1H and 13C NMR spectra of 2-acyl-substituted cyclohexane-1,3-diones (acyl = formyl, 1; 2-nitrobenzoyl, 2; 2-nitro-4-trifluoromethylbenzoyl, 3) and lithium sodium and potassium salts of 1have been measured. The compound 3, known as NTBC, is a life-saving medicine applied in tyrosinemia type I. The optimum molecular structures of the investigated objects in solutions have been found using the DFT method with B3LYP functional and 6-31G** and/or 6-311G(2d,p) basis set. The theoretical values of the NMR parameters of the investigated compounds have been calculated using GIAO DFT B3LYP/6-311G(2d,p) method. The theoretical data obtained for compounds 1−3 have been exploited to interpret their experimental NMR spectra in terms of the equilibrium between different tautomers. It has been found that for these triketones an endo-tautomer prevails. The differences in NMR spectra of the salts of 1 can be rationalized taking into account the size of the cation and the degree of salt dissociation. It seems that in DMSO solution the lithium salt exists mainly as an ion pair stabilized by the chelation of a lithium cation with two oxygen atoms. The activation free energy the of formyl group rotation for this salt has been estimated to be 51.5 kJ/mol. The obtained results suggest that in all the investigated objects, including the free enolate ions, all atoms directly bonded to the carbonyl carbons lie near the same plane. Some observations concerning the chemical shift changes could indicate strong solvation of the anion of 1 by water molecules. Implications of the results obtained in this work for the inhibition mechanism of (4-hydroxyphenyl) pyruvate dioxygenase by NTBC are commented upon.
Home » Posts tagged 'PRIORITY'
Tag Archives: PRIORITY
Pafolacianine

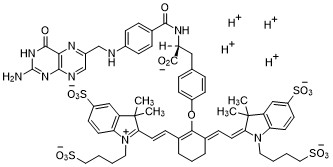
Pafolacianine
OTL-38
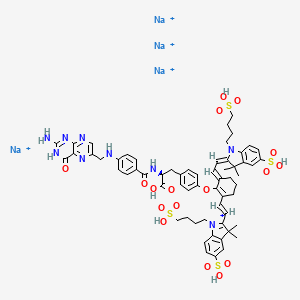
- Molecular FormulaC61H67N9O17S4
- Average mass1326.495 Da
FDA APPROVED NOV 2021
2-{(E)-2-[(3E)-2-(4-{2-[(4-{[(2-Amino-4-oxo-3,4-dihydro-6-pteridinyl)methyl]amino}benzoyl)amino]-2-carboxyethyl}phenoxy)-3-{(2E)-2-[3,3-dimethyl-5-sulfo-1-(4-sulfobutyl)-1,3-dihydro-2H-indol-2-ylidene ]ethylidene}-1-cyclohexen-1-yl]vinyl}-3,3-dimethyl-1-(4-sulfobutyl)-3H-indolium-5-sulfonate OTL-38Tyrosine, N-[4-[[(2-amino-3,4-dihydro-4-oxo-6-pteridinyl)methyl]amino]benzoyl]-O-[(6E)-6-[(2E)-2-[1,3-dihydro-3,3-dimethyl-5-sulfo-1-(4-sulfobutyl)-2H-indol-2-ylidene]ethylidene]-2-[(E)-2-[3,3-dimethy l-5-sulfo-1-(4-sulfobutyl)-3H-indolium-2-yl]ethenyl]-1-cyclohexen-1-yl]-, inner salt
2-(2-(2-(4-((2S)-2-(4-(((2-amino-4-oxo-3,4-dihydropteridin-6-yl)methyl)amino)benzamido)-2-carboxyethyl)phenoxy)-3-(2-(3,3-dimethyl-5-sulfo-1-(4-sulfobutyl)-1,3-dihydro-2H-indol-2-ylidene)ethylidene)cyclohex-1-en-1-yl)ethenyl)-3,3-dimethyl-5-sulfo-1-(4-sulfobutyl)-3H-indolium inner salt,sodium salt (1:4)
- 3H-Indolium, 2-(2-(2-(4-((2S)-2-((4-(((2-amino-3,4-dihydro-4-oxo-6-pteridinyl)methyl)amino)benzoyl)amino)-2-carboxyethyl)phenoxy)-3-(2-(1,3-dihydro-3,3-dimethyl-5-sulfo-1-(4-sulfobutyl)-2H-indol-2-ylidene)ethylidene)-1-cyclohexen-1-yl)ethenyl)-3,3-dimethyl-5-sulfo-1 (4-sulfobutyl)-, inner salt,sodium salt (1:4)
1628423-76-6 [RN]
Pafolacianine sodium [USAN]
RN: 1628858-03-6
UNII: 4HUF3V875C
C61H68N9Na4O17S4+5
- Intraoperative Imaging and Detection of Folate Receptor Positive Malignant Lesions
Pafolacianine, sold under the brand name Cytalux, is an optical imaging agent.[1][2]
The most common side effects of pafolacianine include infusion-related reactions, including nausea, vomiting, abdominal pain, flushing, dyspepsia, chest discomfort, itching and hypersensitivity.[2]
It was approved for medical use in the United States in November 2021.[2][3]
Pafolacianine is a fluorescent drug that targets folate receptor (FR).[1]
Medical uses
Pafolacianine is indicated as an adjunct for intraoperative identification of malignant lesions in people with ovarian cancer.[1][2]
History
The safety and effectiveness of pafolacianine was evaluated in a randomized, multi-center, open-label study of women diagnosed with ovarian cancer or with high clinical suspicion of ovarian cancer who were scheduled to undergo surgery.[2] Of the 134 women (ages 33 to 81 years) who received a dose of pafolacianine and were evaluated under both normal and fluorescent light during surgery, 26.9% had at least one cancerous lesion detected that was not observed by standard visual or tactile inspection.[2]
The U.S. Food and Drug Administration (FDA) granted the application for pafolacianine orphan drug, priority review, and fast track designations.[2][4] The FDA granted the approval of Cytalux to On Target Laboratories, LLC.[2]

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////////////////////////////
SYN

WO 2014149073
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2014149073
In another aspect of the invention, this disclosure provides a method of synthesizing a compound having the formula
[0029] In a fourth embodiment of the invention, this disclosure provides a method of synthesizing a compound having the formula
[0030]
[0032] wherein C is any carbon isotope. In this embodiment, the amino acid linker is selected from a group consisting of methyl 2-di-tert-butyl dicarbonate-amino-3-(4-phenyl)propanoate, 3-(4-hydroxyphenyl)-2-(di-tert-butyl-dicarbonate methylamino)propanoic acid, 2-amino-4-(4-hydroxyphenyl)butanoic acid, and Tert-butyl (2-di-tert-butyl dicarbonate- amino)-3-(4-hydroxyphenyl)propanoate . In a particular embodiment, the aqueous base is potassium hydroxide (KOH). The method of this embodiment may also further include purifying the compound by preparatory HPLC.
EXAMPLE 1 : General synthesis of Pte – L Tyrosine – S0456 (OTL-0038)
[0088] Scheme:
C33H37CIF3N
Reactants for Step I:
[0089] A 500 mL round bottom flask was charged with a stirring bar, pteroic acid
(12.0 g, 29.40 mmol, 1 equiv), (L)-Tyr(-OfBu)-OfBu- HCI (1 1 .63 g, 35.28 mmol, 1 .2
equiv) and HATU (13.45 g, 35.28 mmol, 1 .2 equiv) then DMF (147 mL) was added to give a brown suspension [suspension A]. DIPEA (20.48 mL, 1 17.62 mmol, 4.0 equiv) was added slowly to suspension A at 23 °C, over 5 minutes. The suspension turned in to a clear brown solution within 10 minutes of addition of DIPEA. The reaction was stirred at 23 °C for 2.5 h. Reaction was essentially complete in 30 minutes as judged by LC/MS but was stirred further for 2.5 h. The formation of Pte_N10(TFA)_L_Tyr(-OfBu)-OfBu HCI (Figure 12) was confirmed by LC/MS showing m/z 409→m/z 684. LC/MS method: 0-50% acetonitrile in 20 mM aqueous NH4OAc for 5 min using Aquity UPLC-BEH C18, 1 .7μιη 2.1 * 50 mm column . The reaction mixture was cannulated as a steady stream to a stirred solution of aq. HCI (2.0 L, 0.28 M) over the period of 30 minutes to give light yellow precipitate of Pte_N10(TFA)_L_Tyr(-OfBu)-OfBu HCI. The precipitated Pte_N 10(TFA)_L_Tyr(- OfBu)-OfBu HCI was filtered using sintered funnel under aspirator vacuum, washed with water (8 * 300 mL) until the pH of the filtrate is between 3 and 4. The wet solid was allowed to dry under high vacuum for 12 hours on the sintered funnel. In a separate batch, where this wet solid (3) was dried under vacuum for 48 hours and then this solid was stored at -20 0 C for 48 h. However, this brief storage led to partial decomposition of 3. The wet cake (58 g) was transferred to a 500 mL round bottom flask and was submitted to the next step without further drying or purification.
Reactants for Step II:
The wet solid (58 g) was assumed to contain 29.40 mmol of the desired compound (3) (i. e. quantitative yield for the step I ).
[0090] A 500 mL round bottom flask was charged with a stirring bar, Pte_N10(TFA)_L_Tyr(-OfBu)-OfBu HCI as a wet cake (58 g, 29.40 mmol, 1 equiv). A solution of TFA:TIPS:H20 (95:2.5:2.5, 200 mL) was added at once to give a light brown suspension. The reaction content was stirred at 23°C for 1 .5 hours and was monitored by LC/MS. The suspension became clear dull brown solution after stirring for 5 minutes. LC/MS method: 0-50% acetonitrile in 20 mM aqueous NH4OAc for 5 min using Aquity UPLC-BEH C18, 1 .7μιη 2.1 * 50 mm column. The formation of Pte_TFA_L_Tyr (Figure 12) was confirmed by showing m/z 684→m/z 572. Reaction time varies from 30 min to 1 .5 hours depending on the water content of Pte_N10(TFA)_L_Tyr(-OfBu)-OfBu HCI. The reaction mixture was cannulated as a steady stream to a stirred MTBE (1 .8 L) at 23 °C or 100 °C to give light yellow precipitate of Pte_TFA_L_Tyr. The precipitated Pte_TFA_L_Tyr was filtered using sintered funnel under aspirator vacuum, washed with MTBE (6 * 300 mL) and dried under high vacuum for 8 hours to obtain Pte_TFA_L_Tyr (14.98 g, 83.98% over two steps) as a pale yellow solid. The MTBE washing was tested for absence of residual TFA utilizing wet pH paper (pH between 3-4). The yield of the reaction was between 80-85% in different batches. The deacylated side product was detected in 3.6% as judged by LC/MS. For the different batches this impurity was never more than 5%.
Reactants for Step III:
[0091] A 200 mL round bottom flask was charged with a stirring bar and Pte_TFA_L_Tyr (13.85 g, 22.78 mmol, 1 equiv), then water (95 mL) was added to give a yellow suspension [suspension B]. A freshly prepared solution of aqueous 3.75 M NaOH (26.12 mL, 97.96 mmol, 4.30 equiv), or an equivalent base at a corresponding temperature using dimethylsulfoxide (DMSO) as a solvent (as shown in Table 1 ), was added dropwise to suspension B at 23 °C, giving a clear dull yellow solution over 15 minutes [solution B]. The equivalence of NaOH varied from 3.3 to 5.0 depending on the source of 4 (solid or liquid phase synthesis) and the residual TFA. Trianion 5 (Figure 12) formation was confirmed by LC/MS showing m/z 572→m/z 476 while the solution pH was 9-10 utilizing wet pH paper. The pH of the reaction mixture was in the range of 9-10. This pH is crucial for the overall reaction completion. Notably, pH more than 10 leads to hydrolysis of S0456. Excess base will efficiently drive reaction forward with potential hydrolysis of S0456. The presence of hydrolysis by product can be visibly detected by the persistent opaque purple/blue to red/brown color.
TABLE 1 : Separate TFA deprotection via trianion formation; S0456
[0092] The precipitated OTL-0038 product could also be crashed out by adding the reaction solution steady dropwise to acetone, acetonitrile, isopropanol or ethyl acetate/acetone mixture. Acetone yields optimal results. However, viscous reactions could be slower due to partial insolubility and/or crashing out of S0456. In this reaction, the equivalence of the aqueous base is significant. Excess base will efficiently drive reaction forward with potential hydrolysis of S0456. This solution phase synthesis provides Pte_N10(TFA)_Tyr-OH »HCI salt and desires approximately 4.1 to approximately 4.8 equiv base as a source to hydrolyze the product. Particularly, precipitation of Pte_Tyr_S0456 was best achieved when 1 mL of reaction mixture is added dropwise to the stirred acetone (20 mL). Filtration of the precipitate and washing with acetone (3 x10 mL) gave the highest purity as judged from LC/MS chromatogram.
[0093] During experimentation of this solution-phase synthesis of Pte – L Tyrosine -S0456 (OTL-0038) at different stages, some optimized conditions were observed:
Mode of addition: Separate TFA deprotection via trianion formation; S0456 @ 23 °C; reflux.
Stability data of Pte – L Tyrosine – S0456 (OTL-0038):
Liquid analysis: At 40 °C the liquid lost 8.6% at 270 nm and 1 % at 774 nm. At room temperature the liquid lost about 1 .4% at 270 nm and .5% at 774 nm. At 5 °C the
270 nm seems stable and the 774 nm reasonably stable with a small degradation purity.
Source Purity Linker S0456 Base Solvent Duration % Conversion
4.3-4.6
Solution 0.95
95% 1 equiv equiv H20 15 min 100% phase equiv
K2C03
PATENT
US 20140271482
FDA approves pafolacianine for identifying malignant ovarian cancer lesions
On November 29, 2021, the Food and Drug Administration approved pafolacianine (Cytalux, On Target Laboratories, LLC), an optical imaging agent, for adult patients with ovarian cancer as an adjunct for interoperative identification of malignant lesions. Pafolacianine is a fluorescent drug that targets folate receptor which may be overexpressed in ovarian cancer. It is used with a Near-Infrared (NIR) fluorescence imaging system cleared by the FDA for specific use with pafolacianine.
Efficacy was evaluated in a single arm, multicenter, open-label study (NCT03180307) of 178 women diagnosed with ovarian cancer or with high clinical suspicion of ovarian cancer scheduled to undergo primary surgical cytoreduction, interval debulking, or recurrent ovarian cancer surgery. All patients received pafolacianine. One hundred and thirty-four patients received fluorescence imaging evaluation in addition to standard of care evaluation which includes pre-surgical imaging, intraoperative palpation and normal light evaluation of lesions. Among these patients, 36 (26.9%) had at least one evaluable ovarian cancer lesion detected with pafolacianine that was not observed by standard visual or tactile inspection. The patient-level false positive rate of pafolacianine with NIR fluorescent light with respect to the detection of ovarian cancer lesions confirmed by central pathology was 20.2% (95% CI 13.7%, 28.0%).
The most common adverse reactions (≥1%) occurring in patients were nausea, vomiting, abdominal pain, flushing, dyspepsia, chest discomfort, pruritus, and hypersensitivity.
The recommended pafolacianine dose is 0.025 mg/kg administered intravenously over 60 minutes, 1 to 9 hours before surgery. The use of folate, folic acid, or folate-containing supplements should be avoided within 48 hours before administration of pafolacianine.
View full prescribing information for Cytalux.
This application was granted priority review, fast track designation, and orphan drug designation. A description of FDA expedited programs is in the Guidance for Industry: Expedited Programs for Serious Conditions-Drugs and Biologics.
USFDA approves new drug to help identify cancer lesions
This drug is indicated for use in adult patients with ovarian cancer to help identify cancerous lesions during surgery.By The Health Master -December 2, 2021
The U.S. Food and Drug Administration (USFDA) has approved Cytalux (pafolacianine), an imaging drug intended to assist surgeons in identifying ovarian cancer lesions. The drug is designed to improve the ability to locate additional ovarian cancerous tissue that is normally difficult to detect during surgery.
Cytalux is indicated for use in adult patients with ovarian cancer to help identify cancerous lesions during surgery. The drug is a diagnostic agent that is administered in the form of an intravenous injection prior to surgery.
Alex Gorovets, M.D., deputy director of the Office of Specialty Medicine in the FDA’s Center for Drug Evaluation and Research said, “The FDA’s approval of Cytalux can help enhance the ability of surgeons to identify deadly ovarian tumors that may otherwise go undetected.
By supplementing current methods of detecting ovarian cancer during surgery, Cytalux offers health care professionals an additional imaging approach for patients with ovarian cancer.”
The American Cancer Society estimates there will be more than 21,000 new cases of ovarian cancer and more than 13,000 deaths from this disease in 2021, making it the deadliest of all female reproductive system cancers.
Conventional treatment for ovarian cancer includes surgery to remove as many of the tumors as possible, chemotherapy to stop the growth of malignant cells or other targeted therapy to identify and attack specific cancer cells.
Ovarian cancer often causes the body to overproduce a specific protein in cell membranes called a folate receptor. Following administration via injection, Cytalux binds to these proteins and illuminates under fluorescent light, boosting surgeons’ ability to identify the cancerous tissue.
Currently, surgeons rely on preoperative imaging, visual inspection of tumors under normal light or examination by touch to identify cancer lesions. Cytalux is used with a Near-Infrared fluorescence imaging system cleared by the FDA for specific use with pafolacianine.
The safety and effectiveness of Cytalux was evaluated in a randomized, multi-center, open-label study of women diagnosed with ovarian cancer or with high clinical suspicion of ovarian cancer who were scheduled to undergo surgery.
Of the 134 women (ages 33 to 81 years) who received a dose of Cytalux and were evaluated under both normal and fluorescent light during surgery, 26.9% had at least one cancerous lesion detected that was not observed by standard visual or tactile inspection.
The most common side effects of Cytalux were infusion-related reactions, including nausea, vomiting, abdominal pain, flushing, dyspepsia, chest discomfort, itching and hypersensitivity. Cytalux may cause fetal harm when administered to a pregnant woman.
The use of folate, folic acid, or folate-containing supplements should be avoided within 48 hours before administration of Cytalux. There is a risk of image interpretation errors with the use of Cytalux to detect ovarian cancer during surgery, including false negatives and false positives.
References
- ^ Jump up to:a b c d https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/214907s000lbl.pdf
- ^ Jump up to:a b c d e f g h i “FDA Approves New Imaging Drug to Help Identify Ovarian Cancer Lesions”. U.S. Food and Drug Administration (FDA) (Press release). 29 November 2021. Retrieved 30 November 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ “On Target Laboratories Announces FDA Approval of Cytalux (pafolacianine) injection for Identification of Ovarian Cancer During Surgery”. On Target Laboratories. 29 November 2021. Retrieved 30 November 2021 – via PR Newswire.
- ^ “Pafolacianine Orphan Drug Designations and Approvals”. U.S. Food and Drug Administration (FDA). 23 December 2014. Retrieved 30 November 2021.
External links
- “Pafolacianine”. Drug Information Portal. U.S. National Library of Medicine.
| Clinical data | |
|---|---|
| Trade names | Cytalux |
| Other names | OTL-0038 |
| License data | US DailyMed: Pafolacianine |
| Pregnancy category | Not recommended |
| Routes of administration | Intravenous |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only [1][2] |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 1628423-76-6 |
| PubChem CID | 135565623 |
| DrugBank | DB15413 |
| ChemSpider | 64880249 |
| UNII | F7BD3Z4X8L |
| ChEMBL | ChEMBL4297412 |
| Chemical and physical data | |
| Formula | C61H67N9O17S4 |
| Molar mass | 1326.49 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI |
////////////Pafolacianine, FDA 2021, APPROVALS 2021, Cytalux, OVARIAN CANCER, OTL 38,
[Na+].[Na+].[Na+].[Na+].CC1(C)\C(=C/C=C/2\CCCC(=C2Oc3ccc(C[C@H](NC(=O)c4ccc(NCc5cnc6N=C(N)NC(=O)c6n5)cc4)C(=O)O)cc3)\C=C\C7=[N](CCCCS(=O)(=O)O)c8ccc(cc8C7(C)C)S(=O)(=O)O)\N(CCCCS(=O)(=O)O)c9ccc(cc19)S(=O)(=O)O

NEW DRUG APPROVALS
ONE TIME
$10.00
Brilliant blue G , ブリリアントブルーG ,
Brilliant blue G
FDA 2019, 12/20/2019, TISSUEBLUE, New Drug Application (NDA): 209569
Company: DUTCH OPHTHALMIC, PRIORITY; Orphan
OPQ recommends APPROVAL of NDA 209569 for commercialization of TissueBlue (Brilliant Blue G Ophthalmic Solution), 0.025%
Neuroprotectant
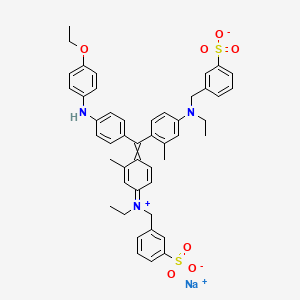
sodium;3-[[4-[[4-(4-ethoxyanilino)phenyl]-[4-[ethyl-[(3-sulfonatophenyl)methyl]azaniumylidene]-2-methylcyclohexa-2,5-dien-1-ylidene]methyl]-N-ethyl-3-methylanilino]methyl]benzenesulfonate
| Formula |
C47H48N3O7S2. Na
|
|---|---|
| CAS |
6104-58-1
|
| Mol weight |
854.0197
|
ブリリアントブルーG, C.I. Acid Blue 90
UNII-M1ZRX790SI
M1ZRX790SI
6104-58-1
Brilliant Blue G
Derma Cyanine G
SYN


////////////Brilliant blue G , ブリリアントブルーG , C.I. Acid Blue 90, FDA 2019, PRIORITY, Orphan
CCN(CC1=CC(=CC=C1)S(=O)(=O)[O-])C2=CC(=C(C=C2)C(=C3C=CC(=[N+](CC)CC4=CC(=CC=C4)S(=O)(=O)[O-])C=C3C)C5=CC=C(C=C5)NC6=CC=C(C=C6)OCC)C.[Na+]
- Benzenemethanaminium, N-[4-[[4-[(4-ethoxyphenyl)amino]phenyl][4-[ethyl[(3-sulfophenyl)methyl]amino]-2-methylphenyl]methylene]-3-methyl-2,5-cyclohexadien-1-ylidene]-N-ethyl-3-sulfo-, hydroxide, inner salt, monosodium salt
- Benzenemethanaminium, N-[4-[[4-[(4-ethoxyphenyl)amino]phenyl][4-[ethyl[(3-sulfophenyl)methyl]amino]-2-methylphenyl]methylene]-3-methyl-2,5-cyclohexadien-1-ylidene]-N-ethyl-3-sulfo-, inner salt, monosodium salt (9CI)
- Brilliant Indocyanine G (6CI)
- C.I. Acid Blue 90 (7CI)
- C.I. Acid Blue 90, monosodium salt (8CI)
- Acid Blue 90
- Acid Blue G 4061
- Acid Blue PG
- Acid Bright Blue G
- Acid Brilliant Blue G
- Acid Brilliant Cyanine G
- Acidine Sky Blue G
- Amacid Brilliant Cyanine G
- Anadurm Cyanine A-G
- BBG
- Benzyl Cyanine G
- Biosafe Coomassie Stain
- Boomassie blue silver
- Brilliant Acid Blue G
- Brilliant Acid Blue GI
- Brilliant Acid Blue J
- Brilliant Acid Cyanine G
- Brilliant Blue G
- Brilliant Blue G 250
- Brilliant Blue J
- Brilliant Indocyanine GA-CF
- Bucacid Brilliant Indocyanine G
- C.I. 42655
- CBB-G 250
- Colocid Brilliant Blue EG
- Coomassie Blue G
- Coomassie Blue G 250
- Coomassie Brilliant Blue G
- Coomassie Brilliant Blue G 250
- Coomassie G 250
- Cyanine G
- Daiwa Acid Blue 300
- Derma Cyanine G
- Derma Cyanine GN 360
- Dycosweak Acid Brilliant Blue G
- Eriosin Brilliant Cyanine G
- Fenazo Blue XXFG
- Impero Azure G
- Kayanol Cyanine G
- Lerui Acid Brilliant Blue G
- Milling Brilliant Blue 2J
- NSC 328382
- Optanol Cyanine G
- Orient Water Blue 105
- Orient Water Blue 105S
- Polar Blue G
- Polar Blue G 01
- Polycor Blue G
- Sandolan Cyanine N-G
- Sellaset Blue B
- Serva Blue G
- Serva Blue G 250
- Silk Fast Cyanine G
- Simacid Blue G 350
- Sumitomo Brilliant Indocyanine G
- Supranol Cyanin G
- Supranol Cyanine G
- TissueBlue
- Triacid Fast Cyanine G
- Water Blue 105
- Water Blue 105S
- Water Blue 150
- Xylene Brilliant Cyanine G
Avapritinib, アバプリチニブ , авапритиниб , أفابريتينيب ,

Avapritinib
BLU-285, BLU285
Antineoplastic, Tyrosine kinase inhibitor
アバプリチニブ
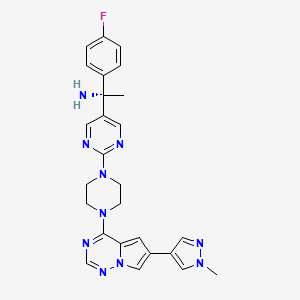
(1S)-1-(4-fluorophenyl)-1-[2-[4-[6-(1-methylpyrazol-4-yl)pyrrolo[2,1-f][1,2,4]triazin-4-yl]piperazin-1-yl]pyrimidin-5-yl]ethanamine
| Formula |
C26H27FN10
|
|---|---|
| CAS |
1703793-34-3
|
| Mol weight |
498.558
|
| No. | Drug Name | Active Ingredient | Approval Date | FDA-approved use on approval date* |
|---|---|---|---|---|
| 1. | Ayvakit | avapritinib | 1/9/2020 | To treat adults with unresectable or metastatic gastrointestinal stromal tumor (GIST) |
PRIORITY; Orphan, NDA 212608
Avapritinib, sold under the brand name Ayvakit, is a medication used for the treatment of tumors due to one specific rare mutation: It is specifically intended for adults with unresectable or metastatic ( y) gastrointestinal stromal tumor (GIST) that harbor a platelet-derived growth factor receptor alpha (PDGFRA) exon 18 mutation.[1]
Common side effects are edema (swelling), nausea, fatigue/asthenia (abnormal physical weakness or lack of energy), cognitive impairment, vomiting, decreased appetite, diarrhea, hair color changes, increased lacrimation (secretion of tears), abdominal pain, constipation, rash. and dizziness.[1]
Ayvakit is a kinase inhibitor.[1]
History
The U.S. Food and Drug Administration (FDA) approved avapritinib in January 2020.[1] The application for avapritinib was granted fast track designation, breakthrough therapy designation, and orphan drug designation.[1] The FDA granted approval of Ayvakit to Blueprint Medicines Corporation.[1]
Avapritinib was approved based on the results from the Phase I NAVIGATOR[2][3] clinical trial involving 43 patients with GIST harboring a PDGFRA exon 18 mutation, including 38 subjects with PDGFRA D842V mutation.[1] Subjects received avapritinib 300 mg or 400 mg orally once daily until disease progression or they experienced unacceptable toxicity.[1] The recommended dose was determined to be 300 mg once daily.[1] The trial measured how many subjects experienced complete or partial shrinkage (by a certain amount) of their tumors during treatment (overall response rate).[1] For subjects harboring a PDGFRA exon 18 mutation, the overall response rate was 84%, with 7% having a complete response and 77% having a partial response.[1] For the subgroup of subjects with PDGFRA D842V mutations, the overall response rate was 89%, with 8% having a complete response and 82% having a partial response.[1] While the median duration of response was not reached, 61% of the responding subjects with exon 18 mutations had a response lasting six months or longer (31% of subjects with an ongoing response were followed for less than six months).[1]
PATENT
WO 2015057873
https://patents.google.com/patent/WO2015057873A1/en
Example 7: Synthesis of (R)-l-(4-fluorophenyl)- l-(2-(4-(6-(l-methyl-lH-pyrazol-4- yl)pyrrolo[2, 1 -f\ [ 1 ,2,4] triazin-4-yl)piperazin- 1 -yl)pyrimidin-5-yl)ethanamine and (S)- 1 – (4- fluorophenyl)- l-(2-(4-(6-(l-methyl-lH-pyrazol-4-yl)pyrrolo[2, l-/] [l,2,4]triazin-4-yl)piperazin- l-yl)pyrimidin-5-yl)ethanamine (Compounds 43 and 44)
Step 1 : Synthesis of (4-fluorophenyl)(2-(4-(6-(l-methyl- lH-pyrazol-4-yl)pyrrolo[2,l- f] [ 1 ,2,4] triazin-4-yl)piperazin- 1 -yl)pyrimidin-5-yl)methanone:
4-Chloro-6-(l-methyl- lH-pyrazol-4-yl)pyrrolo[2,l-/] [l,2,4]triazine (180 mg, 0.770 mmol), (4-fluorophenyl)(2-(piperazin-l-yl)pyrimidin-5-yl)methanone, HC1 (265 mg, 0.821 mmol) and DIPEA (0.40 mL, 2.290 mmol) were stirred in 1,4-dioxane (4 mL) at room temperature for 18 hours. Saturated ammonium chloride was added and the products extracted into DCM (x2). The combined organic extracts were dried over Na2S04, filtered through Celite eluting with DCM, and the filtrate concentrated in vacuo. Purification of the residue by MPLC (25- 100% EtOAc-DCM) gave (4-fluorophenyl)(2-(4-(6-(l-methyl-lH-pyrazol-4-yl)pyrrolo[2,l- ] [l,2,4]triazin-4-yl)piperazin- l-yl)pyrimidin-5-yl)methanone (160 mg, 0.331 mmol, 43 % yield) as an off-white solid. MS (ES+) C25H22FN90 requires: 483, found: 484 [M + H]+.
Step 2: Synthesis of (5,Z)-N-((4-fluorophenyl)(2-(4-(6-(l-methyl- lH-p razol-4-yl)p rrolo[2, l- ] [l,2,4]triazin-4- l)piperazin- l-yl)pyrimidin-5-yl)methylene)-2-methylpropane-2-sulfinamide:
(S)-2-Methylpropane-2-sulfinamide (110 mg, 0.908 mmol), (4-fluorophenyl)(2-(4-(6-(l- methyl- lH-pyrazol-4-yl)pyrrolo[2,l-/][l,2,4]triazin-4-yl)piperazin- l-yl)pyrimidin-5- yl)methanone (158 mg, 0.327 mmol) and ethyl orthotitanate (0.15 mL, 0.715 mmol) were stirred in THF (3.2 mL) at 70 °C for 18 hours. Room temperature was attained, water was added, and the products extracted into EtOAc (x2). The combined organic extracts were washed with brine, dried over Na2S04, filtered, and concentrated in vacuo while loading onto Celite. Purification of the residue by MPLC (0- 10% MeOH-EtOAc) gave (5,Z)-N-((4-fluorophenyl)(2-(4-(6-(l-methyl- lH-pyrazol-4-yl)pyrrolo[2, l-/] [l,2,4]triazin-4-yl)piperazin-l-yl)pyrimidin-5-yl)methylene)-2- methylpropane-2-sulfinamide (192 mg, 0.327 mmol, 100 % yield) as an orange solid. MS (ES+) C29H3iFN10OS requires: 586, found: 587 [M + H]+.
Step 3: Synthesis of (lS’)-N-(l-(4-fluorophenyl)- l-(2-(4-(6-(l-methyl- lH-pyrazol-4- l)pyrrolo[2, l-/] [l,2,4]triazin-4-yl)piperazin-l-yl)pyrimidin-5-yl)ethyl)-2-methylpropane-2-
(lS’,Z)-N-((4-Fluorophenyl)(2-(4-(6-(l-methyl-lH-pyrazol-4-yl)pyrrolo[2,l- ] [l,2,4]triazin-4-yl)piperazin- l-yl)pyrimidin-5-yl)methylene)-2-methylpropane-2-sulfinamide (190 mg, 0.324 mmol) was taken up in THF (3 mL) and cooled to 0 °C. Methylmagnesium bromide (3 M solution in diethyl ether, 0.50 mL, 1.500 mmol) was added and the resulting mixture stirred at 0 °C for 45 minutes. Additional methylmagnesium bromide (3 M solution in diethyl ether, 0.10 mL, 0.300 mmol) was added and stirring at 0 °C continued for 20 minutes. Saturated ammonium chloride was added and the products extracted into EtOAc (x2). The combined organic extracts were washed with brine, dried over Na2S04, filtered, and concentrated in vacuo while loading onto Celite. Purification of the residue by MPLC (0-10% MeOH-EtOAc) gave (lS’)-N-(l-(4-fluorophenyl)-l-(2-(4-(6-(l-methyl- lH-pyrazol-4-yl)pyrrolo[2, l- ] [l,2,4]triazin-4-yl)piperazin- l-yl)pyrimidin-5-yl)ethyl)-2-methylpropane-2-sulfinamide (120 mg, 0.199 mmol, 61.5 % yield) as a yellow solid (mixture of diastereoisomers). MS (ES+) C3oH35FN10OS requires: 602, found: 603 [M + H]+. Step 4: Synthesis of l-(4-fluorophenyl)- l-(2-(4-(6-(l-methyl- lH-pyrazol-4-yl)pyrrolo[2,l- f\ [ 1 ,2,4] triazin-4- l)piperazin- 1 -yl)pyrimidin-5-yl)ethanamine:
(S)-N- ( 1 – (4-Fluorophenyl)- 1 -(2- (4- (6-( 1 -methyl- 1 H-pyrazol-4-yl)pyrrolo [2,1- /] [l,2,4]triazin-4-yl)piperazin- l-yl)pyrimidin-5-yl)ethyl)-2-methylpropane-2-sulfinamide (120 mg, 0.199 mmol) was stirred in 4 M HCl in 1,4-dioxane (1.5 mL)/MeOH (1.5 mL) at room temperature for 1 hour. The solvent was removed in vacuo and the residue triturated in EtOAc to give l-(4-fluorophenyl)- l-(2-(4-(6-(l -methyl- lH-pyrazol-4-yl)pyrrolo[2, l-/][l,2,4]triazin-4- yl)piperazin- l-yl)pyrimidin-5-yl)ethanamine, HCl (110 mg, 0.206 mmol, 103 % yield) as a pale yellow solid. MS (ES+) C26H27FN10requires: 498, found: 482 [M- 17 + H]+, 499 [M + H]+.
Step 5: Chiral separation of (R)-l-(4-fluorophenyl)- l-(2-(4-(6-(l-methyl- lH-pyrazol-4- yl)pyrrolo[2, l-/] [l,2,4]triazin-4-yl)piperazin-l-yl)pyrimidin-5-yl)ethanamine and (5)-1-(4- fluorophenyl)- l-(2-(4-(6-(l-methyl-lH-pyrazol-4-yl)pyrrolo[2, l-/] [l,2,4]triazin-4-yl)piperazin- 1 -yl)pyrimidin- -yl)ethanamine:
The enantiomers of racemic l-(4-fluorophenyl)- l-(2-(4-(6-(l-methyl- lH-pyrazol-4- yl)pyrrolo[2, l-/] [l,2,4]triazin-4-yl)piperazin-l-yl)pyrimidin-5-yl)ethanamine (94 mg, 0.189 mmol) were separated by chiral SFC to give (R)-l-(4-fluorophenyl)- l-(2-(4-(6-(l-methyl-lH- pyrazol-4-yl)pyrrolo[2, l-/][l,2,4]triazin-4-yl)piperazin- l-yl)pyrimidin-5-yl)ethanamine (34.4 mg, 0.069 mmol, 73.2 % yield) and (lS,)-l-(4-fluorophenyl)- l-(2-(4-(6-(l-methyl-lH-pyrazol-4- yl)pyrrolo[2, l-/] [l,2,4]triazin-4-yl)piperazin-l-yl)pyrimidin-5-yl)ethanamine (32.1 mg, 0.064 mmol, 68.3 % yield). The absolute stereochemistry was assigned randomly. MS (ES+)
C26H27FN10 requires: 498, found: 499 [M + H]+.
References
- ^ Jump up to:a b c d e f g h i j k l m “FDA approves the first targeted therapy to treat a rare mutation in patients with gastrointestinal stromal tumors”. U.S. Food and Drug Administration (FDA) (Press release). 9 January 2020. Archived from the original on 11 January 2020. Retrieved 9 January 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ “Blueprint Medicines Announces FDA Approval of AYVAKIT (avapritinib) for the Treatment of Adults with Unresectable or Metastatic PDGFRA Exon 18 Mutant Gastrointestinal Stromal Tumor”. Blueprint Medicines Corporation (Press release). 9 January 2020. Archived from the original on 11 January 2020. Retrieved 9 January 2020.
- ^ “Blueprint Medicines Announces Updated NAVIGATOR Trial Results in Patients with Advanced Gastrointestinal Stromal Tumors Supporting Development of Avapritinib Across All Lines of Therapy”. Blueprint Medicines Corporation (Press release). 15 November 2018. Archived from the original on 10 January 2020. Retrieved 9 January 2020.
Further reading
- Wu CP, Lusvarghi S, Wang JC, et al. (July 2019). “Avapritinib: A Selective Inhibitor of KIT and PDGFRα that Reverses ABCB1 and ABCG2-Mediated Multidrug Resistance in Cancer Cell Lines”. Mol. Pharm. 16 (7): 3040–3052. doi:10.1021/acs.molpharmaceut.9b00274. PMID 31117741.
- Gebreyohannes YK, Wozniak A, Zhai ME, et al. (January 2019). “Robust Activity of Avapritinib, Potent and Highly Selective Inhibitor of Mutated KIT, in Patient-derived Xenograft Models of Gastrointestinal Stromal Tumors”. Clin. Cancer Res. 25 (2): 609–618. doi:10.1158/1078-0432.CCR-18-1858. PMID 30274985.
External links
- “Avapritinib”. Drug Information Portal. U.S. National Library of Medicine (NLM).
| Clinical data | |
|---|---|
| Trade names | Ayvakit |
| Other names | BLU-285, BLU285 |
| License data | |
| Routes of administration |
By mouth |
| Drug class | Antineoplastic agents |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
| Formula | C26H27FN10 |
| Molar mass | 498.570 g·mol−1 |
| 3D model (JSmol) | |
///////Avapritinib, 2020 APPROVALS, PRIORITY, Orphan, BLU-285, BLU285, FDA 2020, Ayvakit, アバプリチニブ , авапритиниб , أفابريتينيب ,
Nitisinone, ニチシノン
Nitisinone
ニチシノン
Orfadin
Launched – 2002, NTBC
SC-0735
SYN-118
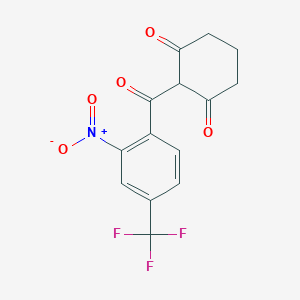
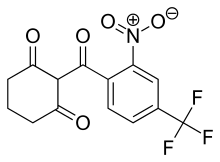
2-(alpha,alpha,alpha-Trifluoro-2-nitro-p-tuluoyl)-1,3-cyclohexanedione
2-(2-Nitro-4-trifluoromethylbenzoyl)cyclohexane-1,3-dione
Priority, Orphan
| Formula |
C14H10F3NO5
|
|---|---|
| CAS |
104206-65-7
|
| Mol weight |
329.2281
|
Nitisinone is a synthetic reversible inhibitor of 4-hydroxyphenylpyruvate dioxygenase. It is used in the treatment of hereditary tyrosinemia type 1. It is sold under the brand name Orfadin.
Nitisinone was first approved by the U.S. Food and Drug Administration (FDA) on January 18, 2002, then approved by the European Medicines Agency (EMA) on February 21, 2005. It was developed and marketed as Orfadin® by Swedish Orphan Biovitrum AB (SOBI) in the US .
The mechanism of action of nitisinone involves reversibile inhibition of 4-Hydroxyphenylpyruvate dioxygenase(HPPD). It is indicated for use as an adjunct to dietary restriction of tyrosine and phenylalanine in the treatment of hereditary tyrosinemia type 1 (HT-1).
Orfadin® is available as capsule for oral use, containing 2, 5 or 10 mg of free Nitisinone. The recommended initial dose is 1 mg/kg/day divided into two daily doses. Maximum dose is 2 mg/kg/day.
Nitisinone was launched in 2002 by Swedish Orphan (now Swedish Orphan Biovitrum) in a capsule formulation as an adjunct to dietary restriction of tyrosine and phenylalanine in the treatment of hereditary tyrosinemia type I. In 2015, this product was launched in Japan for the same indication. The same year, an oral suspension formulation for pediatric patients was registered in the E.U., and launch took place in the United Kingdom shortly after. This formulation was approved in 2016 in the U.S. for the same indication. In 2016, nitisinone tablet formulation developed by Cycle Pharmaceuticals was approved in Canada (this formulation is also available also in the U.S.).
Indication
Used as an adjunct to dietary restriction of tyrosine and phenylalanine in the treatment of hereditary tyrosinemia type 1.
Associated Conditions
EU

Nitisinone MendeliKABS
22 June 2017 EMA/CHMP/502860/2017
Product name, strength, pharmaceutical form: Orfadin • Marketing authorisation holder: Swedish Orphan Biovitrum International AB • Date of authorisation: 21/02/2005
Procedure No. EMEA/H/C/004281/0000
During the meeting on 22 June 2017, the CHMP, in the light of the overall data submitted and the scientific discussion within the Committee, issued a positive opinion for granting a Marketing authorisation to Nitisinone MendeliKABS.
The chemical name of nitisinone is 2-[2-Nitro-4-(trifluoromethyl)benzoyl]-1,3-cyclohexanedione corresponding to the molecular formula C14H10F3NO5. It has a relative molecular mass of 329.23 g/mol and the following structure: Figure 1. Structure of nitisinone.
Nitisinone appears as off-white to yellowish non-hygroscopic fine crystalline powder. It is practically insoluble in unbuffered water. It is freely soluble in dichloromethane, sparingly soluble in ethyl alcohol, slightly soluble in isopropyl alcohol and 70% aqueous isopropyl alcohol and in pH 6.8 phosphate buffer, very slightly soluble in pH 4.5 acetate buffer and practically insoluble at pH 1.1. Solubility in acidified aqueous media depends on the acid counter ion. Solubility increases with increasing pH. Its pKa was found to be around 10. Nitisinone is achiral and does not show polymorphism.
ALSO
2005
Nitisinone is a white to yellowish-white crystalline powder poorly soluble in water. The active substance is a weak acid and it is highly soluble in the pH range 4.5-7.2 in phosphate buffer solutions. Nitisinone has the chemical name 2-(2-nitro-4-trifluoromethylbenzoyl)-cyclohexane-1,3-dione. It does not show polymorphism.
US FDA
https://www.accessdata.fda.gov/drugsatfda_docs/nda/2016/206356Orig1s000ChemR.pdf
Company: Swedish Orphan Biovitrum AB
Application No.: 206356Orig1
Approval Date: April 22, 2016
- Approval Letter(s) (PDF)
- Printed Labeling (PDF)
- Summary Review (PDF)
- Officer/Employee List (PDF)
- Cross Discipline Team Leader Review (PDF)
- Chemistry Review(s) (PDF)
- Pharmacology Review(s) (PDF)
- Clinical Pharmacology Biopharmaceutics Review(s) (PDF)
- Proprietary Name Review(s) (PDF)
- Other Review(s) (PDF)
- Administrative Document(s) & Correspondence (PDF)
Nitisinone (INN), also known as NTBC (an abbreviation of its full chemical name) is a medication used to slow the effects of hereditary tyrosinemia type 1. Since its first use for this indication in 1991, it has replaced liver transplantation as the first-line treatment for this rare condition. It is also being studied in the related condition alkaptonuria. It is marketed under the brand name Orfadin by the company Swedish Orphan Biovitrum (Sobi); it was first brought to market by Swedish Orphan International. It was originally developed as a candidate herbicide.
Uses
Nitisinone is used to treat hereditary tyrosinemia type 1, in combination with restriction of tyrosine in the diet.[1][2][3]
Since its first use for this indication in 1991, it has replaced liver transplantation as the first-line treatment for this rare condition.[4] I It is marketed under the brand name Orfadin.
It has been demonstrated that treatment with nitisinone can reduce urinary levels of homogentisic acid in alkaptonuria patients by 95%.[5] A series of clinical trials run by DevelopAKUre to determine whether nitisinone is effective at treating the ochronosis suffered by patients with alkaptonuria are ongoing.[6] If the trials are successful, DevelopAKUre will try to get nitisinone licensed for use by alkaptonuria patients.[7]
Mechanism of action
The mechanism of action of nitisinone involves reversibile inhibition of 4-Hydroxyphenylpyruvate dioxygenase (HPPD).[8][9] This is a treatment for patients with Tyrosinemia type 1 as it prevents the formation of maleylacetoacetic acid and fumarylacetoacetic acid, which have the potential to be converted to succinyl acetone, a toxin that damages the liver and kidneys.[4] This causes the symptoms of Tyrosinemia type 1 experienced by untreated patients.[10]
Alkaptonuria is caused when an enzyme called homogentisic dioxygenase (HGD) is faulty, leading to a buildup of homogenisate.[11]Alkaptonuria patients treated with nitisinone produce far less HGA than those not treated (95% less in the urine),[5] because nitisinone inhibits HPPD, resulting in less homogenisate accumulation. Clinical trials are ongoing to test whether nitisinone can prevent ochronosisexperienced by older alkaptonuria patients.[6]
Adverse effects
Nitisinone has several negative side effects; these include but are not limited to: bloated abdomen, dark urine, abdominal pain, feeling of tiredness or weakness, headache, light-colored stools, loss of appetite, weight loss, vomiting, and yellow-colored eyes or skin.[12]
Research
Nitisinone is being studied as a treatment for alkaptonuria.[13]
Research at the National Institutes of Health (NIH) has demonstrated that nitisinone can reduce urinary levels of HGA by up to 95% in patients with alkaptonuria. The primary parameter of the NIH trial was range of hip motion, for which the results were inconclusive.[citation needed]
Research done using alkaptonuric mice has shown that mice treated with nitisinone experience no ochronosis in knee joint cartilage. In contrast, all of the mice in the untreated control group developed ochronotic knee joints.[14]
The efficacy of Nitisinone is now being studied in a series international clinical trials called DevelopAKUre.[15] The studies will recruit alkaptonuria patients in Europe.[16] A larger number of patients will be recruited in these trials than in the previous NIH trial.[17] The trials are funded by the European Commission.[18]
Nitisinone has been shown to increase skin and eye pigmentation in mice, and has been suggested as a possible treatment for oculocutaneous albinism.[19][20]
History
Nitisinone was discovered as part of a program to develop a class of herbicides called HPPD inhibitors. It is a member of the benzoylcyclohexane-1,3-dione family of herbicides, which are chemically derived from a natural phytotoxin, leptospermone, obtained from the Australian bottlebrush plant (Callistemon citrinus).[21] HPPD is essential in plants and animals for catabolism, or breaking apart, of tyrosine.[22] In plants, preventing this process leads to destruction of chlorophyll and the death of the plant.[22] In toxicology studies of the herbicide, it was discovered that it had activity against HPPD in rats[23] and humans.[24]
In Type I tyrosinemia, a different enzyme involved in the breakdown of tyrosine, fumarylacetoacetate hydrolase is mutated and doesn’t work, leading to very harmful products building up in the body.[1] Fumarylacetoacetate hydrolase acts on tyrosine after HPPD does, so scientists working on making herbicides in the class of HPPD inhibitors hypothesized that inhibiting HPPD and controlling tyrosine in the diet could treat this disease. A series of small clinical trials attempted with one of their compounds, nitisinone, were conducted and were successful, leading to nitisinone being brought to market as an orphan drug Swedish Orphan International,[8] which was later acquired by Swedish Orphan Biovitrum (Sobi).
Sobi is now a part of the DevelopAKUre consortium. They are responsible for drug supply and regulatory support in the ongoing clinical trials that will test the efficiacy of nitisinone as a treatment for alkaptonuria.[25] It is hoped that if the trials are successful, nitisinone could also be licensed for treatment of alkaptonuria.[7]
Generic versions
There is no generic version of Orfadin in G7 countries. Prior to the market authorization of MDK-Nitisinone in Canada, the only Nitisinone product available globally was Orfadin.[26]Until recently, Nitisinone was not approved in Canada where it was distributed for over 20 years via a Health Canada Special Access Program. In September 2016, MendeliKABS was granted approval of a Priority New Drug Submission (PNDS) by Health Canada for a bioequivalent generic version of Orfadin capsules (MDK-Nitisinone). In November 2016 Cycle Pharma was also granted approval of a PNDS by Health Canada for Nitisinone tablets that are bioequivalent to Orfadin capsules.[27] SOBI was granted approval of a PNDS in December 2016.[28]
PAPER
1H NMR, 13C NMR, and Computational DFT Studies of the Structure of 2-Acylcyclohexane-1,3-diones and Their Alkali Metal Salts in Solution
2-(2-Nitro-4-trifluoromethylbenzoyl)cyclohexane-1,3-dione (NTBC; 3). The compound was prepared in the same manner as 2. The synthesis of an appropriate benzoic acid derivative was started from the transformation of commercially available 2-nitro-4-trifluoromethylaniline into benzonitrile by the classical Sandmeyer method. Then the nitrile was hydrolyzed in 65% sulfuric acid to give 2-nitro-4-trifluoromethylbenzoic acid.13 The obtained triketone 3 had a mp of 140−142 °C (lit.14 141−143 °C). For NMR data, see Supporting Information….. https://pubs.acs.org/doi/suppl/10.1021/jo060583g/suppl_file/jo060583gsi20060420_080852.pdf
NMR data for 2-(2-nitro-4-trifluoromethylbenzoyl)cyclohexane-1,3-dione, 3, in CDCl3

1 H NMR: 16.25 (s, 1H, OH), 8.47 (ddq, 1H, H10, J10,12=1.7 Hz, J10,13=0.4 Hz, J10,F=0.7 Hz), 7.94 (ddq, 1H, H12, J12,13=8.0 Hz, J12,F=0.7 Hz), 7.39 (ddq, 1H, H13, J13,F=0.8 Hz), 2.81 (t-like m, 2H, H4, H4’, JH4,H4’= -18.8 Hz, JH4,H5=5.4 Hz, JH4,H5’=7.3 Hz, JH4,H6=0.7 Hz, JH4,H6’= -0.8 Hz), 2.37 (tlike m, 2H, H6, H6’, JH6,H6’= -16.5 Hz, JH6,H5=4.6 Hz, JH6,H5’=8. 5 Hz), 2.04 (pentet-like m, 2H, H5, H5’, JH5,H5’= -13.6 Hz.

13C NMR: 196.3 (s, C(O)Ph), 195.8 (s, C3), 194.1 (s, C1), 145.5 (s, C9), 139.7 (s, C8), 132.0 (q, C11, J11,F=34.3 Hz), 130.8 (q, C12 J12,F=3.5 Hz), 127.7 (s, C13), 122.6 (q, CF3, JC,F=272.9 Hz), 121.1 (q, C10, J10,F=3.9 Hz), 112.7 (s, C2), 37.3 (s, C6) 31.6 (s, C4), 19.1 (s, C5).
PATENT
EP 186118
US 4780127



The condensation of cyclohexane-1,3-dione (I) with 2-nitro-4-(trifluoromethyl)benzoyl chloride by means of TEA in dichloromethane gives the target Nitisinone.EP 0186118
JP 1986152642, US 4774360, US 4780127
Nitisinone
-
- Synonyms:NTBC, SC 0735
- ATC:A16AX04
- Use:treatment of inherited tyrosinemia type I
- Chemical name:2-[2-nitro-4-(trifluoromethyl)benzoyl]-1,3-cyclohexanedione
- Formula:C14H10F3NO5
- MW:329.23 g/mol
- CAS-RN:104206-65-7
Substance Classes
Synthesis Path
Substances Referenced in Synthesis Path
| CAS-RN | Formula | Chemical Name | CAS Index Name |
|---|---|---|---|
| 504-02-9 | C6H8O2 | cyclohexane-1,3-dione | 1,3-Cyclohexanedione |
| 81108-81-8 | C8H3ClF3NO3 | 2-nitro-4-trifluoromethylbenzoyl chloride |
Trade Names
| Country | Trade Name | Vendor | Annotation |
|---|---|---|---|
| D | Orfadin | Orphan Europe | |
| USA | Orfadin | Swedish Orphan ,2002 |
Formulations
- cps. 2 mg
References
-
- WO 9 300 080 (ICI; 7.1.1993; appl. 18.6.1992; GB-prior. 24.6.1991).
- US 4 774 360 (Stauffer Chemical; 27.9.1988; appl. 29.6.1987).
-
synergistic herbicidal combination:
- WO 9 105 469 (Hoechst AG; 2.5.1991; appl. 12.10.1990; D-prior. 18.10.1989).
-
preparation of benzoylcyclohexanedione herbicides:
- US 4 780 127 (Stauffer Chemical; 25.10.1988; appl. 30.6.1986; USA-prior. 25.3.1982).
-
certain 2-(2-nitrobenzoyl)-1,3-cyclohexanediones:
- EP 186 118 (Stauffer Chemical; 2.7.1986; appl. 18.12.1985; USA-prior. 20.12.1984).
-
stable herbicidal compositions:
- WO 9 727 748 (Zeneca; 7.8.1997; appl. 3.2.1997; USA-prior. 2.2.1996).
PATENT
https://patents.google.com/patent/US9783485B1/en
NTBC is a drug marketed by Swedish Orphan Biovitrum International AB under the brand name Orfadin® and it is used to slow the effects of hereditary tyrosinemia type 1 (HT-1) in adult and pediatric patients. It has been approved by FDA and EMA in January 2002 and February 2005 respectively.
HT-1 disease is due to a deficiency of the final enzyme of the tyrosine catabolic pathway fumarylacetoacetate hydrolase. NTBC is a competitive inhibitor of 4-hydroxyphenylpyruvate dioxygenase (HPPD), an enzyme which precedes fumarylacetoacetate hydrolase. By inhibiting the normal catabolism of tyrosine in patients with HT-1, NTBC prevents the accumulation of the toxic intermediates maleylacetoacetate and fumarylacetoacetate, that in patients with HT-1 are converted to the toxic metabolites succinylacetone and succinylacetoacetate, the former inhibiting the porphyrin synthesis pathway leading to the accumulation of 5-aminolevulinate.
Usefulness of NTBC in the treatment of further diseases has also been documented. A non-comprehensive list is reported hereinafter.
Effectiveness of Orfadin® in the treatment of diseases where the products of the action of HPPD are involved (e.g., HT-1) has been described notably in EP0591275B1 corresponding to U.S. Pat. No. 5,550,165B1. Synthesis of NTBC is also described in this patent.
WO2011106655 reports a method for increasing tyrosine plasma concentrations in a subject suffering from oculocutaneous/ocular albinism, the method comprising administering to the subject a pharmaceutically acceptable composition comprising NTBC in the range of between about 0.1 mg/kg/day to about 10 mg/kg/day.
U.S. Pat. No. 8,354,451B2 reports new methods of combating microbial infections due to fungi or bacteria by means of administration to a subject of a therapeutically active amount of NTBC.
WO2010054273 discloses NTBC-containing compositions and methods for the treatment and/or prevention of restless leg syndrome (RLS).
EP1853241B1 claims the use of NTBC in the treatment of a neurodegenerative disease, notably Parkinson disease.
Introne W. J., et al., disclosed usefulness of nitisinone in the treatment of alkaptonuria (Introne W. J., et al., Molec. Genet. Metab., 2011, 103, 4, 307). The key step of the synthesis reported in EP0591275B1 (now propriety of Swedish Orphan Biovitrum International AB, SE), involves the reaction of 2-nitro-4-trifluromethylbenzoyl chloride and cyclohexane-1,3-dione in the presence of triethylamine and then use of acetone cyanohydrin in order to promote the rearrangement of the key intermediate enol ester. After washing and extraction from CH2Cl2, the crude product is recrystallized from ethyl acetate to get the desired 2-(2-nitro-4-trifluoromethylbenzoyl)-1,3-cyclohexanedione as a solid having a melting point of 88-94° C.
Another patent (U.S. Pat. No. 4,695,673) filed in name of Stauffer Chemical Company disclosed a process of synthesis of acylated 1,3-dicarbonyl compounds in which the intermediate enol ester is isolated prior to its rearrangement into the final product, said rearrangement making use of a cyanohydrin compound derived from alkali metal, methyl alkyl ketone, benzaldehyde, cyclohexanone, C2-C5aliphatic aldehyde, lower alkyl silyl or directly by using hydrogen cyanide.
Yet another patent (U.S. Pat. No. 5,006,158) filed in name of ICI Americas Inc. disclosed a process similar to the one disclosed in U.S. Pat. No. 4,695,673 wherein the intermediate enol ester was isolated prior to its rearrangement into the final product by use of potassium cyanide. Said reaction can optionally be done by concomitant use of a phase transfer catalyst such as Crown ethers. The preferred solvent for conducting such a reaction is 1,2-dichloroethane.
Still a further patent (EP0805791) filed in name of Zeneca Ltd disclosed an alternative synthesis of nitisinone involving the reaction of 1,3-cyclohexanedione and variously substituted benzoyl chloride in the presence of sodium or potassium carbonate in CH3CN or DMF. Best yields were obtained using CH3CN as solvent and sodium carbonate as the base. Reaction was performed at 55-57° C. in 17 hours.
It is well known that one of the problems of the actual drug formulation (i.e., Orfadin® capsules) is its chemical instability. Indeed, even if Orfadin® has to be stored in a refrigerator at a temperature ranging from 2° C. to 8° C., its shelf life is of only 18 months. After first opening, the in-use stability is a single period of 2 months at a temperature not above 25° C., after which it must be discarded. It will be evident that such storage conditions have an impact in the distribution chain of the medicine, in terms of costs and also in terms of logistics for the patient. Therefore, there is an urgent need of more stable formulations, both from a logistic supply chain point of view, and from the patient compliance point of view. Since the formulation of Orfadin® contains only the active ingredient and starch as excipient, relative instability may be attributed to the active pharmaceutical ingredient itself; in other words it can derive from the way it is synthesized and/or the way it is extracted from the reaction mixture, and/or the way it is finally crystallized. Furthermore, some impurities may contribute to render the final product less stable overtime. Consequently, it is of major importance to identify a process of synthesis and/or a crystallization method that enable the reliable production of a highly pure and stable product.
Impurities as herein-above mentioned can derive either from the final product itself (through chemical degradation) or directly from the starting materials/solvents used in the process of synthesis. Regarding the latter option, it is therefore primordial to ascertain that at each step, impurities are completely removed in order not to get them at the final stage, also considering that some of them could potentially be cyto/genotoxic.
The impurities correlated to nitisinone can be either derived from the starting materials themselves (i.e., impurities 1 and 2) or obtained as side products during the process of synthesis and/or under storage conditions (i.e., impurities 3 to 5) and are the following:
-
- 2-nitro-4-(trifluoromethyl) benzoic acid (Impurity no 1),
- 1,3-cyclohexanedione (CHD) (Impurity no 2),
- 4-(trifluoromethyl)salicylic acid (Impurity no 3),
- 2-[3-nitro-4-(trifluoromethyl)benzoyl]-1,3-cyclohexanedione (Impurity no 4), and
- 6-trifluoromethyl-3,4-dihydro-2H-xanthene-1,9-dione (Impurity no 5).
Impurity-2, impurity-3, and impurity-5 have been previously reported in WO2015101794. Strangely, impurity-4 has never been reported, even if it is an obvious side-product which can easily be formed during the coupling reaction between 1,3-cyclohexanedione and 2-nitro-4-(trifluoromethyl) benzoic acid, the latter being not 100% pure but containing some amount of regioisomer 3-nitro-4-(trifluoromethyl) benzoic acid.
Potential genotoxicity of impurity no 4 which possesses an aromatic nitro moiety was assessed using in-silico techniques and resulted to be a potential genotoxic impurity. According to the FDA ICH M7 guidelines, daily intake of a mutagenic impurity (Threshold of Toxicological Concern, TTC) in an amount not greater than 1.5 μg per person is considered to be associated with a negligible risk to develop cancer over a lifetime of exposure. Consequently, assuming a daily dose of 2 mg/kg, for a person weighing 70 kg, the maximum tolerated impurity content of such a compound would be of about 11 ppm, as calculated according to the equation underneath.
concentration limit ( ppm ) = T T C ( µg / day ) Dose ( g / day )
It is therefore of paramount importance to ensure that the process of synthesis of nitisinone and the purification steps of the same give rise to an API devoid of such impurity no 4, or at least far below the threshold of 11 ppm as indicated above. The skilled person will understand that total absence of said impurity is highly desirable.
It is well known in the pharmaceutical field that investigation of potential polymorphism of a solid API is of crucial importance and is also recommended by major regulatory authorities such as FDA.
Notwithstanding the fact that nitisinone has been used for years to treat HT-1 patients, it appears that no NTBC formulation fully satisfies the requisites of stability and/or compliance standard for the patients. Therefore, there is an unmet medical need of long-term pure and stable formulations.
Example 1
Thionyl chloride (162 g, 1.36 mol) was added dropwise into a suspension of 2-nitro-4-trifluoromethylbenzoic acid (228 g, 0.97 mol) in toluene (630 g) at 80° C. The thus obtained solution was kept under stirring at 80° C. for 20 hours, and then cooled to 50° C. The volatiles were removed under reduced pressure in order to get the expected 2-nitro-4-trifluoromethylbenzoyl chloride as an oil. The latter, cooled to 25° C. was added dropwise to a suspension of 1,3-cyclohexanedione (109 g, 0.97 mol) and potassium carbonate (323 g, 2.33 mol) in CH3CN (607 g). After 18 h the mixture was diluted with water (500 ml) and slowly acidified to about pH=1 with HCl 37%. The mixture was then warmed to about 55° C. and the phases were separated. The organic layer was washed with a 10% aqueous solution of sodium chloride and then, concentrated under reduced pressure at a temperature below 55° C. to reach a volume of 380 ml. The thus obtained mixture was stirred at 55° C. for 1 h and then cooled to 0° C. in 16 to 18 h. The resulting solid was filtered and rinsed several times with pre-cooled (0° C.) toluene. The wet solid was dried at 60° C. under vacuum for 6 h to provide nitisinone (164 g) as a white to yellowish solid with a purity of 98.4% as measured by HPLC and a content of potentially genotoxic impurity no 4 of 6.1 ppm measured by HPLC/MS.
Example 2
Nitisinone as obtained from example 1 (164 g) was added to a 3/1 (w/w) mixture of CH3CN/toluene (volume of solvent: 638 ml). The mixture was warmed gently to about 55° C. under stirring until solids were completely dissolved. The solution was then concentrated under reduced pressure maintaining the internal temperature below 50° C. to reach a volume of 290 ml. Then, more toluene (255 g) was added and the solution was concentrated again under reduced pressure until the residual volume reached 290 ml. The solution was heated to about 55° C. for 1 h and successively cooled slowly in 10 to 12 h to 10° C. The resulting solid was filtered and rinsed several times with pre-cooled (0° C.) toluene. The wet solid was dried at about 60° C. under vacuum for 4 h to provide nitisinone (136 g) as a white to yellowish solid, with a purity of 99.94% and a 99.8% assay measured by HPLC and a d(90) particle size between 310 and 350 μm. The content of potential genotoxic impurity no 4 resulted below 1 ppm.
CLIP

References
- ^ Jump up to:a b National Organization for Rare Disorders. Physician’s Guide to Tyrosinemia Type 1Archived 2014-02-11 at the Wayback Machine.
- Jump up^ “Nitisinone (Oral Route) Description and Brand Names”. Mayoclinic.com. 2015-04-01. Retrieved 2015-06-04.
- Jump up^ Sobi Orfadin® (nitisinone)
- ^ Jump up to:a b McKiernan, Patrick J (2006). “Nitisinone in the Treatment of Hereditary Tyrosinaemia Type 1”. Drugs. 66 (6): 743–50. doi:10.2165/00003495-200666060-00002. PMID 16706549.
- ^ Jump up to:a b Introne, Wendy J.; Perry, Monique B.; Troendle, James; Tsilou, Ekaterini; Kayser, Michael A.; Suwannarat, Pim; O’Brien, Kevin E.; Bryant, Joy; Sachdev, Vandana; Reynolds, James C.; Moylan, Elizabeth; Bernardini, Isa; Gahl, William A. (2011). “A 3-year randomized therapeutic trial of nitisinone in alkaptonuria”. Molecular Genetics and Metabolism. 103(4): 307–14. doi:10.1016/j.ymgme.2011.04.016. PMC 3148330
 . PMID 21620748.
. PMID 21620748. - ^ Jump up to:a b “About DevelopAKUre | DevelopAKUre”. Developakure.eu. 2014-06-20. Archived from the original on 2015-05-12. Retrieved 2015-06-04.
- ^ Jump up to:a b “A Potential Drug – Nitisinone”. Akusociety.org. Archived from the original on 2015-05-05. Retrieved 2015-06-04.
- ^ Jump up to:a b Lock, E. A.; Ellis, M. K.; Gaskin, P.; Robinson, M.; Auton, T. R.; Provan, W. M.; Smith, L. L.; Prisbylla, M. P.; Mutter, L. C.; Lee, D. L. (1998). “From toxicological problem to therapeutic use: The discovery of the mode of action of 2-(2-nitro-4-trifluoromethylbenzoyl)-1,3-cyclohexanedione (NTBC), its toxicology and development as a drug”. Journal of Inherited Metabolic Disease. 21 (5): 498–506. doi:10.1023/A:1005458703363. PMID 9728330.
- Jump up^ Kavana, Michael; Moran, Graham R. (2003). “Interaction of (4-Hydroxyphenyl)pyruvate Dioxygenase with the Specific Inhibitor 2-[2-Nitro-4-(trifluoromethyl)benzoyl]-1,3-cyclohexanedione†”. Biochemistry. 42 (34): 10238–45. doi:10.1021/bi034658b. PMID 12939152.
- Jump up^ “Newborn Screening”. Newbornscreening.info. 2013-05-14. Retrieved 2015-06-04.
- Jump up^ “What is Alkaptonuria?”. Akusociety.org. Archived from the original on 2015-04-05. Retrieved 2015-06-04.
- Jump up^ “Nitisinone (Oral Route) Side Effects”. Mayoclinic.com. 2015-04-01. Retrieved 2015-06-04.
- Jump up^ Phornphutkul, Chanika; Introne, Wendy J.; Perry, Monique B.; Bernardini, Isa; Murphey, Mark D.; Fitzpatrick, Diana L.; Anderson, Paul D.; Huizing, Marjan; Anikster, Yair; Gerber, Lynn H.; Gahl, William A. (2002). “Natural History of Alkaptonuria”. New England Journal of Medicine. 347 (26): 2111–21. doi:10.1056/NEJMoa021736. PMID 12501223.
- Jump up^ Preston, A. J.; Keenan, C. M.; Sutherland, H.; Wilson, P. J.; Wlodarski, B.; Taylor, A. M.; Williams, D. P.; Ranganath, L. R.; Gallagher, J. A.; Jarvis, J. C. (2013). “Ochronotic osteoarthropathy in a mouse model of alkaptonuria, and its inhibition by nitisinone”. Annals of the Rheumatic Diseases. 73 (1): 284–9. doi:10.1136/annrheumdis-2012-202878. PMID 23511227.
- Jump up^ “DevelopAKUre”. Developakure.eu. 2014-06-20. Retrieved 2015-06-04.
- Jump up^ “2012-005340-24”. Clinicaltrialsregister.eu. Retrieved 2015-06-04.
- Jump up^ “The Programme | DevelopAKUre”. Developakure.eu. 2014-06-20. Archived from the original on 2015-05-12. Retrieved 2015-06-04.
- Jump up^ “European Commission : CORDIS : Search : Simple”. Cordis.europa.eu. 2012-05-30. Retrieved 2015-06-04.
- Jump up^ Onojafe, Ighovie F.; Adams, David R.; Simeonov, Dimitre R.; Zhang, Jun; Chan, Chi-Chao; Bernardini, Isa M.; Sergeev, Yuri V.; Dolinska, Monika B.; Alur, Ramakrishna P.; Brilliant, Murray H.; Gahl, William A.; Brooks, Brian P. (2011). “Nitisinone improves eye and skin pigmentation defects in a mouse model of oculocutaneous albinism”. Journal of Clinical Investigation. 121 (10): 3914–23. doi:10.1172/JCI59372. PMC 3223618
 . PMID 21968110. Lay summary – ScienceDaily (September 26, 2011).
. PMID 21968110. Lay summary – ScienceDaily (September 26, 2011). - Jump up^ “Nitisinone for Type 1B Oculocutaneous Albinism – Full Text View”. ClinicalTrials.gov. Retrieved 2015-06-04.
- Jump up^ G. Mitchell, D.W. Bartlett, T.E. Fraser, T.R. Hawkes, D.C. Holt, J.K. Townson, R.A. Wichert Mesotrione: a new selective herbicide for use in maize Pest Management Science, 57 (2) (2001), pp. 120–128
- ^ Jump up to:a b Moran, Graham R. (2005). “4-Hydroxyphenylpyruvate dioxygenase”. Archives of Biochemistry and Biophysics. 433 (1): 117–28. doi:10.1016/j.abb.2004.08.015. PMID 15581571.
- Jump up^ Ellis, M.K.; Whitfield, A.C.; Gowans, L.A.; Auton, T.R.; Provan, W.M.; Lock, E.A.; Smith, L.L. (1995). “Inhibition of 4-Hydroxyphenylpyruvate Dioxygenase by 2-(2-Nitro-4-trifluoromethylbenzoyl)-cyclohexane-1,3-dione and 2-(2-Chloro-4-methanesulfonylbenzoyl)-cyclohexane-1,3-dione”. Toxicology and Applied Pharmacology. 133 (1): 12–9. doi:10.1006/taap.1995.1121. PMID 7597701.
- Jump up^ Lindstedt, Sven; Odelhög, Birgit (1987). “4-Hydroxyphenylpyruvate dioxygenase from human liver”. In Kaufman, Seymour. Metabolism of Aromatic Amino Acids and Amines. Methods in Enzymology. 142. pp. 139–42. doi:10.1016/S0076-6879(87)42021-1. ISBN 978-0-12-182042-8. PMID 3037254.
- Jump up^ “Others | DevelopAKUre”. Developakure.eu. 2014-06-20. Retrieved 2015-06-04.
- Jump up^ Pr MDK-Nitisinone Summary Basis of Decisions, Health Canada 2016. http://www.hc-sc.gc.ca/dhp-mps/prodpharma/sbd-smd/drug-med/sbd-smd-2016-mdk-nitisinone-190564-eng.php
- Jump up^ Pr Nitisinone Tablets Regulatory Decision Summary Health Canada, 2016. http://www.hc-sc.gc.ca/dhp-mps/prodpharma/rds-sdr/drug-med/rds-sdr-nitisinone-tab-193770-eng.php
- Jump up^ PrOrfadin Regulatory Decision Summary Health Canada, 2016. http://www.hc-sc.gc.ca/dhp-mps/prodpharma/rds-sdr/drug-med/rds-sdr-orfadin-193226-eng.php
External links
 |
|
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Consumer Drug Information |
| License data |
|
| Routes of administration |
Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | Approximately 54 h |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.218.521 |
| Chemical and physical data | |
| Formula | C14H10F3NO5 |
| Molar mass | 329.228 g/mol |
| 3D model (JSmol) | |
////////////////Nitisinone, ニチシノン , Orfadin, FDA 2002, NTBC , SC-0735 , SYN-118 , JAPAN 2015, JAP 2015, EU 2005, Priority, Orphan
[O-][N+](=O)C1=C(C=CC(=C1)C(F)(F)F)C(=O)C1C(=O)CCCC1=O
FDA Approves Ryanodex for the Treatment of Malignant Hyperthermia

Dantrolene sodium
1-[[[5-(4-nitrophenyl)-2-furanyl]methylene]amino]-2,4-imidazolidinedione
VIEW THIS POST AT BELOW LINK UNTIL FORMATTING IS FIXED
http://www.allfordrugs.com/2014/07/24/fda-approves-ryanodex-for
-the-treatment-of-malignant-hyperthermia/
FDA Approves Ryanodex for the Treatment of Malignant Hyperthermia
WOODCLIFF LAKE, N.J.(BUSINESS WIRE) July 23, 2014 —
Eagle Pharmaceuticals, Inc. (“Eagle” or “the Company”)
(Nasdaq:EGRX) today announced that the U. S. Food and Drug Administration (FDA)
has approved Ryanodex (dantrolene sodium) for injectable
suspension indicated for
the treatment of malignant hyperthermia (MH), along
with the appropriate supportive measures.
MH is an inherited and potentially fatal disorder triggered
by certain anesthesia agents
in genetically susceptible individuals. FDA had designated
Ryanodex as an Orphan Drug in
August 2013. Eagle has been informed by the FDA that it will learn over the next four to
six weeks if it has been granted the seven year Orphan Drug market exclusivity.
read at
http://www.drugs.com/newdrugs/fda-approves-ryanodex-malignant-
hyperthermia-4058.html?utm_source=ddc&utm_medium=email&utm_
news+summary+-+July+23%2C+2014
READ MORE AT
PATENTS, CAS NO ETC
http://www.allfordrugs.com/2014/07/24/fda-approves-ryanodex-

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....