Home » Posts tagged 'darlifarnib'
Tag Archives: darlifarnib
Darlifarnib
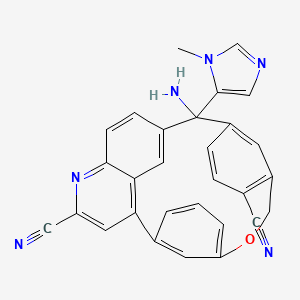
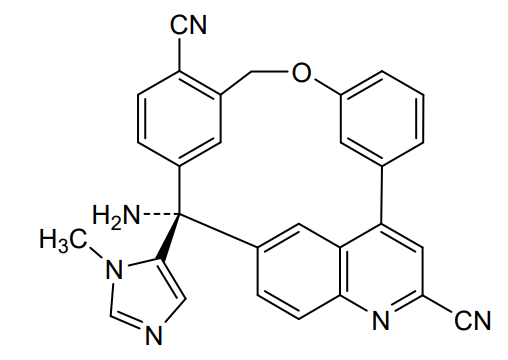
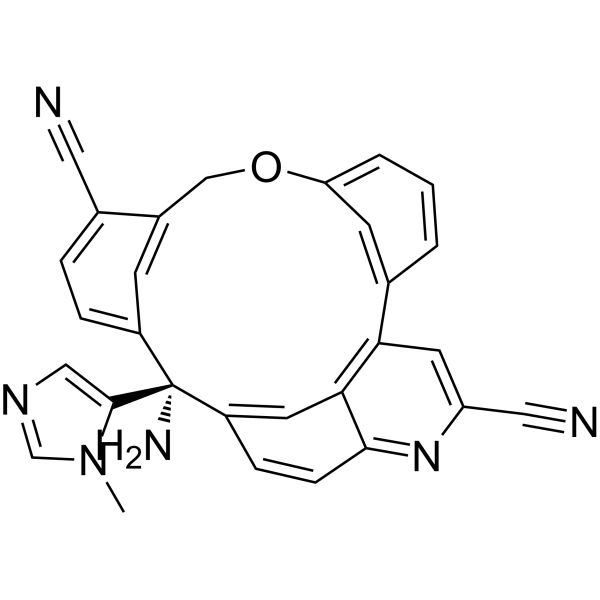
Darlifarnib
CAS 2939824-30-1
MF C29H20N6O MW 468.51
14-amino-14-(3-methylimidazol-4-yl)-7-oxa-19-azapentacyclo[13.6.2.12,6.19,13.018,22]pentacosa-1(22),2(25),3,5,9,11,13(24),15(23),16,18,20-undecaene-10,20-dicarbonitrile
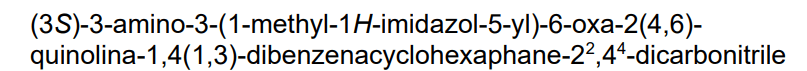
farnesyl transferase inhibitor, antineoplastic, KO-2806, KO 2806, T206317
Darlifarnib (KO-2806) is an investigational, orally active next-generation farnesyl transferase inhibitor (FTI) being developed by Kura Oncology to treat solid tumors, such as clear cell renal cell carcinoma (ccRCC). It inhibits the enzyme farnesyl transferase, blocking KRAS and mTORC1 signaling to induce tumor regression. It is often combined with other agents to overcome resistance.
Key Details About Darlifarnib
- Mechanism of Action: As a FTI, darlifarnib binds to and inhibits farnesyl transferase, which prevents the activation of RAS oncogenes and inhibits downstream mTORC1 signaling, leading to tumor cell death.
- Target Indications: Preclinical and early clinical data show potential in treating KRAS-mutant cancers, including non-small cell lung cancer (NSCLC), colorectal cancer (CRC), and clear cell renal cell carcinoma (ccRCC).
- Combination Therapy: Data from the Phase 1 FIT-001 trial (presented in April 2026) showed that combining darlifarnib with the TKI cabozantinib demonstrated robust activity in patients with pretreated, advanced ccRCC.
- Overcoming Resistance: Darlifarnib is designed to re-sensitize tumors that have become resistant to prior therapies, such as RAS inhibitors and tyrosine kinase inhibitors (TKIs).
- Status: It is an investigational drug and not yet FDA-approved.
- OriginatorKura Oncology
- ClassAntineoplastics; Small molecules
- Mechanism of ActionFarnesyltranstransferase inhibitors
- Phase IAdenocarcinoma; Colorectal cancer; Non-small cell lung cancer; Renal cell carcinoma; Solid tumours
- 12 Jan 2026Kura Oncology plans the one or more expansion cohorts of KO 2806 and cabozantinib in patients with advanced renal cell carcinoma in the first half of 2026
- 22 Oct 2025Pharmacodynamics data from a preclinical trial in Cancer presented at the AACR-NCI-EORTC International Conference on Molecular Targets and Cancer Therapeutics 2025 (AACR-NCI-EORTC-2025)
- 18 Oct 2025Adverse events and efficacy data from a phase I trial in Non-small cell lung cancer, Renal cell carcinoma, Adenocarcinoma released by Kura Oncology
PAT
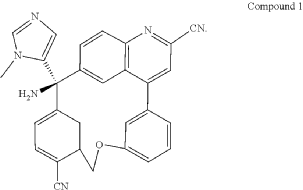
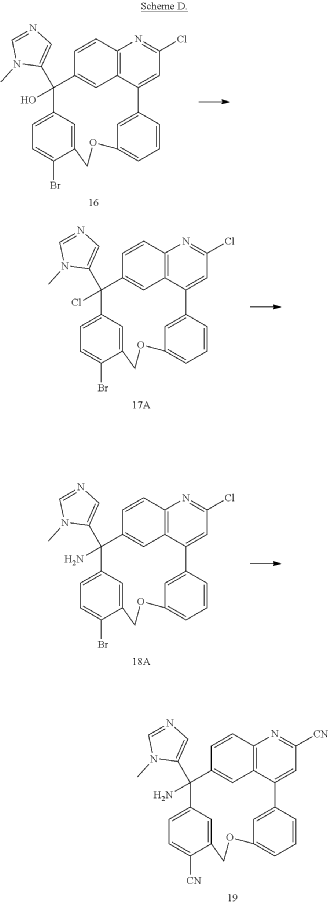
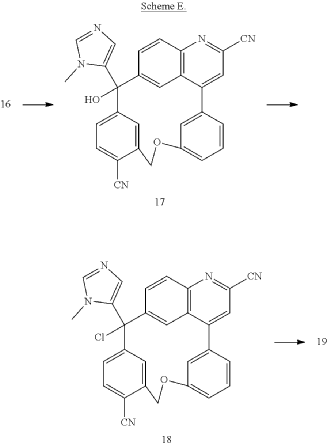
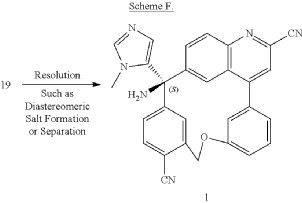
PAT
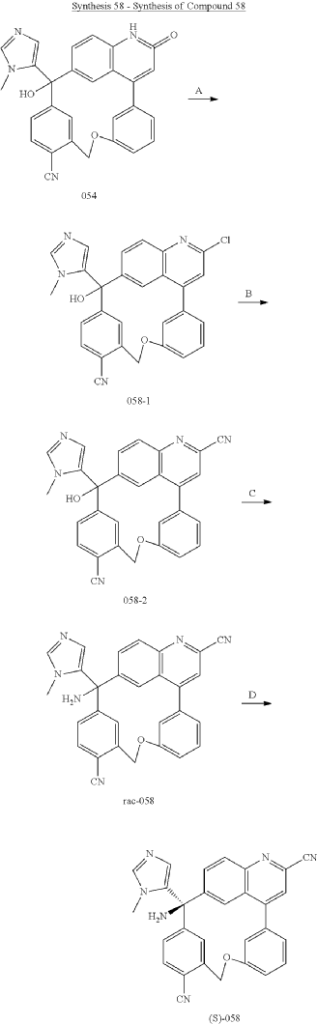
Step A: Preparation of (058-1)
Step B: Preparation of (058-2)
Step C: Preparation of (rac)-3-amino-3-(1-methyl-1H-imidazol-5-yl)-6-oxa-2(4,6)-quinolina-1,4(1,3)-dibenzenacyclohexaphane-22,44-dicarbonitrile (rac-058)
Step D: Preparation of (S)-3-amino-3-(1-methyl-1H-imidazol-5-yl)-6-oxa-2(4,6)-quinolina-1,4(1,3)-dibenzenacyclohexaphane-22,44-dicarbonitrile ((S)-058)
PAT
- Macrocyclic compounds and compositions, and methods of preparing and using the samePublication Number: US-2023322711-A1Priority Date: 2021-11-30
- Macrocyclic compounds and compositions, and methods of preparing and using the samePublication Number: US-12018011-B2Priority Date: 2021-11-30Grant Date: 2024-06-25



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
/////////////darlifarnib, ANAX LAB, farnesyl transferase inhibitor, antineoplastic, KO-2806, KO 2806, T206317

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










