Home » Posts tagged 'RG 6344'
Tag Archives: RG 6344
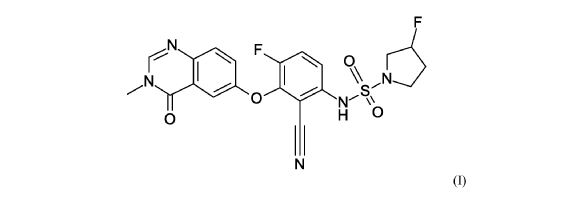
Mosperafenib
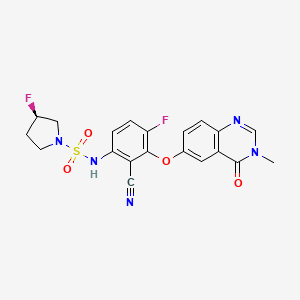
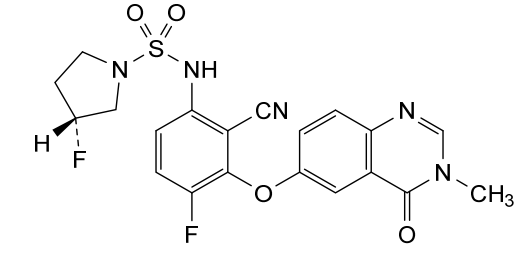
Mosperafenib
CAS 2649372-20-1
MF C20H17F2N5O4S MW 461.4 g/mol
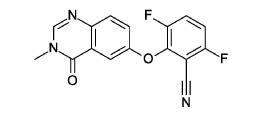
- (3R)-N-{2-cyano-4-fluoro-3-[(3-methyl-4-oxo-3,4-dihydroquinazolin-6-yl)oxy]phenyl}-3-fluoropyrrolidine-1-sulfonamide
- (3R)-N-(2-cyano-4-fluoro-3-((3-methyl-4-oxo-3,4-dihydroquinazolin-6-yl)oxy)phenyl)-3-fluoropyrrolidine-1-sulfonamide
(3R)-N-{2-cyano-4-fluoro-3-[(3-methyl-4-oxo-3,4-dihydroquinazolin-6-yl)oxy]phenyl}-3-fluoropyrrolidine-1-sulfonamide
B-Raf (BRAF) inhibitor, antineoplastic, RG6344, RO7276389, RG 6344, RO 7276389, 881-730-4, B-Raf IN 2
Mosperafenib is a small molecule drug. The usage of the INN stem ‘-rafenib’ in the name indicates that Mosperafenib is a Raf (rapidly accelerated fibrosarcoma) kinase inhibitor. Mosperafenib has a monoisotopic molecular weight of 461.1 Da.
Mosperafenib (RG6344, RO7276389) is an investigational, oral, “paradox-breaker” BRAF inhibitor developed by Roche for treating BRAF-mutated cancers, particularly BRAF V600E-mutant metastatic colorectal cancer. It acts as a potent, selective inhibitor that avoids MAPK pathway overactivation in non-V600E contexts, showing superior preclinical activity and brain penetration compared to existing inhibitors like encorafenib.
Key Aspects of Mosperafenib:
- Mechanism: As a “paradox-breaker” BRAF inhibitor, it avoids the paradoxical MAPK pathway activation seen with earlier inhibitors. It inhibits BRAF mutants (
) and is effective in RAF dimer-mediated resistant models.
- Clinical Development: Currently in Phase I clinical trials for BRAF V600E-mutant colorectal cancer.
- Preclinical Performance: In studies, it demonstrated higher antitumor activity than encorafenib/cetuximab combinations, even in BRAFi-naïve models.
- Combination Potential: It is being evaluated in combination with cetuximab and FOLFOX.
- Targeting: It targets BRAF V600E/K/A/D mutations.
- OriginatorRoche
- ClassAntineoplastics; Fluorinated hydrocarbons; Fluorobenzenes; Nitriles; Phenyl ethers; Pyridones; Pyrrolidines
- Mechanism of ActionProto-oncogene protein b-raf inhibitors
- Phase IMalignant melanoma; Solid tumours
- 18 Sep 2025Chemical structure information added.
- 30 May 2025Efficacy, pharmacokinetics and adverse events data from a phase I trial in Solid tumors presented at the 61st Annual Meeting of the American Society of Clinical Oncology (ASCO-2025)
- 25 Apr 2025Efficacy, pharmacokinetics and adverse events data from a phase I trial in Solid tumors presented at the 116th Annual Meeting of the American Association for Cancer Research (AACR-2025)
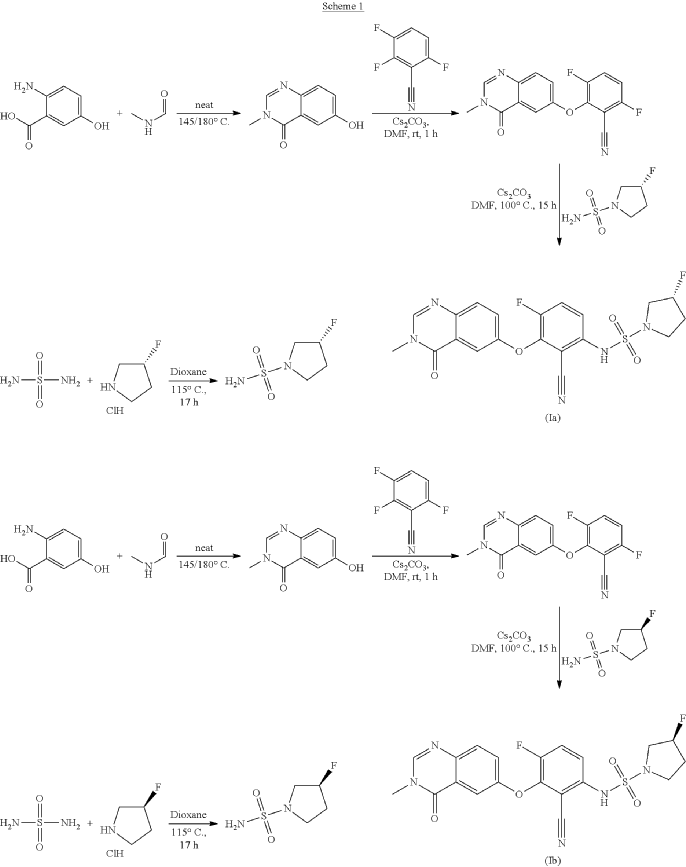
SYN
SYN
3R)-N-[2-cyano-4-fluoro-3-(3-methyl-4-oxo-quinazolin-6-yl)oxy-phenyl]-3-fluoro-pyrrolidine-1-sulfonamide (Example 1)
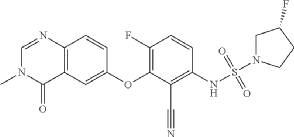
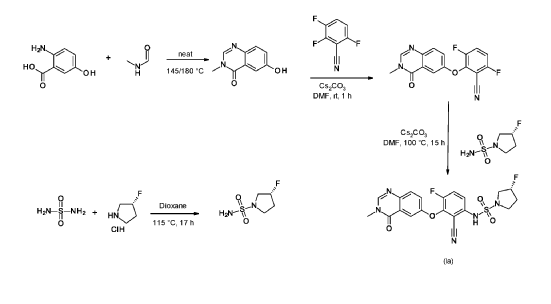
R)-3-Fluoropyrrolidine-1-sulfonamide (1.26 g, 7.51 mmol, Eq: 2.1) and cesium carbonate (2.56 g, 7.87 mmol, Eq: 2.2) were suspended in dry DMF (10.2 ml) under an argon atmosphere. The reaction was stirred at 50° C. for 30 min. The reaction mixture was cooled to rt and a solution of 3,6-difluoro-2-((3-methyl-4-oxo-3,4-dihydroquinazolin-6-yl)oxy)benzonitrile (1.12 g, 3.58 mmol, Eq: 1.0) in DMF (25.5 ml) was added. The reaction mixture was stirred at 100° C. for 15 h, then concentrated in vacuo. The residue was taken up in sat. aq. NH 4Cl (100 mL) and EtOAc (100 mL). The phases were separated, and the aqueous layer was extracted further with 2×100 mL EtOAc. The combined organic layers were washed with water (200 mL) and brine (200 mL), dried (Na 2SO 4), filtered and concentrated in vacuo. The water layer was back-extracted with EtOAc (3×100 mL). The combined organic extracts were washed with brine (200 mL), dried (Na 2SO 4), filtered and concentrated in vacuo. The residue was diluted with DCM and MeOH, and concentrated onto silica. Purification by flash chromatography (120 g, 0.5-2% MeOH/DCM) gave an off-white solid which was triturated with 1:1 heptane/DCM (20 mL) with sonication, then dried in vacuo to give the title compound as a colourless solid (1.087 g, 66% yield). MS (ESI) m/z: 426.2 [M+H] +. Chiral SFC: RT=4.594 min [Chiralpak IC column, 4.6×250 mm, 5 μm particle size (Daicel); gradient of 20-40% MeOH containing 0.2% NHEt 2 over 8 min; flow: 2.5 mL/min; 140 bar backpressure].
SYN
Refences compounds AR-25, AR-30 and AR-31 were prepared according to the synthesis disclosed in WO2012/118492 in example 25, example 30 and example 31 respectively.
6-hydroxy-3-methyl-quinazolin-4-one
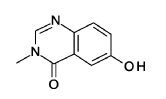
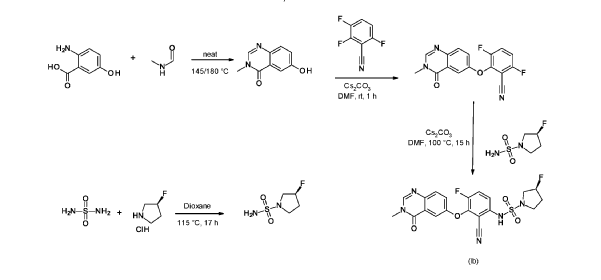
2-Amino-5-hydroxybenzoic acid (10 g, 65.3 mmol, Eq: 1.0) and A-methylformamide (30 g, 29.9 mL, 503 mmol, Eq: 7.7) were heated at 145 °C for 21 h 45 min, then cooled to rt. The reaction mixture was diluted with 50 mL H2O and stirred at rt for 20 min. The resulting precipitate was collected by filtration. The light brown solid was washed 3 × with 20 mL water. The solid was taken up in toluene and evaporated to dryness (3 ×). The solid was dried in vacuo at 40 °C overnight under high vacuum to give the title compound as a light brown solid (10.3 g, 89% yield). MS (ESI) mlz: 177.1 [M+H]+.
3.6-difluoro-2-(3-methyl-4-oxo-quinazolin-6-yl)oxy-benzonitrile
Cesium carbonate (3.22 g, 9.79 mmol, Eq: 1.15) was added at rt to a solution of 6-hydroxy-3-methylquinazolin-4-one (1500 mg, 8.51 mmol, Eq: 1.0) in N,N-dimethylformamide (35 mL). The mixture was stirred for 30 min at rt then 2,3,6-trifluorobenzonitrile (1.47 g, 1.08 ml, 9.37 mmol, Eq: 1.1) was added. After 1 h, the reaction was cooled on ice and diluted with water (120 mL). The resultant solid was collected by filtration, washed with iced water (100 mL) and heptane (100 mL) and suction-dried. The solid was taken up in toluene and evaporated to dryness (3 ×) then dried overnight in vacuo to give the title compound as a light brown solid (2.58 g, 97% yield). MS (ESI) m/z: 314.1 [M+H]+.
(3R)-3 -fluoropyrrolidine- 1 -sulfonamide
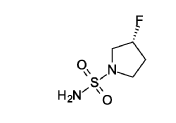
(R)-3 -Fluoropyrrolidine hydrochloride (1.8 g, 14.3 mmol, Eq: 1.2) was added to a solution of sulfuric diamide (1.148 g, 11.9 mmol, Eq: 1.0) and triethylamine (2.42 g, 3.33 mL, 23.9 mmol, Eq: 2) in dioxane (10 mL). The reaction was stirred in a sealed tube at 115 °C for 15.5 h then cooled to rt and concentrated in vacuo. The residue was diluted with DCM, evaporated with silica gel to dryness and transferred to a column. Purification by flash chromatography (40 g silica, 80% EtOAc) gave the title compound as a white crystalline solid (1.82 g, 91% yield). MS (ESI) m/z: 169.1 [M+H]+.
(3S)-3 -fluoropyrrolidine- 1 -sulfonamide
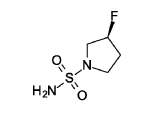
Triethylamine (304 mg, 419 μl, 3.01 mmol, Eq: 2.0) was added to a suspension of sulfuric diamide (146 mg, 1.5 mmol, Eq: 1.0) and (S)-3 -fluoropyrrolidine hydrochloride (234 mg, 1.8 mmol, Eq: 1.2) in dioxane (1.3 ml). The reaction was stirred in a sealed tube at 115°C for 16 h 35 min, then concentrated in vacuo. The residue was diluted with MeOH and evaporated with silica gel to dryness and transferred to a column. Purification by flash chromatography (40 g silica, 0-8% MeOH/DCM) gave the title compound as a light yellow solid (193 mg, 75% yield). MS (ESI) m/z: 169.1 [M+H]+.
(3R)-N-[2-cyano-4-fluoro-3-(3-methyl-4-oxo-quinazolin-6-yl)oxy-phenyl]-3-fluoro-pyrrolidine-1 -sulfonamide (Example 1)
(R)-3-Fluoropyrrolidine-1-sulfonamide (1.26 g, 7.51 mmol, Eq: 2.1) and cesium carbonate (2.56 g, 7.87 mmol, Eq: 2.2) were suspended in dry DMF (10.2 ml) under an argon atmosphere. The reaction was stirred at 50 °C for 30 min. The reaction mixture was cooled to rt and a solution of 3,6-difluoro-2-((3-methyl-4-oxo-3,4-dihydroquinazolin-6-yl)oxy)benzonitrile (1.12 g, 3.58 mmol, Eq: 1.0) in DMF (25.5 ml) was added. The reaction mixture was stirred at 100 °C for 15 h, then concentrated in vacuo. The residue was taken up in sat. aq. NH4Cl (100 mL) and EtOAc (100 mL). The phases were separated, and the aqueous layer was extracted further with 2 x 100 mL EtOAc. The combined organic layers were washed with water (200 mL) and brine (200 mL), dried (Na2SO4), filtered and concentrated in vacuo. The water layer was back-extracted with EtOAc (3 x 100 mL). The combined organic extracts were washed with brine (200 mL), dried (Na2SO4), filtered and concentrated in vacuo. The residue was diluted with DCM and MeOH, and concentrated onto silica. Purification by flash chromatography (120 g, 0.5-2% MeOH/DCM) gave an off-white solid which was triturated with 1 : 1 heptane/DCM (20 mL) with sonication, then dried in vacuo to give the title compound as a colourless solid (1.087 g, 66% yield). MS (ESI) mlz: 426.2 [M+H]+. Chiral SFC: RT = 4.594 min [Chiralpak IC column, 4.6 x 250 mm, 5μm particle size (Daicel); gradient of 20 – 40% MeOH containing 0.2% NHEt2 over 8 min; flow: 2.5 mL/min; 140 bar backpressure],
(3S)-N-[2-cyano-4-fluoro-3-(3-methyl-4-oxo-quinazolin-6-yl)oxy-phenyl]-3-fluoro-pyrrolidine-1 -sulfonamide (Example 2)
(S)-3-Fluoropyrrolidine-1-sulfonamide (181 mg, 1.08 mmol, Eq: 2.1) was dissolved in DMF (1.6 ml). At rt cesium carbonate (368 mg, 1.13 mmol, Eq: 2.2) was added and the reaction mixture was stirred at 50 °C for 30 min. The reaction mixture was cooled to rt and a solution of 3,6-difluoro-2-((3-methyl-4-oxo-3,4-dihydroquinazolin-6-yl)oxy)benzonitrile (160.8 mg, 513 μmol, Eq: 1.0) in DMF (4 ml) was added. The reaction mixture was stirred at 105 °C for 2 h 50 min then concentrated in vacuo. The residue was taken up in DCM and washed with sat. aq. NH4Cl. The aq. layer was back-extracted twice with DCM. The combined organic layers were dried over Na2SO4, filtrated and evaporated. The residue (brown oil) was diluted with DCM and transferred to a column. Purification by flash chromatography (80 g, 0-100% EtOAc in DCM) gave a solid which was further purified by SFC to give the title compound as a light yellow solid (119 mg, 50% yield). MS (ESI) m/z: 426.2 [M+H]+. Chiral SFC: RT = 4.411 min [Chiralpak IC column, 4.6 x 250 mm, 5μm particle size (Daicel); gradient of 20 – 40% MeOH containing 0.2% NHEt2 over 8 min; flow: 2.5 mL/min; 140 bar backpressure].
PAT
New methylquinazolinone derivatives
Publication Number: AU-2020403443-A1
Priority Date: 2019-12-10
- Methylquinazolinone derivativesPublication Number: US-2024174621-A1Priority Date: 2019-12-10
- New methylquinazolinone derivativesPublication Number: EP-4073065-B1Priority Date: 2019-12-10Grant Date: 2025-02-19
- Methylquinazolinone derivativesPublication Number: US-2022298119-A1Priority Date: 2019-12-10
- Novel BRAF inhibitors as anomalous breakersPublication Number: CN-114746405-BPriority Date: 2019-12-10Grant Date: 2024-03-26
- New BRAF inhibitors as paradox breakersPublication Number: AU-2020403082-A1Priority Date: 2019-12-10



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

////////////////mosperafenib, B-Raf (BRAF) inhibitor, antineoplastic, RG6344, RO7276389, RG 6344, RO 7276389, 881-730-4, B-Raf IN 2

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










