Home » Posts tagged 'Quetiapine'
Tag Archives: Quetiapine
Quetiapine

Quetiapine, astrazeneca
111974-69-7 cas
US 5,948,437*PED, NDA 022047 Appr may 17 2007 sustained release formulation
NDA 020639 approved 26.9.1997
patent approved expiry
| United States | 5948437 | 1997-11-28 | 2017-11-28 |
| United States | 4879288 | 1994-09-26 | 2011-09-26 |
| Canada | 2251944 | 2007-04-10 | 2017-05-27 |
| United States | 4879288 | 1994-09-26 | 2011-09-26 |

Quetiapine (/kwɨˈtaɪ.əpiːn/ kwi-ty-ə-peen) (branded as Seroquel, Xeroquel, Ketipinor) is a short-acting atypical antipsychotic approved for the treatment of schizophrenia, bipolar disorder, and along with an antidepressant to treat major depressive disorder.
Annual sales are approximately $5.7 billion worldwide, with $2.9 billion in the United States. The U.S. patent, which expiredv in 2011, received a pediatric exclusivity extension which pushed its expiration to March 26, 2012.The patent has already expired in Canada. Quetiapine was developed by AstraZeneca from 1992-1996 as an improvement from first generation antipsychotics. It was first approved by the FDA in 1997. There are now several generic versions of quetiapine, such as Quepin, Syquel and Ketipinor
Seroquel (quetiapine) is a psychotropic medication that is used to treat schizophrenia in adults and children who are at least 13 years old. Seroquel is also used in the treatment of major depression and bipolar disorder. Side effects of Seroquel may include mood or behavior changes, constipation, drowsiness, headache, and trouble sleeping. Older adults with dementia may have a slightly increased risk of death when taking this medication.
Dosing preparations are 25, 50, 100, 200, 300, and 400 mg tablets. Seroquel may interact with a number of other drugs, including, but not limited to, antidepressant medications, antifungal drugs, steroids, cimetidine (Tagamet), thioridazine (Mellaril), and lorazepam (Ativan). During pregnancy, this medication should be used only when clearly needed. Discuss the risks and benefits with your doctor. Seroquel passes into breast milk and may have undesirable effects on a nursing infant.

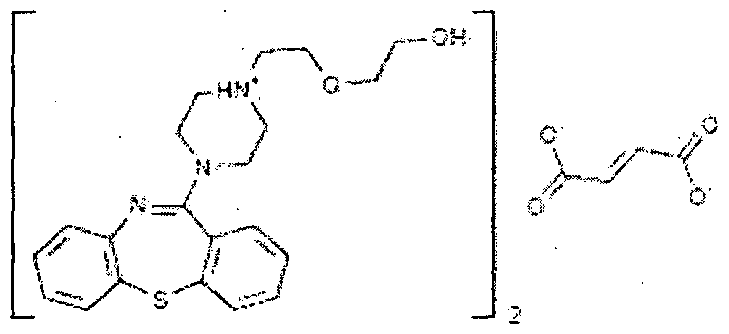
Quetiapine fumarate is a psychotropic agent belonging to a chemical class of dibenzothiazepine derivatives, designated chemically as 2-[2-(4-dibenzo [b,f] [l,4]thiazepin -1 l-yl-l-piperazinyl)ethoxy]-ethanol fumarate (2: l )(salt). Its molecular formula is C42H5oN604S2*C4H404 having a molecular weight of 883.1 1. The structural formula is:
Quetiapine is marketed as immediate release as well as extended release tablets in United States under the trade name Seroquel® and Seroquel XR® by AstraZeneca.
-
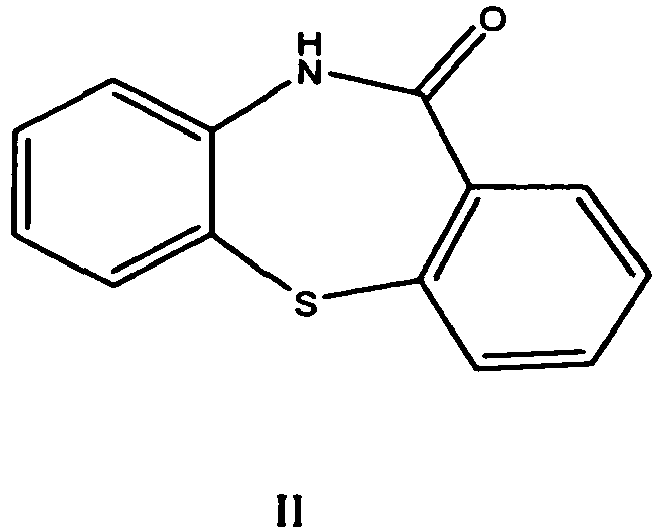
Quetiapine was first described in a patent publication EP 240228 (US 4879288 ). It is prepared starting from dibenzo[b,f][1,4]thiazepin-11-[10H]one of formula II
which is first halogenated with phosphorous oxychloride, then isolated and condensed with 1-(2-hydroxyethoxy) ethyl piperazine to obtainquetiapine. After purification by flash chromatography the yield was 77.7 %. As an alternative to halogenation a process via a thioether in the first step is presented.
-
In the process claimed in EP 282236 the piperazine ring is first condensed with 11-chloro-dibenzo[b,f][1,4] thiazepine and thereafterquetiapine is obtained by its reaction with haloethoxy ethanol. The base is further converted to the hemifumarate salt, which was isolated in 78 % yield.
-
WO 2006/117700 describes a process of EP ‘228 improved by the destruction of phosphorous oxychloride in situ to decrease the amount of hazardous waste. Phosphorous oxychloride is used only about 1 equivalent to the compound of formula II whereas in the process of EP 240228 it was used in about 15 equivalents.
-
A reaction of 11-chlorodibenzo[b,f][1,4]thiazepine with a piperazine moiety in the presence of a halide is the improvement described inWO 2006/113425 . The process is said to yield quetiapine in high purity.
-
In WO 2006/094549 there is described a process which avoids the halogenation step and the use of hazardous phosphorous halogenating agents by the reaction of 10H-dibenzo[b,f][1,4] thiazepin-11-one directly with a piperazine derivative. This is achieved by performing the reaction in the presence of titanium alkoxide. Yields of 50 – 75 % as a fumarate salt are reported. Expensive titanium alkoxide is used from about 2 to 3 fold excess to starting compound of formula II.
-
Also the process of US 2006/0063927 avoids the use of phosphorous compounds in halogenation by using oxalyl chloride as a halogenating agent. The imino chloride is obtained in 66 % yield. The reaction of 11-chloro-dibenzo[b,f][1,4] thiazepine with 1-(2-hydroxyethoxy)ethylpiperazine is performed either in the presence of a base in an organic solvent or in a two-phase system. However, the reagent used, oxalyl chloride is poisonous and requires special attention.
-
A one-pot process for the preparation of quetiapine is described inWO 2007/020011 . Phosphorous oxychloride is used in halogenation step about one equivalent to 10H-dibenzo[b,f][1,4] thiazepin-11-one.
-
WO 2007/004234 describes a process comprising the reaction of chloro ethoxy ethanol with piperazinyl-dibenzo[b,f][1,4] thiazepine dihydrocloride, which is obtained by halogenating the dibenzo[b,f][1,4] thiazepin-11-[10H]one, reacting the imino chloride obtained with piperazine, and treating the obtained compound with an alcoholic solution of hydrogen chloride.
-
All processes described above use dibenzo[b,f][1,4] thiazepin-11-[10H]one as a starting material. Its preparation requires several steps, and in most cases it has to be even halogenated to the imino chloride before the piperazine moiety can be condensed with it. Halogenating reagents, e.g. phosphorous oxychloride have been used in excess and their removal from the reaction mixture requires evaporation of large amounts.
-
A different approach using protected intermediates is used e.g. in routes described in WO 2005/014590 , WO 2005/028457 , WO 2005/028458and WO 2005/028459 . In some cases the reactions may be performed in one pot fashion and no extra purification steps are needed to get a pure product in high yield. However, protection and deprotection steps used lengthen the processes and shorter processes for the preparation of quetiapine are still needed.
-
Similar compounds are prepared in US 3, 539,573
U.S. Patent 4,879,288 discloses 1 l-[4-[2-(2-hydroxyethoxy) ethyl] -1 – piperazinyl] dibenzo [b, f] [1 , 4] thiazepine as an antipsychotic drug of dibenzothiazepine class suitable for treatment of various psychotic disorders.
US patent 5,948,437 discloses sustained release formulations of quetiapine using gelling agents such as hydroxypropyl methylcellulose and its derivatives that create a gel structure after contact with water. US patent 4,547,57 1 describes process for the preparation of carboxymethyl ethyl cellulose (CMEC) polymer.
WO 2004012699 discloses modified release dosage forms prepared by using dual – retard— technique comprising micro matrix particles containing quetiapine and hydrophobic release controlling agents, which are coated with hydrophobic release controlling agents.
WO 2005041935 discloses matrix composition comprising quetiapine and a wax material.
WO 2007086079 discloses sustained release compositions of quetiapinecomprising a channelizer and a rate controlling polymer.
WO 2008060228 discloses extended release compositions comprisingquetiapine, hydroxypropyl methyl cellulose and sodium citrate dihydrate.
WO 20091 13051 discloses sustained release compositions containingquetiapine and one or more non-gelling and/or waxy excipients.
WO 2010001413 discloses sustained release dosage forms comprisingquetiapine or its pharmaceutically acceptable salts and one or more non-gellable release controlling polymers.
WO 2010028794 discloses a matrix formulation in the form of a retard tablet comprising quetiapine, at least one matrix-forming, water-insoluble, non-swellable auxiliary agent, and at least one water-soluble binding agent.
The synthesis of quetiapine begins with a dibenzothiazepinone. The lactam is first treated with phosphoryl chloride to produce a dibenzothiazepine. A nucleophilic substitutionis used to introduce the sidechain.U.S. Patent 4,879,288.
Atypical antipsychotic quetiapine (Quetiapine, drugs used its fumarate) for the treatment of schizophrenia (schizophrenia) and dry depressive disorder (bipolar disorder), trade name Seroquel, produced by AstraZeneca. Star molecule drugs, the global sales of about $ 6 billion.
Quetiapine synthesis o-nitro-chlorobenzene ( 1 ) starting a thiophenol occurred and SNAr reaction, hydrogenation of nitro group to an amino group after reaction with phosgene isocyanate 2 , 2 ring closure in hot sulfuric acid to obtain 3 , 3 with phosphorus oxychloride isomerization chlorinated4 , 4 and 5 SNAr reaction occurs fumarate salt formation with quetiapine fumarate.
The route of the compound 4 is not stable enough, then there are improved route. 6 and the reaction of phenyl chloroformate 7

Quetiapine fumarate, Bis [2-(2-[4-(dibenzo[b,f][1,4]thiazepin-11-yl]ethoxy)ethanol] fumarate (IUPAC)2-[2-(4-dibenzo[b,f][1,4]thiazepin-11-yl-1-piperazinyl)ethoxy]-ethanol-(E)-2-butanedioate (2:1) salt), [ICI 204,636], is a novel dibenzothiazepine antipsychotic developed by Zeneca. It is marketed under the trade name ‘Seroquel’Seroquel. Quetiapine is well tolerated and clinically effective in the treatment of schizophrenia.
The initial hope of investigators was that quetiapine would have antipsychotic potential and that it might share some of the properties of clozapine without its toxicity to white blood cells.
The effective dosage range is usually 300-450 mg/day split into two doses. The dose is titrated upwards from 25 mg twice dailybd from day one to 300mg/daya fuller dosage on day 4. Elderly patients or patients with liver problems should be started on lower doses. It is both superior to placebo and, and comparable to haloperidol in reducing positive symptoms at doses ranging from 150 to 750 mg/day and is an effective treatment for in reducing negative symptoms at a dose of 300 mg/day.
Somnolence is the most common adverse event. Abnormalities of the QTqt interval on ECG appear very infrequently and there is no need for a baseline ECG or blood pressure monitoring as used to be the case with ssertindole. There is no need for haematological monitoring as with clozapine. Quetiapine, across the full dosage range, is associated with no greater extrapyramidal symptoms than placeboThere is a reduced potential for extrapyramidal symptoms compared with conventional antipsychotics.
Quetiapine’s general efficacy and side effect profile suggest that, unless there are unforeseen post-marketing complications, quetiapine deserves a major place in the initial and long term management of schizophreniform disorders.
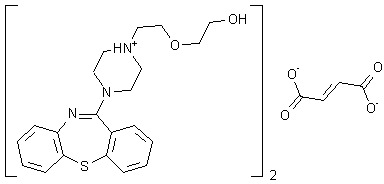
Quetiapine fumarate, Bis [2-(2-[4-(dibenzo[b,f][1,4]thiazepin-11-yl]ethoxy)ethanol] fumarate (IUPAC) 2-[2-(4-dibenzo[b,f][1,4]thiazepin-11-yl-1-piperazinyl)ethoxy]-ethanol-(E)-2-butanedioate (2:1) salt), [ICI 204,636], is a novel dibenzothiazepine antipsychotic developed by Zeneca Pharmaceuticals. It is marketed under the trade name ‘Seroquel’. Quetiapine is well tolerated and clinically effective in the treatment of schizophrenia.


 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....