Home » Posts tagged 'NBA7J58PPP'
Tag Archives: NBA7J58PPP
Deulumateperone
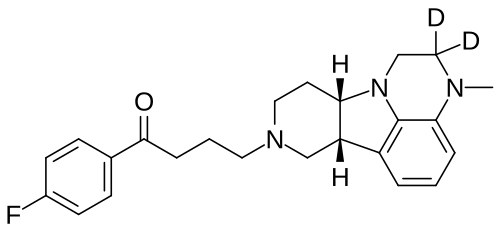
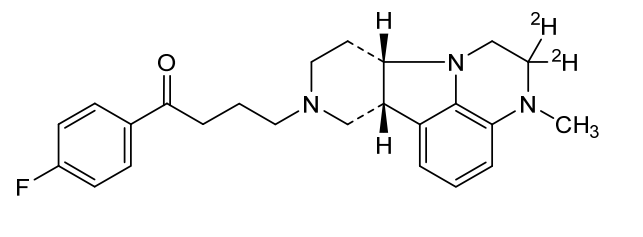
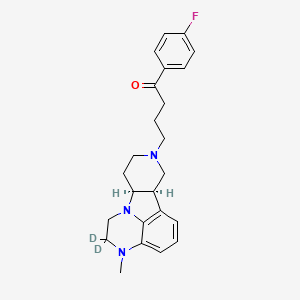
Deulumateperone
CAS 2102683-75-8
MF C24H262H2FN3O MW 395.5 g/mol
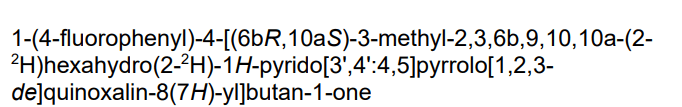
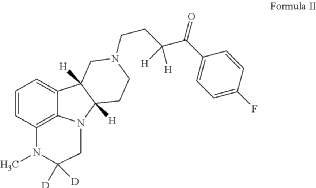
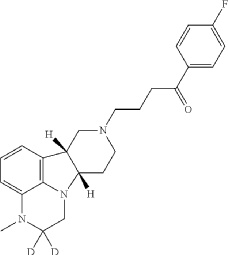
4-[(10R,15S)-3,3-dideuterio-4-methyl-1,4,12-triazatetracyclo[7.6.1.05,16.010,15]hexadeca-5,7,9(16)-trien-12-yl]-1-(4-fluorophenyl)butan-1-one
- 1-(4-Fluorophenyl)-4-[(6bR,10aS)-2,3,6b,9,10,10a-hexahydro-2-d-3-methyl-1H-pyrido[3′,4′:4,5]pyrrolo[1,2,3-de]quinoxalin-8(7H)-yl-2-d]-1-butanone
- 1-Butanone, 1-(4-fluorophenyl)-4-[(6bR,10aS)-2,3,6b,9,10,10a-hexahydro-2-d-3-methyl-1H-pyrido[3′,4′:4,5]pyrrolo[1,2,3-de]quinoxalin-8(7H)-yl-2-d]-
antipsychotic, ITI-1284, ITI 1284, NBA7J58PPP,
Deulumateperone (INNTooltip International Nonproprietary Name; developmental code name ITI-1284) is an experimental antipsychotic of the pyridopyrroloquinoxaline and butyrophenone families as well as a deuterated analogue of lumateperone which is under development as a sublingually administered orally disintegrating tablet (ODT) for the treatment of psychotic disorders, agitation, and generalized anxiety disorder.[2][3][4][1] No recent development has been reported for treatment of depressive disorders, behavioral disorders, and dementia.[2] It is being developed by Intra-Cellular Therapies.[2][3][1] As of January 2025, it has reached phase 2 clinical trials.[2][3]
SYN
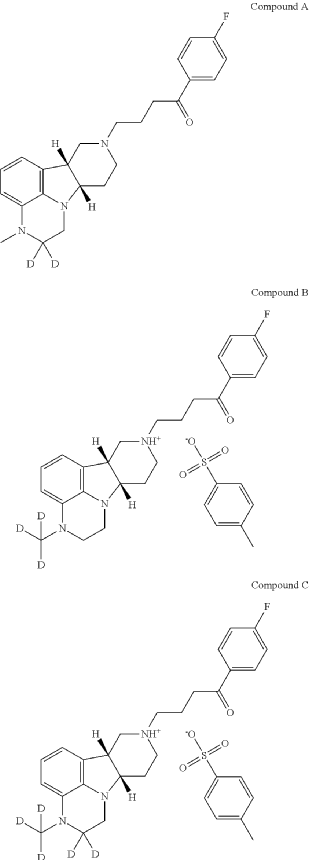
PAT
Example 2
2,2-D2-1-(4-fluorophenyl)-4-((6bR,10aS)-3-methyl-2,3,6b,7,10,10a-hexahydro-1H-pyrido[3′,4′: 4,5]pyrrolo[1,2,3-de]quinoxalin-8(9H)-yl)butan-1-one
| To a suspension of (6bR, 10aS)-3-Methyl-2-oxo-2,3,6b,9,10,10a-hexahydro-1H,7H-pyrido[3′,4′: 4,5]pyrrolo[1,2,3-de]quinoxaline-8-carboxylic acid ethyl ester (945 mg, 3 mmol) in THF (5 mL) is slowly added BD 3-THF (1.0 M in THF, 10 mL, 10 mmol) at room temperature. After completion of the addition, the reaction mixture is stirred at room temperature overnight and then carefully quenched with D 2O (2.0 mL). The solvent is removed under vacuum and the residue is suspended in HCl (12 N, 9 mL). After stirred at 95° C. for 20 h, the reaction mixture is cooled to room temperature and then adjusted to pH of 12 with 50% NaOH. The mixture is concentrated to dryness to give 2,2-d 2-(6bR, 10aS)-3-Methyl-2,3,6b,7,8,9,10,10a-octahydro-1H-pyrido[3′,4′: 4,5]pyrrolo[1, 2,3-de]quinoxaline as a brown solid, which is used directly for next step without further purification. MS (ESI) m/z 232.2 [M+H] +. |
PAT
- Novel compositions and methodsPublication Number: US-2021315891-A1Priority Date: 2018-08-29
- Transmucosal and subcutaneous compositionsPublication Number: US-10716786-B2Priority Date: 2017-03-24Grant Date: 2020-07-21
- Novel compositions and methodsPublication Number: US-2018271862-A1Priority Date: 2017-03-24
- Transmucosal methods for treating psychiatric and neurological conditionsPublication Number: US-11052083-B2Priority Date: 2017-03-24Grant Date: 2021-07-06
- Novel compositions and methodsPublication Number: US-2020375988-A1Priority Date: 2017-03-24
- Novel compositions and methodsPublication Number: US-2021361648-A1Priority Date: 2017-03-24
- Organic compoundsPublication Number: US-2019231780-A1Priority Date: 2016-03-25
- Organic compoundsPublication Number: US-10688097-B2Priority Date: 2016-03-25Grant Date: 2020-06-23
- Organic compoundsPublication Number: US-11096944-B2Priority Date: 2016-03-25Grant Date: 2021-08-24
- Organic compoundsPublication Number: US-2020352949-A1Priority Date: 2016-03-25
- Organic compounds
- Publication Number: US-2022008423-A1
- Priority Date: 2016-03-25



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- Oguma T, Jino K (2024). “Clinical Pipelines for Alzheimer’s Disease Psychosis and Agitation”. Chemical & Pharmaceutical Bulletin. 72 (7) c23-00416: 610–617. doi:10.1248/cpb.c23-00416. PMID 38945937.
- “Intra-Cellular Therapies”. AdisInsight. 29 January 2025. Retrieved 26 February 2025.
- “Delving into the Latest Updates on ITI-1284 with Synapse”. Synapse. 20 February 2025. Retrieved 26 February 2025.
- “Proposed INN: List 132 International Nonproprietary Names for Pharmaceutical Substances (INN)” (PDF). WHO Drug Information. 38 (4): 1073. 2024.
| Clinical data | |
|---|---|
| Other names | Lumateperone deuterated; Deuterated lumateperone; ITI-1284; ITI1284; ITI-1284-ODT-SL |
| Routes of administration | Sublingual (orally disintegrating tablet)[1] |
| Drug class | Atypical antipsychotic |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2102683-75-8 |
| PubChem CID | 140916642 |
| UNII | NBA7J58PPP |
| KEGG | D13268 |
| Chemical and physical data | |
| Formula | C24H26D2FN3O |
| Molar mass | 395.518 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
////////deulumateperone, anax labs, antipsychotic, ITI-1284, ITI 1284, NBA7J58PPP,

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










