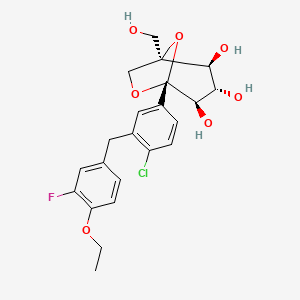
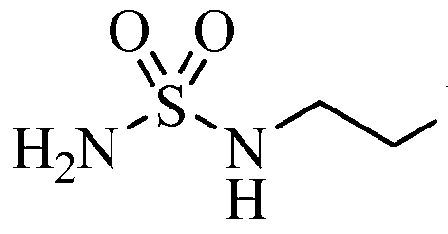INCB24360 (epacadostat)
Molecular Formula, C11H13BrFN7O4S
Average mass438.233 Da
cas 1204669-58-8 (or 1204669-37-3)
| Synonym: | IDO1 inhibitor INCB024360 indoleamine-2,3-dioxygenase inhibitor INCB024360 |
| Code name: | INCB 024360 INCB024360 |
| Chemical structure: | 1,2,5-Oxadiazole-3-carboximidamide, 4-((2-((Aminosulfonyl)amino)ethyl)amino)-N-(3-bromo-4-fluorophenyl)-N’-hydroxy-, (C(Z))- |
- OriginatorIncyte Corporation
- DeveloperFred Hutchinson Cancer Research Center; Incyte Corporation; Merck AG
- ClassAmides; Antineoplastics; Imides; Oxadiazoles; Small molecules
-
- Phase IIFallopian tube cancer; Malignant melanoma; Non-small cell lung cancer; Ovarian cancer; Peritoneal cancer; Solid tumours
Most Recent Events
- 15 Jan 2016Phase-II clinical trials in Solid tumours (Combination therapy, Late-stage disease, Second-line therapy or greater) in USA (PO)
- 11 Jan 2016Phase-II clinical trials in Non-small cell lung cancer (Combination therapy, Late-stage disease, Second-line therapy or greater) in USA (PO)
- 11 Jan 2016The US FDA and Health Canada approve IND application and Clinical Trial Application, respectively, for a phase Ib trial in Ovarian cancer (Combination therapy, Recurrent, Second-line therapy or greater)
In 2016, orphan drug designation was assigned to the compound in the US. for the treatment of stage IIB-IV melanoma
EpacadostatAn orally available hydroxyamidine and inhibitor of indoleamine 2,3-dioxygenase (IDO1), with potential immunomodulating and antineoplastic activities. epacadostat targets and binds to IDO1, an enzyme responsible for the oxidation of tryptophan into kynurenine. By inhibiting IDO1 and decreasing kynurenine in tumor cells, epacadostat increases and restores the proliferation and activation of various immune cells, including dendritic cells (DCs), NK cells, and T-lymphocytes, as well as interferon (IFN) production, and a reduction in tumor-associated regulatory T cells (Tregs). Activation of the immune system, which is suppressed in many cancers, may inhibit the growth of IDO1-expressing tumor cells. IDO1 is overexpressed by a variety of tumor cell types and DCsINCB24360 (epacadostat), An Agent For Cancer Immunotherapy
Incyte and Merck Expand Clinical Collaboration to Include Phase 3 Study Investigating the Combination of Epacadostat with Keytruda® (pembrolizumab) as First-line Treatment for Advanced Melanoma
Pivotal study to evaluate Incyte’s IDO1 inhibitor in combination with Merck’s anti-PD-1 therapy in patients with advanced or metastatic melanoma
WILMINGTON, Del. and KENILWORTH, N.J. — October 13, 2015 — Incyte Corporation (Nasdaq: INCY) and Merck (NYSE:MRK), known as MSD outside the United States and Canada, today announced the expansion of the companies’ ongoing clinical collaboration to include a Phase 3 study evaluating the combination of epacadostat, Incyte’s investigational selective IDO1 inhibitor, with Keytruda® (pembrolizumab), Merck’s anti-PD-1 therapy, as first-line treatment for patients with advanced or metastatic melanoma. The Phase 3 study, which is expected to begin in the first half of 2016, will be co-funded by Incyte and Merck.
“We are very pleased to expand our collaboration with Merck and to move the clinical development program for epacadostat in combination with Keytruda into Phase 3,” said Hervé Hoppenot, President and Chief Executive Officer of Incyte. “We believe the combination of these two immunotherapies shows promise and, if successfully developed, may help to improve clinical outcomes for patients with metastatic melanoma.”
“The initiation of this large Phase 3 study with Incyte in the first-line advanced melanoma treatment setting is an important addition to our robust immunotherapy clinical development program for Keytruda,” said Dr. Roger Dansey, senior vice president and therapeutic area head, oncology late-stage development, Merck Research Laboratories. “We continue to explore the benefit that Keytruda brings to patients suffering from advanced melanoma when used alone, and we are pleased to be able to add this important combination study with epacadostat to our Keytruda development program.”
Under the terms of the agreement Incyte and Merck have also agreed, for a period of two years, not to initiate new pivotal studies of an IDO1 inhibitor in combination with a PD-1/PD-L1 antagonist as first-line therapy in advanced or metastatic melanoma with any third party. During this time, the companies will each offer the other the opportunity to collaborate on any new pivotal study involving an IDO1 inhibitor in combination with a PD-1/PD-L1 antagonist for types of melanoma and lines of therapy outside of the current collaboration agreement.
The agreement is between Incyte and certain subsidiaries and Merck through its subsidiaries.
Epacadostat and Keytruda are part of a class of cancer treatments known as immunotherapies that are designed to enhance the body’s own defenses in fighting cancer; the two therapies target distinct regulatory components of the immune system. IDO1 is an immunosuppressive enzyme that has been shown to induce regulatory T cell generation and activation, and allow tumors to escape immune surveillance. Keytruda is a humanized monoclonal antibody that blocks the interaction between PD-1 and its ligands, PD-L1 and PD-L2. Preclinical evidence suggests that the combination of these two agents may lead to an enhanced anti-tumor immune response compared with either agent alone.
Safety and efficacy data from the ongoing Phase 1/2 study evaluating the combination of epacadostat with Keytruda in patients with advanced malignancies is scheduled to be highlighted as a late-breaking oral presentation (Abstract #142) at the upcoming Society for Immunotherapy of Cancer 30th Anniversary Annual Meeting & Associated Programs, November 4–8, 2015 at the Gaylord National Resort & Convention Center in National Harbor, MD.
Metastatic Melanoma
Melanoma, the most serious form of skin cancer, strikes adults of all ages and accounts for approximately five percent of all new cases of cancer in the United States each year. The number of new cases of melanoma continues to rise by almost three percent each year which translates to 76,000 new cases yearly in the U.S. alone.[i] The 5-year survival rate for late-stage or metastatic disease is 15 percent.[ii]
About Epacadostat (INCB024360)
Indoleamine 2,3-dioxygenase 1 (IDO1) is an immunosuppressive enzyme that has been shown to induce regulatory T cell generation and activation, and allow tumors to escape immune surveillance. Epacadostat is an orally bioavailable small molecule inhibitor of IDO1 that has nanomolar potency in both biochemical and cellular assays and has demonstrated potent activity in enhancing T lymphocyte, dendritic cell and natural killer cell responses in vitro, with a high degree of selectivity. Epacadostat has shown proof-of-concept clinical data in patients with unresectable or metastatic melanoma in combination with the CTLA-4 inhibitor ipilimumab, and is currently in four proof-of-concept clinical trials with PD-1 and PD-L1 immune checkpoint inhibitors in a variety of cancer histologies.

PATENT
WO 2014066834
https://www.google.com/patents/WO2014066834A1?cl=en
EXAMPLE 1
4-({2-[(Aminosulfonyl)amino]ethyl}amino)- V-(3-bromo-4-fluorophenyl)- V -hydroxy- l,2,5-oxadiazole-3-carboximidamide
Step 1: 4-Amino-N’-hydroxy-l,2,5-oxadiazole-3-carboximidamide
[00184] Malononitrile (320.5 g, 5 mol) was added to water (7 L) preheated to 45 °C and stirred for 5 min. The resulting solution was cooled in an ice bath and sodium nitrite (380 g, 5.5 mol) was added. When the temperature reached 10 °C, 6 N hydrochloric acid (55 mL) was added. A mild exothermic reaction ensued with the temperature reaching 16 °C. After 15 min the cold bath was removed and the reaction mixture was stirred for 1.5 hrs at 16-18 °C. The reaction mixture was cooled to 13 °C and 50% aqueous hydroxylamine (990 g, 15 mol) was added all at once. The temperature rose to 26 °C. When the exothermic reaction subsided the cold bath was removed and stirring was continued for 1 hr at 26-27 °C, then it was slowly brought to reflux. Reflux was maintained for 2 hrs and then the reaction mixture was allowed to cool overnight. The reaction mixture was stirred in an ice bath and 6 N hydrochloric acid (800 mL) was added in portions over 40 min to pH 7.0. Stirring was continued in the ice bath at 5 °C. The precipitate was collected by filtration, washed well with water and dried in a vacuum oven (50 °C) to give the desired product (644 g, 90%). LCMS for C3H6N5O2
(M+H)+: m/z = 144.0. 13C MR (75 MHz, CD3OD): δ 156.0, 145.9, 141.3. Step 2: 4-Amino-N-hydroxy-l,2,5-oxadiazole-3-carboximidoyl chloride [00185] 4-Amino-N,-hydroxy-l ,2,5-oxadiazole-3-carboximidamide (422 g, 2.95 mol) was added to a mixture of water (5.9 L), acetic acid (3 L) and 6 Ν hydrochloric acid (1.475 L, 3 eq.) and this suspension was stirred at 42 – 45 °C until complete solution was achieved. Sodium chloride (518 g, 3 eq.) was added and this solution was stirred in an ice/water/methanol bath. A solution of sodium nitrite (199.5 g, 0.98 eq.) in water (700 mL) was added over 3.5 hrs while maintaining the temperature below 0 °C. After complete addition stirring was continued in the ice bath for 1.5 hrs and then the reaction mixture was allowed to warm to 15 °C. The precipitate was collected by filtration, washed well with water, taken in ethyl acetate (3.4 L), treated with anhydrous sodium sulfate (500 g) and stirred for 1 hr. This suspension was filtered through sodium sulfate (200 g) and the filtrate was concentrated on a rotary evaporator. The residue was dissolved in methyl i-butyl ether (5.5 L), treated with charcoal (40 g), stirred for 40 min and filtered through Celite. The solvent was removed in a rotary evaporator and the resulting product was dried in a vacuum oven (45 °C) to give the desired product (256 g, 53.4%). LCMS for C3H4CIN4O2 (M+H)+: m/z = 162.9. 13C NMR (100 MHz, CD3OD): 5 155.8, 143.4, 129.7.
Step 3: 4-Amino-N’-hydroxy-N-(2-methoxyethyl)-l,2,5-oxadiazole-3-carboximidamide [00186] 4-Amino-N-hydroxy-l ,2,5-oxadiazole-3-carboximidoyl chloride (200.0 g, 1.23 mol) was mixed with ethyl acetate (1.2 L). At 0-5 °C 2-methoxyethylamine [Aldrich, product # 143693] (119.0 mL, 1.35 mol) was added in one portion while stirring. The reaction temperature rose to 41 °C. The reaction was cooled to 0 – 5 °C. Triethylamine (258 mL, 1.84 mol) was added. After stirring 5 min, LCMS indicated reaction completion. The reaction solution was washed with water (500 mL) and brine (500 mL), dried over sodium sulfate, and concentrated to give the desired product (294 g, 1 19%) as a crude dark oil.
LCMS for C6Hi2 503 (M+H)+: m/z = 202.3. 1H NMR (400 MHz, DMSO- ): δ 10.65 (s, 1 H), 6.27 (s, 2 H), 6.10 (t, J = 6.5 Hz, 1 H), 3.50 (m, 2 H), 3.35 (d, J = 5.8 Hz, 2 H), 3.08 (s, 3 H).
Step 4: N’-Hydroxy-4-[(2-methoxyethyl)amino]-l,2,5-oxadiazole-3-carboximidamide
[00187] 4-Amino-N-hydroxy-N-(2-methoxyethyl)-l,2,5-oxadiazole-3- carboximidamide (248.0 g, 1.23 mol) was mixed with water (1 L). Potassium hydroxide (210 g, 3.7 mol) was added. The reaction was refluxed at 100 °C overnight (15 hours). TLC with 50% ethyl acetate (containing 1% ammonium hydroxide) in hexane indicated reaction completed (product Rf = 0.6, starting material Rf = 0.5). LCMS also indicated reaction completion. The reaction was cooled to room temperature and extracted with ethyl acetate (3 x 1 L). The combined ethyl acetate solution was dried over sodium sulfate and concentrated to give the desired product (201 g, 81%) as a crude off-white solid. LCMS for C6H12N5O3 (M+H)+: m/z = 202.3 LH NMR (400 MHz, OMSO-d6): δ 10.54 (s, 1 H), 6.22 (s, 2 H), 6.15 (t, J = 5.8 Hz, 1 H), 3.45 (t, J= 5.3 Hz, 2 H), 3.35 (m, 2 H), 3.22 (s, 3 H). Step 5: N-Hydroxy-4-[(2-methoxyethyl)amino]-l,2,5-oxadiazole-3-carboximidoyl chloride
[00188] At room temperature N’-hydroxy-4-[(2-methoxyethyl)amino]- 1 ,2,5- oxadiazole-3-carboximidamide (50.0 g, 0.226 mol) was dissolved in 6.0 M hydrochloric acid aqueous solution (250 mL, 1.5 mol). Sodium chloride (39.5 g, 0.676 mol) was added followed by water (250 mL) and ethyl acetate (250 mL). At 3-5 °C a previously prepared aqueous solution (100 mL) of sodium nitrite (15.0 g, 0.217 mol) was added slowly over 1 hr. The reaction was stirred at 3 – 8 °C for 2 hours and then room temperature over the weekend. LCMS indicated reaction completed. The reaction solution was extracted with ethyl acetate (2 x 200 mL). The combined ethyl acetate solution was dried over sodium sulfate and concentrated to give the desired product (49.9 g, 126%) as a crude white solid. LCMS for
C6HioClN403 (M+H)+: m/z = 221.0. !H NMR (400 MHz, DMSO-d6): δ 13.43 (s, 1 H), 5.85 (t, J= 5.6 Hz, 1 H), 3.50 (t, J= 5.6 Hz, 2 H), 3.37(dd, J= 10.8, 5.6 Hz, 2 H), 3.25 (s, 3 H).
Step 6 : N-(3-Bromo-4-fluorophenyl)-N’-hydroxy-4- [(2-methoxyethyl)amino] – 1 ,2,5- oxadiazole-3-carboximidamide [00189] N-Hydroxy-4-[(2-methoxyethyl)amino]- 1 ,2,5-oxadiazole-3-carboximidoyl chloride (46.0 g, 0.208 mol) was mixed with water (300 mL). The mixture was heated to 60 °C. 3-Bromo-4-fluoroaniline [Oakwood products, product # 013091] (43.6 g, 0.229 mol) was added and stirred for 10 min. A warm sodium bicarbonate (26.3 g, 0.313 mol) solution (300 mL water) was added over 15 min. The reaction was stirred at 60 °C for 20 min. LCMS indicated reaction completion. The reaction solution was cooled to room temperature and extracted with ethyl acetate (2 x 300 mL). The combined ethyl acetate solution was dried over sodium sulfate and concentrated to give the desired product (76.7 g, 98%) as a crude brown solid. LCMS for Ci2Hi4BrF503 (M+H)+: m/z = 374.0, 376.0. 1H NMR (400 MHz, DMSO- tf): δ 11.55 (s, 1 H), 8.85 (s, 1 H), 7.16 (t, J= 8.8 Hz, 1 H), 7.08 (dd, J= 6.1, 2.7 Hz, 1 H), 6.75 (m, 1 H), 6.14 (t, J= 5.8 Hz, 1 H), 3.48 (t, J = 5.2 Hz, 2 H), 3.35 (dd, J= 10.8, 5.6 Hz, 2 H), 3.22 (s, 3 H).
Step 7: 4-(3-Bromo-4-fluorophenyl)-3-{4- [(2-methoxyethyl)amino]-l,2,5-oxadiazol-3- yl}-l,2,4-oxadiazol-5(4H)-one
[00190] A mixture of N-(3-bromo-4-fluorophenyl)-N’-hydroxy-4-[(2- methoxyethyl)amino]-l,2,5-oxadiazole-3-carboximidamide (76.5 g, 0.204 mol), 1,1 ‘- carbonyldiimidazole (49.7 g, 0.307 mol), and ethyl acetate (720 mL) was heated to 60 °C and stirred for 20 min. LCMS indicated reaction completed. The reaction was cooled to room temperature, washed with 1 N HC1 (2 x 750 mL), dried over sodium sulfate, and concentrated to give the desired product (80.4 g, 98%) as a crude brown solid. LCMS for
(M+H)+: m/z = 400.0, 402.0. 1H NMR (400 MHz, DMSO-c½): δ 7.94 (t, J = 8.2 Hz, 1 H), 7.72 (dd, J = 9.1, 2.3 Hz, 1 H), 7.42 (m, 1 H), 6.42 (t, J= 5.7 Hz, 1 H), 3.46 (t, J = 5.4 Hz, 2 H), 3.36 (t, J= 5.8 Hz, 2 H), 3.26 (s, 3 H).
Step 8: 4-(3-Bromo-4-fluorophenyl)-3-{4-[(2-hydroxyethyl)amino]-l,2,5-oxadiazol-3- yl}-l,2,4-oxadiazol-5(4H)-one
[00191] 4-(3-Bromo-4-fluoroplienyl)-3-{4-[(2-metlioxyethyl)amino]-l,2,5-oxadiazol- 3-yl}-l,2,4-oxadiazol-5(4H)-one (78.4 g, 0.196 mol) was dissolved in dichloromethane (600 mL). At -67 °C boron tribromide (37 mL, 0.392 mol) was added over 15 min. The reaction was warmed up to -10 °C in 30 min. LCMS indicated reaction completed. The reaction was stirred at room temperature for 1 hour. At 0 – 5 °C the reaction was slowly quenched with saturated sodium bicarbonate solution (1.5 L) over 30 min. The reaction temperature rose to 25 °C. The reaction was extracted with ethyl acetate (2 x 500 mL, first extraction organic layer is on the bottom and second extraction organic lager is on the top). The combined organic layers were dried over sodium sulfate and concentrated to give the desired product (75 g, 99%) as a crude brown solid. LCMS for Ci2HioBrFN504 (M+H)+: m/z = 386.0, 388.0.
1H NMR (400 MHz, DMSO-^): δ 8.08 (dd, J = 6.2, 2.5 Hz, 1 H), 7.70 (m, 1 H), 7.68 (t, J = 8.7 Hz, 1 H), 6.33 (t, J = 5.6 Hz, 1 H), 4.85 (t, J= 5.0 Hz, 1 H), 3.56 (dd, J= 10.6, 5.6 Hz, 2 H), 3.29 (dd, J= 11.5, 5.9 Hz, 2 H).
Step 9 : 2-({4- [4-(3-Bromo-4-fluorophenyl)-5-oxo-4,5-dihydro- 1 ,2,4-oxadiazol-3-yl] – l,2,5-oxadiazol-3-yl}amino)ethyl methanesulfonate
[00192] To a solution of 4-(3-bromo-4-fluorophenyl)-3-{4-[(2-hydroxyethyl)amino]- l,2,5-oxadiazol-3-yl}-l,2,4-oxadiazol-5(4H)-one (1.5 kg, 3.9 mol, containing also some of the corresponding bromo-compound) in ethyl acetate (12 L) was added methanesulfonyl chloride (185 mL, 2.4 mol) dropwise over 1 h at room temperature. Triethylamine (325 mL, 2.3 mol) was added dropwise over 45 min, during which time the reaction temperature increased to 35 °C. After 2 h, the reaction mixture was washed with water (5 L), brine (1 L), dried over sodium sulfate, combined with 3 more reactions of the same size, and the solvents removed in vacuo to afford the desired product (7600 g, quantitative yield) as a tan solid. LCMS for C HnBrFNsOeS a (M+Na)+: m/z = 485.9, 487.9. !H NMR (400 MHz, DMSO- d6): δ 8.08 (dd, J = 6.2, 2.5 Hz, 1 H), 7.72 (m, 1 H), 7.58 (t, J = 8.7 Hz, 1 H), 6.75 (t, J = 5.9 Hz, 1 H), 4.36 (t, J = 5.3 Hz, 2 H), 3.58 (dd, J = 11.2, 5.6 Hz, 2 H), 3.18 (s, 3 H).
Step 10: 3-{4-[(2-Azidoethyl)amino]-l,2,5-oxadiazol-3-yl}-4-(3-bromo-4-fluorophenyl)- l,2,4-oxadiazol-5(4H)-one
To a solution of 2-({4-[4-(3-bromo-4-f uorophenyl)-5-oxo-4,5-dihydro-l ,2,4- oxadiazol-3-yl]-l ,2,5-oxadiazol-3-yl}amino)ethyl methanesulfonate (2.13 kg, 4.6 mol, containing also some of the corresponding bromo-compound) in dimethylformamide (4 L) stirring in a 22 L flask was added sodium azide (380 g, 5.84 mol). The reaction was heated at 50 °C for 6 h, poured into ice/water (8 L), and extracted with 1 : 1 ethyl acetate:heptane (20 L). The organic layer was washed with water (5 L) and brine (5 L), and the solvents removed in vacuo to afford the desired product (1464 g, 77%) as a tan solid. LCMS for CnHgBrFNsOs a
(M+Na)+: m/z = 433.0, 435.0. !H NMR (400 MHz, DMSO-J6): δ 8.08 (dd, J = 6.2, 2.5 Hz, 1 H), 7.72 (m, 1 H), 7.58 (t, J= 8.7 Hz, 1 H), 6.75 (t, J = 5.7 Hz, 1 H), 3.54 (t, J = 5.3 Hz, 2 H), 3.45 (dd, J= 1 1.1 , 5.2 Hz, 2 H).
Step 11: 3-{4-[(2-Aminoethyl)amino]-l,2,5-oxadiazol-3-yl}-4-(3-bromo-4-fluorophenyl)-
1.2.4- oxadiazol-5(4H)-one hydrochloride
[00194] Sodium iodide (1080 g, 7.2 mol) was added to 3-{4-[(2-azidoethyl)amino]-
1.2.5- oxadiazol-3-yl}-4-(3-bromo-4-fluorophenyl)-l ,2,4-oxadiazol-5(4H)-one (500 g, 1.22 mol) in methanol (6 L). The mixture was allowed to stir for 30 min during which time a mild exotherm was observed. Chlorotrimethylsilane (930 mL, 7.33 mol) was added as a solution in methanol (1 L) dropwise at a rate so that the temperature did not exceed 35 °C, and the reaction was allowed to stir for 3.5 h at ambient temperature. The reaction was neutralized with 33 wt% solution of sodium thiosulfate pentahydrate in water (-1.5 L), diluted with water (4 L), and the pH adjusted to 9 carefully with solid potassium carbonate (250 g – added in small portions: watch foaming). Di-ieri-butyl dicarbonate (318 g, 1.45 mol) was added and the reaction was allowed to stir at room temperature. Additional potassium carbonate (200 g) was added in 50 g portions over 4 h to ensure that the pH was still at or above 9. After stirring at room temperature overnight, the solid was filtered, triturated with water (2 L), and then MTBE (1.5 L). A total of 11 runs were performed (5.5 kg, 13.38 mol). The combined solids were triturated with 1 : 1 THF:dichloromethane (24 L, 4 runs in a 20 L rotary evaporator flask, 50 °C, 1 h), filtered, and washed with dichloromethane (3 L each run) to afford an off- white solid. The crude material was dissolved at 55 °C tetrahydrofuran (5 mL/g), treated with decolorizing carbon (2 wt%) and silica gel (2 wt%), and filtered hot through celite to afford the product as an off-white solid (5122 g). The combined MTBE, THF, and dichloromethane filtrates were concentrated in vacuo and chromatographed (2 kg silica gel, heptane with a 0-100% ethyl acetate gradient, 30 L) to afford more product (262 g). The combined solids were dried to a constant weight in a convection oven (5385 g, 83%).
In a 22 L flask was charged hydrogen chloride (4 N solution in 1 ,4-dioxane, 4 L, 16 mol). tert-Butyl [2-({4-[4-(3-bromo-4-fluorophenyl)-5-oxo-4,5-dihydro-l ,2,4- oxadiazol-3-yl]-l ,2,5-oxadiazol-3-yl}amino)ethyl]carbamate (2315 g, 4.77 mol) was added as a solid in portions over 10 min. The slurry was stirred at room temperature and gradually became a thick paste that could not be stirred. After sitting overnight at room temperature, the paste was slurried in ethyl acetate (10 L), filtered, re-slurried in ethyl acetate (5 L), filtered, and dried to a constant weight to afford the desired product as a white solid (combined with other runs, 5 kg starting material charged, 41 13 g, 95%). LCMS for
Ci2HnBrFN603 (M+H)+: m/z = 384.9, 386.9. 1H NMR (400 MHz, DMSO-^): δ 8.12 (m, 4 H), 7.76 (m, 1 H), 7.58 (t, J = 8.7 Hz, 1 H), 6.78 (t, J = 6.1 Hz, 1 H), 3.51 (dd, J = 1 1.8, 6.1 Hz, 2 H), 3.02 (m, 2 H).
Step 12: tert-Butyl ({[2-({4-[4-(3-bromo-4-nuorophenyl)-5-oxo-4,5-dihydro-l,2,4- oxadiazol-3-yl]-l,2,5-oxadiazol-3-yl}amino)ethyl]amino}sulfonyl)carbamate
A 5 L round bottom flask was charged with chlorosulfonyl isocyanate [Aldrich, product # 142662] (149 mL, 1.72 mol) and dichloromethane (1.5 L) and cooled using an ice bath to 2 °C. teri-Butanol (162 mL, 1.73 mol) in dichloromethane (200 mL) was added dropwise at a rate so that the temperature did not exceed 10 °C. The resulting solution was stirred at room temperature for 30-60 min to provide tert-bvAy\ [chlorosulfonyl]carbamate.
A 22 L flask was charged with 3- {4-[(2-aminoethyl)amino]- 1 ,2,5-oxadiazol-3- yl}-4-(3-bromo-4-fluorophenyl)-l,2,4-oxadiazol-5(4H)-one hydrochloride (661 g, 1.57 mol) and 8.5 L dichloromethane. After cooling to -15 °C with an ice/salt bath, the solution oi tert- Vmtvl i Vi 1 r>rosulfonyl]carbamate (prepared as above) was added at a rate so that the temperature did not exceed -10 °C (addition time 7 min). After stirring for 10 min, triethylamine (1085 mL, 7.78 mol) was added at a rate so that the temperature did not exceed -5 °C (addition time 10 min). The cold bath was removed, the reaction was allowed to warm to 10 °C, split into two portions, and neutralized with 10% cone HC1 (4.5 L each portion). Each portion was transferred to a 50 L separatory funnel and diluted with ethyl acetate to completely dissolve the white solid (-25 L). The layers were separated, and the organic layer was washed with water (5 L), brine (5 L), and the solvents removed in vacuo to afford an off- white solid. The solid was triturated with MTBE (2 x 1.5 L) and dried to a constant weight to afford a white solid. A total of 4113 g starting material was processed in this manner (5409 g, 98%). 1H NMR (400 MHz, DMSO-^): δ 10.90 (s, 1 H), 8.08 (dd, J = 6.2, 2.5 Hz, 1 H), 7.72 (m, 1 H), 7.59 (t, J = 8.6 Hz, 1 H), 6.58 (t, J = 5.7 Hz, 1 H), 3.38 (dd, J= 12.7, 6.2 Hz, 2 H), 3.10 (dd, J= 12.1 , 5.9 Hz, 2 H), 1.41 (s, 9 H).
Step 13: N-[2-({4-[4-(3-Bromo-4-fluorophenyl)-5-oxo-4,5-dihydro-l,2,4-oxadiazol-3-yl]- l,2,5-oxadiazol-3-yl}amino)ethyl]sulfamide
[00198] To a 22 L flask containing 98:2 trifluoroacetic acid:water (8.9 L) was added tert-bvXyl ({[2-({4-[4-(3-bromo-4-fluorophenyl)-5-oxo-4,5-dihydro-l,2,4-oxadiazol-3-yl]- l,2,5-oxadiazol-3-yl}amino)ethyl]amino}sulfonyl)carbamate (1931 g, 3.42 mol) in portions over 10 minutes. The resulting mixture was stirred at room temperature for 1.5 h, the solvents removed in vacuo, and chased with dichloromethane (2 L). The resulting solid was treated a second time with fresh 98:2 trifluoroacetic acid:water (8.9 L), heated for 1 h at 40- 50 °C, the solvents removed in vacuo, and chased with dichloromethane (3 x 2 L). The resulting white solid was dried in a vacuum drying oven at 50 °C overnight. A total of 5409 g was processed in this manner (4990 g, quant, yield). LCMS for C12H12BrFN705S (M+H)+: m/z = 463.9, 465.9. 1H NMR (400 MHz, DMSO- ): δ 8.08 (dd, J = 6.2, 2.5 Hz, 1 H), 7.72 (m, 1 H), 7.59 (t, J= 8.7 Hz, 1 H), 6.67 (t, J = 5.9 Hz, 1H), 6.52 (t, J= 6.0 Hz, 1 H), 3.38 (dd, J = 12.7, 6.3 Hz, 2 H), 3.11 (dd, J = 12.3, 6.3 Hz). Step 14: 4-({2-[(Aminosulfonyl)amino]ethyl}amino)-N-(3-bromo-4-fluorophenyl)-N’- hydroxy-l,2,5-oxadiazole-3-carboximidamide
[00199] To a crude mixture of N-[2-({4-[4-(3-bromo-4-fluorophenyl)-5-oxo-4,5- dihydro-l,2,4-oxadiazol-3-yl]-l,2,5-oxadiazol-3-yl}amino)ethyl]sulfamide (2.4 mol) containing residual amounts of trifluoroacetic acid stirring in a 22 L flask was added THF (5 L). The resulting solution was cooled to 0 °C using an ice bath and 2 N NaOH (4 L) was added at a rate so that the temperature did not exceed 10 °C. After stirring at ambient temperature for 3 h (LCMS indicated no starting material remained), the pH was adjusted to 3-4 with concentrated HC1 (-500 mL). The THF was removed in vacuo, and the resulting mixture was extracted with ethyl acetate (15 L). The organic layer was washed with water (5 L), brine (5 L), and the solvents removed in vacuo to afford a solid. The solid was triturated with MTBE (2 x 2 L), combined with three other reactions of the same size, and dried overnight in a convection oven to afford a white solid (3535 g). The solid was recrystallized (3 x 22 L flasks, 2:1 watenethanol, 14.1 L each flask) and dried in a 50 °C convection oven to a constant weight to furnish the title compound as an off-white solid (3290 g, 78%). LCMS for CnHnBrF yC S (M+H)+: m/z = 437.9, 439.9. i NMR (400 MHz, DMSO-J^): δ 11.51 (s, 1 H), 8.90 (s, 1 H), 7.17 (t, J= 8.8 Hz, 1 H), 7.11 (dd, J= 6.1, 2.7 Hz, 1 H), 6.76 (m, 1 H), 6.71 (t, J = 6.0 Hz, 1 H), 6.59 (s, 2 H), 6.23 (t, J= 6.1 Hz, 1 H), 3.35 (dd, J= 10.9, 7.0 Hz, 2 H), 3.10 (dd, J= 12.1, 6.2 Hz, 2 H).
PATENT
WO 2010005958
https://www.google.com/patents/WO2010005958A2?cl=en
EXAMPLES Example 1
4-({2-[(Aminosulfonyl)amino]ethyl}amino)-7V-(3-bromo-4-fluorophenyl)-iV’-hydroxy- l,2,5-oxadiazole-3-carboximidamide
Step A: 4-Amino-N’-hydroxy-l,2,5-oxadiazole-3-carboximidamide
Malononitrile [Aldrich, product # M1407] (320.5 g, 5 mol) was added to water (7 L) preheated to 45 0C and stirred for 5 min. The resulting solution was cooled in an ice bath and sodium nitrite (380 g, 5.5 mol) was added. When the temperature reached 10 0C, 6 N hydrochloric acid (55 mL) was added. A mild exothermic reaction ensued with the temperature reaching 16 0C. After 15 min the cold bath was removed and the reaction mixture was stirred for 1.5 hrs at 16-18 0C. The reaction mixture was cooled to 13 0C and 50% aqueous hydroxylamine (990 g, 15 mol) was added all at once. The temperature rose to 26 0C. When the exothermic reaction subsided the cold bath was removed and stirring was continued for 1 hr at 26-270C, then it was slowly brought to reflux. Reflux was maintained for 2 hrs and then the reaction mixture was allowed to cool overnight. The reaction mixture was stirred in an ice bath and 6 N hydrochloric acid (800 mL) was added in portions over 40 min to pH 7.0. Stirring was continued in the ice bath at 5 0C. The precipitate was collected by filtration, washed well with water and dried in a vacuum oven (50 0C) to give the desired product (644 g, 90%). LCMS for C3H6N5O2 (M+H)+: m/z = 144.0. 13C NMR (75 MHz, CD3OD): δ 156.0, 145.9, 141.3. Step B: 4-Amino-N-hydroxy-l,2,5-oxadiazole-3-carboximidoyl chloride
4-Amino-N’-hydroxy-l,2,5-oxadiazole-3-carboximidamide (422 g, 2.95 mol) was added to a mixture of water (5.9 L), acetic acid (3 L) and 6 Ν hydrochloric acid (1.475 L, 3 eq.) and this suspension was stirred at 42 – 45 0C until complete solution was achieved. Sodium chloride (518 g, 3 eq.) was added and this solution was stirred in an ice/water/methanol bath. A solution of sodium nitrite (199.5 g, 0.98 eq.) in water (700 mL) was added over 3.5 hrs while maintaining the temperature below 0 0C. After complete addition stirring was continued in the ice bath for 1.5 hrs and then the reaction mixture was allowed to warm to 15 0C. The precipitate was collected by filtration, washed well with water, taken in ethyl acetate (3.4 L), treated with anhydrous sodium sulfate (500 g) and stirred for 1 hr. This suspension was filtered through sodium sulfate (200 g) and the filtrate was concentrated on a rotary evaporator. The residue was dissolved in methyl f-butyl ether (5.5 L), treated with charcoal (40 g), stirred for 40 min and filtered through Celite. The solvent was removed in a rotary evaporator and the resulting product was dried in a vacuum oven (45 0C) to give the desired product (256 g, 53.4%). LCMS for C3H4ClN4O2(M+H)+: m/z = 162.9. 13c NMR (100 MHz, CD3OD): δ 155.8, 143.4, 129.7.
Step C: 4-Amino-N’-hydroxy-N-(2-methoxyethyl)- 1 ,2,5-oxadiazole-3-carboximidamide
4-Amino-N-hydroxy-l,2,5-oxadiazole-3-carboximidoyl chloride (200.0 g, 1.23 mol) was mixed with ethyl acetate (1.2 L). At 0-50C 2-methoxyethylamine [Aldrich, product # 143693] (119.0 mL, 1.35 mol) was added in one portion while stirring. The reaction temperature rose to 41 0C. The reaction was cooled to 0 – 5 °C. Triethylamine (258 mL, 1.84 mol) was added. After stirring 5 min, LCMS indicated reaction completion. The reaction solution was washed with water (500 mL) and brine (500 mL), dried over sodium sulfate, and concentrated to give the desired product (294 g, 119%) as a crude dark oil. LCMS for C6Hi2N5O3 (M+H)+: m/z = 202.3. 1H NMR (400 MHz, DMSO-J6): δ 10.65 (s, 1 H), 6.27 (s, 2 H), 6.10 (t, J= 6.5 Hz, 1 H), 3.50 (m, 2 H), 3.35 (d, J= 5.8 Hz, 2 H), 3.08 (s, 3 H).
Step D: N’-Hydroxy-4-[(2-methoxyethyl)amino]-l ,2,5-oxadiazole-3-carboximidamide
4-Amino-N’-hydroxy-N-(2-methoxyethyl)-l,2,5-oxadiazole-3-carboximidaniide (248.0 g, 1.23 mol) was mixed with water (1 L). Potassium hydroxide (210 g, 3.7 mol) was added. The reaction was refluxed at 100 0C overnight (15 hours). TLC with 50% ethyl acetate (containing 1% ammonium hydroxide) in hexane indicated reaction completed (product Rf= 0.6, starting material Rf = 0.5). LCMS also indicated reaction completion. The reaction was cooled to room temperature and extracted with ethyl acetate (3 x 1 L). The combined ethyl acetate solution was dried over sodium sulfate and concentrated to give the desired product (201 g, 81%) as a crude off-white solid. LCMS for C6H12N5O3 (M+H)+: m/z = 202.3 1H NMR (400 MHz, DMSO-Gk): δ 10.54 (s, 1 H), 6.22 (s, 2 H), 6.15 (t, J= 5.8 Hz, 1 H), 3.45 (t, J= 5.3 Hz, 2 H), 3.35 (m, 2 H), 3.22 (s, 3 H).
Step E: N-Hydroxy-4-[(2-methoxyethyl)amino]-l,2,5-oxadiazole-3-carboximidoyl chloride
Ν. ,Ν O
At room temperature N’-hydroxy-4-[(2-methoxyethyl)amino]-l,2,5-oxadiazole-3- carboximidamide (50.0 g, 0.226 mol) was dissolved in 6.0 M hydrochloric acid aqueous solution (250 mL, 1.5 mol). Sodium chloride (39.5 g, 0.676 mol) was added followed by water (250 mL) and ethyl acetate (250 mL). At 3-5 0C a previously prepared aqueous solution (100 mL) of sodium nitrite (15.0 g, 0.217 mol) was added slowly over 1 hr. The reaction was stirred at 3 – 8 0C for 2 hours and then room temperature over the weekend. LCMS indicated reaction completed. The reaction solution was extracted with ethyl acetate (2 x 200 mL). The combined ethyl acetate solution was dried over sodium sulfate and concentrated to give the desired product (49.9 g, 126%) as a crude white solid. LCMS for C6Hi0ClN4O3 (M+H)+: m/z = 221.0. 1H NMR (400 MHz, DMSO-J6): δ 13.43 (s, 1 H), 5.85 (t, J= 5.6 Hz, 1 H), 3.50 (t, J= 5.6 Hz, 2 H), 3.37(dd, J= 10.8, 5.6 Hz, 2 H), 3.25 (s, 3 H).
Step F: N-(3-Bromo-4-fluorophenyl)-N’-hydroxy-4-[(2-methoxyethyl)amino]- 1 ,2,5- oxadiazole-3 -carboximidamide
N-Hydroxy-4-[(2-methoxyethyl)amino]-l,2,5-oxadiazole-3-carboximidoyl chloride (46.0 g, 0.208 mol) was mixed with water (300 mL). The mixture was heated to 60 °C. 3-Bromo-4- fluoroaniline [Oakwood products, product # 013091] (43.6 g, 0.229 mol) was added and stirred for 10 nrn‘n. A warm sodium bicarbonate (26.3 g, 0.313 mol) solution (300 mL water) was added over 15 min. The reaction was stirred at 60 0C for 20 min. LCMS indicated reaction completion. The reaction solution was cooled to room temperature and extracted with ethyl acetate (2 x 300 mL). The combined ethyl acetate solution was dried over sodium sulfate and concentrated to give the desired product (76.7 g, 98%) as a crude brown solid. LCMS for Ci2Hi4BrFN5O3 (M+H)+: m/z = 374.0, 376.0. 1H NMR (400 MHz, DMSO-J6): δ 11.55 (s, 1 H), 8.85 (s, 1 H), 7.16 (t, J= 8.8 Hz, 1 H), 7.08 (dd, J= 6.1, 2.7 Hz, 1 H), 6.75 (m, 1 H), 6.14 (t, J= 5.8 Hz, 1 H), 3.48 (t, J= 5.2 Hz, 2 H), 3.35 (dd, J= 10.8, 5.6 Hz, 2 H), 3.22 (s, 3 H).
Step G: 4-(3-Bromo-4-fluorophenyl)-3-{4-[(2-methoxyethyl)amino]-l,2,5-oxadiazol-3-yl}- 1 ,2,4-oxadiazol-5(4H)-one
A mixture of N-(3-bromo-4-fluorophenyl)-N’-hydroxy-4-[(2-methoxyethyl)amino]-l,2,5- oxadiazole-3-carboximidamide (76.5 g, 0.204 mol), l,r-carbonyldiimidazole (49.7 g, 0.307 mol), and ethyl acetate (720 mL) was heated to 60 0C and stirred for 20 min. LCMS indicated reaction completed. The reaction was cooled to room temperature, washed with 1 Ν HCl (2 x 750 mL), dried over sodium sulfate, and concentrated to give the desired product (80.4 g, 98%) as a crude brown solid. LCMS for C13H12BrFN5O4 (M+H)+: m/z = 400.0, 402.0. 1H NMR (400 MHz, OMSO-d6): δ 7.94 (t, J= 8.2 Hz, 1 H), 7.72 (dd, J= 9.1, 2.3 Hz, 1 H), 7.42 (m, 1 H), 6.42 (t, J= 5.7 Hz, 1 H), 3.46 (t, J= 5.4 Hz, 2 H), 3.36 (t, J= 5.8 Hz, 2 H), 3.26 (s, 3 H).
Step H: 4-(3-Bromo-4-fluorophenyl)-3-{4-[(2-liydroxyethyl)amino]-l,2,5-oxadiazol-3-yl}- 1 ,2,4-oxadiazol-5(4H)-one
4-(3-Bromo-4-fluorophenyl)-3-{4-[(2-methoxyetliyl)amino]-l,2,5-oxadiazol-3-yl}-l,2,4- oxadiazol-5(4H)-one (78.4 g, 0.196 mol) was dissolved in dichloromethane (600 mL). At -67 0C boron tribromide (37 mL, 0.392 mol) was added over 15 min. The reaction was warmed up to -10 0C in 30 min. LCMS indicated reaction completed. The reaction was stirred at room temperature for 1 hour. At 0 – 5 0C the reaction was slowly quenched with saturated sodium bicarbonate solution (1.5 L) over 30 min. The reaction temperature rose to 25 0C. The reaction was extracted with ethyl acetate (2 x 500 mL, first extraction organic layer is on the bottom and second extraction organic lager is on the top). The combined organic layers were dried over sodium sulfate and concentrated to give the desired product (75 g, 99%) as a crude brown solid. LCMS for C12H10BrFN5O4 (M+H)+: m/z = 386.0, 388.0. 1H NMR (400 MHz, DMSO-^6): δ 8.08 (dd, J= 6.2, 2.5 Hz, 1 H), 7.70 (m, 1 H), 7.68 (t, J= 8.7 Hz, 1 H), 6.33 (t, J= 5.6 Hz, 1 H), 4.85 (t, J= 5.0 Hz, 1 H), 3.56 (dd, J= 10.6, 5.6 Hz, 2 H), 3.29 (dd, J= 11.5, 5.9 Hz, 2 H).
Step I: 2-({4-[4-(3-Bromo-4-fluorophenyl)-5-oxo-4,5-dihydro-l,2,4-oxadiazol-3-yl]-l,2,5- oxadiazol-3-yl}amino)ethyl methanesulfonate
To a solution of 4-(3-bromo-4-fluorophenyl)-3-{4-[(2-hydroxyethyl)amino]-l,2,5-oxadiazol- 3-yl}-l,2,4-oxadiazol-5(4H)-one (1.5 kg, 3.9 mol, containing also some of the corresponding bromo-compound) in ethyl acetate (12 L) was added methanesulfonyl chloride (185 mL, 2.4 mol) dropwise over 1 h at room temperature. Triethylamine (325 mL, 2.3 mol) was added dropwise over 45 min, during which time the reaction temperature increased to 35 0C. After 2 h, the reaction mixture was washed with water (5 L), brine (I L), dried over sodium sulfate, combined with 3 more reactions of the same size, and the solvents removed in vacuo to afford the desired product (7600 g, quantitative yield) as a tan solid. LCMS for
Ci3HnBrFN5O6SNa (M+Na)+: m/z = 485.9, 487.9. 1H NMR (400 MHz, DMSCW6): δ 8.08 (dd, J= 6.2, 2.5 Hz, 1 H), 7.72 (m, 1 H), 7.58 (t, J= 8.7 Hz, 1 H), 6.75 (t, J- 5.9 Hz, 1 H), 4.36 (t, J= 5.3 Hz, 2 H), 3.58 (dd, J= 11.2, 5.6 Hz, 2 H), 3.18 (s, 3 H).
Step J: 3-{4-[(2-Azidoethyl)amino]-l,2,5-oxadiazol-3-yl}-4-(3-bromo-4-fluorophenyl)- 1 ,2,4-oxadiazol-5(4H)-one
To a solution of 2-({4-[4-(3-bromo-4-fluorophenyl)-5-oxo-4,5-dihydro-l,2,4-oxadiazol-3-yl]- l,2,5-oxadiazol-3-yl}amino)ethyl methanesulfonate (2.13 kg, 4.6 mol, containing also some of the corresponding bromo-compound) in dimethylformamide (4 L) stirring in a 22 L flask was added sodium azide (380 g, 5.84 mol). The reaction was heated at 500C for 6 h, poured into ice/water (8 L), and extracted with 1 : 1 ethyl acetate:heptane (20 L). The organic layer was washed with water (5 L) and brine (5 L), and the solvents removed in vacuo to afford the desired product (1464 g, 77%) as a tan solid. LCMS for C12H8BrFN8O3Na (M+Na)+: m/z =
433.0, 435.0. 1H NMR (400 MHz, DMSO-*/*): δ 8.08 (dd, J= 6.2, 2.5 Hz, 1 H), 7.72 (m, 1 H), 7.58 (t, J= 8.7 Hz, 1 H), 6.75 (t, J= 5.7 Hz, 1 H), 3.54 (t, J= 5.3 Hz, 2 H), 3.45 (dd, J= 11.1, 5.2 Hz, 2 H).
Step K: 3-{4-[(2-Aminoethyl)amino]-l,2,5-oxadiazol-3-yl}-4-(3-bromo-4-fluorophenyl)- 1 ,2,4-oxadiazol-5(4H)-one hydrochloride
Sodium iodide (1080 g, 7.2 mol) was added to 3-{4-[(2-azidoethyl)amino]-l,2,5-oxadiazol-3- yl}-4-(3-bromo-4-fluorophenyl)-l,2,4-oxadiazol-5(4H)-one (500 g, 1.22 mol) in methanol (6 L). The mixture was allowed to stir for 30 min during which time a mild exotherm was observed. Chlorotrimethylsilane (930 mL, 7.33 mol) was added as a solution in methanol (1 L) dropwise at a rate so that the temperature did not exceed 35 0C, and the reaction was allowed to stir for 3.5 h at ambient temperature. The reaction was neutralized with 33 wt% solution of sodium thiosulfate pentahydrate in water (~1.5 L), diluted with water (4 L), and the pΗ adjusted to 9 carefully with solid potassium carbonate (250 g – added in small portions: watch foaming). Di-fe/t-butyl dicarbonate (318 g, 1.45 mol) was added and the reaction was allowed to stir at room temperature. Additional potassium carbonate (200 g) was added in 50 g portions over 4 h to ensure that the pΗ was still at or above 9. After stirring at room temperature overnight, the solid was filtered, triturated with water (2 L), and then MTBE (1.5 L). A total of 11 runs were performed (5.5 kg, 13.38 mol). The combined solids were triturated with 1 : 1 TΗF:dichloromethane (24 L, 4 runs in a 20 L rotary evaporator flask, 50 0C, 1 h), filtered, and washed with dichloromethane (3 L each run) to afford an off- white solid. The crude material was dissolved at 55 0C tetrahydrofuran (5 mL/g), treated with decolorizing carbon (2 wt%) and silica gel (2 wt%), and filtered hot through celite to afford the product as an off-white solid (5122 g). The combined MTBE, THF, and dichloromethane filtrates were concentrated in vacuo and chromatographed (2 kg silica gel, heptane with a 0-100% ethyl acetate gradient, 30 L) to afford more product (262 g). The combined solids were dried to a constant weight in a convection oven (5385 g, 83%).
In a 22 L flask was charged hydrogen chloride (4 N solution in 1,4-dioxane, 4 L, 16 mol). fert-Butyl [2-({4-[4-(3-bromo-4-fluorophenyl)-5-oxo-4,5-dihydro-l,2,4-oxadiazol-3-yl]- l,2,5-oxadiazol-3-yl}amino)ethyl]carbamate (2315 g, 4.77 mol) was added as a solid in portions over 10 min. The slurry was stirred at room temperature and gradually became a thick paste that could not be stirred. After sitting overnight at room temperature, the paste was slurried in ethyl acetate (10 L), filtered, re-slurried in ethyl acetate (5 L), filtered, and dried to a constant weight to afford the desired product as a white solid (combined with other runs, 5 kg starting material charged, 4113 g, 95%). LCMS for C12HnBrFN6O3 (M+H)+: m/z
= 384.9, 386.9. 1H NMR (400 MHz, DMSO-J6): δ 8.12 (m, 4 H), 7.76 (m, 1 H), 7.58 (t, J= 8.7 Hz, 1 H), 6.78 (t, J= 6.1 Hz, 1 H), 3.51 (dd, J= 11.8, 6.1 Hz, 2 H), 3.02 (m, 2 H).
Step L: tert-Butyl ({[2-({4-[4-(3-bromo-4-fluorophenyl)-5-oxo-4,5-diliydro-l,2,4-oxadiazol- 3-yl]-l,2,5-oxadiazol-3-yl}amino)ethyl]amino}sulfonyl)carbamate
A 5 L round bottom flask was charged with chlorosulfonyl isocyanate [Aldrich, product #
142662] (149 mL, 1.72 mol) and dichloromethane (1.5 L) and cooled using an ice bath to 2 0C. tert-Butanol (162 mL, 1.73 mol) in dichloromethane (200 mL) was added dropwise at a rate so that the temperature did not exceed 10 0C. The resulting solution was stirred at room temperature for 30-60 min to provide tert-butyl [chlorosulfonyljcarbamate.
A 22 L flask was charged with 3-{4-[(2-aminoethyl)amino]-l,2,5-oxadiazol-3-yl}-4-(3- bromo-4-fluorophenyl)-l,2,4-oxadiazol-5(4H)-one hydrochloride (661 g, 1.57 mol) and 8.5 L dichloromethane. After cooling to -15 0C with an ice/salt bath, the solution of tert-butyl [chlorosulfonyl]carbamate (prepared as above) was added at a rate so that the temperature did not exceed -10 0C (addition time 7 min). After stirring for 10 min, triethylamine (1085 mL, 7.78 mol) was added at a rate so that the temperature did not exceed -5 0C (addition time 10 min). The cold bath was removed, the reaction was allowed to warm to 10 0C, split into two portions, and neutralized with 10% cone HCl (4.5 L each portion). Each portion was transferred to a 50 L separatory funnel and diluted with ethyl acetate to completely dissolve the white solid (~25 L). The layers were separated, and the organic layer was washed with water (5 L), brine (5 L), and the solvents removed in vacuo to afford an off-white solid. The solid was triturated with MTBE (2 x 1.5 L) and dried to a constant weight to afford a white solid. A total of 4113 g starting material was processed in this manner (5409 g, 98%). *Η NMR (400 MHz, OMSO-d6): δ 10.90 (s, 1 H), 8.08 (dd, J= 6.2, 2.5 Hz, 1 H), 7.72 (m, 1 H), 7.59 (t, J= 8.6 Hz, 1 H), 6.58 (t, J= 5.7 Hz, 1 H), 3.38 (dd, J= 12.7, 6.2 Hz, 2 H), 3.10 (dd, J = 12.1, 5.9 Hz, 2 H), 1.41 (s, 9 H). Step M: N-[2-({4-[4-(3-Bromo-4-fluorophenyl)-5-oxo-4,5-dmydro-l ,2,4-oxadiazol-3-yl]- l,2,5-oxadiazol-3-yl}amino)ethyl]sulfamide
To a 22 L flask containing 98:2 trifluoroacetic acid:water (8.9 L) was added tert-butyl ({[2- ({4-[4-(3-bromo-4-fluorophenyl)-5-oxo-4,5-diliydro-l,2,4-oxadiazol-3-yl]-l,2,5-oxadiazol-3- yl}amino)ethyl]amino}sulfonyl)carbamate (1931 g, 3.42 mol) in portions over 10 minutes. The resulting mixture was stirred at room temperature for 1.5 h, the solvents removed in vacuo, and chased with dichloromethane (2 L). The resulting solid was treated a second time with fresh 98:2 trifluoroacetic acid:water (8.9 L), heated for 1 h at 40-50 0C, the solvents removed in vacuo, and chased with dichloromethane (3 x 2 L). The resulting white solid was dried in a vacuum drying oven at 50 0C overnight. A total of 5409 g was processed in this manner (4990 g, quant, yield). LCMS for C]2H12BrFN7O5S (M+H)+: m/z = 463.9, 465.9.
1H NMR (400 MHz, OM$>O-d6): δ 8.08 (dd, J= 6.2, 2.5 Hz, 1 H), 7.72 (m, 1 H), 7.59 (t, J= 8.7 Hz, 1 H), 6.67 (t, J= 5.9 Hz, IH), 6.52 (t, J= 6.0 Hz, 1 H), 3.38 (dd, J= 12.7, 6.3 Hz, 2 H), 3.11 (dd, J= 12.3, 6.3 Hz).
Step N: 4-( {2-[(Aminosulfonyl)amino]ethyl} amino)-N-(3-bromo-4-fluorophenyl)-N- hydroxy-l,2,5-oxadiazole-3-carboximidamide
To a crude mixture of N-[2-({4-[4-(3-bromo-4-fluorophenyl)-5-oxo-4,5-dihydro-l,2,4- oxadiazol-3-yl]-l,2,5-oxadiazol-3-yl}amino)ethyl]sulfamide (2.4 mol) containing residual amounts of trifluoroacetic acid stirring in a 22 L flask was added THF (5 L). The resulting solution was cooled to 0 °C using an ice bath and 2 Ν NaOH (4 L) was added at a rate so that the temperature did not exceed 10 0C. After stirring at ambient temperature for 3 h (LCMS indicated no starting material remained), the pH was adjusted to 3-4 with concentrated HCl (-500 mL). The THF was removed in vacuo, and the resulting mixture was extracted with ethyl acetate (15 L). The organic layer was washed with water (5 L), brine (5 L), and the solvents removed in vacuo to afford a solid. The solid was triturated with MTBE (2 x 2 L), combined with three other reactions of the same size, and dried overnight in a convection oven to afford a white solid (3535 g). The solid was recrystallized (3 x 22 L flasks, 2: 1 water: ethanol, 14.1 L each flask) and dried in a 50 0C convection oven to a constant weight to furnish the title compound as an off-white solid (3290 g, 78%). LCMS for CnH14BrFN7O4S (M+H)+: m/z = 437.9, 439.9. 1H NMR (400 MHz, DMSO-J6): δ 11.51 (s, 1 H), 8.90 (s, 1 H), 7.17 (t, J= 8.8 Hz, 1 H), 7.11 (dd, J= 6.1, 2.7 Hz, 1 H), 6.76 (m, 1 H), 6.71 (t, J= 6.0 Hz, 1 H), 6.59 (s, 2 H), 6.23 (t, J= 6.1 Hz, 1 H), 3.35 (dd, J= 10.9, 7.0 Hz, 2 H), 3.10 (dd, J= 12.1, 6.2 Hz, 2 H).
The final product was an anhydrous crystalline solid. The water content was determined to be less than 0.1% by Karl Fischer titration.
CLIP

Incyte’s Andrew P. Combs presented the company’s clinical candidate for cancer immunotherapy. The basic tenet of this burgeoning field is that the human body’s immune system is a tremendous resource for fighting disease; scientists just need to figure out how to unleash it. One target that’s proven to be particularly attractive for this purpose in recent years is indoleamine-2,3-dioxygenase-1, or IDO1 (C&EN, April 6, page 10).
IDO1 plays a role in signaling the immune system to stand down from attacking foreign bodies it might otherwise go after, such as fetuses. Tumors also produce IDO1 to evade the immune system, so molecules that can inhibit this enzyme could bring the full force of the body’s defenses to bear on these deadly invaders.
Incyte’s search for an IDO1 inhibitor began with a high-throughput screen, which led to a proof-of-concept compound. But the compound had poor oral bioavailability. What’s more, the molecule and its analogs underwent glucuronidation during its metabolism: Enzymes tacked on a glucuronic acid group to the structure’s amidoxime, which was key to its activity.
The chemists reasoned they could block this metabolism by sterically hindering that position. Making such molecules proved to be more difficult than they expected. But then they unearthed a Latvian paper from 1993 that gave them the synthetic method they needed to make the series of compounds that would lead to their clinical candidate INCB24360 (epacadostat).
With its furazan core, as well as its amidoxime, bromide, and sulfuric diamide functional groups, INCB24360 is something of an odd duck, Combs acknowledged. “Some of you in the audience may be looking at this and saying, ‘That molecule does not look like something I would bring forward or maybe even make,’ ” he said, noting that the structure breaks many medicinal chemistry rules. “We’re a data-centric company, and we followed the data, not the rules,” Combs told C&EN.
The compound has completed Phase I clinical trials and is now being used in collaborative studies with several other pharmaceutical companies that combine INCB24360 with other cancer immunotherapy agents.

| WO2007113648A2 * | Mar 26, 2007 | Oct 11, 2007 | Pfizer Products Inc. | Ctla4 antibody combination therapy |
| US20070185165 * | Dec 19, 2006 | Aug 9, 2007 | Combs Andrew P | N-hydroxyamidinoheterocycles as modulators of indoleamine 2,3-dioxygenase |
| US20100055111 * | Feb 14, 2008 | Mar 4, 2010 | Med. College Of Georgia Research Institute, Inc. | Indoleamine 2,3-dioxygenase, pd-1/pd-l pathways, and ctla4 pathways in the activation of regulatory t cells |
| US20120058079 * | Nov 11, 2011 | Mar 8, 2012 | Incyte Corporation, A Delaware Corporation | 1,2,5-Oxadiazoles as Inhibitors of Indoleamine 2,3-Dioxygenase |
REFERENCES
1: Vacchelli E, Aranda F, Eggermont A, Sautès-Fridman C, Tartour E, Kennedy EP, Platten M, Zitvogel L, Kroemer G, Galluzzi L. Trial watch: IDO inhibitors in cancer therapy. Oncoimmunology. 2014 Dec 15;3(10):e957994. eCollection 2014 Nov. Review. PubMed PMID: 25941578; PubMed Central PMCID: PMC4292223.
2: Liu X, Shin N, Koblish HK, Yang G, Wang Q, Wang K, Leffet L, Hansbury MJ, Thomas B, Rupar M, Waeltz P, Bowman KJ, Polam P, Sparks RB, Yue EW, Li Y, Wynn R, Fridman JS, Burn TC, Combs AP, Newton RC, Scherle PA. Selective inhibition of IDO1 effectively regulates mediators of antitumor immunity. Blood. 2010 Apr 29;115(17):3520-30. doi: 10.1182/blood-2009-09-246124. Epub 2010 Mar 2. PubMed PMID: 20197554.
3: Koblish HK, Hansbury MJ, Bowman KJ, Yang G, Neilan CL, Haley PJ, Burn TC, Waeltz P, Sparks RB, Yue EW, Combs AP, Scherle PA, Vaddi K, Fridman JS. Hydroxyamidine inhibitors of indoleamine-2,3-dioxygenase potently suppress systemic tryptophan catabolism and the growth of IDO-expressing tumors. Mol Cancer Ther. 2010 Feb;9(2):489-98. doi: 10.1158/1535-7163.MCT-09-0628. Epub 2010 Feb 2. PubMed PMID: 20124451.
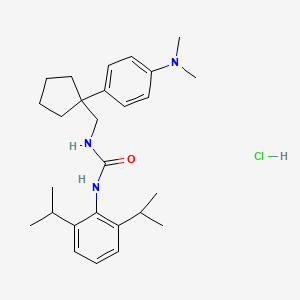
ATR 101

N-(2,6-bis(1-methylethyl)phenyl)-N’-((1-(4-(dimethylamino)phenyl)cyclopentyl) methyl)urea hydrochloride
N-(2,6-BIS(l-METHYLETHYL)PHENYL)-N’-((l-(4- (DIMETHYLAMINO)PHENYL)CYCLOPENTYL)METHYL)UREA
ATR-101; ATR 101; ATR101; PD132301-2; PD-132301-2; PD 132301-2; PD132301; PD-132301; PD 132301.
IUPAC/Chemical Name: 1-(2,6-diisopropylphenyl)-3-((1-(4-(dimethylamino)phenyl)cyclopentyl)methyl)urea hydrochloride
ATR-101 HCl
CAS#: 133825-81-7 (ATR-101 HCl); 133825-80-6 (ATR-101).
| Molecular Formula: | C27H40ClN3O |
|---|---|
| Molecular Weight: | 458.079 g/mol |
| The Regents Of The University Of Michigan, Atterocor, Inc. |
Millendo Therapeutics is developing ATR-101, an ACAT1 inhibitor, for treating adrenal cancers including adrenocortical cancer and congenital adrenal hyperplasia.

ATR-101, also known as PD-132301 (a free base) or PD-132301-2 (a HCl salt), is in clinical development for the treatment of adrenocortical carcinoma (ACC). ATR-101 is a selective inhibitor of ACAT1 (acyl coenzyme A:cholesterol acyltransferase). ACAT1 catalyzes cholesterol ester formation and, in the adrenals, is particularly important in creating a reservoir of substrate for steroid biosynthesis. ATR-101 is uniquely distributed to adrenal tissues and inhibition of adrenal ACAT1 by ATR-101 disrupts steroidogenesis and leads to selective apoptosis of steroid producing adrenocortical-derived cells. Similar effects have been seen in the human ACC cell line, H295R. ATR-101 has shown pre-clinical efficacy in H295R xenograft mouse models. ACC is an ultra-rare malignancy, occurring in about 2 per million population annually.
ATR-101 (Atterocor, Inc., Ann Arbor, MI, USA) is in clinical development for the treatment of adrenocortical carcinoma (ACC). ATR-101 is a selective inhibitor of ACAT1 (acyl coenzyme A:cholesterol acyltransferase). ACAT1 catalyzes cholesterol ester formation and, in the adrenals, is particularly important in creating a reservoir of substrate for steroid biosynthesis. ATR-101 is uniquely distributed to adrenal tissues and inhibition of adrenal ACAT1 by ATR-101 disrupts steroidogenesis and leads to selective apoptosis of steroid producing adrenocortical-derived cells. Similar effects have been seen in the human ACC cell line, H295R. ATR-101 has shown pre-clinical efficacy in H295R xenograft mouse models. ACC is an ultra-rare malignancy, occurring in about 2 per million population annually. ACC is frequently discovered in Stage 4 and the overall disease survival is approximately 17 months. Tumors often overproduce steroids normally produced in the adrenal cortex. Current therapies are toxic, difficult to administer, and poorly effective. Clinical trial information: NCT01898715.
Adrenocortical carcinoma (ACC) generally has poor prognosis. Existing treatments provide limited benefit for most patients with locally advanced or metastatic tumors. We investigated the mechanisms for the cytotoxicity, xenograft suppression and adrenalytic activity of ATR-101 (PD132301-02), a prospective agent for ACC treatment. Oral ATR-101 administration inhibited the establishment and impeded the growth of ACC-derived H295R cell xenografts in mice. ATR-101 induced H295R cell apoptosis in culture and in xenografts. ATR-101 caused mitochondrial hyperpolarization, reactive oxygen release and ATP depletion within hours after exposure, followed by cytochrome c release, caspase-3 activation, and membrane permeabilization. When combined with ATR-101, lipophilic free radical scavengers suppressed the reactive oxygen release, and glycolytic precursors prevented the ATP depletion, abrogating ATR-101 cytotoxicity. ATR-101 directly inhibited F1F0-ATPase activity and suppressed ATP synthesis in mitochondrial fractions. ATR-101 administration to guinea pigs caused oxidized lipofuscin accumulation in the zona fasciculata layer of the adrenal cortex, implicating reactive oxygen release in the adrenalytic effect of ATR-101. These results support the development of ATR-101 and other adrenalytic compounds for the treatment of ACC.
| Company | Millendo Therapeutics Inc. |
| Description | Selective inhibitor of sterol O-acyltransferase 1 (SOAT1; ACAT1) |
| Molecular Target | Sterol O-acyltransferase 1 (SOAT1) (ACAT1) |
PATENT
WO2013142214
https://www.google.co.in/patents/WO2013142214A1?cl=en
PATENT
One such promising agent is N-(2,6-bis( 1 -methylethyl)phenyl)-N’-(( 1 -(4-(dimethyl-amino)phenyl)cyclopentyl)methyl)urea hydrochloride (“ATR-101”). The free base form of ATR-101 has the following chemical structure:

The chemical synthesis of ATR-101 has been previously reported by Trivedi et al. (J. Med. Chem. 37: 1652-1659, 1994). This procedure, however, does not provide for ATR-101 in a form suitable for solid-dosing, particularly with regard to capsule or tablet formation, and does not provide for ATR-101 in high purity.
While significant advances have been made in this field, particularly in the context of ATR-101, there remains a substantial need for improved techniques and products for the oral administration of ATR-101 to patients in need thereof, including patients having ACC and/or other disorders or conditions such as Cushing’s syndrome and congenital adrenal hyperplasia (CAH).

EXAMPLE 1
SYNTHESIS OF SOLID DRUG FORM OF ATR-101

Step 1 : Preparation of Primary Amine 2 from the Nitrile 1

Tetrahyrofuran (THF) and Compound 1 are charged to a reactor vessel and a lithium aluminum hydride (LAH) solution in THF is added slowly. After the addition, the reaction mixture is warmed to 45°C and stirred until in-process HPLC analysis indicates that the reaction is complete. The reaction mixture is cooled to between 0 and 10°C and aqueous NaOH is added slowly while controlling the temperature to between 0 and 10°C. The mixture is then warmed to between 20 and 25°C and any inorganic salts removed by filtration. The solids are then washed with additional THF.
The filtrate is distilled under vacuum. Acetonitrile (MeCN) is added and the distillation continued to reduce the total volume. H20 is added and the solution is cooled to 20°C, and seeded if necessary. Additional water is added to the slurry and cooled to between 0 and 5°C and filtered. The crystallization vessel and filter cake is washed with MeCN and water (1 :2 mixture) and dried under vacuum between 40 to 45°C to produce Compound 2. Typical yield: 85%.
Step 2: Preparation of ATR-101 Free Base

2,6-Diisopropyl aniline hydrochloride (Compound 3) is converted to the corresponding free base by stirring in a mixture of dichloromethane (DCM) and 10% aqueous NaOH. The organic phase is separated and washed with water. The DCM solution containing the aniline free base is concentrated by distillation.
4-dimethylaminopyridine (DMAP) and DCM are charged to a separate reaction vessel. The mixture is cooled and a solution of di-tert-butyl dicarbonate (Boc20) in DCM is slowly added while the temperature is maintained between 0 and 5°C. The aniline free base solution is then slowly added to the reaction vessel. A complete conversion of aniline to the isocyanate is verified by in-process HPLC analysis.
Compound 2 and MeCN are charged to a separate vessel and this solution is cooled to between 0 and 5°C. The isocyanate intermediate solution
(prepared above) is slowly added while the temperature is maintained between 0 and 5°C, and stirred until in-process HPLC indicates that the reaction is complete.
The reaction mixture is distilled under vacuum, and isopropyl alcohol
(IP A) is added and the distillation is continued. The resulting solution is cooled and seeded, if necessary. After crystallization occurs, water is added and the mixture is cooled to between 0 and 5°C, and filtered. The crystallization vessel and filter cake is washed with isopropanol: water (1 : 1) and the product cake is dried under vacuum to yield ATR-101 as the free base. Typical yield: 89 %
Step 3 : Preparation of Solid Drug Form of ATR- 101

The ATR-101 free base is dissolved in acetone and filtered to remove particulates. Additional acetone is used to rinse the dissolution vessel and filter. Concentrated hydrochloric acid (HCl) is added while maintaining the reaction at room temperature. The resultant slurry is filtered and the cake is washed with acetone. The resulting solid is dried under vacuum between 40 and 45°C to obtain the solid drug form of ATR-101. Typical yield: 70-80 %.
EXAMPLE 2
CHARACTERIZATION OF THE SOLID DRUG FORM OF ATR-101
The solid drug form of ATR-101 was analyzed to fully characterize the material and provide proof of structure.
Elemental Analysis
An elemental (CHN) analysis was conducted, in duplicate, of the solid drug form of ATR-101. The results are summarized in Table 1 and are in agreement with the theoretical values calculated for the molecular ATR-101 drug substance formula of C27H39N3O HCl.
Table 1

Chloride Content
The solid drug form of ATR-101 is prepared as its HCl salt. To confirm the chloride content (and the stoichiometry), the hydrochloride salt was analyzed by Ion Chromatography using a validated method. The w/w% result showed 7.8% chloride present. The theoretical value for a mono hydrochloride salt is 7.7%. The experimental result conforms to the theoretical value for the mono-hydrochloride salt.
Mass Spectrometry
Mass spectrometry studies were conducted in accordance with
USP<736> using an AB Sciex API 2000 LC/MS/MS system. The samples were analyzed by electrospray ionization in positive mode. The base peak observed was 422.3 (M+H-HC1), consistent with the parent compound (see Figure 1). Two minor peaks were observed, at 301.3 and 202.3 (uncharacterized fragments). The combined data of the LC/MS and CFIN results support the molecular formula assignment of C27H39N3O and mass of 421.63 g/mol for the free base and C27H39N3O . HCl (mass of 458.09 g/mol) for the mono hydrochloride salt.
Nuclear Magnetic Resonance (NMR) – 1H NMR
The proton NMR spectrum of the solid drug form of ATR-101 was obtained using a Varian Gemini 400 MHz spectrometer and. The sample was dissolved in CD3OD. The resulting proton NMR spectrum is shown in Figure 2.
Two-Dimensional (2D) NMR
The 2D proton NMR spectrum (COSY) shown in Figure 3 confirmed some of the connectivity expected for the solid drug form of ATR-101. In particular the resonance at 1.2 ppm is strongly correlated to the resonance at 3.1. This correlation together with the splitting pattern observed for the peak at 3.1 strongly suggests an isopropyl moiety. Further, the data from these spectra show a strong correlation between each of the broad peaks at 1.6-2.2 ppm, consistent with a cycloalkyl functionality in which no heteroatoms or other non-alkyl substitution is present.
Carbon 13 NMR (13C NMR)
The 100 MHz 13C NMR spectrum of the solid drug form of ATR-101 was obtained using a Varian Gemini 400 MHz spectrometer. The sample was dissolved in CD3OD. The resulting 13C NMR spectrum is shown in Figure 4. The numbering of the carbon atoms for the analysis of the spectrum is shown below, and the interpretation is shown in Table 2. The observed signals are consistent with the structure of ATR-101.

Table 2


Fourier Transform Infrared Spectroscopy (IR)
Infrared (IR) spectroscopy was performed using the soid drug form of ATR-101. The resulting spectrum, shown in Figure 5, is consistent with the structure of ATR-101 drug substance. The major peak assignments are presented in Table_3.
Table 3

EXAMPLE 3
COMPARISON WITH PRIOR ART SYNTHESIS OF ATR-101 (BY TRIVEDI ETAL.. J. MED. CHEM. 37: 1652-1659, 1994)

ATR-101
In this experiment, 10.6 g of ATR-101 was synthesized according to the above procedure, which corresponds to the the procedure set forth in Trivedi et al., J. Med. Chem. 137: 1652-1659, 1994 (hereinafter referred to as the “Trivedi procedure”). The purity of ATR-101 as made by the Trivedi procedure was found to be 94.9%, compared to a purity of 98.3% for ATR-101 obtained by the procedure of Example 1 and as evaluated in Example 2.
Step 1 : Alkylation of p-nitrophenylacetonitrile

52
The initial alkylation reaction was run on 15.0 g scale and, according to the Trivedi procedure, should have given 15.7 g (79%) of product 52. However, several problems occurred, and the yield was much lower than expected (6.0 g, 30% yield), although the purity by 1H NMR and melting point (actual: 71-72°C, reported: 76°C) seemed good. Approximately half way through the addition of 1 ,4-bromobutane and p- nitrophenylacetonitrile to NaH, a black solid precipitated out of the purple solution causing the stirbar in the flask to skip and jump. The rate of stirring had to be monitored throughout the remainder of the addition to maintain a sluggish and inefficient mixing of the solution.
After stirring at ambient temperature overnight to ensure reaction completion, the reaction was worked-up as the procedure indicated. First, excess ether was removed using air bubbling, and the black solid was isolated by filtration. Diethyl ether was then added until all of the solids dissolved to give a clear black solution. However, upon washing the ether solution with 2N HC1, a black amorphous solid precipitated from the solution. There was no note of this black solid in the Trivedi procedure, so the work-up was continued without modification. The black solids ended up in the aqueous washes, or stuck to the seperatory funnel. The remainder of the work-up proceeded as expected, and the hot hexanes extraction of the crude solid resulted in light pink planar crystals.
The procedure was repeated with two changes thought to be responsible for the low yield: the anhydrous solvent (from the bottle) was sieve dried to remove trace water, and the stir bar was replaced with a mechanical stirrer to ensure more even mixing of the solution. The procedure was re-run on 10 g scale, which should have yielded 10.5 g of compound 52. However, despite the changes to the procedure, the resulting product and yield was nearly identical to the first run (4.5 g, 34% yield, 71-72°C melting point).
In an attempt to determine where the bulk of material ended up, the aqueous layer from this reaction was re-extracted with diethyl ether, but only resulted in trace amounts of material. The black solids that formed during the work-up were isolated by filtration, and an NMR was taken of the material. The NMR showed peaks corresponding to compound 52. Presumably, this amorphous black solid that resulted after HC1 formation is the main source of lost material, as there appeared to be several grams of it.
Ste 2: Reduction of Nitro Compound

The conversion of nitro compound 52 to the dimethyl amine 53 was done over two steps: palladium catalyzed hydrogenation of the nitro compound to give the free amine 52b, followed by imine formation & reduction to the dimethylamine 53.
An exploratory small scale reaction was run, using 1/10th of the available material (1.0 g compound 52). The reduction of the nitro compound on the 1 gram scale was very rapid, with hydrogen consumption ceasing after 3-4 hours. A crude NMR of an aliquot of the reaction mixture showed very clean amine (52b). The formaldehyde was added, as well as additional Pd/C, and the hydrogenation was continued. The hydrogen was not consumed as quickly for the imine reduction, and the reaction was still progressing when the vessel was pressurized to 55 psi and left shaking overnight (ca. 16h).
After 16 hours, the pressure in the flask had dropped to 30 psi, indicating that the hydrogenation was still progressing overnight. An aliquot NMR confirmed that the reaction had not proceeded to completion.
On large scale, the nitro reduction proceeded very smoothly, consuming hydrogen at a very rapid rate, and going to completion again within 3-4 hours. The reactor was pressurized to 55 psi and shaken overnight, as indicated in the original procedure, before more Pd/C was added, followed by formaldehyde. Hydrogen consumption was again observed to be very sluggish, so the valve to the hydrogen tank was left open to the vessel, and the reaction was shaken for 24 hours.
After 24 hours of shaking, the valve to the vessel was closed, and a drop of 5 psi was observed over 1 hour, indicating that the reaction had not progressed to completion. TLC also showed several polar products, suggesting that the reaction was only ca. 50% complete. The hydrogenation vessel was pressurized to 55 psi with hydrogen, and the valve again left open for an additional 24 hours of hydrogenation.
After 24 hours, the reaction stopped consuming hydrogen, and the vessel was purged and the contents filtered to remove the palladium catalyst. The work-up was performed similarly to the small scale, and the two reactions were combined prior to purification by column chromatography, giving 5.7g (57.5% yield) of the desired dimethylamine product 53.
Step 3 : Reduction of C ano Compound

A small scale RaNi hydrogenation was done and the test reaction went smoothly. Hydrogen consumption was rapid, and the reaction appeared complete after approximately 2 hours. The consumption of hydrogen had ceased, and TLC indicated that there was no compound 53 remaining. After filtration to remove the Raney Nickel, the reaction completion was confirmed by aliquot NMR.
The remaining material was subjected to reduction using the same conditions, and hydrogen consumption and TLC analysis again indicated reaction completion after 2 hours. The material was filtered and combined with the smaller scale reaction material. After concentration to dryness, the crude yield was found to be 5.5 g (96.5% yield), which was very close to the reported yield (99%>).
Step 4: Formation of Urea Com ound

Urea formation is a straightforward procedure, and the small scale test reaction with the amine 54 (500 mg) being combined with 1.0 equivalent of the
isocyanate in 20 parts ethyl acetate. After stirring for 16 hours, the solution was concentrated to dryness to give a white solid. Crude 1H NMR of the solid confirmed that the spectra matched the reported spectra in the Trivedi procedure.
The remaining material was carried forward to ATR-101 freebase without difficulty, and the lots of product were combined. In an effort to remove the residual ethyl acetate, the solids were dissolved in 10 mL of toluene, followed by concentration under reduced pressure. After drying on high- vacuum, ATR-101 freebase was isolated as a sticky white foam (10.6 g, 99% yield). The 1H NMR of the final product showed trace toluene even after extended drying, and the material was moved on to the HC1 salt formation.
The melting point of the solid was later taken and found to be surprisingly low (50-56°C, expected: 132-133°C). The nature of the solid (oily foam) made the determination of the melting point difficult, but it was judged to be completely melted above 60°C.
Step 5: Formation of HC1 Salt

To the ATR-101 freebase in toluene was added 37% HC1, and a gummy white solid precipitated out immediately. The solution was dried by Dean-Stark apparatus over approximately 3 hours with vigorous stirring and heating (bath temp: 160°C). After drying, the solution was cooled and the fine crystalline solid was isolated by filtration and washed with acetone and diethyl ether. The product ATR-101 was dried until a constant weight was achieved (10.6 g, 92% yield) and fully characterized.
Figure 1 is the LC/MS Mass spectrum of the solid drug form of ATR- 101.
Figure 2 is the proton NMR spectrum of the solid drug form of ATR- 101.
Figure 3 is the 2-D 1H NMR spectrum (COSY) of the solid drug form of ATR-101.
Figure 4 is the 13C NMR spectrum of of the solid drug form of ATR- 101.
Figure 5 is the FT-IR spectrum the solid drug form of ATR-101.
Paper
(J. Med. Chem. 37: 1652-1659, 1994
http://pubs.acs.org/doi/abs/10.1021/jm00037a016
| Patent ID | Date | Patent Title |
|---|---|---|
| EP0474733 | 1994-08-31 | ANTIHYPERLIPIDEMIC AND ANTIATHEROSCLEROTIC UREA COMPOUNDS. |
| WO9015048 | 1990-12-13 | ANTIHYPERLIPIDEMIC AND ANTIATHEROSCLEROTIC UREA COMPOUNDS |
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015087649 | 2015-03-26 | TREATING DISORDERS ASSOCIATED WITH ABERRANT ADRENOCORTICAL CELL BEHAVIOR |
| US2013267550 | 2013-10-10 | Compounds and Methods for Treating Aberrant Adrenocartical Cell Disorders |
| EP0858336 | 2006-12-20 | METHOD AND PHARMACEUTICAL COMPOSITION FOR REGULATING LIPID CONCENTRATION |
| US2005234124 | 2005-10-20 | Carboxyalkylether-ACAT inhibitor combinations |
| US2004072903 | 2004-04-15 | Carboxyalkylether-acat inhibitors combinations |
| US6143755 | 2000-11-07 | Pharmaceutical methods of treatment with ACAT inhibitors and HMG-CoA reductase inhibitors |
| US6124309 | 2000-09-26 | Method and pharmaceutical composition for regulating lipid concentration |
| US6093719 | 2000-07-25 | Method and pharmaceutical composition for regulating lipid concentration |
| WO9716184 | 1997-05-09 | METHOD AND PHARMACEUTICAL COMPOSITION FOR REGULATING LIPID CONCENTRATION |
| EP0474733 | 1994-08-31 | ANTIHYPERLIPIDEMIC AND ANTIATHEROSCLEROTIC UREA COMPOUNDS. |
References
1: Wolfgang GH, MacDonald JR, Vernetti LA, Pegg DG, Robertson DG. Biochemical alterations in guinea pig adrenal cortex following administration of PD 132301-2, an inhibitor of acyl-CoA:cholesterol acyltransferase. Life Sci. 1995 Feb 17;56(13):1089-93. PubMed PMID: 9001442.
2: Saxena U, Ferguson E, Newton RS. Acyl-coenzyme A:cholesterol-acyltransferase (ACAT) inhibitors modulate monocyte adhesion to aortic endothelial cells. Atherosclerosis. 1995 Jan 6;112(1):7-17. PubMed PMID: 7772069.
3: Reindel JF, Dominick MA, Bocan TM, Gough AW, McGuire EJ. Toxicologic effects of a novel acyl-CoA:cholesterol acyltransferase inhibitor in cynomolgus monkeys. Toxicol Pathol. 1994 Sep-Oct;22(5):510-8. PubMed PMID: 7899779.
4: Krause BR, Black A, Bousley R, Essenburg A, Cornicelli J, Holmes A, Homan R, Kieft K, Sekerke C, Shaw-Hes MK, et al. Divergent pharmacologic activities of PD 132301-2 and CL 277,082, urea inhibitors of acyl-CoA:cholesterol acyltransferase. J Pharmacol Exp Ther. 1993 Nov;267(2):734-43. PubMed PMID: 8246149.
5: Dominick MA, McGuire EJ, Reindel JF, Bobrowski WF, Bocan TM, Gough AW. Subacute toxicity of a novel inhibitor of acyl-CoA: cholesterol acyltransferase in beagle dogs. Fundam Appl Toxicol. 1993 Feb;20(2):217-24. PubMed PMID: 8383621.
6: Dominick MA, Bobrowski WA, MacDonald JR, Gough AW. Morphogenesis of a zone-specific adrenocortical cytotoxicity in guinea pigs administered PD 132301-2, an inhibitor of acyl-CoA:cholesterol acyltransferase. Toxicol Pathol. 1993;21(1):54-62. PubMed PMID: 8397438.
///////ATR 101, 133825-81-7, ATR-101 HCl, 133825-80-6, Millendo Therapeutics, ACAT1 inhibitor, treating adrenal cancers, adrenocortical cancer, congenital adrenal hyperplasia, Atterocor, Inc., Ann Arbor, MI, USA
O=C(NCC1(C2=CC=C(N(C)C)C=C2)CCCC1)NC3=C(C(C)C)C=CC=C3C(C)C.[H]Cl
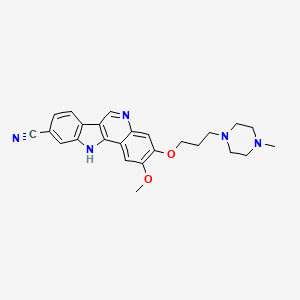
Henagliflozin

Henagliflozin, SHR-3824 ,
CAS 1623804-44-3
C22-H24-Cl-F-O7, 454.8756
PHASE 2 for the treatment of type 2 diabetes
China 20222, approvals 2022
HengRui (Originator)
| Jiangsu Hengrui Medicine Co Ltd |
UNII-21P2M98388; 21P2M98388; Henagliflozin; SHR3824; SHR-3824;
- HENAGLIFLOZIN PROLINE
- 4IO819SW6M
- 570.0 g/mol
- C27H33ClFNO9
- (1R,2S,3S,4R,5R)-5-[4-chloro-3-[(4-ethoxy-3-fluorophenyl)methyl]phenyl]-1-(hydroxymethyl)-6,8-dioxabicyclo[3.2.1]octane-2,3,4-triol;(2R)-pyrrolidine-2-carboxylic acid
In April 2016, Jiangsu Hengrui Medicine is developing henagliflozin (phase 2 clinical trial), a sodium-glucose cotransporter-2 (SGLT-2) inhibitor, for treating type 2 diabetes.
SGLT1 and SGLT2 inhibitors, useful for treating eg diabetes.
Henagliflozin proline is in phase II clinical trials by Jiangsu Hengrui (江苏恒瑞) for the treatment of type 2 diabetes.
1,6-dehydrated-1-C{4-chloro-3-[(3-fluoro-4-ethoxyphenyl)methyl]phenyl}-5-C-(hydroxymethyl)-β-L-idopyranose L-proline
(1 ^ 2345-5- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] -1- (hydroxymethyl) 6,8 – alcohol dioxide
(1R,2S,3S,4R,5R)-5-[4-chloro-3-[(4-ethoxy-3-fluorophenyl)methyl]phenyl]-1-(hydroxymethyl)-6,8-dioxabicyclo[3.2.1]octane-2,3,4-triol
Henagliflozin is a pharmaceutical drug for the treatment of type 2 diabetes.[1] In China, it is approved for adult patients with type 2 diabetes to improve the glycemic control.[2][3]
Henagliflozin, like other drugs of the gliflozin class, inhibits the transporter protein sodium/glucose cotransporter 2 (SGLT2) which leads to a reduction in blood glucose levels.[4]
Shanghai Hengrui Pharmaceutical Co., Ltd., 上海恒瑞医药有限公司, Jiangsu Hengrui Medicine Co., Ltd., 江苏恒瑞医药股份有限公司, Less «
- 01 May 2015 Jiangsu HengRui Medicine Co. initiates enrolment in a phase I drug interaction trial in volunteers in China (NCT02500485)
- 12 Feb 2015 Jiangsu HengRui Medicine plans a phase I trial for Type-2 diabetes mellitus in China (NCT02366377)
- 01 Feb 2015 Jiangsu HengRui Medicine initiates enrolment in a phase I trial for Type-2 diabetes mellitus in China (NCT02366351)
Henagliflozin is a novel sodium-glucose transporter 2 inhibitor and presents a complementary therapy to metformin for patients with T2DM due to its insulin-independent mechanism of action. This study evaluated the potential pharmacokinetic drug-drug interaction between henagliflozin and metformin in healthy Chinese male subjects. 2. In open-label, single-center, single-arm, two-period, three-treatment self-control study, 12 subjects received 25 mg henagliflozin, 1000 mg metformin or the combination. Lack of PK interaction was defined as the ratio of geometric means and 90% confidence interval (CI) for combination: monotherapy being within the range of 0.80-1.25. 3. Co-administration of henagliflozin with metformin had no effect on henagliflozin area under the plasma concentration-time curve (AUC0-24) (GRM: 1.08; CI: 1.05, 1.10) and peak plasma concentration (Cmax) (GRM: 0.99; CI: 0.92, 1.07). Reciprocally, co-administration of metformin with henagliflozin had no clinically significant on metformin AUC0-24 (GRM: 1.09, CI: 1.02, 1.16) although there was an 11% increase in metformin Cmax (GRM 1.12; CI 1.02, 1.23). All monotherapies and combination therapy were well tolerated. 4. Henagliflozin can be co-administered with metformin without dose adjustment of either drug.
PATENT
PATENT
WO2012019496
https://www.google.com/patents/WO2012019496A1?cl=en
Example 4
(1 ^ 2345-5- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] -1- (hydroxymethyl) 6,8 – alcohol dioxide
first step
1-ethoxy-2-fluoro – benzene
A mixture of 2-fluoro-phenol 4a (6.7 g, 60 mmol) was dissolved in 66 mL of acetone, was added iodoethane (6.3 mL,
78 mmol) and potassium carbonate (12.4 g, 90 mmol), at reflux in an oil bath for 5 hours. The reaction solution was concentrated under reduced pressure, was added 100 mL of ethyl acetate and 60 mL of water, separated, the aqueous phase was extracted with ethyl acetate (30 mLx2), the organic phases combined, dried over anhydrous magnesium sulfate, filtered, and the filtrate was concentrated under reduced pressure, to give the title product 1-ethoxy-2-fluoro – benzene 4b (6.9 g, red oil). yield: 82.1%.
MS m / z (ESI): 280.2 [2M + 1]
The second step
(5-bromo-2-chloro – phenyl) – (4-ethoxy-3-fluoro-phenyl) – methanone A mixture of 5-bromo-2-chloro – benzoyl chloride 2a (12.4 g, 48.8 mmol) was dissolved a 100 mL of dichloromethane was added 1-ethoxy-2-fluoro – benzene 4b (6.84 g, 48.8 mmol), cooled to 0 ° C, was added portionwise aluminum (5.86 g, 44 mmol) chloride, 16 h. Was added dropwise under ice-cooling to the reaction mixture 20 mL of 2 M HCl solution, separated, the aqueous phase was extracted with 30 mL of dichloromethane, and the combined organic phase was dried over anhydrous magnesium sulfate, filtered, and the filtrate was concentrated under reduced pressure to give the title The product (5-bromo-2-chloro – phenyl) – (4-ethoxy-3-fluoro-phenyl) – methanone 4c (12.7 g, yellow solid), yield: 72.6%.
MS m / z (ESI): 358.9 [M + l] Step
(5 – bromo-2-chloro – phenyl) – (4-ethoxy-3-fluoro-phenyl) – methanol (5-Bromo-2-chloro – phenyl) – (4-ethoxy -3 – fluoro – phenyl) -methanone 4c (12.7 g, 35.5 mmol) was dissolved in methanol and a 100 mL of tetrahydrofuran (ν: ν = 1: 1) mixed solvent, under an ice bath was added portionwise sodium borohydride (2.68 g, 70 mmol), and reacted at room temperature for 30 minutes. Add 15 mL of acetone, the reaction solution was concentrated under reduced pressure, 150 mL of ethyl acetate was added to dissolve the residue, washed with saturated sodium chloride solution (50 mLx2). The combined organic phase was dried over anhydrous magnesium sulfate, filtered, and concentrated under reduced pressure The filtrate, to give the title product (5-bromo-2-chloro – phenyl) – (4-ethoxy-3-fluoro-phenyl) – methanol 4d (12.7 g, orange oil), was used directly without isolation next reaction.
the fourth step
4 – [(5-bromo-2-chloro-phenyl) – methyl] Small-ethoxy-2-fluoro – benzene (5-bromo-2-chloro – phenyl) – (4-ethoxy -3 – fluoro – phenyl) methanol 4d (12.7 g, 35.3 mmol) was dissolved in a 100 mL of dichloromethane was added triethylsilane (16.9 mL, 106 mmol), was added dropwise boron trifluoride etherate (8.95 mL, 70.6 mmol ), for 3 hours. Was added 50 mL of saturated sodium bicarbonate solution, separated, the aqueous phase was extracted with ethyl acetate (100 mLx2), the organic phases combined, dried over anhydrous magnesium sulfate, filtered, and the filtrate was concentrated under reduced pressure, purified by silica gel column chromatography to elute B surfactant system resulting residue was purified to give the title product 4 – [(5-bromo-2-chloro – phenyl) methyl] -1-ethoxy-2-fluoro – benzene 4e (10 g, as a pale yellow oil ) yield: 82.4%.
1H NMR (400 MHz, CDC1 3 ): δ 7.33-7.27 (m, 3H), 6.95-6.90 (m, 3H), 4.14 (q, 2H), 4.01 (s, 2H), 1.49 (t, 3H)
the fifth step
(2 3R, 4S, 5 ^ 6R) -2- [4- chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] -6- (hydroxymethyl) – 2-methoxy – tetrahydro-pyran-3,4,5-triol
4 – [(5-bromo-2-chloro – phenyl) methyl] -1-ethoxy-2-fluoro – benzene 4e (7.36 g, 21.4 mmol) was dissolved in 30 mL of tetrahydrofuran, cooled to -78 ° C, was added dropwise a solution of n-butyllithium in hexane (10.27 mL, 25.7 mmol), at -78 ° C to react 1 hour, a solution of 20 mL (3R, 4S, 5R, 6R) -3,4,5 – tris (trimethylsilyloxy) -6- (trimethylsilyloxy) tetrahydropyran-2-one 2f (llg, 23.6 mmol) in tetrahydrofuran at -78 ° C under reaction 2 h, 2.8 mL of methanesulfonic acid and 71 mL of methanol, the reaction at room temperature for 16 hours. Was added 100 mL of saturated sodium carbonate solution, the reaction solution was concentrated under reduced pressure, to the residue was added 50 mL of saturated sodium chloride solution, extracted with ethyl acetate (100 mLx3), organic phases were combined, dried over anhydrous magnesium sulfate, filtered, The filtrate was concentrated under reduced pressure, purified by silica gel column chromatography with eluent systems resulting A residue was purified to give the title product (2 3R, 4S, 5 6R) -2- [4- chloro-3 – [(4-ethoxyphenyl 3-fluoro-phenyl) – methyl] phenyl] -6- (hydroxymethyl) -2-methoxy – tetrahydro-pyran-3,4,5-triol 4f (5.7 g, white solid ) yield: 58.3%.
1H NMR (400 MHz, CD 3 OD): δ 7.56 (s, 1H), 7.48 (dd, 1H), 7.37 (dd, 1H), 6.95-6.87 (m, 3H), 4.08-4.07 (m, 4H) , 3.91 (m, 1H), 3.93-3.73 (m, 2H), 3.56-3.53 (m, 1H), 3.45-3.43 (m, 1H), 3.30 (s, 2H), 3.08 (s, 3H), 1.35 (t, 3H)
The sixth step
(2 3R, 4S, 5 6R) -6- [(tert-butyl (dimethyl) silyl) oxymethyl] -2- [4-chloro-3 – [(4-ethoxy-3-fluoro – phenyl) methyl] phenyl] -2-methoxy – tetrahydro-pyran-3,4,5-triol the (2 3R, 4S, 5 6R) -2- [4- chloro-3- [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] -6- (hydroxymethyl) -2-methoxy – 4f tetrahydropyran-3,4,5-triol (5.7 g, 12.5 mmol) was dissolved in 50 mL of pyridine, followed by adding tert-butyldimethylsilyl chloride (2.26 g, 15 mmol) and 4-dimethylaminopyridine (305 mg, 2.5 mmol), for 16 hours. The reaction solution was concentrated under reduced pressure, was added 200 mL of ethyl acetate, washed with a saturated copper sulfate solution (50 mLx3). The combined organic phase was dried over anhydrous magnesium sulfate, filtered, and the filtrate was concentrated under reduced pressure to give the title product (2 3R, 4S, 5 6R) -6- [(tert-butyl (dimethyl) silyl) oxymethyl] -2- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] -2-methoxy – tetrahydro-pyran-3,4,5-triol 4g (7.14 g, colorless oil), without isolation directly used for the next reaction.
Seventh Step
[[(2R, 3R, 4S, 5R, 6 ^ -3,4,5- tris-benzyloxy-6- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl yl] phenyl] -6-methoxy – tetrahydropyran-2-yl] methoxy] – tert-butyl – dimethyl-silane (2 3R, 4S, 5 6R) -6- [(tert butyl (dimethyl) silyl) oxymethyl] -2- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] -2-methoxy yl – tetrahydro-pyran-3,4,5-triol 4g (7.14 g, 12.5 mmol) was dissolved in 100 mL N, N- dimethylformamide was added 60% sodium hydride under ice-cooling (2.5 g , 62.5 mmol), and reacted at room temperature for 40 minutes completed the opening force, was added benzyl bromide (7.5 mL, 62.5 mmol), reaction of 16 hours. 20 mL of methanol, the reaction solution was concentrated under reduced pressure, was added 200 mL of ethyl acetate and 50 mL of water to dissolve the residue, separated, the aqueous phase was extracted with ethyl acetate (50 mL), the organic phase was washed with water (50 mL), washed with saturated sodium chloride solution (50 mL), the combined organic phase was dried over anhydrous magnesium sulfate , filtered, and the filtrate was concentrated under reduced pressure to give the title product [[(2R, 3R, 4S, 5R, 6 ^ -3,4,5- tris-benzyloxy-6- [4-chloro-3 – [(4- ethoxy-3-fluoro-phenyl) – methyl] phenyl] -6-methoxy – tetrahydropyran-2-yl] methoxy] – tert-butyl – dimethylsilane 4h (10.5 g , yellow oil) yield: 99.8%.
Step Eight
[(2R, 3R, 4S, 5R, 6 -3,4,5- tris-benzyloxy-6- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] -6-methoxy – tetrahydropyran-2-yl] methanol
The [[(2R, 3R, 4S, 5R, 6 -3,4,5- tris-benzyloxy-6- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl yl] phenyl] -6-methoxy – tetrahydropyran-2-yl] methoxy] – tert-butyl – dimethylsilane 4h (10.52 g, 12.5 mmol) was dissolved in 50 mL of methanol dropwise add acetyl chloride CO.13 mL, 1.9 mmol), for 1 hour. The reaction solution was concentrated under reduced pressure, purified by silica gel column chromatography with eluent systems B resultant residue was purified to give the title product [(2R, 3R, 4S, 5R, 6 -3,4,5- tris-benzyloxy–6 – [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] -6-methoxy – tetrahydropyran-2-yl] methanol 4i (7.6 g , yellow oil yield: 83.6%.
Step Nine
(2 ^ 3456 3,4,5-tris-benzyloxy-6- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] – 6-methoxy – tetrahydropyran-2-carbaldehyde
Oxalyl chloride (1.17 mL, 13.6 mmol) was dissolved in 20 mL of dichloromethane, cooled to -78 ° C, were added dropwise 20 mL of dimethyl sulfoxide (1.56 mL, 21.9 mmol) in methylene chloride and 50 mL [(2R, 3R, 4S, 5R, 6 -3,4,5- tris-benzyloxy-6- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] -6-methoxy – tetrahydropyran-2-yl] methanol 4i (7.6 g, 10.45 mmol) in methylene chloride, and reacted at -78 ° C for 30 min, triethylamine (7.25 mL, 52.3 mmol), 2 hours at room temperature was added 50 mL 1 M HCl solution, separated, the organic phase was washed with saturated sodium chloride solution (50 mL x 2), the aqueous phase was extracted with dichloromethane (50 mL), the combined organic phase was dried over anhydrous magnesium sulfate, filtered, and the filtrate was concentrated under reduced pressure to give the title product (2 ^ 3456 3,4,5-tris-benzyloxy-6- [4-chloro-3 – [(4 – ethoxy-3-fluoro-phenyl) – methyl] phenyl] -6-methoxy – tetrahydropyran-2-carbaldehyde 4j (7.58 g, colorless oil), was used directly without isolation next reaction.
The tenth step
(2S, 3 4S, 5R, 6 -3,4,5- tris-benzyloxy-6- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl ] -2- (hydroxymethyl) -6-methoxy – tetrahydropyran-2-carbaldehyde
The (23456 3,4,5-tris-benzyloxy-6- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] – 6-methoxy – tetrahydropyran-2-carbaldehyde 4j (7.6 g, 10.45 mmol) was dissolved in 80 mL 1,4- dioxane, followed by adding 15.8 mL 37% aqueous formaldehyde and sodium hydroxide solution (31.35 mL, 31.35 mmol), reacted at 70 ° C for 16 h. Add 50 mL of saturated sodium chloride solution, extracted with ethyl acetate (50 mLx4), the organic phase was washed with saturated sodium bicarbonate solution (50 mL), washed with saturated sodium chloride solution (50 mL), the combined organic phase was dried over anhydrous magnesium sulfate, filtered, and the filtrate was concentrated under reduced pressure to give the title product (23,456 benzyloxy-3,4,5-tris – 6- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] -2- (hydroxymethyl) -6-methoxy – tetrahydropyran – 2- formaldehyde 4k (7.9g, as a colorless oil), without isolation directly used for the next reaction.
Step Eleven
[(3 4S, 5R, 6 -3,4,5- tris-benzyloxy-6- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] 2- (hydroxymethyl) -6-methoxy – tetrahydropyran-2-yl] methanol
The (23456 3,4,5-tris-benzyloxy-6- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] – 2- (hydroxymethyl) -6-methoxy – tetrahydropyran-2-carbaldehyde 4k (7.9 g, 10.45 mmol) was dissolved in 50 mL of tetrahydrofuran and methanol (v: v = 2: 3) mixed solvent , was added sodium borohydride (794 mg, 20.9 mmol), for 30 minutes. Add a small amount of acetone, the reaction solution was concentrated under reduced pressure, purified by silica gel column chromatography with eluent systems resulting A residue was purified to give the title product, 5R, 6 -3,4,5-tris-benzyloxy-6- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] -2- (hydroxymethyl ) -6-methoxy – tetrahydropyran-2-yl] methanol 4m (l.ll g, colorless oil). yield: 14.1%.
Step Twelve
[(12345 ^ -2,3,4-tris-benzyloxy-5- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] 6,8-dioxa-bicyclo [3.2.1] octane-1-yl] methanol
The [(3S, 4S, 5R, 6 -3,4,5- tris-benzyloxy-6- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] benzene yl] -2- (hydroxymethyl) -6-methoxy – tetrahydropyran-2-yl] methanol 4m (l.ll g, 1.46 mmol) was dissolved in 20 mL of dichloromethane, cooled to -10 ° C, was added trifluoroacetic acid (0.23 mL, 3 mmol), and reacted at room temperature for 2 hours. 20 mL of saturated sodium bicarbonate solution, separated, the aqueous phase was extracted with dichloromethane (20 mL> <2), and the combined organic phase was dried over anhydrous magnesium sulfate, filtered, and the filtrate was concentrated under reduced pressure, purified by silica gel column chromatography with eluent systems B resultant residue was purified to give the title product [(1 2 3 4R, 5 -2,3,4- tris-benzyloxy-5- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] 6,8-dioxa-bicyclo [3.2.1] octane-1-yl] methanol 4nC830 mg, colorless oil). yield: 78.3%.
MS m / z (ESI): 742.3 [M + 18]
Thirteenth Step
(12345-5- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] -1- (hydroxymethyl) -6,8 dioxa-bicyclo [3.2.1] octane-2,3,4-triol
The [(1 2 3 4R, 5S) -2,3,4- tris-benzyloxy-5- [4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] benzene yl] -6,8-dioxa-bicyclo [3.2.1] octane-1-yl] methanol 4n (830 mg, 1.14 mmol) was dissolved in 20 mL of tetrahydrofuran and methanol (v: v = l: l) the a mixed solvent of o-dichlorobenzene was added (1.3 mL, 1 1.4 mmol) and Pd / C (500 mg, 10%), purged with hydrogen three times, the reaction for 3 hours. The reaction solution was filtered, rinsed with a small amount of ethyl acetate, the filtrate was concentrated under reduced pressure, purified by silica gel column chromatography with eluent systems resulting A residue was purified to give the title product (1S, 2 3S, 4R, 5 -5- [ 4-chloro-3 – [(4-ethoxy-3-fluoro-phenyl) – methyl] phenyl] -1- (hydroxymethyl) -6,8-dioxa-bicyclo [3.2.1] octane-2,3,4-triol 4 (420 mg, white solid), yield: 81.0% MS m / z (ESI):. 472.2 [m + 18]
1H NMR (400 MHz, CD 3 OD): δ 7.47 (s, 1H), 7.42-7.35 (m, 2H), 6.95-6.87 (m, 3H), 4.16-4.14 (m, 1H), 4.06-4.02 ( m, 4H), 3.85-3.70 (m, 2H), 3.67-3.54 (m, 4H), 1.37 (t, 3H)
References
- Weng J, Zeng L, Zhang Y, Qu S, Wang X, Li P, et al. (August 2021). “Henagliflozin as add-on therapy to metformin in patients with type 2 diabetes inadequately controlled with metformin: A multicentre, randomized, double-blind, placebo-controlled, phase 3 trial”. Diabetes, Obesity & Metabolism. 23 (8): 1754–1764. doi:10.1111/dom.14389. PMID 33769656.
- Wang G (17 February 2022). “Monthly Report: New Drug Approvals in China, January 2022”. BaiPharm.
Henagliflozin Proline Tablets
- “Henagliflozin – Jiangsu HengRui Medicine”. AdisInsight. Springer Nature Switzerland AG.
- He X, Liu G, Chen X, Wang Y, Liu R, Wang C, et al. (July 2023). “Pharmacokinetic and Pharmacodynamic Interactions Between Henagliflozin, a Novel Selective SGLT-2 Inhibitor, and Warfarin in Healthy Chinese Subjects”. Clinical Therapeutics. 45 (7): 655–661. doi:10.1016/j.clinthera.2023.06.002. PMID 37451912.
 |
|
| Clinical data | |
|---|---|
| Trade names | Rui Qin; 瑞沁 |
| Other names | SHR3824; SHR-3824 |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| UNII | |
| Chemical and physical data | |
| Formula | C22H24ClFO7 |
| Molar mass | |
////////Henagliflozin, SHR-3824 , PHASE 2, type 2 diabetes, UNII-21P2M98388, 21P2M98388, SHR 3824, SHR3824, approvals 2022, china 2022, Henagliflozin proline
CCOc1ccc(cc1F)Cc2cc(ccc2Cl)[C@]34[C@@H]([C@H]([C@@H]([C@](O3)(CO4)CO)O)O)O
SYN
Synthesis 2024, 56, 906–943
Henagliflozin (12) (also known as SHR3824), developed by Lexicon Pharmaceuticals (Princeton, NJ, USA), is a potent and selective SGLT inhibitor administered orally. In 2013, the first synthetic route for the preparation of henagliflozin (12) was described and claimed by two pharmaceutical companies: Shanghai Hengrui Pharmaceutical Co., Ltd., and Jiangsu Hengrui Medicine Co., Ltd. Several other C-aryl-glucoside-type derivatives were prepared and registered in the United States under patent application number US8609622B2.67 Among these derivatives, the synthesis of henagliflozin (12) was carried out using a thirteen-step process, resulting in an overall yield of 3% (Schemes 40 and 41). The process consisted of the formation of the key intermediate 215 starting from commercially available 2-fluorophenol (211). In the first step, phenolic compound 211 was converted into 212 in 82% yield using ethyl bromide and po
tassium carbonate in acetone. The Friedel–Crafts reaction of acid chloride 26c′ using AlCl3 in DCM afforded intermediate 213 in 72% yield, which was further reduced to 214 using NaBH4 in a mixture of THF/MeOH. Without further isolation, the reduction of 214 was carried out using Et3SiH and BF3·Et2O in DCM to give 215 (Scheme 40). The intermediate 215 was taken forward for lithium halogen exchange using n-BuLi followed by addition of the lithiated compound to O-silyl-protected compound 22 at
low temperature to afford a lactol intermediate. The obtained lactol intermediate was protected using
MsOH/MeOH to give the desired product 216 in 58% yield. Under the above conditions, deprotection of the O-silylgroups of the C-glucoside 22 was also observed. Further, under basic conditions, the secondary hydroxy group of 216 was silyl protected using tert-butyldimethylsilyl chloride (TBSCl) and DMAP to afford compound 217, which was treated with NaH and BnBr to give benzylated compound
218 in excellent yield. In methanol solution, deprotection of the silyl protecting group of compound 218 using acetylchloride afforded 219. Swern oxidation of the hydroxy compound 219 in the presence of oxalyl chloride and DMSO gave intermediate 220, which was used for the next step without isolation. The crude compound 220 was treated with NaOH and 37% formaldehyde solution to afford 221.
Dihydroxy intermediate 222 was then obtained in low yield via reduction of the aldehyde group of compound 221 with sodium borohydride in THF/MeOH mixture. Next, treatment of 222 with trifluoroacetic acid gave compound 223. Debenzylation of compound 223 was carried out by Pd/C
catalytic hydrogenation to afford the final product henaglifozin (12) (Scheme 41).
The highlight of the synthesis is the design of the route with minimal isolation stages and intermediates possessing unstable functional groups were subjected to subsequent transformations in situ. The drawbacks of the above synthetic process are the use of a protection and deprotection
strategy that led to low throughput and the final compound being obtained in low yield. Reduction of the aldehyde in 221 mediated by sodium borohydride resulted in a poor yield of product 222, and this procedure is not recommend ed for scale-up due to safety concerns. Additionally, the use
of palladium in the last step of the synthesis involves the risk of this toxic metal leaching into the final product. To address the issue with the discovery route, Yongjun and co-workers reported an alternative approach to obtain compound 12 (Scheme 42).68 The authors published the synthesis of henagliflozin proline (12a) starting from TMS protected D-glucolactone 22 and aglycone intermediate The diol 226 was obtained after carrying out a disproportionation reaction on the aldehyde using paraformaldehyde under strong alkaline conditions. Intramolecular etherification of diol 226 using 30% HCl gave henagliflozin
(12) in 95% yield, which was further treated with L-proline to give henagliflozin proline monohydrate 12a. The authors reported several advantages such as easy steps, cost-effective procedures, simple product purification and an overall method that was amenable for commercialization. This Addition of the aglycone intermediate 215 was carried out with 22 followed by mesylation of the OH group to provide 216 in 65% yield. Further, all the secondary hydroxy groups of intermediate 216 were selectively protected us ing TMSCl, imidazole and PPTS to give 224 in 95% yield. The free primary hydroxy group of 224 was oxidized using pyridine sulfur trioxide in triethylamine and DMSO to afford process involves 10 steps and gave an overall yield of 22% of henagliflozin proline (12a) (Schemes 40 and 42)
REF 67, 68
(67) Yang, F.; Tang, P. C.; Dong, Q.; Tu, W.; Fan, J.; Guan, D.; Shen, G.;Wang, Y.; Yuan, J.; Zhang, L. US8609622B2, 2013.
(68) Chun, K.; Peng, Z.; Qichao, L.; Bo, Z.; Zhen, W.; Guorong, Z.;Yongjun, T. CN 112375087A, 2020.
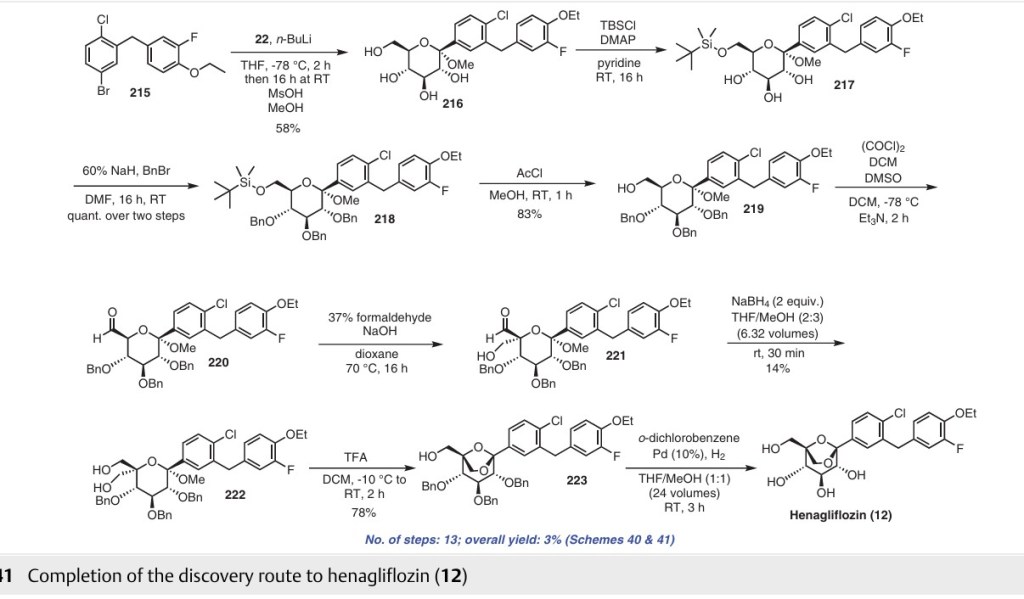
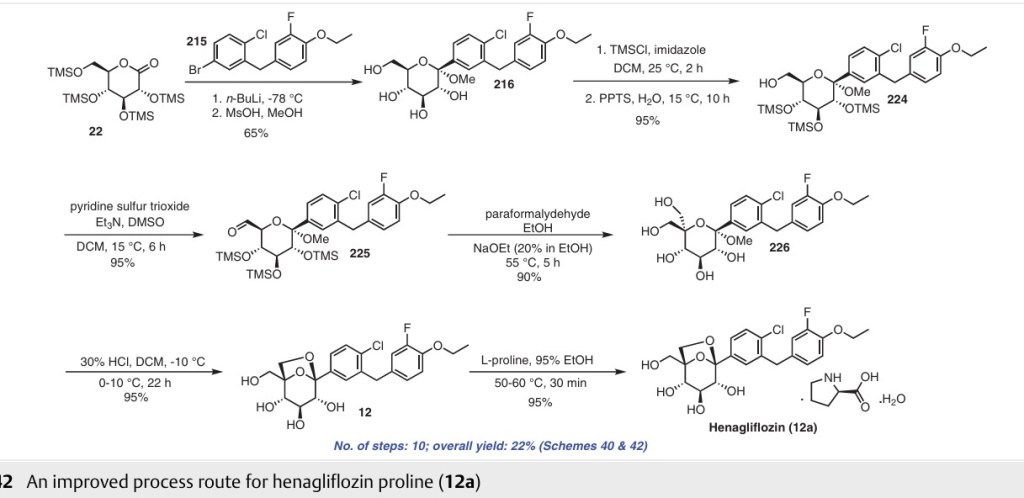
.
TAK-058 (ENV-8058)

TAK-058 , ENV-8058
5-HT 3 receptor antagonist
1-(1-methyl-1H-pyrazol-4-yl)-N-((1R,5S,7S)-9-methyl-3-oxa-9-azabicyclo[3.3.1]nonan-7-yl)-1H-indole-3-carboxamide
l-(l-methyl-lH-pyrazol-4-yl)-N-((lR,5 .7S)-9-methyl-3-oxa-9-azabicyclo[3.3.11nonan-7-yl)-lH-indole-3-carboxamide
1-(1-methyl-1H- pyrazol-4-yl)-N- ((1R,5S,7S)- 9-methyl-3- oxa-9-azabicyclo [3.3.1]nonan-7- yl)-1H-indole-3- carboxamide, 2,2,2- trifluoroacetic acid salt
N-(9-methyl-3-oxa-9-azabicyclo[3.3.1]nonan-7-yl)-1-(1-methylpyrazol-4-yl)indole-3-carboxamide
| Molecular Formula: | C21H25N5O2 |
|---|---|
| Molecular Weight: | 379.4555 g/mol |
https://clinicaltrials.gov/ct2/show/NCT02153099
Phase I Schizophrenia
| Latest Stage of Development | Phase I |
| Standard Indication | Schizophrenia |
| Indication Details | Treat schizophrenia |
- 01 Dec 2015 Phase-I clinical trials in Schizophrenia (Combination therapy) in USA (PO)
- 01 Dec 2015 Takeda completes a phase I trial in Healthy volunteers in USA (NCT02389881)
- 28 Nov 2015 Takeda plans a phase I trial in Schizophrenia (Combination therapy) in USA (NCT02614586)

1 -( 1 -methyl- 1 H-pyrazol-4-yl)-N-((lR,5S,7S)-9-methyl-3-oxa-9-azabicyclo[3.3.1]nonan-7-yl)-lH-indole-3-carboxamide, free base, which is an antagonist of the 5-HT3 receptor. 1 -(1 -Methyl- 1 H-pyrazol-4-yl)-N-((lR,5S,7S)-9-methyl-3-oxa-9-azabicyclo[3.3.1]nonan-7-yl)-lH-indole-3-carboxamide, 2,2,2-trifluoroacetic acid salt, is disclosed in PCT Publication No. WO
2014/014951, published January 23, 2014.
1-(1-methyl-1H-pyrazol-4-yl)-N-((1R,5S,7S)-9-methyl-3-oxa-9-azabicyclo[3.3.1]nonan-7-yl)-1H-indole-3-carboxamide a 5-HT3 receptor antagonist, useful for treating anxiety, depression, eating disorder, schizophrenia, cognitive dysfunction, Parkinson’s disease, Huntington’s Chorea, presenile dementia, Alzheimer’s disease and atherosclerosis.
This compound was originally claimed in WO2014014951, Takeda, following its acquisition of Envoy Therapeutics, is developing TAK-058 (ENV-8058), a 5-HT3 receptor antagonist, as an oral solution for treating schizophrenia, especially cognitive impairment associated with schizophrenia.
In July 2015, the drug was listed as being in phase I development. TAK-058 may have emerged from a schizophrenia therapy program which used Envoy’s bacTRAP translational profiling technology to identify a protein target in the brain.
PATENT
Example 5
Synthesis of l-(l-methyl-lH-pyrazol-4-yl)-N-((lR,5 .7S)-9-methyl-3-oxa-9-azabicyclo[3.3.11nonan-7-yl)-lH-indole-3-carboxamide. 2.2.2-trifluoroacetic acid salt

Step 1 : methyl 1-(1 -methyl- lH-pyrazol-4-yl)-lH-indole-3-carboxylate. TFA
To a sealed tube was added copper(I) iodide (65.2 mg, 0.342 mmol), methyl 1H-indole-3-carboxylate (200 mg, 1.142 mmol) and potassium phosphate (509 mg, 2.397 mmol), then the reaction vessel was evacuated and purged with nitrogen (3x). Next, 4-bromo-l-methyl-lH-pyrazole (184 mg, 1.142 mmol) and (lR,2R)- ,N2-dimethylcyclohexane-l,2-diamine (109 μΐ, 0.685 mmol) were added, followed by toluene (1 142 μΐ). The reaction tube was evacuated and purged with nitrogen, then sealed and heated at 1 10 °C for 24 h. HPLC purification provided the title compound as a colorless oil.
Step 2: 1-(1 -methyl- lH-pyrazol-4-yl)-lH-indole-3-carboxylic acid hydrochloride
To a solution of methyl 1-(1 -methyl- lH-pyrazol-4-yl)-lH-indole-3-carboxylate, TFA
(3.5 mg, 9.48 μιηοΐ) in MeOH (95 μΐ) was added a solution of aq. KOH (33.2 μΐ, 0.066 mmol, 2 M). The reaction mixture was stirred at RT overnight, then acidified with IN HC1.
The solvent was evaporated under reduced pressure and the residue was dried under vacuum overnight. The title compound was used without further purification.
Step 3 : l-(l-methyl-lH-pyrazol-4-yl)-N-((lR,5 .7S)-9-methyl-3-oxa-9-azabicyclor3.3.11nonan-7-yl)-lH-indole-3-carboxamide, 2,2,2-trifluoroacetic acid salt
To a mixture of 1-(1 -methyl- lH-pyrazol-4-yl)-lH-indole-3-carboxylic acid hydrochloride (2.6 mg, 9.36 μιηοΐ) in DMF (187 μΐ) was added HATU (4.27 mg, 0.01 1 mmol) and DIPEA (8.18 μΐ, 0.047 mmol). After the reaction mixture was stirred at RT for 15 min, (lR,5S,7S)-9-methyl-3-oxa-9-azabicyclo[3.3.1]nonan-7-amine, TFA (3.04 mg, 0.01 1 mmol) was added and stirring was continued for 2 h. HPLC purification afforded the title compound as a white solid. MS (ESI, pos. ion) m/z: 380.30 (M+l).
PATENT
EXAMPLE 1 : l-(l-methyl-lH-pyrazol-4-yl)-N-((lR,5S,7S)-9-methyl-3-oxa-9-azabicyclo[3.3.1 ]nonan-7-yl)- lH-indole-3-carboxamide
l-(l-Methyl-lH-pyrazol-4-yl)-lH-indole-3-carboxylic acid (128.7 g, 0.53 mol,) and anhydrous THF (645 mL) was heated to about 43°C. Oxalyl chloride (137.7 g, 92 mL, 1.08 mol) was added dropwise between 40 and 50°C. Gas evolution ceased in approximately 30 minutes. The resulting suspension was stirred for 2 hours at 50°C, allowed to cool to room temperature, and then stirred overnight. The suspension was diluted with heptane (1.5 L), stirred for 10 minutes, and allowed to settle. The supernatant was removed. The addition of heptane (1.5 L), followed by stirring, settling, and decanting was repeated two more times.
The resulting suspension was diluted with anhydrous THF (645 mL) and the ratio between THF and heptane was determined by NMR to be 3:2. The reaction mixture was cooled to 5°C and to the mixture was added DIPEA base (138 g, 1.07 mol) at such a rate that the temperature did not exceed 20°C. Next (li?,55*,7S)-9-methyl-3-oxa-9-azabicyclo[3.3.1]nonan-7-amine (101.4 g, 0.63 mol) in 500 mL of anhydrous THF was added. The reaction mixture was warmed to ambient temperature and stirred at 20 to 23°C overnight to give a suspension.
The suspension was filtered and the cake was dissolved in IN HC1 (2.6 L). The aqueous layer was washed with EtOAc (3 x 2.6 L). The aqueous layer was cooled to 5°C and was basified to pH 12 with aqueous potassium hydroxide (230 g) solution in water (500 mL). The mixture was stirred at 5 to 10°C overnight to give a solid. The product was filtered, washed with water (2 x 1.2 L), followed by MTBE (2 x 1.2 L), and then dried to give 128 g (64%) of the (crude) title compound.
Patent
https://www.google.co.in/patents/US20140024644
1-(1-methyl-1H- pyrazol-4-yl)-N- ((1R,5S,7S)- 9-methyl-3- oxa-9-azabicyclo [3.3.1]nonan-7- yl)-1H-indole-3- carboxamide, 2,2,2- trifluoroacetic acid salt
Synthetic Procedures Reference 1 Synthesis of (1R,5S,7S)-tert-butyl 7-hydroxy-3-oxa-9-azabicyclo[3.3.1]nonane-9-carboxylate
-
Sodium borohydride (259 mg, 6.84 mmol) was added portion-wise to a solution of (1R,5S)-tert-butyl 7-oxo-3-oxa-9-azabicyclo[3.3.1]nonane-9-carboxylate (550 mg, 2.279 mmol) in MeOH (4559 μl) at 0° C. After 5 min, the reaction mixture was allowed to warm to RT then stirred for 30 min. The mixture was concentrated under reduced pressure, dissolved in EtOAc and washed with brine. The combined organic layers were dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure to afford the title compound as a white solid, which was used without further purification.
Example 4 Synthesis of N-((1R,5S,7S)-9-methyl-3-oxa-9-azabicyclo[3.3.1]nonan-7-yl)-1-(1H-pyrazol-4-yl)-1H-indole-3-carboxamide, 2,2,2-trifluoroacetic acid salt
-
A mixture of 1-((1-benzyl-1H-pyrazol-4-yl)-N-((1R,5S,7S)-9-methyl-3-oxa-9-azabicyclo[3.3.1]nonan-7-yl)-1H-indole-3-carboxamide 2,2,2-trifluoroacetate (85 mg, 0.149 mmol) and 10% Pd—C (120 mg) in MeOH (1.0 ml) was stirred at RT under H2 for 2 days. Filtration and concentration afforded the title compound as a white solid. MS (ESI, pos. ion) m/z: 366.20 (M+1).
Example 5 Synthesis of 1-(1-methyl-1H-pyrazol-4-yl)-N-((1R,5S,7S)-9-methyl-3-oxa-9-azabicyclo[3.3.1]nonan-7-yl)-1H-indole-3-carboxamide, 2,2,2-trifluoroacetic acid salt
Step 1: methyl 1-(1-methyl-1H-pyrazol-4-yl)-1H-indole-3-carboxylate, TFA
-
To a sealed tube was added copper(I) iodide (65.2 mg, 0.342 mmol), methyl 1H-indole-3-carboxylate (200 mg, 1.142 mmol) and potassium phosphate (509 mg, 2.397 mmol), then the reaction vessel was evacuated and purged with nitrogen (3×). Next, 4-bromo-1-methyl-1H-pyrazole (184 mg, 1.142 mmol) and (1R,2R)—N1,N2-dimethylcyclohexane-1,2-diamine (109 μl, 0.685 mmol) were added, followed by toluene (1142 μl). The reaction tube was evacuated and purged with nitrogen, then sealed and heated at 110° C. for 24 h. HPLC purification provided the title compound as a colorless oil.
Step 2: 1-(1-methyl-1H-pyrazol-4-yl)-1H-indole-3-carboxylic acid hydrochloride
-
To a solution of methyl 1-(1-methyl-1H-pyrazol-4-yl)-1H-indole-3-carboxylate, TFA (3.5 mg, 9.48 μmol) in MeOH (95 μl) was added a solution of aq. KOH (33.2 μl, 0.066 mmol, 2 M). The reaction mixture was stirred at RT overnight, then acidified with 1N HCl. The solvent was evaporated under reduced pressure and the residue was dried under vacuum overnight. The title compound was used without further purification.
Step 3: 1-(1-methyl-1H-pyrazol-4-yl)-N-((1R,5S,7S)-9-methyl-3-oxa-9-azabicyclo[3.3.1]nonan-7-yl)-1H-indole-3-carboxamide, 2,2,2-trifluoroacetic acid salt
-
To a mixture of 1-(1-methyl-1H-pyrazol-4-yl)-1H-indole-3-carboxylic acid hydrochloride (2.6 mg, 9.36 μmol) in DMF (187 μl) was added HATU (4.27 mg, 0.011 mmol) and DIPEA (8.18 μl, 0.047 mmol). After the reaction mixture was stirred at RT for 15 min, (1R,5S,7S)-9-methyl-3-oxa-9-azabicyclo[3.3.1]nonan-7-amine, TFA (3.04 mg, 0.011 mmol) was added and stirring was continued for 2 h. HPLC purification afforded the title compound as a white solid. MS (ESI, pos. ion) m/z: 380.30 (M+1).
| 15 | TFA |
| 379.456 MW | 380.30 MS +1
|
/////////TAK-058 , ENV-8058, phase I, takeda, 5-HT 3 receptor antagonist, Envoy Therapeutics, Inc., Phase I, Schizophrenia
C12CC(CC(N1C)COC2)NC(c4c3ccccc3n(c4)c5cnn(c5)C)=O
CN1C=C(C=N1)N2C=C(C3=CC=CC=C32)C(=O)NC4CC5COCC(C4)N5C
GSK-525762A
GSK 525762A; 1260907-17-2; I-BET-762; GSK525762A; UNII-5QIO6SRZ2R; 5QIO6SRZ2R;
CAS1260907-17-2
2-[(4S)-6-(4-chlorophenyl)-8-methoxy-1-methyl-4H-[1,2,4]triazolo[4,3-a][1,4]benzodiazepin-4-yl]-N-ethylacetamide
| Molecular Formula: | C22H22ClN5O2 |
|---|---|
| Molecular Weight: | 423.89538 g/mol |
| Solubility: | Soluble in DMSO (84 mg/ml at 25 °C), ethanol (42 mg/ml at 25 °C, warmed), DMF (~30 mg/ml), ethanol:PBS (pH 7.2, 1:1) (~0.5 mg/ml), and water (<1 mg/ml at 25 °C). |
| Storage: | Store at -20° C |
| Density: | ~1.4 g/cm3 (Predicted) |
| Refractive Index: | n20D 1.67 (Predicted) |
| Optical Activity: | α20D 85º±5º, c = 0.3 in methanol |
| IC50: | BRD2: IC50 = 32.5 nM (human); BRD4: IC50 = 36.1 nM (human); BRD3: IC50 = 42.4 nM (human); PBMC: IC50 = 316.23 nM (human); HepG2: EC5050 = 700 nM (human) |
| pK Values: | pKb: 2.43 (Predicted) |
In April 2016, GSK-525762 was reported to be in phase 2 clinical development. GSK-525762 was originally disclosed in WO2011054553, claiming benzodiazepine derivatives as bromodomain inhibitors, useful for treating cancer. See WO2014028547, claiming use of GSK-525762 for treating small cell lung cancer.
GSK 525762A, is a BET Bromodomain Inhibitor, which is now in clinical development. BET bromodomains have emerged as promising drug targets for treatment of cancers, inflammatory diseases, and other medical conditions.
Patent
Patent applications WO201 1/054553 and WO201 1/054845 (both in the name of GlaxoSmithKline LLC) disclose the compound 2-[(4S)-6-(4-chlorophenyl)-1-methyl-8-(methyloxy)-4/-/-[1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]-/V-ethylacetamide as a BET family bromodomain inhibitor and describes therapeutic uses thereof. The chemical structure of this compound is represented by formula (I):

(I)
Scheme 1



Example 1
Preparation of an acetonitrile solvate of 2-[(4S)-6-(4-chlorophenyl)-1 -methyl-8-(methyloxy)-4H-[1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]-yV-ethylacetamide
Amorphous 2-[(4S)-6-(4-chlorophenyl)-1-methyl-8-(methyloxy)-4H-[1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]-/V-ethylacetamide (prepared for instance as described in WO201 1/054553, 1 wt) was dissolved in acetonitrile (20 vol) upon heating (up to reflux). The solution was then distilled to 10 vol keeping the temp 50 °C – 60 °C by adjusting the vacuum. Nucleation occurred during the final stage of the distillation. The slurry was then held at 60 °C before being cooled to 20 °C and filtered. The cake was then washed with
acetonitrile (2 vol). The cake was dried under vacuum with a nitrogen bleed at approximately 60 °C to provide the titled product.
1H-NMR (500 MHz, DMSO-d6, referenced to TMS = 0.00 ppm, T = 25 C) δ ppm 8.22 (1 H, t, J = 5 Hz), 7.79 (1 H, d, J = 9 Hz), 7.53 (2H, d, J = 9 Hz), 7.49 (2H, d, J = 9 Hz), 7.38 (1 H, dd, J = 3 Hz, 9 Hz), 6.87 (1 H, d, J = 3 Hz), 4.49 (1 H, m), 3.79 (3H, s), 3.25 (1 H, m), 3.20-3.06 (3H, several m), 2.54 (3H, s), 2.08 (3H, s), 1 .07 (3H, t, J = 7 Hz).
Example 2
Preparation of a benzene sulphonic acid salt of 2-[(4S)-6-(4-chlorophenyl)-1 -methyl-8-(methyloxy)-4H-[1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]-A/-ethylacetamide in crystalline solid state form
Preparation 1
The acetonitrile solvate of 2-[(4S)-6-(4-chlorophenyl)-1-methyl-8-(methyloxy)-4/-/-[1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]-/V-ethylacetamide (for a preparation see Example 1 , 2.58 g) was slurried in acetonitrile (7 mL) and 2-methyltetrahydrofuran (7 mL). Benzenesulfonic acid (1.17 g) was dissolved in acetonitrile (7 mL). The resulting solution was charged to the slurry of 2-[(4S)-6-(4-chlorophenyl)-1-methyl-8-(methyloxy)-4/-/-[1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]-/V-ethylacetamide acetonitrile solvate in acetonitrile and 2-methyltetrahydrofuran. An additional rinse of acetonitrile (1.4 mL) and 2-methyltetrahydrufran (0.7 mL) was added to the slurry. The slurry was then warmed to 60 °C to dissolve. 2-methyltetrahydrofuran (50 mL) was then added over 30 minutes. Crystals formed during this addition. The resulting suspension was then cooled to 5 °C at a controlled, linear rate of 0.5 °C/minute. The slurry was aged for 1 hour. The crystalline product was then isolated by filtration and rinsed with a 5 to 1 mixture of 2-methyltetrahydrofuran and acetonitrile (15 mL). The product was then dried in a vacuum oven at 55 °C overnight.
Preparation 2
The acetonitrile solvate of 2-[(4S)-6-(4-chlorophenyl)-1 -methyl-8-(methyloxy)-4/-/-[1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]-/V-ethylacetamide (prepared for example in a process such as Example 1 above, 1 wt) was dissolved in 9 vol 2-methyltetrahydrofuran at 65 °C. Once cooled to 20°C the solution was filtered into the crystallization vessel. The dissolution vessel and inline filter were rinsed with 1 vol 2-methyltetrahydrofuran. The solution was then heated to 45 °C.
1 .05 eq of benzene sulphonic acid was dissolved in 1 volume of filtered acetonitrile. 10% of this solution was added to a reactor to which 0.05 wt% of a benzene sulphonic acid
salt of 2-[(4S)-6-(4-chlorophenyl)-1-methyl-8-(methyloxy)-4H-[1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]-/V-ethylacetamide micronized seed (prepared for example as in Preparation 1 above) slurry was charged. The remaining benzene sulphonic acid solution was charged at a steady rate over 2 hours, maintaining the reactor at 45 °C.
The slurry was cooled to 0 °C at no greater than 0.2 °C/minute. The slurry was filtered.
The crystallizer was charged with the first wash, 3 vol of filtered 2-methyltetrahydrofuran, which was cooled to <10 °C while stirring in the crystallizer, before being used to wash the cake. The crystallizer was charged with the second wash, 3 vol of filtered 2-methyltetrahydrofuran, which was cooled to <10 °C while stirring in the crystallizer, before being used to wash the cake. The crystallizer was charged with the third wash, 4 vol of filtered 2-MeTHF, which was cooled to <10 °C while stirring in the crystallizer, before being used to wash the cake. The cake was blown-down until the solvent being removed was reduced to a trickle. The title compound was then dried in a vacuum oven at 50 °C until the loss on drying (LOD) indicates <0.2% wt. loss (LOD method: 10 min at 120 °C). The product was then delumped using a comil.
Example 3
Characterisation of a benzene sulphonic acid salt of 2-[(4S)-6-(4-chlorophenyl)-1 -methyl-8-(methyloxy)-4H-[1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]-yV-ethyl acetamide in crystalline solid state form
XRPD
The X-ray powder diffraction (XRPD) data were acquired on a PANalytical X’Pert Pro powder diffractometer, model PW3050/60, using an X’Celerator detector. The acquisition conditions were: radiation: Cu Ka, generator tension: 45 kV, generator current: 40 mA, step size: 0.017 °2Θ, time per step: 500 seconds, divergence slit type: fixed, divergence slit size: 0.4354 °, measurement temperature: 20-25 °C, goniometer radius: 240 mm. The sample was prepared by packing sample in a 0.9 mm capillary. Peak positions were obtained using PANalytical X’Pert Highscore Plus software. The margin of error is approximately ± 0.1° 2Θ for each of the peak assignments.
The X-ray powder diffraction (XRPD) pattern is shown in Figure 1 and shows characteristic peaks, expressed in degrees 2Θ, at 5.5, 7.4, 9.1 , 10.0, 10.4, 13.3, 13.6, 14.9, 18.7, 20.4, 20.9, 22.8 and 23.1 ° ( ± 0.1 °).
13C Solid State NMR (SSNMR)
A 13C SSNMR spectrum was obtained at 273K on a spectrometer operating at a frequency of 100.56 MHz for 13C observation using a cross-polarization pulse sequence with a Bruker 4-mm triple resonance magic-angle spinning probe at a rotor frequency of 8 kHz. The margin of error is ± 0.2 ppm for each of the peak assignments.
The 13C SSNMR spectrum is shown in Figure 2 and comprises chemical shifts (ppm) at 169.6, 167.5 165.6, 160.1 , 159.4, 157.1 , 155.9, 154.3, 152.4, 146.9, 145.8, 140.0, 137.9, 135.9, 133,4, 132.0, 130.6, 129.9, 128.3, 127.1 , 125.6, 123.5, 120.6, 1 19.1 , 1 14.1 , 1 13.7, 58.0, 53.6, 53.1 , 40.7, 37.0, 34.9, 15.8, 14.7, and 12.0 ( ±0.2 ppm).
PATENT
WO2011054553
http://www.google.com/patents/WO2011054553A1?cl=en
formula (I) which is 2-[(4S)-6-(4- Chlorophenyl)-1-methyl-8-(methyloxy)-4H-[1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]-/V- ethylacetamide
(I)
or a salt thereof.
It will be appreciated that the present invention covers compounds of formula (I) as the free base and as salts thereof, for example as a pharmaceutically acceptable salt thereof.
In one embodiment there is provided a compound which is 2-[(4S)-6-(4-Chlorophenyl)-1- methyl-8-(methyloxy)-4H-[1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]-/V-ethylacetamide.
Because of their potential use in medicine, salts of the compounds of formula (I) are desirably pharmaceutically acceptable. In another embodiment there is provided a compound which is 2-[(4S)-6-(4-Chlorophenyl)-1-methyl-8-(methyloxy)-4H- [1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]-/V-ethylacetamide or a pharmaceutically acceptable salt thereof.
The compound of formula (I) may be prepared according to reaction scheme 1 by reaction of a compound of formula (II) with EtNH2 in the presence of HATU or HBTU and DIEA at room temperature. Alternatively compounds of formula (I) may be prepared by reacting the compound of formula (II) with oxalyl chloride followed by addition of EtNH2 in the presence of triethylamine.
Scheme 1
The compound of formula (II) may be prepared according to reaction Scheme 2. Suitable reaction conditions comprise reacting a compound of formula (III) with alkaline hydroxide preferably sodium hydroxide or lithium hydroxide.
Scheme 2
wherein R represents C-|.galkyl such as methyl.
Compounds of formula (III), may be prepared according to reaction scheme 3 by reacting compounds of formula (IV) with AcOH. Scheme 3
Compounds of formula (IV) may be prepared according to reaction scheme 4 by reacting compounds of formula (VI) with hydrazine below 15 °C followed by reaction of the resulting hydrazone (V) with MeCOCI at 0°C. Generally hydrazone (V) is used without further purification and is reacted with MeCOCI at , for example 0 °C.
Scheme 4
(IV) Compounds of formula (VI) in which R is Ci-6alkyl (such as methyl) may be prepared according to reaction scheme 5 from compounds of formula (VII) by treatment with Lawesson’s reagent or P4Si0. Suitable reaction conditions comprise reacting compounds of formula (VIII) with P4Si0 in 1 ,2-dichloroethane at, for example 70 °C.
Scheme 5
Compounds of formula (VII) may be prepared according to reaction scheme 6, by reacting compounds of formula (IX) with an organic base such as triethylamine followed by reaction of the resulting amine (VIII) with acetic acid. Generally, amine (VIII) is used without further purification and is reacted with AcOH at, for example 60 °C.
Scheme 6
Compounds of formula (IX) may be prepared according to reaction scheme 7, by reacting compounds of formula (XI) with the acylchloride (X) derived from protected aspartic acid. Scheme 7
Compounds of formula (XI) may be prepared according to procedures described in Synthesis 1980, 677-688. Acyl chlorides of formula (X) may be prepared according to procedures described in J. Org. Chem., 1990, 55, 3068-3074 and J. Chem. Soc. Perkin Trans. 1 , 2001 , 1673-1695.
Alternatively the compound of formula (I) may be prepared according to reaction scheme 8.
wherein R represents C-|_4alkyl such as methyl.
The compound of formula (IIIA) may be prepared according to reaction scheme 9 by reacting compounds of formula (IVA) with EtNH2 in the presence of HATU and DIEA at, for example room temperature.
Scheme 9
The compound of formula (IVA) may be prepared according to reaction scheme 10. Suitable reaction conditions comprise reacting compounds of formula (VI) with alkaline hydroxide such as sodium hydroxide. Scheme 10
Example 1 : 2-[(4S)-6-(4-Chlorophenyl)-1 -methyl-8-(methyloxy)-4H-[1 ,2,4]triazolo[4,3-
To a solution of [(4S)-6-(4-Chlorophenyl)-1-methyl-8-(methyloxy)-4H-[1 !2!4]triazolo[4,3- a][1 ,4]benzodiazepin-4-yl]acetic acid (for a preparation see Intermediate 1 )(16.0 g, 40 mmol) in THF at RT was added DIEA (14 mL, 80 mmol) followed by HATU (30.4 g, 80 mmol). The reaction mixture was stirred for 3h at this temperature and ethylamine (40 mL, 2M in THF, 80 mmol) was added. The mixture was stirred for 48h before being concentrated under reduced pressure. The crude material was suspended in water and extracted with DCM. The organic layer was dried over Na2S04, filtered and concentrated in vacuo. The crude solid was purified by chromatography on Si02 (DCM/MeOH 95/5) and the resulting solid recrystallised in MeCN. The solid was then dissolved in DCM and precipited with /‘-Pr20 to give the title compound (8 g, 47% yield) as a white solid.
Rf = 0.48 (DCM/MeOH : 90/10). Mp >140 °C (becomes gummy). 1H NMR (300 MHz, CDCI3) 7.53-7.47 (m, 2H), 7.39 (d, J = 8.9 Hz, 1 H), 7.37-7.31 (m, 2H), 7.20 (dd, J = 2.9 and 8.9 Hz, 1 H), 6.86 (d, J = 2.9 Hz, 1 H), 6.40 (m, 1 H), 4.62 (m, 1 H), 3.80 (s, 3H), 3.51 (dd, J = 7.3 and 14.1 Hz, 1 H), 3.46-3.21 (m, 3H), 2.62 (s, 3H), 1.19 (t, J = 7.3 Hz, 3H). LC/MS : m/z 424 [M(35CI)+H]+, Rt 2.33 min.
Intermediate 1 : [(4S)-6-(4-Chlorophenyl)-1 -methyl-8-(methyloxy)-4H-
[1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]acetic acid
To a solution of methyl [(4S)-6-(4-chlorophenyl)-1 -methyl-8-(methyloxy)-4H- [1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]acetate (for a preparation see Intermediate 2)(28 g, 68 mmol) in THF (450 mL) at RT was added 1 N NaOH (136 mL, 136 mmol). The reaction mixture was stirred at this temperature for 5h before being cooled down and quenched with 1 N HCI (136 mL). THF was removed under reduced pressure and the aqueous layer was extracted with DCM. The combined organic layers were dried over Na2S04, filtered and concentrated under reduced pressure. The crude solid was recrystallised in CH3CN to give the title compound (23.9 g, 89% yield) as a pale yellow powder. 1H NMR (300 MHz, CDCI3) δ 7.55-7.48 (m, 2H), 7.41 (d, J = 8.9 Hz, 1 H), 7.38- 7.31 (m, 2H), 7.22 (dd, J = 2.9 and 8.9 Hz, 1 H), 6.90 (d, J = 2.9 Hz, 1 H), 4.59 (dd, J = 6.9 and 6.9 Hz, 1 H), 3.81 (s, 3H), 3.70 (dd, J = 6.9 and 25.7 Hz, 1 H), 3.61 (dd, J = 6.9 and 25.7 Hz, 1 H), 2.63 (s, 3H). LC/MS: m/z 397 [M(35CI)+H]+, Rt 2.1 1 min.
Intermediate 2: Methyl [(4S)-6-(4-chlorophenyl)-1 -methyl-8-(methyloxy)-4H- [1 ,2,4]triazolo[4,3-a][1 ,4]benz
To crude methyl [(3S)-2-[(1 Z)-2-acetylhydrazino]-5-(4-chlorophenyl)-7-(methyloxy)-3H- 1 ,4-benzodiazepin-3-yl]acetate (for a preparation see Intermediate 3) (34 g, 79 mmol) was suspended in THF (200 mL) and AcOH (200 mL) was added at RT. The reaction mixture was stirred at this temperature overnight before being concentrated to dryness. The residue was suspended in saturated NaHC03 and extracted with DCM. The organic layer was dried over Na2S04, filtered and concentrated in vacuo. The crude solid was purified by chromatography on Si02 (DCM/MeOH : 90/10) to give the title compound (28 g, 86% yield) as a yellow powder.
1H NMR (300 MHz, CDCI3) δ 7.54-7.47 (m, 2H), 7.40 (d, J = 8.8 Hz, 1 H), 7.37-7.31 (m, 2H), 7.22 (dd, J = 2.8 and 8.8 Hz, 1 H), 6.89 (d, J = 2.8 Hz, 1 H), 4.61 (dd, J = 6.4 and 7.8 Hz, 1 H), 3.82 (s, 3H), 3.78 (s, 3H), 3.66 (dd, J = 7.8 and 16.9 Hz, 1 H), 3.60 (dd, J = 6.4 and 16.9 Hz, 1 H), 2.62 (s, 3H). LC/MS m/z 41 1 [M(35CI)+H]+, Rt 2.88 min. Intermediate 3: Methyl [(3S)-2-[2-acetylhydrazino]-5-(4-chlorophenyl)-7-(methyloxy)- 3H-1 ,4-benzodiazepin-3-yl]acetate
To a suspension of methyl [(3S)-5-(4-chlorophenyl)-7-(methyloxy)-2-thioxo-2,3-dihydro- 1 H-1 ,4-benzodiazepin-3-yl]acetate (for a preparation see Intermediate 4)(30.2 g, 77.7 mmol) in THF (800 mL) at 0°C was added hydrazine monohydrate (1 1 .3 ml_, 233 mmol) dropwise. The reaction mixture was stirred for 4h between 0°C and 15°C before being cooled at 0°C. Et3N (32.4 mL, 230 mmol) was then added slowly and AcCI (16.3 mL, 230 mmol) was added dropwise. The mixture was allowed to warm to RT and stir for 1 h then quenched with water and concentrated under reduced pressure. The resulting aqueous layer was then extracted with DCM and the organic layer was dried over Na2S04, filtered and concentrated in vacuo to give the crude title compound (34 g, 100% yield) which was used without further purification. LC/MS: m/z 429 [M(35CI)+H]+, Rt 2.83 min. Intermediate 4: Methyl [(3S)-5-(4-chlorophenyl)-7-(methyloxy)-2-thioxo-2,3-dihydro- 1H-1 ,4-benzodiazepin-3-yl]acetate
A suspension of P4Si0 (85.8 g, 190 mmol) and Na2C03 (20.5 g, 190 mmol) in 1 ,2-DCE (1.5 L) at RT was stirred for 1 h before methyl [(3S)-5-(4-chlorophenyl)-7-(methyloxy)-2- oxo-2,3-dihydro-1 H-1 ,4-benzodiazepin-3-yl]acetate (for a preparation see Intermediate 5) (40 g, 107 mmol) was added. The resulting mixture was stirred at 65°C for 4 h before being cooled and filtered. The solid was washed with DCM and the filtrate washed with sat. NaHC03. The organic layer was dried over Na2S04, filtered and concentrated under reduced pressure. The title compound was precipitated from a DCM//‘-Pr20 mixture and filtered. The filtrate was then concentrated and purified by flash chromatography (DCM/MeOH : 98/2) to afford another batch of product. The title compound was obtained combining the two fractions (30.2 g, 73%) as a yellow powder. LC/MS: m/z 389
[M(35CI)+H]+, Rt 3.29 min.
Intermediate 5: Methyl [(3S)-5-(4-chlorophenyl)-7-(methyloxy)-2-oxo-2,3-dihydro-1H- 1 ,4-benzodiazepin-3-yl]acetat
To a solution of the crude methyl /V1-[2-[(4-chlorophenyl)carbonyl]-4-(methyloxy)phenyl]- /V2-{[(9H-fluoren-9-ylmethyl)oxy]carbonyl}-L-a-asparaginate (for a preparation see Intermediate 6) (assumed 0.2 mol) in DCM (500 mL) was added Et3N (500 mL, 3.65 mol) and the resulting mixture was refluxed for 24h before being concentrated. The resulting crude amine was dissolved in 1 ,2-DCE (1.5 L) and AcOH (104 mL, 1.8 mol) was added carefully. The reaction mixture was then stirred at 60°C for 2h before being concentrated in vacuo and dissolved in DCM. The organic layer was washed with 1 N HCI and the aqueous layer was extracted with DCM (x3). The combined organic layers were washed twice with water, and brine, dried over Na2S04, filtered and concentrated under reduced pressure. The crude solid was recrystallised in MeCN leading to the title compound (51 g) as a pale yellow solid. The filtrate could be concentrated and recrystallised in MeCN to give another 10 g of Intermediate 9 (total: 61 g, 69% yield based on recovered
Intermediate 12). Rf = 0.34 (DCM/MeOH : 95/5). LC/MS m/z 373 [M(35CI)+H]+, Rt 2.76 min.
Intermediate 6: Methyl W^2-[(4 :hlorophenyl)carbonyl]-4-(methyloxy)phenyl] V2– {[(9H-fluoren-9-ylmethyl)oxy]carbonyl}-L-a-asparaginate
A mixture of Methyl /V-{[(9H-fluoren-9-ylmethyl)oxy]carbonyl}-L-a-aspartyl chloride (prepared from J. Org. C em. 1990, 55, 3068-3074 and J. C em. Soc. Perkin Trans. 1 2001 , 1673-1695) (221 g, 0.57 mol) and [2-amino-5-(methyloxy)phenyl](4- chlorophenyl)methanone (for a preparation see Intermediate 7) (133 g, 0.5 mol) in CHCI3 (410 mL) was stirred at 60°C for 1.5h before being cooled and concentrated under reduced pressure and used without further purification. LC/MS: m/z 613 [M(35CI)+H]+, Rt = 3.89 min. Intermediate 7: [2-amino-5-(methyloxy)phenyl](4-chlorophenyl)methanone
To a solution of 2-methyl-6-(methyloxy)-4H-3,1-benzoxazin-4-one (for a preparation see Intermediate 8)(40.0 g, 0.21 mol) in a toluene (560 ml_)/ether (200 mL) mixture at 0°C was added dropwise a solution of 4-chlorophenylmagnesium bromide (170 mL, 1 M in Et20, 0.17 mol). The reaction mixture was allowed to warm to RT and stirred for 1 h before being quenched with 1 N HCI. The aqueous layer was extracted with EtOAc (3 x) and the combined organics were washed with brine, dried over Na2S04, filtered and concentrated under reduced pressure. The crude compound was then dissolved in EtOH (400 mL) and 6N HCI (160 mL) was added. The reaction mixture was refluxed for 2 h before being concentrated under reduced pressure. The resulting solid was filtered and washed twice with ether before being suspended in EtOAc and neutralised with 1 N NaOH. The aqueous layer was extracted with EtOAc (3 x) and the combined organics were washed with brine, dried over Na2S04, filtered and concentrated under reduced pressure. The title compound was obtained as a yellow solid (39 g, 88 % yield) which was used without further purification. Intermediate 8 : 2-methyl-6-(methyloxy)-4H-3,1 -benzoxazin-4-one
A solution of 5-methoxyanthranilic acid (7.8 g, 46.5 mmol) was refluxed in acetic anhydride (60 mL) for 2h15 before being cooled and concentrated under reduced pressure. The crude residue was then concentrated twice in the presence of toluene before being filtered and washed with ether to yield to the title compound (6.8 g, 77% yield) as a beige solid; LC/MS: m/z 192 [M+H]+, Rt 1.69 min.
Preparation of reference compound for use in biological assays
Experimental details of LC-MS methods A and B as referred to herein are as follows:
LC/MS (Method A) was conducted on a Supelcosil LCABZ+PLUS column (3μηΊ, 3.3cm x 4.6mm ID) eluting with 0.1 % HCO2H and 0.01 M ammonium acetate in water (solvent A), and 95% acetonitrile and 0.05% HCO2H in water (solvent B), using the following elution gradient 0-0.7 minutes 0%B, 0.7-4.2 minutes 0→100%B, 4.2-5.3 minutes 100%B, 5.3-5.5 minutes 100→0%B at a flow rate of 3 mL/minute. The mass spectra (MS) were recorded on a Fisons VG Platform mass spectrometer using electrospray positive ionisation [(ES+ve to give [M+H]+ and [M+NH4]+ molecular ions] or electrospray negative ionisation
[(ES-ve to give [M-H]- molecular ion] modes. Analytical data from this apparatus are given with the following format : [M+H]+ or [M-H]-.
LC/MS (Method B) was conducted on an Sunfire C18 column (30mm x 4.6mm i.d. 3.5μηι packing diameter) at 30 degrees centigrade, eluting with 0.1 % v/v solution of Trifluoroacetic Acid in Water (Solvent A) and 0.1 % v/v solution of Trifluoroacetic Acid in Acetonitrile (Solvent B) using the following elution gradient 0-0.1 min 3%B, 0.1- 4.2min 3 – 100% B, 4.2-4.8min 100% B, 4.8-4.9min 100-3%B, 4.9 – 5.0min 3% B at a flow rate of 3ml/min. The UV detection was an averaged signal from wavelength of 210nm to 350nm and mass spectra were recorded on a mass spectrometer using positive electrospray ionization. Ionisation data was rounded to the nearest integer. LC/HRMS: Analytical HPLC was conducted on a Uptisphere-hsc column (3μηι 33 x 3 mm id) eluting with 0.01 M ammonium acetate in water (solvent A) and 100% acetonitrile (solvent B), using the following elution gradient 0-0.5 minutes 5% B, 0.5-3.75 minutes 5→100% B, 3.75-4.5 100% B, 4.5-5 100→5% B, 5-5.5 5% B at a flow rate of 1 .3 mL/minute. The mass spectra (MS) were recorded on a micromass LCT mass spectrometer using electrospray positive ionisation [ES+ve to give MH+ molecular ions] or electrospray negative ionisation [ES-ve to give (M-H)- molecular ions] modes.
TLC (thin layer chromatography) refers to the use of TLC plates sold by Merck coated with silica gel 60 F254.
Silica chromatography techniques include either automated (Flashmaster or Biotage SP4) techniques or manual chromatography on pre-packed cartridges (SPE) or manually- packed flash columns.
Reference compound A : 2-meth -6-(methyloxy)-4H-3,1 -benzoxazin-4-one
A solution of 5-methoxyanthranilic acid (Lancaster) (41.8 g, 0.25 mol) was refluxed in acetic anhydride (230 mL) for 3.5 h before being concentrated under reduced pressure. The crude compound was then concentrated twice in the presence of toluene before being filtered and washed twice with ether to yield to the title compound (33.7 g, 71 % yield) as a brown solid; LC/MS (Method A): m/z 192 [M+H]+, Rt 1.69 min.
Reference compound B: [2-amino- -(methyloxy)phenyl](4-chlorophenyl)methanone
To a solution of 2-methyl-6-(methyloxy)-4H-3,1-benzoxazin-4-one (for a preparation see Reference compound A) (40.0 g, 0.21 mol) in a toluene/ether (2/1 ) mixture (760 mL) at 0°C was added dropwise a solution of 4-chlorophenylmagnesium bromide (170 mL, 1 M in Et20, 0.17 mol). The reaction mixture was allowed to warm to room temperature and stirred for 1 h before being quenched with 1 N HCI (200 mL). The aqueous layer was extracted with EtOAc (3 x 150 mL) and the combined organics were washed with brine (100 mL), dried over Na2S04, filtered and concentrated under reduced pressure. The crude compound was then dissolved in EtOH (400 mL) and 6N HCI (160 mL) was added. The reaction mixture was refluxed for 2 h before being concentrated to one-third in volume. The resulting solid was filtered and washed twice with ether before being suspended in EtOAc and neutralised with 1 N NaOH. The aqueous layer was extracted with EtOAc (3 x 150 mL) and the combined organics were washed with brine (150 mL), dried over Na2S04, filtered and concentrated under reduced pressure. The title compound was obtained as a yellow solid (39 g, 88 % yield); LC/MS (Method A): m/z 262 [M+H]+, Rt 2.57 min.
Reference Compound C: Methyl /^-^-[(^chlorophenyljcarbonyl]^- (methyloxy)phenyl]-yV2-{[(9H-fluoren-9-ylmethyl)oxy]carbonyl}-L-a-asparaginate
Methyl /V-{[(9H-fluoren-9-ylmethyl)oxy]carbonyl}-L-a-aspartyl chloride {Int. J. Peptide Protein Res. 1992, 40, 13-18) (93 g, 0.24 mol) was dissolved in CHCI3 (270 mL) and [2- amino-5-(methyloxy)phenyl](4-chlorophenyl)methanone (for a preparation see Reference compound B) (53 g, 0.2 mol) was added. The resulting mixture was stirred at 60°C for 1 h before being cooled and concentrated at 60% in volume. Ether was added at 0°C and the resulting precipitate was filtered and discarded. The filtrate was concentrated under reduced pressure and used without further purification.
Reference compound D: Methyl [(3S)-5-(4-chlorophenyl)-7-(methyloxy)-2-oxo-2,3- dihydro-1H-1 ,4-benzodiazepin-3-yl]acetate
To a solution of Methyl N1-[2-[(4-chlorophenyl)carbonyl]-4-(methyloxy)phenyl]-N2-{[(9H- fluoren-9-ylmethyl)oxy]carbonyl}-L-a-asparaginate (for a preparation see Reference compound C) (assumed 0.2 mol) in DCM (500 mL) was added Et3N (500 mL, 3.65 mol) and the resulting mixture was refluxed for 24h before being concentrated. The resulting crude amine was dissolved in 1 ,2-DCE (1.5 L) and AcOH (104 mL, 1.8 mol) was added carefully. The reaction mixture was then stirred at 60°C for 2h before being concentrated in vacuo and dissolved in DCM. The organic layer was washed with 1 N HCI and the aqueous layer was extracted with DCM (x3). The combined organic layers were washed twice with water, and brine, dried over Na2S04, filtered and concentrated under reduced pressure. The crude solid was recrystallised in MeCN leading to the title compound (51 g) as a pale yellow solid. The filtrate could be concentrated and recrystallised in MeCN to give to another 10 g of the desired product Rf = 0.34 (DCM/MeOH : 95/5).
HRMS (M+H)+ calculated for C19H18 35CIN204 373.0955; found 373.0957.
Reference compound E: Methyl [(3S)-5-(4-chlorophenyl)-7-(methyloxy)-2-thioxo-2,3- dihydro-1 H-1 ,4-benzodiazepi -3-yl]acetate
A suspension of P4Si0 (36.1 g, 81.1 mmol) and Na2C03 (8.6 g, 81.1 mmol) in 1 ,2-DCE (700 mL) at room temperature was stirred for 2 h before Methyl [(3S)-5-(4-chlorophenyl)- 7-(methyloxy)-2-oxo-2,3-dihydro-1 H-1 ,4-benzodiazepin-3-yl]acetate (for a preparation see Reference compound D) (16.8 g, 45.1 mmol) was added. The resulting mixture was stirred at 70°C for 2 h before being cooled and filtered. The solid was washed twice with DCM and the filtrate washed with sat. NaHC03 and brine. The organic layer was dried over Na2S04, filtered and concentrated under reduced pressure. The crude product was purified by flash-chromatography on silica gel (DCM/MeOH : 99/1 ) to afford the title compound (17.2 g, 98% yield) as a yellowish solid. LC/MS (Method A): m/z 389 [M(35CI)+H]+, Rt 2.64 min
HRMS (M+H)+ calculated for C19H18 35CIN203S 389.0727; found 389.0714. Reference compound F: Methyl [(3S)-2-[2-acetylhydrazino]-5-(4-chlorophenyl)-7- (methyloxy)-3H-1 ,4-benzodiazepin-3- l]acetate
To a suspension of Methyl [(3S)-5-(4-chlorophenyl)-7-(methyloxy)-2-thioxo-2,3-dihydro- 1 H-1 ,4-benzodiazepin-3-yl]acetate (for a preparation see Reference compound E (9.0 g, 23.2 mmol) in THF (300 mL) at 0°C was added hydrazine monohydrate (3.4 mL, 69.6 mmol) dropwise. The reaction mixture was stirred for 5h between 5°C and 15°C before being cooled at 0°C. Et3N (9.7 mL, 69.6 mmol) was then added slowly and acetyl chloride (7.95 mL, 69.6 mmol) was added dropwise. The mixture was then allowed to warm to room temperature for 16h before being concentrated under reduced pressure. The crude product was dissolved in DCM and washed with water. The organic layer was dried over Na2S04, filtered and concentrated in vacuo to give the crude title compound (9.7 g, 98% yield) which was used without further purification. Rf = 0.49 (DCM/MeOH : 90/10).
Reference compound G: Methyl [(4S)-6-(4-chlorophenyl)-1 -methyl-8-(methyloxy)-4H- [1 ,2,4]triazolo[4,3-a][1 ,4]benz
The crude Methyl [(3S)-2-[(1 Z)-2-acetylhydrazino]-5-(4-chlorophenyl)-7-(methyloxy)-3H- 1 ,4-benzodiazepin-3-yl]acetate (for a preparation see Reference compound F) (assumed 9.7 g) was suspended in THF (100 ml) and AcOH (60 mL) was added at room temperature. The reaction mixture was stirred at this temperature for 2 days before being concentrated under reduced pressure. The crude solid was triturated in /‘-Pr20 and filtered to give the title compound (8.7 g, 91 % over 3 steps) as an off-white solid.
HRMS (M+H)+ calculated for C21 H20CIN4O3 41 1.1229; found 41 1.1245.
Reference compound H: [(4S)-6-(4-Chlorophenyl)-1 -methyl-8-(methyloxy)-4H- [1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]acetic acid
To a solution of Methyl [(4S)-6-(4-chlorophenyl)-1 -methyl-8-(methyloxy)-4H- [1 ,2,4]triazolo[4,3-a][1 ,4]benzodiazepin-4-yl]acetate (for a preparation see Reference compound G)(7.4 g, 18.1 mmol) in THF (130 mL) at room temperature was added 1 N NaOH (36.2 mL, 36.2 mmol). The reaction mixture was stirred at this temperature for 5h before being quenched with 1 N HCI (36.2 mL) and concentrated in vacuo. Water is then added and the aqueous layer was extracted with DCM (x3) and the combined organic layers were dried over Na2S04, filtered and concentrated under reduced pressure to give the title compound (7 g, 98% yield) as a pale yellow solid.
PATENT
WO2014028547
http://www.nature.com/nature/journal/v468/n7327/fig_tab/nature09589_F1.html

http://www.google.com/patents/WO2014028547A1?cl=en
Zhao, Y., et al.: J. Med. Chem., 56, 7498 (2013); Mirguet, O., et al.: J. Med. Chem., 56, 7501 (2013);
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015119435 | 2015-04-30 | COMPOUNDS USEFUL FOR PROMOTING PROTEIN DEGRADATION AND METHODS USING SAME |
| US8975417 | 2015-03-10 | Pyrazolopyrrolidine derivatives and their use in the treatment of disease |
| US2014356322 | 2014-12-04 | Compounds & Methods for the Enhanced Degradation of Targeted Proteins & Other Polypeptides by an E3 Ubiquitin Ligase |
| US2014243322 | 2014-08-28 | BIVALENT BROMODOMAIN LIGANDS, AND METHODS OF USING SAME |
| US2014243321 | 2014-08-28 | BIOORTHOGONAL MONOMERS CAPABLE OF DIMERIZING AND TARGETING BROMODOMAINS, AND METHODS OF USING SAME |
| US2014243286 | 2014-08-28 | BROMODOMAIN LIGANDS CAPABLE OF DIMERIZING IN AN AQUEOUS SOLUTION, AND METHODS OF USING SAME |
| US2013079335 | 2013-03-28 | Benzotriazolodiazepine Compounds Inhibitors Of Bromodomains |
| US2012252781 | 2012-10-04 | Benzodiazepine Bromodomain Inhibitor |
| US2012220573 | 2012-08-30 | Benzodiazepine Bromodomain Inhibitor |
| US2012208800 | 2012-08-16 | Bromodomain Inhibitors For Treating Autoimmune And Inflammatory Diseases |
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015366877 | 2015-12-24 | METHODS OF TREATMENT OF HUMAN CYTOMEGALOVIRUS INFECTION AND DISEASES WITH BROMODOMAIN INHIBITORS |
| US2015299210 | 2015-10-22 | Benzodiazepine Bromodomain Inhibitor |
| US2015291562 | 2015-10-15 | IMIDE-BASED MODULATORS OF PROTEOLYSIS AND ASSOCIATED METHODS OF USE |
| US2015246923 | 2015-09-03 | 9H-PYRIMIDO[4,5-B]INDOLES AND RELATED ANALOGS AS BET BROMODOMAIN INHIBITORS |
| US2015238507 | 2015-08-27 | BENZODIAZEPINES FOR TREATING SMALL CELL LUNG CANCER |
| US2015210706 | 2015-07-30 | Benzodiazepine Bromodomain Inhibitor |
| US2015174138 | 2015-06-25 | DIAGNOSTIC AND TREATMENT METHODS IN SUBJECTS HAVING OR AT RISK OF DEVELOPING RESISTANCE TO CANCER THERAPY |
| US2015133436 | 2015-05-14 | Bromodomain Inhibitors For Treating Autoimmune And Inflammatory Diseases |
| US2015133434 | 2015-05-14 | Compositions and Methods for Reactivating Latent Immunodeficiency Virus |
| US2015119435 | 2015-04-30 | COMPOUNDS USEFUL FOR PROMOTING PROTEIN DEGRADATION AND METHODS USING SAME |
/////GSK-525762A, GSK-525762, GSK 525762A, GSK 525762, 1260907-17-2, phase 2,
CCNC(=O)CC1C2=NN=C(N2C3=C(C=C(C=C3)OC)C(=N1)C4=CC=C(C=C4)Cl)C
Asvasiran sodium (ALN-RSV01)

RNA, (C-U-U-G-A-C-U-U-U-G-C-U-A-A-G-A-G-C-C-DT-DT), COMPLEX WITH RNA (G-G-C-U-C-U-U-A-G-C-A-A-A-G-U-C-A-A-G-DT-DT)
Duplex of guanylyl-(3′->5′)-guanylyl-(3′->5′)-cytidylyl-(3′->5′)-uridylyl-(3′->5′)-cytidylyl-(3′->5′)-uridylyl-(3′->5′)-uridylyl-(3′->5′)-adenylyl-(3′->5′)-guanylyl-(3′->5′)-cytidylyl-(3′->5′)-adenylyl-(3′->5′)-adenylyl-(3′->5′)-adenylyl-(3′->5′)-guanylyl-(3′->5′)-uridylyl-(3′->5′)-cytidylyl-(3′->5′)-adenylyl-(3′->5′)-adenylyl-(3′->5′)-guanylyl-(3′->5′)-thymidylyl-(3′->5′)-thymidine and thymidylyl-(5′->3′)-thymidylyl-(5′->3′)-cytidylyl-(5′->3′)-cytidylyl-(5′->3′)-guanylyl-(5′->3′)-adenylyl-(5′->3′)-guanylyl-(5′->3′)-adenylyl-(5′->3′)-adenylyl-(5′->3′)-uridylyl-(5′->3′)-cytidylyl-(5′->3′)-guanylyl-(5′->3′)-uridylyl-(5′->3′)-uridylyl-(5′->3′)-uridylyl-(5′->3′)-cytidylyl-(5′->3′)-adenylyl-(5′->3′)-guanylyl-(5′->3′)-uridylyl-(5′->3′)-uridylyl-(5′->3′)-cytidine
Asvasiran sodium (ALN-RSV01),
C401H500N150O290P40,
CAS 1386946-83-3, 870094-26-1
Alnylam Pharmaceuticals
- Originator Alnylam Pharmaceuticals
- Class Antivirals; Small interfering RNA
- Mechanism of Action Nucleocapsid protein modulators; RNA interference
Treatment of Human Respiratory Syncytial Virus (RSV) Infection
Nucleocapsid protein modulators, RNA interference
- 05 Nov 2014 Alnylam receives patent allowance for RNAi technology in USA
- 20 Feb 2014 Suspended – Phase-II for Respiratory syncytial virus infections in USA (Intranasal) (Alnylam Form 10-K filed in February 2014)
- 20 Feb 2014 Suspended – Phase-I for Respiratory syncytial virus infections in Europe (Intranasal) (Alnylam Form 10-K filed in February 2014)
Aerosolised ALN-RSV01 – Alnylam; ALN RSV01; Intranasal ALN-RSV01 – Alnylam
Alnylam, under license from the University of South Alabama, and with Asian licensee Kyowa Hakko Kirin (formerly Kyowa Hakko Kogyo), is developing a nasally administered formulation of asvasiran sodium (ALN-RSV01), an siRNA that targets the respiratory syncytial virus (RSV) N gene and inhibits viral replication, for the potential treatment or prevention of RSV infection.
.In June 2007, a phase II trial was initiated; in January 2008, top-line data were reported . In March 2013, development was ongoing . In August 2008, Kyowa planned to file the drug for marketing approval in 2014. In March 2013, Alnylam was planning on seeking to outlicense the program to continue to advance the program in other regions .
Alnylam is also developing second-generation agents.
Ex-Asian licensee, Cubist Pharmaceuticals, in collaboration with Alnylam, was previously developing the program for the potential treatment or prevention of RSV infection . However, in February 2013, the deal was terminated . Alnylam was also developing an inhaled formulation of asvasiran sodium; however, in February 2014, the drug was no longer listed on the company’s development pipeline.
| WO-2006074346 | |
| WO-2009076679 | |
| WO-2006062596 | |
| WO-2010048590 |
WO 2016022464
WO 2015173701
WO 2015026792
WO 2014209983
WO 2014031784
US 20130273037
Nucleic Acids Research (2012), 40(21), 10585-10595
WO 2011163518
Drugs of the Future (2009), 34(10), 781-783
Current Opinion in Infectious Diseases (2008), 21(6), 639-643
Antiviral Research (2008), 77(3), 225-231



John Maraganore, president and chief executive officer of Alnylam Pharmaceuticals,

Delivering Value with Integrated Communications led by Cynthia Clayton, Vice President, Investor Relations and Corporate Communications at Alnylam Pharmaceuticals

From the left, Alnylam COO Barry Greene, Adrian Dede, Lauren Virnoche, CEO

Dr. Rachel Meyers, Senior Vice President, Research at Alnylam Pharmaceuticals

Dr. Dinah Sah, Vice President of Research and the head of the Alnylam HD team
//////Asvasiran sodium, ALN-RSV01, PHASE 2, Alnylam
SOME OTHER CHEMISTRY
Figure 6: GalNAc–siRNA conjugates.
From Delivery materials for siRNA therapeutics
- Nature Materials12,967–977(2013)doi:10.1038/nmat3765
- 23 October 2013
http://www.nature.com/nmat/journal/v12/n11/fig_tab/nmat3765_F6.html
 \
\
http://www.google.com/patents/EP2836595A2?cl=en

PF-05387552
PF-05387552
IRAK4
- Molecular Weight429.51
| Molecular Formula: | C25H27N5O2 |
|---|---|
| Molecular Weight: | 429.51418 g/mol |

Synthesis
PAPER
Bioorganic & Medicinal Chemistry Letters (2014), 24(9), 2066-2072
Volume 24, Issue 9, 1 May 2014, Pages 2066–2072
Identification and optimization of indolo[2,3-c]quinoline inhibitors of IRAK4
- L. Nathan Tumeya, , ,
- Diane H. Boschellia,
- Niala Bhagiratha,
- Jaechul Shima,
- Elizabeth A. Murphyb,
- Deborah Goodwinb,
- Eric M. Bennettc,
- Mengmeng Wangd,
- Lih-Ling Linb,
- Barry Pressa,
- Marina Shenb,
- Richard K. Frisbiea,
- Paul Morganb,
- Shashi Mohanb,
- Julia Shinb,
- Vikram R. Raob
- b Pfizer Global R&D, 200 Cambridge Park Dr., Cambridge, MA 02140, USA
- c Pfizer Global R&D, 87 Cambridgepark Dr., Cambridge, MA 02140, USA
- d Pfizer Global R&D, 1 Burtt Rd., Andover, MA 01810, USA
http://www.sciencedirect.com/science/article/pii/S0960894X14002832?np=y
IRAK4 is responsible for initiating signaling from Toll-like receptors (TLRs) and members of the IL-1/18 receptor family. Kinase-inactive knock-ins and targeted deletions of IRAK4 in mice cause reductions in TLR induced pro-inflammatory cytokines and these mice are resistant to various models of arthritis.
Herein we report the identification and optimization of a series of potent IRAK4 inhibitors. Representative examples from this series showed excellent selectivity over a panel of kinases, including the kinases known to play a role in TLR-mediated signaling. The compounds exhibited low nM potency in LPS- and R848-induced cytokine assays indicating that they are blocking the TLR signaling pathway.
A key compound (26) from this series was profiled in more detail and found to have an excellent pharmaceutical profile as measured by predictive assays such as microsomal stability, TPSA, solubility, and c log P. However, this compound was found to afford poor exposure in mouse upon IP or IV administration. We found that removal of the ionizable solubilizing group (32) led to increased exposure, presumably due to increased permeability. Compounds 26 and 32, when dosed to plasma levels corresponding to ex vivo whole blood potency, were shown to inhibit LPS-induced TNFα in an in vivo murine model.
To our knowledge, this is the first published in vivo demonstration that inhibition of the IRAK4 pathway by a small molecule can recapitulate the phenotype of IRAK4 knockout mice.

L. Nathan Tumey, Ph.D., Principal Research Scientist, Pfizer Global R&D
REFERENCES
///////////TLR signaling, Indoloquinoline, IRAK4, Kinase inhibitor, Inflammation, PF-05387552, PF 05387552, 1604034-71-0
N#Cc3ccc4c5cnc2cc(OCCCN1CCN(C)CC1)c(OC)cc2c5nc4c3
ETAMICASTAT

Etamicastat HCl salt
CAS: 677773-32-9 (HCl salt)
CAS 760173-05-5 (free base).
Chemical Formula: C14H16ClF2N3OS
Molecular Weight: 347.8088
Synonym: BIA 5-453; BIA5-453; BIA-5-453; Etamicastat
IUPAC/Chemical Name: (R)-5-(2-aminoethyl)-1-(6,8-difluorochroman-3-yl)-1,3-dihydro-2H-imidazole-2-thione hydrochloride
5-(2-Aminoethyl)-1-((3R)-6,8-difluoro-3,4-dihydro-2H-chromen-3-yl)-1,3-dihydro-2h-imidazole-2-thione
R)-5-(2-aminoethyl)-1-(6,8-difluorochroman-3-yl)-1,3-dihydroimidazole-2-thione hydrochloride,
PHASE 2, Treatment of Heart Failure Therapy, Hypertension
Bial-Portela and Ca, S.A

is a novel peripherally selective dopamine β-hydroxylase (DBH) inhibitor being developed by Bial-Portela and Ca, S.A. for treatment of hypertension and congestive heart failure.(1) The compound was shown to be well tolerated in healthy volunteers.
Etamicastat, also known as BIA 5-453, is a potent, reversible, peripherally selective dopamine β-hydroxylase inhibitor (DBH inhibitor). Chronic dopamine ß-hydroxylase inhibition with etamicastat effectively decreases blood pressure, although does not prevent the development of hypertension in the spontaneously hypertensive rat.

aReagents and conditions: a) Boc2O, EtOH, rt, 2 h; b) TBDMS-Cl, Et3N, DMAP, DCM, rt, 18 h; c) Dess–Martin periodinane, DCM, rt, 1 h; d) 2, KSCN, AcOH, EtOAc, reflux, 7 h; e) 2 N HCl, EtOAc, rt, 2 h.
Paper
Development of the Asymmetric Hydrogenation Step for Multikilogram Production of Etamicastat

The asymmetric hydrogenation of methyl (6,8-difluoro-2H-chromen-3-yl)carbamate is a key step in the manufacturing route to etamicastat. A development of this step including the ruthenium or rhodium catalyst screening and the influence of the catalyst preparation (isolated, preformed in solution or in situ), solvent, temperature, pressure, additive, and concentration on the performance of the given ligand was discussed. Scale-up experiments for the best catalysts under optimized conditions were described.
PAPER
J Med Chem 2006, 49(3): 1191
in the processes .
(J?) -5- (2-Aminoethyl) -1- (6, 8-difluorochroman-3-yl) -1, 3-dihydroimidazole-2 -thione hydrochloride (the compound of formula 1, below) is a potent, non-toxic and peripherally selective inhibitor of ϋβΗ, which can be used for treatment of certain cardiovascular disorders. Compound 1 is disclosed in WO2004/033447 , along with processes for its preparation.

1
The process disclosed in WO2004/033447 involves the reaction of ( R) – 6 , 8 -difluorochroman-3 -ylamine hydrochloride (the structure of ( R) -6, 8-difluorochroman-3 -ylamine is shown below as compound QA) , [4 – ( tert-butyldimethylsilanyloxy) -3 -oxobutyl] carbamic acid tert-butyl ester and potassium thiocyanate .

QA
(R) -6 , 8-difluorochroman- 3 -ylamine (compound QA) is a key intermediate in the synthesis of compound 1. The stereochemistry at the carbon atom to which the amine is attached gives rise to the stereochemistry of compound 1, so it is advantageous that compound QA is present in as pure enantiomeric form as possible. In other words, the (R) -enantiomer of compound QA should be in predominance, with little or no (S) enantiomer present. Thus, the process for preparing compound QA will advantageously produce compound QA with as high enantiomeric excess (ee) as possible.
Advantageous processes for preparing, for example, the compound of formula QA have now been found. In one aspect, the processes involve a biotransformation step. In another aspect, the processes involve chemical transformation. The processes may also be employed in the preparation of similar precursors useful in the production of other peripherally-selective inhibitors of dopamine -β -hydroxylase .
WO2008/136695 discloses a compound of formula YA, its (R) or (S) enantiomer, a mixture of its (R) and (S) enantiomers, or pharmaceutically acceptable salts thereof.

YA
The (R) -enantiomer of the compound of formula YA has been found to be a potent dopamines-hydroxylase inhibitor having high potency and significantly reduced brain access.
As disclosed in WO2008/136695 , the compound of formula YA may be prepared by reacting the compound of formula 1 with benzaldehyde under reductive alkylation conditions. In particular, (R) -5- (2 -aminoethyl ) -1- (6 , 8-difluorochroman-3 -yl) – 1 , 3 -dihydroimidazole-2 -thione and benzaldehyde may be reacted in the presence of a solvent or mixture of solvents, and a reducing agent such as sodium cyanoborohydride or sodium triacetoxyborohydride .
The compound of formula W may be prepared using a process as disclosed herein from the nitro chromene compound M.
The compound of formula WA may also be prepared using a process comprising bromination of 2 , 4 -difluorophenol to give bromophenol, alkylation of bromophenol with 4 -chloro-3 -oxo butanoate to give ketone followed by cyclization and decarboxylation to produce compound WA.

WA
According to an aspect of the present invention, there is provided the following 2 -part synthetic route from the starting material 2 , 4 -difluorophenol to (R) -5- (2 -aminoethyl ) -1- (6 , 8-difluorochroman-3 -yl) -1 , 3 -dihydroimidazole-2 – thione
hydrochloride :
Part (1)


Preferred reagents and conditions:
a) HMTA, CF3COOH, 115°C, 18 hours
b) CH2CHCN, DABCO, DMF, water, 70°C, 16 hours
c) H2S04, AcOH, 100°C, 1 hour
d) NaClO, NaOH, MeOH, 25°C, 24 hours
e) (R) -C3 -TunePhosRu (acac) 2 S/C 3000, 30 bar H2, MeOH, 80°C, 20 hours
f) Water, 2-propanol, reflux to 20°C
g) 40% KOH, MeOH, reflux, 24 hours
h) L-tartaric acid, ethanol, water, RT, 1 hour
Part (2)

![]()

Preferred reagents and conditions
a’) methyl vinyl ketone, t-BuONa, EtOAc, EtOH, 40-50°C, 2-3 hours
Br2, MeOH, 20-25°C, 5 hours
water, reflux, 1 hour
KOH, AcOH, reflux, 1 hour
HCl, water, 2-propanol, 75 °C, 4 hours
KSCN, AcOH, 100°C, 2-4 hours
NaHC03, water, EtOH
NaBH4, 2-propanol, THF, water, 20-25°C, 16 hours
HCl, 2-propanol, water, reflux, 1-2 hours
The ( R ) -5- (2-Aminoethyl) -1- (6, 8-difluorochroman-3 -yl) -1,3-dihydroimidazole-2 – thione hydrochloride
EXAMPLES
Example 1
Nitro chromene synthesis

To 3 , 5-difluoro-2-hydroxybenzaldehyde (lOg, 63mmol, leq) , di-n-butylamine (4.1g, 32mmol, 0.5eq) , phtalic anhydride (18.7g, 126mmol, 2eq) in toluene (500mL) was added nitroethanol (5.75g, 63mmol, leq) . The round bottomed flask fitted with a dean stark apparatus was refluxed for 18h. The mixture was cooled and nitroethanol (5.75g, 63mmol, leq) was added. The resulting reaction mixture was then reflux for 12h. After cooling, the solution was evaporated down to approximately 150mL and purified over silica gel (eluent ethyl acetate : hexane 1:1) this gave several fractions that contained only the product by TLC, these was evaporated under reduced pressure to yield 1.8g which was 100% pure by HPLC aera. Several more fractions were collected containing a mixture of product and starting material. These were combined and washed with 2% NaOH solution (2x50mL) to remove starting material. The organic layer was washed with water (50mL) , dried over sodium sulfate and evaporated under reduced pressure to give 2.49g of brown solid ( 100% pure by HPLC aera) . More fractions were collected. These were combined, washed with 2% NaOH solution (3xl00mL) , water (lOOmL) and dried over sodium sulfate. This was then filtered and evaporated down in vacuum to yield 6.14g of a brown solid which was 91.3% pure by HPLC aera. 6 , 8 -difluoro-3 -nitro-2H-chromene (9.90g, 73.4%) was obtained as a brown solid.
Example 2
Nitro chromene synthesis with column purification
To a solution of isobenzofuran-1 , 3 -dione (4,68 g, 31,6 mmol) , 3 , 5-difluoro-2 -hydroxybenzaldehyde (2,5 g, 15,81 mmol) in Toluene (25 ml) was added 2 -nitroethanol (2,88 g, 31,6 mmol). The resulting mixture was heated to reflux overnight (Dean stark) .
The reaction conversion was checked by TLC (eluent PE/EtOAc 9:1) . A yellow spot was observed and corresponds to the expected product .
Reaction was cooled to room temperature and a plug of silica gel was performed. A pale brown solid (3.9g) was obtained. “””H-NMR showed presence of product and starting material. The solid was dissolved in diethylether and the organic layer was washed with aqueous sodium carbonate, dried over Na2S04, filtered and concentrated under reduced pressure. A pale brown solid (1.7g,) was obtained. The 1H-NMR was indicated no starting material but still polymer from nitroethanol and residue of phtalic anhydride. A second silica plug (eluent: PE/EtOAc 95:5) was done. A pale yellow solid (1.5g) was obtained. 1H-NMR of solid showed only product and polymer. The solid was recrystallized from methanol/water . A pale yellow solid (1.05g, 31.2%) was obtained.
Example 3
Nitro chromene synthesis without column purification
To a solution of isobenzofuran- 1 , 3 -dione (18,74 g, 127 mmol) , 3 , 5-difluoro-2 -hydroxybenzaldehyde (10 g, 63,3 mmol) in Toluene (100 ml) was added 2 -nitroethanol (6,86 ml, 95 mmol) . The resulting mixture was heated to reflux for 24h (Dean stark) .
The reaction conversion was checked by HPLC and by 1H-NMR. Only 50% conversion was obtained.
The reaction mixture was cooled to room temperature and diluted with DCM (lOOmL) and 1M NaOH solution (200mL) .
The biphasic system was stirred for 30 minutes and then separated (very difficult to see phase separation) . The aqueous layer was washed with DCM (50mL) and the combined organic layers were washed twice with water (2x50ml) , dried over sodium sulfate. The filtered organic layer was concentrated under reduced pressure. To the residue was added methanol (50mL) . The methanol was then removed by distillation under reduced pressure. A brown solution precipitated when most of the methanol was removed. More methanol was added and more solid crushed out then few drops of water was added to increase the product precipitation. The brown slurry was stirred for 30 minutes and filtered. The brown solid was washed with methanol/water (1:9, 5mL) and dried in a vacuum oven at 40°C for 12h.6, 8-difluoro-3 -nitro-2H-chroraene (4,9 g, 22,99 mmol,) was obtained as brown solid in 36.3% yield.
HPLC showed a purity of 98% and 1H-NMR confirmed the structure and purity around 95%
Example 4
Reduction of nitro chromene to nitro-alkane (racemic mixture)

To a suspension of 6 , 8 -difluoro-3 -nitro-2H-chromene (213mg, 0,999 mmol) and silica (0,8 g, 0,999 mmol) in a mixture of CHC13 (10 ml) and IPA (3,4 ml) at 0°C was added portion wise sodium borohydride (95 mg, 2,498 mmol). The resulting mixture was stirred at 0°C for 45 minutes. Reaction conversion was checked by HPLC. 1 mL of acetic acid was added at 0°C and the resulting mixture was stirred for 30 minutes at room temperature. The slurry was filtered and the silica was washed with DCM. The filtrate was diluted with ethyl acetate and water and the biphasic system was separated. The aqueous layer was back extracted with ethyl acetate. The combined organic layers were washed with brine, dried over MgS04, filtered and concentrated under reduced pressure.
6 , 8-difluoro-3 -nitrochroman (196mg, 0,911 mmol, 91 % yield) was obtained as a pale yellow oil.
Example 5
Preparation of 6 , 8 -difluorochroman-3 -one from nitro chromene

A solution of 6, 8-difluoro-3 -nitro-2H-chromene (lOOmg, 0,469 mmol) in acetic acid (0.5 ml) is added slowly to a stirred slurry of iron (262 mg, 4,69 mmol) in acetic acid (1 ml) at 60.deg. C. The reaction mixture is stirred at 60. °C for 2 hour then allowed to cool to room temperature and stirred overnight. The reaction mixture is poured onto ice-water (30 ml) and filtered through Celite. The solid was wash with dichloromethane (DCM) (50 ml) . The organic portion is separated and washed with water (2 x 30 ml) and brine (30 ml) , dried over MgS04, filtered and concentrated in vacuo to give a brown oil. 6,8-difluorochroman-3 -one (75 mg, 0,407 mmol, 87 % yield) was obtained as a brown oil.
Example 6
Preparation of 6 , 8-difluorochroman-3 -one from methyl 6,8-difluoro-2H-chromen-3 -yl-carbamate

Methanol (1000m ml) was added to a slurry of methyl fluoro-2H-chromen-3 -yl -carbamate (250 g, 1.037 mol) hydrogen chloride 6N (2000 ml, 12 mol) at room temperature. The resulting mixture was reflux and stirred for 2 hours. Reaction monitored by HPLC.
Reaction was not complete but was stopped in order to avoid degradation of the product. The yellow solution was cooled to room temperature. A slurry (two type of solid) was observed and diluted with diethyl ether (300mL) . The resulting slurry was stirred at 5°C for 1 hour then filtered. The yellow solid was washed with water. The resulting wet yellow solid was suspended in diethylether (400mL) and petroleum ether (PE) (400mL) was added. Slight yellow solid was stirred at room temperature overnight, filtered and washed with PE (300mL) , dried in a vacuum oven at 30 °C for 4h. The wet sample was checked by NMR. No starting material was detected. A pale yellow solid (72.5g, solid 1) was obtained. The mother liquors were concentrated to dryness. A yellow solid was obtained, suspended in diethyl ether and PE. The slurry was then stirred for 4 hours, filtered, washed with PE . A dark yellow solid (4.5g, solid 2) was obtained. Solid 1 (2g) was diluted in DCM and washed with water (pH =6). The organic layer was then dried over Na2S04, filtered, concentrated to dryness. A crystalline pale yellow solid (1.9g, solid 3) was obtained. NMR showed the same purity for solid 3 as for solid 1. The remaining part of solid 1 was then diluted in DCM. The resulting organic layer was washed with water, dried over Na2S04, filtered and then concentrated to dryness. Slight yellow crystalline solid (68.5g, solid 4) was obtained. NMR confirmed high quality material.
Loss on Drying (LOD) : 1.03% .
Example 7
Biotransformation: Transaminases

Codexis transaminases ATA-025, ATA-251 and ATA-P2-A07 recognized 6 , 8 -difluorochroman-3 -one as the substrate and produced the corresponding 6 , 8 -difluorochroman-3 -amine .
References
1: Igreja B, Wright LC, Soares-da-Silva P. Sustained high blood pressure reduction with etamicastat, a peripheral selective dopamine β-hydroxylase inhibitor. J Am Soc Hypertens. 2015 Dec 19. pii: S1933-1711(15)00838-4. doi: 10.1016/j.jash.2015.12.011. [Epub ahead of print] PubMed PMID: 26803288.
2: Loureiro AI, Bonifácio MJ, Fernandes-Lopes C, Pires N, Igreja B, Wright LC, Soares-da-Silva P. Role of P-glycoprotein and permeability upon the brain distribution and pharmacodynamics of etamicastat: a comparison with nepicastat. Xenobiotica. 2015;45(9):828-39. doi: 10.3109/00498254.2015.1018985. Epub 2015 Jun 10. PubMed PMID: 25915108.
3: Loureiro AI, Soares-da-Silva P. Distribution and pharmacokinetics of etamicastat and its N-acetylated metabolite (BIA 5-961) in dog and monkey. Xenobiotica. 2015;45(10):903-11. doi: 10.3109/00498254.2015.1024780. Epub 2015 Apr 14. PubMed PMID: 25869244.
4: Pires NM, Igreja B, Moura E, Wright LC, Serrão MP, Soares-da-Silva P. Blood pressure decrease in spontaneously hypertensive rats folowing renal denervation or dopamine β-hydroxylase inhibition with etamicastat. Hypertens Res. 2015 Sep;38(9):605-12. doi: 10.1038/hr.2015.50. Epub 2015 Apr 9. PubMed PMID: 25854989.
5: Bonifácio MJ, Sousa F, Neves M, Palma N, Igreja B, Pires NM, Wright LC, Soares-da-Silva P. Characterization of the interaction of the novel antihypertensive etamicastat with human dopamine-β-hydroxylase: comparison with nepicastat. Eur J Pharmacol. 2015 Mar 15;751:50-8. doi: 10.1016/j.ejphar.2015.01.034. Epub 2015 Jan 29. PubMed PMID: 25641750.
6: Pires NM, Loureiro AI, Igreja B, Lacroix P, Soares-da-Silva P. Cardiovascular safety pharmacology profile of etamicastat, a novel peripheral selective dopamine-β-hydroxylase inhibitor. Eur J Pharmacol. 2015 Mar 5;750:98-107. doi: 10.1016/j.ejphar.2015.01.035. Epub 2015 Jan 30. PubMed PMID: 25641747.
7: Igreja B, Pires NM, Bonifácio MJ, Loureiro AI, Fernandes-Lopes C, Wright LC, Soares-da-Silva P. Blood pressure-decreasing effect of etamicastat alone and in combination with antihypertensive drugs in the spontaneously hypertensive rat. Hypertens Res. 2015 Jan;38(1):30-8. doi: 10.1038/hr.2014.143. Epub 2014 Oct 9. PubMed PMID: 25298210.
8: Loureiro AI, Bonifácio MJ, Fernandes-Lopes C, Igreja B, Wright LC, Soares-da-Silva P. Etamicastat, a new dopamine-ß-hydroxylase inhibitor, pharmacodynamics and metabolism in rat. Eur J Pharmacol. 2014 Oct 5;740:285-94. doi: 10.1016/j.ejphar.2014.07.027. Epub 2014 Jul 21. PubMed PMID: 25058908.
9: Almeida L, Nunes T, Costa R, Rocha JF, Vaz-da-Silva M, Soares-da-Silva P. Etamicastat, a novel dopamine β-hydroxylase inhibitor: tolerability, pharmacokinetics, and pharmacodynamics in patients with hypertension. Clin Ther. 2013 Dec;35(12):1983-96. doi: 10.1016/j.clinthera.2013.10.012. Epub 2013 Dec 2. PubMed PMID: 24296323.
10: Loureiro AI, Rocha JF, Fernandes-Lopes C, Nunes T, Wright LC, Almeida L, Soares-da-Silva P. Human disposition, metabolism and excretion of etamicastat, a reversible, peripherally selective dopamine β-hydroxylase inhibitor. Br J Clin Pharmacol. 2014 Jun;77(6):1017-26. doi: 10.1111/bcp.12274. PubMed PMID: 24168152; PubMed Central PMCID: PMC4093927.
11: Loureiro AI, Fernandes-Lopes C, Bonifácio MJ, Wright LC, Soares-da-Silva P. N-acetylation of etamicastat, a reversible dopamine-β-hydroxylase inhibitor. Drug Metab Dispos. 2013 Dec;41(12):2081-6. doi: 10.1124/dmd.113.053736. Epub 2013 Sep 6. PubMed PMID: 24013186.
12: Nunes T, Rocha JF, Vaz-da-Silva M, Falcão A, Almeida L, Soares-da-Silva P. Pharmacokinetics and tolerability of etamicastat following single and repeated administration in elderly versus young healthy male subjects: an open-label, single-center, parallel-group study. Clin Ther. 2011 Jun;33(6):776-91. doi: 10.1016/j.clinthera.2011.05.048. PubMed PMID: 21704242.
13: Vaz-da-Silva M, Nunes T, Rocha JF, Falcão A, Almeida L, Soares-da-Silva P. Effect of food on the pharmacokinetic profile of etamicastat (BIA 5-453). Drugs R D. 2011;11(2):127-36. doi: 10.2165/11587080-000000000-00000. PubMed PMID: 21548660; PubMed Central PMCID: PMC3585837.
14: Rocha JF, Vaz-Da-Silva M, Nunes T, Igreja B, Loureiro AI, Bonifácio MJ, Wright LC, Falcão A, Almeida L, Soares-Da-Silva P. Single-dose tolerability, pharmacokinetics, and pharmacodynamics of etamicastat (BIA 5-453), a new dopamine β-hydroxylase inhibitor, in healthy subjects. J Clin Pharmacol. 2012 Feb;52(2):156-70. doi: 10.1177/0091270010390805. PubMed PMID: 21343348.
15: Nunes T, Rocha JF, Vaz-da-Silva M, Igreja B, Wright LC, Falcão A, Almeida L, Soares-da-Silva P. Safety, tolerability, and pharmacokinetics of etamicastat, a novel dopamine-β-hydroxylase inhibitor, in a rising multiple-dose study in young healthy subjects. Drugs R D. 2010;10(4):225-42. doi: 10.2165/11586310-000000000-00000. PubMed PMID: 21171669; PubMed Central PMCID: PMC3585840.
16: Beliaev A, Learmonth DA, Soares-da-Silva P. Synthesis and biological evaluation of novel, peripherally selective chromanyl imidazolethione-based inhibitors of dopamine beta-hydroxylase. J Med Chem. 2006 Feb 9;49(3):1191-7. PubMed PMID: 16451083.
| Cited Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| WO1995007284A1 * | Aug 29, 1994 | Mar 16, 1995 | Smithkline Beecham Plc | Phosphinic acid derivatives with anti-hyper glycemic and/or anti-obesity activity |
| WO2006044293A2 * | Oct 11, 2005 | Apr 27, 2006 | Pharmacopeia Drug Discovery, Inc. | Bicyclic compounds as selective melanin concentrating hormone receptor antagonists for the treatment of obesity and related disorders |
| WO2012007548A1 * | Jul 14, 2011 | Jan 19, 2012 | Dsm Ip Assets B.V. | (r)-selective amination |
| WO2013002660A2 * | Jun 29, 2012 | Jan 3, 2013 | BIAL – PORTELA & Cª, S.A. | Process |
| GR1005093B * | Title not available |
| Reference | ||
|---|---|---|
| 1 | * | AL NEIRABEYEH M. ET AL.: “Methoxy and hydroxy derivatives of 3,4-dihydro-3-(di-n-propylamino)-2H-1-benzopyrans: new synthesis and dopaminergic activity“, EUROPEAN JOURNAL OF MEDICINAL CHEMISTRY, vol. 26, no. 5, 1991, EDITIONS SCIENTIFIQUE ELSEVIER, PARIS; FR, pages 497 – 504, XP023870436, ISSN: 0223-5234, DOI: 10.1016/0223-5234(91)90145-D |
| 2 | * | BELIAEV, A. ET AL.: “Process Research for Multikilogram Production of Etamicastat: A Novel Dopamine ß-Hydroxylase Inhibitor“, ORGANIC PROCESS RESEARCH & DEVELOPMENT, no. 16, 2012, American Chemical Society, Washington; US, pages 704 – 709, XP002731798, DOI: 10.1021/op300012d |
| 3 | * | BOYE, S. ET AL.: “N,N-Disubstituted aminomethyl benzofuran derivatives: synthesis and preliminary binding evaluation“, BIOORGANIC & MEDICINAL CHEMISTRY, no. 7, 1999, ELSEVIER SCIENCE LTD; GB, pages 335 – 341, XP002731795, ISSN: 0968-0896, DOI: 10.1016/S0968-0896(98)00239-9 |
| 4 | * | COMOY, C. ET AL.: “3-Amino-3,4-dihydro-2H-1-benzopyran Derivatives as 5-HT1A Receptor Ligandsand Potential Anxiolytic Agents. 2. Synthesis and QuantitativeStructure-Activity Relationship Studies of Spiro[pyrrolidine- andpiperidine-2,3′(2’H)-benzopyrans]“, JOURNAL OF MEDICINAL CHEMISTRY., vol. 39, no. 21, 1996, AMERICAN CHEMICAL SOCIETY. WASHINGTON; US, pages 4285 – 4298, XP002731797, ISSN: 0022-2623, DOI: 10.1021/JM950861W |
| 5 | * | SHIN, C. ET AL.: “Total Synthesis of Bistratamide G, a Metabolite of the PhilippinesAscidian Lissoclinum bistratum, from Dehydrotripeptides“, CHEMISTRY LETTERS, vol. 33, no. 6, 2004, Chemical Society of Japan, Tokyo; JP, pages 664 – 665, XP002731799, ISSN: 0366-7022, DOI: 10.1246/cl.2004.664 |
| 6 | * | VASSE, J. L. ET AL.: “New efficient conditions for the reduction with NADH models“, SYNLETT, October 1998 (1998-10-01), THIEME INTERNATIONAL, STUTTGART; DE, pages 1144 – 1146, XP002731796, ISSN: 0936-5214, DOI: 10.1055/s-1998-1876 |
| 7 | * | XIAO, G.-Q. ET AL.: “3-Nitro-2H-chromenes as a New Class of Inhibitors against Thioredoxin Reductase and Proliferation of Cancer Cells“, ARCHIV DER PHARMAZIE, no. 345, 2012, VCH VERLAGSGESELLSCHAFT MBH, WEINHEIM; DE, pages 767 – 770, XP002731794, ISSN: 0365-6233, DOI: 10.1002/ardp.201200121 |
////////Etamicastat, BIA-5-453 , PHASE 2, Treatment, Heart Failure Therapy, Hypertension, Bial-Portela and Ca, S.A
SMILES Code: FC1=CC(F)=C(OC[C@H](N2C(CCN)=CNC2=S)C3)C3=C1.[H]Cl
c1c(cc(c2c1C[C@H](CO2)n3c(c[nH]c3=S)CCN)F)F
Zamicastat

- BIAL – PORTELA & CA., S.A. [PT/PT]; À Avenida da Siderurgia Nacional P-4745-457 S. Mamede do Coronado (PT)

- Zamicastat is a dopamine beta-monooxygenase inhibitor in phase I clinical studies at BIAL for the treatment of hypertension and heart failure.
- Zamicastat is a potent and selective dopamine β-mono-oxygenase inhibitor. Zamicastat Prevents the Deterioration of Cardiometabolic and Inflammatory Biomarkers in a Genetic Model of Salt-sensitive Hypertension. Chronic high salt intake deteriorates several cardiometabolic and inflammatory biomarkers in Dahl/SS rats, which can be prevented by dopamine β-hydroxylase inhibition with zamicastat.
- crystalline forms of l-[(3R)-6,8-difluoro- 3,4-dihydro-2H-l-benzopyran-3-yl]-l,3-dihydro-5-[2-[(phenylmethyl)amino]ethyl]-2H- imidazole-2-thione, i.e. the Renantiomer of
and processes for preparing the same. Background and prior art:Interest in the development of inhibitors of dopamines-hydroxylase (ϋβΗ) has centred on the hypothesis that inhibition of this enzyme may provide significant clinical improvements in patients suffering from cardiovascular disorders such as hypertension or chronic heart failure. The rationale for the use of ϋβΗ inhibitors is based on their capacity to inhibit the biosynthesis of noradrenaline, which is achieved via enzymatic hydroxylation of dopamine. Activation of neurohumoral systems, chiefly the sympathetic nervous system, is the principal clinical manifestation of congestive heart failure (Parmley, W.W., Clinical Cardiology, 18: 440-445, 1995). Congestive heart failure patients have elevated concentrations of plasma noradrenaline (Levine, T.B. et al., Am. J. Cardiol., 49: 1659-1666, 1982), increased central sympathetic outflow (Leimbach, W.N. et al., Circulation, 73: 913- 919, 1986) and augmented cardiorenal noradrenaline spillover (Hasking, G.J. et al., Circulation, 73:615-621, 1966). Prolonged and excessive exposure of the myocardium to noradrenaline may lead to down-regulation of cardiac β] -adrenoceptors, remodelling of the left ventricle, arrhythmias and necrosis, all of which can diminish the functional integrity of the heart. Congestive heart failure patients who have high plasma concentrations of noradrenaline also have the most unfavourable long-term prognosis (Cohn, J.N. et al., N. Engl. J. Med., 311 :819-823, 1984). Of greater significance is the observation that plasma noradrenaline concentrations are already elevated in asymptomatic patients with no overt heart failure and can predict ensuing mortality and morbidity (Benedict, C.R. et al., Circulation, 94:690-697, 1996). An activated sympathetic drive is not therefore merely a clinical marker of congestive heart failure, but may contribute to progressive worsening of the disease.
Potent dopamines-hydroxylase inhibitors having high potency and significantly reduced brain access are disclosed in WO 2008/136695. WO 2008/136695 describes compounds of formula I:
I where Rls R2 and R3 are the same or different and signify hydrogens, halogens, alkyl, nitro, amino, alkylcarbonylamino, alkylamino or dialkylamino group; R4 signifies -alkylaryl or – alkylheteroaryl; X signifies CH2, oxygen atom or sulphur atom; n is 2 or 3; including the individual (R)- and (S)-enantiomers or mixtures of enantiomers thereof; and including pharmaceutically acceptable salts and esters thereof, wherein the term alkyl means hydrocarbon chains, straight or branched, containing from one to six carbon atoms, optionally substituted by aryl, alkoxy, halogen, alkoxycarbonyl or hydroxycarbonyl groups; the term aryl means a phenyl or naphthyl group, optionally substituted by alkyl, alkyloxy, halogen or nitro group; the term halogen means fluorine, chlorine, bromine or iodine; the term heteroaryl means heteroaromatic group. In particular, WO 2008/136695 describes l-[(3R)-6,8-difluoro-3,4-dihydro-2H-l-benzopyran-3-yl]-l,3-dihydro-5-[2- [(phenylmethyl)amino]ethyl]-2H-Imidazole-2-thione.
Processes for the preparation of compounds of formula I, and in particular l-[(3R)-6,8- difluoro-3,4-dihydro-2H-l-benzopyran-3-yl]-l,3-dihydro-5-[2-[(phenylmethyl)amino] ethyl] -2H-Imidazole-2-thione, are described in WO 2008/136695 and are incorporated by reference herein. It is known that polymorphic forms of the same drug may have substantially different pharmaceutically important properties such as dissolution characteristics and bioavailability as well as stability of the drug. Furthermore, different forms may have different particle size, hardness and glass transition temperature. Thus, one form may provide significant advantages over other forms of the same drug in solid dosage form manufacture processes, such as accurate measurement of the active ingredients, easier filtration, or improved stability during granulation or storage. Furthermore, a particular process suitable for one form may also provide drug manufacturers several advantages such as economically or environmentally suitable solvents or processes, or higher purity or yield of the desired product.
PATENT
http://www.google.com/patents/WO2012087174A2?cl=en
Preparation of compound 2
[0090] Six lots of compound 2 (designated as lots 1, 2, 3, 4, 5 and 6) were prepared. The starting materials were prepared according to the following experimental protocols.
Lot 1 (Form A)
To a suspension of (R)-5-(2-aminoethyl)-l-(6,8-difluorochroman-3-yl)-lH- imidazole-2(3H)-thione (6.23 g, 20 mmol) in a mixture of Dichloromethane (DCM – 40 ml) and Methanol (40.0 ml) was added BENZALDEHYDE (2.230 ml, 22.00 mmol). To the resulting clear solution SODIUM CYANOBOROHYDRIDE (1.9 g, 28.7 mmol) was added in portions at 20-25°C to avoid intensive foaming and the solution was stirred at 20- 25°C for 40 h. The solution was quenched at 20-25°C with IN HC1 (35 ml), neutralised with 3N NaOH (35 ml), the mixture was extracted with DCM (200 ml). The organic phase was washed with brine, dried (MgS04), evaporated to dryness. The oily residue crystallised from 2-propanol (40 ml) at 20-25°C over a week-end. The crystals were collected, washed with 2-propanol, dried to give 5.2 g of the crude product. Re- crystallisation from 2-propanol-DCM hasn’t removed all impurities. Everything collected, evaporated with silica, applied on a column, eluted with Ethyl Acetate (EA)->EA-MeOH 9:1->4: 1, fractions 8-25 collected to give 3.8 g. Re-crystallised from 2-propanol (45 ml) and DCM (120 ml, removed on a rotavap) to give 2.77 g => initial lot (a) (HPLC 98.3% area) and 0.3 g of undissolved filtered off, by TLC right product. Initial lot (a) re- crystallised from 2-propanol (35 ml) and DCM (95 ml, removed on a rotavap) to give 2.51 g => initial lot (b) (HPLC 98.3% area). Combined with the above undissolved, re- crystallised from acetonitrile (200 ml, reflux to ice bath) to give 2.57 g => initial lot (c) (HPLC 98.8% area). Re-crystallised from acetonitrile (180 ml, reflux to 15°C) to give 2.25 g => Lot 1 (HPLC 99.2% area), mp 190-92°C. Lot 2 (Form A)
[0092] (R)-5-(2-(benzylamino)ethyl)-l-(6,8-difluorochroman-3-yl)-lH-imidazole- 2(3H)-thione (12 g, 29.9 mmol) was dissolved with heating to reflux in Tetrahydrofuran (300 ml), the solution was cooled to 5-10°C, Water (510 ml) was added slowly (approx 10 min) with stirring. The mixture was stirred for 1 h, solid was collected, washed with water, dried to give 11.73 g of product, by HPLC 1% of (R)-5-(2-Aminoethyl)-l-(6,8- difluorochroman-3-yl)-l,3-dihydroimidazole-2-thione hydrochloride and 1% of less polar impurity. The product was dissolved in Tetrahydrofuran (300 ml) with heating to reflux, 2- Propanol (150 ml) was added, the solution was concentrated to approx 100 ml (crystallisation occured), stirred in ice for 1.5 h. Solid was collected, washed with 2- propanol, dried to give 11.2 g of product, by HPLC 0.8% of (R)-5-(2-aminoethyl)-l-(6,8- difluorochroman-3-yl)-lH-imidazole-2(3H)-thione hydrochloride and 0.5% of less polar impurity. The product was dissolved in Tetrahydrofuran (300 ml) with heating to reflux, 2- Propanol (150 ml) was added, the solution was concentrated to approx 100 ml (crystallisation occured), stirred at 20-25°C for 1 h. Solid was collected, washed with 2- propanol, dried to give (R)-5-(2-(benzylamino)ethyl)-l-(6,8-difluorochroman-3-yl)-lH- imidazole-2(3H)-thione (10.22 g, 25.5 mmol, 85 % yield).,
Lot 3 (form B)
To (R)-5-(2-aminoethyl)-l-(6,8-difluorochroman-3-yl)-lH-imidazole-2(3H)- thione (2.36 g, 7.58 mmol) in a mixture of Methanol (15.00 ml) and Dichloromethane (15 ml) was added BENZALDEHYDE (0.845 ml, 8.34 mmol). To the resulting clear solution SODIUM CYANOBOROHYDRIDE (0.702 g, 10.61 mmol) was added in portions at 20- 25°C to avoid intensive foaming and the solution was stirred at 20-25°C for 40 h. The solution was quenched at 20-25°C with IN HC1 (12 ml), neutralised with 3N NaOH (12 ml), the mixture was extracted with DCM (100 ml). The organic phase was washed with brine, dried (MgS04), evaporated to dryness. The residue was purified on a column with EA-MeOH 9: 1 as eluent, fractions collected, concentrated to approx 20 ml, cooled in ice. The precipitate collected, washed with Ethyl Acetate-Petroleum Ether 1 : 1, dried on air to give (R)-5-(2-(benzylamino)ethyl)-l-(6,8-difluorochroman-3-yl)-lH-imidazole-2(3H)- thione (1.55 g, 3.86 mmol, 50.9 % yield). Lot 4 (Form A)
To a 500 mL flask set up for atmospheric distillation was added (R)-5-(2- (benzylamino)ethyl)-l-(6,8-difluorochroman-3-yl)-lH-imidazole-2(3H)-thione (20 g, 49,8 mmol) and Tetrahydrofuran (400 ml) to afford a suspension. The suspension was heated until full dissolution was achieved (61°C) whereupon it was filtered. The resulting solution was then heated to 66°C in order to commence the distillation. A mixture of Water (125 ml) & 2-Propanol (125 ml) was added at the same rate as the distillate was collected. The distillation was continued until 400 mL of distillate was collected. Crystallisation commenced after ~320 mL of distillate was collected. The suspension was cooled to 20°C and aged for 45 min. before filtering and washing with additional 2- propanol (80 mL) and then dried under vacuum at 50°C overnight to give (R)-5-(2- (benzylamino)ethyl)-l-(6,8-difluorochroman-3-yl)-lH-imidazole-2(3H)-thione (18.79 g, 94%). Lot 5 (Form A)
To a mixture of Methanol (66 L) and Water (10 L) at 20°C was added purified (R)-5-(2-(benzylamino)ethyl)-l-(6,8-difluorochroman-3-yl)-lH-imidazole-2(3H)-thione hydrochloride (4.37 kg, 9.98 mol) to afford a suspension. The reaction mixture was then heated to 67°C to affect complete dissolution, whereupon IN Sodium hydroxide (10.48 Ls 10.48 mol, 1.05 eq) was added in a single portion. The reaction mixture was adjusted back to 67°C and held at 67°C for 30 min. The reaction mixture was then cooled to 20°C and aged at 20°C for at least 30 min. The reaction was then filtered and the filter cake washed with aqueous Methanol (1 : 1 v/v, 20 L), sucked down for 15 min. and then dried at 45°C under vacuum, to afford (R)-5-(2-(benzylamino)ethyl)-l-(6,8-difluorochroman-3-yl)-lH- imidazole-2(3H)-thione (3.855 kg, 96%) as a pale tan crystalline solid.
PATENT
WO 2015038022
http://www.google.com/patents/WO2015038022A1?cl=en
processes .
(J?) -5- (2-Aminoethyl) -1- (6, 8-difluorochroman-3-yl) -1, 3-dihydroimidazole-2 -thione hydrochloride (the compound of formula 1, below) is a potent, non-toxic and peripherally selective inhibitor of ϋβΗ, which can be used for treatment of certain cardiovascular disorders. Compound 1 is disclosed in WO2004/033447 , along with processes for its preparation.

1
The process disclosed in WO2004/033447 involves the reaction of ( R) – 6 , 8 -difluorochroman-3 -ylamine hydrochloride (the structure of ( R) -6, 8-difluorochroman-3 -ylamine is shown below as compound QA) , [4 – ( tert-butyldimethylsilanyloxy) -3 -oxobutyl] carbamic acid tert-butyl ester and potassium thiocyanate .

QA
(R) -6 , 8-difluorochroman- 3 -ylamine (compound QA) is a key intermediate in the synthesis of compound 1. The stereochemistry at the carbon atom to which the amine is attached gives rise to the stereochemistry of compound 1, so it is advantageous that compound QA is present in as pure enantiomeric form as possible. In other words, the (R) -enantiomer of compound QA should be in predominance, with little or no (S) enantiomer present. Thus, the process for preparing compound QA will advantageously produce compound QA with as high enantiomeric excess (ee) as possible.
Advantageous processes for preparing, for example, the compound of formula QA have now been found. In one aspect, the processes involve a biotransformation step. In another aspect, the processes involve chemical transformation. The processes may also be employed in the preparation of similar precursors useful in the production of other peripherally-selective inhibitors of dopamine -β -hydroxylase .
WO2008/136695 discloses a compound of formula YA, its (R) or (S) enantiomer, a mixture of its (R) and (S) enantiomers, or pharmaceutically acceptable salts thereof.

YA
The (R) -enantiomer of the compound of formula YA has been found to be a potent dopamines-hydroxylase inhibitor having high potency and significantly reduced brain access.
As disclosed in WO2008/136695 , the compound of formula YA may be prepared by reacting the compound of formula 1 with benzaldehyde under reductive alkylation conditions. In particular, (R) -5- (2 -aminoethyl ) -1- (6 , 8-difluorochroman-3 -yl) – 1 , 3 -dihydroimidazole-2 -thione and benzaldehyde may be reacted in the presence of a solvent or mixture of solvents, and a reducing agent such as sodium cyanoborohydride or sodium triacetoxyborohydride .
process comprises the following steps:

The route from 2 , 4-difluorophenol may be as described 9/064210.
Preferably, the reagents and conditions are:
(i) H2S04, acetic acid
(ii) NaOCl, MeOH/water
(iii) Ru-based catalyst, H2, 30 bars, MeOH
(iv) aqueous KOH, MeOH, L-tartaric acid
(v) KSCN, AcOH/lPA
(vi) NaBH4, BF3.THF complex, THF then IPA
n one aspect, the process comprises the following steps


i. KOH, Thioglycolic acid or cysteine
ii. MEK
According to an aspect of the present invention, there is provided the following 2 -part synthetic route from the starting material 2 , 4 -difluorophenol to (R) -5- (2 -aminoethyl ) -1- (6 , 8-difluorochroman-3 -yl) -1 , 3 -dihydroimidazole-2 – thione
hydrochloride :
Part (1)


Preferred reagents and conditions:
a) HMTA, CF3COOH, 115°C, 18 hours
b) CH2CHCN, DABCO, DMF, water, 70°C, 16 hours
c) H2S04, AcOH, 100°C, 1 hour
d) NaClO, NaOH, MeOH, 25°C, 24 hours
e) (R) -C3 -TunePhosRu (acac) 2 S/C 3000, 30 bar H2, MeOH, 80°C, 20 hours
f) Water, 2-propanol, reflux to 20°C
g) 40% KOH, MeOH, reflux, 24 hours
h) L-tartaric acid, ethanol, water, RT, 1 hour
Part (2)

![]()

Preferred reagents and conditions
a’) methyl vinyl ketone, t-BuONa, EtOAc, EtOH, 40-50°C, 2-3 hours
Br2, MeOH, 20-25°C, 5 hours
water, reflux, 1 hour
KOH, AcOH, reflux, 1 hour
HCl, water, 2-propanol, 75 °C, 4 hours
KSCN, AcOH, 100°C, 2-4 hours
NaHC03, water, EtOH
NaBH4, 2-propanol, THF, water, 20-25°C, 16 hours
HCl, 2-propanol, water, reflux, 1-2 hours
The ( R ) -5- (2-Aminoethyl) -1- (6, 8-difluorochroman-3 -yl) -1,3-dihydroimidazole-2 – thione hydrochloride may then be used to
prepare (R) -5- (2- (benzylamino) ethyl) -1- (6, 8-difluorochroman-3 -yl) -lH-imidazole-2 (3H) -thione as follows.

Preferred reaction conditions/reagents:
q) NaBH(OAc)3, PhCHO, IPA;
t) NaOH, MeOH , H20
Either r) and s) :
r) HCI aq;
s) MeOH/Toluene;
Or n) , o) and p) :
n) HCI aq;
o) MeOH, toluene;
p) IPA.
EXAMPLES
Example 1
Nitro chromene synthesis

To 3 , 5-difluoro-2-hydroxybenzaldehyde (lOg, 63mmol, leq) , di-n-butylamine (4.1g, 32mmol, 0.5eq) , phtalic anhydride (18.7g, 126mmol, 2eq) in toluene (500mL) was added nitroethanol (5.75g, 63mmol, leq) . The round bottomed flask fitted with a dean stark apparatus was refluxed for 18h. The mixture was cooled and nitroethanol (5.75g, 63mmol, leq) was added. The resulting reaction mixture was then reflux for 12h. After cooling, the solution was evaporated down to approximately 150mL and purified over silica gel (eluent ethyl acetate : hexane 1:1) this gave several fractions that contained only the product by TLC, these was evaporated under reduced pressure to yield 1.8g which was 100% pure by HPLC aera. Several more fractions were collected containing a mixture of product and starting material. These were combined and washed with 2% NaOH solution (2x50mL) to remove starting material. The organic layer was washed with water (50mL) , dried over sodium sulfate and evaporated under reduced pressure to give 2.49g of brown solid ( 100% pure by HPLC aera) . More fractions were collected. These were combined, washed with 2% NaOH solution (3xl00mL) , water (lOOmL) and dried over sodium sulfate. This was then filtered and evaporated down in vacuum to yield 6.14g of a brown solid which was 91.3% pure by HPLC aera. 6 , 8 -difluoro-3 -nitro-2H-chromene (9.90g, 73.4%) was obtained as a brown solid.
Example 2
Nitro chromene synthesis with column purification
To a solution of isobenzofuran-1 , 3 -dione (4,68 g, 31,6 mmol) , 3 , 5-difluoro-2 -hydroxybenzaldehyde (2,5 g, 15,81 mmol) in Toluene (25 ml) was added 2 -nitroethanol (2,88 g, 31,6 mmol). The resulting mixture was heated to reflux overnight (Dean stark) .
The reaction conversion was checked by TLC (eluent PE/EtOAc 9:1) . A yellow spot was observed and corresponds to the expected product .
Reaction was cooled to room temperature and a plug of silica gel was performed. A pale brown solid (3.9g) was obtained. “””H-NMR showed presence of product and starting material. The solid was dissolved in diethylether and the organic layer was washed with aqueous sodium carbonate, dried over Na2S04, filtered and concentrated under reduced pressure. A pale brown solid (1.7g,) was obtained. The 1H-NMR was indicated no starting material but still polymer from nitroethanol and residue of phtalic anhydride. A second silica plug (eluent: PE/EtOAc 95:5) was done. A pale yellow solid (1.5g) was obtained. 1H-NMR of solid showed only product and polymer. The solid was recrystallized from methanol/water . A pale yellow solid (1.05g, 31.2%) was obtained.
Example 3
Nitro chromene synthesis without column purification
To a solution of isobenzofuran- 1 , 3 -dione (18,74 g, 127 mmol) , 3 , 5-difluoro-2 -hydroxybenzaldehyde (10 g, 63,3 mmol) in Toluene (100 ml) was added 2 -nitroethanol (6,86 ml, 95 mmol) . The resulting mixture was heated to reflux for 24h (Dean stark) .
The reaction conversion was checked by HPLC and by 1H-NMR. Only 50% conversion was obtained.
The reaction mixture was cooled to room temperature and diluted with DCM (lOOmL) and 1M NaOH solution (200mL) .
The biphasic system was stirred for 30 minutes and then separated (very difficult to see phase separation) . The aqueous layer was washed with DCM (50mL) and the combined organic layers were washed twice with water (2x50ml) , dried over sodium sulfate. The filtered organic layer was concentrated under reduced pressure. To the residue was added methanol (50mL) . The methanol was then removed by distillation under reduced pressure. A brown solution precipitated when most of the methanol was removed. More methanol was added and more solid crushed out then few drops of water was added to increase the product precipitation. The brown slurry was stirred for 30 minutes and filtered. The brown solid was washed with methanol/water (1:9, 5mL) and dried in a vacuum oven at 40°C for 12h.6, 8-difluoro-3 -nitro-2H-chroraene (4,9 g, 22,99 mmol,) was obtained as brown solid in 36.3% yield.
HPLC showed a purity of 98% and 1H-NMR confirmed the structure and purity around 95%
Example 4
Reduction of nitro chromene to nitro-alkane (racemic mixture)

To a suspension of 6 , 8 -difluoro-3 -nitro-2H-chromene (213mg, 0,999 mmol) and silica (0,8 g, 0,999 mmol) in a mixture of CHC13 (10 ml) and IPA (3,4 ml) at 0°C was added portion wise sodium borohydride (95 mg, 2,498 mmol). The resulting mixture was stirred at 0°C for 45 minutes. Reaction conversion was checked by HPLC. 1 mL of acetic acid was added at 0°C and the resulting mixture was stirred for 30 minutes at room temperature. The slurry was filtered and the silica was washed with DCM. The filtrate was diluted with ethyl acetate and water and the biphasic system was separated. The aqueous layer was back extracted with ethyl acetate. The combined organic layers were washed with brine, dried over MgS04, filtered and concentrated under reduced pressure.
6 , 8-difluoro-3 -nitrochroman (196mg, 0,911 mmol, 91 % yield) was obtained as a pale yellow oil.
Example 5
Preparation of 6 , 8 -difluorochroman-3 -one from nitro chromene

A solution of 6, 8-difluoro-3 -nitro-2H-chromene (lOOmg, 0,469 mmol) in acetic acid (0.5 ml) is added slowly to a stirred slurry of iron (262 mg, 4,69 mmol) in acetic acid (1 ml) at 60.deg. C. The reaction mixture is stirred at 60. °C for 2 hour then allowed to cool to room temperature and stirred overnight. The reaction mixture is poured onto ice-water (30 ml) and filtered through Celite. The solid was wash with dichloromethane (DCM) (50 ml) . The organic portion is separated and washed with water (2 x 30 ml) and brine (30 ml) , dried over MgS04, filtered and concentrated in vacuo to give a brown oil. 6,8-difluorochroman-3 -one (75 mg, 0,407 mmol, 87 % yield) was obtained as a brown oil.
Example 6
Preparation of 6 , 8-difluorochroman-3 -one from methyl 6,8-difluoro-2H-chromen-3 -yl-carbamate

Methanol (1000m ml) was added to a slurry of methyl fluoro-2H-chromen-3 -yl -carbamate (250 g, 1.037 mol) hydrogen chloride 6N (2000 ml, 12 mol) at room temperature. The resulting mixture was reflux and stirred for 2 hours. Reaction monitored by HPLC.
Reaction was not complete but was stopped in order to avoid degradation of the product. The yellow solution was cooled to room temperature. A slurry (two type of solid) was observed and diluted with diethyl ether (300mL) . The resulting slurry was stirred at 5°C for 1 hour then filtered. The yellow solid was washed with water. The resulting wet yellow solid was suspended in diethylether (400mL) and petroleum ether (PE) (400mL) was added. Slight yellow solid was stirred at room temperature overnight, filtered and washed with PE (300mL) , dried in a vacuum oven at 30 °C for 4h. The wet sample was checked by NMR. No starting material was detected. A pale yellow solid (72.5g, solid 1) was obtained. The mother liquors were concentrated to dryness. A yellow solid was obtained, suspended in diethyl ether and PE. The slurry was then stirred for 4 hours, filtered, washed with PE . A dark yellow solid (4.5g, solid 2) was obtained. Solid 1 (2g) was diluted in DCM and washed with water (pH =6). The organic layer was then dried over Na2S04, filtered, concentrated to dryness. A crystalline pale yellow solid (1.9g, solid 3) was obtained. NMR showed the same purity for solid 3 as for solid 1. The remaining part of solid 1 was then diluted in DCM. The resulting organic layer was washed with water, dried over Na2S04, filtered and then concentrated to dryness. Slight yellow crystalline solid (68.5g, solid 4) was obtained. NMR confirmed high quality material.
Loss on Drying (LOD) : 1.03% .
Example 7
Biotransformation: Transaminases

Codexis transaminases ATA-025, ATA-251 and ATA-P2-A07 recognized 6 , 8 -difluorochroman-3 -one as the substrate and produced the corresponding 6 , 8 -difluorochroman-3 -amine .
PATENT
WO 2014077715
WO 2013002660
WO 2008136695
REFERNCES
International Journal of Pharmaceutics (Amsterdam, Netherlands) (2016), 501(1-2), 102-111.
| WO2012087174A2 | Dec 21, 2011 | Jun 28, 2012 | BIAL – PORTELA & Cª., S.A. | Crystalline forms and processes for their preparation |
| WO2012087174A3 * | Dec 21, 2011 | May 10, 2013 | BIAL – PORTELA & Cª., S.A. | Crystalline forms and processes for their preparation |
| WO2013002660A2 | Jun 29, 2012 | Jan 3, 2013 | BIAL – PORTELA & Cª, S.A. | Process |
| WO2014077715A1 * | Nov 14, 2013 | May 22, 2014 | BIAL – PORTELA & Cª, S.A. | 1,3-dihydroimidazole-2-thione derivatives for use in the treatment of pulmonary arterial hypertension and lung injury |
| US8481582 | May 6, 2008 | Jul 9, 2013 | Bial-Portela & Ca, S.A. | 1,3-dihydroimidazole-2-thione derivatives as inhibitors of dopamine-beta-hydroxylase |
| US8865913 | Jun 19, 2013 | Oct 21, 2014 | Bial-Portela & Ca, S.A. | Crystalline forms and processes for their preparation |
| WO1995007284A1 * | Aug 29, 1994 | Mar 16, 1995 | Smithkline Beecham Plc | Phosphinic acid derivatives with anti-hyper glycemic and/or anti-obesity activity |
| WO2006044293A2 * | Oct 11, 2005 | Apr 27, 2006 | Pharmacopeia Drug Discovery, Inc. | Bicyclic compounds as selective melanin concentrating hormone receptor antagonists for the treatment of obesity and related disorders |
| WO2012007548A1 * | Jul 14, 2011 | Jan 19, 2012 | Dsm Ip Assets B.V. | (r)-selective amination |
| WO2013002660A2 * | Jun 29, 2012 | Jan 3, 2013 | BIAL – PORTELA & Cª, S.A. | Process |
| GR1005093B * | Title not available |
///////Zamicastat, BIA-5-1058, dopamine beta-monooxygenase inhibitor, phase I, clinical studies, BIAL, treatment of hypertension , heart failure.
S=C4NC=C(CCNCc1ccccc1)N4[C@@H]2Cc3cc(F)cc(F)c3OC2
FDA approves new drug Venclexta (venetoclax) for chronic lymphocytic leukemia in patients with a specific chromosomal abnormality

April 11, 2016
Release
The U.S. Food and Drug Administration today approved Venclexta (venetoclax) for the treatment of patients with chronic lymphocytic leukemia (CLL) who have a chromosomal abnormality called 17p deletion and who have been treated with at least one prior therapy. Venclexta is the first FDA-approved treatment that targets the B-cell lymphoma 2 (BCL-2) protein, which supports cancer cell growth and is overexpressed in many patients with CLL.
According to the National Cancer Institute, CLL is one of the most common types of leukemia in adults, with approximately 15,000 new cases diagnosed each year. CLL is characterized by the progressive accumulation of abnormal lymphocytes, a type of white blood cell. Patients with CLL who have a 17p deletion lack a portion of the chromosome that acts to suppress cancer growth. This chromosomal abnormality occurs in approximately 10 percent of patients with untreated CLL and in approximately 20 percent of patients with relapsed CLL.
“These patients now have a new, targeted therapy that inhibits a protein involved in keeping tumor cells alive,” said Richard Pazdur, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research. “For certain patients with CLL who have not had favorable outcomes with other therapies, Venclexta may provide a new option for their specific condition.”
The efficacy of Venclexta was tested in a single-arm clinical trial of 106 patients with CLL who have a 17p deletion and who had received at least one prior therapy. Trial participants took Venclexta orally every day, beginning with 20 mg and increasing over a five-week period to 400 mg. Results showed that 80 percent of trial participants experienced a complete or partial remission of their cancer.
Venclexta is indicated for daily use after detection of 17p deletion is confirmed through the use of the FDA-approved companion diagnostic Vysis CLL FISH probe kit.
The most common side effects of Venclexta include low white blood cell count (neutropenia), diarrhea, nausea, anemia, upper respiratory tract infection, low platelet count (thrombocytopenia) and fatigue. Serious complications can include pneumonia, neutropenia with fever, fever, autoimmune hemolytic anemia, anemia and metabolic abnormalities known as tumor lysis syndrome. Live attenuated vaccines should not be given to patients taking Venclexta.
The FDA granted the Venclexta application breakthrough therapy designation, priority review status, and accelerated approval for this indication. These are distinct programs intended to facilitate and expedite the development and review of certain new drugs in light of their potential to benefit patients with serious or life-threatening conditions. Venclexta also received orphan drug designation, which provides incentives such as tax credits, user fee waivers and eligibility for exclusivity to assist and encourage the development of drugs for rare diseases.
Venclexta is manufactured by AbbVie Inc. of North Chicago, Illinois, and marketed by AbbVie and Genentech USA Inc. of South San Francisco, California. The Vysis CLL FISH probe kit is manufactured by Abbott Molecular of Des Plaines, Illinois.

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....