EU approves Takeda’s bowel drug Entyvio
Hot on the heels of an approval in the US, regulators in Europe have now also given Takeda’s Entyvio (vedolizumab) the nod for two inflammatory bowel diseases. The European Commission has granted Marketing Authorisation for use of the gut-selective humanised monoclonal antibody to treat adults with moderately to severely active ulcerative colitis (UC) and adults with moderately to severely active Crohn’s disease (CD).
Read more at: http://www.pharmatimes.com/Article/14-05-27/EU_approves_Takeda_s_bowel_drug_Entyvio.aspx#ixzz334DL7xQJ
Unusual structural and electronic properties make skin pigments superior radical scavengers

A black and insoluble biopolymer called eumelanin and other types of melanin together determine skin and hair color, particularly for dark phenotypes. Eumelanin is also a soft, biocompatible nanomaterial with technological potential. However, previous studies of this substance have primarily been carried out with synthetic samples. In the journal Angewandte Chemie, Italian researchers have now revealed why natural eumelanin is significantly superior to the synthetic version as a radical scavenger, antioxidant, and photo-protectant.
Thanks to its unusual optoelectronic, dielectric, metal-binding, and radical-scavenging properties, eumelanin could be useful for a variety of technical applications, including organic electronic components or antioxidants for plastics. However, it was recently discovered that the properties of synthetic eumelanin are significantly different from those of the natural product.
Within pigment cells, eumelanin is produced enzymatically from tyrosine or DOPA. An important intermediate step in this process is the isomerization of dopachrome to 5,6-dihydroxyindole-2-carboxylic acid (DHICA). In…
View original post 279 more words
Brain scans spot possible clues to chronic fatigue syndrome
Seeking better insight into chronic fatigue syndrome, a new brain scan investigation has pinpointed what could be the first evidence of a connection between nerve cell inflammation and the onset of this debilitating and somewhat mysterious illness, researchers say.
The finding stems from a small PET scan study, led by Yasuhito Nakatomi of the RIKEN Center for Life Science Technologies in Hyogo, Japan. The study involved just nine patients with chronic fatigue syndrome and 10 healthy participants.
However, the investigators believe that their initial results are the first to show that neuro-inflammation is a distinct feature of chronic fatigue syndrome. This inflammation affects specific areas of the brain that are commonly linked with the kind of fatigue, pain, depression, and thought-process difficulties long associated with the syndrome, the researchers noted.
“While the results will need to be confirmed in larger studies, it is a very exciting finding,” said Suzanne…
View original post 563 more words
Chlorination with convenience: Palau’chlor
I came across this article in JACS recently. I remember using chlorine gas myself and being a little trepidacious with it. It is a nasty gas to deal with. I had to remember to clean out the regulator after every use so it didn’t corrode and make it difficult to close at the most inopportune time. I used thionyl chloride a lot and that had a tendency to wreak havoc on my Rotovap at the time. Not nice stuff. Anyways.
What caught my eye with this article was that it was written by Phil Baran at Scripps and Martin Eastgate at Bristol-Myers Squibb in Chem. Dev., so it was worth a gander to take a look and see if this paper was a keeper for later use. The article I am referring to is “Palau’chlor: A Practical and Reactive Chlorinating Reagent”, by Phil S. Baran et al, J. Am. Chem…
View original post 109 more words
In battle of IPF drugs, Boehringer Ingelheim’s nintedanib impresses

NINTEDANIB
656247-17-5
-
UNII-G6HRD2P839
-
Vargatef
- BIBF 1120
methyl (3Z)-3-{[(4-{methyl[(4-methylpiperazin-1-yl)acetyl]amino}phenyl)amino](phenyl)methylidene}-2-oxo-2,3-dihydro-1H-indole-6-carboxylate
Boehringer Ingelheim International Gmbh

In battle of IPF drugs, BI’s nintedanib impresses
As an eagerly-anticipated debate kicks off at the American Thoracic Society conference in San Diego on drugs for idiopathic pulmonary fibrosis, Boehringer Ingelheim has posted promising late-stage data on its offering, nintedanib.
Read more at: http://www.pharmatimes.com/Article/14-05-18/In_battle_of_IPF_drugs_BI_s_nintedanib_impresses.aspx#ixzz32K0f3NqQ
Follow us: @PharmaTimes on Twitter
Nintedanib, a triple angiokinase inhibitor, is currently being evaluated against advanced HCC in phase I/II clinical trials. Here, we report the underlying molecular mechanism by which nintedanib (BIBF-1120) induces an anti-HCC effect.
NCI: An orally bioavailable, indolinone-derived, receptor tyrosine kinase (RTK) inhibitor with potential antiangiogenic and antineoplastic activities. Multitargeted tyrosine kinase inhibitor BIBF 1120 selectively binds to and inhibits vascular endothelial growth factor receptor (VEGFR), fibroblast growth factor receptor (FGFR) and platelet-derived growth factor receptor (PDGFR) tyrosine kinases, which may result in the induction of endothelial cell apoptosis; a reduction in tumor vasculature; and the inhibition of tumor cell proliferation and migration. In addition, this agent also inhibits members of the Src family of tyrosine kinases, including Src, Lck, Lyn, and FLT-3 (fms-like tyrosine kinase 3). VEGFR, FGFR and PDGFR RTKs play key roles in tumor angiogenesis
Nintedanib (formerly BIBF 1120; trade name Vargatef) is a small molecule of angiokinase inhibitor class inhibiting vascular endothelial growth factor receptor (VEGFR), fibroblast growth factor receptor (FGFR) and platelet derived growth factor receptor(PDGFR) being developed by Boehringer Ingelheim for use as an anti-vascular anti-cancer agent.
Mechanism of action
Nintedanib is an indolinone-derived drug that inhibits the process of blood vessel formation (angiogenesis) in tumours. Angiogenesis inhibitors stop the formation and reshaping of blood vessels in and around tumours, which reduces the tumour’s blood supply, starving tumour cells of oxygen and nutrients leading to cell death and tumour shrinkage. Unlike conventional anti-cancer chemotherapy which has a direct cell killing effect on cancer cells, angiogenesis inhibitors starve the tumour cells of oxygen and nutrients which results in tumour cell death. One of the advantages of this method of anti-cancer therapy is that it is more specific than conventional chemotherapy agents, therefore results in fewer and less severe side effects than conventional chemotherapy.[citation needed]
The process of new blood vessel formation (angiogenesis) is essential for the growth and spread of cancers. It is mediated by signaling molecules (growth factors) released from cancer cells in response to low oxygen levels. The growth factors cause the cells of the tumour’s blood vessel to divide and reorganize resulting in the sprouting of new vessels in and around the tumour, improving its blood supply.[citation needed]
Angiogenesis is a process that is essential for the growth and spread of all solid tumours, blocking it prevents the tumour from growing and may result in tumour shrinkage as well as a reduction in the spread of the cancer to other parts of the body. Nintedanib exerts its anti-cancer effect by binding to and blocking the activation of cell receptors involved in blood vessel formation and reshaping (i.e. VEGFR 1-3, FGFR 1-3 AND PDGFRα and β). Inhibition of these receptors in the cells that make up blood vessels (endothelial cells, smooth muscle cells and pericytes) by Nintedanib leads to programmed cell death, destruction of tumor blood vessels and a reduction in blood flow to the tumour. Reduced tumour blood flow inhibits tumor cell proliferation and migration hence slowing the growth and spread of the cancer.[1]
Adverse effects
Preclinical studies have shown that nintedanib binds in a highly selective manner to the ATP binding domain of its three target receptors, without binding to similarly shaped ATP domains in other proteins, which reduces the potential for undesirable side effects.[2]
The most common side effects observed with nintedanib were reversible elevation in liver enzymes (10-28% of patients) and gastrointestinal disturbance (up to 50%). Side effects observed with nintedanib were worse with the higher 250 mg dose, for this reason subsequent trials have used the equally clinically effective 200 mg dose.[1][2][3][4][5][6][7][8][9]
Nintedanib inhibits the growth and reshaping of blood vessels which is also an essential process in normal wound healing and tissue repair. Therefore a theoretical side effect of nintedanib is reduced wound healing however, unlike other anti-angiogenic agents, this side effect has not been observed in patients receiving nintedanib.[citation needed]
Studies
Preclinical studies have demonstrated that nintedanib selectively binds to and blocks the VEGF, FGF and PDGF receptors, inhibiting the growth of cells that constitute the walls of blood vessels (endothelial and smooth muscle cells and pericytes) in vitro. Nintedanib reduces the number and density of blood vessels in tumours in vivo, resulting in tumour shrinkage.[1][2] Nintedanib also inhibits the growth of cells that are resistant to existing chemotherapy agents in vitro, which suggests a potential role for the agent in patients with solid tumours that are unresponsive to or relapse following current first line therapy.[10]
Early clinical trials of nintedanib have been carried out in patients with non-small cell lung, colorectal, uterine, endometrial, ovarian and cervical cancer and multiple myeloma.[4][5][7][8][9] These studies reported that the drug is active in patients, safe to administer and is stable in the bloodstream. They identified that the maximum tolerated dose of nintedanib is 20 0 mg when taken once a day.

Clinical studies
In the first human trials, nintedanib halted the growth of tumours in up to 50% of patients with non-small cell lung cancer and 76% of patients with advanced colorectal cancer and other solid tumours.[4][8] A complete response was observed in 1/26 patients with non-small cell lung and 1/7 patients with ovarian cancer treated with nintedanib. A further 2 patients with ovarian cancer had partial responses to nintedanib.[8][9]
Two phase II trials have been carried out assessing the efficacy, dosing and side effects of nintedanib in non-small cell lung and ovarian cancer. These trials found that nintedanib delayed relapse in patients with ovarian cancer by two months[6] and that overall survival of patients with non-small cell lung who received nintedanib was similar to that observed with the FDA approved VEGFR inhibitor sorafenib. These trials also concluded that increasing the dose of the nintedanib has no effect on survival.[3]
Current clinical trials
Nintedanib is currently undergoing investigation in phase II and III clinical trials and is yet to be licensed by the FDA. Angiogenesis inhibitors such as nintedanib may be effective in a range of solid tumour types including; lung, ovarian, metastatic bowel, liver and brain cancer.
Several further phase I and II clinical trials with nintedanib are underway. Patients are also being recruited for three phase III clinical trials that will evaluate the potential benefit of nintedanib when added to existing 1st line treatments in patients with ovarian.[11] and 2nd line treatment in non-small cell lung cancer [12][13] The phase III trials of nintedanib in lung cancer have been named LUME-Lung 1 and LUME-Lung 2.
Current phase II trials are investigating the effect of nintedanib in patients with metastatic bowel cancer, liver cancer and the brain tumour: glioblastoma multiforme.[14]
Phase III trials are investigating the use of nintedanib in combination with the existing chemotherapy agents permexetred and docetaxel in patients with non-small cell lung cancer,[15] and in combination with carboplatin and paclitaxel as a first line treatment for patients with ovarian cancer.[16]
A phase III clinical trial is also underway examining the safety and efficacy of nintedanib on patients with the non-cancerous lung condition idiopathic pulmonary fibrosis.[17]
……………………….
http://pubs.acs.org/doi/abs/10.1021/jm900431g

Inhibition of tumor angiogenesis through blockade of the vascular endothelial growth factor (VEGF) signaling pathway is a new treatment modality in oncology. Preclinical findings suggest that blockade of additional pro-angiogenic kinases, such as fibroblast and platelet-derived growth factor receptors (FGFR and PDGFR), may improve the efficacy of pharmacological cancer treatment. Indolinones substituted in position 6 were identified as selective inhibitors of VEGF-, PDGF-, and FGF-receptor kinases. In particular, 6-methoxycarbonyl-substituted indolinones showed a highly favorable selectivity profile. Optimization identified potent inhibitors of VEGF-related endothelial cell proliferation with additional efficacy on pericyctes and smooth muscle cells. In contrast, no direct inhibition of tumor cell proliferation was observed. Compounds 2 (BIBF 1000) and 3 (BIBF 1120) are orally available and display encouraging efficacy in in vivo tumor models while being well tolerated. The triple angiokinase inhibitor 3 is currently in phase III clinical trials for the treatment of nonsmall cell lung cancer.
References
- Hilberg, F.; G. J. Roth, M. Krssak, S. Kautschitsch, W. Sommergruber, U. Tontsch-Grunt, P. Garin-Chesa, G. Bader, A. Zoephel, J. Quant, A. Heckel, W. J. Rettig (2008). “BIBF 1120: triple angiokinase inhibitor with sustained receptor blockade and good antitumor efficacy”. Cancer Res 68 (12): 4774–82. doi:10.1158/0008-5472.CAN-07-6307. ISSN 1538-7445. PMID 18559524.
- Hilberg, F.; U. Tontsch-Grunt, F. Colbatzky, A. Heckel, R. Lotz, J.C.A. van Meel, G.J. Roth (2004). “BIBF1120 a novel, small molecule triple angiokinase inhibitor: profiling as a clinical candidate for cancer therapy”. European Journal of Cancer Supplements 2(50).
- Reck, M.; R. Kaiser, C. Eschbach, M. Stefanic, J. Love, U. Gatzemeier, P. Stopfer, J. von Pawel (2011). “A phase II double-blind study to investigate efficacy and safety of two doses of the triple angiokinase inhibitor BIBF 1120 in patients with relapsed advanced non-small-cell lung cancer”. Ann Oncol. ISSN 1569-8041.
- Okamoto, I.; H. Kaneda, T. Satoh, W. Okamoto, M. Miyazaki, R. Morinaga, S. Ueda, M. Terashima, A. Tsuya, A. Sarashina, K. Konishi, T. Arao, K. Nishio, R. Kaiser, K. Nakagawa (2010). “Phase I safety, pharmacokinetic, and biomarker study of BIBF 1120, an oral triple tyrosine kinase inhibitor in patients with advanced solid tumors”. Mol Cancer Ther 9 (10): 2825–33. doi:10.1158/1535-7163.MCT-10-0379. ISSN 1538-8514. PMID 20688946.
- Mross, K.; M. Stefanic, D. Gmehling, A. Frost, F. Baas, C. Unger, R. Strecker, J. Henning, B. Gaschler-Markefski, P. Stopfer, L. de Rossi, R. Kaiser (2010). “Phase I study of the angiogenesis inhibitor BIBF 1120 in patients with advanced solid tumors”.Clin Cancer Res 16 (1): 311–9. doi:10.1158/1078-0432.CCR-09-0694. ISSN 1078-0432. PMID 20028771.
- Ledermann, J.A. (2009). “A randomised phase II placebo-controlled trial using maintenance therapy to evaluate the vascular targeting agent BIBF 1120 following treatment of relapsed ovarian cancer (OC)”. J Clin Oncol 27 (15s): (suppl; abstr 5501).
- Kropff, M.; J. Kienast, G. Bisping, W. E. Berdel, B. Gaschler-Markefski, P. Stopfer, M. Stefanic, G. Munzert (2009). “An open-label dose-escalation study of BIBF 1120 in patients with relapsed or refractory multiple myeloma”. Anticancer Res 29 (10): 4233–8.ISSN 1791-7530. PMID 19846979.
- Ellis, P. M.; R. Kaiser, Y. Zhao, P. Stopfer, S. Gyorffy, N. Hanna (2010). “Phase I open-label study of continuous treatment with BIBF 1120, a triple angiokinase inhibitor, and pemetrexed in pretreated non-small cell lung cancer patients”. Clin Cancer Res 16 (10): 2881–9. doi:10.1158/1078-0432.CCR-09-2944. ISSN 1078-0432.PMID 20460487.
- du Bois, A.; J. Huober, P. Stopfer, J. Pfisterer, P. Wimberger, S. Loibl, V. L. Reichardt, P. Harter (2010). “A phase I open-label dose-escalation study of oral BIBF 1120 combined with standard paclitaxel and carboplatin in patients with advanced gynecological malignancies”. Ann Oncol 21 (2): 370–5. doi:10.1093/annonc/mdp506.ISSN 1569-8041. PMID 19889612.
- Xiang, Q. F.; F. Wang, X. D. Su, Y. J. Liang, L. S. Zheng, Y. J. Mi, W. Q. Chen, L. W. Fu (2011). “Effect of BIBF 1120 on reversal of ABCB1-mediated multidrug resistance”.Cell Oncol (Dordr) 34 (1): 33–44. doi:10.1007/s13402-010-0003-7. ISSN 2211-3436.
- “Boehringer Ingelheim – AGO-OVAR 12 / LUME-Ovar 1 Trial Information”. 2011.
- “Boehringer Ingelheim – LUME-Lung 2 Trial Information”. 2011.
- “Boehringer Ingelheim – LUME-Lung 1 Trial Information”. 2011.
- http://clinicaltrials.gov/ct2/results?term=++%09+BIBF+1120&phase=1
- http://clinicaltrials.gov/ct2/show/NCT00805194 Phase III LUME-Lung 1: BIBF 1120 Plus Docetaxel as Compared to Placebo Plus Docetaxel in 2nd Line Non Small Cell Lung Cancer
- http://clinicaltrials.gov/ct2/show/NCT01015118 Phase III BIBF 1120 or Placebo in Combination With Paclitaxel and Carboplatin in First Line Treatment of Ovarian Cancer
- http://clinicaltrials.gov/ct2/show/NCT01335477 Safety and Efficacy of BIBF 1120 at High Dose in Idiopathic Pulmonary Fibrosis Patients II
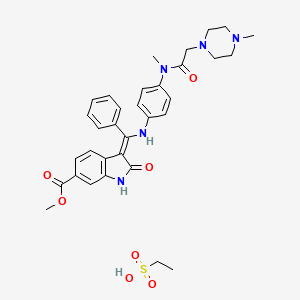
Ethanesulfonic acid – methyl (3Z)-3-{[(4-{methyl[(4-methyl-1-piperazinyl)acetyl]amino}phenyl)amino](phenyl)methylene}-2-oxo-6-indolinecarboxylate (1:1)
Nintedanib esylate
656247-18-6 [RN]
Methyl (3Z)-3-[({4-[N-methyl-2-(4-methylpiperazin-1-yl)acetamido]phenyl}amino)(phenyl)methylidene]-2-oxo-2,3-dihydro-1H-indole-6-carboxylate ethanesulfonate
Nintedanib esylate [USAN]
(3Z)-2,3-Dihydro-3-[[[4-[methyl[2-(4-methyl-1-piperazinyl)acetyl]amino]phenyl]amino]phenylmethylene]-2-oxo-1H-indole-6-carboxylic acid methyl ester ethanesulfonate
Cardamom: Courage in a Bottle
 I don’t think there could be a more perfect herb to help counteract the windy, rainy, cold, warm spring of Colorado than Cardamom. Weather you are looking for some courage and grounding or want get rid of a little excess Kapha, Cardamom can do that and a whole lot more. In ancient Egypt soldiers would put on Cardamom essential oil to increase their courage, before going into battle, so maybe you could use some before work.
I don’t think there could be a more perfect herb to help counteract the windy, rainy, cold, warm spring of Colorado than Cardamom. Weather you are looking for some courage and grounding or want get rid of a little excess Kapha, Cardamom can do that and a whole lot more. In ancient Egypt soldiers would put on Cardamom essential oil to increase their courage, before going into battle, so maybe you could use some before work.
Cardamom V-P+K- (Having trouble understanding these symbols? Click here.)
Cardamom can be used as an Essential Oil for smelling or as a spice in food.
Foods that use cardamom: Chai, Pumpkin Pie, Rice Pudding and Oatmeal. Spices are great for stoking the digestive fire and Cardamom is known for getting rid of Vata gas and Kapha mucous. Side note: its antispasmodic qualities, can be helpful for getting rid of hiccups, and intestinal cramping (Dillute…
View original post 54 more words
Herpes-loaded stem cells used to kill brain tumors

This shows tumor cells in green. This shows oHSV-loaded stem cells in red. This shows oHSV-infected tumor cells in yellow. Credit: Khalid Shah/Massachusetts General Hospital
Harvard Stem Cell Institute (HSCI) scientists at Massachusetts General Hospital have a potential solution for how to more effectively kill tumor cells using cancer-killing viruses. The investigators report that trapping virus-loaded stem cells in a gel and applying them to tumors significantly improved survival in mice with glioblastoma multiform, the most common brain tumor in human adults and also the most difficult to treat.
The work, led by Khalid Shah, MS, PhD, an HSCI Principal Faculty member, is published in the Journal of the National Cancer Institute. Shah heads the Molecular Neurotherapy and Imaging Laboratory at Massachusetts General Hospital.
Cancer-killing or oncolytic viruses have been used in numerous phase 1 and 2 clinical trialsfor brain tumors but with limited success. In preclinical studies…
View original post 535 more words
Virus combination effective against deadly brain tumor
The finding means that barriers to treating the disease, such as resistance to the drug temozolomide, may be overcome. The study, by Forsyth and colleagues in Canada, Texas and Florida, appeared in a recent issue ofNeuro-Oncology.
“Although temozolomide improves survival for patients with glioblastoma multiforme, drug resistance is a significant obstacle,” said Forsyth, the study lead author. “Oncolytic viruses that infect and break down cancer cellsoffer promising possibilities for overcoming resistance to targeted therapies.”
The authors note that oncolytic viruses have the potential to provoke a multipronged attack…
View original post 571 more words
Orphan Drugs: Top 2013 Selling Drugs Launched
.
.
Last week, GEN (Genetic Engineering & Biotechnology News), publishes an online list of the top 18 best-selling drugs that are launched in 2013, for those companies who provide 2013 sales information.
The chart below shows the 8 orphan drugs and the following data fields:
• GEN ranking out of 18
• Manufacturer
• 2013 Sales in $Millions
• FDA and EC approval dates
• Indication.
Top 8 Selling Orphan Drugs Launched in 2013
| GEN # | Product | Manufacturer | 2013 Sales | FDA App/EC App Dts | Indication |
| 2 | Pomalyst (Pomalidomide) | Celgene | $305 | 02.08.13/ 08.09.13 | Multiple Myeloma |
| 6 | Juxtapid (Lomitapide) | Aegerion Pharmaceuticals | $48.5 | 12.21.12/ 07.31.13 | Homozygous Familial
Hypercholester-olemia (HoFH) |
| 7 | Iclusig (Ponatinib) | Ariad Pharmaceuticals | $45.2 | 12.14.12/ 07.03.13 | CML/ Ph+ ALL |
| 10 | Gattex (Teduglutide) | NPS Pharmaceuticals | $31.8 | 12.21.12/ 08.30.12 | Short Bowel
Syndrome (SBS) |
| 11 | Tafinlar (Dabrafenib) * | GlaxoSmithKline | $26.9 | 05.29.13/ 09.02.13 | Metastatic
Melanoma with BRAF V600E mutation |
| 13 | Mekinist (Trametinib) * | GlaxoSmithKline | $16.8 | 05.29.13/ 09.02.13 |
View original post 172 more words

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....