Ono Pharmaceutical Co has become the first company in the world to get an approval for a PD-1 checkpoint inhibitor, as regulators in Japan gave the green light to nivolumab, developed with Bristol-Myers Squibb, as a treatment for melanoma.
Home » Posts tagged 'melanoma'
Tag Archives: melanoma
BQ-788
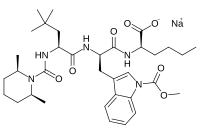
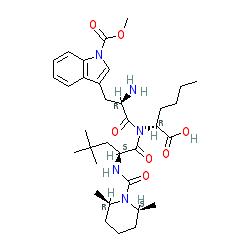
BQ-788
- Molecular FormulaC34H50N5NaO7
- Average mass663.780 Da
SP ROT +3.8 ° Conc: 1.032 g/100mL; methanol; Wavlenght: 589.3 nm, Development of an efficient strategy for the synthesis of the ETB receptor antagonist BQ-788 and some related analogues
Peptides (New York, NY, United States) (2005), 26, (8), 1441-1453., https://doi.org/10.1016/j.peptides.2005.03.022
FOR FREE FORM +19.6 °, Conc: 0.998 g/100mL; : N,N-dimethylformamide; 589.3 nm
BQ-788 is a selective ETB antagonist.[1]
presumed to be under license from Banyu , was investigating BQ-788, a selective endothelin receptor B (ETRB) antagonist, for treating metastatic melanoma. By December 2009, the drug was in validation.
Also claimed is their use as an ETBR antagonist and for treating cancers, such as brain cancer, pancreas cancer, colon cancer, breast cancer, ovary cancer, prostate cancer, glioblastoma, solid tumor, melanoma and squamous cell carcinoma. Represent a first filing from ENB Therapeutics Inc and the inventors on these deuterated forms of BQ-788. Melcure SarL ,

SYN
By Brosseau, Jean-Philippe et alFrom Peptides (New York, NY, United States), 26(8), 1441-1453; 2005
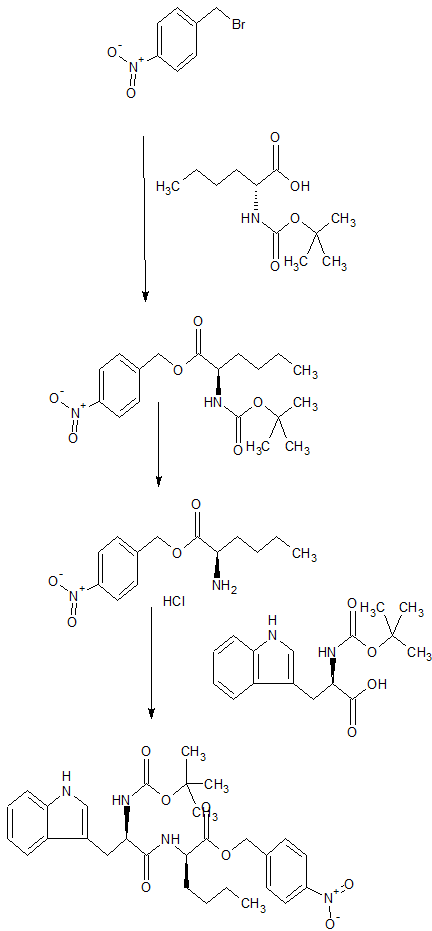
CONTD…………

PAPER
https://pubs.acs.org/doi/pdf/10.1021/jo00130a028

N-(cw-2,6-Dimethylpiperidinocarbonyl)-y-methylleucylD-l-(methoxycarbonyl)tryptophanyl-D-norleucine Sodium Salt (1, BQ-788). To a solution of 15 (3.5 g, 5.5 mmol) in methanol (50 mL) was slowly added 5% aqueous NaHCOs (300 mL) over a period of 30 min. The solution was stirred until clarity was achieved (30 min, 23 °C). The solution was diluted with water (200 mL), and the resulting solution was passed through a C18 (60 mL) cartridge preequilbrated in water. BQ-788 (1) was eluted with methanol (2 x 50 mL), concentrated under reduced pressure, resuspended in water (50 mL), and lyophilized to quantitatively yield compound 1 as a white powder:
HPLC £r = 16.4 (gradient A, > 99%);
NMR (400 MHz, DMSO-d6) ó 0.80 (s, 9H), 0.74-0.85 (m, 3H), 1.00 (d, 3H), 1.02 (d, 3H), 1.10-1.25 (m, 6H), 1.30-1.55 (m, 6H), 1.60-1.75 (m, 2H), 2.92 (dd, 1H), 3.12 (dd, 1H), 3.78 (m, 1H), 3.95 (s, 3H), 4.08 (m, 1H), 4.13 (m, 1H), 4.29 (m, 1H), 4.50 (m, 1H), 5.98 (d, 1H), 7.22 (t, 1H), 7.32 (t, 1H), 7.50 (s, 1H), 7.58 (br d, 1H), 7.65 (d, 1H), 8.05 (d, 1H), 8.15 (br d, 1H) ESMS m/z 640.6 (M).
PATENT
WO-2019140324
Novel deuterated analogs of a substituted heterocyclic compound, particularly BQ-788 , processes for their preparation and compositions and combinations comprising them are claimed.
PAPER
https://www.sciencedirect.com/science/article/abs/pii/S0196978105001415

PAPER
By He, John X.; Cody, Wayne L.; Doherty, Annette M., From Journal of Organic Chemistry (1995), 60(25), 8262-6
Journal of medicinal chemistry (1996), 39(12), 2313-30.
References
 |
|
| Names | |
|---|---|
| Systematic IUPAC name
Sodium N-{[(2R,6S)-2,6-dimethyl-1-piperidinyl]carbonyl}-4-methyl-L-leucyl-N-[(1R)-1-carboxylatopentyl]-1-(methoxycarbonyl)-D-tryptophanamide
|
|
| Identifiers | |
|
3D model (JSmol)
|
|
| ChemSpider | |
|
PubChem CID
|
|
|
CompTox Dashboard (EPA)
|
|
| Properties | |
| C34H50N5NaO7 | |
| Molar mass | 663.792 g·mol−1 |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
|
|
///////////BQ-788, BQ 788, BQ788, ETBR antagonist, cancers, brain cancer, pancreas cancer, colon cancer, breast cancer, ovary cancer, prostate cancer, glioblastoma, solid tumor, melanoma, squamous cell carcinoma, PEPTIDE
CCCC[C@H](C(=O)O)NC(=O)[C@@H](Cc1cn(c2c1cccc2)C(=O)OC)NC(=O)[C@H](CC(C)(C)C)NC(=O)N3[C@@H](CCC[C@@H]3C)C
Japan approves world’s first PD-1 drug, nivolumab
http://www.pharmatimes.com/Article/14-07-07/Japan_approves_world_s_first_PD-1_drug_nivolumab.aspx
old article cut paste

NIVOLUMAB
Anti-PD-1;BMS-936558; ONO-4538
PRONUNCIATION nye vol’ ue mab
THERAPEUTIC CLAIM Treatment of cancer
CHEMICAL DESCRIPTION
A fully human IgG4 antibody blocking the programmed cell death-1 receptor (Medarex/Ono Pharmaceuticals/Bristol-Myers Squibb)
MOLECULAR FORMULA C6362H9862N1712O1995S42
MOLECULAR WEIGHT 143.6 kDa
SPONSOR Bristol-Myers Squibb
CODE DESIGNATION MDX-1106, BMS-936558
CAS REGISTRY NUMBER 946414-94-4
Bristol-Myers Squibb announced promising results from an expanded phase 1 dose-ranging study of its lung cancer drug nivolumab
Nivolumab (nye vol’ ue mab) is a fully human IgG4 monoclonal antibody designed for the treatment of cancer. Nivolumab was developed by Bristol-Myers Squibb and is also known as BMS-936558 and MDX1106.[1] Nivolumab acts as an immunomodulator by blocking ligand activation of the Programmed cell death 1 receptor.
A Phase 1 clinical trial [2] tested nivolumab at doses ranging from 0.1 to 10.0 mg per kilogram of body weight, every 2 weeks. Response was assessed after each 8-week treatment cycle, and were evaluable for 236 of 296 patients. Study authors concluded that:”Anti-PD-1 antibody produced objective responses in approximately one in four to one in five patients with non–small-cell lung cancer, melanoma, or renal-cell cancer; the adverse-event profile does not appear to preclude its use.”[3]
Phase III clinical trials of nivolumab are recruiting in the US and EU.[4]
- Statement On A Nonproprietary Name Adopted By The USAN Council – Nivolumab, American Medical Association.
- A Phase 1b Study of MDX-1106 in Subjects With Advanced or Recurrent Malignancies (MDX1106-03), NIH.
- Topalian SL, et al. (June 2012). “Safety, Activity, and Immune Correlates of Anti–PD-1 Antibody in Cancer”. New England Journal of Medicine 366. doi:10.1056/NEJMoa1200690. Lay summary – New York Times.
- Nivolumab at ClinicalTrials.gov, A service of the U.S. National Institutes of Health.
The PD-1 blocking antibody nivolumab continues to demonstrate sustained clinical activity in previously treated patients with advanced non-small cell lung cancer (NSCLC), according to updated long-term survival data from a phase I trial.
Survival rates at one year with nivolumab were 42% and reached 24% at two years, according to the median 20.3-month follow up. Additionally, the objective response rate (ORR) with nivolumab, defined as complete or partial responses by standard RECIST criteria, was 17% for patients with NSCLC. Results from the updated analysis will be presented during the 2013 World Conference on Lung Cancer on October 29.
“Lung cancer is very difficult to treat and there continues to be a high unmet medical need for these patients, especially those who have received multiple treatments,” David R. Spigel, MD, the program director of Lung Cancer Research at the Sarah Cannon Research Institute and one of the authors of the updated analysis, said in a statement.
“With nivolumab, we are investigating an approach to treating lung cancer that is designed to work with the body’s own immune system, and these are encouraging phase I results that support further investigation in larger scale trials.”
In the phase I trial, 306 patients received intravenous nivolumab at 0.1–10 mg/kg every-other-week for ≤12 cycles (4 doses/8 week cycle). In all, the trial enrolled patients with NSCLC, melanoma, renal cell carcinoma, colorectal cancer, and prostate cancer.
The long-term follow up focused specifically on the 129 patients with NSCLC. In this subgroup, patients treated with nivolumab showed encouraging clinical activity. The participants had a median age of 65 years and good performance status scores, and more than half had received three or more prior therapies. Across all doses of nivolumab, the median overall survival was 9.9 months, based on Kaplan-Meier estimates.
In a previous update of the full trial results presented at the 2013 ASCO Annual Meeting, drug-related adverse events of all grades occurred in 72% of patients and grade 3/4 events occurred in 15%. Grade 3/4 pneumonitis related to treatment with nivolumab emerged early in the trial, resulting in 3 deaths. As a result, a treatment algorithm for early detection and management was developed to prevent this serious side effect.
Nivolumab is a fully human monoclonal antibody that blocks the PD-1 receptor from binding to both of its known ligands, PD-L1 and PD-L2. This mechanism, along with early data, suggested an associated between PD-L1 expression and response to treatment.
In separate analysis presented at the 2013 World Conference on Lung Cancer, the association of tumor PD-L1 expression and clinical activity in patients with NSCLC treated with nivolumab was further explored. Of the 129 patients with NSCLC treated with nivolumab in the phase I trial, 63 with NSCLC were tested for PD-L1 expression by immunohistochemistry (29 squamous; 34 non-squamous).
Gene Therapy for Melanoma: Progress and Perspectives

FIGURE 1.
Schematic representation of the wild type counterpart of the typically used recombinant viral vectors. (A) Gammaretroviruses and (B) lentiviruses share similar structures, but differ greatly in their genomes and their impact on cellular function. Gag, pro, pol and env genes encode structural proteins of the capsid, protease, reverse transcriptase and envelope proteins, respectively. The additional lentiviral genes perform regulatory functions as well as alter cellular function. (C) The serotype 5 adenovirus has a protein capsid (non-enveloped) and a large, complex genome that encodes critical genes for viral replication (E1a, E1b) as well as structural and functional genes that regulate both viral and cellular activities.
Introduction
Gene therapy, the therapeutic transfer of genetic information to a target cell, continues to be a promising alternative in the fight against cancer. In the case of melanoma, the use of an experimental treatment is justified since this disease is incurable in its advanced stages. Is gene therapy a viable option for the treatment of melanoma patients? In this chapter, we will attempt to answer this question by exploring the intersection between the technology of gene therapy and the biology of melanoma, a point at which opportunities for intervention are revealed.
Gene Therapy for Melanoma: Progress and Perspectives
[1] Cancer Institute of Sao Paulo, University of Sao Paulo School of Medicine, Brazil
[2] University of Sao Paulo, Biomedical Sciences Institute, Brazil

……………….
The New Jersey drug giant Merck announced this morning that its investigational cancer drug Pembrolizumab, lambrolizumab (MK-3475) had received the breakthrough blessing in recognition of the dramatic clinical benefits observed in an open-label, phase 1 trial involving people with advanced melanoma.
STRUCTURAL FORMULA
Heavy chain
QVQLVQSGVE VKKPGASVKV SCKASGYTFT NYYMYWVRQA PGQGLEWMGG 50
INPSNGGTNF NEKFKNRVTL TTDSSTTTAY MELKSLQFDD TAVYYCARRD 100
YRFDMGFDYW GQGTTVTVSS ASTKGPSVFP LAPCSRSTSE STAALGCLVK 150
DYFPEPVTVS WNSGALTSGV HTFPAVLQSS GLYSLSSVVT VPSSSLGTKT 200
YTCNVDHKPS NTKVDKRVES KYGPPCPPCP APEFLGGPSV FLFPPKPKDT 250
LMISRTPEVT CVVVDVSQED PEVQFNWYVD GVEVHNAKTK PREEQFNSTY 300
RVVSVLTVLH QDWLNGKEYK CKVSNKGLPS SIEKTISKAK GQPREPQVYT 350
LPPSQEEMTK NQVSLTCLVK GFYPSDIAVE WESNGQPENN YKTTPPVLDS 400
DGSFFLYSRL TVDKSRWQEG NVFSCSVMHE ALHNHYTQKS LSLSLGK 447
Light chain
EIVLTQSPAT LSLSPGERAT LSCRASKGVS TSGYSYLHWY QQKPGQAPRL 50′
LIYLASYLES GVPARFSGSG SGTDFTLTIS SLEPEDFAVY YCQHSRDLPL 100′
TFGGGTKVEI KRTVAAPSVF IFPPSDEQLK SGTASVVCLL NNFYPREAKV 150′
QWKVDNALQS GNSQESVTEQ DSKDSTYSLS STLTLSKADY EKHKVYACEV 200′
THQGLSSPVT KSFNRGEC 218′
Disulfide bridges
22-96 22”-96” 23′-92′ 23”’-92”’ 134-218′ 134”-218”’ 138′-198′ 138”’-198”’
147-203 147”-203” 226-226” 229-229” 261-321 261”-321” 367-425 367”-425”
Glycosylation sites (N)
Asn-297 Asn-297”
lambrolizumab, or MK-3475
| C6504H10004N1716O2036S46 (peptide) | |
| Mol. mass | 146.3 kDa (peptide) |
Pembrolizumab, Lambrolizumab (also known as MK-3475) is a drug in development by Merck that targets the PD-1 receptor. The drug is intended for use in treating metastatic melanoma.
http://www.ama-assn.org/resources/doc/usan/lambrolizumab.pdf structureof lambrolizumab, or MK-3475
https://download.ama-assn.org/resources/doc/usan/x-pub/pembrolizumab.pdf
Statement on a Nonproprietary Name Adopted by the USAN Council. November 27, 2013.
see above link for change in name
may 2, 2013,
An experimental drug from Merck that unleashes the body’s immune system significantly shrank tumors in 38 percent of patients with advanced melanoma, putting the company squarely in the race to bring to market one of what many experts view as the most promising class of drugs in years.
The drugs are attracting attention here at the annual meeting of the American Society of Clinical Oncology, even though they are still in the early stage of testing. Data from drugs developed by Bristol-Myers Squibb and by Roche had already been released.
The drugs work by disabling a brake that prevents the immune system from attacking cancer cells. The brake is a protein on immune system cells called programmed death 1 receptor, or PD-1.
Merck’s study, which was presented here Sunday and also published in the New England Journal of Medicine, involved 135 patients. While tumors shrank in 38 percent of the patients over all, the rate was 52 percent for patients who got the highest dose of the drug, which is called lambrolizumab, or MK-3475.
Hamid, O; Robert, C; Daud, A; Hodi, F. S.; Hwu, W. J.; Kefford, R; Wolchok, J. D.; Hersey, P; Joseph, R. W.; Weber, J. S.; Dronca, R; Gangadhar, T. C.; Patnaik, A; Zarour, H; Joshua, A. M.; Gergich, K; Elassaiss-Schaap, J; Algazi, A; Mateus, C; Boasberg, P; Tumeh, P. C.; Chmielowski, B; Ebbinghaus, S. W.; Li, X. N.; Kang, S. P.; Ribas, A (2013). “Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma”. New England Journal of Medicine 369 (2): 134–44. doi:10.1056/NEJMoa1305133. PMID 23724846

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










