Home » Posts tagged 'MDX-1106'
Tag Archives: MDX-1106
NKTR 214
CAS 946414-94-4
- BMS 936558
- MDX 1106
- NKTR 214
- ONO 4538
- Opdivio
- NIVOLUMAB
Pegylated engineered interleukin-2 (IL-2) with altered receptor binding
NKTR-214 is a cytokine (investigational agent) that is designed to target CD122, a protein which is found on certain immune cells (known as CD8+ T Cells and Natural Killer Cells) to expand these cells to promote their anti-tumor effects. Nivolumab is a full human monoclonal antibody that binds to a molecule called PD-1 (programmed cell death protein 1) on immune cells and promotes anti-tumor effects.
Protein Sequence
Sequence Length: 1308, 440, 440, 214, 214multichain; modified (modifications unspecified)
NKTR-214 is a CD122-biased cytokine in phase II clinical trials at the M.D. Anderson Cancer Center for the treatment of advanced sarcoma in combination with nivolumab.
M.D. Anderson Cancer Center, PHASE 2, SARCOMA
NKTR-214 in combination with OPDIVO® (nivolumab)
RESEARCH FOCUS: Immuno-oncology
DISCOVERED AND WHOLLY OWNED BY NEKTAR
About NKTR-214, Nektar’s Lead Immuno-oncology Candidate
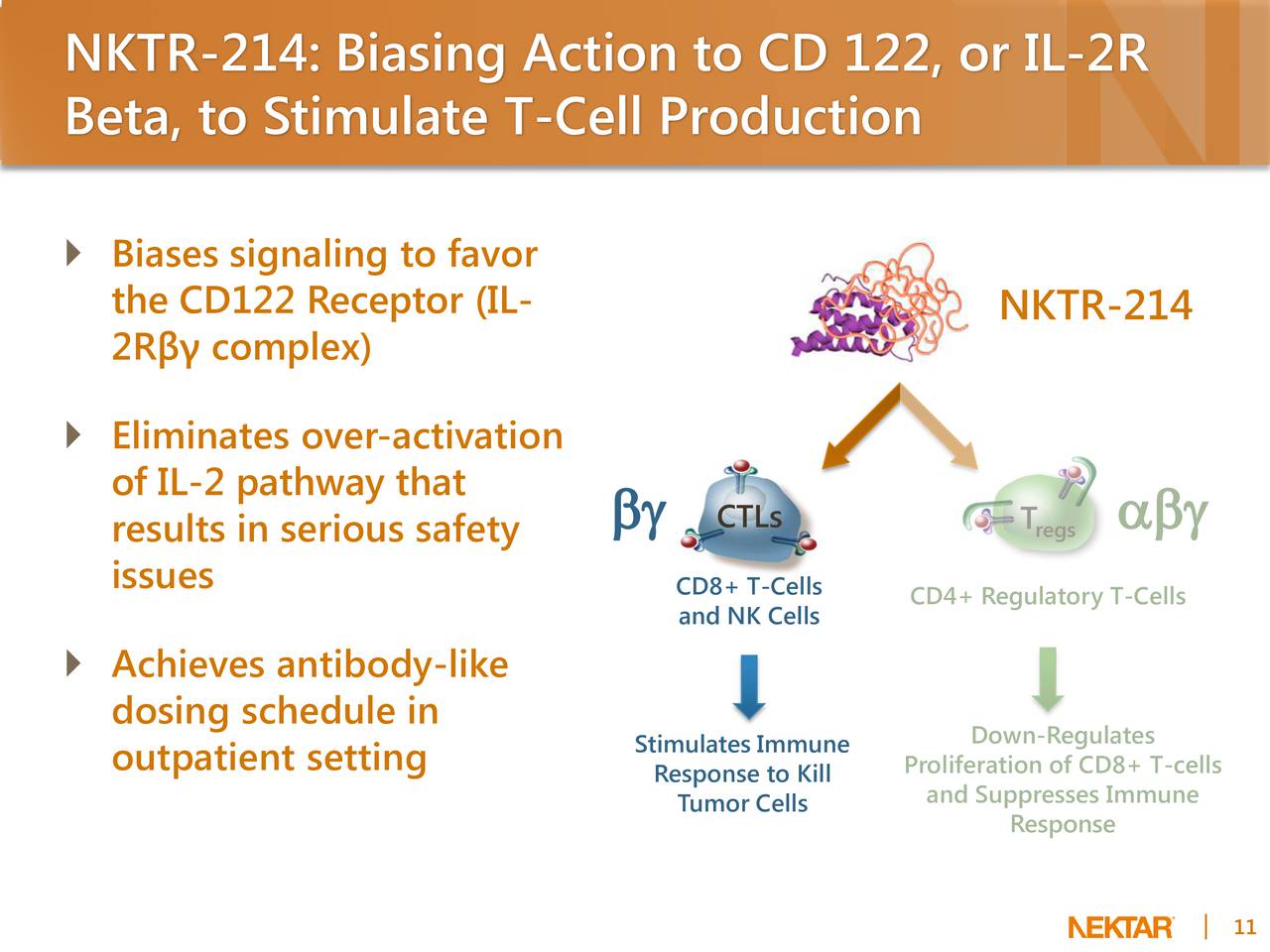
NKTR-214 is a CD122-biased agonist designed to stimulate the patient’s own immune system to fight cancer. NKTR-214 is designed to grow specific cancer-killing T cells and natural killer (NK) cell populations in the body which fight cancer, which are known as endogenous tumor-infiltrating lymphocytes (TILs). NKTR-214 stimulates these cancer-killing immune cells in the body by targeting CD122 specific receptors found on the surface of these immune cells, known as CD8+ effector T cells and Natural Killer (NK) cells. CD122, which is also known as the Interleukin-2 receptor beta subunit, is a key signaling receptor that is known to increase proliferation of these effector T cells.1 In preclinical studies, treatment with NKTR-214 results in a rapid expansion of these cells and mobilization into the tumor micro-environment. NKTR-214 has an antibody-like dosing regimen similar to the existing checkpoint inhibitor class of approved medicines.
In preclinical studies, NKTR-214 demonstrated a mean ratio of 450:1 within the tumor micro-environment of CD8-positive effector T cells, which promote tumor destruction, compared with CD4-positive regulatory T cells, which are a type of cell that can suppress tumor-killing T cells.2Furthermore, a single dose of NKTR-214 resulted in a 500-fold AUC exposure within the tumor compared with an equivalent dose of the existing IL-2 therapy, enabling, for the first time, an antibody-like dosing regimen for a cytokine.2 In dosing studies in non-human primates, there was no evidence of severe side effects such as low blood pressure or vascular leak syndrome with NKTR-214 at predicted clinical therapeutic doses.2 NKTR-214 has a range of potential uses against multiple tumor types, including melanoma (the most serious type of skin cancer), kidney cancer and non-small cell lung cancer (the most common form of lung cancer).
A Phase 1 study evaluating NKTR-214 as a single agent in patients with locally recurrent or metastatic solid tumors including melanoma, renal cell carcinoma (RCC), bladder, colorectal and other solid tumors is ongoing with patient enrollment complete. Results from this Phase 1 trial were presented at the Society for Immunotherapy of Cancer (SITC) 2016 Annual Meeting and showed encouraging evidence of anti-tumor activity, and a favorable safety and tolerability profile. (Poster #387)
In September 2016, Nektar entered into a clinical collaboration with Bristol-Myers Squibb to evaluate NKTR-214 as a potential combination treatment regimen with Opdivo (nivolumab) in five tumor types and eight potential indications. The Phase 1/2 PIVOT clinical trials, known as PIVOT-02 and PIVOT-04 will enroll up to 260 patients and will evaluate the potential for the combination of Opdivo (nivolumab) and NKTR-214 to show improved and sustained efficacy and tolerability above the current standard of care in melanoma, kidney, triple-negative breast cancer, bladder and non-small cell lung cancer patients.
In May 2017, Nektar entered into a research collaboration with Takeda to explore the combination of NKTR-214 with five oncology compounds from Takeda’s cancer portfolio including a SYK-inhibitor and a proteasome inhibitor. The collaboration will explore the anti-cancer activity of NKTR-214 combined with five different targeted mechanisms in preclinical tumor models of lymphoma, melanoma and colorectal cancer to identify which combination treatment regimens show the most promise for possible advancement into the clinic.
Under the terms of the collaboration, the companies will share costs related to the preclinical studies and each will contribute their respective compounds to the research collaboration. Nektar and Takeda will each maintain global commercial rights to their respective drugs and/or drug candidates.
Additional development plans for NKTR-214 include combination studies with additional checkpoint inhibitors, cell therapies and vaccines.
![]()
About the Excel NKTR-214 Phase 1/2 Study
The dose-escalation stage of the Excel Phase 1/2 study is designed to evaluate safety, efficacy, and define the recommended Phase 2 dose of NKTR-214 in approximately 20 patients with solid tumors. In addition to a determination of the recommended Phase 2 dose, the study will assess preliminary anti-tumor activity, including objective response rate (ORR). The immunologic effect of NKTR-214 on tumor-infiltrating lymphocytes (TILs) and other immune infiltrating cells in both blood and tumor tissue will also be assessed. Enrollment in the dose escalation study is completed. More information on the Excel Phase 1/2 study can be found on clinicaltrials.gov.

About the PIVOT Phase 1/2 Program: NKTR-214 in combination with OPDIVO® (nivolumab)
The dose escalation stage of the PIVOT program (PIVOT-02 Phase 1/2 study) is underway and will determine the recommended Phase 2 dose of NKTR-214 administered in combination with nivolumab. The study is first evaluating the clinical benefit, safety, and tolerability of combining NKTR-214 with nivolumab in approximately 30 patients with melanoma, renal cell carcinoma or non-small cell lung cancer. Once the recommended Phase 2 dose is achieved, the study will expand into additional patients for each tumor type. The second phase of the expansion cohorts in the PIVOT program (PIVOT-04 Phase 2 study) will evaluate safety and efficacy of the combination in up to 260 patients, in five tumor types and eight indications, including first and second-line melanoma, second-line renal cell carcinoma in immune-oncology therapy (IO) naïve and IO-relapsed patients, second-line non-small cell lung cancer in IO-naïve and IO-relapsed patients, first-line urothelial carcinoma, and second-line triple negative breast cancer. This study is expected to initiate in the second quarter of 2017.
Information on the PIVOT-02 study can be found on clinicaltrials.gov.
![]()
About the PROPEL Phase 1/2 Program: NKTR-214 in combination with TECENTRIQ® (atezolizumab) or KEYTRUDA®(pembrolizumab)
The dose escalation stage of the PROPEL program will determine the recommended Phase 2 dose of NKTR-214 administered in combination with anti-PD-L1 agent, atezolizumab or anti-PD-1 agent, pembrolizumab. The study will evaluate the clinical benefit, safety and tolerability of combining NKTR-214 with atezolizumab or pembrolizumab and will enroll patients into two separate arms concurrently. The first arm will evaluate an every three-week dose regimen of NKTR-214 in combination with atezolizumab in up to 30 patients in approved treatment settings of atezolizumab, including patients with non-small cell lung cancer or bladder cancer. The second arm will evaluate an every three-week dose regimen of NKTR-214 in combination with pembrolizumab in up to 30 patients in approved treatment settings of pembrolizumab, including patients with melanoma, non-small cell lung cancer or bladder cancer.
Information on the PROPEL study can be found on clinicaltrials.gov.
References
1Boyman, J., et al., Nature Reviews Immunology, 2012, 12, 180-190.
2Charych, D., et al., Clin Can Res; 22(3) February 1, 2016
http://www.nektar.com/application/files/7714/7887/7212/2016_SITC_NKTR-214-clinical_poster.pdf
https://www.google.co.in/patents/WO2015125159A1?cl=en
| Inventors | Murali Krishna Addepalli, Deborah H. Charych, Seema Kantak, Steven Robert Lee |
| Applicant | Nektar Therapeutics (India) Pvt. Ltd., Nektar Therapeutics |
////////////946414-94-4, BMS 936558, MDX 1106, NKTR 214, ONO 4538, Opdivio, NIVOLUMAB, PHASE 2
Bristol-Myers Squibb announced promising results from an expanded phase 1 dose-ranging study of its lung cancer drug nivolumab

NIVOLUMAB
Anti-PD-1;BMS-936558; ONO-4538
PRONUNCIATION nye vol’ ue mab
THERAPEUTIC CLAIM Treatment of cancer
CHEMICAL DESCRIPTION
A fully human IgG4 antibody blocking the programmed cell death-1 receptor (Medarex/Ono Pharmaceuticals/Bristol-Myers Squibb)
MOLECULAR FORMULA C6362H9862N1712O1995S42
MOLECULAR WEIGHT 143.6 kDa
SPONSOR Bristol-Myers Squibb
CODE DESIGNATION MDX-1106, BMS-936558
CAS REGISTRY NUMBER 946414-94-4
Bristol-Myers Squibb announced promising results from an expanded phase 1 dose-ranging study of its lung cancer drug nivolumab
Nivolumab (nye vol’ ue mab) is a fully human IgG4 monoclonal antibody designed for the treatment of cancer. Nivolumab was developed by Bristol-Myers Squibb and is also known as BMS-936558 and MDX1106.[1] Nivolumab acts as an immunomodulator by blocking ligand activation of the Programmed cell death 1 receptor.
A Phase 1 clinical trial [2] tested nivolumab at doses ranging from 0.1 to 10.0 mg per kilogram of body weight, every 2 weeks. Response was assessed after each 8-week treatment cycle, and were evaluable for 236 of 296 patients. Study authors concluded that:”Anti-PD-1 antibody produced objective responses in approximately one in four to one in five patients with non–small-cell lung cancer, melanoma, or renal-cell cancer; the adverse-event profile does not appear to preclude its use.”[3]
Phase III clinical trials of nivolumab are recruiting in the US and EU.[4]
- Statement On A Nonproprietary Name Adopted By The USAN Council – Nivolumab, American Medical Association.
- A Phase 1b Study of MDX-1106 in Subjects With Advanced or Recurrent Malignancies (MDX1106-03), NIH.
- Topalian SL, et al. (June 2012). “Safety, Activity, and Immune Correlates of Anti–PD-1 Antibody in Cancer”. New England Journal of Medicine 366. doi:10.1056/NEJMoa1200690. Lay summary – New York Times.
- Nivolumab at ClinicalTrials.gov, A service of the U.S. National Institutes of Health.
The PD-1 blocking antibody nivolumab continues to demonstrate sustained clinical activity in previously treated patients with advanced non-small cell lung cancer (NSCLC), according to updated long-term survival data from a phase I trial.
Survival rates at one year with nivolumab were 42% and reached 24% at two years, according to the median 20.3-month follow up. Additionally, the objective response rate (ORR) with nivolumab, defined as complete or partial responses by standard RECIST criteria, was 17% for patients with NSCLC. Results from the updated analysis will be presented during the 2013 World Conference on Lung Cancer on October 29.
“Lung cancer is very difficult to treat and there continues to be a high unmet medical need for these patients, especially those who have received multiple treatments,” David R. Spigel, MD, the program director of Lung Cancer Research at the Sarah Cannon Research Institute and one of the authors of the updated analysis, said in a statement.
“With nivolumab, we are investigating an approach to treating lung cancer that is designed to work with the body’s own immune system, and these are encouraging phase I results that support further investigation in larger scale trials.”
In the phase I trial, 306 patients received intravenous nivolumab at 0.1–10 mg/kg every-other-week for ≤12 cycles (4 doses/8 week cycle). In all, the trial enrolled patients with NSCLC, melanoma, renal cell carcinoma, colorectal cancer, and prostate cancer.
The long-term follow up focused specifically on the 129 patients with NSCLC. In this subgroup, patients treated with nivolumab showed encouraging clinical activity. The participants had a median age of 65 years and good performance status scores, and more than half had received three or more prior therapies. Across all doses of nivolumab, the median overall survival was 9.9 months, based on Kaplan-Meier estimates.
In a previous update of the full trial results presented at the 2013 ASCO Annual Meeting, drug-related adverse events of all grades occurred in 72% of patients and grade 3/4 events occurred in 15%. Grade 3/4 pneumonitis related to treatment with nivolumab emerged early in the trial, resulting in 3 deaths. As a result, a treatment algorithm for early detection and management was developed to prevent this serious side effect.
Nivolumab is a fully human monoclonal antibody that blocks the PD-1 receptor from binding to both of its known ligands, PD-L1 and PD-L2. This mechanism, along with early data, suggested an associated between PD-L1 expression and response to treatment.
In separate analysis presented at the 2013 World Conference on Lung Cancer, the association of tumor PD-L1 expression and clinical activity in patients with NSCLC treated with nivolumab was further explored. Of the 129 patients with NSCLC treated with nivolumab in the phase I trial, 63 with NSCLC were tested for PD-L1 expression by immunohistochemistry (29 squamous; 34 non-squamous).

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










