Home » Posts tagged 'egalognastat'
Tag Archives: egalognastat
Egalognastat
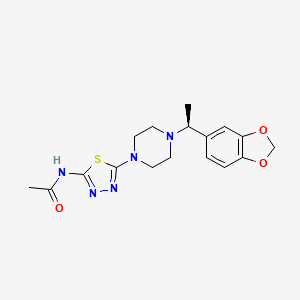
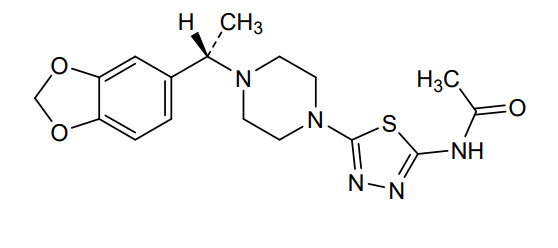
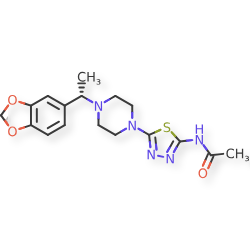
Egalognastat
CAS 1884154-02-2
MF C17H21N5O3S MW375.45
(S)-N-(5-(4-(1-(BENZO(D)(1,3)DIOXOL-5-YL)ETHYL)PIPERAZIN-1-YL)-1,3,4-THIA-DIAZOL-2-YL)ACETAMIDE
ACETAMIDE, N-(5-(4-((1S)-1-(1,3-BENZODIOXOL-5-YL)ETHYL)-1-PIPERAZINYL)-1,3,4-THIADIAZOL-2-YL)-
N-(5-{4-[(1S)-1-(2H-1,3-benzodioxol-5-yl)ethyl]piperazin-1-yl}-1,3,4-thiadiazol-2-yl)acetamide
O-GlcNAcase enzyme inhibitor, ASN90, ASN 90, E9QIS63WUM, ASN 120290
Egalognastat (ASN90) is a potent, selective, and brain-penetrant O-GlcNAcase (OGA) inhibitor (\(IC_{50} = 10.2 \text{ nM}\)). It acts as a disease-modifying agent in pre-clinical studies for neurodegenerative conditions by enhancing protein O-GlcNAcylation, which regulates tau and \(\alpha \)-synuclein pathology. Egalognastat is under investigation for diseases like Alzheimer’s and Parkinson’s.
Key Details on Egalognastat (ASN90):
- Mechanism: As a substrate-competitive OGA inhibitor, it binds to the OGA enzyme and reduces the removal of O-GlcNAc from proteins.
- Disease Targets: It is primarily studied for tauopathies and \(\alpha \)-synucleinopathies.
- Efficacy: Preclinical data shows it raises O-GlcNAcylation of brain proteins and has shown therapeutic potential in models of neurodegeneration.
- Distinction: Unlike earlier sugar-based inhibitors (like Thiamet G), Egalognastat is chemically distinct.
- Status: It is primarily used for research and preclinical development.
- Related Research: Recent studies (2025) have analyzed the potential synaptotoxic effects of OGA inhibitors, including Egalognastat (ASN90) and Ceperognastat, indicating that while they are effective for removing misfolded proteins, they may interfere with synaptic plasticity.
SYN
Publication Name: Journal of Medicinal Chemistry
Publication Date: 2024-08-07
PMID: 39109492
DOI: 10.1021/acs.jmedchem.4c01132
SYN
PAT
PAT
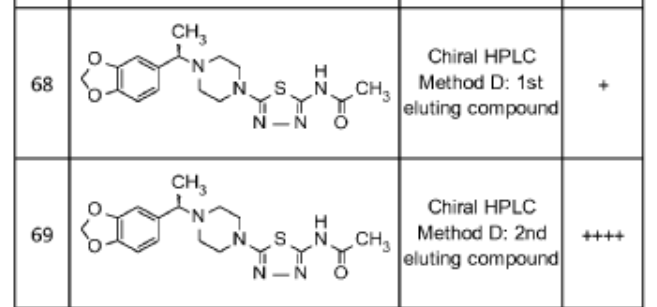

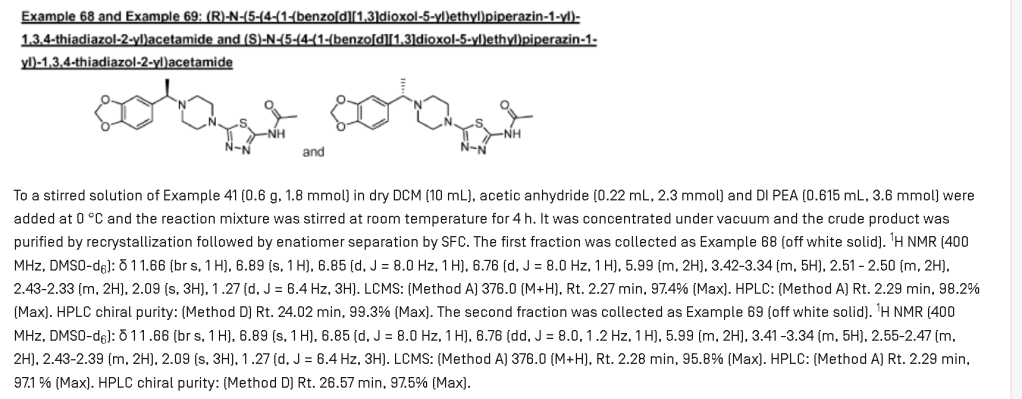
PAT
- Glycosidase inhibitorsPublication Number: US-12187741-B2Priority Date: 2014-08-28Grant Date: 2025-01-07
- Glucosidase inhibitorsPublication Number: ES-2893289-T3Priority Date: 2014-08-28Grant Date: 2022-02-08
- Glycosidase inhibitorsPublication Number: EP-3186243-B1Priority Date: 2014-08-28Grant Date: 2021-07-21
- Glycosidase inhibitorsPublication Number: US-10336775-B2Priority Date: 2014-08-28Grant Date: 2019-07-02
- Glycosidase inhibitorsPublication Number: US-11046712-B2Priority Date: 2014-08-28Grant Date: 2021-06-29
- Glycosidase inhibitorsPublication Number: EP-3868752-A1Priority Date: 2014-08-28
- Glycosidase InhibitorsPublication Number: CN-107108601-APriority Date: 2014-08-28
- Glycosidase inhibitorsPublication Number: US-2019367533-A1Priority Date: 2014-08-28
- Glycosidase inhibitorsPublication Number: WO-2016030443-A1Priority Date: 2014-08-28
- Glycosidase inhibitorsPublication Number: EP-3186243-A1Priority Date: 2014-08-28
- Glycosidase inhibitorPublication Number: JP-2019206554-APriority Date: 2014-08-28
- Glycosidase inhibitorsPublication Number: US-2017298082-A1Priority Date: 2014-08-28



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
////////egalognastat, anax labs, O-GlcNAcase enzyme inhibitor, ASN90, ASN 90, E9QIS63WUM, ASN 120290

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










