Home » Posts tagged 'dirozalkib'
Tag Archives: dirozalkib
Dirozalkib
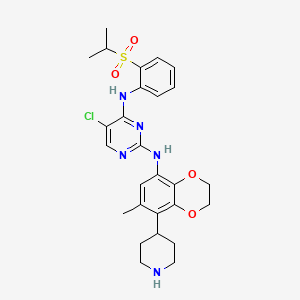
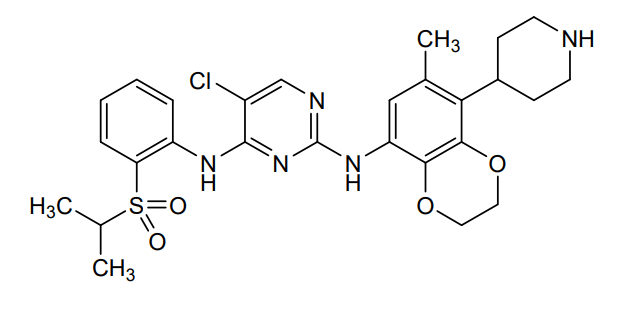
Dirozalkib
CAS 1893419-37-8
MF C27H32ClN5O4S MW558.1 g/mol
5-chloro-2-N-(6-methyl-5-piperidin-4-yl-2,3-dihydro-1,4-benzodioxin-8-yl)-4-N-(2-propan-2-ylsulfonylphenyl)pyrimidine-2,4-diamine

anaplastic lymphoma kinase (ALK) inhibitor, antineoplastic, XZP-3621, XZP 3621, Xuanzhu Biopharmaceutical, 2FH56C28YT
Dirozalkib (XZP-3621) is a novel, potent, and highly selective ALK/ROS1 tyrosine kinase inhibitor developed by Xuanzhu Biopharmaceutical to treat advanced ALK-positive non-small cell lung cancer (NSCLC). It demonstrated high efficacy (47.4% ORR, up to 89.3% in naive patients) in clinical trials and is designed to overcome resistance to earlier inhibitors.
Key Aspects of Dirozalkib
- Indication: Treatment of adult patients with ALK-positive locally advanced or metastatic non-small cell lung cancer (NSCLC).
- Mechanism: Acts as a dual-target ALK/ROS1 tyrosine kinase inhibitor (TKI), effective against ALK fusion-positive cells and various resistance mutations.
- Clinical Efficacy (Phase I/II): In studies, the drug showed significant antitumor activity with an Objective Response Rate (ORR) of 47.4% and an 89.3% ORR in ALK inhibitor-naive patients at 500 mg/day.
- Safety Profile: No dose-limiting toxicities occurred; the maximum tolerated dose was 600 mg/day, with a recommended dose of 500 mg/day. Common adverse events included diarrhea.
- Status: As of early 2026, the NDA (New Drug Application) for Dexitinib (Dirozalkib) was accepted by China’s NMPA, with potential for further market expansion.
- OriginatorXuanzhu Biopharmaceutical
- Class2 ring heterocyclic compounds; Amines; Aniline compounds; Antineoplastics; Chlorinated hydrocarbons; Piperidines; Pyrimidines; Small molecules; Sulfones
- Mechanism of ActionAnaplastic lymphoma kinase inhibitors
- RegisteredNon-small cell lung cancer
- 26 Aug 2025Chemical structure information added.
- 22 Aug 2025Registered for Non-small cell lung cancer (Late-stage disease) in China (PO) – First global approval
- 22 Aug 2025Efficacy and adverse events data from a phase III trial in Non-small cell lung cancer released by Xuanzhu Biopharmaceutical
- A Phase I Study of XZP-3621 in Chinese Patients With ALK or ROS1 Rearrangement Non-small Cell Lung CancerCTID: NCT05055232Phase: Phase 1Status: CompletedDate: 2025-07-24
- Food Effect and Mass Balance Study of XZP-3621 TabletsCTID: NCT05034120Phase: Phase 1Status: CompletedDate: 2025-05-25
- A Study of XZP-3621 in Chinese Patients With ALK Positive NSCLCCTID: NCT05482087Phase: Phase 2Status: Unknown statusDate: 2022-08-01
- A Study to Evaluate and Compare the Efficacy and Safety of XZP-3621 Versus CrizotinibCTID: NCT05204628Phase: Phase 3Status: Unknown statusDate: 2022-01-24
PAT
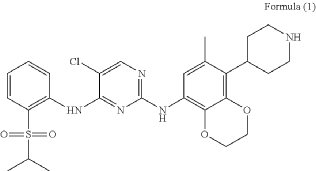
PAT
Example 3 Preparation of 2-((5-chloro-2-((7-methyl-8-(piperidin-4-yl)-2,3-dihydrobenzo[b][1,4]dioxin- 5-yl)amino)pyrimidin-4-yl)amino)-N,N-dimethylbenzenesulfonamide (compound 3)
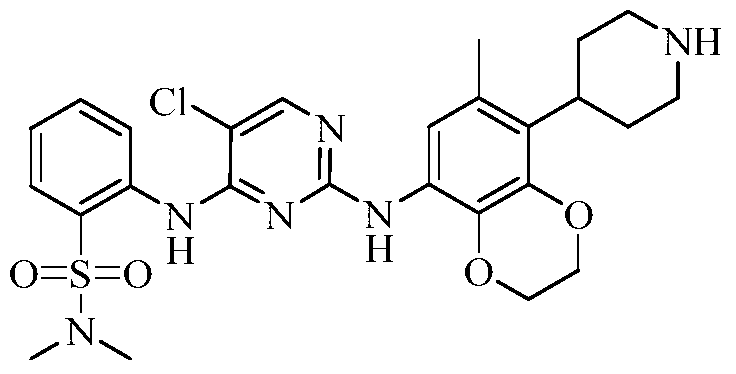
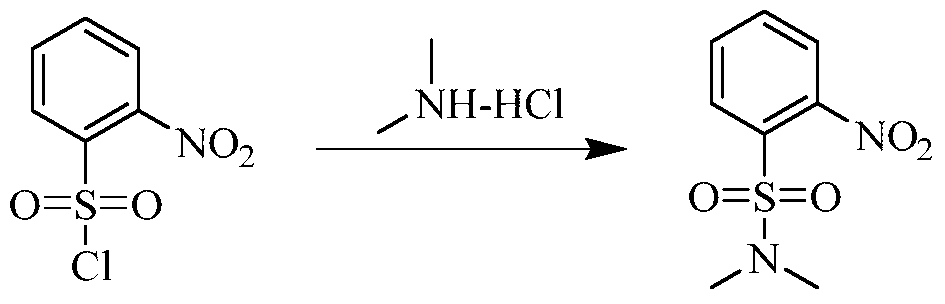
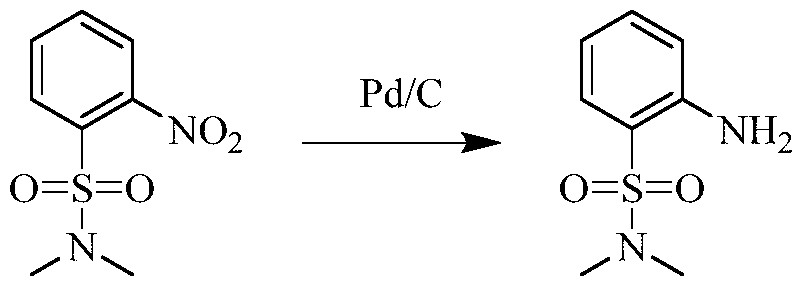
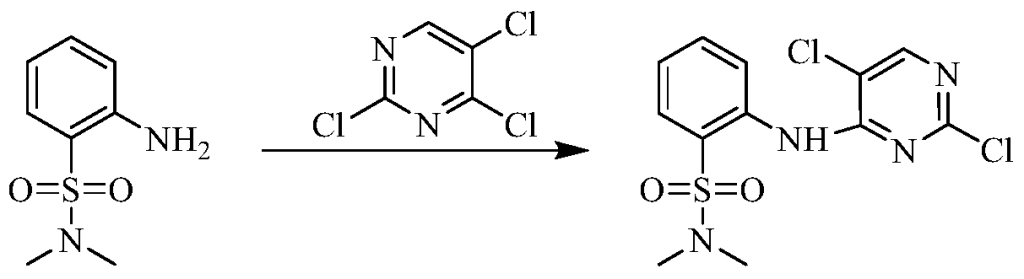
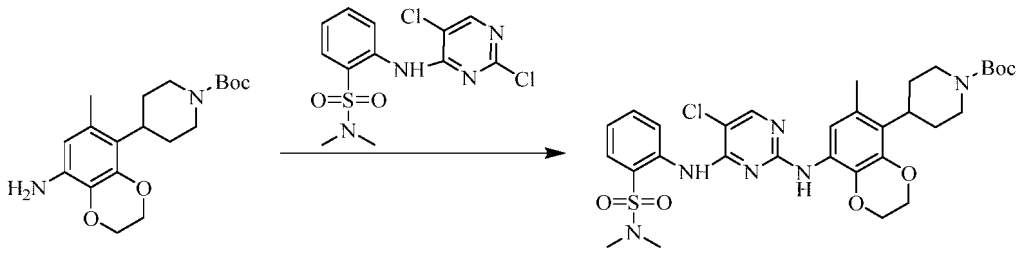
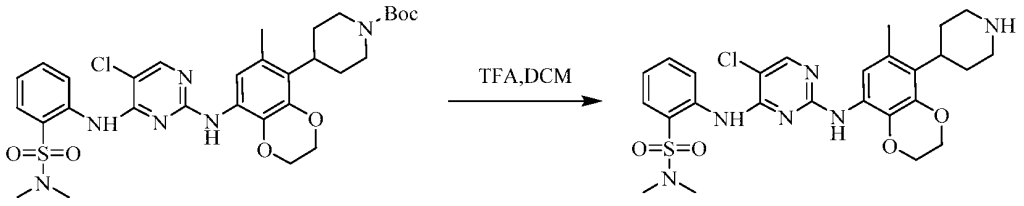
(5) Preparation of 2-((5-chloro-2-((7-methyl-8-(piperidin-4-yl)-2,3-dihydrobenzo[b][1,4]dioxin-5-yl)amino)pyrimidin-4-yl)amino)-N,N-dimethylbenzenesulfonamide
75 mg (0.114 mmol) of tert-butyl 4-(8-((5-chloro-4-((2-(N,N-dimethylaminosulfonyl)phenyl)amino)pyrimidin-2-yl)amino)-6-methyl-2,3-dihydrobenzo[b][1,4]dioxin-5-yl)piperidine-1-carboxylic acid ester was dissolved in dichloromethane (10 mL), and trifluoroacetic acid (1 mL) was added. The mixture was stirred at room temperature for 12 hours. The starting material disappeared as detected by TLC. Water (20 mL) was added, and the mixture was separated. The aqueous phase was extracted twice with dichloromethane (20 mL × 2). The organic phases were combined, dried over anhydrous sodium sulfate, and the solvent was removed by rotary evaporation. The crude product was purified by silica gel column chromatography (methanol:dichloromethane = 1:50) to obtain the final product (30 mg, yield 47.2%).
[0415]Molecular formula:
C26H31ClN6O4S Molecular weight: 559.08 LC-MS (m / z): 280.2 [ M /2+H ] +
[0416]
1H-NMR(400MHz,MeOD)δ:8.44(d,1H,J=1.2),8.11(s,1H),7.86(d,1H,J=1.2),7.56-7.60(m,1H),7.28-7.35(m,2H),4.26(s,4H),3.45-3.48(m,2H),3.06-3.15(m,3H),2.56-2.74(m,8H),2.17(s,3H),1.76-1.80(m,2H).



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: US-10011592-B2Priority Date: 2014-09-29Grant Date: 2018-07-03
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: US-2018086745-A9Priority Date: 2014-09-29
- Polycyclic anaplastic lymphoma kinase inhibitorPublication Number: EP-3202765-A1Priority Date: 2014-09-29
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: KR-20170055555-APriority Date: 2014-09-29
- Crystal form of polycyclic anaplastic lymphoma kinase inhibitorPublication Number: US-2023348443-A1Priority Date: 2020-01-17
- Crystal form of polycyclic anaplastic lymphoma kinase inhibitorPublication Number: US-12441717-B2Priority Date: 2020-01-17Grant Date: 2025-10-14
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: US-2017240534-A1Priority Date: 2014-09-29
- Polycyclic inhibitor of anaplastic lymphoma kinasePublication Number: KR-101909404-B1Priority Date: 2014-09-29Grant Date: 2018-10-17
- Polycyclic anaplastic lymphoma kinase inhibitorPublication Number: WO-2016050171-A1Priority Date: 2014-09-29
- Pharmaceutical composition of anaplastic lymphoma kinase inhibitor and preparation method thereforPublication Number: WO-2025140560-A1Priority Date: 2023-12-28
- CRYSTALLINE FORM OF A POLYCYCLIC ANAPLASIC LYMPHOMA KINASE INHIBITORPublication Number: EP-4092021-A4Priority Date: 2020-01-17
- Polycyclic anaplastic lymphoma kinase inhibitor crystalline formPublication Number: CN-113135905-BPriority Date: 2020-01-17Grant Date: 2023-11-21
- Crystal form of polycyclic anaplastic lymphoma kinase inhibitorPublication Number: WO-2021143819-A1Priority Date: 2020-01-17
- Crystal form of polycyclic anaplastic lymphoma kinase inhibitorPublication Number: EP-4092021-A1Priority Date: 2020-01-17
/////////dirozalkib, anax labs, anaplastic lymphoma kinase (ALK) inhibitor, antineoplastic, XZP-3621, XZP 3621, Xuanzhu Biopharmaceutical, 2FH56C28YT

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










