Home » Posts tagged 'congenital adrenal hyperplasia'
Tag Archives: congenital adrenal hyperplasia
Atumelnant




Atumelnant
CAS 2392970-97-5
MF C33H42F3N5O3 MW 613.7 g/mol
CRN04894, NR57FH6U1N
CRINETICS PHARMA, Orphan Drug Status, Congenital adrenal hyperplasia
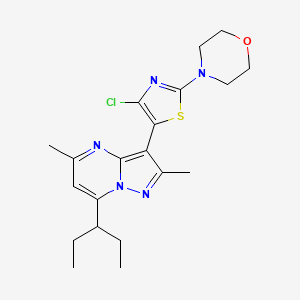
N-[(3S)-1-azabicyclo[2.2.2]octan-3-yl]-6-(2-ethoxyphenyl)-3-[(2R)-2-ethyl-4-[1-(trifluoromethyl)cyclobutanecarbonyl]piperazin-1-yl]pyridine-2-carboxamide
N-[(3S)-1-azabicyclo[2.2.2]octan-3-yl]-6-(2-ethoxyphenyl)-3-{(2R)-2-ethyl-4-[1-(trifluoromethyl) cyclobutane-1-carbonyl]piperazin-1-yl}pyridine-2-carboxamide
Adrenocorticotropic hormone receptor antagonist
- OriginatorCrinetics Pharmaceuticals
- ClassAmides; Antineoplastics; Antisecretories; Benzene derivatives; Cyclobutanes; Ethers; Fluorocarbons; Ketones; Piperazines; Pyridines; Quinuclidines; Small molecules
- Mechanism of ActionMelanocortin type 2 receptor antagonists
- Orphan Drug StatusYes – Congenital adrenal hyperplasia
- Phase IICongenital adrenal hyperplasia; Cushing syndrome
- No development reportedEctopic ACTH syndrome
- 21 Aug 2025Atumelnant receives Orphan Drug status for Congenital adrenal hyperplasia in the US
- 07 Aug 2025Crinetics pharmaceuticals plans phase II/III clinical trial for Cushing’s disease in 1H 2026
- 08 May 2025Crinetics Pharmaceuticals plans the phase III CALM-CAH trial for Congenital adrenal hyperplasia (In adults) (PO), in the second half of 2025
Atumelnant (INNTooltip International Nonproprietary Name; developmental code name CRN04894) is an investigational new drug developed by Crinetics Pharmaceuticals for the treatment of adrenocorticotropic hormone (ACTH)-dependent endocrine disorders.[1] It is a selective antagonist of the melanocortin type 2 receptor (MC2R), also known as the ACTH receptor, which is primarily expressed in the adrenal glands.[1][2] The drug is orally active.[1] Atumelnant is being evaluated to treat conditions such as congenital adrenal hyperplasia (CAH) and ACTH-dependent Cushing’s syndrome caused for example by pituitary adenomas.[3]
Atumelnant is an orally bioavailable nonpeptide antagonist of the adrenocorticotropic hormone (ACTH) receptor (ACTHR; melanocortin receptor 2; MC2R), with potential steroid hormone production inhibitory activity. Upon oral administration, atumelnant competes with ACTH for receptor binding to MC2R in the adrenal cortex and inhibits ACTH signaling. This may inhibit the synthesis and secretion of steroid hormones. MC2R, a member of the melanocortin receptor subfamily of type 1 G protein-coupled receptors, plays a key role in adrenal steroidogenesis.
PAPER
Discovery of CRN04894: A Novel Potent Selective MC2R Antagonist
Publication Name: ACS Medicinal Chemistry Letters
Publication Date: 2024-03-19, PMCID: PMC11017392, PMID: 38628803
DOI: 10.1021/acsmedchemlett.3c00514
PATENTS
- Melanocortin subtype-2 receptor antagonists and uses thereofPublication Number: IL-279152-B2Priority Date: 2018-06-05
- Melanocortin subtype-2 receptor (mc2r) antagonists and uses thereofPublication Number: US-2024300920-A1Priority Date: 2018-06-05
- Melanocortin subtype-2 receptor antagonists and uses thereofPublication Number: IL-279152-B1Priority Date: 2018-06-05
- Melanocortin subtype-2 receptor (mc2r) antagonists and uses thereofPublication Number: JP-2024009837-APriority Date: 2018-06-05
- Melanocortin subtype-2 receptor (MC2R) antagonists and uses thereofPublication Number: KR-102695210-B1Priority Date: 2018-06-05Grant Date: 2024-08-13
- Melanocortin subtype-2 receptor (mc2r) antagonists and uses thereofPublication Number: US-2024109866-A1Priority Date: 2018-06-05
- Melanocortin subtype-2 receptor (MC2R) antagonists and uses thereofPublication Number: CN-112533904-BPriority Date: 2018-06-05Grant Date: 2024-10-29
- Melanocortin subtype-2 receptor (MC2R) antagonists and uses thereofPublication Number: US-10981894-B2Priority Date: 2018-06-05Grant Date: 2021-04-20
- Melanocortin subtype-2 receptor (mc2r) antagonists and uses thereofPublication Number: US-2021002254-A1Priority Date: 2018-06-05
- Melanocortin subtype-2 receptor (mc2r) antagonists and uses thereofPublication Number: US-2021238164-A1Priority Date: 2018-06-05
- Melanocortin subtype-2 receptor (MC2R) antagonists and uses thereofPublication Number: US-11566015-B2Priority Date: 2018-06-05Grant Date: 2023-01-31
- Melanocortin subtype-2 receptor (MC2R) antagonists and their usesPublication Number: JP-7359783-B2Priority Date: 2018-06-05Grant Date: 2023-10-11
- Melanocortin subtype-2 receptor (mc2r) antagonists and uses thereofPublication Number: US-2020216415-A1Priority Date: 2018-06-05
- Melanocortin subtype-2 receptor (MC2R) antagonists and uses thereofPublication Number: US-10766877-B2Priority Date: 2018-06-05Grant Date: 2020-09-08
- Melanocortin subtype-2 receptor (MC2R) antagonists and uses thereofPublication Number: CN-112533904-APriority Date: 2018-06-05
- Melanocortin subtype-2 receptor (mc2r) antagonists and uses thereofPublication Number: EP-3802500-A1Priority Date: 2018-06-05
- Melanocortin subtype-2 receptor (MC2R) antagonists and uses thereofPublication Number: KR-20210005995-APriority Date: 2018-06-05
- Melanocortin subtype-2 receptor (MC2R) antagonists for the treatment of diseasePublication Number: CN-117043146-APriority Date: 2021-03-19
- Melanocortin subtype-2 receptor (MC2R) antagonists and uses thereofPublication Number: US-10562884-B2Priority Date: 2018-06-05Grant Date: 2020-02-18
- Melanocortin subtype-2 receptor (MC2R) antagonists and uses thereofPublication Number: US-10604507-B2Priority Date: 2018-06-05Grant Date: 2020-03-31
- Melanocortin subtype-2 receptor (mc2r) antagonists and uses thereofPublication Number: US-2019367481-A1Priority Date: 2018-06-05
- Melanocortin subtype-2 receptor (mc2r) antagonists and uses thereofPublication Number: US-2020010452-A1Priority Date: 2018-06-05
Melanocortin subtype-2 receptor (mc2r) antagonist for the treatment of diseasePublication Number: US-2022313691-A1Priority Date: 2021-03-19 - Melanocortin subtype-2 receptor (mc2r) antagonist for the treatment of diseasePublication Number: WO-2022197798-A1Priority Date: 2021-03-19
- Melanocortin subtype-2 receptor (mc2r) antagonist for the treatment of diseasePublication Number: TW-202302108-APriority Date: 2021-03-19
- Melanocortin subtype-2 receptor (mc2r) antagonist for the treatment of diseasePublication Number: AU-2022240609-A1Priority Date: 2021-03-19
- Melanocortin subtype-2 receptor (mc2r) antagonist for the treatment of diseasePublication Number: EP-4308553-A1Priority Date: 2021-03-19
- Melanocortin subtype-2 receptor (mc2r) antagonist for the treatment of acth-dependent cushing’s syndromePublication Number: WO-2024211343-A1Priority Date: 2023-04-05
- Crystalline melanocortin subtype-2 receptor (mc2r) antagonistPublication Number: TW-202430167-APriority Date: 2022-12-16
- Crystalline melanocortin subtype-2 receptor (mc2r) antagonistPublication Number: US-2024208963-A1Priority Date: 2022-12-16
- Crystalline melanocortin subtype-2 receptor (mc2r) antagonistPublication Number: WO-2024130091-A1Priority Date: 2022-12-16
- Treatment of congenital adrenal hyperplasia and polycystic ovary syndromePublication Number: WO-2023163945-A1Priority Date: 2022-02-2
SYN
PATENT
https://patentscope.wipo.int/search/en/detail.jsf?docId=US278278493&_cid=P22-MFXDN2-76849-1






Example 31: N-[(3S)-1-azabicyclo[2.2.2]octan-3-yl]-6-(2-ethoxyphenyl)-3-[(2R)-2-ethyl-4-[1-(trifluoromethyl)cyclobutanecarbonyl]piperazin-1-yl]pyridine-2-carboxamide (Compound 1-410)

Step 31-1, Preparation of 6-(2-ethoxyphenyl)-3-[(2R)-2-ethyl-4-[1-(trifluoromethyl)cyclobutanecarbonyl]piperazin-1-yl]pyridine-2-carboxylic acid
Step 31-2, Preparation of N-[(3S)-1-azabicyclo[2.2.2]octan-3-yl]-6-(2-ethoxyphenyl)-3-[(2R)-2-ethyl-4-[1-(trifluoromethyl)cyclobutanecarbonyl]piperazin-1-yl]pyridine-2-carboxamide



AS ON JUNE2025 4.45 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
References
- “Crinetics Pharmaceuticals”. AdisInsight. 21 January 2025. Retrieved 25 February 2025.
- “Atumelnant (CRN04894)”. crinetics.com. 14 August 2020.
- Varlamov EV, Gheorghiu ML, Fleseriu M (December 2024). “Pharmacological management of pituitary adenomas – what is new on the horizon?”. Expert Opinion on Pharmacotherapy. 26 (2): 119–125. doi:10.1080/14656566.2024.2446625. PMID 39718553.
| Clinical data | |
|---|---|
| Other names | CRN04894 |
| Routes of administration | Oral[1] |
| Drug class | Melanocortin MC2 receptor antagonist[1] |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2392970-97-5 |
| PubChem CID | 146361282 |
| IUPHAR/BPS | 13339 |
| ChemSpider | 129750231 |
| UNII | NR57FH6U1N |
| KEGG | D13102 |
| Chemical and physical data | |
| Formula | C33H42F3N5O3 |
| Molar mass | 613.726 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
////////Atumelnant, CRN04894, CRN 04894, NR57FH6U1N, CRINETICS PHARMA, Orphan Drug Status, Congenital adrenal hyperplasia, PHASE 3
TILDACERFONT
TILDACERFONT
| Synonyms: |
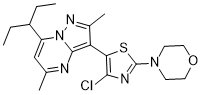
Tildacerfont 1014983-00-6 3-(4-Chloro-2-morpholin-4-yl-thiazol-5-yl)-7-(1-ethyl-propyl)-2,5-dimethyl-pyrazolo[1,5-a]pyrimidine 7-(1-ethyl-propyl)-3-(4-chloro-2-morpholin-4-yl-thiazol-5-yl)-2,5-dimethyl-pyrazolo[1,5-a]pyrimidine |
|---|---|
| MW/ MF | 420 g/mol/ C20H26ClN5OS |
- Originator Spruce Biosciences
- Class2 ring heterocyclic compounds; Morpholines; Pyrazoles; Pyrimidines; Small molecules; Thiazoles
- Mechanism of Action Corticotropin receptor antagonists
- Orphan Drug Status Yes – Congenital adrenal hyperplasia
- New Molecular Entity Yes
- Phase II Congenital adrenal hyperplasia
- 09 Jul 2020 Spruce Biosciences initiates a phase II trial in Congenital adrenal hyperplasia in USA (PO) (NCT04457336)
- 24 Sep 2019 Spruce Biosciences completes a phase II trial in Congenital adrenal hyperplasia in USA (NCT03687242)
- 19 Sep 2019 Updated safety and efficacy data from a phase II trial in Congenital adrenal hyperplasia release by Spruce Biosciences
Deuterated pyrazolo[1,5-a]pyrimidine derivatives, particularly tildacerfont (SPR-001), useful as CRF antagonists for treating congenital adrenal hyperplasia. Spruce Bioscience is developing tildacerfont under license from Lilly as an oral capsule formulation for the treatment of congenital adrenal hyperplasia; in July 2017, a phase II trial for CAH was initiated.
Corticotropin releasing factor (CRF) is a 41 amino acid peptide that is the primary physiological regulator of proopiomelanocortin (POMC) derived peptide secretion from the anterior pituitary gland. In addition to its endocrine role at the pituitary gland, immunohistochemical localization of CRF has demonstrated that the hormone has a broad extrahypothalamic distribution in the central nervous system and produces a wide spectrum of autonomic, electrophysiological and behavioral effects consistent with a neurotransmitter or neuromodulator role in the brain. There is also evidence that CRF plays a significant role in integrating the response in the immune system to physiological, psychological, and immunological stressors.
PATENT
Product case, WO2008036579 ,
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2008036579
Example 16
3-(4-Chloro-2-morpholin-4-yl-thiazol-5-yl)-7-(l-ethyl-propyl)-2,5-dimethyl- pyrazolo [ 1 ,5 -α]pyrimidine
Under a nitrogen atmosphere dissolve 3-(4-bromo-2-morpholin-4-yl-thiazol-5-yl)-7-(l-ethyl-propyl)-2,5-dimethyl-pyrazolo[l,5-α]pyrimidine (116 mg, 0.25 mmol) in THF (1.5 mL) and chill to -78 0C. Add n-butyl lithium (0.1 mL. 2.5 M in hexane, 0.25 mmol) and stir at -78 0C for 30 min. Add N-chlorosuccinimide (33.4 mg, 0.25 mmol) and stir for another 30 min, slowly warming to room temperature. After stirring overnight, quench the reaction by adding a solution of saturated ammonia chloride and extract with ethyl acetate. Wash the organic layer with brine, dry over sodium sulfate, filter, and concentrate to a residue. Purify the crude material by flash chromatography, eluting with hexanes:dichloromethane: ethyl acetate (5:5:2) to provide the title compound (54 mg). MS (APCI) m/z (35Cl) 420.6 (M+l)+; 1H NMR (400 MHz, CDCl3): 6.44 (s, IH), 3.79 (t, 4H, J=4.8 Hz), 3.63-3.56 (m, IH), 3.47 (t, 4H, J=4.8 Hz), 2.55 (s, 3H), 2.45 (s, 3H), 1.88-1.75 (m, 4H), 0.87 (t, 6H, J=7.5 Hz).
Alternate Preparation from Preparation 6:
Combine 7-(l-ethyl-propyl)-3-iodo-2,5-dimethyl-pyrazolo[l,5-α]pyrimidine, (9 g,
26.2 mmol) and 4-chloro-2-morpholino-thiazole (7.5 g, 36.7 mmol) in
dimethylformamide (90 mL) previously degassed with nitrogen. Add cesium carbonate (17.8 g, 55 mmol), copper iodide (250 mg, 1.31 mmol), triphenylphosphine (550 mg, 2.09 mmol) and palladium acetate (117 mg, 0.52 mmol). Heat the mixture to 125 0C for 16 h and then cool to 22 0C. Add water (900 mL) and extract with methyl-?-butyl ether (3 x 200 mL). Combine the organic portions and evaporate the solvent. Purify by silica gel chromatography eluting with hexanes/ethyl acetate (4/1) to afford the title compound (6.4 g, 62%). ES/MS m/z (35Cl) 420 (M+l)+.
Example 16a
3-(4-Chloro-2-morpholin-4-yl-thiazol-5-yl)-7-(l-ethyl-propyl)-2,5-dimethyl- pyrazolo[l,5-α]pyrimidine, hydrochloride
Dissolve 3-(4-chloro-2-morpholin-4-yl-thiazol-5-yl)-7-(l-ethyl-propyl)-2,5-dimethyl-pyrazolo[l,5-α]pyrimidine (1.40 g, 3.33 mmol) in acetone (10 mL) at 50 0C and cool to room temperature. Add hydrogen chloride (2 M in diethyl ether, 2.0 mL, 4.0 mmol) and stir well in a sonicator. Concentrate the solution a little and add a minimal amount of diethyl ether to crystallize the HCl salt. Cool the mixture in a refrigerator overnight. Add additional hydrogen chloride (2 M in diethyl ether, 2.0 mL, 4.0 mmol) and cool in a refrigerator. Filter the crystalline material and dry to obtain the title compound (1.15 g, 75%). ES/MS m/z (35Cl) 420 (M+l)+; 1H NMR(CDCO): 9.18 (br, IH), 6.86 (s, IH), 3.72 ( m, 4H), 3.49(m, IH), 3.39 (m, 4H), 2.48 (s, 3H), 2.38(s, 3H), 1.79 (m, 4H), 0.79 (m, 6H).
PATENT
US-20200255436
PATENT
WO2019210266
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2019210266
claiming the use of CRF-1 antagonists (eg tildacerfont).
PATENT
WO 2010039678
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2010039678
EXAMPLES
Example 1 : 7-(l-ethyl-propyl)-3-(‘2,4-dichloro-thiazol-5-yl)-2,5-dimethyl-pyrazolori ,5-alpyrimidine nthroline
Charge 7-(l-ethyl-propyl)-3-iodo-2,5-dimethyl-pyrazolo[l,5-a]pyrimidine (1.03 g, 3.00 mmoles), K3PO4 (1.95 g, 9.00 mmoles), 2,4-dichlorothiazole (0.58 g, 3.75 mmoles), 1,10 phenanthroline (0.05 g, 0.30 mmoles) and anhydrous DMAC (5 mL) to a round bottom flask equipped with a magnetic stir bar, thermal couple and N2 inlet. Degas the yellow heterogeneous reaction mixture with N2 (gas) for 30 min. and then add CuI (0.06 g, 0.30 mmoles) in one portion followed by additional 30 min. degassing with N2 (gas). Stir the reaction mixture at 120 0C for about 6 hr. Cool the reaction mixture to room temperature overnight, add toluene (10 mL) and stir for 1 hr. Purify the mixture through silica gel eluting with toluene (10ml). Extract with 1 M HCl (10 mL), water (10 mL), brine (10 mL) and concentrate under reduced pressure to give a yellow solid. Recrystallize the solid from methanol (5ml) to yield the title compound as a yellow crystalline solid. (0.78 g, 70% yield, >99% pure by LC) MS(ES) = 369 (M+ 1). 1H NMR (CDCl3)= 6.5 (IH, s); 3.6 (IH, m); 2.6 (3H, s); 2.5 (3H, s); 1.9 (4H, m); 0.9 (6H, t).
Example 2: 7-(l-ethyl-propyl)-3-(4-chloro-2-morpholin-4-yl-thiazol-5-yl)-2,5-dimethyl-pyrazolol“! ,5-aipyrimidine
Charge 7-(l-ethyl-propyl)-3-(2,4-dichloro-thiazol-5-yl)-2,5-dimethyl-pyrazolo[l,5-a]pyrimidine (0.37 g, 1.00 mmoles), K2CO3 (0.28 g, 2.00 mmoles) and anhydrous morpholine (3 mL) to a round bottom flask equipped with a magnetic stir bar and N2 inlet. Stir the yellow mixture at 100 0C for about 4 hr., during which time the reaction becomes homogeneous. Cool the reaction mixture to room temperature, add H2O (10 mL) and stir the heterogeneous reaction mixture overnight at room temperature. Collected the yellow solid by filtration, wash with H2O and allowed to air dry overnight to give the crude title compound (391mg). Recrystallize from isopropyl alcohol (3 mL) to yield the title compound as a light yellow crystalline solid (380 mg, 90.6% yield, >99% by LC). MS(ES) = 420 (M+l). 1H NMR (CDCl3)= 6.45 (IH, s); 3.81 (m, 4H); 3.62 (IH, m); 3.50 (m, 4H); 2.6 (3H, s); 2.45 (3 H, s); 1.85 (4H, m); 0.9 (6H, t).
Example 3 :
The reactions of Example 1 are run with various other catalysts, ligands, bases and solvents, which are found to have the following effects on yield of 7-(l-ethyl-propyl)-3-(2,4-dichloro-thiazol-5-yl)-2,5-dimethyl-pyrazolo[l,5-a]pyrimidine. (See Tables 1 – 4).
Table 1 : Evaluation of different li ands
(Reactions are carried out in parallel reactors with 1.2 mmol 2,4-dichlorothiazole, 1 mmol 7-(l-ethyl-propyl)-3-iodo-2,5-dimethyl-pyrazolo[l,5-a]pyrimidine, 0.5 mmol CuI, 0.5 mmol ligand and 2.1 mmol Cs2CO3 in 4 mL DMAC. The reactions are degassed under N2 for 30 min. and then heated at between 80 and
1000C overnight under N2. Percent product is measured as the percent of total area under the HPLC curve for the product peak. Longer reaction times are shown in parenthesis) Table 2: Evaluation of various solvents
(Reactions are carried out in parallel reactors with 1.2 mmol 2,4-dichlorothiazole 1 mmol 7-(l-ethyl-propyl)-3-iodo-2,5-dimethyl-pyrazolo[l,5-a]pyrimidine, 0.25 mmol CuI, 0.25 mmol 1,10-phenanthroline and 2.1 mmol Cs2CO3 in 3 mL specified solvent. The reactions are degassed under N2 for 30 minutes and then heated at 1000C overnight under N2. Percent product is measured as the percent of total area under the HPLC curve for the product peak.)
Table 3 : Evaluation of different copper sources
(Reactions are carried out in in parallel reactors with 1 mmol 2,4-dichlorothiazole 1 mmol 7-(l-ethyl-propyl)-3-iodo-2,5-dimethyl-pyrazolo[l,5-a]pyrimidine, 0.05 mmol CuX, 0.01 mmol 1,10-phenanthroline and 3 equivalents K3PO4 in 3 mL DMAC. The reactions are degassed under N2 for 30 minutes and then heated at 1000C overnight under N2. Percent product is measured as the percent of total area under the HPLC curve for the product peak.)
Table 4: Evaluation of various inorganic bases
(Reactions are carried out in in parallel reactors with 1 mmol 2,4-dichlorothiazole 1 mmol 7-(l-ethyl-propyl)-3-iodo-2,5-dimethyl-pyrazolo[l,5-a]pyrimidine, 0.1 mmol CuI, 0.1 mmol 1,10-phenanthroline and 2.1 mmol base and degassed for 30 minutes prior to the addition of 3 mL DMAC. The reactions are degassed under N2 for 10 minutes and then heated at 1000C overnight under N2. Percent product is measured as the percent of total area under the HPLC curve for the product peak.)
Example 4. Use of morpholine both as a reactant and base in 2-MeTHF as solvent.
solvent
7-(l-ethyl-propyl)-3-(2,4-dichloro-thiazol-5-yl)-2,5-dimethyl-pyrazolo[l,5-ajpyrimidine (15.2 g, 41.16 mmoles) is charged into a 250 mL 3-necked round bottomed flask, followed by addition of 2-MeTHF (61 mL, 4.0 volumes), the yellowish brown slurry is stirred at about 20 0C for 5 min. Then morpholine (19 g, 218.18 mmoles) is added over 2-5 minutes. Contents are heated to reflux and maintained at reflux for 12 hr. The slurry is cooled to 25 0C, followed by addition of 2-MeTHF (53 mL, 3.5 volumes) and water ( 38 mL 2.5 volumes). The reaction mixture is warmed to 40 0C, where upon a homogenous solution with two distinct layers formed. The layers are separated, the organic layer is filtered and concentrated to ~3 volumes at atmospheric pressure. Four volumes 2-propanol (61 mL) are added. The solution is concentrated to ~3 volumes followed by addition of 4 volumes 2-propanol (61 mL), re-concentrated to ~3 volumes, followed by addition of another 6 volumes 2-propanol (91 mL), and refluxed for 15 min. The clear solution is gradually cooled to 75 0C, seeded with 0.45 g 7-(l-ethyl-propyl)-3-(4-chloro-2-morpholin-4-yl-thiazol-5-yl)-2,5-dimethyl-pyrazolo[l,5-a]pyrimidine slurried in 2 mL 2-propanol, rinsed with an additional 2 mL 2-propanol and transferred to a crystallization flask. The slurry is cooled to between 0-5 0C, maintained for 1 hr, filtered and the product rinsed with 2-propanol (30 mL, 2 volumes). The solid is dried at 60 0C in a vacuum oven to afford 16.92 g 7-(l-ethyl-propyl)-3-(4-chloro-2-morpholin-4-yl-thiazol-5-yl)-2,5-dimethyl-pyrazolo[l,5-a]pyrimidine. Purity of product by HPLC assay is 100.00 %. XRPD and DSC data of product is consistant with reference sample. MS(ES) = 420 (M+ 1).
Example 5. Use of morpholine as both reactant and base in 2-propanol as solvent.
7-(l-ethyl-propyl)-3-(2,4-dichloro-thiazol-5-yl)-2,5-dimethyl-pyrazolo[l,5-ajpyrimidine (11.64 mmoles) is charged into a 100 mL 3 -necked round bottomed flask followed by addition of 2-propanol ( 16 mL, 3.72 volumes). The yellowish brown slurry is stirred at about 20 0C for 5 min. Then morpholine (3.3 g, 37.84 mmoles) is added over 2-5 minutes. Contents are refluxed for 6 hr. The slurry is cooled to 25 0C. 2-Propanol ( 32 mL, 7.44 volumes) and water ( 8.6 mL, 2.0 volumes) are added and the mixture warmed to 70-75 0C, filtered and concentrated to ~ 9 volumes at atmospheric pressure. The clear solution is gradually cooled to 55 0C, seeded with 0.06 g of crystalline 7-(l-ethyl-propyl)-3-(4-chloro-2-morpholin-4-yl-thiazol-5-yl)-2,5-dimethyl-pyrazolo[l,5-a]pyrimidine slurried in 0.5 mL 2-propanol, rinsed with additional 0.5 mL 2-propanol and added to crystallization flask. The slurry is cooled to 0-5 0C, maintained for 1 hr., filtered and the product rinsed with 2-propanol ( 9 mL, 2.1 volumes). Suctioned dried under vacuum at 60 0C to afford 4.6 g of dry 7-(l-ethyl-propyl)-3-(4-chloro-2-morpholin-4-yl-thiazol-5-yl)-2,5-dimethyl-pyrazolo[l,5-a]pyrimidine (88.8 % yield, purity by HPLC assay is 99.88 % ). MS(ES) = 420 (M+ 1).
Example 6: 7-(l-ethyl-propyl)-3-(2,4-dichloro-thiazol-5-yl)-2,5-dimethyl-pyrazolori ,5-alpyrimidine
7-(l-ethyl-propyl)-3-iodo-2,5-dimethyl-pyrazolo[l,5-a]pyrimidine (10 g, 29.17 mmoles), 2, 4-dichlorothiazole (5.2 g , 33.76 mmoles), cesium carbonate(19.9g, 61.07 mmoles) and 1,10-phenanthroline (1 g, 5.5 mmoles) are charged into a 250 mL 3-necked round bottomed flask, followed by 2-MeTHF (36 mL, 3.6 volumes). The reaction mixture is degassed with nitrogen and then evacuated. Cuprous chloride (0.57 g, 5.7 mmoles), DMAC (10 mL, 1 volume) and 2-MeTHF (4 mL, 0.4 volumes) are added in succession. The reaction mixture is degassed with nitrogen and then evacuated. The contents are refluxed for 20 hr. The reaction mixture is cooled to -70 0C and 2-MeTHF (100 mL, 10 volumes) is added. The contents are filtered at ~70 0C and the residual cake is washed with 2-MeTHF (80 mL, 8 volumes) at about 65-72°C. The filtrate is transferred into a separatory funnel and extracted with water. The organic layer is separated and washed with dilute HCl. The resulting organic layer is treated with Darco G60, filtered hot (600C). The filtrate is concentrated at atmospheric pressure to -2.8 volumes. 25 mL 2-propanol is added, followed by re-concentration to -2.8 volumes. An additional 25 mL 2-propanol is added, followed again by re-concentration to -2.8 volumes. Finally, 48 mL 2-propanol is added. The contents are cooled to -7 0C, maintained at -7 0C for 1 hr., filtered and rinsed with 20 mL chilled 2-propanol. Product is suction dried and then vacuum dried at 60 0C to afford 9.41 g 7-(l-ethyl-propyl)-3-(2,4-dichloro-thiazol-5-yl)-2,5-dimethyl-pyrazolo[l,5-a]pyrimidine (purity of product by HPLC assay is 95.88 %). MS(ES) = 369 (M+ 1).
Example 7. Synthesis of 7-(l-ethyl-propyl)-3-(2, 4-dichloro-thiazol-5-yl)-2,5-dimethyl-pyrazolori,5-a1pyrimidine using 1,4-Dioxane solvent and CuCl catalyst
Add dioxane (9.06X), Cs2CO3 (2.00X), 7-(l-ethyl-propyl)-3-iodo-2,5-dimethyl-pyrazolo[l,5-a]pyrimidine (1.0 equivalent), 2,4-dichlorothiazole (0.54 equivalent) to a reactor under N2. Purge the reactor with N2 three times, degas with N2 for 0.5-1 hr., and then add 1,10-phenanthroline (0.3 eq) and CuCl (0.3eq) under N2 , degassing with N2 for 0.5-1 hr. Heat the reactor to 1000C -1100C under N2 . Stir the mixture for 22-24 hr. at 100 0C -1100C. Cool to 10~20°C and add water (10V) and CH3OH (5V), stir the mixture for 1-1.5 hr. at 10~20°C. Filter the suspension, resuspend the wet cake in water, stirr for 1-1.5 hr. at 10~20°C, and filter the suspension again. Charge the wet cake to n-heptane (16V) and EtOAc (2V) under N2. Heat the reactor to 40 °C~500C under N2.
Active carbon (0. IX) is added at 40 °C~500C. The reactor is heated to 55°C~650C under N2 and stirred at 55 °C~650C for 1-1.5 hr. The suspension is filtered at 40~55°C through diatomite (0.4 X). The cake is washed with n-heptane (2.5V). The filtrate is transferred to another reactor. EtOAc (10V) is added and the the organic layer washed with 2 N HCl (10V) three times, followed by washing two times with water (10X, 10V). The organic layer is concentrated to 3-4V below 500C. The mixture is heated to 80-90 0C. The mixture is stirred at this temperature for 40-60 min. The mixture is cooled to 0~5°C, stirred for 1-1.5 hr. at 0~5°C and filtered. The cake is washed with n-heptane (IV) and vacuum dried at 45-500C for 8-10 hr. The crude product is dissolved in 2-propanol (7.5V) under N2, and re-crystallized with 2-propanol. The cake is dried in a vacuum oven at 45°C~50°C for 10-12 hr. (55-80% yield). 1H NMR56.537 (s, IH) 3.591-3.659 (m, IH, J=6.8Hz), 2.593 (s, 3H), 2.512 (s, 3H), 1.793-1.921(m, 4H), 0.885-0.903 (m, 6H).
REFERENCES
1: Zorrilla EP, Logrip ML, Koob GF. Corticotropin releasing factor: a key role in the neurobiology of addiction. Front Neuroendocrinol. 2014 Apr;35(2):234-44. doi: 10.1016/j.yfrne.2014.01.001. Epub 2014 Jan 20. Review. PubMed PMID: 24456850; PubMed Central PMCID: PMC4213066.
/////////////tildacerfont, SPR 001, Orphan Drug Status, Congenital adrenal hyperplasia, SPRUCE BIOSCIENCES, PHASE 2
CCC(CC)C1=CC(=NC2=C(C(=NN12)C)C3=C(N=C(S3)N4CCOCC4)Cl)C
ATR 101

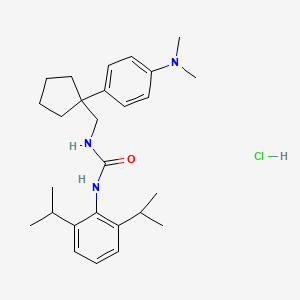
N-(2,6-bis(1-methylethyl)phenyl)-N’-((1-(4-(dimethylamino)phenyl)cyclopentyl) methyl)urea hydrochloride
N-(2,6-BIS(l-METHYLETHYL)PHENYL)-N’-((l-(4- (DIMETHYLAMINO)PHENYL)CYCLOPENTYL)METHYL)UREA
ATR-101; ATR 101; ATR101; PD132301-2; PD-132301-2; PD 132301-2; PD132301; PD-132301; PD 132301.
IUPAC/Chemical Name: 1-(2,6-diisopropylphenyl)-3-((1-(4-(dimethylamino)phenyl)cyclopentyl)methyl)urea hydrochloride
ATR-101 HCl
CAS#: 133825-81-7 (ATR-101 HCl); 133825-80-6 (ATR-101).
| Molecular Formula: | C27H40ClN3O |
|---|---|
| Molecular Weight: | 458.079 g/mol |
| The Regents Of The University Of Michigan, Atterocor, Inc. |
Millendo Therapeutics is developing ATR-101, an ACAT1 inhibitor, for treating adrenal cancers including adrenocortical cancer and congenital adrenal hyperplasia.

ATR-101, also known as PD-132301 (a free base) or PD-132301-2 (a HCl salt), is in clinical development for the treatment of adrenocortical carcinoma (ACC). ATR-101 is a selective inhibitor of ACAT1 (acyl coenzyme A:cholesterol acyltransferase). ACAT1 catalyzes cholesterol ester formation and, in the adrenals, is particularly important in creating a reservoir of substrate for steroid biosynthesis. ATR-101 is uniquely distributed to adrenal tissues and inhibition of adrenal ACAT1 by ATR-101 disrupts steroidogenesis and leads to selective apoptosis of steroid producing adrenocortical-derived cells. Similar effects have been seen in the human ACC cell line, H295R. ATR-101 has shown pre-clinical efficacy in H295R xenograft mouse models. ACC is an ultra-rare malignancy, occurring in about 2 per million population annually.
ATR-101 (Atterocor, Inc., Ann Arbor, MI, USA) is in clinical development for the treatment of adrenocortical carcinoma (ACC). ATR-101 is a selective inhibitor of ACAT1 (acyl coenzyme A:cholesterol acyltransferase). ACAT1 catalyzes cholesterol ester formation and, in the adrenals, is particularly important in creating a reservoir of substrate for steroid biosynthesis. ATR-101 is uniquely distributed to adrenal tissues and inhibition of adrenal ACAT1 by ATR-101 disrupts steroidogenesis and leads to selective apoptosis of steroid producing adrenocortical-derived cells. Similar effects have been seen in the human ACC cell line, H295R. ATR-101 has shown pre-clinical efficacy in H295R xenograft mouse models. ACC is an ultra-rare malignancy, occurring in about 2 per million population annually. ACC is frequently discovered in Stage 4 and the overall disease survival is approximately 17 months. Tumors often overproduce steroids normally produced in the adrenal cortex. Current therapies are toxic, difficult to administer, and poorly effective. Clinical trial information: NCT01898715.
Adrenocortical carcinoma (ACC) generally has poor prognosis. Existing treatments provide limited benefit for most patients with locally advanced or metastatic tumors. We investigated the mechanisms for the cytotoxicity, xenograft suppression and adrenalytic activity of ATR-101 (PD132301-02), a prospective agent for ACC treatment. Oral ATR-101 administration inhibited the establishment and impeded the growth of ACC-derived H295R cell xenografts in mice. ATR-101 induced H295R cell apoptosis in culture and in xenografts. ATR-101 caused mitochondrial hyperpolarization, reactive oxygen release and ATP depletion within hours after exposure, followed by cytochrome c release, caspase-3 activation, and membrane permeabilization. When combined with ATR-101, lipophilic free radical scavengers suppressed the reactive oxygen release, and glycolytic precursors prevented the ATP depletion, abrogating ATR-101 cytotoxicity. ATR-101 directly inhibited F1F0-ATPase activity and suppressed ATP synthesis in mitochondrial fractions. ATR-101 administration to guinea pigs caused oxidized lipofuscin accumulation in the zona fasciculata layer of the adrenal cortex, implicating reactive oxygen release in the adrenalytic effect of ATR-101. These results support the development of ATR-101 and other adrenalytic compounds for the treatment of ACC.
| Company | Millendo Therapeutics Inc. |
| Description | Selective inhibitor of sterol O-acyltransferase 1 (SOAT1; ACAT1) |
| Molecular Target | Sterol O-acyltransferase 1 (SOAT1) (ACAT1) |
PATENT
WO2013142214
https://www.google.co.in/patents/WO2013142214A1?cl=en
PATENT
One such promising agent is N-(2,6-bis( 1 -methylethyl)phenyl)-N’-(( 1 -(4-(dimethyl-amino)phenyl)cyclopentyl)methyl)urea hydrochloride (“ATR-101”). The free base form of ATR-101 has the following chemical structure:

The chemical synthesis of ATR-101 has been previously reported by Trivedi et al. (J. Med. Chem. 37: 1652-1659, 1994). This procedure, however, does not provide for ATR-101 in a form suitable for solid-dosing, particularly with regard to capsule or tablet formation, and does not provide for ATR-101 in high purity.
While significant advances have been made in this field, particularly in the context of ATR-101, there remains a substantial need for improved techniques and products for the oral administration of ATR-101 to patients in need thereof, including patients having ACC and/or other disorders or conditions such as Cushing’s syndrome and congenital adrenal hyperplasia (CAH).

EXAMPLE 1
SYNTHESIS OF SOLID DRUG FORM OF ATR-101

Step 1 : Preparation of Primary Amine 2 from the Nitrile 1

Tetrahyrofuran (THF) and Compound 1 are charged to a reactor vessel and a lithium aluminum hydride (LAH) solution in THF is added slowly. After the addition, the reaction mixture is warmed to 45°C and stirred until in-process HPLC analysis indicates that the reaction is complete. The reaction mixture is cooled to between 0 and 10°C and aqueous NaOH is added slowly while controlling the temperature to between 0 and 10°C. The mixture is then warmed to between 20 and 25°C and any inorganic salts removed by filtration. The solids are then washed with additional THF.
The filtrate is distilled under vacuum. Acetonitrile (MeCN) is added and the distillation continued to reduce the total volume. H20 is added and the solution is cooled to 20°C, and seeded if necessary. Additional water is added to the slurry and cooled to between 0 and 5°C and filtered. The crystallization vessel and filter cake is washed with MeCN and water (1 :2 mixture) and dried under vacuum between 40 to 45°C to produce Compound 2. Typical yield: 85%.
Step 2: Preparation of ATR-101 Free Base

2,6-Diisopropyl aniline hydrochloride (Compound 3) is converted to the corresponding free base by stirring in a mixture of dichloromethane (DCM) and 10% aqueous NaOH. The organic phase is separated and washed with water. The DCM solution containing the aniline free base is concentrated by distillation.
4-dimethylaminopyridine (DMAP) and DCM are charged to a separate reaction vessel. The mixture is cooled and a solution of di-tert-butyl dicarbonate (Boc20) in DCM is slowly added while the temperature is maintained between 0 and 5°C. The aniline free base solution is then slowly added to the reaction vessel. A complete conversion of aniline to the isocyanate is verified by in-process HPLC analysis.
Compound 2 and MeCN are charged to a separate vessel and this solution is cooled to between 0 and 5°C. The isocyanate intermediate solution
(prepared above) is slowly added while the temperature is maintained between 0 and 5°C, and stirred until in-process HPLC indicates that the reaction is complete.
The reaction mixture is distilled under vacuum, and isopropyl alcohol
(IP A) is added and the distillation is continued. The resulting solution is cooled and seeded, if necessary. After crystallization occurs, water is added and the mixture is cooled to between 0 and 5°C, and filtered. The crystallization vessel and filter cake is washed with isopropanol: water (1 : 1) and the product cake is dried under vacuum to yield ATR-101 as the free base. Typical yield: 89 %
Step 3 : Preparation of Solid Drug Form of ATR- 101

The ATR-101 free base is dissolved in acetone and filtered to remove particulates. Additional acetone is used to rinse the dissolution vessel and filter. Concentrated hydrochloric acid (HCl) is added while maintaining the reaction at room temperature. The resultant slurry is filtered and the cake is washed with acetone. The resulting solid is dried under vacuum between 40 and 45°C to obtain the solid drug form of ATR-101. Typical yield: 70-80 %.
EXAMPLE 2
CHARACTERIZATION OF THE SOLID DRUG FORM OF ATR-101
The solid drug form of ATR-101 was analyzed to fully characterize the material and provide proof of structure.
Elemental Analysis
An elemental (CHN) analysis was conducted, in duplicate, of the solid drug form of ATR-101. The results are summarized in Table 1 and are in agreement with the theoretical values calculated for the molecular ATR-101 drug substance formula of C27H39N3O HCl.
Table 1

Chloride Content
The solid drug form of ATR-101 is prepared as its HCl salt. To confirm the chloride content (and the stoichiometry), the hydrochloride salt was analyzed by Ion Chromatography using a validated method. The w/w% result showed 7.8% chloride present. The theoretical value for a mono hydrochloride salt is 7.7%. The experimental result conforms to the theoretical value for the mono-hydrochloride salt.
Mass Spectrometry
Mass spectrometry studies were conducted in accordance with
USP<736> using an AB Sciex API 2000 LC/MS/MS system. The samples were analyzed by electrospray ionization in positive mode. The base peak observed was 422.3 (M+H-HC1), consistent with the parent compound (see Figure 1). Two minor peaks were observed, at 301.3 and 202.3 (uncharacterized fragments). The combined data of the LC/MS and CFIN results support the molecular formula assignment of C27H39N3O and mass of 421.63 g/mol for the free base and C27H39N3O . HCl (mass of 458.09 g/mol) for the mono hydrochloride salt.
Nuclear Magnetic Resonance (NMR) – 1H NMR
The proton NMR spectrum of the solid drug form of ATR-101 was obtained using a Varian Gemini 400 MHz spectrometer and. The sample was dissolved in CD3OD. The resulting proton NMR spectrum is shown in Figure 2.
Two-Dimensional (2D) NMR
The 2D proton NMR spectrum (COSY) shown in Figure 3 confirmed some of the connectivity expected for the solid drug form of ATR-101. In particular the resonance at 1.2 ppm is strongly correlated to the resonance at 3.1. This correlation together with the splitting pattern observed for the peak at 3.1 strongly suggests an isopropyl moiety. Further, the data from these spectra show a strong correlation between each of the broad peaks at 1.6-2.2 ppm, consistent with a cycloalkyl functionality in which no heteroatoms or other non-alkyl substitution is present.
Carbon 13 NMR (13C NMR)
The 100 MHz 13C NMR spectrum of the solid drug form of ATR-101 was obtained using a Varian Gemini 400 MHz spectrometer. The sample was dissolved in CD3OD. The resulting 13C NMR spectrum is shown in Figure 4. The numbering of the carbon atoms for the analysis of the spectrum is shown below, and the interpretation is shown in Table 2. The observed signals are consistent with the structure of ATR-101.

Table 2


Fourier Transform Infrared Spectroscopy (IR)
Infrared (IR) spectroscopy was performed using the soid drug form of ATR-101. The resulting spectrum, shown in Figure 5, is consistent with the structure of ATR-101 drug substance. The major peak assignments are presented in Table_3.
Table 3

EXAMPLE 3
COMPARISON WITH PRIOR ART SYNTHESIS OF ATR-101 (BY TRIVEDI ETAL.. J. MED. CHEM. 37: 1652-1659, 1994)

ATR-101
In this experiment, 10.6 g of ATR-101 was synthesized according to the above procedure, which corresponds to the the procedure set forth in Trivedi et al., J. Med. Chem. 137: 1652-1659, 1994 (hereinafter referred to as the “Trivedi procedure”). The purity of ATR-101 as made by the Trivedi procedure was found to be 94.9%, compared to a purity of 98.3% for ATR-101 obtained by the procedure of Example 1 and as evaluated in Example 2.
Step 1 : Alkylation of p-nitrophenylacetonitrile

52
The initial alkylation reaction was run on 15.0 g scale and, according to the Trivedi procedure, should have given 15.7 g (79%) of product 52. However, several problems occurred, and the yield was much lower than expected (6.0 g, 30% yield), although the purity by 1H NMR and melting point (actual: 71-72°C, reported: 76°C) seemed good. Approximately half way through the addition of 1 ,4-bromobutane and p- nitrophenylacetonitrile to NaH, a black solid precipitated out of the purple solution causing the stirbar in the flask to skip and jump. The rate of stirring had to be monitored throughout the remainder of the addition to maintain a sluggish and inefficient mixing of the solution.
After stirring at ambient temperature overnight to ensure reaction completion, the reaction was worked-up as the procedure indicated. First, excess ether was removed using air bubbling, and the black solid was isolated by filtration. Diethyl ether was then added until all of the solids dissolved to give a clear black solution. However, upon washing the ether solution with 2N HC1, a black amorphous solid precipitated from the solution. There was no note of this black solid in the Trivedi procedure, so the work-up was continued without modification. The black solids ended up in the aqueous washes, or stuck to the seperatory funnel. The remainder of the work-up proceeded as expected, and the hot hexanes extraction of the crude solid resulted in light pink planar crystals.
The procedure was repeated with two changes thought to be responsible for the low yield: the anhydrous solvent (from the bottle) was sieve dried to remove trace water, and the stir bar was replaced with a mechanical stirrer to ensure more even mixing of the solution. The procedure was re-run on 10 g scale, which should have yielded 10.5 g of compound 52. However, despite the changes to the procedure, the resulting product and yield was nearly identical to the first run (4.5 g, 34% yield, 71-72°C melting point).
In an attempt to determine where the bulk of material ended up, the aqueous layer from this reaction was re-extracted with diethyl ether, but only resulted in trace amounts of material. The black solids that formed during the work-up were isolated by filtration, and an NMR was taken of the material. The NMR showed peaks corresponding to compound 52. Presumably, this amorphous black solid that resulted after HC1 formation is the main source of lost material, as there appeared to be several grams of it.
Ste 2: Reduction of Nitro Compound

The conversion of nitro compound 52 to the dimethyl amine 53 was done over two steps: palladium catalyzed hydrogenation of the nitro compound to give the free amine 52b, followed by imine formation & reduction to the dimethylamine 53.
An exploratory small scale reaction was run, using 1/10th of the available material (1.0 g compound 52). The reduction of the nitro compound on the 1 gram scale was very rapid, with hydrogen consumption ceasing after 3-4 hours. A crude NMR of an aliquot of the reaction mixture showed very clean amine (52b). The formaldehyde was added, as well as additional Pd/C, and the hydrogenation was continued. The hydrogen was not consumed as quickly for the imine reduction, and the reaction was still progressing when the vessel was pressurized to 55 psi and left shaking overnight (ca. 16h).
After 16 hours, the pressure in the flask had dropped to 30 psi, indicating that the hydrogenation was still progressing overnight. An aliquot NMR confirmed that the reaction had not proceeded to completion.
On large scale, the nitro reduction proceeded very smoothly, consuming hydrogen at a very rapid rate, and going to completion again within 3-4 hours. The reactor was pressurized to 55 psi and shaken overnight, as indicated in the original procedure, before more Pd/C was added, followed by formaldehyde. Hydrogen consumption was again observed to be very sluggish, so the valve to the hydrogen tank was left open to the vessel, and the reaction was shaken for 24 hours.
After 24 hours of shaking, the valve to the vessel was closed, and a drop of 5 psi was observed over 1 hour, indicating that the reaction had not progressed to completion. TLC also showed several polar products, suggesting that the reaction was only ca. 50% complete. The hydrogenation vessel was pressurized to 55 psi with hydrogen, and the valve again left open for an additional 24 hours of hydrogenation.
After 24 hours, the reaction stopped consuming hydrogen, and the vessel was purged and the contents filtered to remove the palladium catalyst. The work-up was performed similarly to the small scale, and the two reactions were combined prior to purification by column chromatography, giving 5.7g (57.5% yield) of the desired dimethylamine product 53.
Step 3 : Reduction of C ano Compound

A small scale RaNi hydrogenation was done and the test reaction went smoothly. Hydrogen consumption was rapid, and the reaction appeared complete after approximately 2 hours. The consumption of hydrogen had ceased, and TLC indicated that there was no compound 53 remaining. After filtration to remove the Raney Nickel, the reaction completion was confirmed by aliquot NMR.
The remaining material was subjected to reduction using the same conditions, and hydrogen consumption and TLC analysis again indicated reaction completion after 2 hours. The material was filtered and combined with the smaller scale reaction material. After concentration to dryness, the crude yield was found to be 5.5 g (96.5% yield), which was very close to the reported yield (99%>).
Step 4: Formation of Urea Com ound

Urea formation is a straightforward procedure, and the small scale test reaction with the amine 54 (500 mg) being combined with 1.0 equivalent of the
isocyanate in 20 parts ethyl acetate. After stirring for 16 hours, the solution was concentrated to dryness to give a white solid. Crude 1H NMR of the solid confirmed that the spectra matched the reported spectra in the Trivedi procedure.
The remaining material was carried forward to ATR-101 freebase without difficulty, and the lots of product were combined. In an effort to remove the residual ethyl acetate, the solids were dissolved in 10 mL of toluene, followed by concentration under reduced pressure. After drying on high- vacuum, ATR-101 freebase was isolated as a sticky white foam (10.6 g, 99% yield). The 1H NMR of the final product showed trace toluene even after extended drying, and the material was moved on to the HC1 salt formation.
The melting point of the solid was later taken and found to be surprisingly low (50-56°C, expected: 132-133°C). The nature of the solid (oily foam) made the determination of the melting point difficult, but it was judged to be completely melted above 60°C.
Step 5: Formation of HC1 Salt

To the ATR-101 freebase in toluene was added 37% HC1, and a gummy white solid precipitated out immediately. The solution was dried by Dean-Stark apparatus over approximately 3 hours with vigorous stirring and heating (bath temp: 160°C). After drying, the solution was cooled and the fine crystalline solid was isolated by filtration and washed with acetone and diethyl ether. The product ATR-101 was dried until a constant weight was achieved (10.6 g, 92% yield) and fully characterized.
Figure 1 is the LC/MS Mass spectrum of the solid drug form of ATR- 101.
Figure 2 is the proton NMR spectrum of the solid drug form of ATR- 101.
Figure 3 is the 2-D 1H NMR spectrum (COSY) of the solid drug form of ATR-101.
Figure 4 is the 13C NMR spectrum of of the solid drug form of ATR- 101.
Figure 5 is the FT-IR spectrum the solid drug form of ATR-101.
Paper
(J. Med. Chem. 37: 1652-1659, 1994
http://pubs.acs.org/doi/abs/10.1021/jm00037a016
| Patent ID | Date | Patent Title |
|---|---|---|
| EP0474733 | 1994-08-31 | ANTIHYPERLIPIDEMIC AND ANTIATHEROSCLEROTIC UREA COMPOUNDS. |
| WO9015048 | 1990-12-13 | ANTIHYPERLIPIDEMIC AND ANTIATHEROSCLEROTIC UREA COMPOUNDS |
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015087649 | 2015-03-26 | TREATING DISORDERS ASSOCIATED WITH ABERRANT ADRENOCORTICAL CELL BEHAVIOR |
| US2013267550 | 2013-10-10 | Compounds and Methods for Treating Aberrant Adrenocartical Cell Disorders |
| EP0858336 | 2006-12-20 | METHOD AND PHARMACEUTICAL COMPOSITION FOR REGULATING LIPID CONCENTRATION |
| US2005234124 | 2005-10-20 | Carboxyalkylether-ACAT inhibitor combinations |
| US2004072903 | 2004-04-15 | Carboxyalkylether-acat inhibitors combinations |
| US6143755 | 2000-11-07 | Pharmaceutical methods of treatment with ACAT inhibitors and HMG-CoA reductase inhibitors |
| US6124309 | 2000-09-26 | Method and pharmaceutical composition for regulating lipid concentration |
| US6093719 | 2000-07-25 | Method and pharmaceutical composition for regulating lipid concentration |
| WO9716184 | 1997-05-09 | METHOD AND PHARMACEUTICAL COMPOSITION FOR REGULATING LIPID CONCENTRATION |
| EP0474733 | 1994-08-31 | ANTIHYPERLIPIDEMIC AND ANTIATHEROSCLEROTIC UREA COMPOUNDS. |
References
1: Wolfgang GH, MacDonald JR, Vernetti LA, Pegg DG, Robertson DG. Biochemical alterations in guinea pig adrenal cortex following administration of PD 132301-2, an inhibitor of acyl-CoA:cholesterol acyltransferase. Life Sci. 1995 Feb 17;56(13):1089-93. PubMed PMID: 9001442.
2: Saxena U, Ferguson E, Newton RS. Acyl-coenzyme A:cholesterol-acyltransferase (ACAT) inhibitors modulate monocyte adhesion to aortic endothelial cells. Atherosclerosis. 1995 Jan 6;112(1):7-17. PubMed PMID: 7772069.
3: Reindel JF, Dominick MA, Bocan TM, Gough AW, McGuire EJ. Toxicologic effects of a novel acyl-CoA:cholesterol acyltransferase inhibitor in cynomolgus monkeys. Toxicol Pathol. 1994 Sep-Oct;22(5):510-8. PubMed PMID: 7899779.
4: Krause BR, Black A, Bousley R, Essenburg A, Cornicelli J, Holmes A, Homan R, Kieft K, Sekerke C, Shaw-Hes MK, et al. Divergent pharmacologic activities of PD 132301-2 and CL 277,082, urea inhibitors of acyl-CoA:cholesterol acyltransferase. J Pharmacol Exp Ther. 1993 Nov;267(2):734-43. PubMed PMID: 8246149.
5: Dominick MA, McGuire EJ, Reindel JF, Bobrowski WF, Bocan TM, Gough AW. Subacute toxicity of a novel inhibitor of acyl-CoA: cholesterol acyltransferase in beagle dogs. Fundam Appl Toxicol. 1993 Feb;20(2):217-24. PubMed PMID: 8383621.
6: Dominick MA, Bobrowski WA, MacDonald JR, Gough AW. Morphogenesis of a zone-specific adrenocortical cytotoxicity in guinea pigs administered PD 132301-2, an inhibitor of acyl-CoA:cholesterol acyltransferase. Toxicol Pathol. 1993;21(1):54-62. PubMed PMID: 8397438.
///////ATR 101, 133825-81-7, ATR-101 HCl, 133825-80-6, Millendo Therapeutics, ACAT1 inhibitor, treating adrenal cancers, adrenocortical cancer, congenital adrenal hyperplasia, Atterocor, Inc., Ann Arbor, MI, USA
O=C(NCC1(C2=CC=C(N(C)C)C=C2)CCCC1)NC3=C(C(C)C)C=CC=C3C(C)C.[H]Cl

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....