Home » Uncategorized (Page 51)
Category Archives: Uncategorized
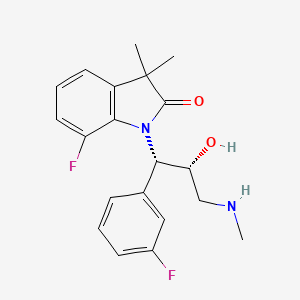
QbD: Controlling CQA of an API
DRUG REGULATORY AFFAIRS INTERNATIONAL
The importance of Quality by Design (QbD) is being realized gradually, as it is gaining popularity among the generic companies. However, the major hurdle faced by these industries is the lack of common guidelines or format for performing a risk-based assessment of the manufacturing process. This article tries to highlight a possible sequential pathway for performing QbD with the help of a case study. The main focus of this article is on the usage of failure mode and effect analysis (FMEA) as a tool for risk assessment, which helps in the identification of critical process parameters (CPPs) and critical material attributes (CMAs) and later on becomes the unbiased input for the design of experiments (DoE). In this case study, the DoE was helpful in establishing a risk-based relationship between critical quality attributes (CQAs) and CMAs/CPPs. Finally, a control strategy was established for all of the CPPs and CMAs…
View original post 158 more words
A Concise and Highly Efficient Synthesis of Praziquantel as an Anthelmintic Drug
PAPER
HETEROCYCLES
An International Journal for Reviews and Communications in Heterocyclic Chemistry
Web Edition ISSN: 1881-0942
Published online: 11th October, 2016
Paper | Regular issue | Prepress
DOI: 10.3987/COM-16-13538
■ A Concise and Highly Efficient Synthesis of Praziquantel as an Anthelmintic Drug
Zhezhou Yang, Lin Zhang, Huirong Jiao, Rusheng Bao, Weiwei Xu, and Fuli Zhang*
*Shanghai Institute of Pharmaceutical Industry, China State Institute of Pharmaceutical Industry, 285 Gebaini Road, Shanghai 201203, China
Abstract
A concise and practical synthesis of praziquantel as anthelmintic drug is described. The key steps include a monoalkylation of ethanolamine for the preparation of 2-(2-hydroxyethylamino)-N-phenethylacetamide and a mild oxidation protocol with SO3-Py/DMSO as oxidant to transform alcohol into the corresponding aza-acetal. The telescoped synthesis is composed of five steps without purification of the intermediates, providing an overall yield of 80% with 99.8% purity after crystallization.
///////////////Praziquantel, Anthelmintic Drug
Critical Impurities in Pharmaceutical Water
DRUG REGULATORY AFFAIRS INTERNATIONAL

The quality of the source water used to produce pharmaceutical water plays an important role for both the design of the treatment and the validation of the water system. FDA Warning Letters over the past few years have shown that compliance with the specification of pharmaceutical water is not enough. A validation of the treatment process is expected. This includes documentation of the process capacity to produce pharmaceutical water according to specification. If we do not know the quality of the source water, however, the purification capacity is not known either. As a consequence, fluctuations of the quality of the source (feed) water quality may lead to water that does not comply with the specification after purification. Or it is not known up to which quality level of the source water pharmaceutical water that complies with the specification can be produced. Therefore, it is important to know the impurities respectively their concentration…
View original post 487 more words
FDA presentation at the ECA Conference Particles in Parenterals
DRUG REGULATORY AFFAIRS INTERNATIONAL

At the Particles in Parenterals Conference Dr Stephen Langille from the US FDA gave a talk on the FDA’s current thinking with regard to the visual inspection of medicinal products for parenteral use.
Dr Stephen Langille from the US FDA gave a talk on the FDA’s current thinking with regard to the visual inspection of medicinal products for parenteral use. In his presentation he showed the number of recalls caused by visible particulate matter over the last 11 years. For him, most of the recalls were justified when the types of particles found were taken into consideration. He also emphasized that something is possibly wrong in the visual inspection process if particles found in the market are bigger than 1000 µm.
The prevention of particles is very important to him. From his perspective the best particle is one which is not in the product. Also important to him…
View original post 179 more words
The impact of the FDA Combination Products Guidance on Nasal and Oral Inhalation Drug Products
DRUG REGULATORY AFFAIRS INTERNATIONAL

The FDA draft guidance for combination products has a substantial impact on the development of Oral Inhalation and Nasal Drug Products (OINDPs) as it requires that the manufacturers have to be compliant not only with CGMPs for the drugs (21 CFR Parts 210 and 211) but also with the quality system (QS) regulations for devices (21 CFR Part 820). Find out more about the FDA Draft Guidance for Combination Products.
Based on the CGMP requirements for single-entity and co-packaged combination products (21 CFR Part 4) the manufacturers of Oral Inhalation and Nasal Drug Products (OINDPs) have to be compliant with CGMPs for the drug constituent part(s) (21 CFR Parts 210 and 211) and the quality system (QS) regulations for device constituent part(s) (21 CFR Part 820).
This can be achieved either by a drug CGMP-based streamlined approach (21 CFR 4.4(a)) or a QS regulation-based streamlined approach (21 CFR 4.4(b)). Following the…
View original post 125 more words
Counterfeit of medicines causes 37,000 job losses in EU Pharma Industry
DRUG REGULATORY AFFAIRS INTERNATIONAL
Counterfeit medicine is an increasing problem for public health and economy. This is no longer a problem of certain regions such as Asia and Africa. It has now also become an issue in the EU and US. The European Union Intellectual Property Office (EUIPO) published a press release on 29 September 2016 in which they state that fake medicines cost the EU pharmaceutical sector 10.2 billion Euro every year. Read more about the latest figures on counterfeit medicines
Counterfeit medicine is an increasing problem for public health and economy. This is no longer a problem of certain regions such as Asia and Africa. It has now also become an issue in the EU and the US. In the past, counterfeit medicines could not enter the legal supply chain in the EU and US. But the problem has now also been arising in western countries. A number ofcases of counterfeit medicines were…
View original post 158 more words
Onions contain a powerful cancer fighting compound — ClinicalNews.Org
Onions contain a powerful cancer fighting compound Onions contain a powerful cancer fighting compound We review the study” Anti-cancer effects found in natural compound derived from onions ” This study was done in regard to Ovarian Cancer, but should have potential in a variety of cancers. * Tsuboki, J. et al. Onionin A inhibits ovarian […]
via Onions contain a powerful cancer fighting compound — ClinicalNews.Org
BMS-442608

BMS-442608
8-Azaspiro(4.5)decane-7,9-dione, 6-hydroxy-8-(4-(4-(2-pyrimidinyl)-1-piperazinyl)butyl)-, (6R)-
(6R)-6-Hydroxy-8-[4-[4-(2-pyrimidinyl)-1-piperazinyl]butyl]-8-azaspiro[4.5]decane-7,9-dione
(R)-6-Hydroxybuspirone, UNII-93881477KV, CAS 477930-30-6,
Molecular Formula, C21-H31-N5-O3, Molecular Weight, 401.5079
BMS-442608 is a 5-HT1A partial agonist. BMS-442608 is the R-enantiomer. (R)-Enantiomer showed higher affinity and selectivity for the 5HT1A receptor compared to the (S)-enantiomer. (S)-Enantiomer has advantage of being cleared more slowly from blood compared to the (R)-enantiomer.
PAPER
Enantioselective α-Hydroxylation of 2-Arylacetic Acid Derivatives and Buspirone Catalyzed by Engineered Cytochrome P450 BM-3
http://pubs.acs.org/doi/abs/10.1021/ja061261x

Here we report that an engineered microbial cytochrome P450 BM-3 (CYP102A subfamily) efficiently catalyzes the α-hydroxylation of phenylacetic acid esters. This P450 BM-3 variant also produces the authentic human metabolite of buspirone, R-6-hydroxybuspirone, with 99.5% ee.

PATENT
US 20020193380
http://www.google.st/patents/US20020193380
PATENT
WO 2003009851
https://google.com/patents/WO2003009851A1?cl=en
PAPER
Tetrahedron: Asymmetry (2005), 16(16), 2711-2716.
Volume 16, Issue 16, 22 August 2005, Pages 2711–2716
Preparation of (R)- and (S)-6-hydroxybuspirone by enzymatic resolution or hydroxylation
- Department of Process Research and Development, Bristol-Myers Squibb Pharmaceutical Research Institute, One Squibb Drive, New Brunswick, NJ 08903, USA
http://www.sciencedirect.com/science/article/pii/S0957416605005549
http://dx.doi.org/10.1016/j.tetasy.2005.07.020
Abstract
6-Hydroxybuspirone is an active metabolite of the antianxiety drug buspirone. The (R)- and (S)-enantiomers of 6-hydroxybuspirone were prepared using an enzymatic resolution process. l-Amino acid acylase from Aspergillus melleus (Amano Acylase 30000) was used to hydrolyze racemic 6-acetoxybuspirone to (S)-6-hydroxybuspirone in 95% ee after 45% conversion. The remaining (R)-6-acetoxybuspirone with 88% ee was converted to (R)-6-hydroxybuspirone by acid hydrolysis. The ee of both enantiomers could be improved to 99% by crystallization as a metastable polymorph. (S)-6-Hydroxybuspirone was also obtained in 88% ee and 14.5% yield by hydroxylation of buspirone using Streptomyces antibioticus ATCC 14890.
Graphical abstract
Tetrahedron: Asymmetry (2005), 16(16), 2778-2783
http://dx.doi.org/10.1016/j.tetasy.2005.07.015
Abstract
The enantioselective microbial reduction of 6-oxo-8-[4-[4-(2-pyrimidinyl)-1-piperazinyl]butyl]-8-azaspiro[4.5]decane-7,9-dione 1 to either of the corresponding (R)- or (S)-6-hydroxy-8-[4-[4-(2-pyrimidinyl)-1-piperazinyl]butyl]-8-azaspiro[4.5]decane-7,9-diones 2 and 3 is described.
PAPER
Enzyme and Microbial Technology (2006), 39(7), 1441-1450.
http://dx.doi.org/10.1016/j.enzmictec.2006.03.033
Abstract
The enantioselective microbial reduction of 6-oxo-8-[4-[4-(2-pyrimidinyl)-1-piperazinyl]butyl]-8-azaspiro[4.5]decane-7,9-dione (1) to either of the corresponding (S)- and (R)-6-hydroxy-8-[4-[4-(2-pyrimidinyl)-1-piperazinyl]butyl]-8-azaspiro[4.5]decane-7,9-diones (2 and 3, respectively) is described. The NADP+-dependent (R)-reductase (RHBR) which catalyzes the reduction of 6-ketobuspirone (1) to (R)-6-hydroxybuspirone (3) was purified to homogeneity from cell extracts of Hansenula polymorpha SC 13845. The subunit molecular weight of the enzyme is 35,000 kDa based on sodium dodecyl sulfate gel electrophoresis and the molecular weight of the enzyme is 37,000 kDa as estimated by gel filtration chromatography. (R)-reductase from H. polymorpha was cloned and expressed in Escherichia coli. To regenerate the cofactor NADPH required for reduction we have cloned and expressed the glucose-6-phosphate dehydrogenase gene from Saccharomyces cerevisiae in E. coli. The NAD+-dependent (S)-reductase (SHBR) which catalyzes the reduction of 6-ketobuspirone (1) to (S)-6-hydroxybuspirone (2) was purified to homogeneity from cell extracts of Pseudomonas putida SC 16269. The subunit molecular weight of the enzyme is 25,000 kDa based on sodium dodecyl sulfate gel electrophoresis. The (S)-reductase from P. putida was cloned and expressed in E. coli. To regenerate the cofactor NADH required for reduction we have cloned and expressed the formate dehydrogenase gene from Pichia pastoris in E. coli. RecombinantE. coli expressing (S)-reductase and (R)-reductase catalyzed the reduction of 1 to (S)-6-hyroxybuspirone (2) and (R)-6-hyroxybuspirone (3), respectively, in >98% yield and >99.9% e.e.
PATENT
https://www.google.com/patents/US6686361
| Inventors | Joseph P. Yevich, Robert F. Mayol, Jianqing Li,Frank Yocca |
| Original Assignee | Bristol-Myers Squibb Company |
The present invention relates to methods of treating anxiety and depression using R-6-hydroxy-buspirone and pharmaceutical compositions containing R-6-hydroxy-buspirone.
Buspirone, chemically: 8-[4-[4-(2-pyrimidinyl)1-piperazinyl]butyl-8-azaspiro(4,5)-decane-7,9-dione, is approved for the treatment of anxiety disorders and depression by the United States Food and Drug Administration. It is available under the trade name BUSPAR® from Bristol-Myers Squibb Company.
Studies have shown that buspirone is extensively metabolized in the body. (See, for example, Mayol, et al., Clin. Pharmacol. Ther., 37, p. 210, 1985). One of the metabolites is 6-hydroxy-8-[4-[4-(2-pyrimidinyl)1-piperazinyl]butyl-8-azaspiro(4,5)-decane-7,9-dione having Formula I. This metabolite is also known as BMS 28674, BMS 442608, or
as 6-hydroxy-buspirone. This compound is believed to be the active metabolite of buspirone and its use in treating anxiety disorders and depression is disclosed in U.S. Pat. No. 6,150,365. The specific stereochemistry of 6-hydroxy-buspirone has not been described previously. Neither racemic 6-hydroxy-buspirone nor its enantiomers are commercially available at the present time.
Preclinical studies demonstrate that 6-hydroxy-buspirone, like buspirone, demonstrates a strong affinity for the human 5-HT1A receptor. In functional testing, 6-hydroxy-buspirone produced a dose-dependent anxiolytic response in the rat pup ultrasonic vocalization test, a sensitive method for assessment of anxiolytic and anxiogenic effects (Winslow and Insel, 1991, Psychopharmacology, 105:513-520).
Clinical studies in volunteers orally dosed with buspirone demonstrate that 6-hydroxy-buspirone blood plasma levels were not only 30 to 40 times higher but were sustained compared to buspirone blood plasma levels. The time course of 6-hydroxy-buspirone blood plasma levels, unlike buspirone blood plasma levels, correlate more closely with the sustained anxiolytic effect seen following once or twice a day oral dosing with buspirone.
Although buspirone is an effective treatment for anxiety disorders and depression symptomatology in a significant number of patients treated, about a third of patients get little to no relief from their anxiety and responders often require a week or more of buspirone treatment before experiencing relief from their anxiety symptomatology. Further, certain adverse effects are reported across the patient population. The most commonly observed adverse effects associated with the use of buspirone include dizziness, nausea, headache, nervousness, lightheadedness, and excitement. Also, since buspirone can bind to central dopamine receptors, concern has been raised about its potential to cause unwanted changes in dopamine-mediated neurological functions and a syndrome of restlessness, appearing shortly after initiation of oral buspirone treatment, has been reported in small numbers of patients. While buspirone lacks the prominent sedative effects seen in more typical anxiolytics such as the benzodiazepines, patients are nonetheless advised against operating potentially dangerous machinery until they experience how they are affected by buspirone.
It can be seen that it is desirable to find a medicament with buspirone’s advantages but which demonstrates more robust anxiolytic potency with a lack of the above described adverse effects.
Formation of 6-hydroxy-buspirone occurs in the liver by action of enzymes of the P450 system, specifically CYP3A4. Many substances such as grapefruit juice and certain other drugs; e.g. erythromycin, ketoconazole, cimetidine, etc., are inhibitors of the CYP3A4 isozyme and may interfere with the formation of this active metabolite from buspirone. For this reason it would be desirable to find a compound with the advantages of buspirone but without the drug—drug interactions when coadministered with agents affecting the activity level of the CYP3A4 isozyme.
R-6-hydroxy-buspirone may be prepared utilizing methods of synthesis and enantiomeric separation known to one skilled in the art. One method of preparation (Scheme 1) utilizes buspirone as a starting material to produce racemic 6-hydroxy-buspirone that is separated into the two enantiomers by chiral chromatographic techniques.
An improved one-step synthesis of racemic 6-hydroxy-buspirone is set forth in Scheme 2. Again, enantiomeric separation provides R-6-hydroxy-buspirone.
EXAMPLE 1 Preparation of 6-Hydroxy-buspirone
A. Di-4-nitrobenzyl Peroxydicarbonate (V)
Di-4-nitrobenzyl peroxydicarbonate was prepared using a modification of the literature procedure1. Thus, to an ice-cold solution of 4-nitrobenzyl chloroformate (10.11 g, 4.7 mmol) in acetone (20 mL) was added dropwide over 30 min an ice-cold mixture of 30% H2O2 (2.7 mL, 24 mmol) and 2.35 N NaOH (20 mL, 47 mmol). The mixture was vigorously stirred for 15 min and then it was filtered and the filter-cake was washed with water and then with hexane. The resulting damp solid was taken up in dichloromethane, the solution was dried (Na2SO4) and then it was diluted with an equal volume of hexane. Concentration of this solution at 20° C. on a rotary evaptor gave a crystalline precipitate which was filtered, washed with hexane and dried in vacuo to give compound III (6.82 g, 74%) as pale yellow microcrystals, mp 104° C. (dec).
1F. Strain, et al., J. Am. Chem. Soc., 1950, 72, 1254
Di-4-nitrobenzyl peroxydicarbonate was found to be a relatively stable material which decomposed as its melting point with slow gas evolution. In comparison, dibenzyl peroxydicarbonate2 decomposed with a sudden vigorous expulsion of material from the melting point capillary.
2Cf. M. P. Gove, J. C. Vedaras, J. Org. Chem., 1986, 51, 3700
B. 6-(4-Nitrobenzyl peroxydicarbonatyl)-8-[4-[4-(2-pyrimidinyl)-piperazinyl]-butyl]-8-azaspiro[4.5]-7,9-dione (III)
To a solution of 8-[4-[4-(2-pyrimidinyl)-piperazinyl]-butyl]-8-azaspiro[4.5]-7,9-dione (buspirone: 10 g, 26 mmole) in dry THF (250 mL) was added LiN (Me3Si)2 (28.5 mL of a 1 M THF solution) at 78° C. and stirred for 3 h and then a solution of di-4-nitrobenzyl peroxydicarbonate (11.2 g) in dry THF (150 mL) was added dropwide over 1 h. Stirring was continued at −78° C. for 1 h.
The cooling bath was removed and the reaction solution was poured into a mixture of H2O and EtOAc. The organic phase was separated and washed with H2O and then brine. The organic base was dried and then evaporated to a viscous oil. Flash chromatography of this oil, eluting the silica column with MeCN-EtOAc (1:2) gave crude product which was washed with acetone, to remove unreacted buspirone, leaving 6.23 g of a white solid (46%) product (III).
C. 6-Hydroxy-8-[4-[4-(2-pyrimidinyl)-piperazinyl]-butyl]-8-azaspiro[4.5]-7,9-dione (I; 6-Hydroxy-buspirone)
A mixture of III (4.0 g; 6.9 mmole) and 10% Pd/C (about 1 g) in MeOH (100 mL) was hydrogenated in a Parr shaker at 40-45 psi for 1 h. The hydrogenation mixture was filtered through a Celite pad which was then washed with EtOAc. The filtrate was evaporated to a gum which was purified by flash chromatography through a silica gel column eluting with EtOAc to give 0.41 g of an off-white solid (I).
Anal. Calcd. for C21H31N5O3: C, 62.82; H, 7.78; N, 17.44. Found: C, 62.84; H, 7.81; N, 17.33.
EXAMPLE 2 Enantiomeric Separation
Preparative Chiral HPLC Purification Procedure for 6-hydroxy-buspirone
1.1 g 6-Hydroxy-buspirone is dissolved in 55 mL HPLC grade methanol (20 mg/mL). Repetitive 0.5 mL injections of the solution are made on a Chirobiotic-Vancomycin Chiral HPLC column, 22.1 mm×250 mm, 10 um particle size (Advanced Separation Technologies, Inc., Whippany, N.J.) equilibrated with a mobile phase of MeOH/acetic acid/triethylamine, 100/0.2/0.1, v/v/v, at a flow rate of 20 mL/minute. The UV trace is monitored at 236 nm. Each enantiomer (RTs˜10.9 and ˜13.4 minutes, respectively) is collected in ˜1000 mL of mobile phase and condensed separately under reduced pressure at 40° C. ˜2 mL of clear solution resulting from the evaporation of methanol is diluted with 5 mL of H2O. The pH of these solutions is adjusted from 5 to ˜8 with NH4OH, upon which a white precipitate is observed. The precipitates are centrifuged, and the aqueous layers extracted three times with equal volumes of methylene chloride. The methylene chloride layers are evaporated and any remaining solid is re-chromatographed. The centrifuged precipitates are washed three times with H2O to remove any residual salts and air dried at room temperature.
The basic form of R-6-hydroxy-buspirone can be converted to the hydrochloride salt by treatment of an ethanol solution of R-6-hydroxy-buspirone with ethanolic HCl.
EXAMPLE 3 One-Step Synthesis of 6-Hydroxy-buspirone (I)
Buspirone (19.3 g, 50 mmole) was dissolved in dry THF (400 mL) and the resulting solution was cooled to −78° C. A solution of KN(SiMe3)2 in toluene (100 mL, 1 M) was added slowly. After the reaction mixture was stirred at −78° C. for 1 h, a solution of 2-(phenylsulfonyl)-3-phenyloxaziridine (Davis reagent, prepared according to literature method: F. A. Davis, et al., Org. Synth., 1988, 66, 203) (17.0 g, 65 mmole) in dry THF (150 mL, precooled to −78° C.) was added quickly via a cannular. After stirred for 30 mins at −78° C., the reaction was quenched with 1 N HCl solution (500 mL). It was extracted with EtOAc (3×500 mL). The aqueous layer was separated, neutralized with saturated sodium bicarbonate solution, and extracted with EtOAc (3×500 mL). The combined organic extracts were dried over Na2SO4, filtered, and concentrated under reduced pressure to give a white solid residue which was subjected to column chromatography using CH2Cl2/MeOH/NH4OH (200:10:1) as the eluent to give pure 6-hydroxy-buspirone (I, 7.2 g) and a mixture of buspirone and 6-hydroxy-buspirone (I). The mixture was purified by above column chromatography to afford another 3.3 g of pure 6-hydroxy-buspirone (I).
1H NMR (CDCl3) δ8.30 (d, J=4.7 Hz, 2H), 6.48 (t, J=4.7 Hz, 1H), 4.20 (s, 1H), 3.83-3.72 (m, 5H), 3.55 (s, 1H), 2.80 (d, J=17.5 Hz, 1H), 2.55-2.40 (m, 7H), 2.09-2.03 (m, 1H), 1.76-1.54 (m, 10 H), 1.41-1.36 (m, 1H), 1.23-1.20 (m, 1H).
EXAMPLE 4 5-HT1A Receptor Binding Assay
Membranes are prepared for binding using the human 5-HT1 A receptor expressed in HEK293 cells. Cells are collected and ruptured using a dounce homogenizer. The cells are spun at 18000×g for 10 minutes and the pellet is resuspended in assay buffer, frozen in liquid nitrogen and kept at −80° C. until the day of the assay.
A total of 30 ug protein is used per well. The assay is carried out in 96-deep-well plates. The assay buffer is 50 mM HEPES containing 2.5 mM MgCl2 and 2 mM EGTA. The membrane preparation is incubated at 25° C. for 60 minutes with 0.1 nM to 1000 nM test compound and 1 nM 3H-8-OH-DPAT. 10 mM serotonin serves as blocking agent to determine non-specific binding. The reaction is terminated by the addition of 1 ml of ice cold 50 mM HEPES buffer and rapid filtration through a Brandel Cell Harvester using Whatman GF/B filters. The filter pads are counted in an LKB Trilux liquid scintillation counter. IC50 values are determined using non-linear regression by Excel-fit.
EXAMPLE 5 Rat Pup Isolation-Induced Ultrasonic Vocalization Test
Harlan Sprague-Dawley rat pups (male and female) were housed in polycarbonate cages with the dam until 9-11 days old. Thirty minutes before testing, pups were removed from the dam, placed into a new cage with a small amount of home bedding and brought into the lab and placed under a light to maintain body temperature at 37° C. Pups were then weighed, sexed, marked and returned to the litter group until behavioral assessment. Testing took place in a Plexiglas recording chamber that contained a metal plate maintained at (18-20° C.) with a 5×5 cm grid drawn on the plate. A microphone was suspended 10 cm above the plate to record ultrasonic vocalizations. Ultrasonic calls were recorded using the Noldus UltraVox system providing online analysis of the frequency and duration of calls. The number of grid cells entered by the pup was also collected by visual scoring. Pups that failed to emit at least 60 calls during a 5 minute pretest session were excluded from pharmacological assessment. Immediately following the collection of the baseline measures, pups were injected with vehicle or drug subcutaneously at the nape of the neck and returned to its littermates. Thirty minutes later, pups were retested on each of the dependent measures (vocalization and grid cell crossings) to assess drug effects. Unless otherwise specified, each pup was used only once. Baseline differences and percent change from baseline for the frequency of ultrasonic vocalizations and grid cell crossings were analyzed using a one-way ANOVA. Bonferroni/Dunn post hoc comparisons were performed to assess the acute drug effects with vehicle control. Log-probit analysis was used to estimate the dose (milligrams per kilogram) of each agonist predicted to inhibit isolation-induced ultrasonic vocalizations by 50% (ID50). All comparison were made with an experimental type I error rate (α) set at 0.05.
Doses for each drug were administered in an irregular order across several litters. R-6-hydroxy-buspirone and racemic 6-hydroxy-buspirone were dissolved in physiological saline (0.9% NaCl; vehicle). All injections were administered subcutaneously in a volume of 10 ml/kg. Doses of the drug refer to weight of the salt.
Clip
Pharmacokinetics of 6-hydroxybuspirone and its enantiomers administered individually or following buspirone administration in humans
The objective of this study was to assess the pharmacokinetics of 6-hydroxybuspirone (6OHB) when given orally via three forms: racemate (BMS-528215), S-enantiomer (BMS-442606) and R-enantiomer (BMS-442608), versus following the administration of buspirone. A double-blind, randomized, four-period, four-treatment, crossover study balanced for residual effects in healthy subjects was conducted (n=20). Subjects received single 10 mg doses of each compound in a randomized fashion with pharmacokinetics determined over a 24 h period. There was a 4-day washout between each dosing period. All three forms of 6OHB (racemate, S-enantiomer and R-enantiomer) were well tolerated. There was nterconversion between enantiomers. The dominant enantiomer was the S-enantiomer no matter which form of 6OHB was administered. All three forms of 6OHB produced approximately 2- to 3-fold greater exposure to total 6OHB than did buspirone. All three forms produced equal exposure to 1-(2-pyrimidinyl)-piperazine (1-PP) which was approximately 30% less than the 1-PP exposure derived from buspirone administration. All three forms of 6OHB produced approximately 3-fold higher 6OHB:1-PP ratios and approximately 2.5-fold higher total 6OHB exposures than did buspirone administration. All compounds were well tolerated. There seemed to be no advantage of one of the enantiomers of 6OHB over the racemate. Therefore, the racemate was chosen for further clinical development. Copyright © 2007 John Wiley & Sons, Ltd.
| Cited Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| US6150365 | Jun 6, 2000 | Nov 21, 2000 | Bristol-Myers Squibb Company | Anxiety method |
| Reference | ||
|---|---|---|
| 1 | Mayol, et al., “Pharmacokinetics and Disposition of 14C-Buspirone HCI After Intravenous and Oral Dosing in Man,” Clin. Pharmacol. Ther., 37, p. 210, 1985. | |
| 2 | * | Robichaud et al. in Annual Reports in Medicinal Chemistry, vol. 35,pp. 11-20 (2000).* |
| 3 | Winslow, et al., “Serotonergic modulation of the rat pup ultrasonic isolation call: studies with 5HT1 and 5HT2 subtype-selective agonists and antagonists,” Psychopharmacology, 105, pp. 513-520, 1991. | |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| US20090023744 * | Jun 17, 2008 | Jan 22, 2009 | The General Hospital Corporation | Combination therapy for depression |
| WO2015197079A1 | Jun 25, 2015 | Dec 30, 2015 | Contera Pharma Aps | Use of buspirone metabolites |
REFERENCES
1: Dockens RC, Tran AQ, Zeng J, Croop R. Pharmacokinetics of 6-hydroxybuspirone and its enantiomers administered individually or following buspirone administration in humans. Biopharm Drug Dispos. 2007 Oct;28(7):393-402. PubMed PMID: 17668416.
///////////////BMS-442608, BMS 442608, (R)-6-Hydroxybuspirone, UNII-93881477KV, CAS 477930-30-6
c1cnc(nc1)N2CCN(CC2)CCCCN3C(=O)CC4(CCCC4)[C@H](C3=O)O
MW: 401.5079
S FORM
ANDA Submissions – Prior Approval Supplements Under GDUFA, FDA Guidance document
DRUG REGULATORY AFFAIRS INTERNATIONAL
ANDA Submissions – Prior Approval Supplements Under GDUFA, FDA Guidance document, oct 2016, Generics
 The presentation will load below
The presentation will load below
//////ANDA Submissions, Prior Approval Supplements, GDUFA, FDA Guidance document
WAY-315193
7-fluoro-1-[(1S,2R)-1-(3-fluorophenyl)-2-hydroxy-3-(methylamino)propyl]-3,3-dimethyl-1,3-dihydro-2H-indol-2-one

7-f luoro-1 -[(1 S,2R)-1 -(3-fluorophenyl)-2-hydroxy-3- (methylamino)propyl]-3,3- dimethyl-1 ,3-dihydro-2H-indol-2-one hydrochloride
WAY-315193
| 7-fluoro-1-[(1S,2R)-1-(3-fluorophenyl)-2-hydroxy-3-(methyl amino)propyl]-3,3-dimethyl-1,3-dihydro-2H-indol-2-one; | |
| Molecular Formula: | C20H22F2N2O2 |
|---|---|
| Molecular Weight: | 360.397686 g/mol |
7-Fluoro-1-[(1S,2R)-1-(3-fluorophenyl)-2-hydroxy-3-(methylamino)propyl]-3,3-dimethylindolin-2-one Hydrochloride
Drugs that possess norepinephrine reuptake inhibition, either selectively or in combination with serotonin reuptake inhibition, have been used for multiple indications including major depressive disorder, attention deficit hyperactivity disorder, stress urinary incontinence, vasomotor symptoms, and pain disorders such as diabetic neuropathy and fibromyalgia.1 In the search for new candidates with improvements in both potency and selectivity, one of the lead compounds in the 1-(3-amino- 2-hydroxy-1-phenylpropyl)indolin-2-one series, WAY-315193 (1), was identified.2
Vasomotor symptoms (VMS), referred to as hot flushes and night sweats, are the most common symptoms associated with menopause, occurring in 60% to 80% of all women following ‘ natural or surgically-induced menopause. VMS are likely to be an adaptive response of the central nervous system (CNS) to declining sex steroids. To date, the most effective therapies for VMS are hormone-based treatments, including estrogens and/or some progestins. Hormonal treatments are very effective at alleviating VMS, but they are not appropriate for all women. It is well recognized that VMS are caused by fluctuations of sex steroid levels and can be disruptive and disabling in both males and females. A hot flush can last up to thirty minutes and vary in their frequency from several times a week to multiple occurrences per day. The patient experiences a hot flash as a sudden feeling of heat that spreads quickly from the face to the chest and back and then over the rest of the body. It is usually accompanied by outbreaks of profuse sweating. It may sometimes occur several times an hour, and it often occurs at night. Hot flushes and outbreaks of sweats occurring during the night can cause sleep deprivation. Psychological and emotional symptoms observed, such as nervousness, fatigue, irritability, insomnia, depression, memory loss, headache, anxiety, nervousness or inability to concentrate are considered to be caused by the sleep deprivation following hot flush and night sweats (Kramer et al., In: Murphy et al., 3rd Int’l Symposium on Recent Advances in Urological Cancer Diagnosis and Treatment- Proceedings, Paris, France: SCI: 3-7 (1992)).
Hot flushes may be even more severe in women treated for breast cancer for several reasons: 1) many survivors of breast cancer are given tamoxifen, the most prevalent side effect of which is hot flush, 2) many women treated for breast cancer undergo premature menopause from chemotherapy, 3) women with a history of breast cancer have generally been denied estrogen therapy because of concerns about potential recurrence of breast cancer (Loprinzi, et al., Lancet, 2000, 356(9247): 2059-2063).
Men also experience hot flushes following steroid hormone (androgen) withdrawal. This is true in cases of age-associated androgen decline (Katovich, et al., Proceedings of the Society for Experimental Biology & Medicine, 1990, 193(2): 129-35) as well as in extreme cases of hormone deprivation associated with treatments for prostate cancer (Berendsen, et al., European Journal of Pharmacology, 2001, 419(1): 47-54. As many as one-third of these patients will experience persistent and frequent symptoms severe enough to cause significant discomfort and inconvenience.
The precise mechanism of these symptoms is unknown but generally is thought to represent disturbances to normal homeostatic mechanisms controlling thermoregulation and vasomotor activity (Kronenberg et al., “Thermoregulatory Physiology of Menopausal Hot Flashes: A Review,” Can. J. Physiol. Pharmacol., 1987, 65:1312-1324).
The fact that estrogen treatment (e.g. estrogen replacement therapy) relieves the symptoms establishes the link between these symptoms and an estrogen deficiency. For example, the menopausal stage of life is associated with a wide range of other acute symptoms as described above and these symptoms are generally estrogen responsive.
It has been suggested that estrogens may stimulate the activity of both the norepinephrine (NE) and/or serotonin (5-HT) systems (J. Pharmacology & Experimental Therapeutics, 1986, 236(3) 646-652). It is hypothesized that estrogens modulate NE and 5-HT levels providing homeostasis in the thermoregulatory center of the hypothalamus. The descending pathways from the hypothalamus via brainstem/spinal cord and the adrenals to the skin are involved in maintaining normal skin temperature. The action of NE and 5-HT reuptake inhibitors is known to impinge on both the CNS and peripheral nervous system (PNS). The pathophysiology of VMS is mediated by both central and peripheral mechanisms and, therefore, the interplay between the CNS and PNS may account for the efficacy of dual acting SRI/NRIs in the treatment of thermoregulatory dysfunction. In fact, the physiological aspects and the CNS/PNS involvement in VMS may account for the lower doses proposed to treat VMS (Loprinzi, et al., Lancet, 2000, 356:2059-2063; Stearns et al., JAMA, 2003, 289:2827-2834) compared to doses used to treat the behavioral aspects of depression. The interplay of the CNS/PNS in the pathophysiology of VMS and the presented data within this document were used to support the claims that the norepinephrine system could be targeted to treat VMS.
Although VMS are most commonly treated by hormone therapy (orally, transdermally, or via an implant), some patients cannot tolerate estrogen treatment (Berendsen, Maturitas, 2000, 36(3): 155-164, Fink et al., Nature, 1996, 383(6598): 306). In addition, hormone replacement therapy is usually not recommended for women or men with or at risk for hormonally sensitive cancers (e.g. breast or prostate cancer). Thus, non-hormonal therapies (e.g. fluoxetine, paroxetine [SRIs] and clonidine) are being evaluated clinically. WO9944601 discloses a method for decreasing hot flushes in a human female by administering fluoxetine. Other options have been studied for the treatment of hot flashes, including steroids, alpha- adrenergic agonists, and beta-blockers, with varying degree of success (Waldinger et al., Maturitas, 2000, 36(3): 165-168).
It has been reported that α2-adrenergic receptors play a role in thermoregulatory dysfunctions (Freedman etal., Fertility & Sterility, 2000, 74(1): 20- 3). These receptors are located both pre- and post-synaptically and mediate an inhibitory role in the central and peripheral nervous system. There are four distinct subtypes of the adrenergicα2 receptors, i.e., are 2A, O2B, 0.2c and α2D (Mackinnon et al., TIPS, 1994, 15: 119; French, Pharmacol. Ther., 1995, 68: 175). It has been reported that a non-select 2-adrenoceptor antagonist, yohimbine, induces a flush and an 2-adrenergic receptor agonist, clonidine, alleviates the yohimbine effect (Katovich, et al., Proceedings of the Society for Experimental Biology & Medicine, 1990, 193(2): 129-35, Freedman et al., Fertility & Sterility, 2000, 74(1): 20-3). Clonidine has been used to treat hot flush. However, using such treatment is associated with a number of undesired side effects caused by high doses necessary to abate hot flash described herein and known in the related arts.
Patent
https://www.google.com/patents/WO2005097744A1?cl=en
invention relates to phenylaminopropanol derivatives, compositions containing these derivatives, and methods of their use for the prevention and treatment of conditions ameliorated by monoamine reuptake including, inter alia, vasomotor symptoms (VMS), sexual dysfunction, gastrointestinal and genitourinary disorders, chronic fatigue syndrome, fibromylagia syndrome, nervous system disorders, and combinations thereof, particularly those conditions selected from the group consisting of major depressive disorder, vasomotor symptoms, stress and urge urinary incontinence, fibromyalgia, pain, diabetic neuropathy, and combinations thereof.
EXAMPLE 101 : 7-f luoro-1 -[(1 S,2R)-1 -(3-fluorophenyl)-2-hydroxy-3- (methylamino)propyl]-3,3- dimethyl-1 ,3-dihydro-2H-indol-2-one hydrochloride
[0538] Step 1 : A mixture of 7-fluoro-3, 3-dimethyl-1 ,3-dihydro-2H-indol-2-one (EXAMPLE 99, step 5, 1.0 g; 5.58 mmol) and sodium tert-butoxide (1.0 g, 11.16 mmol) in dry dichloromethane (15 mL) was stirred at room temperature under nitrogen for 20 minutes. Titanium isopropoxide (2.0 mL, 6.70 mmol) was added to a solution of [(2R,3R)-3-(3-fluorophenyl)oxiran-2-yl]methanol (EXAMPLE 47, step 3, 844 mg, 5.02 mmol) in dry dichloromethane (6 mL) and stirred for 20 minutes at room temperature. The epoxide complex was added drop-wise to the mixture of tert- butoxide and allowed to stir for 4 days. The reaction mixture was poured into a 2N aqueous solution of hydrochloric acid and diluted with ethyl acetate. The layers were separated, and the organic layer was washed with water and brine. The organic layer was dried over anhydrous sodium sulfate, filtered and concentrated in vacuo to give 2.0 g of crude product. The crude product was purified via Isco chromatography (RediSep, silica, gradient of 0% to 100% ethyl acetate in hexane) to yield 600 mg (31 %) of (2S,3S)-7-Fluoro-1 -[1 -(3-fluoro-phenyl)-2,3-dihydroxy-propyl]-3,3-dimethyl- 1 ,3-dihydro-indol-2-one as an oil. MS (ESI) m/z 348 ([M+Hf).
[0539] Step 2: In an analogous manner to EXAMPLE 1 , step 2 (2S, 3S)-toluene- 4-sulfonic acid 3-(7-fluoro-3,3-dimethyl-2-oxo-2,3-dihydro-indol-1 -yl)-3-(3-fluoro- phenyl)-2-hydroxy-propyl ester was prepared from (2S,3S)-7-fluoro-1-[1-(3-fluoro- phenyl)-2,3-dihydroxy-propyl]-3,3-dimethyl-1 ,3-dihydro-indol-2-one. MS (ESI) m/z 502 ([M+Hf).
10 A. Kende, Synth. Comm. 1 : 12 (1982) [0540] Step 3: In an analogous manner to EXAMPLE 5 7-fluoro-1-[(1S,2R)-1-(3- fluorophenyl)-2-hydroxy-3-(methylamino)propyl]-3,3- dimethyl-1 ,3-dihydro-2H-indol- 2-one hydrochloride was prepared from (2S,3S)-toluene-4-sulfonic acid 3-(7-fluoro- 3,3-dimethyl-2-oxo-2,3-dihydro-indol-1-yl)-3-(3-fluoro-phenyl)-2-hydroxy-propyl ester. MS (ESI) m/z 360 ([M+Hf), HRMS: calcd for C20H22F2N2O2 + H+, 361.17221; found (ESI, [M+Hf), 361.1719.
PATENT
https://www.google.com/patents/WO2008024492A2?cl=en
Scheme A
cat.
Scheme B
Acetylbromidθ
EtOH
Scheme C (cat.)
1. TSCI1 TEA1 CH3CN A + B dlbutyltln oxide cat. 1. 33 wt% CH3NH2In EtOH. MeOH
1.35 equiv. 2. NaOH aq., toluene 2. 5N HCI in IPA, toluene
HCI salt
Example 1 :
Preparation of (1 R3/?)-3-(3-fluoropheπv0-2-(hvdroxymethv0oxirane
[0117] A thoroughly dried 5-L jacketed reactor was equipped with a mechanical stirrer, a 500-mL addition funnel, a temperature probe and a nitrogen inlet. The reactor was charged with D-(-)-DIPT (13.0 g 46 mmol), 4-A 5-μm molecular sieves (90 g) and dichloromethane (4.00 L) and then it was purged with nitrogen. The contents of the reactor were cooled to -15°C. Titanium isopropoxide (12.19 g, 43 mmol) was added rapidly to the reaction mixture via the addition funnel and the reaction mixture was further cooled to -200C. A solution of allylic alcohol (127 g, 0.854 mol) in CH2CI2 (380 mL) was added to the reaction mixture via the addition funnel at a rate to keep the temperature in the reactor below -200C. The resulting mixture was allowed to stir at -200C for 10 minutes. A solution of TBHP in CH2CI2 (4.5 M, 380 mL, 1.71 mol) was added to the reaction mixture via the addition funnel at a rate to maintain the temperature below -200C and above -25°C (addition rate 7 ml/min). The reaction mixture was stirred at -200C for 4 hours. Reaction progress was monitored by HPLC: an aliquot was drawn out of the reactor and diluted with MeCN-water. The reaction was deemed complete when the amount of the starting olefin fell below 1 %.
[0118] The reaction mixture was transferred from the reactor into a 6-L flask containing a solution of FeSCU x 7H2O (356 g, 1.28 mol), citric acid monohydrate (93 g, 0.39 mol) and de-ionized water (to the total volume of 1.0 L) chilled in an ice bath to 00C. The rate of transfer was adjusted to maintain the temperature of the mixture below 100C. The flask with the resulting mixture was equipped with a mechanical stirrer and the mixture was stirred for 25 minutes.
[0119] The organic layer was separated and filtered through a pad of Celite. The aqueous phase was extracted with MTBE (2 x 300 mL). Combined organic solutions were cooled to 00C in an ice bath. A 30% solution of NaOH (100 mL) in brine (prepared by dissolving 5 g of NaCI in a solution of NaOH (30.0 g) in 90 mL of water) was cooled in an ice bath to 00C and then added to the combined organic phases. The resulting mixture was stirred rapidly for 2 hours at 00C. Water (400 mL) was added to the mixture and the layers were separated. The aqueous layer was extracted with MTBE (2 x 250 mL). The combined organic layers were dried with Na2SO4 (300 g), the drying agent was filtered off through a paper filter and the filtrate was evaporated on rotary evaporator. The oily residue was mixed with 700 mL of toluene and the solvent was removed on a rotary evaporator. The residue after evaporation: Weight 125.9 g.
HPLC purity (area% 215 nm): 94%, impurities: toluene (3.1 %), starting olefin (1.0%), 3 unknown impurities (< 0.7% each).
1H NMR (CDCI3). Impurities: toluene (1.7 weight%), DIPT (1.1 weight%), t-BuOH (0.4 weight%).
Example 2: Preparation of 7-fluoro-1-r(1 S,2SM-(3-fiuorophenv[)-2,3-dihvdroxypropyπ-3.3- dimethylindolin-2-one
[0120] 7-Fluoro-3,3-dimethyl-1 ,3-dihydro-2H-indol-2-one (60 g, 335 mmol) was mixed under nitrogen with anhydrous dimethylformamide (DMF) (10.8 ml_). To the resulting viscous solution, cooled to 5-7°C, was added via syringe a solution of LiHMDS in THF (1 M in THF, 140 ml, 140 mmol) at a rate to keep the reaction mixture temperature below 7-1O0C (addition of the first 60 ml was very exothermic, later the rate of addition could be increased). The resulting purple-red clear solution was allowed to warm up to 100C.
[0121] In a separate flask, [(2/?,3fl)-3-(3-fluorophenyl)oxiran-2-yl]methanol (59.1 g, 352 mmol, 1.05eq.) was dissolved in 600 ml_ of anhydrous THF, the flask was purged with nitrogen and the solution was cooled to 5-7°C. Titanium isopropoxide (104 ml, 100 g, 584 mmol) was added to the epoxide solution dropwise via syringe maintaining the temperature in the 7-12°C range. The resulting bright-yellow solution was stirred for 40 minutes, allowing it to warm up to room temperature.
[0122] The contents of the second flask, the epoxy-titanium solution, were transferred to the solution of the indolinone salt via cannula maintaining the temperature of the mixture below 15°C. The resulting mixture was stirred at room temperature. The reaction progress was monitored by HPLC: after 20 hours, about 17 area% of indolinone was left, while no epoxide was detectable. Additional amount of the epoxide-titanium isopropoxide complex was prepared from epoxide (9.85 g, 58.4 mmol) and titanium isopropoxide (17.3 ml, 16.6 g, 58.4 mmol) in THF (100 mL) as described above and added slowly to the reaction mixture. The mixture was kept at room temperature for 20 hours longer, at which point HPLC analysis showed 4 area% of the unreacted indolinone and no detectable amount of the epoxide.
[0123] The reaction mixture was transferred into 1.80 L of cold (00C) 2 M aqueous HCI solution (Exotherm. The rate of addition was adjusted to keep the temperature below 15°C). The resulting clear solution was extracted with MTBE (3 x 800 ml), the combined organic phase were washed with brine (800 ml), dried over magnesium sulfate and filtered through a pad of magnesol. The filtrate was evaporated, diluted with toluene (600 ml), and evaporated again to remove maximum amount of solvents. The residue (133 g) contained a sufficiently pure product to be used in the next step without further purification.
HPLC purity (area% at 215 nm): 95%, impurities: indolinone (3.5%).
1H NMR (CDCI3). Impurities: residual solvents (DMF, toluene, MTBE).
Example 3:
Preparation of 7-fluoro-1 -f(SH3-f luorophenylUf S)-oxiran-2-yl)methv0-3.3- dimethylindolin-2-one
[0124] A 2-L round bottom flask, equipped with a mechanical stirrer, a 100-mL addition funnel, a temperature probe and a nitrogen inlet, was charged with a solution of 7-fluoro-1 -[(1 S,2S)-1 -(3-fluorophenyl)-2,3-dihydroxypropyl]-3,3- dimethylindolin-2-one (50.0 g, 144 mmol) in CH2CI2 (500 mL), triethylamine (62 mL, 0.433 mol), solid dibutyltin oxide (716 mg, 2.9 mmol) and DMAP (1.74 g, 14.4 mmol). Tosyl chloride (28.23 g, 148 mmol) was dissolved in CH2CI2 (60 mL) and the solution was added slowly to the reaction mixture (addition rate 5.6 mL/min). Temperature range 200C to 23°C. The reaction flask was cooled in an ice water bath during the addition to keep the temperature below 25°C. After the addition was finished, the bath was removed and the reaction mixture was stirred at room temperature. The reaction progress was monitored by HPLC.
[0125] After about one hour, the amount of the diol fell below 10%. A solution of NaOH, prepared by diluting 72 mL of 10 M aqueous NaOH with 360 mL of deionized water, was added rapidly to the reaction mixture. Solid Bu4N+ CP hydrate (2.05 g, 7.2 mmol) was added and the reaction mixture was stirred rapidly at room temperature. The progress of the epoxide closure was monitored by HPLC. After 2 hour, all fosylate was consumed.
[0126] The layers were separated. The aqueous layer was extracted with 100 mL of CH2CI2. Combined organic solutions were washed with 100 mL portions of 0.5 M aqueous HCI until pH of the washes fell below 5, then with 50 mL of 0.5 M aqueous NaOH, then it was dried with Na2SO-I. The solution was gravity-filtered through a pad of Silica gel (150 g, thickness of the pad 5 cm) prepared in a glass filter funnel. The drying agent and the pad were washed with dichloromethane. The washing continued until no more epoxide was detectable in the eluent (by HPLC). The filtrate was evaporated to dryness on rotary evaporator (room temp. bath).
[0127] The residue after evaporation: weight 42.6 g. HPLC purity 82%, impurities: bis-tosylate (12%), diol (2.5%), indolinone (2.4%). The crude intermediate was used in the next step without further purification.
Example 4:
Preparation of 7-fluoro-1-iY1 S.2fl)-1-(3-fluorophenyl)-2-hvdroxy-3-(methyl amino)propyπ-3,3-dimethylindolin-2-one hydrochloride
[0128] The residue from the previous step (42.6 g) was dissolved in ethanol (160 mL) and the solution was placed into a 1-L round bottom flask equipped with a mechanical stirrer and a temperature probe. Aqueous methylamine (40 weight%, 240 mL, 2.74 mol) was added to the solution and the resulting suspension was stirred at room temperature. The reaction was monitored by HPLC. After 15 hours, the amount of the epoxide fell below 1%. Ethanol was removed on rotary evaporator (bath temperature 27°C). The residue was mixed with MTBE (250 mL) and water (100 mL). The layers were separated. The aqueous layer was extracted with 50 mL of MTBE. Combined organic solutions were washed with 100 mL of water. Small amount of brine was added to speed up the phase separation. The resulting organic solution was extracted with aqueous HCI (200 m L of 2 M solution, then 50 mL of 1 M solution). Combined acidic extracts were washed with 50 mL of MTBE.
[0129] MTBE (200 mL) was added to the aqueous solution. Aqueous NaOH (10 M solution^ 50 mL, 500 mmol) was added to the bi-phasic mixture. The mixture was shaken and the layers were separated. The aqueous layer was extracted with MTBE (100 mL). Combined organic solutions were dried with Na2SO4 (75 g). The drying agent was filtered off and the filtrate was evaporated in vacuum. [0130] The residue (38.0 g) was mixed with 70 ml_ of ethanol and the solvent was removed on rotary evaporator. The residue was re-dissolved in 100 ml_ of ethanol. With magnetic stirring, 2 M HCI in Et2O (57 mL, 114 mmol) was added to the solution. The acidity of the solution was checked by placing a drop of the solution on a wet pH paper to ensure the solution is strongly acidic. The resulting solution was seeded with crystals of 7-fluoro-1-[(1 S,2R)-1-(3-fluorophenyl)-2-hydroxy-3-(methyl amino) propyl]-3,3-dimethyl-1 ,3-dihydro-2H-indol-2-one hydrochloride salt which caused slow crystallization of the salt in about 30 minutes. The slurry was stirred at room temperature for 1 hour.
[0131] The reaction flask was placed into a 0°C bath (equipped with thermostat) and the slurry was stirred magnetically for 21 hours. The cold slurry was filtered through a paper filter. The solid was washed with a 1 :1 mixture of EtOH-Et2O (70 mL) and then was dried on the filter in a stream of air for 2 hours.
[0132] Weight of the crystals 29.7 g (54% from theoretical yield calculated from the diol).
HPLC purity (area% at 215 nm): 98.2%, impurities (relative retention time): 1.05
(0.46%), 0.98 (0.42%), 1.07 (0.15%), 2.05 (0.14%).
Enantiomeric purity 99.4% ee. m.p. 209.5-211.20C.
[a)? = -,10.7°.
1H NMR (D2O, 400 MHz), δ: 7.45-7.30 (m, 3H), 7.16-6.97 (m, 4H), 5.53-5.30 (2H1 broad m), 3.35-3.24 (2H, broad m), 2.82 (s, 3H), 1.41 (s, 3H), 1.27 (broad s, 3H).
Impurities: ethanol (0.3 weight%).
ES+ MS, m/z 361 (MH+).
Anal, calc’d for C20H23CIF2N2O2 (396.9): C, 60.53; H, 5.84; N, 7.06. Found: C, 60.43;
H, 5.69; N, 6.84.
Sn content: 3 ppm.
Example 5:
Preparation of 7-fluoro-3. 3-dimethyl-oxindole via selective C-methylation of 7- fluoro-oxindole [0133] To a stirred slurry of potassium tert-butoxide (185 g, 1.65 mol) in tθtrahydrofuran (1350 mL) was added 7-fluoro-oxindole (50 g, 0.33 mol) and copper (I) bromide-dimethyl sulfide complex (7 g, 0.033 mol). Methyl iodide (150 g, 1.06 mol.) was added to the mixture at 5-100C. The reaction mixture was stirred at 20- 25°C for 1 hour. 10% NH4CI (1000 mL) was added to the reaction mixture. The two layers were separated. The organic layer was concentrated via vacuum distillation at 25-400C to reach a volume of 250 mL. The aqueous layer is extracted with tert- butyl methyl ether (2 x 500 mL). The concentrated organic layer and tert-butyl methyl ether extraction layers were combined and washed with 15% NaCI (250 mL). The organic solution was filtered through silica gel (100 g). Heptane (1250 mL) was added to the filtrate. The mixture was concentrated under atmosphere at 60-950C to reach a volume of 700 mL. The concentrate was cooled to 0-50C from 85-95°C over 2 hours to crystallize. Solid was filtered, washed with heptane (100 mL), and oven- dried to give 41 g (69.4%) of a beige solid 7-fluoro-3, 3-dimethyl-oxindole, 92% w/w purity by HPLC.
Example 6:
Preparation of 3-(3-fluoro-phenyl)-prop-2-en-1-ol
[0134] A 5-L reactor equipped with a mechanical stirrer, thermocouple, and nitrogen inlet was charged with MeOH (1.40 L) and 3-fluorocinnamic acid (0.20 kg, 1.20 mol). To the slurry charged p-TSA (0.023 kg, 0.120 mol) at 200C to 25°C. The suspension was refluxed at 65°C to 68°C for 3-5 hours. The mixture was concentrated via atmospheric distillation to reach a volume of 700 mL. Methanol was then chased off by adding toluene (1.8 L) and was further concentrated to a solution (about 1.5 L). The reaction mixture then washed successively with 5% aqueous NaHCU3 (1.5) and water (1.5 L). The organic mixture was concentrate via atmospheric distillation to a minimum volume of 500 mL. HPLC analysis indicates that the solution strength 53.5% KF 0.17%, 98.8% area HPLC purity of the product. [0135] A 3-L reactor equipped with a mechanical stirrer, thermocouple, and nitrogen inlet was charged with diisobutylaluminum hydride 25% w/w in Toluene (1.56 kg, 1 ,85 L12.75 mol). The solution was cooled to -25°C. To the reactor was then added using FMI pump a solution of 3-(3-Fluoro-phenyl)-acrylic acid methyl ester (0.41 kg, 0.40 L, 1.20 mol) in toluene while maintaining the internal temperature between -15°C to -8°C. The reaction mixture was stirred at -15 to -8°C for 60 minutes. The reaction mixture was then quenched in a 5-L reactor into a solution of concentrated HCI (0.40 L, 0.48 kg; 4.87 mol) in water (0.75 kg) maintaining internal temperature at 400C to 45°C. The biphasic mixture was separated. The lower aqueous layer was washed with Toluene (0.34 kg, 0.40 L). The combined organic phase was successively washed with a 5% aqueous solution of sodium bicarbonate (0.7 L) and 10% brine (0.7 L). The*organic solution was concentrated via atmospheric distillation to reach a volume of 500 mL. HPLC analysis indicates that the solution strength is 53%, 169 g (93% Y), Al: 9 ppm, KF: 0.04%, 99% area HPLC purity of the allylic alcohol.
Example 7:
Preparation of r3-(3-fluoro-phenyl)-oxiranyπ-methanol
[0136] A 3-L reactor equipped with a mechanical stirrer, thermocouple, and nitrogen inlet was charged with toluene (200 mL) and pre-activated molecular sieves powder (4A, 70 g). The resultant slurry was cooled to — 35°C. To the reactor was then added a solution of D-(-)-diisopropyl tartrate (21.6 g, 92.0 mmol) in toluene (25 mL), followed by addition of titanium (IV) isopropoxide (18.7 g, 65.7 mmol). The temperature of the reaction mixture was maintained between -300C to -400C during the addition. To the reactor was then charged with a solution of 3-(3-fluoro-phenyl)- prop-2-en-1-ol (100 g, 657 mmol) in toluene (490 mL) while maintaining the temperature of the reaction mixture between -300C to -400C. The reaction mixture was stirred at -35°C for 30 minutes. To the reactor was then added a solution of 5.5 M tert-butyl hydroperoxide in decane (240 mL, 1310 mmol) while maintaining the temperature of the reaction mixture between -300C to -400C. The reaction mixture was stirred at -35°C for 6 hours, followed by 8 hours at -200C. The reaction mixture was warmed to room temperature and filtered through a thin layer of celite. The filter cake was washed with toluene (2 x 100 ml_). The combined filtrate and washes were cooled to 00C and a solution of 30% sodium hydroxide saturated with sodium chloride (100 mL) was then added. The reaction mixture is stirred at 0°C for 2 hours. To the reaction mixture was then added a solution of sodium metabisulfite (69 g) and citric acid (50 g) in water (600 mL). The biphasic mixture was stirred at room temperature for 1 hour and the phases were separated. The organic phase was successively washed with a 5% sodium bicarbonate (500 mL) and 10% brine (500 mL). The organic solution was then concentrated under vacuum to reach a volume of 500 mL. HPLC analysis indicates that the solution contains 90.3 g (81.7%) of the epoxy alcohol product.
Example 8:
Preparation of r3-(3-fluoro-pheny[)-oxiranyll-methanol
[0137] A 1-L reactor equipped with a mechanical stirrer, thermocouple, and nitrogen inlet is charged with toluene (140 mL) and pre-activated molecular sieves powder (4A, 14 g). The resultant slurry was cooled to -35°C. To the reactor is then added a solution of D-(-)-diisopropyl tartrate (4.31 g, 18.4 mmol) in toluene (20 mL), followed by addition of titanium (IV) isopropoxide (3.74, 13.1 mmol). The temperature of the reaction mixture was maintained between -300C to -400C during the addition. To the reactor is then charged with a solution of 3-(3-fluoro-phenyl)- prop-2-en-1 -ol (20 g, 131 mmol) in toluene (80 mL) while maintaining the temperature of the reaction mixture between — 300C to — 400C. The reaction mixture is stirred at -35°C for 30 minutes. To the reactor is then added a solution of cumene hydroperoxide (88% purity, 45.5 g, 263 mmol) while maintaining the temperature of the reaction mixture between -300C to -400C. The reaction mixture is stirred at – 35°C for 16 hours. A solution of 30% sodium hydroxide saturated with sodium chloride (20 mL) is charged while maintaining temperature of the reaction mixture below -200C. To the reaction mixture is then added a solution of sodium metabisulfite (13.7 g) in water (60 mL) while maintaining the reaction mixture temperature below 25°C. The biphasic mixture is stirred at room temperature for 1 hour. To the reaction mixture is added Celite (70 g) and the mixture is filtered. The filter cake is washed with toluene (2 x 50 mL). The filtrate is successively washed with 5% sodium bicarbonate (100 mL) and 10% brine (100 mL). The organic solution is then concentrated under vacuum to reach a volume of 100 mL.
Example 9:
Preparation of r3-(3-fluoro-phenyl)-oxiranvn-methanol
[0138] A 5-L jacketed reactor equipped with a mechanical stirrer, addition funnel, temperature probe, and nitrogen inlet. All equipment must be rigorously dry. The reactor was charged with D-(-)-DIPT (10.0 mL, 11.0 g, 46 mmol), 4-A, 5-um molecular sieves (49.3 g), dichloromethane (3 L). The flask was purged with nitrogen. The contents of the flask were cooled to 00C. Titanium isopropoxide (9.34 g, 9.73 mL was added rapidly to the flask via an addition funnel. The reaction mixture was cooled to -200C. A solution of allylic alcohol (100 g, 0.657 mol) in CH2CI2 (300 mL) was added to the reaction mixture via an addition funnel while keeping the temperature below -200C.
[0139] The reaction mixture was stirred at -200C for 10 minutes. A solution of TBHP in CH2CI2 (188 mL, 5.7 M) was added to the reaction mixture via an addition funnel while maintaining the temperature between -200C to -25°C. The reaction mixture was stirred at -200C for 4 hours. Reaction progress was monitored by HPLC. A solution prepared from FeSO4 x 6H2O (217 g, 0.79 mol), citric acid monohydrate (72 g, 0.39 mol) and de-ionized water to the total volume of 660 mL, was chilled in an ice bath to 00C.
[0140] The reaction mixture was quenched into the chilled solution of FeSO4 and citric acid in water. The mixture was stirred for 30-60 minutes. The organic layer was checked for the presence of organic peroxides. The layers were separated. The aqueous phase was extracted with MTBE (2 x 200 mL). Combined organic solutions were cooled to 00C in an ice bath. [0141] A 30% solution of NaOH (60 ml_) in brine (prepared by dissolving 5 g of NaCI in a solution of NaOH (30.0 g) in 90 mL of water) was cooled in an ice bath to 00C and then added to the combined organic phases. The resulting mixture was stirred rapidly for 1-2 hours at 00C. Water (300 mL) was added to the mixture. The two layers were separated. The aqueous layer was extracted with MTBE (2 x 250 mL). The combined organic layers were evaporated on. rotary evaporator. HPLC analysis indicates that the solution contains 90.5 g (81.5%) of the epoxy alcohol product with chiral purity 95.6/4.4 and chemical purity 96.5 area %.
Example 10:
Preparation of 7-fluoro-1- f(1S, 2S)-1-(3-fluorophenv0-2,3-dihvdroxypropyl1-3,3- dimethvM . 3- dihvdro-2H-indol-2-one
[0142] To a suspension of 7-fluoro-3, 3-dimethyl-oxindole (35 g, 0.195 mol) in N, N dimethylformamide (36 g, 0.49 mol) and toluene (200 mL) was added (1 M / toluene) lithium bis (trimethylsilyl) amide (585 mL, 0.585 mol). To the resulting mixture was added a solution of (20% / toluene) [3-(3-fluoro-phenyl)-oxiranyl]-methanol (210 g, 0.253 mol) and titanium (IV) isopropoxϊde (72g, 0.253 mol) in toluene (300 mL) at 5- 100C. The reaction mixture was stirred for 3-4 hours at 40-45°C. To the reaction mixture was added 37% HCI (460 g, 2.34 mol) and water (500 mL) at 20-250C to give a bi-phasic mixture. The organic layer was separated. The aqueous layer was extracted with toluene (1000 mL). The combined organic layers were washed with 1N NaOH (200 g), and then with 10% NaCI (200 g). The organic layer was concentrated via atmospheric distillation at 100-1100C to a volume of (1800 mL). The concentrated solution was filtered through silica gel (150 g). The silica gel plug was rinsed with ethyl acetate (850 mL). The filtrate was concentrated via atmospheric distillation at 80-1100C to reach a volume of (250 mL). The concentrate was cooled to 0-50C from 100-1 100C over 4 hours to crystallize. Solid was filtered, washed with heptane (150 mL), and oven-dried to give 50.6 g (74.7%) of a beige solid, 97.4% w/w purity by HPLC.
Example 11 : Preparation of 7-fluoro-1-rf1S.2R)-1-(3-ftuorophenyl)-2-hvdroxy-3-fmethyl amino) propyl1-3.3-dirnethyl-1.3-dihvdro-2H-indol-2-one:
[0143] To the solution of the diol (52 g, 0.144 mol) in MeCN (500 mL) was added Bu2SnO (0.39 g, 1.44 mmol) and TsCI (28.8 g, 0.151 mol). To the resulting solution was added Et3N (29 g, 0.288 mol) dropwise at 0-50C. The reaction was stirred for 1 hour at 0-50C until the tosylation was complete by HPLC. To the reaction containing the mono-tosylate was added a solution of NaOH (58 g, 0.72 mol) in water (400 mL) at 00C. At the end of the epoxide formation, toluene (800 mL) and NaCI (25 g) in water (150 mL) were added to form a bi-phasic reaction mixture. The two layers were separated. The organic layer was washed with 37% w/w HCI (56 g) in water (256 mL) followed by NaCI (50 g) in water (300 mL). The organic layer was diluted with toluene (700 mL) and concentrated to a volume of about 900 mL. The resulting concentrated solution was filtered through a silica gel (200 g) plug. The silica gel plug was eluted with toluene (1.5 L). The combined filtrate was concentrated under vacuum to about 300 mL. Methylamine in EtOH (33 weight %, 245 mL, 2.0 mol) and Ca(OTf)2 (15 g, 43 mmol) were added to the toluene solution. The reaction mixture was stirred at 20-250C for 12 hours then concentrated via vacuum distillation to about 200 mL. MTBE (500 mL) and water (500 mL) were added. The two layers were separated. 37% w/w HCI (160 g,) in water (340 g) was added to the organic layer. Stirred and the two layers were separated. The aqueous organic layer was washed with MTBE (500 mL). To the acidic aqueous layer was charged MTBE (500 mL) then the mixture was cooled to 0-50C and basified with NaOH (50% w/w, 150 g, 100 mL). Reaction mixture was stirred for 20 minutes then the two layers were separated. The organic layer was washed with 15% NaCI (170 mL) then concentrated to about 250 mL via atmospheric distillation. To the MTBE concentrate was added EtOH (2B) (150 mL) followed by HCI (5.7 N in EtOH, 45 mL, 0.26 mol). The mixture was stirred at 20 to 25°C for a minimum of 2 hours and then cooled to 0 to 5°C over 1 hour. The suspension was filtered and washed with MTBE (50 mL) to give 26 g (45%) of an off-white solid.
Example 12:
Preparation of (2EKH3,5-difluorophenvQprop-2-en-1-ol [0144] A 5-L reactor equipped with a mechanical stirrer, thermocouple, and nitrogen inlet was charged with MeOH (1.40 L), 3, 5-difluorocinnamic acid (0.20 kg, 1.09 mol) and p-TSA (0.0207 kg, 0.109 mol) at 200C to 25°C. The suspension was refluxed at 65°C to 68°C for 4-6 hours. The mixture was concentrated via atmospheric distillation to reach a volume of about 700 mL. Methanol was then chased off by adding toluene (1.8 L) and was further concentrated to a solution (about 1.5 L). The reaction was cooled to 500C to 55°C then washed successively with 5% aqueous NaHCO3 (1.5 L) and water (1.5 L). The organic mixture was concentrated via atmospheric distillation to a minimum volume of about 1.5 L. KF 0.17%.
[0145] A 3-L reactor equipped with a mechanical stirrer, thermocouple, and nitrogen inlet was charged with diisobutylaluminum hydride 25% w/w in toluene (1.42 kg, 1.68 L, 2.31 mol). The solution was cooled to -25°C. To the reactor was then added using FMI pump a solution of 3-(3, 5-difluoro-phenyl)-acrylic acid methyl ester (1.4 L, 1.09 mol) in toluene while maintaining the internal temperature between -15°C to -8°C. The reaction mixture was stirred at that temperature for 60 minutes then quenched into a 5-L reactor with a solution of concentrated HCI (0.40 L, 0.48 kg; 4.87 mol) in water (0.70 kg) while maintaining the internal temperature at 400C to 45°C. The biphasic mixture was separated. The lower aqueous layer was washed with toluene (0.34 kg, 0.40 L). The combined organic phase was successively washed with a 5% aqueous solution of sodium bicarbonate (0.70 L) and 10% brine (0.70 L). The organic solution was concentrated via atmospheric distillation to reach a volume of 0.386 Kg, about 500 mL. HPLC analysis indicates that the solution contains 170 g, 91% yield of (2E)-3-(3,5-difluorophenyl)prop-2-en-1-ol. Al: 1 ppm, KF: 0.12%, 99.8% area HPLC purity.
Example 13:
Preparation of r(2ff,3/?)-3-(3.5-difluorophenyl)oxiran-2-vH-methanol [0146] A 3-L reactor equipped with a mechanical stirrer, thermocouple, and nitrogen inlet was charged with toluene (100 mL) and pre-activated molecular sieves powder (4A, 70 g). The resultant slurry was cooled to -35°C. To the reactor was then added a solution of D-(-)-diisopropyl tartrate (19.3 g, 0.082 mol) in toluene (25 mL), followed by addition of titanium (IV) isopropoxide (16.7 g, 0.059 mol). The temperature of the reaction mixture was maintained between -300C to -400C during the addition. To the reactor was then added a solution of 3-(3,5-difluoro-phenyl)- prop-2-en-1-ol (100 g, 0.588 mol) in toluene (250 mL) while maintaining the temperature of the reaction mixture between -300C to -400C. The reaction mixture was stirred at -35°C for 30 min. To the reactor was then added a solution of 5.5 M tert-butyl hydroperoxide in decane (173 g, 1.18 mol) while maintaining the temperature of the reaction mixture between -300C to -400C. The reaction mixture was stirred at -35°C for 6 hours, followed by 8 hours at -25°C. The reaction mixture was warmed to room temperature and filtered through a thin bed of celite (25 g). The filter cake was washed with toluene (2 x 200 mL). The combined filtrate and washes were cooled to 00C and a solution of 30% sodium hydroxide saturated with sodium chloride (100 mL) was then added. The reaction mixture is stirred at 00C for 3 h. To the reaction mixture was then added a solution of sodium metabisulfite (61.5 g) and citric acid (44.5 g) in water (600 mL). The biphasic mixture was stirred at room temperature for 1 hour and the phases were separated. The organic phase was successively washed with a 5% sodium bicarbonate (500 mL) and 10% brine (500 mL). The organic solution was then concentrated under vacuum to reach a volume of about 400 mL. A small portion of the concentrate was taken out for seed generation at 25-300C. To the suspension was then charged 3 volume parts of heptane (300-400 mL). The mixture was cooled to 5-100C then filtered to give 71.3 g, 65% yield of [(2R,3R)-3-(3,5-difluorophenyl)oxiran-2-yl]-methanol as an off-white solid with chiral purity 94 %ee, mp: 48-50°C.
Example 14:
Preparation of 1-r(7S,2S>-1-(3,5-difluorophenyl)-2,3-dihvdroxypropyH-7-fluoro- 3,3-dimethyl-1,3-dihvdro-2H-indol-2-one [0147] To a suspension of dimethyl oxidole (68 g of 74 % strength crude, 280 mmol) in DMF (51 g, 700 mmol.) and toluene (200 rriL), a toluene solution of (Me3Si)2NLi (840 ml_, 1 M, 840 mmol) was added dropwise while keeping the mixture below 100C to give a dark solution. A solution of epoxy alcohol (76 g of 85% strength, 350 mmol) and Ti(OiPr)4 (103 g, 360 mmol) in toluene (400 ml_) was added to the above dark solution at below 100C. The reaction mixture was stirred for 20 hours at 200C before cooling to 00C. A solution of HCI (660 g, 37% in water) in water (750 g) was added at below 200C to give a bi-phasic mixture. The two layers were separated. The organic layer was washed with NaOH (400 ml_, 0.7 N in water, 280 mmol), and brine (230 g). The organic layer was filtered through a silica gel (150 g) plug. The silica gel plug was rinsed with. EtOAc (1100 ml_). The filtrate was concentrated in vacuo at 50°C to a volume of 240 ml_. This concentrate was diluted with CH3CN (300 ml_) to give 1-[(1S,2S)-1-(3,5-difluorophenyl)-2,3-dihydroxypropyl]- 7-fluoro-3,3-dimethyl-1 ,3-dihydro-2H-indol-2-one as a CH3CN solution, 431 g of a 20.8% strength solution, yield: 88%.
Example 15:
Preparation of 1-r(7S..g/?)-1-(3,5-d.fluorophenvπ-2-hvdroxy-3-(methylamϊno) propyπ-7-fluoro-3,3-dimethyl-1,3-dihvdro-2H-indol-2-one
[0148] To the solution of 1-[( 7S,2S)-1 -(3,5-difluorophenyl)-2,3-dihydroxypropyl]-7- fluoro-3,3-dimethyl-1 ,3-dihydro-2H-indol-2-one in acetonitrile (394 g of 20.8% strength solution, 224 mmol) at 200C, tosyl chloride (56g, 269 mmol) and Bu2SnO (1.4 g 5.6 mmol) were added. The reaction mixture was cooled to 5°C, and then Et3N (45 g, 448 mmol) was added dropwise. The reaction mixture was stirred for about 1 hour at 200C until tosylation is complete.
[0149] A solution of NaOH (90 g of 50% w/w solution in water, 1120 mmol) in water (492 g) was added at 5°C. The reaction mixture was stirred for 1 hour. Toluene (1312 ml_) was added to the reaction mixture to give a bi-phasic mixture. The organic layer was separated and washed with HCI (44 g of 37% solution in water, 448 mmol) in water (32OmL) then with brine (400 mL). The organic layer was then concentrated to a volume of (400 mL) under vacuum keeping the temperature below 500C. The concentrate was diluted with toluene (1120 mL). The resulting solution was filtered through a silica gel (320 g) plug. The silica gel plug was eluted with toluene (2400 ml_). The filtrate was concentrated to a volume of 400 ml_ in vacuo keeping the temperature below 500C.
[0150] Methanol (1200 mL) was charged to the mixture then concentrated down to about 400 mL in vacuo while keeping temperature below 500C. To the concentrate was added methanol (1600 mL) and methylamine (252 g of 33 wt % solution in ethanol, 2688 mmol.). The reaction mixture was stirred for 20 hours at 400C until the aminolysis is complete. The mixture was concentrated down to about 400 mL in vacuo. Toluene (960 mL) was added to the concentrate. The mixture was concentrated in vacuo down to about 400 mL.
[0151] HCI (40 g of 5N solution in isopropanol, 224 mmol.) in IPA was added to the mixture. Stirred at 200C for 2 hours. The resulting slurry was filtered then dissolved in acetone (1230 mL) at 400C. Heptane (1640 mL) was added. The resulting solution was concentrated at 700C to a volume of (1230 mL). The resulting slurry was filtered and dried for 18 hours at 55°C to give 46.5 g, 50% overall yield of 1- [(1 S,2R)-1-(3,5-difluorophenyl)-2-hydroxy-3-(methylamino)propyl]-7-fluoro-3,3- dimethyl-1 ,3-dihydro-2H-indol-2-one as a white solid.
Example 16:
Preparation of 7-fluoro-1-fπSH3-fluorophenvθr(2S)-oxiran-2-vπmethyl}-3.3- dimethyl-1.3-dihvdro-2H-indol-2-one
[0152] Diethyl-azodicarboxylate (100 g, 572 mmol) was added dropwise to a solution of 1-[(1S, 2S)-1-(3,5-difluorophenyl)-2,3-dihydroxypropyl]-7-fluoro-3,3- dimethyl-1 ,3-dihydro-2H-indol-2-one (90 g, 260 mmol) and Ph3P (129g, 520 mmol) in toluene (1042 mL) at 250C. The mixture was stirred for 1 hour at 800C. Ph3P (7 g, 26 mmol) was added to the mixture at 800C. The mixture was stirred for 8 hours at 😯0C. Diethyl-azodicarboxylate (9 g, 52 mmol) was added to the mixture at 800C. The mixture was stirred for about 2 hours at 800C until the reaction is complete. Heptane (3120 mL) was added to the reaction mixture at 800C. The mixture was cooled to 100C and then filtered through a silica gel (720 g) plug. The filtrate was discarded. The silica gel plug was rinsed with a solution of ethyl acetate (1100 mL) in heptane (3300 mL). The filtrate was concentrated to dryness at 500C to give 56 g, 80% purity, 52% yield of 7-fluoro-1-{(1 S)-(3-fluorophenyl)[(2S)-oxiran-2-yl]methyl}- 3,3-dimethyl-1 ,3-dihydro-2H-indol-2-one.
Example 17:
Preparation of 7-fluoro-1-rf1S.2R)-1-f3-fluorophenvπ-2-hydroxy-3-(methyl amino) propyll-3,3-dimethyl-1 ,3-dihydro-2H-indol-2-one:
[0153] In a flask with 7-fluoro-1-[(1 S,2S)-1-(3-fluorophenyl)-2,3-dihydroxypropyl]- 3,3-dimethylindolin-2-one (10 g, 0.0288 mol) and para-toluenesulfonic acid (pTSA) (0.0438 g, 0.023 mol) in THF (50 mL), trimethyl orthoacetate (4.15 g, 4.3 mL, 0.0346 moles) was added dropwise. The amber color solution was stirred at room temperature for 1 hour. The reaction mixture was concentrated to oil then THF (50 mL) was added. Cooled to 00C to 5°C then acetyl bromide (8.50 g, 0.0692 mol) was added. The resulting mixture was stirred at room temperature for 3 to 4 hours then concentrated to oil and charged with THF (25 mL) and EtOH 2B (25 mL) followed by K2CO3 -325 (39.8 g, 0.288 mol). The mixture was stirred at room temperature then the mixture was concentrated in vacuo to oil. MTBE (100 mL) and H2O (170 mL) were added to dissolve the oil. The two layers were separated. The aqueous layer was extracted with MTBE (2 x 100 mL). The combined organic layer was concentrated to oil then 33% solution of methylamine in ethanol (15 eq.) was added and stirred at room temperature. At the completion of the reaction, the mixture was concentrated to oil. MTBE 100 mL) and H2O (100 mL) were added. The two layers were separated. The organic layer was extracted with 37% concentrated HCI (30.7g) in H2O (65 g). The lower aqueous layer was extracted with MTBE (100 mL) then cooled to 0-50C. MTBE (100 mL) and a solution of 50% NaOH (30 g) in H2O (30 g) were added to the aqueous layer. The mixture was stirred for 20 minutes at room temperature and the layers were separated. The aqueous layer was back extracted with MTBE (50 mL). The combined organic layer was washed with a 15% NaCI (23 mL) solution. The organic layer was concentrated to give as oil (8.4 g, about 90% by LC/MS, 60% yield). [0154] When ranges are used herein for physical properties, such as molecular weight, or chemical properties, such as chemical formulae, all combinations and subcombinations of ranges specific embodiments therein are intended to be included.
Paper
Organic Process Research & Development 2009, 13, 880–887
Large-Scale Synthesis of a Selective Inhibitor of the Norepinephrine Transporter:
Mechanistic Aspects of Conversion of Indolinone Diol to Indolinone Aminoalcohol
and Process Implications
Asaf Alimardanov,* Alexander Gontcharov, Antonia Nikitenko, Anita W. Chan, Zhixian Ding, Mousumi Ghosh,
Mahmut Levent, Panolil Raveendranath,† Jianxin Ren, Maotang Zhou, Paige E. Mahaney,‡ Casey C. McComas,‡
Joseph Ashcroft, and John R. Potoski
Wyeth Research, 401 North Middletown Road, Pearl RiVer, New York 10965, U.S.A., and Wyeth Research, 500 Arcola Road,
CollegeVille, PennsylVania 19426, U.S.A.
TREATMENT OF GYNECOLOGICAL DISORDERS
WAY-315193 (Wyeth Pharmaceuticals)
Development of a scalable synthesis of WAY-315193 is described.
Use of LiHMDS as a base and Ti(O-i-Pr)4 as a Lewis acid was optimal for efficient and reproducible addition of indolinone anion to epoxyalcohol. Conversion of indolinone diol to indolinone aminoalcohol was achieved via monotosylationmethylamination.
The possibility of selective formation of the amidine side product, as well as its utilization for alternative selective preparation of the target aminoalcohol, was demonstrated.
The synthetic route used initially for preparation of 1 is shown in Scheme 1. The key step of the synthesis was the
Sharpless epoxidation of fluorocinnamic alcohol 3 which selectively introduced both relative and absolute configurations at the C-2 and C-3 positions. At the early stages of the project, allylic alcohol 3 was prepared in two steps from commercially available fluorocinnamic acid 2 by treatment with MeI in the presence of Cs2CO3 in acetone, followed by DIBAL reduction at -78 °C. The epoxide 4 was opened with the sodium salt of dimethylfluoroindolinone in DMF to afford the diol. The diol 6 was further elaborated into the final aminoalcohol hydrochloride 1 in 30-34% yield via tosylation with p-toluenesulfonyl chloride (TsCl) in pyridine, isolation of the intermediate monotosylate, treatment with MeNH2, and conversion to HCl salt. Dimethylfluoroindolinone was prepared by reduction and bis-methylation of 7-fluoroisatin by a process developed earlier as described in a prior publication.3
white solid (58% yield). Mp 209-212 °C.
[R]D25°)+10.7°.
1H NMR (D2O, 400 MHz) δ: 7.40-7.25 (m,3H), 7.16-6.97 (m, 4H), 5.47-5.25 (2H, broad m), 3.27-3.20
(2H, broad m), 2.76 (s, 3H), 1.37 (s, 3H), 1.24 (broad s, 3H).
ES+ MS, m/z 361 (MH+). Anal. Calc’d for C20H23ClF2N2O2:C, 60.53; H, 5.84; N, 7.06. Found: C, 60.43; H, 5.69; N, 6.84.
Sn content: <1 ppm. Enantiomeric purity: 99.1% ee. Chiral SFCanalysis conditions: column: Chiralcel OF 250 mm × 4.6 mm;mobile phase: 30% ethanol, 0.4% diethylamine in CO2; detection wavelength: 254 nm; 2 mL/min, 40 °C.
* Corresponding author. E-mail: alimara@wyeth.com.
† Deceased.
‡ Wyeth Research, Collegeville, PA.
(1) (a) For a review on norepinephrine reuptake inhibitors, see: Babu,R. P. K.; Maiti, S. N. Heterocycles 2006, 69, 539. (b) Krell, H. V.;Leuchter, A. F.; Cook, I. A.; Abrams, M. Psychosomatics 2005, 46,379. (c) Hajos, M.; Fleishaker, J. C.; Filipiak-Reisner, J. K.; Brown,M. T.; Wong, E. H. W. CNS Drug ReV. 2004, 10, 23. (d) McCormack,
P. L.; Keating, G. M. Drugs 2004, 64, 2567.
(2) Kim, C. Y.; Mahaney, P. E.; Trybulski, E. J.; Zhang, P.; Terefenko,E. A.; McComas, C. C.; Marella, M. A.; Coghlan, R. D.; Heffernan,G. D.; Cohn, S. T.; Vu, A. T.; Sabatucci, J. P.; Ye, F. Phenylaminopropanol
Derivatives and Methods of Their Use. U.S. Patent 7,517,899,2009.
(3) Wu, Y.; Wilk, B. K.; Ding, Z.; Shi, X.; Wu, C. C.; RaveendranathP.; Durutlic, H. Process for the Synthesis of Progesterone ReceptorModulators. U.S. Patent Publ. Appl. US 2007/027327, 2007.
(4) (a) Gao, Y.; Hanson, R. M.; Klunder, J. M.; Ko, S. Y.; Masamune,H.; Sharpless, K. B. J. Am. Chem. Soc. 1987, 109, 5765. (b) For a recent example of large-scale asymmetric epoxidation, see: Henegar,
K. E.; Cebula, M. Org. Process Res. DeV. 2007, 11, 354.
(5) (a) For indolinone deprotonation for epoxide opening, see: Proudfoot,J. R.; Regan, J. R.; Thomson, D. S.; Kuzmich, D.; Lee, T. W.;Hammach, A.; Ralph, M. S.; Zindell, R.; Bekkali, Y. Preparation ofPropanol and Propylamine Derivatives and Their Use as Glucocorticoid Ligands. WO 2004/063163, 2004. (b) Gillman, K.; Bocchino, D. M.
Preparation of Monosaccharides Prodrugs of Fluorooxindoles Useful in Treatment of Disorders Which are Responsive to the Opening of Potassium Channels. U.S. Patent Publ. Appl. US 2004/0152646, 2004.
(c) For amide deprotonation for epoxide opening, see: Albanese, D.; Landini, D.; Penso, M. Tetrahedron 1997, 53, 4787. (d) Chan, W. N.; Evans, J. M.; Hadley, M. S.; Herdon, H. J.; Jerman, J. C.; Morgan,H. K. A.; Stean, T. O.; Thompson, M.; Upton, N.; Vong, A. K. J. Med.Chem. 1996, 39, 4537.
(6) Bordwell, F. G.; Fried, H. E. J. Org. Chem. 1991, 56, 4218.
(7) (a) Smith, J. G. Synthesis 1984, 629. (b) Parker, R. E.; Isaacs, N. S.Chem. ReV. 1959, 59, 737.
| Patent ID | Date | Patent Title |
|---|---|---|
| US7595338 | 2009-09-29 | Process for preparing 3, 3-disubstituted oxindoles and thio-oxindoles |
| US2009099164 | 2009-04-16 | Phenylaminopropanol Derivatives and Methods of Their Use |
| US7517899 | 2009-04-14 | Phenylaminopropanol derivatives and methods of their use |
| US2009093469 | 2009-04-09 | Phenylaminopropanol Derivatives and Methods of Their Use |
| US2008146645 | 2008-06-19 | Process for Preparing Indolinone Phenylaminopropanol Derivatives |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| WO2006118955A2 * | Apr 27, 2006 | Nov 9, 2006 | Wyeth | Process for preparing 3,3-disubstituted oxindoles and thio-oxindoles |
| WO2006118955A3 * | Apr 27, 2006 | Jan 11, 2007 | Bogdan Kazimierz Wilk | Process for preparing 3,3-disubstituted oxindoles and thio-oxindoles |
| WO2007041023A1 * | Sep 27, 2006 | Apr 12, 2007 | Wyeth | 1- (1h- indol- 1-yl) -3- (methylamino) -1- phenylpropan-2-ol derivatives and related compounds as modulators of the monoamine reuptake for the treatment of vasomotor symptoms (vms) |
| WO2008024492A2 * | Aug 22, 2007 | Feb 28, 2008 | Wyeth | Indolinone phenylaminopropanol derivatives and process for the preparation thereof |
| WO2008024492A3 * | Aug 22, 2007 | May 15, 2008 | Wyeth Corp | Indolinone phenylaminopropanol derivatives and process for the preparation thereof |
| US9403807 | Dec 2, 2014 | Aug 2, 2016 | Merck Sharp & Dohme Corp. | Mineralocorticoid receptor antagonists |
| WO2005097744A1 * | Mar 29, 2005 | Oct 20, 2005 | Wyeth | 1-(1h-indol-1-yl)-3-(4-methylpiperazin-1-yl)-1-phenyl propan-2-ol derivatives and related compounds as modulators of the norepinephrine (ne) and the serotonine (5-ht) activity and the monoamine reuptake for the treatment of vasomotor symptoms (vms) |
| WO2005097761A1 * | Mar 29, 2005 | Oct 20, 2005 | Wyeth | Heterocyclic phenylaminopropanol derivatives as modulators of the monoamine reuptake for the treatment of vasomotor symptoms (vms) |
| WO2007041023A1 * | Sep 27, 2006 | Apr 12, 2007 | Wyeth | 1- (1h- indol- 1-yl) -3- (methylamino) -1- phenylpropan-2-ol derivatives and related compounds as modulators of the monoamine reuptake for the treatment of vasomotor symptoms (vms) |
| US20070072897 * | Sep 27, 2006 | Mar 29, 2007 | Wyeth | Phenylaminopropanol derivatives and methods of their use |
//////////WAY-315193
c1c2c(c(cc1)F)N(C(C2(C)C)=O)[C@@H](c3cc(ccc3)F)[C@@H](CNC)O

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










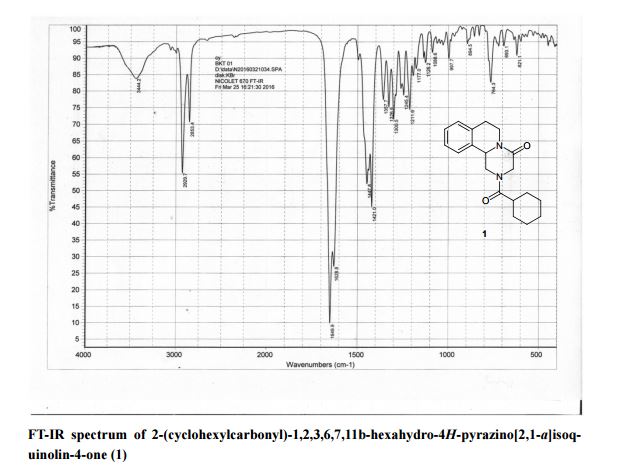
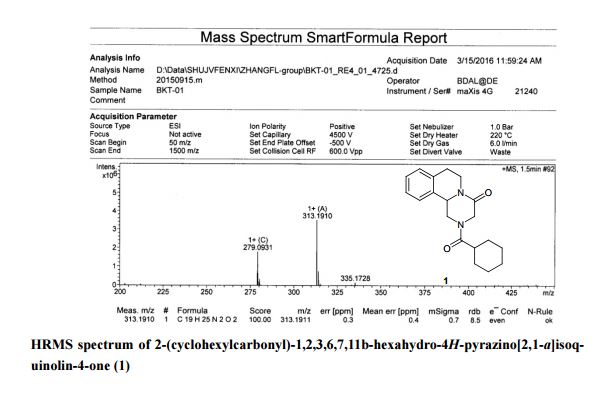





 (S)-6-Hydroxybuspirone
(S)-6-Hydroxybuspirone