75 g), ethyltrifluoroacetate (37.95 g), sodium methoxide (25% in methanol, 53.28 g) and 4′-methylacetophenone (27.43 g). The reaction mixture was heated to 50-55° C. and held for at least 2 hours. To a separate 1 L reactor which had been purged with nitrogen and fitted with a mechanical stirrer and a chilled condenser, was charged 4-SAPH•HCl (45.96 g), isopropyl alcohol (101.2 g), water (74 g) and trifluoroacetic acid (23.43 g). The 4-SAPH•HCl was heated to 50° C. with agitation. At the completion of the 2 hour reaction period, the contents of the first reactor was transferred to the second reactor containing the 4-SAPH•HCl over a period of at least five minutes and the reaction mixture was then brought to 55° C. and maintained at that temperature for at least 30 minutes. The pH of the reaction mixture was then adjusted to be within the range of 3 to 9 followed by the addition of water (95 g). The contents were then heated to 65° C. and the pH was again adjusted to be within the range of 3 to 9. Water (90 g) was then added to the mixture over a time period of about 20 minutes while maintaining the temperature at about 65° C. The reaction mixture was then cooled to about 20° C. over a period of 12 to 14 hours to produce celecoxib (62-65 g) with less than 0.05% regio-isomer and undetectable regioisomer.
Example 8Preparation of Celecoxib with Anhydrous Hydrazine Reactant
To a 250 mL reactor which had been purged with nitrogen and which had been fitted with a mechanical stirrer and a chilled condenser was charged while stirring, isopropyl alcohol (50.75 g), ethyltrifluoroacetate (37.95 g), sodium methoxide (25% in methanol, 53.28 g) and 4′-methylacetophenone (27.43 g). The reaction mixture was heated to 50-55° C. and held for at least 2 hours. To a separate 1 L reactor which had been purged with nitrogen and fitted with a mechanical stirrer and a chilled condenser, was charged 4-SAPH•HCl (45.96 g), isopropyl alcohol (101.2 g) and trifluoroacetic acid (23.43 g). The 4-SAPH•HCl was heated to 50° C. with agitation. At the completion of the 2 hour reaction period, the contents of the first reactor was transferred to the second reactor containing the 4-SAPH•HCl over a period of at least five minutes and the reaction mixture was then brought to 55° C. and maintained at that temperature for at least 30 minutes. The pH of the reaction mixture was then adjusted to be within the range of 3 to 9 followed by the addition of water (95 g). The contents were then heated to 65° C. and the pH was again adjusted to be within the range of 3 to 9. Water (90 g) was then added to the mixture over a time period of about 20 minutes while maintaining the temperature at about 65° C. The reaction mixture was then cooled to about 20° C. over a period of 12 to 14 hours to produce celecoxib (62-65 g) with less than 0.05% regio-isomer. Analysis of the reaction mixture prior to initiation of crystallization indicated that the regio-isomer content was less than 0.5 mole percent of the reaction products.
Example 9Preparation of Celecoxib by Addition of Diketone Salt to 4-SAPH-HCl
To a 250 mL reactor, fitted with a mechanical stirrer and maintained under a nitrogen atmosphere, was added isopropyl alcohol (54.8 g, 0.912 moles), ethyl trifluoroacetate (38.0 g, 0.267 moles) and 25% sodium methoxide in methanol (53.3 g, 0.246 moles). To the agitated reactor was added 4-methylacetophenone (27.6 g, 0.206 moles). The reaction mixture was heated to 50° C. and maintained for 2 hours. To a second (1 liter) reactor was added 4-sulphamidophenyl hydrazine hydrochloride (46.0 g, 0.206 moles), isopropyl alcohol (101.3 g, 1.685 moles) and trifluoroacetic acid (11.7 g, 0.103 moles) with stirring. The reaction mixture was heated to approximately 45° C. Upon completion of the 2-hour reaction period in the 250 mL reactor, the contents was added to the second reactor over approximately 10 minutes. The reaction mixture maintained at 55° C. for 30 minutes. The pH was adjusted with 50% aqueous sodium hydroxide to a pH of 5-6. The reaction mixture was heated to 65° C. and water was added (95 g, 5.3 moles). The pH was again adjusted with 50% aqueous sodium hydroxide to a value of 5-6. Water (90 g, 5.0 moles) was added over 20 minutes while maintaining the temperature at 65° C. The reaction mixture was then cooled over 9 hours to 20° C. The reaction mixture was filtered, washed twice with 50% aqueous isopropyl alcohol and dried in a vacuum over for 16 hours to yield celecoxib (65.6 g) whose HPLC retention time was identical to that of authentic celecoxib. Regio-isomer was not detected by HPLC.
Example 10Preparation of Celecoxib by the Addition of 4-SAPH-HCl to Diketone
To a 1 L reactor fitted with a mechanical stirrer and maintained under a nitrogen atmosphere, was added isopropyl alcohol (54.7 g, 0.912 moles), ethyl trifluoroacetate (37.7 g, 0.267 moles), and 25% sodium methoxide in methanol (53.3 g, 0.247 moles). To the agitated reactor was added 4-methylacetophenone (27.32 g, 0.205 moles). The reaction mixture was heated to 50° C. and maintained for 2 hours. Trifluoroacetic acid (36.69, 0.321 moles) was added to the reaction mixture over a period of five minutes. 4-SAPH-HCl (46.0 g, 0.205 moles) was added through a power addition funnel over a period of 10 minutes. The reaction mixture was brought to 55° C. and maintained for one hour. Isopropyl alcohol (81.5 g, 1.36 moles) was added followed by the addition of 50% sodium hydroxide (18.5 g, 0.231 moles) to achieve a pH of 7. Water (87.8 g, 4.88 moles) was added and the reaction mixture heated to 65° C. Water (90.0 g 5.00 moles) was added over ten minutes. The reaction mixture was cooled to 20° C. over nine hours. The slurry was filtered and washed twice with 50% (weight) aqueous isopropyl alcohol (100 g). The solid was dried in a vacuum oven for 16 hours to yield celecoxib (67.2 g) whose HPLC retention time was identical to that of authentic material. Regio-isomer was not detected by HPLC.

4-[5-(4-Methylphenyl)-3-(trifluoromethyl)-1H-pyrazol-1-yl]benzenesulfonamide (Celecoxib)
E-mail: prataprp@ drreddys.com. Fax: 914044346285. Telephone: 9989997176


HPLC Conditions:
 29.2 min
29.2 min 30.9 min
30.9 min11
Bioorganic and Medicinal Chemistry, , vol. 15, # 4 p. 1802 – 1807

![]()
..celecoxib
………
http://www.google.com/patents/WO2014012074A2?cl=en
4,4,4-Trifluoro-l-(p-tolyl)butane-l,3-dione (C01)
25% sodium methoxide in methanol (51.3 ml, 223.5 mmol) and ethyl
trifluoroacetate (24.4 ml, 204.9 mmol) were dissolved in 110 mL methyl tert-butyl ether under N2, at room temperature. 4′-methyl acetophenone (25.0 ml, 186.3 mmol) was added and stirred at room temperature overnight. The reaction was washed with 3M HC1 and dried over magnesium sulfate. The solution was then evaporated and the resulting oil dried under vacuum overnight. The resulting light orange crystalline solid was washed with cold isooctane and dried under vacuum to yield an off white crystalline solid (37.3 g, 87% yield). LC tr=3.49 minutes (C- 18 column, 5 to 95% acetonitrile/water over 6 minutes at 1.7 mL/min with detection 254 nm, at 23 °C).
4-(5-(p-Tolyl)-3-(trifluoromethyl)-lH-pyrazol-l-yl)benzenesulfonamide (C02)
] C01 (23.55 g, 102.3 mmol) was refluxed with 4-sulphonamidophenyl
hydrazine HCl (23.95 g, 127.9 mmol) in 700 mL ethanol overnight. The reaction was evaporated, dissolved in 700 mL ethyl acetate, washed with water and brine, dried over magnesium sulfate and evaporated to -100 mL ethyl acetate. The product was crystalized by the addition of ~ 400 mL isooctane. After 15 minutes, the white crystalline solid was broken up, washed with isooctane and dried under vacuum (35.15 g, 90% yield). 1H NMR (400 MHz, CDC13) δ 7.94-7.91 (m, 2H), 7.51-7.49 (m, 2H), 7.21-7.20 (m, 2H), 7.15-7.13 (m, 2H), 6.77 (s, 1H), 2.41 (s, 3H). LC tr=4.27 minutes (C-18 column, 5 to 95% acetonitrile/water over 6 minutes at 1.7 mL/min with detection 254 nm, at 23° C). ES(neg)MS m/z 380 (M-H calcd for C17H14F3N302S requires 380).
![]()

http://www.slideshare.net/kennytirorx/celebrex-ms-report
………………………………….
http://www.google.co.in/patents/WO2011055233A2?cl=en
The preparation and use as COX-2 inhibitors of benzenesulfonamide derivatives such as celecoxib is described in US Patent No. 5,466,823. Processes for the preparation of celecoxib are also described in U.S. Patent Nos. 5,760,068 and 5,910,597.
As per the process exemplified in the U.S. Patent No. 5,760,068 (hereinafter referred to as the Ό68 patent), celecoxib is prepared by the reaction of l-(4-methylphenyl)- 4,4,4-trifluorobutane-l,3-dione with 4-sulphonamidophenyl hydrazine hydrochloride in absolute ethanol at reflux temperature under argon for 24 hours. The resulting mass is cooled to room temperature, followed by filtering and concentrating the reaction mixture to afford an orange solid, which is then recrystallized from a solvent system containing methylene chloride/hexane to produce the product as a pale yellow solid (melting point: 157° – 159°C).
The recrystallization process for preparing celecoxib described in the ‘608 patent suffers from disadvantages since the recrystallization process requires large volumes of solvents (more than 20 volumes each of methylene chloride and hexane solvents per gram of celecoxib), which is not commercially and environmentally, advisable for scale up operations. Moreover, the use of methylene chloride is hazardous to the environment and human health. The use of n-hexane is not advisable because it causes an ignition and fire risk due to its electrostatic charge accumulation property.
PCT Publication No. WO 01/42222 (hereinafter referred to as the ‘222 application) discloses three polymorphic forms (Form I, Form II and Form III) of celecoxib, pharmaceutical compositions, and methods of use thereof. The crystalline forms are characterized by powder X-ray diffraction (P-XRD), differential scanning calorimetry (DSC) and Infrared (IR) spectroscopy. The ‘222 application further teaches that the crystalline Form III of celecoxib is more thermodynamically stable than Form I and Form II. The ‘222 application also teaches that crystalline Form III of celecoxib is produced by crystallization of celecoxib from a solvent comprising isopropanol and water, for example, as described in U.S. Patent No. 5,910,597.
According to the ‘222 application, the polymorphic Form I is characterized by an X-ray powder diffraction pattern having peaks expressed as 2-theta at about 5.5, 5.7, 7.2 and 16.6 degrees; a melting point of about 162.5°C to about 163°C; a differential scanning calorimetry (DSC) endotherm maximum at about 163.3°C; and an Infra Red (IR) spectrum with peaks at about 3256 and 3356 cm-1. The polymorphic Form II is characterized by an X- ray powder diffraction pattern having peaks expressed as 2-theta at about 10.3, 13.8 and 17.7 degrees, a melting point of about 161°C to about 162°C; a differential scanning calorimetry (DSC) endotherm maximum at about 162°C. The polymorphic Form III is characterized by a melting point of about 160.8°C.
However, it has been observed by the present inventors that the celecoxib obtained after crystallization from isopropanol and water is fluffier resulting in low bulk density and poor flow properties. Moreover, it has also been observed that the particles of the crystalline form III of celecoxib obtained by the aforementioned crystallization processes are static or cohesive thereby increasing the difficulties of formulation scientists.
PCT Publication No. WO 01/42221 discloses an amorphous form of celecoxib, and processes for preparing amorphous celecoxib using crystallization inhibitors. Amorphous celecoxib exhibits an apparent glass transition at 111.4°C (onset).
EP Patent No. 1167355 (hereinafter referred to as the ‘355 patent) discloses a crystalline form, designated as Form I, of celecoxib, processes for the preparation, and pharmaceutical compositions thereof. The crystalline form is characterized by powder X-ray diffraction (P-XRD) and scanning electron microscopy (SEM). According to the ‘355 patent, the crystalline Form I is characterized by an X-ray powder diffraction pattern having peaks expressed as 2-theta at about 14.8, 16.05, 17.875, 19.615, 21.455, 22.080, 22.385, 23.425, 25.33 and 29.355 degrees. The crystalline Form I is further characterized by an X-ray powder diffraction pattern having additional peaks expressed as 2-theta at about 10.67, 10.97, 12.985, 13.855, 18.340, 18.685, 20.425, 20.67, 23.185, 24.51, 24.93, 25.73, 26.915, 27.63, 28.185, 29.955, 30.375, 31.405, 34.915, 35.585, 37.895, 44.070 and 45.250 degrees. The ‘355 patent teaches that the crystalline Form I has improved properties over prior art crystal form which is used for formulating celecoxib as disclosed in International Application No. WO 95/15316. The ‘355 patent teaches that the prior art crystal form (designated as Form II) has several disadvantages, caused by its crystal structure, since it has low bulk density and a crystal morphology that tends to form long cohesive needles.
According to the ‘355 patent, the celecoxib crystalline Form I is prepared by dissolving celecoxib in a solvent system comprising at least one amide solvent selected from the group consisting of N,N-dimethylformamide, Ν,Ν-dimethylacetamide, and mixtures thereof; and isolating the crystals of Form I by adding a non-solvent, especially water, to the solution.
The process for the preparation of the celecoxib crystalline Form I described in the ‘355 patent also suffers from drawbacks since the use of amide solvents in the purification/crystallization of celecoxib leads to the formation of solvates (for example, the formation of solvates of celecoxib with amide solvents such as dimethylacetamide and dimethylformamide can be found in the preparative examples of the WO 01/42222). It is well known that the removal of these residual amide solvents from the celecoxib crystalline form is very difficult and requires high temperatures.
Formation of solvates of celecoxib with amide solvents such as dimethylacetamide and dimethylformamide is also described in PCT Publication No. WO2005/014546. Moreover, celecoxib obtained by the crystallization process using amide solvents described in the ‘355 patent does not have satisfactory purity for pharmaceutical use.
The solvated forms of celexicob are not acceptable from regulatory point of view since they include substantial amounts of organic solvents, and thus are not acceptable for clinical use. It is well known that impurities and residual solvents in celecoxib or any active pharmaceutical ingredient (API) are undesirable and might be harmful. Purity standards are set by regulatory authorities with the intention of ensuring that an API is as free of impurities and residual solvents as possible, and, thus, are as safe as possible for clinical use. Furthermore, it is required to control the levels of these impurities in the final drug compound obtained by the manufacturing process and to ensure that the impurities and/or residual solvents are present in the lowest possible levels.
celecoxib characterized by a powder X-ray diffraction (P-XRD) pattern having peaks (designated as d-values) at about 16.0, 15.3, 12.3, 10.6 ± 0.2 A. According to the ‘340 application, the crystalline Form N of celecoxib is prepared by suspending celecoxib Form III in a hydrocarbon solvent selected from the group consisting of n-tetradecane, and n-decane, heating the suspension at high temperatures (about 165°C) while stirring, stirring the resulting emulsion at the same high temperature, followed by cooling to 145°C. The resulting mass is then reheated to about 165°C, followed by cooling to about 110°C, filtering the separated crystals, and drying at 100°C under the vacuo for 12 hours to produce celecoxib Form N.
The crystallization process for preparing celecoxib described in the ‘340 application also suffers from disadvantages since the processes involve tedious and cumbersome procedures such as the use of high boiling point solvents, large amounts of solvents (about 20 volumes of high boiling point solvents per gram of celecoxib), high temperatures (about 165°C), high drying temperatures (about 100°C), and prolonged periods of drying at a high temperature, resulting in formation of unwanted impurities, thereby making the process industrially unfeasible.
PCT Publication No. WO 05/089511 discloses a hydrate of celecoxib sodium salt characterized by a powder X-ray diffraction (P-XRD) pattern having peaks at 3.05, 8.91 and 10.77 degrees 2-theta.
PCT Publication No. WO 2006079923A1 discloses a crystalline Form IV of celecoxib characterized by a powder X-ray diffraction (P-XRD) pattern having peaks at about 4.46, 13.13, 18.29, 20.21, 21.83 and 26.24 degrees 2-theta. [0020] Based on the aforementioned drawbacks, the prior art crystallization processes may be unsuitable for the preparation of celecoxib, especially in crystalline Form III, in commercial scale operations.
| Patent | Submitted | Granted |
|---|---|---|
| 5-oxo-pyrrolidine-2-carboxylic acid hydroxamide derivatives [EP1004578] | 2000-05-31 | 2004-02-25 |
| ANALGESIC COMPOSITIONS COMPRISING ANTI-EPILEPTIC COMPOUNDS AND METHODS OF USING SAME [EP1011658] | 2000-06-28 | 2005-12-07 |
| SYNERGISTIC ANALGESIC COMBINATION OF OPIOID ANALGESIC AND CYCLOOXYGENASE-2 INHIBITOR [EP1014886] | 2000-07-05 | 2004-11-24 |
| BIARYLALKANOIC ACIDS AS CELL ADHESION INHIBITORS [EP1017382] | 2000-07-12 | 2006-03-01 |
| BIOADHESIVE COMPOSITIONS AND METHODS FOR TOPICAL ADMINISTRATION OF ACTIVE AGENTS [EP1021204] | 2000-07-26 | 2005-12-28 |
| Implantable medical device with enhanced biocompatibility and biostability [EP1023879] | 2000-08-02 | 2005-04-06 |
| NOVEL COMBINATION [EP1027052] | 2000-08-16 | |
| CYCLIC AMINO ACID DERIVATIVES AS CELL ADHESION INHIBITORS [EP1033983] | 2000-09-13 | |
| SUBSTITUTED BETA-ALANINE DERIVATIVES AS CELL ADHESION INHIBITORS [EP1034164] | 2000-09-13 | 2004-05-19 |
| Delayed-release oral formulation of dihydroxy open acid simvastatin and salts and esters thereof [EP1036563] | 2000-09-20 |
| Patent | Submitted | Granted |
|---|---|---|
| Crystalline hydrated dihydroxy open-acid simvastatin calcium salt [EP1036783] | 2000-09-20 | 2003-05-21 |
| CYCLOOXYGENASE-2 INHIBITION CYCLOOXYGENASE-2 INHIBITION [EP1039914] | 2000-10-04 | |
| Dioxocyclopentyl hydroxamic acids [EP1041072] | 2000-10-04 | 2003-07-16 |
| PHARMACEUTICAL ORAL DOSAGE FORM COMPRISING A COMBINATION OF AN OPIOID AGONIST AND NALTREXONE [EP1041987] | 2000-10-11 | 2006-04-19 |
| Method and compositions for the treatment and prevention of pain and inflammation [US2005101563] | 2005-05-12 | |
| Compositions of a cyclooxygenase-2 selective inhibitor and a neurotrophic factor-modulating agent for the treatment of central nervous system mediated disorders [US2005148589] | 2005-07-07 | |
| Compositions and methods of treatment involving peroxisome proliferator-activated receptor-gamma agonists and cyclooxygenase-2 selective inhibitors [US2003220374] | 2003-11-27 | |
| Methods for treating carbonic anhydrase mediated disorders [US2003220376] | 2003-11-27 | |
| Method of using a Cox-2 inhibitor and a 5-HT1A receptor modulator as a combination therapy [US2004147581] | 2004-07-29 | |
| Tyrosine kinase inhibitors [US2002137755] | 2002-09-26 |
| Cited Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| WO1995015316A1 | 14 Nov 1994 | 8 Jun 1995 | Stephen R Bertenshaw | Substituted pyrazolyl benzenesulfonamides for the treatment of inflammation |
| WO2001042221A1 | 6 Dec 2000 | 14 Jun 2001 | Michael J Hageman | Solid-state form of celecoxib having enhanced bioavailability |
| WO2001042222A1 | 1 Dec 2000 | 14 Jun 2001 | Leonard J Ferro | Polymorphic crystalline forms of celecoxib |
| WO2002000627A1 * | 26 Jun 2001 | 3 Jan 2002 | Mehmet Bahar | A crystalline form of celecoxib |
| WO2005014546A1 | 8 Aug 2003 | 17 Feb 2005 | Hetero Drugs Ltd | Novel crystalline forms of celecoxib |
| WO2005089511A2 | 17 Mar 2005 | 29 Sep 2005 | Transform Pharmaceuticals Inc | Novel pharmaceutical forms, and methods of making and using the same |
| WO2006051340A1 | 21 Jul 2005 | 18 May 2006 | Pliva Istrazivanje I Razvoj D | Novel form of celecoxib |
| WO2006079923A2 | 19 Jan 2006 | 3 Aug 2006 | Pharmacia & Upjohn Co Llc | Form iv crystalline celecoxib |
| EP1167355A1 | 15 Mar 2001 | 2 Jan 2002 | Fako Ilaclari A.S. | A crystalline form of celecoxib |
| US5466823 | 30 Nov 1993 | 14 Nov 1995 | G.D. Searle & Co. | Antiinflammatory agents; FDA Orange book listed patent for Celebrex |
| US5760068 | 14 Nov 1994 | 2 Jun 1998 | G.D. Searle & Co. | Substituted pyrazolyl benzenesulfonamides for the treatment of inflammation |
| US5910597 | 14 Oct 1998 | 8 Jun 1999 | G.D. Searle & Co. | Process for preparing 3-haloalkyl-1H-pyrazoles |
………..

Dr Raghupathi Reddy Anumula
Managing Director- Chemikos Laboratories Private Limited

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....






















 +
+

 +
+





