Home » Posts tagged 'purinoreceptor (P2X) antagonist'
Tag Archives: purinoreceptor (P2X) antagonist
Zanvipixant
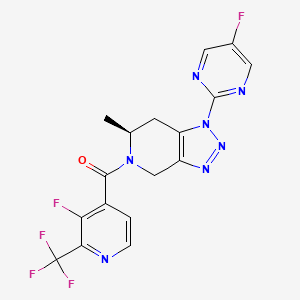
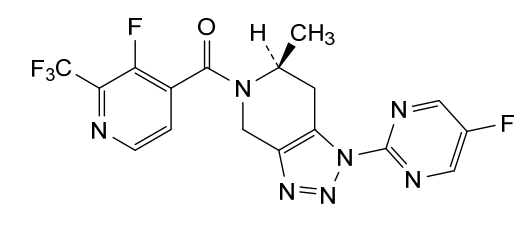
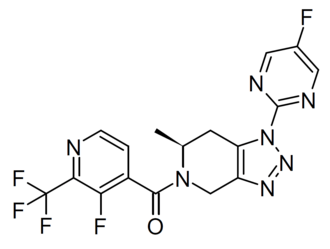
Zanvipixant
CAS 2166558-11-6
MF C17H12F5N7O MW 425.3 g/mol
[(6S)-1-(5-fluoropyrimidin-2-yl)-6-methyl-6,7-dihydro-4H-triazolo[4,5-c]pyridin-5-yl]-[3-fluoro-2-(trifluoromethyl)-4-pyridinyl]methanone
[(6S)-1-(5-fluoropyrimidin-2-yl)-6-methyl-1,4,6,7-tetrahydro-5H-[1,2,3]triazolo[4,5-c]pyridin-5-yl][3-fluoro-2-
(trifluoromethyl)pyridin-4-yl]methanone
purinoreceptor (P2X) antagonist, JNJ-55308942, JNJ 55308942, B7YN3CQ7S7,
JNJ-55308942 is under investigation in clinical trial NCT05328297 (A Study of JNJ-55308942 in the Treatment of Bipolar Depression).
JNJ-55308942 is an investigational drug that works as a P2X7 antagonist with a downstream effect of reducing interleukin-1β release.[1][2][3] It is developed by Janssen Pharmaceuticals for bipolar depression.[4]
Zanvipixant (JNJ-55308942) is an investigational small-molecule, brain-penetrant, and potent antagonist of the P2X7 receptor developed by Janssen (J&J). It is primarily studied for its potential to treat neuroinflammation-related conditions, including mood disorders and depression, by inhibiting P2X7-mediated IL-1β release.
Key Details for Zanvipixant:
- Target: P2X7 Receptor (P2X7R).
- Mechanism: Brain-penetrant antagonist reducing neuroinflammation, specifically inhibiting microglial IL-1β release.
- Chemical Properties: Structure typically includes a triazolopyridine moiety.
- Development Status: Evaluated as a potential CNS treatment for conditions related to neuroinflammation, with research highlighting its pharmacological profile, including good CNS partitioning.
- Alternative Name: JNJ-55308942.
Research indicates that the P2X7 receptor is activated by high levels of extracellular ATP during stress, leading to inflammation and behavioral deficits associated with depression. Zanvipixant has been studied in this context as a potential therapeutic intervention.
- A Study of JNJ-55308942 in the Treatment of Bipolar DepressionCTID: NCT05328297Phase: Phase 2Status: CompletedDate: 2025-07-11
- A Study in Healthy Participants to Evaluate the Effects of Multiple Doses of JNJ-55308942 on Cytochrome P450 Substrate Activity and on the Pharmacokinetics of Levonorgestrel/Ethinyl EstradiolCTID: NCT03547024Phase: Phase 1Status: CompletedDate: 2025-04-27
- A Positron Emission Tomography (PET) Study to Investigate P2X7 Receptor Occupancy by JNJ-55308942 Using [18F]-JNJ-64413739CTID: NCT03437590Phase: Phase 1Status: CompletedDate: 2025-04-27
- A Study to Investigate the Safety, Tolerability, and Pharmacokinetics of JNJ-55308942 in Healthy Male and Female ParticipantsCTID: NCT03151486Phase: Phase 1Status: CompletedDate: 2025-04-27
- A Randomized, Stratified, Double-blind, Placebo-Controlled Study to Investigate the Efficacy, Safety and Tolerability of JNJ-55308942 in Bipolar Depression
- EudraCT: 2021-004790-31
- Phase: Phase 2
- Status: Ongoing, Completed
- Date: 2022-05-31
SYN
Publication Name: Journal of Medicinal Chemistry
Publication Date: 2017-12-20
PMID: 29211470
DOI: 10.1021/acs.jmedchem.7b01279
SYN
Example 228
(S*)-(3-fluoro-2-(trifluoromethyl)pyridin-4-yl)(1-(5-fluoropyrimidin-2-yl)-6-methyl-6,7-dihydro-1H-[1,2,3]triazolo[4,5-c]pyridin-5(4H)-yl)methanone
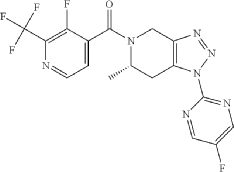
MS (ESI) mass calcd C 17H 12F 5N 7O, 425.1 m/z. found, 426.1 [M+H] +.
PAT
Example 228 (S)-(3 -fluoro-2-(trifluoromethyl)pyridin-4-yl)( 1 -(5 -fluoropyrimidin-2-yl)-6-methyl-6J-dihvdro-lH-ri,2,31triazolor4,5-c1pyridin-5(4H)-yl)methanone
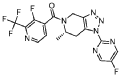
he title compound was prepared as described in Example 65, substituting (S)-l-(5-fluoropyrimidin-2-yl)-6-methyl-4,5,6,7-tetrahydro-lH-[l,2,3]triazolo[4,5-c]pyridine-hydrochloride salt (prepared as Step 2 intermediate of method II synthesis) in Example 220 for l-pyrimidin-2-yl-4,5,6,7-tetrahydro-lH-[l,2,3]triazolo[4,5-c]pyridine, 3-fluoro-2-(trifluoromethyl)isonicotinic acid for 2-chloro-3-(trifluoromethyl)benzoic acid and Hunig’s base (3.0 equiv) for Et3N. The powder x-ray diffraction pattern for this compound is shown in Figure 2. Alternatively, the title compound was synthesized using the following procedure:
Step 1 : (S)-l-(3-fluoro-2-(trifluoromethyl)isonicotinoyl)-2-methylpiperidin-4-one
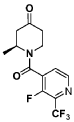
(S)-l-(3-fluoro-2-(trifluoromethyl)isonicotinoyl)-2-methylpiperidin-4-one.: (S)-2-methylpiperidin-4-one, TFA salt (108 g, 478 mmol, 1.0 equiv.) was suspended in DCM (1.4 L). Et3 (265 mL, 1.9 mol, 4.0 equiv.) and 3-fluoro-2-(trifluoromethyl)isonicotinoyl
chloride (1 19 g, 526 mmol, 1.1 equiv.) were added sequentially. The reaction solution was stirred at room temperature for 1 h. The precipitated solid was filtered and washed with EtOAc. The filtrate solution was concentrated and the residue was re-dissolved in EtOAc (500 mL). The organic layer was washed with saturated aHC03 aqueous solution, water and brine, dried over Na2S04, and concentrated. The crude product was triturated from EtOAc/hexanes to afford (S)-l-(3-fluoro-2-(trifluoromethyl)isonicotinoyl)-2-methylpiperidin-4-one (103 g, 368 mmol, 77%), which was used without further purification. MS = 305.1 (positive mode)
Step 2: (S)-(3-fluoro-2-(trifluoromethyl)pyridin-4-yl)(l-(5-fluoropyrimidin-2-yl)-6-methyl-6,7-dihvdro-lH-ri ,2,31triazolor4,5-c1pyridin-5(4H)-yl)methanone and (S)-(3-fluoro-2-(trifluoromethyl)pyridin-4-yl)(l-(5-fluoropyrimidin-2-yl)-4-methyl-6J-dihydro-lH
[ 1.2.3 Itriazolo [4.5 -c]pyridin-5 (4H)-yl)methanone
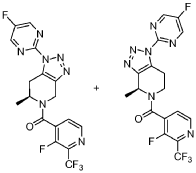
In a 5 L, three-neck, round-bottom flask equipped with a mechanic stirring, Dean-Stark trap, reflux condenser and an internal thermometer, to the solution of (S)-l-(3-fluoro-2-(trifluoromethyl)isonicotinoyl)-2-methylpiperidin-4-one, prepared in Step 1 above (100 g, 328 mmol, 1.0 equiv.) in toluene (1.5 L), -toluenesulphonic acid (0.62 g, 3.29 mmol, 0.01 equiv.), pyrrolidine (33 mL, 394 mmol, 1.2 equiv.) and 2-azido-5-fluoropyrimidine (59.4 g, 427 mmol, 1.3 equiv.) were added sequentially. The reaction mixture was heated to reflux temperature for 4 hours and then cooled to room temperature. aHC03 (55.2 g, 657 mmol, 2.0 equiv.) and mCPBA (162 g, 657 mmol, 2.0 equiv.) solution in EtOAC (-250 mL) were added sequentially. After stirring at room temperature for 2 hours, water and EtOAc were added. The organic layer was washed sequentially with 1M a2S03, saturated aHC03 aqueous solution, and brine, dried over Na2S04, and concentrated. The crude product was purified via column chromatography to afford a mixture of (S)-(3-fluoro-2-
(trifluoromethyl)pyridin-4-yl)(l-(5-flu^
[l,2,3]triazolo[4,5-c]pyridin-5(4H)-yl)methanone and (S)-(3-fluoro-2- (trifluoromethyl)pyridin-4-yl)(l-(5-fl^^
[l,2,3]triazolo[4,5-c]pyridin-5(4H)-yl)methanone in a 10: 1 ratio (97 grams).
Step 3 : (S)-(3-fluoro-2-(trifluoromethyl)pyridin-4-yl)(l -(5-fluoropyrimidin-2-yl)-6-methyl-6.7-dihvdro-lH-ri.2.31triazolor4.5-c1pyridin-5(4H)-yl)methanone
HPLC purification of the mixture from Step 2 was performed via achiral SFC (Stationary phase: Chiralcel OD-H 5μιη 250x30mm), (Mobile phase: 75% C02, 25% MeOH) to afford pure (S)-(3-fluoro-2-(trifluoromethyl)pyridin-4-yl)(l-(5-fluoropyrimidin-2-yl)-6-methyl-6,7-dihydro-lH-[l,2,3]triazolo[4,5-c]pyridin-5(4H)-yl)methanone (73 gram, 172 mmol, 52%). MS (ESI) mass calcd Ci7H12F5 70, 425.1 m/z found, 426.1 [M+H]+. XH NMR (500 MHz, CDC13) δ 8.84 – 8.69 (m, 2H), 8.68 – 8.57 (m, 1H), 7.71 – 7.49 (m, 1H), 5.90 -5.68 (d, J = 16.4 Hz, 0.5H), 5.66 – 5.54 (m, 0.5H), 4.76 – 4.58 (d, J = 15.8 Hz, 0.5H), 4.58 – 4.48 (m, 0.5H), 4.45 – 4.33 (m, 0.5H), 4.20 – 4.09 (m, 0.5H), 3.56 – 3.12 (m, 2H), 1.48 -1.18 (m, 3H).
2-chloro-3-(trifluoromethyl)benzoic acid and Hunig’s base (3.0 equiv) for Et3N. MS (ESI) mass calcd Ci7H12F5 70, 425.1 m/z found, 426.1 [M+H]+.
PAT
- P2X7 modulatorsPublication Number: US-9464084-B2Priority Date: 2013-03-14Grant Date: 2016-10-11
- P2X7 MODULATORSPublication Number: PT-2970267-TPriority Date: 2013-03-14
- P2X7 modulatorsPublication Number: US-11225478-B2Priority Date: 2013-03-14Grant Date: 2022-01-18
- P2X7 modulatorPublication Number: JP-6293861-B2Priority Date: 2013-03-14Grant Date: 2018-03-14
- P2X7 modulatorsPublication Number: AU-2019203186-A1Priority Date: 2013-03-14
- P2X7 modulatorsPublication Number: TW-201446762-APriority Date: 2013-03-14
- P2X7 regulatorsPublication Number: CN-110003200-APriority Date: 2013-03-14
- P2X7 modulatorsPublication Number: US-9066946-B2Priority Date: 2013-03-14Grant Date: 2015-06-30
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
“CTG Labs – NCBI”. clinicaltrials.gov. Retrieved 27 November 2023.
Bhattacharya, Anindya; Lord, Brian; Grigoleit, Jan-Sebastian; He, Yingbo; Fraser, Ian; Campbell, Shannon N.; Taylor, Natalie; Aluisio, Leah; O’Connor, Jason C.; Papp, Mariusz; Chrovian, Christa; Carruthers, Nicholas; Lovenberg, Timothy W.; Letavic, Michael A. (December 2018). “Neuropsychopharmacology of JNJ-55308942: evaluation of a clinical candidate targeting P2X7 ion channels in animal models of neuroinflammation and anhedonia”. Neuropsychopharmacology. 43 (13): 2586–2596. doi:10.1038/s41386-018-0141-6. ISSN 0893-133X. PMC 6224414. PMID 30026598.
Bhattacharya, Anindya; Ceusters, Marc (January 2020). “Targeting neuroinflammation with brain penetrant P2X7 antagonists as novel therapeutics for neuropsychiatric disorders”. Neuropsychopharmacology. 45 (1): 234–235. doi:10.1038/s41386-019-0502-9. ISSN 0893-133X. PMC 6879571. PMID 31477815.
Kolb, Hartmuth C.; Barret, Olivier; Bhattacharya, Anindya; Chen, Gang; Constantinescu, Cristian; Huang, Chaofeng; Letavic, Michael; Tamagnan, Gilles; Xia, Chunfang A.; Zhang, Wei; Szardenings, Anna Katrin (August 2019). “Preclinical Evaluation and Nonhuman Primate Receptor Occupancy Study of 18 F-JNJ-64413739, a PET Radioligand for P2X7 Receptors”. Journal of Nuclear Medicine. 60 (8): 1154–1159. doi:10.2967/jnumed.118.212696. ISSN 0161-5505. PMID 30733317. S2CID 73454130.
| Clinical data | |
|---|---|
| Trade names | Zanvipixant |
| Legal status | |
| Legal status | Investigational New Drug |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2166558-11-6 |
| PubChem CID | 90408860 |
| DrugBank | DB19110 |
| ChemSpider | 76771276 |
| UNII | B7YN3CQ7S7 |
| ChEMBL | ChEMBL3914857 |
| Chemical and physical data | |
| Formula | C17H12F5N7O |
| Molar mass | 425.323 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
////////zanvipixant, ANAX LAB, purinoreceptor (P2X) antagonist, JNJ-55308942, JNJ 55308942, B7YN3CQ7S7,
Relicpixant
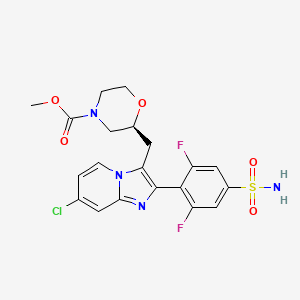
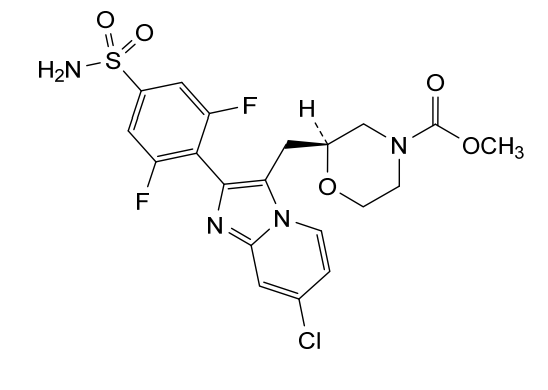
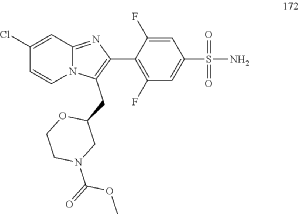
Relicpixant
CAS 2445366-94-7
MF C20H19ClF2N4O5S, Mw 500.9 g/mol
methyl (2S)-2-{[7-chloro-2-(2,6-difluoro-4-sulfamoylphenyl)imidazo[1,2-a]pyridin-3-yl]methyl}morpholine-4-
carboxylate
purinoreceptor (P2X) antagonist, 3NWJ8FHG2R
Relicpixant is a small molecule drug. The usage of the INN stem ‘-pixant’ in the name indicates that Relicpixant is a purinoreceptor (P2X) antagonist. Relicpixant has a monoisotopic molecular weight of 500.07 Da.
SYN
Example 172
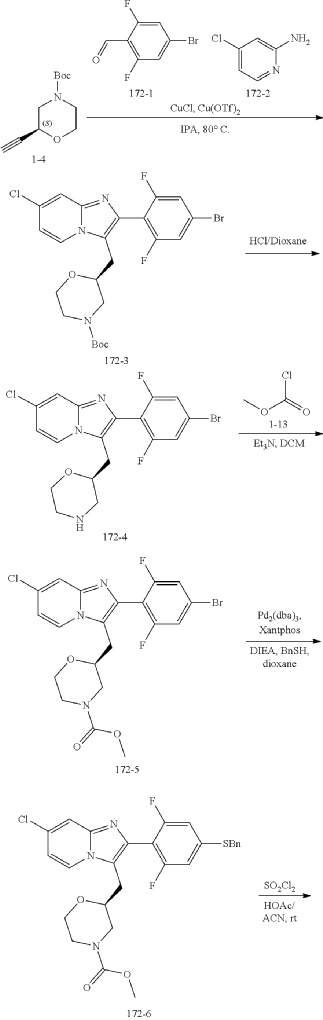
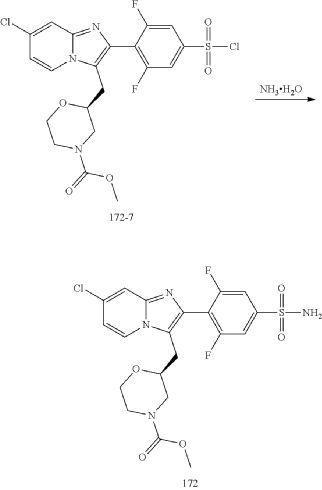
Step (8) Preparation of methyl (S)-2-((7-chloro-2-(2,6-difluoro-4-sulfamoylphenyl)imidazo[1,2-a]pyridin-3-yl)methyl)morpholine-4-carboxylate
| Aqueous ammonia (2 mL) was diluted with acetonitrile (1 mL) and added dropwise to the above reaction system at 0° C. The reaction system was reacted at room temperature for 0.5 h. The starting material was consumed completely, and a target product was generated as detected by LCMS. The reaction system was extracted with water and ethyl acetate twice, washed with saturated brine, dried over anhydrous sodium sulfate, and concentrated. The residue was purified by preparative chromatography to give compound 172 (185 mg, 99.74% purity) in the form of a white solid. LC-MS: [M+H] +=501.1. |
PAT
Example 1: Preparation of Compound of Formula A
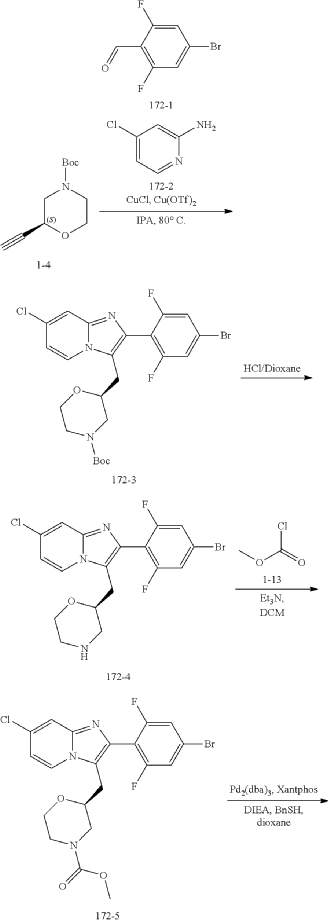
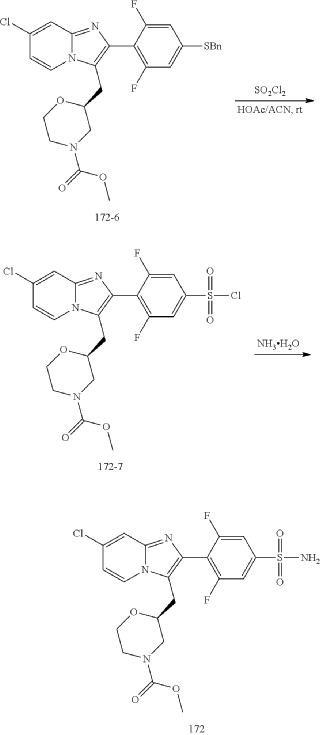
Step 6: Preparation of methyl (S)-2-((7-chloro-2-(2,6-difluoro-4-sulfamoylphenyl)imidazo[1,2-a]pyridin-3-yl)methyl)morpholine-4-carboxylate AS ABOVE
PAT
PAT
- Preparation method for heterocycloalkyl compound, and intermediate and application thereof heterocycloalkyl compoundPublication Number: WO-2022268218-A1Priority Date: 2021-06-24
- Pharmaceutical composition, preparation and preparation method and application thereofPublication Number: CN-115463133-BPriority Date: 2021-06-10Grant Date: 2024-03-01
- Pharmaceutical composition, preparation, and preparation method therefor and use thereofPublication Number: US-2024293420-A1Priority Date: 2021-06-10
- Pharmaceutical composition, preparation, and preparation method therefor and use thereofPublication Number: WO-2022258059-A1Priority Date: 2021-06-10
- Crystal form of heterocyclic compound, preparation method and application thereofPublication Number: CN-113929677-BPriority Date: 2020-06-29Grant Date: 2025-05-30
- Crystalline form of heterocyclic compound, preparation method therefor and application thereofPublication Number: EP-4169921-A1Priority Date: 2020-06-29
- Crystalline form of heterocyclic compound, preparation method therefor and application thereofPublication Number: WO-2022001820-A1Priority Date: 2020-06-29
- Crystalline form of heterocyclic compound, preparation method therefor and application thereofPublication Number: US-2023406850-A1Priority Date: 2020-06-29
- Heterocyclic compound intermediate, preparation method therefor and application thereofPublication Number: EP-3889154-A1Priority Date: 2018-12-29
- Heterocyclic compound intermediate, preparation method therefor and application thereofPublication Number: WO-2020135771-A1Priority Date: 2018-12-29
- Heterocyclic compound, intermediate, preparation method therefor and application thereofPublication Number: US-2023068538-A1Priority Date: 2018-12-29
- Heterocyclic compound, intermediate, preparation method and application thereofPublication Number: CN-113272301-BPriority Date: 2018-12-29Grant Date: 2024-04-26
- Heterocyclic compounds, intermediates, methods and applications thereof The present application applies to the Chinese patent application CN 2018116442319 with a filing date of December 29, 2018, and the Chinese patent application CN201910440214.3 with a filing date of May 24, 2019. Japan claims priority based on Chinese patent application CN200911016158.7 on October 24, 2019. In addition, the full text of the above Chinese patent application is incorporated in this application.Publication Number: JP-2022515879-APriority Date: 2018-12-29
- Heterocyclic compound, intermediate, preparation method therefor and application thereofPublication Number: US-2024034729-A9Priority Date: 2018-12-29
PAT



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

///////relicpixant, purinoreceptor (P2X) antagonist, 3NWJ8FHG2R

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










