Home » Posts tagged 'pixavir marboxil'
Tag Archives: pixavir marboxil
Pixavir marboxil
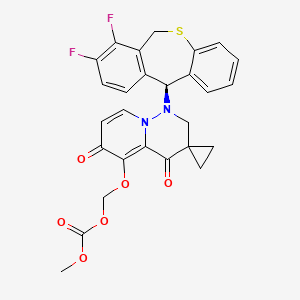
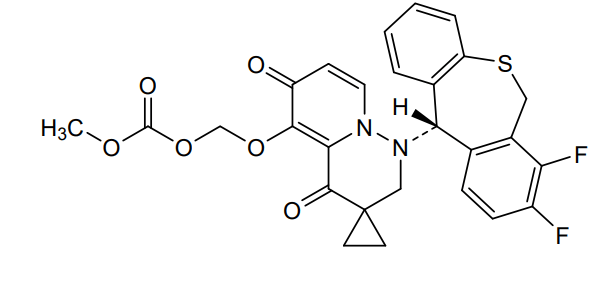
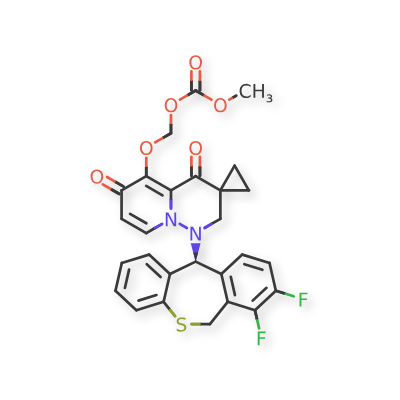
Pixavir marboxil
CAS 2365473-17-0
MF C27H22F2N2O6S MW540.535
(1-((11S)-7,8-DIFLUORO-6,11-DIHYDROBENZO(C)(1)BENZOTHIEPIN-11-YL)-4,6-DIOXOSPIRO(2H-PYRIDO(1,2-B)PYRIDAZINE-3,1′-CYCLOPROPANE)-5-YL)OXYMETHYL METHYL CARBONATE
({1′-[(11S)-7,8-difluoro-6,11-dihydrodibenzo[b,e]thiepin-11-yl]-4′,6′-dioxo1′,2′,4′,6′-tetrahydrospiro[cyclopropane-1,3′-pyrido[1,2-b]pyridazin]-5′-yl}oxy)methyl methyl carbonate
antiviral, TG 1000, Yi Li Kang, SV42843XSX, Cap-dependent endonuclease-IN-1, Influenza virus infections, TaiGen Biotechnology
- OriginatorTaiGen Biotechnology
- Class3-ring heterocyclic compounds; Antivirals; Benzene derivatives; Carbonates; Cyclopropanes; Dibenzothiepins; Esters; Ethers; Fluorobenzenes; Organic sulfur compounds; Pyridazines; Pyridones; Small molecules; Spiro compounds
- Mechanism of ActionEndonuclease inhibitors; Virus replication inhibitors
- MarketedInfluenza virus infections
- 27 Feb 2026Launched for Influenza virus infections (In adults, In adolescents) in China (PO), prior to February 2026 (TaiGen Biotechnology pipeline, February 2026)
- 26 Jan 2026Pixavir marboxil licensed to Boryung Biopharma for commercialization in South Korea
- 16 Dec 2025Chemical structure information added.
Pixavir marboxil (also known as TG-1000) is an investigational antiviral drug designed to treat and inhibit influenza virus infections. It belongs to a class of compounds known as cap-dependent endonuclease (CEN) inhibitors, which target a key viral enzyme necessary for influenza virus replication.
Mechanism of Action
- Blocks viral replication: Pixavir marboxil works by inhibiting the influenza virus’s cap-dependent endonuclease, a part of the viral RNA polymerase complex the virus needs to “snatch” capped RNA fragments from host cell mRNA. Without this process, the virus cannot efficiently produce its own viral proteins or replicate.
What Viruses It Targets
Pixavir marboxil has shown activity against:
- Influenza A viruses
- Influenza B viruses
- Certain drug-resistant influenza strains
This broad spectrum makes it useful for seasonal flu and potentially strains less responsive to older antiviral drugs
Clinical Development & Approval Status
Phase Trials & Results
- Completed Phase III: Clinical trials in adults and adolescents (age ≥12) showed that a single dose shortened time to symptom relief compared to placebo (e.g., median ~60.9 h vs ~87.9 h).
- Symptom relief benefits: The data indicated statistically significant improvement in flu symptoms and faster viral inactivation in treated patients versus placebo.
- Pediatric Formulation: China’s health authority approved pediatric Phase III studies for Pixavir (children <12), indicating further development for younger patients.
Regulatory Filings
- NDA (New Drug Application): Pixavir marboxil has been submitted for approval to the National Medical Products Administration (NMPA) in mainland China based on Phase III results.
- Generic Name Approved: The drug has been officially recognized with the generic name “Pixavir marboxil,” moving it closer to commercialization.
Pixavir marboxil is a small molecule drug. The usage of the INN stem ‘-xavir’ in the name indicates that Pixavir marboxil is a influenza CAP-dependent endonuclease inhibitor. Pixavir marboxil has a monoisotopic molecular weight of 540.12 Da.
SYN
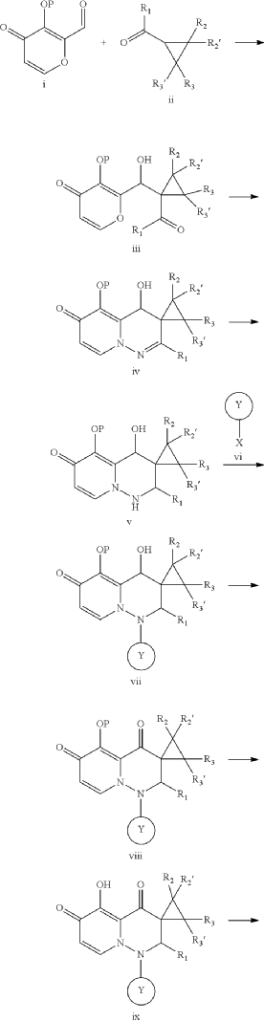
PAT
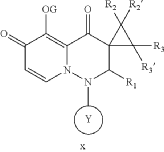
- Cap-dependent endonuclease inhibitorsPublication Number: US-10596171-B2Patent Family: AU-2019209426-A1; AU-2019209426-B2; BR-112020014810-A2; CA-3078391-A1; CA-3078391-C; CL-2020001919-A1; CN-110300753-A; CN-110300753-B; CO-2020006411-A2; EA-202090658-A1; EP-3743424-A1; EP-3743424-A4; IL-274199-A; IL-274199-B; JO-P20200159-A1; JP-2021512146-A; JP-6994121-B2; KR-102432975-B1; KR-20200086385-A; MX-2020007722-A; MY-197875-A; NZ-763248-A; PE-20211240-A1; PH-12020550921-A1; SG-11202003014V-A; TW-201938166-A; TW-I714951-B; US-10596171-B2; US-2019224198-A1; WO-2019144089-A1; ZA-202002037-BPriority Date: 2018-01-22Grant Date: 2020-03-24Inventor(s): LIN CHU-CHUNG; CHEN HUNG-CHUAN; CHIANG CHIAYN; YEN CHI-FENG; HSU MING-CHUAssignee(s): TAIGEN BIOTECHNOLOGY CO LTD; HSU MING CHUClassification: A61K31/5025; A61P31/16Abstract: Provided is a compound of Formula (I) below, or a pharmaceutically acceptable salt, metabolite, or prodrug thereof: n nwherein: A 1 is CR 4 or N; A 2 is CR 5 R 6 or NR 7 ; A 3 is CR 5 ′R 6 ′ or NR 7 ′; each of R 1 , R 2 , R 2 ′, R 3 , R 3 ′, R 4 , R 5 , R 5 ′, R 6 , R 6 ′, R 7 , and R 7 ′, independently, is hydrogen, deuterium, halogen, cyano, hydroxyl, carboxyl, amino, formyl, nitro, C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-6 alkoxy, C 2-6 alkenyloxy, C 1-6 alkylcarbonyl, C 1-6 alkyloxycarbonyl, C 1-6 alkylamine, C 3-20 carbocyclyl, or C 3-20 heterocyclyl; or R 5 and R 6 , R 5 ′ and R 6 ′, or R 5 and R 5 ′, together with the adjacent atom to which they are each attached, form C 3-10 carbocyclyl or C 3-10 heterocyclyl. Further provided are a method of using the above-described compound, or the pharmaceutically acceptable salt, metabolite, or prodrug thereof for treating influenza and a pharmaceutical composition containing same.Linked Compounds: 274Linked Substances: 411
- Cap-dependent endonuclease inhibitorsPublication Number: US-2019224198-A1Priority Date: 2018-01-22Linked Compounds: 275Linked Substances: 411



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

////////////pixavir marboxil, antiviral, TG 1000, Yi Li Kang, SV42843XSX, Cap-dependent endonuclease-IN-1, Influenza virus infections, TaiGen Biotechnology

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










