Home » Posts tagged 'dencatistat'
Tag Archives: dencatistat
Dencatistat
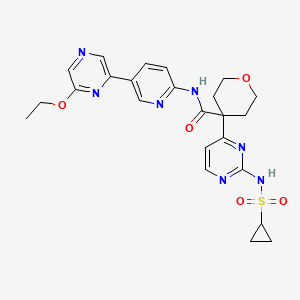
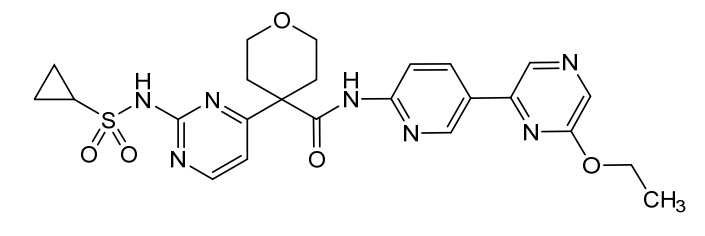
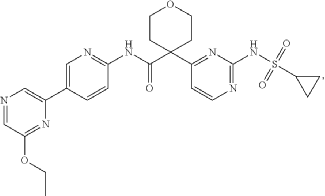
Dencatistat
CAS 2377000-84-3
MFC24H27N7O5S MW 525.6 g/mol
4-[2-(cyclopropylsulfonylamino)pyrimidin-4-yl]-N-[5-(6-ethoxypyrazin-2-yl)-2-pyridinyl]oxane-4-carboxamide
4-[2-(cyclopropanesulfonamido)pyrimidin-4-yl]-N-[5-(6-ethoxypyrazin-2-yl)pyridin-2-yl]oxane-4-carboxamide
CTP synthase 1 inhibitor, antineoplastic, STP 938, CTPS1-IN-2, QG9C9SZZ3T
Dencatistat (formerly known as STP938) is a first-in-class, orally bioavailable cancer drug designed to target specific blood cancers and solid tumours
Dencatistat is an orally bioavailable, small molecule inhibitor of cytidine triphosphate synthase 1 (CTPS1), with potential antineoplastic activity. Upon oral administration, dencatistat targets, binds to and inhibits the activity of CTPS1, thereby decreasing the production of cytidine triphosphate (CTP), an essential building block of deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). This may disrupt DNA and RNA synthesis and trigger apoptosis. CTPS1, an enzyme that catalyzes the rate-limiting step in pyrimidine synthesis, plays an important and nonredundant role in B-cell and T-cell proliferation. CTPS1 is required for rapid cell division in certain types of cancers that arise from blood cells.
Mechanism of Action
It works by inhibiting CTPS1 (Cytidine Triphosphate Synthase 1), a key enzyme that cancer cells “addicted” to for DNA synthesis.
- Targeted approach: It aims to kill cancer cells while leaving healthy cells unharmed by exploiting a “synthetic lethal” dependency in certain tumours.
- Precision medicine: It is particularly being tested in patients whose tumours lack CTPS2, a backup enzyme, which makes them highly vulnerable to dencatistat.
🏥 Clinical Status
Developed by Step Pharma, the drug is currently in several clinical trials:
- Lymphoma: Phase 1/2 trials for relapsed or refractory T-cell and B-cell lymphomas.
- Solid Tumours: Phase 1 studies for patients with solid tumours, specifically ovarian and endometrial cancers.
- Essential Thrombocythaemia: A Phase 1b trial for this blood disorder was initiated in 2025.
- Orphan Drug Status: Received FDA Orphan Drug Designation for T-cell lymphoma in May 2025.
- OriginatorStep Pharma
- ClassAnti-inflammatories; Antineoplastics; Antirheumatics; Antithrombotics; Small molecules
- Mechanism of ActionCTPS1 protein inhibitors
- Orphan Drug StatusYes – T-cell lymphoma
- Phase I/IIB-cell lymphoma; T-cell lymphoma
- Phase ISolid tumours; Thrombocytosis
- PreclinicalGraft-versus-host disease; Inflammation
- No development reportedRheumatoid arthritis
- 23 Feb 2026Step Pharma plans phase II trials for Gynaecological cancer
- 10 Feb 2026Preclinical development in Inflammation is till ongoing in France (PO) (Step Pharma pipeline, February 2026)
- 15 Oct 2025Adverse event data from a phase I/II trial in T-cell lymphoma/B-cell lymphoma released by Step Pharma
SYN
US20250177394, Compound CTPS1-IA
PAT
PAT
A. Preparation of Active Ingredient
20 Example A1 – Preparation of crude 4-(2-(cyclopropanesulfonamido)pyrimidin-4-yl)-N-(5- (6-ethoxypyrazin-2-yl)pyridin-2-yl)tetrahydro-2H-pyran-4-carboxamide
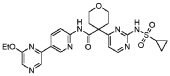
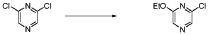
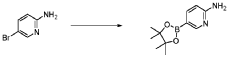
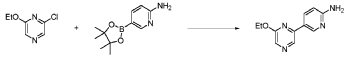
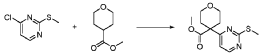
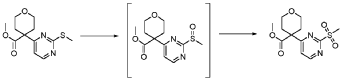
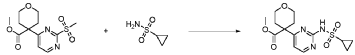
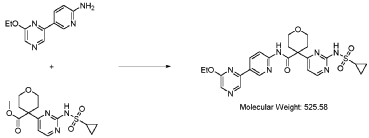
Step 4 – Preparation of crude 4-(2-(cyclopropanesulfonamido)pyrimidin-4-yl)-N-(5-(6- ethoxypyrazin-2-yl)pyridin-2-yl)tetrahydro-2H-pyran-4-carboxamide
4-(2-(cyclopropanesulfonamido)pyrimidin-4-yl)tetrahydro-2H-pyran-4-carboxylate (1.76 kg, 5.15 mol, 1.00 equiv.) and 5-(6-ethoxypyrazin-2-yl)pyridin-2-amine (1.22 kg, 5.65 mol, 1.10 equiv.) were suspended in a mixture of THF (27.1 L, 15.5 rel. vol.) and DMSO (2.63 L, 1.50 rel. vol.) and stirred until the solids were evenly dispersed. The mixture was concentrated by
STP-P3718PCT
102
distillation at atmospheric pressure and approximately 70 oC to a volume of 15 L. The temperature was adjusted to 20 ± 5 oC, potassium tert-butoxide (6.92 kg 20 wt% solution in THF, 12.3 mol, 2.40 equiv.) was added over 1 h and the reaction mixture stirred at 20 ± 5 oC for 70 minutes until completion. THF (880 mL, 0.500 rel vol.) was charged, followed by acetic acid (780 5 mL, 820 g, 13.6 mol, 2.64 equiv.) over 10 minutes, followed by methanol (4.40 L, 2.50 rel. vol.), followed by water (13.2 L, 7.50 rel. vol.) over 35 minutes. The mixture was stirred at 20 ± 5 oC for 15 minutes and then 16 h at 0 ± 5 oC. The resulting suspension was filtered and washed with water (2 × 8.80 L, 2 × 5.00 rel. vol.), followed by methanol (4.40 L, 2.50 rel. vol.) The filter cake was dried at 35 oC under a flow of nitrogen for 20 h to afford crude 4-(2-10 (cyclopropanesulfonamido)pyrimidin-4-yl)-N-(5-(6-ethoxypyrazin-2-yl)pyridin-2-yl)tetrahydro- 2H-pyran-4-carboxamide (“CTPS1-IA”).
PAT
- Aminopyrimidine derivatives as CTPS1 inhibitorsPublication Number: JP-7428692-B2Priority Date: 2018-03-23Grant Date: 2024-02-06
- Aminopyrimidine derivatives as ctps1 inhibitorsPublication Number: EP-3768674-A1Priority Date: 2018-03-23
- Aminopyrimidine derivative as a CTPS1 inhibitorPublication Number: JP-2021518436-APriority Date: 2018-03-23
- Aminopyrimidine derivatives as ctps1 inhibitorsPublication Number: EP-3768674-B1Priority Date: 2018-03-23Grant Date: 2024-01-03
- Aminopyrimidine derivatives as CTPS1 inhibitorsPublication Number: CN-111868051-APriority Date: 2018-03-23
- Aminopyrimidine derivatives as CTPS1 inhibitorsPublication Number: ES-2974445-T3Priority Date: 2018-03-23Grant Date: 2024-06-27
- CompoundsPublication Number: US-2021024507-A1Priority Date: 2018-03-23
- CompoundsPublication Number: US-2021387965-A1Priority Date: 2018-10-23
- CompoundsPublication Number: US-2023192673-A1Priority Date: 2018-06-04
- Aminopyrimidine derivatives as CTPS1 inhibitorsPublication Number: CN-111868051-BPriority Date: 2018-03-23Grant Date: 2024-04-09
- Aminopyrimidine derivatives as ctps1 inhibitorsPublication Number: WO-2019180244-A1Priority Date: 2018-03-23
- Aminopyrimidine derivatives as ctps1 inhibitorsPublication Number: WO-2019179652-A1Priority Date: 2018-03-23



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
////////dencatistat, anax lab, CTP synthase 1 inhibitor, antineoplastic, STP 938, CTPS1-IN-2, QG9C9SZZ3T

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










