An efficient process for the preparation of 1-(2-methoxyphenoxy)-2,3-epoxypropane, a key intermediate for the synthesis of ranolazine is described.
Home » Uncategorized (Page 52)
Category Archives: Uncategorized
EMA issues new Guideline on “Chemistry of Active Substances”
DRUG REGULATORY AFFAIRS INTERNATIONAL

The new EMA “Guideline on the chemistry of active substances” represents the current state of the art in regulatory practice and fits into the context of the ICH Guidelines Q8-11. Find out what information regarding active substances European authorities expect in an authorization application.
A medicinal product authorization application requires comprehensive information on origin and quality of an active substance. What information is required was defined in two Guidelines so far: the Guideline “Chemistry of Active Substances” (3AQ5a) from 1987 and the “Guideline on the Chemistry of New Active Substances” from 2004. Because both Guidelines’ content do not take into account the ICH Guidelines Q8-11 issued in the meantime and do thus not meet the current state of the art in sciences and in regulatory practice, the EMA Quality Working Party (QWP) developed an updated document entitled “Guideline on the chemistry of active substances” (EMA/454576/2016), which was issued…
View original post 738 more words
MODANAFIL

MODANAFIL
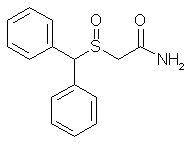
| Modafinil; 68693-11-8; Provigil; Modiodal; 2-[(diphenylmethyl)sulfinyl]acetamide; Modafinilum [Latin] | |
| Molecular Formula: | C15H15NO2S |
|---|---|
| Molecular Weight: | 273.35 g/mol |
Patent EP0966962 and Patent US2002043207.
Modafinil (INN,[6] USAN, BAN, JAN) is a wakefulness-promoting agent (or eugeroic) used for treatment of disorders such as narcolepsy, shift work sleep disorder, and excessive daytime sleepiness associated with obstructive sleep apnea.[7] It has also seen widespread off-label use as a purported cognition-enhancing agent. In English-speaking countries it is sold under the brand names Alertec, Modavigil, and Provigil. In the United States modafinil is classified as a schedule IV controlled substance and restricted in availability and usage, due to concerns about possible addiction potential. In most other countries it is a prescription drug but not otherwise legally restricted.
Although the mechanism of action of modafinil was initially unknown, it now appears that the drug acts as a selective, relatively weak, atypical dopamine reuptake inhibitor. However, it appears that other additional mechanisms may also be at play.

History
Modafinil was originally developed in France by neurophysiologist and emeritus experimental medicine professor Michel Jouvet and Lafon Laboratories. Modafinil originated with the late 1970s invention of a series of benzhydryl sulfinyl compounds, including adrafinil, which was first offered as an experimental treatment for narcolepsy in France in 1986. Modafinil is the primary metabolite of adrafinil, lacking the polar -OH group on its terminal amide,[77] and has similar activity to the parent drug but is much more widely used. It has been prescribed in France since 1994 under the name Modiodal, and in the US since 1998 as Provigil.
In 1998, modafinil was approved by the U.S. Food and Drug Administration[78] for the treatment of narcolepsy and in 2003 for shift work sleep disorder and obstructive sleep apnea/hypopnea[79] even though caffeine and amphetamine were shown to be more wakefulness promoting on the Stanford Sleepiness Test Score than modafinil.[80]
It was approved for use in the UK in December 2002. Modafinil is marketed in the US by Cephalon Inc., who originally leased the rights from Lafon, but eventually purchased the company in 2001.
Cephalon began to market the R-enantiomer armodafinil of modafinil in the U.S. in 2007. After protracted patent litigation and negotiations (see below), generic versions of modafinil became available in the U.S. in 2012.
That’s how it went…

2-benzhydryl-sulfanylacetamide.
Diphenylbromomethane (4,95g = 0.02 moles) and thiourea (1,52g=0.02moles) were refluxed in 20mls water for 30mins. As the synth from Rh’s says, a clear solution must have been formed in 5 mins, but in the end we still had a lot of oil at the bottom (the reasion to blame was old, semidecomposed diphenylbromomethane – when we opened the can, it emitted HBr). We were too lazy to separate the oil , so 2.5g (0.04moles) KOH in 15mls water was added straight and the reflux continued for 30 more mins. A disgusting stench filled the lab.
Thus obtained solution of potassium salt of diphenylmercaptane was cooled to 50-60 C and 1.9g (0.02moles) of chloroacetamide was added thereto. The mixtr was left to its own devices for 2hours – the precipitated oil crystallized. The xtals were filtered, washed thrice w/water, thrice w/ether (removing all benzhydrol). After drying there was obtained 1.9g (37%) of finely divided crystals with mp of 111 C.
With fresh diphenylbromomethane this will give not less than 80% – otherwise I’ll bee a reddish (this is an idiom which I am again unable to translate![]() ).
).
Modafinil
Into the solution of 3.6g benzhydrylsulfanylacetamide (0.014moles) in 15mls of GAA there was added 3mls (~0.03moles) 30% hydrogene peroxide. The mixture was left at RT (15 Ñ in our case, better not to heat above) for 20 hrs. Then into the solution there was added 30mls aqua, scratching the walls with a glass rod. After 1 hr the precipitate was filtered, washed w/water twice, then w/ether and dried. Yield – 2,3g (61%), mp – 158-159 C. After some time the mother liquor yielded some more product but we were too lazy to work it up.
PATENT
Patent US2002183552
This is a part of the experimental section:
Preparation of isothiouronium Salt (IV).
Diphenylmethanol (130 g, 0.7 mole) and thiourea (65 g, 0.85 mole) are added in 0.5 L reactor charging with water (325 ml). The mixture is heated to 95°C. (an emulsion is obtained) and 48% HBr (260 gr. 3.22 mole, 4.6 equivalents) is then added gradually during 0.5 hour. The mixture is heated under reflux {106-107°C) for 0.5 hour and cooled to 80-85°C. At this temperature, the mixture is seeded with several crystals of the product and the mixture is stirred at that temperature for 0.5 hour and then cooled to 25°C. The colorless crystals are collected by filtration, washed with water (200 ml) yielding about 240 gr. of wet crude isothiouronium salt.
Preparation of diphenylmethylthioacetamide.
A 2 L reactor was charged with diphenylmethylisothiouronium bromide crude wet obtained (240 gr.) and water {700 mL) under nitrogen. The suspension was heated to 60°C and 46% aqueous NaOH solution (98 ml, 1.68 mole, 2.4 eq.) was added. The reaction mixture was heated to 85°C and stirred until all the solid was dissolved. Then, it was cooled to 60°C and chloroacetamide (80 g, O.84 mole, 1.2 eq.) was added in five portions hour at 60-70°C during one hour. The suspension is stirred at 70°C for 4-5 hours. The mixture was filtered while warm and the cake was washed with hot water (250 ml). Diphenylmethylthioacetamide crude wet is obtained [220 gr., HPLC assay: 78%, HPLC purity: 95%, yield: 95% (from diphenylmethanol.)]
20 gr. of the product was recrystallized twice from ethyl acetate, dried in vacuo to give 15 gr. of pure title compound.
Preparation of Modafinil.
A 1.0 L reactor was charged with diphenylmethylthioacetamide crude wet (220 gr.) obtained above and glacial acetic acid (610 mL). The mixture was heated to 40°C and stirred until full dissolution is achieved. 5.8% H2O2 solution (500g, 1.2 eq) was added dropwise during 0.5 hours at 40-45°C. The reaction mixture was stirred at 40-45°C for 4 hours. Then sodium metabisulfite (18.3g) in 610 mL water was added in order to quench the unreacted H2O2 and the suspension was stirred for 0.5 hours. Then the reaction mixture was cooled to 15°C and filtered. The cake was washed with water (610 mL) and dried on air to obtain crude wet Modafinil (205 g). Reslurry in refluxing ethyl acetate, followed by recrystallization from methanol:water (4:1) solution afforded pure Modafinil [125 g, HPLC assay: 99.9%, HPLC purity: 99.9%, yield: 67% (from diphenylmethanol)].![]()
CLIP
Anti-Narcoleptic Agent Modafinil and Its Sulfone: A Novel
Facile Synthesis and Potential Anti-Epileptic Activity
Nithiananda Chatterjie, James P. Stables, Hsin Wang, and George J. Alexander
Neurochemical Research, Vol. 29, No. 8, August 2004 (© 2004), pp. 1481–1486

Abstract:
We report a facile procedure to synthesize racemic modafinil (diphenylmethylsulfinylacetamide), which is now being used in pharmacotherapy, and its achiral oxidized derivative (diphenylmethylsulfonyl acetamide). Modafinil is of interest more than for its potential anti-narcoleptic activity. It has also been reported to have neuroprotective properties and may potentially be effective in the enhancement of vigilance and cognitive performance. Finally, it may also protect from subclinical seizures that have been implicated as causative factors in autistic spectrum disorders and other neurodegenerative conditions. This agent can now be synthesized simply and in larger amounts than previously, making it more readily available for testing in various research modalities. The described procedure also lends itself to production of several other amides of potential interest. We are currently in the process of synthesizing and testing several new derivatives in this series. The anticonvulsant properties of modafinil and its sulfone derivative have not previously been extensively described in the literature. It may be of interest to note that the oxidized derivative of modafinil is also nontoxic and almost as effective as an anticonvulsant as the parent.
Experimental
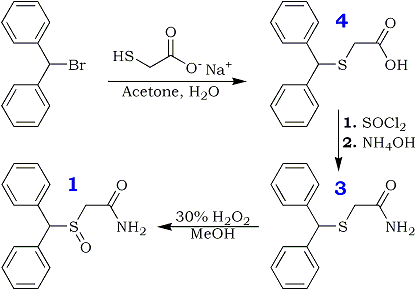
Diphenylmethylthioacetic Acid (3)
Benzhydryl bromide (14.78 gm, 0.059 mole) was dissolved in 75 ml of acetone in a 250-ml round-bottomed flask. To this solution was added dropwise sodium mercaptoacetate (6.59 g, 0.058 mole) in about 60 ml of H2O; the mixture was stirred under N2 for 2 h at room temperature and was thereafter warmed at about 60–70°C for 1 h. The reaction mixture was evaporated to dryness and taken up in CH2Cl2 and saturated aqueous NaHCO3. The organic extract was rejected, and the aqueous phase was treated with acid to pH 2 and chilled. Suction filtration gave the 6.9 g of the acid (3, 46%), mp 125°C. Rf 0.2. Recrystallization from MeOH/H2O gave mp 126–128°C.
Diphenylmethylthioacetamide (4)
Diphenylmethylthioacetic acid (19.5 g, 0.076 mole)
in 114 ml of dry benzene was taken in a 250-ml roundbottomed
flask attached to a reflux condenser, under N2 gas. To this was added thionyl chloride (19.5 ml, 0.097 mole) with a dropping funnel. The mixture was stirred at room temperature with a magnetic stirrer and refluxed for 1 h. Thereafter, the mixture was evaporated under low pressure to give a yellow oil that was taken up in about 100 ml of CH2Cl2 and filtered to yield a clear orange solution. This was chilled in ice water and added slowly to an ice-cold solution of concentrated NH4OH in H2O (40:40 ml). The ensuing mixture was stirred for 1 h and shaken well in a separatory funnel. The organic layer was dried (Na2SO4) and evaporated to dryness to give 14.39 g (54%) of the amide (4), mp 108–109°C (lit2 110°C). Rf 0.8. Recrystallization from CH3OH/H2O gave mp 109–110°C.
Diphenylmethylsulfinylacetamide (modafinil, 1)
Diphenylthioacetamide (3.46 g, 0.013 mole) was taken in glacial acetic acid (14 ml) with stirring; to this was added 1.34 ml of 30% H2O2 with chilling in ice water. The mixture was left in the refrigerator for 4 h and thereafter worked up by treating it with 70 ml of ice-cold water. The precipitated material was filtered under suction and washed with ice-cold water to give 1.5 g of white crystals (43%), mp 159–160°C. Rf 0.6. Recrystallization from hot MeOH gave mp 161–162°C
Diphenylmethylsufonylacetamide (2)
Diphenylmethylthioacetamide (2.5 g, 0.009 mole) (reg. No. 118779-53-6) was dissolved in about 12 ml of glacial acetic acid and 3 ml of 30% H2O2 and set aside overnight (16 h or more). The next day, the mixture was diluted with 100 ml of H2O and set aside to cool in the refrigerator. Upon filtration and drying, 2.1 g (80%) of 2 was obtained as a white powder. Rf 0.89. The melting point of sample after recrystallization from absolute EtOH was 195–197°C.
One aspect of our preparation of modafinil needs further mention. When diphenylmethylthioacetamide (4) is being oxidized by H2O2, care must be taken to keep the reaction mixture cool, and workup should be done in a timely manner. Allowing the reaction to go to 24 h or longer at room temperature results in the formation of the sulfone (2). The paper by Mu et al. (3) does not discuss this possibility. In our hands, the procedure stated therein led to the higher melting sulfone and not the modafinil. Our NMR data for the newly prepared modafinil preparation are in consonance with the data of the patented commercial product. It should be noted that the methylene protons in modafinil are geminally
coupled and appear as a pair of doublets. This is due to the fact that the adjacent sulfoxide moiety is chiral, and therefore the methylene protons adjacent to it wind up being diastereotopic with different chemical shifts and coupling. In the sulfone 2, the methylene protons appear as a singlet due to the fact that the adjacent sulfone moiety is achiral, thus making the two protons equivalent. Modafinil 1 is, however, an equal mixture of enantiomers, as in the reported patent and publication (2,3).
RESULTS
The chemical pathway leading to modafinil may be
represented in Scheme 1.

see pdf for further information and references,
CLIP
Synthesis and determination of the absolute configuration of the enantiomers of modafinil
Thomas Prisinzanoa, John Podobinskia, Kevin Tidgewella, Min Luoa and Dale Swensonb
Tetrahedron: Asymmetry 15(6), 1053-1058 (2004) (../rhodium/pdf /modafinil.enantiomers.pdf)
DOI:10.1016/j.tetasy.2004.01.039
a Division of Medicinal & Natural Products Chemistry, College of Pharmacy, The University of Iowa, Iowa City, Iowa 52242-1112, USA
b Department of Chemistry, The University of Iowa, Iowa City, Iowa 52242, USA
Abstract
The asymmetric synthesis of both enantiomers of modafinil, a unique CNS stimulant with a reduced abuse liability, is described. This approach effectively prepares modafinil on a multigram scale in several steps from benzhydrol. The described synthetic route has also been used to produce the more water soluble analogue, adrafinil. X-ray crystallographic analysis on (-)-(diphenylmethanesulfinyl)acetic acid has determined the absolute configuration to be R.
Graphical Abstract

Stereochemistry Abstract

(S)-(+)-(Diphenylmethanesulfinyl)acetic acid
C15H14O3S
[alpha]D22 + 40.2 (c=1.11, MeOH)
Source of chirality: resolution via diastereomeric salt formation with (R)-(+)-alpha-methylbenzylamine
Absolute configuration: S CLIP
Narcolepsy is a debilitating neurological disorder which is characterized by chronic sleepiness and is marked to be disorganization of sleep and wake patterns. Every six out of ten thousand people in Western Europe and North America are affected by this disorder. Modafinil (Provigil®) is approved by the Food and Drug Administration for the treatment of narcolepsy. It is commonly used in opposition to Ritalin®, however Ritalin® has an associated dependency issue. Modafinil, a central nervous system stimulant, has reported to have little abuse potential. Modafinil has the ability to act like wake-promoting sympathomimetic agents which includes amphetamine. At relevant pharmacological concentrations modafinil lacks binding ability to receptors for sleep/wake regulation, which includes the ones used for norepinephrine and serotonin. The precise mechanism of action of modafinil is unknown and is presently being researched. Modafinil contains a chiral sulfoxide moiety but is prescribed as a racemate. In collaboration with faculty from the Psychology department at Western Michigan University we were to synthesize modafinil for behavioral studies with animals. Therefore a large scale of pure modafinil was synthesized.
The tetrahedral sulfur atom acts as a chiral center (being surrounded by two dissimilar carbon atoms, an oxygen atom and an electron lone pair (Figure 1). Unlike most analogous trisubstituted amines that undergo umbrella-like inversion at the nitrogen atom, sulfoxides are configurationally stable.
The initial target of this synthesis was to prepare the 2-(diphenylmethylthio)acetamide (1) (Scheme I). The reaction of benzyhydral chloride and thiourea are reacted with potassium iodide, water, heat, 30% sodium hydroxide, 2-chloroacetamide and triethylamine. The procedure required the 2-(diphenylmethylthio) acetamide (1) to be recrystallized to remove any impurities with methanol:water solution 60:40 . After recrystallization (Figure 2) the ¹H NMR spectrum of the synthesized 2-(diphenylmethylthio)acetamide (1) provides evidence that the recrystallization did not purify the compound. In addition recrystallization significantly reduced the percent yield from 78.3-79.2% to 56%. If the compound were pure it would only show peaks at the following locations (ppm): 3.05 (s, 2H), 5.18 (s, 1H), 6.53 (s, 1H), 7.21-7.44(m, 10H).
To produce pure 2-(diphenylmethylthio)acetamide (1) elimination of the recrystallization step and 2-(diphenylmethylthio)acetamide (1) was then purified via column chromatography using dichloromethane:ether 80:20 as an eluent with the stationary phase (silica gel). After testing several of the fractions from the column using thin layer chromatography the fractions where able to be identified that contained 2- (diphenylmethylthio)acetamide (1). Once 2-(diphenylmethylthio)acetamide (1) was isolated it was oxidized with peracetic acid. The oxidation process was extended to three hours due to lack of desired product (±) modafinil (Figure 1).
With the procedure we used and modified through experimentation a new procedure was developed that increased the percent yield from 56% to 78.3-79.2%. We encountered a few problems that lead to the removal of the recrystallization step and the use of column chromatography was performed to purify 2-(diphenylmethylthio)acetamide (1) . Over- oxidation could have occurred but would have showed up at 3.7-3.8 (ppm), this did not occur in our experiment. The peak at 1.5 (ppm) is a water peak that was not fully removed during the rotovep procedure. After a precise and confident procedure was perfected then we were able to upscale the reaction and sythesize12gs of pure (±) modafinil.
FROM EROWID………
Benzhydrylsulphinylacetamide (Modafinil)2
Benzhydrylthioacetyl chloride
19.5g (0.076 mol) of benzhydrylthioacetic acid in 114 ml of benzene are placed in a three-necked flask provided with a condenser and a dropping funnel. The mixture is heated and 19 ml of thionyl chloride are added drop by drop. Once the addition is complete, the reflux is continued for about 1 hour, cooling and filtering are carried out and the benzene and the excess thionyl chloride and then evaporated. In this way, a clear orange oil is obtained.
Benzhydrylthioacetamide
35 ml of ammonia in 40 ml of water are introduced into a three-necked flask provided with a condenser and a dropping funnel and the benzhydrylthioacetyl chloride dissolved in about 100 ml of methylene chloride is added drop by drop. Once the addition is complete, the organic phase is washed with a dilute solution of soda and dried over Na2SO4, the solvent is evaporated and the residue is taken up in diisopropyl ether; in this way, the benzhydrylthioacetamide is crystallized. 16.8 g of product (yield 86%) are obtained. M.p. 110°C.
Modafinil (CRL 40,476)
14.39 g (0.056 mol) of benzhydrylthioacetamide are placed in a balloon flask and 60 ml of acetic acid and 5.6 ml of H2O2 (about 110 volumes, 33%) are added. The mixture is left in contact for one night at 40°C. and about 200 ml of water are then added; the CRL 40476 crystallizes. By recrystallization from methanol, 11.2 g of benzhydrylsulphinylacetamide are obtained. Yield: 73%. M.p. 164-66°C.
Novel Synthesis of Modafinil and its sulfone analog3
Our interest in synthesis of modified neuroactive compounds has led us to consider Modafinil (1), a stimulant and anti-narcoleptic agent that is finding increasing use in a number of neurological areas. The compound was originally prepared by a rather tedious route described in a procedure patented by L. Lafon2. More recently, its preparation has been reported by Mu et al.4 We believe that this compound has many interesting properties and possible alternative uses in addition to its recognized anti-narcoleptic actions.
Fig 1.
The chemical pathway leading to modafinil
Not having been able to obtain it from the patent holder, we proceeded to explore alternate synthetic pathways and settled on a convenient synthesis, which permitted us to produce this compound along with a primary derivative, the sulfone (2) in sufficient quantities for whole-animal studies. The current, more facile method starts with benzhydryl bromide and sodium thiolacetate in aqueous acetone, which reacts directly to form diphenylmethylthioacetic acid (3), possibly by an ionic mechanism. This resultant compound can be converted to its acid chloride that, in turn, may be used to acylate ammonia. The ensuing primary amide (4) may be gently oxidized by H2O2 to form the corresponding sulfoxide (Modafinil, 1) and, under more vigorous conditions, the modafinil sulfone (2), whose anticonvulsant and biological properties have not been described extensively in the literature. Additionally, this procedure is also uniquely suitable for large-scale preparation of Modafinil and its congeners.
One aspect of our preparation of modafinil needs further mention. When diphenylmethylthioacetamide (4) is being oxidized by H2O2, care must be taken to keep the reaction mixture cool, and workup should be done in a timely manner. Allowing the reaction to go to 24 h or longer at room temperature results in the formation of the sulfone (2). The paper by Mu et al.4 does not discuss this possibility. In our hands, the procedure stated therein led to the higher melting sulfone and not the modafinil. Our NMR data for the newly prepared modafinil preparation are in consonance with the data of the patented commercial product. It should be noted that the methylene protons in modafinil are geminally coupled and appear as a pair of doublets. This is due to the fact that the adjacent sulfoxide moiety is chiral, and therefore the methylene protons adjacent to it wind up being diastereotopic with different chemical shifts and coupling. In the sulfone 2, the methylene protons appear as a singlet due to the fact that the adjacent sulfone moiety is achiral, thus making the two protons equivalent. Modafinil 1 is, however, an equal mixture of enantiomers, as in the reported patent and publication2,4.
Experimental
The new compounds were prepared according to modified procedures published in the patent literature. Starting materials and solvents were obtained commercially from Fluka and/or Aldrich Chemical Corp. Thin layer chromatography (TLC) was performed on silica gel plates. Solvent system was EtOAc:MeOH:NH4OH, 100:10:3 by volume. Melting points are uncorrected.
Diphenylmethylthioacetic Acid (3)
Benzhydryl bromide (14.78 gm, 0.059 mole) was dissolved in 75 ml of acetone in a 250-ml round-bottomed flask. To this solution was added dropwise sodium mercaptoacetate (6.59 g, 0.058 mole) in about 60 ml of H2O; the mixture was stirred under N2 for 2 h at room temperature and was thereafter warmed at about 60–70°C for 1 h. The reaction mixture was evaporated to dryness and taken up in CH2Cl2 and saturated aqueous NaHCO3. The organic extract was rejected, and the aqueous phase was treated with acid to pH 2 and chilled. Suction filtration gave the 6.9 g of the acid (3, 46%), mp 125°C. Rf 0.2. Recrystallization from MeOH/H2O gave mp 126–128°C.
Diphenylmethylthioacetamide (4)
Diphenylmethylthioacetic acid 3 (19.5 g, 0.076 mole) in 114 ml of dry benzene was taken in a 250-ml roundbottomed flask attached to a reflux condenser, under N2 gas. To this was added thionyl chloride (19.5 ml, 0.097 mole) with a dropping funnel. The mixture was stirred at room temperature with a magnetic stirrer and refluxed for 1 h. Thereafter, the mixture was evaporated under low pressure to give a yellow oil that was taken up in about 100 ml of CH2Cl2 and filtered to yield a clear orange solution. This was chilled in ice water and added slowly to an ice-cold solution of concentrated NH4OH in H2O (40:40 ml). The ensuing mixture was stirred for 1 h and shaken well in a separatory funnel. The organic layer was dried (Na2SO4) and evaporated to dryness to give 14.39 g (54%) of the amide (4), mp 108–109°C (lit4 110°C). Rf 0.8. Recrystallization from CH3OH/H2O gave mp 109–110°C.
Diphenylmethylsulfinylacetamide (Modafinil, 1)
Diphenylmethylthioacetamide 4 (3.46 g, 0.013 mole) was taken in glacial acetic acid (14 ml) with stirring; to this was added 1.34 ml of 30% H2O2 with chilling in ice water. The mixture was left in the refrigerator for 4 h and thereafter worked up by treating it with 70 ml of ice-cold water. The precipitated material was filtered under suction and washed with ice-cold water to give 1.5 g of white crystals (43%), mp 159–160°C. Rf 0.6. Recrystallization from hot MeOH gave mp 161–162°C
Diphenylmethylsulfonylacetamide (2)
Diphenylmethylthioacetamide (2.5 g, 0.009 mole) (CAS No. 118779-53-6) was dissolved in about 12 ml of glacial acetic acid and 3 ml of 30% H2O2 and set aside overnight (16 h or more). The next day, the mixture was diluted with 100 ml of H2O and set aside to cool in the refrigerator. Upon filtration and drying, 2.1 g (80%) of 2 was obtained as a white powder. Rf 0.89. The melting point of sample after recrystallization from absolute EtOH was 195–197°C.
High-yield Synthesis of Modafinil from Benzhydrol5
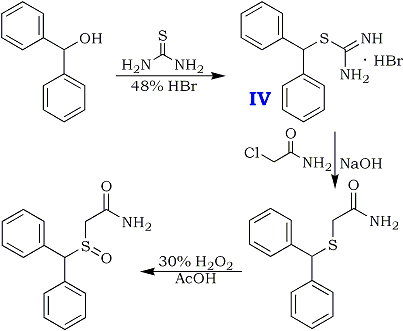
A recent patent5 describes a very easy two-step route to the Modafinil precursor diphenylmethanethioacetamide from benzhydrol (diphenylmethanol) in 90% yield and with 95% purity. A 200g batch is made in a 2000 mL vessel using water as reaction medium and ethyl acetate for recrystallization of the product.
Diphenylmethylbromide is prepared in situ from benzhydrol and react it with thiourea in a one-pot reaction to form the corresponding isothiouronium salt. The crude salt is then reacted with chloroacetamide (by generating the thiolate cation in situ), and after filtration and washing, diphenylmethylthioacetamide is isolated in excellent yield and good purity. After oxidation of the thioacetamide with hydrogen peroxide, followed by recrystallization, the overall yield of Modafinil is 67% from the benzhydrol.
(Chimimanie’s Voice:) The synthesis works just as great without the nitrogen inert atmosphere (most patents do not use it at all), step two is only a hydrolysis of the thiouronium salt to the thiolate. You just have to put the salt, NaOH and heat till you got a homogenous solution, with no more solid material floating around. The following chloroacetamide SN2 reaction is a breeze too. Sometime a blue solution can bee obtained, it is nothing to worry about. In the final step, you just have to filter off the solid which did not dissolve when the crude thioacetamide is put in the GAA/H2O2, bee4 crashing the soluble one with water.
Do not forget to slurry the modafinil in EtOAc and then recrystallize it from aqueous MeOH, as the crystalline shape of modafinil is important for the kinetic and quality of effects, at least according to the patents EP0966962 and US2002043207.
Experimental
Preparation of isothiouronium Salt (IV)
Diphenylmethanol (130 g, 0.7 mole) and thiourea (65 g, 0.85 mole) are added in 0.5 L reactor charging with water (325 ml). The mixture is heated to 95°C. (an emulsion is obtained) and 48% HBr (260 gr. 3.22 mole, 4.6 equivalents) is then added gradually during 0.5 hour. The mixture is heated under reflux (106-107°C) for 0.5 hour and cooled to 80-85°C. At this temperature, the mixture is seeded with several crystals of the product and the mixture is stirred at that temperature for 0.5 hour and then cooled to 25°C. The colorless crystals are collected by filtration, washed with water (200 ml) yielding about 240 gr. of wet crude isothiouronium salt.
(Antoncho’s Voice:) Assholium successfully made Modafinil by this method, but there turned out to be a mistake in the original patent text – In the preparation of IV, the quantity of HBr stated here is excessive and leads to complete hydrolysis of the initially formed isothiouronium salt. The acid should bee added until the reaction mixture turns completely clear (about half as much as the patent says) – a sort of titration. Further addition will result in precipitation of heavy stinky oil, benzhydrylmethanethiol.
Preparation of diphenylmethylthioacetamide
A 2 L reactor was charged with diphenylmethylisothiouronium bromide crude wet obtained (240 gr.) and water (700 mL) under nitrogen. The suspension was heated to 60°C and 46% aqueous NaOH solution (98 ml, 1.68 mole, 2.4 eq.) was added. The reaction mixture was heated to 85°C and stirred until all the solid was dissolved. Then, it was cooled to 60°C and chloroacetamide (80 g, 0.84 mole, 1.2 eq.) was added in five portions hour at 60-70°C during one hour. The suspension is stirred at 70°C for 4-5 hours. The mixture was filtered while warm and the cake was washed with hot water (250 ml). Diphenylmethylthioacetamide crude wet is obtained [220 gr., HPLC assay: 78%, HPLC purity: 95%, yield: 95% from diphenylmethanol]. 20g of the product was recrystallized twice from ethyl acetate, dried in vacuo to give 15g of pure title compound.
Preparation of Modafinil
A 1.0 L reactor was charged with diphenylmethylthioacetamide crude wet (220 gr.) obtained above and glacial acetic acid (610 mL). The mixture was heated to 40°C and stirred until full dissolution is achieved. 5.8% H2O2 solution (500g, 1.2 eq) was added dropwise during 0.5 hours at 40-45°C. The reaction mixture was stirred at 40-45°C for 4 hours. Then sodium metabisulfite (18.3g) in 610 mL water was added in order to quench the unreacted H2O2 and the suspension was stirred for 0.5 hours. Then the reaction mixture was cooled to 15°C and filtered. The cake was washed with water (610 mL) and dried on air to obtain crude wet Modafinil (205 g). Reslurry in refluxing ethyl acetate, followed by recrystallization from methanol:water (4:1) solution afforded pure Modafinil [125 g, HPLC assay: 99.9%, HPLC purity: 99.9%, yield: 67% (from diphenylmethanol)].
References
- US Pat 4,066,686
- L. Lafon, US Pat 4,177,290 (1979); L. Lafon, Eur. Pat. 283,362 (1988)
- Nithiananda Chatterjie, James P. Stables, Hsin Wang, and George J. Alexander, Anti-Narcoleptic Agent Modafinil and Its Sulfone: A Novel Facile Synthesis and Potential Anti-Epileptic Activity, Neurochemical Research, 29(8), 1481–1486 (2004)
- Mu, B., Lei, G., He, X., and Du, X., Synthesis of central stimulant modafinil. Zhongguo Yaowu Huaxue Zazhi, 9(2), 132–134 (1999)
- US Pat. 6,649,796 (2002)

(R)-(−)-modafinil (armodafinil; top)
(S)-(+)-modafinil (bottom) |
|
| Clinical data | |
|---|---|
| Trade names | Provigil, others (see below) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a602016 |
| License data | |
| Pregnancy category |
|
| Dependence liability |
Psychological: Very low[1] Physical: Negligible[1] |
| Addiction liability |
Very low to low[2] |
| Routes of administration |
Oral (tablets) |
| ATC code | N06BA07 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Not determined due to the aqueous insolubility |
| Protein binding | 62% |
| Metabolism | Hepatic (primarily via amide hydrolysis;[3] CYP1A2, CYP2B6, CYP2C9, CYP2C19, CYP3A4, CYP3A5 involved [4] |
| Biological half-life | 15 hours (R-enantiomer), 4 hours (S-enantiomer)[5] |
| Excretion | Urine (80%) |
| Identifiers | |
| Synonyms | CRL-40476; Diphenylmethylsulfinylacetamide |
| CAS Number | 68693-11-8 |
| PubChem (CID) | 4236 |
| IUPHAR/BPS | 7555 |
| DrugBank | DB00745 |
| ChemSpider | 4088 |
| UNII | R3UK8X3U3D |
| KEGG | D01832 |
| ChEBI | CHEBI:31859 |
| ChEMBL | CHEMBL1373 |
| ECHA InfoCard | 100.168.719 |
| Chemical and physical data | |
| Formula | C15H15NO2S |
| Molar mass | 273.35 g/mol |
| 3D model (Jmol) | Interactive image |
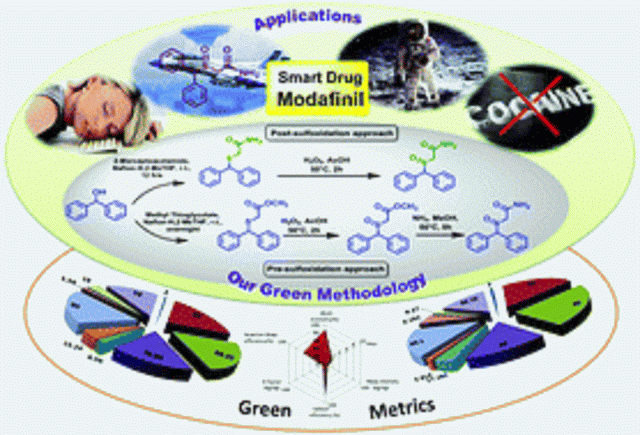
DOI: 10.1039/C6GC02623K, Communication
We developed a post-sulfoxidation protocol for the synthesis of Modafinil that exhibits improved sustainability credentials, utilizing the recyclable heterogeneous catalyst Nafion-H.
Efficient atom and step economic (EASE) synthesis of the “smart drug” Modafinil
E-mail: dratulsax@gmail.com, atul_kumar@cdri.res.in
DOI: 10.1039/C6GC02623K


Atul Kumar


////////////
Efficient atom and step economic (EASE) synthesis of the “smart drug” Modafinil
Efficient atom and step economic (EASE) synthesis of the “smart drug” Modafinil
DOI: 10.1039/C6GC02623K, Communication
We developed a post-sulfoxidation protocol for the synthesis of Modafinil that exhibits improved sustainability credentials, utilizing the recyclable heterogeneous catalyst Nafion-H.
Efficient atom and step economic (EASE) synthesis of the “smart drug” Modafinil
E-mail: dratulsax@gmail.com, atul_kumar@cdri.res.in
DOI: 10.1039/C6GC02623K


Atul Kumar
Professor, Academy of Scientific and Innovative Research (AcSIR)/ Senior Principal Scientist at CSIR-CDRI
View original post 206 more words
Selection and justification of starting materials: new Questions and Answers to ICH Q11 published
DRUG REGULATORY AFFAIRS INTERNATIONAL

The ICH Q11 Guideline describing approaches to developing and understanding the manufacturing process of drug substances was finalised in May 2012. Since then the pharmaceutical industry and the drug substance manufacturers had time to get familiar with the principles outlined in this guideline. However, experience has shown that there is some need for clarification. Thus the Q11 Implementation Working Group recently issued a Questions and Answers Document.
The ICH Q11 Guideline describes approaches to developing and understanding the manufacturing process of drug substances. It was finalised in May 2012 and since then the pharmaceutical industry and the drug substance manufacturers had time to get familiar with the principles outlined in this guideline. However, experiences during implementation of these principles within this 4 years period have shown that there is need for clarification in particular with regard to the selection and justification of starting materials.
On 30 November 2016 the ICH published a Questions and Answers…
View original post 2,105 more words
Lumefantrine
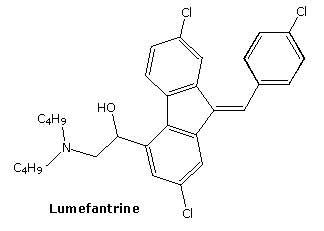
lumefantrine
- Molecular FormulaC30H32Cl3NO
- Average mass528.940 Da
- Benflumetol
- dl-Benflumelol
UNIIF38R0JR742
CAS number82186-77-4
2-(dibutylamino)-1-[(9Z)-2,7-dichloro-9-[(4-chlorophenyl)methylidene]-9H-fluoren-4-yl]ethan-1-ol
| (±)-2,7-Dichloro-9-((Z)-p-chlorobenzylidene)-α-((dibutylamino)methyl)fluorene-4-methanol |
| 2-Dibutylamino-1-[2,7-dichloro-9-(4-chloro-benzylidene)-9H-fluoren-4-yl]-ethanol |
| 2-Dibutylamino-1-{2,7-dichloro-9-[1-(4-chloro-phenyl)-meth-(Z)-ylidene]-9H-fluoren-4-yl}-ethanol |
| Benflumetol |
| dl-Benflumelol |
UNII F38R0JR742
CAS number 82186-77-4
Weight Average: 528.94
Monoisotopic: 527.154947772
Chemical Formula C30H32Cl3NO
Lumefantrine (or benflumetol) is an antimalarial drug. It is only used in combination with artemether. The term “co-artemether” is sometimes used to describe this combination.[1] Lumefantrine has a much longer half-life compared to artemether and so is therefore thought to clear any residual parasites that remain after combination treatment.[2]
Lumefantrine, along with pyronaridine and naphtoquine, were synthesized in course of the Project 523 antimalaria drug research initiated in 1967; these compounds are all used in combination antimalaria therapies.[3][4][5]

Lumefantrine is an antimalarial drug chemically known as 2-(dibutylamino)-1-[(9Z)-2, 7-dichloro-9-(4- chlorobenzylidene)-9H-floren-4-yl] ethanol, which is used in the prevention and treatment of Malaria in worm blooded animals. Lumefantrine is using the combination of β-Artemether in the treatment of Malaria
SYN
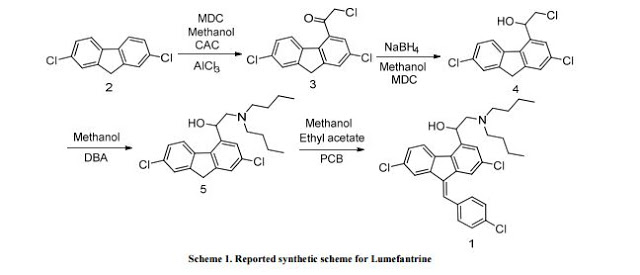
Synthetic Reference
Beutler, Ulrich; Fuenfschilling, Peter C.; Steinkemper, Andreas. An Improved Manufacturing Process for the Antimalaria Drug Coartem. Part II. Organic Process Research & Development. Volume 11. Issue 3. Pages 341-345. 2007.

SYN 2
Synthetic Reference
Rao, Dharmaraj Ramachandra; Kankan, Rajendra Narayanrao; Phull, Manjinder Singh. Process for preparation of lumefantrine as antimalarial agent with improved method. Assignee Cipla Co., Ltd., India. CN 1865227. (2006).

SYN 3
Synthetic Reference
Sethi, Madhuresh Kumar; Gonuguntla, Anantavena Rani; Arikatla, Siva Lakshmi Devi; Mulukutla, Suryanarayana; Yerramalla, Rajakrishna; Bontalakoti, Jaganmohanarao; Vemula, Lakshminarayana; Thirunavukarasu, Jayaprakash. Synthesis and characterization of novel related substances of Lumefantrine, an anti-malarial drug. Pharma Chemica. Volume 8. Issue 3. Pages 91-100. 2016.

SYN4
Synthetic Reference
Mathur, Prafull; Mathur, Suvigya; Vishwanath, Kannan; Mishra, Anand Kumar. Preparation of lumefantrine. Assignee Aanjaneya Lifecare Limited, India. IN 2013MU00611. (2015).

SYN 5
Synthetic Reference
Wu, Guang-liang; Dai, Ying-jie; Kang, Cong-min; Zi, Yan. A new synthetic technology of anti-malarial drug lumefantrine. Zhongguo Xinyao Zazhi. Volume 21. Issue 24. Pages 2944-2947. 2012.

SYN 6
Synthetic Reference
Krishna, Bettadapura Gundappa; Verma, Sudhakar; Krishna, Sujatha; Naik, Gajanan; Arulmoli, Thangavel. A process for preparation of lumefantrine. Assignee SeQuent Scientific Limited, India. IN 2012CH00470. (2012).

SYN 7
Synthetic Reference
Bansi, Lal; Genbhau, Gund Vitthal; Prabhakar, Bapat Chintamani; Popat, Bochiya Pravin; Banshi, Punde Dnyanadeo; Venkata, Reddy Prabhakar Gorla. Improved one pot process for the synthesis of lumefantrine. Assignee Calyx Chemicals and Pharmaceuticals Ltd., India. IN 2009MU01437. (2010).

SYN 8
Synthetic Reference
Shailesh, Singh; Dhaval, Vashi; Vinod, Gaikwad; Sanjay, Chowkekar; Sanjay, Bute. Process for the preparation of lumefantrine. Assignee Ajanta Pharma Ltd., India. IN 2008MU01677. (2010).

SYN 9
Synthetic Reference
Rawalnath, Sakhardande Rajiv; Kanji, Khatri Navin; Nilkanth, Firake Pandharinath; Vasant, Panchal Rajesh; Nagesh, Babrekar Chandan; Madhukar, Mohite Dhanaji. Preparation of lumefantrine. Assignee Saxena, Alok, India. IN 2006MU00260. (2007).

///////////////////////////////////////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
PATENT
https://patents.google.com/patent/CN107501316A/en
(19) (PDF) Process for high purity lumefantrine. Available from: https://www.researchgate.net/publication/221933417_Process_for_high_purity_lumefantrine [accessed Feb 06 2022].
(19) (PDF) Process for high purity lumefantrine. Available from: https://www.researchgate.net/publication/221933417_Process_for_high_purity_lumefantrine [accessed Feb 06 2022].
(19) (PDF) Process for high purity lumefantrine. Available from: https://www.researchgate.net/publication/221933417_Process_for_high_purity_lumefantrine [accessed Feb 06 2022].
PAPER
Tropical Journal of Pharmaceutical Research October 2013; 12 (5): 791-798 ISSN: 1596-5996 (print); 1596-9827 (electronic) © Pharmacotherapy Group, Faculty of Pharmacy, University of Benin, Benin City, 300001 Nigeria. All rights reserved. Available online at http://www.tjpr.org
CLIP
NMR

1H NMR

COSY

NOESY

HSQC

DEPT

13C NMR

DSC

CLIP
CLIP
Click to access DPC-2016-8-3-91-100.pdf
REFERENCES
[1] Ulrich Beutler, C Peter.; Fuenfschilling.; and Andreas, Steinkemper.; Novartis Pharma AG; Chemical and Analytical Development: CH-4002 Basel, Switzerland, Organic Process Research & Development 2007, 11, 341- 345.
[2] Boehm, M. Fuenfschilling.; Krieger, P. C.; Kuesters, E. M.; Struber, F.; Org. Process Res. DeV. 2007, 11, 336- 340.
[3] (a) Rao, D. R.; Kankan, R. N.; Phull, M. S.; Patent Application CN 1009-3724 20060424, 2005. (b) Deng, R.; Zhong, J.; Zhao, D.; Wang, J.; Yaoxue, X. 2000, 35 (1), 22. (c) Allmendinger, Th.; Wernsdorfer, W. H. PCT WO 99/67197.
[4] Perrumattam, J.; Shao, Ch.; Confer, W. L. Synthesis 1994, 1181.
[5] Fuenfschilling, P. C.; Hoehn P.; Mutz J.-P. Organic Process Res. Dev. 2007, 11, 13.
[6] Di Nunno, L.; Scilimati, A. Tetrahedron 1988, 44, 3639.
[7] Pharmacopeial Forum, Vol. 36(2) [Mar.-Apr. 2010]
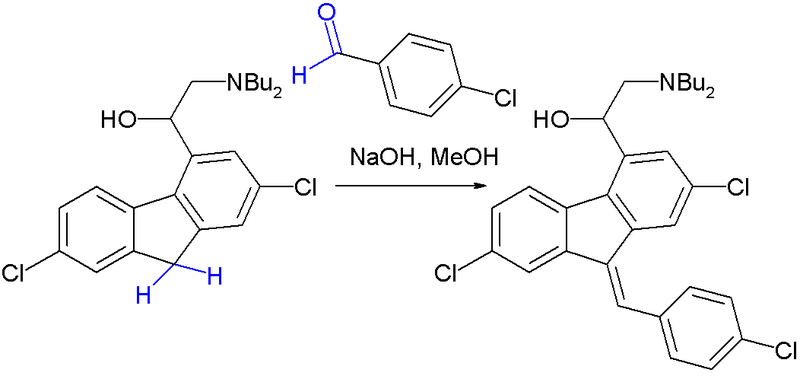
Preparation of 2-(dibutylamino)-1-[(9Z)-2, 7-dichloro-9-(4-chlorobenzylidene)-9H-floren-4-yl] ethanol (Lumefantrine) 1.
To a stirred solution of NaOH (1.97 g 0.0492 mol) in methanol (100 ml) there was added 1-(2, 7- dichloro-9 H-fluren-4-yl)-2-(dibutyl amino) ethanol (10 g, 0.0246 mol) and para chloro benzaldehyde (5.24 g 0.0372). The suspension obtained was stirred at reflux temperature till the absence of starting material by TLC. After confirming the product formation reaction mixture was cooled to room temperature and further stirred at same temperature for overnight. The precipitated solids were filtered and washed with methanol and dried under vacuum at 50°C to get desired compound. (Purity by HPLC: 99%).
IR (cm-1): 3408, 3092, 2953, 2928, 2870, 2840, 1634, 1589, 1487, 1465, 1443, 1400, 1365, 1308, 1268, 1241, 1207, 1173, 1156, 1085, 1071, 1014, 980, 933, 874, 839, 815, 806, 770;
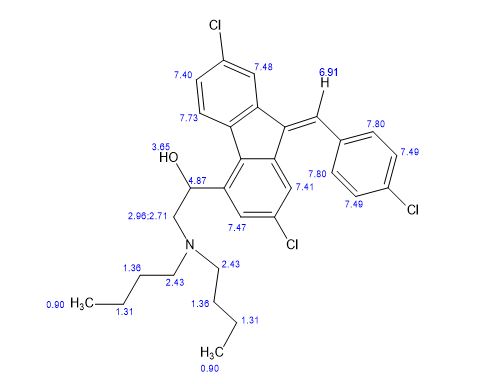
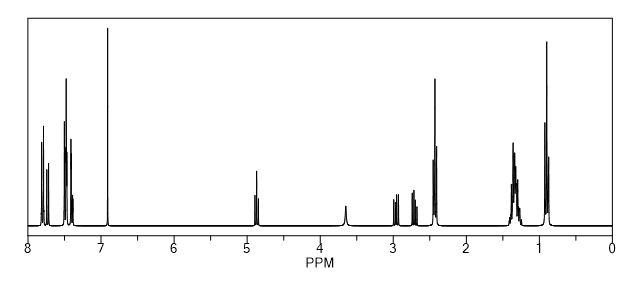
1H NMR (CDCl3, δ ppm): 7.75 (d, 1H, CH, J 1.5 Hz), 7.68 (d, 1H, CH, J 1.5 Hz), 7.60-7.63 (m, 1H, CH), 7.32-7.35 (dd, 1H, CH, J 1.7,8.3 Hz), 7.45-7.50 (m, 1H, CH), 5.35-5.39 (dd, 1H, CH, J 3.0,9.9 Hz), 2.41-2.74 (m, 1H, CH2Ha), 2.86-2.92 (m, 1H, CH2Hb), 2.41-2.74 (m, 4H, CH2), 1.25-1.56 (m, 8H, CH2), 0.97 (t, 1H, CH, J 7.2 Hz), 7.60-7.63 (m, 1H, CH), 7.45-7.50 (m, 4H, CH), 4.54 (broad, 1H, OH),
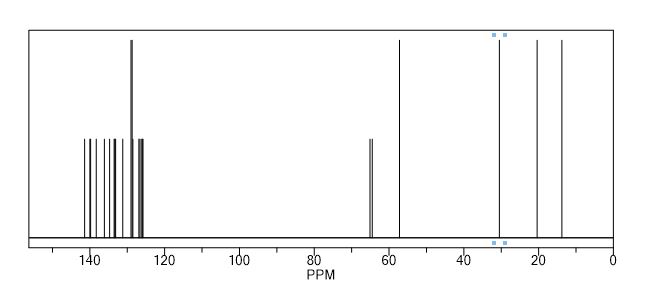
13C NMR (CDCl3, δ ppm): 138.2, 141.5, 120.6, 133.2, 126.3, 135.0, 135.0, 136.4, 123.9, 128.3, 132.8, 123.0, 139.8, 65.5, 60.0, 53.5, 29.1, 20.6, 14.0, 127.6, 134.7, 130.5, 129.1, 133.2;
MS: m/z: 528 [M+H]+ ; Analysis calcd. for C30H32Cl3NO: C, 68.12; H, 6.10; N, 2.65% Found: C, 68.38; H, 6.14; N, 2.63 %.
CLIP
One-dimensional 1H NMR spectrum of B) a lumefantrine standard,
A CLIP
http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0103-50532012000100010&lng=en&nrm=iso




CLIP
A simple and precise method for quantitative analysis of lumefantrine …
[2–4] Thus, today lumefantrine is a drug of choice in antimalarial treatment against P. …. The NMRspectra observed triplet at 0.943-0.989 (methyl protons of alkyl …



References
- Jump up^ Toovey S, Jamieson A, Nettleton G (2003). “Successful co-artemether (artemether-lumefantrine) clearance of falciparum malaria in a patient with severe cholera in Mozambique”. Travel medicine and infectious disease. 1 (3): 177–9. doi:10.1016/j.tmaid.2003.09.002. PMID 17291911.
- Jump up^ White, Nicholas J.; van Vugt, Michele; Ezzet, Farkad (1999). “Clinical Pharmacokinetics and Pharmacodynamics of Artemether-Lumefantrine”. Clinical Pharmacokinetics. 37 (2): 105–125. doi:10.2165/00003088-199937020-00002. ISSN 0312-5963.
- Jump up^ Cui, Liwang; Su, Xin-zhuan (2009). “Discovery, mechanisms of action and combination therapy of artemisinin”. Expert Review of Anti-infective Therapy. 7 (8): 999–1013. doi:10.1586/eri.09.68. PMC 2778258
 . PMID 19803708.
. PMID 19803708. - Jump up^ http://aac.asm.org/content/56/5/2465.full
- Jump up^ Laman, M; Moore, BR; Benjamin, JM; Yadi, G; Bona, C; Warrel, J; Kattenberg, JH; Koleala, T; Manning, L; Kasian, B; Robinson, LJ; Sambale, N; Lorry, L; Karl, S; Davis, WA; Rosanas-Urgell, A; Mueller, I; Siba, PM; Betuela, I; Davis, TM (2014). “Artemisinin-naphthoquine versus artemether-lumefantrine for uncomplicated malaria in Papua New Guinean children: an open-label randomized trial”. PLoS Med. 11: e1001773. doi:10.1371/journal.pmed.1001773. PMC 4280121
 . PMID 25549086.
. PMID 25549086.
 |
|
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| MedlinePlus | a609024 |
| Routes of administration |
Oral |
| ATC code | P01BF01 (WHO) (combination with artemether) |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | 82186-77-4 |
| PubChem (CID) | 6437380 |
| DrugBank | DB06708 |
| ChemSpider | 4941944 |
| UNII | F38R0JR742 |
| KEGG | D03821 |
| ChEBI | CHEBI:156095 |
| ChEMBL | CHEMBL38827 |
| Chemical and physical data | |
| Formula | C30H32Cl3NO |
| Molar mass | 528.939 g/mol |
| 3D model (Jmol) | Interactive image |
///////////lumefantrine, lumefantrene, Antimalarial, CPG-56695, CPG 56695,
CCCCN(CCCC)CC(O)C1=C2C(=CC(Cl)=C1)\C(=C/C1=CC=C(Cl)C=C1)C1=C2C=CC(Cl)=C1
Designing Polymers for Amorphous Solid Dispersions — AAPS Blog
By: Laura I. Mosquera-Giraldo and Lynne S. Taylor Imagine spending billions of dollars in the discovery of a new drug, and then realizing that it is impractical to administer it orally because it cannot reach the systemic circulation and achieve a therapeutic effect. This is the case for many emerging drugs that are insoluble in water, […]
via Designing Polymers for Amorphous Solid Dispersions — AAPS Blog
Ranolazine Intermediate, An Efficient Synthesis of 1-(2-Methoxyphenoxy)-2,3-epoxypropane: Key Intermediate of β-Adrenoblockers

http://pubs.acs.org/doi/suppl/10.1021/op300056k
Preparation of 1-(2-Methoxyphenoxy)-2,3-epoxypropane 4.
To a stirring solution of 2-methoxy phenol 2 (10 kg, 80.55 mol) and water (40 L) at about 30 °C was added sodium hydroxide (1.61 kg, 40.25 mol) and water (10 L). After stirring for 30−45 min, epichlorohydrin 3 (22.35 kg, 241.62 mol) was added and stirred for 10−12 h at 25−35 °C. Layers were separated, and water (40 L) was added to the organic layer (bottom layer) containing product. Sodium hydroxide solution (3.22 kg, 80.5 mol) and water (10 L) were added at 27 °C and stirred for 5−6 h at 27 °C.
The bottom product layer was separated and washed with sodium hydroxide solution (3.0 kg 75 mol) and water (30 L). Excess epichlorohydrin (3) was recovered by distillation of the product layer at below 90 °C under vacuum (650−700 mmHg) to give 13.65 kg (94%) of title compound with 98.3% purity by HPLC, 0.2% of 2- methoxy phenol 2, 0.1% of epichlorohydrin 3, 0.1% of chlorohydrin 11, 0.3% of dimer 12 and 0.3% of dihydroxy 13.
1 H NMR (400 MHz, CDCl3, δ) 6.8−7.0 (m, 4H), 4.3 (dd, J = 5.6 Hz, 5.4 Hz, 1H), 3.8 (dd, J = 5.6 Hz, 5.3 Hz, 1H), 3.7 (s, 3H), 3.2−3.4 (m, 1H), 2.8 (dd, J = 5.6 Hz, 5.4 Hz, 1H), 2.7 (dd, J = 5.6 Hz, 5.3 Hz, 1H);
IR (KBr, cm−1 ) 2935 (C−H, aliphatic), 1594 and 1509 (CC, aromatic), 1258 and 1231 (C−O−C, aralkyl ether), 1125 and 1025 (C−O−C, epoxide);
MS (m/z) 181 (M+ + H).
Compound Details
CAS 2210-74-4
| Glycidyl 2-methoxyphenyl ether | |
| Guaiacol glycidyl ether |
1H NMR PREDICT
13C NMR PREDICT
COSY PREDICT

CREDIT……….http://www.molbase.com/en/synthesis_2210-74-4-moldata-95563.html
DR REDDYS LABORATORIES
An Efficient Synthesis of 1-(2-Methoxyphenoxy)-2,3-epoxypropane: Key Intermediate of β-Adrenoblockers
† Innovation Plaza, IPD, R&D, Dr. Reddy’s Laboratories Ltd., Survey Nos. 42, 45,46, and 54, Bachupally, Qutubullapur – 500073, Andhra Pradesh, India
‡ Institute of Science and Technology, Center for Environmental Science, JNT University, Kukatpally, Hyderabad – 500 072, Andhra Pradesh, India
Org. Process Res. Dev., 2012, 16 (10), pp 1660–1664
DOI: 10.1021/op300056k
Publication Date (Web): September 14, 2012
Copyright © 2012 American Chemical Society
*Telephone: +91 4044346000. Fax: +91 4044346285. E-mail: rakeshwarb@drreddys.com.
ADRAFINIL
ADRAFINIL
2-((diphenylmethyl)sulfinyl)-acetohydroxamicaci;2-((diphenylmethyl)sulfinyl)-n-hydroxy-acetamid;2-((diphenylmethyl)sulfinyl)-n-hydroxyacetamide;2-(benzhydrylsulfinyl)acetohydroxamicacid;ADRAFINIL;2-[(DIPHENYLMETHYL)SULFINYL]ACETOHYDROXAMIC ACID;CRL 40028;OLMIFON
- CAS 63547-13-7
- MF:C15H15NO3S
- MW:289.35
- EINECS:264-303-1
WATCH THIS POST AS DETAILS LIKE SYNTHESIS ARE UPDATED………….
Adrafinil is touted mainly for its stimulant properties and ability to provide alertness and wakefulness.
- Stay up late/stay awake during normal sleeping hours: Adrafinil may be helpful for night workers who need a kick-start adapting their body’s natural circadian rhythm of wakefulness in the daytime and sleepiness in the evening to their job needs. This can also make it helpful for periodic late-night study sessions. Adrafinil is best taken in the afternoon or evening for nighttime wakefulness.
- Boost energy, alertness, and focus during the day time: Adrafinil can also be used as an energy-boost during waking hours.
- CONTACT SKYPE CATHERINESSPC WICKR
Adrafinil (INN) (brand name Olmifon)[2] is a discontinued wakefulness-promoting agent (or eugeroic) that was formerly used inFrance to promote vigilance (alertness), attention, wakefulness, mood, and other parameters, particularly in the elderly.[3][4] It was also used off-label by individuals who wished to avoid fatigue, such as night workers or others who needed to stay awake and alert for long periods of time. Additionally, “adrafinil is known to a larger nonscientific audience, where it is considered to be a nootropic agent.”[3] Adrafinil is a prodrug; it is primarily metabolized in vivo to modafinil, resulting in very similar pharmacological effects.[3] Unlike modafinil, however, it takes time for the metabolite to accumulate to active levels in the bloodstream. Effects usually are apparent within 45–60 minutes when taken orally on an empty stomach. Adrafinil was marketed in France under the trade name Olmifon[2] until September 2011 when it was voluntarily discontinued.[4]
Pharmacology
Pharmacodynamics
Because α1-adrenergic receptor antagonists were found to block effects of adrafinil and modafinil in animals, “most investigators assume[d] that adrafinil and modafinil both serve as α1-adrenergic receptor agonists.”[3] However, adrafinil and modafinil have not been found to bind to the α1-adrenergic receptor and they lack peripheral sympathomimetic side effects associated with activation of this receptor;[5] hence, the evidence in support of this hypothesis is weak, and other mechanisms are probable.[3] Modafinil was subsequently screened at a variety of targets in 2009 and was found to act as a weak, atypical blocker of the dopamine transporter(and hence as a dopamine reuptake inhibitor), and this action may explain some or all of its pharmacological effects.[6][7][8] Relative to adrafinil, modafinil possesses greater specificity in its action, lacking or having a reduced incidence of many of the common side effects of the former (including stomach pain, skin irritation, anxiety, and elevated liver enzymes with prolonged use).[9][10][11] There is a case report of two patients that adrafinil may increase interest in sex.[3] A case report of adrafinil-induced orofacial dyskinesia exists.[12][13] Reports of this side effect also exist for modafinil.[12]
Pharmacokinetics
In addition to modafinil, adrafinil also produces modafinil acid (CRL-40467) and modafinil sulfone (CRL-41056) as metabolites, which form from metabolic modification of modafinil.
History
Adrafinil was discovered in 1974 by two chemists working for the French pharmaceutical company Laboratoires Lafon who were screening compounds in search of analgesics.[14] Pharmacological studies of adrafinil instead revealed psychostimulant-like effects such as hyperactivity and wakefulness in animals.[14] The substance was first tested in humans, specifically for the treatment of narcolepsy, in 1977–1978.[14] Introduced by Lafon (now Cephalon), it reached the market in France in 1984,[4] and for the treatment of narcolepsy in 1985.[14][15] In 1976, two years after the discovery of adrafinil, modafinil, its active metabolite, was discovered.[14] Modafinil appeared to be more potent than adrafinil in animal studies, and was selected for further clinical development, with both adrafinil and modafinil eventually reaching the market.[14] Modafinil was first approved in France in 1994, and then in the United States in 1998.[15] Lafon was acquired by Cephalon in 2001.[16] As of September 2011, Cephalon has discontinued Olmifon, its adrafinil product, while modafinil continues to be marketed.[4]
Society and culture
Regulation
Athletic doping
Adrafinil and its active metabolite modafinil were added to the list of substances prohibited for athletic competition according to World Anti-Doping Agency in 2004.[17]
New Zealand
In 2005 a Medical Classification Committee in New Zealand recommended to MEDSAFE NZ that adrafinil be classified as a prescription medicine due to risks of it being used as a party drug. At that time adrafinil was not scheduled in New Zealand.[18]
Research
In a clinical trial with clomipramine and placebo as active comparators, adrafinil showed efficacy in the treatment of depression.[3] In contrast to clomipramine however, adrafinil was well-tolerated, and showed greater improvement in psychomotor retardation in comparison.[3] As such, “further investigations of the antidepressive effects of adrafinil are warranted.”[3]
/////////////
SYNTHESIS
Adrafinil (CAS NO.63547-13-7) was discovered in the late 1970s by scientists working with the French pharmaceutical company Group Lafon. First offered in France in 1986 as an experimental treatment for narcolepsy, Lafon later developed modafinil, the primary metabolite of adrafinil. Modafinil possesses greater selective alpha-1 adrenergic activity than adrafinil without the side effects of stomach pain, skin irritations, feelings of tension, and increases in liver enzyme levels.
It is important to monitor the liver of an individual using adrafinil. It can cause liver damage in some instances.
The Adrafinil with CAS registry number of 63547-13-7 is also known as 2-[(Diphenylmethyl)sulfinyl]-N-hydroxyacetamide. The IUPAC name is 2-Benzhydrylsulfinyl-N-hydroxyacetamide. It belongs to product categories of Aromatics Compounds; Aromatics; Intermediates & Fine Chemicals; Pharmaceuticals; Sulfur & Selenium Compounds. This chemical is a light pink solid and its EINECS registry number is 264-303-1. In addition, the formula is C15H15NO3S and the molecular weight is 289.35. This chemical is harmful if swallowed.
Physical properties about Adrafinil are: (1)ACD/LogP: 1.596; (2)ACD/LogD (pH 5.5): 1.60; (3)ACD/LogD (pH 7.4): 1.53; (4)ACD/BCF (pH 5.5): 9.60; (5)ACD/BCF (pH 7.4): 8.34; (6)ACD/KOC (pH 5.5): 175.52; (7)ACD/KOC (pH 7.4): 152.63; (8)#H bond acceptors: 4; (9)#H bond donors: 2; (10)#Freely Rotating Bonds: 6; (11)Index of Refraction: 1.653; (12)Molar Refractivity: 78.858 cm3; (13)Molar Volume: 215.542 cm3; (14)Polarizability: 31.262 10-24cm3; (15)Surface Tension: 67.25 dyne/cm; (16)Density: 1.342 g/cm3
Preparation of Adrafinil: it is prepared by reaction of diphenyl methyl bromide with thiourea. This reaction needs reagent NaOH. After reacting with chloroacetic acid, hydrochloric acid amine and hydrogen peroxide, the product is obtained. The yield is about 73%.

Uses of Adrafinil: it is used as non-amphetamine-type psychostimulant and can wake up and raise awareness. For the elderly arousal disorder and depressive symptoms in symptomatic treatment.


Benzhydrylsulphinyl-acetohydroxamic Acid (Adrafinil)1
Diphenylmethanethiol
15.2 g (0.2 mol) of thiourea and 150 ml of demineralized water are introduced into a 500 ml three-neck flask equipped with a central mechanical stirrer, and with a dropping funnel and a condenser on the (respective) side-necks.The temperature of the reaction mixture is brought to 50°and 49.4g (0.2 mol) of bromodiphenyl- methane are added all at once whilst continuing the heating. After refluxing for about 5 minutes, the solution, which has become limpid, is cooled to 20°C and 200 ml of 2.5 N NaOH are then added dropwise whilst maintaining the said temperature. The temperature is then again kept at the reflex for 30 minutes after which, when the mixture has returned to ordinary temperature (15-25°C), the aqueous solution is acidified with 45 ml of concentrated hydrochloric acid. The supernatant oil is extracted with 250 ml of diethyl ether and the organic phase is washed with 4×80 ml of water and then dried over magnesium sulphate. 39 g of crude diphenylmethane-thiol are thus obtained. Yield 97.5%.
Benzhydryl-thioacetic acid
10 g (0.05 mol) of diphenylmethane-thiol and 2g (0.05 mol) of NaOH dissolved in 60 ml of demineralised water are introduced successively into a 250 ml flask equipped with a magnetic stirrer and a reflux condenser. The reactants are left in contact for 10 minutes whilst stirring, and a solution consisting of 7g (0.075 mol) of chloroacetic acid, 3g (0.075 mol) of NaOH pellets and 60 ml of demineralized water is then added all at once. The aqueous solution is gently warmed to about 50°C for 15 minutes, washed with 50 ml of ether, decanted and acidified with concentrated hydrochloric acid. after filtration, 10.2g of benzhydryl-thioacetic acid are thus obtained. Melting point 129-130°C. Yield 79%.
Ethyl benzhydryl-thioacetate
The following reaction mixture is heated under reflux for 7 hours: 10.2 g (0.0395 mol) of benzhydryl-thioacetic acid, 100 ml of anhydrous ethanol and 2 ml of sulphuric acid. When heating has been completed, the ethanol isevaporated in vacuo; the oily residue is taken up in 100 ml of ethyl ether and the organic solution is then washed with water, with an aqueous sodium carbonate solution and then with water until the wash waters have a neutral pH. After drying over sodium sulphate, the solvent is evaporated. 10.5g of ethyl benzhydryl- thioacetate are thus obtained. Yield 93%.
Benzhydryl-thioacetohydroxamic acid
The following three solutions are prepared:
- Ethyl Benzhydryl-thioacetate 10.8 g (0.0378 mol) in 40 ml methanol
- Hydroxylamine hydrochloride 5.25 g (0.0756 mol) in 40 ml methanol
- Potassium Hydroxide pellets 7.3 g (0.0134 mol) in 40 ml methanol
The solutions are heated, if necessary, until they become limpid, and when the temperatures have again fallen to below 40°C, the solution of potassium hydroxide in methanol is poured into the solution of hydroxylamine hydrochloride in alcohol. Finally, at a temperature of about 5° to 10°C, the solution of ethyl benzhydryl- thioacetate is added in its turn. After leaving the reactants in contact for 10 minutes, the sodium chloride is filtered off the limpid solution obtained is kept for about 15 hours at ordinary temperature. The methanol is then evaporated under reduced pressure, the residual oil is taken up in 100 ml of water and the aqueous solution is acidified with 3 N hydrochloric acid. The hydroxamic acid which has crystallized is filtered off, washed with water and then dried. 9.1 g of product are obtained. Yield = 87.5%. Melting point 118-120°C.
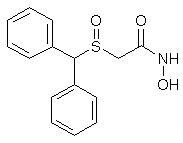
Adrafinil (CRL 40,028)
10.4g (0.038 mol) of benzhydryl-thioacetohydroxamic acid are oxidized at 40°C, over the course of 2 hours, by means of 3.8 ml (0.038 mol) of hydrogen peroxide of 110 volumes strength (33%), in 100 ml of acetic acid.
When the oxidation has ended, the acetic acid is evaporated under reduced pressure and the residual oil is taken up in 60 ml of ethyl acetate. The product which has crystallized is filtered off and then purified by recrystallisation from a 3:2 (by volume) mixture of ethyl acetate and isopropyl alcohol.
8g (73%) of Adrafinil, mp 159-160°C, are thus obtained. H2O Solubility
CLIP

Figure 2: GC/MS extracted ion chromatogram (a) and mass spectrum (b) of derivatized adrafinil in the electron ionization mode (monitoring the m/z 167, 165 and 152 ions; all the four peaks are derivatised adrafinil products).

Figure 4: LC/ESI-MS full scan chromatogram of adrafinil and its metabolites (a) (modafinil acid RT 3.8 min, adrafinil RT 4.0 min, modafinil RT 4.1 min), and LC/ESI-MS full scan mass spectra of modafinil acid (b), adrafinil (c), and (d) modafinil. (b, c and d showing the similar ions at m/z 167, 165, 152 together with the appropriate sodium and potassium adducts).

NMR
1H NMR PREDICT
13C NMR PREDICT
Patent
https://www.google.com/patents/US6180678 below
FIG. 1 shows the structure of adrafinil and its metabolites.
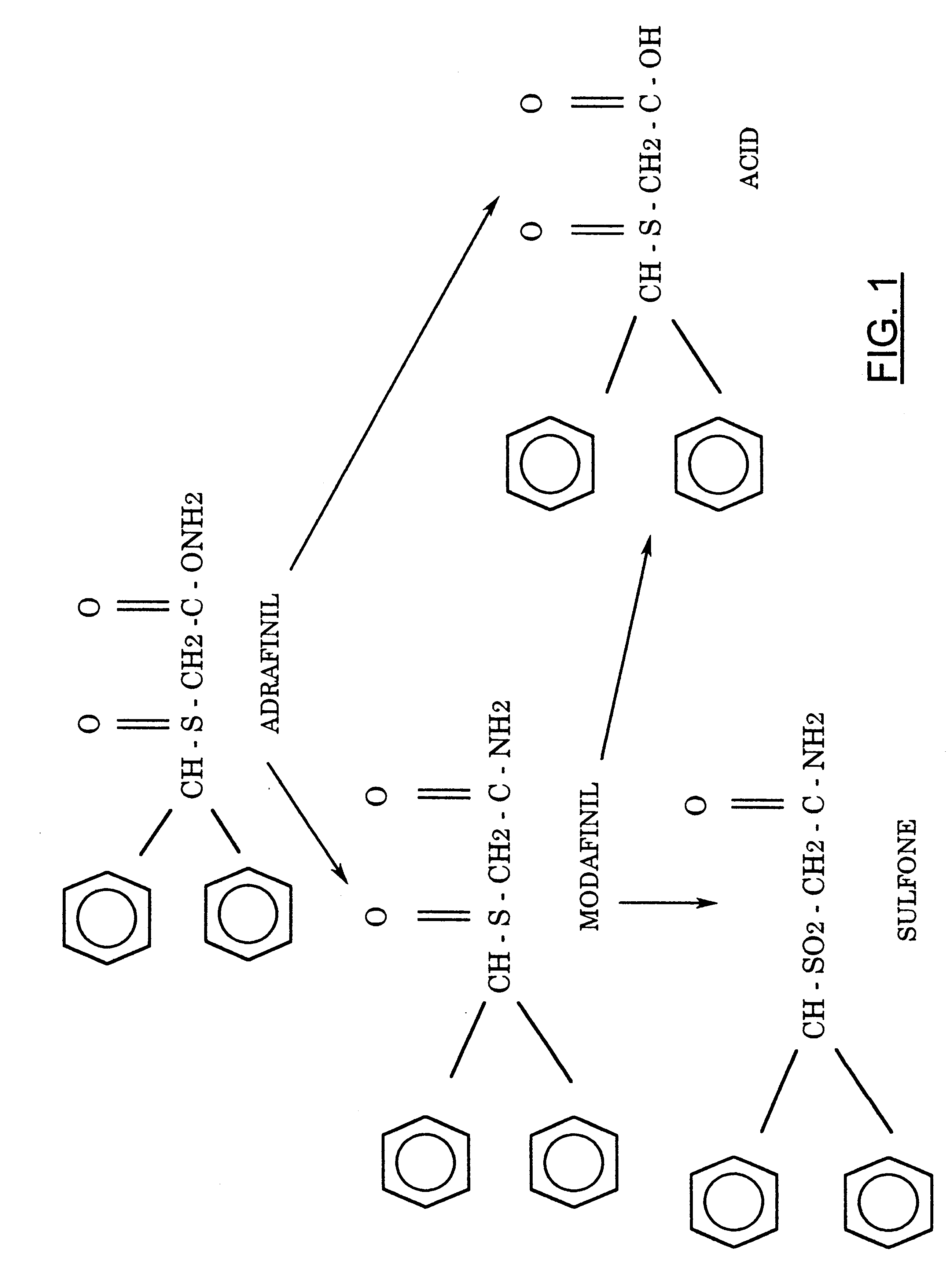
FIG. 2 shows the chemical synthesis of adrafinil.

//////////////
References
- Jump up^ Robertson P, Hellriegel ET (2003). “Clinical pharmacokinetic profile of modafinil”. Clin Pharmacokinet. 42 (2): 123–37. doi:10.2165/00003088-200342020-00002.PMID 12537513.
- ^ Jump up to:a b Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 20–. ISBN 978-3-88763-075-1.
- ^ Jump up to:a b c d e f g h i Milgram, Norton (1999). “Adrafinil: A Novel Vigilance Promoting Agent”.CNS Drug Reviews. 5 (3): 193–212. doi:10.1111/j.1527-3458.1999.tb00100.x. Retrieved2 October 2014.
- ^ Jump up to:a b c d AFSSAPS (2011). “Point d’information sur les dossiers discutés en commission d’AMM Séance du jeudi 1er décembre 2011 – Communiqué”.
- Jump up^ Simon P, Chermat R, Puech AJ (1983). “Pharmacological evidence of the stimulation of central alpha-adrenergic receptors”. Prog. Neuropsychopharmacol. Biol. Psychiatry. 7 (2-3): 183–6. doi:10.1016/0278-5846(83)90105-7. PMID 6310690.
- Jump up^ Zolkowska D, Jain R, Rothman RB, Partilla JS, Roth BL, Setola V, Prisinzano TE, Baumann MH (May 2009). “Evidence for the involvement of dopamine transporters in behavioral stimulant effects of modafinil”. The Journal of Pharmacology and Experimental Therapeutics. 329 (2): 738–46. doi:10.1124/jpet.108.146142.PMC 2672878
 . PMID 19197004.
. PMID 19197004. - Jump up^ Reith ME, Blough BE, Hong WC, Jones KT, Schmitt KC, Baumann MH, Partilla JS, Rothman RB, Katz JL (Feb 2015). “Behavioral, biological, and chemical perspectives on atypical agents targeting the dopamine transporter”. Drug and Alcohol Dependence. 147: 1–19. doi:10.1016/j.drugalcdep.2014.12.005. PMC 4297708
 . PMID 25548026.
. PMID 25548026. - Jump up^ Quisenberry AJ, Baker LE (Dec 2015). “Dopaminergic mediation of the discriminative stimulus functions of modafinil in rats”. Psychopharmacology. 232 (24): 4411–9.doi:10.1007/s00213-015-4065-0. PMID 26374456.
- Jump up^ Ballas, Christos A; Deborah Kim; Claudia F Baldassano; Nicholas Hoeh (July 2002). “Modafinil: past, present and future”. Expert Review of Neurotherapeutics. 2 (4): 449–57.doi:10.1586/14737175.2.4.449. PMID 19810941.
- Jump up^ Alan F. Schatzberg; Charles B. Nemeroff (2009). The American Psychiatric Publishing Textbook of Psychopharmacology. American Psychiatric Pub. pp. 850–. ISBN 978-1-58562-309-9.
- Jump up^ Ballas, Christos A; Kim, Deborah; Baldassano, Claudia F; Hoeh, Nicholas (2002). “Modafinil: past, present and future”. Expert Review of Neurotherapeutics. 2 (4): 449–457.doi:10.1586/14737175.2.4.449. ISSN 1473-7175. PMID 19810941.
- ^ Jump up to:a b Jeffrey K Aronson (31 December 2012). Side Effects of Drugs Annual: A worldwide yearly survey of new data in adverse drug reactions. Newnes. pp. 6–. ISBN 978-0-444-59503-4.
- Jump up^ Thobois S, Xie J, Mollion H, Benatru I, Broussolle E (2004). “Adrafinil-induced orofacial dyskinesia”. Mov. Disord. 19 (8): 965–6. doi:10.1002/mds.20154. PMID 15300665.
- ^ Jump up to:a b c d e f Antonio Guglietta (28 November 2014). Drug Treatment of Sleep Disorders. Springer. pp. 212–. ISBN 978-3-319-11514-6.
- ^ Jump up to:a b Jie Jack Li; Douglas S. Johnson (27 March 2013). Modern Drug Synthesis. John Wiley & Sons. pp. 2–. ISBN 978-1-118-70124-9.
- Jump up^ url=http://www.bloomberg.com/research/stocks/private/snapshot.asp?privcapId=1366624
- Jump up^ World Anti-Doping Agency – 2007 Prohibited List
- Jump up^ MCC Minutes Out of Session Meeting. Medsafe.govt.nz (2013-05-23). Retrieved on 2013-12-18.
External links
- “SID 184744 – PubChem Substance Summary“. PubChem Project. National Center for Biotechnology Information. Retrieved 7 December 2005.
- “Adrafinil – Bank of Automated Data on Drugs“. Bank of Automated Data on Drugs. VIDAL. Archived from the original on 5 October 2008. Retrieved 4 October 2008.
- Milgram, Norton W.; Callahan, Heather; Siwak, Christina (September 1992). “Adrafinil: A Novel Vigilance Promoting Agent” (PDF). CNS Drug Reviews. 5 (3): 193–212.doi:10.1111/j.1527-3458.1999.tb00100.x.

- Thobois, S. P.; Xie, J.; Mollion, H.; Benatru, I.; Broussolle, E. (August 2004). “Adrafinil-induced orofacial dyskinesia”. Movement Disorders. 19 (8): 965–966.doi:10.1002/mds.20154. PMID 15300665.
 |
|
 |
|
| Clinical data | |
|---|---|
| Trade names | Olmifon |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral |
| ATC code | N06BX17 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 80% |
| Metabolism | 75% (Liver) |
| Metabolites | Modafinil |
| Biological half-life | 1 hour (T1/2 is 12–15 hours for modafinil)[1] |
| Excretion | Kidney |
| Identifiers | |
|
Systematic (IUPAC) name: (±)-2-Benzhydrylsulfinylethanehydroxamic acid
|
|
| Synonyms | CRL-40028 |
| CAS Number | 63547-13-7 |
| PubChem (CID) | 3033226 |
| DrugBank | DB08925 |
| ChemSpider | 2297976 |
| UNII | BI81Z4542G |
| KEGG | D07348 |
| ChEMBL | CHEMBL93077 |
| Chemical and physical data | |
| Formula | C15H15NO3S |
| Molar mass | 289.351 g/mol |
| 3D model (Jmol) | Interactive image |
////////////ADRAFINIL
-
O=S(C(c1ccccc1)c2ccccc2)CC(=O)NO
Update……………..
Adrafinil
-
- Synonyms:CRL-40028
- ATC:N06BX17
- Use:α-adrenoceptor agonist (for symptomatic treatment of vigilance and depressive manifestations), stimulant
- Chemical name:2-[(diphenylmethyl)sulfinyl]-N-hydroxyacetamide
- Formula:C15H15NO3S
- MW:289.36 g/mol
- CAS-RN:63547-13-7
- EINECS:264-303-1
- LD50:>2048 mg/kg (M, i.p.); 1950 mg/kg (M, p.o.)
Substance Classes
Synthesis Path
Substances Referenced in Synthesis Path
| CAS-RN | Formula | Chemical Name | CAS Index Name |
|---|---|---|---|
| 91-01-0 | C13H12O | benzhydrol | Benzenemethanol, α-phenyl- |
| 63547-24-0 | C15H14O3S | (benzhydrylsulfinyl)acetic acid | Acetic acid, [(diphenylmethyl)sulfinyl]- |
| 63547-22-8 | C15H14O2S | (benzhydrylthio)acetic acid | Acetic acid, [(diphenylmethyl)thio]- |
| 79-11-8 | C2H3ClO2 | chloroacetic acid | Acetic acid, chloro- |
| 77-78-1 | C2H6O4S | dimethyl sulfate | Sulfuric acid, dimethyl ester |
| 4237-48-3 | C13H12S | diphenylmethanethiol | Benzenemethanethiol, α-phenyl- |
| 63547-22-8 | C15H14O2S | 2-(diphenylmethylthio)acetic acid | Acetic acid, [(diphenylmethyl)thio]- |
| 63547-44-4 | C15H15NO2S | 2-[(diphenylmethyl)thio]-N-hydroxyacetamide | Acetamide, 2-[(diphenylmethyl)thio]-N-hydroxy- |
| 7803-49-8 | H3NO | hydroxylamine | Hydroxylamine |
| 62-56-6 | CH4N2S | thiourea | Thiourea |
Trade Names
| Country | Trade Name | Vendor | Annotation |
|---|---|---|---|
| F | Olmifon | Cephalon |
Formulations
- cps. 300 mg
References
-
- DOS 2 642 511 (Lab. Lafon; appl. 22.9.1976; GB-prior. 2.10.1975).
- US 4 066 686 (Lab. Lafon; 3.1.1978; GB-prior. 2.10.1975).
- US 4 098 824 (Lab. Lafon; 4.7.1978; GB-prior. 2.10.1975).

THANKS AND REGARD’S
DR ANTHONY MELVIN CRASTO Ph.D
web link

 |
Dr. Anthony Melvin Crasto
Principal Scientist, Glenmark Pharma |
![]()
Generics: FDA´s New Guidance on Prior Approval Supplements
DRUG REGULATORY AFFAIRS INTERNATIONAL

Generics: The US Food and Drug Administration (FDA) recently published a new Guidance regarding Prior Approval Supplements (PAS). Read more about FDA´s Guidance for Industry “ANDA Submissions – Prior Approval Supplements Under GDUFA“.
On October 14, 2016, the US Food and Drug Administration (FDA) published a new Guidance regarding Prior Approval Supplements (PAS).
FDA says that “this guidance is intended to assist applicants preparing to submit to FDA prior approval supplements (PASs) and amendments to PASs for abbreviated new drug applications (ANDAs)”.
Specifically, the guidance describes how the Generic Drug User Fee Amendments of 2012 (GDUFA) performance metric goals apply to:
- A PAS subject to the refuse-to-receive (RTR) standards;
- A PAS that requires an inspection;
- A PAS for which an inspection is not required;
- An amendment to a PAS;
- Other PAS-related matters.
GDUFA is designed to speed the delivery of safe and effective generic drugs to the…
View original post 679 more words
Doxercalciferol, доксэркальциферол , دوكساركالسيفيرول , 度骨化醇
Doxercalciferol
- Molecular FormulaC28H44O2
- Average mass412.648
доксэркальциферол [Russian]
دوكساركالسيفيرول [Arabic]
度骨化醇 [Chinese]
1,3-Cyclohexanediol, 4-methylene-5-[(2E)-2-[(1R,3aS,7aR)-octahydro-7a-methyl-1-[(1R,2E,4R)-1,4,5-trimethyl-2-hexen-1-yl]-4H-inden-4-ylidene]ethylidene]-, (1R,3S,5Z)-
54573-75-0
|
Title: Doxercalciferol CAS Registry Number: 54573-75-0 CAS Name: (1a,3b,5Z,7E,22E)-9,10-Secoergosta-5,7,10(19),22-tetraene-1,3-diol Additional Names: 1a-hydroxyvitamin D2; 1-hydroxyergocalciferol Trademarks: Hectorol (Bone Care) Molecular Formula: C28H44O2 Molecular Weight: 412.65 Percent Composition: C 81.50%, H 10.75%, O 7.75% Literature References: Synthetic vitamin D prohormone. Prepn: H.-Y. P. Lam et al., Science 186, 1038 (1974); eidem, Steroids30, 671 (1977); H. E. Paaren et al., J. Org. Chem. 45, 3253 (1980). Comparative activity and toxicity: G. Sjöden et al., Proc. Soc. Exp. Biol. Med. 178, 432 (1985). Metabolism to bioactive form: J. C. Knutson et al., Endocrinology 136, 4749 (1995). Pharmacology: J. W. Coburn et al., Nephrol. Dial. Transplant. 11, Suppl. 3, 153 (1996). Clinical trial for suppression of secondary hyperparathyroidism in hemodialysis: J. M. Frazao et al., ibid. 13, Suppl. 3, 68 (1998). Properties: Crystals, mp 138-140°. uv max (ethanol): 265 nm (e 18300). LD50 orally in rats: 3.5-6.5 mg/kg (Sjöden). Melting point: mp 138-140° Absorption maximum: uv max (ethanol): 265 nm (e 18300) Toxicity data: LD50 orally in rats: 3.5-6.5 mg/kg (Sjöden) Therap-Cat: Antihyperparathyroid. Keywords: Antihyperparathyroid. |

CLIP

Doxercalciferol (1α-hydroxyvitamin D2) is a commercially approved vitamin D derivative used to treat chronic kidney disease (CKD) patients whose kidneys cannot metabolically introduce a hydroxyl group at C1. A new process for the production of doxercalciferol from ergocalciferol was developed using a continuous photoisomerization of a known vitamin D intermediate as the key step, thus circumventing the limitations of batch photoisomerization processes. Doxercalciferol is produced in an overall yield of about 10% from ergocalciferol.
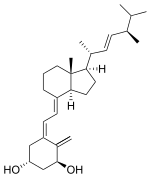
Doxercalciferol
1H NMR (CDCl3) δ 6.40 (d, 1H, J = 11.2), 6.04 (d, 1H, J = 11.2), 5.35 (s, 1H), 5.15–5.29 (m, 2H), 5.03 (s, 1H), 4.45 (dd, 1H, J = 7.3, 4.0), 4.21–4.31 (m, 1H), 2.81–2.90 (m, 1H), 2.62 (d, 1H, J = 13.3), 2.34 (dd, 1H, J = 13.3, 6.5), 1.83–2.11(m, 6H), 1.42–1.79 (m, 7H), 1.21–1.40 (m, 3H), 1.04 (d, 3H, J = 6.6), 0.94 (d, 3H, J = 6.8), 0.86 (t, 6H, J = 7.3), 0.58 (s, 3H) ppm.
Doxercalciferol (trade name Hectorol) is drug for secondary hyperparathyroidism and metabolic bone disease.[1] It is a synthetic analog of ergocalciferol (vitamin D2). It suppresses parathyroid synthesis and secretion.[2]
PATENT


CLIP

References
 |
|
| Names | |
|---|---|
| IUPAC name
(1S,3R,5Z,7E,22E)-9,10-Secoergosta-5,7,10,22-tetraene-1,3-diol |
|
| Other names
1-Hydroxyergocalciferol; 1-Hydroxyvitamin D2; 1α-Hydroxyergocalciferol; 1α-Hydroxyvitamin D2; Hectorol; TSA 840 |
|
| Identifiers | |
| 54573-75-0 |
|
| 3D model (Jmol) | Interactive image |
| ChEMBL | ChEMBL1200810 |
| ChemSpider | 4444554 |
| DrugBank | DB06410 |
| ECHA InfoCard | 100.170.997 |
| 2790 | |
| PubChem | 5281107 |
| UNII | 3DIZ9LF5Y9 |
| Properties | |
| C28H44O2 | |
| Molar mass | 412.66 g·mol−1 |
| Pharmacology | |
| H05BX03 (WHO) | |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). |
|
/////////////

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....