Home » Uncategorized (Page 11)
Category Archives: Uncategorized
Padoprazan


Padoprazan
CAS 2756367-23-2
MF C19H20FN3O4S MW 405.4 g/mol
1-[5-(2-fluorophenyl)-4-methoxy-1-(6-methoxypyridine-3-sulfonyl)-1Hpyrrol-3-yl]-N-methyl methanamine
1-[5-(2-fluorophenyl)-4-methoxy-1-[(6-methoxy-3-pyridinyl)sulfonyl]pyrrol-3-yl]-N-methylmethanamine
1-(5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1 H -pyrrol-3-yl)- N -methylmethanamine
proton pump inhibitor, 95BJ28E2RP, ID-120040002, ID 120040002
Padoprazan is a new-generation potassium-competitive acid blocker (P-CAB) used to treat acid-related disorders like gastroesophageal reflux, according to MedchemExpress.com and Patsnap Synapse. It works by inhibiting the proton pump in the stomach and is different from traditional proton pump inhibitors (PPIs) because it is not dependent on acid activation. Padoprazan is currently undergoing Phase 3 clinical trials in Korea, notes THE BIO (더바이오).
Key facts about padoprazan
- Drug class: Potassium-competitive acid blocker (P-CAB), a type of proton pump inhibitor, according to DrugBank and GlpBio.
- Mechanism: It inhibits the proton pump in the stomach to reduce acid production and is not acid-activated like older PPIs, per DrugBank.
- Indications: Used for acid-related conditions like gastroesophageal reflux, reports Patsnap Synapse.
- Status: Currently undergoing Phase 3 clinical trials in Korea, says THE BIO (더바이오).
- Development: It is a new-generation drug being developed by companies like Daewon Pharmaceutical.
Padoprazan is a small molecule drug. The usage of the INN stem ‘-prazan’ in the name indicates that Padoprazan is a proton pump inhibitor, not dependent on acid activation. Padoprazan has a monoisotopic molecular weight of 405.12 Da.
PAT
- NEW INHIBITOR OF ACID SECRETION AND USE OF THE SAMEPublication Number: PE-20231652-A1Priority Date: 2020-06-17
- Novel acid secretion inhibitors and use thereofPublication Number: TW-I839161-BPriority Date: 2020-06-17Grant Date: 2024-04-11
- Novel acid secretion inhibitor and use thereofPublication Number: US-2023373954-A1Priority Date: 2020-06-17
- Novel acid secretion inhibitor and use thereofPublication Number: EP-4148050-B1Priority Date: 2020-06-17Grant Date: 2024-12-18
- Novel acid secretion inhibitors and use thereofPublication Number: TW-I797645-BPriority Date: 2020-06-17Grant Date: 2023-04-01
- Acid secretion inhibitor and use thereofPublication Number: US-11767311-B2Priority Date: 2020-06-17Grant Date: 2023-09-26
- Novel Acid Secretion Inhibitor and use thereofPublication Number: AU-2021293694-B2Priority Date: 2020-06-17Grant Date: 2023-12-21
- Novel acid secretion inhibitor and use thereofPublication Number: CN-115884968-BPriority Date: 2020-06-17Grant Date: 2024-06-21
- Novel acid secretion inhibitors and their usesPublication Number: JP-7404561-B2Priority Date: 2020-06-17Grant Date: 2023-12-25
- Novel acid secretion inhibitors and use thereofPublication Number: KR-102432523-B1Priority Date: 2020-06-17Grant Date: 2022-08-16
- Novel acid secretion inhibitor and use thereofPublication Number: CN-115884968-APriority Date: 2020-06-17
- Novel acid secretion inhibitor and use thereofPublication Number: JP-2023524172-APriority Date: 2020-06-17
- Novel acid secretion inhibitor and use thereofPublication Number: US-2023192650-A1Priority Date: 2020-06-17
- Novel acid secretion inhibitors and use thereofPublication Number: TW-202325702-APriority Date: 2020-06-17
- Novel acid secretion inhibitor and use thereofPublication Number: WO-2021256861-A1Priority Date: 2020-06-17
- Novel acid secretion inhibitors and use thereofPublication Number: TW-202214588-APriority Date: 2020-06-17
- Novel Acid Secretion Inhibitor and use thereofPublication Number: AU-2021293694-A1Priority Date: 2020-06-17
- Novel acid secretion inhibitor and use thereofPublication Number: CA-3182882-A1Priority Date: 2020-06-17
- Novel acid secretion inhibitor and use thereofPublication Number: EP-4148050-A1Priority Date: 2020-06-17
- Novel salt of 1-sulfonyl pyrrole derivative, preparation method thereof and pharmaceutical composition comprising thereofPublication Number: TW-I828476-BPriority Date: 2021-12-15Grant Date: 2024-01-01
- Novel salt of 1-sulfonyl pyrrole derivative, preparation method thereof and pharmaceutical composition comprising thereofPublication Number: WO-2023113458-A1Priority Date: 2021-12-15
- Novel salt of 1-sulfonyl pyrrole derivative, method for preparing same, and pharmaceutical composition including samePublication Number: WO-2023113474-A1Priority Date: 2021-12-15
- Novel salt of 1-sulfonyl pyrrole derivative, method for preparing same, and pharmaceutical composition including samePublication Number: US-2025042872-A1Priority Date: 2021-12-15
- Novel acid secretion inhibitors and use thereofPublication Number: KR-20210156234-APriority Date: 2020-06-17
- Novel salt of 1-sulfonylpyrrole derivative, preparation method thereof and pharmaceutical composition comprising the samePublication Number: CN-118541361-APriority Date: 2021-12-15
- Novel salt of 1-sulfonyl pyrrole derivative, preparation method thereof and pharmaceutical composition comprising thereofPublication Number: KR-20230091056-APriority Date: 2021-12-15
- Novel salts of 1-sulfonyl pyrrole derivatives, methods for producing the same, and pharmaceutical compositions containing the samePublication Number: KR-20240119083-APriority Date: 2021-12-15
- Novel salt of 1-sulfonyl pyrrole derivative, preparation method thereof and pharmaceutical composition comprising thereofPublication Number: TW-202334114-APriority Date: 2021-12-15
- Novel formulation comprising acid secretion inhibitorsPublication Number: KR-20240161598-APriority Date: 2023-05-04
- Method for preparation of 6-methoxypyridine-3-yl derivativesPublication Number: TW-202411216-APriority Date: 2022-05-23
- Method for preparing 6-methoxypyridin-3-yl derivativesPublication Number: WO-2023229322-A1Priority Date: 2022-05-23
- Method for preparation of 6-methoxypyridine-3-yl derivativesPublication Number: KR-20230163283-APriority Date: 2022-05-23
- NOVEL SALT OF A DERIVATIVE OF 1-SULFONYLPYRROL, METHOD OF PREPARATION THEREOF AND PHARMACEUTICAL COMPOSITION THAT INCLUDES THE SAMEPublication Number: AR-127964-A1Priority Date: 2021-12-15
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2021256861&_cid=P22-MI13VU-05837-1



Synthesis Example 1. Synthesis of Example 1
[267]
[Example 1] 1-(5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1 H -pyrrol-3-yl)- N -methylmethanamine
[268]
(1) Synthesis of step methyl 5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1 H -pyrrole-3-carboxylate
[269]Methyl 5-(2-fluorophenyl)-4-methoxy-1
H -pyrrole-3-carboxylate (intermediate 1, 1.0 eq., 1.2 g, 4.8 mmol) was dissolved in THF (20.0 mL), and NaH (2.0 eq., 384.8 mg, 9.6 mmol) was added dropwise at 0 °C and stirred at room temperature for 10 min. 6-Methoxypyridine-3-sulfonyl chloride (1.5 eq., 1.6 g, 7.2 mmol) was added and stirred at room temperature for 1 h. Water was added to the reaction solution, and the mixture was extracted with EA. The organic layer was dried over anhydrous magnesium sulfate, filtered, concentrated, and purified by column chromatography to obtain methyl 5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1
H -pyrrole-3-carboxylate as a light brown solid. (1.85 g, 91.6%)
[270]
(2) Synthesis of step 5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1 H -pyrrol-3-yl)methanol
[271]Methyl 5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1
H -pyrrole-3-carboxylate (1.0 eq., 1.0 g, 2.38 mmol) was dissolved in THF (5.0 mL), and 1.0 M DIBAL in
n -hexane solution (5.0 eq., 11.9 mL, 11.9 mmol) was added dropwise at 0 °C, followed by stirring at room temperature for 1 h. The reaction solution was cooled to 0 °C, quenched with an aqueous Rochelle salt solution, and extracted with EA. The organic layer was dried over anhydrous magnesium sulfate, filtered, concentrated, and purified by column chromatography to obtain 5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1
H -pyrrol-3-yl)methanol as a yellow oil. (654.8 mg, 70.2%)
[272]
(3) Synthesis of step 5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1 H -pyrrole-3-carbaldehyde
[273]5-(2-Fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1
H -pyrrol-3-yl)methanol (1.0 eq., 500.0 mg, 1.3 mmol) and Dess-Martin periodinane (1.0 eq., 540.4 mg, 1.3 mmol) were dissolved in DCM (10.0 mL) and stirred at room temperature for 1 h. The reaction mixture was concentrated and purified by column chromatography to give 5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1
H -pyrrole-3-carbaldehyde as a pale brown solid. (388.2 mg, 78.1%)
[274]
(4) Step 1 Synthesis of (5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1 H -pyrrol-3-yl)- N -methylmethanamine
[275]5-(2-Fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1
H -pyrrole-3-carbaldehyde (1.0 eq., 385.0 mg, 0.99 mmol) was dissolved in THF (5.0 mL), and 2.0 M methylamine in THF (10 eq., 4.9 mL, 9.9 mmol) was added. After stirring at room temperature for 1 h, the reaction mixture was cooled to 0 °C, and NaBH
4 (10 eq., 373.4 mg, 9.9 mmol) was added, followed by stirring at room temperature for 1 h. 6.0
N aqueous hydrogen chloride solution was slowly added dropwise to the reaction solution, and the resulting solid was filtered. The filtered solid was dissolved in water, 1
N aqueous sodium hydroxide solution was added, and extraction was performed with EA. The organic layer was dried over anhydrous magnesium sulfate, filtered, and concentrated to obtain 1-(5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1 H -pyrrol-3-yl)-
N -methylmethanamine as a white solid. (125.8 mg, 28.3%) [M+H] + : 405
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2023113474&_cid=P22-MI1405-08231-1

7) Preparation of 1-(5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1H-pyrrol-3-yl)-N-methylmethanamine free base[211]
(1) Step: Synthesis of methyl 5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1 H -pyrrole-3-carboxylate[212]Methyl 5-(2-fluorophenyl)-4-methoxy-1
H -pyrrole-3-carboxylate (intermediate 1, 1.0 eq., 920 g, 3.69 mol) was dissolved in DMF (9.2 L), and t-BuOK (2.0 eq., 828 g, 7.38 mmol) was added dropwise at 0 °C and stirred for 30 min. 6-Methoxypyridine-3-sulfonyl chloride (1.5 eq., 1.15 kg, 5.54 mol) was added and stirred at 0 °C for 1 h. Water was added to the reaction solution, which was then extracted with ethyl acetate. The organic layer was dried over anhydrous magnesium sulfate, filtered, concentrated, and purified by column chromatography to obtain methyl 5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1
H -pyrrole-3-carboxylate as a white solid. (1.20 kg, 77.4%) [213]
(2) Step: Synthesis of 5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1 H -pyrrol-3-yl)methanol[214]Methyl 5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1
H -pyrrole-3-carboxylate (1.0 eq., 1.1 kg, 2.62 mol) was dissolved in THF (11.0 L), and DIBAL 2.0 M in THF solution (3.0 eq., 3.93 L, 7.86 mol) was added dropwise at 0 °C, followed by stirring for 30 min. The reaction solution was quenched with 5% aqueous Rochelle’s salt solution and extracted with ethyl acetate. The organic layer was dried over anhydrous magnesium sulfate, filtered, and concentrated to obtain 5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1
H -pyrrol-3-yl)methanol as a light yellow oil. (870 g, 84.8%) [215]
(3) Step: Synthesis of 5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1 H -pyrrole-3-carbaldehyde[216]5-(2-Fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1
H -pyrrol-3-yl)methanol (1.0 eq., 830 g, 2.12 mol) and TEA (4.0 eq., 1.59 kg, 15.7 mol) were dissolved in DMSO (4.15 L), and SO
3 -pyridine (4.0 eq., 1.35 kg, 8.48 mol) was added dropwise, and the mixture was stirred at room temperature for 1.5 h. Water was added to the reaction mixture at 0 °C, and the mixture was extracted with ethyl acetate. The organic layer was dried over anhydrous magnesium sulfate, filtered, and concentrated to obtain 5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1
H -pyrrole-3-carbaldehyde as a yellow solid. (722 g, 87.6%) [217]
(4) Step: Synthesis of 1-(5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1 H -pyrrol-3-yl)- N -methylmethanamine[218]5-(2-Fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1
H -pyrrole-3-carbaldehyde (1.0 eq., 715 g, 1.83 mol) was dissolved in methanol (7.2 L), and methylamine in methanol (5.0 eq., 916 g, 9.16 mol) was added. After stirring at room temperature for 1 h, the reaction mixture was concentrated, dissolved in ethanol (7.2 L), cooled to 0 °C, and NaBH
4 (2.0 eq., 139 g, 3.66 mol) was added, and stirred at 0 °C for 1 h. Water was added to the reaction solution, and extracted with ethyl acetate. The organic layer was dried over anhydrous magnesium sulfate, filtered, concentrated, and purified by column chromatography to obtain 1-(5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1 H -pyrrol-3-yl)-
N -methylmethanamine as a brown oil. (347 g, 46.7%)
<Example 1> Preparation of hydrochloric acid salt of 1-(5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1H-pyrrol-3-yl)-N-methylmethanamine About 500 mg of the free base of 1-(5-(2-fluorophenyl)-4-methoxy-1-((6-methoxypyridin-3-yl)sulfonyl)-1H-pyrrol-3-yl)-N-methylmethanamine was weighed and placed in a glass vial, and then dissolved in 2 mL of ethanol while heating at 25°C. Then, 647.44 μL (2 M) hydrochloric acid was added to the vial. The sample was continuously stirred on a magnetic stirrer at room temperature for 24 hours, and after stirring for 24 hours, the solid precipitate was separated by centrifugation. Subsequently, the wet solid was dried at 40°C for 20 hours to obtain a grayish white dried powder.
SYN



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
/////////Padoprazan, proton pump inhibitor, 95BJ28E2RP, ID-120040002, ID 120040002
Ofirnoflast


Ofirnoflast
CAS 2731294-23-6
MFC23H19F4N7O2 MW501.4 g/mol
N-[4-(4-amino-7-cyclopropyl-7H-pyrrolo[2,3-d]pyrimidin-5-yl)-2-fluorophenyl]-N’-{5-[1-
(trifluoromethyl)cyclopropyl]-1,2-oxazol-3-yl}urea
N-(4-(4-AMINO-7-CYCLOPROPYL-7H-PYRROLO(2,3-D)PYRIMIDIN-5-YL)-2-FLUOROPHENYL)-N’-(5-(1-(TRIFLUOROMETHYL)CYCLOPROPYL)-3-ISOXAZOLYL)UREA
N-(4-(4-AMINO-7-CYCLOPROPYL-7H-PYRROLO(2,3-D)PYRIMIDIN-5-YL)-2-FLUOROPHENYL)-N’-(5-(1-(TRIFLUOROMETHYL)CYCLOPROPYL)-1,2-OXAZOL-3-YL)UREA
OFIRNOLAST [USAN]
OFIRNOFLAST
UREA, N-(4-(4-AMINO-7-CYCLOPROPYL-7H-PYRROLO(2,3-D)PYRIMIDIN-5-YL)-2-FLUOROPHENYL)-N’-(5-(1-(TRIFLUOROMETHYL)CYCLOPROPYL)-3-ISOXAZOLYL)-
OFIRNOFLAST [INN]
serine/ threonine-protein kinase Nek7 inhibitor, antiinflammatory, HT-6184, HT 6184, 54PY2PBN7S
Ofirnoflast is an investigational drug, a NEK7 inhibitor, that targets and disrupts the formation of the NLRP3 inflammasome, a key driver of chronic inflammation. Developed by Halia Therapeutics, it is being explored for conditions like myelodysplastic syndromes (MDS), obesity, and Alzheimer’s disease. The drug’s unique mechanism aims to address inflammation at a root cause level, potentially offering a new approach to treating these diseases.
How it works
- Ofirnoflast is a “first-in-class” molecule that selectively inhibits the NEK7 protein.
- NEK7 is essential for the assembly of the NLRP3 inflammasome, a molecular complex that causes chronic inflammation.
- By inhibiting NEK7, ofirnoflast prevents the inflammasome from forming and promotes its disassembly.
- This approach aims to reduce inflammation without causing broad immunosuppression.
Therapeutic applications
- Myelodysplastic Syndromes (MDS): Ofirnoflast has completed a Phase 2 study for this condition and received Orphan Drug Designation from the FDA. It is being investigated for its potential to improve blood cell production by targeting the underlying inflammation.
- Obesity: An ongoing Phase 2 study is exploring ofirnoflast in combination with semaglutide to target inflammation and metabolic issues.
- Alzheimer’s Disease: Ofirnoflast is part of an early-stage program looking into its potential for this disease.
Ofirnoflast is a first-in-class, orally bioavailable NEK7 inhibitor currently undergoing Phase 2 clinical evaluation. It disrupts NLRP3 inflammasome assembly by targeting NEK7’s scaffolding function—blocking complex formation independently of NLRP3 activation status, upstream of caspase activation, pyroptosis, and inflammatory cytokine release. This mechanism offers a novel therapeutic approach for chronic inflammation. Unlike NSAIDs, corticosteroids, cytokine-neutralising biologics, and NLRP3-directed small molecules—which are frequently limited by off-target effects, immunosuppression, or incomplete efficacy—ofirnoflast provides a targeted approach with fewer anticipated liabilities
- A Ph2 Study to Evaluate the Safety, Efficacy and Tolerability of HT-6184 and Semaglutide in Obese Participants With T2DMCTID: NCT07172867Phase: Phase 2Status: Not yet recruitingDate: 2025-09-15
- HT-6184 in Subjects With MDSCTID: NCT07052006Phase: Phase 2Status: Active, not recruitingDate: 2025-07-14
- Evaluating Ability of HT-6184 to Reduce Inflammation and Pain After Third Molar ExtractionCTID: NCT06241742Phase: Phase 2Status: CompletedDate: 2025-03-30
- Study to Evaluate HT-6184 in Healthy SubjectsCTID: NCT05447546Phase: Phase 1Status: CompletedDate: 2023-08-28
SYN
https://www.tandfonline.com/doi/full/10.1080/1061186X.2025.2542856
SYN
COMPD 10
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2021242505&_cid=P11-MHZPDU-32878-1


INTERMEDIATE D1
5-(4-AMINO-3-FLUOROPHENYL)-7-CYCLOPROPYL-7H-PYRROLO[2,3-D]PYRIMIDIN-4- AMINE

A mixture of 7-cyclopropyl-5-iodo-7H-pyrrolo[2,3-d]pyrimidin-4-amine (C1, 0.160 g, 0.533 mmol), 2-fluoro-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)aniline (0.190 g, 0.800 mmol), and K2CO3 (0.221 g, 1.599 mmol) in 1,4-dioxane (1 mL) and water (0.3 mL) was purged with N2 for 10 min. Pd(PPh3)4 (0.062 g, 0.053 mmol) was then added and the reaction mixture was stirred at 100 °C for 12 h. Following completion of the reaction (as indicated by TLC), the mixture was filtered through a pad celite which was then rinsed with EtOAc (2 x 10 mL). The combined filtrates were concentrated under reduced pressure to yield crude material which was purified by flash chromatography (silica gel 230-400 mesh, eluting with 3% MeOH in DCM), affording
the title compound as a yellow solid (0.110 g, 73% yield).1H NMR (400 MHz, DMSO-d6) δ = 8.14 (s, 1H), 7.13 (s, 1H), 7.05-7.09 (m, 1H), 6.95-6.98 (m, 1H), 6.82-6.86 (m, 1H), 6.10 (bs, 2H), 5.22 (bs, 2H), 3.52-3.58 (m, 1H), 1.00-1.04 (m, 4H). LCMS: 284.1 [M+H].

3-(1-(Trifluoromethyl)cyclopropyl)isoxazol-5-amine (precursor to E6) and 5-(1-(trifluoromethyl)cyclopropyl)isoxazol-3-amine (precursor to E7) were synthesized as reported in Synthesis 2013, 45, 171–173
EXAMPLE 5
1-(4-(4-AMINO-7-CYCLOPROPYL-7H-PYRROLO[2,3-D]PYRIMIDIN-5-YL)-2- FLUOROPHENYL)-3-(3-(1-(TRIFLUOROMETHYL)CYCLOPROPYL)ISOXAZOL-5-YL)UREA

The title compound was prepared following the general procedure for urea formation (Method A), starting from 5-(4-amino-3-fluorophenyl)-7-cyclopropyl-7H-pyrrolo[2,3-d]pyrimidin-4-amine (D1, 0.080 g, 0.282 mmol) and phenyl (3-(1-(trifluoromethyl)cyclopropyl)isoxazol-5-yl)carbamate (E6, 0.088 g, 0.282 mmol), and was obtained as a white solid (0.031 g, 22% yield).1H NMR (400 MHz, DMSO-d6) δ = 10.59 (bs, 1H), 8.84 (bs, 1H), 8.11-8.17 (m, 2H), 7.26-7.37 (m, 3H), 6.20 (s, 1H), 6.16 (bs, 2H), 3.55-3.61 (m, 1H), 1.45-1.49 (m, 2H), 1.38-1.43 (m, 2H), 1.03-1.08 (m, 4H). LCMS: 502.1 [M+H].
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2024249257&_cid=P11-MHZP9H-30149-1



PAT
- Targeted nek7 inhibition for modulation of the nlrp3 inflammasomePublication Number: US-2023210853-A1Priority Date: 2020-05-08
- Inhibitors of NEK7 kinasePublication Number: US-11713321-B2Priority Date: 2020-05-08Grant Date: 2023-08-01
- Inhibitors of nek7 kinasePublication Number: EP-4146348-B1Priority Date: 2020-05-08Grant Date: 2024-07-03
- Inhibitors of nek7 kinasePublication Number: US-2023416259-A1Priority Date: 2020-05-08
- Inhibitors of NEK7 kinasePublication Number: US-12091413-B2Priority Date: 2020-05-08Grant Date: 2024-09-17
- Inhibitors of nek7 kinasePublication Number: TW-202208356-APriority Date: 2020-05-08
- Inhibitors of NEK7 kinasePublication Number: AU-2021280893-A1Priority Date: 2020-05-08
- Inhibitors of NEK7 kinasePublication Number: CN-115843272-APriority Date: 2020-05-08
- Inhibitors of nek7 kinasePublication Number: EP-4146348-A1Priority Date: 2020-05-08
- Inhibitors of NEK7 kinasePublication Number: KR-20230008763-APriority Date: 2020-05-08
- Polymorphs of nek 7 inhibitorsPublication Number: WO-2024249257-A1Priority Date: 2023-05-26
- Inhibitors of NEK7 kinasePublication Number: US-11161852-B1Priority Date: 2020-05-08Grant Date: 2021-11-02
- Inhibitors of nek7 kinasePublication Number: US-2021355130-A1Priority Date: 2020-05-08
- Inhibitors of nek7 kinasePublication Number: US-2022064173-A1Priority Date: 2020-05-08
- Inhibitors of nek7 kinasePublication Number: WO-2021242505-A1Priority Date: 2020-05-08



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
///////////ofirnoflast, serine/ threonine-protein kinase Nek7 inhibitor, antiinflammatory, HT-6184, HT 6184, 54PY2PBN7S
Nurandociguat


Nurandociguat
CAS 2781965-75-9
MF C30H36ClF2N5O2 MW 572.1 g/mol
1-[(3R)-1-{4-chloro-4′-[4-(2-methylpropyl)piperazin-1-yl][1,1′-biphenyl]-2-yl} piperidin-3-yl]-5-(difluoromethyl)-1H-pyrazole-4-carboxylic acid
1-[(3R)-1-[5-chloro-2-[4-[4-(2-methylpropyl)piperazin-1-yl]phenyl]phenyl]piperidin-3-yl]-5-(difluoromethyl)pyrazole-4-carboxylic acid
guanylate cyclase activator, BAY 3283142, LPU8429UK5
Nurandociguat is a small molecule drug candidate, previously known as BAY 3283142, that is a guanylate cyclase activator being developed by Bayer for cardiovascular conditions. The “ciguat” stem in its name indicates its function as a guanylate cyclase activator, a mechanism that is also being investigated for related drugs like runcaciguat. It is currently in clinical trials, including a Phase 2 program for chronic kidney disease (CKD).
- Drug class: Guanylate cyclase activator
- Developer: Bayer
- Previous name: BAY 3283142
- Indication: Investigated for cardiovascular conditions
- Current status: In clinical development, including a Phase 2 study for chronic kidney disease (CKD)
- OriginatorBayer
- ClassAntihypertensives; Cardiovascular therapies; Hepatoprotectants; Urologics
- Mechanism of ActionGuanylate cyclase stimulants
- Phase IIRenal failure
- Phase ICardiovascular disorders; Diabetic retinopathy; Hypertension; Liver disorders
- 28 Sep 2025No recent reports of development identified for phase-I development in Renal-failure in Germany (PO, Immediate release)
- 16 Sep 2025(CTIS2024-510856-11-00) (EudraCT2024-510856-11-00): Trial initiation and completion info added; updated DevT; Corrected intro to match DevT as most of the info about indication and countries missing
- 28 May 2025No recent reports of development identified for phase-I development in Renal-failure(In volunteers, In adults) in Japan (PO, Immediate release)
Nurandociguat is a small molecule drug. The usage of the INN stem ‘-ciguat’ in the name indicates that Nurandociguat is a guanylate cyclase activator and stimulator. Nurandociguat has a monoisotopic molecular weight of 571.25 Da.
PAT
- Soluble guanylate cyclase activators for use in the treatment of heart failure with preserved ejection fraction in womenPublication Number: WO-2023237577-A1Priority Date: 2022-06-09
- Substituted pyrazolo piperidine carboxylic acidsPublication Number: WO-2022122910-A1Priority Date: 2020-12-10
- The use of sgc activators for the treatment of ophthalmologic diseasesPublication Number: WO-2022122917-A1Priority Date: 2020-12-10
- Use of sGC activators for the treatment of ophthalmic diseasesPublication Number: CN-115175681-APriority Date: 2020-12-10
- Use of sgc activators for the treatment of ophthalmologic diseasesPublication Number: US-2022241273-A1Priority Date: 2020-12-10
- Use of sGC activators for the treatment of ophthalmic diseasesPublication Number: JP-2023514928-APriority Date: 2020-12-10
- The use of sgc activators for the treatment of ophthalmologic diseasesPublication Number: EP-4259140-A1Priority Date: 2020-12-10
- Use of sGC activators for the treatment of ophthalmic diseasesPublication Number: KR-20230118143-APriority Date: 2020-12-10
- Substituted pyrazolo piperidine carboxylic acidsPublication Number: US-2023265072-A1Priority Date: 2020-12-10
- Use of sGC activators for the treatment of ophthalmic diseasesPublication Number: JP-2024073585-APriority Date: 2020-12-10
- Use of sGC activators for the treatment of ophthalmological diseasesPublication Number: JP-7458683-B2Priority Date: 2020-12-10Grant Date: 2024-04-01
- Use of sgc activators for the treatment of ophthalmologic diseasesPublication Number: US-2023346777-A1Priority Date: 2020-12-10
- Use of sGC activators for treating ophthalmic diseasesPublication Number: CN-115175681-BPriority Date: 2020-12-10Grant Date: 2024-10-25
SYN
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2022122917&_cid=P20-MHVQYD-96133-1
soluble guanylate cyclase (sGC) activators for use in the treatment and/or prophylaxis of ophthalmologic diseases, including non-proliferative diabetic retinopathy (NPDR), diabetic macular edema (DME), retinal ganglion cell/photoreceptor neurodegeneration and cataract, especially wherein the soluble guanylate cyclase (sGC) activators are compounds selected from the group consisting of





Example 1
1 – [ 1 – { 4-Chloro-4′- [4-(2-methylpropyl)piperazin- 1 -yl] [1,1 ’-biphenyl] -2-yl }piperidin-3-yl] -5- (difluoromethyl)-lH-pyrazole-4-carboxylic acid hydrochloride (Enantiomer 1)

Ethyl 1 – [ 1 – { 5-chloro-2- [(trifluoromethanesulfonyl)oxy]phenyl }piperidin-3-yl] -5-(difluoromethyl)- 1 H-pyrazole-4-carboxylate (prepared in analogy to Example 11A, Enantiomer 1, 80.0 mg, 147 pmol) and l-(2-methylpropyl)-4- [4-(4,4,5 ,5-tetramethyl- 1 ,3,2-dioxaborolan-2-yl)phenyl]piperazine (Example 18 A 62.8 mg, 97 % purity, 177 pmol) were placed under argon in toluene/ethanol (820/820 pl). 2 M sodium carbonate solution (220 pl, 2.0 M, 440 pmol) and tetrakis(triphenylphosphine)palladium(0) (8.52 mg, 7.37 pmol) were added and the mixture was stirred at 100°C. overnight. The reaction mixture was diluted with ethyl acetate and 1 M hydrochloric acid was added. The aqueous phase was extracted three times with ethyl acetate. The organic phase was dried with sodium sulfate, filtered off and evaporated. The crude mixture was dissolved with THF/ethanol (2.0/0.2 ml), 1 M lithium hydroxide solution (1.5 ml, 1.5 mmol) was added and the mixture was stirred at room temperature overnight. A I M lithium hydroxide solution (740 pl, 740 pmol) was added again. After about 6 h the reaction mixture was evaporated at 50°C. The residue was dissolved in
SUBSTITUTE SHEET (RULE 26)
acetonitrile/water/0.25 ml trifluoroacetic acid and purified by preparative HPLC (RP18 column, acetonitrile/water gradient with the addition of 0.1% trifluoroacetic acid). The crude product was purified by means of thick layer chromatography (dichloromethane/methanol/formic acid: 10/1/0.1). The silica gel mixture was stirred with dichloromethane/1 M hydrochloric acid in dioxane (10/1) in ethanol, filtered off and carefully evaporated at 30°C and lyophilized. 34 mg of the target compound (36% of theory, purity 95%) were obtained.
LC-MS (Method 6): Rt = 1.23 min; MS (ESIpos): m/z = 572 [M-HC1+H]+
‘H-NMR (600 MHz, DMSO-d6) 5 [ppm]: 1.004 (15.87), 1.015 (16.00), 1.500 (0.51), 1.521 (0.57), 1.728 (0.73), 1.750 (0.61), 1.897 (0.57), 1.917 (0.62), 1.975 (0.79), 2.122 (0.42), 2.133 (0.84), 2.144 (1.02), 2.156
(0.79), 2.571 (0.47), 2.587 (0.91), 2.610 (0.52), 3.004 (0.84), 3.022 (2.01), 3.026 (2.20), 3.038 (3.72), 3.048
(2.50), 3.065 (0.75), 3.154 (2.66), 3.161 (2.75), 3.169 (2.36), 3.177 (1.88), 3.224 (0.84), 3.237 (0.70), 3.589
(1.41), 3.602 (1.80), 3.825 (1.02), 3.841 (0.78), 3.866 (1.05), 3.882 (0.75), 4.223 (2.57), 4.445 (0.68), 4.463
(0.97), 4.481 (0.57), 7.045 (0.55), 7.055 (3.63), 7.070 (3.72), 7.084 (2.72), 7.087 (3.09), 7.110 (1.47), 7.113
(1.11), 7.123 (2.19), 7.127 (2.02), 7.163 (3.67), 7.177 (2.19), 7.215 (0.46), 7.428 (0.83), 7.495 (4.24), 7.510
(4.02), 7.515 (2.07), 7.602 (0.82), 7.959 (4.79), 9.484 (0.54).
Example 2
1 – [ 1 – { 4-Chloro-4′- [4-(2-methylpropyl)piperazin- 1 -yl] [1,1 ’-biphenyl] -2-yl }piperidin-3-yl] -5-(difluoromethyl)-lH-pyrazole-4-carboxylic acid (Enantiomer 2)

Method A
A solution of ethyl l-[l-{4-chloro-4′-[4-(2-methylpropyl)piperazin-l-yl][l,T-biphenyl]-2-yl}piperidin-3-yl]-5-(difluoromethyl)-lH-pyrazole-4-carboxylate (prepared in analogy to Example 17A, Enantiomer 2, 50.8 g, 84.6 mmol) in a THF/methanol mixture 9:1 (1.0 1) was treated with an aqueous solution of lithium hydroxide (850 ml, 1.0 M, 850 mmol) and stirred overnight at room temperature. The reaction mixture was
SUBSTITUTE SHEET (RULE 26)
concentrated, diluted with dichloromethane (1.5 1) and adjusted to pH = 2 with an aqueous solution of hydrogen chloride (2N). The resulting suspension was stirred 45 minutes at room temperature. The solid was filtered, washed with water and dried under vacuum affording 43 g (90 % yield) of the title compound.
LC-MS (Method 7): Rt = 1.27 min; MS (ESIpos): m/z = 572 [M+H]+
‘H-NMR (600 MHz, DMSO-d6) 5 [ppm]: 1.002 (15.68), 1.013 (16.00), 1.080 (0.57), 1.092 (1.18), 1.103 (0.63), 1.498 (0.74), 1.519 (0.83), 1.719 (1.03), 1.741 (0.88), 1.902 (0.78), 1.908 (0.74), 1.922 (0.88), 1.928
(0.83), 1.943 (0.45), 1.978 (1.13), 1.994 (0.74), 2.102 (0.71), 2.112 (0.85), 2.123 (0.70), 2.571 (1.40), 2.591
(0.77), 2.882 (1.10), 3.018 (1.27), 3.035 (3.01), 3.053 (2.14), 3.239 (2.40), 3.254 (2.32), 3.368 (1.13), 3.379
(1.40), 3.391 (1.33), 3.403 (0.92), 3.493 (0.76), 4.463 (0.65), 4.482 (1.12), 4.500 (0.62), 7.033 (4.22), 7.048
(4.45), 7.074 (3.47), 7.077 (4.04), 7.100 (1.85), 7.103 (1.52), 7.113 (2.53), 7.117 (2.34), 7.162 (4.18), 7.175
(2.71), 7.439 (1.03), 7.481 (4.88), 7.495 (4.57), 7.526 (2.04), 7.613 (0.91), 7.952 (5.28).
Method B
1 – { 1 – [4-Chloro-4′-(4-isobutylpiperazin- 1 -yl) [biphenyl] -2-yl]piperidin-3-yl } -5-(difluoromethyl)- 1 H-pyrazole-4-carboxylic acid hydrochloride (prepared in analogy to Example 3, Enantiomer 2, 31.2 mg, 51.3 pmol) were dissolved in 17 ml of dichloromethane and 1 ml of methanol. The solution was shaken once with 1.5 ml of saturated, aqueous sodium bicarbonate solution. The phases were separated. 5 ml of dichloromethane and 3 ml of methanol were added to the organic phase. The organic phase was then dried over sodium sulfate, filtered, evaporated and purified by preparative HPLC (RP18 column, acetonitrile/water gradient, neutral without acid addition). Product fractions were combined and lyophilized. 22 mg of the target compound (74% of theory) were obtained.
LC-MS (Method 3): Rt = 1.73 min; MS (ESIpos): m/z = 572 [M+H]+
‘H-NMR (600 MHz, DMSO-d6) 5 [ppm]: 0.887 (15.60), 0.898 (16.00), 1.493 (0.64), 1.514 (0.70), 1.695 (0.89), 1.718 (0.74), 1.799 (0.48), 1.811 (0.88), 1.822 (1.12), 1.833 (0.92), 1.844 (0.48), 1.890 (0.68), 1.910
(0.74), 1.977 (0.93), 1.995 (0.62), 2.118 (3.91), 2.130 (3.66), 2.516 (5.14), 3.017 (1.09), 3.035 (2.76), 3.053
(1.94), 3.181 (5.03), 3.185 (5.02), 3.267 (1.53), 4.473 (0.55), 4.491 (0.96), 4.509 (0.54), 6.963 (3.96), 6.977
(4.06), 7.048 (3.13), 7.051 (3.31), 7.081 (1.60), 7.084 (1.26), 7.095 (2.21), 7.098 (1.89), 7.152 (3.52), 7.165
(2.42), 7.434 (4.45), 7.448 (4.50), 7.533 (1.51), 7.621 (0.67), 7.930 (4.14).
Example 3
1 – { 1 – [4-Chloro-4′-(4-isobutylpiperazin- 1 -yl) [biphenyl] -2-yl]piperidin-3-yl } -5-(difluoromethyl)- 1 H-pyrazole-4-carboxylic acid hydrochloride (Enantiomer 2)
SUBSTITUTE SHEET (RULE 26)

Method A
A suspension of 1 – [ 1 – { 4-chloro-4′- [4-(2-methylpropyl)piperazin- 1 -yl] [1,1 ’-biphenyl] -2-yl }piperidin-3-yl] -5-(difluoromethyl)-lH-pyrazole-4-carboxylic acid (prepared in analogy to Example 2, Enantiomer 2, 43.5 g, 76.0 mmol) in diethyl ether (870 ml) was treated with a solution of hydrogen chloride in diethyl ether (84 ml, 1.0 M, 84 mmol). The resulting mixture was stirred overnight at room temperature and evaporated affording 46.1 g (quant.) of the title compound.
LC-MS (Method 3): Rt = 1.72 min; MS (ESIpos): m/z = 572 [M+H]+
‘H-NMR (600 MHz, DMSO-d6) 5 [ppm]: 1.026 (15.64), 1.037 (16.00), 1.497 (0.56), 1.519 (0.61), 1.722 (0.78), 1.743 (0.65), 1.903 (0.59), 1.910 (0.53), 1.924 (0.66), 1.930 (0.61), 1.978 (0.82), 1.994 (0.50), 2.142
(0.45), 2.154 (0.91), 2.165 (1.11), 2.176 (0.89), 2.187 (0.45), 2.557 (0.64), 2.577 (1.02), 2.594 (0.55), 2.992
(1.81), 3.002 (2.77), 3.012 (1.87), 3.018 (1.15), 3.036 (2.40), 3.054 (1.60), 3.133 (1.12), 3.148 (1.19), 3.168
(0.53), 3.237 (0.88), 3.250 (0.76), 3.338 (0.81), 3.360 (1.42), 3.379 (0.88), 3.580 (1.61), 3.791 (0.89), 3.819
(1.25), 3.844 (0.81), 4.463 (0.89), 4.474 (0.97), 4.481 (1.26), 4.488 (0.99), 4.499 (0.88), 7.051 (3.56), 7.065
(3.77), 7.077 (2.72), 7.080 (3.14), 7.103 (1.42), 7.106 (1.13), 7.116 (2.00), 7.120 (1.84), 7.165 (3.40), 7.178
(2.22), 7.443 (0.84), 7.489 (4.04), 7.504 (3.79), 7.531 (1.66), 7.618 (0.72), 7.954 (4.33), 10.519 (0.49).
Method B
Ethyl 1 – [ 1 – { 5-chloro-2- [(trifluoromethanesulfonyl)oxy]phenyl }piperidin-3-yl] -5-(difluoromethyl)- 1 H-pyrazole-4-carboxylate (prepared in analogy to Example 14A, Enantiomer 2, 80.0 mg, 150 pmol) and l-(2-methylpropyl)-4- [4-(4,4,5 ,5-tetramethyl- 1 ,3,2-dioxaborolan-2-yl)phenyl]piperazine (Example 18 A 64.1 mg, 97 % purity, 180 pmol) were dissolved under argon in toluene/ethanol (0.83/0.83 ml). Tetrakis(triphenylphosphine)palladium(0) (8.69 mg, 7.52 pmol) and 2 M sodium carbonate solution (226 pl, 452 pmol) were added and the mixture was stirred at 100°C overnight. The reaction mixture was diluted with ethyl acetate and water. The aqueous phase was acidified with 1 M hydrochloric acid. The phases were
SUBSTITUTE SHEET (RULE 26)
separated and the aqueous phase was extracted twice with ethyl acetate. The combined organic phases were dried over sodium sulfate, filtered and evaporated. The crude product was dissolved in THF/ethanol (3.9/0.39 ml), 1 M aqueous lithium hydroxide solution (1.5 ml, 1.5 mmol) was added and the mixture was stirred overnight at room temperature. The mixture was evaporated, the residue was dissolved in acetonitrile/TFA/water and purified using preparative HPLC (RP18 column, acetonitrile/water gradient with the addition of 0.1% TFA). The product fractions were combined and evaporated. The residue was mixed with 0.1 M hydrochloric acid in dioxane, carefully evaporated at 30°C (twice) and then lyophilized. 53 mg of the target compound (55% of theory, purity 95%) were obtained.
LC-MS (Method 4): Rt = 0.91 min; MS (ESIpos): m/z = 572 [M-HC1+H]+
‘H-NMR (400 MHz, DMSO-d6) 5 [ppm]: 1.004 (15.46), 1.020 (16.00), 1.491 (0.44), 1.522 (0.50), 1.722 (0.68), 1.753 (0.55), 1.890 (0.47), 1.920 (0.55), 1.967 (0.84), 2.129 (0.76), 2.146 (0.96), 2.163 (0.76), 2.582
(0.91), 2.613 (0.48), 2.999 (0.86), 3.010 (1.71), 3.025 (3.88), 3.041 (2.30), 3.131 (0.88), 3.161 (1.25), 3.177
(2.08), 3.213 (1.75), 3.242 (1.16), 3.467 (1.06), 3.496 (0.84), 3.503 (0.60), 3.519 (0.54), 3.525 (0.50), 3.549
(0.75), 3.555 (0.84), 3.572 (1.57), 3.582 (1.48), 3.589 (1.38), 3.601 (2.78), 3.608 (1.89), 3.633 (0.44), 3.640
(0.41), 3.811 (0.94), 3.847 (1.32), 3.878 (0.71), 4.329 (0.49), 4.439 (0.46), 4.466 (0.73), 4.477 (0.52), 4.839
(0.49), 7.047 (3.30), 7.070 (3.64), 7.082 (2.61), 7.087 (3.29), 7.104 (1.46), 7.109 (0.86), 7.124 (2.34), 7.129
(2.03), 7.160 (3.99), 7.181 (1.96), 7.388 (0.88), 7.490 (4.02), 7.512 (3.81), 7.519 (2.20), 7.650 (0.72), 7.959
(3.78), 9.708 (0.41).
[OC]D20 = -73.05°, c = 0.465g/100cm3, trichloromethane.
Enantiomer 2 has an absolute configuration of R as shown in example 3 A below.
1 – { 3(2?)- 1 – [4-Chloro-4′-(4-isobutylpiperazin- 1 -yl) [biphenyl] -2-yl]piperidin-3-yl } -5-(difluoromethyl)- 1H-pyrazole-4-carboxylic acid hydrochloride
Example 3A
1 – { 3(7?)- 1 – [4-Chloro-4′-(4-isobutylpiperazin- 1 -yl) [biphenyl] -2-yl]piperidin-3-yl } -5-(difluoromethyl)- 1H-pyrazole-4-carboxylic acid hydrochloride hemihydrate
SUBSTITUTE SHEET (RULE 26)

100 mg 1 – { 1 – [4-Chloro-4′-(4-isobutylpiperazin- 1 -yl) [biphenyl] -2-yl]piperidin-3-yl } -5-(difluoromethyl)-lH-pyrazole-4-carboxylic acid hydrochloride (Enantiomer 2) (example 3) were solved at 60°C in 3,5 ml 2 -propanol, wherein the 2-propanol was dosed portion wise in lOOpl -portions at 60°C until a clear solution was obtained. Afterwards the vessel was closed with a septum and placed into a slowly cooling sand bath from 60°C to roomtemperature over the weekend -> small amounts of solids were detected. Thereafter the septum was provided with a canula, in order to slowly let the solvent evaporate. After 4 weeks crystals were collected and inspected under a microscope.



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
/////////Nurandociguat, guanylate cyclase activator, BAY 3283142, LPU8429UK5
Muvadenant


Muvadenant
CAS 2459881-03-7
MF C21H26N4O4S , 430.5 g/mol
(5S)-N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy[1,3]thiazolo[4,5-c]pyridin-2-yl]-7-oxa-2-azaspiro[4.5] decane-2-carboxamide
(5S)-N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-[1,3]thiazolo[4,5-c]pyridin-2-yl]-7-oxa-2-azaspiro[4.5]decane-2-carboxamide
adenosine receptor antagonist, antineoplastic, 6LSF69F6A8, M1069 , M 1069
Muvadenant is a small molecule drug. The usage of the INN stem ‘-adenant’ in the name indicates that Muvadenant is a adenosin receptor antagonist. Muvadenant has a monoisotopic molecular weight of 430.17 Da.
Adenosine is an ubiguitous modulator of numerous physiological activities, particularly within the cardiovascular, nervous and immune systems. Adenosine is related both structurally and metabolically to the bioactive nucleotides adenosine triphosphate (ATP), adenosine diphosphate (ADP), adenosine monophosphate (AMP) and cyclic adenosine monophosphate (cAMP), to the biochemical methylating agent S-adenosyl-L-methione (SAM) and structurally to the coenzymes NAD, FAD and coenzym A and to RNA.
Via cell surface receptors, adenosine modulates diverse physiological functions including induction of sedation, vasodilatation, suppression of cardiac rate and contractility, inhibition of platelet aggregability, stimulation of gluconeogenesis and inhibition of lipolysis. Studies show that adenosine is able to activate adenylate cyclases, open potassium channels, reduce flux through calcium channels, and inhibit or stimulate phosphoinositide turnover through receptor-mediated
mechanisms (Muller C. E. and Stein B., Current Pharmaceutical Design, 2: 501 , 1996; Muller C. E., Exp. Opin. Ther. Patents, 7(5): 419, 1997).
Adenosine receptors belong to the superfamily of G-protein-coupled receptors (GPCRs). Four major subtypes of adenosine receptors have been
pharmacologically, structurally and functionally characterized (Fredholm et al., Pharm. Rev., 46: 143-156, 1994) and referred to as A1, A2A, A2B and A3. Though the same adenosine receptor can couple to different G-proteins, adenosine A1 and A3 receptors usually couple to inhibitory G-proteins referred to as G, and Go which inhibit adenylate cyclase and down-regulate cellular cAMP levels. In contrast, the adenosine A2A and A2B receptors couple to stimulatory G-proteins referred to as Gs that activate adenylate cyclase and increase intracellular levels of cAMP (Linden J., Annu. Rev. Pharmacol. Toxicol., 41 : 775-87 2001).
PAT
- Thiazolopyridine derivatives as adenosine receptor antagonistsPublication Number: US-2022119412-A1Priority Date: 2019-01-22
- Thiazolopyridine derivatives as adenosine receptor antagonistsPublication Number: EP-3914600-B1Priority Date: 2019-01-22Grant Date: 2024-08-07
- Thiazolopyridine derivatives as adenosine receptor antagonistsPublication Number: CN-113939520-BPriority Date: 2019-01-22Grant Date: 2024-11-22
- Thiazolopyridine derivatives as adenosine receptor antagonistsPublication Number: ES-2992081-T3Priority Date: 2019-01-22Grant Date: 2024-12-09
- Thiazolopyridine derivatives as adenosine receptor antagonists.Publication Number: JP-7600119-B2Priority Date: 2019-01-22Grant Date: 2024-12-16
- Thiazolopyridine derivatives as adenosine receptor antagonistsPublication Number: AU-2020211697-A1Priority Date: 2019-01-22
- Thiazolopyridine derivatives as adenosine receptor antagonistsPublication Number: KR-20210116572-APriority Date: 2019-01-22
- Thiazolopyridine derivatives as adenosine receptor antagonistsPublication Number: CN-113939520-APriority Date: 2019-01-22
- Thiazolopyridine derivatives as adenosine receptor antagonistsPublication Number: EP-3914600-A1Priority Date: 2019-01-22
- Thiazolopyridine derivative as an adenosine receptor antagonistPublication Number: JP-2022524914-APriority Date: 2019-01-22
- Combination therapy for cancerPublication Number: CN-117858723-APriority Date: 2021-06-07
- Combination treatment of cancerPublication Number: EP-4351640-A1Priority Date: 2021-06-07
- Combination Treatment of CancerPublication Number: JP-2024520764-APriority Date: 2021-06-07
- Combination Treatment of CancerPublication Number: US-2024279338-A1Priority Date: 2021-06-07
- Thiazolopyridine derivatives as adenosine receptor antagonistsPublication Number: WO-2020152132-A1Priority Date: 2019-01-22
- Novel crystalline forms of (s)-7-oxa-2-aza-spiro[4.5]decane-2-carboxylic acid [7-(3,6-dihydro-2h-pyran-4-yl)-4-methoxy-thiazolo[4,5-c]pyridin-2-yl]-amide and co-crystal forms thereofPublication Number: TW-202415667-APriority Date: 2022-08-02
- Novel crystalline forms of (s)-7-oxa-2-aza-spiro[4.5]decane-2-carboxylic acid [7-(3,6-dihydro-2h-pyran-4-yl)-4-methoxy-thiazolo[4,5-c]pyridin-2-yl]-amide and co-crystal forms thereofPublication Number: WO-2024028273-A1Priority Date: 2022-08-02
- Combination treatment of cancerPublication Number: WO-2022258622-A1Priority Date: 2021-06-07
- Combination treatment of cancerPublication Number: AU-2022288571-A1Priority Date: 2021-06-07
- Combination treatment of cancerPublication Number: CA-3220380-A1Priority Date: 2021-06-07
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2020152132&_cid=P10-MHPOEV-06540-1



1. (5R)-N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-thiazolo[4,5-c]pyridin- 2-yl]-7-oxa-2-azaspiro[4.5]decane-2-carboxamide 24
and (5S)-N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-thiazolo[4,5- c]pyridin-2-yl]-7-oxa-2-azaspiro[4.5]decane-2-carboxamide 25

a. 4-chloro-5-iodo-3-nitropyridin-2-ol
Into a 250-mL round-bottom flask was placed 4-chloro-3-nitropyridin-2-ol (10.0 g, 54.4 mmol, 95%), N-lod-succinimid (NIS, 14.2 g, 59.9 mmol, 95%) in acetonitrile (115 mL). The solution was stirred for 1 h overnight at 80°C in an oil bath. The mixture was concentrated and the precipitate formed collected by filtration. The residue was washed with twice with petrol ether (500 mL) dried under vacuum at 60°C overnight. This resulted in 4-chloro-5-iodo-3-nitropyridin-2-ol (16.5 g, 97.9%, 97% purity) as a yellow solid. MS: m/z = 300.9 [M+H]+.
b. 4-chloro-5-iodo-2-methoxy-3-nitropyridine
Into a 500-mL round-bottom flask was placed 4-chloro-5-iodo-3-nitropyridin-2-ol (16.5 g, 53.3 mmol, 97%), Ag2CO3 (15.5 g, 53.3 mmol, 95%) in toluene (310 mL). To this suspension CH3I (15.9 g, 107 mmol, 95%) was added at 50°C and the mixture was stirred at 80°C for 4 h. The precipitate was collected by filtration and discarded. The filtrate was evaporated to dryness under vacuum and the residue purified by silica gel chromatography with ethyl acetate/petroleum ether (15:85).
This resulted in 4-chloro-5-iodo-2-methoxy-3-nitropyridine (9.90 g, 52.6%, 89% purity) as a light yellow solid. MS: m/z = 315.5 [M+H]+.
c. 4-chloro-5-iodo-2-methoxypyridin-3-amine
Into a 250-mL 3-necked round-bottom flask was placed 4-chloro-5-iodo-2-methoxy-3-nitropyridine (9.90 g, 28.0 mmol, 89%), iron (16.5 g, 281 mmol, 95%) and NH 4C (9.40 g, 174 mmol, 99%) in ethanol (152 mL) and water (30 mL). The mixture was stirred for 2 h at 80°C in an oil bath. The reaction mixture was filtered over Celite, washed with ethanol and the mother liquor was concentrated to dryness. The residue was stirred for 30 min. with 100 ml water at 60°dried in vacuo. This resulted in 4-chloro-5-iodo-2-methoxypyridin-3-amine (7.20 g, 75%, 83% purity) as an off-white solid. It was used without further purification in the next step. MS: m/z = 285.9 [M+H]+.
d. N-[7-iodo-4-methoxy-[1,3]thiazolo[4,5-c]pyridin-2-yl]benzamide
Into a 500-mL round-bottom flask was placed 4-chloro-5-iodo-2-methoxypyridin-3-amine (7.20 g, 21.0 mmol, 83%) in acetone (150 mL) and benzoyl isothiocyanate (5.21 g, 31.5 mmol, 99%) was added dropwise at room temperature. The solution was stirred for 1 h at 50 °C in an oil bath. The solids were collected by filtration, washed with acetone and dried in vacuo to give N-[7-iodo-4-methoxy-[1 ,3]thiazolo[4,5-c]pyridin-2-yl]benzamide (8.73 g, 91 %, 90% purity) as a white solid. MS: m/z = 412.2 [M+H]+.
e. N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-[1,3]thiazolo[4,5-c]pyridin- 2-yl]benzamide
To a solution of N-[7-iodo-4-methoxy-[1 ,3]thiazolo[4,5-c]pyridin-2-yl]benzamide (6.00 g, 13.1 mmol, 90%) and 2-(3,6-dihydro-2H-pyran-4-yl)-4,4,5,5-tetramethyl-1 ,3,2-dioxaborolane (6.13 g, 27.7 mmol, 95%) in dioxane (200 mL) and water (40.00 mL) were added NaOH (2.90 g, 68.9 mmol, 95%) and Pd(dppf)Cl2* dichloromethane (1.20 g, 1.40 mmol, 95%). After stirring for 1 h at 100°C under a nitrogen atmosphere, the mixture was concentrated to dryness under vacuo. The residue was purified by silica gel chromatography with ethyl acetate/hexane (95:5). This resulted in 3.32 g (62%, 90% purity) of N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-[1 ,3]thiazolo[4,5-c]pyridin-2-yl]benzamide as colorless solid. MS: m/z = 368.1 [M+H]+.
f. 7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-[1,3]thiazolo[4,5-c]pyridin-2- amine
To a stirred mixture of N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-[1 ,3]thiazolo[4,5-c]pyridin-2-yl]benzamide (3.27 g, 8.00 mmol, 90%) in water/methanol (1 :1 , 300 mL) was added NaOH (3.36 g, 80.0 mmol, 95%) at room temperature under nitrogen atmosphere. The mixture was stirred for overnight at 90°C under nitrogen atmosphere and evaporated to dryness. The residue was taken up in water and extracted 3 times with dichloromethane (100 mL). The combined organic layers were dried over anhydrous Na2SO4, filtered and evaporated to dryness. The residue was purified by silica gel column chromatography, eluted with petrol ether/ethyl acetate (1 :1) to afford 7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-[1 ,3]thiazolo[4,5-c]pyridin-2-amine (1.50 g, 68%, 96% purity) as a light brownish solid. MS: m/z = 264.1 [M+H]+.
g. phenyl N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy- [1,3]thiazolo[4,5c]pyridin-2-yl]-N-(phenoxycarbonyl)carbamate
To a stirred solution of 7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-[1 ,3]thiazolo[4,5-c]pyridin-2-amine (600 mg, 2.19 mmol, 96%) and phenyl chloroformate (1.81 g,
11.0 mmol, 95%) in THF (50 mL) was added K2CO3 (1.59 g, 11.0 mmol, 95%) and pyridine (913 mg, 11.0 mmol, 95%) at room temperature under nitrogen
atmosphere. The mixture was stirred for 6 h at 50° and then after re-cooling to room temperature quenched by the addition of water (300 mL). The mixture was extracted 3 times with dichloromethane (200 mL), the combined organic layers were washed once with brine (200 mL), dried over anhydrous Na2SO4, filtered, and evaporated to dryness under reduced pressure. This resulted in phenyl N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-[1 ,3]thiazolo[4,5-c]pyridin-2-yl]-N-(phenoxycarbonyl)carbamate (1.00 g, 69%, 76% purity) as a light yellow solid. The crude product was used in the next step directly without further purification. MS: m/z = 504.1 [M+H]+.
h. N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-[1,3]thiazolo[4,5-c]pyridin- 2-yl]-7-oxa-2-azaspiro[4.5]decane-2-carboxamide
To a mixture of phenyl N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-[1 ,3]thiazolo[4,5-c]pyridin-2-yl]-N-(phenoxycarbonyl)carbamate (1.00 g, 1.52 mmol, 76.) and bis(7-oxa-2-azaspiro[4.5]decane), oxalic acid (1.19 g, 3.03 mmol, 95%) in THF (50 mL) was added diisopropylethyl amine (1.24 g, 9.09 mmol, 95%) at room temperature under nitrogen atmosphere. The mixture was stirred for 1 h at 60°. After re-cooling to room temperature, the mixture was extracted twice with dichloromethane (100 mL). The combined organic layers were dried over anhydrous Na2SO4, filtered and evaporated to dryness. The residue was purified by silica gel column chromatography, eluted with petrol ether/ethyl acetate (1 :1) to afford N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-[1 ,3]thiazolo[4,5-c]pyridin-2-yl]-7-oxa-2-azaspiro[4.5]decane-2-carboxamide (600 mg, 92%) as a white solid. HPLC: 99.9 % purity, RT = 1.17 min. MS: m/z = 431.1 [M+H]+. 1 H NMR (300 MHz, DMSO-d6) d 1 1.37 (s, 1 H), 7.95 (s, 1 H), 6.25 (s, 1 H), 4.30-4.29 (m, 2H), 3.99 (s, 3H), 3.89 (t, J=5.4Hz, 2H), 3.61-3.29 (m, 8H), 2.55-2.51 (m, 2H), 1.82-1.54 (m, 6H).
i. (5R)-N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-thiazolo[4,5-c]pyridin- 2-yl]-7-oxa-2-azaspiro[4.5]decane-2-carboxamide 24
and (5S)-N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-thiazolo[4,5- c]pyridin-2-yl]-7-oxa-2-azaspiro[4.5]decane-2-carboxamide 25
N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-[1 ,3]thiazolo[4,5-c]pyridin-2-yl]-7-oxa-2-azaspiro[4.5]decane-2-carboxamide (450 mg, 1.044 mmol, 1 equiv, 99.9%) was purified by chiral-preparative HPLC (Preparative HPLC-032, column: ChiralPak IA, 2*25cm, 5 mm; mobile phase, dichloromethane:ethanol (20:80); detector, UV). This resulted in (5R)-N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-[1 ,3]thiazolo[4,5-c]pyridin-2-yl]-7-oxa-2-azaspiro[4.5]decane-2-carboxamide (178 mg, 39%) as a white solid. HPLC: 99.7 % purity, RT (chiral) = 3.86 min, 100% ee. MS: m/z = 431.2 [M+H]+. 1 H NMR (400 MHz, DMSO-d6) d 1 1.36 (s, 1 H), 7.94 (s, 1 H), 6.24 (s, 1 H), 4.29-4.27 (m, 2H), 3.97 (s,3H), 3.88 (t, J=5.2 Hz, 2H), 3.51-3.19 (m, 8H), 2.55-2.50 (m, 2H), 1.83-1.53 (m, 6H) and (5S)-N-[7-(3,6-dihydro-2H-pyran-4-yl)-4-methoxy-[1 ,3]thiazolo[4,5-c]pyridin-2-yl]-7-oxa-2-azaspiro[4.5]decane-2-carboxamide (171 mg, 38%) as a white solid. HPLC: 99.8 % purity, RT (chiral) = 5.23 min, 99.9% ee. MS: m/z = 431.2 [M+H]+. 1 H NMR (400 MHz, DMSO-d6) d 1 1.35 (s, 1 H), 7.94 (s, 1 H), 6.24 (s, 1 H), 4.29-4.28 (m, 2H), 3.99 (s, 3H), 3.88-3.85 (m, 2H), 3.61-3.29 (m, 8H), 2.55-2.50 (m,2H), 1.83-1.53 (m,6H).
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2024028273&_cid=P10-MHPOFP-06905-1



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
//////////muvadenant, adenosine receptor antagonist, antineoplastic, 6LSF69F6A8, M1069 , M 1069
Matsupexole





Matsupexole
CAS 1399442-97-7
MF C22H34N6O2S, Molecular Weight, 446.61
(4aR,6R,8aR)-2-amino-3-cyano-N-{[2-(dimethylamino)ethyl]carbamoyl}-8-methyl-N-propyl 4,4a,5,6,7,8,8a,9-octahydrothieno[3,2-g]quinoline-6-carboxamide
(4aR,6R,8aR)-2-amino-3-cyano-N-[2-(dimethylamino)ethylcarbamoyl]-8-methyl-N-propyl-4a,5,6,7,8a,9-hexahydro-4H-thieno[3,2-g]quinoline-6-carboxamide
dopamine receptor agonist, Phase 2, Parkinson’s disease, K4UEG65HTX
- OriginatorKissei Pharmaceutical
- DeveloperAffaMed Therapeutics; Kissei Pharmaceutical
- ClassAmides; Amines; Antiparkinsonians; Dimethylamines; Ethylenediamines; Nitriles; Quinolines; Small molecules; Thiophenes; Urea compounds
- Mechanism of ActionDopamine receptor agonists
- Phase IIParkinson’s disease
- 28 Aug 2025Chemical structure information added.
- 06 Sep 2021Kissei Pharmaceutical completes a phase II trial in Parkinson’s disease (In adults, In elderly) in Japan (PO) (NCT04867551)
- 04 Aug 2021Phase-II clinical trials in Parkinson’s disease in China (PO) (Kissei Pharmaceutical pipeline, August 2021)
PAT
- Novel octahydrothienoquinoline derivative, pharmaceutical composition comprising derivative, and use of thesePublication Number: WO-2012124649-A1Priority Date: 2011-03-14
- COMPOUNDS DERIVED FROM OCTAIDROTHIENOQUINOLINE, PHARMACEUTICAL COMPOSITION AND PHARMACEUTICAL AGENT COMPRISING SUCH COMPOUNDSPublication Number: BR-112013023575-B1Priority Date: 2011-03-14
- New octahydrothienoquinoline derivative, pharmaceutical composition containing derivative, and using themPublication Number: RU-2573399-C2Priority Date: 2011-03-14Grant Date: 2016-01-20
- Novel octahydrothienoquinoline derivative, pharmaceutical composition comprising derivative, and use of thesePublication Number: SG-193400-A1Priority Date: 2011-03-14
- Novel octahydrothienoquinoline derivatives, pharmaceutical compositions containing the same, and their usesPublication Number: TW-I537274-BPriority Date: 2011-03-14Grant Date: 2016-06-11
- Novel octahydrothienoquinoline derivative, pharmaceutical composition comprising derivative, and use of thesePublication Number: US-2014243311-A1Priority Date: 2011-03-14
- Octahydrothienoquinoline derivative, pharmaceutical composition comprising derivative, and use of thesePublication Number: US-9138434-B2Priority Date: 2011-03-14Grant Date: 2015-09-22
- Novel octahydrothienoquinoline derivative, pharmaceutical composition comprising derivative, and use of thesePublication Number: HU-E033449-T2Priority Date: 2011-03-14
- Novel octahydrothienoquinoline derivatives, pharmaceutical compositions containing them and their usePublication Number: JP-5563716-B2Priority Date: 2011-03-14Grant Date: 2014-07-30
- Novel octahydrothienoquinoline derivatives, pharmaceutical compositions containing them and their usePublication Number: JP-WO2012124649-A1Priority Date: 2011-03-14
- Novel octahydrothienoquinoline derivatives, pharmaceutical compositions containing them and uses thereofPublication Number: KR-20140010137-APriority Date: 2011-03-14
- NEW OCTAHYDROTHYENOCHINOLINE DERIVATIVE, PHARMACEUTICAL COMPOSITION CONTAINING A DERIVATIVE AND THEIR APPLICATIONPublication Number: RU-2013145799-APriority Date: 2011-03-14
- Novel octahydrothienoquinoline derivatives, pharmaceutical compositions comprising said derivatives and their usesPublication Number: CN-103443106-BPriority Date: 2011-03-14Grant Date: 2015-09-30
- New octahydrothienoquinoline derivative, pharmaceutical composition comprising the derivative, and use thereofPublication Number: DK-2687532-T3Priority Date: 2011-03-14Grant Date: 2017-02-20
- Novel octahydrothienoquinoline derivative, pharmaceutical composition comprising derivative, and use of thesePublication Number: EP-2687532-A1Priority Date: 2011-03-14
- Novel octahydrothienoquinoline derivative, pharmaceutical composition comprising derivative, and use of thesePublication Number: EP-2687532-B1Priority Date: 2011-03-14Grant Date: 2016-12-14
- Novel octahydrothienoquinoline derivative, pharmaceutical composition comprising the derivative, and use thereofPublication Number: ES-2613658-T3Priority Date: 2011-03-14Grant Date: 2017-05-25
- Novel dopamine D2 receptor agonistPublication Number: JP-2014074013-APriority Date: 2012-09-12
- Novel dopamine D2 receptor agonistPublication Number: JP-6177061-B2Priority Date: 2012-09-12Grant Date: 2017-08-09
- Novel octahydrothienoquinoline derivative, pharmaceutical composition comprising derivative, and use of thesePublication Number: AU-2012227428-A1Priority Date: 2011-03-14
- Novel octahydrothienoquinoline derivative, pharmaceutical composition comprising derivative, and use of thesePublication Number: AU-2012227428-B2Priority Date: 2011-03-14Grant Date: 2016-05-05
- Novel octahydrothienoquinoline derivatives, pharmaceutical compositions comprising said derivatives and their usesPublication Number: CN-103443106-APriority Date: 2011-03-14
- Succinate of octahydrothienoquinoline compound, and crystals thereofPublication Number: WO-2022009815-A1Priority Date: 2020-07-06
- Succinate of octahydrothienoquinoline compound and its crystalPublication Number: CN-115803329-APriority Date: 2020-07-06
- Succinate salts of octahydrothienoquinoline compounds and crystals thereofPublication Number: KR-20230035050-APriority Date: 2020-07-06
- Succinate of octahydrothienoquinoline compound, and crystals thereofPublication Number: EP-4177257-A1Priority Date: 2020-07-06
- Succinate salts of octahydrothienoquinoline compound and crystals thereofPublication Number: US-2023286998-A1Priority Date: 2020-07-06
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2022009815&_cid=P22-MHO952-66657-1
[0018]Example 11-{[(4aR,6R,8aR)-2-amino-3-cyano-8-methyl-4,4a,5,6,7,8,8a,9-octahydrothieno[3,2-g]quinolin-6-yl]carbonyl}-3-[2-(dimethylamino)ethyl]-1-propylurea sesquisuccinate monohydrate (Form I crystals of salt (A-1)) 102.8 g of acetone was added to 1-{[(4aR,6R,8aR)-2-amino-3-cyano-8-methyl-4,4a,5,6,7,8,8a,9-octahydrothieno[3,2-g]quinolin-6-yl]carbonyl}-3-[2-(dimethylamino)ethyl]-1-propylurea (22.00 g), the mixture was suspended, and the suspension was heated and stirred at an external temperature of 52°C to dissolve the suspension. Activated carbon (2.2 g) was added to this solution and stirred for 10 minutes. This suspension was hot filtered and washed with 35.2 g of acetone. 220.0 g of acetone was then added, and the reaction solution was heated to an external temperature of 52°C and stirred. Next, 44.0 g of water was added to the reaction solution. Separately, 8.73 g of succinic acid was dissolved in a mixed solution of 156.1 g of acetone and 19.8 g of water. This succinic acid solution was added dropwise to the reaction solution over approximately 10 minutes. The dropping funnel was washed with a mixed solution of 17.4 g of acetone and 2.2 g of water and then added dropwise to the reaction solution. The reaction solution was stirred at an internal temperature of 50°C for 1 hour and cooled to 15°C over 30 minutes. The reaction solution was stirred at an external temperature of 10°C for 2 hours, and the crystals were collected by filtration. The crystals were washed twice with 52.8 g of acetone. The obtained wet crystals were dried under reduced pressure at 50°C for 37 hours and then returned to room temperature under reduced pressure over 3 hours. The crystals were stored under air for 24 hours to obtain crystals (27.75 g) of the title compound.
1 H-NMR (DMSO-d6) (δ (ppm)): 0.85 (3H, t, J = 7.4Hz), 1.32 (1H, ddd, J=12.2Hz, 12.2Hz, 12.2Hz), 1.42-1.57 (2H, m), 1.57-1.70 (1H, m ), 1.89-2.00 (2H, m), 2.20-2.13 (1H, m), 2.13-2.28 (2H, m), 2.21 (3H, s), 2.24 (6H, s ), 2.35-2.48 (1H, m), 2.40 (6H, s), 2.46 (2H, t, J = 6.4Hz), 2.81-2.96 (2H, m), 3.00-3 .12 (1H, m), 3.21-3.33 (2H, m), 3.47-3.66 (2H, m), 6.99 (2H, s), 8.50-8.90 (1H, br).
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2012124649&_cid=P22-MHO8UB-55660-1



[0422]Example 1-11-{[(4aR,6R,8aR)-2-amino-3-cyano-8-methyl-4H,4aH,5H,6H,7H,8H,8aH,9H-thieno[3,2-g]quinolin-6-yl]carbonyl}-3-[2-(dimethylamino)ethyl]-1-propylurea (Compound 1-1) To a mixture of 1-{[(3R,4aR,8aR)-1-methyl-6-oxodecahydroquinolin-3-yl]carbonyl}-3-[2-(dimethylamino)ethyl]-1-propylurea (Reference Example 10-1) (1.602 g) and ethanol (44 mL) were added malononitrile (435 mg), morpholine (0.572 mL), and then elemental sulfur (282 mg) with stirring at room temperature, and the mixture was heated to 55°C and stirred for 1.5 hours. After cooling to room temperature, the reaction mixture was concentrated under reduced pressure, and the residue was purified by column chromatography on aminopropyl silica gel (eluent: 0%-5% methanol/ethyl acetate, gradient elution) to give the title compound (1.479 g) as a solid.
1 H-NMR (CDCl
3 ) δ ppm: 0.94(3H, t, J=7.4Hz), 1.45-1.85(4H, m), 1.95-2.15(2H, m), 2.15-2.30(7H, m), 2.30-2.55(7H, m), 2.60-2.75(1H, m), 2.90-3.00(2H, m), 3.00-3.10(1H, m), 3.35-3.45(2H, m), 3.60-3.85(2H, m), 4.65(2H, s), 9.27(1H, br)[α]
D 29 =-105.54°(c=0.30, MeOH)

[0311]Reference Example 10-11-{[(3R,4aR,8aR)-1-methyl-6-oxodecahydroquinolin-3-yl]carbonyl}-3-[2-(dimethylamino)ethyl]-1-propylurea 1-{[(3’R,4’aR,8’aR)-1′-methyloctahydro-1’H-spiro[1,3-dioxolane-2,6′-quinoline]-3′-yl]carbonyl}-3-[2-(dimethylamino)ethyl]-1-propylurea (Reference Example 8-1) (2.366 g) was added to 2 mol/L hydrochloric acid (30 mL), and the mixture was stirred at room temperature for 2 hours. The reaction mixture was washed with diethyl ether, and then potassium carbonate was added to the aqueous layer to make it alkaline. The mixture was extracted with a methylene chloride/methanol mixed solvent (methylene chloride:methanol = 9:1). The organic layer was dried over anhydrous sodium sulfate and concentrated under reduced pressure to give the title compound (1.605 g).
1 H-NMR (CDCl
3 ) δ ppm: 0.94 (3H, t, J=7.4 Hz), 1.45-1.90 (6H, m), 1.95-2.05 (1H, m), 2.10-2.55 (17H, m), 2.90-3.10 (2H, m), 3.30-3.45 (2H, m), 3.60-3.80 (2H, m), 9.22 (1H, brs).[α]
D 28 =-37.56° (c=0.38, MeOH).

[0198]Reference Example 8-1To a mixture of phenyl 1-{[(3’R,4’aR,8’aR)-1′-methyloctahydro-1’H-spiro[1,3-dioxolane-2,6′-quinoline]-3′-yl]carbonyl}-3-[2-(dimethylamino)ethyl]-1-propylurea N-{[(3’R,4’aR,8’aR)-1′-methyloctahydro-1’H-spiro[1,3-dioxolane-2,6′-quinoline]-3′-yl]carbonyl}-N-propylcarbamate (Reference Example 6-1) (2.401 g) and 2-propanol (30 mL), N,N-dimethylethylenediamine (1.26 mL) was added with stirring at room temperature, and the mixture was heated to 53°C and stirred for 13 hours. After cooling to room temperature, the reaction mixture was concentrated under reduced pressure. The residue was purified by aminopropyl silica gel column chromatography (eluent: 0%-100% ethyl acetate/hexane, gradient elution) to give the title compound (2.383 g).
1 H-NMR (CDCl
3 ) δ ppm: 0.92(3H, t, J=7.4Hz), 1.35-1.50(3H, m), 1.50-1.90(8H, m), 2.00-2.15(1H, m), 2.26(6H, s), 2.31(3H, s), 2.37(1H, t, J=11.2Hz), 2.46(2H, t, J=6.4Hz), 2.85-3.10(2H, m), 3.35-3.45(2H, m), 3.60-3.70(1H, m), 3.70-3.80(1H, m), 3.90-4.00(4H, m), 9.33(1H, br)[α]
D 28 =-6.62°(c=0.31, MeOH)



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
///////matsupexole, dopamine receptor agonist, Phase 2, Parkinson’s disease, K4UEG65HTX
Lunresertib



Lunresertib
CAS 2719793-90-3
MF C18H20N4O2 MW 324.4 g/mol
(1P)-2-amino-1-(3-hydroxy-2,6-dimethylphenyl)-5,6-dimethyl1H-pyrrolo[2,3-b]pyridine-3-carboxamide
serine/ threonine kinase inhibitor, antineoplastic, N95U3A7N57, RP-6306, RP 6306
2-Amino-1-(3-hydroxy-2,6-dimethylphenyl)-5,6-dimethylpyrrolo[2,3-b]pyridine-3-carboxamide
Lunresertib is an investigational new drug that is being evaluated for the treatment of cancer. It is an oral small molecule inhibitor of PKMYT1, developed by Repare Therapeutics.[1] This drug targets cell cycle regulation in tumors with specific genetic alterations, including CCNE1 amplifications or FBXW7 and PPP2R1A loss of function mutations. It is currently in phase 1/2 clinical trials, both as monotherapy or in combination with camonsertib, an ATR inhibitor.[2]
Lunresertib is an orally bioavailable inhibitor of the human membrane-associated tyrosine– and threonine-specific cdc2-inhibitory kinase (PKMYT1), with potential antineoplastic activity. Upon oral administration, lunresertib targets, binds to and inhibits the activity of PKMYT1. This results in the inhibition of CDK1 phosphorylation, which may promote both premature mitosis and a prolonged mitotic arrest, and lead to the accumulation of unrepaired DNA damage and apoptosis in susceptible tumor cells, such as CCNE1-overexpressing tumor cells. PKMYT1 phosphorylates CDK1 specifically when CDK1 is complexed to cyclins, which blocks progression from G2 into mitosis.NCI Thesaurus (NCIt)
- Study of RP-6306 With FOLFIRI in Advanced Solid TumorsCTID: NCT05147350Phase: Phase 1Status: TerminatedDate: 2025-08-20
- Study of RP-6306 Alone or in Combination With RP-3500 or Debio 0123 in Patients With Advanced Solid TumorsCTID: NCT04855656Phase: Phase 1Status: RecruitingDate: 2025-08-06
- RP-6306 in Patients With Advanced CancerCTID: NCT05605509Phase: Phase 2Status: Active, not recruitingDate: 2025-07-14
- Study of RP-6306 With Gemcitabine in Advanced Solid TumorsCTID: NCT05147272Phase: Phase 1Status: TerminatedDate: 2025-06-17
- Liquid-biopsy Informed Platform Trial to Evaluate CDK4/6-inhibitor Resistant ER+/HER2- Metastatic Breast CancerCTID: NCT05601440Phase: Phase 2Status: RecruitingDate: 2025-01-14
- Phase 1 Study of RP-6306 With Carboplatin and Paclitaxel in TP53 Ovarian and Uterine Cancer
- CTID: NCT06107868
- Phase: Phase 1
- Status: Active, not recruiting
- Date: 2024-03-22
PAT
- Compounds, Pharmaceutical Compositions, and Methods of Preparing and Using CompoundsPublication Number: JP-2023521633-APriority Date: 2020-04-01
- Compounds, pharmaceutical compositions, and methods of preparing compounds and of their usePublication Number: US-2023151014-A1Priority Date: 2020-04-01
- Methods of using myt1 inhibitorsPublication Number: US-2023158022-A1Priority Date: 2020-04-01
- Compounds, pharmaceutical compositions, and methods of preparing compounds and of their usePublication Number: EP-4126879-A1Priority Date: 2020-04-01
- Compounds, pharmaceutical compositions, and methods of preparing compounds and of their usePublication Number: IL-296934-APriority Date: 2020-04-01
- Compounds, pharmaceutical compositions, and methods of making the compounds and methods of using themPublication Number: KR-20230011279-APriority Date: 2020-04-01
- Compounds, pharmaceutical compositions and methods of making compounds and methods of their usePublication Number: CN-115916783-APriority Date: 2020-04-01
- Methods of using MYT1 inhibitorsPublication Number: JP-2023519430-APriority Date: 2020-04-01
- Methods of using myt1 inhibitorsPublication Number: WO-2021195782-A1Priority Date: 2020-04-01
- Compounds, pharmaceutical compositions, and methods of preparing compounds and of their usePublication Number: AU-2021250744-A1Priority Date: 2020-04-01
- Methods of using myt1 inhibitorsPublication Number: CA-3173955-A1Priority Date: 2020-04-01
- Methods of using MYT1 inhibitorsPublication Number: CN-115811976-APriority Date: 2020-04-01
- Methods of using myt1 inhibitorsPublication Number: EP-4125907-A1Priority Date: 2020-04-01
SYN
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2021195781&_cid=P20-MHLE6P-37080-1








Step 9. To a suspension of 2-amino-1-(3-methoxy-2,6-dimethyl-phenyl)-5,6-dimethyl-pyrrolo[2,3-b]pyridine-3-carboxamide (2.22 g, 6.56 mmol, 77% purity) in DCM (25 mL) was added tribromoborane in DCM (1 M, 26 mmol, 26 mL) dropwise. The reaction mixture was stirred at RT for 45 min, then concentrated to dryness. The crude product was taken in DCM and placed in an ice bath and MeOH was added carefully (exotherm). The mixture was concentrated to dryness then co-evaporated twice with MeOH. The residue was triturated with saturated aqueous NaHCO3. The solids were collected by filtration on a Buchner funnel, washed with H2O and air-dried. The still wet solid was dissolved in DCM/MeOH, concentrated to dryness and triturated in 20% MeOH/DCM (50 mL). The solid was collected by filtration, washed with 20% MeOH/DCM, air-dried then dried in vacuo to afford 2-amino-1-(3-hydroxy-2,6-dimethyl-phenyl)-5,6-dimethyl-pyrrolo[2,3-b]pyridine-3-carboxamide (1.60g, 75% yield) as a light beige solid. MS: [M+1]: 325.1. A different batch was purified by preparative HPLC to yield 2-amino-1-(3-hydroxy-2,6-dimethyl-phenyl)-5,6-dimethyl-pyrrolo[2,3-b]pyridine-3-carboxamide (63% yield) as an off-white fluffy solid.
1H NMR (400 MHz, DMSO-d6) δ 9.51 (s, 1H), 7.82 (s, 1H), 7.05 (d, J = 8.3 Hz, 1H), 6.90 (d, J =
8.2 Hz, 1H), 6.71 (br s, 2H), 6.64 (br s, 2H), 2.26 (s, 3H), 2.23 (s, 3H), 1.74 (s, 3H), 1.65 (s, 3H). MS: [M+1]: 325.1.
Chiral SFC separation of Compound 181 (1.60g, 4.93 mmol) (Instrument: Waters Prep 100 SFC-MS; Column: Phenomenex Lux Cellulose-2, 30 x 250 mm, 5 μm; Conditions: isocratic at 55% IPA + 10mM Ammonium Formate with 45% CO2 ; Flow Rate: 70 mL/min) provided
Compound 182 and Compound 183.

Compound 182 from SFC separation of 181. Peak 1 (retention time 3.94 min, 99.86%): (S)-2- amino-1-(3-hydroxy-2,6-dimethyl-phenyl)-5,6-dimethyl-pyrrolo[2,3-b]pyridine-3-carboxamide (381 mg) was obtained as an off white fluffy solid. 1H NMR (400 MHz, DMSO-d6) δ 9.50 (s, 1H), 7.83 (s, 1 H), 7.05 (d, J = 8.3 Hz, 1H), 6.90 (d, J = 8.3 Hz, 1H), 6.72 (s, 2H), 6.65 (s, 2H), 2.26 (s, 3H), 2.24 (s, 3H), 1.74 (s, 3H), 1.65 (s, 3H). MS: [M+1]: 325.1.

Compound 183 from SFC separation of 181. Peak 2 (retention time 4.35 min, 98.09%): (R)-2- amino-1-(3-hydroxy-2,6-dimethyl-phenyl)-5,6-dimethyl-pyrrolo[2,3-b]pyridine-3-carboxamide (495 mg) was obtained as an off white fluffy solid. 1H NMR (400 MHz, DMSO-d6) δ 9.50 (s, 1H), 7.83 (s, 1 H), 7.05 (d, J = 8.2 Hz, 1H), 6.90 (d, J = 8.2 Hz, 1H), 6.72 (s, 2H), 6.66 (s, 2H), 2.26 (s, 3H), 2.24 (s, 3H), 1.74 (s, 3H), 1.65 (s, 3H). MS: [M+1]: 325.1.
SYN
https://pubs.acs.org/doi/full/10.1021/acs.oprd.4c00493

REF
- The Science and Art of Structure-Based Virtual ScreeningPublication Name: ACS Medicinal Chemistry LettersPublication Date: 2024-03-25PMCID: PMC11017385PMID: 38628791DOI: 10.1021/acsmedchemlett.4c00093
- Discovery of an Orally Bioavailable and Selective PKMYT1 Inhibitor, RP-6306Publication Name: Journal of Medicinal ChemistryPublication Date: 2022-07-26PMCID: PMC9837800PMID: 35880755DOI: 10.1021/acs.jmedchem.2c00552
- CCNE1 amplification is synthetic lethal with PKMYT1 kinase inhibitionPublication Name: NaturePublication Date: 2022-04-20PMCID: PMC9046089PMID: 35444283DOI: 10.1038/s41586-022-04638-9
- Contributions in the domain of cancer research: Review¶Negative regulators of cyclin-dependent kinases and their roles in cancersPublication Name: Cellular and molecular life sciences : CMLSPublication Date: 2001-11PMCID: PMC11337304PMID: 11766887DOI: 10.1007/pl00000826



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
| Clinical data | |
|---|---|
| Other names | RP-6306 |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2719793-90-3 |
| PubChem CID | 156869388 |
| ChemSpider | 115008046 |
| UNII | N95U3A7N57 |
| KEGG | D12736 |
| ChEMBL | ChEMBL5199076 |
| Chemical and physical data | |
| Formula | C18H20N4O2 |
| Molar mass | 324.384 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
References
- Szychowski J, Papp R, Dietrich E, Liu B, Vallée F, Leclaire ME, et al. (August 2022). “Discovery of an Orally Bioavailable and Selective PKMYT1 Inhibitor, RP-6306”. Journal of Medicinal Chemistry. 65 (15): 10251–10284. doi:10.1021/acs.jmedchem.2c00552. PMC 9837800. PMID 35880755.
- Previtali V, Bagnolini G, Ciamarone A, Ferrandi G, Rinaldi F, Myers SH, et al. (July 2024). “New Horizons of Synthetic Lethality in Cancer: Current Development and Future Perspectives”. Journal of Medicinal Chemistry. 67 (14): 11488–11521. doi:10.1021/acs.jmedchem.4c00113. PMC 11284803. PMID 38955347.
///////lunresertib, Serine/ threonine kinase inhibitor, antineoplastic, N95U3A7N57, RP-6306, RP 6306
Lomedeucitinib



Lomedeucitinib
CAS 2328068-29-5
MF C18H172H3N6O4S
MW 419.5 g/mol

4-{[3-(methanesulfonyl)pyridin-2-yl]amino}-N-(2H3)methyl-6-[(1R)-spiro[2.2]pentane-1-carboxamido]pyridazine-3-carboxamide
4-[(3-methylsulfonyl-2-pyridinyl)amino]-6-[[(2R)-spiro[2.2]pentane-2-carbonyl]amino]-N-(trideuteriomethyl)pyridazine-3-carboxamide
Janus kinase inhibitor, anti-inflammatory, BMS-986322, BMS 986322, EYQ7KA55XA
Lomedeucitinib is an investigational new drug that is being evaluated for the treatment of psoriasis and psoriatic arthritis. It is a tyrosine kinase 2 (TYK2) inhibitor.[1]
- A Study to Evaluate Effectiveness and Safety of BMS-986322 in Participants With Moderate-to-Severe PsoriasisCTID: NCT05730725Phase: Phase 2Status: CompletedDate: 2024-09-19
- A Study to Evaluate the Drug Levels, Metabolism, and Removal of BMS-986322 in Healthy Adult Male ParticipantsCTID: NCT06088264Phase: Phase 1Status: CompletedDate: 2024-03-29
- A Study Investigating Interactions Between BMS-986322 and Rosuvastatin, Metformin and Methotrexate in Healthy ParticipantsCTID: NCT05615012Phase: Phase 1Status: CompletedDate: 2024-03-27
- A Study to Investigate the Interaction of BMS-986322 and a Combined Oral Hormonal Contraceptive (Ethinyl Estradiol [EE]/Norethindrone [NET]) in Healthy Female ParticipantsCTID: NCT05579574Phase: Phase 1Status: CompletedDate: 2023-08-18
- A Study to Assess the Safety and Tolerability of BMS-986322 in Healthy Participants of Japanese DescentCTID: NCT05546151Phase: Phase 1Status: CompletedDate: 2023-06-22
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=US333829535&_cid=P10-MHIXWK-98212-1
General Scheme for Examples 252 and 253:

Example 252
Step 1

| A mixture of cesium carbonate (149 mg, 0.457 mmol), Xantphos (14.43 mg, 0.025 mmol), Pd 2(dba) 3 (11.42 mg, 0.012 mmol), 6-chloro-N-(methyl-d3)-4-((3-(methylthio)pyridin-2-yl)amino)pyridazine-3-carboxamide (65 mg, 0.208 mmol), and (R)-spiro[2.2]pentane-1-carboxamide (50.8 mg, 0.457 mmol) in dioxane (3 mL) was degassed using a vacuum/N2 fill cycle three times. The reaction was heated at 110° C. for 16 hours. The reaction was diluted with water and DCM. The DCM layer was separated and washed two more times with water and then dried (Na 2SO 4), filtered and concentrated. Purification via automated flash chromatography, eluting with methanol in DCM from 0 to 10%, gave the title compound (R)—N-(methyl-d3)-4-((3-(methylthio)pyridin-2-yl)amino)-6-(spiro[2.2]pentane-1-carboxamido)pyridazine-3-carboxamide (54 mg, 67% yield). 1H NMR (400 MHz, CHLOROFORM-d) δ 12.15 (br s, 1H), 9.88 (s, 1H), 8.68 (br s, 1H), 8.36 (br d, J=3.5 Hz, 1H), 8.25 (br s, 1H), 7.72 (br d, J=7.4 Hz, 1H), 6.97 (br dd, J=7.0, 5.1 Hz, 1H), 2.51 (s, 3H), 2.21-2.09 (m, 1H), 1.58-1.10 (m, 6H), 1.08-0.93 (m, 5H). |
| LCMS (ESI) m/e 388.1 [(M+H) +, calc’d C 18H 18D 3N 6O 2S 1, 388.1]; LC/MS retention time (method D): t R=0.80 min. |
Step 2

To a suspension of hydrogen peroxide (30% solution in water, 0.258 mL, 2.52 mmol) and (R)—N-(methyl-d3)-4-((3-(methylthio)pyridin-2-yl)amino)-6-(spiro[2.2]pentane-1-carboxamido)pyridazine-3-carboxamide (0.0489 g, 0.126 mmol) in AcOH (1 mL) was added sodium tungstate dihydrate (0.042 g, 0.126 mmol) at room temperature. After stirring at room temperature for 1 hour, the reaction was diluted with water, basified with Na 2CO 3 powder and extracted three times with DCM. The DCM layers were combined, washed with Na 2S 2O 3 (5% solution), dried (Na 2SO 4), filtered and concentrated. The crude product was purified using reverse phase prepHPLC to give the title compound (R)—N-(methyl-d3)-4-((3-(methylsulfonyl)pyridin-2-yl)amino)-6-(spiro[2.2]pentane-1-carboxamido)pyridazine-3-carboxamide (16.2 mg, 31%) as a colorless solid. 1H NMR (500 MHz, DMSO-d 6) δ 12.07 (s, 1H), 11.22 (s, 1H), 9.49 (s, 1H), 9.16 (s, 1H), 8.63 (dd, J=4.6, 1.5 Hz, 1H), 8.29 (dd, 0.1=7.8, 1.4 Hz, 1H), 7.34 (dd, 0.1=7.8, 4.7 Hz, 1H), 2.48-2.43 (m, 1H), 1.46-1.41 (m, 1H), 1.42-1.36 (m, 1H), 0.95-0.82 (m, 3H), 0.80-0.73 (m, 1H). (3H methyl sulfone was buried under DMSO peak). LCMS (ESI) m/e 420.0 [(M+H) +, calc’d C 18H 18D 3N 6O 4S, 420.1]; LC/MS retention time (method E): t R=1.38 min; OR: −205.39 (20° C.).
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=US242383764&_cid=P10-MHIXVD-97150-1
PAT
- Sulfone pyridine alkyl amide-substituted heteroaryl compoundsPublication Number: US-11787779-B2Priority Date: 2017-11-21Grant Date: 2023-10-17
- Sulfone pyridine alkyl amide-substituted heteroaryl compoundsPublication Number: US-2024002364-A1Priority Date: 2017-11-21
- Sulfone pyridine alkyl amide-substituted heteroaryl compoundsPublication Number: KR-102702228-B1Priority Date: 2017-11-21Grant Date: 2024-09-02
- Sulfone pyridine alkyl amide-substituted heteroaryl compoundsPublication Number: NZ-805343-APriority Date: 2017-11-21
- Sulfonepyridine alkylamide-substituted heteroaryl compoundsPublication Number: JP-2023098942-APriority Date: 2017-11-21
- Sulfone pyridine alkyl amide-substituted heteroaryl compoundsPublication Number: AU-2023255024-A1Priority Date: 2017-11-21
- Heteroaryl compounds substituted with sulfone pyridinylalkylamidesPublication Number: CN-111315737-BPriority Date: 2017-11-21Grant Date: 2024-06-18
- The heteroaryl compounds are substituted with sulfone-pyridine alkyl amidesPublication Number: IL-274816-B2Priority Date: 2017-11-21
- Sulfonepyridine alkylamide substituted heteroaryl compoundsPublication Number: JP-7490107-B2Priority Date: 2017-11-21Grant Date: 2024-05-24
- Sulfone pyridine alkyl amide-substituted heteroaryl compoundsPublication Number: AU-2022228101-A1Priority Date: 2017-11-21
- Sulfone pyridine alkyl amide-substituted heteroaryl compoundsPublication Number: TW-I776994-BPriority Date: 2017-11-21Grant Date: 2022-09-11
- Sulfonepyridine alkylamide-substituted heteroaryl compoundsPublication Number: JP-7258903-B2Priority Date: 2017-11-21Grant Date: 2023-04-17
- Sulfone pyridine alkyl amide-substituted heteroaryl compoundsPublication Number: AU-2022228101-B2Priority Date: 2017-11-21Grant Date: 2023-08-03
- The heteroaryl compounds are substituted with sulfone-pyridine alkyl amidesPublication Number: IL-274816-B1Priority Date: 2017-11-21
- Sulfone pyridine alkyl amide-substituted heteroaryl compoundsPublication Number: US-2019152948-A1Priority Date: 2017-11-21
- Sulfone pyridine alkyl amide-substituted heteroaryl compoundsPublication Number: CA-3083122-A1Priority Date: 2017-11-21
- Sulfone pyridine alkyl amide-substituted heteroaryl compoundsPublication Number: KR-20200089706-APriority Date: 2017-11-21
- Sulfone pyridine alkyl amide-substituted heteroaryl compoundsPublication Number: US-11021462-B2Priority Date: 2017-11-21Grant Date: 2021-06-01
- Sulfone pyridine alkyl amide-substituted heteroaryl compoundsPublication Number: US-2021253554-A1Priority Date: 2017-11-21



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
| Clinical data | |
|---|---|
| Other names | BMS-986322 |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2328068-29-5 |
| PubChem CID | 138620496 |
| IUPHAR/BPS | 13210 |
| UNII | EYQ7KA55XA |
| KEGG | D12725 |
| ChEMBL | ChEMBL5314608 |
| Chemical and physical data | |
| Formula | C18H17D3N6O4S |
| Molar mass | 419.47 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
References
- Ahsan S, Degener R, Schlamp M (2024). “Non-Invasive Treatments Invade the Psoriasis Pipeline”. Drugs in Context. 13: 2024–5–6. doi:10.7573/dic.2024-5-6. PMC 11313207. PMID 39131603.
////////lomedeucitinib, Janus kinase inhibitor, anti-inflammatory, BMS-986322, BMS 986322, EYQ7KA55XA
Linustedastat




Linustedastat
CAS 2254299-48-2
MFC26H29F2N3O2 MW 453.5 g/mol
FOR-6219, OG-6219, FOR 6219, OG 6219, PP3PLL7GZY, Phase 2, Endometriosis
3-[(8R,9S,13S,14S,15R,17E)-4-fluoro-17-hydroxyimino-13-methyl-7,8,9,11,12,14,15,16-octahydro-6H-cyclopenta[a]phenanthren-15-yl]-N-(5-fluoro-2-pyridinyl)propanamide
- (15beta,17E)-4-Fluoro-N-(5-fluoro-2-pyridinyl)-17-(hydroxyimino)estra-1,3,5(10)-triene-15-propanamide
- 3-[(17E)-4-fluoro-17-(hydroxyimino)estra-1,3,5(10)-trien-15beta-yl]-N-(5-fluoropyridin-2-yl)propanamide
- Estra-1,3,5(10)-triene-15-propanamide, 4-fluoro-N-(5-fluoro-2-pyridinyl)-17-(hydroxyimino)-, (15beta,17E)-
3-[(17E)-4-fluoro-17-(hidroxiimino)estra-1,3,5(10)-trien-15β-il]-N-(5-fluoropiridin-2-il)propanamida
inhibidor de la hidroxiesteroide 17-beta deshidrogenasa 1(HSD17B1)
- OriginatorHormos Medical; Solvay Pharmaceuticals B.V.; University of Turku
- DeveloperOrganon
- ClassSmall molecules
- Mechanism of ActionEstradiol dehydrogenase inhibitors
- Phase IIEndometriosis
- 02 Jul 2025Efficacy data from the phase II ELENA trial in Endometriosis released by Organon
- 28 May 2025Organon completes a phase-II clinical trials in Endometriosis (In adults) in Latvia, Sweden, Poland, Italy, France, Hungary, Germany, Czech Republic, Czech Republic, Bulgaria, Belgium, USA (PO) (NCT05560646)
- 28 Nov 2023No recent reports of development identified for phase-I development in Endometriosis(In volunteers) in United Kingdom (PO)
Linustedastat (developmental code names FOR-6219 and OG-6219) is a 17β-hydroxysteroid dehydrogenase 1 (17β-HSD1; HSD17B1) inhibitor which is under development for the treatment of endometriosis.[1][2][3][4][5] It is a steroidal compound derived from estrone and works by preventing the formation of the more potent estrogen estradiol from the minimally active precursor estrone.[1][2][5] This in turn results in antiestrogenic effects that may be useful in the treatment of estrogen-dependent conditions.[1][2][5] As of November 2023, the drug is in phase 2 clinical trials for endometriosis.[1][2] It is also under preclinical investigation for treatment of breast cancer and endometrial cancer.[5]
A Study to Investigate Efficacy and Safety of OG-6219 BID in 3 Dose Levels Compared With Placebo in Participants Aged 18 to 49 With Moderate to Severe Endometriosis-related Pain
CTID: NCT05560646
Phase: Phase 2
Status: Completed
Date: 2025-05-29
Pat
WO2018224736
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2018224736&_cid=P21-MHFVBM-49409-1


Compound 26
3-((13S,15R,E)-3-fluoro-17-(hydroxyimino)-13-methyl-7,8,9,11,12,13,14,15,16,17-decahydro-6H-cyclopenta[a]phenanthren-15-yl)-N-(5-fluoropyridin-2-yl)propanamide

Example 26 was prepared in 94% yield from the compound 25 by the same method as with Example 2 in three hours reaction time.
1H NMR (200 MHz, DMSO-d6): 1.03 (s, 3 H), 1.12 – 2.48 (m, 15 H), 2.57 – 2.78 (m, 1 H), 2.80 – 2.95 (m, 2 H), 6.79 – 7.01 (m, 2 H), 7.18 – 7.38 (m, 1 H), 7.72 (td, 1 H), 8.15 (dd, 1 H), 8.31 (d, 1 H), 10.18 (s, 1 H), 10.64 (s, 1 H). MS m/z (TOF ES+): 454 (M+1).
SYNTHESIS




AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
| Clinical data | |
|---|---|
| Other names | FOR-6219; OG-6219; 3-[(17E)-4-Fluoro-17-(hydroxyimino)estra-1,3,5(10)-trien-15β-yl]-N-(5-fluoropyridin-2-yl)propanamide |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2254299-48-2 |
| PubChem CID | 171390018 |
| UNII | PP3PLL7GZY |
| KEGG | D13078 |
| Chemical and physical data | |
| Formula | C26H29F2N3O2 |
| Molar mass | 453.534 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
References
- “FOR 6219”. AdisInsight. 28 November 2023. Retrieved 15 August 2024.
- “Delving into the Latest Updates on Linustedastat with Synapse”. Synapse. 3 August 2024. Retrieved 15 August 2024.
- Barra F, Romano A, Grandi G, Facchinetti F, Ferrero S (June 2019). “Future directions in endometriosis treatment: discovery and development of novel inhibitors of estrogen biosynthesis”. Expert Opin Investig Drugs. 28 (6): 501–504. doi:10.1080/13543784.2019.1618269. hdl:11380/1201688. PMID 31072144.
- Perrone U, Evangelisti G, Laganà AS, Bogliolo S, Ceccaroni M, Izzotti A, Gustavino C, Ferrero S, Barra F (December 2023). “A review of phase II and III drugs for the treatment and management of endometriosis”. Expert Opin Emerg Drugs. 28 (4): 333–351. doi:10.1080/14728214.2023.2296080. PMID 38099328.
- Rižner TL, Romano A (2023). “Targeting the formation of estrogens for treatment of hormone dependent diseases-current status”. Front Pharmacol. 14 1155558. doi:10.3389/fphar.2023.1155558. PMC 10175629. PMID 37188267.
Several compounds with inhibitory action on the enzyme HSD17B1 have been developed and one steroidal compound, a competitive HSD17B1 inhibitor (OG-6219) recently entered the clinical phase for endometriosis […] and it is in the preclinical phase for endometrial and breast cancer (Husen et al., 2006a; Husen et al., 2006b; Konings et al., 2018b; Jarvensivu et al., 2018; Xanthoulea et al., 2021). […] Only the C15 estrone derivative developed by Organon Finland, former Forendo pharma (compound FOR-6219/OR-6219) reached the clinical phase for endometriosis with three clinical trials registered in the database Clinical Trails (Table 2). Phase 1 and 1b trials NCT04686669 and NCT03709420 determined the bio-availability of the compound administered orally as gelatine capsule in 12 subjects (NCT04686669) and then the safety, tolerability, food interactions, the pharmacokinetics and pharmacodynamics of escalating doses of the drug in 87 subjects (NCT03709420). The phase 2 randomized, double-blind, Elena study (NCT05560646) is currently recruiting patients and aims at evaluating the efficacy and safety of OG-6219 in women with moderate to severe endometriosis […]
External links
//////////Linustedastat, FOR-6219, OG-6219, FOR 6219, OG 6219, PP3PLL7GZY, Phase 2, Endometriosis
Imocitrelvir




Imocitrelvir
CAS 343565-99-1
MFC26H29N5O7 MW523.5 g/mol
ethyl (2E,4S)-4-{(2S)-2-[3-(5-methyl-1,2-oxazole-3-carboxamido)-2-oxopyridin-1(2H)-yl]pent-4-ynamido}-5-[(3S)-2-oxopyrrolidin-3-yl]pent-2-enoate
ethyl (E,4S)-4-[[(2S)-2-[3-[(5-methyl-1,2-oxazole-3-carbonyl)amino]-2-oxo-1-pyridinyl]pent-4-ynoyl]amino]-5-[(3S)-2-oxopyrrolidin-3-yl]pent-2-enoate
protease inhibitor, antiviral, AG-7404, V-7404, AG 7404, V 7404, VQ1AN3OO42
Imocitrelvir is an investigational new drug that is being evaluated for the treatment of viral infections. It is a 3C protease inhibitor in picornaviruses. Originally developed by Pfizer for treating human rhinovirus infections,[1] this small molecule has shown promise against a broader range of viruses, including polioviruses.[2][3]
SYN
Publication Name: Journal of Medicinal Chemistry
Publication Date: 2003-09-17
PMID: 14521419
DOI: 10.1021/jm030166l
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2016044656&_cid=P21-MHBDH2-20719-1
PAT
PAT
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2001040189&_cid=P21-MHBDI9-21481-1





EXAMPLE 21
Preparation of Compound 22: tra«5-(4S,3″”S)-4-(2′-{3″-[(5′”-Methylisoxazole-3′”-carbonyl)amino]-2″-oxo-2″H-pyridin- 1 “-yl} acetylamino)-5-(2″”-oxopyrrilidin-3″”-yl)pent-2-enoic Acid Ethyl Ester

Preparation of Intermediate {3-[(5′-Methylisoxazole-3′-carbonyl)amino]-2-oxo-2H-pyridin-l-yl} acetic Acid tert-Butyl Ester
To a solution of 5-methylisoxazole-3-carboxylic acid (2′-hydroxy-4′-methylpyridin-3′-yl)amide (F2, Example 19) (0.520 g, 2.37 mmol, 1 equiv) in TΗF (20 mL) at 0 °C was added NaΗ (0.095 g, 2.37 mmol, 1.0 equiv). The resulting mixture was stirred at 0 °C for 20 min, and then t-butyl bromoacetate (0.385 mL, 2.61 mmol, 1.1 equiv) was added. The reaction mixture was stirred and warmed to room temperature for 30 min, then was partitioned between 0.5 N ΗC1 (100 mL) and EtOAc (2 x 100 mL). The combined organic layers were dried over Na2SO and were concentrated. Purification of the residue by flash column chromatography (30% EtOAc in hexanes) provided the title intermediate (0.628 g, 79%) as a white solid: IR (cm-1) 3343, 1743, 1651, 1581, 1156; Η NMR (CDC13) δ 1.52 (s, 9H), 2.53 (s, 3H), 4.65 (s, 2H), 6.32 (t, 1H, 7= 7.2), 6.51 (s, IH), 7.01 (dd, 1H, 7= 6.9, 1.8), 8.50 (dd, 1H, 7= 7.5, 1.8), 9.63 (s, br. IH); Anal. C16H19N3O5: C, H, N.
Preparation of Compound 22
The preceding intermediate was transformed into Compound 22 by a process that was analogous to that described in Example 25 for the transformation of V3 to product R3: mp = 102-106 °C; IR (cm”1) 3336, 1684, 1534, 1457; JH NMR (CDCI3) δ 1.27 (t, 3H, 7= 7.2), 1.67-1.75 (m, IH), 1.98-2.09 (m, IH), 2.37-2.49 (m, IH), 2.53 (s, 3H), 2.55-2.61 (m, IH), 3.34-3.46 (m, 2H), 3.51-3.52 (m, IH), 4.17 (q, 2H, 7= 7.2), 4.61-4.78 (m, 3H), 5.98 (dd, IH, 7 = 15.6, 1.5), 6.20 (s, br. IH), 6.35 (t, 1H, 7= 7.8), 6.51 (s, IH), 6.85 (dd, IH, 7= 15.6, 5.1), 7.17 (d, IH, 7= 7.2), 8.33 (d, IH, 7= 7.2), 8.49 (d, IH, 7= 7.5), 9.57 (s, br. IH); Anal.
C23H27N5O7: C, H, N.
EXAMPLE 24
Preparation of Compound 25: trans-(2’S,3″”‘S,4S)-4-(3,-(4″-Fluorophenyl)-2′-{3″‘-[(5″”-methylisoxazole-3″”-carbonyl)amino]-2′”-oxo-2′”H-pyridin- “-yl}propionylamino)-5-(2″ oxopyrrolidin-3′””-yl)pent-2-enoic Acid Ethyl Ester

The title compound was prepared from F2 (Example 19) in a manner analogous to that described for the conversion of U2 to 13 in Example 23 utilizing intermediate Y2 (Example 25) where appropriate: IR (cm-1) 3331, 1690, 1590, 1531, 1455; !H NMR (CDCI3) δ 1.30 (t, 3H, 7= 7.0), 1.45-1.55 (m, IH), 1.64-1.75 (m, IH), 2.03-2.31 (m, 3H), 2.49 (s, 3H), 3.10 (dd, IH, 7= 13.7, 7.9), 3.20-3.46 (m, 3H), 4.20 (q, 2H, 7= 7.0), 4.36-4.47 (m, IH), 5.67 (dd, IH, 7 = 15.7, 1.4), 5.85-5.92 (m, IH), 6.29 (t, 1H, 7= 7.2), 6.45 (s, IH), 6.70 (dd, IH, 7= 15.7, 5.7), 6.86 (s, IH), 6.90-6.97 (m, 2H), 7.10-7.16 (m, 2H), 7.60 (dd, IH, 7= 7.2, 1.6), 8.37 (dd, IH, 7 = 7.2, 1.6), 8.51 (d, IH, 7= 6.6), 9.47 (s, IH).
EXAMPLE 25
Preparation of Compound 26: tr_.«5-(2’S,3″”S,4S)-4-(2′-{3″-[(5″‘-Methyl-isoxazole-3′”-carbonyl)amino]-2″-oxo-2″H-pyridin-l”-yl}butyrylamino)-5-(2″”-oxopyrrolidin-3″”-yl)pent-2-enoic Acid Ethyl Ester (R3)

Preparation of Intermediate (2R)-2-Trifluoromethanesulfonyl-oxybutyric acid tert-butyl ester (U3)
Commercially available T3 (0.575 g, 3.59 mmol, 1 equiv) was dissolved in CH2CI2 (25 mL) and cooled in an ice bath. 2,6-Lutidine (0.836 mL, 7.18 mmol, 2 equiv) and trifluoromethanesulfonic anhydride (1.15 mL, 6.84 mmol, 1.9 equiv) were added and the reaction mixture was stirred 30 min. It was then diluted with MTBE (400 mL), washed with a mixture of brine and 1 N HCl (2:1, 100 mL) and brine (100 mL), dried over Na2SO4 and evaporated to provide the title intermediate which was used without further purification.
Preparation of Intermediate (2S)-2- {3′-[(5″-Methylisoxazole-3″-carbonyl)amino]-2′-oxo-2’H-pyri din- l’-yl} butyric Acid tert-Butyl Ester (V3)
Intermediate F2 from above (0.200 g, 0.912 mmol, 1.1 equiv) was suspended in TΗF (6 mL). Sodium hydride (60% dispersion in mineral oil, 0.0332 g, 0.830 mmol, 1 equiv) was added in one portion. After stirring 30 min, a solution of intermediate U3 (0.830 mmol, 1 equiv, based on T3) in TΗF (7 mL) was added dropwise. The resulting mixture was stirred 2 hours, then diluted with EtOAc (200 mL) and washed with brine (2 x 50 mL). The organic phase was dried over MgSO4 and evaporated. The residue was purified by flash column chromatography (25% EtOAc in hexanes) to provide the title intermediate (0.178 g, 59%) as an oil: R/= 0.30 (25% EtOAc in hexanes); IR (cm”1) 3331, 1731, 1690, 1649, 1602, 1531 ; *Η NMR (CDCI3) δ 0.93 (t, 3H, 7= 7.3), 1.45 (s, 9H), 1.83-2.01 (m, IH), 2.17-2.31 (m, IH), 2.50 (s, 3H), 5.44-5.51 (m, IH), 6.32 (t, IH, 7= 7.2), 6.48 (s, IH), 7.10 (dd, IH, 7= 7.2, 1.8), 8.45 (dd, 1H, 7= 7.2, 1.8), 9.64 (s, IH); Anal. C18H23N3O5: C, H, N.
Preparation of Intermediate (2S)-2- {3′-[(5″-Methylisoxazole-3″-carbonyl)amino]-2′-oxo-2’H-pyridin-l’-yl}butyric Acid (W3)
Intermediate V3 from above (0.143 g, 0.397 mmol, 1 equiv) was stirred for 1 h in a solution of TFA (2 mL) in CΗ2CI2 (3 mL). The volatiles were evaporated. The residue was suspended in toluene (10 mL) and concentrated to dryness, providing the title intermediate which was used without further purification.
Preparation of Intermediate trα«5-(3’S,4S)-4-Amino-5-(2′-oxopyrrolidin-3′-yl)pent-2-enoic Acid Ethyl Ester (Y2)
Intermediate X2, prepared according to the method disclosed in the co-pending application, U.S. Provisional Patent Application No. 60/150,358, filed August 24, 1999(0.130 g, 0.398 mmol, 1 equiv), was stirred for 30 min in a solution of TFA (2 mL) in CH2CI2 (3 mL). The volatiles were evaporated. The residue was suspended in toluene (10 mL) and concentrated to dryness, providing the title intermediate which was used without further purification.
Preparation of Product R3 (Compound 26)
Intermediates W3 and Y2 (as prepared above) were combined in CH2CI2 (7 mL) and cooled in an ice bath. HOBt (0.064 g, 0.47 mmol, 1.2 equiv), iP^NEt (0.484 mL, 2.78 mmol, 7 equiv) and EDC (0.084 g, 0.44 mmol, 1.1 equiv) were added sequentially. The reaction mixture was allowed to warm to 23 °C overnight, then diluted with EtOAc (500 mL) and washed with 5% KHSO4 , half saturated NaHCO3, and brine (100 mL each). The organic phase was dried over MgSO4 and evaporated. The residue was purified by flash column chromatography (gradient elution, 2→3% CH3OH in CH2CI2) to provide the title intermediate (0.119 g, 58%) as a white foam: IR (cm”1) 3331, 1684, 1649, 1590, 1531; JH NMR (CDCI3) δ 0.92 (t, 3H, J = 7.3), 1.29 (t, 3H, J = 7.1), 1.47-1.58 (m, IH), 1.62-1.77 (m, IH), 1.85-2.00 (m, IH), 2.08-2.33 (m, 4H), 2.49 (s, 3H), 3.25-3.42 (m, 2H), 4.19 (q, 2H, J = 7.1), 4.39-4.50 (m, IH), 5.73 (dd, IH, J = 8.8, 6.8), 5.97 (dd, IH, J = 15.7, 1.4), 6.34 (t, IH, J = 7.2), 6.46 (s, IH), 6.86 (dd, IH, J = 15.7, 5.9), 7.18 (s, IH), 7.59 (dd, IH, J = 7.2, 1.8), 8.42 (dd, IH, J = 7.2, 1.8), 8.58-8.62 (m, IH), 9.56 (s, 1); Anal. C25H31N5O7O.5OH2O: C, H, N.
PAT
- Treatment of infection by human enterovirus d68Publication Number: US-2020016243-A1Priority Date: 2014-09-17
- Treatment of infection by human enterovirus d68Publication Number: WO-2016044656-A1Priority Date: 2014-09-17
- Treatment of infection by human enterovirus d68Publication Number: US-2021052708-A1Priority Date: 2014-09-17
- Treatment of infection by human enterovirus D68Publication Number: US-11191817-B2Priority Date: 2014-09-17Grant Date: 2021-12-07
- Therapeutic compounds and methodsPublication Number: US-2025051283-A1
- Protease Inhibitors for Treatment or Prevention of Coronavirus DiseasePublication Number: US-2023192660-A1Priority Date: 2020-05-08
- Composition and combined medication method for treating enterovirus infectionPublication Number: US-2019030027-A1Priority Date: 2016-01-29
- Composition and combined medication method for treating enterovirus infectionPublication Number: US-10864210-B2Priority Date: 2016-01-29Grant Date: 2020-12-15
- Treatment of infection by human enterovirus D68Publication Number: US-10328128-B2Priority Date: 2014-09-17Grant Date: 2019-06-25
- Treatment of infection by human enterovirus d68Publication Number: US-2017290893-A1Priority Date: 2014-09-17
- Nucleotide and nucleoside therapeutic compositions, combinations and related uses thereofPublication Number: CN-117881402-APriority Date: 2021-05-05
- Nucleotide and nucleoside therapeutic compositions, combinations and uses related theretoPublication Number: EP-4333859-A1Priority Date: 2021-05-05
- Nucleotide and nucleoside therapeutic compositions, combinations, and related usesPublication Number: JP-2024517807-APriority Date: 2021-05-05
- Nucleotide and nucleoside therapeutic compositions, combinations and uses related theretoPublication Number: WO-2022235874-A1Priority Date: 2021-05-05
- Protease inhibitors for treatment or prevention of coronavirus diseasePublication Number: EP-4146267-A1Priority Date: 2020-05-08
- 4′-substituted nucleosides and nucleotides as antiviral agentsPublication Number: WO-2024227159-A2Priority Date: 2023-04-28
- Therapeutic compoundsPublication Number: WO-2024206284-A2Priority Date: 2023-03-27
- Antibody molecules binding to sars-cov-2Publication Number: WO-2024168061-A2Priority Date: 2023-02-07
- Predictive model for variants associated with drug resistance and theranostic applications thereofPublication Number: WO-2023172635-A1Priority Date: 2022-03-08
- Nucleotide and nucleoside therapeutic compositions, combinations and uses related theretoPublication Number: CA-3216679-A1Priority Date: 2021-05-05
LIT
- Structure and inhibition of SARS-CoV-1 and SARS-CoV-2 main proteases by oral antiviral compound AG7404Publication Name: Antiviral ResearchPublication Date: 2022-12PMCID: PMC9632241PMID: 36336176DOI: 10.1016/j.antiviral.2022.105458
- Bioisosteres of the Phenyl Ring: Recent Strategic Applications in Lead Optimization and Drug DesignPublication Name: Journal of Medicinal ChemistryPublication Date: 2021-09-30PMID: 34591488DOI: 10.1021/acs.jmedchem.1c01215
- In Vitro Antiviral Activity of New Oxazoline Derivatives as Potent Poliovirus InhibitorsPublication Name: Journal of Medicinal ChemistryPublication Date: 2018-12-04PMCID: PMC9169555PMID: 30512950DOI: 10.1021/acs.jmedchem.8b01482
- A Novel Series of Highly Potent Small Molecule Inhibitors of Rhinovirus ReplicationPublication Name: Journal of Medicinal ChemistryPublication Date: 2017-06-15PMID: 28581749DOI: 10.1021/acs.jmedchem.7b00175
- Anti-poliovirus activity of protease inhibitor AG-7404, and assessment of in vitro activity in combination with antiviral capsid inhibitor compoundsPublication Name: Antiviral ResearchPublication Date: 2013-05PMID: 23499651DOI: 10.1016/j.antiviral.2013.03.003



AS ON JUNE2025 4.45 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
| Clinical data | |
|---|---|
| Other names | AG-7404, V-7404 |
| Identifiers | |
| IUPAC name | |
| CAS Number | 343565-99-1 |
| PubChem CID | 5280053 |
| IUPHAR/BPS | 13223 |
| UNII | VQ1AN3OO42 |
| ChEMBL | ChEMBL141157 |
| Chemical and physical data | |
| Formula | C26H29N5O7 |
| Molar mass | 523.546 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
References
- “Imocitrelvir”. PatSnap.
- Xie H, Rhoden EE, Liu HM, Ogunsemowo F, Mainou BA, Burke RM, et al. (November 2024). “Antiviral Development for the Polio Endgame: Current Progress and Future Directions”. Pathogens. 13 (11). Basel, Switzerland: 969. doi:10.3390/pathogens13110969. PMC 11597170. PMID 39599522.
- Bandyopadhyay AS, Burke RM, Hawes KM (June 2024). “Polio Eradication: Status, Struggles and Strategies”. The Pediatric Infectious Disease Journal. 43 (6): e207-211. doi:10.1097/INF.0000000000004330. PMID 38564755.
////////Imocitrelvir, protease inhibitor, antiviral, AG-7404, V-7404, AG 7404, V 7404, VQ1AN3OO42
Ilantimod




Ilantimod
CAS 2242464-44-2
MF C18H18ClN5O3 MW 387.82
6-(4-chlorophenyl)-N-[(2S)-1-hydroxypropan-2-yl]-2-(1-methyl-1H-pyrazol-4-yl)-3-oxo-2,3-dihydropyridazine-4-carboxamide
(S)-6-(4-chlorophenyl)-N-(1-hydroxypropan-2-yl)-2-(1-methyl-1H-pyrazol-4-yl)-3-oxo-2,3-dihydropyridazine-4-carboxamide
immunomodulator, BAY-2416964, BAY 2416964, Y87V4WXQ4Z
Ilantimod is an orally available formulation containing a small molecule antagonist of the aryl hydrocarbon receptor (AhR; class E basic helix-loop-helix protein 76; bHLHe76) with potential immunomodulating and antineoplastic activities. Upon oral administration, ilantimod specifically binds to AhR, inhibits AhR activation, and prevents AhR-mediated signaling. Abrogation of AhR activation prevents the activation of immune-tolerant dendritic cells (DCs) and regulatory T-cells (Tregs) in the tumor microenvironment (TME). This may restore the immune response against tumor cells. AhR, a member of the basic helix-loop-helix/Per-Arnt-Sim (bHLH/PAS) family of transcription factors, has important roles in regulating immunity and cellular differentiation. AhR can exhibit both pro-oncogenic and tumor suppressor-like functions depending on the tumor type; therefore, its expression may serve as a negative or positive prognostic factor.
- A Study to Learn How Safe the Study Drug BAY 2416964 (AhR Inhibitor) in Combination With the Treatment Pembrolizumab is, How This Combination Affects the Body, the Maximum Amount That Can be Given, How it Moves Into, Through and Out of the Body and Its Action Against Advanced Solid Cancers in AdultsCTID: NCT04999202Phase: Phase 1Status: TerminatedDate: 2025-02-10
- A First-in-Humans Dose Finding Study for an Aryl Hydrocarbon Receptor Inhibitor (AhRi) in Patients With Advanced CancerCTID: NCT04069026Phase: Phase 1Status: CompletedDate: 2024-03-06
SYN
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2018146010&_cid=P11-MHAFJG-41587-1


Example 17
6-(4-Chlorophenyl)-/V-[(2S)-1 -hydroxypropan-2-yl]-2-(1 -methyl-1 H-pyrazol-4-yl)-3-oxo-2,3-dihydropyridazine-4-carboxamide

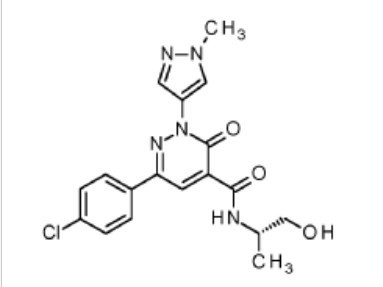
A solution of 80 mg intermediate 1 1 , 29.1 mg (2S)-2-aminopropan-1 -ol, 1 10 mg HATU and 0.1 mL ethyldiisopropylamine in 5 mL of DMF was stirred at room temperature for 14 hours. Then the reaction was quenched by water, and the mixture was extracted with dichloromethane two times. The combined organic phases were dried over sodium sulfate and evaporated to dryness. The residue was subjected to RP-HPLC ((column: X-Bridge C18 5μηι 100x30mm, mobile phase: acetonitrile / water (0.1 vol% formic acid)-gradient)) to yield 50 mg 6-(4-chlorophenyl)-/V-[(2S)-1 -hydroxypropan-2-yl]-2-(1 -methyl-1 H-pyrazol-4-yl)-3-oxo-2,3-dihydropyridazine-4-carboxamide
1H-NMR (400 MHz, CDC ): δ [ppm] = 1.34 (d, 3H); 2.73-2.82 (m, 1 H); 3.66-3.73 (m, 1 H); 3.77-3.84 (m, 1 H); 3.98 (s, 3H); 4.26-4.36 (m, 1 H); 7.49 (d, 2H); 7.87 (d, 2H); 8.12 (s, 1 H); 8.33 (s, 1 H); 8.69 (s, 1 H); 9.82 (bd, 1 H).
PAT
https://patentscope.wipo.int/search/en/detail.jsf?docId=US438191125&_cid=P11-MHAFQQ-47913-1
SEE EX 17
PAT
- 2-Heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamide for the treatment of cancerPublication Number: KR-102627266-B1Priority Date: 2017-02-09Grant Date: 2024-01-24
- 2-heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamides for the treatment of cancerPublication Number: US-11795164-B2Priority Date: 2017-02-09Grant Date: 2023-10-24
- 2-heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamides for the treatment of cancerPublication Number: US-2024294505-A1Priority Date: 2017-02-09
- 2-heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamidesPublication Number: TW-I770113-BPriority Date: 2017-02-09Grant Date: 2022-07-11
- 2-Heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamides for the treatment of cancerPublication Number: CN-110678459-BPriority Date: 2017-02-09Grant Date: 2023-04-04
- 2-heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamides for the treatment of cancerPublication Number: US-2023121195-A1Priority Date: 2017-02-09
- 2-Heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamides for the treatment of cancerPublication Number: CN-116531380-APriority Date: 2017-02-09
- 2-Heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamides for the treatment of cancerPublication Number: CN-116554152-APriority Date: 2017-02-09
- 2-heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamides for the treatment of cancerPublication Number: WO-2018146010-A1Priority Date: 2017-02-09
- 2-heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamides for the treatment of cancerPublication Number: EP-3580211-B1Priority Date: 2017-02-09Grant Date: 2020-12-02
- 2-HETEROARYL-3-OXO-2,3-DIHYDROPYRIDAZINE-4-CARBOXAMIDES FOR THE TREATMENT OF CANCERPublication Number: HR-P20210143-T1Priority Date: 2017-02-09
- 2-heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamides for the treatment of cancerPublication Number: AU-2018217860-B2Priority Date: 2017-02-09Grant Date: 2021-07-08
- 2-Troaril-3-oxo-3,2-dihydropyridazine-4-carboxamides for the treatment of cancerPublication Number: IL-268469-APriority Date: 2017-02-09
- 2-heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamides for the treatment of cancerPublication Number: CA-3052718-A1Priority Date: 2017-02-09
- 2-Heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamide for the treatment of cancerPublication Number: CN-110678459-APriority Date: 2017-02-09
- 2-heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamides for the treatment of cancerPublication Number: EP-3580211-A1Priority Date: 2017-02-09
- 2-heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamide for the treatment of cancerPublication Number: KR-20190115460-APriority Date: 2017-02-09
- 2-heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamidePublication Number: TW-201840549-APriority Date: 2017-02-09
- Combination of an ahr-inhibitor and an pd1-inhibitor antibody and its use in the treatment of cancerPublication Number: EP-4076462-A1Priority Date: 2019-12-16
- Combinations of AHR inhibitors and PD1 inhibitor antibodies and their use in the treatment of cancerPublication Number: JP-2023505907-APriority Date: 2019-12-16
- Combinations of AHR-inhibitors and PD1-inhibitor antibodies and their use in the treatment of cancerPublication Number: KR-20220128622-APriority Date: 2019-12-16
- Combination of an ahr-inhibitor and an pd1-inhibitor antibody and its use in the treatment of cancerPublication Number: US-2023084899-A1Priority Date: 2019-12-16
- 2-heteroaryl-3-oxo-2,3-dihydropyridazine-4-carboxamides for the treatment of cancerPublication Number: AU-2018217860-A1Priority Date: 2017-02-09
- Methods and compositions for treating inflammatory and fibrotic pulmonary disordersPublication Number: US-2021401987-A1Priority Date: 2020-03-20
- Methods and compositions for treating inflammatory and fibrotic pulmonary disordersPublication Number: EP-4121111-A1Priority Date: 2020-03-20
- Combination of an ahr-inhibitor and an pd1-inhibitor antibody and its use in the treatment of cancerPublication Number: WO-2021122434-A1Priority Date: 2019-12-16
- Combination of an AhR-inhibitor and an PD1-inhibitor antibody and its use in the treatment of cancerPublication Number: AU-2020403801-A1Priority Date: 2019-12-16
- Combinations of AHR inhibitor and PD1 inhibitor antibodies and their use in cancer therapyPublication Number: CN-114786674-APriority Date: 2019-12-16
- Compositions and methods for treating myelin deficiency by rejuvenating glial progenitor cellsPublication Number: US-2023190961-A1Priority Date: 2021-10-20
- Prophylactic or therapeutic agent for severe pulmonary hypertension, refractory pulmonary hypertension, or drug-induced pulmonary hypertensionPublication Number: WO-2022149605-A1Priority Date: 2021-01-08
- Deuterated 2-arylheterocycle-3-oxo-2,3-dihydropyridazine-4-carboxamide inhibitor and preparation method therefor and application thereofPublication Number: EP-4253374-A1Priority Date: 2020-11-27
- Heteroaromatic ahr inhibitorPublication Number: WO-2022078356-A1Priority Date: 2020-10-15
- Methods and compositions for treating inflammatory and fibrotic pulmonary disordersPublication Number: WO-2021188849-A1Priority Date: 2020-03-20



AS ON JUNE2025 4.45 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

……
///////////Ilantimod, immunomodulator, BAY-2416964, BAY 2416964, Y87V4WXQ4Z

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










