Home » Posts tagged 'TUU8G1CX9O'
Tag Archives: TUU8G1CX9O
Danifexor
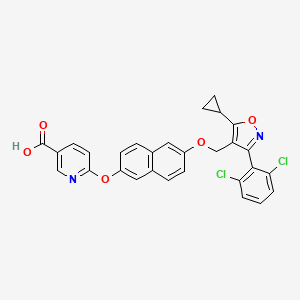
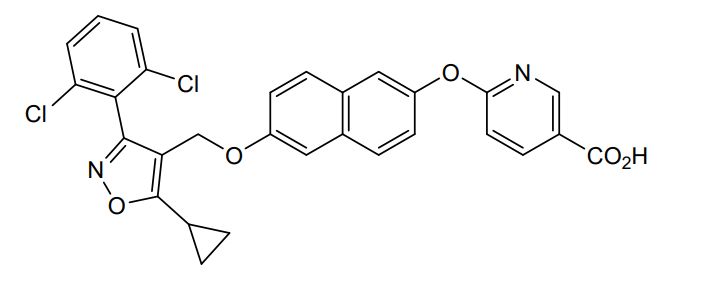
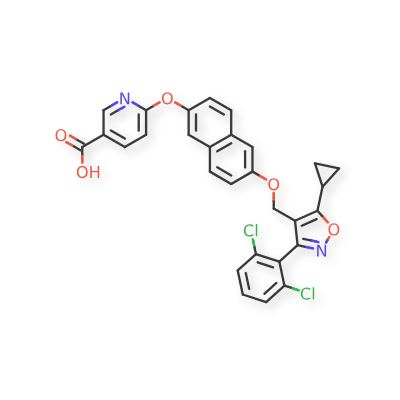
Danifexor
CAS 2648738-68-3
MF C29H20Cl2N2O5 MW547.386
6-[6-[[5-cyclopropyl-3-(2,6-dichlorophenyl)-1,2-oxazol-4-yl]methoxy]naphthalen-2-yl]oxypyridine-3-carboxylic acid
| 3-Pyridinecarboxylic acid, 6-[[6-[[5-cyclopropyl-3-(2,6-dichlorophenyl)-4-isoxazolyl]methoxy]-2-naphthalenyl]oxy]- |

farnesoid X receptor agonist, TUU8G1CX9O, HEC 96719, ASC42
Danifexor is an investigational drug that acts as a potent and selective agonist for the farnesoid X receptor (FXR). It was primarily being developed for the treatment of liver diseases such as Primary Biliary Cholangitis (PBC). ProbeChem +1
However, recent reports from April 2024 indicate that development for Danifexor has been discontinued because it was deemed non-competitive against other emerging therapies for PBC.
Key Properties and Identifiers
Danifexor is a non-steroidal molecule with specific chemical markers used in laboratory research:
- Target: Farnesoid X receptor (FXR).
Therapeutic Context
The drug was designed to target the FXR pathway, which regulates bile acid, lipid, and glucose metabolism.
- Primary Goal: Treatment of Primary Biliary Cholangitis (PBC), a chronic liver disease.
- Mechanism: As an agonist, it binds to and activates FXR to help reduce the toxic buildup of bile acids in the liver.
SYN
Example 1
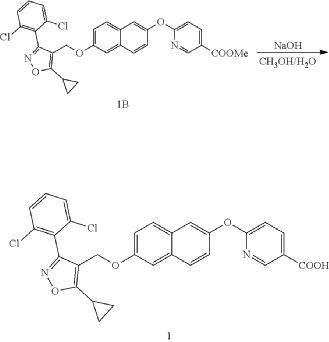
Preparation of 6-((6-((5-cyclopropyl-3-(2,6-dichlorophenyl)isoxazol-4-yl) methoxy)naphthalen-2-yl)oxy)nicotinic acid (Compound 1)
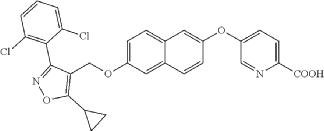
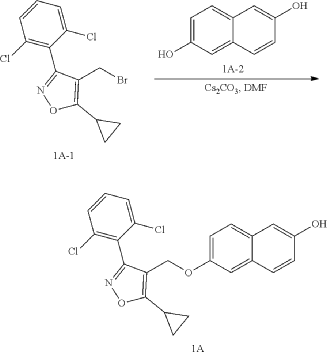
(a) Referring to the following reaction equation (Route A), Compound 1A-1 (1.0 g, 2.88 mmol, 1 eq.), Compound 1A-2 (0.46 g, 2.88 mmol, 1 eq.) and cesium carbonate (1.88 g, 5.76 mmol, 2 eq.) were dissolved in DMF (10 ml). The reaction was carried out at 65° C. for 2 h. After cooling, 10 ml water and 10 ml EA (ethyl acetate) were added for extraction, and the organic phase was washed with water and concentrated to dryness to give Compound 1A, 6-((5-cyclopropyl-3-(2,6-dichlorophenyl)isoxazol-4-yl)methoxy)naphthalen-2-ol, 0.8 g, yield: 65.0%. LCMS (ESI): calculated for C 23H 17C 12NO 3; [M+H] +: 426.1, found: 426.1.
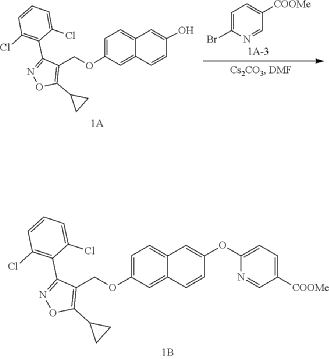
b) Referring to the following reaction equation, Compound 1A (0.2 g, 0.47 mmol, 1 eq.), 6-bromonicotinic acid methyl ester (0.1 g, 0.47 mmol, 1 eq.) and cesium carbonate (0.306 g, 0.94 mmol, 2 eq.) were dissolved in DMF (10 ml). The reaction was carried out at 65° C. for 2 h. After cooling, 10 ml water and 10 ml EA were added for extraction, and the organic phase was washed with water and concentrated to dryness to give Compound 1B, methyl 6((6((5-cyclopropyl-3-(2,6-dichlorophenyl)isoxazol-4-yl)methoxy)naphthalene-2-yl)oxy)nicotinate, 0.21 g, yield: 80.0%. LCMS (ESI): calculated for C 30H 22C 12N 2O 5; [M+H] +: 561.1, found: 561.1.
PAT
PAT
- Use and pharmaceutical composition of phenylisoxazolyl methylene-naphthalene-ether derivativesPublication Number: WO-2021109713-A1Priority Date: 2019-12-03
- Compounds for modulating activity of fxr and uses thereofPublication Number: WO-2021109712-A1Priority Date: 2019-12-03
- Uses and pharmaceutical compositions of phenylisoxazolylmethylene-naphthalene-ether derivativesPublication Number: CN-112891348-APriority Date: 2019-12-03
- Compounds that modulate FXR activity and their applicationsPublication Number: CN-112898289-APriority Date: 2019-12-03
- Compounds that modulate FXR activity and their applicationsPublication Number: CN-112898289-BPriority Date: 2019-12-03Grant Date: 2022-11-04
- Uses and pharmaceutical compositions of phenylisoxazolyl methylene-naphthalene-ether derivativesPublication Number: KR-20220101697-APriority Date: 2019-12-03
- Compounds for modulating activity of fxr and uses thereofPublication Number: EP-4073070-A1Priority Date: 2019-12-03
- Use and pharmaceutical composition of phenylisoxazolyl methylene-naphthalene-ether derivativesPublication Number: EP-4073071-A1Priority Date: 2019-12-03
- Compounds for modulating activity of fxr and uses thereofPublication Number: WO-2021108974-A1Priority Date: 2019-12-03



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
////////danifexor, ANAX LAB, farnesoid X receptor agonist, TUU8G1CX9O, HEC 96719, ASC42

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










