Home » Posts tagged 'pulmonary arterial hypertension'
Tag Archives: pulmonary arterial hypertension
Iptakalim Hydrochloride 盐酸埃他卡林

Iptakalim Hydrochloride 盐酸埃他卡林
NDA Filed china
A K(ir) 6.1/SUR2B activator potentially for the treatment of pulmonary arterial hypertension.

179.7, C9H21N.HCl
CAS No. 642407-44-1(Iptakalim)
642407-63-4(Iptakalim Hydrochloride)
N-(1-methylethyl)-2,3-dimethyl-2-butylamine
| Catholic Healthcare West (D/B/A/ St. Joseph’s Hospital And Medical Center) |
Hypertension is a multifactorial disorder, and effective blood pressure control is not achieved in most individuals. According to the most recent report of the American Heart Association, for 2010, the estimated direct and indirect financial burden for managing hypertension is estimated to be $76.6 billion. Overall, almost 75% of adults with cardiovascular diseases/comorbidities have hypertension, which is associated with a shorter overall life expectancy. Alarmingly, rates of prehypertension and hypertension are increasing among children and adolescents due, in part, to the obesity epidemic we currently face. There is also the problem of an aging population and the growing rates of diabetes and obesity in adults, all factors that are associated with high blood pressure.Thus, the need is great for novel drugs that target the various contributing causes of hypertension and the processes leading to end organ damage.
Iptakalim (IPT), chemically 2, 3–dimethyl-N-(1-methylethyl)-2-butanamine hydrochloride, is novel adenosine triphosphate–sensitive potassium (KATP) channel opener. KATP channels are composed of discrete pore-forming inward rectifier subunits (Kir6.1s) and regulatory sulphonylurea subunits (SUR).IPT shows high selectivity for cardiac KATP (SUR2A/Kir6.2) and vascular KATP (SUR2B/Kir6.1 or SUR6B/Kir6.2). Because of this high selectivity, IPT does not exhibit the adverse side effects associated with the older nonspecific K+ channel openers, which limit their use to the treatment of severe or refractory hypertension. IPT produces arteriolar and small artery vasodilatation, with no significant effect on capacitance vessels or large arteries. Vasodilatation is induced by causing cellular hyperpolarization via the opening of K+ channels, which in turn decreases the opening probability of L-type Ca2+ channels. Of particular note, IPT is very effective in lowering the blood pressure of hypertensive humans but not of those with normal blood pressure.
-
The present compd relates generally to a novel method for decreasing a human’s cravings for cigarettes and reducing instances of relapse during detoxification once smoking abstinence has been achieved, and more specifically, to a method for decreasing nicotine use by treating a human with a novel type of nicotinic acetylcholine receptor antagonist, iptakalim hydrochloride (IPT).
-
Cigarette smoking is a prevalent, modifiable risk factor for increased morbidity and mortality in the United States, and perhaps in the world. Smokers incur medical risks attributable to direct inhalation. Bystanders, termed passive smokers, also incur medical risks from second-hand smoke. Society, as a whole, also bears the economic costs associated with death and disease attributable to smoking. Although the majority of smokers have tried repeatedly to quit smoking, eighty percent of smokers return to tobacco in less than two years after quitting. Therefore, tobacco dependence is a health hazard for millions of Americans.
-
Nicotine is the biologically active substance that is thought to promote the use of tobacco products by approximately one-quarter of the world populations. Tobacco-related disease is personally and economically costly to the any nation. Unfortunately, once use of tobacco has begun, it is hard for a smoker to quit because of nicotinic dependence and addiction.
-
The initiation and maintenance of tobacco dependence in a human is due to certain bio-behavioral and neuromolecular mechanisms. Nicotinic acetylcholine receptors (nAChRs) in humans are the initial binding sites for nicotine. The binding of nicotine to nAChRs is thought to modulate the brain’s “reward” function by triggering dopamine release in the human brain. The nAChRs exist as a diverse family of molecules composed of different combinations of subunits derived from at least sixteen genes. nAChRs are prototypical members of the ligand-gated ion channel superfamily of neurotransmitter receptors. nAChRs represent both classical and contemporary models for the establishment of concepts pertaining to mechanisms of drug action, synaptic transmission, and structure and function of transmembrane signaling molecules.
-
Basic cellular mechanisms of nicotinic dependence also involve the functional state changes during repeated nicotinic agonists exposure and receptor changes in the number of receptors during chronic nicotinic exposure. nAChRs can exist in many different functional states, such as resting, activated, desensitized or inactivated The activation and/or desensitization of nAChRs plays an important role in initiating nicotinic tolerance and dependence. Recovery from receptor activation and/or desensitization contributes to nicotinic withdrawal symptoms.
-
The most abundant form of nAChRs in the brain contains α4 and β2 subunits. α4β2-nAChRs bind nicotine with high affinity and respond to levels of nicotine found in the plasma of smokers. α4β2-nAChR also have been implicated in nicotine self-administration, reward, and dependence. Therefore, selective drug action at nAChRs, especially at those containing α4 subunits, is thought to be an ideal way for nicotine cessation and reducing nicotine withdrawal syndrome. Unfortunately, thus far, no optimal compound can meet this purpose. The brain-blood-barrier permeable nAChR antagonist, mecamylamine is popularly used systemically but exhibits much less nAChR subtype selectivity.
-
Although a variety of psychopharmacological effects contribute to drug reinforcement, actions on the mesolimbic dopaminergic pathway is the predominant hypothesis for mechanisms of nicotinic reward. The mesolimbic dopaminergic pathway originates in the ventral tegmental area (VTA) of the midbrain and projects to forebrain structures including the prefrontal cortex and to limbic areas such as the olfactory tubercle, the amygdala, the septal region, and the nucleus accumbens. Many studies have indicated that dopamine release in the nucleus accumbens of the human brain is “rewarding” or signals an encounter with a “reward” from the environment. Other substances, such as alcohol, cocaine, and opiates, operate in the same manner, resulting in a cycle of substance or alcohol abuse.
-
Therefore, a considerable need exists for a novel compound that can selectively block α4 subtypes of nAChRs to prevent smoking-induced “reward”, to limit increasing nicotine-induced dopamine release, and/or to diminish nicotinic withdrawal symptoms.
https://www.google.com/patents/US20040266822
Example 1
-
Production of N-(1-methylethyl)-2,3-dimethyl-2-butylamine (Compound 1): Method 1. The solution of 7.6 g (0.0745 mole) 2,3-dimethyl-2-butanol in 3.24 mL glacial acetic acid was cooled and maintained at −5 to −8 degree of centigrade (° C.), then was added 7.3 g (0.49 mole) of powdered potassium cyanide in several times under stirring. 32.4 mL concentrated sulfuric acid was added dropwise while keeping the temperatue below 20° C., after which, the reaction mixture was stirred for 3.5 hours below 20° C. and another 6 hours at room temperature, then stood overnight. After poured into ice colded water, the mixture was adjusted to pH10 with 20% aqueous sodium hydroxide solution, and extracted with ether (×4). The extract was dried over anhydrous sodium sulfate. After filtration on the next day, the dessicator was removed, and the filtrate was evaporated off the ether, then distilled in vacuum to give 8.8 g (yield 91.6%) N-[2-(2,3-dimethylbutyl)]-fomide; bp 105-108° C./5 mmHg.
-
To the mixture of 7.7 g (0.0597 mole) N-[2-(2,3-dimethylbutyl)]-formide, 6.2 mL ethanol and 51.6 mL wate, 17.4 mL concentrated hydrochloric acid was added. The reaction mixture was refluxed for 4 hours in the oil bath, then distilled off ethanol in vacuum. The residue was adjusted to above pH12 with 40% aqueous sodium hydroxide solution, and extracted with ether. The extract was dried over anhydrous potassiun carbonate. After recovering the ether, The residue was distilled at atmosphere to give 3.75 g (yield 62.2%) 2,3-dimethyl-2-butylamine, bp 97-104° C.
-
The mixture of 10.6 g (0.15 mole) 2,3-dimethyl-2-butylamine, 6.45 g (0.0524 mole) 2-bromopropane, 3.0 mL glycol and 22.0 mL toluene was added into an autoclave, and heated with stirring for 17 hours at temperature of 170° C., after which, the organic layer was separated and extracted with 6N hydrochloric acid (15 mL×4). The extract was combined and washed once with toluene, then adjusted to pH 12-13 with 4% aqueous sodium hydroxide in the ice bath. The mixture was extracted with ether and then dried over anhydrous potassium carbonate. After recovering the ether, The filtrate was distilled to yield the fraction of bp 135-145° C. (yield 68.8%). The hydrochloride’s Mp is 228-230° C. (1-PrOH-Et2O). Elemental analysis for C9H22ClN(%): Calculated C, 60.14; H, 12.34; N, 7.79, Cl 19.73; Found C, 60.14; H, 12.48; N, 7.31, Cl 19.67.
-
1H-NMR(D2O, ppm) 0.98(d, J=6.75H, 6H), 1.33(s, 6H), 1.37(d, J=6.46, 6H), 2.10(m, 1H), 3.70(m, 1H). MS(m/z) 143 (M+), 100(B).
-
Method 2. To the mixture of 288 mL glacial acetic acid, 412 g (6.86 mole) urea and 288 g (3.43 mole) 2,3-dimethyl-2-butene, the solution of 412 mL concentrated sulfuric acid and 412 mL of glacial acetic acid was added dropwise under stirring, while maintaining the reaction temperature at the range of 45° C. to 50° C., then stirred for 5 hours at the temperature of 50-55° C. The mixture stood overnight. Next day, the mixture was reacted for another 7 hours at the temperature of 50-55° C., then poured into the solution of 1200 g (30 mole) sodium hydroxide in 8L glacial water. The resulting solid was filtered, washed with water (200 mL×5) and dried to give 404 g (yield 81.8%) N-(2,3-dimethyl-2-butyl)urea as white solid, mp 175-176° C. Elemental analysis for C7H16N2O(%): Calculated C 58.30, H 11.18, N 19.42; Found C, 58.70; H, 11.54; N, 19.25, 1H-NMR(CDCl3, ppm) 0.88-0.91(d, 6H, 2×CH3), 1.26(s, 6H, 2×CH3), 2.20-2.26(m, 1H, CH), 4,45(br, 2H), 4.65(br, 1H). MS(m/z) 145.0, 144.0(M+), 143.0, 129.1, 101.0, 86.1, 69.1, 58.0(B).
-
To the mixture of 196 g (1.36 mole) N-(2,3-dimethyl-2-butyl)urea and 392 mL glycol or tri-(ethanol)amine, a solution of 118 g (2.95 mole) sodium hydroxide in 118 mL water was added. The reaction mixture was heated for 8 hours in an oil bath at temperature of 120° C., then distilled at atmosphere to collect the fraction of bp 95-102° C. To the fraction, 75 g anhydrous potassium carbonate and. 40 g sodium hydroxide were added. The resulting mixture was distilled to give 88.5 g (yield 64.3%) 2,3-dimethyl-2-butylamine as colorless liquid, bp 99-101° C.
-
1H-NMR(CDCl3, ppm) 0.88-0.91(d, 6H, 2×CH3), 1.04 (s, 6H, 2×CH3), 1.53(m, 1H, CH).
-
To a 50.0 ml autoclave, 10.6 g (0.15 mole) 2,3-dimethyl-2-butylamine, 6.45 g (0.0524 mol) 2-bromopropane, 3.0 ml glycol and 22.0 ml toluene were added, and heated with stirring for 17 hours at 170° C., after which the organic layer was seperated and extracted with 6N hydrochloric acid (15 ml×4). The extract was combined and washed once with toluene, then adjusted to pH 12-13 with 4% aqueous sodium hydroxide in the ice bath. The mixture was extracted with ether and then dried over anhydrous potassium carbonate the ether was recovered, and distilled to give the fraction of bp 135-145° C. (yield 68.8%). mp of the hydrochloride is 228-230° C., (i-PrOH: Et2O). Elemental analysis for C9H22ClN(%): Calculated C, 60.14; H, 12.34; N, 7.79, Cl 19.73; Found C 60.14, H 12.48, N 7.31, Cl 19.67. 1H-NMR(D2O, ppm) 0.98(d, J=6.75H, 6H), 1.33(s, 6H), 1.37(d, J=6.46, 6H), 2.10(m, 1H), 3.70(m, 1H). MS(m/z) 143 (M+), 100(B).
-
Method 3. a solution of 0.10 mole enamine (prepared from the condensation of methyl iso-propyl ketone and iso-propylamine) in 20 mL hexane was filled with N2 and added dropwise to a solution containing 0.10 mole lithium methide with stirring in ice bath. After the reaction is complete, the mixture was poured into 500 g glacial water, and stirred. The aqueous layer was extracted with ether (×2). The resulting organic layer was concentrated. 3N hydrochloric acid was added to acified the organic layer to pH<1. The mixture was kept minutes and adjusted to pH>11 with 10% aqueous sodium hydroxide, then extracted with ether (×3). The extract was dried over anhydrous potassium carbonate and filtered. The filtrate was distilled at atmosphere to give a fraction of bp 140-145° C. with a yield of 80%.
REF
http://www.google.com/patents/US20060293393
//////Iptakalim Hydrochloride, 盐酸埃他卡林 , K(ir) 6.1/SUR2B activator, pulmonary arterial hypertension, nda
see……….http://apisynthesisint.blogspot.in/2015/12/iptakalim-hydrochloride.html
Sitasentan TBC 11251
Sitasentan,TBC 11251
210421-64-0
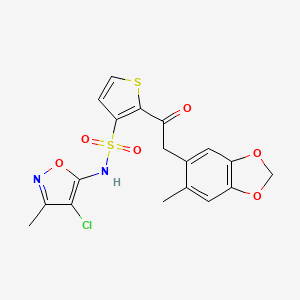
N-(4-chloro-3-methyl-1,2-oxazol-5-yl)-2-[2-(6-methyl-1,3-benzodioxol-5-yl)acetyl]thiophene-3-sulfonamide
Sitaxentan sodium (TBC-11251) is a medication for the treatment of pulmonary arterial hypertension (PAH).[1] It was marketed as Thelin by Encysive Pharmaceuticals until Pfizer purchased Encysive in February 2008. In 2010, Pfizer voluntarily removed sitaxentan from the market due to concerns about liver toxicity.[2]
Sitaxentan belongs to a class of drugs known as endothelin receptor antagonists (ERAs). Patients with PAH have elevated levels of endothelin, a potent blood vessel constrictor, in their plasma and lung tissue. Sitaxentan blocks the binding of endothelin to its receptors, thereby negating endothelin’s deleterious effects.
Mechanism of action
Sitaxentan is a small molecule that blocks the action of endothelin (ET) on the endothelin-A (ETA) receptor selectively (by a factor of 6000 compared to the ETB).[3] It is a sulfonamide class endothelin receptor antagonist (ERA) and is undergoing Food and Drug Administration (FDA) review for treating pulmonary hypertension. The rationale for benefit compared to bosentan, a nonselective ET blocker, is negligible inhibition of the beneficial effects of ETB stimulation, such as nitric oxide production and clearance of ET from circulation. In clinical trials, the efficacy of sitaxentan has been much the same as bosentan, but the hepatotoxicity of sitaxentan outweighs its benefits. Dosing is once daily, as opposed to twice daily for bosentan.
Regulatory status
On December 10, 2010 Pfizer announced it would be withdrawing sitaxentan worldwide (both from marketing and from all clinical study use), citing that it is a cause of fatal liver damage.[2]
Sitaxentan was approved for marketing in the European Union in 2006, in Canada in 2006[4] and in Australia in 2007. By February 2008 it had been launched commercially in Germany, Austria, The Netherlands, the United Kingdom, Ireland, France, Spain and Italy.
In March 2006, the FDA recommended an approvable status to sitaxentan but said it would not yet approve the product. In July 2006, sitaxentan received a second approvable letter stating that efficacy outcome issues raised in the context of the STRIDE-2 study were still unresolved. In July 2007, Encysive commenced a formal dispute resolution process in a preliminary meeting with the FDA. In September 2007 the company announced that it was making preparations for another phase III clinical trial (intended to be named STRIDE-5) to overcome the FDA’s concerns.[5] The takeover by Pfizer resulted in a reconfiguration and extension of these plans, to include combination therapy with sildenafil. The Sitaxentan Efficacy and Safety Trial With a Randomized Prospective Assessment of Adding Sildenafil (SR-PAAS) was an ongoing program of three clinical trials conducted in the United States (ClinicalTtrials.gov identifiers: NCT00795639, NCT00796666 and NCT00796510) with anticipated completion dates between June 2010 and January 2014.

N-(4-Chloro-3-methyl-5-isoxazolyl)-2-[2-(6-methyl-1,3-benzodioxol-5-yl)acetyl]-3-thiophenesulfonamide sodium salt, Sitaxsentan sodium salt, TBC-11251 sodium salt, Thelin
- CAS Number 210421-74-2
- Empirical Formula C18H14ClN2NaO6S2
- Molecular Weight 476.89
Adverse effects
Adverse effects observed with sitaxentan are class effects of endothelin receptor antagonists, and include :
- liver enzyme abnormalities (increased ALT and AST)
- headache
- edema
- constipation
- nasal congestion
- upper respiratory tract infection
- dizziness
- insomnia
- flushing
Because sitaxentan inhibits metabolism of warfarin, a decreased dose of warfarin is needed when co-administered with sitaxentan. This is because warfarin acts to prevent blood from clotting, and if it remains unmetabolized, it can continue to thin the blood.
http://www.google.com/patents/WO2007149568A2?cl=en
As used herein “sitaxsentan” refers to N-(4-chloro-3-methyl-5-isoxazolyl)-2-[2- methyl-4,5-(methylenedioxy)phenylacetyl]-thiophene-3-sulfonamide. Sitaxsentan is also known as TBCl 1251. Other chemical names for sitaxsentan include 4-chloro-3-methyl-5-(2- (2-(6-methylbenzo[d][l ,3]dioxol-5-yl)acetyl)-3-thienylsulfonamido)isoxazole and N-(4- chloro-3-methyl-5-isoxazolyl)-2-[3,4-(methylenedioxy)-6-methylphenylacetyl]-thiophene-3- sulfonamide.
The chemical name for sitaxsentan is N-(4-chloro-3-methyl-5-isoxazolyl)-2-[2- methyl-4,5-(methylenedioxy)phenylacetyl]-thiophene-3-sulfonamide, and its structural formula is as follows:
Sitaxsentan
Sitaxsentan is a potent endothelin receptor antagonist that has oral bioavailability in several species, a long duration of action, and high specificity for ETA receptors.
EXAMPLE 1
Preparation of 4-chloro-3-methyl-5-(2-(2-(6-methylbenzo[d] [l,3|dioxol-5-yl)aeetyl)-3- thienylsulfonamido)isoxazole, or N-(4-chloro-3-methyl-5-isoxazolyl)-2-[2-methy 1-4,5- (methylenedioxy)phenylacetyl]-thiophene-3-sulfonamide, or N-(4-chIoro-3-methyl-5- isoxazolyl)-2-[3,4-(methylenedioxy)-6-methylphenylacetyl]-thiophene-3-sulfonamide.
A. Preparation of (4-chIoro-3-methyl-5-(2-(2-(6-methylbenzo[d] [l,3]dioxol-5-yl)acetyl)- 3-thienylsuIfonamido)isoxazole 1. Preparation of 5-chloromethyI-6-methylbenzo[d][l,3]dioxole
To a mixture of methylene chloride (130 L), concentrated HCl (130 L), and tetrabuylammonium bromide (1.61 Kg) was added 5-methylbenzo[d][l,3]dioxole (10 Kg) followed by the slow addition of formaldehyde (14 L, 37 wt% in water). The mixture was stirred overnight. The organic layer was separated, dried with magnesium sulfate and concentrated to an oil. Hexane (180 L) was added and the mixture heated to boiling. The hot hexane solution was decanted from a heavy oily residue and evaporated to give almost pure 5-chloromethyl-6-methylbenzo[d][l,3]dioxole as a white solid. Recrystallization from hexane (50 L) gave 5-chloromethyl-6-methylbenzo[d][l,3]dioxole (80% recovery after recrystallization). 2. Formation of (4-chloro-3-methyl-5-(2-(2-(2-methyIbenzo[d][l,3]dioxol-5-yl) acetyl)-3-thienylsulfonamido)isoxazole
A portion of a solution of 5-chloromemyl-6-methylbenzo[d][l,3]di-oxole (16.8 g, 0.09 mol) in tetrahydrofuran (THF)(120 mL) was added to a well stirred slurry of magnesium powder, (3.3 g, 0.136 g-atom, Alfa, or Johnson-Mathey, -20 +100 mesh) in THF (120 mL) at room temperature. The resulting reaction admixture was warmed up to about 40-450C for about 2-3 min, causing the reaction to start. Once the heating activated the magnesium, and the reaction began, the mixture was cooled and maintained at a temperature below about 8 0C. The magnesium can be activated with dibromoethane in place of heat.
A flask containing the reaction mixture was cooled and the remaining solution of 5- chloromethlybenzo[d][l,3]dioxole added dropwise during 1.5 hours while maintaining an internal temperature below 8 0C. Temperature control is important: if the Grignard is generated and kept below 8 0C5 Wurtz coupling is suppressed. Longer times at higher temperatures promote the Wurtz coupling pathway. Wurtz coupling can be avoided by using high quality Mg and by keeping the temperature of the Grignard below about 8 0C and stirring vigorously. The reaction works fine at -20 0C, so any temperature below 8 0C is acceptable at which the Grignard will form. The color of the reaction mixture turns greenish.
The reaction mixture was stirred for an additional 5 min at 0 0C, while N2-methoxy- N2-methyl-3-(4-chloro-3-methyl-5-isoazolylsulfamoyl)-2-thiophenecarboxamide (6.6 g, 0.018 mol) in anhydrous THF (90 mL) was charged into the addition funnel. The reaction mixture was degassed two times then the solution of N2-methoxy-N2-methyl-3-(4-chloro-3- methyl-5-isoxazolylsulfamoyl)-2-thiophenecarboxamide was added at 0 0C over 5 min. TLC of the reaction mixture (Silica, 12% MeOHZCH2Cl2) taken immediately after the addition shows no N2-methoxy-N2-methyl-3-(4-chloro-3-methyl-5-isoxazolysulfamoyl)-2-thio- phenecarboxamide. The reaction mixture was transferred into a flask containing IN HCl (400 mL, 0.4 mol
HCl, ice-bath stirred), and the mixture stirred for 2 to 4 min, transferred into a separatory funnel and diluted with ethyl acetate (300 mL). The layers were separated after shaking. The water layer was extracted with additional ethyl acetate (150 mL) and the combined organics washed with half-brine. Following separation, THF was removed by drying the organic layer over sodium sulfate and concentrating under reduced pressure at about 39 0C to obtain the title compound. EXAMPLE 2
1.0 g Sitaxentan was dissolved in 10 ml ethyl acetate and 5 ml hexanes were added. The formed suspension was heated until a clear solution was obtained. Upon cooling light yellow plates were formed. After filtration and drying under vacuum 515 mg of sitaxentan polymorph A was obtained as light yellow plates in very high purity.
EXAMPLE 3
Preparation of 4-chloro-3-methyl-5-(2-(2-(6-methyIbenzo[dJ [l,3]dioxol-5-yl)acetyl)-3- thienylsulfonamido)isoxazole, Sodium Salt
The crystalline sitaxsentan from Example 2 is dissolved in ethyl acetate and washed with saturated NaHCO3 (5 x 10 mL). The solution is washed with brine, dried over Na2SO4 and concentrated in vacuo to obtain a solid residue. 10 mL OfCH2Cl2 is added and the mixture is stirred under nitrogen for 5 to 10 minutes. Ether (15 mL) is added and the mixture stirred for about 10 min. The product is isolated by filtration, washed with a mixture of CH2Cl2 /ether (1 :2) (10 mL) then with ether (10 mL) and dried under reduced pressure to obtain 4-Chloro-3-methyl-5-(2-(2-(6-methyIbenzo[d][l ,3]dioxol-5-yl)acetyl)-3- thienylsulfonamido)isoxazole, sodium salt.
………………………..
Yingyong Huaxue (2007), 24, (11), 1310-1313. Publisher: (Kexue Chubanshe, ) CODEN:YIHUED ISSN:1000-0518.
References
- 2
- Citing liver damage, Pfizer withdraws Thelin, Associated Press, December 12, 2010
- 3
- Girgis, RE; Frost, AE; Hill, NS; Horn, EM; Langleben, D; McLaughlin, VV; Oudiz, RJ; Robbins, IM et al. (2007). “Selective endothelinA receptor antagonism with sitaxsentan for pulmonary arterial hypertension associated with connective tissue disease”. Annals of the rheumatic diseases 66 (11): 1467–72. doi:10.1136/ard.2007.069609. PMC 2111639. PMID 17472992.
- 4
- “UPDATE 1-Encysive gets Canadian approval for hypertension drug”. Reuters. 30 May 2007. Retrieved 2007-07-08.
- 5
- “Encysive Pharmaceuticals to Conduct Phase III Study With Thelin (Sitaxsentan Sodium) in Pulmonary Arterial Hypertension”. PrimeNewswire via COMTEX News Network. 29 September 2007. Retrieved 2007-12-12.
![]()
External links
- http://www.drugs.com/nda/thelin_050714.html
- Mucke HAM: Sitaxsentan for the Oral Treatment of Pulmonary Arterial Hypertension: Benefits from Endothelin Receptor Subtype Selectivity? Clinical Medicine: Therapeutics 2009:1 111–121.
- ATS 2005. The International Conference of the American Thoracic Society. 20–25 May 2005. San Diego, CA.
- Robyn J. Barst, MD; Stuart Rich, MD, FCCP; Allison Widlitz, MS, PA; Evelyn M. Horn, MD; Vallerie McLaughlin, MD and Joyce McFarlin, RN : Clinical Efficacy of Sitaxsentan, an Endothelin-A Receptor Antagonist, in Patients With Pulmonary Arterial Hypertension Chest. 2002;121:1860-1868.
- American Heart Association. Primary or Unexplained Pulmonary Hypertension
| US20010021714 * | Apr 4, 1996 | Sep 13, 2001 | Ming Fai Chan | Compounds such as n-(4-bromo-3-methyl-5-isoxazolyl)-2-n-benzylbenzo(b)thiophene-3-sufonamide administered as endothelin peptide receptor antagonists |
| Reference | ||
|---|---|---|
| 1 | * | WU C ET AL: “Discovery of TBC11251, a Potent, Long Acting, Orally Active Endothelin Receptor-A Selective Antagonist” JOURNAL OF MEDICINAL CHEMISTRY, AMERICAN CHEMICAL SOCIETY. WASHINGTON, US, vol. 40, no. 11, 23 May 1997 (1997-05-23), pages 1690-1697, XP002164198 ISSN: 0022-2623 |
| Patent | Submitted | Granted |
|---|---|---|
| ANTIHYPERTENSIVE THERAPY METHOD [US2007293552] | 2007-12-20 | |
| Crystalline N-(4-chloro-3-methyl-5-isoxazolyl)-2-[2-methyl-4.5-(methylenedioxy)phenylacetyl]-thiophene-3-sulfonamide [US2008026061] | 2008-01-31 | |
| Gnrh agonist combination drugs [US2005215528] | 2005-09-29 | |
| THIENYL-, FURYL-, PYRROLYL- AND BIPHENYLSULFONAMIDES AND DERIVATIVES THEREOF THAT MODULATE THE ACTIVITY OF ENDOTHELIN [WO9631492] | 1996-10-10 | |
| SULFONAMIDES FOR TREATMENT OF ENDOTHELIN-MEDIATED DISORDERS [WO9849162] | 1998-11-05 |
| Patent | Submitted | Granted |
|---|---|---|
| Respiratory Drug Condensation Aerosols and Methods of Making and Using Them [US2009258075] | 2009-10-15 | |
| Method and Composition for Treating Alzheimer’s Disease and Dementias of Vascular Origin [US2010173872] | 2010-07-08 | |
| Method and Composition for Treating Alzheimer’s Disease and Dementias of Vascular Origin [US2010184725] | 2010-07-22 | |
| Formulations of sitaxsentan sodium [US2008076812] | 2008-03-27 | |
| Methods and compositions for treatment of sleep apnea [US2008085313] | 2008-04-10 | |
| Processes for the preparation of 4-chloro-3-methyl-5-(2-(2-(6-methylbenzo[d][1,3]dioxol-5-yl)acetyl)-3-thienylsulfonamido)isoxazole [US2008086010] | 2008-04-10 | |
| Method and composition for treating alzheimer’s disease and dementias of vascular origin [US2004092427] | 2004-05-13 | |
| Method for preventing or treating pulmonary inflammation by administering an endothelin antagonist [US2003004199] | 2003-01-02 | |
| Methods and Compositions for Treatment of an Interstitial Lung Disease [US2009004268] | 2009-01-01 | |
| Methods and compositions for treatment of diastolic heart failure [US2007232671] | 2007-10-04 | |
| Patent | Submitted | Granted |
|---|---|---|
| Isoxazolyl endothelin antagonists [US6043265] | 2000-03-28 | |
| Aminoguanidine hydrazone derivatives, process for producing the same and drugs thereof [US6350749] | 2002-02-26 | |
| Method for preventing or treating pain by administering an endothelin antagonist [US6573285] | 2002-06-27 | 2003-06-03 |
| Method for preventing or treating erectile dysfunction by administering an endothelin antagonist [US6268388] | 2001-07-31 | |
| Method and composition for potentiating the antipyretic action of a nonopioid analgesic [US7351692] | 2003-12-25 | 2008-04-01 |
| Method and Composition for Potentiating an Opiate Analgesic [US8114896] | 2010-05-06 | 2012-02-14 |
| SUBSTITUTED THIOPHENES [US7863308] | 2008-10-16 | 2011-01-04 |
| Respiratory drug condensation aerosols and methods of making and using them [US7550133] | 2004-06-03 | 2009-06-23 |
| SUBSTITUTED THIOPHENES [US2010280086] | 2010-11-04 | |
| Method and Composition for Potentiating an Opiate Analgesic [US2010311665] | 2010-12-09 |
 |
|
| Systematic (IUPAC) name | |
|---|---|
| N-(4-chloro-3-methyl-1,2-oxazol-5-yl)-2-[2-(6-methyl-2H-1,3-benzodioxol-5-yl)acetyl]thiophene-3-sulfonamide | |
| Clinical data | |
| AHFS/Drugs.com | International Drug Names |
| Licence data | EMA:Link |
| Legal status | |
| Routes | Oral |
| Pharmacokinetic data | |
| Bioavailability | 70 to 100% |
| Protein binding | >99% |
| Metabolism | Hepatic (CYP2C9– and CYP3A4-mediated) |
| Half-life | 10 hours |
| Excretion | Renal (50 to 60%) Fecal (40 to 50%) |
| Identifiers | |
| CAS number | 184036-34-8 210421-64-0 (sodium salt) |
| ATC code | C02KX03 |
| PubChem | CID 216235 |
| IUPHAR ligand | 3950 |
| DrugBank | DB06268 |
| ChemSpider | 21106381 |
| UNII | J9QH779MEM |
| KEGG | D07171 |
| ChEMBL | CHEMBL282724 |
| Synonyms | Sitaxsentan; TBC-11251 |
| Chemical data | |
| Formula | C18H15ClN2O6S2 |
| Molecular mass | 454.906 g/mol |
![]()

Structures and observed activities of the ETA receptor antagonists for the HipHop training set
 COCK WILL TEACH YOU NMR
COCK WILL TEACH YOU NMR COCK SAYS MOM CAN TEACH YOU NMR
COCK SAYS MOM CAN TEACH YOU NMR

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE amcrasto@gmail.com
amcrasto@gmail.com
TAKE A TOUR
KISUMU, KENYA

Kisumu – Wikipedia, the free encyclopedia
Kisumu is a port city in Kisumu County, Kenya 1,131 m (3,711 ft), with a population of 409,928 (2009 census). It is the third largest city in Kenya, the principal city …
Kisumu CountyKisumu County is one of the new devolved counties of Kenya. Its …
|
Kisumu International AirportKisumu International Airport is an airport in Kisumu, Kenya (IATA …
|
Boat riding in Kisumu
Local inhabitants near Kisumu, 1911


Clockwise: Lake Victoria Panorama, Kisumu Panorama, sunset at Oginga Odinga street, Downtown, Kiboko Point, Nighttime in Kisumu and Jomo Kenyatta Stadium.
|
|
Coordinates:  0°6′S 34°45′E 0°6′S 34°45′E |
|
| Country | |
|---|---|
| County | Kisumu County |
Kisumu panorama, viewed from Lake Victoria