Home » Uncategorized (Page 70)
Category Archives: Uncategorized
GMP/GDP: When will I be inspected by the Authorities?
DRUG REGULATORY AFFAIRS INTERNATIONAL

Various competent authorities are performing inspections. But who is subject to such an inspection?
GMP Inspections are carried out at Manufacturer Licence Holders
A manufacturer of medicinal products must meet Good Manufacturing Practice (GMP) standards. These standards are defined in various laws and regulations. In the EU the compliance with these regulations is checked and assessed by the national competent authorities. The overall goal is to have medicinal products of consistent high quality that meet the requirements of the marketing authorisation (MA) or product specification.
If a company supplies product to the USA, the U.S. Food and Drug Administration (FDA) might inspect the site assuring that drugs, medical devices, certain active pharmaceutical ingredients (APIs) and biological products manufactured in foreign countries and intended for U.S. distribution are in compliance with the applicable U.S. law and regulations.
GDP Inspections are carried out at Wholesale Dealer Licence Holders
Good Distribution Practice…
View original post 312 more words
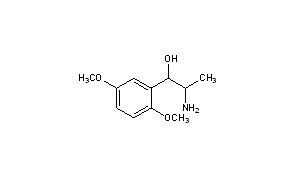
1R,2S-Methoxamine

1R,2S-methoxamine, also known as L-erythro-methoxamine
CAS 13699-29-1
- Molecular Weight, 211.26, C11 H17 N O3
HYDROCHLORIDE
(1R,2S)-isomer HCl salt of 1 -(2,5-dimethoxyphenyl)-2-amino-1 -propanol also called as (1R, 2S)methoxamine hydrochloride
CAS 16122-04-6
Used as a pressor agent, as a vasoconstrictor, as a nasal decongestant, in ophthalmology and also found very effective in the treatment of faecal incontinence.
treatment of relief of fecal incontinence and anal itch (pruritis ani) , particularly for patients who have had a major bowel resection and reanastomosis .
Anal or fecal incontinence is the inability to voluntarily control the passage of feces or gas through the anus. It may occur either as fecal soiling or as rare episodes of incontinence for gas or watery stools. It is a very distressing condition that can result in self-inflicted social isolation and despair.
Conventional treatments for fecal incontinence include drug therapy to improve stool consistency, such as morphine, loperamide and codeine phosphate to reduce gut motility, and laxatives to soften stools and relieve constipation. Biofeedback training is another treatment which involves muscle strengthening exercises to improve anal canal resting pressure, and squeeze pressure, and to teach symmetry of anal canal function. The most common form of treatment however, is surgical repair, such as the creation of a neo-sphincter which involves grafting on muscle from other parts of the anus, or a colostomy. (Gastroenterology in Practice, Summer 1995, pl8- 21; Dig Dis 1990; 8:179-188; and The New England Journal of Medicine, April 1992, pl002-1004) . In mild cases of anal leakage, the patient will often try and plug the anus with a ball of cotton wall.
In Gut, 1991, 32, p.345-346 it was reported that two thirds of patients with idiopathic faecal incontinence had a decreased anal resting pressure resulting from an abnormal internal sphincter function. In many incontinent patients, the internal anal sphincter was found to be abnormally thin, while others had an external anal sphincter defect. It has also been reported that in vi tro contractile response of the internal anal sphincter to noradrenaline is decreased in incontinence, (Br. J. Surg. 1992, vol 79, August, p829-832; Digestive Diseases and Sciences, vol 38, no. 11, Nov. 1993, pl961-1969) . A further discussion of the innervation and control of the internal anal sphincter and drugs which can increase or decrease the normal anal resting pressure, is discussed in the text book Coloproctology and the Pelvic Floor (Butterworths) , second edition, 1992, at chapter 3 p37-53; Automic Control of Internal Anal Sphincter; and Journal of Clinical Investigation 1990, 86: p424-429.
In Surgery 1990; 107: p311-315 sodium valproate was found to be useful in the treatment of minor incontinence after ileoanal anastomosis.
It has now surprisingly been found that fecal incontinence and anal itch can be resolved by treatment with α adrenergic agonists, nitric oxide synthase inhibitors, prostaglandins F2α, dopamine, morphine, β-blockers such as propranolol, and 5-Hydroxytryptamine (5-HT) .
This is surprising since it was always thought that once an anal sphincter began functioning abnormally, the patient would require major surgery.
In this way the anal leakage is reduced or eliminated without the patient having to undergo major surgery.
Accordingly in a first aspect of the invention there is provided use of a physiologically active agent selected from an α adrenergic agonist, nitric oxide synthase inhibitor, prostaglandin F2α, dopamine, morphine, β-blockers, and 5- Hydroxytryptamine in the preparation of a medicament for the treatment or prophylaxis of fecal incontinence or anal itch.
The agents of the invention appear to at least partially treat the incontinence by increasing the resting pressure of the internal anal sphincter. Preferred agents are λ adrenergic agonists, nitric oxide synthase inhibitors, and prostaglandins F2α.
Examples of suitable aλ adrenergic agonists are nor- adrenalin, methoxamine, but particularly preferred is phenylephrine .
Examples of suitable F2α prostaglandin are dinoprost and carboprost.
Examples of suitable NO synthase inhibitors are
NG-monnoommeetthhyyll–LL–aarrggiinn:ine (L-NMMA) , and NG-nitro-L-arginine methyl ester ( -NAME)
The medicament can contain a single active agent or a combination of any of the above active agents.
Nitric Oxide (NO) synthase inhibitors such as LNMMA have previously been suggested for the therapeutic treatment of septic shock.
The prostaglandins, along with thromboxanes and leukotrienes are all derived from 20 -carbon polyunsaturated fatty acids and are collectively termed eicosanoids. F2α prostaglandins are derived in vivo from the endoperoxide prostaglandin H2which is in turn derived from leukotrienes. Clinically, F2α prostaglandins such as dinoprost and carboprost are used as uterine stimulants in the termination of pregnancy, missed abortion or the induction of labour.
Phenylephrine (an αx adrenergic agonist) is used as a mydriatic in ophthalmology, and as a decongestant , for example, in cold and flu remedies.
However there has been no suggestion to the inventors knowledge of using any of these active agents to treat fecal incontinence or anal itch. As used herein “fecal incontinence” includes all types of anal leakage from minor leakage or ‘spotting’ through moderate leakage, to major instances of faecal incontinence, and includes neurogenic, active, urge and passive incontinence.
More particularly the class of incontinent patients who will benefit most from the present invention are those with idiopathic incontinence and those whose incontinence is at least partly due to a weakness of either the internal or external anal sphincter, especially those with a normal or low maximum anal pressure and a structurally intact internal anal sphincter muscle, such as with an abnormally thin sphincter. However patients with minor structural damage such as a fragmented sphincter would still benefit from the invention. Not only incontinent patients with a damaged or abnormal internal sphincter can be treated, but also patients with a damaged or abnormal external sphincter since the increase in the internal anal resting tone induced by the invention will compensate for a poorly functioning external sphincter.
Another class of patients who particularly benefit from the invention are post-surgical patients who have had major bowel resection and reanastomosis . For example patients with ileoanal pouch (restorative proctocolectomy) , coloanal (with or without colonic pouch) anostomosis, lower anterior resection, and colectomy with ileorectal anastomosis.
The damage to the sphincter could be caused by trauma, such as experienced in child birth, surgical operations, or road traffic accidents. Furthermore it is also believed that incontinence caused by primary internal anal degeneration can also be relieved by the invention.
Anal leakage also often leads to pruritis of the anus and therefore by reducing or eliminating the leakage, the pruritis or anal itch is also relieved or prevented. Furthermore, as a result of the increased anal resting pressure, the patient no longer has the discomfort of distended anal sphincter muscles.
Methoxamine contains two chiral carbons and thus exists in four isomeric forms. Of all the isomeric forms, the studies revealed (1R,2S)- isomer to be therapeutically active.
US patent 2359707 describes the process for the synthesis of racemic β-(2,5-dimethoxy phenyl)-P-hydroxy-isopropyl amine in neutral, acid salt and its derivative from 2,5- dimethoxy propiophenone by treatment with methylnitrite in diethyl ether medium to obtain 2,5-dimethoxy-a-isonitrosopropiophenone hydrochloride. It is further reduced with palladium on carbon to yield β-(2,5-dimethoxyphenyl)-p-ketoisopropylamine hydrochloride and then with platinum black to get p-(2,5-dimethoxyphenyl)-β- hydroxyisopropyl amine hydrochloride. The described process for di-methoxamine HC1 is not cost-effective, due to the use of two expensive catalysts (platinum black and palladium carbon), solvent diethyl ether and involves more number of steps. The other drawback being it is racemic mixture and cannot be used directly as drug. The process described did not specify the quality of the product.
In US patent 3284490 the processes for racemic N-alkyl derivatives of methoxamine are described from dl-methoxamine.
JP 63165348 describes process for production of optically active l-(2,5- dimethoxyphenyl)-2-aminophenol by resolving racemic compound with the use of optically active L-N-acetylleucine as resolving agent. The disadvantages of the process are less yield, low quality and use of expensive naturally occurring amino acid, which prevents from employing this method on commercial scale.
WO 03/055474 A1 discloses mainly, the use of (1R, 2S)-methoxamine in the treatment of faecal incontinence at low doses without local or systemic side effects when used topically. The patent also described the synthesis of (1R, 2S)-methoxamine, from L- alanine, by protecting the amino group using methylchloroformate, converting carboxy
group of the N-protected alanine into an acid chloride insitu followed by reaction with an amine to produce an N-protected (S)-alanine amide and coupling that compound with a brominated 2,5-dimethoxybenzene in the presence of n-butyllithium or a magnesium based reagent to give (S)-amino-l-(2,5-dimethoxy-phenyl)-l-propanone, the amino group of which is protected .The reduction of the N-protected propanone was carried out using dimethylphenylsilane and the protecting group was removed by treatment with potassium hydroxide. Other method adopted in the patent to isolate (1R,2S)methoxamine is by separation of racemic methoxamine using chiral column.

The prior art suffers with some of the disadvantages like using n-butyllithium, which is pyrophoric, expensive and causes hazards to commercial scale. Also, the separation of racemic Methoxamine using chiral column mentioned in the patent can be considered for
isolating small quantities of the required isomer for analytical purposes but cannot be adopted on commercial scale for production of the drug.
US Patent 5962737 described stereospecific synthesis of the racemic threo isomers of 2- nitro-1 -phenylpropanols by reacting benzaldehyde derivative with nitroalkane in the presence of a tertiary amine and reducing 2-nitro-l-phenylpropanols with lithium aluminium hydride to 2-amino-l-phenylpropanols. Also described is phase transfer resolution of racemic mixtures of 2-amino-l-phenylpropanol and its derivatives into their optically pure isomers by reacting with the mono alkali metal salt of tartaric acid ester in a two phase system of a hydrocarbon and water. The specification further describes optically pure isomer D-threo 2-amino-( 1 -dialkoxy or alkoxy)phenylpropanol by resolution of dl- threo 2-amino-( 1 -dialkoxy or alkoxy)phenylpropanol by using dibenzoyltartaric acid. The synthesis of the product (lS,2S)-threo 2-amino-(l-dialkoxy or alkoxy) phenyl propanol involves the use of expensive and hazardous chemicals like LAH making the process technically and commercially difficult for implementation.
Paper
Journal of the American Chemical Society (1984), 106(16), 4629-30
http://pubs.acs.org/doi/pdf/10.1021/ja00328a062
PATENT
http://www.google.com/patents/EP2275099A1?cl=en
EXAMPLE 3Synthesis of 1R,2S-Methoxamine(S)-N-Methoxycarbonyl alanine
To a stirred solution of L-alanine (300g, 3.37 mol sodium hydroxide (1N, 1800 cm3) at 0°C in an ice bath was added dropwise, over 2 hours, methyl chloroformate (274 cm3, 3.54 mol). The pH of the solution was maintained at 9 by the addition of sodium hydroxide (5N). The reaction mixture was stirred at 0°C for 3 hours whereupon it was acidified to pH 1 by the addition of phosphoric acid solution (15%) and extracted with diethyl ether (5 x 1000 cm3). The combined organic extracts were dried (MgSO4) and concentrated under reduced pressure to yield the product as a viscous green oil (386 g, 78%). 1H NMR (250 MHz; C2HCl3) 1.48 (3H, d, J7.25, CH3), 3.72 (3 H, s, COCH3), 4.40 (1 H, quintet, J7.25, CH), 5.31 (1 H, bs, NH).
(S)-N-Methoxycarbonyl alaninedimethylamide
To a stirred solution of MeOC-alanine (227 g, 1.54 mol) and dimethylformamide (DMF) (25 cm3) in dry dichlorourethane (DCM) (2000 cm3) at 0°C was added dropwise oxalyl chloride (146 cm3, 1.62 mol) over a period of 2 hours. The solution was stirred at 0°C until the evolution of gasses ceased whereupon a basic solution of dimethylamine (676 g, 7.70 mol) in NaOH (3 N, 2000 cm3) was added. The aqueous layer was extracted with diethyl ether (2 x 500 cm3) and the combined organic layers dried (MgSO4) and concentrated under reduced pressure to give the product as a white crystalline solid which required no further purification (230 g, 86%). 1H NMR (250 MHz; C2HCl3) 1.33 (3 H, d, J6.75, CH3), 2.99 3 H, s, OCH3) 3.08, (3 H, s, OCH3), 3.66 (3 H, s, COCH3), 4.66 (H, quintet, J7.00, CH), 5.75 (1 H, d, J5.75, NH).
(S)-2-[(Methoxycarbonyl)amino]-1-(2,5-dimethoxyphenyl)-1-propanone.
To a THF (1000 cm3) solution of bromo-2,5-dimethoxybenzene (55 g, 0.25 mol) at -20°C under nitrogen was addedn-butyl lithium (100 cm3, 2.5 M in hexanes, 0.25 mol). The mixture was stirred at -20°C for 0.75 hours, whereupon a THF (100 cm3) solution of amide (30 g, 0.17 mol) was added via cannula. The solution was stirred at -20°C for 2 hours and was then allowed to warm to room temperature over 1 hour and quenched by the addition of ammonium chloride solution (700 cm3). The solution was diluted with diethyl ether (1000 cm3) and the organic layer was dried (MgSO4) and concentrated under reduced pressure to give a yellow oil. The product was purified by dry flash chromatography on silica (eluant 4:1 hexane/ethyl acetate then 3:2 hexane/ethyl acetate) to give the product as a white crystalline solid (45 g, 98%). 1H NMR (250 MHz; C2HCl3) 1.36 (3 H, d, J7.0, CH3), 3.70 (3 H, s, COCH3), 3.82 (3 H, s, OCH3), 3.92 (3 H, s, OCH3), 5.43 (1 H, quintet, J 7.3, H-2), 5.80 (1 H, bs, NH), 6.94 (1 H, d, J 9.0, ArH), 7.10 (1 H, dd, J 9.0, 3.3, ArH), 7.32 (1 H, d, J 3.3, ArH).
(1R,2S)-2-[(Methoxycarbonyl)amino]-1-(2,5-dimethoxyphenyl)-1-propanol.
To a stirred solution of ketone i.e. (S)-2-[(methoxycarbonyl)amino]-1-(2,5-dimethoxyphenyl)-1-propanone (20 g, 74.9 mmol) and dimethylphenyl silane (10.7 g, 78.6 mmol) in dry DCM (500 cm3) at 0°C in an ice bath was added dropwise trithioroacetic acid (TFA) (50 cm3). The solution was stirred at 0°C for 1 h and then quenched by the addition of sodium hydroxide (500 cm3, 1 N). The organic layer was dried and concentrated under reduced pressure to give a yellow oil which solidified on standing. This solid was crystallized from ether/hexane to give the product as a white crystalline solid (15.6 g, 75%).1H NMR (250 MHz; C2HCl3) 1.03 (3 H, d, J7.0, CH3), 3.04 (1 H, d, J4.3, OH), 3.68 (3 H, s, COCH3), 3.78 (3 H, s, OCH3), 3.80 (3 H, s, OCH3), 3.94-3.99 (1 H, m, H-2), 5.05-5.15 (2 H, m, H-1 and NH), 6.72-6.85 (2 H, m, ArH) 6.97 (1 H, d, J 2.0, ArH).
(1,R,2S)-Methoxamine.
To a stirred solution of methoxycarbonyl (MeOC) protected alcohol i.e. (1R,2S)-2-[(methoxycarbonyl)amino]-1-(2,5-dimethoxyphenyl)-1-propanol (4.0 g, 14.9 mmol) in methanol (175 cm3) was added a solution of KOH (4.06 g, 72.8 mmol in water (60 cm3). The solution was cooled and acidified with phosphoric acid (15% v/v). The solution was extracted with DCM (2 x 50 cm3) and the aqueous layer basified by the addition of K2CO3. The aqueous layer was extracted with diethyl ether (5 x 50 cm3) and the combined ethereal extracts dried (MgSO4) and concentrated under reduced pressure to give the product as a clear yellow oil (1.9 g, 61%), 1H NMR (250 MHz; C2HCl3) 0.84 (3 H, d, J 7.0, CH3), 3.19-3.22 (1 H, m, H-2), 3.71 (6 H, s, 2 x OCH3), 4.67 (1 H, d, J 5.0, H-1), 6.66-6.72 (2 H, m, ArH), 6.92 (1 H, d, J 2.5, ArH).
(1R, 2S)-Methoxamine hydrochloride.
To an ice cooled solution of (1R,2S)-methoxamine (1.9 g, 9.00 mmol) in anhydrous diethyl ether (30 cm3) was passed a stream of dry HCl gas for 45 mins. The resultant precipitate was filtered by suction, washed with cold diethyl ether and dried under nitrogen to yield the title compound as a white solid. (1.5 g, 68%). 1H NMR (250 MHz; [C2H3]2SO) 0.89 (3 H, d, J 6.8, CH3), 3.37-3.42 (1 H,m,H-2), 3.71 (3 H, s, OCH3), 3.75 (3 H, s, OCH3), 5.12 (1 H, s, H-1), 5.92 (1 H, d, J 4.3, OH), 6.84 (1 H, dd, J 8.8, 3.0, ArH), 6.92-7.00 (2 H, m, ArH); HPLC.
Analytical Method for the Analysis of Methoxamine
The following method was used to analyse methoxamine samples.
Method
-
Column : Cyclobond I RSP 250 x 4.6 mm Column temperature : 23°C Mobile phase : 0.1% Tetraethylammonium pH 4.1* 95%v/v : Acetonitrile 5%v/v Flow rate : 0.6 ml/min Solution
Concentration :5 mg/l Injection volume : 2.5 µl to 20 µl Detection : UV 230 nm *Tetraethylammonium acetate pH 4.1 was prepared fresh daily.
Example 2 above allows the complete assignment of the methoxamine isomers as shown below:
PATENT
INDIAN 1020/CHE/2011
BY


The Managing Director of Malladi Drugs & Pharmaceuticals, Prashant Malladi (left), with the Chief Executive Officer, V. N. Gopalakrishnan

V.N Gopalakrishnan
CEO at Malladi Drugs & Pharmaceuticals Ltd

Prabhakaran Ranganathan
Vice President (Operations) at Malladi Drugs and Pharmaceuticals Limited

The present invention further provides an improved process for the preparation of (JS, 2S)-Methoxamine HC1 of formula (6) from (1R, 2S)-methoxamine by treating with acetic anhydride in toluene medium followed by acid hydrolysis and basification to obtain (IS, 2S)-Methoxamine base which is further acidified to form (1S,2S)- Methoxamine HC1 (6).
The present invention further provides an improved process for the preparation of (1R, 2R)-Methoxamine HC1 of formula (5) from its diastereomer (1S, 2R)-methoxamine HC1 of formula (2) by treating with acetic anhydride in toluene medium followed by acid hydrolysis and basification to obtain (1R, 2R)-Methoxamine base which is further acidified to form (1R, 2R)-Methoxamine HC1 (5).
The following examples illustrate the invention.
EXAMPLES
Example 1
Preparation of l-(2,5-Dimethoxyphenyl)propan-l-one (8)
Aluminium chloride (127.4 g; 0.955 mol) was added to dichloromethane (420 mL) in a round bottomed flask under nitrogen atmosphere. The reaction mixture was cooled to -5 °C; 1,4-dimethoxybenzene (100 g; 0.724 mol) was added slowly within 15-30 minutes. Propionic chloride (87 g; 0.94 mol) dissolved in dichloromethane (245 mL) was added slowly within 2 hours. The reaction mass was allowed to stir for 2 hours and then was quenched in crushed ice (1 kilo) and HC1 (75 mL) at 0 – 5 °C. Separated the layers and the organic layer was washed with 5% sodium hydroxide solution, dried and concentrated (140 g; colorless liquid); Purity by HPLC : 99.04%
Spectroscopic interpretation
The structure of the product, l-(2,5-Dimethoxyphenyl)propan-l-one was confirmed with the help of the following spectroscopic data.
a) IR (cm-1) (KBr)
Aromatic C-H stretch at 3071, aliphatic C – H stretch at 2938, C = O stretch at 1674, benzenoid bands at 1609 and 1584, C – O stretch at 1223, C – H out of plane bending of tri-substituted benzene ring at 814,719.
b) 1H NMR(CDCb, 300 MHz) (δH)
1.16 (3H, t, -CH2-CH3), 3.0 (2H, q, -CH2-CH3), 3.78 (3H, s, -OCH3), 3.85 (3H, s, -OCH3), 6.83 – 7.72 (3H, m, aromatic protons)
c) 13C NMR (CDCb, 300 MHz) (δC)
8.44 (-CH2-CH3), 37.03 (-CH2-CH3), 55.74 (-OCH3), 56.01 (-OCH3), 113.09 – 153.41 (aromatic carbons), 202.96 (C=O)
d) Mass spectrum (ESI, methanol)
[M+Na]+ at m/z 217 (9), [M+H]+ at m/z 195 (100).
Example 2
Preparation of l-(2,5-Dimethoxyphenyl)-2-nitrosopropan-l-one (9) l-(2,5-Dimethoxyphenyl)propan-l-one (100 g; 0.515 mol) was added to dichloromethane (660 mL) in a round bottomed flask under nitrogen atmosphere. Butylnitrite (46.6 g; 0.52 mol) was slowly added in about 30 minutes at 30 – 35 °C. Diethyl ether (60.2 mL) was added to the reaction mixture and dry HC1 gas was purged for about 4 hours at 30 – 35 °C. The reaction mass was maintained for 12 hours and then concentrated under vacuum The residue obtained (60 g; Pale yellow crystalline powder); Purity by HPLC: 99.81%; mp: 104-107 °C
Spectroscopic interpretation
The structure of the product, l-(2,5-Dimethoxyphenyl)-2-nitrosopropan-l-one was confirmed with the help of the following spectroscopic data
a) IR (cm1) (KBr)
O-H stretch at 3250 (broad), aromatic C-H stretch at 3024, aliphatic C – H stretch at 2934, C = O stretch at 1688, C = N stretch at 1645, benzenoid bands at 1589 and 1504, C-O stretch at 1231, C-H out of plane bending of tri-substituted benzene ring at 745,702.
b) 1H NMR(CDCb, 300 MHz) (δh)
2.07 (3H, s, -C-CH3), 3.72 (3H, s, -OCH3), 3.76 (3H, s, -OCH3), 6.84-6.99 (3H, m, aromatic protons), 8.89 (1H, bs, OH)
c) 13C NMR (CDCb, 300 MHz) (δC)
9.16 (-C-CH3), 55.81 (-OCH3), 56.34 (-OCH3), 113.09 – 153.27 (aromatic carbons), 157.07 (C=N-OH); 193.32 (CO)
d) Mass spectrum (ESI, methanol) [M+H]+ at m/z 224 (100)
Example 3
Preparation of dl-erythro-methoxamine HC1 (10)
Raney nickel (50 g); iso-propyl alcohol (250 mL) were added to the autoclave. l-(2,5- Dimethoxyphenyl)-2-nitrosopropan-1 -one (100 g; 0.448 mol) was added slowly at 50 – 55 °C by simultaneously purging the flask with hydrogen at 2-3 Kilo pressure. When hydrogen consumption ceases, the catalyst was filtered and the filtrate was concentrated. iso-Propyl alcohol (200 mL) was added to the concentrated mass followed by acidification with HC1 to obtaindl-erythro-methoxamine HC1 (70 g; white crystalline solid)
Spectroscopic interpretation
The structure of the product, dl-erythro-methoxaxmne HC1 was confirmed with the help of the following spectroscopic data.
a) IR (cm1) (KBr)
O-H stretch at 3409, aromatic C-H stretch at 3010, aliphatic C – H stretch at 2914, HN-H str. at 2574 and 2467, benzenoid bands at 1615 and 1569, C-N stretch at 1279, C-O stretch at 1216, C-H out of plane bending of 1,2,4-tri- substituted benzene ring at 812.
b) 1H NMR (DMSO-d6, 300 MHz) (δH)
1.0 (3H,d, -CH-CH3), 3.74 (3H, s, -OCH3), 3.77 (3H, s, -OCH3), 4.89 (1H, q, -CH-CH3),6.1 (1H, d, -CH-OH), 6.87-7.01 (3H, m, aromatic protons), 8.06 (3H, bs, HN-H) The -OH proton appears to have exchanged with the solvent.
c) 13C NMR (DMSO-d6, 300 MHz) (δc)
14.75 (-CH-CH3), 52.12 (-OCH3), 55.70 (-OCH3), 55.70 (-CH-CH3), 67.25 (CH-OH), 111.89 – 153.16 (aromatic carbons)
d) Mass spectrum (ESI, methanol)
[M+H)+ at m/z 212 (100), [M-H2O]+ at m/z 194 (56).
Example 4
Preparation of(JR,2S)-Metboxamine HC1 (1) and (1S, 2R)-Methoxamine HC1 (2) dl-erythro-methoxamine HC1 (117g; 0.47 mol) was dissolved in water (350 mL) at 30-35 °C. The clear solution obtained was basified using 50% sodium hydroxide solution. dl-erythro-Methoxaumne (3) was extracted into dichloromethane (150 mL) and concentrated. Mixture of methanol/DMSO (4:1; 1650 mL) was added and the mass was heated to 50 °C. L-(+)-Tartaric acid (71.1g; 0.47mol) was added slowly and the temperature of the mass was further raised to 70 °C for complete dissolution. The mass was cooled to 35 °C and maintained for 48 hours. (IR,2.S)-Methoxamine tartrate complex (80 g) precipitated was filtered. From the filtrate on concentration was obtained (1S,2R)- methoxamine tartrate complex (82 g) (IR,25)-Methoxamine tartrate complex was added to water (250 mL) at 35 °C, basified to 12 – 13 pH with 50% sodium hydroxide solution. Dichloromethane (200 mL) was added and stirred for 30 min. Separated the org layer, dried over sodium sulphate and concentrated completely under vacuum at 45° C. Iso-Propyl alcohol (150 mL) was added, charcaolized and filtered. The clear filtrate was acidified with 20%IPA HC1 to yield (1R, 2S)-Methoxamine HC1 which was filtered and dried (48 g); White crystalline powder; Purity by HPLC : 100%; Chiral purity : 100 %; mp : 172-175 °C; [α]D: -47.94° (c = 2% in MeOH)
Spectroscopic interpretation
The structure of the product, (1R,2S)-Methoxamine HC1 was confirmed with the help of the following spectroscopic data.
a) IR (cm1) (KBr)
O-H stretch at 3300, aromatic C-H stretch at 3065, aliphatic C-H stretch at 2938, HN-H str. at 2693 and 2580, benzenoid bands at 1609 and 1578, C-N stretch at 1277, C-O stretch at 1217, C-H out of plane bending of 1,2,4-tri- substituted benzene ring at 818.
b) 1H NMR (DMSO-d6 300 MHz) (δH)
0.91 (3H,d, -CH-CH3), 3.71 (3H, s, -OCH3), 3.75 (3H, s, -OCH3), 5.14 (1H, m, -CH- NH3+), 5.95 (1H, d, -CH-OH), 6.83-7.01 (3H, m, aromatic protons), 8.25 (3H, bs, HN-H) The -OH proton appears to have exchanged with the solvent.
c) 13C NMR (DMSO-d6, 300 MHz) (δC)
II. 44 (-CH-CH3), 49.22 (-OCH3), 55.24 (-OCH3), 55.70 (-CH-CH3), 66.49 (CH-OH),
III. 41 – 153.03 (aromatic carbons)
d) Mass spectrum (ESI, methanol)
[M+H]+ at m/z 212 (100), [M-H2O]+ at m/z 194 (15).
(IS, 2i?)-Methoxamine tartrate complex was added to water (275 mL) at 35 °C, basified
to 12 – 13 pH with 50% sodium hydroxide solution. Dichloromethane (250 mL) was added and stirred for 30 min. Separated the organic layer, dried over sodium sulphate and concentrated completely under vacuum at 45 °C. Iso-Propyl alcohol (175 mL) was added, charcaolized and filtered. The clear filtrate was acidified with 20%IPA HC1 to yield (1S, 2R)-Methoxamine HC1 which was filtered and dried (51 g) White crystalline powder; Purity by HPLC : 99.99%; Chiral purity . 100 %; mp . 172-175 °C;[α]D : + 47.9° (c = 2% in MeOH)
Spectroscopic interpretation
The structure of the product, (1S, 2R)-Methoxamine HC1 was confirmed with the help of the following spectroscopic data.
a) m (cm1) (KBr)
O-H stretch at 3265, aromatic C-H stretch at 3059, aliphatic C-H stretch at 2997, HN-H str. at 2658 and 2567, benzenoid bands at 1611 and 1587,
C-N stretch at 1294, C-O stretch at 1217, C-H out of plane bending of 1,2,4-tri- substituted benzene ring at 818.
b) 1H NMR (DMSO-d6,300 MHz) (δH)
0.91 (3H,d, -CH-CH3), 3.71 (3H, s, -OCH3), 3.75 (3H, s, -OCH3), 5.14 (1H, m, -CH- NH3+), 5.97 (1H, d, -CH-OH), 6.83-7.01 (3H, m, aromatic protons), 8.19 (3H, bs, HN-H) The -OH proton appears to have exchanged with the solvent.
c) 13C NMR (DMSO-d6,300 MHz) (δc)
II. 46 (-CH-CH3), 49.18 (-OCH3), 55.23 (-OCH3), 55.68 (-CH-CH3), 66.45 (CH-OH),
III. 42 – 153.02 (aromatic carbons)
d) Mass spectrum (ESI, methanol)
[M+H]+ at m/z 212 (100), [M-H2O]+ at m/z 194 (15).
Example 5
Preparation of dl-threo-methoxamine HC1 (11)
dl-erythro-methoxamine HC1 (120g; 0.48 mol) was dissolved in DM water (500 mL) at 30 – 35 °C and cooled to 10 – 15 °C. The clear solution was basified using 50 % sodium hydroxide solution and extracted in dichloromethane (250 mL). The organic layer was separated and concentrated under vacuum. The residue thus obtained was dissolved in toluene (200 mL) and was added slowly to acetic anhydride (120 g; 1.17mol) at 65 – 70 °C. The reaction mass was maintained under stirring and further cooled to 10 – 20 °C. Conc.Sulphuric acid (57.6g; 0.58mol) was added to the reaction mass slowly by maintaining the reaction mass at 10 – 200 C. The reaction mass was heated to 35 – 400 C for 3 hours and concentrated under vacuum at below 80 °C.
The reaction mass was cooled to 10 – 15 °C and was dissolved in DM water (250 mL). The mass was maintained for 3 h at reflux temperature and again cooled to 10 – 15 °C.
The pH was adjusted to 12 – 13 using 50% sodium hydroxide solution and extracted the d/-threo-Methoxamine base in dichloromethane (250 mL). Separated the organic layer and concentrated under vacuum. The concentrated mass was triturated with iso-Propyl alcohol (150 mL); acidified using 20% HC1 in iso-propyl alcohol. Distilled the iso- propyl alcohol completely to the final traces and acetone (300 mL) was added. The material precipitated, crude dl-threo-methoxamine HC1 was filtered. (85 g) Off white powder; Purity by HPLC: 99.4%; mp: 221-223 °C Spectroscopic interpretation
The structure of the product, di-threo-methoxamine HC1 was confirmed with the help of the following spectroscopic data.
a) IR (cm”1) (KBr)
O-H stretch at 3401, aromatic C-H stretch at 3005, aliphatic C-H stretch at 2924, HN-H str. at 2581 and 2490, benzenoid bands at 1609 and 1578, C-N stretch at 1277, C-0 stretch at 1215, C-H out of plane bending of 1,2,4-tri- substituted benzene ring at 802.
b) NMR (DMSO-d6,300 MHz) (δH)
1.2 (3H,d, -CH-CHs), 3.72 (3H, s, -OCH3), 3.75 (3H, s, -OCH3), 4.87 (1H, q, -CH-CH3),6.3 (1H, d, -CH-OH), 6.83-6.99 (3H, m, aromatic protons), 8.03 (3H, bs, HN-H) The -OH proton appears to have exchanged with the solvent.
c) 13C NMR (DMSO-d6, 300 MHz) (δC)
14.76 (-CH-CH3), 52.15 (-OCH3), 55.89 (-OCH3), 67.34 (CH-OH), 111.96 – 153.21 (aromatic carbons)
d) Mass spectrum (ESI, methanol)
[M+H]+ at m/z 212 (100), [M-H2O]+ at m/z 194 (52).
Example 6
Preparation of (1S,2S)- Methoxamine HC1 (6)
(IR, 2S)-Methoxamine HC1 (120 g; 0.48 mol) was dissolved in DM water (500 mL) at 30 -35 °C and cooled to 10 – 15 °C. The clear solution was basified using 50 % sodium hydroxide solution and extracted in dichloromethane (250 mL). The organic layer was separated and concentrated under vacuum. The residue thus obtained was dissolved in toluene (200 mL) and was added slowly to acetic anhydride (120 g; 1.17 mol) at 65 – 70 °C. The reaction mass was maintained under stirring and further cooled to 10 – 20 °C. Conc.sulphuric acid (57.6 g; 0.58 mol) was added to the reaction mass slowly by maintaining the reaction mass at 10 – 20 °C. The reaction mass was heated to 35 – 40 °C for 3 hours and concentrated under vacuum at below 80 °C.
The reaction mass was cooled to 10-15°C and was dissolved in DM water (250 mL). The mass was maintained for 3 h at reflux temperature and again cooled to 10 – 15 °C. The pH was adjusted to 12-13 using 50% sodium hydroxide solution and extracted the (1S, 2S)-Methoxamine base in dichloromethane (250 mL). Separated the organic layer and concentrated under vacuum The concentrated mass was triturated with iso-Propyl alcohol (150 mL); acidified using 20% HC1 in iso-propyl alcohol. Distilled the iso- propyl alcohol completely to the final traces and acetone (300 mL) was added. The material precipitated, crude (IS, 2S)-methoxamine HC1 was filtered. (86 g); White crystalline powder; Purity by HPLC . 99.8%; Chiral purity : 99.7%; mp : 172-175 °C; [α]D: + 30.739° (c = 2% in MeOH)
Spectroscopic interpretation
The structure of the product, (IS, 2S)-methoxamine HC1 was confirmed with the help of the following spectroscopic data.
a) IR (cm1) (KBr)
O-H stretch at 3356, aromatic C-H stretch at 3080, aliphatic C-H stretch at 2999, HN-H str. at 2641 and 2583, benzenoid bands at 1611 and 1506, C-N stretch at 1302, C-O stretch at 1229, C-H out of plane bending of 1,2,4-tri- substituted benzene ring at 812.
b) 1H NMR (DMSO-d6 300 MHz) (δH)
1.04 (3H,d, -CH-CH3), 3.72 (3H, s, -OCH3), 3.75 (3H, s, -OCH3), 4.90 (1H, m, -CH- CH3),6.07 (1H, d, -CH-OH), 6.84-7.01 (3H, d, aromatic protons), 8.15 (3H, bs, HN-H)
The -OH proton appears to have exchanged with the solvent.
c) 13C NMR (DMSO-d6, 300 MHz) (δC)
14.75 (-CH-CH3), 52.18 (-OCH3), 55.21 (-OCH3), 55.69 (-CH-CH3), 67.32 (CH-OH), 111.38 -153.01 (aromatic carbons)
d) Mass spectrum (ESI, methanol)
[M+H]+ at m/z 212 (100), [M-H2O]+ at m/z 194 (48).
Example 7
Preparation of (1R, 2R)-Methoxamine HC1 (5)
(IS, 2R)Methoxamine HC1 (120g; 0.48 mol) was dissolved in DM water (500 mL) at 30 – 35 °C and cooled to 10 – 15 °C. The clear solution was basified using 50 % sodium hydroxide solution and extracted in dichloromethane (250 mL). The organic layer was separated and concentrated under vacuum. The residue thus obtained was dissolved in toluene (200 mL) and was added slowly to acetic anhydride (120 g; 1.17mol) at 65 – 70 °C. The reaction mass was maintained under stirring and further cooled to 10 – 20 °C. Cone.Sulphuric acid (57.6g; 0.58mol) was added to the reaction mass slowly by maintaining the reaction mass at 10 – 20 °C. The reaction mass was heated to 35 – 40 °C for 3 hours and concentrated under vacuum at below 80 °C.
The reaction mass was cooled tol0-15°C and was dissolved in DM water (250 mL). The mass was maintained for 3 h at reflux temperature and again cooled to 10 – 15 °C. The pH was adjusted to 12-13 using 50% sodium hydroxide solution and extracted the (IR, 2i?)-Methoxamine base in dichloromethane (250 mL). Separated the organic layer and concentrated under vacuum. The concentrated mass was triturated with iso-Propyl alcohol (150 mL); acidified using 20% HC1 in iso-propyl alcohol Distilled the iso- propyl alcohol completely to the final traces and acetone (300 mL) was added. The material precipitated, crude (1R, 2R)-methoxamine HC1 was filtered. (90 g) White crystalline powder; Purity by HPLC: 99.1%, Chiral purity. 100%; mp: 172-175 °C;[α]D: -29.04° (c – 2% in MeOH)
Spectroscopic interpretation
The structure of the product, (1R, 2R)methoxamine HC1 was confirmed with the help of the following spectroscopic data.
a) IR (cm1) (KBr)
O-H stretch at 3356, aromatic C-H stretch at 3078, aliphatic C-H stretch at 2999, HN-H str. at 2619 and 2500, benzenoid bands at 1611 and 1508, C-N stretch at 1302, C-O stretch at 1229, C-H out of plane bending of 1,2,4-tri- substituted benzene ring at 812.
b) 1H NMR(DMSO-d6 300 MHz) (δH)
I. 04 (3H,d, -CH-CHa), 3.72 (3H, s, -OCH3), 3.75 (3H, s, -OCH3), 4.90 (1H, m, -CH- CH3),6.07 (1H, d, -CH-OH), 6.83-7.01 (3H, d, aromatic protons), 8.13 (3H, bs, HN-H) The -OH proton appears to have exchanged with the solvent.
c) 13C NMR (DMSO-d6 300 MHz) (δe)
II. 41 (-CH-CH3), 52.16 (-OCH3), 55.22 (-OCH3), 55.70 (-CH-CH3), 67.32 (CH-OH), III. 39-153.15 (aromatic carbons)
d) Mass spectrum (ESI, methanol)
[M+H]+ at m/z 212 (100), [M-H2O]+ at m/z 194 (44).
PATENT
http://www.google.com/patents/US8491931
(1,R,2S)-Methoxamine
To a stirred solution of methoxycarbonyl (MeOC) protected alcohol i.e. (1R,2S)-2-[(methoxycarbonyl)amino]-1-(2,5-dimethoxyphenyl)-1-propanol (4.0 g, 14.9 mmol) in methanol (175 cm3) was added a solution of KOH (4.06 g, 72.8 mmol in water (60 cm3). The solution was cooled and acidified with phosphoric acid (15% v/v). The solution was extracted with DCM (2×50 cm3) and the aqueous layer basified by the addition of K2CO3. The aqueous layer was extracted with diethyl ether (5×50 cm3) and the combined ethereal extracts dried (MgSO4) and concentrated under reduced pressure to give the product as a clear yellow oil (1.9 g, 61%), 1H NMR (250 MHz; C2HCl3) 0.84 (3H, d, J 7.0, CH3), 3.19-3.22 (1H, m, H-2), 3.71 (6H, s, 2×OCH3), 4.67 (1H, d, J 5.0, H-1), 6.66-6.72 (2H, m, ArH), 6.92 (1H, d, J 2.5, ArH).
(1R,2S)-Methoxamine hydrochloride
To an ice cooled solution of (1R,2S)-methoxamine (1.9 g, 9.00 mmol) in anhydrous diethyl ether (30 cm3) was passed a stream of dry HCl gas for 45 mins. The resultant precipitate was filtered by suction, washed with cold diethyl ether and dried under nitrogen to yield the title compound as a white solid. (1.5 g, 68%). 1H NMR (250 MHz; [C2H3]2SO) 0.89 (3H, d, J 6.8, CH3), 3.37-3.42 (1H,M,H-2), 3.71 (3H, s, OCH3), 3.75 (3H, s, OCH3), 5.12 (1H, s, H-1), 5.92 (1H, d, J 4.3, OH), 6.84 (1H, dd, J 8.8, 3.0, ArH), 6.92-7.00 (2H, m, ArH); HPLC.



//1R,2S-methoxamine
RACEMIC
 |
|
Title: Methoxamine
CAS Registry Number: 390-28-3
CAS Name: a-(1-Aminoethyl)-2,5-dimethoxybenzenemethanol
Additional Names: a-(1-aminoethyl)-2,5-dimethoxybenzyl alcohol; 2-amino-1-(2,5-dimethoxyphenyl)-1-propanol; b-hydroxy-b-(2,5-dimethoxyphenyl)isopropylamine; b-(2,5-dimethoxyphenyl)-b-hydroxyisopropylamine; 2,5-dimethoxynorephedrine
Molecular Formula: C11H17NO3
Molecular Weight: 211.26
Percent Composition: C 62.54%, H 8.11%, N 6.63%, O 22.72%
Literature References: a1-Adrenergic agonist. Prepn: Baltzly et al., US 2359707 (1944 to Burroughs Wellcome). Metabolism: A. Klutch, M. Bordun, J. Med. Chem. 10, 860 (1967). Clinical pharmacology: N. T. Smith, C. Whitcher, Anesthesiology 28, 735 (1967); P. D. Snashall et al., Clin. Sci. Mol. Med. 54, 283 (1978). HPLC determn in plasma: I. A. Al-Meshal et al., J. Liq. Chromatogr. 12, 1589 (1989). Therapeutic use: P. M. C. Wright et al., Anesth. Analg. 75, 56 (1992); L. Cabanes et al., N. Engl. J. Med. 326, 1661 (1992). Comprehensive description: A. M. Al-Obaid, M. M. El-Domiaty, Anal. Profiles Drug Subs. 20, 399-431 (1991).
Derivative Type: Hydrochloride
CAS Registry Number: 61-16-5
Trademarks: Vasoxine (Burroughs Wellcome); Vasoxyl (Burroughs Wellcome); Vasylox (Burroughs Wellcome)
Molecular Formula: C11H17NO3.HCl
Molecular Weight: 247.72
Percent Composition: C 53.33%, H 7.32%, N 5.65%, O 19.38%, Cl 14.31%
Properties: Crystals, mp 212-216°. pKa (25°C) 9.2. Very sol in water: One gram dissolves in 2.5 ml water, in 12 ml ethanol. Practically insol in ether, benzene, chloroform. pH of a 2% aq soln between 4.5 and 5.5.
Melting point: mp 212-216°
pKa: pKa (25°C) 9.2
Therap-Cat: Antihypotensive.
Keywords: a-Adrenergic Agonist; Antihypotensive.
|
Regulatory Approval Pathways: EU vs US
DRUG REGULATORY AFFAIRS INTERNATIONAL

Regulatory Approval Pathways: EU vs US
 Drug Authorization Procedures in the EU
Drug Authorization Procedures in the EU
Sponsors have several options when seeking market approval for a new drug in Europe: a national authorization procedure, a decentralized procedure, a mutual recognition procedure and a centralized procedure. Depending on a product’s eligibility, each of these authorization routes offers different advantages and disadvantages to the sponsor, and these should be considered when setting up the market strategy of a product.
National Procedure
This procedure is used whenever a company wants to commercialize a product in only one EU Member State.
The National procedure is specific to each country. That is, each country within the EU has its own procedures for authorizing a marketing application for a new drug. Sponsors can find information regarding the requirements and procedure of each country on the websites of the regulatory agencies.
ADVANTAGES of National Procedure
There are some advantages in submitting…
View original post 3,049 more words
Enasidenib (AG-221)

Enasidenib (AG-221)
1446502-11-9
Chemical Formula: C19H17F6N7O
Exact Mass: 473.13988
AG-221; AG 221; AG221; CC-90007; CC 90007; CC90007; Enasidenib
IUPAC/Chemical Name: 2-methyl-1-((4-(6-(trifluoromethyl)pyridin-2-yl)-6-((2-(trifluoromethyl)pyridin-4-yl)amino)-1,3,5-triazin-2-yl)amino)propan-2-ol
2-methyl-1-(4-(6-(trifluoromethyl)pyridin-2-yl)-6-(2-(trifluoromethyl)pyridin-4-ylamino)-1,3,5-triazin-2-ylamino)propan-2-ol
Agios Pharmaceuticals, Inc. innovator
Enasidenib, aslo known as AG-221 and CC-90007, is a potent and selective IDH2 inhibitor with potential anticancer activity (IDH2 = Isocitrate dehydrogenase 2). The mutations of IDH2 present in certain cancer cells result in a new ability of the enzyme to catalyze the NAPH-dependent reduction of α-ketoglutarate to R(-)-2-hydroxyglutarate (2HG). The production of 2HG is believed to contribute to the formation and progression of cancer . The inhibition of mutant IDH2 and its neoactivity is therefore a potential therapeutic treatment for cancer
AG-221 is an orally available, selective, potent inhibitor of the mutated IDH2 protein, making it a highly targeted investigational medicine for the potential treatment of patients with cancers that harbor an IDH2 mutation. AG-221 has received orphan drug and fast track designations from the U.S. FDA. In September 2013, Agios initiated a Phase 1 multicenter, open-label, dose escalation clinical trial of AG-221 designed to assess the safety and tolerability of AG-221 in advanced hematologic malignancies. In October 2014, Agios initiated four expansion cohorts as part of the ongoing Phase 1 study and expanded its development program with the initiation of a Phase 1/2 study of AG-221 in advanced solid tumors. For the detailed information of AG-221, the solubility of AG-221 in water, the solubility of AG-221 in DMSO, the solubility of AG-221 in PBS buffer, the animal experiment (test) of AG-221, the cell expriment (test) of AG-221, the in vivo, in vitro and clinical trial test of AG-221, the EC50, IC50,and affinity,of AG-221, For the detailed information of AG-221, the solubility of AG-221 in water, the solubility of AG-221 in DMSO, the solubility of AG-221 in PBS buffer, the animal experiment (test) of AG-221, the cell expriment (test) of AG-221, the in vivo, in vitro and clinical trial test of AG-221, the EC50, IC50,and affinity,of AG-221,
Agios Announces New Data from Ongoing Phase 1 Dose Escalation and Expansion Trial of AG-221 Showing Durable Clinical Activity in Patients with Advanced Hematologic Malignancies
IDH2-Mutant Inhibitor Shows Durable Responses of More than 15 Months in Patients with Advanced Acute Myeloid Leukemia (AML) and Other Blood Cancers
Proof-of-Concept Demonstrated in Myelodysplastic Syndrome (MDS) and Untreated AML
125-Patient Expansion Cohort and Global Registration-Enabling Program Remain on Track
Company to Host Conference Call and Webcast Today
CAMBRIDGE, Mass. & VIENNA–(BUSINESS WIRE)–Jun. 12, 2015– Agios Pharmaceuticals, Inc. (Nasdaq:AGIO), a leader in the fields of cancer metabolism and rare genetic disorders of metabolism, today announced new data from the dose-escalation phase and expansion cohorts from the ongoing Phase 1 study evaluating single agent AG-221, a first-in-class, oral, selective, potent inhibitor of mutant isocitrate dehydrogenase-2 (IDH2), in advanced hematologic malignancies. The data will be presented at the 20th Congress of the European Hematology Association (EHA) taking place June 11-14, 2015 in Vienna.
Data as of May 1, 2015 from 177 patients (104 in dose escalation and 73 from the first four expansion cohorts) with advanced hematologic malignancies treated with single agent AG-221 showed durable clinical activity and a favorable safety profile. More than half of the 177 patients remain on treatment. The study had an overall response rate of 40 percent (63 of 158 response-evaluable patients, using the criteria below) and a complete remission rate of 16 percent (26 of 158 response-evaluable patients). Patients responding to AG-221 continue to show durable clinical activity on treatment for more than 15 months, with an estimated 76 percent of responders staying on treatment for six months or longer. The overall safety profile observed was consistent with previously reported data with more than 100 additional patients treated as of the last analysis.
This new data reflects responses in the evaluable population, which includes all patients with a pre-AG-221 screening assessment and day 28 or later response assessment or an earlier discontinuation for any reason. Patients with a screening assessment who were still on treatment, but had not reached the day 28 disease assessment, were excluded.
“The clinical profile of AG-221 continues to be impressive from the perspectives of response rate, durability, safety and unique mechanism of action,” said Courtney DiNardo, M.D., lead investigator and assistant professor, leukemia atUniversity of Texas MD Anderson Cancer Center. “Additionally, it is encouraging to see early proof-of-concept in myelodysplastic syndrome (MDS) and untreated acute myeloid leukemia (AML) given the need for more effective therapies for these patients.”
“As the data from the AG-221 study continue to mature, we are compiling a robust dataset to quickly move this program into global registration studies later this year in collaboration with Celgene,” said Chris Bowden, M.D., chief medical officer of Agios. “We are excited about the speed of enrollment we’ve seen to date in our four expansion cohorts and are on track to enroll our recently announced fifth expansion cohort of 125 patients with relapsed and/or refractory AML. With this progress, we are executing on our strategy to combine speed and breadth to reach people with hematologic malignancies in urgent need of better treatments.”
About the Ongoing Phase 1 Trial for AG-221 in Advanced Hematologic Malignancies
AG-221 is currently being evaluated in an ongoing Phase 1 trial that includes a dose-escalation phase and four expansion cohorts of 25 patients each, evaluating patients with relapsed or refractory AML who are 60 years of age and older and transplant ineligible; relapsed or refractory AML patients under age 60; untreated AML patients who decline standard of care chemotherapy; and patients with other IDH2-mutant positive hematologic malignancies. Data reported here are from patients receiving AG-221 administered from 60 mg to 450 mg total daily doses in the dose escalation arm and 100 mg once daily in the first four expansion arms, as of May 1, 2015. The median age of these patients is 69 (ranging from 22-90). Treatment with AG-221 showed substantial reduction in the plasma levels of the oncometabolite 2-hydroxglutarate (2HG) to the level observed in healthy volunteers.
Safety Data
A safety analysis was conducted for all 177 treated patients as of May 1, 2015.
- The majority of adverse events reported by investigators were mild to moderate, with the most common being nausea, fatigue, increased blood bilirubin and diarrhea.
- The majority of serious adverse events (SAE) were disease related; SAEs possibly related to study drug were reported in 27 patients.
- A maximum tolerated dose (MTD) has not been reached.
- The all-cause 30-day mortality rate was 4.5 percent.
Efficacy Data
Sixty-three out of 158 response-evaluable patients achieved investigator-assessed objective responses for an overall response rate of 40 percent as of May 1, 2015.
- Of the 63 patients who achieved an objective response, there were 26 (16 percent) complete remissions (CR), three CRs with incomplete platelet recovery (CRp), 14 marrow CRs (mCR), two CRs with incomplete hematologic recovery (CRi) and 18 partial remissions (PR).
- Of the 111 patients with relapsed or refractory AML, 46 (41 percent) achieved an objective response, including 20 (18 percent) CRs, one CRp, 16 PRs, eight mCRs and one CRi.
- Of the 22 patients with AML that had not been treated, seven achieved an objective response, including three CRs, two PRs, one mCR and one CRi.
- Of the 14 patients with myelodysplastic syndrome (MDS), seven achieved an objective response, including two CRs, one CRp and four mCRs.
- Responses were durable, with duration on study drug more than 15 months and ongoing. As of the analysis date, an estimated 88 percent of responses lasted three months or longer, and 76 percent of responses lasted six months or longer.
Upcoming Milestones for AG-221
Agios studies in IDH2-mutated solid and hematologic tumors are ongoing or planned for 2015 to further support development of AG-221.
- Continue to enroll patients in the fifth expansion cohort of 125 patients with IDH2 mutant-positive AML who are in second or later relapse, refractory to second-line induction or re-induction treatment, or have relapsed after allogeneic transplantation.
- Initiate combination trials to evaluate AG-221 as a potential frontline treatment for patients with AML and a broad range of hematologic malignancies in the second half of 2015.
- Initiate a global Phase 3 registration-enabling study in relapsed/refractory AML patients that harbor an IDH2 mutation in the second half of 2015.
- Continue dose escalation in the Phase 1/2 trial in patients with advanced solid tumors, including glioma and angioimmunoblastic T-cell lymphoma (AITL) that carry an IDH2 mutation in 2015.
Conference Call Information
Agios will host a conference call and webcast from the congress to review the data on Friday, June 12, 2015, beginning at 8:00 a.m. ET (2:00 p.m. CEST). To participate in the conference call, please dial (877) 377-7098 (domestic) or (631) 291-4547 (international) and refer to conference ID 53010830. The webcast will be accessible live or in archived form under “Events & Presentations” in the Investors and Media section of the company’s website at www.agios.com.
About Agios/Celgene Collaboration
AG-221, the IDH1-mutant inhibitor AG-120 and the pan-IDH mutant inhibitor AG-881 are part of Agios’ global strategic collaboration with Celgene Corporation. Under the terms of the collaboration, Celgene has worldwide development and commercialization rights for AG-221. Agios continues to conduct clinical development activities within the AG-221 development program and is eligible to receive up to $120 million in payments on achievement of certain milestones and royalties on net sales. For AG-120, Agios retains U.S. development and commercialization rights. Celgene has an exclusive license outside the United States. Celgene is eligible to receive royalties on net sales in the U.S. Agios is eligible to receive royalties on net sales outside the U.S. and up to $120 million in payments on achievement of certain milestones. For AG-881, the companies have a joint worldwide development and 50/50 profit share collaboration, and Agios is eligible to receive regulatory milestone payments of up to $70 million.
About IDH Mutations and Cancer
IDH1 and IDH2 are two metabolic enzymes that are mutated in a wide range of hematologic and solid tumor malignancies, including AML. Normally, IDH enzymes help to break down nutrients and generate energy for cells. When mutated, IDH increases production of an oncometabolite 2-hydroxyglutarate (2HG) that alters the cells’ epigenetic programming, thereby promoting cancer. 2HG has been found to be elevated in several tumor types. Agios believes that inhibition of the mutated IDH proteins may lead to clinical benefit for the subset of cancer patients whose tumors carry them.
About Acute Myelogenous Leukemia (AML)
AML, a cancer of blood and bone marrow characterized by rapid disease progression, is the most common acute leukemia affecting adults. Undifferentiated blast cells proliferate in the bone marrow rather than mature into normal blood cells. AML incidence significantly increases with age, and according to the American Cancer Society, the median age of onset is 66. Less than 10 percent of U.S. AML patients are eligible for bone marrow transplant, and the vast majority of patients do not respond to chemotherapy and progress to relapsed/refractory AML. The five-year survival rate for AML is approximately 20 to 25 percent. IDH2 mutations are present in about 9 to 13 percent of AML cases.
About Myelodysplastic Syndrome (MDS)
MDS comprises a diverse group of bone marrow disorders in which immature blood cells in the bone marrow do not mature or become healthy blood cells. The National Cancer Institute estimates that more than 10,000 people are diagnosed with MDS in the United States each year. Failure of the bone marrow to produce mature healthy cells is a gradual process, and reduced blood cell and/or reduced platelet counts may be accompanied by the loss of the body’s ability to fight infections and control bleeding. For roughly 30 percent of the patients diagnosed with MDS, this bone marrow failure will progress to AML. Chemotherapy and supportive blood products are used to treat MDS.
About Agios Pharmaceuticals, Inc.
Agios Pharmaceuticals is focused on discovering and developing novel investigational medicines to treat cancer and rare genetic disorders of metabolism through scientific leadership in the field of cellular metabolism. In addition to an active research and discovery pipeline across both therapeutic areas, Agios has multiple first-in-class investigational medicines in clinical and/or preclinical development. All Agios programs focus on genetically identified patient populations, leveraging our knowledge of metabolism, biology and genomics. For more information, please visit the company’s website at agios.com.
clips
AG-221, Inhibitor Of IDH2 Mutants

The enzyme isocitrate dehydrogenase (IDH) is probably most famous for its role in the central cellular metabolic pathway, the Krebs cycle. The enzyme catalyzes the oxidative decarboxylation of isocitrate to α-ketoglutarate. One subtype of the enzyme, IDH1, is found in cells’ cytoplasm, and another, IDH2, is found in their mitochondria.

Target: IDH2
People with certain mutations in IDH end up making R-2-hydroxyglutarate (2-HG) instead of α-ketoglutarate. 2-HG is known to make cancer cells flourish. In fact, IDH mutations have been implicated in about 70% of brain cancers and have also been identified in solid tumors and blood cancers, such as acute myeloid leukemia.
Jeremy M. Travins of Agios Pharmaceuticals spoke about how scientists at the company found compounds based on substituted triazines that can cut down on 2-HG production by inhibiting a dimer of mutant IDH2. Using structure-activity relationships and a crystal structure of a lead compound bound to the mutant IDH2 dimer, they managed to develop a clinical candidate: AG-221. It turns out that AG-221 doesn’t bind to the active site of mutant IDH2. Rather, the compound binds to the spot where the two enzymes meet in the dimer.
Hitting this position in just the right way is tricky, Travins explained. Hydrogen-bonding interactions from the triazine and the two amino groups that flank it are critical.
The compound is in Phase I clinical trials, Travins said, and it’s been shown to lower 2-HG levels to those seen in people without cancer. What’s more, he noted, the drug candidate has few side effects, giving patients a higher quality of life than standard chemotherapeutic agents do.
Patent
http://www.google.com/patents/US20130190287
Compound 409—2-methyl-1-(4-(6-(trifluoromethyl)pyridin-2-yl)-6-(2-(trifluoromethyl)pyridin-4-ylamino)-1,3,5-triazin-2-ylamino)propan-2-ol
1H NMR (METHANOL-d4) δ 8.62-8.68 (m, 2H), 847-8.50 (m, 1H), 8.18-8.21 (m, 1H), 7.96-7.98 (m, 1H), 7.82-7.84 (m, 1H), 3.56-3.63 (d, J=28 Hz, 2H), 1.30 (s, 6H). LC-MS: m/z 474.3 (M+H)+.
| Patent ID | Date | Patent Title |
|---|---|---|
| US2013190287 | 2013-07-25 | THERAPEUTICALLY ACTIVE COMPOUNDS AND THEIR METHODS OF USE |
REFERENCES
1: Caino MC, Altieri DC. Molecular Pathways: Mitochondrial Reprogramming in Tumor Progression and Therapy. Clin Cancer Res. 2016 Feb 1;22(3):540-5. doi: 10.1158/1078-0432.CCR-15-0460. Epub 2015 Dec 9. PubMed PMID: 26660517; PubMed Central PMCID: PMC4738153.
2: Stein EM. IDH2 inhibition in AML: Finally progress? Best Pract Res Clin Haematol. 2015 Jun-Sep;28(2-3):112-5. doi: 10.1016/j.beha.2015.10.016. Epub 2015 Oct 19. Review. PubMed PMID: 26590767.
3: Rowe JM. Reasons for optimism in the therapy of acute leukemia. Best Pract Res Clin Haematol. 2015 Jun-Sep;28(2-3):69-72. doi: 10.1016/j.beha.2015.10.002. Epub 2015 Oct 22. Review. PubMed PMID: 26590761.
4: Stein EM. Molecular Pathways: IDH2 Mutations-Co-opting Cellular Metabolism for Malignant Transformation. Clin Cancer Res. 2016 Jan 1;22(1):16-9. doi: 10.1158/1078-0432.CCR-15-0362. Epub 2015 Nov 9. PubMed PMID: 26553750.
5: Kiyoi H. Overview: A New Era of Cancer Genome in Myeloid Malignancies. Oncology. 2015;89 Suppl 1:1-3. doi: 10.1159/000431054. Epub 2015 Nov 10. Review. PubMed PMID: 26551625.
6: Tomita A. [Progress in molecularly targeted therapies for acute myeloid leukemia]. Rinsho Ketsueki. 2015 Feb;56(2):130-8. doi: 10.11406/rinketsu.56.130. Japanese. PubMed PMID: 25765792.
/////////Enasidenib, AG-221,
CC(O)(C)CNC1=NC(C2=NC(C(F)(F)F)=CC=C2)=NC(NC3=CC(C(F)(F)F)=NC=C3)=N1
PF-06260414
PF-06260414
CAS: 1612755-71-1
Chemical Formula: C14H14N4O2S
Exact Mass: 302.0837
PF-06260414; PF 06260414; PF06260414; PF6260414; PF-6260414; PF 6260414.
IUPAC/Chemical Name: (R)-6-(4-methyl-1,1-dioxido-1,2,6-thiadiazinan-2-yl)isoquinoline-1-carbonitrile
- 6-[(4R)-4-Methyl-1,1-dioxido-1,2,6-thiadiazinan-2-yl]isoquinoline-1-carbonitrile
https://clinicaltrials.gov/ct2/show/NCT02070939
- 28 Jul 2015Discontinued – Phase-I for Cachexia in USA (PO)
- 27 Apr 2015Pfizer terminates a phase I trial (In volunteers) in USA (NCT02393807)
- 26 Mar 2015Pfizer plans a phase I pharmacokinetic trial for Healthy volunteers in USA (NCT02393807)
| Company | Pfizer Inc. |
| Description | Selective androgen receptor modulator |
| Molecular Target | Androgen receptor |
| Mechanism of Action | |
| Therapeutic Modality |
| Latest Stage of Development | Phase I |
| Standard Indication | Cachexia |
| Indication Details | Treat cachexia |
PF-06260414 is a selective androgen receptor modulator, or SARM, which is developed to treat muscle weakening. Testosterone’s anabolic properties help develop muscle mass, and its androgenic activity is associated with reproduction. Improving muscle mass would improve quality of life and may even prolong survival in certain patient populations.
PATENT
WO 2015173684
The androgen receptor (“AR”) is a ligand-activated transcriptional regulatory protein that mediates induction of male sexual development and function through its activity with endogenous androgens. Androgenic steroids play an important role in many physiologic processes, including the development and maintenance of male sexual characteristics such as muscle and bone mass, prostate growth,
spermatogenesis, and the male hair pattern. The endogenous steroidal androgens include testosterone and dihydrotestosterone (“DHT”). Steroidal ligands which bind the AR and act as androgens (e.g. testosterone enanthate) or as antiandrogens (e.g.
cyproterone acetate) have been known for many years and are used clinically.
6-[(4f?)-4-Methyl-1 , 1-dioxido-1 ,2,6-thiadiazinan-2-yl]isoquinoline-1-carbonitrile (Formula I), in its free base form, has the chemical formula C14H14N4SO2 and the following structural formula:

Formula I
Synthesis of 6-[(4f?)-4-methyl-1 , 1-dioxido-1 ,2,6-thiadiazinan-2-yl]isoquinoline-1-carbonitrile is disclosed in co-pending international patent application,
PCT/IB2013/060381 , filed 25th November 2013, and published as WO 2014/087298 on 12th June 2014, assigned to the assignee of the present invention and which is incorporated herein by reference in its entirety. 6-[(4f?)-4-Methyl-1 , 1-dioxido-1 ,2,6-thiadiazinan-2-yl]isoquinoline-1-carbonitrile is known to be active as a selective androgen receptor modulator (SARM) and, as such, is useful for treating and/or preventing a variety of hormone-related conditions, for example, conditions associated with androgen decline, such as, inter alia, anaemia; anorexia; arthritis; bone disease; musculoskeletal impairment; cachexia; frailty; age-related functional decline in the elderly; growth hormone deficiency; hematopoietic disorders; hormone replacement; loss of muscle strength and/or function; muscular dystrophies; muscle loss following surgery; muscular atrophy; neurodegenerative disease; neuromuscular disease;
obesity; osteoporosis; and, muscle wasting.
Identification of new solid forms of a known pharmaceutical active ingredient provide a means of optimising either the physicochemical, stability, manufacturability and/or bioperformance characteristics of the active pharmaceutical ingredient without modifying its chemical structure. Based on a chemical structure, one cannot predict with any degree of certainty whether a compound will crystallise, under what conditions it will crystallise, or the solid state structure of any of those crystalline forms. The specific solid form chosen for drug development can have dramatic influence on the properties of the drug product. The selection of a suitable solid form is partially dictated by yield, rate and quantity of the crystalline structure. In addition, hygroscopicity, stability, solubility and the process profile of the solid form such as compressibility, powder flow and density are important considerations.
The general reaction schemes provided herein illustrate the preparation of 6-[(4f?)-4-methyl-1 , 1-dioxido-1 ,2,6-thiadiazinan-2-yl]isoquinoline-1-carbonitrile (Formula I).

Example 1

Procedure:
Into a 2L 3-neck round bottom flask equipped with a mechanical stirrer, reflux condenser and thermocouple with heating mantle was placed 2-methyltetrahydrofuran (2-MeTHF) (10 mL/g; 8.15 moles; 817 ml_; 702 g) followed by racemic-2,2′-bis(diphenylphosphino)-1 ,1 ‘-binaphthyl (BINAP) (0.04 equiv (molar); 14.0 mmol; 8.74 g) and bis(dibenzylideneacetone)palladium (Pd2(dba)3) (0.04 equiv (molar); 14.0 mmol;
8.07 g). The mixture was degassed by pulling vacuum and refilling with nitrogen three times then heated to 75 °C for 15 minutes and cooled to ambient temperature. In a separate flask, (S)-3-amino-2-methylpropan-1-ol (1.60 equiv; 561 mmol; 50.0 g, prepared using literature methods, for example as disclosed in EP-A-0,089, 139 published on 21st September 1983) was dissolved in 2-methyltetrahydrofuran (5 ml_/g;
4.08 moles; 409 ml_; 351 g) and degassed by pulling vacuum and refilling with nitrogen three times. Into the pot containing the catalyst was added 6-(bromoisoquinoline-1- carbonitrile) (1.00 equiv; 351 mmol; 81.75 g) and cesium carbonate (1.6 equiv (molar); 561 mmol; 185 g) in single portions followed by the solution of the aminoalcohol via addition funnel. The reaction mixture was again degassed by pulling vacuum and refilling with nitrogen three times. The reaction was heated to 70 °C for 3 hours. The reaction was cooled to ambient temperature and filtered through a pad of Celite. The contents of the flask were rinsed out with three 100 mL portions of 2-methyltetrahydrofuran. The filtrate was transferred into a 2L round bottom flask equipped with a thermocouple and mechanical stirrer under nitrogen. Silica Gel (Silicylate SiliaMet® Thiol) (0.4 g/g-pure-LR; 544 mmol; 32.7 g) was charged and the flask was stirred at 40 °C overnight. The following morning, the reaction was cooled to < 30 °C and filtered again through Celite. The pad was washed with 100ml_ of 2-methyltetrahydrofuran (or until no yellow color persisted in the filtrate). The filtrate was placed into a 3L round bottom flask equipped with a magnetic stir bar, distillation head (with condenser and receiving flask), and thermocouple. The mixture was heated to 60 °C and placed under vacuum (-450-500 mbar) to distil out 1.3 L total of 2-methyltetrahydrofuran. 500 mL of toluene was added to precipitate the desired product. The heating mantle was removed and the reaction was allowed to reach ambient temperature. The mixture was stirred for 1 hour at ambient temperature and then the solids were collected by vacuum filtration on a sintered glass funnel. The cake was dried overnight on the funnel under vacuum. The following morning, the solids were transferred into an amber bottle and weighed (71.9 g; 298 mmol). The product was used in the next step without further purification.
Example 2

Procedure:
In a 1 L reactor equipped with a temperature probe and overhead stirring was added the product of Example 1 (20.0 g; 1.00 equiv; 82.9 mmol) and 2-methyltetrahydrofuran (2-MeTHF) (30 mL/g-pure-LR; 5.98 moles; 600 mL; 515 g). The reaction mixture was
gently warmed to 40°C to achieve partial solubility. The reaction was cooled to 0°C. Once the reaction reached 0°C methanesulfonyl chloride (MsCI) (1.4 equiv (molar); 1 16 mmol; 8.98 mL; 13.3 g) was added in a single portion followed immediately by triethylamine (TEA) (1.4 equiv (molar); 116 mmol; 16.2 mL; 11.7 g) dropwise via syringe over a period of 15 minutes. The reaction mixture was further stirred for 30 min at 0°C and then warmed to 23°C for 60 minutes. The product (26.47 g; 1.00 equiv; 82.88 mmol; 26.47 g; 100% assumed yield) was then used without purification for the sulfonylation reaction.
Example 3
t-BuOH, 2-MeTHF
o 0 °C to 23 °C o
CI-S-N=C=0 CI-S-NHBoc
0 O
Procedure:
To a solution of t-butyl alcohol (t-BuOH) (1 equiv (molar); 116 mmol; 1 1.0 mL; 8.60 g) in 2-methyltetrahydrofuran (2-MeTHF) (1 M; 1.16 moles; 116 mL; 99.6 g) at 0°C was added chlorosulfonyl isocyanate (116 mmol; 1.00 equiv; 10.1 mL; 16.4 g) dropwise. The homogeneous solution was stirred for 30 minutes at ambient temperature and then used directly in the sulfonylation reaction.
Example 4

Sulfonylation Reaction Procedure:
A previously prepared solution of the product of Example 3 (1.4 equiv (molar); 1 16 mmol; 116 g) in 2-methyltetrahydrofuran was added to a suspension of the product of Example 2 (1.00 equiv; 82.89 mmol; 26.5 g) at 0°C. The mixture was warmed to ambient temperature over 30 minutes. HPLC analysis revealed the reaction was complete. The reaction was quenched with a 10% sodium carbonate solution (2 equiv
(molar); 165 mmol; 101 mL; 1 17 g) and water (to dissolve salts) (5 L/kg; 7.35 moles; 132 mL; 132 g). The top organic layer was removed and passed through a plug of Carbon (Darco G60) (0.5 g/g) on a filter. A significant improvement in color (dark orange to yellow) was observed. The solution was concentrated to 10 total volumes and used in the next step without purification.
Example 5

Procedure:
A solution of the product of Example 4 (1.OOequiv; 82.9 mmol; 41.3 g) in 2-methyltetrahydrofuran (2-MeTHF) (10ml_/g; 4.12 moles; 413 mL; 355 g) was placed into a 1 L reactor equipped with an overhead stirrer and temperature probe. Next, potassium carbonate (K2CO3) (325 mesh) (6 equiv (molar); 497 mmol; 69.4 g) and water (0.0 L/100-g-bulk-LR; 459 mmol; 8.26 mL; 8.26 g) were added and the mixture heated to 40°C (jacket temperature) and stirred overnight. The reaction was cooled to ambient temperature and water (4L/kg-pure-LR; 9.17 moles; 165 mL; 165 g]) was added. The biphasic reaction was stirred for 1 hour at 23 °C. The aqueous layer was extracted and removed. The organic layer was passed through a plug of Carbon (Darco G60) (0.5 g/g-pure-LR; 20.7g) in a disposable filter. The 2-methyltetrahydrofuran solution was switched to a 10 volume solution of toluene via a constant strip-and-replace distillation to no more than 1 % 2-methyltetrahydrofuran. The toluene solution of the reaction product (1.00 equiv; 82.9 mmol; 33.4 g; 100% assumed yield) was used as-is in the next step without further purification.
Example 6

Procedure:
To a 1 L reactor under nitrogen and equipped with overhead stirring and a temperature probe was added the product of Example 5 (1.00 equiv; 78.7 mmol; 33.4 g) as a solution in toluene (10 mL/g-pure-LR; 3.00 moles; 317 ml_; 276 g). Next, trifluoroacetic acid (TFA) (10 equiv (molar); 787 mmol; 59.5 ml_; 89.8 g) was added to the reaction over a period of 1 hour keeping the internal temperature below 30°C. The dark red mixture was stirred for 1 hour. The reaction was quenched at 23 °C by the addition of sodium carbonate (5 equiv (molar); 394 mmol; 240 ml_; 278 g). The reaction was quenched slowly, over a period of 1 hour to form the TFA salt of the product. Once the charge was complete, the mixture was cooled to 0°C, held for 1 hour and filtered. The next morning, the solid product (6-[(4R)-4-methyl-1 , 1-dioxido-1 ,2,6-thiadiazinan-2-yl]isoquinoline-1-carbonitrile in its free base form) was weighed (0.89 equiv; 70.0 mmol; 21.2 g; 89.0% yield) and used in the next step without further purification.
Example 7
Crystalline 6-[(4f?)-4-methyl-1 , 1-dioxido-1 ,2,6-thiadiazinan-2-yl]isoquinoline-1-carbonitrile free base (Form (1)) was prepared as follows.
In a 1 L 3-neck round bottom flask was added 6-[(4R)-4-methyl-1 , 1-dioxido-1 ,2,6-thiadiazinan-2-yl]isoquinoline-1-carbonitrile free base (1.00 equiv; 70.0 mmol; 21.2 g) a magnetic stir bar and acetone (40ml_/g; 1 1.5 moles; 847 ml_; 669 g). The mixture was heated to reflux (approximately 57°C) and stirred for 1 hour. The mixture was concentrated by atmospheric distillation (heating mantle set at 65°C) and 40ml_ of acetone was collected into a graduated cylinder. Next, water (25 mL/g; 29.4 moles; 530 ml_; 530 g) was charged over a period of one hour. The mixture was stirred at ambient temperature for 60min before being cooled to 0°C at 1 °C /min for 1 hour. The solids were collected by filtration in a disposable funnel. Crystalline 6-[(4f?)-4-methyl-1 , 1-dioxido-1 ,2,6-thiadiazinan-2-yl]isoquinoline-1-carbonitrile (Form (1), 0.88 equiv; 61.9 mmol; 18.7 g; 88.3% yield) was dried under vacuum overnight at 40 °C. Typical purity after crystallization is 98%.
PATENT
Step 1. Synthesis of 6-bromoisoquinoline (#A1). A mixture of 4-bromobenzaldehyde (300.0 g, 1620.0 mmol) and amino acetaldehyde dimethyl acetal (170.4 g, 1620 mmol) in anhydrous toluene (1.5 L) was refluxed under a Dean-Stark condenser for 12 h. The solution was concentrated under vacuum. The residue was dissolved in anhydrous THF and cooled to —10° C. Ethyl chloroformate (193.3 mL, 1782 mmol) was added and stirred for 10 min at −10° C., and then allowed to warm to room temperature. Subsequently trimethyl phosphite (249.6 mL, 1782.0 mmol) was added dropwise to the reaction mixture and stirred for 10 h at room temperature. The solvent was evaporated under vacuum and the residue was dissolved in anhydrous DCM (1.5 L) and stirred for 30 minutes. The reaction mixture was cooled to 0° C., and titanium tetrachloride (1.2 L, 6480 mmol) was added dropwise. The reaction mixture was stirred at 40° C. for 6 days. The reaction mixture was poured into ice and pH was adjusted to 8-9 with aqueous 6N NaOH solution. The suspension was extracted three times with EtOAc. The organic layer was extracted with 3 M HCl. The acidic aqueous solution was adjusted to pH to 7-8 with 3N NaOH solutions and extracted two times with EtOAc. The organic layer was dried over anhydrous sodium sulfate and concentrated under reduced pressure to provide the product. Crude compound was dissolved in minimum amount of DCM and mixed with pentane to get compound #A1 as light brown solid. Yield: 90 g (35%). Rf: 0.6 (30% EtOAc in petroleum ether).
LCMS m/z=209 (M+1). 1H NMR (400 MHz, d6-DMSO): δ 7.82 (m, 2H), 8.11 (d, J=8.8 Hz, 2H), 8.30 (br s, 1H), 8.56 (d, J=6.0 Hz, 1H), 9.35 (s, 1H).
Step 2. Synthesis of 6-bromoisoquinoline 2-oxide (#A2). m-Chloroperoxybenzoic acid (120.0 g, 720.0 mmol) was added to a solution of #A1 (90.0 g, 480.0 mmol) in DCM (500 mL) at room temperature, and the reaction mixture was stirred for 16 h. 1N NaOH was added to the stirred reaction mixture to adjust the pH to 7-8. The layers were separated and the aqueous layer was extracted with DCM. The organic layer was dried over anhydrous sodium sulfate and concentrated under reduced pressure to render crude product. The solid product was triturated with the mixture of n-pentane and ethanol (8:2) to get the #A2 as white solid. Yield: 65 g (60%). Rf: 0.2 (EtOAc).
LCMS m/z=225 (M+1). 1H NMR (400 MHz, d6-DMSO): δ 7.83 (m, 2H), 7.91 (d, J=6.8 Hz, 1H), 8.21 (dd, J=8.0, 1.2 Hz, 1H), 8.26 (br s, 1H), 8.97 (s, 1H).
Step 3. Synthesis of 6-bromoisoquinoline-1-carbonitrile (#A3). Trimethylsilyl cyanide (52.0 mL, 580.0 mmol) was added dropwise to the stirred solution of #A2 (65.0 g, 290.0 mmol) and DBU (50.0 mL, 348.0 mmol) in THF (500 mL) at room temperature over a period of 15 minutes. The reaction mixture was stirred at room temperature for 1 h. Water was added to the reaction mixture, and the solution was extracted with DCM. The organic layer was dried over anhydrous Na2SO4 and concentrated under reduced pressure to give crude product. The product was purified by column chromatography using silica gel (100-200 mesh) with 0-4% EtOAc in petroleum ether as an eluent to give #A3 as white solid. Yield: 41 g (61%). Rf: 0.6 (30% EtOAc in petroleum ether).
LCMS m/z=233 (M+1). 1H NMR (400 MHz, d6-DMSO): δ 8.07 (dd, J=11.2, 2.0 Hz, 1H), 8.21 (m, 2H), 8.55 (br s, 1H), 8.77 (d, J=7.6 Hz, 1H).
A General Procedure to Prepare Intermediates of #A4, #A5, #A6 and #1, #2, #3, #4, #6, #7.
Step 4. A solution of #A3 (1 eq.) in toluene (50 mL) was degassed by bubbling with argon gas for 15 min and then Pd2dba3 (0.03 eq.), BINAP (0.06 eq.) and Cs2CO3(3 eq.) were added to the solution followed by the addition aminoalcohol (2 eq.). The mixture was heated at 100° C. under argon atmosphere for 3 h. Reaction mixture was cooled to room temperature, diluted with EtOAC and washed with water and brine. The organic layer was dried over Na2SO4 and concentrated to get crude product. The crude compounds were purified by silica gel (100-200 mesh) column chromatography by using 0-5% MeOH in DCM. Yields: 25-45%.
Step 5. MsCl (1 eq.) was added dropwise to a solution of #A4 (1 eq.) and Et3N (2 eq.) in DCM (10 mL) at 0° C. and was stirred at room temperature for 3 h. The reaction mixture was diluted with DCM, washed with water and brine. The organic layer was dried over Na2SO4 and concentrated. Crude products were used in next step without further purification.
Step 6. t-Butanol (2 eq.) was slowly added to a solution of chloro sulfonyl isocyanate (2 eq.) in toluene (1 mL/1 mmol) at 0° C. The reaction mixture was stirred at room temperature for 45 min. This solution (t-butyl chlorosulfonylcarbamate) was then added to a solution of #A5 (1 eq.) and DIPEA (4 eq.) in THF and stirred at room temperature for 12 h. Reaction mixture was diluted with water and extracted with EtOAc. Organic layer was washed with water, brine, then dried over anhydrous Na2SO4 and concentrated. Crude products were purified by silica gel (100-200 mesh) column chromatography using 0-40% EtOAc in petroleum ether.
Step 7. TFA was added to a solution of #A6 (1 eq.) in DCM (8 mL) at 0° C. and stirred at room temperature for 2 h. Reaction mixture was concentrated, diluted with water, neutralized with sat. aq. NaHCO3 soln. then extracted with DCM. The organic layer was washed with water and dried over Na2SO4 then concentrated. The crude products were purified by triturating with DCM and pentane to provide the compound. In the case of racemic materials, the enantiomers were separated by chiral preparative HPLC.
Column: CHIRALPAK IA, 4.6 mm×250, 5 μm; Mobile phase: n-Hexane: EtOH (65:35) (For X3: 35:65; For X2: 70:30); Flow rate: 1 mL/min; Eluent: EtOH.
EXAMPLE 16-[(3S)-3-methyl-1,1-dioxido-1,2,5-thiadiazolidin-2-yl]isocluinoline-1-carbonitrile (#1; R═CH3)
LCMS m/z=289.1 (M+1). 1H NMR (400 MHz, d6-DMSO): δ 1.37 (d, J=6.3 Hz, 3H), 3.27 (m, 1H), 3.74 (m, 1H), 4.63 (m, 1H), 7.17 (d, J=5.7 Hz, 1H), 7.72 (m, 1H), 7.89 (dd, J=10.7, 2.1 Hz, 1H), 8.26 (m, 2H), 8.62 (d, J=5.7 Hz, 1H).
PATENT
example 9
6 – [(3S) -3-methyl-1, 1 -dioxido-1, 2,5-thiadiazolidin-2-carbonitrile 1-yl1naphthalene
(Stereochemistry is arbitrarily Assigned)
LCMS m / z = 286.0 (M – H). 1 H NMR (400 MHz, cf 6 -DMSO): δ 1 .31 (d, J = 6.2 Hz, 3H), 3.13 – 3.25 (m, 1H), 3.71 (dt, J = 12.5, 6.8 Hz, 1H), 4.49 – 4.62 (m, 1H), 7.62 – 7.70 (m, 1H), 7.75 – 7.83 (m, 2H), 7.99 (t, J = 7.8 Hz, 1H), 8.07 (d, J = 6.6 Hz, 1H), 8.14 (d, J = 8.9 Hz, 1H), 8.28 (d, J = 8.4 Hz, 1H). Chiral HPLC purity: 99.1% (retention time 17.12 minutes)

Step 1. Synthesis of amino ester (# D1). Thionylchlride (8.5 mL, 1 16.5 mmol) Was added to the solution of amino acid (4.0 g, 38.8 mmol) in MeOH (170 mL) at 0 ° C, and the reaction mixture Was Stirred for 6 h at room temperature. The reaction Was monitored by TLC, and after-disappearance of the starting material It was cooled to room temperature and solid NaHC0 3 Was added. The reaction mixture Was filtered, concentrated in vacuo and the resulting and residue Was triturated with diethyl ether to crude obtenir # D1 (4 g, 90%) as a white solid. R f : 0.4 (f-BuOH: AcOH: H 2 0 (4: 0.5: 0.5)).
GCMS m / z 1 17.1 (M +). 1 H NMR (400 MHz, cf 6 -DMSO): δ 1.17 (d, J = 6.8Hz, 3H), 2.83 – 2.88 (m, 2H), 3.03 – 3.05 ( m, 1H), 3.65 (s, 3H), 8.02 – 8.30 (br s, 3H).
Step 2. Synthesis of aminoalcohol (# D2). # D1 (2.0 g, 13.0 mmol) Was added
portionwise to a suspension of LiAlH 4 (1.4 g, 39.2 mmol) in THF (75 mL) under nitrogen atmosphere at 0 ° C. The reaction mixture Was Stirred for 30 minutes and allowed to stir Then at room temperature for Reviews another 30 minutes. The reaction mixture Was Refluxed for 2 h, And Then It was cooled to -10 ° C and quenched with ice cold water Carefully (1.4 mL). 10% NaOH solution (2.8 mL) and ice cold water (4.2 mL) Were added, and the mixture Was Stirred for 15 minutes. It was filtered, and the filtrate washed with EtOAc (3 x 100 mL), dried over anhydrous Na 2 S0 4 and Concentrated under vacuum to obtenir # D2 (1.2 g, 86%) as a pale yellow liquid. R f: 0.2 (20% MeOH in DCM).
1 H NMR (400 MHz, cf 6 -DMSO): δ 0.78 (d, J = 6.8Hz, 3H), 1.46 – 1.54 (m, 1H), 2.41 -2.45 (m, 2H), 2.50 – 2.54 (m , 1H), 3.22 – 3.34 (m, 4H).
Step 3. Synthesis of coupling product (# D3). K 3 P0 4 (6.1 g, 28.8 mmol), BINAP (0.44 g, 0.72 mmol) and Pd 2 (dba) 3 (0.32.0 g, 0.36 mmol) Was added to the degassed
suspension of 6-bromo-1 -cyanoisoquinoline # A3 (1.7 g, 7.2 mmol), # D2 (1.2 g, 14.5 mmol) in DMSO at room temperature. The reaction mixture Was heated at 105 ° C for 2 h. The reaction Was cooled to room temperature, water (500 mL) Followed by EtOAc (100 mL) Were added, and the mixture Was Stirred for 10 minutes. The biphasic mixture Was filtered through a Celite ™ pad and washed with EtOAc (100 mL). The organic layer Was separated, and the aqueous layer Was Extracted with EtOAc (3 x 100 mL). The combined organic layers Were dried over anhydrous Na 2 S0 4 , concentrated under Reduced pressure to get a crude material. Reviews This was purified by column chromatography on 100-200 mesh silica gel, using 50-70% EtOAc in petroleum ether as the eluent to obtenir # D3 (0.5 g, 48.5%) as a yellow solid. R f : 0.4 (60% EtOAc in petroleum ether).
LCMS m / z = 242.0 (M + H). 1 H NMR (400 MHz, cf 6 -DMSO): δ 0.97 (d, J = 6.4Hz, 3H), 1.87 – 1.99 (m, 1H), 2.92 – 2.99 (m, 1H), 3.20 – 3.27 (m, 1H), 3.38 – 3.42 (m, 2H), 4.59 (t, J = 5.2 Hz, 1H), 6.77 (d, J = 2.0, 1H ), 7.01 (t, J = 5.6 Hz, 1H), 7.34 (dd, J = 9.2 Hz, J = 2.0 Hz, 1H), 7.73 (d, J = 6.0 Hz, 1H), 7.88 (d, J = 8.8 Hz, 1H), 8.312 (d, J = 6.0 Hz, 1H).
Step 4. Methanesulfonated coupling product (# D4). Triethylamine (0.44 mL, 3.1 mmol) Was added to a solution of # D3 (0.50 g, 2.0 mmol) in DCM at 0 ° C.
Methanesulfonylchloride (0.25 mL, 3.1 mmol) Was added over 10 minutes, and the reaction mixture Was Stirred for 1 h at room temperature. After disappearance of the starting material by TLC, It was diluted with DCM and washed with water. The organic layer Was separated, dried over Na 2 S0 4 , concentrated under pressure to obtenir Reduced crude # D4 (0.6 g, crude) as yellow solid. Reviews This was used for next step Without Any purification. R f : 0.6 (50% EtOAc in petroleum ether).
LCMS m / z = 320.0 (M + H). 1 H NMR (400 MHz, CDCl 3 ): δ 1.17 (d, J = 6.8Hz, 3H), 2.32 – 2.37 (m, 1H), 3.06 (s, 3H), 3.26 – 3.41 (m, 2H), 4.16 – 4.20 (m, 1H), 4.33 – 4.37 (m, 1H), 4.75 (br s, 1H), 6.70 (d, J = 2.4, 1 H), 7.09 (dd, J = 9.2 Hz, 2.4 Hz, 1H), 7.57 (d, J = 6.0 Hz, 1H), 8.05 (d, J = 9.2 Hz, 1H), 8.39 (d, J = 5.6 Hz, 1H).
Step 5. cyclized and uncyclized intermediates (# D5, D6 #). Chlorosulfonyl isocyanate (1.2 mL, 13.1 mmol) Was added dropwise to a solution of f-BuOH (1.4 mL, 13.1 mmol) in toluene (4.0 mL) at -5 ° C. The reaction mixture Was Stirred at room temperature for 20 minutes, And Then THF (1 mL) Was added to the resulting suspension to obtenir clear solution. In Reviews another flask, DIPEA (2.3 mL, 13.1 mmol) Was added to a solution of # D4 (0.6 g, 2.6 mmol crude) in dry THF (3 mL). The Above Prepared reagent (CIS0 2 NH-Soc) Was added to this reaction mixture dropwise at room temperature over a period of 20 minutes. The resulting and reaction mixture Was Then Stirred for 16 h at room temperature. The mixture Was diluted with EtOAc (100 mL) and washed with water (100 mL). The aqueous layer Was washed with EtOAc (2 x 100 mL), combined all the organic layers, dried over Na 2 S0 4 , concentrated under Reduced pressure to obtenir the crude product (LCMS shows Desired # D6 and uncyclized # D5. This crude Was purified by column chromatography on 100-200 mesh silica gel, using 10-30% EtOAc in petroleum ether as an eluent to obtenir Desired # D6 (0.35 g, 47.8%), and uncyclized # D5 (0.22 g, crude).
The uncyclized # D5 (0.22 g, crude) Was Dissolved in THF (1 mL) and DIPEA (0.6 ml) Was added to the solution. The reaction mixture Was Stirred Reviews another for 12 h at room temperature. After qui time, It was diluted with EtOAc (100 mL) and washed with water (100 mL). The aqueous layer Was washed with EtOAc (2 x 100 mL), combined all the organic layers, dried over Na 2 S0 4 , concentrated under pressure to obtenir Reduced crude product. Was this crude purified by column chromatography on 100-200 mesh silica gel, using 10-30% EtOAc in petroleum ether as an eluent to obtenir Desired # D6 (1 .1 g, 13.2%). Total amount of # D6 Was (0.5 g, 60% for two steps, 82% purity LCMS). R f : 0.8 (60% EtOAc in petroleum ether).
LCMS m / z = 403.1 (M + H). 1 H NMR (400 MHz, CDCl3): δ 1 .04 (d, J = 6.8 Hz, 3H), 1 .50 (s, 9H), 2.38 – 2.48 ( m, 1H), 3.65 – 3.82 (m, 2H), 3.92 – 4.02 (m, 1H), 4.30 – 4.38 (m, 1H), 7.79 – 7.81 (m, 1H), 7.86 – 7.88 (m , 2H), 8.34 – 8.37 (d, J = 9.2 Hz, 1H), 8.67 (d, J = 6.0 Hz, 1H).
Step 6. Racemate # D7 and final products (# 10, # 11). TFA (5 mL) Was added to a solution of # D6 (0.15 g, 0.37 mmol) in DCM (100 mL) at 0 ° C. The reaction mixture Was Stirred for 1 h at 0 ° C. The solution Was Neutralized with saturated aqueous NaHC03 solution at 0 ° C. The mixture Was diluted with water, Extracted with DCM (3 x 100 mL). The combined organic layers Were dried over anhydrous Na 2 S0 4 and Concentrated under pressure Reduced to obtenir racemic # D7 (0.10 mg, 73%).
LCMS m / z = 303.0 (M + H). R f : 0.3 (60% EtOAc in petroleum ether).
Enantiomeric separation: # D7 Was Submitted for chiral separation to obtenir final compounds # 10 (0.015 mg) and # 11 (0.016 mg).
Column: CHIRALPAK IA, 4.6 χ 250 mm, 5 m; Mobile phase: n-Hexane / / -PrOH / DCM (60% / 15% / 15%); Flow rate: 0.8 mL / min.
example 10
6 – [(4R) -4-methyl-1, 1 -dioxido-1, 2,6-thiadiazinan-2-yl1isoquinoline-1-carbonitrile (# 10; R = (R) -CH 3 )
LCMS m / z = 303.0 (M + 1). 1 H NMR (400 MHz, cf 6 -DMSO): δ 0.98 (d, J = 6.4Hz, 3H), 2.22 – 2.26 (m, 1H), 3.16 – 3.22 (m, 1H), 3.34 – 3.39 (m, 1H), 3.59 – 3.65 (m, 1H), 3.77 – 3.81 (m, 1H), 7.75 – 7.79 (m, 1H, Disappeared in D20 exchange), 7.95 (dd, J = 8.8 Hz, J = 2.0 Hz, 1H), 8.06 (d, J = 1 .6 Hz, 1H), 8.23 – 8.27 (m, 2H), 8703 (d, J = 5.2 Hz, 1H). R f : 0.3 (60% EtOAc in petroleum ether). Chiral HPLC purity: 98.2% (retention time on January 1 .43 minutes).
CLIP
PF-06260414, A Treatment For Muscle Diseases

Target: Androgen receptors
Disease: Muscular dystrophy, atrophy, sarcopenia

There aren’t many options when it comes to treating weakening muscles caused either by a disease such as muscular dystrophy or atrophy or by sarcopenia, the natural muscle weakening that comes with age. Doctors’ primary option is to give patients testosterone—a hormone with serious unwanted side effects on reproductive organs, the liver, and kidneys.


Pfizer’s Eugene Chekler spoke about PF-06260414, a selective androgen receptor modulator, or SARM, the company developed to treat muscle weakening. The idea, Chekler told C&EN, was to develop a nonsteroidal small molecule that would target androgen receptors but wouldn’t have any of testosterone’s negative side effects.

Testosterone’s anabolic properties help develop muscle mass, and its androgenic activity is associated with reproduction. To discover their SARM, Pfizer’s scientists used a novel screening strategy in which they decoupled anabolic and androgenic properties in vitro, Chekler said. Compounds that performed well in the muscle assay but had little effect in an assay that predicts androgenic response were developed further.
PF-06260414’s key pharmacophore is an isoquinoline with a pendant cyano group. The molecule also features a cyclic sulfuric diamide. It has completed Phase I clinical trials. “The market potential for this kind of treatment is huge,” Chekler said. “Improving muscle mass would improve quality of life and may even prolong survival in certain patient populations.”
Many answers from a first in human (FIH) study: Safety, tolerability, pharmacokinetics (PK) and pharmacodynamics (PD) of PF-06260414 in healthy Western and Japanese males
Annu Meet Am Soc Clin Pharmacol Ther (ASCPT) (March 8-12, San Diego) 2016, Abst PI-021
/////////////////////PF-06260414
N#CC1=NC=CC2=C1C=CC(N(C[C@H](C)CN3)S3(=O)=O)=C2
ND 0126
ND 0126
CAS 1240322-54-6
| Molecular Formula: | C29H25F3N6O3 |
|---|---|
| Molecular Weight: | 562.54241 g/mol |
methyl 5-[[2-methyl-5-[[3-(4-methylimidazol-1-yl)-5-(trifluoromethyl)benzoyl]amino]phenyl]methylamino]-1H-pyrrolo[2,3-b]pyridine-2-carboxylate
5-{2-Methyl-5-[3-(4-methyl-imidazol-1-yl)-5-trifluoromethyl-benzoylamino]-benzylamino}-1H-pyrrolo[2,3-b]pyridine-2-carboxylic Acid Methyl Ester
Potent dual ABL/SRC inhibitors based on a 7-azaindole core with the aim of developing compds. that demonstrate a wider activity on selected oncogenic kinases. Multi-Targeted Kinase Inhibitors (MTKIs) were then derived, focusing on kinases involved in both angiogenesis and tumorigenesis processes.
Dysfunction/deregulation of protein kinases (PK) is the cause of a large number of pathologies including oncological, immunological, neurological, metabolic and infectious diseases. This has generated considerable interest in the development of small molecules and biological kinase inhibitors for the treatment of these disorders.
Numerous PK are particularly deregulated during the process of tumorigenesis. Consequently protein kinases are attractive targets for anticancer drugs, including small molecule inhibitors that usually act to block the binding of ATP or substrate to the catalytic domain of the tyrosine kinase and monoclonal antibodies that specifically target receptor tyrosine kinases (RTK) and their ligands. In solid malignancies, it is unusual for a single kinase abnormality to be the sole cause of disease and it is unlikely that tumors are dependent on only one abnormally activated signaling pathway. Instead multiple signaling pathways are dysregulated. Furthermore, even single molecular abnormalities may have multiple downstream effects. Multi targeted therapy using a single molecule (MTKI = “Multi-Targeted Kinase Inhibitors”) which targets several signaling pathways simultaneously, is more effective than single targeted therapy. Single targeted therapies have shown activity for only a few indications and most solid tumors show deregulation of multiple signaling pathways. For example, the combination of a vascular endothelial growth factor receptor (VEGFR) inhibitor and platelet derived growth factor receptor (PDGFR) inhibitor results in a cumulative antitumor efficacy (Potapova et al, Mol Cancer Ther 5, 1280-1289, 2006).
Tumors are not built up solely of tumor cells. An important part consists of connective tissue or stroma, made up of stromal cells and extracellular matrix, which is produced by these cells. Examples of stromal cells are fibroblasts, endothelial cells and macrophages. Stromal cells also play an important role in the carcinogenesis, where they are characterized by upregulation or induction of growth factors and their receptors, adhesion molecules, cytokines, chemokines and proteolytic enzymes (Hofmeister et al., Immunotherapy 57, 1-17, 2007; Raman et al, Cancer Letters 256, 137-165, 2007; Fox et al, The Lancet Oncology 2, 278-289, 2001) The receptor associated tyrosine kinase VEGFR on endothelial and tumor cells play a central role in the promotion of cancer by their involvement in angiogenesis (Cebe-Suarez et al, Cell Mol Life Sci 63, 601-615, 2006). In addition, the growth factors TGF-β, PDGF and FGF2 secreted by cancer cells transform normal fibroblasts into tumor associated fibroblasts, which make their receptors a suitable target for inhibition by kinase inhibitors (Raman et al, 2007).
Moreover, increasing evidence suggests a link between the EGF receptor (EGFR) and HER2 pathways and VEGF-dependent angiogenesis and preclinical studies have shown both direct and indirect angiogenic effects of EGFR signaling (Pennell and Lynch, The Oncologist 14, 399-411, 2009). Upregulation of tumor pro -angiogenic factors and EGFR- independent tumor-induced angiogenesis have been suggested as a potential mechanism by which tumor cells might overcome EGFR inhibition. The major signaling pathways regulated by EGFR activation are the PI3K, MAPK and Stat pathways that lead to increased cell proliferation, angiogenesis, inhibition of apoptosis and cell cycle progression. EGFR is overexpressed in a wide variety of solid tumors, such as lung, breast, colorectal and cancers of the head and neck (Cook and Figg, CA Cancer J Clin 60, 222-243 2010). Furthermore, higher expression of EGFR has been shown to be associated with metastasis, decreased survival and poor prognosis.
c-Src, a membrane-associated non receptor tyrosine kinase, is involved in a number of important signal transduction pathways and has pleiotropic effects on cellular function. c-Src integrates and regulates signaling from multiple transmembrane receptor-associated tyrosine kinases, such as the EGFR, PDGFR, IGF1R, VEGFR, HER2. Together, these actions modulate cell survival, proliferation, differentiation, angiogenesis, cell motility, adhesion, and invasion (Brunton and Frame, Curr Opin Pharmacol 8, 427-432, 2008). Overexpression of the protein c-Src as well as the increase in its activity were observed in several types of cancers including colorectal, gastrointestinal (hepatic, pancreatic, gastric and oesophageal), breast, ovarian and lung (Yeatman, Nat Rev Cancer 4, 470-480, 2004).
The activation in EGFR or KRAS in cancers leads to a greatly enhanced level of Ras- dependent Raf activation. Hence, elimination of Raf function is predicted to be an effective treatment for the numerous cancers initiated with EGFR and KRAS lesions (Khazak et al, Expert Opin. Ther. Targets 11, 1587-1609, 2007). Besides activation of Raf signaling in tumors, a number of studies implicate the activation of the Ras-Raf-MAPK signaling pathway as a critical step in vasculo genesis and angiogenesis. Such activation is induced by growth factor receptors such as VEGFR2, FGFR2 and thus inhibition of Raf activation represents a legitimate target for modulation of tumor angiogenesis and vascularization.
Although VEGFR, PDGFR, EGFR, c-Src and Raf are important targets on both tumor cells and tumor stroma cells, other kinases such as FGFR only function in stromal cells and other oncogenes often only function in tumor cells.
Protein kinases are fundamental components of diverse signaling pathways, including immune cells. Their essential functions have made them effective therapeutic targets. Initially, the expectation was that a high degree of selectivity would be critical; however, with time, the use of “multikinase” inhibitors has expanded. Moreover, the spectrum of diseases in which kinase inhibitors are used has also expanded to include not only malignancies but also immune-mediated diseases / inflammatory diseases. The first step in signaling by multi-chain immune recognition receptors is mediated initially by Src family protein tyrosine kinases. MTKI targeting kinases involved in immune function are potential drugs for autoimmune diseases such as rheumatoid arthritis, psoriasis and inflammatory bowel diseases (Kontzias et al. , F 1000 Medicine Reports 4, 2012)
Protein kinases mentioned previously are also key components of many other physiological and pathological mechanisms such as neurodegeneration and neuroprotection (Chico et al, Nature Reviews Drug Discovery 8, 892-909, 2009), atherosclerosis, osteoporosis and bone resorption, macular degeneration, pathologic fibrosis, Cystogenesis (human autosomal dominant polycystic kidney disease…).
In WO2010/092489 and related patents/patent applications, we identified several compounds which exhibited interesting properties for such applications. However, we have discovered that some of these compounds could be enhanced in their properties by selectively working on particular regions of their structures. However, the mechanism of action of these structures on kinases was not precisely elucidated at the time of WO2010/092489’s filing and thus it was unexpectedly that we found the high activities of the structures disclosed in the present application. The subject matter of the present invention is to offer novel multi-targeted kinase inhibitors, having an original backbone, which can be used therapeutically in the treatment of pathologies associated with deregulation of protein kinases including tumorigenesis, human immune disorders, inflammatory diseases, thrombotic diseases, neurodegenerative diseases, bone diseases, macular degeneration, fibrosis, cystogenesis. The inhibitors of the present invention can be used in particular for the treatment of numerous cancers and more particularly in the case of liquid tumors such hematological cancers (leukemias) or solid tumors including but not limited to squamous cell cancer, small- cell lung cancer, non-small cell lung cancer, gastric cancer, pancreatic cancer, glial cell tumors such as glioblastoma and neurofibromatosis, cervical cancer, ovarian cancer, liver cancer, bladder cancer, breast cancer, melanoma, colorectal cancer, endometrial carcinoma, salivary gland carcinoma, renal cancer, prostate cancer, vulval cancer, thyroid cancer, sarcomas, astrocytomas, and various types of hyperproliferative diseases.
Efforts were made to improve a series of potent dual ABL/SRC inhibitors based on a 7-azaindole core with the aim of developing compounds that demonstrate a wider activity on selected oncogenic kinases. Multi-targeted kinase inhibitors (MTKIs) were then derived, focusing on kinases involved in both angiogenesis and tumorigenesis processes. Antiproliferative activity studies using different cellular models led to the discovery of a lead candidate (6z) that combined both antiangiogenic and antitumoral effects. The activity of 6z was assessed against a panel of kinases and cell lines including solid cancers and leukemia cell models to explore its potential therapeutic applications. With its potency and selectivity for oncogenic kinases, 6z was revealed to be a focused MTKI that should have a bright future in fighting a wide range of cancers.
5-{2-Methyl-5-[3-(4-methyl-imidazol-1-yl)-5-trifluoromethyl-benzoylamino]-benzylamino}-1H-pyrrolo[2,3-b]pyridine-2-carboxylic Acid Methyl Ester (6z)
Rational Design, Synthesis, and Biological Evaluation of 7-Azaindole Derivatives as Potent Focused Multi-Targeted Kinase Inhibitors
https://www.google.com/patents/WO2010092489A1?cl=en
Example 91: Preparation of methyl 5-(5-(3-(trifluoromethγl)-5~(4-methyl-1 H-imidazol-1 – yl)benzamido)-2-methγlbenzylamino)-1H-pyrrolo[2,3-blpyridine-2-carboχylate (ND0126)
Step 1 : preparation of methyl 5-(3-(trifluoromethyl)-5-(4-methyl-1 H-imidazol-1 – yl)benzamido)-2-methylbenzoate
The compound is obtained using the procedures of example 88 (step 4) replacing the 4-((3-(dimethylamino)pyrrolidin-1-yl)methyl)-3-(trifluoromethyl)-benzoic acid
(Shakespeare W. C, WO2007133562) by the 3-(trifluoromethyI)-5-(4-methyl-1H- imidazol-1-yl)benzoic acid.
Step 2: preparation of 3-(tπϊluoromethyl)-N-(3-formyl-4-methylphenyl)-5-(4- methyl-1H-imidazol-1-yl)benzamide
The compound is obtained by using the procedures of examples 83 (steps 1 and 2) replacing the methyl 5-(4-((4-methylpiperazin-1-yl)methyl)benzamido)-2- methylbenzoate with the methyl 5-(3-(trifluorometny))-5-(4-metbyl-1H-imidazol-1- yl)benzamido)-2-methylbenzoate.
Step 3: preparation of methyl 5-(5-(3-(trifluoromethyl)-5-(4-methyl-1 H-imidazol- 1-yl)benzamido)-2-methylbenzylamino)-1H-pyrrolo[2,3-bJpyridine-2-carboxylate (ND0126)
The composed is obtained according to example 83 (step 3) replacing N-(3-formyl-4- methylphenyl)-4-((4-methylpiperazin~1-yl)methyl)-benzamide with the 3- (trifluoromethyl)-N-(3-formyl-4-methylphenyl)-5-(4-methyl-1 H-imidazol-1-yl)benzamide.
PATENT
REFERENCES
| WO2005063747A1 * | Dec 23, 2004 | Jul 14, 2005 | Pfizer Italia S.R.L. | PYRROLO[2,3-b] PYRIDINE DERIVATIVES ACTIVE AS KINASE INHIBITORS, PROCESS FOR THEIR PREPARATION AND PHARMACEUTICAL COMPOSITION COMPRISING THEM |
| WO2008028617A1 * | Sep 4, 2007 | Mar 13, 2008 | F. Hoffmann-La Roche Ag | Heteroaryl derivatives as protein kinase inhibitors |
| WO2008124849A2 * | Apr 10, 2008 | Oct 16, 2008 | Sgx Pharmaceuticals, Inc. | Pyrrolo-pyridine kinase modulators |
| WO2008144253A1 * | May 9, 2008 | Nov 27, 2008 | Irm Llc | Protein kinase inhibitors and methods for using thereof |
| WO2014102376A1 * | Dec 30, 2013 | Jul 3, 2014 | Oribase Pharma | Protein kinase inhibitors |
| WO2014102377A1 * | Dec 30, 2013 | Jul 3, 2014 | Oribase Pharma | Azaindole derivatives as multi kinase inhibitors |
| WO2014102378A1 * | Dec 30, 2013 | Jul 3, 2014 | Oribase Pharma | Azaindole derivatives as inhibitors of protein kinases |
| US20150353540 * | Dec 30, 2013 | Dec 10, 2015 | Oribase Pharma | Azaindole derivatives as inhibitors of protein kinases |
| US2011312959 | 2011-12-22 | Derivatives of Azaindoles as Inhibitors of Protein Kinases ABL and SRC |
///////ND 0126, 1240322-54-6, PRECLINICAL
O=C(OC)c1cc2cc(cnc2n1)NCc3cc(ccc3C)NC(=O)c4cc(cc(c4)n5cc(C)nc5)C(F)(F)F
CC1=C(C=C(C=C1)NC(=O)C2=CC(=CC(=C2)N3C=C(N=C3)C)C(F)(F)F)CNC4=CN=C5C(=C4)C=C(N5)C(=O)OC
AMG 337
PIC CREDIT.BETHANY HALFORD



Name: AMG-337(AMG337; AMG 337)
Cas 1173699-31-4
Formula: C23H22FN7O3
M.Wt: 463.46
Chemical Name: 6-[(1R)-1-[8-fluoro-6-(1-methylpyrazol-4-yl)-[1,2,4]triazolo[4,3-a]pyridin-3-yl]ethyl]-3-(2-methoxyethoxy)-5-methylidene-1,6-naphthyridine
(R)-6-(1-(8-fluoro-6-(1-methyl-1H-pyrazol-4-yl)-[1,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)-1,6-naphthyridin-5(6H)-one
(R)-6-(1-(8-Fluoro-6-(1-methyl-1H-pyrazol-4-yl)-[1,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)-1,6-naphthyridin-5(6H)-one
6-{ (lR)-l-[8-fluoro-6-(l-methyl-lH-pyrazol-4-yl)[l,2,4]triazolo[4,3-a]pyridin-3-yl]ethyl}-3-(2-methoxyethoxy)-l,6-naphthyridin-5(6H)-one (“Compound M”),
PHASE 2 CANCER OF ESOPHAGUS
AMG-337 is a potent and highly selective small molecule ATP-competitive MET kinase inhibitor. AMG 337 inhibits MET kinase activity with an IC50 of < 5nM in enzymatic assays.
IC50 value: < 5nM [1]
Target: MET
in vitro: AMG-337 demonstrates exquisite selectivity for MET when profiled against a diverse panel of over 400 protein and lipid kinases in a competitive binding assay. In cellular assays, AMG 337 inhibits HGF-dependent MET phosphorylation with an IC50 of < 10 nM. [1] AMG 337 is a selective inhibitor of Met, which inhibits multiple mechanisms of Met activation. [2]
in vivo: AMG-337 demonstrates robust activity in MET-dependent cancer models. Oral administration of AMG 337 results in robust dose-dependent anti-tumor efficacy in MET amplified gastric cancer xenograft models, with inhibition of tumor growth consistent with the pharmacodynamic modulation of MET signaling
AMG 337 is a potent and highly selective small molecule ATP-competitive MET kinase inhibitor that demonstrates robust activity in MET-dependent cancer models. In enzymatic assays, AMG 337 inhibited MET kinase activity with an IC50 less than 5 nM. AMG 337 demonstrated exquisite selectivity for MET when profiled against a diverse panel of over 400 protein and lipid kinases in a competitive binding assay. In cellular assays, AMG 337 inhibited HGF-dependent MET phosphorylation with an IC50 of less than 10 nM [1].
AMG 337 was profiled in cell viability assays using a diverse panel of over 200 cancer cell lines where on treatment with AMG 337 affected the viability of only two gastric cancer cell lines (SNU-5 and Hs746T), both of which harbor amplification of the MET gene. The AMG 337 IC50 in the two sensitive cell lines was less than 50 nM, and greater than 10 µM in all other tested cell lines.
The receptor tyrosine kinase c-Met and its natural ligand, hepatocyte growth factor (HGF), are involved in cell proliferation, migration, and invasion and are essential for normal embryonic development. Deregulation of c-Met/HGF signaling can lead to tumorigenesis and metastasis and has been implicated in a variety of cancers. Several mechanisms lead to deregulation, including overexpression of c-Met and/or HGF, amplification of the MET gene, or activating mutations of c-Met, all of which have been found in human cancers.
AMG 337 is a potent and highly selective inhibitor of wild-type and some mutant forms of MET. In a competitive binding assay conducted on 402 human kinases, AMG 337 bound only to MET. In a cell viability study, the only cell lines that responded to an AMG 337 analog were gastric cancer cells harboring MET gene amplification. None of the other cell lines were sensitive to the AMG 337 analog and none harbored MET gene amplification. In secondary pharmacology assays with transporters, enzymes, ion channels, and receptors, binding to the adenosine transporter was the only activity inhibited.
In vivo, oral administration of AMG 337 resulted in robust dose-dependent anti-tumor efficacy in MET amplified gastric cancer xenograft models, with inhibition of tumor growth consistent with the pharmacodynamic modulation of MET signaling. Further studies in an expanded panel of additional cancer cell lines derived from gastric, NSCLC, and esophageal cancer confirmed that the in-vitro anti-proliferative activity of AMG 337 correlated with amplification of MET. In those cell lines, treatment with AMG 337 inhibited downstream PI3K and MAPK signaling pathways, which translated into growth arrest as evidenced by an accumulation of cells in the G1 phase of the cell cycle, a concomitant reduction in DNA synthesis, and the induction of apoptosis [1].
In a small subset of patients with MET-amplified gastrointestinal (GI) tumors, monotherapy with the investigational agent AMG 337 produced a “dramatic” response. Of the 13 patients with MET-amplified gastric and esophageal cancers, eight experienced a response. The overall response rate in this group of patients was 62%. Response was rapid, with time to response being 4 weeks in most cases. Patients achieved tumor shrinkage and symptomatic improvement. One patient achieved a complete response and is still on treatment at 155 weeks; the others achieved partial responses or stable disease. This has led to further trials, including Phase II trials MET amplified gastric/esophageal adenocarcinoma or other solid tumors.
PAPER
Discovery of (R)-6-(1-(8-Fluoro-6-(1-methyl-1H-pyrazol-4-yl)-[1,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)-1,6-naphthyridin-5(6H)-one (AMG 337), a Potent and Selective Inhibitor of MET with High Unbound Target Coverage and Robust In Vivo Antitumor Activity.
Boezio, A.A., Copeland, K.W., Rex, K., K Albrecht, B., Bauer, D., Bellon, S.F., Boezio, C., Broome, M.A., Choquette, D., Coxon, A., Dussault, I., Hirai, S., Lewis, R., Lin, M.H., Lohman, J., Liu, J., Peterson, E.A., Potashman, M., Shimanovich, R., Teffera, Y., Whittington, D.A., Vaida, K.R., Harmange, J.C.
(2016) J.Med.Chem. 59: 2328-2342
http://pubs.acs.org/doi/abs/10.1021/acs.jmedchem.5b01716
Deregulation of the receptor tyrosine kinase mesenchymal epithelial transition factor (MET) has been implicated in several human cancers and is an attractive target for small molecule drug discovery. Herein, we report the discovery of compound 23 (AMG 337), which demonstrates nanomolar inhibition of MET kinase activity, desirable preclinical pharmacokinetics, significant inhibition of MET phosphorylation in mice, and robust tumor growth inhibition in a MET-dependent mouse efficacy model.
(R)-6-(1-(8-Fluoro-6-(1-methyl-1H-pyrazol-4-yl)-[1,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)-1,6-naphthyridin-5(6H)-one (23)
PATENT
WO 2009091374
http://www.google.com/patents/WO2009091374A2?cl=en
Example 515
(SV6-(l-f8-fluoro-6-(3-methvIisoxazol-5-vn-|l,2,41triazoIo[4,3-a1pyridin-3-vncthvn-3-(f2- methoxyethoxy)methv.)-l,6-naphthyridin-5(6HVone Synthesized in the same general manner as that previously described for example 509 using General Method N. Chiral separation by preparative SFC (Chiralpak® AD-H (20 x 150 mm, 5Dm), 25% MeOH, 75% CO2, 0.2% DEA; 100 bar system pressure; 75 mL/min; tr 4.75min). On the basis of previous crystallographic data and potency recorded for related compound in the same program, the absolute stereochemistry has been assigned to be the S enantiomer. M/Z – 465.2 [M+H], calc 464.16 for C23H2iFN6O4
Example 516 ri?)-6-ri-(8-fluoro-6-(l-methyl-lH-pyrazol-4-vn-H.2.41triazolo[4,3-alpyridin-3-yl)ethyl)- 3-(2-methoxyethoxy)-l,6-naphthyridin-5(6H)-one The title compound was synthesized using General Method N. Chiral separation by preparative SFC (Chiralpak® AS-H (20 x 150 mm, 5 Dm), 20% iPrOH, 80% CO2; 100 bar system pressure, 50 mL/min; tr 1.67 min). On the basis of previous crystallographic data and potency recorded for related compound in the same program, the absolute stereochemistry has been assigned to be the R enantiomer. M/Z = 464.2 [M+H], calc 463.18 for C23H22FN7O3. 1H NMR (400 MHz, CHLOROFORM-^ D ppm 2.15 (d, J=7.14 Hz, 3 H) 3.49 (s, 3 H) 3.80 – 3.90 (m, 2 H) 3.97 (s, 3 H) 4.27 – 4.39 (m, 2 H) 6.83 (d, J=7.73 Hz, 1 H) 7.00 – 7.13 (m, 2 H) 7.42 (d, J=7.82 Hz, 1 H) 7.61 (s, 1 H) 7.72 (s, 1 H) 8.15 (d, J=2.84 Hz, 1 H) 8.31 (s, 1 H) 8.72 (d, J=3.03 Hz, 1 H).
6-{ (lR)-l-[8-fluoro-6-(l-methyl-lH-pyrazol-4-yl)[l,2,4]triazolo[4,3-a]pyridin-3-yl]ethyl}-3-(2-methoxyethoxy)-l,6-naphthyridin-5(6H)-one (“Compound M”), which is a selective inhibitor of the c-Met receptor, and useful in the treatment, prevention, or amelioration of cancer:

PATENT
https://www.google.com/patents/WO2014210042A2?cl=en
The overall scheme for the preparation of Compound A is shown below. The optical purity of Compound A is controlled during the synthetic process by both the quality of the incoming starting materials and the specific reagents used for the transformations. Chiral purity is preserved during both the coupling reaction (the second step) and the dehydration reaction (the third step).

NAPH (S)-halopropionic NAPA
acid/ester

PREPARATION OF COMPOUND A
In one aspect, provided herein is a method for preparing Compound A, salts of Compound A, and the monohydrate form of Compound A. Compound A can be prepared from the NAPH, PYRH, and S-propionic acid/ester starting materials in three steps. First, NAPH and ^-propionic acid/ester undergo an S 2 alkylation reaction to result in (R)-2-(3-(2-methoxyethoxy)-5-oxo-l,6-naphthyridin-6(5H)-yl)propanoic acid/ester. The ^-propionic acid starting material produces (R)-2-(3-(2-methoxyethoxy)-5-oxo-l,6-naphthyridin-6(5H)-yl)propanoic acid (“NAPA”) in one step. The ^-propionic ester starting material first produces the ester analog of NAPA, and is subsequently hydrolyzed to form NAPA. During workup, the acid can optionally form a salt (e.g., HC1 or 2-naphthalenesulfonic acid).
Step 1:

NAPH (S)-2-halopropionic
acid/ester
1 2
wherein R is Br, CI, I, or OTf; and R is COOH or Ci-salkyl ester, and
when R is Ci^alkyl ester, the method of forming the NAPA or salt thereof further comprises hydrolyzing the Ci-salkyl ester to form an acid.
Second, NAPA and PYRH are coupled together to form (R)-N’-(3-fluoro-5-(lmethyl-lH-pyrazol-4-yl)pyridin-2-yl)-2-(3-(2-methoxyethoxy)-5-oxo- l,6-naphthyridin- 6(5H)yl)propanehydrazide (“HYDZ”).
Step 2:

Third, HYDZ is dehydrated to form Compound A.

The free base form of Compound A can be crystallized as a salt or a monohydrate.
Step 1: Alkylation of NAPH to form NAPA
The first step in the preparation of Compound A is the alkylation of NAPH to form NAPA. The NAPA product of the alkylation reaction is produced as a free base and is advantageously stable.
Thus, one aspect of the disclosure provides a method for preparing NAPA comprising admixing 3-(2-methoxyethoxy)-l,6-naphthyridin-5(6H)-one (“NAPH”):

Me
‘1 R2 , and a base, under conditions sufficient to form NAPA:

wherein R1 is Br, CI, I, or OTf; and
R2 is COOH or C^alkyl ester;
and when R2 is Ci_3alkyl ester, the method of forming the NAPA or salt thereof further comprises hydrolyzing the Ci-3alkyl ester to form an acid.
Me
The compound, R1 R2 , represents an (^-propionic acid and/or (S)- propionic ester
Me
(“(S)-propionic acid/ester”). When R1 R2 is an acid (i.e., R2 is COOH), NAPA is formed in one step:

-prop on c ac
Me
When R1 R2 is an ester (i.e., R2 is C1-3 alkyl ester), then the NAPA ester analog is formed, which can be hydrolyzed to form NAPA.


The SN2 alkylation of NAPH to form NAPA occurs with an inversion of
EXAMPLE 1
SYNTHESIS OF (R)-2-(3-(2-METHOXYETHOXY)-5-OXO-l,6-NAPHTHYRIDIN-6(5H)- YL)PROPANOIC ACID NAPHTHALENE-2-SULFONATE (NAPA)

Scheme 1: Synthesis of naphthyridinone acid 2-napsylate (NAPA)
NAPA was synthesized according to Scheme 1 by the following procedure. A jacket reactor (60 L) was charged with 3000 g (1.0 equivalent) of 3-(2-methoxyethoxy)-l,6-naphthyridin-5(6H)-one and 4646 g (2.0 equivalents) of magnesium ie/t-butoxide. 12 L (4.0 Vol) tetrahydrofuran was added to the reactor and an N2sweep and stirring were initiated. 2213 g (1.5 equivalents) of S-2-bromopropionic acid was added over at least 30 min, controlling the addition such that the batch temperature did not rise above 30 °C. The charge port was rinsed with tetrahydrofuran (0.5 Vol) after addition. The batch was then aged for at least 5 min at 25 °C. 1600 g (1.05 equivalents) of potassium iert-butoxide was added to the reactor in four portions (approximately equal) such that the batch temperature did not rise above 30 °C. The charge port was again rinsed with tetrahydrofuran (1.5 L, 0.5 Vol). The batch temperature was adjusted to 35+5 °C and the batch was aged for at least 12 h.
A separate 100 L reactor was charged with 6 L of 2-Metetrahydrofuran (2-MeTHF) (2.0 Vol), 8.4 L of water (1.5 Vol) and 9.08 L (4.0 equivalents) of 6 N HC1. The mixture from the 60 L reactor was pumped into the 100 L reactor, while maintaining the batch temperature at less than 45 °C.
The batch temperature was then adjusted to 20+5°C. The pH of the batch was adjusted with 6N HC1 (or 2N NaOH) solution until the pH was 1.4 to 1.9. The aqueous layer was separated from the product-containing organic layer. The aqueous layer was extracted with 2-MeTHF (2 Vol), and the 2-MeTHF was combined with the product stream in the reactor. The combined organic stream was washed with 20% brine (1 Vol). The organic layer was polish-filtered through a < ΙΟμιη filter into a clean vessel.
In a separate vessel, 1.1 equivalents of 2-Naphthalenesulfonic acid hydrate was dissolved in THF (2 Vol). The solution was polish-filtered prior to use. The 2-naphthalenesulfonic acid hydrate THF solution was added into the product organic solution in the vessel over at least 2 h at 25+5 °C. The batch temperature was adjusted to 60+5 °C and the batch was aged for 1+0.5 h. The batch temperature was adjusted to 20+5 °C over at least 2 h. The batch was filtered to collect the product. The collected filter cake was washed with THF (5.0 Vol) by displacement. The product cake was dried on a frit under vacuum/nitrogen stream until the water content was < lwt% by LOD.
The yield of the product (R)-2-(3-(2-methoxyethoxy)-5-oxo- l,6-naphthyridin-6(5H)-yl)propanoic acid naphthalene-2-sulfonate, was 87%. The chiral purity was determined using chiral HPLC and was found to be 98-99% ee. The purity was determined using HPLC, and was found to be > 98%.
Thus, Example 1 shows the synthesis of NAPA according to the disclosure.
EXAMPLE 2
SYNTHESIS OF (R)-N’-(3-FLUORO-5-(l-METHYL-lH-PYRAZOL-4-YL)PYRIDIN-2- YL)-2-(3-(2-METHOXYETHOXY)-5-OXO-l,6-NAPHTHYRIDIN-6(5H)- YL)PROPANEHYDRAZIDE (HYDZ)

Scheme 2: Synthesis of (R)-N’-(3-fluoro-5-(l-methyl- lH-pyrazol-4-yl)pyridin-2-yl)-2-(3-(2-methoxyethoxy)-5-oxo-l,6-naphthyridin-6(5H)-yl)propanehydrazide
HYDZ was synthesized according to Scheme 2 by the following procedure. A 60 L jacket reactor was charged with 2805.0 g (1.0 equivalent) of (R)-2-(3-(2-methoxyethoxy)-5-oxo-l,6-naphthyridin-6(5H)-yl)propanoic acid 2-napsylate (NAPA) and N,N-dimethylacetamide (DMAC) (4.6 mL DMAC per gram of NAPA). Stirring and an N2 sweep were initiated. 1.05 equivalents of N,N-diisopropylethylamine (DIPEA) was added while maintaining the batch temperature at less than 35°C. Initially the NAPA dissolves. A white precipitate formed while aging, but the precipitate had no impact on the reaction performance. 2197 g (1.10 equivalents) of 3-fluoro-2-hydrazinyl-5-(l-methyl- lH-pyrazol-4-yl)pyridine (PYRH) was added to the batch. The batch temperature was adjusted to 10+5 °C. 2208 g (1.2 equivalents) of N-(3-dimethylaminopropyl)-N-ethylcarbodiimide hydrochloride (EDC) was added in four portions (approximately equal) over at least 1 h (about 20 min interval per portion) at 10+5 °C.
The batch was aged until the amide conversion target was met. If the amide conversion target was not reached within 2 h, additional EDC was added until the conversion target was met. Once the target was met, the batch was heated to 55 °C until the solution was homogeneous. The batch was filtered through a <20 μ in-line filter into a reactor. The vessel and filter were rinsed with DMAC (0.2 mL DMAC/g of NAPA). The batch temperature was adjusted to 45+5 °C.
The reactor was charged with a seed slurry of (R)-N’-(3-fluoro-5-(l-methyl-lH-pyrazol-4-yl)pyridin-2-yl)-2-(3-(2-methoxyethoxy)-5-oxo-l,6-naphthyridin-6(5H)-yl)propanehydrazide (HYDZ) (0.01 equivalents) in water (0.3 mL/g).
The batch was aged at 50+5 °C for at least 30 min. The batch temperature was adjusted to 20+5°C over at least 2 h. The batch was aged at 20+5°C for at least 30 min. 2.90 mL water per g was added at 25+5 °C over at least 2 h. The batch was aged at 20+5 °C for at least 1 h. The batch slurry was filtered to collect the product. The product was washed with 30% DMAC/H20 (0.5 Vol) by displacement. The product cake was washed with water (3 Vol) by displacement. The product cake was dried on the frit under vacuum/nitrogen stream until the water content was < 0.2 wt% as determined by Karl Fischer titration (KF). The product was a white, crystalline solid. The yield was about 83-84%. The ee was measured by HPLC and was found to be > 99.8%ee. The purity was determined by HPLC and was found to be >99.8 LCAP (purity by LC area percentage).
Thus, Example 2 demonstrates the synthesis of HYDZ according to the disclosure.
EXAMPLE 3
SYNTHESIS OF (R)-6-(l-(8-FLUORO-6-(l-METHYL-lH-PYRAZOL-4-YL)- [l,2,4]TRIAZOLO[4,3-A]PYRIDIN-3-YL)ETHYL)-3-(2-METHOXYETHOXY)-l,6- NAPHTHYRIDIN-5(6H)-ONE HYDROCHLORIDE SALT (COMPOUND A-HCL) – ROUTE 1

Scheme 3 Route 1 – Synthesis of (R)-6-(l-(8-fluoro-6-(l-methyl- lH-pyrazol-4-yl)- [l,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)- l,6-naphthyridin-5(6H)-one hydrochloride
(R)-6-(l-(8-fluoro-6-(l-methyl-lH-pyrazol-4-yl)-[l,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)- l,6-naphthyridin-5(6H)-one hydrochloride salt (Compound A-
HC1) was synthesized according to Scheme 3, Route 1 by the following procedure. A 15 L reactor, Reactor 1, was charged with 750 g HYDZ and the reactor jacket temperature was adjusted to 20+5 °C. A nitrogen sweep was initiated in Reactor 1 and the condenser coolant (at 5+5 °C) was started. Acetonitrile (3.4 L, 4.5 Vol) was added to Reactor 1 and stirring was initiated. 420 g (2.5 equivalents) of 2,6-lutidine was added to the reactor.
A solution of diphenylphosphinyl chloride Ph2P(0)(Cl) was prepared by combining 850 g (2.3 equivalents) of Ph2P(0)(Cl) and 300 g acetonitrile in an appropriate container. The contents of the PH2P(0)(C1) solution were added to Reactor 1. The jacket temperature was adjusted over 60+30 min until the reflux temperature of the batch (approximately 85 °C) was reached. The reaction was stirred for 14+6 h. The batch temperature was reduced to 75+5 °C and the batch was sampled for IPT analysis. The expected result was < 2% HYDZ remaining. If the target was not met, the heating at reflux temperature was continued for 9+6 h. Sampling, analysis, and heating was repeated until a satisfactory conversion assay result was obtained (< 10% HYDZ was considered satisfactory, < 1% was actually achieved). The final sample was assayed for optical purity by HPLC, and was found to be > 99.5% ee.
A K2CO3/KCI quench solution (5.0 Vol) was prepared in advance by combining 555 g (3.1 equivalents) of potassium carbonate with 335 g (2.9 equivalents) of potassium chloride and 3450 g of water in an appropriate container. The quench solution was added to Reactor 1 over at least 15 min, maintaining the batch temperature at 60+5 °C. As the aqueous base reacted with excess acid some bubbling (C02) occurred. 3.0 L (4.0 Vol) of toluene was added to Reactor 1 at 65+5 °C. A sample of the batch was taken for IPT analysis. The lower (aqueous) phase of the sample was assayed by pH probe (glass electrode). The pH was acceptable if in the range of pH 8-11. The upper (organic) phase of the sample was assayed by HPLC.
The batch was agitated for 20+10 min at 65+5 °C. Stirring was stopped and the suspension was allowed to settle for at least 20 min. The aqueous phase was drained from Reactor 1 via a closed transfer into an appropriate inerted container. The remaining organic phase was drained from Reactor 1 via a closed transfer to an appropriate inerted container. The aqueous phase was transferred back into Reactor 1.
An aqueous cut wash was prepared in advance by combining 2.3 L (3.0 Vol) acetonitrile and 2.3 L(3.0 Vol) toluene in an appropriate container. The aqueous cut wash was added to Reactor 1. The batch was agitated for 20+10 min at 65+5 °C. The stirring was stopped and the suspension was allowed to settle for at least 20 min. The lower (aqueous) phase was drained from Reactor 1 via a closed transfer into an appropriate inerted container. The organic phase was drained from Reactor 1 via a closed transfer to the inerted container containing the first organic cut. The combined mass of the two organic cuts was measured and the organic cuts were transferred back to Reactor 1. Agitation was initiated and the batch temperature was adjusted to 60+10 °C. A sample of the batch was taken and tested for Compound A content by HPLC. The contents of Reactor 1 were distilled under vacuum (about 300-450 mmHg) to approximately 8 volumes while maintaining a batch temperature of 60+10 °C and a jacket temperature of less than 85 °C. The final volume was between 8 and 12 volumes.
The nitrogen sweep in Reactor 1 was resumed and the batch temperature adjusted to 70+5 °C. A sample of the batch was taken to determine the toluene content by GC. If the result was not within 0-10% area, the distillation was continued and concomitantly an equal volume of 2-propanol, up to 5 volumes, was added to maintain constant batch volume. Sampling, analysis, and distillation was repeated until the toluene content was within the 0-10% area window. After the distillation was complete, 540 g (450 mL, 3.5 equivalents) of hydrochloric acid was added to Reactor 1 over 45+15 min while maintaining a batch temperature at 75+5 °C.
A Compound A-HC1 seed suspension was prepared in advance by combining 7.5 g of Compound A-HC1 and 380 mL (0.5 Vol) of 3 propanol in an appropriate container. The seed suspension was added to Reactor 1 at 75+5 °C. The batch was agitated for 60+30 min at 75+5 °C. The batch was cooled to 20+5 °C over 3+1 h. The batch was agitated for 30+15 min at 20+5 °C. 2.6 L (3.5 Vol) of heptane was added to the batch over 2+1 h. The batch was then agitated for 60+30 min at 20+5 °C. A sample of the batch was taken and filtered for IPT analysis. The filtrate was assayed for Compound A-HC1. If the amount of Compound A-HC1 in the filtrate was greater than 5.0 mg/mL the batch was held at 20 °C for at least 4 h prior to filtration. If the amount of Compound A-HC1 in the filtrate was in the range of 2-5 mg.ML, the contents of Reactor 1 were filtered through a < 25 μιη PTFE or PP filter cloth, sending the filtrate to an appropriate container.
A first cake wash was prepared in advance by combining 1.5L (2.0 Vol) of 2-propanol and 1.5L (2.0 Vol) of heptane in an appropriate container. The first cake wash was added to Reactor 1 and the contents were agitated for approximately 5 min at 20+5 °C. The contents of Reactor 1 were transferred to the cake and filter. A second cake wash of 3.0L (4.0 Vol) of heptane was added to Reactor 1 and the contents were agitated for approximately 5 min at 20+5 °C. The contents of Reactor 1 were transferred to the cake and filter. The wet cake was dried under a flow of nitrogen and vacuum until the heptane content was less than 0.5 wt% as determined by GC. The dried yield was 701g, 85% as a yellow powder. The dried material was assayed for chemical purity and potency by HPLC and for residual solvent content by GC. The isolated product was 88.8% Compound A-HC1, having 99.8% ee and 0.6% water.
Thus, Example 3 shows the synthesis of Compound A-HCL according to the disclosure.
EXAMPLE 4
SYNTHESIS OF (R)-6-(l-(8-FLUORO-6-(l-METHYL-lH-PYRAZOL-4-YL)- [l,2,4]TRIAZOLO[4,3-A]PYRIDIN-3-YL)ETHYL)-3-(2-METHOXYETHOXY)-l,6- NAPHTHYRIDIN-5(6H)-ONE HYDROCHLORIDE SALT (COMPOUND A-HCL) – ROUTE 2

HYDZ A HCI
Scheme 4: Route 2 – Synthesis of (R)-6-(l-(8-fluoro-6-(l-methyl- lH-pyrazol-4-yl)- [l,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)- l,6-naphthyridin-5(6H)-one hydrochloride
(R)-6-(l-(8-fluoro-6-(l-methyl-lH-pyrazol-4-yl)-[l,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)- l,6-naphthyridin-5(6H)-one hydrochloride salt was synthesized according to Scheme 4, Route 2, by the following procedure. A clean and dry 60 L reactor was fitted with a reflux condenser, nitrogen inlet, and vented to a scrubber (Reactor 1). The jacket temperature of Reactor 1 was set to 20 °C. A scrubber was set up to the vent of Reactor 1, and aqueous bleach solution was charged to the scrubber. The circulating pump (commercial 5.25% NaOCl) was initiated. The scrubber pump was turned on and N2 sweep on Reactor 1 was started. Reactor 1 was charged with 2597 g (0.52 equivalents) of Lawesson’s reagent. Reactor 1 was then charged with 6000 g (1.0 equivalent) of HYDZ and 30 L (5.0 vol) acetonitrile (MeCN). Agitation of Reactor 1 was initiated. The reactor was heated to 50+5 °C and aged until an LC assay showed consumption of HYDZ (> 99% conversion).
The jacket temperature of a second clean and dry reactor, Reactor 2, was set to 50 °C. The contents of Reactor 1 were transferred to Reactor 2 through a 5 micron inline filter. Reactor 1 was rinsed with MeCN, and the rinse was transferred through the inline filter to Reactor 2. Reactor 2 was charged with toluene. (31.7 Kg)
In a separate container a solution of 16.7% K2C03 was prepared by adding 7200 g K2C03 and 36 L water to the container and shaking the container well until all the solid was dissolved. Half of the contents of the K2C03 solution was added to Reactor 2 over at least 10 min. The batch temperature of Reactor 2 was adjusted to 50+5 °C. The batch in Reactor 2 was agitated at 50+5 °C for at least 1 h. The agitation was stopped and the batch in Reactor 2 was allowed to phase separate. The aqueous phase was removed. The remaining contents of the K2C03 solution was added to Reactor 2 over at least 10 min. The batch temperature in Reactor 2 was adjusted to 50+5 °C. The batch in Reactor 2 was agitated at 50+5 °C for at least 1 h. The agitation was stopped and the batch in Reactor 2 was allowed to phase separate. The aqueous phase was removed.
The jacket temperature of a clean and dry reactor, Reactor 3, was set to 50 °C. The contents of Reactor 2 were transferred to Reactor 3 through a 5 micron in-line filter. The contents of Reactor 3 were distilled at reduced pressure. Isopropyl alcohol (IP A, 23.9 kg) was charged to Reactor 3 and then the batch was distilled down. IPA (23.2 kg) was again added to Reactor 3. The charge/distillation/charge cycle was repeated. The batch temperature in Reactor 3 was adjusted to 70+15 °C. Reactor 3 was then charged with DI water (1.8 L). Concentrated HC1 (1015 mL) was added to Reactor 3 over at least 15 min at 70+15 °C.
A seed of the Compound A-HCl was prepared by combining a seed and IPA in a separate container. The Compound A-HCl seed was added to Reactor 3 as a slurry. The batch in Reactor 3 was aged at 70+15 °C for at least 15 min to ensure that the seed held. The batch in Reactor 3 was cooled to 20+5 °C over at least 1 h. Heptane (24.5 kg) was added to Reactor 3 at 20+5 °C over at least 1 h. The batch was aged at 20+5 °C for at least 15 min. The contents of Reactor 3 were filtered through an Aurora filter fitted with a <25 μιη PTFE or PP filter cloth. The mother liquor was used to rinse Reactor 3.
A 50% v/v IP A/heptane solution was prepared, in advance, in a separate container by adding the IPA and heptane to the container and shaking. The filter cake from Reactor 3 was washed with the 50% IP A/heptane solution. If needed, the IP A/heptane mixture, or heptane alone, can be added to Reactor 3 prior to filtering the contents through the Aurora filter. The cake was washed with heptane. The cake was dried under nitrogen and vacuum until there was about < 0.5 wt% heptane by GC analysis. The product was analyzed for purity and wt% assay by achiral HPLC, for wt% by QNMR, for water content by KF, for form by XRD, for chiral purity by chiral HPLC, and for K and P content by ICP elemental analysis.
Compound A-HCl had a purity of 99.56 area% and 88.3 wt% assay by achiral HPLC, and 89.9 wt% by QNMR. The water content was 0.99 wt% as determined by KF. The chiral purity was 99.9%ee as determined by chiral HPLC. The P and K content was found to be 171 ppm and 1356 ppm, respectively, as determined by ICP elemental analysis.
Thus, Example 4 shows the synthesis of Compound A-HCl according to the disclosure.
EXAMPLE 5
SYNTHESIS OF (R)-6-(l-(8-FLUORO-6-(l-METHYL-lH-PYRAZOL-4-YL)- [l,2,4]TRIAZOLO[4,3-A]PYRIDIN-3-YL)ETHYL)-3-(2-METHOXYETHOXY)-l,6- NAPHTHYRIDIN-5(6H)-ONE (COMPOUND A) – ROUTE 3

Scheme 5: Route 3 – Synthesis of (R)-6-(l-(8-fluoro-6-(l-methyl-lH-pyrazol-4-yl)-[l,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)- l,6-naphthyridin-5(6H)-one (compound A)
(R)-6-(l-(8-fluoro-6-(l-methyl-lH-pyrazol-4-yl)-[l,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)- l,6-naphthyridin-5(6H)-one was synthesized according to Scheme 5, Route 3, by the following procedure. 0.760 g (1.6 mmol) N’-iS-fluoro-S-il-methyl-lH-pyrazol-4-yl)pyridin-2-yl)-2-(3-(2-methoxyethoxy)-5-oxo- l,6-naphthyridin-6(5H)-yl)propanehydrazide (HYDZ) and 0.62 g (2.4 mmol) triphenylphosphine were taken up in 16 mL THF. 0.31 mL (2.4 mmol) trimethylsilyl (TMS)-azide was added, followed by addition of 0.37 mL (2.4 mmol) DEAD, maintaining the reaction temperature below 33 °C. The reaction was stirred at room temperature for 50 minutes. The reaction mixture was concentrated in vacuo.
The crude material was taken up in dichloromethane and loaded onto silica gel. The crude material was purified via medium pressure liquid chromatography using a 90: 10: 1 DCM : MeOH : NH4OH solvent system. 350 mg, (48% yield) of (R)-6-(l-(8-fluoro-6-(l-methyl-lH-pyrazol-4-yl)-[l,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)-l,6-naphthyridin-5(6H)-one was collected as a tan solid. The (S) isomer was also collected. The product had a purity of 97% by HPLC.
Thus, Example 5 shows the synthesis of enantiomerically pure Compound A according to the disclosure.
EXAMPLE 6
SYNTHESIS OF (R)-6-(l-(8-FLUORO-6-(l-METHYL-lH-PYRAZOL-4-YL)- [l,2,4]TRIAZOLO[4,3-A]PYRIDIN-3-YL)ETHYL)-3-(2-METHOXYETHOXY)-l,6-NAPHTHYRIDIN-5(6H)-ONE (COMPOUND A) AND THE HYDROCHLORIDE SALT- ROUTE 3

Scheme 6: Route 3 – Synthesis of (R)-6-(l-(8-fluoro-6-(l-methyl-lH-pyrazol-4-yl)-[l,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)-l,6-naphthyridin-5(6H)-one (compound A) and the hydrochloride salt
(R)-6-(l-(8-fluoro-6-(l-methyl-lH-pyrazol-4-yl)-[l,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)-l,6-naphthyridin-5(6H)-one was synthesized according to Scheme 6, Route 3, by the following procedure. Benzothiazyl disulfide (3.31 g, 9.97 mmol), HYDZ (4.0 g, 8.31 mmol), and a stir bar were added to a 50 mL 3-neck flask fitted with a reflux condenser topped with a nitrogen inlet, a thermocouple and a septum. The flask headspace was purged with nitrogen, and the solids were suspended in MeCN (20.00 mL, 5 mL/g) at ambient conditions. The flask contents were heated to 50 °C on a heating mantle. Finally,
trimethylphosphine, solution in THF (9.97 ml, 9.97 mmol) was added dropwise by syringe pump with stirring over 1 h. An ice pack was affixed to the side of the flask in lieu of a reflux condenser. After about 0.5 h from addition, the resulting suspension was sampled and analyzed by, showing about 99% conversion of penultimate, and about 94% Compound A vs.
benzothiazole-2-thiol (“BtSH”) adduct selectivity.
After about 0.75 h from addition, the yellow reaction mixture was cooled to 0 °C in an ice bath, and 30% hydrogen peroxide in water (2.037 mL, 19.94 mmol) was added dropwise over 2 hours. The reaction solution was allowed to warm to room temperature overnight.
The suspension was heated to 30 °C, held at that temperature for 3 h and then cooled to room temperature. After cooling was complete, an aliquot was filtered and the filtrate was analyzed by liquid chromatography, showing 99% Compound A vs. BtSH adduct (91% purity for Compound A overall).
A Celite filtration pad about 0.5″ thick was set up on a 50 mL disposable filter frit and wetted with toluene (32.0 mL, 8 mL/g). The reaction suspension was transferred to the Celite pad and filtered to remove BtSH-related byproducts, washing with MeCN (2.000 mL, 0.5 mL/g). The filtrate was transferred to a 100 mL round bottom flask, and treated with 30 mL (7.5 Vol) of an aqueous quench solution consisting of sodium bicarbonate (7.5 ml, 8.93 mmol) and sodium thiosulfate (3.75 ml, 4.74 mmol) at overall about 5 wt% salt. The suspension was stirred for about 15 min and then the layers were allowed to separate. Once the layers were cut, the aqueous waste stream was analyzed by LC, showing 8% loss. The organic stream was similarly analyzed, showing 71% assay yield, implying about 20% loss to waste cake.
The organic cut was transferred to a 3-neck 50 mL round bottom flask with magnetic stir bar, thermocouple, and a shortpath distillation head with an ice-cooled receiving flask. The boiling flask contents were distilled at 55 °C and 300 torr pressure. The volume was reduced to 17 mL. The distillation was continued at constant volume with concomitant infusion of IPA (about 75 mL). The resulting thin suspension was filtered into a warm flask and water (0.8 mL) was added. The solution was heated to 80 °C. After this temperature had been reached, hydrochloric acid, 37% concentrated (0.512 ml, 6.23 mmol) was added, and the solution was seeded with about 30 mg (about 1 wt%) Compound A-HC1 salt. The seed held for 15 min. Next the suspension was cooled to 20 °C over 2 h. Finally heptane (17 mL, 6 Vol) was added over 2 h by syringe pump. The suspension was allowed to stir under ambient conditions overnight.
The yellow-green solid was filtered on an M-porosity glass filter frit. The wet cake was washed with 1: 1 heptane/IPA (2 Vol, 5.5 mL) and then with 2 Vol additional heptane (5.5 mL). The cake was dried by passage of air. The dried cake (3.06 g , 78.5 wt%, 94 LC area% Compound A, 62% yield) was analyzed by chiral LC showing optical purity of 99.6% ee.
Thus, Example 6 shows the synthesis of enantiomerically pure Compound A and the hydrochloric salt thereof, according to the disclosure.
EXAMPLE 7
RE-CRYSTALLIZATION OF COMPOUND A

A-HCI A monohydrate
Scheme 7: Re-crystallization of Compound A
Compound A-HCI was recrystallized to Compound A. A (60 L) jacketed reactor, Reactor 1, with a jacket temperature of 20 °C was charged with 5291 g, 1.0 equivalent of Compound A-HCI. 2 Vol (10.6 L) of IPA and 1 Vol (5.3 L) of water were added to Reactor 1 and agitation of Reactor 1 was initiated.
An aqueous NaHC03 solution was prepared in advance by charging NaHC03 (1112 g) and water (15.87 L, 3 Vol) into an appropriate container and shaking well until all solids were dissolved. The prepared NaHC03 solution was added to Reactor 1 over at least 30 min, maintaining the batch temperature below 30 °C. The batch temperature was then adjusted to about 60 °C. The reaction solution was filtered by transferring the contents of Reactor 1 through an in-line filter to a second reactor, Reactor 2, having a jacket temperature of 60+5 °C. Reactor 2 was charged with water (21.16 L) over at least 30 min through an in-line filter, maintaining the batch temperature at approximately 60 °C. After the addition, the batch temperature was adjusted to approximately 60 °C.
A seed was prepared by combining Compound A seed (0.01 equivalents) and IP A/water (20:80) in an appropriate container, in an amount sufficient to obtain a suspension. The seed preparation step was performed in advance. Reactor 2 was charged with the seed slurry. The batch was aged at 55-60 °C for at least 15 min. The batch was cooled to 20+5 °C over at least 1 h. The batch from Reactor 2 was recirculated through a wet mill for at least 1 h, for example, using 1 fine rotor stator at 60 Hz, having a flow rate of 4 L/min, for about 150 min.
The reaction mixture was sampled for particle size distribution during the milling operation. The solids were analyzed by Malvern particle size distribution (PSD) and
microscopic imaging. At the end of the milling operation a sample of the reaction mixture was again analyzed. The supernatant concentration was analyzed by HPLC, and the solids were analyzed by Malvern PSD and microscopic imaging to visualize the resulting crystals.
The batch temperature was adjusted to 35+5 °C and the batch was aged for at least 1 h. The batch was cooled to 20+5 °C over at least 2 h. The reaction mixture was sampled to determine the amount of product remaining in the supernatant. The supernatant concentration was analyzed by HPLC for target of <5 mg/mL Compound A in the supernatant. The contents of Reactor 2 were filtered through an Aurora filter fitted with a <25 μιη PTFE or PP filter cloth.
A 20% v/v IP A/water solution was prepared and the filter cake from Reactor 2 was washed with the 20% IP A/water solution. The cake was then washed with water. If needed, the IP A/water solution, or water alone, can be added to Reactor 2 prior to filtering to rinse the contents of the reactor. The cake was dried under moist nitrogen and vacuum until target residual water and IPA levels were reached. The product had 3.2-4.2% water by KF analysis. The product was analyzed by GC for residual IPA (an acceptable about less than or equal to about 5000 ppm). The yield and purity were determined to be 100% and 99.69% (by HPLC), respectively.
Thus, Example 6 shows the recrystallization of Compound A from the HC1 salt, Compound A-HC1, according to the disclosure.
EXAMPLE 8
SYNTHESIS OF (R)-6-(l-(8-FLUORO-6-(l-METHYL-lH-PYRAZOL-4-YL)- [l,2,4]TRIAZOLO[4,3-A]PYRIDIN-3-YL)ETHYL)-3-(2-METHOXYETHOXY)-l,6- NAPHTHYRIDIN-5(6H)-ONE (COMPOUND A)

HYDZ A
Scheme 8 Synthesis of (R)-6-(l-(8-fluoro-6-(l-methyl-lH-pyrazol-4-yl)-[l,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)-l,6-naphthyridin-5(6H)-one
(R)-6-(l-(8-fluoro-6-(l-methyl-lH-pyrazol-4-yl)-[l,2,4]triazolo[4,3-a]pyridin-3-yl)ethyl)-3-(2-methoxyethoxy)-l,6-naphthyridin-5(6H)-one was synthesized according to Scheme 8 by the following procedure. A clean and dry 60 L reactor was fitted with a reflux condenser, nitrogen inlet, and vented to a scrubber (Reactor 1). The jacket temperature of Reactor 1 was set to 20 °C. A scrubber was set up to the vent of Reactor 1, and aqueous bleach solution was charged to the scrubber. The circulating pump (commercial 5.25% NaOCl) was initiated. The scrubber pump was turned on and N2 sweep on Reactor 1 was started. Reactor 1 was charged with 1599.5 g (0.52 equivalents) of Lawesson’s reagent. Reactor 1 was then charged with 24.4 L acetonitrile (MeCN). Agitation of Reactor 1 was initiated. 3664.7 g (1.0 equivalent) of HYDZ was added to the reactor in portions over 1+0.5 h, using acetonitrile (5 L) as rinse. The reactor was heated to 50+5 °C and aged until an LC assay shows consumption of HYDZ (> 99% conversion).
The reactor was cooled to 20 °C and the reaction was assayed by HPLC for
Compound A. The assay showed a 99% crude yield of Compound A.
The contents of Reactor 1 were transferred to second reactor, Reactor 2, through a 1 micron inline filter. Reactor 2 was charged with 2 L of water. Reactor 2 was connected to a batch concentrator and vacuum distilled until a final volume of about 10 L. The jacket temperature was 50 °C during distillation and the pot temperature was maintained below 50 °C. The batch was then cooled to 20 °C.
In a separate container a solution of 10% K2CO3 was prepared by adding 1160 g K2CO3 and 10450 mL water to the container and shaking the container well until all the solid was dissolved. The K2CO3 solution was added to Reactor 2 through an in-line filter (5 μηι). 13 kg of purified water was added to the reactor through the in-line filter (5 μηι).
A Compound A seed was added to the reactor through an addition port. The resulting slurry was aged for one hour during which crystallization was observed. The reactor was placed under vacuum and charged with 16 L of water. The resulting slurry was aged at 20 °C overnight. The product slurry was filtered through a 25 μιη filter cloth and washed with 10 L of a 10% MeCN in water solution, followed by 12 L of water. The product was dried on a frit under a stream of ambient humidity filtered air.
Compound A was isolated as a monohydrate crystalline solid which reversibly dehydrates at < 11% RH. After drying, there was 3.9 wt.% water present in constant weight solid as determined by KF. 3.317 kg, 89% yield, of Compound A was isolated as a pale yellow solid. The product had a purity of 99.4 wt.% as determined by LCAP.
EXAMPLE 9
SYNTHESIS OF NAPH – ROUTE 1
CuBr (5-10%)

ethyl 5-bromo-2- Bromonaphthyridinone Naphthyridinone ether methylnicotinate
Scheme 9: Synthesis of NAPH – Route 1
The NAPH starting material for the synthesis of Compound A was synthesized according to Scheme 9, Route 1 by the following procedure. The jacket temperature of a 6 L jacketed reactor, Reactor 1, was set to 22 °C. 2409 g (1.0 equiv) of ethyl 5-bromo-2-methylnicotinate, 824 g (1.0 equivalent) of triazine, and 3.6 L dimethyl sulfoxide (DMSO) were added to the reactor. The jacket temperature was adjusted to 45 °C. The reactor was agitated until a homogenous solution resulted. Once complete dissolution has occurred (visually) the jacket of Reactor 1 was cooled to 22 °C.
A second, 60 mL reactor, Reactor 2, was prepared. 8.0 L of water was charged to a scrubber. 4.0 L of 10 N sodium hydroxide was added to the scrubber and the scrubber was connected to Reactor 2. The cooling condenser was started. 6411.2 g of cesium carbonate and 12.0 L of DMSO were added to Reactor 2. Agitation of Reactor 2 was initiated. The batch temperature of Reactor 2 was adjusted to 80 °C. The solution from Reactor 1 was added slowly over 1 h at 80 °C, while monitoring the internal temperature. 1.2 L of DMSO was added to Reactor 1 as a rinse. The DMSO rinse was transferred from Reactor 1 to Reactor 2 over 6 min. Reactor 2 was agitated for more than 1 h and the conversion to 3-bromo-l,6-naphthyridin-5(6H)-one was monitored by HPLC until there was < 1.0% ethyl 5-bromo-2-methylnicotinate remaining. When the reaction was complete the batch temperature was adjusted to 60 °C. 24.0 L (10V) of water was added to Reactor 2 over 2 h, maintaining a reaction temperature of 60+5 °C, using a peristaltic pump at 192 mL/min. Reactor 2 was cooled to 22 °C over 1 h 10 min. Stirring was continued at 22+5 °C until the supernatant assays for less than 3mg/mL of 3-bromo-l,6-naphthyridin-5(6H)-one (analyzed by HPLC). The crystallized product was filtered through an Aurora filter fitted with 25 μιη polypropylene filter cloth. The reactor and filter cake were washed with a 75 wt% H20-DMSO solution (3 Vol made from 1.6 L DMSO and 5.6 L water), followed by water (7.2 L, 3 Vol), and finally toluene (7.2 L, 3 Vol). The product cake was dried on the aurora filter under vacuum with a nitrogen stream at ambient temperature. The product was determined to be dry when the KF was < 2.0 wt% water. 2194 g of 3-bromo-l,6-naphthyridin-5(6H)-one was isolated as a beige solid. The chemical purity was 99.73%. The adjusted yield was 2031.6 g (91.9%).
The jacket temperature of a 100 L reactor, Reactor 3, was set to 15+5 °C. 6.45 L of 2-methoxyethanol was added to the reactor and agitation was initiated. (8107 g) lithium tert-butoxide was added portion- wise to the reactor, maintaining the reactor temperature in a range of 15 °C to 24 °C. 3795 g of 3-bromo-l,6-naphthyridin-5(6H)-one was added to the reactor. 4 mL of 2-methoxyethanol was added to rinse the solids on the wall of the reactor. The reactor contents were stirred for at least 5 min. The reaction mixture was heated to distillation to remove i-BuOH and water, under 1 atm of nitrogen (jacket temperature 145 °C). Distillation continued until the pot temperature reached 122+3 °C. The reactor contents were sampled and analyzed for water content by KF. The reaction mixture was cooled to less than 35 °C. 243 g CuBr was added to the reactor. The reaction mixture was de-gassed by applying vacuum to 50 torr and backfilling with nitrogen three times. The batch was heated to 120+5 °C while maintaining the jacket temperature below 150 °C. The batch was agitated (174 RPM) for 15.5 h. A sample of the reaction was taken and the reaction progress was monitored by HPLC. When the remaining 3-bromo-l,6-maphthyridin-5(6H)-one was less than 1%, the jacket temperature was cooled down to 25 °C.
An Aurora filter was equipped with a 25 μιη PTFE cloth and charged with Celite®. The reactor content was transferred onto the filter cloth and the filtrate was collected in the reactor. 800 mL of 2-methoxyethanol was added to the reactor and agitated. The reactor contents were transferred onto the filter and the filtrate was collected in the reactor. 5.6 L of acetic acid was added to the reactor to adjust the pH to 6.5, while maintaining the temperature at less than 32 °C. The batch was then heated to 80 °C. The reaction mixture was concentrated to 3.0+5 Vol (about 12 L) at 80+5 °C via distillation under vacuum.
In a separate container labeled as HEDTA Solution, 589.9 g of N-(2-hydroxyethyl)ethylenediaminetriacetic acid trisodium salt hydrate and 7660 mL water were mixed to prepare a clear solution. The HEDTA solution was slowly added to the reactor while maintaining the temperature of the batch at about 80-82 °C. The batch was then cooled to 72 °C.
An aqueous seed slurry of NAPH (31.3g) in 200 mL of water was added to the reactor. The slurry was aged for 30+10 min. 20 L of water was slowly added to the reactor to maintain the temperature at 65+5 °C. The batch was aged at 65+5 °C for 30 min. The batch was cooled to 20 °C over 1 h. The reactor contents were purged with compressed air for 1 h, and then the batch was further cooled to – 15 °C and aged for 12.5 h. The batch was filtered through a centrifuge fitted with 25 μιη PTFE filter cloth. 5.31 Kg of wet cake was collected (60-62 wt ). The wet cake was reslurried in 6V HEDTA solution and filtered through the centrifuge. The collected wet cake was dried in the centrifuge, and transferred to an Aurora filter for continued drying.
2.82 kg (76% isolated yield) of NAPH was collected having a 2.7% water content by KF.
Thus, Example 8 shows the synthesis of NAPH according to the examples.
EXAMPLE 10
SYNTHESIS OF NAPH – ROUTE 2


Scheme 10: Synthesis of NAPH via Route 2
The NAPH starting material for the synthesis of Compound A was synthesized according to Scheme 10, Route 2, by the following procedure.
Preparation of protected 2-methoxy-pyridin-4ylamine. A 1600 L reactor was flushed with nitrogen and charged with 120 L of N,N-dimethylacetamide, 100.0 kg 2-methoxy-pyridin-4-ylamine, and 89.6 kg triethylamine, maintaining the temperature of the reactor at less than 20 °C. In a separate container, 103.0 kg pivaloyl chloride was dissolved in 15.0 L of N,N-dimethylacetamide and cooled to less than 10 °C. The pivaloyl chloride solution was added to the reactor using an addition funnel over 3.2 hours while maintaining the reactor temperature between 5 °C and 25 °C. The addition funnel was washed with 15.0 L of N,N-dimethylacetamide, which was added to the reactor. The reaction was stirred for 2.3 hours at 20-25 °C. A sample of the reaction was taken and analyzed for 2-methoxy-pyridin-4ylamine by TLC. No 2-methoxy-pyridin-4ylamine remained in the solution and the reaction was aged at 20-25 °C under nitrogen over night. 1200 L of deionized water was added to the reaction over 2
hours at while the reaction was maintained at 5-15 °C. The resulting mixture was stirred at 15 °C for 2 hours and then cooled to 5 °C. The reaction was centrifugated at 700-900 rpm in 3 batches. Each batch was washed 3 times with deionized water (3x 167 L) at 800 rpm. The wet solids obtained were dried under vacuum at 55 °C for 18 hours in 2 batches, sieved and dried again under vacuum at 55 °C for 21 hours until the water content was < 0.2% as determined by KF. 80.4 kg (89.7% yield) of the protected 2-methoxy-pyridin-4ylamine was collected as a white solid.
Preparation of protected 3-formyl-4-amino-2-methoxypyridine. A 1600 L reactor was flushed with nitrogen and charged with 1000 L of THF and 70.5 kg of the protected 2-methoxy-pyridin-4ylamine. The reaction was stirred for 10 min at 15-25°C. The reaction was cooled to -5 °C and 236.5 kg of w-hexyllithium (solution in hexane) was added over 11.5 hours while maintaining the temperature of the reaction at <-4°C. The reaction was maintained at <-4°C for 2 hours. A sample of the reaction was quenched with D20 and the extent of the ortho-lithiation was determined by 1H NMR (98.2% conversion). 61.9 kg dimethylforaiamide (DMF) was added at <-4°C over 3.2 h. After stirring 7.5 hours at <-4°C, a sample of the reaction was assayed for conversion by HPLC (98.5% conversion).
A 1600 L reactor, Reactor 2, was flushed with nitrogen and charged with 145 L THF and 203.4 kg of acetic acid. The resulting solution was cooled to -5 °C. The content of the first reactor was transferred to Reactor 2 over 2.5 hours at 0 °C. The first reactor was washed with 50 L THF and the washing was transferred into Reactor 2. 353 L deionized water was added to Reactor 2 while maintaining the temperature at less than 5 °C. After 15 min of decantation, the aqueous layer was removed and the organic layer was concentrated at atmospheric pressure over 5 hours until the volume was 337 L. Isopropanol (350 L + 355 L) was added and the reaction was again concentrated at atmospheric pressure until the volume was 337 L. Distillation was stopped and 90 L of isopropanol was added to the reactor at 75-94 °C. 350 L of deionized water was added to the reactor at 60-80 °C over 1 h (the temperature was about 60-65 °C at the end of the addition). The reaction was cooled to 0-5 °C. After 1 hour, the resulting suspension was filtered. Reactor 2 was washed twice with deionized water (2x 140L). The washings were used to rinse the solid on the filter. The wet solid was dried under vacuum at 50 °C for 15 h. 71.0 kg (80% yield) of the protected 3-formyl-4-amino-2-methoxypyridine was produced. The purity of the formyl substituted pyridine was found to be 92.7% by LCAP.
A 1600 L reactor, Reactor 3, was flushed with nitrogen and successively charged with 190 L ethanol, 128.7 kg of protected 3-formyl-4-amino-2-methoxypyridine, 144 L of deionized water and 278.2 kg of sodium hydroxide. The batch was heated to 60-65°C and 329.8 kg of the bisulfite adduct was added over 1 h. After lh of stirring, a sample was taken for HPLC analysis which showed 100% conversion. The batch was aged 2 hours at 60-65 °C, then was allowed to slowly cool down to 20-25 °C. The batch was aged 12 h at 20-25 °C. The batch was filtered and the reactor was washed with water (2x 125 L). The washings were used to rinse the solid on the filter. The wet solid was transferred to the reactor with 500 L deionized water and heated to 45-50 °C for 1 h. The batch was allowed to return to 20-25 °C (24 h). The solid was filtered and the reactor was washed with deionized water (2x 250 L). The washings were used to rinse the solid on the filter. 112.5 kg of wet white solid was obtained (containing 85.1 Kg (dry) of the naphthyridine, 72.3% yield, greater than 97% purity as determined by HPLC). The wet product was used directly in the next step, without drying.
A 1600 L reactor was flushed with nitrogen and charged with 417 L of deionized water and 112.5 kg of the wet napthyridine. The scrubber was filled with 700 L of water and 92.2 kg monoethanolamine. A solution of hydrochloric acid (46.6 kg diluted in 34 L of deionized water) was added to the reactor at 15-20 °C over 10 minutes. The batch was heated to 60-65 °C for 3 h. A sample of the batch was taken and contained no remaining starting material as determined by TLC. A solution of concentrated sodium hydroxide (58.2 kg in 31 L of deionized water) was added to the reactor at 60-65 °C. 65% of the solution was added over 15 min and then the batch was seeded with crystallized NAPH. Crystallization was observed after 2.5 h and then the remaining35% of the sodium hydroxide solution was added (pH – 11.1). The batch was cooled to 25-30 °C and a solution of sodium phosphate monobasic (1.8 kg in 2.9 L of deionized water) was added over 25 min at 25-30 °C) (pH = 6.75). The batch was stirred at 15-20 °C for 12 hours and filtered. The reactor was washed twice with deionized water (2x 176 L). The washings were used to rinse the solid on the filter. The wet solid was dried under vacuum at 50 °C until the water content was < 5% (by KF), to give 78.1 kg (73.8% yield, > 95%)) of NAPH as a beige powder.
Thus, Example 9 shows the synthesis of NAPH according to the disclosure.
EXAMPLE 11
SYNTHESIS OF (R)-2-(3-(2-METHOXYETHOXY)-5-OXO-l,6-NAPHTHYRIDIN-6(5H)- YL)PROPANOIC ACID NAPHTHALENE-2-SULFONATE (NAPA)

6N HCI/ THF 80C

Scheme 11: Synthesis of NAPA, Route 3
NAPA was synthesized according to Scheme 11, Route 3 by the following procedure. 4.75 g of 3-(2-Methoxyethoxy)-l,6-naphthyridin-5(6H)-one was suspended in 45 mL of DMF. 2.58 mL (s)-methyl lactate and 9.05 g triphenylphosphine were added to the suspension. The reaction mixture was cooled to 0 °C. 5.12 mL diethyl azodicarboxylate (DEAD) was added dropwise via syringe. The mixture was stirred at 0 °C for 1 h. A sample of the reaction was taken and the reaction was determined to be complete by LCMS. The reaction mixture was concentrated under vacuum to give crude material as a yellow oil.
1 g of the crude material was loaded in dichloromethane onto a silia gel pre-column. The sample was purified using the Isco Combi-Flash System; column 40 g, solvent system hexane/ethyl acetate, gradient 0-100% ethyl acetate over 15 minutes. Product eluted at 100% ethyl acetate. The product fractions were combined and concentrated under vacuum. 256 mg of (R)-methyl 2-(3-(2-methoxyethoxy)-5-oxo-l,6-naphthyridin-6(5H)-yl)propanoate was collected as a pale yellow oil.
The remaining residue was partitioned between benzene and 6N aq hydrochloric acid (35.9 mL). The acidic layer was extracted with benzene (3x), diethyl ether (2x), ethyl acetate (2x) and dichloromethane (lx). The dichloromethane layer was back extracted with 6N aq. Hydrochloric acid (2x). The aqueous layer was diluted with THF (80 mL). The mixture was heated at 80 °C for 3 h. The reaction mixture was concentrated to remove the THF. The remaining acidic water layer was extracted with ethyl acetate and dichloromethane. The aqueous layer was concentrated under vacuum. The remaining solid was triturated with methanol. The mixture was filtered to remove the solid (naphthyridone). The methanol layer was concentrated under vacuum. The remaining solid was dried overnight on a freeze drier. 10.2 g of material was collected as a yellow solid. NAPA made up 72% of the material as determined by HPLC.
1.0 g of the crude material was dissolved in minimal hot iPrOH then filtered and cooled to RT. Crystallization didn’t occur; therefore the solution was cooled in the freezer overnight. A yellow precipitate formed. The solid was collected on a glass frit and was washed with minimal iPrOH. 171 mg of yellow solid was collected, which was NAPA with a small amount of naphthyridone by LC-MS and 1H NMR.
Acid-base extraction. About 1 g of the crude material was dissolved in saturated aqueous sodium bicarbonate. The crude material was extracted with dichloromethane. The pH of the aqueous layer was adjusted to 6-7 with acetic acid then extracted with dichloromethane. 11 mg of the product was isolated; the majority of the product remained in the aqueous layer. The pH was reduced to approximately 4-5 with additional acetic acid. The aqueous layer was extracted with dichloromethane, ethyl acetate, and 15% methanol/dichloromethane. The organic layers were concentrated under vacuum to yield 260 mg of NAPA as the free base, as determined by LC-MS.
Thus, Example 10 shows the synthesis of NAPA according to the disclosure.
EXAMPLE 12
SYNTHESIS OF BISULFITE ADDUCT
DMSO
(COCI)2
MeCX ,ΟΗ Et3N
O

aqueous solution
Scheme 12: Synthesis of bisulfite adduct
Method 1
The bisulfite adduct was synthesize according to Method 1 of Scheme 12 by the following procedure. A 2L round-bottom flask (RBF) was purged with nitrogen and charged with 73.1 mL of reagent grade oxalyl chloride and 693 mL methylene chloride. The batch was cooled to less than -40 °C. 88 mL of dimethyl sulfoxide was added to the flask via an addition funnel at less than -40 °C. After the addition, the batch was stirred for 10 in at -60 °C. 97 mL diethylene glycol monomethyl ether was added to the flask at less than -50 °C over 10 min. The resulting white slurry was stirred at -60 °C for 30 min. 229 mL triethylamine was added to the flask via an addition funnel at less than -30 °C over 1 h. The batch was warmed to RT. 300 mL MTBE was added to the flask and the batch was stirred for 15 min. The slurry was filtered through a fritted funnel and the cake was washed with 300 mL MTBE. The filtrate was concentrated to 350-400g and then filtered again to remove triethylamine-HCl salt, and the solid was rinsed with MTBE, resulting in 357.7 g of a slightly yellow filtrate solution. The solution was assayed by QNMR and comprised 19 wt (68 g) of the desired aldehyde (70% crude yield). The solution was concentrated to 150.2 g.
A 500 mL RBF was charged with 60.0 g sodium bisulfite and 150 mL of water to give a clear solution. The concentrated aldehyde solution was added to the aqueous bisulfite solution over 5 min. An exothermic temperature rising was observed up to 60 °C from 18 °C. The solution was rinsed with 15 mL water. The resulting yellow solution was cooled to RT and was stirred under a sweep of nitrogen overnight.. A QNMR of the solution was taken. The solution contained 43 wt.% of the bisulfite adduct (300 g, 70% yield).
Method 2
The bisulfite adduct was synthesized according to Method 2 of Scheme 12 by the following procedure. A 2500 L reactor was flushed with nitrogen and charged with 657.5 L of 2-methoxyethanol. 62.6 kg of lithium hydroxide monohydrate was added to the reactor while maintaining the temperature at less than 30 °C. The reactor was heated to 113+7 °C. 270 L of solvent were distilled over 1 h and then the reactor temperature was adjusted to 110 °C. 269.4 kg of bromoacetaldehyde diethyl acetal was added over 16 minutes, maintaining the temperature between 110 and 120 °C. The reaction was heated to reflux (115-127°C) for 13 hours. A sample of the reaction was assayed and conversion to 2-(2-methoxyethoxy)acetaldehyde was found to be 98.3%. The reaction was cooled to 15-20°C and 1305 L of methyl ie/t-butyl ether (MTBE) and 132 L of deionized water was added to the reactor. The reaction was stirred for 20 min and then was decanted. The aqueous layer was transferred into a 1600 L reactor and the organic layer was kept in the first reactor. The aqueous layer was extracted with 260 L of MTBE for 10 min. After 10 min decantation, the aqueous layer was removed and the organic layer was transferred to the first reactor. The mixed organic layers were washed twice, 15 min each, with a mixture of concentrated sodium hydroxide solution (2x 17.3 kg) diluted in deionized water (2x 120 L). The aqueous layers were removed, and the organic layer was concentrated at atmospheric pressure at 60-65 °C until the volume was 540 L. The organic layer was cooled down to 15-20 °C to give 2-(2-methoxyethoxy)acetaldehyde as an orange liquid solution (417.4 kg) containing 215.2 kg of pure product (87.3% yield) as determined by 1H NMR and HPLC assay.
A 1600 L reactor, Reactor 3, was flushed with nitrogen and charged with 595 L deionized water followed by 37.8 kg sulfuric acid over 25 minutes via addition funnel, while maintaining the temperature below 25 °C. The addition funnel was washed with 124 L of deionized water and the washing was added to Reactor 3.
A 2500 L reactor, Reactor 4, was flushed with nitrogen and charged with 417.4 kg of the solution of the 2-(2-methoxyethoxy)acetaldehyde. The content of Reactor 3 was transferred into Reactor 4 over 25 min while maintaining the temperature of Reactor 4 below 35 °C. The batch was aged at 30-35 °C for 3 hours. A sample of the batch was taken and assayed for 2-(2- methoxyethoxy)acetaldehyde. No 2-(2-methoxyethoxy)acetaldehyde remained. The batch was aged 5 h then cooled to 15-20 °C.
A solution of sodium carbonate (39.2 kg) in deionized water (196 L) was prepared in Reactor 3. The sodium carbonate solution was transferred to Reactor 4 over 25 min while maintaining the temperature of Reactor 4 below 30 °C. The pH of the resulting mixture was pH 5-6. 1.0 kg sodium carbonate was added by portion until the pH was about 7-8. A solution of sodium bisulfite (116.5 kg) in deionized water (218 L) was prepared in Reactor 3. The sodium bisulfite solution was transferred to Reactor 4 over 20 min while maintaining the temperature of Reactor 4 below 30 °C. Reactor 3 was washed with deionized water (15 L) and the washing was added to Reactor 4. The batch was stirred for 1.2 hours. 23.3 kg sodium bisulfite was added to Reactor 4 and the batch was aged overnight. The batch was concentrated under vacuum at 30-50 °C over 6.5 hours until precipitation was observed. The batch was cooled to 0-10°C at atmospheric pressure. After 30 min at 0-10 °C, the suspension was filtered on 2 filters. Reactor 4 was washed with deionized water (2x 23 L). The first washing was used to rinse the solid on the first filter and the second washing was used to rinse the solid on the second filter. Filtrates were joined to give 473.9 kg of an aqueous solution of the bisulfite adduct (202.5 kg of pure product, 76.3% yield) as a yellow liquid.
Thus, Example 11 shows the synthesis of the bisulfite adduct according to the invention.
EXAMPLE 13
SYNTHESIS OF 2,3-DIFLUORO-5-(l-METHYL-lH-PYRAZOL-4-YL)PYRIDINE

Scheme 13: Synthesis of 2,3-difluoro-5-(l-methyl-lH-pyrazol-4-yl)pyridine, precursor to PYRH
2,3-Difluoro-5-(l-methyl-lH-pyrazol-4-yl)pyridine was synthesized according to Scheme 13 by the following procedure. A boronic-ate complex slurry was prepared in a first 3-neck-2-L round-bottom flask (RBF #1). RBF #1 was charged with 141 g (66.4 wt%, 0.9 equivalents based on boronic ester) of lithium 2-hydroxy-4,4,5,5-tetramethyl-2-(l-methyl-lH-pyrazol-4-yl)-l,3,2-dioxaborolan-2-uide. 120 mL (1.6 Vol relative to 5-chloro-2,3-difluoropyridine) of nitrogen- sparged (2 h) 2-BuOH and 120 mL (1.6 Vol) nitrogen-sparged (2 h) water were added to RBF #1. Agitation and N2 sweep were initiated. The reaction was aged at 20 °C for at least 30 min (reactions aged to 24 h were also successful).
] A second 3-neck-2-L round-bottom flask (RBF #2) was charged with 1.48 g (0.004 equivalents) of Xphos-palladacycle and 450 mL (6 Vol relative to 5-chloro-2,3-difluoropyridine) of nitrogen- sparged (2 h) 2-BuOH. Vacuum/N2 flush was cycled through RBF #2 three times to inert the RBF with N2. The batch in RBF #2 was heated to 80 °C. 75 g (1.0 equivalents) of 5-chloro-2,3-difluoropyridine was added to RBF #2.
The slurry of boronic-ate complex was transferred from RBF #1 to a 500 mL dropping funnel. RBF #1 was rinsed with 30 mL (0.4 Vol) 2-BuOH. Using the dropping funnel, the slurry of boronic-ate complex was added over 1 h to the hot solution mixture in RBF #2. After 1 h, 95% conversion was observed. If greater than 90% conversion was not observed, additional boronic-ate complex slurry was added (0.1 equivalents at a time with 1.6 Vol of 1: 1 2-BuOH/water relative to boronic-ate complex). After the conversion was complete, the batch was cooled to 50 °C. While cooling, 600 mL (8 Vol) of toluene was added to RBF #2. 300 mL (4 Vol) of 20% w/v NaHS03 in water was added to RBF #2 and the batch was stirred at 50 °C for at least 1 h. The batch was polish filtered using a 5 micron Whatman filter at 50 °C, into a 2-L Atlas reactor. RBF #2 was rinsed with 30 mL (4.0 Vol) of a 1: 1 2-BuOH:toluene solution. The temperature of the batch was adjusted to 50 °C in the Atlas reactor while stirring. The stirring was stopped and the phases were allowed to settle for at least 15 min while maintaining the batch at 50 °C. The bottom, aqueous layer was separated from the batch. The Atlas reactor was charged with 300 mL (4 Vol) of a 20% w/v NaHS03 solution and the batch was stirred at 50°C for 1 h. The agitation was stopped and the phases were allowed to settle for at least 15 min at 50 °C. The bottom, aqueous layer was removed. Agitation was initiated and the Atlas reactor was charged with 200 mL (4 Vol) of 0.5 M KF while keeping the batch at 50 °C for at least 30 min. The agitation was stopped and the phases were allowed to settle for at least 15 min at 50 °C. The bottom, aqueous layer was removed. Agitation was initiated and the reactor was charged with 300 mL (4 Vol) of water. The batch was aged at 50 °C for at least 30 min. Agitation was stopped and the phases were allowed to settle for at least 15 min at 50 °C. The bottom, aqueous later was removed.
The organic phase was concentrated by distillation under reduced pressure (180 torr, jacket temp 70°C, internal temp about 50 °C) to a minimal stir volume (about 225 mL). 525 mL (7 Vol) of 2-BuOH was added to the Atlas reactor. The organic batch was again concentrated using reduced pressure (85-95 torr, jacket temp 75 °C, internal temp about 55 °C) to a minimal stir volume (about 125 mL). The total volume of the batch was adjusted to 250 mL with 2-BuOH.
525 mL (7 Vol) heptane was added to the slurry mixture in the Atlas reactor. The jacket temperature was adjusted to 100 °C and the batch was aged for more than 15 min, until the batch became homogeneous. The batch was cooled to 20 °C over at least 3 h. A sample of the mixture was taken and the supernatant assayed for 2,3-difluoro-5-(l-methyl-lH-pyrazol-4-yl)pyridine. If the concentration was greater than 10 mg/mL, the aging was continued for at least 1 h until the supernatant concentration was less than 10 mg/mL. The batch was filtered using a medium frit. The filter cake was washed with 150 mL (2 Vol) 30% 2-BuOH/heptane solution followed by 150 mL (2 Vol) heptane. The filter cake was dried under N2/vacuum. 76.64 g of 2,3-difluoro-5-(l-methyl-lH-pyrazol-4-yl)pyridine was isolated as a white solid (87% yield).
A 60 L jacketed reactor was fitted with a reflux condenser. The condenser cooling was initiated at 0+5 °C. The reactor was charged with 2612 g (1 equivalent) of 2,3-difluoro-5-(l-methyl-lH-pyrazol-4-yl)pyridine and placed under an atmosphere of nitrogen. 31.7 L (12.2 Vol) water was added to the reactor and the resulting slurry was nitrogen sparged for 1 h with agitation. 7221 mL (6 equivalents) of hydrazine (35 wt% in water) was added to the reactor under a nitrogen atmosphere. The reactor was heated to 100 °C for 2+2 h until reaction was complete by HPLC analysis. The reactor was cooled to 20 °C over 2+1 h at a rate of 40°C/h. The reactor contents were stirred for 10+9 hours until the desired supernatant assay (< 2mg/mL PYRH in mother liquor). The reactor contents were filtered through an Aurora filter fitted with 25 μιη polypropylene filter cloth. The collected filter cake was washed with 12.0 L (4.6 V) of water in three portions. The filter cake was dried on the Aurora filter for 4-24 h at 22+5 °C, or until the product contained less than 0.5% water as determined by KF. The dry product was collected. 2.69 kg (97% yield) 2,3-Difluoro-5-(l-methyl-lH-pyrazol-4-yl)pyridine was collected as a white crystalline solid. The solid had a water content of 12 ppm as determined by KF.
Thus, Example 12 shows the synthesis of 2,3-Difluoro-5-(l-methyl-lH-pyrazol-4-yl)pyridine, a precursor to PYRH, according to the disclosure.
EXAMPLE 14
SYNTHESIS OF PYRH – ROUTE 2

Scheme 14: Synthesis of 3-fluoro-2-hydrazinyl-5-(l-methyl-lH-pyrazol-4-yl)-pyridine (PYRH)
3-fluoro-2-hydrazinyl-5-(l-methyl-lH-pyrazol-4-yl)-pyridine was synthesized according to Scheme 14 by the following procedure. A 60 L jacketed reactor was fitted with a 5 L addition funnel and the jacket temperature was set to 20+5 °C. 36.0 L (15 Vol) of 2-methyltetrahydrofuran was added to the reactor via a 20 μιη inline filter with vacuum using polypropylene transfer lines. The solution was sparged by bubbling nitrogen through a dipstick in the solution for 1+0.5 h with agitation. After 1 h the dipstick was removed but the nitrogen sweep continued. 1.55 kg of sparged 2-MeTHF was removed to be used as rinse volumes. 36.7 g of Pd2dba3, 75.6 g X-Phos, 259 g of tetrabutylammonium bromide, and 7397 g of potassium phosphate tribasic were added to the reactor. The manhole was rinsed with 0.125 kg of sparged 2-MeTHF. The reactor was agitated and the nitrogen sweep continued for 1+0.5 h. Then the nitrogen sweep was stopped and the reaction left under a positive pressure of nitrogen.
3.6 L (1.5 Vol) of sparged water was prepared in advance by bubbling nitrogen through a 4 L bottle of water for 1+0.5 h. The nitrogen sparged water was transferred to the 5 L addition funnel via a 20 μηι inline filter with vacuum using polypropylene transfer lines, then slowly added to the reaction while maintaining the internal temperature at 20+5 °C. The 5 L addition funnel was replaced with a 2 L addition funnel. 2412 g of 5-chloro-2,3-difluoropyridine was added to the 2 L addition funnel. The 5-chloro-2,3-difluoropyridine was then added to the reaction through the 2 L addition funnel. The 2L addition funnel was rinsed with 0.060 kg of sparged 2-MeTHF. 83.8 g (1.15 equivalents) of l-methylpyrazole-4-boronic acid, pinacol ester was added to reactor, the reactor was swept with nitrogen for 1+0.5 h, then left under a positive pressure of nitrogen. The internal temperature of the reactor was adjusted to 70+5 °C. The batch was agitated at 70+5 °C for at least 4 hours after the final reagent was added. A sample was taken from the reaction and the reaction progress assayed for conversion. The progress of the reaction was checked every 2 hours until the reaction was completed (e.g., greater than 99% conversion). The batch was cooled to 20+5 °C.
A 20% w/v sodium bisulfite solution (12.0 L, 5 Vol) was prepared by charging 12.0 L of water then 2411 g sodium bisulfite to an appropriate container and agitating until
homogeneous. The 20% sodium bisulfite solution was transferred into the reactor and agitated for 30 minutes. The agitation was stopped, the phases allowed to settle, and the aqueous phase was removed. A 0.5 M potassium fluoride solution (12.0 L, 5 Vol) was prepared by charging 12.0 L of water and 348 g of potassium fluoride to an appropriate container and agitating until homogenous. The 0.5 M potassium fluoride solution was transferred into the reactor and agitated for 30 min. The agitation was stopped, the phases were allowed to settle, and the aqueous phase was removed. A 25% w/v sodium chloride solution (12.0 L, 5 Vol) was prepared by charging an appropriate container with 12.0 L of water and 2999 g of sodium chloride and agitating until homogeneous. The 25% sodium chloride solution was transferred into the reactor and agitated for 30 min. The agitation was stopped, the phases were allowed to settle, and the aqueous phase was removed from the reactor.
The organic phase was distilled at constant volume (36 L, 15 Vol) while maintaining the internal temperature of the reactor at 50+5 °C by adjusting the vacuum pressure until no more than 0.3% of water remained. 2-Methyltetrahydrofuran was added to the reactor as needed to
maintain constant volume. The batch was cooled to 20 °C and transferred into drums. The batch was transferred using a polish filter (using a 5 μιη inline filter) into a 60 L jacketed reactor with a batched concentrator attached. 1.2 L of 2-MeTHF was used to rinse the drums. The batch was concentrated to about 9 Vol while maintaining the internal temperature of the vessel at 50+5 °C by adjusting the vacuum pressure. The batch was then distilled at constant volume (22.0 L, 9Vol) while maintaining the internal temperature of the vessel at 50+5 °C by adjusting the vacuum pressure. Heptane was added with residual vacuum until a 15% 2-MeTHF:heptane supernatant mixture was obtained. The pressure was brought to atmospheric pressure under nitrogen. The reactor was cooled to 20+5 °C over 2+2 h. The batch was agitated at 20+5 °C until an assay of the supernatant indicated that the amount of product was 7 mg/mL 2,3-difluoro-5-(l-methyl-lH-pyrazol-4-yl)pyridine.
A 10% 2-MeTHF:heptane (7.2 L, 3 Vol) wash solution was prepared by mixing 720 mL of 2-MeTHF and 6.5 L of heptane. The batch slurry was filtered through an Aurora filter fitted with a 25 μιη polypropylene filter cloth, resulting in heavy crystals that required pumping with a diaphragm pump using polypropylene transfer lines through the top of the reactor while stirring. The mother liquor was recycled to complete the transfer. The reactor and filter cake were washed with two portions of the 10% 2-MeTHF:heptane wash solution (3.6 L each). The product cake was dried on a frit under a nitrogen stream at ambient temperature. The 2,3-difluoro-5-(l-methyl-lH-pyrazol-4-yl)pyridine was determined to be dry when the 1H NMR assay was < 0.05+0.05. 2.635 kg was isolated as an off white crystalline solid (85% yield).
A 60 L jacketed reactor was fitted with a reflux condenser. The condenser cooling was initiated at 0+5 °C. The reactor was charged with 2612 g (1 equivalent) of 2,3-difluoro-5-(l-methyl-lH-pyrazol-4-yl)pyridine and placed under an atmosphere of nitrogen. 31.7 L (12.2 Vol) water was added to the reactor and the resulting slurry was nitrogen sparged for 1 h with agitation. 7221 mL (6 equivalents) of hydrazine (35 wt% in water) was added to the reactor under a nitrogen atmosphere. The reactor was heated to 100 °C for 2+2 h until reaction was complete by HPLC analysis. The reactor was cooled to 20 °C over 2+1 h at a rate of 40°C/h. The reactor contents were stirred for 10+9 hours until the desired supernatant assay was reached (< 2mg/mL PYRH in mother liquor). The reactor contents were filtered through an Aurora filter fitted with 25 μιη polypropylene filter cloth. The collected filter cake was washed with 12.0 L
(4.6 V) of water in three portions. The filter cake was dried on the Aurora filter for 4-24 h at 22+5 °C, or until the product contained less than 0.5% water as determined by KF. The dry product was collected. 2.69 kg was isolated as a white crystalline solid (97% yield). The water content was determined to be 12 ppm by KF.
| WO2007075567A1 * | Dec 18, 2006 | Jul 5, 2007 | Janssen Pharmaceutica, N.V. | Triazolopyridazines as tyrosine kinase modulators |
| WO2007138472A2 * | May 18, 2007 | Dec 6, 2007 | Pfizer Products Inc. | Triazolopyridazine derivatives |
| WO2008008539A2 * | Jul 13, 2007 | Jan 17, 2008 | Amgen Inc. | Fused heterocyclic derivatives useful as inhibitors of the hepatocyte growth factor receptor |
| WO2008051805A2 * | Oct 18, 2007 | May 2, 2008 | Sgx Pharmaceuticals, Inc. | Triazolo-pyridazine protein kinase modulators |
| WO2008155378A1 * | Jun 19, 2008 | Dec 24, 2008 | Janssen Pharmaceutica Nv | Polymorphic and hydrate forms, salts and process for preparing 6-{difluoro[6-(1-methyl-1h-pyrazol-4-yl)[1,2,4]triazolo[4,3-b]pyridazin-3-yl]methyl}quinoline |
References:
1. Hughes, P. E.; et. al. Abstract 728: AMG 337, a novel, potent and selective MET kinase inhibitor, has robust growth inhibitory activity in MET-dependent cancer models. Cancer Res 2014, 74, 728.
2. Boezio, A. A.; et. al. Discovery and optimization of potent and selective triazolopyridazine series of c-Met inhibitors. Bioorg Med Chem Lett 2009, 19(22), 6307-6312.
3. ClinicalTrials.gov Phase 2 Study of AMG 337 in MET Amplified Gastric/Esophageal Adenocarcinoma or Other Solid Tumors. NCT02016534 (retrieved 10-06-2015)
4. ClinicalTrials.gov A Study of AMG 337 in Subjects With Advanced Solid Tumors. NCT01253707 (retrieved 10-06-2015)
/////////// AMG-337, AMG337, AMG 337, 1173699-31-4, AMGEN, ESOPHAGUS
O=C1C2=C(N=CC(OCCOC)=C2)C=CN1[C@@H](C3=NN=C4C(F)=CC(C5=CN(C)N=C5)=CN43)C
ATR 101

N-(2,6-bis(1-methylethyl)phenyl)-N’-((1-(4-(dimethylamino)phenyl)cyclopentyl) methyl)urea hydrochloride
N-(2,6-BIS(l-METHYLETHYL)PHENYL)-N’-((l-(4- (DIMETHYLAMINO)PHENYL)CYCLOPENTYL)METHYL)UREA
ATR-101; ATR 101; ATR101; PD132301-2; PD-132301-2; PD 132301-2; PD132301; PD-132301; PD 132301.
IUPAC/Chemical Name: 1-(2,6-diisopropylphenyl)-3-((1-(4-(dimethylamino)phenyl)cyclopentyl)methyl)urea hydrochloride
ATR-101 HCl
CAS#: 133825-81-7 (ATR-101 HCl); 133825-80-6 (ATR-101).
| Molecular Formula: | C27H40ClN3O |
|---|---|
| Molecular Weight: | 458.079 g/mol |
| The Regents Of The University Of Michigan, Atterocor, Inc. |
Millendo Therapeutics is developing ATR-101, an ACAT1 inhibitor, for treating adrenal cancers including adrenocortical cancer and congenital adrenal hyperplasia.

ATR-101, also known as PD-132301 (a free base) or PD-132301-2 (a HCl salt), is in clinical development for the treatment of adrenocortical carcinoma (ACC). ATR-101 is a selective inhibitor of ACAT1 (acyl coenzyme A:cholesterol acyltransferase). ACAT1 catalyzes cholesterol ester formation and, in the adrenals, is particularly important in creating a reservoir of substrate for steroid biosynthesis. ATR-101 is uniquely distributed to adrenal tissues and inhibition of adrenal ACAT1 by ATR-101 disrupts steroidogenesis and leads to selective apoptosis of steroid producing adrenocortical-derived cells. Similar effects have been seen in the human ACC cell line, H295R. ATR-101 has shown pre-clinical efficacy in H295R xenograft mouse models. ACC is an ultra-rare malignancy, occurring in about 2 per million population annually.
ATR-101 (Atterocor, Inc., Ann Arbor, MI, USA) is in clinical development for the treatment of adrenocortical carcinoma (ACC). ATR-101 is a selective inhibitor of ACAT1 (acyl coenzyme A:cholesterol acyltransferase). ACAT1 catalyzes cholesterol ester formation and, in the adrenals, is particularly important in creating a reservoir of substrate for steroid biosynthesis. ATR-101 is uniquely distributed to adrenal tissues and inhibition of adrenal ACAT1 by ATR-101 disrupts steroidogenesis and leads to selective apoptosis of steroid producing adrenocortical-derived cells. Similar effects have been seen in the human ACC cell line, H295R. ATR-101 has shown pre-clinical efficacy in H295R xenograft mouse models. ACC is an ultra-rare malignancy, occurring in about 2 per million population annually. ACC is frequently discovered in Stage 4 and the overall disease survival is approximately 17 months. Tumors often overproduce steroids normally produced in the adrenal cortex. Current therapies are toxic, difficult to administer, and poorly effective. Clinical trial information: NCT01898715.
Adrenocortical carcinoma (ACC) generally has poor prognosis. Existing treatments provide limited benefit for most patients with locally advanced or metastatic tumors. We investigated the mechanisms for the cytotoxicity, xenograft suppression and adrenalytic activity of ATR-101 (PD132301-02), a prospective agent for ACC treatment. Oral ATR-101 administration inhibited the establishment and impeded the growth of ACC-derived H295R cell xenografts in mice. ATR-101 induced H295R cell apoptosis in culture and in xenografts. ATR-101 caused mitochondrial hyperpolarization, reactive oxygen release and ATP depletion within hours after exposure, followed by cytochrome c release, caspase-3 activation, and membrane permeabilization. When combined with ATR-101, lipophilic free radical scavengers suppressed the reactive oxygen release, and glycolytic precursors prevented the ATP depletion, abrogating ATR-101 cytotoxicity. ATR-101 directly inhibited F1F0-ATPase activity and suppressed ATP synthesis in mitochondrial fractions. ATR-101 administration to guinea pigs caused oxidized lipofuscin accumulation in the zona fasciculata layer of the adrenal cortex, implicating reactive oxygen release in the adrenalytic effect of ATR-101. These results support the development of ATR-101 and other adrenalytic compounds for the treatment of ACC.
| Company | Millendo Therapeutics Inc. |
| Description | Selective inhibitor of sterol O-acyltransferase 1 (SOAT1; ACAT1) |
| Molecular Target | Sterol O-acyltransferase 1 (SOAT1) (ACAT1) |
PATENT
WO2013142214
https://www.google.co.in/patents/WO2013142214A1?cl=en
PATENT
One such promising agent is N-(2,6-bis( 1 -methylethyl)phenyl)-N’-(( 1 -(4-(dimethyl-amino)phenyl)cyclopentyl)methyl)urea hydrochloride (“ATR-101”). The free base form of ATR-101 has the following chemical structure:

The chemical synthesis of ATR-101 has been previously reported by Trivedi et al. (J. Med. Chem. 37: 1652-1659, 1994). This procedure, however, does not provide for ATR-101 in a form suitable for solid-dosing, particularly with regard to capsule or tablet formation, and does not provide for ATR-101 in high purity.
While significant advances have been made in this field, particularly in the context of ATR-101, there remains a substantial need for improved techniques and products for the oral administration of ATR-101 to patients in need thereof, including patients having ACC and/or other disorders or conditions such as Cushing’s syndrome and congenital adrenal hyperplasia (CAH).

EXAMPLE 1
SYNTHESIS OF SOLID DRUG FORM OF ATR-101

Step 1 : Preparation of Primary Amine 2 from the Nitrile 1

Tetrahyrofuran (THF) and Compound 1 are charged to a reactor vessel and a lithium aluminum hydride (LAH) solution in THF is added slowly. After the addition, the reaction mixture is warmed to 45°C and stirred until in-process HPLC analysis indicates that the reaction is complete. The reaction mixture is cooled to between 0 and 10°C and aqueous NaOH is added slowly while controlling the temperature to between 0 and 10°C. The mixture is then warmed to between 20 and 25°C and any inorganic salts removed by filtration. The solids are then washed with additional THF.
The filtrate is distilled under vacuum. Acetonitrile (MeCN) is added and the distillation continued to reduce the total volume. H20 is added and the solution is cooled to 20°C, and seeded if necessary. Additional water is added to the slurry and cooled to between 0 and 5°C and filtered. The crystallization vessel and filter cake is washed with MeCN and water (1 :2 mixture) and dried under vacuum between 40 to 45°C to produce Compound 2. Typical yield: 85%.
Step 2: Preparation of ATR-101 Free Base

2,6-Diisopropyl aniline hydrochloride (Compound 3) is converted to the corresponding free base by stirring in a mixture of dichloromethane (DCM) and 10% aqueous NaOH. The organic phase is separated and washed with water. The DCM solution containing the aniline free base is concentrated by distillation.
4-dimethylaminopyridine (DMAP) and DCM are charged to a separate reaction vessel. The mixture is cooled and a solution of di-tert-butyl dicarbonate (Boc20) in DCM is slowly added while the temperature is maintained between 0 and 5°C. The aniline free base solution is then slowly added to the reaction vessel. A complete conversion of aniline to the isocyanate is verified by in-process HPLC analysis.
Compound 2 and MeCN are charged to a separate vessel and this solution is cooled to between 0 and 5°C. The isocyanate intermediate solution
(prepared above) is slowly added while the temperature is maintained between 0 and 5°C, and stirred until in-process HPLC indicates that the reaction is complete.
The reaction mixture is distilled under vacuum, and isopropyl alcohol
(IP A) is added and the distillation is continued. The resulting solution is cooled and seeded, if necessary. After crystallization occurs, water is added and the mixture is cooled to between 0 and 5°C, and filtered. The crystallization vessel and filter cake is washed with isopropanol: water (1 : 1) and the product cake is dried under vacuum to yield ATR-101 as the free base. Typical yield: 89 %
Step 3 : Preparation of Solid Drug Form of ATR- 101

The ATR-101 free base is dissolved in acetone and filtered to remove particulates. Additional acetone is used to rinse the dissolution vessel and filter. Concentrated hydrochloric acid (HCl) is added while maintaining the reaction at room temperature. The resultant slurry is filtered and the cake is washed with acetone. The resulting solid is dried under vacuum between 40 and 45°C to obtain the solid drug form of ATR-101. Typical yield: 70-80 %.
EXAMPLE 2
CHARACTERIZATION OF THE SOLID DRUG FORM OF ATR-101
The solid drug form of ATR-101 was analyzed to fully characterize the material and provide proof of structure.
Elemental Analysis
An elemental (CHN) analysis was conducted, in duplicate, of the solid drug form of ATR-101. The results are summarized in Table 1 and are in agreement with the theoretical values calculated for the molecular ATR-101 drug substance formula of C27H39N3O HCl.
Table 1

Chloride Content
The solid drug form of ATR-101 is prepared as its HCl salt. To confirm the chloride content (and the stoichiometry), the hydrochloride salt was analyzed by Ion Chromatography using a validated method. The w/w% result showed 7.8% chloride present. The theoretical value for a mono hydrochloride salt is 7.7%. The experimental result conforms to the theoretical value for the mono-hydrochloride salt.
Mass Spectrometry
Mass spectrometry studies were conducted in accordance with
USP<736> using an AB Sciex API 2000 LC/MS/MS system. The samples were analyzed by electrospray ionization in positive mode. The base peak observed was 422.3 (M+H-HC1), consistent with the parent compound (see Figure 1). Two minor peaks were observed, at 301.3 and 202.3 (uncharacterized fragments). The combined data of the LC/MS and CFIN results support the molecular formula assignment of C27H39N3O and mass of 421.63 g/mol for the free base and C27H39N3O . HCl (mass of 458.09 g/mol) for the mono hydrochloride salt.
Nuclear Magnetic Resonance (NMR) – 1H NMR
The proton NMR spectrum of the solid drug form of ATR-101 was obtained using a Varian Gemini 400 MHz spectrometer and. The sample was dissolved in CD3OD. The resulting proton NMR spectrum is shown in Figure 2.
Two-Dimensional (2D) NMR
The 2D proton NMR spectrum (COSY) shown in Figure 3 confirmed some of the connectivity expected for the solid drug form of ATR-101. In particular the resonance at 1.2 ppm is strongly correlated to the resonance at 3.1. This correlation together with the splitting pattern observed for the peak at 3.1 strongly suggests an isopropyl moiety. Further, the data from these spectra show a strong correlation between each of the broad peaks at 1.6-2.2 ppm, consistent with a cycloalkyl functionality in which no heteroatoms or other non-alkyl substitution is present.
Carbon 13 NMR (13C NMR)
The 100 MHz 13C NMR spectrum of the solid drug form of ATR-101 was obtained using a Varian Gemini 400 MHz spectrometer. The sample was dissolved in CD3OD. The resulting 13C NMR spectrum is shown in Figure 4. The numbering of the carbon atoms for the analysis of the spectrum is shown below, and the interpretation is shown in Table 2. The observed signals are consistent with the structure of ATR-101.

Table 2


Fourier Transform Infrared Spectroscopy (IR)
Infrared (IR) spectroscopy was performed using the soid drug form of ATR-101. The resulting spectrum, shown in Figure 5, is consistent with the structure of ATR-101 drug substance. The major peak assignments are presented in Table_3.
Table 3

EXAMPLE 3
COMPARISON WITH PRIOR ART SYNTHESIS OF ATR-101 (BY TRIVEDI ETAL.. J. MED. CHEM. 37: 1652-1659, 1994)

ATR-101
In this experiment, 10.6 g of ATR-101 was synthesized according to the above procedure, which corresponds to the the procedure set forth in Trivedi et al., J. Med. Chem. 137: 1652-1659, 1994 (hereinafter referred to as the “Trivedi procedure”). The purity of ATR-101 as made by the Trivedi procedure was found to be 94.9%, compared to a purity of 98.3% for ATR-101 obtained by the procedure of Example 1 and as evaluated in Example 2.
Step 1 : Alkylation of p-nitrophenylacetonitrile

52
The initial alkylation reaction was run on 15.0 g scale and, according to the Trivedi procedure, should have given 15.7 g (79%) of product 52. However, several problems occurred, and the yield was much lower than expected (6.0 g, 30% yield), although the purity by 1H NMR and melting point (actual: 71-72°C, reported: 76°C) seemed good. Approximately half way through the addition of 1 ,4-bromobutane and p- nitrophenylacetonitrile to NaH, a black solid precipitated out of the purple solution causing the stirbar in the flask to skip and jump. The rate of stirring had to be monitored throughout the remainder of the addition to maintain a sluggish and inefficient mixing of the solution.
After stirring at ambient temperature overnight to ensure reaction completion, the reaction was worked-up as the procedure indicated. First, excess ether was removed using air bubbling, and the black solid was isolated by filtration. Diethyl ether was then added until all of the solids dissolved to give a clear black solution. However, upon washing the ether solution with 2N HC1, a black amorphous solid precipitated from the solution. There was no note of this black solid in the Trivedi procedure, so the work-up was continued without modification. The black solids ended up in the aqueous washes, or stuck to the seperatory funnel. The remainder of the work-up proceeded as expected, and the hot hexanes extraction of the crude solid resulted in light pink planar crystals.
The procedure was repeated with two changes thought to be responsible for the low yield: the anhydrous solvent (from the bottle) was sieve dried to remove trace water, and the stir bar was replaced with a mechanical stirrer to ensure more even mixing of the solution. The procedure was re-run on 10 g scale, which should have yielded 10.5 g of compound 52. However, despite the changes to the procedure, the resulting product and yield was nearly identical to the first run (4.5 g, 34% yield, 71-72°C melting point).
In an attempt to determine where the bulk of material ended up, the aqueous layer from this reaction was re-extracted with diethyl ether, but only resulted in trace amounts of material. The black solids that formed during the work-up were isolated by filtration, and an NMR was taken of the material. The NMR showed peaks corresponding to compound 52. Presumably, this amorphous black solid that resulted after HC1 formation is the main source of lost material, as there appeared to be several grams of it.
Ste 2: Reduction of Nitro Compound

The conversion of nitro compound 52 to the dimethyl amine 53 was done over two steps: palladium catalyzed hydrogenation of the nitro compound to give the free amine 52b, followed by imine formation & reduction to the dimethylamine 53.
An exploratory small scale reaction was run, using 1/10th of the available material (1.0 g compound 52). The reduction of the nitro compound on the 1 gram scale was very rapid, with hydrogen consumption ceasing after 3-4 hours. A crude NMR of an aliquot of the reaction mixture showed very clean amine (52b). The formaldehyde was added, as well as additional Pd/C, and the hydrogenation was continued. The hydrogen was not consumed as quickly for the imine reduction, and the reaction was still progressing when the vessel was pressurized to 55 psi and left shaking overnight (ca. 16h).
After 16 hours, the pressure in the flask had dropped to 30 psi, indicating that the hydrogenation was still progressing overnight. An aliquot NMR confirmed that the reaction had not proceeded to completion.
On large scale, the nitro reduction proceeded very smoothly, consuming hydrogen at a very rapid rate, and going to completion again within 3-4 hours. The reactor was pressurized to 55 psi and shaken overnight, as indicated in the original procedure, before more Pd/C was added, followed by formaldehyde. Hydrogen consumption was again observed to be very sluggish, so the valve to the hydrogen tank was left open to the vessel, and the reaction was shaken for 24 hours.
After 24 hours of shaking, the valve to the vessel was closed, and a drop of 5 psi was observed over 1 hour, indicating that the reaction had not progressed to completion. TLC also showed several polar products, suggesting that the reaction was only ca. 50% complete. The hydrogenation vessel was pressurized to 55 psi with hydrogen, and the valve again left open for an additional 24 hours of hydrogenation.
After 24 hours, the reaction stopped consuming hydrogen, and the vessel was purged and the contents filtered to remove the palladium catalyst. The work-up was performed similarly to the small scale, and the two reactions were combined prior to purification by column chromatography, giving 5.7g (57.5% yield) of the desired dimethylamine product 53.
Step 3 : Reduction of C ano Compound

A small scale RaNi hydrogenation was done and the test reaction went smoothly. Hydrogen consumption was rapid, and the reaction appeared complete after approximately 2 hours. The consumption of hydrogen had ceased, and TLC indicated that there was no compound 53 remaining. After filtration to remove the Raney Nickel, the reaction completion was confirmed by aliquot NMR.
The remaining material was subjected to reduction using the same conditions, and hydrogen consumption and TLC analysis again indicated reaction completion after 2 hours. The material was filtered and combined with the smaller scale reaction material. After concentration to dryness, the crude yield was found to be 5.5 g (96.5% yield), which was very close to the reported yield (99%>).
Step 4: Formation of Urea Com ound

Urea formation is a straightforward procedure, and the small scale test reaction with the amine 54 (500 mg) being combined with 1.0 equivalent of the
isocyanate in 20 parts ethyl acetate. After stirring for 16 hours, the solution was concentrated to dryness to give a white solid. Crude 1H NMR of the solid confirmed that the spectra matched the reported spectra in the Trivedi procedure.
The remaining material was carried forward to ATR-101 freebase without difficulty, and the lots of product were combined. In an effort to remove the residual ethyl acetate, the solids were dissolved in 10 mL of toluene, followed by concentration under reduced pressure. After drying on high- vacuum, ATR-101 freebase was isolated as a sticky white foam (10.6 g, 99% yield). The 1H NMR of the final product showed trace toluene even after extended drying, and the material was moved on to the HC1 salt formation.
The melting point of the solid was later taken and found to be surprisingly low (50-56°C, expected: 132-133°C). The nature of the solid (oily foam) made the determination of the melting point difficult, but it was judged to be completely melted above 60°C.
Step 5: Formation of HC1 Salt

To the ATR-101 freebase in toluene was added 37% HC1, and a gummy white solid precipitated out immediately. The solution was dried by Dean-Stark apparatus over approximately 3 hours with vigorous stirring and heating (bath temp: 160°C). After drying, the solution was cooled and the fine crystalline solid was isolated by filtration and washed with acetone and diethyl ether. The product ATR-101 was dried until a constant weight was achieved (10.6 g, 92% yield) and fully characterized.
Figure 1 is the LC/MS Mass spectrum of the solid drug form of ATR- 101.
Figure 2 is the proton NMR spectrum of the solid drug form of ATR- 101.
Figure 3 is the 2-D 1H NMR spectrum (COSY) of the solid drug form of ATR-101.
Figure 4 is the 13C NMR spectrum of of the solid drug form of ATR- 101.
Figure 5 is the FT-IR spectrum the solid drug form of ATR-101.
Paper
(J. Med. Chem. 37: 1652-1659, 1994
http://pubs.acs.org/doi/abs/10.1021/jm00037a016
| Patent ID | Date | Patent Title |
|---|---|---|
| EP0474733 | 1994-08-31 | ANTIHYPERLIPIDEMIC AND ANTIATHEROSCLEROTIC UREA COMPOUNDS. |
| WO9015048 | 1990-12-13 | ANTIHYPERLIPIDEMIC AND ANTIATHEROSCLEROTIC UREA COMPOUNDS |
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015087649 | 2015-03-26 | TREATING DISORDERS ASSOCIATED WITH ABERRANT ADRENOCORTICAL CELL BEHAVIOR |
| US2013267550 | 2013-10-10 | Compounds and Methods for Treating Aberrant Adrenocartical Cell Disorders |
| EP0858336 | 2006-12-20 | METHOD AND PHARMACEUTICAL COMPOSITION FOR REGULATING LIPID CONCENTRATION |
| US2005234124 | 2005-10-20 | Carboxyalkylether-ACAT inhibitor combinations |
| US2004072903 | 2004-04-15 | Carboxyalkylether-acat inhibitors combinations |
| US6143755 | 2000-11-07 | Pharmaceutical methods of treatment with ACAT inhibitors and HMG-CoA reductase inhibitors |
| US6124309 | 2000-09-26 | Method and pharmaceutical composition for regulating lipid concentration |
| US6093719 | 2000-07-25 | Method and pharmaceutical composition for regulating lipid concentration |
| WO9716184 | 1997-05-09 | METHOD AND PHARMACEUTICAL COMPOSITION FOR REGULATING LIPID CONCENTRATION |
| EP0474733 | 1994-08-31 | ANTIHYPERLIPIDEMIC AND ANTIATHEROSCLEROTIC UREA COMPOUNDS. |
References
1: Wolfgang GH, MacDonald JR, Vernetti LA, Pegg DG, Robertson DG. Biochemical alterations in guinea pig adrenal cortex following administration of PD 132301-2, an inhibitor of acyl-CoA:cholesterol acyltransferase. Life Sci. 1995 Feb 17;56(13):1089-93. PubMed PMID: 9001442.
2: Saxena U, Ferguson E, Newton RS. Acyl-coenzyme A:cholesterol-acyltransferase (ACAT) inhibitors modulate monocyte adhesion to aortic endothelial cells. Atherosclerosis. 1995 Jan 6;112(1):7-17. PubMed PMID: 7772069.
3: Reindel JF, Dominick MA, Bocan TM, Gough AW, McGuire EJ. Toxicologic effects of a novel acyl-CoA:cholesterol acyltransferase inhibitor in cynomolgus monkeys. Toxicol Pathol. 1994 Sep-Oct;22(5):510-8. PubMed PMID: 7899779.
4: Krause BR, Black A, Bousley R, Essenburg A, Cornicelli J, Holmes A, Homan R, Kieft K, Sekerke C, Shaw-Hes MK, et al. Divergent pharmacologic activities of PD 132301-2 and CL 277,082, urea inhibitors of acyl-CoA:cholesterol acyltransferase. J Pharmacol Exp Ther. 1993 Nov;267(2):734-43. PubMed PMID: 8246149.
5: Dominick MA, McGuire EJ, Reindel JF, Bobrowski WF, Bocan TM, Gough AW. Subacute toxicity of a novel inhibitor of acyl-CoA: cholesterol acyltransferase in beagle dogs. Fundam Appl Toxicol. 1993 Feb;20(2):217-24. PubMed PMID: 8383621.
6: Dominick MA, Bobrowski WA, MacDonald JR, Gough AW. Morphogenesis of a zone-specific adrenocortical cytotoxicity in guinea pigs administered PD 132301-2, an inhibitor of acyl-CoA:cholesterol acyltransferase. Toxicol Pathol. 1993;21(1):54-62. PubMed PMID: 8397438.
///////ATR 101, 133825-81-7, ATR-101 HCl, 133825-80-6, Millendo Therapeutics, ACAT1 inhibitor, treating adrenal cancers, adrenocortical cancer, congenital adrenal hyperplasia, Atterocor, Inc., Ann Arbor, MI, USA
O=C(NCC1(C2=CC=C(N(C)C)C=C2)CCCC1)NC3=C(C(C)C)C=CC=C3C(C)C.[H]Cl
PF-05387552
PF-05387552
IRAK4
- Molecular Weight429.51
| Molecular Formula: | C25H27N5O2 |
|---|---|
| Molecular Weight: | 429.51418 g/mol |

Synthesis
PAPER
Bioorganic & Medicinal Chemistry Letters (2014), 24(9), 2066-2072
Volume 24, Issue 9, 1 May 2014, Pages 2066–2072
Identification and optimization of indolo[2,3-c]quinoline inhibitors of IRAK4
- L. Nathan Tumeya, , ,
- Diane H. Boschellia,
- Niala Bhagiratha,
- Jaechul Shima,
- Elizabeth A. Murphyb,
- Deborah Goodwinb,
- Eric M. Bennettc,
- Mengmeng Wangd,
- Lih-Ling Linb,
- Barry Pressa,
- Marina Shenb,
- Richard K. Frisbiea,
- Paul Morganb,
- Shashi Mohanb,
- Julia Shinb,
- Vikram R. Raob
- b Pfizer Global R&D, 200 Cambridge Park Dr., Cambridge, MA 02140, USA
- c Pfizer Global R&D, 87 Cambridgepark Dr., Cambridge, MA 02140, USA
- d Pfizer Global R&D, 1 Burtt Rd., Andover, MA 01810, USA
http://www.sciencedirect.com/science/article/pii/S0960894X14002832?np=y
IRAK4 is responsible for initiating signaling from Toll-like receptors (TLRs) and members of the IL-1/18 receptor family. Kinase-inactive knock-ins and targeted deletions of IRAK4 in mice cause reductions in TLR induced pro-inflammatory cytokines and these mice are resistant to various models of arthritis.
Herein we report the identification and optimization of a series of potent IRAK4 inhibitors. Representative examples from this series showed excellent selectivity over a panel of kinases, including the kinases known to play a role in TLR-mediated signaling. The compounds exhibited low nM potency in LPS- and R848-induced cytokine assays indicating that they are blocking the TLR signaling pathway.
A key compound (26) from this series was profiled in more detail and found to have an excellent pharmaceutical profile as measured by predictive assays such as microsomal stability, TPSA, solubility, and c log P. However, this compound was found to afford poor exposure in mouse upon IP or IV administration. We found that removal of the ionizable solubilizing group (32) led to increased exposure, presumably due to increased permeability. Compounds 26 and 32, when dosed to plasma levels corresponding to ex vivo whole blood potency, were shown to inhibit LPS-induced TNFα in an in vivo murine model.
To our knowledge, this is the first published in vivo demonstration that inhibition of the IRAK4 pathway by a small molecule can recapitulate the phenotype of IRAK4 knockout mice.

L. Nathan Tumey, Ph.D., Principal Research Scientist, Pfizer Global R&D
REFERENCES
///////////TLR signaling, Indoloquinoline, IRAK4, Kinase inhibitor, Inflammation, PF-05387552, PF 05387552, 1604034-71-0
N#Cc3ccc4c5cnc2cc(OCCCN1CCN(C)CC1)c(OC)cc2c5nc4c3
Curis and Aurigene’s CA 4948, AU 4948
Example 13 WO2015104688
6-(6-aminopyridin-3-yl)-N-(2-morpholin-4-yl-1,3-benzothiazol-6-yl)pyridine-2-carboxamide
| Molecular Formula: | C22H20N6O2S |
|---|---|
| Molecular Weight: | 432.4982 g/mol |
PROBABLE STRUCTURE

Example 1 ……..6′-amino-N-(2-morpholinooxazolo[4,5-b]pyridin-6-yl)-[2,3′-bipyridine]-6-carboxamideWO2015104688
Compound-6: 6′-amino-N-(5-(cyclopropyIamino)-2-morpholinobenzo [d]oxazoI-6-yl)-[2,3′-bipyridine]-6-carboxamide.WO2013042137
PROBABLE CA 4948, AU 4948,AU-4948, CA-4948
STRUCTURE AND SYNTHESIS COMING……..
| Latest Stage of Development | Preclinical |
| Standard Indication | B cell lymphoma |
| Indication Details | Treat diffuse large B cell lymphoma (DLBCL) |
| Regulatory Designation | |
| Partner | Curis Inc. |
Interleukin-1 Receptor Associated Kinase-4 (IRAK-4) is a serine/threonine protein kinase belonging to tyrosine like kinase (TLK) family. IRAK-4 is one of the important signalling components downstream of IL-1/Toll family of receptors (IL-1R, IL-18R, IL-33R, Toll-like receptors). Recent studies have reported occurrence of oncogenic mutations in MYD88 in 30% of ABC diffuse large B cell lymphomas (ABC DLBCL) and 90% of Waldenstrom’s macroglobulinemia (WM). Most of ABC DLBCLs have a single amino acid substitution of proline for the leucine at position 265 (L265P) in the TIR domain of MYD88 protein resulting in constitutive activation of IRAK-4. Thus, IRAK4 is an attractive therapeutic target for the treatment of B-cell lymphomas with activating MYD88 L265P mutation. We have designed, synthesized and tested small molecule IRAK-4 inhibitors based on hits originating from Aurigene’ s compound library. These novel compounds were profiled for IRAK4 kinase inhibition, anti-proliferative activity, kinase selectivity, and drug-like properties. Furthermore, selected compounds were tested in a proliferation assay and pIRAK1 mechanistic assay using ABC-DLBCL cell lines with activating MYD88 L265P mutation, OCI-lLy10 and OCI-lLy3. We have identified a series of novel bicyclic heterocycles as potent inhibitors of IRAK-4. Aurigene Lead compound exhibited potent inhibitory activity for IRAK-4 with an IC50 of 3nM in biochemical assay. Aurigene Lead compound inhibited pIRAK1 levels, and proliferation of OCI-Ly3 and OCI-Ly10 cells with an IC501of 132nM and 52nM respectively. To the best of our knowledge, Aurigene Lead compound represents the most potent IRAK4 inhibitor reported for target modulation and anti-proliferative activity in DLBCL cell lines with activating MYD88 L265P mutation. Aurigene Lead compound has good oral pharmacokinetic profile in mice and has demonstrated excellent pharmacodynamic effect in an in vivo LPS induced TNF-α model with an ED50 of 3.8 mg/Kg in mice. Preliminary in vitro tox studies indicated clean safety profile. Demonstration of efficacy in OCI-lLy10 mouse tumor model is ongoing. In summary, a series of potent IRAK-4 inhibitors belonging to 3 different chemical series have been discovered and are being evaluated for treatment of B-cell lymphomas.
Curis with the option to exclusively license Aurigene’s orally-available small molecule inhibitor of Interleukin-1 receptor-associated kinase 4 (IRAK4) in the precision oncology field. Curis expects to exercise its option to obtain exclusive licenses to both programs and file IND applications for a development candidate from each in 2015.
Recent studies have also shown that alterations of the MYD88 gene lead to dysregulation of its downstream target IRAK4 in a number of hematologic malignancies, including Waldenström’s Macroglobulinemia and a subset of diffuse large B-cell lymphomas, making IRAK4 an attractive target for the treatment of these cancers.
Curis and Aurigene Announce Collaboration, License and Option Agreement to Discover, Develop and Commercialize Small Molecule Antagonists for Immuno-Oncology and Precision Oncology Targets
— Agreement Provides Curis with Option to Exclusively License Aurigene’s Antagonists for Immuno-Oncology, Including an Antagonist of PD-L1 and Selected Precision Oncology Targets, Including an IRAK4 Kinase Inhibitor —
— Investigational New Drug (IND) Application Filings for Both Initial Collaboration Programs Expected this Year —
— Curis to issue 17.1M shares of its Common Stock as Up-front Consideration —
— Management to Host Conference Call Today at 8:00 a.m. EST —
LEXINGTON, Mass. and BANGALORE, India, Jan. 21, 2015 (GLOBE NEWSWIRE) — Curis, Inc. (Nasdaq:CRIS), a biotechnology company focused on the development and commercialization of innovative drug candidates for the treatment of human cancers, and Aurigene Discovery Technologies Limited, a specialized, discovery stage biotechnology company developing novel therapies to treat cancer and inflammatory diseases, today announced that they have entered into an exclusive collaboration agreement focused on immuno-oncology and selected precision oncology targets. The collaboration provides for inclusion of multiple programs, with Curis having the option to exclusively license compounds once a development candidate is nominated within each respective program. The partnership draws from each company’s respective areas of expertise, with Aurigene having the responsibility for conducting all discovery and preclinical activities, including IND-enabling studies and providing Phase 1 clinical trial supply, and Curis having responsibility for all clinical development, regulatory and commercialization efforts worldwide, excluding India and Russia, for each program for which it exercises an option to obtain a license.
The first two programs under the collaboration are an orally-available small molecule antagonist of programmed death ligand-1 (PD-L1) in the immuno-oncology field and an orally-available small molecule inhibitor of Interleukin-1 receptor-associated kinase 4 (IRAK4) in the precision oncology field. Curis expects to exercise its option to obtain exclusive licenses to both programs and file IND applications for a development candidate from each in 2015.
“We are thrilled to partner with Aurigene in seeking to discover, develop and commercialize small molecule drug candidates generated from Aurigene’s novel technology and we believe that this collaboration represents a true transformation for Curis that positions the company for continued growth in the development and eventual commercialization of cancer drugs,” said Ali Fattaey, Ph.D., President and Chief Executive Officer of Curis. “The multi-year nature of our collaboration means that the parties have the potential to generate a steady pipeline of novel drug candidates in the coming years. Addressing immune checkpoint pathways is now a well validated strategy to treat human cancers and the ability to target PD-1/PD-L1 and other immune checkpoints with orally available small molecule drugs has the potential to be a distinct and major advancement for patients. Recent studies have also shown that alterations of the MYD88 gene lead to dysregulation of its downstream target IRAK4 in a number of hematologic malignancies, including Waldenström’s Macroglobulinemia and a subset of diffuse large B-cell lymphomas, making IRAK4 an attractive target for the treatment of these cancers. We look forward to advancing these programs into clinical development later this year.”
Dr. Fattaey continued, “Aurigene has a long and well-established track record of generating targeted small molecule drug candidates with bio-pharmaceutical collaborators and we have significantly expanded our drug development capabilities as we advance our proprietary drug candidates in currently ongoing clinical studies. We believe that we are well-positioned to advance compounds from this collaboration into clinical development.”
CSN Murthy, Chief Executive Officer of Aurigene, said, “We are excited to enter into this exclusive collaboration with Curis under which we intend to discover and develop a number of drug candidates from our chemistry innovations in the most exciting fields of cancer therapy. This unique collaboration is an opportunity for Aurigene to participate in advancing our discoveries into clinical development and beyond, and mutually align interests as provided for in our agreement. Our scientists at Aurigene have established a novel strategy to address immune checkpoint targets using small molecule chemical approaches, and have discovered a number of candidates that modulate these checkpoint pathways, including PD-1/PD-L1. We have established a large panel of preclinical tumor models in immunocompetent mice and can show significant in vivo anti-tumor activity using our small molecule PD-L1 antagonists. We are also in the late stages of selecting a candidate that is a potent and selective inhibitor of the IRAK4 kinase, demonstrating excellent in vivo activity in preclinical tumor models.”
In connection with the transaction, Curis has issued to Aurigene approximately 17.1 million shares of its common stock, or 19.9% of its outstanding common stock immediately prior to the transaction, in partial consideration for the rights granted to Curis under the collaboration agreement. The shares issued to Aurigene are subject to a lock-up agreement until January 18, 2017, with a portion of the shares being released from the lock-up in four equal bi-annual installments between now and that date.
The agreement provides that the parties will collaborate exclusively in immuno-oncology for an initial period of approximately two years, with the option for Curis to extend the broad immuno-oncology exclusivity.
In addition Curis has agreed to make payments to Aurigene as follows:
- for the first two programs: up to $52.5 million per program, including $42.5 million per program for approval and commercial milestones, plus specified approval milestone payments for additional indications, if any;
- for the third and fourth programs: up to $50 million per program, including $42.5 million per program for approval and commercial milestones, plus specified approval milestone payments for additional indications, if any; and
- for any program thereafter: up to $140.5 million per program, including $87.5 million per program in approval and commercial milestones, plus specified approval milestone payments for additional indications, if any.
Curis has agreed to pay Aurigene royalties on any net sales ranging from high single digits to 10% in territories where it successfully commercializes products and will also share in amounts that it receives from sublicensees depending upon the stage of development of the respective molecule.
About IRAK4:
Interleukin-1 receptor-associated kinase 4, or IRAK4 is a signaling kinase that becomes inappropriately activated in certain cancers including activated B cell-diffuse large B cell lymphoma (ABC-DLBCL), an aggressive form of lymphoma with poor prognosis. There appears to be a mechanistic link with IRAK4 in ABC-DLBCL where these tumors from approximately 35% of patients harbor oncogenic mutations in the MYD88 gene, which encodes an adaptor protein that interacts directly with IRAK4. MYD88 mutations appear to constitutively activate the IRAK4 kinase complex, driving pro-survival pathways in ABC-DLBCL disease. Oncogenic MYD88 mutations have also been identified in other cancers, including in over 90% of patients with Waldenström’s Macroglobulinemia as well as in a subset of patients with chronic lymphocytic leukemia (CLL).
About Curis, Inc.
Curis is a biotechnology company focused on the development and commercialization of novel drug candidates for the treatment of human cancers. Curis’ pipeline of drug candidates includes CUDC-907, a dual HDAC and PI3K inhibitor, CUDC-427, a small molecule antagonist of IAP proteins, and Debio 0932, an oral HSP90 inhibitor. Curis is also engaged in a collaboration with Genentech, a member of the Roche Group, under which Genentech and Roche are developing and commercializing Erivedge®, the first and only FDA-approved medicine for the treatment of advanced basal cell carcinoma. For more information, visit Curis’ website at www.curis.com.
About Aurigene
Aurigene is a specialized, discovery stage biotechnology company, developing novel and best-in-class therapies to treat cancer and inflammatory diseases. Aurigene’s Programmed Death pathway program is the first of several immune checkpoint programs that are at different stages of discovery and preclinical development. Aurigene has partnered with several large- and mid-pharma companies in the United States and Europe and has delivered multiple clinical compounds through these partnerships. With over 500 scientists, Aurigene has collaborated with 6 of the top 10 pharma companies. Aurigene is an independent, wholly owned subsidiary of Dr. Reddy’s Laboratories Ltd. (NYSE:RDY). For more information, please visit Aurigene’s website at http://aurigene.com/.
Small Molecule IRAK4 Kinase Inhibitor)
Innate immune responses mediated through Toll-like receptors or certain interleukin receptors are important mediators of the body’s initial defense against foreign antigens, while their dysregulation is associated with certain inflammatory conditions. Toll-like receptor and interleukin receptor signaling through the adaptor protein MYD88, results in the assembly and activation of IRAK4, initiating a signaling cascade that induces cytokine and survival factor expression mediated by the transcription factor NFκB. More recently, components of this pathway are recognized to be genetically altered and have important roles in specific human cancers. Toll-like receptor and interleukin receptor signaling through the adaptor protein MYD88, results in the assembly and activation of IRAK4, initiating a signaling cascade that induces cytokine and survival factor expression mediated by the transcription factor NFκB. MYD88 gene mutations are shown to occur in approximately 30% of Activated B-Cell (ABC) subtype of diffuse large B-cell lymphomas (DLBCL)1,2 and in over 90% of the B-cell malignancy Waldenstrom’s macroglobulinemia.3 Due to IRAK4’s central role in these signaling pathways, it is considered an attractive target for generation of therapeutics to treat these B-cell malignancies as well as certain inflammatory diseases.
As part of the collaboration with Aurigene, in October 2015 we exercised our option to exclusively license a program of orally-available, small molecule inhibitors of IRAK4 kinase, including the development candidate, CA-4948. Curis expects to file an IND and initiate clinical testing of CA-4948 in patients with advanced hematologic cancers during the second half of 2016.
1Nature. 2011; 470(7332):115–1192Immunology and Cell Biology. 2011; 89(6):659–6603N Engl J Med. 30, 2012; 367(9):826–833
CLIP
In November 2015, preclinical data were presented at the 2015 AACR-NCI-EORTC Molecular Targets and Cancer Therapeutics Conference in Boston, MA
Aurigene Collaboration (IRAK4 Inhibitor):
In October 2015, Curis exercised its option to exclusively license a program of orally available small molecule inhibitors of IRAK4 kinase, a serine/threonine kinase involved in innate immune responses as well as in certain hematologic cancers. The Company has since designated the development candidate as CA-4948 and expects to file an IND application for this molecule during 2016.
In November 2015, Curis’ collaborator Aurigene presented preclinical data from the IRAK4 program at the 2015 AACR-NCI-EORTC Molecular Targets and Cancer Therapeutics Conference in Boston, MA. This presentation included data from chemically distinct series of small molecule compounds with potent IRAK4 inhibitory activity in biochemical assays as well as in in vivo preclinical models, including MYD88 mutant DLBCL xenograft tumor models as well as a model of inflammatory disease.
CLIP
In April 2014, preclinical data presented at the CHI’s Ninth Drug Discovery Chemistry Conference in San Diego, CA, showed the compounds in vivo to have activity down to 10 mg/kg .
CLIP
April 24-25 2014
Drug Discovery Chemistry – CHI’s Ninth Annual Conference: Fifth Annual Kinase inhibitor Chemistry, San Diego, CA, USA
Novel IRAK4 inhibitors
Susanta Samajdar from Aurigene Discovery Technologies presented the discovery of new IRAK4 (IL-1 receptor-associated kinase 4) inhibitors. Research began with a HTS campaign using two types of libraries: rationally designed novel scaffolds by hopping and morphing of known IRAK4 inhibitors and novel scaffolds identified by virtual screening of drug-like commercial library. A benzoxazol series was identified and crystallography was used to help their design. Lead optimization culminated in the identification of very potent compounds (AU-2807 and AU-2202) in cell assay (inflammation pathway and oncology pathway, respectively). The compounds were also active against Flt3 and KDR. Some PD in vivo data using LPS and TNFalpha release were presented in which the compound showed activity down to 10 mg/kg: no other in vivo model data were disclosed, but it was mentioned that studies in the CIA (collagen induced arthritis) model was ongoing. Dr Samajdar answered to three questions, one related to IRAK1 selectivity (the answer was that the compound is fully selective against IRAK1 and IRAK2). It was also mentioned that the compounds have a PBB higher than 98%. And the last question was related to the synergetic effect with BTK inhibitor in activated B-cell like diffuse large B-cell lymphoma, and this effect was observed with these compounds.

Research Director at Aurigene Discovery Technologies
PATENT
http://www.google.com/patents/WO2013042137A1?cl=en
Compound-6: Synthesis of 6′-amino-N-(5-(cyclopropyIamino)-2-morpholinobenzo [d]oxazoI-6-yl)-[2,3′-bipyridine]-6-carboxamide.
Step_l^N-cyclopropyl-2-morpholino-6-nitrobenzo[d]oxazol-5-amine.
N-cyclopropyl-2-moφholino-6-nitrobenzo[d]oxazol-5-amine(0.7g,70%) was prepared from 5-fluoro-2-mo holino-6-nitrobenzo[d]oxazole(lg,Intermediate-2) by treating with cyciopropanamine in sealed tube at 100°C for 8-14h. The progress of the reaction was monitored by TLC. After the reaction was completed, it was extracted with water (15ml) and dichioromethane (2x 15ml). The organic layer was collected, washed with brine, dried over sodium sulfate and concentrated under reduced pressure to get the crude. MS (ES) m/e 305(M+1, 50%).
Steg2:6-bromo-N-(5-(cyclopropylamino)-2-morpholinobenzo[d]oxazol-6-yl)
picolinamide.
Step Π and ii):The process of these steps are adopted from step 2 and step 3 of compound- 1.
Step3:6′-amino-N-(5-(cvclopropvlamino)-2-morpholinobenzord]oxazol-6-yl)-r2,3′- bipyridine]-6-carboxamide.
(i) N-(4-methoxybenzyl)-5-(4,4,5,5-tetramethyl-l ,3,2-dioxaborolan-2-yl)pyridin
Na2C03, Pd(dppf)Cl2, ACN, H20, 80-100°C, 8-14h; TFA, 60-70°C, 8-14h.
6′-amino-N-(5-(cyclopropylamino)-2-mo holinobenzo[d]oxazol-6-yl)-[2,3′-bipyridine]-6- carboxamide (0.03g,61%) was prepared from 6-bromo-N-(5-(cyclopropyIamino)-2- moφholinobenzo[d]o azoI-6-yl)picolinamide(0.07g, step-3) by following the same process used in step-1 and 2 of compound-3.
Ή NMR (400 MHz, DMSO-< ):6 1 1.63 (s, IH), 8.90 (s, IH), 8.61 (s, IH), 8.55 (s, IH), 8.37- 8.03 (m, 2H), 7.39 (s, IH), 6.80-6.62 (s, IH), 3.80-3.59 (m, 15H), 2.88-2.64 (m, 2H). MS (ESI): 472 (M+l , 60%).
PATENT
Example 13
6′-amino-N-(2-morphol ne]-6-carboxamide

Step-1: Synthesis of 6-chloro thiazolo[4,5-c]pyridine-2(3H)-thione
Using the same reaction conditions as described in step 1 of example 1, 4,6-dichloropyridin-3-amine (1.3 g, 7 mmol) was cyclised using potassium ethyl xanthate (2.55 g, 15 mmol) in DMF (25mL) at 150°C for 8h to afford the title compound (1.3 g, 86.6 %) as a light brown solid.
1HNMR (400 MHz, DMSO-d6): δ 14.2-14.0 (b, 1H), 8.274 (s, 1H), 7.931 (s, 1H); LCMS: 100%, m/z = 201.3 (M+l)+.
Step-2: Synthesis of 4-(6-chloro thiazolo[4,5-c]pyridin-2-yl) morpholine
To a suspension of 6-chlorothiazolo[4,5-c]pyridine-2(3H)-thione (0.3 g, 1.16 mmol) in
DCM (4 mL), oxalyl chloride (0.2 mL, 2.38 mmol) and DMF (1.5 mL) were added at 0°C. The resulting mixture was slowly allowed to warm to room temperature and stirred there for 1 h. The reaction mixture was again cooled to 0°C and triethyl amine (0.66 mL, 4.76 mmol) and morpholine (0.13 mL, 1.75 mmol) were added. The reaction mixture was stirred at RT for 1 h and quenched with water and extracted with ethyl acetate. The combined organic layers were washed with water, brine, dried over sodium sulphate and concentrated under reduced pressure. The crude material was purified by column chromatography (EtOAc/n-hexanes 3:7) to afford the title compound (0.14 g, 39.6 %) as a light brown solid.
1H NMR (400 MHz, DMSO-d6): δ 8.47 (s, 1H), 8.04 (s, 1H), 3.74-3.72 (m, 4H), 3.61-3.59 (m, 4H); LCMS: m/z = 256.1 (M+l)+.
Step-3: Synthesis of 6′-amino-/V-(2-morpholino thiazolo [4,5-c]pyridin-6-yl)-[2,3′-bipyridine]-6-carboxamide
Using the same reaction conditions as described in step 4 of example 12, 4-(6-chlorothiazolo[4,5-c] pyridin-2-yl) morpholine (0.081 g, 0.32 mmol), was coupled with tert-butyl (6-carbamoyl-[2,3′-bipyridin]-6′-yl)carbamate (intermediate 2) (0.1 g, 0.32 mmol) using cesium carbonate (0.21 g, 0.64 mmol), XantPhos (0.028g, 0.047mmol) and Pd2(dba)3 (0.015 mg, 0.015 mmol) in toluene : dioxane (2:2mL) to get the crude product. The resultant crude was purified by 60-120 silica gel column chromatography using 2% methanol in DCM as eluent. Further the resultant crude was purified by prep HPLC to afford title compound (0.01 g, 6 %) as an off-white solid.
1H NMR (400 MHz, DMSO-d6): δ 10.65 (s, 1H), 8.88 (d, 1H), 8.85 (dd, 1H), 8.71 (s, 1H), 8.55 (s, 1H), 8.22-8.13 (m, 4 H), 7.09 (d, 1H), 3.73 (t, 4H), 3.58 (t, 4H). LCMS: 100%, m/z = 434.2 (M+l)+.
Example 11
(S)-2-(2-methylpyridin-4-yl)-N-(2-morpholino-5-(pyrrolidin-3-ylamino)oxazolo[4,5-b]pyridin-6-yl)oxazole-4-carboxamide

Step l:Preparation of (S)-tert-butyl 3-((2-morpholino-6-nitrooxazolo[4,5-b]pyridin-5-yl)amino)pyrrolidine- 1 -carboxylate
A solution of 5-chloro-2-morpholino-6-nitrooxazolo[4,5-b]pyridine (300mg, 1.0563 mmol) (S)-tert-butyl 3 -aminopyrrolidine- 1 -carboxylate (237mg, 1.267 mmol) and potassium carbonate (292mg, 2.112 mmol) in DMF (2mL) was heated at 100°C for 2h. Reaction was quenched with ice water and filtered the solid. The resultant crude was purified by 60-120 silica gel column chromatography using 1 % methanol in DCM as eluent to obtain the title compound (350mg, 76.25%). LCMS: m/z: 435.4 (M+l)+.
Step 2:Preparation of (S)-tert-butyl 3-((6-amino-2-morpholinooxazolo[4,5-b]pyridin-5-yl)amino)pyrrolidine- 1 -carboxylate
Using the same reaction conditions as described in step 5 of example 1, (S)-tert-butyl 3- ((2-morpholino-6-nitrooxazolo[4,5-b]pyridin-5-yl)amino)pyrrolidine-l -carboxylate (350mg, 0.806 mmol) was reduced with zinc dust (422mg, 6.451 mmol) and ammonium chloride (691mg, 12.903 mmol) in THF/methanol/H20 (10mL/2mL/lmL) to get the title compound (240mg, 71.8%). LCMS: m/z: 405.2 (M+l)+.
Step 3:Preparation of (S)-tert-butyl 3-((6-(2-(2-methylpyridin-4-yl)oxazole-4-carboxamido)-2-morpholinooxazolo[4,5-b]pyridin-5-yl)amino)pyrrolidine-l-carboxylate
Using the same reaction conditions as described in step 6 of example 1, (S)-tert-butyl 3-((6-amino-2-morpholinooxazolo[4,5-b]pyridin-5-yl)amino)pyrrolidine-l -carboxylate (115mg, 0.284 mmol), was coupled with 2-(2-methylpyridin-4-yl)oxazole-4-carboxylic acid (70mg, 0.341 mmol) using EDCI.HCl (82mg, 0.426 mmol), HOBt (58mg, 0.426 mmol), DIPEA (0.199mL, 1.138 mmol) in DMF (2mL) to afford the title compound (lOOmg, 59.52%). LCMS: m/z: 591.4 (M+l)+.
Step 4: Preparation of (S)-2-(2-methylpyridin-4-yl)-N-(2-morpholino-5-(pyrrolidin-3-ylamino)oxazolo[4,5-b]pyridin-6-yl)oxazole-4-carboxamide
Using the same reaction conditions as described in step 8 of example 1, (S)-tert-butyl 3- ((6-(2-(2-methylpyridin-4-yl)oxazole-4-carboxamido)-2-morpholinooxazolo[4,5-b]pyridin-5-yl)amino)pyrrolidine-l -carboxylate (lOOmg, 0.169 mmol) was deprotected using methanolic HC1 (5mL) to get the crude product. This was then purified by prep HPLC to get the title compound (9mg, 10.84%).
1HNMR (CDCI3, 400MHz): δ 9.91 (s, 1H), 8.78 (s, 1H), 8.74-8.73 (d, 1H), 8.45 (s, 1H), 7.82 (s, 1H), 7.76-7.74 (d, 1H), 4.50 (s, 1H), 4.04-4.03 (d, 4H), 3.30-3.00 (m, 7H), 2.70 (s, 3H), 2.40-1.80 (m, 4H), 1.00-0.08 (m, 1H). LCMS: 100%, m/z = 491.3 (M+l)+.
REFERENCES
http://www.curis.com/images/stories/pdfs/posters/Aurigene_IRAK4_AACR-NCI-EORTC_2015.pdf
http://www.curis.com/images/stories/pdfs/posters/Aurigene_IRAK4_AACR_20150421.pdf
1Nature. 2011; 470(7332):115–119
2Immunology and Cell Biology. 2011; 89(6):659–660
3N Engl J Med. 30, 2012; 367(9):826–833
April 2014, preclinical data presented at the CHI’s Ninth Drug Discovery Chemistry Conference in San Diego, CA
November 2015, preclinical data were presented at the 2015 AACR-NCI-EORTC Molecular Targets and Cancer Therapeutics Conference in Boston, MA
http://pubs.acs.org/doi/abs/10.1021/jm5016044
http://cancerres.aacrjournals.org/content/75/15_Supplement/3646
2015 Apr 18-22; Philadelphia, PA. Philadelphia (PA): AACR; Cancer Res 2015;75(15 Suppl):Abstract nr 3646. doi:10.1158/1538-7445.AM2015-3646
////////IRAK4 Kinase Inhibitor, Curis, Aurigene, CA 4948, AU 4948, CA-4948, AU-4948, 1428335-77-6
c21ccc(cc1sc(n2)N3CCOCC3)NC(c4nc(ccc4)c5ccc(nc5)N)=O
सुकून उतना ही देना प्रभू, जितने से जिंदगी चल जाये। औकात बस इतनी देना, कि औरों का भला हो जाये।
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO …..FOR BLOG HOME CLICK HERE
Join me on Facebook FACEBOOK
 LIONEL MY SON
LIONEL MY SON
सुकून उतना ही देना प्रभू, जितने से
जिंदगी चल जाये।
औकात बस इतनी देना,
कि औरों का भला हो जाये।
Read all about Organic Spectroscopy on ORGANIC SPECTROSCOPY INTERNATIONAL 



















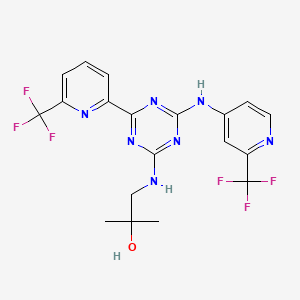

















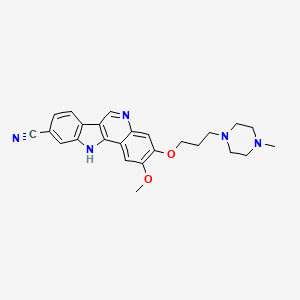









 Susanta Samajdar, Ph.D., Research Director, Medicinal Chemistry, Aurigene Discovery Technologies Limited
Susanta Samajdar, Ph.D., Research Director, Medicinal Chemistry, Aurigene Discovery Technologies Limited





