Home » Uncategorized (Page 86)
Category Archives: Uncategorized
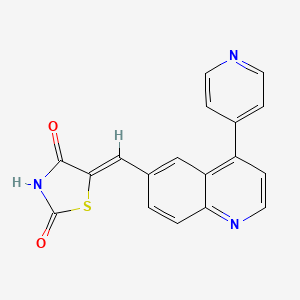
GSK 1059615
GSK1059615; 958852-01-2; GSK-1059615; UNII-07YMO87363;
- GSK 615
(5Z)-5-[(4-pyridin-4-ylquinolin-6-yl)methylidene]-1,3-thiazolidine-2,4-dione
5-[[4-(4-Pyridinyl)-6-quinolinyl]methylene]-2,4-thiazolidenedione
| C18H11N3O2S | |
| Molecular Weight: | 333.36384 |
|---|
CAS 958852-01-2
nmr……..http://file.selleckchem.com/downloads/nmr/S136001-GSK1059615-NMR-Selleck.pdf
GSK1059615 is a potent, ATP-competitive inhibitor of PI 3-kinase alpha (PI3Kα) with IC50 of 2 nM. Phosphatidylinositol-3 kinases (PI3K) are critical for malignant cellular processes including growth, proliferation, and survival. GSK1059615 is also a novel inhibitor of PI3Kβ, PI3Kδ, PI3Kγ and mTOR with IC50 of 0.6 nM, 2 nM, 5 nM and 12 nM, respectively. GSK1059615 (25 mg/kg) effectively inhibits tumor growth in xenograft mice models of BT474 or HCC1954 breast cancer cells and attenuates MAPK signaling.
GSK1059615 is a phosphoinositide 3-kinase (PI3K) inhibitor with potential antineoplastic activity. PI3K inhibitor GSK1059615 inhibits PI3K in the PI3K/AKT kinase signaling pathway, which may trigger the translocation of cytosolic Bax to the mitochondrial outer membrane and an increase in mitochondrial membrane permeability, followed by apoptosis. Bax is a member of the proapoptotic Bcl-2 family of proteins. PIK3, an enzyme often overexpressed in cancer cells, plays a crucial role in tumor cell regulation and survival.


Patent
http://www.google.com/patents/US20090306074
http://www.google.com/patents/US20090306074
Example 1 (5Z)-5-{[4-(4-pyridinyl)-6-quinolinyl]methylidene}-1,3-thiazolidine-2,4-dione
a) 4-chloro-6-ethenylquinoline
A mixture of 6-bromo-4-chloroquinoline (6.52 g, 26.88 mmol; see J. Med. Chem., 21, 268 (1978)), tributyl(vinyl)tin (8.95 g, 28.22 mmol), and tetrakistriphenylphosphine palladium (0) (0.62 g, 0.54 mmol) in 1,4-dioxane (150 mL) was refluxed for 2.0 h, cooled to room temperature, and concentrated in vacuo. The residue was purified by flash chromatography on silica gel (0-4% MeOH:CH2Cl2) to give the title compound (5.1 g) as a pale yellow solid. MS (ES)+m/e 190 [M+H]+. This material was used directly in the next step.
b) 4-chloro-6-quinolinecarbaldehyde
A mixture of 4-chloro-6-ethenylquinoline (5.1 g, 26.88 mmol), 2,6-lutidine (5.76 g, 53.75 mmol), sodium (meta) periodate (22.99 g, 107.51 mmol), and osmium tetroxide (5.48 g of a 2.5% solution in tert-butanol, 0.538 mmol) in 1,4-dioxane:H2O (350 mL of 3:1 mixture) was stirred for 3.5 h at room temperature and concentrated in vacuo. The residue was purified by flash chromatography on silica gel (CH2Cl2) to give the title compound (4.26 g, 83% for 2 steps) as a pale yellow solid. MS (ES)+ m/e 192 [M+H]+.
c) 4-(4-pyridinyl)-6-quinolinecarbaldehyde
A mixture of 4-chloro-6-quinolinecarbaldehyde (3.24 g, 16.92 mmol), 4-pyridylboronic acid (3.12 g, 25.38 mmol), tetrakistriphenylphosphine palladium (0) (0.978 g, 0.846 mmol), and 2M aqueous K2CO3 (7.02 g, 50.76 mmol, 25.4 mls of 2M solution) in DMF (100 mL) was heated at 100° C. for 3.0 h and cooled to room temperature. The mixture was filtered through celite and the celite was washed with EtOAc. The filtrate was transferred to a separatory funnel, washed with water and saturated NaCl, dried (Na2SO4), filtered and concentrated in vacuo. The residue was purified by flash chromatography on silica gel (5% MeOH:CH2Cl2) to give the title compound (2.03 g, 51%) as a tan solid. MS (ES)+ m/e 235 [M+H]+.
d) (5Z)-5-{[4-(4-pyridinyl)-6-quinolinyl]methylidene}-1,3-thiazolidine-2,4-dione
A mixture of 4-(4-pyridinyl)-6-quinolinecarbaldehyde (0.108 g, 0.463 mmol), 2,4-thiazolidinedione (0.0417 g, 0.356 mmol), piperidine (0.0303 g, 0.356 mmol), and acetic acid (0.0214 g, 0.356 mmol) in EtOH (5 mL) was heated at 150° C. for 30 minutes in a microwave oven. The reaction was cooled to room temperature and the resulting precipitate was filtered and dried in a Buchner funnel to give the title compound (0.0594 g, 50%) as a tan solid. MS (ES)+ m/e 334 [M+H]+. 1H NMR (400 MHz, DMSO-d6) □ ppm 9.08 (d, J=4.42 Hz, 1H) 8.80-8.88 (m, 2H) 8.25 (d, J=8.72 Hz, 1H) 8.00-8.07 (m, 2H) 7.98 (s, 1H) 7.65-7.68 (m, 2H) 7.63 (d, J=4.42 Hz, 1H).
……………..
http://www.google.com/patents/WO2007136940A2?cl=en
Schemes/Experimentals
Scheme I:
Conditions: a) Tributyl(vinyl)tin, Pd(PPh3)4, dioxane, reflux; b) OsO4, NaIO4, 2,6- lutidine, f-BuOH, dioxane, H2O, rt; c) heteroaryl (R) boronic acid, Pd(PPh3)4, 2 M K2CO3, DMF, 10O 0C; d) 2,4-thiazolidinedione, piperidine, AcOH, EtOH, μwave, 150 0C.
Examples:
Example 1 : (5Z)-5-ff4-(4-pyridinyl)-6-quinolinvnmethylidene}-1 ,3-thiazolidine-
2,4-dione
a) 4-chloro-6-ethenylquinoline
A mixture of 6-bromo-4-chloroquinoline (6.52 g, 26.88 mmol; see J. Med. Chem., 21_, 268 (1978) ), tributyl(vinyl)tin (8.95 g, 28.22 mmol), and tetrakistriphenylphosphine palladium (0) (0.62 g, 0.54 mmol) in 1 ,4-dioxane (150 ml.) was refluxed for 2.0 h, cooled to room temperature, and concentrated in vacuo. The residue was purified by flash chromatography on silica gel (0-4% MeOH:CH2CI2) to give the title compound (5.1 g) as a pale yellow solid. MS(ES)+ m/e 190 [M+H]+. This material was used directly in the next step.
b) 4-chloro-6-quinolinecarbaldehyde
A mixture of 4-chloro-6-ethenylquinoline (5.1 g, 26.88 mmol), 2,6-lutidine (5.76 g, 53.75 mmol), sodium (meta) periodate (22.99 g, 107.51 mmol), and osmium tetroxide (5.48 g of a 2.5% solution in tert-butanol, 0.538 mmol) in 1 ,4- dioxane:H2O (350 ml. of 3:1 mixture) was stirred for 3.5 h at room temperature and concentrated in vacuo. The residue was purified by flash chromatography on silica gel (CH2CI2) to give the title compound (4.26 g, 83% for 2 steps) as a pale yellow solid. MS(ES)+ m/e 192 [M+H]+.
c) 4-(4-pyridinyl)-6-quinolinecarbaldehyde
A mixture of 4-chloro-6-quinolinecarbaldehyde (3.24 g, 16.92 mmol), 4- pyridylboronic acid (3.12 g, 25.38 mmol), tetrakistriphenylphosphine palladium (0) (0.978 g, 0.846 mmol), and 2M aqueous K2CO3 (7.02 g, 50.76 mmol, 25.4 mis of 2M solution) in DMF (100 ml.) was heated at 1000C for 3.0 h and cooled to room temperature. The mixture was filtered through celite and the celite was washed with EtOAc. The filtrate was transferred to a separatory funnel , washed with water and saturated NaCI, dried (Na2SO4), filtered and concentrated in vacuo. The residue was purified by flash chromatography on silica gel (5% MeOHiCH2CI2) to give the title compound (2.03 g, 51%) as a tan solid. MS(ES)+ m/e 235 [M+H]+.
d) (5Z)-5-{[4-(4-pyridinyl)-6-quinolinyl]methylidene}-1 ,3-thiazolidine-2,4-dione
A mixture of 4-(4-pyridinyl)-6-quinolinecarbaldehyde (0.108 g, 0.463 mmol), 2,4-thiazolidinedione (0.0417 g, 0.356 mmol), piperidine (0.0303 g, 0.356 mmol), and acetic acid (0.0214 g, 0.356 mmol) in EtOH (5 ml.) was heated at 15O0C for 30 minutes in a microwave oven. The reaction was cooled to room temperature and the resulting precipitate was filtered and dried in a Buchner funnel to give the title compound (0.0594 g, 50%) as a tan solid. MS(ES)+ m/e 334 [M+H]+. 1 H NMR (400 MHz, DMSO-d6) D ppm 9.08 (d, J=4.42 Hz, 1 H) 8.80 – 8.88 (m, 2 H) 8.25 (d, J=8.72 Hz, 1 H) 8.00 – 8.07 (m, 2 H) 7.98 (s, 1 H) 7.65 – 7.68 (m, 2 H) 7.63 (d, J=4.42 Hz, 1 H).
| Patent | Submitted | Granted |
|---|---|---|
| THIAZOLIDINEDIONE DERIVATIVES AS PI3 KINASE INHIBITORS [US2008255115] | 2008-10-16 | |
| THIAZOLIDINEDIONE DERIVATIVES AS P13 KINASE INHIBITORS [US2009306074] | 2009-12-10 | |
| Role of PI3K p110 delta Signaling in Retroviral Infection and Replication [US2011135655] | 2011-06-09 | |
| PI3 KINASE INHIBITORS AND USES THEREOF [US2011230476] | 2011-09-22 |
Identification of druggable targets for radiation mitigation using a small interfering RNA screening assay.
Zellefrow CD,et al. Radiat Res. 2012 Sep;178(3);150-9. PMID: 22747550.
Saadia et al (2009) Phosphatidylinositol-3-kinase as a therapeutic target in melanoma. Clin.Cancer Res. 15 3029. PMID: 19383818.
Knight et al (2010) Discovery of GSK2126458, a highly potent inhibitor of PI3K and the mammalian target of rapamycin. ACS Med.Chem.Lett. 1 39.
////////GSK 1059615, GSK 615
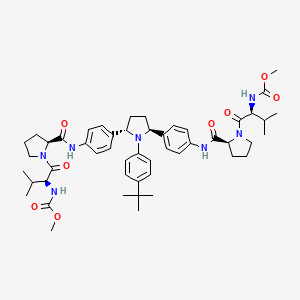
Ombitasvir

Ombitasvir; ABT-267; ABT 267; UNII-2302768XJ8; 1258226-87-7;
| C50H67N7O8 | |
| Molecular Weight: | 894.10908 g/mol |
|---|
Anti-Viral Compounds [US2010317568]
Dimethyl (2S,2′S)-1,1′-((2S,2′S)-2,2′-(4,4′-((2S,5S)-1-(4-tert-Butylphenyl)pyrrolidine-2,5-diyl)bis(4,1-phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2,1-diyl))bis(3-methyl-1-oxobutane-2,1-diyl)dicarbamate
methyl N-[(2S)-1-[(2S)-2-[[4-[(2S,5S)-1-(4-tert-butylphenyl)-5-[4-[[(2S)-1-[(2S)-2-(methoxycarbonylamino)-3-methylbutanoyl]pyrrolidine-2-carbonyl]amino]phenyl]pyrrolidin-2-yl]phenyl]carbamoyl]pyrrolidin-1-yl]-3-methyl-1-oxobutan-2-yl]carbamate
Ombitasvir is an antiviral drug for the treatment of hepatitis C virus (HCV) infection. In the United States, it is approved by theFood and Drug Administration for use in combination with paritaprevir, ritonavir and dasabuvir in the product Viekira Pak for the treatment of HCV genotype 1,[1][2] and with paritaprevir and ritonavir in the product Technivie for the treatment of HCV genotype 4.[3][4]
Ombitasvir is in phase II clinical development at AbbVie for the treatment of chronic hepatitis C infection in combination with ABT-450/ritonavir and, in combination with peginterferon alpha-2a/ribavirin (pegIFN/RBV) in treatment naïve Hepatitis C virus (HCV) genotype 1 infected patients.
Ombitasvir is part of a fixed-dose formulation with ABT-450/ritonavir that is approved in the U.S. and the E.U.
Ombitasvir acts by inhibiting the HCV protein NS5A.[5]
In 2013, breakthrough therapy designation was assigned in the U.S. for the treatment of genotype 1 hepatitis C in combination with ABT-450, ritonavir and ABT-333, with and without ribavirin.
DeGoey, DA, Discovery of ABT-267, a Pan-genotypic Inhibitor of HCV NS5A, J. Med. Chem., 2014, 57 (5), pp 2047-2057
http://pubs.acs.org/doi/full/10.1021/jm401398x
http://pubs.acs.org/doi/suppl/10.1021/jm401398x/suppl_file/jm401398x_si_001.pdf

We describe here N-phenylpyrrolidine-based inhibitors of HCV NS5A with excellent potency, metabolic stability, and pharmacokinetics. Compounds with 2S,5S stereochemistry at the pyrrolidine ring provided improved genotype 1 (GT1) potency compared to the 2R,5Ranalogues. Furthermore, the attachment of substituents at the 4-position of the central N-phenyl group resulted in compounds with improved potency. Substitution with tert-butyl, as in compound 38 (ABT-267), provided compounds with low-picomolar EC50 values and superior pharmacokinetics. It was discovered that compound 38 was a pan-genotypic HCV inhibitor, with an EC50 range of 1.7–19.3 pM against GT1a, -1b, -2a, -2b, -3a, -4a, and -5a and 366 pM against GT6a. Compound 38 decreased HCV RNA up to 3.10 log10 IU/mL during 3-day monotherapy in treatment-naive HCV GT1-infected subjects and is currently in phase 3 clinical trials in combination with an NS3 protease inhibitor with ritonavir (r) (ABT-450/r) and an NS5B non-nucleoside polymerase inhibitor (ABT-333), with and without ribavirin.
Dimethyl (2S,2′S)-1,1′-((2S,2′S)-2,2′-(4,4′-((2S,5S)-1-(4-tert-Butylphenyl)pyrrolidine-2,5-diyl)bis(4,1-phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2,1-diyl))bis(3-methyl-1-oxobutane-2,1-diyl)dicarbamate (38)…desired
and
Dimethyl (2S,2′S)-1,1′-((2S,2′S)-2,2′-(4,4′-((2R,5R)-1-(4-tert-Butylphenyl)pyrrolidine-2,5-diyl)bis(4,1-phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2,1-diyl))bis(3-methyl-1-oxobutane-2,1-diyl)dicarbamate (39)…….undesired
………..
PATENT
WO 2011156578
dimethyl (2S,2,S)-l,l ‘-((2S,2’S)-2,2′-(4,4’-((2S,5S)-l-(4-fert-butylphenyl)pyrrolidine- 2,5-diyl)bis(4, 1 -phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3- methyl- l-oxobutane-2,l-diyl)dicarbamate
hereinafter Compound IA),..http://www.google.com/patents/WO2011156578A1?cl=en
……………………………..
PATENT
US 20100317568
https://www.google.co.in/patents/US20100317568
Example 34
Dimethyl (2S,2’S)-l,r-((2S,2’S)-2,2′-(4,4′-((2S,5S)-1-(4-tert-butylphenyl)pyrrolidine-2,5-diyl)bis(4,l- phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 -oxobutane-2, 1 – diyl)dicarbamate and
Dimethyl (2S,2’S)-l,r-((2S,2’S)-2,2′-(4,4′-((2R,5R)-1-(4-ter/’-butylphenyl)pyrrolidine-2,5- diyl)bis(4, 1 -phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 – oxobutane-2, 1 -diyl)dicarbamate
Example 34A l-(4-fer?-butylphenyl)-2,5-bis(4-nitrophenyl)pyrrolidine The product from Example 1C (3.67 g, 7.51 mmol) and 4-tert-butylaniline (11.86 ml, 75 mmol) in DMF (40 ml) was stirred under nitrogen at 50 °C for 4 h. The resulting mixture was diluted into ethyl acetate, treated with IM HCl, stirred for 10 minutes and filtered to remove solids. The filtrate organic layer was washed twice with brine, dried with sodium sulfate, filtered and evaporated. The residue was purified by chromatography on silica gel eluting with ethyl acetate in hexane (5% to 30%) to give a solid. The solid was triturated in a minimal volume of 1 :9 ethyl acetate/hexane to give a light yellow solid as a mixture of trans and cis isomers (1.21 g, 36%).
Example 34B 4,4′-((2S,5S)-1-(4-tert-butylphenyl)pyrrolidine-2,5-diyl)dianiline and 4,4′-((2R,5R)-1-(4-fert- butylphenyl)pyrrolidine-2,5-diyl)dianiline To a solution of the product from Example 34A (1.1 g, 2.47 mmol) in ethanol (20 ml) and
THF (20 ml) was added PtC>2 (0.22 g, 0.97 mmol) in a 50 ml pressure bottle and stirred under 30 psi hydrogen at room temperature for 1 h. The mixture was filtered through a nylon membrane and evaporated. The residue was purified by chromatography on silica gel eluting with ethyl acetate in hexane (20% to 60%). The title compound eluted as the first of 2 stereoisomers (trans isomer, 0.51 g, 54%).
Example 34C
(2S,2’S)-tert-Butyl 2,2′-(4,4′-((2S,5S)-1-(4-fer/’-butylphenyl)pyrrolidine-2,5-diyl)bis(4,l- phenylene))bis(azanediyl)bis(oxomethylene)dipyrrolidine- 1 -carboxylate and (2S,2’S)-tert-Butyl 2,2′- (4,4′-((2R,5R)-1-(4-tert-butylphenyl)pyrrolidine-2,5-diyl)bis(4,l- phenylene))bis(azanediyl)bis(oxomethylene)dipyrrolidine-1-carboxylate To a mixture of the product from Example 34B (250 mg, 0.648 mmol), (S)-1-(tert- butoxycarbonyl)pyrrolidine-2-carboxylic acid (307 mg, 1.427 mmol) and HATU (542 mg, 1.427 mmol) in DMSO (10 ml) was added Hunig’s base (0.453 ml, 2.59 mmol). The reaction mixture was stirred at room temperature for 1 h. The mixture was partitioned with ethyl acetate and water. The organic layer was washed with brine, dried with sodium sulfate, filtered and evaporated. The residue was purified by chromatography on silica gel eluting with ethyl acetate in hexane (10% to 50%) to give the title compound (500 mg, 99%).
Example 34D
(2S,2’S)-N,N’-(4,4′-((2S,5S)-1-(4-ter/’-butylphenyl)pyrrolidine-2,5-diyl)bis(4,l- phenylene))dipyrrolidine-2-carboxamide and (2S,2’S)-N,N’-(4,4′-((2R,5R)-1-(4-tert- butylphenyl)pyrrolidine-2,5-diyl)bis(4,l-phenylene))dipyrrolidine-2-carboxamide To the product from Example 34C (498 mg, 0.638 mmol) in dichloromethane (4 ml) was added TFA (6 ml). The reaction mixture was stirred at room temperature for 1 h and concentrated in vacuo. The residue was partitioned between 3: 1 CHCl3dsopropyl alcohol and saturated aq. NaHCO3. The aqueous layer was extracted by 3: 1 CHCl3:isopropyl alcohol again. The combined organic layers were dried over
filtered and concentrated to give the title compound (345 mg, 93%).
Example 34E Dimethyl (2S,2’S)-l,r-((2S,2’S)-2,2′-(4,4′-((2S,5S)-1-(4-fert-butylphenyl)pyrrolidine-2,5-diyl)bis(4,l- phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 -oxobutane-2, 1 – diyl)dicarbamate and
Dimethyl (2S,2’S)-1, r-((2S,2’S)-2,2′-(4,4′-((2R,5R)-1-(4-fert-butylphenyl)pyrrolidine-2,5- diyl)bis(4, 1 -phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 – oxobutane-2, 1 -diyl)dicarbamate
The product from Example 34D (29.0 mg, 0.050 mmol), (S)-2-(methoxycarbonylamino)-3- methylbutanoic acid (19.27 mg, 0.110 mmol), EDAC (21.09 mg, 0.110 mmol), HOBT (16.85 mg,
0.110 mmol) and N-methylmorpholine (0.027 ml, 0.250 mmol) were combined in DMF (2 ml). The reaction mixture was stirred at room temperature for 3 h. The mixture was partitioned with ethyl acetate and water. The organic layer was washed with brine twice, dried with sodium sulfate, filtered and evaporated. The residue was purified by chromatography on silica gel eluting with ethyl acetate in hexane (50% to 80%) to give a solid. The solid was triturated with ethyl acetate/hexane to give the title compound (13 mg, 29%). 1H NMR (400 MHz, DMSO-D6) δ ppm 0.85 – 0.95 (m, 12 H) 1.11 (s, 9 H) 1.59 – 1.65 (m, 2 H) 1.79 – 2.04 (m, 8 H) 2.10 – 2.18 (m, 2 H) 2.41-2.46 (m, 2H) 3.52 (s, 6 H)
3.57 – 3.67 (m, 2 H) 3.76 – 3.86 (m, 2 H) 4.00 (t, J=7.56 Hz, 2 H) 4.39 – 4.46 (m, 2 H) 5.15 (d, J=7.00
Hz, 2 H) 6.17 (d, J=7.70 Hz, 2 H) 6.94 (d, J=8.78 Hz, 2 H) 7.13 (d, J=7.37 Hz, 4 H) 7.30 (d, J=8.20
Hz, 2 H) 7.50 (d, J=8.24 Hz, 4 H) 9.98 (s, 2 H); (ESI+) m/z 895 (M+H)+. The title compound showed an EC50 value of less than about 0.1 nM in HCV lb-Conl replicon assays in the presence of 5% FBS.
Example 35
Dimethyl (2S,2’S)-l,r-((2S,2’S)-2,2′-(4,4′-((2S,5S)-1-(4-tert-butylphenyl)pyrrolidine-2,5-diyl)bis(4,l- phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 -oxobutane-2, 1 – diyl)dicarbamate
The product from Example 34E was purified by chiral chromatography on a Chiralpak AD-H semi-prep column eluting with a 2:1 mixture of hexane:(2: l isopropyl alcohol: EtOH). The title compound was the first of the 2 diastereomers to elute. 1H NMR (400 MHz, DMSO-D6) δ ppm 0.88 (d, J=6.61 Hz, 6 H) 0.93 (d, J=6.72 Hz, 6 H) 1.11 (s, 9 H) 1.63 (d, J=5.42 Hz, 2 H) 1.80 – 2.04 (m, 8 H) 2.09 – 2.19 (m, 2 H) 2.44 – 2.47 (m, 2 H) 3.52 (s, 6 H) 3.59 – 3.66 (m, 2 H) 3.77 – 3.84 (m, 2 H) 4.02 (t, J=8.40 Hz, 2 H) 4.42 (dd, J=7.86, 4.83 Hz, 2 H) 5.14 (d, J=6.18 Hz, 2 H) 6.17 (d, J=8.67 Hz, 2 H) 6.94 (d, J=8.78 Hz, 2 H) 7.13 (d, J=8.46 Hz, 4 H) 7.31 (d, J=8.35 Hz, 2 H) 7.50 (d, J=8.35 Hz, 4 H) 9.98 (s, 2 H). The title compound showed an EC50 value of less than about 0.1 nM in HCV Ib- Conl replicon assays in the presence of 5% FBS.
Example 36 Dimethyl (2S,2’S)-1, r-((2S,2’S)-2,2′-(4,4′-((2R,5R)-1-(4-fert-butylphenyl)pyrrolidine-2,5- diyl)bis(4, 1 -phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 – oxobutane-2, 1 -diyl)dicarbamate
The product from Example 34E was purified by chiral chromatography on a Chiralpak AD-H semi-prep column eluting with a 2:1 mixture of hexane:(2: l isopropyl alcohol: EtOH). The title compound was the second of 2 diastereomers to elute. 1H NMR (400 MHz, DMSO-D6) δ ppm 0.87
(d, J=6.51 Hz, 6 H) 0.92 (d, J=6.72 Hz, 6 H) 1.11 (s, 9 H) 1.63 (d, J=5.53 Hz, 2 H) 1.82 – 2.04 (m, 8
H) 2.09-2.18 (m, 2 H) 2.41 – 2.47 (m, 2 H) 3.52 (s, 6 H) 3.58 – 3.67 (m, 2 H) 3.75 – 3.84 (m, 2 H) 4.02
(t, J=7.26 Hz, 2 H) 4.43 (dd, J=7.92, 4.88 Hz, 2 H) 5.14 (d, J=6.18 Hz, 2 H) 6.17 (d, J=8.78 Hz, 2 H) 6.94 (d, J=8.67 Hz, 2 H) 7.12 (d, J=8.46 Hz, 4 H) 7.31 (d, J=8.35 Hz, 2 H) 7.49 (d, J=8.46 Hz, 4 H)
9.98 (s, 2 H). The title compound showed an EC50 value of less than about 0.1 nM in HCV lb-Conl replicon assays in the presence of 5% FBS.
Example 37 Dimethyl (2S,2’S)-l,r-((2S,2’S)-2,2′-(4,4′-((2S,5S)-1-(4-fert-butylphenyl)pyrrolidine-2,5-diyl)bis(4,l- phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 -oxobutane-2, 1 – diyl)dicarbamate
Example 37A (S)-2,5-dioxopyrrolidin-1-yl 2-(methoxycarbonylamino)-3-methylbutanoate To a mixture of (S)-2-(methoxycarbonylamino)-3-methylbutanoic acid (19.66 g, 112 mmol) and N-hydroxysuccinimide (13.29g, 116 mmol) was added ethyl acetate (250 ml), and the mixture was cooled to 0-5 °C. Diisopropylcarbodiimide (13.88 g, 110 mmol) was added and the reaction mixture was stirred at 0-5 °C for about 1 hour. The reaction mixture was warmed to room temperature. The solids (diisopropylurea by-product) were filtered and rinsed with ethyl acetate. The filtrate was concentrated in vacuo to an oil. Isopropyl alcohol (200 ml) was added to the oil and the mixture was heated to about 50 °C to obtain a homogeneous solution. Upon cooling, crystalline solids formed. The solids were filtered and washed with isopropyl alcohol (3 x 20 ml) and dried to give the title compound as a white solid (23.2 g, 77% yield).
Example 37B
(S)- 1 -((S)-2-(methoxycarbonylamino)-3-methylbutanoyl)pyrrolidine-2-carboxylic acid To a mixture of L-proline (4.44g, 38.6 mmol), water (20 ml), acetonitrile (20 ml) and DIEA (9.5 g, 73.5 mmol) was added a solution of the product from Example 37A (1Og, 36.7 mmol) in acetonitrile (20 inL) over 10 minutes. The reaction mixture was stirred overnight at room temperature. The solution was concentrated under vacuum to remove the acetonitrile. To the resulting clear water solution was added 6N HCl (9 ml) until pH ~ 2 .The solution was transferred to a separatory funnel and 25% NaCl (10 ml) was added and the mixture was extracted with ethyl acetate (75 ml), and then again with ethyl acetate (6 x 20 ml), and the combined extracts were washed with 25% NaCl (2 x 10ml). The solvent was evaporated to give a thick oil. Heptane was added and the solvent was evaporated to give a foam, which was dried under high vacuum. Diethyl ether was added and the solvent was evaporated to give a foam, which was dried under high vacuum to give the title compound (10.67g) as a white solid.
The compound of Example 37B can also be prepreared according to the following procedure: To a flask was charged L- valine (35 g, 299 mmol), IN sodium hydroxide solution (526 ml,
526 mmol) and sodium carbonate (17.42 g, 164 mmol). The mixture was stirred for 15 min to dissolve solids and then cooled to 15 °C. Methyl chloroformate (29.6 g, 314 mmol) was added slowly to the reaction mixture. The mixture was then stirred at rt for 30 min. The mixture was cooled to 15 °C and pH adjusted to -5.0 with concentrated HCl solution. 100 inL of 2-methytetrahydrofuran (2- MeTHF) was added and the adjustment of pH continued until the pH reached ~ 2.0. 150 mL of 2- MeTHF was added and the mixture was stirred for 15 min. Layers were separated and the aqueous layer extracted with 100 mL of 2-MeTHF. The combined organic layer was dried over anhyd Na2SC^ and filtered, and Na2SC^ cake was washed with 50 mL of 2-MeTHF. The product solution was concentrated to ~ 100 mL, chased with 120 mL of IPAc twice. 250 mL of heptanes was charged slowly and then the volume of the mixture was concentrated to 300 mL. The mixture was heated to 45 °C and 160 mL of heptanes charged. The mixture was cooled to rt in 2h, stirred for 30 min, filtered and washed with 2-MeTHF/heptanes mixture (1:7, 80 inL). The wetcake was dried at 55 °C for 24 h to give 47.1 g of Moc-L- VaI-OH product as a white solid (90%).
Moc-L- VaI-OH (15O g, 856 mmol), HOBt hydrate (138 g, 899 mmol) and DMF (1500 ml) were charged to a flask. The mixture was stirred for 15 min to give a clear solution. EDC hydrochloride (172 g, 899 mmol) was charged and mixed for 20 min. The mixture was cooled to 13
°C and (L)-proline benzyl ester hydrochloride (207 g, 856 mmol) charged. Triethylamine (109 g,
1079 mmol) was then charged in 30 min. The resulting suspension was mixed at rt for 1.5 h. The reaction mixture was cooled to 15 °C and 1500 mL of 6.7% NaHCO3 charged in 1.5 h, followed by the addition of 1200 mL of water over 60 min. The mixture was stirred at rt for 30 min, filtered and washed with water/DMF mixture (1 :2, 250 mL) and then with water (1500 mL). The wetcake was dried at 55 °C for 24 h to give 282 g of product as a white solid (90%).
The resulting solids (40 g) and 5% Pd/ Alumina were charged to a Parr reactor followed by THF (160 mL). The reactor was sealed and purged with nitrogen (6 x 20 psig) followed by a hydrogen purge (6 x 30 psig). The reactor was pressurized to 30 psig with hydrogen and agitated at room temperature for approximately 15 hours. The resulting slurry was filtered through a GF/F filter and concentrated to approximately 135 g solution. Heptane was added (120 mL), and the solution was stirred until solids formed. After an addition 2 – 3 hours additional heptane was added drop-wise (240 mL), the slurry was stirred for approximately 1 hour, then filtered. The solids were dried to afford the title compound.
Example 37C
(lR,4R)-1,4-bis(4-nitrophenyl)butane-1,4-diyl dimethanesulfonate
The product from Example 32 (5.01 g, 13.39 mmol) was combined with 2- methyltetrahydrofuran (70 mL) and cooled to -5 °C, and N,N-diisopropylethylamine (6.81 g, 52.7 mmol) was added over 30 seconds. Separately, a solution of methanesulfonic anhydride (6.01 g, 34.5 mmol) in 2-methyltetrahydrofuran (30 mL) was prepared and added to the diol slurry over 3 min., maintaining the internal temperature between -15 °C and -25 °C. After mixing for 5 min at -15 °C, the cooling bath was removed and the reaction was allowed to warm slowly to 23 °C and mixed for 30 minutes. After reaction completion, the crude slurry was carried immediately into the next step.
Example 37D
(2S,5S)-1-(4-tert-butylphenyl)-2,5-bis(4-nitrophenyl)pyrrolidine
To the crude product solution from Example 37C (7.35 g, 13.39 mmol) was added 4-tert- butylaniline (13.4 g, 90 mmol) at 23 °C over 1 minute. The reaction was heated to 65 °C for 2 h. After completion, the reaction mixture was cooled to 23 °C and diluted with 2-methyltetrahydrofuran (100 mL) and 1 M HCl (150 mL). After partitioning the phases, the organic phase was treated with 1 M HCl (140 mL), 2-methyltetrahydrofuran (50 mL), and 25 wt% aq. NaCl (100 mL), and the phases were partitioned. The organic phase was washed with 25 wt% aq. NaCl (50 mL), dried over MgSO/t, filtered, and concentrated in vacuo to approximately 20 mL. Heptane (30 mL) and additional 2- methyltetrahydrofuran were added in order to induce crystallization. The slurry was concentrated further, and additional heptane (40 mL) was slowly added and the slurry was filtered, washing with 2- methyltetrahydrofuran:heptane (1:4, 20 mL). The solids were suspended in MeOH (46 mL) for 3 h, filtered, and the wet solid was washed with additional MeOH (18 mL). The solid was dried at 45 °C in a vacuum oven for 16 h to provide the title compound (3.08 g, 51% 2-step yield).
Example 37E
4,4′-((2S,5S)-1-(4-tert-butylphenyl)pyrrolidine-2,5-diyl)dianiline
To a 160 ml Parr stirrer hydrogenation vessel was added the product from Example 37D (2 g, 4.49 mmol), followed by 60 ml of THF, and Raney Nickel Grace 2800 (1 g, 50 wt% (dry basis)) under a stream of nitrogen. The reactor was assembled and purged with nitrogen (8 x 20 psig) followed by purging with hydrogen (8 x 30 psig). The reactor was then pressurized to 30 psig with hydrogen and agitation (700 rpm) began and continued for a total of 16 h at room temperature. The slurry was filtered by vacuum filtration using a GF/F Whatman glass fiber filter. Evaporation of the filtrate to afford a slurry followed by the addition heptane and filtration gave the crude title compound, which was dried and used directly in the next step.
Example 37F dimethyl (2S,2’S)-l,r-((2S,2’S)-2,2′-(4,4′-((2S,5S)-1-(4-tert-butylphenyl)pyrrolidine-2,5-diyl)bis(4, l- phenylene)bis(azanediyl)bis(oxomethylene))bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 -oxobutane-2, 1 – diy 1) die arb amate To a solution of the product from Example 37E (1.64 g, 4.25 mmol) in DMF (20 ml), the product from Example 37B (2.89 g, 10.63 mmol), and HATU (4.04 g, 10.63 mmol) in DMF (15OmL) was added triethylamine (1.07 g, 10.63 mmol), and the solution was stirred at room temperature for 90 min. To the reaction mixture was poured 20 mL of water, and the white precipitate obtained was filtered, and the solid was washed with water (3×5 mL). The solid was blow dried for Ih. The crude material was loaded on a silica gel column and eluted with a gradient starting with ethyl acetate/ heptane (3/7), and ending with pure ethyl acetate. The desired fractions were combined and solvent distilled off to give a very light yellow solid, which was dried at 45 °C in a vacuum oven with nitrogen purge for 15 h to give the title compound (2.3 g, 61% yield). 1H NMR (400 MHz, DMSO- D6) δ ppm 0.88 (d, J=6.61 Hz, 6 H) 0.93 (d, J=6.72 Hz, 6 H) 1.11 (s, 9 H) 1.63 (d, J=5.42 Hz, 2 H) 1.80 – 2.04 (m, 8 H) 2.09 – 2.19 (m, 2 H) 2.44 – 2.47 (m, 2 H) 3.52 (s, 6 H) 3.59 – 3.66 (m, 2 H) 3.77 – 3.84 (m, 2 H) 4.02 (t, J=8.40 Hz, 2 H) 4.42 (dd, J=7.86, 4.83 Hz, 2 H) 5.14 (d, J=6.18 Hz, 2 H) 6.17 (d, J=8.67 Hz, 2 H) 6.94 (d, J=8.78 Hz, 2 H) 7.13 (d, J=8.46 Hz, 4 H) 7.31 (d, J=8.35 Hz, 2 H) 7.50 (d, J=8.35 Hz, 4 H) 9.98 (s, 2 H).
Alternately, the product from example 37E (11.7 g, 85 wt%, 25.8 mmol) and the product from example 37B (15.45 g, 56.7 mmol) are suspended in EtOAc (117 mL), diisopropylethylamine (18.67 g, 144 mmol) is added and the solution is cooled to 0 °C. In a separate flask, 1-propanephosphonic acid cyclic anhydride (T3P®) (46.0 g, 50 wt% in EtOAc, 72.2 mmol) was dissolved in EtOAc (58.5 mL), and charged to an addition funnel. The T3P solution is added to the reaction mixture drop-wise over 3-4 h and stirred until the reaction is complete. The reaction is warmed to room temperature,and washed with IM HCl/7.5 wt% NaCl (100 mL), then washed with 5% NaHCO3 (100 mL), then washed with 5% NaCl solution (100 mL). The solution was concentrated to approximately 60 mL, EtOH (300 mL) was added, and the solution was concentrated to 84 g solution.
A portion of the EtOH solution of product (29 g) was heated to 40 °C, and added 134 g 40 w% EtOH in H2O. A slurry of seeds in 58 wt/wt% EtOH/H2O was added, allowed to stir at 40 °C for several hours, then cooled to 0 °C. The slurry is then filtered, and washed with 58wt/wt% EtOH/H2O. The product is dried at 40 – 60 °C under vacuum, and then rehydrated by placing a tray of water in the vacuum oven to give the title compound. The title compound showed an EC50 value of less than about 0.1 nM in HCV lb-Conl replicon assays in the presence of 5% FBS.
……………..
PATENT
http://www.google.com/patents/EP2337781A2?cl=en
Example 34
Dimethyl (2S,2’S)-l,r-((2S,2’S)-2,2′-(4,4′-((2S,5S)-1-(4-tert-butylphenyl)pyrrolidine-2,5-diyl)bis(4,l- phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 -oxobutane-2, 1 – diyl)dicarbamate and
Dimethyl (2S,2’S)-l,r-((2S,2’S)-2,2′-(4,4′-((2R,5R)-1-(4-ter/’-butylphenyl)pyrrolidine-2,5- diyl)bis(4, 1 -phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 – oxobutane-2, 1 -diyl)dicarbamate
Example 34A l-(4-fer?-butylphenyl)-2,5-bis(4-nitrophenyl)pyrrolidine The product from Example 1C (3.67 g, 7.51 mmol) and 4-tert-butylaniline (11.86 ml, 75 mmol) in DMF (40 ml) was stirred under nitrogen at 50 °C for 4 h. The resulting mixture was diluted into ethyl acetate, treated with IM HCl, stirred for 10 minutes and filtered to remove solids. The filtrate organic layer was washed twice with brine, dried with sodium sulfate, filtered and evaporated. The residue was purified by chromatography on silica gel eluting with ethyl acetate in hexane (5% to 30%) to give a solid. The solid was triturated in a minimal volume of 1 :9 ethyl acetate/hexane to give a light yellow solid as a mixture of trans and cis isomers (1.21 g, 36%).
Example 34B 4,4′-((2S,5S)-1-(4-tert-butylphenyl)pyrrolidine-2,5-diyl)dianiline and 4,4′-((2R,5R)-1-(4-fert- butylphenyl)pyrrolidine-2,5-diyl)dianiline To a solution of the product from Example 34A (1.1 g, 2.47 mmol) in ethanol (20 ml) and
THF (20 ml) was added PtC>2 (0.22 g, 0.97 mmol) in a 50 ml pressure bottle and stirred under 30 psi hydrogen at room temperature for 1 h. The mixture was filtered through a nylon membrane and evaporated. The residue was purified by chromatography on silica gel eluting with ethyl acetate in hexane (20% to 60%). The title compound eluted as the first of 2 stereoisomers (trans isomer, 0.51 g, 54%).
Example 34C
(2S,2’S)-tert-Butyl 2,2′-(4,4′-((2S,5S)-1-(4-fer/’-butylphenyl)pyrrolidine-2,5-diyl)bis(4,l- phenylene))bis(azanediyl)bis(oxomethylene)dipyrrolidine- 1 -carboxylate and (2S,2’S)-tert-Butyl 2,2′- (4,4′-((2R,5R)-1-(4-tert-butylphenyl)pyrrolidine-2,5-diyl)bis(4,l- phenylene))bis(azanediyl)bis(oxomethylene)dipyrrolidine-1-carboxylate To a mixture of the product from Example 34B (250 mg, 0.648 mmol), (S)-1-(tert- butoxycarbonyl)pyrrolidine-2-carboxylic acid (307 mg, 1.427 mmol) and HATU (542 mg, 1.427 mmol) in DMSO (10 ml) was added Hunig’s base (0.453 ml, 2.59 mmol). The reaction mixture was stirred at room temperature for 1 h. The mixture was partitioned with ethyl acetate and water. The organic layer was washed with brine, dried with sodium sulfate, filtered and evaporated. The residue was purified by chromatography on silica gel eluting with ethyl acetate in hexane (10% to 50%) to give the title compound (500 mg, 99%).
Example 34D
(2S,2’S)-N,N’-(4,4′-((2S,5S)-1-(4-ter/’-butylphenyl)pyrrolidine-2,5-diyl)bis(4,l- phenylene))dipyrrolidine-2-carboxamide and (2S,2’S)-N,N’-(4,4′-((2R,5R)-1-(4-tert- butylphenyl)pyrrolidine-2,5-diyl)bis(4,l-phenylene))dipyrrolidine-2-carboxamide To the product from Example 34C (498 mg, 0.638 mmol) in dichloromethane (4 ml) was added TFA (6 ml). The reaction mixture was stirred at room temperature for 1 h and concentrated in vacuo. The residue was partitioned between 3: 1 CHCl3dsopropyl alcohol and saturated aq. NaHCO3. The aqueous layer was extracted by 3: 1 CHCl3:isopropyl alcohol again. The combined organic layers were dried over
filtered and concentrated to give the title compound (345 mg, 93%).
Example 34E Dimethyl (2S,2’S)-l,r-((2S,2’S)-2,2′-(4,4′-((2S,5S)-1-(4-fert-butylphenyl)pyrrolidine-2,5-diyl)bis(4,l- phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 -oxobutane-2, 1 – diyl)dicarbamate and
Dimethyl (2S,2’S)-1, r-((2S,2’S)-2,2′-(4,4′-((2R,5R)-1-(4-fert-butylphenyl)pyrrolidine-2,5- diyl)bis(4, 1 -phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 – oxobutane-2, 1 -diyl)dicarbamate
The product from Example 34D (29.0 mg, 0.050 mmol), (S)-2-(methoxycarbonylamino)-3- methylbutanoic acid (19.27 mg, 0.110 mmol), EDAC (21.09 mg, 0.110 mmol), HOBT (16.85 mg,
0.110 mmol) and N-methylmorpholine (0.027 ml, 0.250 mmol) were combined in DMF (2 ml). The reaction mixture was stirred at room temperature for 3 h. The mixture was partitioned with ethyl acetate and water. The organic layer was washed with brine twice, dried with sodium sulfate, filtered and evaporated. The residue was purified by chromatography on silica gel eluting with ethyl acetate in hexane (50% to 80%) to give a solid. The solid was triturated with ethyl acetate/hexane to give the title compound (13 mg, 29%). 1H NMR (400 MHz, DMSO-D6) δ ppm 0.85 – 0.95 (m, 12 H) 1.11 (s, 9 H) 1.59 – 1.65 (m, 2 H) 1.79 – 2.04 (m, 8 H) 2.10 – 2.18 (m, 2 H) 2.41-2.46 (m, 2H) 3.52 (s, 6 H)
3.57 – 3.67 (m, 2 H) 3.76 – 3.86 (m, 2 H) 4.00 (t, J=7.56 Hz, 2 H) 4.39 – 4.46 (m, 2 H) 5.15 (d, J=7.00
Hz, 2 H) 6.17 (d, J=7.70 Hz, 2 H) 6.94 (d, J=8.78 Hz, 2 H) 7.13 (d, J=7.37 Hz, 4 H) 7.30 (d, J=8.20
Hz, 2 H) 7.50 (d, J=8.24 Hz, 4 H) 9.98 (s, 2 H); (ESI+) m/z 895 (M+H)+. The title compound showed an EC50 value of less than about 0.1 nM in HCV lb-Conl replicon assays in the presence of 5% FBS.
Example 35
Dimethyl (2S,2’S)-l,r-((2S,2’S)-2,2′-(4,4′-((2S,5S)-1-(4-tert-butylphenyl)pyrrolidine-2,5-diyl)bis(4,l- phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 -oxobutane-2, 1 – diyl)dicarbamate
The product from Example 34E was purified by chiral chromatography on a Chiralpak AD-H semi-prep column eluting with a 2:1 mixture of hexane:(2: l isopropyl alcohol: EtOH). The title compound was the first of the 2 diastereomers to elute. 1H NMR (400 MHz, DMSO-D6) δ ppm 0.88 (d, J=6.61 Hz, 6 H) 0.93 (d, J=6.72 Hz, 6 H) 1.11 (s, 9 H) 1.63 (d, J=5.42 Hz, 2 H) 1.80 – 2.04 (m, 8 H) 2.09 – 2.19 (m, 2 H) 2.44 – 2.47 (m, 2 H) 3.52 (s, 6 H) 3.59 – 3.66 (m, 2 H) 3.77 – 3.84 (m, 2 H) 4.02 (t, J=8.40 Hz, 2 H) 4.42 (dd, J=7.86, 4.83 Hz, 2 H) 5.14 (d, J=6.18 Hz, 2 H) 6.17 (d, J=8.67 Hz, 2 H) 6.94 (d, J=8.78 Hz, 2 H) 7.13 (d, J=8.46 Hz, 4 H) 7.31 (d, J=8.35 Hz, 2 H) 7.50 (d, J=8.35 Hz, 4 H) 9.98 (s, 2 H). The title compound showed an EC50 value of less than about 0.1 nM in HCV Ib- Conl replicon assays in the presence of 5% FBS.
Example 36 Dimethyl (2S,2’S)-1, r-((2S,2’S)-2,2′-(4,4′-((2R,5R)-1-(4-fert-butylphenyl)pyrrolidine-2,5- diyl)bis(4, 1 -phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 – oxobutane-2, 1 -diyl)dicarbamate
The product from Example 34E was purified by chiral chromatography on a Chiralpak AD-H semi-prep column eluting with a 2:1 mixture of hexane:(2: l isopropyl alcohol: EtOH). The title compound was the second of 2 diastereomers to elute. 1H NMR (400 MHz, DMSO-D6) δ ppm 0.87
(d, J=6.51 Hz, 6 H) 0.92 (d, J=6.72 Hz, 6 H) 1.11 (s, 9 H) 1.63 (d, J=5.53 Hz, 2 H) 1.82 – 2.04 (m, 8
H) 2.09-2.18 (m, 2 H) 2.41 – 2.47 (m, 2 H) 3.52 (s, 6 H) 3.58 – 3.67 (m, 2 H) 3.75 – 3.84 (m, 2 H) 4.02
(t, J=7.26 Hz, 2 H) 4.43 (dd, J=7.92, 4.88 Hz, 2 H) 5.14 (d, J=6.18 Hz, 2 H) 6.17 (d, J=8.78 Hz, 2 H) 6.94 (d, J=8.67 Hz, 2 H) 7.12 (d, J=8.46 Hz, 4 H) 7.31 (d, J=8.35 Hz, 2 H) 7.49 (d, J=8.46 Hz, 4 H)
9.98 (s, 2 H). The title compound showed an EC50 value of less than about 0.1 nM in HCV lb-Conl replicon assays in the presence of 5% FBS.
Example 37 Dimethyl (2S,2’S)-l,r-((2S,2’S)-2,2′-(4,4′-((2S,5S)-1-(4-fert-butylphenyl)pyrrolidine-2,5-diyl)bis(4,l- phenylene))bis(azanediyl)bis(oxomethylene)bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 -oxobutane-2, 1 – diyl)dicarbamate
Example 37A (S)-2,5-dioxopyrrolidin-1-yl 2-(methoxycarbonylamino)-3-methylbutanoate To a mixture of (S)-2-(methoxycarbonylamino)-3-methylbutanoic acid (19.66 g, 112 mmol) and N-hydroxysuccinimide (13.29g, 116 mmol) was added ethyl acetate (250 ml), and the mixture was cooled to 0-5 °C. Diisopropylcarbodiimide (13.88 g, 110 mmol) was added and the reaction mixture was stirred at 0-5 °C for about 1 hour. The reaction mixture was warmed to room temperature. The solids (diisopropylurea by-product) were filtered and rinsed with ethyl acetate. The filtrate was concentrated in vacuo to an oil. Isopropyl alcohol (200 ml) was added to the oil and the mixture was heated to about 50 °C to obtain a homogeneous solution. Upon cooling, crystalline solids formed. The solids were filtered and washed with isopropyl alcohol (3 x 20 ml) and dried to give the title compound as a white solid (23.2 g, 77% yield).
Example 37B
(S)- 1 -((S)-2-(methoxycarbonylamino)-3-methylbutanoyl)pyrrolidine-2-carboxylic acid To a mixture of L-proline (4.44g, 38.6 mmol), water (20 ml), acetonitrile (20 ml) and DIEA (9.5 g, 73.5 mmol) was added a solution of the product from Example 37A (1Og, 36.7 mmol) in acetonitrile (20 inL) over 10 minutes. The reaction mixture was stirred overnight at room temperature. The solution was concentrated under vacuum to remove the acetonitrile. To the resulting clear water solution was added 6N HCl (9 ml) until pH ~ 2 .The solution was transferred to a separatory funnel and 25% NaCl (10 ml) was added and the mixture was extracted with ethyl acetate (75 ml), and then again with ethyl acetate (6 x 20 ml), and the combined extracts were washed with 25% NaCl (2 x 10ml). The solvent was evaporated to give a thick oil. Heptane was added and the solvent was evaporated to give a foam, which was dried under high vacuum. Diethyl ether was added and the solvent was evaporated to give a foam, which was dried under high vacuum to give the title compound (10.67g) as a white solid.
The compound of Example 37B can also be prepreared according to the following procedure: To a flask was charged L- valine (35 g, 299 mmol), IN sodium hydroxide solution (526 ml,
526 mmol) and sodium carbonate (17.42 g, 164 mmol). The mixture was stirred for 15 min to dissolve solids and then cooled to 15 °C. Methyl chloroformate (29.6 g, 314 mmol) was added slowly to the reaction mixture. The mixture was then stirred at rt for 30 min. The mixture was cooled to 15 °C and pH adjusted to -5.0 with concentrated HCl solution. 100 inL of 2-methytetrahydrofuran (2- MeTHF) was added and the adjustment of pH continued until the pH reached ~ 2.0. 150 mL of 2- MeTHF was added and the mixture was stirred for 15 min. Layers were separated and the aqueous layer extracted with 100 mL of 2-MeTHF. The combined organic layer was dried over anhyd Na2SC^ and filtered, and Na2SC^ cake was washed with 50 mL of 2-MeTHF. The product solution was concentrated to ~ 100 mL, chased with 120 mL of IPAc twice. 250 mL of heptanes was charged slowly and then the volume of the mixture was concentrated to 300 mL. The mixture was heated to 45 °C and 160 mL of heptanes charged. The mixture was cooled to rt in 2h, stirred for 30 min, filtered and washed with 2-MeTHF/heptanes mixture (1:7, 80 inL). The wetcake was dried at 55 °C for 24 h to give 47.1 g of Moc-L- VaI-OH product as a white solid (90%).
Moc-L- VaI-OH (15O g, 856 mmol), HOBt hydrate (138 g, 899 mmol) and DMF (1500 ml) were charged to a flask. The mixture was stirred for 15 min to give a clear solution. EDC hydrochloride (172 g, 899 mmol) was charged and mixed for 20 min. The mixture was cooled to 13
°C and (L)-proline benzyl ester hydrochloride (207 g, 856 mmol) charged. Triethylamine (109 g,
1079 mmol) was then charged in 30 min. The resulting suspension was mixed at rt for 1.5 h. The reaction mixture was cooled to 15 °C and 1500 mL of 6.7% NaHCO3 charged in 1.5 h, followed by the addition of 1200 mL of water over 60 min. The mixture was stirred at rt for 30 min, filtered and washed with water/DMF mixture (1 :2, 250 mL) and then with water (1500 mL). The wetcake was dried at 55 °C for 24 h to give 282 g of product as a white solid (90%).
The resulting solids (40 g) and 5% Pd/ Alumina were charged to a Parr reactor followed by THF (160 mL). The reactor was sealed and purged with nitrogen (6 x 20 psig) followed by a hydrogen purge (6 x 30 psig). The reactor was pressurized to 30 psig with hydrogen and agitated at room temperature for approximately 15 hours. The resulting slurry was filtered through a GF/F filter and concentrated to approximately 135 g solution. Heptane was added (120 mL), and the solution was stirred until solids formed. After an addition 2 – 3 hours additional heptane was added drop-wise (240 mL), the slurry was stirred for approximately 1 hour, then filtered. The solids were dried to afford the title compound.
Example 37C
(lR,4R)-1,4-bis(4-nitrophenyl)butane-1,4-diyl dimethanesulfonate
The product from Example 32 (5.01 g, 13.39 mmol) was combined with 2- methyltetrahydrofuran (70 mL) and cooled to -5 °C, and N,N-diisopropylethylamine (6.81 g, 52.7 mmol) was added over 30 seconds. Separately, a solution of methanesulfonic anhydride (6.01 g, 34.5 mmol) in 2-methyltetrahydrofuran (30 mL) was prepared and added to the diol slurry over 3 min., maintaining the internal temperature between -15 °C and -25 °C. After mixing for 5 min at -15 °C, the cooling bath was removed and the reaction was allowed to warm slowly to 23 °C and mixed for 30 minutes. After reaction completion, the crude slurry was carried immediately into the next step.
Example 37D
(2S,5S)-1-(4-tert-butylphenyl)-2,5-bis(4-nitrophenyl)pyrrolidine
To the crude product solution from Example 37C (7.35 g, 13.39 mmol) was added 4-tert- butylaniline (13.4 g, 90 mmol) at 23 °C over 1 minute. The reaction was heated to 65 °C for 2 h. After completion, the reaction mixture was cooled to 23 °C and diluted with 2-methyltetrahydrofuran (100 mL) and 1 M HCl (150 mL). After partitioning the phases, the organic phase was treated with 1 M HCl (140 mL), 2-methyltetrahydrofuran (50 mL), and 25 wt% aq. NaCl (100 mL), and the phases were partitioned. The organic phase was washed with 25 wt% aq. NaCl (50 mL), dried over MgSO/t, filtered, and concentrated in vacuo to approximately 20 mL. Heptane (30 mL) and additional 2- methyltetrahydrofuran were added in order to induce crystallization. The slurry was concentrated further, and additional heptane (40 mL) was slowly added and the slurry was filtered, washing with 2- methyltetrahydrofuran:heptane (1:4, 20 mL). The solids were suspended in MeOH (46 mL) for 3 h, filtered, and the wet solid was washed with additional MeOH (18 mL). The solid was dried at 45 °C in a vacuum oven for 16 h to provide the title compound (3.08 g, 51% 2-step yield).
Example 37E
4,4′-((2S,5S)-1-(4-tert-butylphenyl)pyrrolidine-2,5-diyl)dianiline
To a 160 ml Parr stirrer hydrogenation vessel was added the product from Example 37D (2 g, 4.49 mmol), followed by 60 ml of THF, and Raney Nickel Grace 2800 (1 g, 50 wt% (dry basis)) under a stream of nitrogen. The reactor was assembled and purged with nitrogen (8 x 20 psig) followed by purging with hydrogen (8 x 30 psig). The reactor was then pressurized to 30 psig with hydrogen and agitation (700 rpm) began and continued for a total of 16 h at room temperature. The slurry was filtered by vacuum filtration using a GF/F Whatman glass fiber filter. Evaporation of the filtrate to afford a slurry followed by the addition heptane and filtration gave the crude title compound, which was dried and used directly in the next step.
Example 37F dimethyl (2S,2’S)-l,r-((2S,2’S)-2,2′-(4,4′-((2S,5S)-1-(4-tert-butylphenyl)pyrrolidine-2,5-diyl)bis(4, l- phenylene)bis(azanediyl)bis(oxomethylene))bis(pyrrolidine-2, 1 -diyl))bis(3-methyl- 1 -oxobutane-2, 1 – diy 1) die arb amate To a solution of the product from Example 37E (1.64 g, 4.25 mmol) in DMF (20 ml), the product from Example 37B (2.89 g, 10.63 mmol), and HATU (4.04 g, 10.63 mmol) in DMF (15OmL) was added triethylamine (1.07 g, 10.63 mmol), and the solution was stirred at room temperature for 90 min. To the reaction mixture was poured 20 mL of water, and the white precipitate obtained was filtered, and the solid was washed with water (3×5 mL). The solid was blow dried for Ih. The crude material was loaded on a silica gel column and eluted with a gradient starting with ethyl acetate/ heptane (3/7), and ending with pure ethyl acetate. The desired fractions were combined and solvent distilled off to give a very light yellow solid, which was dried at 45 °C in a vacuum oven with nitrogen purge for 15 h to give the title compound (2.3 g, 61% yield). 1H NMR (400 MHz, DMSO- D6) δ ppm 0.88 (d, J=6.61 Hz, 6 H) 0.93 (d, J=6.72 Hz, 6 H) 1.11 (s, 9 H) 1.63 (d, J=5.42 Hz, 2 H) 1.80 – 2.04 (m, 8 H) 2.09 – 2.19 (m, 2 H) 2.44 – 2.47 (m, 2 H) 3.52 (s, 6 H) 3.59 – 3.66 (m, 2 H) 3.77 – 3.84 (m, 2 H) 4.02 (t, J=8.40 Hz, 2 H) 4.42 (dd, J=7.86, 4.83 Hz, 2 H) 5.14 (d, J=6.18 Hz, 2 H) 6.17 (d, J=8.67 Hz, 2 H) 6.94 (d, J=8.78 Hz, 2 H) 7.13 (d, J=8.46 Hz, 4 H) 7.31 (d, J=8.35 Hz, 2 H) 7.50 (d, J=8.35 Hz, 4 H) 9.98 (s, 2 H).
Alternately, the product from example 37E (11.7 g, 85 wt%, 25.8 mmol) and the product from example 37B (15.45 g, 56.7 mmol) are suspended in EtOAc (117 mL), diisopropylethylamine (18.67 g, 144 mmol) is added and the solution is cooled to 0 °C. In a separate flask, 1-propanephosphonic acid cyclic anhydride (T3P®) (46.0 g, 50 wt% in EtOAc, 72.2 mmol) was dissolved in EtOAc (58.5 mL), and charged to an addition funnel. The T3P solution is added to the reaction mixture drop-wise over 3-4 h and stirred until the reaction is complete. The reaction is warmed to room temperature,and washed with IM HCl/7.5 wt% NaCl (100 mL), then washed with 5% NaHCO3 (100 mL), then washed with 5% NaCl solution (100 mL). The solution was concentrated to approximately 60 mL, EtOH (300 mL) was added, and the solution was concentrated to 84 g solution.
A portion of the EtOH solution of product (29 g) was heated to 40 °C, and added 134 g 40 w% EtOH in H2O. A slurry of seeds in 58 wt/wt% EtOH/H2O was added, allowed to stir at 40 °C for several hours, then cooled to 0 °C. The slurry is then filtered, and washed with 58wt/wt% EtOH/H2O. The product is dried at 40 – 60 °C under vacuum, and then rehydrated by placing a tray of water in the vacuum oven to give the title compound. The title compound showed an EC50 value of less than about 0.1 nM in HCV lb-Conl replicon assays in the presence of 5% FBS.
Intermediates
Example 32
( 1 R,4R)- 1 ,4-bis(4-mtrophenyl)butane- 1 ,4-diol
To (S)-(-)-α,α-diphenyl-2-pyrrohdinemethanol (2 71 g, 10 70 mmol) was added THF (80 mL) at 23 °C The very thin suspension was treated with t11methyl borate (1 44 g, 13 86 mmol) over 30 seconds, and the resulting solution was mixed at 23 °C for 1 h The solution was cooled to 16-19 °C, and N,N-diethylanilme borane (21 45 g, 132 mmol) was added dropwise via syringe over 3-5 mm (caution vigorous H2 evolution), while the internal temperature was maintained at 16-19 °C After 15 mm, the H2 evolution had ceased To a separate vessel was added the product from Example IA (22 04 g, 95 wt%, 63 8 mmol), followed by THF (80 mL), to form an orange slurry After cooling the slurry to 11 °C, the borane solution was transferred via cannula into the dione slurry over 3-5 min During this period, the internal temperature of the slurry rose to 16 °C After the addition was complete, the reaction was maintained at 20-27 °C for an additional 2 5 h After reaction completion, the mixture was cooled to 5 °C and methanol (16 7 g, 521 mmol) was added dropwise over 5-10 mm, maintaining an internal temperature <20 °C (note vigorous H2 evolution) After the exotherm had ceased (ca 10 mm), the temperature was adjusted to 23 °C, and the reaction was mixed until complete dissolution of the solids had occurred Ethyl acetate (300 mL) and 1 M HCl (120 mL) were added, and the phases were partitioned The organic phase was then washed successively with 1 M HCl (2 x 120 mL), H2O (65 mL), and 10% aq NaCl (65 mL) The orgamcs were dried over MgSO4, filtered, and concentrated in vacuo Crystallization of the product occurred during the concentration The slurry was warmed to 50 °C, and heptane (250 inL) was added over 15 min. The slurry was then allowed to mix at 23 °C for 30 min and filtered. The wet cake was washed with 3: 1 heptane:ethyl acetate (75 mL), and the orange, crystalline solids were dried at 45 °C for 24 h to provide the title compound (15.35 g, 99.3% ee, 61% yield), which was contaminated with 11% of the meso isomer (vs. dl isomer).
References
- “VIEKIRA PAK™ (ombitasvir, paritaprevir and ritonavir tablets; dasabuvir tablets), for Oral Use. Full Prescribing Information”(PDF). AbbVie Inc., North Chicago, IL 60064. Retrieved 30 July 2015.
- “FDA approves Viekira Pak to treat hepatitis C”. Food and Drug Administration. December 19, 2014.
- “TECHNIVIE™ (ombitasvir, paritaprevir and ritonavir) Tablets, for Oral Use. Full Prescribing Information” (PDF). AbbVie Inc., North Chicago, IL 60064. Retrieved 28 July 2015.
- “FDA approves Technivie for treatment of chronic hepatitis C genotype 4”. Food and Drug Administration. July 24, 2015.
- Jordan J. Feld, Kris V. Kowdley, Eoin Coakley, Samuel Sigal, David R. Nelson, Darrell Crawford, Ola Weiland, Humberto Aguilar, Junyuan Xiong, Tami Pilot-Matias, Barbara DaSilva-Tillmann, Lois Larsen, Thomas Podsadecki, and Barry Bernstein (2014). “Treatment of HCV with ABT-450/r–Ombitasvir and Dasabuvir with Ribavirin”. N Engl J Med 370: 1594–1603.doi:10.1056/NEJMoa1315722.
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
Dimethyl ({(2S,5S)-1-[4-(2-methyl-2-propanyl)phenyl]-2,5-pyrrolidinediyl}bis{4,1-phenylenecarbamoyl(2S)-2,1-pyrrolidinediyl[(2S)-3-methyl-1-oxo-1,2-butanediyl]})biscarbamate
|
|
| Clinical data | |
| Trade names | Viekira Pak (with ombitasvir, paritaprevir, ritonavir and dasabuvir), Technivie (with ombitasvir, paritaprevir, and ritonavir) |
| Legal status |
|
| Routes of administration |
Oral |
| Pharmacokinetic data | |
| Bioavailability | not determined |
| Protein binding | ~99.9% |
| Metabolism | amide hydrolysis followed by oxidation |
| Onset of action | ~4 to 5 hours |
| Biological half-life | 21 to 25 hours |
| Excretion | mostly with feces (90.2%) |
| Identifiers | |
| CAS Registry Number | 1258226-87-7 |
| PubChem | CID: 54767916 |
| ChemSpider | 31136214 |
| ChEBI | CHEBI:85183 |
| Synonyms | ABT-267 |
| Chemical data | |
| Formula | C50H67N7O8 |
| Molecular mass | 894.11 g/mol |
rx list
VIEKIRA PAK is ombitasvir, paritaprevir, ritonavir fixed dose combination tablets copackaged with dasabuvir tablets.
Ombitasvir, paritaprevir, ritonavir fixed dose combination tablet includes ahepatitis C virus NS5A inhibitor (ombitasvir), a hepatitis C virus NS3/4Aprotease inhibitor (paritaprevir), and a CYP3A inhibitor (ritonavir) that inhibits CYP3A mediated metabolism of paritaprevir, thereby providing increased plasma concentration of paritaprevir. Dasabuvir is a hepatitis C virus nonnucleoside NS5B palm polymerase inhibitor, which is supplied as separate tablets in the copackage. Both tablets are for oral administration.
Ombitasvir
The chemical name of ombitasvir is Dimethyl ([(2S,5S)-1-(4-tert-butylphenyl) pyrrolidine-2,5diyl]bis{benzene-4,1-diylcarbamoyl(2S)pyrrolidine-2,1-diyl[(2S)-3-methyl-1-oxobutane-1,2diyl]})biscarbamate hydrate. The molecular formula is C50H67N7O8•4.5H2O (hydrate) and the molecular weight for the drug substance is 975.20 (hydrate). The drug substance is white to light yellow to light pink powder, and is practically insoluble in aqueous buffers but is soluble in ethanol. Ombitasvir has the following molecular structure:
Paritaprevir
The chemical name of paritaprevir is (2R,6S,12Z,13aS,14aR,16aS)-N-(cyclopropylsulfonyl)-6{[(5-methylpyrazin-2-yl)carbonyl]amino}-5,16-dioxo-2-(phenanthridin-6-yloxy)1,2,3,6,7,8,9,10,11,13a,14,15,16,16a-tetradecahydrocyclopropa[e]pyrrolo[1,2-a][1,4] diazacyclopentadecine-14a(5H)-carboxamide dihydrate. The molecular formula is C40H43N7O7S•2H2O (dihydrate) and the molecular weight for the drug substance is 801.91 (dihydrate). The drug substance is white to off-white powder with very low water solubility. Paritaprevir has the following molecular structure:
 |
Ritonavir
The chemical name of ritonavir is [5S-(5R*,8R*,10R*,11R*)]10-Hydroxy-2-methyl-5-(1methyethyl)-1-[2-(1-methylethyl)-4-thiazolyl]-3,6-dioxo-8,11-bis(phenylmethyl)-2,4,7,12tetraazatridecan-13-oic acid,5-thiazolylmethyl ester. The molecular formula is C37H48N6O5S2 and the molecular weight for the drug substance is 720.95. The drug substance is white to off white to light tan powder practically insoluble in water and freely soluble in methanol and ethanol. Ritonavir has the following molecular structure:
Ombitasvir, Paritaprevir, Ritonavir Fixed-Dose Combination Tablets
Ombitasvir, paritaprevir, and ritonavir film-coated tablets are co-formulated immediate release tablets. The tablet contains copovidone, K value 28,vitamin E polyethylene glycol succinate, propylene glycol monolaurate Type I, sorbitan monolaurate, colloidal silicon dioxide/colloidal anhydrous silica, sodium stearyl fumarate, polyvinyl alcohol, polyethylene glycol 3350/macrogol 3350, talc, titanium dioxide, and iron oxide red. The strength for the tablet is 12.5 mg ombitasvir, 75 mg paritaprevir, 50 mg ritonavir.
Dasabuvir
The chemical name of dasabuvir is Sodium 3-(3-tert-butyl-4-methoxy-5-{6[(methylsulfonyl)amino]naphthalene-2-yl}phenyl)-2,6-dioxo-3,6-dihydro-2H-pyrimidin-1-ide hydrate (1:1:1). The molecular formula is C26H26N3O5S•Na•H2O (salt, hydrate) and the molecular weight of the drug substance is 533.57 (salt, hydrate). The drug substance is white to pale yellow to pink powder, slightly soluble in water and very slightly soluble in methanol and isopropyl alcohol. Dasabuvir has the following molecular structure:
 |
Dasabuvir is formulated as a 250 mg film-coated, immediate release tablet containing microcrystalline cellulose (D50-100 um), microcrystalline cellulose (D50-50 um), lactose monohydrate, copovidone, croscarmellose sodium, colloidal silicon dioxide/anhydrous colloidal silica, magnesium stearate, polyvinyl alcohol, titanium dioxide, polyethylene glycol 3350/macrogol 3350, talc, and iron oxide yellow, iron oxide red and iron oxide black. Each tablet contains 270.3 mg dasabuvir sodium monohydrate equivalent to 250 mg dasabuvir.
//////////fda 2014, Ombitasvir, orphan drug, Abbvie Inc.
IPI 926, Saridegib, Patidegib


IPI 926, Saridegib, Patidegib
C29H48N2O3S
Exact Mass: 504.33856
1037210-93-7
-
Patidegib hydrochloride
-
Saridegib hydrochloride
-
-
C29-H48-N2-O3-S.Cl-H
- 541.2361
-
Methanesulfonamide, N-((2S,3R,3’R,3aS,4’aR,6S,6’aR,6’bS,7aR,12’aS,12’bS)-2′,3′,3a,4,4′,4’a,5,5′,6,6′,6’a,6’b,7,7′,7a,8′,10′,12′,12’a,12’b-eicosahydro-3,6,11′,12’b-tetramethylspiro(furo(3,2-b)pyridine-2(3H),9′(1’H)-naphth(2,1-a)azulen)-3′-yl)-, hydrochloride (1:1)
CAS 1169829-40-6 HCL
Saridegib also known as IPI-926 is an experimental drug candidate undergoing clinical trials for the treatment of various types of cancer, including hard to treat hematologic malignancies such as myelofibrosis and ligand-dependant tumors such as chondrosarcoma.[1] IPI-926 exhibits its pharmacological effect by inhibition of the G protein-coupled receptor smoothened, a component of the hedgehog signaling pathway.[2]
Chemically, it is a semi-synthetic derivative of the alkaloid cyclopamine. The process begins with cyclopamine extracted from harvested Veratrum californicum which is taken through a series of alterations resulting in an analogue of the natural product cyclopamine, making IPI-926 the only compound in development/testing that is not fully synthetic.[2]
Saridegib is a member of a class of anti-cancer compounds known as hedgehog inhibitors (Hhi). Most of these compounds affect thehedgehog signaling pathway via inhibition of smoothened (Smo), a key component of the pathway. Depending on when a Hh inhibiting compound is approved by the U.S. Food and Drug Administration (FDA), there may be a perceived need for one to be differentiated over another for marketing purposes, which could lead to different nomenclature (e.g., a Hhi or an agonist of Smo).
This marketing technique is more of a differentiation strategy than a scientific property of these compounds, as the mechanism of action (MOA) in the end is inhibition of the Hh pathway, targeting cancer stem cells. However, as these new compounds are further studied, identification of differences in a compound’s MOA, could lead to hypotheses regarding the stage at which Smo is inhibited, where along the pathway the compound binds, or specific binding properties of a compound.
If these hypotheses are proven, claims could be made regarding a specific compound’s MOA and how it affects efficacy, safety, combinability with other cancer treatments, etc. Scientific data in support of such hypotheses have not been published to date.
SARIDEGIB

N-[(3R,3’R,3’aS,4aR,6’S,6aR,6bS,7’aR,9S,12aS,12bS)-3′,6′,11,12b-tetramethylspiro[1,2,3,4,4a,5,6,6a,6b,7,8,10,12,12a-tetradecahydronaphtho[2,1-a]azulene-9,2′-3a,4,5,6,7,7a-hexahydro-3H-furo[3,2-b]pyridine]-3-yl]methanesulfonamide
There are currently no drugs in the Hhi class FDA approved, however IPI-926 and GDC-0449 are the 2 leading compounds in the class. IPI-926, GDC-0449, and LDE-225 are the only compounds that have generic names passed by the United States Adopted Name (USAN) council (Infinity IPI-926/saridegib, Genentech GDC-0449/vismodegib, and Novartis LDE-225/erismodegib). Although Infinity is further along in chondrosarcoma, myelofibrosis, and AML, Roche/Genentech recently submitted an NDA for GDC-0449 for the treatment of adults with advanced basal cell carcinoma (BCC) when surgery is no longer an option, and the FDA has accepted and has filed the NDA, giving it priority review status. Thus it appears that Roche/Genentech will be the first Hhi to market with GDC-0449, if approved, for the treatment of advanced BCC, with Infinity second to market with IPI-926 for treatment in chondrosarcoma. It appears Infinity will not pursue an indication for BCC and focus on cancers with high unmet needs.[1][3][4][5][6]
Other Hhi-class compounds not as far along in development as IPI-926 and GDC-0449 include:[7]
- Novartis’ LDE-225 (USAN generic name erismodegib)
- Exelixis/Bristol-Myers Squibb’s BMS-833923 (XL139)
- Millennium Pharmaceuticals’s TAK-441
- Pfizer’s PF-04449913

Fig 1. Chemical structure comparison between IPI-926 and cyclopamine
IPI-926 is currently developed by Infinity Pharmaceuticals, Inc. Malignant activation of the Hedgehog pathway is implicated in multiple cancer settings and Infinity’s development strategy is designed to enable IPI-926 to target a broad range of critical oncology targets – from the tumor cell to the cancer microenvironment. This broadly applicable, targeted approach represents an innovative method for fighting cancer and has potential in treating a range of cancers, including pancreatic cancer, small cell lung cancer, ovarian cancer, bladder cancer, medulloblastoma, basal cell carcinoma, and certain hematological malignancies.
The hedgehog pathway inhibitor IPI-926 has been in clinical investigation for basal cell carcinoma, chondrosarcoma, and pancreatic cancer. In the final step of the synthesis of IPI-926 the drug substance (DS) is isolated as the hydrochloride salt of the 2-propanol (2-PrOH) solvate
A design of experiments (DoE) approach was taken to optimize purity and reaction yield of the final debenzylation and hydrochloride salt formation of IPI-926. The study involved a careful dissection of the different process steps to enable an independent investigation of these steps while ensuring that process streams were representative. The results enabled a streamlined process from the final chemical transformation to the salting and isolation and led to the elimination of variability in the process as well as a robust control of impurities. The optimized process was applied to production and demonstrated on the kilogram scale.
A Design of Experiments Approach to a Robust Final Deprotection and Reactive Crystallization of IPI-926, A Novel Hedgehog Pathway Inhibitor
The product was dried at a jacket temperature of 45 °C until an LOD <2.30% (w/w) was achieved. Yield: 11.5 kg (73% from compound 1, correcting for the seed). HPLC purity: 99.9% area (compound 2 content: 0.08% w/w). Assay: 83.7% w/w (as-is), 99.1% w/w (anhydrous, solvent-free). Moisture content: 1.6% w/w. Chlorine content: 5.72% w/w. Residual solvents: acetone (720 ppm); acetonitrile (<41 ppm); 2-MeTHF (none detected); 2-propanol (81 147 ppm); toluene (<90 ppm). Residual metals: palladium (0 ppm); platinum (0 ppm); ruthenium (0 ppm). Additional data for the IPI-926 free base:
1H NMR (400 MHz, CDCl3) 6.90 (br s, 1H), 3.31 (dt, J = 10.6, 3.8 Hz, 1H), 3.20 (br s, 1H), 3.10 (dd, J = 13.7, 4.5 Hz, 1H), 2.91 (s, 3H), 2.62 (dd,J = 9.9, 7.6 Hz, 1H), 2.33 (br d, J = 14.5 Hz, 1H), 2.27–2.15 (m, 1H), 2.10 (dd, J = 14.5, 6.9 Hz, 1H), 1.99–1.17 (m, 28H), 1.05 (q, J = 11.6 Hz, 1H), 0.93 (d, J = 7.4 Hz, 3H), 0.88 (d, J = 6.6 Hz, 3H), 0.86 (s, 3H) ppm.
13C NMR (100 MHz, CDCl3) 140.47, 124.53, 82.48, 76.97, 63.73, 54.08, 53.87, 50.12, 49.98, 47.19, 44.73, 42.27, 42.10, 40.24, 37.55, 37.44, 36.04, 34.44, 31.87, 31.33, 30.46, 29.79, 28.37, 27.94, 26.26, 24.19, 22.70, 18.92, 10.19 ppm;
MS: m/z = 505.29 [M + H]+.
PAPER
Tremblay, M. R.; Lescarbeau, A.; Grogan, M. J.; Tan, E.; Lin, G.; Austad, B. C.; Yu, L.-C.;Behnke, M. L.; Nair, S. J.; Hagel, M.; White, K.; Conley, J.; Manna, J. D.; Alvarez-Diez, T. M.; Hoyt, J.; Woodward, C. N.; Sydor, J. R.; Pink, M.; MacDougall, J.; Campbell, M. J.;Cushing, J.; Ferguson, J.; Curtis, M. S.; McGovern, K.; Read, M. A.; Palombella, V. J.;Adams, J.; Castro, A. C. J. Med. Chem. 2009, 52, 4400– 4418, DOI: 10.1021/jm900305z

Recent evidence suggests that blocking aberrant hedgehog pathway signaling may be a promising therapeutic strategy for the treatment of several types of cancer. Cyclopamine, a plant Veratrum alkaloid, is a natural product antagonist of the hedgehog pathway. In a previous report, a seven-membered D-ring semisynthetic analogue of cyclopamine, IPI-269609 (2), was shown to have greater acid stability and better aqueous solubility compared to cyclopamine. Further modifications of the A-ring system generated three series of analogues with improved potency and/or solubility. Lead compounds from each series were characterized in vitro and evaluated in vivo for biological activity and pharmacokinetic properties. These studies led to the discovery of IPI-926 (compound 28), a novel semisynthetic cyclopamine analogue with substantially improved pharmaceutical properties and potency and a favorable pharmacokinetic profile relative to cyclopamine and compound2. As a result, complete tumor regression was observed in a Hh-dependent medulloblastoma allograft model after daily oral administration of 40 mg/kg of compound 28.
28 (4.06 g, 8.05 mmol, 95% for two steps). NMR δH (400 MHz, CDCl3) 6.90 (br s, 1H), 3.31 (dt, J = 10.6, 3.8 Hz, 1H), 3.20 (br s, 1H), 3.10 (dd, J = 13.7, 4.5 Hz, 1H), 2.91 (s, 3H), 2.62 (dd, J = 9.9, 7.6 Hz, 1H), 2.33 (br d, J = 14.5 Hz, 1H), 2.27−2.15 (m, 1H), 2.10 (dd, J = 14.5, 6.9 Hz, 1H), 1.99−1.17 (m, 28H), 1.05 (q, J = 11.6 Hz, 1H), 0.93 (d, J = 7.4 Hz, 3H), 0.88 (d, J = 6.6 Hz, 3H), 0.86 (s, 3H); NMR δC (100 MHz, CDCl3) 140.47, 124.53, 82.48, 76.97, 63.73, 54.08, 53.87, 50.12, 49.98, 47.19, 44.73, 42.27, 42.10, 40.24, 37.55, 37.44, 36.04, 34.44, 31.87, 31.33, 30.46, 29.79, 28.37, 27.94, 26.26, 24.19, 22.70, 18.92, 10.19; m/z = 505.29 [M + H]+; HPLC 99.1 a/a % at 215 nm.
Click on images for clear view……………..
Paper

A design of experiments (DoE) approach was taken to optimize purity and reaction yield of the final debenzylation and hydrochloride salt formation of IPI-926. The study involved a careful dissection of the different process steps to enable an independent investigation of these steps while ensuring that process streams were representative. The results enabled a streamlined process from the final chemical transformation to the salting and isolation and led to the elimination of variability in the process as well as a robust control of impurities. The optimized process was applied to production and demonstrated on the kilogram scale.
Development of a Multi Kilogram-Scale, Tandem Cyclopropanation Ring-Expansion Reaction en Route to Hedgehog Antagonist IPI-926

The formation of the d-homocyclopamine ring system in IPI-926 is the key step in its semisynthesis and proceeds via a chemoselective cyclopropanation followed by a stereoselective acid-catalyzed carbocation rearrangement. In order to perform large-scale cyclopropanation reactions, we developed new iodomethylzinc bis(aryl)phosphate reagents that were found to be both effective and safe. These soluble reagents can be prepared under mild conditions and are stable during the course of the reaction. Importantly, they have favorable energetics relative to other cyclopropanating agents such as EtZnCH2I. Herein, we describe the process optimization studies that led to successful large-scale production of the d-homocyclopamine core necessary for IPI-926.
References
- “Pipeline: IPI-926”. Infinity Pharmaceuticals.
- Tremblay, MR; Lescarbeau, A; Grogan, MJ; Tan, E; Lin, G; Austad, BC; Yu, LC; Behnke, ML et al. (2009). “Discovery of a potent and orally active hedgehog pathway antagonist (IPI-926)”. Journal of Medical Chemistry 52 (14): 4400–18. doi:10.1021/jm900305z. PMID 19522463.
- “Pipeline”. Infinity Pharmaceuticals.
- “Genentech Pipeline”. Genentech.
- “USAN Stem List” (PDF). AMA.
- “Names under consideration”. AMA.
- “Search results for Hh clinical trials”. United National Institute of Health’s ClinicalTrials.gov.
- 1. Tremblay MR, Lescarbeau A, Grogan MJ, Tan E, Lin G, Austad BC, Yu LC, Behnke ML, Nair SJ, Hagel M et al.. (2009)
Discovery of a potent and orally active hedgehog pathway antagonist (IPI-926).
J. Med. Chem., 52 (14): 4400-18.
 |
|
 |
|
| Names | |
|---|---|
| IUPAC name
N-((2S,3R,3aS,3′R,4a′R,6S,6a′R,6b′S,7aR,12a&prmie;S,12b′S)-3,6,11′,12b′-tetramethyl-2′,3a,3′,4,4′,4a′,5,5&prmie;,6,6′,6a′,6b′,7,7a,7′,8′,10′,12′,12a′,12b′-icosahydro-1′H,3H-spiro[furo[3,2-b]pyridine-2,9′-naphtho[2,1-a]azulen]-3′-yl)methanesulfonamide
|
|
| Other names
saridegib
|
|
| Identifiers | |
| 1037210-93-7 |
|
| ChEMBL | ChEMBL538867 |
| ChemSpider | 26353073 |
| 8198 | |
| Jmol-3D images | Image |
| PubChem | 25027363 |
| UNII | JT96FPU35X |
| Properties | |
| C29H48N2O3S | |
| Molar mass | 504.77 g·mol−1 |
| Pharmacology | |
| Legal status |
|
/////Saridegib, IPI-926
EU: New GMP Implementing Act published
DRUG REGULATORY AFFAIRS INTERNATIONAL

The EU Commission has published a new public consultation on an Implementing Act on GMP principles and guidelines for medicinal products for human use.
The EU Commission has published a new public consultation on an Implementing Act on Principles and guidelines on good manufacturing practices for medicinal products for human use.
The reason is that once Regulation (EU) No 536/2014 on clinical trials becomes applicable, manufacture and import of Investigational Medicinal Products (IMPs) for the use in clinical trials carried out under that Regulation cannot follow GMP for IMPs set out in Directive 2003/94/EC. They then have to be manufactured or imported under regulations laid down by the Delegated Act or other specified regulation. It is therefore necessary that Directive 2003/94/EC is revised by a new Implementing Directive on principles and guidelines of good manufacturing practice for medicinal products for human use (without IMPs).
The EU Commission…
View original post 60 more words
WO 2015129603, NEW PATENT, Daiichi Sankyo Co Ltd, Edoxaban

HIGH-PURITY CRYSTALS OF ACTIVE BLOOD COAGULATION FACTOR X (FXA) INHIBITOR
DAIICHI SANKYO COMPANY,LIMITED [JP/JP]; 3-5-1,Nihonbashi Honcho,Chuo-ku, Tokyo 1038426 (JP)
Claims highly pure crystalline form of edoxaban p-toluenesulfonate monohydrate. Useful for treating thrombotic diseases. Daiichi Sankyo had developed and launched edoxaban for treating non-valvular atrial fibrillation, deep vein thrombosis and pulmonary embolism, the drug was recently launched in US (in February 2015) and approved in Europe (in June 2015).
The present invention addresses the problem of providing high-purity crystals of a compound which is represented by formula (1a) and is an active blood coagulation factor X (FXa) inhibitor. High-purity crystals of a compound represented by formula (1a) which: are characterised by being obtained by a step for dissolving crystals in a solvent and thereafter performing recrystallisation; have a 0.03% or less maximum content of one impurity as the impurity content by percentage; and have a 0.13% or less total impurity content.
In N represented 1 – (5-Chloro-2-yl) -N 2 – ((1S, 2R, 4S) -4 – [(dimethylamino) carbonyl] -2 – {[(5-methyl-4 , 5,6,7-tetrahydro thiazolone [5,4-c] pyridin-2-yl) carbonyl] amino} cyclohexyl) Etanjiamido p- toluenesulfonic acid monohydrate [hereinafter, may be referred to as compound (1a) is there
The obtained compound, in analysis using HPLC, as impurities, a peak of more impurities (both 0.03 wt%) is confirmed, the total of the impurities was 0.16 wt.% Since, its purity was 99.84% (Note that the content of% refers to% of the HPLC area value of the free form of formula (1a) compound).1 H-NMR (DMSO-d6) delta: 1.45-1.54 (1H, M), 1.66-1.78 (3H, M), 2.03-2.10 (2H, M), 2.28 (3H, s), 2.79 (3H, s), 2.91-3.02 (1H, m), 2.93 (3H, s), 2.99 (3H, s), 3 .13-3.24 (2H, m), 3.46-3.82 (2H, m), 3.98-4.04 (1H, m), 4.43-4.80 (3H, m) , 7.11 (2H, d, J = 7.8Hz), 7.46 (2H, d, J = 8.2Hz), 8.01 (2H, d, J = 1.8Hz), 8.46 ( 1H, t, J = 1.8Hz), 8.75 (1H, d, J = 6.9Hz), 9.10-9.28 (1H, br.s), 10.18 (1H, br.s ), 10.29 (1H, s).
Found: C; 50.25%, H; 5.36%, N; 13.32%

/////////////WO 2015129603, NEW PATENT, Daiichi Sankyo Co Ltd, Edoxaban
Indian pharma’s struggle to tighten standards paves way for M&A deals
MUMBAI – India’s smaller generic drugmakers, struggling to cope with a bruised reputation and tougher regulation in the United States, are under pressure to consider branching out to new, less-profitable markets or sell out to larger rivals.
Two years after its most high-profile regulatory setback to date in the United States – Ranbaxy’s $500 million U.S. fine for drug safety violations – India’s $15 billion a year generic drug industry is still rebuilding its image in its biggest market.
Many of its top firms are facing sanctions at some of their factories, as the U.S. Food and Drug Administration (FDA) tightens checks and its approvals process.
Combined with government-mandated price controls on drugs at home, that is piling pressure on smaller players.
“If they want to have a presence globally, they have to make investments. If they can’t, then they’ll have to focus on other markets or scale back their ambition outside of India, and that’s probably what will happen,” said Subhanu Saxena, CEO of Cipla , India’s fourth-largest drugmaker by revenue.
Ashok Anand, president of Hikal Ltd , a Mumbai-based drugmaker with a market value of $167 million, said some peers were putting themselves on the block.
“If they cannot deal with the stricter regulations, they might just prefer to sell out,” he said.
Pressure on U.S. sales has been felt across the Indian industry, with all drugmakers hit by delays in FDA approvals as the U.S. safety body overhauls its review process. Growth in U.S. revenue for drugmakers slowed to 14 percent in the year to March 2015, less than half what it was in the year to March 2012, according to brokerage Edelweiss.
But for larger players who want to plug gaps or, for the likes of Glenmark and Aurobindo who aim to grow in the United States, this pressure has lowered prices and could pave the way for attractive deals, bankers said.
“Now that some of the smaller companies are reeling under intensive regulatory scrutiny and want to cash out on their investments, valuations would be much more realistic,” said the head of India M&A at a large European bank in Mumbai.
SPENDING SPREE
Indian manufacturers say they have spent millions in high-end testing equipment, improved training and have hired larger teams in quality control since Ranbaxy was fined for manipulating clinical data.
Some consultants estimate spending on compliance has more than doubled to reach about 6 to 7 percent of sales for the larger companies.
But while the number of U.S. export bans issued to Indian companies fell to eight in 2014 from 21 in 2013, according to FDA data, the agency continues to find manufacturing violations at the plants of some of the biggest drugmakers in the country, an indication of the pervasiveness of the problem.
Sun Pharmaceutical Industries , Wockhardt , Dr Reddy’s Laboratories and Cadila Healthcarehave all faced FDA rebukes over the past year.
Smaller firms Ipca and Aarti Drugs faced FDA bans on their plants this year.
These failures – which executives blame on India’s “quick fix” culture and consultants blame on a failure to prioritize compliance – have clouded short-term growth prospects and added to pressure on smaller players, pushing some to look elsewhere.
“They can choose to be in lesser-regulated markets, such as Latin America, where there is a lot of demand. But they will have to live with much thinner margins,” said the finance director of a small Indian drugmaker, who did not want to be named. “It’s survival of the fittest.” REUTERS
http://m.todayonline.com/business/indian-pharmas-struggle-tighten-standards-paves-way-ma-deals
///////
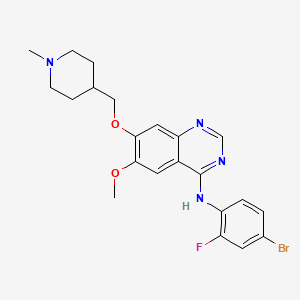
Vandetanib
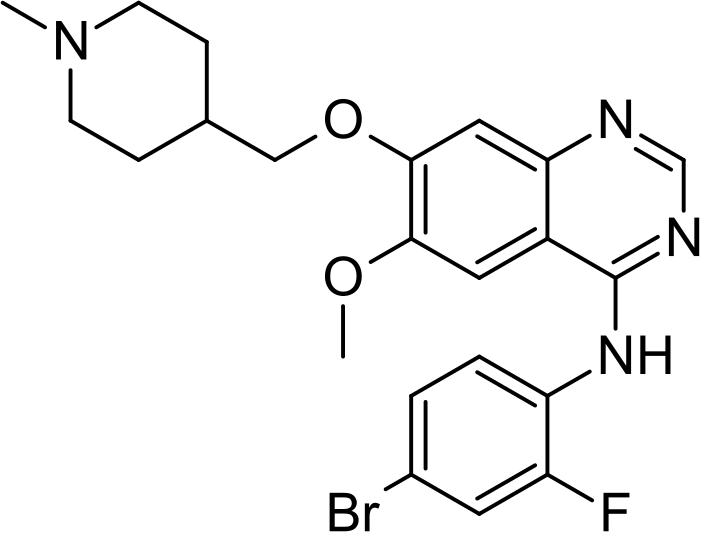
Vandetanib; 443913-73-3; Zactima; ZD6474; Caprelsa; ZD 6474; ch 331, azd 6474
cas 338992-00-0 free form
338992-48-6 HCl
338992-53-3 monotrifluoroacetate
N-(4-Bromo-2-fluorophenyl)-6-methoxy-7-(1-methylpiperidin-4-ylmethoxy)quinazolin-4-amine
Vandetanib (INN, trade name Caprelsa) is an anti-cancer drug that is used for the treatment of certain tumours of the thyroid gland. It acts as a kinase inhibitor of a number of cell receptors, mainly the vascular endothelial growth factor receptor (VEGFR), theepidermal growth factor receptor (EGFR), and the RET-tyrosine kinase.[1][2] The drug was developed by AstraZeneca.
Orphan drug designation has been assigned in the E.U. for the treatment of medullary thyroid carcinoma. In 2005, orphan drug designation was also assigned in the U.S. for several indications, including treatment of patients with follicular thyroid carcinoma, medullary thyroid carcinoma, anaplastic thyroid carcinoma, and locally advanced and metastatic papillary thyroid carcinoma. In 2013, orphan drug designation has been assigned in Japan as well for the treatment of thyroid cancer.
Approvals and indications
Vandetanib was the first drug to be approved by FDA (April 2011) for treatment of late-stage (metastatic) medullary thyroid cancer in adult patients who are ineligible for surgery.[3] Vandetanib was first initially marketed without a trade name,[4] and is being marketed under the trade name Caprelsa since August 2011.[5]
Vandetanib is an orally active vascular endothelial growth factor receptor-2 (VEGFR-2/KDR) tyrosine kinase inhibitor, originally developed by AstraZeneca, which was filed for approval in the U.S. and the E.U. for the treatment of non-small cell lung cancer (NSCLC) in combination with chemotherapy, in patients previously treated with one prior anticancer therapy.
However, in late 2009 the company withdrew both the U.S and the EU applications. In 2010, AstraZeneca discontinued development of this compound for the treatment of NSCLC. In 2011, the FDA approved vandetanib for the treatment of medullary thyroid cancer. Also in 2011, a positive opinion was assigned to the regulatory application filed in the E.U. for this indication and in Japan was filed for approval.
Final EMA approval was granted in February 2012 and first E.U. launch took place in the U.K. in 2012.
2011 年 4 月 6 by the FDA-approved surgical resection can not be used for locally advanced or metastatic medullary thyroid cancer (medullary thyroid cancer, MTC) of the drug. Vandetanib is vascular endothelial growth factor receptors (vascular endothelial growth factor receptor, VEGFR) and epidermal growth factor receptor (epidermal growth factor receptor, EGFR) antagonists, tyrosine kinase inhibitors (tyrosine kinase inhibitor). Produced by AstraZeneca.
The synthetic route is as follows:
………………
………………………..
……….
Design and structure-activity relationship of a new class of potent VEGF receptor tyrosine kinase inhibitors
J Med Chem 1999, 42(26): 5369
http://pubs.acs.org/doi/abs/10.1021/jm990345w
………………………
Radiosynthesis of [(11)C]Vandetanib and [(11)C]chloro-Vandetanib as new potential PET agents for imaging of VEGFR in cancer
Bioorg Med Chem Lett 2011, 21(11): 3222
Novel 4-anilinoquinazolines with C-7 basic side chains: Design and structure activity relationship of a series of potent, orally active, VEGF receptor tyrosine kinase inhibitors
J Med Chem 2002, 45(6): 1300
A novel approach to quinazolin-4(3H)-one via quinazoline oxidation: An improved synthesis of 4-anilinoquinazolines
Tetrahedron 2010, 66(4): 962
………………………………
CN 104098544
http://www.google.com/patents/CN104098544A?cl=en
Vandetanib is a synthetic Anilinoquinazoline, advanced medullary thyroid cancer can not be used for the treatment of surgical treatment (medullary thyroid cancer), chemical name: 4- (4-bromo-2- fluoroanilino) _6_ methoxy -7 – [(l- methylpiperidin-4-yl) methoxy] quinazoline, having the following structural formula I:
[0004] The present method of synthesizing the compound are as follows:
[0005] US Patent US7173038 AstraZeneca announced the following methods:
[0006] Method One:
[0007]
Method two:
A structure in which the synthesis of compounds of formula as follows:
the process is cumbersome, long synthetic route, therefore a need to provide a new synthetic way to overcome these problems.
An aspect provides a compound having the structure of formula II:
Another aspect provides a process for preparing a compound of the structural formula II, a compound of formula III with a compound of formula IV in the presence of a base to give a compound of the structural formula II,
where Μ for methylphenylsulfonyl, methylsulfonyl.
Example: 4- (4-bromo-2-fluoroanilino) -6_ methoxy-7 – [(1-formyl-4-yl) methoxy] quinazoline preparation
[0026] in 50mL two-neck flask was added 4- (4-bromo-2-fluoroanilino) -6-methoxy-7-hydroxy-quinazoline (3. 64g, 0 · Olmol), 1- formyl- 4-p methylsulfonyloxy- methylpiperazine steep (3. 56g, 0 · 012mol) and potassium carbonate (4. 14g, 0.03mol), yellow turbid solution was stirred and heated to 100 ° C, TLC detection to feed completion of the reaction. Down to room temperature, the reaction mixture was slowly poured into l〇〇mL water, stirred, filtered, then the filter cake was washed with 50mL water, 15mL of ethyl acetate and then slurried, filtered and dried to give a pale green solid 4- (4- bromo-2-fluoroanilino) -6-methoxy -7 – [(l- carboxylic acid piperidin-4-yl) methoxy] quinazoline 3. 9g, 80% yield.
[0027] ^ NMR (400Mz, DMS0): δ = 1 1〇-1 29 (m, 2H), δ = 1 40-1 43 (m, 2H), δ = 2 15 (s,….. 1H), δ = 2. 64-2. 73 (m, 1H), δ = 3. 06-3. 12 (m, 1H), δ = 3. 71-3. 74 (d, 1H), δ = 3. 95 (s, 3H), δ = 4 • 03-4. 05 (d, 2H), δ = 4. 20-4. 23 (d, 1H), δ = 7. 20 (s, 1H), δ = 7. 46-7. 48 (m, 1H), δ = 7. 51-7 • 53 (m, 1H), δ = 7. 65-7. 67 (d, 1H), δ = 7. 80 (s, 1H), δ = 8. 01 (s, 1H), δ = 8. 35 (s, 1H), δ = 9. 54 (s, 1H).
[0028] Example 2: Preparation of 4- (4-bromo-2-fluoroanilino) -6-methoxy-7 – [(1-methyl-piperidin-4-yl) methoxy] quinazoline preparation
[0029] 4- (4-bromo-2-fluoroanilino) in 100mL three-necked flask, 6-methoxy-7 – [(1-formyl-4-yl) methoxy] quinoline oxazoline (0 · 98g, 2. Ommol), zinc (0 · 6g, 4. 4mmol) and tetrahydrofuran (20mL), stirred pale yellow turbid liquid. At room temperature was added portionwise sodium borohydride (0. 15g, 4. OmmoL), little change in the temperature. Heating
……………………………….
CN 104211649
http://www.google.com/patents/CN104211649A?cl=en
Pharmacokinetics
Vandetanib is well absorbed from the gut, reaches peak blood plasma concentrations 4 to 10 hours after application, and has a half-life of 120 hours days on average, per Phase I pharmacokinetic studies. It has to be taken for about three months to achieve a steady-state concentration. In the blood, it is almost completely (90–96%) bound to plasma proteins such as albumin. It is metabolised to N-desmethylvandetanib via CYP3A4 and to vandetanib-N-oxide via FMO1 and 3. Both of these are active metabolites. Vandetanib is excreted via the faeces (44%) and the urine (25%) in form of the unchanged drug and the metabolites.[2][9][10]
Metabolites of vandetanib (top left): N-desmethylvandetanib (bottom left, via CYP3A4), vandetanib-N-oxide (bottom right, via FMO1 andFMO3), both pharmacologically active, and a minor amount of aglucuronide.[10]
Clinical trials
Non-small cell lung cancer
The drug underwent clinical trials as a potential targeted treatment for non-small-cell lung cancer. There have been some promising results from a phase III trial withdocetaxel.[11] There have also been ambivalent results when used with pemetrexed.[12] Another trial with docetaxel was recruiting in July 2009.[13]
AstraZeneca withdrew EU regulatory submissions for vandetanib (under the proposed trade name Zactima) in October 2009 after trials showed no benefit when the drug was administered alongside chemotherapy.[14]
References
- “Definition of vandetanib”. NCI Drug Dictionary. National Cancer Institute.
- “Vandetanib Monograph”. Drugs.com. Retrieved 29 August 2012.
- “FDA approves new treatment for rare form of thyroid cancer”. Retrieved 7 April 2011.
- “FDA approves orphan drug vandetanib for advanced medullary thyroid cancer” (Press release). AstraZeneca. Retrieved 2011-08-17.
- “AstraZeneca announces trade name CAPRELSA® for vandetanib” (Press release). AstraZeneca. Retrieved 2011-08-17.
- Khurana V, Minocha M, Pal D, Mitra AK (March 2014). “Role of OATP-1B1 and/or OATP-1B3 in hepatic disposition of tyrosine kinase inhibitors.”. Drug Metabol Drug Interact.0 (0): 1–11. doi:10.1515/dmdi-2013-0062. PMID 24643910.
- Haberfeld, H, ed. (2012). Austria-Codex (in German). Vienna: Österreichischer Apothekerverlag.
- Khurana V, Minocha M, Pal D, Mitra AK (May 2014). “Inhibition of OATP-1B1 and OATP-1B3 by tyrosine kinase inhibitors.”. Drug Metabol Drug Interact. 0 (0): 1–11.doi:10.1515/dmdi-2014-0014. PMID 24807167.
- Martin, P.; Oliver, S.; Kennedy, S. J.; Partridge, E.; Hutchison, M.; Clarke, D.; Giles, P. (2012). “Pharmacokinetics of Vandetanib: Three Phase I Studies in Healthy Subjects”.Clinical Therapeutics 34 (1): 221–237. doi:10.1016/j.clinthera.2011.11.011.PMID 22206795.
- “Clinical Pharmacology Review: Vandetanib” (PDF). US Food and Drug Administration, Center for Drug Evaluation and Research. 20 August 2010. Retrieved29 August 2012.
- “Vandetanib Shows Clinical Benefit When Combined With Docetaxel For Lung Cancer”. ScienceDaily. 3 June 2009.
- “IASLC: Vandetanib Fails to Improve NSCLC Outcomes with Pemetrexed”. Medpage today. 5 Aug 2009.
- Clinical trial number NCT00687297 for “Study of Vandetanib Combined With Chemotherapy to Treat Advanced Non-small Cell Lung Cancer” at ClinicalTrials.gov
- “Zactima”. European Medicines Agency.
External links
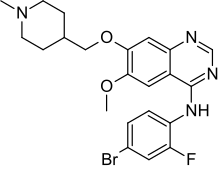 |
|
| Systematic (IUPAC) name | |
|---|---|
|
N-(4-bromo-2-fluorophenyl)-6-methoxy-7-[(1-methylpiperidin-4-yl)methoxy]quinazolin-4-amine
|
|
| Clinical data | |
| Trade names | Caprelsa |
| AHFS/Drugs.com | Consumer Drug Information |
| MedlinePlus | a611037 |
| Licence data | US FDA:link |
| Pregnancy category |
|
| Legal status |
|
| Routes of administration |
Oral |
| Pharmacokinetic data | |
| Protein binding | 90–96% |
| Metabolism | CYP3A4, FMO1, FMO3 |
| Biological half-life | 120 hours (mean) |
| Excretion | 44% faeces, 25% urine |
| Identifiers | |
| CAS Registry Number | 443913-73-3 |
| ATC code | L01XE12 |
| PubChem | CID: 3081361 |
| IUPHAR/BPS | 5717 |
| DrugBank | DB08764 |
| ChemSpider | 2338979 |
| UNII | YO460OQ37K |
| ChEBI | CHEBI:49960 |
| ChEMBL | CHEMBL24828 |
| Synonyms | ZD6474 |
| Chemical data | |
| Formula | C22H24BrFN4O2 |
| Molecular mass | 475.354 g/mol |
//////
DABIGATRAN PART 2/3

-
Dabigatran etexilate (a compound of formula (I)) is the international commonly accepted non-proprietary name for ethyl 3-{[(2-{[(4-{[(hexyloxy)carbonyl]carbamimidoyl}phenyl)amino]methyl}-1-methyl-1H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino}propanoate, which has an empirical formula of C34H41N7O5 and a molecular weight of 627.73.
-
Dabigatran etexilate is the pro-drug of the active substance, dabigatran, which has a molecular formula C25H25N7O3 and molecular mass 471.51. The mesylate salt (1:1) of dabigatran etexilate is known to be therapeutically useful and is commercially marketed as oral hard capsules in the United States and in Europe under the trade mark Pradaxa™ for the prevention of stroke and systemic embolism in patients with non-valvular atrial fibrillation. Additionally, it is also marketed in Europe under the same trade mark for the primary prevention of venous thromboembolic events in adult patients who have undergone elective total hip replacement surgery or total knee replacement surgery.
-
Dabigatran etexilate was first described in U.S. Patent No. 6,087,380 , according to which the synthesis of dabigatran etexilate was carried out in three synthetic steps (see Scheme 1). Example 58 describes the condensation between ethyl 3-{[3-amino-4-(methylamino)benzoyl](pyridin-2-yl)amino}propanoate (compound II) and N-(4-cyanophenyl)glycine (compound III) in the presence of N,N‘-carbonyldiimidazole (CDI) in tetrahydrofuran to give the hydrochloride salt of ethyl 3-{[(2-{[(4-cyanophenyl)amino]methyl}-1-methyl-1H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino}propanoate (compound IV), which is subsequently reacted with ethanolic hydrochloric acid, ethanol and ammonium carbonate to give the hydrochloride salt of ethyl 3-{[(2-{[(4-carbamimidoylphenyl)amino]methyl}-1-methyl-1H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino}propanoate (compound V). Finally, example 113 describes the reaction between compound V and n-hexyl chloroformate (compound VI), in the presence of potassium carbonate, in a mixture of tetrahydrofuran and water, to give dabigatran etexilate after work-up and chromatographic purification. However, no information is given about the purity of the isolated dabigatran etexilate.
-
U.S. Patent No. 7,202,368 describes an alternative process for the synthesis of dabigatran etexilate (see Scheme 2). Example 3 describes the condensation between ethyl 3-{[3-amino-4-(methylamino)benzoyl](pyridin-2-yl)amino}propanoate (compound II) and 2-[4-(1,2,4-oxadiazol-5-on-3-yl)phenylamino]acetic acid (compound VII) in the presence of a coupling agent such as N,N‘-carbonyldiimidazole (CDI), propanephosphonic anhydride (PPA), or pivaloyl chloride, to give ethyl 3-{[(2-{[(4-{1,2,4-oxadiazol-5-on-3-yl}phenyl)amino]methyl}-1-methyl-1H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino}propanoate (compound VIII), which is subsequently hydrogenated (Example 4) in the presence of a palladium catalyst to give ethyl 3-{[(2-{[(4-carbamimidoylphenyl)amino]methyl}-1-methyl-1H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino}propanoate (compound V). Then, Example 5 describes the acylation of compound V with n-hexyl chloroformate (compound VI) to give dabigatran etexilate. Finally, Example 6 describes the conversion of dabigatran etexilate into its mesylate salt. Although the patent describes the HPLC purities of intermediate compounds II, VII, VIII and V, no information is given neither about the purity of the isolated dabigatran etexilate nor about its mesylate salt.
-
European Patent Applications EP 1966171A and EP 1968949Adescribe similar processes for the synthesis of dabigatran etexilate to that depicted in Scheme 2, but without isolating some of the intermediate compounds. HPLC purities higher than 99% are described for both dabigatran etexilate (see Examples 6B and 6C ofEP 1966171A ) and its mesylate salt (see Example 9 ofEP 1966171A and Example 7 ofEP 1968949A). However, no information is given about the structure of the impurities present in dabigatran etexilate and / or its mesylate salt.
-
PCT Patent Application WO 2010/045900 describes the synthesis of dabigatran etexilate mesylate with 99.5% purity by HPLC (Examples 3 and 4) by following a similar synthetic process to that described in Scheme 1. However, no information is given about the structure of the impurities present in the mesylate salt of dabigatran etexilate.
-
The Committee for Medicinal Products for Human use (CHMP) assessment report for Pradaxa (i.e. dabigatran etexilate mesylate salt 1:1) reference EMEA/174363/2008, as published in the European Medicines Agency website on 23/04/2008, describes (page 8) that the proposed specifications for impurities in the active substance are for some specified impurities above the qualification threshold of the ICH guideline “Impurities in new drug substances”, i.e. above 0.15%. However, no information is given about the structure of the impurities present in the mesylate salt of dabigatran etexilate.
…………..
Patent
http://www.google.com/patents/EP2522662A1?cl=en
There is still further provided by the present invention a process of preparing dabigatran etexilate mesylate, which process comprises the following synthetic steps:
wherein X is a leaving group, such as chloro.
Typically, intermediate (I) is prepared, preferably as a hydrochloride salt, by the following intermediate steps.
Example 1: Synthesis of dabigatran etexilate mesylate
a) 4-(Methylamino)-3-nitrobenzoic acid
-
300 g (1.49 mol) of 4-chloro-3-nitrobenzoic acid were suspended in 769 g of a 25-30% aqueous solution of methylamine. After heating to reflux temperature, a clear solution was obtained. The solution was kept at reflux temperature for 2 hours and total consumption of 4-chloro-3-nitrobenzoic acid was checked by TLC. The solution was cooled to room temperature, and pH was adjusted to about 1 by addition of 2M aqueous sulphuric acid. Precipitation of a yellow solid was observed, which was isolated by filtration. The filtered cake was washed with water and subsequently with methanol to obtain 331 g of wet 4-(methylamino)-3-nitrobenzoic acid as a yellow powder. Purity (HPLC, method 2): 99.1 %.
b) Ethyl 3-(2-pyridylamino)propanoate
-
75.2 g (0.80 mol) of 2-aminopyridine and 88.0 g (0.88 mol) of ethyl acrylate were dissolved in 20 mL of acetic acid. The mixture was heated to 80°C and stirred for 24 hours at the same temperature. Solvent was removed under vacuum, and the title compound was isolated by vacuum distillation (b.p. 160-172°C, 10-15 mmHg) to obtain 77.0 g of ethyl 3-(2-pyridylamino)propionate as a white solid. Yield: 49.6 %.
c) Ethyl 3-{[{1-(methylamino)-2-nitrophen-4-yl}carbonyl](pyridyn-2-yl)aminolpropanoate hydrochloride
-
50 g (0.25 mol) of 4-(methylamino)-3-nitrobenzoic acid as obtained in step (a) were suspended in a mixture of 459.2 g of thionyl chloride and 3 mL of N,N-dimethylformamide. The mixture was stirred at reflux temperature for 45 minutes. Excess thionyl chloride was removed by vacuum distillation. The residue was dissolved in 300 mL of toluene, which was subsequently removed by vacuum distillation to remove completely any residual thionyl chloride. The brownish crystalline residue obtained was dissolved in 280 mL of tetrahydrofuran at 60°C. At this point, 35.1 g of triethylamine were added to the solution. Then, a solution of 45 g (0.23 mol) of ethyl 3-(2-pyridylamino)propanoate as obtained in step (b) in 95 mL of tetrahydrofuran was added dropwise over the reaction mixture, keeping the temperature at about 30°C. The resulting mixture was stirred overnight at room temperature. Solvent was removed by vacuum distillation, and the residue was dissolved in 1 L of dichloromethane. The resulting solution was washed with 500 mL of water, 500 mL of 2M hydrochloric acid, 500 mL of saturated sodium bicarbonate and 500 mL of water. The organic phase was dried with anhydrous sodium sulfate and concentrated under vacuum. The residue was dissolved with 600 mL of ethyl acetate, and dry hydrogen chloride was bubbled into the solution until precipitation was completed. The solid was isolated by filtration and dried to obtain 63 g of the title compound, which was recrystallized in a mixture of 450 mL of ethanol and 50 mL of acetonitrile at reflux temperature. After cooling to 10°C, solid was isolated by filtration and dried to yield 44.7 g of ethyl 3-{[{1-(methylamino)-2-nitrophen-4-yl}carbonyl](pyridyn-2-yl)amino}propanoate hydrochloride as a yellow solid. Yield: 47.2 %. Purity (HPLC, method 1): 97.6 %.
d) Ethyl 3-{[{2-amino-1-(methylamino)phen-4-yl}carbonyl](pyridyn-2-yl)amino}propanoate (compound II)
-
82.2 g (0.20 mol) of ethyl 3-{[{1-(methylamino)-2-nitrophen-4-yl}carbonyl](pyridyn-2-yl)amino}propanoate hydrochloride as obtained in step (c) were suspended in 1.1 L of isopropanol, in the presence of 126.7 g of ammonium formate and 17.5 g of a 5 % Pd/C catalyst (55% water content). The reaction mixture was stirred at reflux temperature for 2.5 hours. After cooling to room temperature, the catalyst was removed by filtration, the filtrate was concentrated under vacuum, and the residue was dissolved in 1.5 L of ethyl acetate. The resulting solution was washed with 800 mL of saturated sodium bicarbonate and with 800 mL of water. The organic phase was dried with anhydrous sodium sulfate and was concentrated under vacuum to yield 44 g of ethyl 3-{[{2-amino-1-(methylamino)phen-4-yl}carbonyl](pyridyn-2-yl)amino}propanoate as a dark oil. Yield: 63.9 %. Purity (HPLC, method 2): 90.8 %.
e) 2-(4-Cyanophenylamino)acetic acid (compound III)
-
54.0 g (0.46 mol) of 4-aminobenzonitrile and 106.5 g (0.92 mol) of sodium chloroacetate were suspended in 750 mL of water, and the resulting mixture was stirred at reflux temperature for 4 hours. After cooling to room temperature, pH was adjusted to 8-9 with sodium bicarbonate. The resulting solution was washed with 2 x 200 mL of ethyl acetate, and 5M hydrochloric acid was added to the aqueous phase until pH=3. The precipitated solid was isolated by filtration, washed with 100 mL of water and dried to yield 57.1 g of 2-(4-cyanophenylamino)acetic acid as an off-white solid. Yield: 70.9 %. Purity (HPLC, method 3): 88.4 %.
f) Ethyl 3-{[(2-{[(4-cyanophenyl)amino]methyl}-1-methyl-1H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino}propanoate oxalate (salt of compound IV)
-
[0081]
-
25.7 g (0.15 mol) of 2-(4-cyanophenylamino)acetic acid as obtained in step (e) and 22.8 g (0.14 mol) of 1,1′-carbonyldiimidazole were suspended in 720 mL of tetrahydrofuran. The mixture was stirred at reflux temperature for 1 hour. Then, a solution of 44.0 g (0.13 mol) of ethyl 3-{[{2-amino-1-(methylamino)phen-4-yl}carbonyl](pyridyn-2-yl)amino}propanoate as obtained in step (d) in 180 mL of tetrahydrofuran was added dropwise over the reaction mixture. The resulting mixture was stirred overnight at reflux temperature, and the solvent was removed by distillation under vacuum. The resulting residue was dissolved in 486 mL of acetic acid and heated to reflux temperature for 1 hour. After cooling to room temperature, solvent was removed by distillation under vacuum. The resulting residue was dissolved in 450 mL of ethyl acetate, and the solution was washed with 450 mL of water. The organic phase was dried with anhydrous sodium sulfate and heated to 50-60°C. At this temperature, 15.1 g (0.17 mol) of oxalic acid were added, and the resulting mixture was stirred for 1 hour at 50-60°C. After cooling to room temperature, the precipitated solid was filtered and dried under vacuum, to yield 47.7 g of ethyl 3-{[(2-{[(4-cyanophenyl)amino]methyl}-1-methyl-1H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino}propanoate oxalate as a brownish solid. Yield: 64.8 %. Purity (HPLC, method 1): 87.9 %
g) Ethyl 3-{[(2-{[(4-{carbamimidoyl}phenyl)amino]methyl}-1-methyl-1H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino}propanoate (compound V)
-
[0084]47.7 g (83 mmol) of ethyl 3-{[(2-{[(4-cyanophenyl)amino]methyl}-1-methyl-1H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino}propanoate oxalate as obtained in step (f) and 21.8 g of p-toluenesulfonic acid were suspended in 142 g of a 10M hydrogen chloride solution in ethanol. The mixture was stirred at room temperature for 24 hours. At this point, 400 mL of ethanol were added and the resulting mixture was cooled to 0°C. Ammonia gas was bubbled at this temperature until formation of precipitate was completed. The mixture was stirred at 10°C for 2 hours, and then was stirred at room temperature overnight. Solvent was removed by distillation under vacuum. The residue was dissolved in a mixture of 400 mL of ethanol, 400 mL of water and 2.3 g of sodium hydroxide at 55°C, and was stirred at this temperature for 45 minutes. After cooling to 10°C, the mixture was stirred at this temperature for 1 hour. The solid was removed by filtration and discarded. The mother liquors were concentrated under vacuum to remove ethanol. The precipitated solid was isolated by filtration, washed with 200 mL of water and with 2 x 100 mL of acetone, to yield 34.7 g of ethyl 3-{[(2-{[(4-{carbamimidoyl}phenyl)amino]methyl}-1-methyl-1H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino}propanoate as an off-white solid. Yield: 83.4 %. Purity (HPLC, method 3): 83 %.
h) Dabigatran etexilate
-
33.7 g (67 mmol) of ethyl 3-{[(2-{[(4-{carbamimidoyl}phenyl)amino]methyl}-1-methyl-1H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino}propanoate as obtained in step (g) and 24.7 g of potassium carbonate were suspended in a mixture of 280 mL of water and 1.4 L of tetrahydrofuran. After stirring at room temperature for 15 minutes, 9.2 g (56 mmol) of hexyl chloroformate were added dropwise. The resulting mixture was stirred at room temperature for 1 hour. The organic phase was extracted, washed with 400 mL of brine and dried with anhydrous sodium sulfate. The solvent was removed under vacuum, and the resulting solid was purified by column chromatography eluting with ethyl acetate, to yield 24.9 g of dabigatran etexilate as an off-white solid. Yield: 71.0 %. Purity (HPLC, method 1): 96.3 %.
i) Dabigatran etexilate mesylate
-
18.7 g (30 mmol) of dabigatran etexilate as obtained in step (h) were suspended in 103 g of acetone. The mixture was heated to 45°C. After cooling to 36°C, a solution of 2.83 g of methanesulfonic acid in 11.6 g of acetone at 0°C was added dropwise over the reaction mixture. The reaction was stirred at 23-33°C for 90 minutes and at 17-23°C for 60 minutes. The resulting solid was isolated by filtration, washed with 97 mL of acetone and dried at 50°C under vacuum, to yield 18.7 g of dabigatran etexilate mesylate as a pale yellow solid. Yield: 86.7 %. Purity (HPLC, method 1): 98.8 %.
…………….
PATENT
http://www.google.com/patents/WO2010045900A1?cl=en
One of the advanced intermediates during the production of dabigatran is the substance of formula VI.
VI
The compound of formula VI is prepared by a reaction of substance IV with reagent V as shown in Scheme 1.
Scheme 1
The procedure described in WO 9837075 produces compound VI in the form of its base or acetate. Both these products require chromatographic purification, which is very difficult to apply in the industrial scale. This purification method burdens the process economy very much and has a negative impact on the yield.
In the next stage acidic hydrolysis of the nitrile function of compound VI and a reaction with ammonium carbonate is performed to produce the substance of formula VII. The reaction is shown in Scheme 2.
Vl VII
Scheme 2 The procedure in accordance with WO 9837075 produces substance VII in the monohydro chloride form.
When reproducing the procedure of WO 9837075 we found out, in line with WO 9837075, that compound VII prepared by this method required subsequent chromatographic purification as it was an oily substance with a relatively high content of impurities. We did not manage to find a solvent that would enable purification of this substance by crystallization.
The last stage is a reaction of intermediate VII with hexyl chloroformate producing dabigatran and its transformation to a pharmaceutically acceptable salt; in the case of the above mentioned patent application it is the methanesulfonate.
Scheme 3.
EtOH
DABIGATRAN
Example 3: Preparation of dabigatran mesylate
To 9.1 g of compound VII-2HC1 (0.016 mol) 270 ml of chloroform and 9 ml (0.064 mol) of triethylamine are added. Then, a solution of 3.1 ml (0.018 mol) of hexyl chloroformate in chloroform is added dropwise at the laboratory temperature. After one hour the reaction mixture is shaken with brine and the organic layer is separated, which is dried with sodium sulfate and concentrated. The obtained evaporation residue is crystallized from ethyl acetate. Yield: 8.6 g (86%)
This product is dissolved in acetone and an equimolar amount of methanesulfonic acid is added dropwise. The separated precipitate is aspirated and dried at the laboratory temperature. Yield: 75%; content according to HPLC: 99.5%. 27
Example 4:
Preparation of dabigatran mesylate
9 g of compound VII-HCl (0.017 mol) were dissolved in 300 ml of chloroform. 6, ml of triethylamine were added to this solution and then a solution of 3.4 ml (0.02 mol) of hexyl chloroformate in chloroform was added dropwise. After one hour the reaction mixture is shaken with brine, the organic layer is separated, which is dried with sodium sulfate and concentrated. The obtained evaporation residue is crystallized from ethyl acetate. Yield: 9.6 g (90%)
This product is dissolved in acetone and an equimolar amount of methanesulfonic acid is added dropwise. The separated evaporation residue is aspirated and dried at the laboratory temperature. Yield: 73%; content according to HPLC: 99.5%.
………………..
PATENT
http://www.google.com/patents/WO2014009966A2?cl=en
DabigatranEtexilateMesylate chemically know as N-[[2-[[[4-[[[(hexyloxy) carbonyl] amino]-iminomethyl] phenyl] amino] . methyl]-l -methyl-lH- benzimidazol-5-yl] carbonyI]-N-2- pyridinyl-beta-Alanine ethyl ester methanesulfonate having the formula I as provided below,
Formula I
is a direct thrombin inhibitor having anti – coagulant activity when administered orally.
DabigatranEtexilate is first time reported in the US patent 6087380 (hereinafter referred as US’380) in which the process fo the preparation of DabigatranEtexilate is disclosed in the Example 49, 58a and Example 59, said process for the preparation of DabigatranEtexilate is depicted below:
Dabigatran etexilate
In accordance to the process in the Patent US’380 the substance requires complex purifying operations, such as chromatography for the production of high- quality API. Further the chromatographic purification is expensive and difficult to implement in large scale. The impurity in the Dabigatran single prodrug and Dabigatran Etexilate affects the purity of the final product DabigatranEtexilateMesylate.. Hence there is a necessity to maintain the purity level of every intermediate involved in the preparation of DabigatranEtexilateMesylate.
The patent application US201 1082299 discloses a process for the preparation Dabigatran from 3- ([2-[(4-cyanophenyl amino)-methyl]- l-methyl- l H-benzimidazole-5-carbonyl]-pyridin-2-yl-amino) ethyl propionate oxalate as one of the intermediate in order to overcome the problem of the process depicted in the product pate
The patent US81 19810 discloses the process for the preparation Dabigatran from 3- ([2-[(4-cyanophenylamino)-methyl]-l-methyl-lH- benzimidazole-5-carbonyl]-pyridin-2-yl-amino) ethyl propionate hydro bromide as one of the intermediate in order to overcome the problem of the process depicted in the product patent.
The single prodrug of Dabigatran having the formula-II,
and double
which is DabigatranEtexilate are exemplified in the examples of the patent US’380. The patent US’380 has no information about the solid state properties of the single prodrug of Dabigatran and DabigatranEtexilate. However, a similar process described in a publication of Hauel et al in Journal of Medicinal Chemistry, 2002, 45, .1757 – 1766, wherein DabigatranEtexilate is characterized by 128 – 129°C.
The PCT publication WO2006131491 discloses the anhydrous form [ of DabigatranEtexilate having the melting point 135°C, anhydrous form II of DabigatranEtexilate having the melting point 150°C, and hydrate form of DabigatranEtexilate having the melting point 90°C.
The PCT publication WO2008059029 discloses anhydrous form III of DabigatranEtexilate having melting point 128°C, anhydrous form IV of DabigatranEtexilate having the melting point 133°C, and mono hydrate form I of DabigatranEtexilate having melting point 128°C and mono hydrate form II of DabigatranEtexilate having melting point 123°C.
The different forms of the single prodrug of Dabigatran and/or the DabigatranEtexilate are disclosed in the patent applications of WO2012027543, WO2012004396 and WO 2012044595.
The patent application US2007185333 discloses the process ; for the preparation of DabigatranEtexilateMesylate from the DabigatranEtexilate by adding acetone solution of , methanesulfonic acid in an acetone solution of DabigatranEtexilate.
The patent application US 200601 83779 discloses the process for the preparation of DabigatranEtexilateMesylate from the DabigatranEtexilate by adding ethylacetate solution of methanesulfonic acid in an ethylacetate solution of DabigatranEtexilate.
Example-9: Process for the preparation of DabigatranEtexilateMesylate from DabigatranEtexilate
[0086] The DabigatranEtexilate (0.04 mol) was dissolved in acetone (250.0 ml) and added Methanesulfonic acid (0.04 mol) in Ethyl acetate (25 ml) at 25-30°C. Stirred the reaction mass for 3 hrs at the same temperature, the isolated solid was filtered and washed with acetone, dried under vacuum to get the DabigatranEtexilateMesylate. Yield: 85 %, Purity: Not less than 99.0%
Example 10: Process for the preparation of DabigatranEtexilateMesylate
[0087] To a solution of DabigatranEtexilate (0.04 mol) in Acetone (8 volumes) and Ethanol (2 volumes), Methanesulfonic acid solution [Methanesulfonic acid (0.04 mol) was dissolved in Ethyl acetate (25 ml) was added at 25-30°C and stirred for 3 hrs at the same temperature. After completion of the reaction, the resultant solid was filtered, washed with acetone and dried under vacuum. Yield: 93%
……………
http://www.google.com/patents/WO2013150545A2?cl=en
l-methyl-2-|Tvi-[4-(TSi-n-hexyloxycarbonylamidino)phenyl]aminomethyl]benzimidazole- 5-yl-carboxylicacid-N-(2-pyridyl)-N-(2-ethoxycarbonylethyl)amide is commonly known as Dabigatran etexilate. Dabigatran is an anticoagulant from the class of the direct thrombin inhibitors developed by Boehringer Ingelheim and is used for the treatment of thrombosis, cardiovascular diseases, and the like. Dabigatran etexilalte mesylate was approved in both US and Europe and commercially available under the brand name Pradaxa.
Dabigatran etexilate and process for its preparation was first disclosed in WO 98/37075.
The disclosed process involves the reaction of ethyl 3-(3-amino-4-(methylamino)-N-(pyridin-2- yl)benzamido)propanoate with 2-(4-cyanophenylamino) acetic acid in the presence of N,N- carbonyldiimidazole in tetrahydrofuran to provide ethyl 3-(2-((4-cyanophenylamino)methyl)-l- methyl-N-(pyridin-2-yl)-lH-benzo[d] imidazole-5-carboxamido)propanoate, which is further converted into l-methyl-2-[N-[4-amidinophenyl]aminomethyl]benzimidazol-5-ylcarboxylicacid- N-(2-pyridyl)-N-(2-ethoxycarbonylethyl)amide hydrochloride by reacting with ammonium carbonate in ethanol, followed by treating with ethanolic hydrochloric acid. The obtained compound was reacted with n-hexyl chloroformate in presence of potassium carbonate in tetrahydrofuran/water provides Dabigatran etexilate and further conversion into its mesylate salt was not disclosed. The purity of Dabigatran etexilate prepared as per the disclosed process is not satisfactory, and also the said process involves chromatographic purification which is expensive and difficult to implement in the large scale. Hence the said process is not suitable for commercial scale up.
Moreover, the said process proceeds through the l-methyl-2-[N-[4-amidinophenyl] aminomethyl]benzimidazol-5-ylcarboxylicacid-N-(2-pyridyl)-N-(2-ethoxycarbonylethyl)amide hydrochloride (herein after referred as “Dabigatran hydrochloride”), which degrades to form impurities and resulting in the formation of Dabigatran etexilate with low purity. In view of intrinsic fragility of Dabigatran hydrochloride, there is a need in the art to develop a novel salt form of 1 -methyl-2-[N-[4-amidinophenyl]aminomethyl]benzimidazol-5-ylcarboxylicacid-N-(2- pyridyl)-N-(2-ethoxycarbonyl ethyl)amide, which enhances the purity of the final compound.
The prior reported processes disclosed in WO2012004396 and WO2008095928 Al involves the usage of inorganic salts like hydrochloride and hydrobromide salts of ethyl 3-(2-((4- cyanophenylamino)methyl)- 1 -methyl -N-(pyridin-2-yl)- 1 H-benzo[d]imidazole-5-carboxamido) propanoate (herein after referred as “cyano intermediate”) and ethyl 3-(2-((4-carbamimidoyl phenylamino)methyl)- 1 -methyl -N-(pyridin-2-yl)- 1 H-benzo[d]imidazole-5-carboxamido) propanoate (herein after referred as “amidino intermediate”). The inorganic acid addition salts are less stable when compared to the organic acid addition salts and also the process for the preparation of organic acid addition salts is very much easy when compared to inorganic acid addition salt. Inorganic acid addition salts of amidine intermediate seem to be hygroscopic in nature. Therefore, organic acid addition salts are always preferable to synthesize stable salts which in-turn enhances the purity of the final compound.
The oxalate salt of cyano intermediate was disclosed in WO2009111997. However as on date, there is no other organic acid addition salts of cyano intermediate were reported in the prior art for preparing pure Dabigatran etexilate. Henceforth, there is a need to develop a novel organic acid addition salt of cyano intermediate compound which is very much efficient when compared to its corresponding oxalate salt and that result in the formation of final compound with high purity and yield.
The process disclosed in WO 98/37075 also involves the reduction of, ethyl 3-(4- (methylamino)-3-nitro-N-(pyridin-2-yl)benzamido)propanoate (herein after referred as “nitro compound”) using Pd-C in a mixture of dichloromethane and methanol under hydrogen pressure to provide ethyl 3-(3-amino-4-(methylamino)-N-(pyridin-2-yl)benzamido)propanoate (herein after referred as “diamine compound”).
The reduction of nitro compound through catalytic hydrogenation in the presence of tertiary amine under hydrogen pressure was also disclosed in WO2009153214; and in presence of inorganic base under hydrogen pressure was also disclosed in WO2012004397.
However, most of the prior art processes proceed through catalytic hydrogenation which involves the pressure reactions. Handlings of these pressure reactions are not suitable for the large scale process. Therefore, there is a significant need in the art to provide a simple reduction process which avoids the difficulties associated with catalytic hydrogenation.
JMC, 2002, 45(9), 1757-1766 disclosed a process for the preparation of ethyl 3-(3-amino- 4-(methylamino)-N-(pyridin-2-yl)benzamido)propanoate starting from 4-(methylamino)-3- nitrobenzoic acid. The disclosed process involves the conversion of 4-(methylamino)-3- nitrobenzoic acid into its acid chloride using thionyl chloride and the obtained compound was reacted with ethyl 3-(pyridin-2-ylamino)propanoate to provide nitro compound, followed by catalytic reduction using Pd-C to provide diamine compound.
However, particularly in large scale synthesis the reduction reaction occasionally stops due to catalyst poisoning which leads to incomplete reaction and requires additional catalyst to complete the reaction. Moreover the sulfur impurities which are present in nitro compound formed due to the reaction with thionyl chloride in the previous stages of the synthesis of diamine compound are strongly influence the reaction time, quality and catalyst consumption in the manufacturing process.
Surprisingly, the problem associated with the catalytic hydrogenation and catalyst poisoning is solved by the present invention by adopting a suitable reducing agent such as Fe- acetic acid and Fe-hydrochloric acid.
The crystalline forms-I, II, V and VI of Dabigatran etexilate oxalate were disclosed in WO2008043759 and WO2011110876.
The crystalline forms-Ill, IV and V of Dabigatran etexilate fumarate were disclosed in WO2008043759 and WO2011110876.
Various different salts for Dabigatran etexilate and their polymorphs were reported in WO98/37075, WO03074056, WO2005028468, WO2006114415, WO2008043759, WO2011110876, WO2012027543 and WO2012044595.
The process for the preparation of crystalline form-I of Dabigatran etexilate mesylate was described in WO2005028468 and WO2012027543.
HPLC analysis of Innovator Tablet
The present inventors has also analyzed the Pradaxa 110 mg tablet having Lot no: 808809 and compared with dabigatran etexilate mesylate obtained from the present invention and found that, the impurity profile of both the products are similar to each other i.e., amide impurity, despyridyl ethyl ester etc. are well present even in Pradaxa tablet. Henceforth, we can presume that these impurities are known from the art.
Amide Impurity: 0.31%; Despyridyl ethyl ester: 0.10%; Deshexyl Impurity: 0.08%. HPLC Method of Analysis:
a) Dabigatran etexilate (Formula-1) and Dabigatran etexilate mesylate (Formula-la):
Apparatus: A liquid chromatographic system is to be equipped with variable wavelength
UV-detector; Column: Zorbax Eclipse XDB CI 8, 100 X 4.6mm, 3.5 μιη θΓ Equivalent; Flow Rate: 1.0 mL/min; Wavelength : 300 nm; Column temperature: 25°C; Injection volume: 5 μΐ,; Run time: 50 minutes; Auto sampler temperature: 5°C; Buffer: Dissolve 0.63gm of Ammonium formate in lOOOmL of Milli-Q- Water and mix well. Adjust its pH to 8.2 with Ammonia and filtered through 0.22 μιη nylon membrane and degas it. Mobile phase-A: Buffer; Mobile phase- B: Acetonitrile: Water (80:20) v/v; Diluent: N,N-Dimethylformamide; Needle wash: Diluent; Elution: Gradient. b) Ethyl 3-(2-((4-cyanophenylamino)methyl)-l-methyl-N-(pyridin-2-yl)-lH-benzo[d] imidazole-5-carboxamido)propanoate methanesulfonate (Formula-10)
Apparatus : A liquid chromatograph is equipped with variable wavelength UV- Detector; Column: Zorbax SB CN 150 x 4.6mm, 5μπι (or) Equivalent (Make: Agilent and PNo: 883975- 905); Flow Rate: 1.0 mL / min; Column temperature: 25°C; Wave length: 290 nm; Injection volume: 5 μΐ-.; Run time: 60 minutes; Elution: Gradient; Diluent: Water: Acetonitrile (70:30) v/v; Needle wash: Diluent; Buffer: Weigh accurately about 2 g of 1 -Octane sulphonic acid sodium salt anhydrous and add 5 mL of Ortho phosphoric acid in 1000 mL of Milli-Q- Water and mix well, filter this solution through 0.22 μηι^ΐοη membrane and sonicate to degas; Mobile Phase- A: Buffer(100%);Mobile Phase- B: Acetonitrile: Methanol (90: 10) v/v. c) Ethyl 3-(2-((4-carbamimidoylphenylamino)methyl)-l-methyl-N-(pyridin-2-yl)-lH- benzo[d] imidazole-5-carboxamido)propanoate methanesulfonate (Formula-11)
Apparatus : A liquid chromatographic system is to be equipped with variable wavelength UV- Detector and Integrator; Column : Zodiac CI 8 250 X 4.6 mm, 5 μηι (or) equivalent (Make: Zodiac and PNo. ZLS.C18.46.250.0510 ); Flow Rate: 1.0 mL/min; Wavelength: 290 nm; Column temperature: 25°C; Injection Volume: 5μί; Run time: 55 min; Elution: Gradient;
Buffer: Take 5 mL of Ortho phosphoric acid(85%) and 2 g of 1 -Octane sulfonic acid sodium salt anhydrous in 1000 mL of Milli-Q-water and adjust its pH to 2.5 with Triethyl amine filter, through 0.22 μπι Nylon membrane filter paper and sonicate to degas it; Mobile Phase-A: Buffer(l 00%) Mobile Phase-B: Acetonitrile: Water (90: 10) v/v; Diluent : Water: Acetonitrile (80:20) v/v.
Morphology: Method of analysis: Samples were mounted on aluminium stubs using double adhesive tape, coated with gold using HUS-5GB vacuum evaporation and observed in Hitachi S-3000 N SEM at an acceleration voltage of 10KV.
Following are the impurities observed during the preparation of Dabigatran etexilate mesylate.
Deshexyl Impurity Despyridyl Ethyl Ester
Methyl Carbamate Ethyl Carbamate
The present invention is schematically represented as follows:
Formula-2 ene
Formula-6
Fe-AcOH
Formula-7
Dabigatran etexilate Dabigatran etexilate Mesylate The process described in the present invention was demonstrated in examples illustrated below.
Example-13: Preparation of Dabigatran etexilate (Formula-1)
n-hexanol (30.8 g) was added to a solution of N, N-carbonyldiimidazole (61.15 g) and dichloromethane (360 ml) at 15-25°C and stirred for 3 hours. The organic layer was washed with water followed by sodium chloride solution. Distilled off the solvent from the organic layer completely under reduced pressure to get amide compound. Acetonitrile (157.5 ml) was added to the obtained amide compound. This was added to a mixture of ethyl 3-(2-((4- carbamimidoylphenylamino)methyl)-l-methyl-N-( yridin-2-yl)-lH-benzo[d]imidazole-5- carboxamido)propanoate mesylate compound of formula- 11 (90 g), potassium carbonate (62.5 g), acetonitrile (378 ml) and water (252 ml) at 25-35°C. The reaction mixture was heated to 40- 50°C and stirred for 8 hours. After completion of the reaction, both the organic and aqueous layers were separated; the organic layer was cooled to -5 to +5°C and stirred for 2 hours. Filtered the precipitated solid washed with acetonitrile and water. The obtained compound was dissolved in a mixture of acetone (270 ml) and acetonitrile (270 ml) at 45-50°C. Cooled the reaction mixture to 25-30°C and water (360 ml) was added to it. Filtered the obtained solid and dissolved in the mixture of dichloromethane and sodium chloride solution at 35-40°C. Both the organic and aqueous layers were separated; the organic layer was distilled under reduced pressure and then co-distilled with ethyl acetate. The obtained crude compound was dissolved in ethyl acetate (540 ml) by heating it to 70-80°C and stirred for 30 minutes. Filtered the reaction mixture, the filtrate was cooled to 35-45°C and ethanol (8 ml) was added to the reaction mixture. The reaction mixture was again cooled to 25-35°C and stirred for 3 hours. Filtered the precipitated solid and then dried to get pure title compound.
Yield: 44 g; MR: 128-131 °C. Purity by HPLC: 99.63%.
……………….
http://www.google.com/patents/WO2014020555A2?cl=en
EXAMPLE 6
Preparation of dabigatran etexilate mesylate: l-methyl-2-[N-[4-( -n-hexyloxycarbonylamidino)phenyl] amino methyl]benzimidazol-5- yl-carboxylicacid-N-(2-pyridyl)-N-(2-ethoxycarbonyl ethyl) amide (100 gm) was dissolved acetone (1000 ml) under heating at 25-35 °C. A solution of methane sulfonic acid (13.77 gm) in acetone (100 ml) was added to the reaction mixture. The solution is filtered and after the addition of acetone cooled to approximately 20° C. The precipitated product was filtered and washed with acetone then dried at 50° C under reduced pressure.
Wet weight : 0.120-0.140 kg
Dry weight : 0.90-1.0 kg
Yield (W/W) : 0.90-1.0
Theoretical Yield (w/w) : 1.15
Percentage Yield : 78.2-86.9%
………………….
| US20050095293 * | Sep 3, 2004 | May 5, 2005 | Boehringer Ingelheim Pharma Gmbh Co. Kg | Administration form for the oral application of poorly soluble drugs |
| US20070185173 * | Dec 21, 2006 | Aug 9, 2007 | Georg Zerban | Process for the Preparation of the Salts of 4-(Benzimidazolylmethylamino)-Benzamides |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| WO2014020555A2 * | Jul 31, 2013 | Feb 6, 2014 | Alembic Pharmaceuticals Limited | An improved process for the preparation of dabigatran etexilate mesylate |
| WO2014009966A2 * | Jul 5, 2013 | Jan 16, 2014 | Rao Davuluri Ramamohan | An improved process for the preparation of dabigatran etexilate mesylate and its intermediates thereof |
| WO2014009966A3 * | Jul 5, 2013 | Mar 6, 2014 | Rao Davuluri Ramamohan | An improved process for the preparation of dabigatran etexilate mesylate and its intermediates thereof |
| EP1966171A1 | Dec 20, 2006 | Sep 10, 2008 | Boehringer Ingelheim International GmbH | Improved process for the preparation of 4-(benzimidazolylmethylamino)-benzamides and the salts thereof |
| EP1968949A1 | Dec 20, 2006 | Sep 17, 2008 | Boehringer Ingelheim International GmbH | Improved process for the preparation of the salts of 4-(benzimidazolylmethylamino)-benzamides |
| US6087380 | Feb 18, 1998 | Jul 11, 2000 | Boehringer Ingelheim Pharma Kg | Disubstituted bicyclic heterocycles, the preparations and the use thereof as pharmaceutical compositions |
| US7202368 | Jun 9, 2005 | Apr 10, 2007 | Boehringer Ingelheim International Gmbh | Process for the preparation of 4-(benzimidazolymethylamino) benzamidines |
| WO2000005207A1 * | Jul 20, 1999 | Feb 3, 2000 | Boehringer Ingelheim Pharma | Substituted phenylamidines with antithrombotic action |
| WO2007071742A1 * | Dec 20, 2006 | Jun 28, 2007 | Boehringer Ingelheim Int | Improved process for the preparation of 4-(benzimidazolylmethylamino)-benzamides and the salts thereof |
| WO2010045900A1 | Oct 26, 2009 | Apr 29, 2010 | Zentiva, K.S. | A method for the preparation of dabigatran and its intermediates |
| Reference | ||
|---|---|---|
| 1 | * | European Medicines Agency (EMEA): “CHMP ASSESSMENT REPORT FOR Pradaxa“, , 1 January 2008 (2008-01-01), pages 1-36, XP55003938, London Retrieved from the Internet: URL:http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/000829/WC500041062.pdf [retrieved on 2011-08-01] |
| 2 | * | HAUEL N H ET AL: “STRUCTURE-BASED DESIGN OF NOVEL POTENT NONPEPTIDE THROMBIN INHIBITORS“, JOURNAL OF MEDICINAL CHEMISTRY, AMERICAN CHEMICAL SOCIETY, US, vol. 45, no. 9, 1 January 2002 (2002-01-01), pages 1757-1766, XP001098844, ISSN: 0022-2623, DOI: DOI:10.1021/JM0109513 |
| CN103058920A * | Jan 21, 2013 | Apr 24, 2013 | 上海应用技术学院 | Preparation method of 3-(2-pyridineamino)ethyl propionate |
| CN1861596A * | May 18, 2006 | Nov 15, 2006 | 复旦大学 | Process for synthesizing antithrombin inhibitor of non-asymmetric non-peptide kind |
| CN101875626A * | Nov 6, 2009 | Nov 3, 2010 | 广东光华化学厂有限公司;北京理工大学 | Method for synthesizing N-benzyl maleimide from immobilized supported acid catalyst |
| EP2522662A1 * | May 11, 2011 | Nov 14, 2012 | Medichem, S.A. | Dabigatran etexilate and related substances, processes and compositions, and use of the substances as reference standards and markers |
| JP2004315371A * | Title not available |
See full gatran series at………………http://apisynthesisint.blogspot.in/p/argatroban.html
Lemborexant

Lemborexant
E2006
CAS Number: 1369764-02-2
MF C22 H20 F2 N4 O2
MW 410.42
Chemical Name: (1R, 2S) -2 – {[(2,4-dimethylpyrimidin-5-yl) oxy] methyl} -2- (3-fluorophenyl ) N (5-fluoropyridin-2-yl) cyclopropanecarboxamide
Cyclopropanecarboxamide, 2-[[(2,4-dimethyl-5-pyrimidinyl)oxy]methyl]-2-(3-fluorophenyl)-N-(5-fluoro-2-pyridinyl)-, (1R,2S)-
(1R,2S)-2-{[(2,4-dimethylpyrimidin-5-yl)oxy]methyl}-2-(3-fluorophenyl)-N-(5-fluoropyridin-2-yl)cyclopropanecarboxamide
Indication: Insomnia
Company: Eisai
Orexin receptors are G-protein coupled receptors found predominately in the brain. Their endogenous ligands, orexin-A and orexin-B, are expressed by neurons localized in the hypothalamus. Orexin-A is a 33 amino acid peptide; orexin-B consists of 28 amino acids. (Sakurai T. et al., Cell, 1998, 92, 573-585). There are two subtypes of orexin receptors, OXi and OX2; OX) binds orexin-A preferentially, while OX2 binds both orexin-A and -B. Orexins stimulate food consumption in rats, and it has been suggested that orexin signaling could play a role in a central feedback mechanism for regulating feeding behavior (Sakurai et al., supra). It has also been observed that orexins control wake-sleep conditions (Chemelli R.M. et al., Cell, 1999, 98, 437-451). Orexins may also play roles in brain changes associated with opioid and nicotine dependence (S.L. Borgland et al, Neuron, 2006, 49, 598-601; C.J. Winrow et al., Neuropharmacology, 2010, 58, 185-194), and ethanol dependence (J.R. Shoblock et al, Psychopharmacology, 2011, 215, 191-203). Orexins have additionally been suggested to play a role in some stress reactions (T. Ida et al, Biochem. Biophys. Res. Commun., 2000, 270, 318- 323).
Compounds such as (lR,2S)-2-(((2,4-dimethylpyrimidin-5-yl)oxy)methyl)-2-(3- fluorophenyl)-N-(5-fluoropyridin-2-yl)cyclopropanecarboxamide (Compound A, below) have been found to be potent orexin receptor antagonists, and may be useful in the treatment of sleep disorders such as insomnia, as well as for other therapeutic uses.


The orexin/hypocretin receptors are a family of G protein-coupled receptors and consist of orexin-1 (OX1) and orexin-2 (OX2) receptor subtypes. Orexin receptors are expressed throughout the central nervous system and are involved in the regulation of the sleep/wake cycle. Because modulation of these receptors constitutes a promising target for novel treatments of disorders associated with the control of sleep and wakefulness, such as insomnia, the development of orexin receptor antagonists has emerged as an important focus in drug discovery research. Here, we report the design, synthesis, characterization, and structure–activity relationships (SARs) of novel orexin receptor antagonists. Various modifications made to the core structure of a previously developed compound (–)-5, the lead molecule, resulted in compounds with improved chemical and pharmacological profiles. The investigation afforded a potential therapeutic agent, (1R,2S)-2-{[(2,4-dimethylpyrimidin-5-yl)oxy]methyl}-2-(3-fluorophenyl)-N-(5-fluoropyridin-2-yl)cyclopropanecarboxamide (E2006), an orally active, potent orexin antagonist. The efficacy was demonstrated in mice in an in vivo study by using sleep parameter measurements.

E. Preparation of Compounds of Formula V
((lR,2S)-2-(((2,4-dimethylpyrimidin-5-yI)oxy)methyl)-2-(3-fluorophenyl)-cyclopropyl) methanol (11). ((lR,2S)-2-(3-fluorophenyl)-2-((tosyloxy)methyl)cyclopropyl)metliyl acetate (8, 11.05 g, 0.028 mol, 1.0 equiv.), 2,4-dimethylpyrimidin-5-ol (3.74 g, 0.030 mol, 1.07 equiv.), and cesium carbonate (22.94 g, 1.8 equiv.) were dissolved in ACN (110.5 mL), under nitrogen. The solution was stirred vigorously and heated to 65-70 °C for 2-3 hours. The reaction was monitored by HPLC and TLC (EtO Ac/Heptane = 1/1). Once complete, aqueous 1 N NaOH solution (71.82 mL) was added to the reaction mixture. The reaction mixture was stirred at 20-25 °C for 10-16 h, and was monitored by HPLC and TLC (EtO Ac/Heptane = 1/1). Once the hydrolysis reaction was complete, the reaction mixture was diluted with MTBE (110.50 mL) and stirred for at least 15 min. The aqueous layer was back extracted once with MTBE (55.25 mL). The organic layers were combined and washed once with saturated aqueous NaCl solution (33.15 mL). The solvent was removed under reduced pressure to afford the title compound; ((lR,2S)-2-(((2,4- dimethylpyrimidin-5-yl)oxy)methyl)-2-(3-fluorophenyl)cyclopi pyl)methanol: (11, 8.51 g).
((lR,2S)-2-(((2,4-dimethylpyrimidin-5-yl)oxy)methyl)-2-(3-fluorophenyl)- cyclopropyl)methanol: 1H NMR (500 MHz, DMSO-d6) δ 8.21 (s, 1H), 7.33 (td, J = 8.0, 6.5 Hz, 1H), 7.20 (d, J= 7.9 Hz, 1H), 7.19 – 7.14 (m, 1H), 7.01 (ddd, J= 8.3, 2.6, 1.2 Hz, 1H), 4.63 (t, J = 5.4 Hz, 1H), 4.36 (dd, J= 22.5, 10.5 Hz, 2H), 3.72 – 3.61 (m, 2H), 2.45 (s, 3H), 2.22 (s, 3H), 1.51 – 1.43 (m, 1H), 1.23 (dd, J= 8.9, 5.0 Hz, 1H), 1.01 (dd, J= 6.0, 5.3 Hz, 1H). 13C NMR (126 MHz, DMSO-dfi) δ 162.48 (d, JCF = 243.0 Hz), 158.91, 156.26, 149.51, 147.47 (d, JCF = 7.5 Hz), 139.85, 130.35 (d, JCF = 8.5 Hz), 124.72 (d, JCF = 2.5 Hz), 115.54 (d, JCF = 21.3 Hz), 113.43 (d, JCF = 20.9 Hz), 72.73, 60.70, 29.23, 28.64, 24.94, 18.77, 17.06.
HRMS Calculated for C17H20FN2O2 [M+H]+ 303.1590; found 303.1517.
F. Preparation of Compounds of Formula VII
(lR,2S)-2-(((2,4-dimethylpyrimidin-5-yl)oxy)methyl)-2-(3-fluorophenyl)cyclopropane- carboxylic acid (13). ((lR,2S)-2-(((2,4-dimethylpyrimidin-5-yl)oxy)methyl)-2-(3- fluorophenyl)cyclopropyl)methanol (11, 87.5 g, 290 mmol, 1.0 equiv.) was dissolved in toluene (390 mL). To the mixture was added pH 7 buffer (107 g, prepared from 4.46 g of sodium phosphate dibasic and 7.79 g of sodium phosphate monobasic in 94.4 mL of water) and 2,2,6,6- tetramethylpiperidine 1-oxyl (TEMPO) (0.93 g, 5.9 mmol, 0.02 equiv.). The mixture was cooled to 0 °C and sodium hypochlorite solution (5% active chlorine, 383 mL, 304 mmol, 1.05 equiv.) was added dropwise, maintaining the internal temperature below 9 °C. The mixture was allowed to warm to room temperature and stirred for 2 h. To the mixture was added aqueous hydrochloric acid (2.0 M, 8.73 mL, 0.05 equiv.) followed by a solution of sodium chlorite (36.0 g, 318 mmol, 1.1 equiv.) in water (87 mL), maintaining the internal temperature below 26 °C. The mixture was stirred at room temperature for 4 h, and then cooled to 10 °C. A solution of sodium thiosulfate (92 g, 579 mmol, 2.0 equiv.) in water (177 mL) was added, maintaining the internal temperature below 20 °C. The mixture was stirred for 20 min, and then aqueous sodium hydroxide solution (4 N, 87 mL, 348 mmol, 1.2 equiv.) was added to achieve ca. pH = 13. The mixture was heated to 80 °C for 4 hours, then cooled to room temperature. Stirring was halted and the phases allowed to split. The lower aqueous phase was collected and the upper organic phase was washed once with 4 N sodium hydroxide solution (17 mL). The combined aqueous phases were acidified with aqueous hydrochloric acid solution (4 N, 17 mL) to pH = 4 and extracted with ethyl acetate (2 x 470 mL). The combined organic phases were washed with ca. 20% aqueous NaCl solution (175 mL). The organic phases were concentrated by rotary evaporation to yield 96.84 g of crude oil. A portion (74 g) of this crude oil was dissolved in acetonitrile (400 mL) and concentrated to dryness by rotary evaporation. Another portion of acetonitrile (400 mL) was added and the mixture was again concentrated to dryness. To the residue was added acetonitrile (370 mL). The mixture was heated to 65 °C resulting in a clear solution. The mixture was cooled to room temperature, then to 0 °C and held at this temperature for 6 h. The mixture was filtered and the wet cake was washed with acetonitrile (2 x 74 mL). The cake was dried under vacuum with a nitrogen sweep, then in a vacuum oven at 20 torr and 40 °C to afford (lR,2S)-2-(((2,4- dimethylpyrimidin-5-yl)oxy)methyl)-2-(3-fluorophenyl)cyclopropanecarboxylic acid (13, 56.9 g, 80% yield) as an off-white crystalline solid.
(lR,2S)-2-(((2,4-dimethylpyrimidin-5-yl)oxy)methyl)-2-(3-fluoi phenyl)- cyclopropanecarboxylic acid: 1H NMR (500 MHz, DMSO-d6) δ 12.47 (s, 1H), 8.17 (s, 1H), 7.39 (td, J= 8.0, 6.4 Hz, 1H), 7.29 (d, J= 7.9 Hz, 1H), 7.27 – 7.22 (m, 1H), 7.10 (td, J – 8.3, 2.1 Hz, 1H), 4.63 (d, J= 10.2 Hz, 1H), 4.30 (d, J= 10.2 Hz, 1H), 2.46 (s, 3H), 2.26 (s, 3H), 2.13 (dd, J= 7.7, 6.6 Hz, 1H), 1.63 – 1.54 (m, 2H); 13C NMR (126 MHz, DMSO-d6) δ 172.65, 162.48 (d, JCF = 243.6 Hz), 159.08, 156.24, 149.45, 145.15 (d, JCF = 7.5 Hz), 139.60, 130.71 (d, JCF = 8.5 Hz), 124.79 (d, JCF = 2.6 Hz), 115.60 (d, JCF = 21.8 Hz), 114.32 (d, JCF = 20.8 Hz), 71.15, 33.92 (d, JCF = 2.0 Hz), 26.46, 24.96, 19.72, 18.70.
HRMS Calculated for Ci7Hi8FN203 [M+H]+ 317.1301; found 317.1298.
G. Preparation of Compounds of Formula IX
(lR,2S)-2-(((2,4-dimethylpyrimidin-5-yl)oxy)methyl)-2-(3-fluorophenyl)-N-(S- fluoropyridin-2-yl)cyclopropanecarboxamide (14). (lR,2S)-2-(((2,4-dimethylpyrimidin- 5-yl)oxy)methyl)-2-(3-fluorophenyl)-cyclopropanecarboxylic acid (13, 12.80 g, 0.040 mol, 1.0 equiv.), and 2-amino-5-fluoiOpyridine (4.76 g, 0.0425 mol, 1.05 equiv.) were dissolved in ethyl acetate (102.4 mL), under nitrogen. The solution was cooled to 0-5 °C, and N,N- diisopropylethylamine (14.10 mL, 0.081 mol, 2.0 equiv.) was added to the reaction mixture while maintaining the internal temperature at 0-15 °C. The reaction mixture was stirred at 0-10 °C for 20-30 minutes. n-Propylphosphonic anhydride (T3P; 50% w/w solution in ethyl acetate, 36.1 g, 1.4 equiv.) was added to the reaction mixture while maintaining the internal temperature at 0-15 °C. The reaction was stirred at 20-25 °C for at least 20-24 hour and monitored by HPLC and TLC (EtO Ac/Heptane = 1/1). Upon completion of the reaction, the reaction mixture was cooled to 0-5 °C and then was quenched with water (64.0 mL) while maintaining the internal temperature below 10-15 °C. The aqueous layer was back extracted once with MTBE (76.8 mL). The organic layers were combined and washed once with saturated aqueous NaHC03 solution (38.4 mL) and once with water (38.4 mL). The organic layer was polish filtered and the filter rinsed with MTBE (12,8 mL). The organic layer was then concentrated under reduced pressure to a minimum stirrable volume. Ethyl acetate (60.8 mL) was added to the reaction mixture and the mixture was heated to no more than 50 °C to achieve a clear solution. n-Heptane (86.3 mL) was added slowly with agitation. The reaction mixture was cooled to 20-25 °C, and the suspension was stirred for at least 1 h at 20-25 °C and then stirred at least for 1 h at 0-5 °C. The suspension was filtered and the cake was washed two times with 5 : 1 heptane/ethyl acetate (2 x
12.8 mL). The cake was dried under nitrogen and/or vacuum to provide the title compound, (lR,2S)-2-(((2,4-dimethylpyrimidin-5-yl)oxy)methyl)-2-(3-fluoiOphenyl)-N-(5-fiuoropyridin^ yl)cyclopropanecarboxamide, (14, 12.54 g, >99% ee) as a white to off white solid.
(lR,2S)-2-(((2,4-dimethylpyrimidin-5-yl)oxy)methyl)-2-(3-fluoiOphenyl)-N-(5- fluoropyridin-2-yl)cyclopropanecarboxamide:
1H NMR (500 MHz, DMSO-d6) δ 11.19 (s, 1H), 8.31 (d, J = 3.0 Hz, 1H), 8.12 (s, 1H), 7.94 – 7.85 (m, 1H), 7.62 (tt, J = 8.7, 3.1 Hz, 1H), 7.44 (dd, J = 10.6, 1.5 Hz, 1H), 7.41 – 7.40 (m, 1H), 7.39 (s, 1H), 7.14 – 7.06 (m, 1H), 4.67 (d, J = 10.2 Hz, 1H), 4.29 (t, J= 9.9 Hz, 1H), 2.63 (t, J= 7.0 Hz, 1H), 2.38 (s, 3H), 2.03 (s, 3H), 1.76 – 1.64 (m, 1H), 1.49 (dd, J = 8.0, 4.8 Hz, 1H); 13C NMR (125 MHz, DMSO-d6) δ 168.68, 161.98 (d, JcF = 242.3 Hz), 158.46, 155.15, 155.38 (d, JCF = 247.9 Hz), 148.90, 148.51, 145.00 (d, JCF = 7.7 Hz), 139.37, 135.15 (d, JCF = 24.9 Hz), 130.06 (d, JCF = 8.4 Hz), 125.05 (d, JCF = 19.5 Hz), 124.70 (d, JCF = 2.6 Hz), 115.71 (d, JCF = 21.7 Hz), 114.20 (d, JCF = 4.1 Hz), 113.70 (d, JCF =
20.9 Hz), 70.80, 34.09 (d, JCF = 1.9 Hz), 26.90, 24.38, 18.37, 17.78.
HRMS Calculated for C22H21F2N402 [M+H]+ 411.1627; found 411.1632.
Production Example 14
(1R, 2S) -2 – Synthesis of {[(2,4-dimethyl-pyrimidin-5-yl) oxy] methyl} -2- (3-fluorophenyl) cyclopropanecarboxylic acid (Prep14-6)
(1) (1S, 5R) -1- (3- fluorophenyl) -3-hexane-2-one to oxabicyclo [3.1.0] (Prep14-1)
3-fluorophenyl acetonitrile (70g) was dissolved in THF (500ml), ice – salt bath under cooling, was added dropwise NaHMDS (1000ml, 1.06M). After allowed to stir 1 hour, R – (-) – it was added dropwise epichlorohydrin (40.6ml) (approximately 10 minutes, the internal temperature <10 ℃). After it was allowed to stirred for 2 hours (maintained before and after the internal temperature 0 ℃), and stirred at room temperature for 14 hours. The reaction was I was dropping a small amount of water cooled with ice. The reaction solution was concentrated under reduced pressure, the residue in ethanol (700ml), 1N potassium hydroxide aqueous solution (1000ml) was added and heated to reflux for 5 hours. After returning to room temperature, it was added 5N hydrochloric acid (400ml), and stirred for 1 hour at 60 ℃. The reaction mixture was concentrated under reduced pressure, it was added thereto to carry out a liquid separation with ethyl acetate and water. The organic layer saturated aqueous sodium hydrogen carbonate solution, it was washed successively with saturated sodium chloride aqueous solution. Dried over magnesium sulfate, and the solvent was concentrated under reduced pressure. The residue was purified by silica gel column chromatography to obtain a purified by (n- heptane-ethyl acetate) The title compound (84.9g).
1 H-NMR (400MHz, CDCl 3) δ (ppm): 1.41 (t, J = 5.2Hz, 1H), 1.64 (dd, J = 8.0,5.2Hz, 1H), 2 .56-2.63 (m, 1H), 4.30 (d, J = 9.2Hz, 1H), 4.47 (dd, J = 9.2,4.8Hz, 1H), 6.96- 7.02 (m, 1H), 7.16-7.21 (m, 2H), 7.28-7.35 (m, 1H).
(2) (1S, 2R) -1- (3- fluorophenyl) cyclopropane-1,2-dimethanol (Prep14-2)
THF- methanol compound Prep14-1 (72.7g) (440ml-220ml) sodium borohydride solution (25g) was added at 0 ℃, and the mixture was stirred for 65 hours at room temperature. Under ice-cooling, water and 5N hydrochloric acid were added to the reaction solution, followed by extraction with ethyl acetate. The organic layer was washed with a saturated sodium chloride aqueous solution, and then dried with magnesium sulfate. The solvent was concentrated under reduced pressure, the residue was purified by silica gel column chromatography to obtain a purified by (n- heptane-ethyl acetate) The title compound (72.7g).
1 H-NMR (400MHz, CDCl 3) δ (ppm): 0.80 (t, J = 5.0Hz, 1H), 1.10 (dd, J = 8.6,5.0Hz, 1H), 1 .62-1.71 (m, 1H), 3.41 (t, J = 11.4Hz, 1H), 3.58 (d, J = 12.0Hz, 1H), 4.12-4.25 ( m, 2H), 6.90-6.96 (m, 1H), 7.08-7.14 (m, 1H), 7.16-7.21 (m, 1H) 7.24-7.32 (m, 1H).
(3) {(1S, 2R) – [2- (tert- butyldiphenylsilyloxy) -1- (3-fluorophenyl) cyclopropyl]} methanol (Prep14-3)
Compound Prep14-2 a (42.4g) was dissolved triethylamine (33.0ml) in dichloromethane (216ml), was cooled to -20 ℃, was added dropwise tert- butyldiphenylsilyl chloride (56.3ml) (about 30 minute, almost at the same time insoluble matter is deposited with the completion of the dropping). After stirring for 1 hour, further stirred at room temperature for 20 hours.Water was added to the reaction mixture, and the mixture was extracted with dichloromethane. Washed with water and dried over anhydrous magnesium sulfate. The solvent was evaporated under reduced pressure, and the residue was purified by silica gel column chromatography to obtain a purified by (n- heptane ethyl acetate) The title compound (67.8g).
1 H-NMR (400MHz, CDCl 3) δ (ppm): 0.73 (t, J = 5.2Hz, 1H), 1.04 (dd, J = 8.4,5.2Hz, 1H), 1 .09 (s, 9H), 1.48-1.53 (m, 1H), 3.52 (t, J = 12.0Hz, 1H), 3.56 (dd, J = 9.6,1. 6Hz, 1H), 3.70 (dd, J = 9.6,1.6Hz, 1H), 4.18 (t, J = 12.0Hz, 1H), 4.20 (dd, J = 12.0 , 5.2Hz, 1H), 6.93 (tdd, J = 8.0,2.4,1.2Hz, 1H), 7.11 (dt, J = 9.6,2.4Hz, 1H), 7.20 (dt, J = 8.0,1.2Hz, 1H), 7.28 (td, J = 8.0,6.0Hz, 1H), 7.37-7.49 (m, 6H) , 7.69-7.74 (m, 4H).
(4) {(1R, 2S) -2 – {[(-5- 2,4- dimethyl-pyrimidin-yl) oxy] methyl} -2- (3-fluorophenyl) cyclopropyl} methanol (Prep14-4)
Compound Prep14-3 (581mg), triphenylphosphine (1.3g) and Preparation Example 4 to give 2,4-dimethyl – THF (10ml) solution of diisopropyl azodicarboxylate pyrimidin-5-ol (183mg) ( The 0.316ml) was added dropwise at 0 ℃, and the mixture was stirred at room temperature for 2 days. The reaction mixture was concentrated under reduced pressure, silica gel column chromatography (n- heptane: ethyl acetate = 19: 1 → 7: 3) was purified by. The resulting (1S, 2R) -2- (tert- butyldiphenylsilyloxy-methyl) -1 – {[(2,4-dimethyl-pyrimidin-5-yl) oxy] methyl} -1- (3-fluorophenyl) cyclopropane was dissolved in THF (15ml), tetrabutylammonium fluoride (1M-THF solution: 1.61ml) was added dropwise at room temperature and stirred at room temperature for 14 hours. The reaction mixture was concentrated under reduced pressure, silica gel column chromatography (n- heptane: ethyl acetate = 10: 1 → 0: 1) to obtain purified by the title compound (238mg).
1 H-NMR (400MHz, CDCl 3) δ (ppm): 1.00 (t, J = 5.6Hz, 1H), 1.25-1.33 (m, 1H), 1.78-1.88 (m, 1H), 2.39 (s, 3H), 2.61 (s, 3H), 3.58 (dd, J = 12.0,9.6Hz, 1H), 4.02-4.11 (m, 1H), 4.12 (d, J = 10.4Hz, 1H), 4.43 (d, J = 9.6Hz, 1H), 6.92-6.98 (m, 1H), 7 .10-7.16 (m, 1H), 7.18-7.23 (m, 1H), 7.29 (td, J = 8.0,6.0Hz, 1H), 8.00 (s, 1H).
(4 alternative method)
((1R, 2S) -2 – {[(2,4- dimethyl-pyrimidin-5-yl) oxy]} methyl] -2- (3-fluorophenyl) cyclopropyl} methanol (Prep14-4) (alternative method)
Triethylamine (14.5ml) was added in dichloromethane (200ml) solution of compound Prep14-3 (41.3g), cooled to 0 ℃. It was added dropwise methanesulfonyl chloride (7.34ml), and stirred for 1 hour. Water was added to the reaction mixture, and the mixture was extracted with dichloromethane. Dried over anhydrous sodium sulfate, and the solvent was evaporated under reduced pressure. The resulting residue in acetonitrile (200ml) solution obtained in Production Example 4- (2) 2,4-dimethyl – pyrimidin-5-ol (14.1g) and cesium carbonate (61.8g) was added, 70 ℃ It was heated to. After 4 hours of stirring at 70 ℃, the reaction solution was cooled to 0 ℃, tetrabutylammonium fluoride (1M-THF solution: 190ml) was added dropwise, and the mixture was stirred for 1 hour at room temperature. Water was added to the reaction mixture, and the mixture was extracted with ethyl acetate. Dried over anhydrous sodium sulfate, and the solvent was evaporated under reduced pressure. The residue was purified by NH- silica gel column chromatography (n- heptane: ethyl acetate = 9: 1 to 1: 1) to give the title compound (20.7g) was purified by.
(5) (1R, 2S) -2 – {[(2,4- dimethyl-pyrimidin-5-yl) oxy] methyl} -2- of (3-fluorophenyl) cyclopropane carbaldehyde (Prep14-5)
Oxalyl dichloromethane solution of chloride (137μl) a (7ml) was cooled to -78 ℃, there was added dropwise dimethyl sulfoxide (226μl) (internal temperature below -60 ℃). After stirring for 10 minutes at the same temperature, dichloromethane (3ml) solution of the compound to the reaction mixture Prep14-4 (238mg) was dropped at -78 ℃, and the mixture was stirred at the same temperature for 30 minutes. After stirring for 15 minutes triethylamine (671μl) was added to the reaction mixture, and the temperature was raised to room temperature. Saturated sodium chloride aqueous solution was added to the reaction mixture, and the mixture was extracted with ethyl acetate. The organic layer was dried anhydrous magnesium sulfate and concentrated under reduced pressure to give the crude title compound (236mg).
1 H-NMR (400MHz, CDCl 3) δ (ppm): 1.67 (dd, J = 8.0,4.8Hz, 1H), 1.96-2.00 (m, 1H), 2.36 (s, 3H), 2.49-2.55 (m, 1H), 2.59 (s, 3H), 4.19 (d, J = 9.6Hz, 1H), 4.44 (d, J = 10.0Hz, 1H), 6.97-7.04 (m, 1H), 7.14-7.20 (m, 1H), 7.21-7.25 (m, 1H), 7.30 -7.37 (m, 1H), 7.95 (s, 1H), 9.87 (d, J = 3.2Hz, 1H).
(6) (1R, 2S) -2 – {[(2,4- dimethyl-pyrimidin-5-yl) oxy] methyl} -2- (3-fluorophenyl) cyclopropanecarboxylic acid (Prep14-6)Compound Prep14- 5 (18.9g) and 2-methyl-2-butene (26.1ml), sodium dihydrogen phosphate the (9.07g) was dissolved in acetone-water mixed solvent (200ml · 40ml), sodium chlorite ( 6.26g) and I were added little by little. After stirring for 2 hours at room temperature, the reaction solution was concentrated under reduced pressure. The precipitated solid was filtered off, washed with dichloromethane, and the solvent was evaporated under reduced pressure. The residue was purified by silica gel column chromatography (n- heptane: After 1, ethyl acetate: ethyl acetate = 1: 1-0 methanol = 10: 1) to give the title compound (16.2g) was purified by.
1 H-NMR (400MHz, CDCl 3) δ (ppm): 1.55 (dd, J = 8.4,5.6Hz, 1H), 1.76 (t, J = 5.6Hz, 1H), 2 .25 (dd, J = 8.4,6.4Hz, 1H), 2.33 (s, 3H), 2.55 (s, 3H), 4.47 (t, J = 9.6Hz, 1H) , 4.50 (d, J = 9.6Hz, 1H), 6.99 (tdd, J = 8.0,2.4,1.2Hz, 1H), 7.21 (dt, J = 9.6 , 2.4Hz, 1H), 7.26 (td, J = 8.0,1.2Hz, 1H), 7.32 (td, J = 8.0,6.0Hz, 1H), 8.21 ( s, 1H).
Compound Prep14-6 can be prepared directly by the following method from the compound Prep14-4.
Compound Prep14-4 (300mg) and TEMPO (5mol%, 7.74mg) was dissolved in phosphate buffer solution of acetonitrile · pH6.4 (5ml · 5ml), 2N- hydrochloric acid (150μl), sodium chlorite (180mg ) and it was added. After heating to 40 °, 5w% of the hypochlorite solution (2mol%, 26.5μl) were added and stirred for 2 hours. Cooled to room temperature, the reaction mixture was stirred for 5 minutes was added an excess of 2-methyl-2-butene in. The reaction solution was extracted with dichloromethane, the solvent was distilled off under reduced pressure, the residue was purified by silica gel column chromatography (n- heptane: ethyl acetate = 1: 1 to 0: After 1, ethyl acetate: methanol = 9: 1) in was purified to give the title compound (215mg).
Example 95
(1R, 2S) -2 – {[(2,4- dimethyl-pyrimidin-5-yl) oxy] methyl} -2- (3-fluorophenyl) -N- (5- fluoro-2-yl) cyclopropane The synthesis of carboxamide (95)
Acid Prep14-6 a (226mg) was dissolved in dichloromethane (10ml), oxalyl chloride (122μl), and stirred for 1 hour at room temperature was added DMF (a few drops). The reaction mixture was concentrated under reduced pressure to give the crude acid chloride. N in THF (10ml) solution of 2-amino-5-fluoro pyridine (96.1mg), N- diisopropylethylamine (283μl) was added mixture was heated to 60 ℃, the temperature of intact dropwise a THF solution of the crude acid chloride in it was allowed to stir for 1 hour. The reaction mixture was allowed to cool to room temperature and allowed to stir for 1 hour, after which the reaction mixture was concentrated under reduced pressure, partitioned between ethyl acetate and water, the organic layer was separated. The organic layer was dried over anhydrous magnesium sulfate, and the filtrate was concentrated under reduced pressure. The residue was purified by NH- silica gel column chromatography (n- heptane: ethyl acetate = 2: 1) to give diethyl ether to the obtained target compound was added. The precipitated solid was filtered dried to give the title compound (130mg).
1 H-NMR (400MHz, d-DMSO) δ (ppm): 1.46-1.50 (m, 1H), 1.68 (t, J = 6.0Hz, 1H), 2.01 (s, 3H), 2.36 (s, 3H), 2.59-2.63 (m, 1H), 4.27 (d, J = 10.4Hz, 1H), 4.66 (d, J = 10. 4Hz, 1H), 7.06-7.11 (m, 1H), 7.37-7.44 (m, 3H), 7.60-7.65 (m, 1H), 7.85-7. 89 (m, 1H), 8.11 (s, 1H), 8.30 (d, J = 3.2Hz, 1H), 11.20 (brs, 1H)
MS [M + H] + = 411
Synthesis coming…….watch out
References
- Christopher, John A (2014). “Small-molecule antagonists of the orexin receptors”. Pharmaceutical Patent Analyst 3 (6): 625–638.doi:10.4155/ppa.14.46. ISSN 2046-8954.
- Cristoph Boss, Catherine Ross (2015). “Recent Trends in Orexin Research – 2010 to 2015”. ScienceDirect.doi:10.1016/j.bmcl.2015.05.012.
- Boss, Christoph (2014). “Orexin receptor antagonists – a patent review (2010 to August 2014)”. Expert Opinion on Therapeutic Patents 24 (12): 1367–1381.doi:10.1517/13543776.2014.978859. ISSN 1354-3776.
- AdisInsight. “Lemborexant”. Springer. Retrieved 2015-05-23.

External links

| Systematic (IUPAC) name | |
|---|---|
|
(1R,2S)-2-[(2,4-dimethylpyrimidin-5-yl)oxymethyl]-2-(3-fluorophenyl)-N-(5-fluoropyridin-2-yl)cyclopropane-1-carboxamide
|
|
| Clinical data | |
| Legal status |
|
| Identifiers | |
| CAS Registry Number | 1369764-02-2 |
| ATC code | None |
| PubChem | CID: 56944144 |
| ChemSpider | 34500836 |
| Chemical data | |
| Formula | C22H20F2N4O2 |
| Molecular mass | 410.417 g/mol |
////////
How to document a Product Transfer? Example templates!
DRUG REGULATORY AFFAIRS INTERNATIONAL

All participants of the GMP training course “GMP-compliant Product Transfer” will receive a special version of the Guideline Manager CD including documents and templates useable for site change projects. Read more.
According to the European GMP-Rules, written procedures for tranfser activities and their documentation are required. For example, a Transfer SOP, a transfer plan and a report are now mandatory and will be checked during inspections.
As a participant of the GMP education course “GMP-compliant Product Transfer” in Prague, from 20-22 October 2015 you will receive a special version of the Guideline Manager CD with a special section concerning product transfers. This section contains, amongst others, a Transfer SOP and a template for a Transfer Plan. Both documents are in Word format and can immediately be used after adoption to your own situation.

Regulatory Guidance Documents like the WHO guideline on transfer of technology in pharmaceutical manufacturing and the…
View original post 36 more words

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....