Home » Posts tagged 'enzalutamide'
Tag Archives: enzalutamide
Enzalutamide
4-(6-chloro-1-oxo-3-thioxo-9,9a-dihydro-1H-imidazo[1,5-a]indol-2(3H)-yl)-2-(trifluoromethyl)-benzonitrile
- Drug Name:
- Enzalutamide
- Research Code:
- MDV-3100
- Trade Name:
- Xtandi®
- MOA:
- Androgen receptor inhibitor
- Indication:
- Prostate cancer
- Status:
- Approved
- Company:
- Medivation (Originator) , Astellas
- Sales:
- $2,100.8 Million (Y2015);

$1,247.3 Million (Y2014);
$546 Million (Y2013);
$148.8 Million (Y2012); - ATC Code:
- L01
| Approval Date | Approval Type | Trade Name | Indication | Dosage Form | Strength | Company | Review Classification |
|---|---|---|---|---|---|---|---|
| 2012-08-31 | Marketing approval | Xtandi | Prostate cancer | Capsule, Liquid filled | 40 mg | Astellas | Priority |
| Approval Date | Approval Type | Trade Name | Indication | Dosage Form | Strength | Company | Review Classification |
|---|---|---|---|---|---|---|---|
| 2013-06-21 | Marketing approval | Xtandi | Prostate cancer | Capsule | 40 mg | Astellas |
| Approval Date | Approval Type | Trade Name | Indication | Dosage Form | Strength | Company | Review Classification |
|---|---|---|---|---|---|---|---|
| 2014-03-24 | Marketing approval | Xtandi | Prostate cancer | Capsule | 40 mg | Astellas |
WILL BE UPDATED………
Enzalutamide was approved by the U.S. Food and Drug Administration (FDA) on August 31, 2012, then approved by European Medicine Agency (EMA) on June 21, 2013, and approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on March 24, 2014. It was developed by Medivation and Astellas and marketed as Xtandi® by Astellas.
Enzalutamide is an androgen receptor inhibitor that decreases proliferation and induces death of prostate cancer cells. It is indicated for the treatment of patients with metastatic castration-resistant prostate cancer who have previously received docetaxel.
Xtandi® is available as capsule for oral use, containing 40 mg of free Enzalutamide, and the recommended doe is 160 mg once daily.
Prostate cancer, one of the most malignant tumors worldwide, is the second leading cause of cancer deaths among men in America . Although androgen deprivation therapy (ADT) has been proved to be effective initially, the tumor will eventually progress and develop into the lethal castration resistant prostate cancer (CRPC) . The androgen receptor (AR) is a ligand-dependent transcription factor belonging to the nuclear receptor superfamily and plays a critical role in the progression of normal prostate cells. However, overexpression of AR was found in most CRPC, which is essential for CRPC to adapt to the low levels of androgens. As AR contributes significantly to the resistance to castration, it has been recognized as an attractive target for the treatment of CRPC
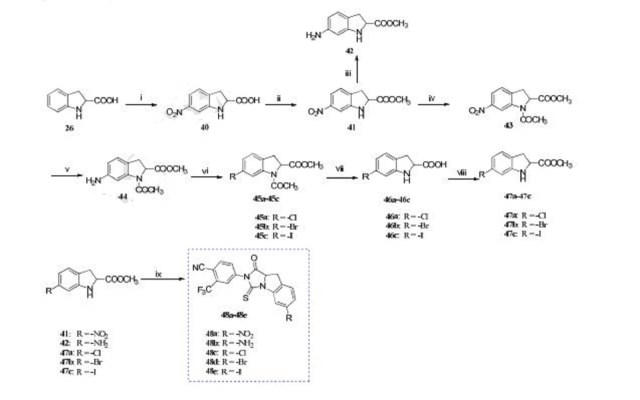
Reagents and conditions: (i) HNO3 , H2SO4 , -5 oC, 3 h; (ii) SOCl2 , MeOH, reflux, 12 h; (iii) H2 , Pd/C, MeOH, rt, 12 h; (iv) (CH3CO)2O, TEA, 50 oC, 6 h; (v) H2 , Pd/C, MeOH, rt, 12 h; (vi) acetone, HCl (6 mol/L), -10 oC, 0.5 h, NaNO2 , H2O, -10 oC, 1 h, CuCl/CuBr/KI, 0 oC, 3 h; (vii) HCl, 50 oC, 3 h; (viii) SOCl2 , MeOH, reflux, 12 h; (ix) 2, DMF, TEA, 60 oC, 1 h.
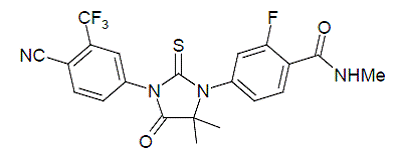
4-(6-chloro-1-oxo-3-thioxo-9,9a-dihydro-1H-imidazo[1,5-a]indol-2(3H)-yl)-2-(trifluoromethyl)-benzonitrile (48c). It was obtained as a yellow solid
m.p. 220-222 oC;
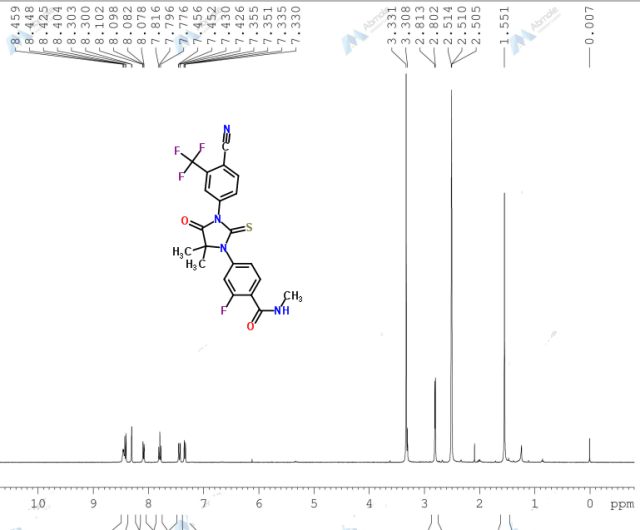
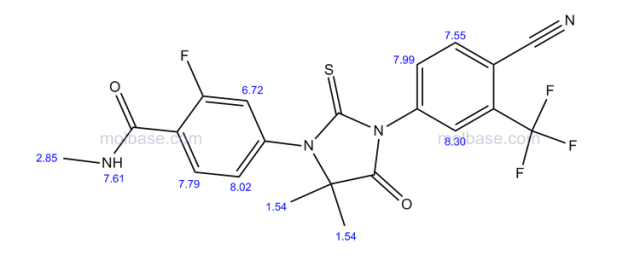
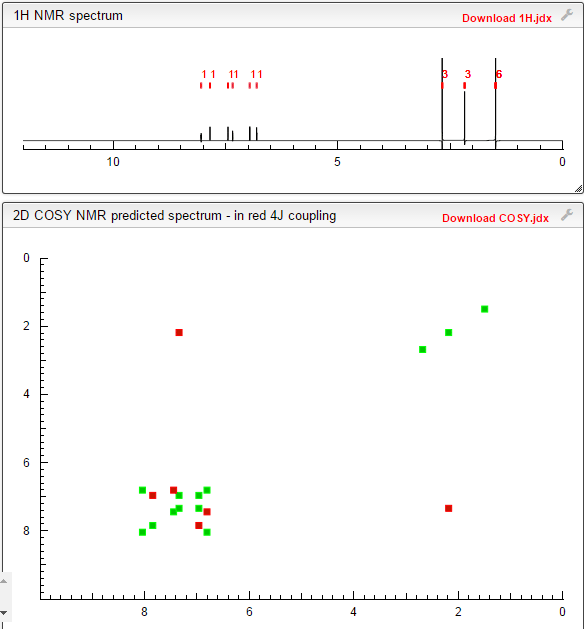
1H-NMR (300 MHz,DMSO-d6): δ 8.40 (d, J = 8.1 Hz, 1H, Ar-H), 8.19 (s, 1H, Ar-H), 8.02-7.92 (m, 2H, Ar-H), 7.49-7.46 (m, 1H, Ar-H), 7.34-7.32 (m, 1H, Ar-H), 5.56 (t, J = 9.6 Hz, 1H, -CH-), 3.58 (d, J = 9.6 Hz, 2H, -CH2-) ppm;
13C-NMR (75 MHz, DMSO-d6): δ 184.1, 172.1, 142.3, 138.5, 136.8, 134.7, 131.9, 131.5, 128.0, 126.1 (q, J = 267.9 Hz, CF3), 117.2, 115.4, 66.9, 39.9 ppm;
IR (KBr): 3094, 2232, 1763, 1607, 1499, 1270, 1136, 1052, 998, 786 cm-1;
HRMS (ESI): m/z, calculated for C18H9ClF3N3OS 408.0180 (M + H)+ , found 408.0173.
Paper
A series of indoline thiohydantoin derivatives were synthesized and evaluated in vitro.The most potent compound 48c shows comparable ability with enzalutamide in proliferation inhibition of LNCaP cells.Compound 48c has less cytotoxic to AR-negative cells compared with Enzalutamide.
The bicalutamide-resistant mechanism was clarified and overcome by compound 48c.
Abstract
A novel scaffold of indoline thiohydantoin was discovered as potent androgen receptor (AR) antagonist through rational drug designation. Several compounds showed good biological profiles in AR binding and higher selective toxicity than enzalutamide toward LNCaP cells (AR-rich) versus DU145 cells (AR-deficient). In addition, the docking studies supported the rationalization of the biological evaluation. Among these compounds, the representative compound 48c exhibited the strongest inhibitory effect on LNCaP growth and also acted as a competitive AR antagonist. Further preliminary mechanism study confirmed that 48c exerted its AR antagonistic activity through impairing AR nuclear translocation. All these results indicated that the novel scaffold compounds demonstrated AR antagonistic behaviour and promising candidates for future development were identified.
Available online 22 October 2016
- Department of Medicinal Chemistry, China Pharmaceutical University, 24 Tongjiaxiang, Nanjing 210009, PR China
- zhiyuli@cpu.edu.cn
http://www.sciencedirect.com/science/article/pii/S0223523416309114
http://dx.doi.org/10.1016/j.ejmech.2016.10.049
Paper


SYN 1
. WO2006124118A1.
2. WO2007127010A2 / US8110594B2.
3. J. Med. Chem. 2010, 53, 2779-2796.
4. WO2013087004A1 / US20140371284A1.

SYN 2
WO2011106570A1 / US20130190507A1.
2. WO2014041487A2.
3. CN103980141A.
4. CN104016924A.

SYN 3
WO2011106570A1 / US20130190507A1.

SYN 4
CN103910679A.
2. Fine Chem. Intermed. 2012, 42, 34-36.

CHINA PATENTS
////////Prostate cancer, Androgen receptor, Antagonist, Indoline thiohydantoin derivatives, indoline thiohydantoin derivatives, enzalutamide, antiproliferative agents, prostate cancer
c1c(cc(c(c1)C#N)C(F)(F)F)N2C(C3Cc4ccc(cc4N3C2=S)Cl)=O
ENZALUTAMIDE
1H NMR PREDICT AND 13 C NMR PREDICT BELOW
Synthesis pics
……………………..
PATENT
http://www.google.com/patents/WO2015063720A1?cl=en

…………………………
PAPER
J Med Chem 2010, 53(7): 2779
http://pubs.acs.org/doi/full/10.1021/jm901488g
A structure−activity relationship study was carried out on a series of thiohydantoins and their analogues 14 which led to the discovery of 92 (MDV3100) as the clinical candidate for the treatment of hormone refractory prostate cancer.
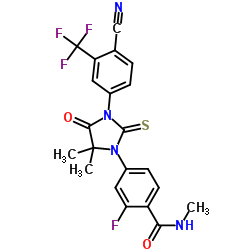
N-Methyl-4-[3-(4-cyano-3-trifluoromethylphenyl)-5,5-dimethyl-4-oxo-2-thioxoimidazolidin-1-yl]-2-fluorobenzamide, 92
REF
MEDIVATION PROSTATE THERAPEUTICS, INC.; JAIN, Rajendra, Parasmal; ANGELAUD, Remy; THOMPSON, Andrew; LAMBERSON, Carol; GREENFIELD, Scott Patent: WO2011/106570 A1, 2011 ; Location in patent: Page/Page column 46
Regents of the University of California Patent: US2007/254933 A1, 2007 ; Location in patent: Page/Page column 7 ;
WO2011/106570 A1,
J Med Chem 2010, 53(7): 2779
| WO2013067151A1 * | Nov 1, 2012 | May 10, 2013 | Medivation Prostate Therapeutics, Inc. | Treatment methods using diarylthiohydantoin derivatives |
| WO2014041487A2 * | Sep 11, 2013 | Mar 20, 2014 | Dr. Reddy’s Laboratories Limited | Enzalutamide polymorphic forms and its preparation |
| WO2014066799A2 * | Oct 25, 2013 | May 1, 2014 | Memorial Sloan-Kettering Cancer Center | Modulators of resistant androgen receptor |
| WO2014167428A3 * | Mar 5, 2014 | Feb 19, 2015 | Shilpa Medicare Limited | Amorphous 4-(3-(4-cyano-3-(trifluoromethyl)phenyl)-5,5-dimethyl-4-oxo-2-thioxoimidazolidin-1-yl)-2-fluoro-n-methylbenzamide |
| EP2536708A2 * | Feb 16, 2011 | Dec 26, 2012 | Aragon Pharmaceuticals, Inc. | Androgen receptor modulators and uses thereof |















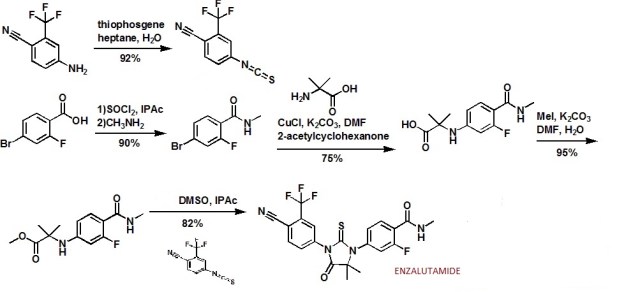
From 2-fluoro-4-bromo – benzoic acid s-1, firstly the carboxylic acid is converted to acid chloride with SOCl2 s-2, and then methylamine to give the enamine compound s-3, and s-3 bromide aminoisobutyric acid Ullmann (Goldberg) in CuCl catalyzed reaction to give the compound s-4, followed by reaction of the carboxylic acid and methyl iodide to give the corresponding methyl ester compound s-5.
Aniline compound s-6 in the sulfur phosgene primary amine is converted to the isothiocyanate s-7.
Finally, the nitrogen atom of the compound s-5 attack isothiocyanate s-7, followed by transesterification ring closure to give the final Xtandi (Enzalutamide, uh miscellaneous Lu amine). Scheme: WO2011106570A1

//////////
UK launch for Astellas’ prostate cancer drug
http://www.pharmatimes.com/Article/13-07-19/UK_launch_for_Astellas_prostate_cancer_drug.aspx
UK patients with advanced prostate cancer have been given access to a new treatment that could prolong survival following the launch of Astella’s Xtandi in the country.
Xtandi (enzalutamide) was licensed in Europe this month for the treatment of men with advanced prostate cancer whose disease has become resistant to first-line hormonal treatments and has progressed following docetaxel chemotherapy.
Enzalutamide is an androgen receptor inhibitor. The chemical name is 4-{3-[4-cyano-3-(trifluoromethyl)phenyl]-5,5dimethyl-4-oxo-2-sulfanylideneimidazolidin-1-yl}-2-fluoro-N-methylbenzamide.
The molecular weight is 464.44 and molecular formula is C21H16F4N4O2S. The structural formula is:
 |
Enzalutamide is a white crystalline non-hygroscopic solid. It is practically insoluble in water.
XTANDI is provided as liquid-filled soft gelatin capsules for oral administration. Each capsule contains 40 mg of enzalutamide as a solution in caprylocaproyl polyoxylglycerides. The inactive ingredients are caprylocaproyl polyoxylglycerides, butylated hydroxyanisole, butylated hydroxytoluene, gelatin, sorbitol sorbitan solution, glycerin, purified water, titanium dioxide, and black iron oxide.
Astellas’ Xtandi approved in EU for prostate cancer
enzalutamide
XTANDI
Regulators in European have given the green light to Astellas Pharma and Medivation’s oral prostate cancer drug Xtandi.
Specifically, the European Commission has approved Xtandi (enzalutamide) capsules for the treatment of men with metastatic castration-resistant prostate cancer whose disease has progressed on or after docetaxel therapy. The thumbs-up comes a couple of months after the European Medicines Agency’s Committee for Human Medicinal Productsissued a positive recommendation on the treatment….. READ ALL AT
http://www.pharmatimes.com/Article/13-06-24/Astellas_Xtandi_approved_in_EU_for_prostate_cancer.aspx
Enzalutamide (marketed as Xtandi and formerly known as MDV3100) is an androgen receptor antagonist drug developed by the pharmaceutical company Medivation for the treatment of metastatic castration-resistant prostate cancer. Medivation has reported up to an 89% decrease in prostate specific antigen serum levels after a month of taking the medicine. Early preclinical studies also suggest that enzalutamide inhibits breast cancer cell growth. In August of 2012, the U.S. Food and Drug Administrationapproved enzalutamide for the treatment of castration-resistant prostate cancer.
Enzalutamide was discovered by Charles Sawyers who is now at Memorial Sloan–Kettering Cancer Center and Michael Jung at UCLA.

A model of the interaction between the prostate cancer drug enzalutamide and the androgen receptor
Astellas Pharma and Medivation have announced the submission of application for marketing approval of enzalutamide in Japan for the treatment of prostate cancer
enzalutamide
read all at
![]()
Enzalutamide, 20128.31FDA-approved treatment for advanced prostate cancer drugs. Enzalutamide the androgen receptor antagonist for the treatment of castration resistant prostate cancer (castration-ressitant prostate cancer). Enzalutamide Medivation developed by the trade name Xtandi.
Enzalutamide synthesis of amino acids by the compounds 1 and 2 obtained by the Ullmann coupling with methyl esterification of 3,3 isothiocyanate 4 (can be retrieved by the corresponding aniline and phosgene prepared sulfur) cyclization Enzalutamide
Prostate cancer is a threat to men’s health killer, according to incomplete statistics, their deaths after lung cancer. Metastatic prostate cancer refers to cancer cells by the prostate and surrounding tissue spread to other tissues or organs. Metastatic prostate cancer is usually divided into two phases: hormone sensitive period (the hormone-sensitive state) and castration tolerant phase (the castration-resistant state). Testosterone (testosterone) and other male hormones can provide nourishment prostate cancer cell growth.

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....