Home » Posts tagged 'MYK 224'
Tag Archives: MYK 224
Delocamten
Delocamten
CAS 2417411-02-8
MFC19H21F2N3O3 MW377.4 g/mol
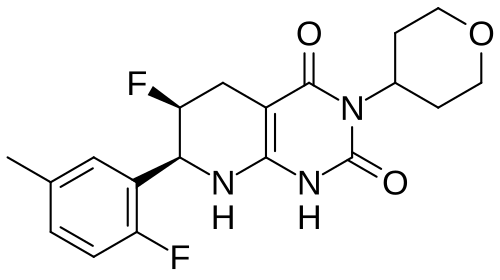
(6S,7S)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3-(oxan-4-yl)-5,6,7,8-tetrahydro-1H-pyrido[2,3-d]pyrimidine-2,4-dione
(6S,7S)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3-(oxan-4-yl)-5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4(1H,3H)-dione
cardiac myosin inhibitor, MYK-224; BMS-986435, MYK 224, BMS 986435, IE5886BN8T
Delocamten (development code MYK-224) is a small-molecule cardiac myosin inhibitor developed by Bristol Myers Squibb for hypertrophic cardiomyopathy.[1][2][3]
Delocamten is a small molecule drug. Delocamten is under investigation in clinical trial NCT06122779 (Study to Evaluate Safety, Tolerability and Drug Levels of BMS-986435/MYK-224 in Participants With Heart Failure With Preserved Ejection Fraction (HFpEF)). Delocamten has a monoisotopic molecular weight of 377.16 Da.
SYN
Example 1-3: Preparation of (6S,7S)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3-(tetrahydro-2H-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2, 4 (1H, 3H)-dione (3)
Step 7. Synthesis of (6S,7S)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3-(tetrahydro-2H-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2, 4 (1H, 3H)-dione (3). A mixture of crude 3G (1.0 g, 2.53 mmol) in CH 3CN (15 mL) was put into a microwave reactor with stirring at 120° C. for 30 min. Subsequently, the mixture was concentrated and the residue was purified by preparative HPLC (column: C18 silica gel; mobile phase: CH3CN:H 2O=20:80 (v v) increasing to CH3CN:H 2O=80:20 (v v) within 40 min; detector: UV 254 nm) to give compound 3 (302 mg, 32%), as a white solid, which was identified as Form 1 polymorph (see Example 2). LC-MS (ES, m/z): 378 [M+H] +; 1H NMR (300 MHz, d-DMSO): δ 10.20 (s, 1H), 7.38-7.05 (m, 3H), 6.45 (s, 1H), 5.11-4.81 (m, 3H), 3.89 (dd, J=10.8, 3.9 Hz, 2H), 3.34-3.27 (m, 3H), 2.76-2.48 (m, 4H), 2.28 (s, 3H), 1.39-1.36 (m, 2H); 19F NMR (376 MHz, d 6-DMSO): δ −123.51 (t, J=86.5 Hz), −191.57 (d, J=129.34 Hz).
PAT
Example 1-3: Preparation of (6S,7S)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3- (tetrahydro-2H-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2, 4 (1H, 3H)-dione (3).
Scheme 3
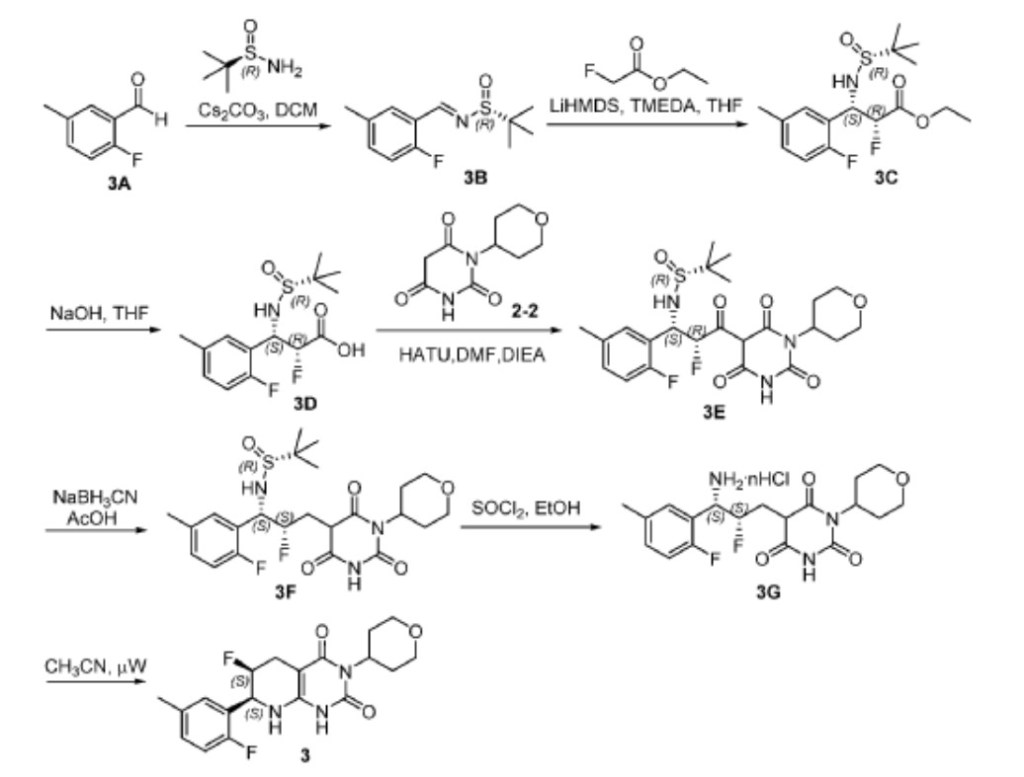
[0165] Step 1. Synthesis of (R,E)-N-(2-fluoro-5-methylbenzylidene)-2-methylpropane-2-sulfinamide (3B). The reaction mixture was filtered and the filtrate was diluted with ether (150 mL). Subsequently, the resulting suspension was filtered. The filtrate was concentrated and the residue was dried in vacuo to give 3B (8.7 g, 97%) as a yellow oil. LC-MS (ES, m/z): 242 [M+H] + ; 1 H NMR (400 MHz, d 6 -DMSO): d 8.87 (s, 1H), 7.76 (m, 1H), 7.29 (m, 1H), 7.03 (m, 1H), 2.37 (d, J = 1.0 Hz, 3H), 1.27 (s, 9H).
[0166] Step 2. Synthesis of ethyl (2R,3S)-3-(((R)-tert-butylsulfinyl)amino)-2-fluoro-3-(2-fluoro-5-methylphenyl)propanoate (3C). To a solution of 3B (4 g, 16.6 mmol), ethyl 2- fluoroacetate (2.6 g, 24.6 mmol), and TMEDA (4.8 mL) in anhydrous THF (40 mL) was added LiHMDS (1 M in THF, 24.6 mL, 24.6 mmol) dropwise at -78 o C over 30 min under an atmosphere of Ar. After stirring at -78 o C for 1 h, the reaction was quenched by adding 1 N aq.
HCl (50 mL), while maintaining the inner temperature of the mixture at < -20 o C. Subsequently, the mixture was concentrated to remove most of the organic solvent and then extracted with EtOAc (100 mL x 3). The combined organic extracts were washed with brine (100 mL) and dried over anhydrous Na2SO4. The solvent was removed and the residue was dried in vacuo to give crude 3C (6.0 g) as a yellow oil, which was used for the next step without further purification. LC-MS (ES, m/z): 348 [M+H] + .
[0167] Step 3. Synthesis of (2R,3S)-3-(((R)-tert-butylsulfinyl)amino)-2-fluoro-3-(2-fluoro-5-methylphenyl)propanoic acid (3D). To a solution of 3C (6.0 g, 17.3 mmol) in THF (40 mL) was added 1N aq. NaOH (34.6 mL, 34.6 mmol) at rt. After stirring at rt for 1 h, the reaction mixture was added ice water (50 mL). The resulting mixture was extracted with EtOAc (100 mL x 2). The aqueous layer was adjusted to pH 5 with sat. aq. citric acid, followed by extraction with EtOAc (100 mL x 3). Subsequently, the combined organic extracts were washed with brine (100 mL) and dried over anhydrous Na 2 SO 4 . The solvent was removed and the residue was purified by preparative HPLC (Column: LC-MS (ES, m/z): 320 [M+H] + ; 1 H NMR (400 MHz, d 6 -DMSO): d 13.57 (br, 1H), 7.55 (dd, J = 7.5, 2.2 Hz, 1H), 7.23– 6.94 (m, 2H), 6.04 (d, J = 10.8 Hz, 1H), 5.37– 4.86 (m, 2H), 2.29 (s, 3H), 1.12 (s, 9H).
[0168] Step 4. Synthesis of (R)-N-((1S,2R)-2-fluoro-1-(2-fluoro-5-methylphenyl)-3-oxo-3- (2,4,6-trioxo-1-(tetrahydro-2H-pyran-4-yl)hexahydropyrimidin-5-yl)propyl)-2-methylpropane-2-sulfinamide (3E). A solution of 3D (700 mg, 2.19 mmol), 2-2 (698 mg, 3.29 mmol), and HATU (1.25 g, 3.29 mmol) in DMF (10 mL) was added DIEA (849 mg, 6.57 mmol) at 0 o C under an atmosphere of Ar. aq. sodium bicarbonate (30 mL) and the resulting solution was extracted with ethyl acetate (50 mL x3). The combined organic extracts were washed with brine (50 mL x 2) and dried over anhydrous Na 2 SO 4 . The solvent was removed and the residue was dried in vacuo to give crude 3E (1.3 g) as a white solid, which was used for the next step without further purification. LC-MS (ES, m/z): 514 [M+H] + ; 1 H NMR (400 MHz, d 6 -DMSO): d 12.16 (br, 1H), 7.66– 7.45 (m, 1H), 7.23– 6.98 (m, 2H), 6.37 (m, 1H), 6.13 (d, J = 10.7 Hz, 1H), 5.22 (m, 1H), 4.79 (m, 1H), 3.94 (m, 2H), 3.35 (t, J = 11.7 Hz, 2H), 2.52– 2.39 (m, 2H), 2.29 (s, 3H), 1.49 (d, J = 12.2 Hz, 2H), 1.04 (s, 9H).
[0169] Step 5. Synthesis of (R)-N-((1S,2S)-2-fluoro-1-(2-fluoro-5-methylphenyl)-3-(2,4,6- trioxo-1-(tetrahydro-2H-pyran-4-yl)hexahydropyrimidin-5-yl)propyl)-2-methylpropane-2-sulfinamide (3F). A solution of crude 3E (1.3 g, 2.53 mmol) in AcOH (10 mL) was added NaBH3CN (398 mg, 6.33 mmol) at 0 o C under an atmosphere of Ar. After stirring at rt for 1 h, the reaction mixture was added ice water (20 mL) and the resulting solution was extracted with EtOAc (50 mL x 3). Next, the combined organic extracts were washed with brine (50 mL) and
dried over anhydrous Na2SO4. The solvent was removed and the residue was dried in vacuo to give crude 3F (1.3 g) as a white solid, which was used for the next step without further purification. LC-MS (ES, m/z): 500 [M+H] + ; 1 H NMR (400 MHz, d 6 -DMSO): d 11.31 (d, J = 28.1 Hz, 1H), 7.41 (d, J = 7.4 Hz, 1H), 7.27– 6.84 (m, 2H), 6.11– 5.78 (m, 2H), 5.08– 4.43 (m, 3H), 3.87 (m, 3H), 2.29 (s, 6H), 1.99 (s, 1H), 1.53– 1.28 (m, 2H), 1.10 (d, J = 2.1 Hz, 10H).
[0170] Step 6. Synthesis of 5-((2S,3S)-3-amino-2-fluoro-3-(2-fluoro-5-methylphenyl)propyl)-1-(tetrahydro-2H-pyran-4-yl)pyrimidine-2, 4, 6 (1H, 3H, 5H)-trione (3G). A solution of crude 3F (1.3 g, 2.60 mmol) in ethanol (10 mL) was added thionyl chloride (334 mg) at 0 o C. After stirring at rt for 1 h, the reaction mixture was concentrated and the residue was dried in vacuo to give crude 3G (1.0 g) as a white solid, which was used for the next step without further purification. LC-MS (ES, m/z): 396 [M+H] + .
[0171] Step 7. Synthesis of (6S,7S)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3-(tetrahydro-2H-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2, 4 (1H, 3H)-dione (3). A mixture of crude 3G (1.0 g, 2.53 mmol) in CH 3 CN (15 mL) was put into a microwave reactor with stirring at 120 o C for 30 min. Subsequently, the mixture was concentrated and the residue was purified by preparative HPLC (column: C18 silica gel; mobile phase: CH3CN:H2O = 20:80 (v/v) increasing to CH3CN:H2O = 80:20 (v/v) within 40 min; detector: UV 254 nm) to give compound 3 (302 mg, 32%), as a white solid, which was identified as Form 1 polymorph (see Example 2). LC-MS (ES, m/z): 378 [M+H] + ; 1 H NMR (300 MHz, d 6 -DMSO): d 10.20 (s, 1H), 7.38– 7.05 (m, 3H), 6.45 (s,1H), 5.11– 4.81 (m, 3H), 3.89 (dd, J = 10.8, 3.9 Hz, 2H), 3.34– 3.27 (m, 3H), 2.76–2.48 (m, 4H), 2.28 (s, 3H), 1.39–1.36 (m, 2H); 19 F NMR (376 MHz, d 6 -DMSO): d -123.51 (t, J = 86.5 Hz), -191.57 (d, J = 129.34 Hz).
PAT
- Tetrahydropyran (thp)-substituted bicyclic-pyrimidinedione compoundsPublication Number: EP-4464321-A2Priority Date: 2018-10-29
- Tetrahydropyran-substituted bicyclic pyrimidinedione compounds (THP)Publication Number: ES-2986923-T3Priority Date: 2018-10-29Grant Date: 2024-11-13
- Substituted 5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4-diones for treating cardiac diseasesPublication Number: US-2024025894-A1Priority Date: 2018-10-29
- Substituted 5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4-diones for treating cardiac diseasesPublication Number: US-12344607-B2Priority Date: 2018-10-29Grant Date: 2025-07-01
- Substituted 5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4-diones for treating cardiac diseasesPublication Number: US-2025282779-A1Priority Date: 2018-10-29
- Tetrahydropyran (THP) Substituted Bicyclic Pyrimidinedione CompoundsPublication Number: CN-113056465-APriority Date: 2018-10-29
- Tetrahydropyrane (THP) -substituted bicyclic pyrimidinedione compoundsPublication Number: CN-119977963-APriority Date: 2018-10-29
- Tetrahydropyran (thp)-substituted bicyclic-pyrimidinedione compoundsPublication Number: EP-3873904-B1Priority Date: 2018-10-29Grant Date: 2024-07-10
- Substituted 5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4-diones for treating cardiac diseasesPublication Number: US-11034693-B2Priority Date: 2018-10-29Grant Date: 2021-06-15
- Substituted 5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4-diones for treating cardiac diseasesPublication Number: US-2022106314-A1Priority Date: 2018-10-29
- Tetrahydropyran (THP)-Substituted Bicyclic-Pyrimidinedione CompoundsPublication Number: JP-2024063091-APriority Date: 2018-10-29
- Tetrahydropyran (thp)-substituted bicyclic-pyrimidinedione compoundsPublication Number: TW-202426449-APriority Date: 2018-10-29
- Tetrahydropyrane (THP) -substituted bicyclic pyrimidinedione compoundsPublication Number: CN-113056465-BPriority Date: 2018-10-29Grant Date: 2025-01-28
- Tetrahydropyran (thp)-substituted bicyclic-pyrimidinedione compoundsPublication Number: US-2020165247-A1Priority Date: 2018-10-29
- BICYCLIC PYRIMIDINODIONA COMPOUNDS REPLACED WITH TETRAHYDROPYRAN, POLYMORPHIC FORM OF THE SAME AND THE USE OF THE SAME FOR THE TREATMENT OF HCMPublication Number: AR-116880-A1Priority Date: 2018-10-29
- Substituted 5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4-diones for treating cardiac diseasesPublication Number: US-2022340569-A1Priority Date: 2018-10-29
- Tetrahydropyran (thp)-substituted bicyclic-pyrimidinedione compoundsPublication Number: EP-3873904-A1Priority Date: 2018-10-29
- CRYSTALLINE FORMS OF (6S,7S)-6-FLUORO-7-(2-FLUORO-5-METHYLPHENYL)- 3-(TETRAHYDRO-2H-PYRAN-4-YL)-5,6,7,8-TETRAHYDROPYRIDO[2,3- d]PYRIMIDINE-2,4(1H,3H)-DIONEPublication Number: WO-2024026058-A8Priority Date: 2022-07-29
- Crystalline forms of (6s,7s)-6-fluoro-7-(2-fluoro-5-methylphenyl)- 3-(tetrahydro-2h-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3- d]pyrimidine-2,4(1h,3h)-dionePublication Number: EP-4561697-A1Priority Date: 2022-07-29
- Methods of Administering Myosin InhibitorsPublication Number: US-2023338378-A1Priority Date: 2022-04-26
- Methods of treatment with myosin modulatorPublication Number: US-2023158027-A1Priority Date: 2019-11-10
- tetrahydropyran-substituted bicyclic pyrimidinedione compounds (thp)Publication Number: BR-112021008077-A2Priority Date: 2018-10-29
- CRYSTALLINE FORMS OF (6S,7S)-6-FLUORO-7-(2-FLUORO-5-METHYLPHENYL)-3-(TETRAHYDRO-2H-PYRAN-4-YL)-5,6,7,8-TETRAHYDROPYRIDO[2,3-d]PYRIMIDINE-2,4(1H,3H)-DIONEPublication Number: US-2025034129-A1Priority Date: 2023-07-28
- CRYSTALLINE FORMS OF (6S,7S)-6-FLUORO-7-(2-FLUORO-5-METHYLPHENYL)-3-(TETRAHYDRO-2H-PYRANO-4-IL)-5,6,7,8-TETRAHYDROPYRIDE[2 ,3-D]PYRIMIDINE-2,4(1H,3H)-DIONEPublication Number: AR-130058-A1Priority Date: 2022-07-29
- Crystalline forms of (6s,7s)-6-fluoro-7-(2-fluoro-5-methylphenyl)- 3-(tetrahydro-2h-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3- d]pyrimidine-2,4(lh,3h)-dionePublication Number: WO-2024026058-A1Priority Date: 2022-07-29
- Crystalline form of (6S,7S)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3-(tetrahydro-2H-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4(1H,3H)-dionePublication Number: CN-119855816-APriority Date: 2022-07-29
- Crystalline forms of (6s,7s)-6-fluoro-7-(2-fluoro-5-methylphenyl)-3-(tetrahydro-2h-pyran-4-yl)-5,6,7,8-tetrahydropyrido[2,3-d]pyrimidine-2,4(1h,3h)-dionePublication Number: TW-202412788-APriority Date: 2022-07-29



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- Lehman, Sarah J.; Crocini, Claudia; Leinwand, Leslie A. (June 2022). “Targeting the sarcomere in inherited cardiomyopathies”. Nature Reviews Cardiology. 19 (6): 353–363. doi:10.1038/s41569-022-00682-0. ISSN 1759-5010. PMC 9119933. PMID 35304599.
- Sebastian, Sneha Annie; Padda, Inderbir; Lehr, Eric J.; Johal, Gurpreet (September 2023). “Aficamten: A Breakthrough Therapy for Symptomatic Obstructive Hypertrophic Cardiomyopathy”. American Journal of Cardiovascular Drugs. 23 (5): 519–532. doi:10.1007/s40256-023-00599-0. PMID 37526885. S2CID 260348901.
- Packard, Elizabeth; de Feria, Alejandro; Peshin, Supriya; Reza, Nosheen; Owens, Anjali Tiku (December 2022). “Contemporary Therapies and Future Directions in the Management of Hypertrophic Cardiomyopathy”. Cardiology and Therapy. 11 (4): 491–507. doi:10.1007/s40119-022-00283-5. PMC 9652179. PMID 36243823.
////////delocamten, ANAX LAB, cardiac myosin inhibitor, MYK-224; BMS-986435, MYK 224, BMS 986435, IE5886BN8T

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










