Home » Uncategorized (Page 64)
Category Archives: Uncategorized
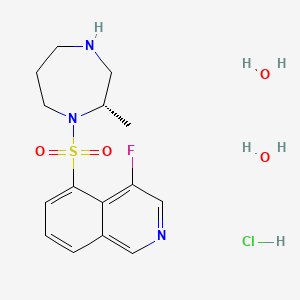
MK 8876

MK 8876
CAS 1426960-33-9
2-(4-Fluorophenyl)-5-(11-fluoro-6H-pyrido[2′,3′:5,6][1,3]oxazino[3,4-a]indol-2-yl)-N-methyl-6-(N-methylmethanesulfonamido)-1-benzofuran-3-carboxamide
| 2-(4-Fluorophenyl)-5-(11-fluoro-6H-pyrido[2′,3′:5,6][1,3]oxazino[3,4-a]indol-2-yl)-N-methyl-6-[methyl(methylsulfonyl)amino]-3-benzofurancarboxamide |
| Molecular Formula | C32H24F2N4O5S | |
| Molecular Weight | 614.62 |
- Originator Merck & Co
- Class Antivirals
- Phase I Hepatitis C
Most Recent Events
- 11 Oct 2013 Phase-I clinical trials in Hepatitis C in Germany (PO)
- 11 Oct 2013 Phase-I clinical trials in Hepatitis C in Moldova (PO)
- 23 Aug 2013 Preclinical trials in Hepatitis C in USA (PO)
DATA
2-(4-Fluorophenyl)-5-(11-fluoro-6H-pyrido[2′,3′:5,6][1,3]oxazino[3,4-a]indol-2-yl)-N-methyl-6-(N-methylmethanesulfonamido)-1-benzofuran-3-carboxamide
MK-8876 off-white solid
1H NMR (500 MHz, DMSO-d6) δ 8.56 (q, J = 4.7 Hz, 1H), 8.06–8.01 (m, 2H), 8.05 (s, 1H), 7.86 (s, 1H), 7.71 (d, J = 8.5 Hz, 1H), 7.62 (d, J = 8.5 Hz, 1H), 7.52 (d, J = 8.3 Hz, 1H), 7.46–7.40 (m, 2H), 7.29–7.22 (m, 1H), 7.11 (s, 1H), 6.94 (dd, J = 10.6, 7.9 Hz, 1H), 6.27 (s, 2H), 3.31 (s, 3H), 2.96 (s, 3H), 2.85 (d, J = 4.7 Hz, 3H);
13C NMR (125.7 MHz, DMSO-d6) δ 162.86, 162.82 (d, JC–F = 248.5 Hz), 155.74 (d, JC–F = 246.1 Hz), 153.80, 152.43, 152.28, 147.20, 137.08, 137.00 (d, JC–F = 10.8 Hz), 136.36, 136.20, 132.37, 129.50 (d, JC–F = 8.6 Hz), 127.17, 125.45 (d, JC–F = 3.1 Hz), 125.08, 125.02, 123.70 (d, JC–F = 7.7 Hz), 122.28, 117.23 (d, JC–F = 22.4 Hz), 116.01 (d, JC–F = 21.9 Hz), 113.65, 111.76, 106.90 (d,JC–F = 3.5 Hz), 105.32 (d, JC–F = 18.5 Hz), 94.16, 73.57, 39.39, 37.24, 26.16;
HR-ESI-MS m/zcalcd for C32H25N4O5SF2+ [M + H]+ 615.1514, found 615.1500.
| HPLC Method | |
|---|---|
| column | Ascentis Express C18 2.7 μm (fused core), 100 mm × 4.6 mm |
| detection | UV at 210 nm |
| column temperature | 40 °C |
| flow rate | 1.8 mL/min |
| injection volume | 5.0 μL |
| gradient | 90% A to 5% A over 11 min, hold at 5% A for 2 min, 5% A back to 90% A over the next 0.1 min, and then hold at 90% A for 2.9 min |
| run time | 16 min |
| data collection | acquisition for the first 13 min |
| mobile phases | solvent A: water with 0.1% H3PO4 |
| solvent B: acetonitrile | |
| Retention Time Data | |
|---|---|
| identity | tR (min) |
| boronic acid 27 | 4.24 |
| desbromoarene 28 | 5.33 |
| MK-8876 (1) | 7.89 |
| chloropyridine starting material 2 | 8.03 |
| BHT | 10.22 |
SYNTHESIS



CONTD……………
MK 8876













MK 8876
Patent
Scheme 1
Scheme 2
Scheme 3
Q
Scheme 4
EXAMPLES
Example 1
Preparation of Compound 1
Step 1 – Synthesis of 2,6-dichloropyridin-3-ol
Η202 (1.60 g, 47.12 mmol) was added slowly to the solution of compound 2,6- dichloropyridin-3-ylboronic acid (3 g, 15.71 mmol) in CH2CI2 (30 mL) at 0 °C. After stirred at room temperature for about 15 hours, the mixture was quenched with sat. Na2S203 aqueous (50 mL) and adjusted to pH < 7 with IN HC1. The mixture was extracted with EtOAc (40 mL x 3). The organic layer was washed with brine (100 mL), dried over Na2S04, filtered and the solvent was evaporated to provide2,6-dichloropyridin-3-ol (2.34 g, yield: 91.4%). 1H-NMR (CDC13, 400 MHz) δ 7.30 (d, / = 8.4 Hz, 1H), 7.19 (d, / = 8.4 Hz, 1H), 5.70 (br, 1H).
– Synthesis of 2,6-dichloro- -methoxypyridine
To a solution of 2,6-dichloropyridin-3-ol (16.3 g, 0.1 mol) and K2C03 (41.4 g, 0.3 mol) in DMF (200 mL) were added Mel (21.3 g, 0.15 mol). The mixture was allowed to stir at 80 °C for 2 hours. The mixture was then diluted with water (200 mL) and extracted with EtOAc (200 mL x 3). The organic layer was washed with brine (200 mL x 3), dried over Na2S04, filtered and the solvent was evaporated to provide 2,6-dichloro-3-methoxypyridine (17.0 g, yield: 96.0%). 1H-NMR (CDC13, 400 MHz) δ 7.12-7.18 (m, 2H), 3.86 (s, 3H). Step 3 – Synthesis of2-(6-chloro-3-methoxypyridin-2-yl)-lH-indole
To a degassed solution of compound 2,6-dichloro-3-methoxypyridine (8.9 g, 0.05 mol), (l-(tert-butoxycarbonyl)-lH-indol-2-yl)boronic acid (13 g, 0.05 mol) and K3PO4 (31.8 g, 3.0 mol) in DMF (100 mL) was added Pd(dppf)Cl2 (3.65 g, 0.005 mol) under N2. The mixture was heated at 60 °C for about 15 hours. The reaction mixture was cooled to room temperature, diluted with EtOAc and filtered. The filtrate was washed with H20, brine, dried over Na2S04. After being concentrated in vacuo, the resulting residue was purified using prep-HPLC to provide the desired product of 2-(6-chloro-3-methoxypyridin-2-yl)-lH-indole (9.0 g, yield:
69.8%). 1H-NMR (CDC13, 400 MHz) δ 9.52 (s, 1H), 7.65 (d, / = 7.6 Hz, 1H), 7.38-7.43 (m, 2H), 7.07-7.26 (m, 4H), 4.03 (s, 3H).
Step 4 – Synthesis of6-chlor -2-(lH-indol-2-yl)pyridin-3-ol
BBr3 (0.4 mL, 0.39 mmol) was added to the solution of 2-(6-chloro-3- methoxypyridin-2-yl)-lH-indole (50 mg, 0.194 mmol) in CH2C12 (0.5 mL) at -78 °C under N2. The mixture was allowed to stir at room temperature for 3 hours. The mixture was then quenched with CH3OH (10 mL) at -78 °C. After being concentrated in vacuo, the resulting residue was purified using prep-TLC (PE : EtOAc = 2.5 : 1) to afford the desired product of 6- chloro-2-(lH-indol-2-yl)pyridin-3-ol (40 mg, yield: 85.1%). 1H-NMR (CDC13, 400 MHz) δ 10.09 (s, 1H), 9.72 (s, 1H), 7.50 (d, / = 7.9 Hz, 1H), 7.17-7.32 (m, 3H), 7.08-7.14 (m, 1H), 6.87-6.96 (m, 2H).
Step 5 – Synthesis of 2-chlo -6H-pyrido[2′ ,3′ : 5 ,6] [ 1 ,3]oxazino[3 ,4-a]indole
To a solution of chloroiodomethane (3.51 g, 20.0 mmol) and K2CO3 (1.38 g, 10.0 mmol) in DMF (50 mL) was allowed to stir at 100 °C, 6-chloro-2-(lH-indol-2-yl)pyridin-3-ol (480 mg, 2.0 mmol) in DMF (50 mL) was added dropwise. After addition, the mixture was allowed to stir for another 0.5 hours. The mixture was then diluted with water (100 mL) and extracted with EtOAc (100 mL x 3). The organic layer was washed with brine (100 mL x 3), dried over Na2S04 and concentrated. The residue was purified using prep-TLC (PE : EtOAc = 3 1) to afford the desired product of 2-chloro-6H-pyrido[2′,3′:5,6][l,3]oxazino[3,4-a]indole (260 mg, yield: 50.7%). 1H-NMR (CDC13, 400 MHz) δ 7.63 (d, / = 8.0 Hz, 1H), 7.22-7.27 (m, 3H), 7.19 (d, / = 2.4 Hz, 1H), 7.08-7.12 (m, 2H), 5.86 (s, 2H).
Step 6 – Synthesis of2-(4-fluowphenyl)-N-methyl-6-(N-methylmethylsulfonamido)-5-(6H- pyridol 2 ‘,3’:5,6][ l, mpound 1 )
To a degassed solution of 2-(4-fluorophenyl)-N-methyl-6-(N- methylmethylsulfonamido)-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)benzofuran-3- carboxamide (502 mg, 1.0 mmol), 2-chloro-6H-pyrido[2′,3′:5,6][l,3]oxazino[3,4-a]indole (256 mg, 1.0 mmol) and K3PO4 (636 mg, 3.0 mmol) in dioxane : H20 (1.5 mL : 0.4 mL) was added Pd2(dba)3 (91 mg, 0.1 mmol) and X-phos (91 mg, 0.2 mmol) under N2. The mixture was heated to 110 °C for 3 hours. The reaction mixture was cooled to room temperature, diluted with EtOAc and filtered. The filtrate was washed with H20, brine, dried over Na2S04. After being concentrated in vacuo, the resulting residue was purified using prep-HPLC to provide the desired product of Compound 1 (275 mg, yield: 46.1%). 1H-NMR (CDC13, 400 MHz) δ 7.88-7.94 (m, 3H), 7.61-7.63 (m, 2H), 7.40 (s, 2H), 7.09-7.28 (m, 6H), 5.94 (s, 2H), 5.86 (d, / = 4.4 Hz, 1H), 3.29 (s, 3H), 2.92 (d, / = 5.2 Hz, 3H), 2.65 (s, 3H). MS (M+H)+: 596.
Compounds 2-15, depicted in the table below, were prepared using the method described above.
COMPD 2 IS MK 8876

PATENT
Example 81
Preparation of Compound 2
Synthesis of ethyl 3- 4-fluorophenyl)-3-oxopropanoate
Diethyl carbonate (130 g, 1.1 mol) was dissolved in a suspension ofNaH (60% in oil, 50.2 g, 1.3 mol) in anhydrous tetrahydrofuran (1.5 L), and then l-(4-fluorophenyl)ethanone (150 g, 1.09 mol) was added dropwise at 70 °C. The resulting mixture was stirred at 70 °C for 3 hours. After the reaction mixture was cooled to room temperature and poured into HCl (1 N). The mixture was extracted with EtOAc, the organic phase was dried with anhydrous NaS04 and concentrated in vacuo. The resulting residue was purified using column chromatography (eluted with petroleum ether / EtOAc = 50 / 1) to provide ethyl 3-(4-fluorophenyl)-3-oxopropanoate (217 g, yield: 95%). 1H-NMR (CDC13, 400 MHz) δ 7.92-7.97 (m, 2H), 7.07-7.13 (m, 2H), 4.14-4.20 (m, 2H), 3.93 (s, 2H), 1.22 (d, J= 7.2 Hz, 3H). MS (M+H)+: 211. Step 2 – Synthesis of ethyl 5-bromo-2-(4-fluorophenyl)benzofuran-3-carboxylate
A solution of ethyl 3-(4-fluorophenyl)-3-oxopropanoate (130 g, 0.6 mol), 4- bromophenol (311 g, 1.8 mol) and FeCl3-6H20 (19.5 g, 0.09 mol) in DCE (700 mL) was heated to reflux, and then 2-(tert-butylperoxy)-2-methylpropane (193 g, 1.32 mol) was added dropwise under nitrogen. After 6 hours of refluxing, the mixture was cooled to RT, quenched with saturated NaHS03 and extracted with dichloromethane. The organic phases were washed with water, brine and dried over Na2S04, filtered and concentrated in vacuo. The resulting residue was purified using column chromatography (petroleum ether / dichloromethane = 15 / 1) to provide the crude product, which was crystallized from cold MeOH to provde ethyl 5-bromo-2- (4-fluorophenyl)benzofuran-3-carboxylate (37 g, yield: 14.3%) as solid. 1H- MR (CDC13, 400 MHz) δ 8.12 (s, 1H), 7.97-8.01 (m, 2H), 7.37 (d, J= 4.0 Hz, 1H), 7.32 (d, J= 8.0 Hz, 1H), 7.11 (t, J= 8.0 Hz, 2H), 4.32-4.38 (m, 2H), 1.36 (t, J= 8.0 Hz, 3H). MS (M+H)+: 363 / 365.
Step 3 – Synthesis of eth l 5-bromo-2-(4-fluorophen -6-nitrobenzofuran-3-carboxylate
To a solution of ethyl 5-bromo-2-(4-fluorophenyl)benzofuran-3-carboxylate (50 g,
137.6 mmol) in CHC13 (500 mL), fuming HN03 (50 mL) was added dropwise at -15 °C and the mixture was stirred for 0.5 hour. The reaction mixture was poured into ice water and extracted with CH2C12. The organic layer was washed with a.q. sat. NaHC03 and brine, after removed the most of solvent, the resulting residue was crystallized with petroleum ether / dichloromethane = 20 / 1 to provide product of ethyl 5-bromo-2-(4-fluorophenyl)-6-nitrobenzofuran-3-carboxylate (35 g, yield: 66%). 1H- MR (CDC13, 400 MHz) δ 8.36 (s, 1H), 8.02-8.04 (m, 3H), 7.13-7.18 (m, 2H), 4.36-4.41 (m, 2H), 1.37 (t, J= 4.0 Hz, 3H). MS (M+H)+: 408 / 410.
Step 4 – Synthesis of ethyl 6-amino-5-bromo-2-(4-fluorophenyl)benzofuran-3-carboxylate
A mixture of ethyl 5-bromo-2-(4-fluorophenyl)-6-nitrobenzofuran-3-carboxylate (52 g, 127 mmol), iron filings (21.3 g, 382.2 mmol) and H4C1 (41 g, 764.4 mmol) in MeOH / THF / H20 (2 / 2 / 1, 500 mL) was stirred at reflux for 3 hour. After filtered and concentrated, the resulting residue was purified using column chromatography (petroleum ether / EtOAc / dichloromethane = 20 : 1 : 20) to provide ethyl 6-amino-5-bromo-2-(4-fluorophenyl) benzofuran-3-carboxylate (40 g, yield: 82%). 1H- MR (CDC13, 400 MHz) δ 8.01 (s, 1H), 7.94-7.98 (m, 2H), 7.08 (t, J= 8.0 Hz, 2H), 6.83 (s, 1H), 4.32-4.36 (m, 2H), 4.18 (s, 2H), 1.35 (t, J= 8.0 Hz, 3H). MS (M+H)+: 378 / 380.
Step 5 – Synthesis of 5-Bromo-2-(4-fluoro-phenyl)-6-methanesulfonylamino-benzofuran-3- carboxylic acid eth l ester
MsCI (31.7 g, 277.5 mmol) was added to a solution of ethyl 6-amino-5-bromo-2- (4-fluorophenyl)benzofuran-3-carboxylate (35 g, 92.5 mmol) and pyridine (60 mL) in
dichloromethane (300 mL) at 0 °C. After stirred overnight at room temperature, the mixture was diluted with water and extracted with dichloromethane. The organic layer was washed with brine, dried over Na2S04, filtered and concentrated in vacuo, the resulting residue was purified using crystallized with EtOAc to provde the pure product of ethyl 5-bromo-2-(4-fluorophenyl)-6- (methylsulfonamido)benzofuran-3-carboxylate (35 g, yield: 82%). 1H- MR (CDC13, 400 MHz) δ 8.27 (s, 1H), 8.01-8.05 (m, 2H), 7.87 (s, 1H), 7.15-7.19 (m, 2H), 6.87 (s, 1H), 4.38-4.43 (m, 2H), 3.00 (s, 3H), 1.40 (t, J= 40 Hz, 3H). MS (M+H)+: 456 / 458.
Step 6 – Synthesis of 5-Bromo-2-(4-fluoro-phenyl)-6-methanesulfonylamino-benzofuran-3- carboxylic acid
To a solution of ethyl 5-bromo-2-(4-fluorophenyl)-6-(methylsulfonamido) benzofuran-3-carboxylate (53 g, 0.23 mol) in dioxane / H20 (5 / 1, 600 mL) was added
LiOH-H20 (25 g, 1.17 mol), and the mixture was stirred at 100 °C for 3 hours. After
concentrated, the resulting residue was dissolved in H20, 1 N HCl was added until pH reached 3, and the mixture was extracted with EtOAc. The organic layer was washed with brine, dried over Na2S04 and filtered. The solvent was removed to provide the product of 5-bromo-2-(4- fluorophenyl)-6-(methylsulfonamido)benzofuran-3-carboxylic acid (48 g, yield: 96%).1H- MR (DMSO- e, 400 MHz) δ 13.49 (s, 1H), 9.67 (s, 1H), 8.30 (s, 1H), 8.12-8.17 (m, 2H), 7.87 (s, 1H), 7.45-7.50 (m, 2H), 3.16 (s, 3H). MS (M+H)+: 428 / 430. Step 7 – Synthesis of 5-Bromo-2-(4-fluoro-phenyl)-6-methanesulfonylamino-benzofuran-3- carboxylic acid methylamide
A solution of 5-bromo-2-(4-fluorophenyl)-6-(methylsulfonamido) benzofuran-3- carboxylic acid (33 g, 77 mmol), HOBT (15.6 g, 115.5 mmol) and EDCI (22.2 g, 115.5 mmol) in DMF (250 mL) was stirred at room temperature. After 2 hours, Et3N (50 mL) and CH3 H2 (HC1 salt, 17.7 g, 231 mmol) was added to the mixture, and the mixture was stirred overnight. After the solvent was removed, H20 was added and the mixture was extracted with ethyl acetate. The combined organic layer was washed with H20, brine and concentrated in vacuo. The resulting residue was washed with EtOAc to provide the product of 5-bromo-2-(4-fluorophenyl)-N- methyl-6-(methylsulfonamido)benzofuran-3-carboxamide (32 g, yield: 94%). 1H- MR (DMSO- ck, 400 MHz) δ 9.55 (br s, 1H), 8.46-8.48 (m, 1H), 8.12-8.17 (m, 2H), 7.96 (s, 1H), 7.87 (s, 1H), 7.45-7.50 (m, 2H), 3.16 (s, 3H), 2.93 (d, J= 8.4 Hz, 3H). MS (M+H)+: 441 / 443.
Step 8 – Synthesis of 5-bromo-2-(4-fluorophenyl)-N-methyl-6-(N- methylmethylsulfonamido benzofuran-3-carboxamide
CH3I (31.6 g, 223 mmol) was added to a mixture of 5-bromo-2-(4-fluorophenyl)- N-methyl-6-(methylsulfonamido)benzofuran-3-carboxamide (32 g, 74 mmol), K2C03 (25.6 g, 186 mmol) and KI (246 mg, 1.5 mmol) in DMF (150 mL) under N2 protection. The mixture was stirred at 80-90 °C overnight. After concentrated in vacuo, the resulting residue was washed with water (200 mL) and EtOAc (200 mL) to provide the product of 5-bromo-2-(4- fluorophenyl)-N-methyl-6-(N-methylmethylsulfonamido)benzofuran-3-carboxamide (31.5 g, 94%). 1H- MR (CDCI3, 400 MHz) δ 8.16 (s, 1H), 7.88-7.92 (m, 2H), 7.70 (s, 1H), 7.18-7.23 (m, 2H), 5.78 (br s, 1H), 3.34 (s, 3H), 3.09 (s, 3H), 3.00 (d, J= 4.8 Hz, 3H). MS (M+H)+: 455 / 457. Step 9 – Synthesis of 2-(4-fluorophenyl)-N-methyl-6-(N-methylmethylsulfonamido)-5-(4, 4, 5, 5- tetramethyl-1 -dioxaborolan-2-yl)benzofuran-3-carboxamide
a degassed solution of 5-bromo-2-(4-fluorophenyl)-N-methyl-6-(N- methylmethylsulfonamido)benzofuran-3-carboxamide (1.0 g, 2.2 mmol) and pinacol diborane (2.79 g, 11.0 mmol) in 1,4-Dioxane (25 mL) was added KOAc (647 mg, 6.6 mmol) under N2 and stirred for 4 hours at room temperature. Then Pd(dppf)Cl2 (60 mg) was added, and the mixture was stirred for another 30 minutes. Then the mixture was put into a pre-heated oil-bath at 130 °C and stirred for another 1 hour under N2. The reaction mixture was cooled to room
temperatureand concentrated and extracted with EtOAc. The organic layers were washed with brine, dried over Na2S04. After concentrated, the crude product of the boronic ester was purified using column chromatography (petroleum ether / EtOAc = 5 / 1 to 2 / 1) to obtain 2-(4- fluorophenyl)-N-methyl-6-(N-methylmethylsulfonamido)-5-(4,4,5,5-tetramethyl-l,3,2- dioxaborolan-2-yl)benzofuran-3-carboxamide as white solid (700 mg, yield: 64%). 1H- MR (CDCI3, 400 ΜΗζ) δ 8.17 (s, 1H), 7.87-7.91 (m, 2H), 7.52 (s, 1H), 7.11 (t, 7= 7.6 Hz, 2H), 5.81 (d, 7= 2.8 Hz, 1H), 3.30 (s, 3H), 2.97 (d, 7= 5.2 Hz, 3H), 2.90 (s, 3H), 1.31 (s, 12H). MS (M+H)+: 503.
Step 10 – Synthesis of tert-butyl 4-fluoro-lH-indole-l -car boxy late
To a solution of 4-fluoro-lH-indole (5 g, 0.11 mol) and DMAP (150 mg, 3%Wt) in THF (50 mL) was added (Boc)20 (8.5 g, 0.04 mol) dropwise. The mixture was stirred at room temperature for 2 hours. The organic solvent was removed in vacuo, and the resulting residue was purified using column chromatography (pure petroleum ether) to provide tert-butyl 4-fluoro- lH-indole-l-carboxylate (8.3 g, yield: 96%). 1H- MR (CDC13, 400 MHz) δ 7.92 (d, J= 8.4 Hz, 1H), 7.55 (d, J= 3.6 Hz, 1H), 7.23 (m, 1H), 6.90 (m, 1H), 6.66 (d, J= 3.6 Hz, 1H), 1.67 (s, 9H). MS (M+H)+: 236.
Step 11 – Synthesis of (l-(tert-butoxycarbonyl)-4-fluoro-lH-indol-2-yl)boronic acid
To a solution of diisopropylamine (7.5 mL, 0.11 mol) in THF (35 mL) at 0 °C was added «-BuLi (21 mL, 0.055 mol) dropwise. The mixture was stirred at 0 °C for 40 minutes. Then the mixture was cooled to -78 °C. Tert-butyl 4-fluoro-lH-indole-l-carboxylate (5 g, 0.02 mol) in THF (13 mL) was added dropwise slowly. After addition, the mixture was stirred at -78 °C for 2 hours. Then triisopropyl borate (3.29 g, 0.03 mol) was added. The mixture was stirred at -78 °C for another 40 minutes. The reaction was monitored using TLC. When the reaction was completed, the mixture was adjusted to pH = 6 with 1 N HC1. After extracted with EtOAc (25 mL x 3), the combined organic layers were washed with brine (50 mL), dried over Na2S04, filtered and concentrated in vacuo. The obtained solid was recrystallized with EtOAc and petroleum ether to provide (l-(tert-butoxycarbonyl)-4-fluoro-lH-indol-2-yl)boronic acid (4.5 g, yield: 76.7%, which might be unstable at high temp, work up, store in fridge). 1H- MR (CDC13, 400 MHz) δ 7.77 (d, J= 8.4 Hz, 1H), 7.57 (s, 1H), 7.44 (s, 2H), 7.24 (m, 1H), 6.90 (m, 1H), 1.66 (s, 9H). MS (M+H)+: 280.
Step 12 – Synthesis of 6-chloro-2-iodopyridin-3-ol
6-chloropyridin-3-ol (5.0 g, 38.6 mmol) was dissolved in water (50 mL) and placed under an N2 atmosphere. Na2C03 (8.2 g, 77.4 mmol) was added followed by iodine (9.8 g, 38.8 mmol). The reaction mixture was stirred at room temperature for 2 hours. The mixture was poured into 1M Na2S203 and extracted with EtOAc. The combined organic phases were washed with brine, dried over Na2S04 and concentrated to provide the product of 6-chloro-2- iodopyridin-3-ol (7.0 g, yield: 70.9%). 1H- MR (CDC13, 400 MHz) δ 7.17 (d, J= 8.4 Hz, 1H), 7.06 (d, J= 8.4 Hz, 1H). MS (M+H)+: 256 / 258.
Step 13 – Synthesis of 6-chloro-2-(4-fluoro-lH-indol-2-yl)pyridin-3-ol
A mixture of (l-(tert-butoxycarbonyl)-4-fluoro-lH-indol-2-yl)boronic acid (5 g, 18.0 mmol), 6-chloro-2-iodopyridin-3-ol (3.82 g, 15.0 mol) and NaHC03 (3.78 g, 45.0 mol) in 1, 4-dioxane (76 mL) and water (7 mL) was stirred at room temperature for 15 minutes. Then Pd(PPh3)2Cl2 (527 mg, 0.75 mmol) was added under nitrogen atmosphere, and the mixture was heated at 100 °C under N2 for 16 hours. The reaction mixture was cooled to room temperature, diluted with EtOAc (50 mL), filtered and concentrated in vacuo. The resulting residue was diluted with H20 (60 mL) and EtOAc (30 mL), and the layer was separated, the aqueous layer was extracted with EtOAc (3*30 mL). The combined organic layers were washed with brine (50 mL), dried over Na2S04, filtered and concentrated in vacuo. The resulting residue was purified using column chromatography (petroleum ether / EtOAc = 20 / 1 ~ 3 / 1) to provide 6-chloro-2- (4-fluoro-lH-indol-2-yl)pyridin-3-ol (3 g, yield: 76.5%). 1H- MR (MeOD, 400 MHz) δ 7.36 (s, 1H), 7.23-7.27 (m, 2H), 7.03-7.11 (m, 2H), 6.63-6.68 (m, 1H). MS (M+H)+: 263 / 265.
Ste 14 – Synthesis of 2-chloro-ll-fluoro-6H-pyrido[2′,3′:5, 6][l,3]oxazino[3,4-a]indole
A solution of 6-chloro-2-(4-fluoro-lH-indol-2-yl)pyridin-3-ol (2 g, 7.6 mmol) and Cs2C03 (7.46 g, 22.89 mmol) in DMF (100 mL) was stirred at 100 °C (internal temperature) for 15 min, and then chloroiodomethane (2.85 g, 15.3 mmol) in DMF (2 mL) was added dropwise. After the reaction was completed, the mixture was filtered and concentrated in vacuo. The resulting residue was diluted with water (50 mL) and extracted with ethyl acetate (30 mL x 3). The organic layer was washed with brine, dried over Na2S04 and concentrated in vacuo. The resulting residue was purified using column chromatography (petroleum ether:EA=10: l) to provde 2-chloro-l l-fluoro-6H-pyrido[2′,3′:5,6][l,3]oxazino[3,4-a]indole (1.8 g, yield: 86.1%). 1H- MR (DMSO-i¾, 400 MHz) δ 7.64 (d, J= 8.8 Hz, 1H), 7.39-7.46 (m, 2H), 7.21-7.25 (m, 1H), 7.06 (s, 1H), 6.88-6.92 (m, 1H), 6.18 (s, 2H). MS (M+H)+: 275 / 277. Step 15 – Synthesis of5-(ll-fluoro-6H-pyrido[2 3′:5, 6][l,3]oxazino[3,4-a]indol-2-yl)-2-(4- fluorophenyl)-N-methyl-6-(N-methylmethylsulfonamido)benzofuran-3-carboxam
To a degassed solution of 2-(4-fluorophenyl)-N-methyl-6-(N- methylmethylsulfonamido)-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)benzofuran-3- carboxamide (100 mg, 0.199 mmol), 2-chloro-l l-fluoro-6H-pyrido[2′,3′:5,6][l,3]oxazino[3,4- a]indole (56 mg, 0.199 mmol) and Κ3Ρ04·3Η20 (159 mg, 0.597 mmol) in dioxane / H20 (0.8 mL / 0.2 mL) was added Pd2(dba)3 (9 mg, 0.01 mmol) and X-Phos (9 mg, 0.02 mmol) under N2. The mixture was heated at 80 °C for 1 hour. The mixture was then diluted with water (30 mL) and extracted with EtOAc (15 mL x 3). The organic layer was washed with brine (20 mL), dried over Na2S04 and concentrated in vacuo. The resulting residue was purified using prep-TLC (petroleum ether / EtOAc = 1 : 1.5) to provde the pure product of 5-(l l-fluoro-6H- pyrido [2′, 3 ‘ : 5 , 6] [ 1 , 3 ]oxazino [3 ,4-a]indol-2-yl)-2-(4-fluorophenyl)-N-methyl-6-(N- methylmethylsulfonamido)benzofuran-3-carboxamide (60 mg, 48.8%). 1H- MR (CDC13, 400 MHz) δ: 7.99 (s, 1H), 7.93-7.96 (m, 2H), 7.65 (s, 1H), 7.45-7.50 (m, 2H), 7.17-7.21 (m, 4H), 7.10 (d, J= 8.0 Hz, 1H), 6.81-6.85 (m, 1H), 5.98 (s, 3H), 3.35 (s, 3H), 2.98 (d, J= 4.8 Hz, 3H), 2.72 (s, 3H). MS (M+H)+: 615.
Paper

We describe the route development and multikilogram-scale synthesis of an HCV NS5B site D inhibitor, MK-8876. The key topics covered are (1) process improvement of the two main fragments; (2) optimization of the initially troublesome penultimate step, a key bis(boronic acid) (BBA)-based borylation; (3) process development of the final Suzuki–Miyaura coupling; and (4) control of the drug substance form. These efforts culminated in a 28 kg delivery of the desired active pharmaceutical ingredient.
Process Development of the HCV NS5B Site D Inhibitor MK-8876
*E-mail: qinghao.chen@merck.com
http://pubs.acs.org/doi/abs/10.1021/acs.oprd.5b00405
PAPER

Using the Teasdale method, purge factor estimates for six impurities identified as mutagenic alerts in the synthesis of MK-8876 are compared to actual measured amounts of these impurities determined via appropriate analytical methods. The results from this comparison illustrate the conservative nature of purge factor estimates, meaning that overprediction of mutagenic impurity purging is unlikely when using this method. Industry and regulatory acceptance of the purge factor estimation method may help minimize analytical burden in pharmaceutical development projects.
Evaluation and Control of Mutagenic Impurities in a Development Compound: Purge Factor Estimates vs Measured Amounts
*E-mail: mark_mclaughlin@merck.com.
http://pubs.acs.org/doi/abs/10.1021/acs.oprd.5b00263?journalCode=oprdfk
| WO2004041201A2 * | Oct 31, 2003 | May 21, 2004 | Viropharma Incorporated | Benzofuran compounds, compositions and methods for treatment and prophylaxis of hepatitis c viral infections and associated diseases |
| WO2011106992A1 * | Mar 2, 2011 | Sep 9, 2011 | Merck Sharp & Dohme Corp. | Inhibitors of hepatitis c virus ns5b polymerase |
| WO2004041201A2 * | Oct 31, 2003 | May 21, 2004 | Viropharma Incorporated | Benzofuran compounds, compositions and methods for treatment and prophylaxis of hepatitis c viral infections and associated diseases |
| WO2010030592A1 * | Sep 8, 2009 | Mar 18, 2010 | Bristol-Myers Squibb Company | Compounds for the treatment of hepatitis c |
| WO2011106992A1 * | Mar 2, 2011 | Sep 9, 2011 | Merck Sharp & Dohme Corp. | Inhibitors of hepatitis c virus ns5b polymerase |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| WO2014123794A1 * | Feb 3, 2014 | Aug 14, 2014 | Merck Sharp & Dohme Corp. | Tetracyclic heterocycle compounds and methods of use thereof for the treatment of hepatitis c |
| WO2014123795A2 * | Feb 3, 2014 | Aug 14, 2014 | Merck Sharp & Dohme Corp. | Tetracyclic heterocycle compounds and methods of use thereof for the treatment of hepatitis c |
| WO2014123795A3 * | Feb 3, 2014 | Oct 30, 2014 | Merck Sharp & Dohme Corp. | Tetracyclic heterocycle compounds and methods of use thereof for the treatment of hepatitis c |
| US9242998 | Feb 3, 2014 | Jan 26, 2016 | Merck Sharp & Dohme Corp. | Tetracyclic heterocycle compounds and methods of use thereof for the treatment of hepatitis C |
//////MK-8876, 1426960-33-9, Merck & Co, Antivirals, Phase I, Hepatitis C
Fc7cccc6c7cc2n6COc1ccc(nc12)c3cc4c(cc3N(C)S(C)(=O)=O)oc(c4C(=O)NC)c5ccc(F)cc5
BRISTOL-MYERS SQUIBB’S TRICYCLOHEXADECAHEXAENE DERIVATIVES FOR USE IN THE TREATMENT OF HEPATITIS C VIRUS
 .
.
TRICYCLOHEXADECAHEXAENE DERIVATIVES FOR USE IN THE TREATMENT OF HEPATITIS C VIRUS
WO2015026454, COMBINATIONS COMPRISING TRICYCLOHEXADECAHEXAENE DERIVATIVES FOR USE IN THE TREATMENT OF HEPATITIS C VIRUS
BRISTOL-MYERS SQUIBB COMPANY [US/US]; Route 206 and Province Line Road Princeton, New Jersey 08543 (US)
PATENT WO2015026454 [LINK]
WANG, Alan Xiangdong; (US).
LOPEZ, Omar D.; (US).
TU, Yong; (US).
BELEMA, Makonen; (US)
Example B-l

Example B-l Step a

To a solution of 4-bromobenzene-l,2-diamine (2.5 g, 13.37 mmol) in DCM (30 mL) was added (S)-2-((tert-butoxycarbonyl)amino)-3,3-dimethylbutanoic acid (3.09 g, 13.37 mmol), DIPEA (2.334 mL, 13.37 mmol) and HATU (5.08 g, 13.37 mmol). The reaction mixture was stirred at room temperature for 18 h. The reaction mixture was diluted with water and extracted with DCM. The organic phase was washed with brine, dried over Na2S04, filtered and concentrated. The crude material was purified by ISCO using 40 g Redisep silica column, CHCl3/MeOH as eluant to obtain (S)-tert-butyl ( 1 -((2-amino-4-bromophenyl)amino)-3 ,3 -dimethyl- 1 -oxobutan-2-yl) carbamate (1.82 g) as yellow solid. LC (Condition 1): Rt = 2.13 min. LC/MS: Anal. Calcd. for [M+H20]+ Ci7H27BrN204 : 402.12; found 402.2. 1H NMR (DMSO-d6, δ = 2.50 ppm, 400 MHz): δ 9.35 – 9.21 (m, 1 H), 7.07 (d, J= 8.5 Hz, 1 H), 6.91 (d, J= 2.0 Hz, 1 H), 6.80 – 6.60 (m, 1 H), 5.25 – 5.01 (m, 2 H), 4.07 – 3.89 (m, 1 H), 1.52 – 1.34 (m, 9 H), 1.02 – 0.86 (m, 9 H).
Example B-l, Step b

Acetic acid (15 mL) was added to (S)-tert-butyl (l-((2-amino-4-bromo phenyl)amino)-3,3-dimethyl-l-oxobutan-2-yl)carbamate (1.8 g, 4.50 mmol) and the reaction mixture was heated to 65 °C for overnight. The volatile component was removed in vacuo, and the residue was co-evaporated with dry CH2C12 (2 x 15 mL). The organic phase was washed with saturated NaHC03 solution, brine, dried over Na2S04 and concentrated to obtain (S)-tert-butyl (l-(6-bromo-lH-benzo[d] imidazol-2-yl)-2,2-dimethyl propyl)carbamate (1.68 g) as yellow solid. LC (Condition 1): Rt = 2.19 min. LC/MS: Anal. Calcd. for [M+H]+ Ci7H25BrN302 : 381.11; found 382.2. 1H NMR (DMSO-dg, δ = 2.50 ppm, 300 MHz): δ 12.46 – 12.27 (m, 1 H), 7.82 – 7.65 (m, 1 H), 7.59 – 7.41 (m, 1 H), 7.29 (dt, J= 1.9, 8.5 Hz, 1 H), 7.12 – 6.90 (m, 1 H), 4.64 (d, J= 9.8 Hz, 1 H), 1.44 – 1.27 (m, 9 H), 0.88 (br. s., 9 H).
-1 Step c

To a solution of (S)-tert-butyl (l-(6-bromo-lH-benzo[d]imidazol-2-yl)-2,2-dimethyl propyl) carbamate (1.57 g, 4.11 mmol) in dioxane (25 mL) was added bis (pinacolato)diboron (1.564 g, 6.16 mmol) and potassium acetate (1.209 g, 12.32 mmol). The reaction mixture was purged with argon for 10 min then PdCl2(dppf) (0.150 g, 0.205 mmol) was added to the above reaction mixture and again purged with argon for 5 min. The reaction mixture was heated to 90 °C for overnight. The reaction mixture was diluted with water (15 ml) and extracted with EtOAc (2 x 25 ml). The combined organic phase was washed with brine, dried over Na2S04 and concentrated in vacuo. The crude material was purified by ISCO using 40 g Redisep column, hexane/ethyl acetate as eluant to afford (S)-tert-butyl (2,2-dimethyl-l-(6-(4,4,5 ,5-tetramethyl- 1 ,3 ,2-dioxaborolan-2-yl)- 1 H-benzo[d]imidazol-2-yl)propyl) carbamate (1.35 g) as yellow solid. LC (Condition 1): Rt = 2.21 min. LC/MS: Anal. Calcd. for [M+H]+ C23H37BN304 : 430.29; found 430.4. 1H NMR (CD3OD, δ = 3.34 ppm, 400 MHz): δ 7.98 (s, 1 H), 7.65 (dd, J= 1.0, 8.5 Hz, 1 H), 7.53(d, J= 8.5 Hz, 1 H), 4.73 (br. s., 1 H), 1.37 (s, 12 H), 1.24 (m, 9 H), 1.01 (s, 9 H).
-1 Step d

To a solution of (S)-tert-butyl (2,2-dimethyl-l-(6-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)-lH-benzo[d]imidazol-2-yl)propyl)carbamate (1.114 g, 2.59 mmol) and 4,16-dibromo[2,2]paracyclophane (0.38g, 1.038 mmol) in dioxane (10 mL) was added Cs2C03 (0.845 g, 2.59 mmol) in water (2 mL) and degassed for 10 min.
PdCl2(dppf) (0.038 g, 0.052 mmol) was added to the above reaction mixture and again degassed for 5 min. The reaction mixture was heated to 90 °C for 12 h. Then the reaction mixture was filtered to get Example B-1 Step d which was taken for next step without further purification. LC (Condition 1): Rt = 2.54 min. LC/MS: Anal. Calcd. for [M+H]+ ^0Η63Ν6Ο4 : 811.49; found 811.6. 1H NMR (DMSO-d6, δ = 2.50 ppm, 300 MHz): δ 12.36 (br. s., 2 H), 7.85 – 7.52 (m, 4 H), 7.32 (d, J= 7.9 Hz, 2 H), 7.05 (br. s., 2 H), 6.89 – 6.67 (m, 4 H), 6.54 (br. s., 2 H), 4.72 (d, J= 8.7 Hz, 2 H), 3.57 – 3.44 (m, 2 H), 3.07 (br. s., 2 H), 2.83 (br. s., 2 H), 2.65 (br. s., 2 H), 1.36 (s, 18 H), 1.08 – 0.91 (m, 18 H).
-1 Step e

HC1 in dioxane (4 mL, 24.00 mmol) was added to Example B-1 Step d (0.1 g,
0.102 mmol), and the reaction mixture was allowed to stir at RT for 2 h. Completion of the reaction was monitored by LCMS. The volatile component was removed in vacuo and the residue was washed with diethyl ether and dried to afford Example B-1 Step e (0.07 g) as yellow solid. LC (Condition 1): R, = 2.54 min. LC/MS: Anal.
Calcd. for [M+H]+ C40H47N6 : 611.39; found 611.4. 1H NMR (CD3OD, δ = 3.34 ppm, 400 MHz): δ 7.90 (d, J= 13.1 Hz, 2 H), 7.83 (d, J= 8.5 Hz, 2 H), 7.61 (d, J= 8.5 Hz, 2 H), 6.84 (d, J= 6.5 Hz, 2 H), 6.78 (s, 2 H), 6.70 – 6.65 (m, 2 H), 4.54 (d, J= 1.0 Hz, 2 H), 3.54 – 3.46 (m, 2 H), 3.18 – 3.10 (m, 2 H), 2.98 – 2.86 (m, 2 H), 2.71 (br. s., 2 H), 1.25 – 1.22 (m, 18 H).
To a solution of Example B-1 Step e (0.04 g, 0.053 mmol) in DMF (5 mL) was added 4,4-difluorocyclohexanecarboxylic acid (0.017 g, 0.106 mmol), DIPEA (0.055 mL, 0.317 mmol) and HATU (0.030 g, 0.079 mmol). After being stirred for 2 h at room temperature, the volatile component was removed in vacuo and the residue was dissolved in DCM (10 mL), washed with saturated solution of NH4C1, 10% NaHC03 solution, brine, dried over Na2S04 and concentrated in vacuo. The crude was purified by reverse phase HPLC purification to give Example B-1 as a white solid. LC (Condition 1): R, = 2.37 min. LC/MS: Anal. Calcd. for [M+H]+
C54H63F4N602: 903.49; found 903.4. 1H NMR (DMSO-d6, δ = 2.50 ppm, 400 MHz): δ 12.53 – 12.32 (m, 2 H), 8.41 – 8.21 (m, 2 H), 7.84 – 7.50 (m, 4 H), 7.43 – 7.24 (m, 2 H), 6.90 – 6.67 (m, 4 H), 6.60 – 6.44 (m, 2 H), 5.14 – 4.97 (m, 2 H), 3.44 (br. s., 2 H), 3.08 (br. s., 2 H), 2.93 – 2.77 (m, 2 H), 2.73 – 2.56 (m, 4 H), 2.20 – 1.98 (m, 3 H), 1.96 – 1.49 (m, 13 H), 1.02 (s, 18 H).
Starting materials can be obtained from commercial sources or prepared by well-established literature methods known to those of ordinary skill in the art. Acid precursors for the final step can be prepared according to the methods described in U.S. Patent Application Serial No. 13/933495, filed July 2, 2013.
LC/MS Condition 1
Column = Ascentis Express C18, 2.1 X 50 mm, 2.7 um
Solvent A = CH3CN (2%) + 10 mM NH4COOH in H20 (98%)
Solvent B = CH3CN (98%) + 10 mM NH4COOH in H20 (2%)
Start %B = 0; Final %B = 100
Gradient time = 1.4 min; Stop time = 4 min
Stop time = 4 min
Flow Rate = 1 mL/min; Wavelength = 220 nm
LC/MS Condition 2
Column = Waters BEH CI 8, 2.0 x 50 mm, 1.7 μιη
Slovent A = ACN (5%) + H20 (95%) containing 10 mM NH4OAc
Solvent B = ACN (95%) + H20 (5%) containing 10 mM NH4OAc
Start %B = 0; Final %B = 100
Gradient time = 3 min
Flow Rate = 1 mL/min
Wavelength = 220 nm
Temperature = 50 °C
LC/MS Condition 3
Column: Waters Phenomenex CI 8, 2.0 x 30 mm, 3 μιη particle
Mobile Phase A: 10% MeOH:90% Water :0.1%TFA
Mobile Phase B: 90% MeOH: 10% Water :0.1%TFA
Gradient: 0%B, 0-100% B over 3 minutes, then a 1 -minute hold at 100% B Flow: 0.8mL/min
Detection: 220 nm
Temperature: 40 °C
LC/MS Condition 4
Column: Waters BEH CI 8, 2.0 x 50 mm, 1.7 μιη particle
Mobile Phase A: 5:95 acetonitrile: water with 10 mM ammonium acetate Mobile Phase B: 95:5 acetonitrile: water with 10 mM ammonium acetate Gradient: 0%B, 0-100% B over 3 minutes, then a 0.5-minute hold at 100% B Flow: 1 mL/min
Detection: UV at 220 nm
Temperature: 50 °C
/////////1663477-91-5, BRISTOL-MYERS SQUIBB, TRICYCLOHEXADECAHEXAENE DERIVATIVES, TREATMENT OF HEPATITIS C VIRUS
FC1(F)CCC(CC1)C(=O)N[C@H](c2nc3ccc(cc3n2)c9cc4ccc9CCc5ccc(CC4)c(c5)c6ccc7nc(nc7c6)[C@@H](NC(=O)C8CCC(F)(F)CC8)C(C)(C)C)C(C)(C)C
Ripasudil hydrochloride hydrate 塩酸塩水和物 , リパスジル
Ripasudil hydrochloride hydrate
4-fluoro-5-[[(2S)-2-methyl-1,4-diazepan-1-yl]sulfonyl]isoquinoline;dihydrate;hydrochloride
4-Fluoro-5-[2(S)-methylperhydro-1,4-diazepin-1-ylsulfonyl]isoquinoline hydrochloride dihydrate
(S)-4-Fluoro-5-(2-methyl-1,4-diazepan-1-ylsulfonyl)isoquinoline hydrochloride dihydrate
Cas 223645-67-8 FREE
| M.Wt | 395.88 | OR C15H18FN3O2S·HCl·2H2O | |
|---|---|---|---|
| Formula | C15H23ClFN3O4S | ||
| CAS No | 887375-67-9 .HCL 2 H2O |
016TTR32QF, K 115
LAUNCHED 2014 Kowa
JAPAN 2014-09-26, Glanatec

リパスジル塩酸塩水和物
Ripasudil Hydrochloride Hydrate

C15H18FN3O2S.HCl.2H2O : 395.88
[887375-67-9]
SEE http://pdf.irpocket.com/C4576/GpH7/tLM4/sJIT.pdf
Ripasudil
- Molecular FormulaC15H18FN3O2S
- Average mass323.386
| Company | D. Western Therapeutics Institute Inc. |
| Description | Selective rho kinase inhibitor |
| Molecular Target | Rho kinase |
| Mechanism of Action | Rho kinase inhibitor |
SEE
NMR ETC
Published online: 24th May, 2011
■ A Practical Synthesis of Novel Rho-Kinase Inhibitor, (S)-4-Fluoro-5-(2-methyl-1,4-diazepan-1-ylsulfonyl)isoquinoline
Noriaki Gomi, Tadaaki Ohgiya, Kimiyuki Shibuya,* Jyunji Katsuyama, Masayuki Masumoto, and Hitoshi Sakai
*Pharmaceutical Division, Tokyo New Drug Research Laboratories, Kowa Co., Ltd., 2-17-43, Noguchicho, Higashimurayama, Tokyo 189-0022, Japan
Abstract
A practical synthesis of novel Rho-kinase inhibitor, (S)-4-fluoro-5-(2-methyl-1,4-diazepan-1-ylsulfonyl)isoquinoline hydrochloride dihydrate (1) was achieved in a pilot-scale production. We have demonstrated the regioselective chlorosulfonylation of 4-fluoroisoquinoline in an one-pot reaction to afford 4-fluoroisoquinoline-5-sulfonyl chloride and the asymmetric construction of the (S)-2-methyl-1,4-diazepane moiety as key steps.
K-115, an isoquinolinesulfonamide compound, is a highly selective and potent (IC50 = 31 nM) Rho-kinase inhibitor; is in Phase II clinical development in patients with POAG or ocular hypertension.Ripasudil hydrochloride hydrate (Glanatec® ophthalmic solution 0.4 %; hereafter referred to as ripasudil) is a small-molecule, Rho-associated kinase inhibitor developed by Kowa Company, Ltd. for the treatment of glaucoma and ocular hypertension. This compound, which was originally discovered by D. Western Therapeutics Institute, Inc., reduces intraocular pressure (IOP) by directly acting on the trabecular meshwork, thereby increasing conventional outflow through the Schlemm’s canal.
Ripasudil hydrochloride hydrate was first approved by Pharmaceuticals and Medical Devices Agency of Japan (PMDA) on Sept 26, 2014. It was developed and marketed as Glanatek® by Kowa Pharmaceuticals.
Ripasudil hydrochloride hydrate is the first drug that can inhibit the rho-associated, coiled-coil containing protein kinase (ROCK). It is indicated for the treatment of glaucoma and ocular hypertension.
Glanatek® is available as solution (0.4%) for ophthalmic use, containing 4 mg of free Ripasudil per millimeter, and the recommended dose is one drop twice daily.
As a result of this mechanism of action, ripasudil may offer additive effects in the treatment of glaucoma and ocular hypertension when used in combination with agents such as prostaglandin analogues (which increase uveoscleral outflow) and β blockers (which reduce aqueous production).
The eye drop product has been approved in Japan for the twice-daily treatment of glaucoma and ocular hypertension, when other therapeutic agents are not effective or cannot be administered. Phase II study is underway for the treatment of diabetic retinopathy.
K-115 is a Rho-kinase inhibitor as ophthalmic solution originally developed by Kowa and D Western Therapeutics Institute (DWTI). The product candidate was approved and launched in Japan for the treatment of glaucoma and ocular hypertension in 2014.
In 2002, the compound was licensed to Kowa Pharmaceutical by D Western Therapeutics Institute (DWTI) in Japan for the treatment of glaucoma. The compound is currently in phase II clinical trials at the company for the treatment of age-related macular degeneration and diabetic retinopathy.
Use of (S)-(-)-1-(4- fluoro-5-isoquinoline-sulfonyl)-2-methyl-1,4-homopiperazine (ripasudil hydrochloride, first disclosed in WO9920620), in the form of eye drops, for the treatment of retinal diseases, particularly diabetic retinopathy or age-related macular degeneration.
Follows on from WO2012105674 by claiming a combination of the same compound. Kowa, under license from D Western Therapeutics Institute, has developed the Rho kinase inhibitor ripasudil hydrochloride hydrate (presumed to be Glanatek) as an eye drop formulation for the treatment of glaucoma and ocular hypertension which was approved in Japan in September 2014..
The company is also developing the agent for the treatment of diabetic retinopathy, for which it is in phase II trial as of October 2014.
Ripasudil (Glanatec) is a drug used for the treatment of glaucoma and ocular hypertension. It is approved for use in Japan as a 0.4% ophthalmic solution.[1]
Ripasudil, a derivative of fasudil, is a rho kinase inhibitor.[2]
Paper
A Practical Synthesis of (S)-tert-butyl 3-methyl-1,4-diazepane-1-carboxylate, the key intermediate of Rho-kinase inhibitor K-115
Synthesis (Stuttgart) 2012, 44(20): 3171
https://www.thieme-connect.com/products/ejournals/abstract/10.1055/s-0032-1316771
practical synthesis of (S)-tert-butyl 3-methyl-1,4-diazepane-1-carboxylate has been established for supplying this key intermediate of Rho–kinase inhibitor K-115 in a multikilogram production. The chiral 1,4-diazepane was constructed by intramolecular Fukuyama–Mitsunobu cyclization of a N-nosyl diamino alcohol starting from the commercially available (S)- or (R)-2-aminopropan-1-ol. In the same manner, an enantiomeric pair of a structural isomer were prepared for demonstration of the synthetic utility.
SEE
WO 2006137368 http://www.google.com/patents/WO2006137368A1?cl=en
PATENT
WO 2012026529
http://www.google.com/patents/WO2012026529A1?cl=en
The including prevention and treatment cerebral infarction, cerebral hemorrhage, subarachnoid hemorrhage, cerebrovascular disorders such as cerebral edema, the present invention relates to a salt thereof or isoquinoline derivatives useful as therapeutic agents, particularly glaucoma.
(S) – (-) -1 – (4 – fluoro-iso-5 – yl) sulfonyl – 2 – methyl -1,4 – diazepane the following formula (1):
It is a compound represented by the particular it is a crystalline water-soluble, not hygroscopic, because it is excellent in chemical stability, it is useful as a medicament has been known for its hydrochloride dihydrate ( refer to Patent Documents 1 and 2). -5 Isoquinoline of these – the sulfonamide compounds, that prophylactic and therapeutic agents for cerebral infarction, cerebral hemorrhage, subarachnoid hemorrhage, cerebrovascular disorders such as cerebral edema, is useful as a therapeutic agent for preventing and glaucoma in particular is known (1-5 see Patent Document 1).
Conventionally, for example, a method of manufacturing by the method described in Patent Document 1, as shown in the following production process has been reported preparation of said compound (Production Method 1-A).
That is, (S)-1-tert-butoxycarbonyl – 3 – by reacting the presence of triethylamine in methylene chloride-fluoro-isoquinoline (2) – methyl -1,4 – diazepane and 5 (3) – chloro-sulfonyl -4 by adding trifluoroacetic acid in methylene chloride compound (the first step), obtained following (4) to synthesize a compound (4) by deprotection to (second step) the desired compound (1) This is a method of manufacturing.
It is also an important intermediate for preparing the compound (1) (S)-1-tert-butoxycarbonyl – 3 – methyl-1 ,4 – diazepane to (3), for example, in the following manner (; see JP Production Process 1-B) that can be produced is known.
Further, on the other hand, the compound (1) (see Patent Document 1) to be manufactured manufacturing routes such as: Any (Process 2) are known.
WO 1999/20620 pamphlet WO 2006/057397 pamphlet WO 1997/028130 pamphlet JP Patent Publication No. 2006-348028 JP Patent Publication No. 2006-290827
However, it is possible to produce in the laboratory of a small amount scale, but you place the point of view for mass industrial production, environmentally harmful halogenated hydrocarbon solvent in the compound of the above-mentioned process for producing 1-A is ( problem because it is carried out coupling step (3) and 2), giving significant adverse environmental exists. Therefore, solvent of halogenated hydrocarbon other than those listed to the specification of the patent document 1, for example, I tried actually dioxane, tetrahydrofuran and the like, but the present coupling reaction will be some progress indeed, Problems reaction is not completed raw material remained even after prolonged reaction time, yield undesirably stays in at most 30% was found. Furthermore, it is hard to decompose in the environment, elimination is also difficult to dioxane is not preferred irritating to humans, and are known as compounds that potentially harmful brain, kidney and liver .
When we actually produced compound (3) by the above production method 1-B, can be obtained desired compound in good yield merged with reproducibility is difficult has further been found that. That is, in the production path, 1,4 – and is used sodium hydride with dimethyl sulfoxide in forming a diazepane ring, except that I actually doing this step, Tsu than the reproducibility of the desired compound It could not be obtained in high yield Te. Also, that this is due to the synthetic route through the unstable intermediate, that it would be converted into another compound easily found this way. limitations and potential problems of the present production process is exposed since this stability may affect the reproducibility of the reaction.
Meanwhile, an attempt to carry out mass production is actually in the Process 2, it encounters various problems. For example, it is stored as an impurity whenever I repeat step, by-products formed in each stage by tandem production process ranging from step 8 gave more complex impurity profile. Depending, it is necessary to repeat a complicated recrystallization purity obtained as a medicine until the purification, the yield in the laboratory be a good overall yield is significantly reduced in the mass production of actual example be away, it does not have industrial utility of true was found. It can be summarized as follows: Considering from the viewpoint of GMP process control required for pharmaceutical production these problems.
Requires control process and numerous complex ranging 1) to 8 step, 3 2) third step – amino-1 – in the step of reacting a propanol, a difficult to remove positional isomers are mixed, 3) The fourth step water is mixed by the minute liquid extraction operation at the time of return to the free base from oxalate require crystallization purification by oxalate in the removal of contaminants of positional isomers, in 4) fifth step, 5) sixth step The Mitsunobu by reproducibility poor require water control in the Mitsunobu reaction used in the ring closure compounds to (1) compounds in (6), 6) ring closure reaction, departing management of the reagent added or the like is generated, in 7) Seventh Step it takes a complicated purification in impurity removal after the reaction, resulting in a decrease in isolated yield. These are issues that must be solved in order to provide a stable supply of raw material for pharmaceuticals high chemical purity is required.
Thus, gentle salt thereof, or the environment isoquinoline derivative comprising a compound represented by the formula (1), the present invention provides a novel production method having good reproducibility and high purity easily and in high yield I intended.
As a result of intensive studies in view of such circumstances, the present inventors, in the manufacturing process of the final target compound shown by the following expression
(Wherein represents a fluorine, chlorine, bromine or iodine, may, R 3 and 1, R 2 R represents a C 1-4 alkyl group be the same or different from each other, and P, X 1 is a protecting group shows a, 0 to m represents an integer of 3, 0 to n is. represents an integer of 3)
Is a urea-based solvents nitrile solvents, amide solvents, sulfoxide or solvents, the solvent may be preferably used in the coupling step of the compound (III) and (II) are generally very short time With these solvents It has been found that can be converted to the desired product quantitatively. It is possible to carry out the coupling step Volume scale while maintaining a high yield by using these solvents, there is no need to use a halogenated hydrocarbon solvent to give significant adverse environment. In consideration of the process such as removal of the solvent after the reaction was further found that acetonitrile is the best among these solvents. Also, since by using hydrochloric acid with ethyl acetate solvent in step deprotection can be isolated as crystal of hydrochloride desired compound (I), without going through the manipulation of solvent evaporation complicated , it has been found that it is possible to obtain the object compound (I) is a simpler operating procedure. Since there is no need to use a halogenated hydrocarbon solvent in this deprotection step further, there is no possibility of harming the environment.
It has been found that it is possible in mass production of (II), leading to the target compound purity, in high yield with good reproducibility as compared with the conventional method compounds are important intermediates in the coupling step further. That is, was it possible to lead to the intermediate high purity and in high yield by eliminating the production of a harmful halogenated hydrocarbon solvent to the environment in this manner. 1,4 addition – in order to avoid the problems encountered in the reaction using sodium hydride in dimethyl sulfoxide in forming the diazepane ring, in order to allow the cyclization reaction at mild conditions more, as a protecting group By performing the Mitsunobu reaction using Noshiru group instead of the carbobenzyloxy group, in addition to one step shorten the manufacturing process of the whole, without deteriorating the optical purity was successfully obtained the desired compound desired.

SEE
CLIP
Ripasudil hydrochloride hydrate (Glanatec)
Ripasudil hydrochloride hydrate (Glanatec) was approved in Japan in 2014 for the treatment of glaucoma and ocular hypertension.
219 Originally discovered by D. Western Therapeutics Institute,Inc. and licensed by the Kowa Company, Ltd, ripasudil
functions as a selective Rho-kinase inhibitor and reduces intraocular pressure by stimulation of aqueous humour drainage of the
trabecular meshwork.219–221
While this recent approval allows for use of ripasudil as a twice-daily monotherapy treatment when
other drugs cannot be used or are not effective, clinical trials using ripasudil as a combination therapy with other glaucoma
drugs have shown promising results in the treatment of primary open-angle glaucoma or ocular hypertension.222,223 Currently, the
Kowa Company is also pursuing trials focused on the use of ripasudil for the treatment of diabetic retinopathy and diabetic macular edema.224
While initial synthetic routes to ripasudil were carried out via a stepwise functionalization of 4-fluoroisoquinoline-5-sulfonylchloride (238),225,226 more recent reports describe an efficient route to ripasudil employing a late stage-coupling of Boc-diazepane
(237) with 4-fluoroisoquinoline-5-sulfonyl chloride (238), enabling synthesis on multi-kilogram scale and isolation of the
drug in high purity (Scheme 40).221,227,228 This optimized route to ripasudil begins with 2-nitrobenzene sulfonyl chloride (NsCl)-
mediated protection of (S)-2-amino-1-propanol (234) in 82% yield.
In this case, use of the NaHCO3/THF/H2O conditions were essential for preventing bis-nosylation.228 Alcohol activation with methanesulfonyl chloride (MsCl) in N-methyl morpholine (NMM) took place smoothly to give the corresponding mesylate 235 in 91%
yield. Direct mesylate displacement with 3-aminopropanol and subsequent amine protection as the carbamate ((Boc)2O) in a
one-pot fashion provided the corresponding Boc-amino propanol product 236 in 95% yield over 2 steps.
With the acyclic diazepane precursor 236 in hand, employment of the intramolecular Fukuyama-Mitsunobu N-alkyl cyclization conditions (diisopropylazodicarboxylate (DIAD)/PPh3) allowed generation of the diazepane in 75% yield. Nosyl group cleavage with thiophenol/K2CO3provided the Boc-diazepane 237 in 65% overall yield and 98% purity following a pH-controlled aqueous workup.
Finally, 4-fluoroisoquinoline- 5-sulfonyl chloride (238)—prepared via subjection of 4- fluoroisoquinoline (239, Scheme 41)229 to sulfur trioxide and sulfuric acid followed by treatment with thionyl chloride and finally 4 N HCl in ethyl acetate—was involved in a 1-pot, two-step procedure in which this sulfonyl chloride was coupled with diazepane 237 (TEA/MeCN) to access the ripasudil framework in quantitative yield.
Synthesis of the final drug target by deprotection with 4 MHCl in ethyl acetate followed by neutralization with aqueoussodium hydroxide provided the free base of ripasudil in 93% yield and 99.8% purity. Conversion to the more stable hydrochloride dihydrate form could be performed by treatment of the free base with 1 M HCl/EtOH and subsequent heating of the hydrochloride in H2O/acetone to provide ripasudil hydrochloride dihydrate XXIX in 83% yield.230,231
219. Garnock, J. P. K. Drugs 2014, 74, 2211.
220. Isobe, T.; Mizuno, K.; Kaneko, Y.; Ohta, M.; Koide, T.; Tanabe, S. Curr. Eye Res.2014, 39, 813.
221. Sumi, K.; Inoue, Y.; Nishio, M.; Naito, Y.; Hosoya, T.; Suzuki, M.; Hidaka, H.
Bioorg. Med. Chem. Lett. 2014, 24, 831.
222. Mizuno, K. WO Patent 2,012,105,674, 2012.
223. Mizuno, K.; Matsumoto, J. WO Patent 2,007,007,737, 2007.
224. http://clinicaltrials.jp/user/cteDetail.jsp.
225. Gomi, N.; Ohgiya, T.; Shibuya, K. WO Patent 2,012,026,529, 2012.
226. Hidaka, H.; Nishio, M.; Sumi, K. US Patent 20,080,064,681, 2008.
227. Gomi, N.; Kouketsu, A.; Ohgiya, T.; Shibuya, K. Synthesis 2012, 44, 3171.
228. Gomi, N.; Ohgiya, T.; Shibuya, K.; Katsuyama, J.; Masumoto, M.; Sakai, H.Heterocycles 2011, 83, 1771.
229. Sakai, H.; Masunoto, M.; Katsuyama, J.; Onogi, K. WO Patent 2006090783A1,2006.
230. Hidaka, H.; Matsuura, A. WO Patent 1999020620A1, 1999.
231. Ohshima, T.; Hidaka, H.; Shiratsuchi, M.; Onogi, K.; Oda, T. US Patent7858615B2, 2008.
![4-fluoro-5-[[(2S)-2-methyl-1,4-diazepan-1-yl]sulfonyl]isoquinoline NMR spectra analysis, Chemical CAS NO. 223645-67-8 NMR spectral analysis, 4-fluoro-5-[[(2S)-2-methyl-1,4-diazepan-1-yl]sulfonyl]isoquinoline H-NMR spectrum](https://i0.wp.com/pic11.molbase.net/nmr/nmr_image/2014-11-28/002/466/2466109_1h.png) CAS NO. 223645-67-8, 4-fluoro-5-[[(2S)-2-methyl-1,4-diazepan-1-yl]sulfonyl]isoquinoline H-NMR spectral analysis |
![4-fluoro-5-[[(2S)-2-methyl-1,4-diazepan-1-yl]sulfonyl]isoquinoline NMR spectra analysis, Chemical CAS NO. 223645-67-8 NMR spectral analysis, 4-fluoro-5-[[(2S)-2-methyl-1,4-diazepan-1-yl]sulfonyl]isoquinoline C-NMR spectrum](https://i0.wp.com/pic11.molbase.net/nmr/nmr_image/2014-11-28/002/466/2466109_13c.png) CAS NO. 223645-67-8, 4-fluoro-5-[[(2S)-2-methyl-1,4-diazepan-1-yl]sulfonyl]isoquinoline C-NMR spectral analysis |
| WO1997028130A1 | Jan 31, 1997 | Aug 7, 1997 | Hiroyoshi Hidaka | Isoquinoline derivatives and drugs |
| WO1999020620A1 | Oct 22, 1998 | Apr 29, 1999 | Hiroyoshi Hidaka | Isoquinoline derivative and drug |
| WO2006057397A1 | Nov 29, 2005 | Jun 1, 2006 | Hiroyoshi Hidaka | (s)-(-)-1-(4-fluoroisoquinolin-5-yl)sulfonyl-2-methyl-1,4homopiperazine hydrochloride dihydrate |
| JP2006290827A | Title not available | |||
| JP2006348028A | Title not available | |||
| JPH11171885A * | Title not available | |||
| JPS61227581A * | Title not available |
References
- Garnock-Jones, K. P. (2014). “Ripasudil: First global approval”. Drugs 74 (18): 2211–5. doi:10.1007/s40265-014-0333-2.PMID 25414122.
- Tanihara, H; Inoue, T; Yamamoto, T; Kuwayama, Y; Abe, H; Suganami, H; Araie, M; the K-115 Clinical Study Group (2014). “Intra-ocular pressure-lowering effects of a Rho kinase inhibitor, ripasudil (K-115), over 24 hours in primary open-angle glaucoma and ocular hypertension: A randomized, open-label, crossover study”. Acta Ophthalmologica: n/a. doi:10.1111/aos.12599. PMID 25487877.
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
4-Fluoro-5-{[(2S)-2-methyl-1,4-diazepan-1-yl]sulfonyl}isoquinoline
|
|
| Clinical data | |
| Trade names | Glanatec |
| Identifiers | |
| PubChem | CID 9863672 |
| ChemSpider | 8039366 |
| Synonyms | K-115 |
| Chemical data | |
| Formula | C15H18FN3O2S |
| Molar mass | 323.39 g/mol |
///////////////// , Ripasudil hydrochloride hydrate, Ripasudil, 223645-67-8, 塩酸塩水和物 , リパスジル
O=S(=O)(c2c1c(F)cncc1ccc2)N3[C@H](CNCCC3)C
Annex 16: How a QP should handle unexpected Deviations
DRUG REGULATORY AFFAIRS INTERNATIONAL

In a recent blog of the MHRA, the inspectorate looks at one aspect of the new Annex 16 – the handling of unexpected deviations.
In a recent blog of the U.K. Medicines and Healthcare products Regulatory Agency (MHRA), the inspectorate looks at one aspect of the new Annex 16 – the handling of unexpected deviations.
Before Annex 16 was revised, the handling of minor deviations from defined processes was discussed in the European Medicines Agency’s “reflection paper” EMEA/INS/GMP/227075/2008. However, the status of this paper was not always clear, and its use was not consistently applied. Now section 3 of the new Annex 16 provides guidance on when a Qualified Person (QP) may consider confirming compliance or certifying a batch where an unexpected deviation (concerning the manufacturing process and/or the analytical control methods) from the MA and/or GMP has occurred.
Pre-requisites
Before a QP releases a batch these pre-requisites…
View original post 226 more words
FDA publishes Technical Guide on Quality Metrics
DRUG REGULATORY AFFAIRS INTERNATIONAL

The FDA has published a supplementing Guide on Quality Metrics. This is a very unusual step as the contents of the guide are planned to be integrated into the Guideline on Quality Metrics which hasn’t been finalised yet. Read more about the Technical Quality Metrics Guide.
The FDA has published a supplementing Guide on Quality Metrics. This is a very unusual step as the contents of the guide are planned to be integrated into the Guideline on Quality Metrics which hasn’t been finalised yet.
The so-called FDA Quality Metrics Technical Conformance Guide should supplement the Guidance for Industry: Request for Quality Metrics published on 28 July 2015 which is currently still in the draft version. We have recently published a GMP News about a Quality Metrics Case Study at Aenovaregarding a possible implementation. Now, the Technical Guide defines how the industry should submit Quality Metrics to the…
View original post 218 more words
Yonkenafil

Yonkenafil
Mw 487.61, MF C₂₄H₃₃N₅O₄S,
Cas 804518-63-6
4H-Pyrrolo[2,3-d]pyrimidin-4-one, 2-[2-ethoxy-5-[(4-ethyl-1-piperazinyl)sulfonyl]phenyl]-3,7-dihydro-5-methyl-7-propyl-,
2- [2-ethoxy –5- (4 – ethylpiperazine -1– sulfonyl) phenyl] -5 – methyl – 7 – n-Propyl-3 7 – PYRROLINE [2, 3 – d] pyrimidin – 4 – one
Phase2 Erectile dysfunction
扬子江药业 (Originator), 天士力制药 (Originator)
phosphodiesterase type 5 (PDE5) inhibitor
- Originator Tasly Pharmaceutical Group; Yangtze River Pharmaceutical Group
- Class Erectile dysfunction therapies
- Mechanism of Action Type 5 cyclic nucleotide phosphodiesterase inhibitors




Yonkenafil Hydrochloride
| Molecular Weight | 524.08 | |
| Formula | C24H33N5O4S • HCl |
804518-63-6 (Yonkenafil);
804519-64-0 (Yonkenafil Hydrochloride);
4H-Pyrrolo[2,3-d]pyrimidin-4-one, 2-[2-ethoxy-5-[(4-ethyl-1-piperazinyl)sulfonyl]phenyl]-3,7-dihydro-5-methyl-7-propyl-, hydrochloride (1:1)
2- [2-ethoxy –5- (4 – ethylpiperazine -1– sulfonyl) phenyl] -5 – methyl – 7 – n-Propyl-3 7 – PYRROLINE [2, 3 – d] pyrimidin – 4 – one
Yonkenafil hydrochloride, useful for treating erectile dysfunction and other PDE-5 mediated diseases eg female sexual dysfunction, benign prostatic hyperplasia, hypertension, allergic asthma, bronchitis, glaucoma, gastrointestinal motility disorders or Alzheimer’s Ydisease.
Yangtze River Pharmaceutical, under license from Jilin University, is developing yonkenafil (appears to be first disclosoed in WO2004108726), a PDE-5 inhibitor, for treating male erectile dysfunction.
In June 2016, yonkenafil was reported to be in phase 2 clinical development.
Yonkenafil hydrochloride is in phase II clinical trials for the treatment of erectile dysfunction (ED).
The compound was co-developed by Yangtze River Pharmaceutical and Tianjin Tasly Pharm.
Yonkenafil is a novel phosphodiesterase type 5 (PDE5) inhibitor. Here we evaluated the effect of yonkenafil on ischemic injury and its possible mechanism of action. Male Sprague-Dawley rats underwent middle cerebral artery occlusion, followed by intraperitoneal or intravenous treatment with yonkenafil starting 2h later. Behavioral tests were carried out on day 1 or day 7 after reperfusion. Nissl staining, Fluoro-Jade B staining and electron microscopy studies were carried out 24h post-stroke, together with an analysis of infarct volume and severity of edema. Levels of cGMP-dependent Nogo-66 receptor (Nogo-R) pathway components, hsp70, apaf-1, caspase-3, caspase-9, synaptophysin, PSD-95/neuronal nitric oxide synthases (nNOS), brain-derived neurotrophic factor (BDNF)/tropomyosin-related kinase B (TrkB) and nerve growth factor (NGF)/tropomyosin-related kinase A (TrkA) were also measured after 24h. Yonkenafil markedly inhibited infarction and edema, even when administration was delayed until 4h after stroke onset. This protection was associated with an improvement in neurological function and was sustained for 7d. Yonkenafil enlarged the range of penumbra, reduced ischemic cell apoptosis and the loss of neurons, and modulated the expression of proteins in the Nogo-R pathway. Moreover, yonkenafil protected the structure of synapses and increased the expression of synaptophysin, BDNF/TrkB and NGF/TrkA. In conclusion, yonkenafil protects neuronal networks from injury after stroke.

Erectile dysfunction (Erectile dysfunction, ED) refers to the duration can not be achieved, and (or) maintain an erection sufficient for satisfactory sexual life. ED can be divided according to different causes psychogenic, organic and mixed three categories, which are closely related to the aging process, but it is not inevitable disease with age.
The primary risk factors for ED include: high blood pressure, high cholesterol, diabetes, coronary and peripheral vascular disease, spinal cord injury or pelvic organs or surgery. According to statistics worldwide about 150 million men suffer from varying degrees of ED, 2025 the number of patients will double. More ED treatment options, such as oral medications phosphodiesterase 5 (PDE5) inhibitors, dopaminergic activator, a receptor blocker, intracavernous injection therapy, vacuum devices treatment, penile prosthesis treatment Wait. Wherein the selective phosphodiesterase 5 (PDE5) inhibitors are the most sophisticated study based on ED treatment, clinical treatment for ED is the first-line drugs. Has now approved the listing of these drugs were five sildenafil (Sildenafil), Tadalafil (Tadalafil), vardenafil (Vardenafil), to that of non-black (Udenafil) and Miro that non-( Mirodenafil).
In 2004 the Chinese patent CN03142399. X discloses a series pyrrolopyrimidine ketone compound of the structure and for the treatment of sexual dysfunction in animals, including humans, in particular male erectile dysfunction and TOE5 function-related diseases use; wherein the compound 1-HC1, i.e. 2- [2_ ethoxy-5- (4-ethyl-piperazine-1-sulfonyl) phenyl] -5-methyl-7-n-propyl -3 , 7-dihydro-pyrrolo [2, 3-d] pyrimidine-4-one monohydrochloride salt has been used as CN03142399. X Example features are disclosed compound named hydrochloride that non-gifted grams. This patent only to the preparation of the compounds have been described
PATENT
WO2004108726
http://www.google.co.in/patents/WO2004108726A1?cl=en
Example 1
Preparation of 2-[2-ethoxyl-5-(4-ethylpiperazinyl-1-sulfonyl)phenyl] -5-methyl-7-n-propyl-3,7-dihydropyrrolo[2,3-d]pyrimidin-4-one, its monohydrochloride and dihydrochloride
Route of synthesis
-
- (1a)2-amino-3-cyano-4-methylpyrrole;
- (1b)N-propyl-2-amino-3-cyano-4-methylpyrrole;
- (2)2-ethoxylbenzoyl chloride;
- (3a)N-(3-cyano-4-methyl-1H-pyrrol-2-yl)-2-ethoxylbenzamide;
- (3b)N-(3-cyano-4-methyl-1-n-propyl-1H-pyrrol-2-yl)-2-ethoxylbenzami de;
- (4a) 2-(2-ethoxylbenzamido)-4-methyl-1H-pyrrolo-3-formamide;
- (4b) 2-(2-ethoxylbenzamido)-4-methyl-1-n-propyl-1H-pyrrolo-3-formamide;
- (5) 2-(2-ethoxylphenyl)-5-methyl-3,7-dihydro-pyrrolo[2,3-d]pyrimidin -4-one;
- (6)2-(2-ethoxylphenyl)-5-methyl-7-n-propyl-3,7-dihydropyrrolo[2,3-d ]pyrimidin-4-one;
- (7)4-ethoxyl-3-(5-methyl-4-oxy-7-n-propyl-3,7-dihydropyrrolo[2,3-d] pyrimidin-2-yl)benzenesulfonyl chloride;
- (8)2-[2-ethoxyl-5-(4-ethylpiperazinyl-1-sulfonyl)phenyl]-5-methyl-7 -n-propyl-3,7-dihydropyrrolo[2,3-d]pyrimidin-4-one.
Preparation 1N-(3-cyano-4-methyl-1H-pyrrol-2-yl)-2-ethoxylbenzamide (3a) and N-(3-cyano-4-methyl-1-n-propyl-1H-pyrrol-2-yl)-2-ethoxylbenzamide (3b)
2-ethoxyl benzoic acid (10.0g, 60.2mmol) was added into thionyl chloride (20ml), and the mixture was refluxed with agitation for 40 minutes, and the excess amount of thionyl chloride was evaporated under reduced pressure. The residual was dissolved into dichloromethane (150ml). Within 30 minutes and being stirred on ice bath, the afore-obtained solution of 2-ethoxyl benzoyl chloride was dropped into the compound (1a) (7.0g, 56.8mmol) dissolved in tetrahydrofuran (80ml) and triethylamine (8.5ml, 61.0mmol). After completion, the mixture was stirred for 1 hour at 0°C . After being washed with water and filtrated with diatomaceous earth, the reaction solution was mixed with 20g of silica gel and evaporated to dryness. The resulting residual was eluted with dichloromethane by using silica gel(80g) column to obtain 7.5g of solid product (3a) with the yield of 48%. Furthermore, the sample for analysis was prepared by column chromatography (developing agent: dichloromethane: n-hexane=1:2) and recrystallization (dichloromethane: n-hexane=1:5).
mp 183~184°C (sublimation 162°C);\
IR (cm-1) : 3326, 3309, 2981, 2938, 2915, 2854, 2208, 1647, 1593, 1471, 1309, 1302, 1232, 1039, 923, 727, 655, 648;1H NMR (CDCl3) : δ 1.70 (t, J=7.0Hz, 3H), 2.15 (s, 3H), 4.32 (q, J=7.0Hz, 2H), 6.24 (s, 1H), 7.04 (d, 1H), 7.10 (m, 1H), 7.51 (dd, 1H), 8.20 (dd, J=7.9 and 1.8Hz, 1H), 10.69 (brs, 1H), 10.80 (s, 1H);13CNMR (CDCl3) : δ (CH3) 10.6, 15.0; (CH2) 65.7; (CH) 110.3, 112.3, 121.4132.1, 134.2; (C) 78.7, 115.6, 119.2, 119.4, 136.7, 157.0, 163.2;
MS (ES+) : m/z 287 (M+NH4) .
Elemental analysis (C15H15N3O2) : C 66.90%; H 5.61%; N 15.60%; 0 11.88%. The compound (3b) was prepared from compound (1b) according to the above-mentioned method with the yield of 41%.
mp 58~61°C;
IR (cm-1) : 3596, 3336, 2969, 2937, 2877, 2216, 1676, 1658, 1603, 1571, 1537, 1475, 1431, 1292, 1232, 1122, 1037, 927, 789, 752, 577;1H NMR (CDCl3): δ 0.88 (t, J=7.4Hz, 3H), 1.58 (t, J=7.0Hz, 3H), 1.75(m, 2H), 2.16 (s, 3H), 3.73 (t, J=7.4Hz, 2H),4.30 (q, J=7.0Hz, 2H), 6.36 (s, 1H), 7.04 (d, 1H), 7.11 (m, 1H), 7.48 (dd, 1H), 8.23 (dd, J=7.9 and 1.8Hz, 1H), 9.62 (brs, 1H) ;13C NMR (CDCl3) : δ (CH3) 11.1, 14.8; (CH2) 23.6, 48.3, 65.2; (CH) 112.5,117.0, 121.3, 132.5, 134.1; (C) 89.2, 115.6, 119.8, 120.5, 131.2, 157.1, 165.0;MS (ES+): m/z 329 (M+NH4).
Preparation 2
2-(2-ethoxylbenzamido)-4-methyl-1H-pyrrolo-3-formamide (4a) and 2-(2-ethoxylbenzamido)-4-methyl-1-n-propyl-1H-pyrrolo-3-formamide(4 b);
A mixture of N-(3-cyano-4-methyl-1H-pyrrol-2-yl)-2-ethoxylbenzamide(3a) (2.00g, 7.44mmol) or N-(3-cyano-4-methyl-1-n-propyl-1H-pyrrol-2-yl)-2 -ethoxylbenzamide(3b) (2.30g, 7.44mmol) of preparation 1 and 85% phosphoric acid (14.8ml) was stirred for 20 minutes at 130°C, cooled and poured into crushed ice (80g). The precipitations were filtrated and dried to give dark red solid of compound (3a) or (3b) with the yield of 80%. The product(3a) and (3b) of this step may be directly used for the next step without further purification.
Preparation 32-(2-ethxoylphenyl)-5-methyl-3,7-dihydropyrrolo[2,3-d]pyrimidin-4-one(5) and 2-(2-ethoxylphenyl)-5-methyl-7-n-propyl -3,7-dihydropyrrolo[2,3-d]pyrimidin-4-one(6)
A mixture of 2-(2-ethoxylbenzamido)-4-methyl-1H-pyrrolo-3-formamide (4a) (7.0g, 25.5mmol) of preparation 2 and dimethyl cyclohexylamine (20ml) was refluxed with agitation for 11 hours in N,N-dimethyl formamide (100ml). After evaporation the solvent by distillation under reduced pressure, the residual was extracted with dichloromethane, and the dichloromethane extraction was washed with water. the resultant extraction was dried with anhydrous sodium sulfate. n-hexane (80ml) was added into the residual and ground to give product (5) (6.0g) by filtration with the yield of 91%.
mp 219~221°C
IR (cm-1) : 3187, 3114, 3062, 2978, 2923, 1658, 1587, 1460, 1321, 1292, 1250, 1044, 771, 763;
1H NMR (DMSO-d6) : δ 1.35 (t, J=6.9Hz, 3H), 2.29 (s, 3H), 4.13 (q, J=7.0Hz, 2H), 6.79 (s, 1H), 7.05 (t, 1H), 7.14 (d, 1H), 7.45 (dd, 1H), 7.76 (dd, 1H), 11.35 (brs, 1H), 11.54 (brs, 1H);
13C NMR (DMSO-d6) : δ (CH3) 11.2, 14.5; (CH2) 64.2; (CH) 113.0, 118.0, 120.6, 130.1, 131.9, (C) 105.0, 113.6, 121.9, 148.5, 149.8, 156.5, 159.2; MS(ES+) : m/z 287 (M+NH4) .
The compound (6) was prepared from compound(4b) according to the above-mentioned method with the yield of 85%
mp 124~127°C
IR (cm-1) : 3234, 3184, 3141, 3103, 3056, 2956, 2943, 2869, 1654, 1595, 1567, 1468, 1311, 1267, 1243, 1191, 1118, 1047, 758;
1H NMR (CDCl3) : δ 0.88 (t, J=7.5Hz, 3H), 1.23 (t, 3H), 1 . 80 (q, 2H), 2. 42 (s, 3H), 4.08 (t, J=7.2Hz, 2H), 4.22 (q, 2H), 6.60 (s, 1H), 7.01 (d, J=8.3Hz, 1H), 7.08 (t, 1H), 7.40 (m, 1H), 8.35 (dd, J=8.0 and 1.9 Hz, 1H), 11.02 (brs, 1H).
Preparation 42-(2-ethxoylphenyl)-5-methyl-7-n-propyl-3,7-dihydro-pyrrolo[2,3-d] pyrimidin-4-one(6):
A mixture of compound (5) (1.5g, 5.57mmol) of preparation 3, n-propyl bromide (2.0g, 16.3mmol) and potassium carbonate (5g, 36.2mmol) was dissolved in acetone (15ml), refluxed with agitation by heating for 15 hours, after the solids were filtrated out, the filtrate was dried under reduced pressure. The resultant was developed by column chromatography, using dichloromethane as mobile phase to obtain 0.6g of product (6) with yield of 35%. The physical/chemical data were identical with that of the above-mentioned.
Preparation 54-ethoxyl-3-(5-methyl-4-oxy-7-n-propyl-4,7-dihydropyrrolo[2,3-d] pyrimidin-2-yl)benzenesulfonyl chloride(7):
2-(2-ethxoylphenyl)-5-methyl-7-n-propyl-3,7-dihydropyrrolo[2,3-d] pyrimidin-4-one(6) (1.25g, 4.01mmol) of preparation 4 was added into chlorosulfonic acid (4ml) that was dissolved in acetic ether (20ml), stirred at 0°C by two batches. The obtained solution was stirred at 0 °C for 30 minutes, and then reacted with agitation at room temperature for 3 hours. The resultant solution was poured into the a mixture of icy water (50ml) and acetic ether (50ml) . The organic layer was separated, washed with cold water (5ml), desiccated with anhydrous sodium sulfate and concentrated to dryness to afford 1.33g of product as yellow foam. The yield was 81%. The product was used directly for the next reaction.
Compound 1:
BASE
2-[2-ethoxyl-5-(4-ethyl-piperazinyl-1-sulfonyl)phenyl]-5-methyl-7-n -propyl-3,7-dihydropyrrolo[2,3-d]pyrimidin-4-one (8):
4-ethoxyl-3-(5-methyl-4-oxy-7-n-propyl-4,7-dihydro-3H-pyrrolo[2,3-d ]pyrimidin-2-yl)benzenesulfonyl chloride(7) (1.00g, 2.44mmol) of Preparation 5 was dissolved into dichloromethane (20ml), stirred at 0 °C, into which 1-ethyl piperazine (0.78ml, 6.10mmol) was added slowly. Reactant solution was stirred at 0°C for 5 minutes, and then sequentially stirred at room temperature for 5 hours. The crude product was washed with water and dried with anhydrous sodium sulfate to give 1. 2g of product as yellow foam. Continuously, the product was refined by column chromatography (acetic ether: methanol=20:1) to afford 0.89g of product as a yellow solid with yield of 75%.
mp: 174~176°C (EtOAc);
IR (cm-1) : 3324, 2960, 2923, 2869, 2862, 2767, 1682, 1560, 1458, 1355, 1282, 1247, 1172, 1149, 739, 615, 588, 555;
1H NMR(CDCl3) : δ 0.89(t,J=7.4Hz, 3H), 0.99(t, J=7.2Hz, 3H), 1.61(t,J=7.0Hz,3H),1.77-1.86(m, 2H), 2.35(m, 2H), 2.41(s, 3H), 2.50(brs, 4H), 3.05(brs,4H), 4.08(t, J=7.0Hz, 2H), 4.29-4.37(q, 2H), 6.61(s, 1H), 7.11(d, J=8.8Hz,1H), 7.77(dd, J=8.7 and2.2Hz, 1H), 8.74(d, J=2.2, 1H), 10.63(brs, 1H);
13C NMR(CDCl3) : δ (CH3) 11.0, 11.3, 11.8, 14.3; (CH2)23.8, 45.9, 46.1, 51.6, 51.7, 65.8; (CH)112.9, 121.1, 130.6, 131.3;(C)105.7,114.6, 121.4, 127.8, 146.8, 147.3, 159.3, 159.6;MS(ES+): m/z 505(M+NH4).
Elemental analysis (C24H33N5O4S) : theoretical value C 59.12%; H 6.82%; N 14.36%; practically measured value C59.38%; H 7.10%; N 14.12%.
Compound 1-HCl:
2-[2-ethoxyl-5-(4-ethylpiperazinyl-1-sulfonyl)phenyl]-5-methyl-7-n-propyl-3,7-dihydropyrrolo[2,3-d]pyrimidin-4-one monohydrochloride (9) :
The free alkali (compound 1) (1.00g, 2.05mmol) was dissolved into ether (10ml) and dichloromethane (10ml), into which the solution of 4M hydrochloric acid (HC1)- dioxane (0.51ml, 2.04mmol) diluted with ethyl ether (10ml) was dropped with agitation. After completion, the resulting solution was continued to stir at room temperature for 20 minutes, filtrated and dried to give 1.01g of monohydrochloride with yield of 94%.
mp: 147~150°C;
IR(cm-1): 2964, 2931, 2675, 2599, 2462, 1668, 1574, 1456, 1348, 1167, 933, 588;
1H NMR(D2O): δ 0.72(m, 3H),1.24(t, J=7.3Hz, 3H), 1.45(m, 3H), 1.59(m, 2H), 2.04(s, 3H), 2.77-3.81(all brs, 8H), 3.20(q, 2H), 3.75(m, 2H), 4.20(m, 2H), 6.62(m, 1H), 7.17(m, 1H), 7.73(m, 1H), 8.22(s, 1H).
Elemental analysis (C24H33N5O4S. HCl) : theoretical value C 55.00%; H 6.54%; N 13.36%; practically measured value C55.28%; H 6.41%; N 13.07%.
PATENT
WO 2016095650
https://patentscope.wipo.int/search/en/detail.jsf?docId=WO2016095650&redirectedID=true
SEE
https://www.google.com/patents/CN1552714A?cl=en
Spectral Analysis
13C NMR PREDICT
COSY PREDICT
| CN1552714A * | Jun 6, 2003 | Dec 8, 2004 | 天津倍方科技发展有限公司 | 2-substituted benzyl-5,7-dihydrocarbyl-3,7-dihydro pyrroline [2,3-d] pyromidine-4-one derivative ,its preparation and medicinal use |
| CN102970965A * | Apr 4, 2011 | Mar 13, 2013 | Sk化学公司 | Composition containing PDE5 inhibitor for relieving skin wrinkles |
| WO2007067570A1 * | Dec 5, 2006 | Jun 14, 2007 | Biomarin Pharmaceutical Inc. | Methods and compositions for the treatment of disease |
//////////yonkenafil, Phase 2, Erectile dysfunction , phosphodiesterase type 5 (PDE5) inhibitor, Tasly Pharmaceutical Group; Yangtze River Pharmaceutical Group
Cc4cn(CCC)c1c4N/C(=N\C1=O)c2cc(ccc2OCC)S(=O)(=O)N3CCN(CC3)CC
Gisadenafil
Tenatoprazole, テナトプラゾール
Tenatoprazole
テナトプラゾール
泰妥拉唑
| Tenatoprazole; 113712-98-4; Ulsacare; Protop; TU 199; TU-199; | |
| Molecular Formula: | C16H18N4O3S |
|---|---|
| Molecular Weight: | 346.40412 g/mol |
5-methoxy-2-[(4-methoxy-3,5-dimethylpyridin-2-yl)methylsulfinyl]-1H-imidazo[4,5-b]pyridine
2-[2-(3,5-Dimethyl)pyridylmethylsulfinyl]-5-methoxyimidazo[4,5-b]pyridine
Phase I
PHASE 1 FOR ………..A proton pump inhibitor potentially for the treatment of gastroesophageal reflux disease.
![]()
Research Code TU-199
CAS No. 113712-98-4
Mitsubishi Tanabe Pharma and was licensed to Negma Laboratories

Tenatoprazole is a proton pump inhibitor drug candidate that was undergoing clinical testing as a potential treatment for refluxoesophagitis and peptic ulcer as far back as 2003.[1] The compound was invented by Mitsubishi Tanabe Pharma and was licensed to Negma Laboratories (part of Wockhardt as of 2007[2]).[3]:22
Mitsubishi reported that tenatoprazole was still in Phase I clinical trials in 2007[4]:27 and again in 2012.[3]:17
Tenatoprazole has an imidazopyridine ring in place of the benzimidazole moiety found in other proton pump inhibitors, and has a half-life about seven times longer than other PPIs.[5]

Tenatoprazole, or (+)-5-methoxy-2-{[(4-methoxy-3,5-dimethyl-2-pyridyl) methyl] sulfinyl} imidazo-[4,5-b] pyridine, is described in Patent No. EP 254,588. It belongs to the group of drugs considered as proton pump inhibitors, which inhibit the secretion of gastric acid and are useful in the treatment of gastric and duodenal ulcers. It can also be used to treat gastro-oesophageal reflux, digestive bleeding and dyspepsia, because of its relatively long elimination half-life, as described in the application for French patent No. FR 02. 13113.
The first known derivative of this series of proton pump inhibitors was omeprazole, described in Patent No. EP 001,529, which is endowed with properties which inhibit the secretion of gastric acid and is widely employed as an anti-ulcerative in human therapeutics.
In addition to omeprazole, other proton pump inhibitors are well known, and particular mention can be made of rabeprazole, pantoprazole and lansoprazole, which all exhibit structural analogy and lansoprazole, which all exhibit structural analogy and belong to the group of pyridinyl methyl sulfinyl benzimidazoles. These compounds are sulfoxides presenting with asymmetry at the level of the sulphur atom, and therefore generally take the form of a racemic mixture of two enantiomers.
Like omeprazole and other sulfoxide with an analogue structure, tenatoprazole has an asymmetric structure and may therefore be present in the form of a racemic mixture or of its enantiomers. Thus it may exist in the form of its two enantiomers with R and S configurations, or (+) or (−), respectively.
Recent studies have shown that, unlike all the other proton pump inhibitors such as, for example, omeprazole or lansoprazole, and unexpectedly, tenatoprazole is endowed with a markedly prolonged duration of action, resulting from a plasma half-life which is about seven times longer. Thus the clinical data collected have shown that tenatoprazole enables a degree of symptom relief and healing of gastric lesions which is superior to that achieved by other drugs belonging to the same therapeutic category of proton pump inhibitors, which thus allows its effective use in the treatment of atypical and oesophageal symptoms of gastro-oesophageal reflux, digestive bleeding and dyspepsia, as indicated above.
Studies performed by the application have made it possible to show that the two enantiomers contribute differently to the properties of tenatoprazole, and that the two enantiomers, (+) and (−) exhibit significantly different pharmacokinetic properties. Thus it is possible to prepare medicinal products with specific activity by isolating the enantiomers, and these enantiomers themselves exhibit a different pharmacokinetic profile from that of the known racemic mixture. It then becomes possible to use each of these enantiomers more effectively in precise indications for the treatment of perfectly identified pathologies.
Anti-ulcer drug
tenatoprazole (tenatoprazole) is a new proton pump inhibitor, by the Japanese company Tokyo Tanabe, Japan’s Mitsubishi Corporation and Japan’s Hokuriku pharmaceutical companies jointly developed, has passed Phase II clinical trials. It acts on gastric parietal cells, reducing treatment of gastric ulcer, duodenal ulcer, reflux wall cell H + / K + -ATP activity, inhibition of gastric acid secretion, and H. pylori antibacterial activity, mainly for esophagitis and Zhuo – Ellison syndrome and gastric acid secretion disorders related diseases. Compared with the same types of drugs, Tenatoprazole suppress H + / K + -ATP enzyme activity is stronger, more stable, its efficacy than similar products currently widely used in clinical omeprazole strong 7 times. It has not been in the domestic market, nor ratified the production, with broad market prospects and development potential.
Proton pump inhibitors (proton pump inhibitors) for the treatment of acid-related diseases, the past ten years a wide range of clinical applications, better effect of the drug. It can quickly pass through the stomach wall membrane, gathered in a strongly acidic secretory tubules, and H + / K + -ATP enzyme (proton pump) thiol groups covalently bonded to form a disulfide bond, proton pump inactivation, inhibition of the enzyme H + / K + transport, so as to achieve the effect of acid suppression. Proton pump inhibitors and conventional clinical application of gastric acid suppression drugs H2 receptor antagonists compared with different sites of action and have different characteristics, namely acid-suppressing effect at night is good, rapid onset of acid inhibition strong and long time, easy to take these drugs can quickly and efficiently inhibit gastric acid secretion and clearance of Helicobacter pylori, it is widely used gastric ulcer, duodenal ulcer, reflux esophagitis and Zhuo – Ellison syndrome and other diseases treatment. Currently, proton pump inhibitors has been listed on the main omeprazole, lansoprazole, pantoprazole, rabeprazole and esomeprazole.Physical and
chemical properties ofwhite or white crystalline powder, melting point 174 ~ 175 ℃. Soluble in chloroform, insoluble in alcohol and water.
This product and other proton pump inhibitors as compared to chemically stable. China had 34 omeprazole preparations from Portugal, Brazil, India, China and other 13 countries, the stability of the measurements were made. The results showed that only six products (18%) during the trial showing good physical and chemical stability of. 27 products (79%) less (including Chinese product), the active ingredient a significant chemical decomposition, color and physical properties such as dissolution, are also a corresponding change. The results of a stability test designed to compare the various proton pump inhibitors show investigated eight days at 60 ℃, relative humidity of 75%, after omeprazole decomposition only 3% of the active ingredient, the tenatoprazole 77% of the data, said Alpha pantoprazole stability far superior to omeprazole, is already developed similar products in the most promising products.
Synthesis
Matsuishi, N.; Takeda, H.; Iizumi, K.; Murakami, K.; Hisamitsu, A. US Patent 4,808,596, 1989
Synthesis of Tenatoprazole 1 commences with the coupling of 2-mercapto-5-methoxyimidazo[4,5-b]pyridine 2 with 2-chloromethyl-4-methoxy-3,5-dimethyl pyridine hydrochloride 3 in the presence of potassium hydroxide affords 4 with 73% yield in ethanol and chloroform. The oxidation of the penultimate sulfide intermediate4 with m-CPBA in chloroform (100 vol) afforded 1
Syn 2
synthesis of 1 begins with the solvent-free condensation of 2-mercapto-5-methoxyimidazo[4,5-b]pyridine 2 with 2-chloromethyl-4-methoxy-3,5-dimethyl pyridine hydrochloride 3 to deliver the sulfide intermediate4 with 98% yield.
The final step of the synthesis is the oxidation of the sulfide intermediate with m-CPBA to form tenatoprazole 1. The sulfide intermediate 4 on treatment with 0.9 equiv of m-chloroperbenzoic acid (m-CPBA) at −10 to −15 °C afforded the crude tenatoprazole which was isolated as its sodium salt. The sodium salt of tenatoprazole 5 was purified by recrystallsation using dimethyl formamide and ethyl acetate (2:1 ratio) to yield the pure crystalline tenatoprazole sodium 5. Treatment of tenatoprazole sodium 5 with dil. HCl in the presence of acetone and water afforded the pure tenatoprazole 1
PATENT
CN 1861600
CN 1982311
WO 2009116072
CN 101429192
WO 2010043601
IN 2010CH00462
IN 251400
CN 102304127
WO 2012004802
CN 102703922
IN 2009DE01392
WO 2014111957
IN 2013MU00181
IN 2014CH01419
PAPER
Dai, Liyan; Synthetic Communications 2008, V38(4), P576-582
Advanced Materials Research (Durnten-Zurich, Switzerland) (2011), 233-235(Pt. 1, Fundamental of Chemical Engineering), 160-164.
Organic Process Research & Development (2013), 17(10), 1293-1299
Enantiomeric separation of proton pump inhibitors on new generation chiral columns using LC and supercritical fluid chromatography
Journal of Separation Science (2013), 36, (18), 3004-3010………http://onlinelibrary.wiley.com/doi/10.1002/jssc.201300419/abstract
PATENT
CN 102304127
https://www.google.com/patents/CN102304127A?cl=en
Tenatoprazole is a new type of gastric H + / K + -ATP enzyme inhibitors (proton pump inhibitor PPI), the chemical name 5-methoxy-2- (4-methoxy-3, 5-dimethyl-2-methylsulfinyl) imidazole and W, 5-b] pyridine, useful in the treatment of gastric ulcer, duodenal ulcer, reflux esophagitis and Zhuo – Ai syndrome and gastric acid secretion disorders related diseases. The drug was developed by Japan’s Tokyo Tanabe, Japan’s Mitsubishi Corporation and Japan’s Hokuriku pharmaceutical companies. Compared with other varieties of the same type, which inhibit H + / K + -ATP enzyme activity is stronger, ulcers of various tests are effective, and significantly improve the stability compared with other proton pump inhibitors.
US patent US4808596 “hidazo [4,5_b] pyridine compounds and pharmaceutical compositions containing same)) synthesis process disclosed Tenatoprazole the below formula:

By The route of 2-chloro-3,5-dimethyl-4-methoxypyridine hydrochloride with 2-mercapto-5-methoxy-imidazole, 5-b] pyridine under basic conditions condensation of Intermediate 2- [2- (3,5_-dimethyl) -4-methoxy-5-methoxy-pyridylmethyl sulfide] imidazo W, 5-b] pyridine, and then oxidizing the Thai duly omeprazole. This route for the synthesis of pull azole classic line, many pull azoles such as omeprazole can be synthesized by a similar route, this route mild condition, simple operation. But the route condensation, oxidation treatment after use of large amounts of toxic solvent chloroform, is not conducive to industrial scale; lower oxidation yields, the resulting Tenatoprazole containing unreacted starting materials 2- [2_ (3,5 – dimethyl) -4-methoxy-5-methoxy-pyridylmethyl sulfide] imidazo W, 5-b] pyridine, further comprising a sulfone by-product, N- oxide, N- oxide sulfone, These by-products may interfere with purification of tenatoprazole.
Japanese Patent invention Wo 丨 J JP05222038 “5_methoxy-2- [[(4_methoxy-3, 5-dimethyl-2-pyridyl) methyl] thio] imidazo [4,5 ~ b] pyridine and intermediates)) male
Synthesis open Tenatoprazole the below formula:
4-chloro-2-chloromethyl-3,5-dimethylpyridine -N- oxide 2_ mercapto _5_ methoxy-imidazo – [4, 5-b] pyridine in alkaline under condensation of Intermediate 5-Methoxy-2- (4-chloro-3,5-dimethyl-2-methylthio Bi) imidazo W, 5-b] pyridine-oxide -N- ( yield 82%), then refluxed in a solution of sodium methoxide in methanol to give 5-methoxy-2- (4-oxo-3,5-dimethyl-2-methyl sulfide) imidazo W , 5-b] pyridine -N- oxide (income ¥ 71%), and then at room temperature in methylene chloride, phosphorus trichloride treated with deoxy (yield 95%), and finally oxidation in Tenatoprazole (income Rate not reported). The novel synthetic route, mild reaction conditions, simple operation, the yield of each step is higher, but the route is too long resulting in a total yield is not high, prolonged and rising production costs.
Reaction route is as follows:
Example 1:
] a) 2- [2- (3,5-dimethyl) -4-methoxy-picolyl thioether _5_ methoxy] imidazo [4,5_b] pyridine:
To a reaction flask was added 2-mercapto-5-methoxy-imidazole, 5-b] pyridine 18. lg, 12g of sodium hydroxide and water 144. 8g, stirred and dissolved at 25 ° C, was added dropwise within Ih 20g of the 2-chloromethyl-dimethyl-4-methoxy _3,5- pyridine hydrochloride and 60g of water were mixed solution dropwise at 25 ° C the reaction 2h, the reaction is completed, filtered, washed with water 144. 8g, 36. 2mL ethanol and washed to obtain a wet powder; wet powder was dried at 50 ° C in vacuo to constant weight to give 2- [2_ (3,5-dimethyl) -4-methoxy-pyridylmethyl sulfide -5 – methoxy] imidazo [4,5-b] pyridine 32. Og;
2) Preparation of tenatoprazole lithium salt: To a reaction flask was added 2- [2- (3,5_-dimethyl) -4-methoxy-5-methoxy-pyridylmethyl sulfide] imidazo W, 5-b] pyridine 30g, dichloromethane 300g, methanol 15g, and dissolved with stirring; cooled to -10 ° C, was added dropwise the 15g and 485g m-chloroperbenzoic acid in methylene chloride mixed solution, dropwise addition the reaction temperature was controlled at -10 ° C, the dropping time of the pool; the dropwise addition, the temperature control at -10 ° C, the reaction 30min; completion of the reaction, at 10 ° C by the dropwise addition of lithium hydroxide and 135g water 15g mixed solution, drip complete, insulation stirred Ih; filtered cake was washed with acetone 60mL, get wet powder; wet powder was dried at 35 ° C under vacuum to constant weight to give Tenatoprazole lithium salt ^ g;
3) Preparation Tenatoprazole: To a reaction flask 加入泰 pantoprazole lithium salt 25g, acetone 63mL, water IOOmL, cooling M0 ° C, dropping lmol within lh / L hydrochloric pH7 0, drops. Albert, stirring 30min; the filter cake washed with water 50mL, washed with acetone and 50mL, wet powder was dried at 35 ° C under vacuum to constant weight to give Tenatoprazole 19. Sg.
Example 2:
a) 2- [2- (3,5-dimethyl) -4-methoxy-5-methoxy-pyridylmethyl sulfide] imidazo W, 5_b] pyridine (4) Preparation: To the reaction flask was added 2-mercapto-5-methoxy-imidazo 44,5-b] pyridine 18. lg, 11. 2g of potassium hydroxide and water 217mL, stirred and dissolved at! 35 ° C, was added dropwise within 2h by the 33. 3g of 2-chloro-3,5-dimethyl-4-methoxypyridine hydrochloride and 133. 2mL water mixed solution, dropwise at 35 ° C the reaction 4h, the reaction is completed, filtration, water 217mL, 72. 4mL ethanol and washed to obtain a wet powder; wet powder was dried at 60 ° C in vacuo to constant weight to give 2- [2- (3,5-dimethyl) -4-methoxy-pyridylmethyl sulfide -5-methoxy-yl] imidazo W, 5-b] pyridine 33. Ig;
2) Preparation of tenatoprazole lithium salt: To a reaction flask was added 2- [2- (3,5_-dimethyl) -4-methoxy-5-methoxy-pyridylmethyl sulfide] imidazole and W, 5-b] pyridine 30g, dichloromethane 400mL, methanol 50mL, stirring to dissolve; cooled to _15 ° C, was added drop by the m-chloroperoxybenzoic acid 16g of mixed solution of dichloromethane and 400mL , the process reactor temperature control was added dropwise at -20 ° C, the dropping time 2. 5h; the dropwise addition, the temperature control _15 ° C, the reaction 35min; completion of the reaction, at 15 ° C by the dropwise addition of 20g of hydrogen Lithium oxide and 200mL water mixed solution, drip completed, insulation mixing 1. 5h; filtration, the filter cake washed with acetone 90mL, get wet powder; wet powder was dried at 40 ° C under vacuum to constant weight to give Tenatoprazole lithium salt 28. 6g;
3) Preparation Tenatoprazole: To a reaction flask 加入泰 pantoprazole lithium salt 25g, ethanol 75mL, water 150mL, cooled to 10 ° C, dropping 6mol / L hydrochloric pH8 0 within 2h,. drops Albert, stirring 40min; the filter cake washed with water 100mL, washed with acetone IOOmL, wet powder was dried at 40 ° C under vacuum to constant weight, yield powder was Tenatoprazole 19. 5g.
Example 3:
a) 2- [2- (3,5-dimethyl) -4-methoxy-5-methoxy-pyridylmethyl sulfide] imidazo W, 5_b] pyridine (4) Preparation: To the reaction flask was added 2-mercapto-5-methoxy-imidazo 44,5-b] pyridine 18. lg, 8.4g of lithium hydroxide and water 180ml, stirred and dissolved at 30 ° C, was added dropwise within 1. 5h by the Guang .6g 2-chloro-3,5-dimethyl-4-methoxy-pyridine hydrochloride and 90mL water mixed solution, drop end at 30 ° C reaction 3h, the reaction is complete, filtration, water 217mL , washed with 85mL ethanol to obtain a wet powder; wet powder was dried at 55 ° C in vacuo to constant weight to give 2- [2- (3,5-dimethyl) -4-methoxy-5-pyridylmethyl sulfide oxy] imidazo [4,5-b] pyridine 32. 4g;
2) Preparation of tenatoprazole lithium salt: To a reaction flask was added 2- [2- (3,5_-dimethyl) -4-methoxy-5-methoxy-pyridylmethyl sulfide] imidazole and W, 5-b] pyridine 30g, dichloromethane 600mL, methanol 60mL, stirring to dissolve; cooled to -20 ° C, was added drop by the m-chloroperoxybenzoic acid 18g of mixed solution of dichloromethane and 600mL , dropwise addition the reaction temperature is controlled at _20 ° C, the dropping time of the pool; the dropwise addition, the temperature control at _20 ° C, the reaction 40min; completion of the reaction, at 20 ° C by the dropwise addition of lithium hydroxide and 300mL 30g water mixed solution, drip complete insulation mixing tank; filter, the filter cake washed with acetone and 120mL, get wet powder; wet powder was dried at 40 ° C under vacuum to constant weight to give Tenatoprazole lithium salt 28. 7g;
3) Preparation Tenatoprazole: To a reaction flask 加入泰 pantoprazole lithium salt 25g, methanol 75mL, water 120mL, cooled to 5 ° C, dropping dilute hydrochloric acid within 1 5h tune pH7 5,.. drops Albert, stirring 35min; the filter cake washed with water 75mL, 75mL acetone washed, wet powder was dried at 40 ° C under vacuum to constant weight, yield powder was Tenatoprazole 19. 6g.
Example 4:
a) 2- [2- (3,5-dimethyl) -4-methoxy-picolyl thioether _5_ methoxy] imidazo [4,5_b] pyridine ⑷ Preparation of: To a solution The reaction flask was added 2-mercapto-5-methoxy imidazole -½, 5-b] pyridine 18. lg, IOg sodium hydroxide and water 150ml, stirred and dissolved at 30 ° C, the 1. 5h dropwise added from 21 . 5g of 2-chloro-3,5-dimethyl-4-methoxypyridine hydrochloride and 90mL water mixed solution, dropwise at 30 ° C the reaction 3h, completion of the reaction, was filtered, washed with water 217mL, The wet powder was washed with ethanol to give 85mL; wet powder was dried at 55 ° C in vacuo to constant weight to give 2- [2- (3,5_-dimethyl) -4-methoxy-5-methoxy-pyridylmethyl sulfide ] imidazo [4,5-b] pyridine 32. 3g;
2) Preparation of tenatoprazole lithium salt: To a reaction flask was added 2- [2- (3,5_-dimethyl) -4-methoxy-5-methoxy-pyridylmethyl sulfide] imidazole and W, 5-b] pyridine 30g, dichloromethane 500mL, methanol 60mL, stirring to dissolve; cooled to -20 ° C, was added drop by the m-chloroperoxybenzoic acid 18g of mixed solution of dichloromethane and 500mL , the process reactor temperature control was added dropwise at -20 ° C, the dropping time pool; the dropwise addition, the temperature control in -20 ° C, the reaction 40min; completion of the reaction, at 20 ° C by the dropwise addition of lithium hydroxide 30g and 300mL water mixed solution, drip complete insulation mixing tank; filter, the filter cake washed with acetone and 120mL, get wet powder; wet powder was dried at 40 ° C under vacuum to constant weight to give Tenatoprazole lithium salt 28. 6g;
3) Preparation Tenatoprazole: To a reaction flask 加入泰 pantoprazole lithium salt 25g, isopropanol 75mL, water 120mL, cooled to 5 ° C, dropping 3mol / L hydrochloric within 1 5h. . pH7 5, drops Albert, stirring 35min; the filter cake washed with water 75mL, 75mL acetone washed, wet powder was dried at 40 ° C under vacuum to constant weight, yield powder was Tenatoprazole 19. 7g.
PAPER
An Improved Synthesis of Antiulcerative Drug: Tenatoprazole
http://pubs.acs.org/doi/full/10.1021/op800173u
, ‡Osmania University.
, §Dr. Reddy’s Laboratory Ltd.

An efficient, cost-effective and multikilogram-scale process for the synthesis of tenatoprazole 1, an antiulcerative drug, is described. The key steps in this synthesis involve the coupling of 2-mercapto-5-methoxyimidazo[4,5-b]pyridine 2 with 2-chloromethyl-4-methoxy-3,5-dimethyl pyridine hydrochloride 3 to yield 4 and its subsequent oxidation with m-CPBA to produce sulfoxide 1. The process has been scaled up for the multikilogram-scale of compound 1 with an overall yield of 72%. The new process requires no purification process and affords the target compound 1 with 99.8% purity by HPLC.
2-[2-(3,5-dimethyl)pyridylmethylsulfinyl]-5-methoxyimidazo[4,5-b]pyridine (1, 15.5 kg, 74%). Purity by HPLC 99.8%; 1H NMR (200 MHz, DMSO) δ 2.2 (s, 6H), 3.8 (s, 6H), 4.8 (s, 2H), 6.6 (d, 1H), 7.8 (d, 1H), 8.2 (s, 1H), 13.0 (s, 1H).
PATENT
http://www.google.co.in/patents/US7507746
the (+) enantiomer of tenatoplazole can be obtained by using chloroform, an industrially acceptable solvent, in accordance with the method proposed by Umemura et al. (J. Org. Chem. 1993, 58, 4592) as follows:
Example 1 (−)-5-methoxy-2-{(4-methoxy-3,5-dimethyl-2-pyridyl)methyl]sulfinyl}-1H-imidazo[4,5-b]pyridineThe conditions for preparative chromatography, shown as an example, are as follows:
Column: 265×110 mm ChiralPak®
Chiral Stationary Phase selector of the Amylose tris type [(S)-a methylbenzylcarbamate]
Flow rate: 570 ml/min
Detection: UV 240 nm
Temperature: Ambient temperature
These conditions are implemented on a liquid preparative chromatography apparatus.
Introduce approximately 2 g of the racemic mixture if tenatoprazole exhibiting purity higher than 99.5%. The (−) enantiomer is identified by measuring the angle of optical rotation, which must be laevogyre. This measurement can be performed directly on the column, the product being dissolved in the solvent (acetonitrile).
Example 2 (+)-5-methoxy-2-{(4-methoxy-3, 5-dimethyl-2-pyridyl)methyl]sulfinyl}-1H-imidazo[4,5-b]pyridine(R)-(+)-binaphthol 85 g (0.311 mol, 0.2 equivalence), ortho titanic acid isopropyl 42 g (0.148 mol, 0.1 equivalence), water 55 g (3.06 mol) and chloroform 7.5 L were stirred for 1 hour at room temperature. To the resultant, 5-methoxy-2-{(4-methoxy-3,5-dimethyl-2-pyridyl)methyl]thio}imidazo[4,5-b]pyridine (MPI), 0.5 kg, was added and stirred for 0.5 hours at room temperature. The thus-prepared mixture was cooled to 5° C. and then 70% aqueous solution of tert-butylhydroperoxide, 0.4 L (approx. 3.0 mol, 2.0 equivalence) was added and stirred for 72 hours at the same temperature as above. After the reaction endpoint was confirmed by HPLC, an aqueous solution of sodium hydroxide was added thereto to separate the aqueous layer, thus removing foreign matter. Then, the resultant was concentrated. Ethyl acetate was added to concentrated residues, which were then heated and suspended. The thus-prepared crude crystalline substances were dissolved in water and neutralized to pH 6.8 with a diluted sulfuric acid solution which was chilled with ice. Deposited crystals were filtered, dried and recrystallized by addition of ethanol to obtain (+)-5-methoxy-2-{(4-methoxy-3,5-dimethyl-2-pyridyl)methyl]sulfinyl}-1H-imidazo[4,5-b]pyridine {(+)-TU-199}
Yield: 77%
Optical purity: 96.6% ee
Chemical purity: 94.5%
Melting point: 135° C.
Optical rotation: +184° (conditions: C=1.0, N,N-dimethylformaldehyde solution)
Ultraviolet absorption spectrum: (10 μg/mL)λmax (nm): 316, 273, 206
When measurements were carried out, for a solubility of (+), (−) forms and a racemic form (±) of tenatoprazole in relation to water, it was found that the (+) form dissolved almost 3 times greater than the racemic body and (−) form dissolved over 2 times greater than the racemic form, exhibiting favorable physical properties in preparing drugs (refer to Table 2 below).
| TABLE 2 | |||
| (+) form | (−) form | (±)racemic form | |
| Solubility (water) μg/mL | 93.0 | 74.4 | 34.6 |
CLIPS

Tenatoprazole is a pyridinylmethylsulfinyl imidazopyridine compound, which is a weak base. This compound has three pKas. One is the pyridine pKa of pyridinylmethyl moiety and the others are the imidazole pKa and the pyridine pKa of the imidazopyridine moiety. The pyridine pKa1 enables tenatoprazole accumulation in the acidic canaliculus of the parietal cell. Protonation of the imidazopyridine ring enhances electron deficiency at the C-2 position, allowing intramolecular rearrangement to the active form. The active form is the sulfenic acid and/or cyclic sulfonamide, and reacts with luminal cysteine thiols of the enzyme to inhibit the enzyme activity
Synthesis route
from 2-mercapto-5-methoxy-imidazo [4,5-b] pyridine (2) and 2-chloro-3,5-dimethyl-4-methoxypyridine hydrochloride ( 3) by nucleophilic substitution synthesis of 2- (4-methoxy-3,5-dimethyl-2-methylthio) -5-methoxy-imidazo [4,5-b] pyridine (4) the oxidation of 4 1. Figure 1 is a synthesis route of tenatoprazole

References
- DataMonitor. March 2003. Gastrointestinal Disease Update: Digestive Disease Week 2003
- Economic Times. 3 March, 2011. Investors unwilling to forgive Wockhardt, promoter for failings
- Mitsubishi Tanabe Pharma State of New Product Development (as of May 8, 2012)
- Mitsubishi Tanabe Pharma FY2007 Interim Financial Results
- Li H et al. H+/K+-ATPase inhibitors: a patent review. Expert Opin Ther Pat. 2013 Jan;23(1):99-111. PMID 23205582
| US4808596 * | 24 Jul 1987 | 28 Feb 1989 | Tokyo Tanabe Company, Ltd. | Imidazo[4,5-b]pyridine compounds and pharmaceutical compositions containing same |
| US5753265 * | 7 Jun 1995 | 19 May 1998 | Astra Aktiebolag | Multiple unit pharmaceutical preparation |
| US5798120 * | 6 Oct 1994 | 25 Aug 1998 | Tokyo Tanabe Company Limited | Enteric granule-containing tablets |
| EP0124495A2 | 28 Feb 1984 | 7 Nov 1984 | Aktiebolaget Hässle | Omeprazole salts |
| EP0254588A1 | 24 Jul 1987 | 27 Jan 1988 | Tokyo Tanabe Company Limited | Imidazo[4,5-b] pyridine compounds, process for preparing same and pharmaceutical compositions containing same |
| Reference | ||
|---|---|---|
| 1 | * | Andersson et al., Pharmacology & Therapeutics, 2005, vol. 108, pp. 294-307. |
| 2 | * | Anon et al., Drugs in R&D, 2002, vol. 3, pp. 276-277. |
| 3 | Kakinoki et al., Methods and Findings in Experimental and Clinical Pharmacology, 21(3): 179-187 (1999). | |
| 4 | Komatsu et al., J. Org. Chem., 58(17): 4529-4533 (1993). | |
| 5 | Uchiyama et al., Journal of Pharmacy and Pharmacology, 51(4): 457-464 (1999). | |
| 6 | Uchiyama et al., Methods and Findings in Experimental and Clinical Pharmacology, 21(2): 115-122 (1999). | |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| US20120220623 * | 30 Aug 2012 | Mitsubishi Tanabe Pharma Corporation | The enantiomer of tenatoprazole and the use thereof in therapy |
| CN1453278A * | May 10, 2002 | Nov 5, 2003 | 中国人民解放军军事医学科学院放射医学研究所 | Omprazole compound and its prepn and application |
| CN1861600A * | Jun 14, 2006 | Nov 15, 2006 | 浙江大学 | Preparation process of taytrolazole |
| Reference | ||
|---|---|---|
| 1 | * | 《Organic Process Research & Development》 20081112 Somaiah Sripathi et al. An Improved Synthesis of Antiulcerative Drug:Tenatoprazole 第804-806页 1-6 第13卷, |
| 2 | * | 《Synthetic Communication》 20080101 Liyan Dai et al. Improved Synthetic Approach to Tenatoprazole 第576-582页 1-6 第38卷, |
| 3 | * | 《中国药物化学杂志》 20061231 赵冬梅等 抗溃疡药泰妥拉唑的合成 第360-362页 1-6 第16卷, 第6期 |
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
5-methoxy-2-[(4-methoxy-3,5-dimethylpyridin-2-yl)methylsulfinyl]-1H-imidazo[4,5-b]pyridine
|
|
| Clinical data | |
| Routes of administration |
Oral |
| Pharmacokinetic data | |
| Metabolism | Hepatic (CYP2C19-mediated) |
| Biological half-life | 4.8 to 7.7 hours |
| Identifiers | |
| CAS Number | 113712-98-4 |
| ATC code | none |
| PubChem | CID 636411 |
| ChemSpider | 552196 |
| UNII | RE0689TX2K |
| Chemical data | |
| Formula | C16H18N4O3S |
| Molar mass | 346.405 g/mol |
| Chirality | Racemic mixture |
テナトプラゾール
Tenatoprazole

C16H18N4O3S : 346.4
[113712-98-4]
/////////////Tenatoprazole, 113712-98-4, TU-199, proton pump inhibitor, treatment of gastroesophageal reflux disease, Mitsubishi Tanabe Pharma, Negma Laboratories, PHASE 1, テナトプラゾール
CC1=CN=C(C(=C1OC)C)CS(=O)C2=NC3=C(N2)C=CC(=N3)OC
Roxadustat

ROXADUSTAT
ASP1517; ASP 1517; ASP-1517; FG-4592; FG 4592; FG4592; Roxadustat.
CAS 808118-40-3
Chemical Formula: C19H16N2O5
Exact Mass: 352.10592
THERAPEUTIC CLAIM
Treatment of anemia
Roxadustat nonproprietary drug name
CHEMICAL NAMES
(4-hydroxy-1-methyl-7-phenoxyisoquinoline-3-carbonyl)glycine
1. Glycine, N-[(4-hydroxy-1-methyl-7-phenoxy-3-isoquinolinyl)carbonyl]-
2. N-[(4-hydroxy-1-methyl-7-phenoxyisoquinolin-3-yl)carbonyl]glycine
MF C19H16N2O5
MW 352.3
SPONSOR FibroGen
CODE FG-4592; ASP1517
CAS 808118-40-3
WHO NUMBER 9717
Roxadustat, also known as ASP1517 and FG-4592, is an HIF α prolyl hydroxylase inhibitor in a cell-free assay. It stabilizes HIF-2 and induces EPO production and stimulates erythropoiesis. Roxadustat transiently and moderately increased endogenous erythropoietin and reduced hepcidin
|
FG-4592 (also known as ASP1517), 2-(4-hydroxy-1-methyl-7-phenoxyisoquinoline-3-carboxamido)acetic acid,
is a potent small molecule inhibitor of hypoxia-inducible factor prolyl hydroxylase (HIF-PH),
an enzyme up-regulating the expression of endogenous human erythropoietin (Epo). It is currently being investigated as an oral treatment for anemia associated with chronic kidney disease (CKD). Unlike other anemia treating agents, erythropoiesis-stimulating agents (ESAs),
FG-4592 inhibits HIF, through a distinctive mechanism, by stabilization of HIF. According to previous studies,
FG-4592 is capable of correcting and maintaining hemoglobin levels in CKD patients not
receiving dialysis and in patients of end-stage renal disease who receives dialysis but do not need intravenous iron supplement.
Reference
1. Luis Borges. Different modalities of erythropoiesis stimulating agents.
Port J Nephrol Hypert 2010; 24(2): 137-145 2. “FibroGen and Astellas announce initiation of phase 3 trial of FG-4592/ASP1517 for treatment of anemia of chronic kidney disease” Fibrogen Press Release. Dec 11 2012
3. “FibroGen announces initiation of phase 2b studies of FG-4592, an oral HIF prolyl hydroxylase inhibitor, for treatment of anemia”
|
- Originator FibroGen
- Developer Astellas Pharma; AstraZeneca; FibroGen
- Class Amides; Antianaemics; Carboxylic acids; Isoquinolines; Small molecules
- Mechanism of Action Basic helix loop helix transcription factor modulators; Hypoxia-inducible factor-proline dioxygenase inhibitors
- Phase III Anaemia
- Discontinued Sickle cell anaemia
Most Recent Events
- 09 Jun 2016 Phase-III clinical trials in Anaemia in Japan (PO)
- 20 May 2016 In collaboration with FibroGen, Astellas Pharma plans a phase III trial for Anaemia (In chronic kidney disease patients undergoing peritoneal dialysis) in Japan (PO) (NCT02780726)
- 19 May 2016 In collaboration with FibroGen, Astellas Pharma plans a phase III trial for Anaemia (In erythropoiesis stimulating agent-naive, chronic kidney disease patients undergoing haemodialysis) in Japan (PO) (NCT02780141)


Roxadustat (FG-4592) is a novel new-generation oral hypoxia-induciblefactor (HIF) prolyl hydroxylase inhibitor (PHI) for the treatment of ane-mia in patients with chronic kidney disease (CKD). HIF is a cytosolic tran-scription factor that induces the natural physiological response to lowoxygen conditions, by stimulating erythropoiesis and other protectivepathways. Roxadustat has been shown to stabilize HIF and induce ery-thropoiesis. Consequently, it corrects anemia and maintains hemoglo-bin levels without the need for intravenous iron supplementation in CKDpatients not yet receiving dialysis and in end-stage renal disease pa-tients receiving dialysis. There are many concerns about the use of ery-thropoiesis-stimulating agents (ESA) to treat anemia as they causesupra-physiologic circulating erythropoietin (EPO) levels and are asso-ciated with adverse cardiovascular effects and mortality. Available clin-ical data show that modest and transient increases of endogenous EPOinduced by HIF-PHI (10- to 40-fold lower than ESA levels) are sufficientto mediate erythropoiesis in CKD patients. Evidence suggests that rox-adustat is well tolerated and, to date, no increased risk of cardiovascu-lar events has been found. This suggests that roxadustat provides adistinct pharmacological and clinical profile that may provide a saferand more convenient treatment of CKD anemia
FG-4592 is a new-generation hypoxia-inducible factor prolyl hydroxylase inhibitor in early clinical trials at FibroGen for the oral treatment of iron deficiency anemia and renal failure anemia. Preclinical studies are ongoing for the treatment of sickle cell anemia.
The investigational therapy is designed to restore balance to the body’s natural process of erythropoiesis through mechanisms including: natural EPO production, suppression of the effects of inflammation, downregulation of the iron sequestration hormone hepcidin, and an upregulation of other iron genes, ensuring efficient mobilization and utilization of the body’s own iron stores. In April 2006, FG-4592 was licensed to Astellas Pharma by originator FibroGen in Asia, Europe and South Africa for the treatment of anemia. FibroGen retains rights in the rest of the world. In 2007, the FDA put the trial on clinical hold due to one case of death by fulminant hepatitis during a phase II clinical trial for patients with anemia associated with chronic kidney disease and not requiring dialysis. However, in 2008, the FDA informed the company that clinical trials could be resumed. Phase II/III clinical trials for this indication resumed in 2012. In 2013, the compound was licensed to AstraZeneca by FibroGen for development and marketing in US, CN and all major markets excluding JP, Europe, the Commonwealth of Independent States, the Middle East and South Africa, for the treatment of anemia associated with chronic kidney disease (CKD) and end-stage renal disease (ESRD).
PATENTS
WO 2004108681
WO 2008042800
WO 2009058403
WO 2009075822
WO 2009075824
WO 2012037212
WO 2013013609
WO 2013070908
PATENT
CN 104892509
MACHINE TRANSLATED
Connaught orlistat (Roxadustat) by the US company Phibro root (FibroGen) R & D, Astellas AstraZeneca and licensed by a hypoxia-inducible factor (HIF) prolyl hydroxylase small molecule inhibitors, codenamed FG-4592.As a first new oral drug, FG-4592 is currently in Phase III clinical testing stage, for the treatment of chronic kidney disease and end-stage renal disease related anemia. Because the drug does not have a standard Chinese translation, so the applicant where it is transliterated as “Connaught Secretary him.”
Connaught orlistat (Roxadustat, I) the chemical name: N_ [(4- hydroxy-1-methyl-7-phenoxy-3-isoquinolinyl) carbonyl] glycine, its structural formula is:
The original research company’s international patent W02004108681 Division provides a promise he was prepared from the intermediate and intermediate Connaught Secretary for his synthetic route:
Zhejiang Beida company’s international patent W02013013609 preparation and acylation of core intermediate was further optimized synthesis route is:
n PhO. eight XOOH
original research company’s international patent W02014014834 and W02014014835 also provides another synthetic route he Connaught Secretary prepared:
Analysis of the above synthetic route, although he continued to Connaught Division to improve and optimize the synthesis, but its essence rings manner that different form quinoline ring is basically the same mother. Especially methyl isoquinoline replaced either by way of introducing the Suzuki reaction catalyzed by a noble metal element, either through amine reduction achieved. Moreover, the above reaction scheme revelation raw materials are readily available, many times during the reaction need to be protected and then deprotected. Clearly, the preparation process is relatively complicated, high cost, industrial production has brought some difficulties.
Example One:
tyrosine was added to the reaction flask and dried (18. lg, 0.1 mmol) and methanol 250mL, cooling to ice bath 0_5 ° C, was added dropwise over 1 hour a percentage by weight of 98% concentrated sulfuric acid 10g. Drops Albert, heating to reflux. The reaction was stirred for 16-20 hours, TLC the reaction was complete. Concentrated under atmosphere pressure, the residue was added water 100mL, using 10% by weight sodium hydroxide to adjust the pH to 6. 5-7.0, precipitated solid was filtered, washed with methanol and water chloro cake (I: 1) and dried in vacuo tyrosine methyl ester as a white solid (11) 15.38, yield 78.5% out 1–] \ ^ 111/2: 196 [] \ 1 + 1] +!.
Example Two:
[0041] a nitrogen atmosphere and ice bath, was added to the reaction flask tyrosine methyl ester (II) (9. 8g, 50mmol), potassium methoxide (3. 5g, 50mmol) and methanol 50mL, until no gas generation after, was heated to reflux, the reaction was stirred for 2 hours. Concentrated under atmosphere pressure to remove the solvent, the residue was added dimethylsulfoxide 25mL, freshly prepared copper powder (0.2g, 3. Lmmol), was slowly warmed to 150-155 ° C, for about half an hour later, a solution of bromobenzene ( 7. 9g, 50mmol), continue to heat up to 170-175 ° C, the reaction was stirred for 3 hours, TLC detection of the end of the reaction. Was cooled to 60 ° C, and methanol was added to keep micro-boiling, filtered while hot, the filter cake washed three times with hot ethanol, and the combined organic phases, was cooled to square ° C, filtered, and dried in vacuo to give a white solid of 2-amino-3- ( 4-phenoxyphenyl) propanoate (111) 8 11.5, yield 84.9% as 1 -] \ ^ 111/2:! 272 [] \ 1 + 1] +.
Example Three:
in the reaction flask was added 2-amino-3- (4-phenoxyphenyl) propionic acid methyl ester (III) (10. 8g, 40mmol), 40% by weight acetaldehyde (20g, 0. 2mol ) and the percentage by weight of 35% concentrated hydrochloric acid 50mL, refluxed for 1 hour. Continue 40% by weight was added acetaldehyde (10g, 0.1mol), and the percentage by weight of 35% concentrated hydrochloric acid 25mL, and then the reaction was refluxed for 3-5 hours. Was cooled to 4-7 ° C, ethyl acetate was added, and extracted layers were separated. The aqueous layer was adjusted with sodium hydroxide solution to pH 11-12, extracted three times with ethyl acetate. The combined organic phase was dried over anhydrous sodium sulfate, and concentrated under reduced pressure to give a white solid of 1-methyl-3-carboxylate -7- phenoxy-1,2,3,4-tetrahydroisoquinoline (IV) 8 4g, 70.7% yield; Mass spectrum (EI): EI-MS m / z: 298 [M + H] + .
Example Four:
Under ice bath, the reaction flask was added methyl 3-carboxylate I- -7- phenoxy-1,2, 3,4-tetrahydro-isoquinoline (IV) (5. 9g, 20mmol) and dichloromethane 100mL, 0 ° C and under stirring added potassium carbonate (13. 8g, 0. lmol), p-toluenesulfonyl chloride (11. 4g, 60mmol), the addition was completed, the ice bath was removed and stirred at room temperature 3 hour. Water was added 30mL, after stirring standing layer, the organic phase was washed with dilute hydrochloric acid, water and saturated brine, and concentrated, the resulting product was added a 30% by weight sodium hydroxide solution (8. 0g, 60mmol) and dimethyl sulfoxide 60mL, gradually warming to 120-130 ° C, the reaction was stirred for 2-4 hours to complete the reaction by TLC. Cooled to room temperature, water was added lOOmL, extracted three times with ethyl acetate, the combined organic phase was successively washed with water and saturated brine, dried over anhydrous magnesium sulfate, and concentrated, the resulting oil was treated with ethyl acetate and n-hexane (1: 3) recrystallization, vacuum dried to give an off-white solid 1-methyl-3-carboxylate 7-phenoxyheptanoic isoquinoline (V) 5. 25g, yield 89. 6%; EI-MS m / z: 294 [M + H] VH NMR (DMS0-d6) δ 2. 85 (s, 3H), 3 · 97 (s, 3H), 7 · 16-7. 24 (m, 3H), 7 · 49-7. 60 (m, 4Η), 8 · 35 (d, J = 9 · 0,1Η), 8 · 94 (s, 1Η).
Example five:
[0047] added 1-methyl-3-carboxylic acid methyl ester 7-phenoxyheptanoic isoquinoline (V) (2. 93g, IOmmol) and glacial acetic acid 50mL reaction flask, stirring solution of 30% by weight hydrogen peroxide 5mL, warmed to 60-70 ° C, was slowly added dropwise within 10 hours the percentage by weight of a mixture of 30% hydrogen peroxide 2mL and 12mL of glacial acetic acid, a dropping was completed, the reaction was continued for 20-24 hours. Concentrated under reduced pressure, ethanol was added, distillation is continued to be divisible remaining glacial acetic acid. The residue was dissolved with dichloromethane, washed with 5% by weight of sodium bicarbonate, the organic phase was separated, dried over anhydrous sodium sulfate. Filtered and the resulting solution was added p-toluenesulfonyl chloride (3. 8g, 20mmol), was heated to reflux, the reaction was stirred for 3-4 hours, TLC detection completion of the reaction. The solvent was distilled off under reduced pressure, cooled to room temperature, methanol was added, the precipitated solid, cooled to square ° C, allowed to stand overnight. Filtered, the filter cake washed twice with cold methanol and vacuum dried to give an off-white solid 1- methyl-3-methyl-4-hydroxy-phenoxy-isoquinoline -7- (VI) I. 86g, yield 60.2 %; EI-MS m / z:.. 310 [M + H] +, 1H NMR (DMS0-d6) δ 2.90 (s, 3H), 4.05 (s, 3H), 7 17-7 26 (m, 3H ), 7. 49-7. 61 (m, 4H), 8. 38 (d, J = 9. 0,1H), 11. 7 (s, 1H) 〇
Example VI:
in the reaction flask with magnetic stirring and pressure to join I- methyl-3-methyl-4-hydroxy-7-phenoxyheptanoate isoquinoline (VI) (1.55g, 5mmol), glycine (I. 13g, 15mmol) and sodium methoxide (3. 25g, 6mmol) in methanol (30mL).Sealed, slowly heated to 120 ° C, the reaction was stirred for 8-10 hours to complete the reaction by TLC. Cooled to room temperature, solid precipitated. Filtration, and the resulting solid was recrystallized from methanol, acetone and then beating the resulting solid was dried under vacuum to give a white solid Connaught orlistat 1.40g, yield 79.5%;
EI-MS m / z: 353 [M + H] +,
1H NMR (DMS0-d6) S2.72 (s, 3H), 3 · 99 (d, J = 6 · 0, 2H), 7 · 18-7. 28 (m, 3H), 7 · 49-7. 63 (m, 4H), 8 · 31 (d, J = 8 · 8,1H), 9 · 08 (s, lH), 13.41 (brs, lH).
PATENT

Example 10. Preparation of Compound A
a) 5-Phenoxyphthalide
[0200] A reactor was charged with DMF (68 Kg), and stirring was initiated. The reactor was then charged with phenol (51 Kg), acetylacetone (8 Kg), 5-bromophthalide (85 Kg), copper bromide (9 Kg), and potassium carbonate (77 Kg). The mixture was heated above 85 °C and maintained until reaction completion and then cooled. Water was added. Solid was filtered and washed with water. Solid was dissolved in dichloromethane, and washed with aqueous HCl and then with water. Solvent was removed under pressure and methanol was added. The mixture was stirred and filtered. Solid was washed with methanol and dried in an oven giving 5- phenoxyphthalide (Yield: 72%, HPLC: 99.6%). b) 2-Chloromethyl-4-phenoxybenzoic acid methyl ester
[0201] A reactor was charged with toluene (24 Kg), and stirring was initiated. The reactor was then charged with 5-phenoxyphthalide (56 Kg), thionyl chloride (41 Kg), trimethyl borate (1
Kg), dichlorotriphenylphosphorane (2.5 Kg), and potassium carbonate (77 Kg). The mixture was heated to reflux until reaction completion and solvent was removed leaving 2-chloromethyl-4- phenoxybenzoyl chloride. Methanol was charged and the mixture was heated above 50 °C until reaction completion. Solvent was removed and replaced with DMF. This solution of the product methyl 2-chloromethyl-4-phenoxybenzoic acid methyl ester in DMF was used directly in the next step (HPLC: 85%). c) 4-Hydroxy-7-phenoxyisoquinoline-3-carboxylic acid methyl ester (la)
[0202] A reactor was charged with a solution of 2-chloromethyl-4-phenoxybenzoic acid methyl ester (~68 Kg) in DMF, and stirring was initiated. The reactor was then charged with p- toluenesulfonylglycine methyl ester (66 Kg), potassium carbonate (60 Kg), and sodium iodide (4 Kg). The mixture was heated to at least 50 °C until reaction completion. The mixture was cooled. Sodium methoxide in methanol was charged and the mixture was stirred until reaction completion. Acetic acid and water were added, and the mixture was stirred, filtered and washed with water. Solid was purified by acetone trituration and dried in an oven giving la (Yield from step b): 58%; HPLC: 99.4%). 1H NMR (200 MHz, DMSO-d6) δ 11.60 (s, 1 H), 8.74 (s, 1H),
8.32 (d, J = 9.0 Hz, 1 H), 7.60 (dd, J = 2.3 & 9.0 Hz, 1H), 7.49 (m, 3 H), 7.24 (m, 3 H), 3.96 (s, 3 H); MS-(+)-ion M+l = 296.09 d) Methyl l-((dimethylamino)methyl)-4-hydroxy-7-phenoxyisoquinoline-3-carboxylate
(lb)
[0203] A flask was charged with la (29.5 g) and acetic acid (44.3 g ± 5%), and then stirred. Bis-dimethylaminomethane (12.8 g ± 2%) was slowly added. The mixture was heated to 55 ± 5 °C and maintained until reaction completion. The reaction product was evaluated by MS, HPLC and 1H NMR. 1H NMR (200 MHz, DMSO-d6) δ 11.7 (s, 1 H), 8.38 (d, J = 9.0 Hz, 1 H), 7.61 (dd, J = 9.0, 2.7 Hz, 1 H), 7.49 (m, 3 H), 7.21 (m, 3 H), 5.34 (s, 2 H), 3.97 (s, 3 H), 1.98 (s, 3 H); MS-(+)-ion M+l = 368.12. e) Methyl l-((acetoxy)methyl)-4-hydroxy-7-phenoxyisoquinoline-3-carboxylate (lc)
[0204] The solution of lb from a) above was cooled below 25 °C, at which time acetic anhydride (28.6 g ± 3.5 %) was added to maintain temperature below 50 °C. The resulting mixture was heated to 100 ± 5 °C until reaction completion.
[0205] The solution of lc and Id from above was cooled to less than 65 ± 5 °C. Water (250 mL) was slowly added. The mixture was then cooled to below 20 ± 5 °C and filtered. The wet cake was washed with water (3 x 50 mL) and added to a new flask. Dichloromethane (90 mL) and water (30 mL) were added, and the resulting mixture was stirred. The dichloromethane layer was separated and evaluated by HPLC.
[0206] The organic layer was added to a flask and cooled 5 ± 5 °C. Morpholine was added and the mixture was stirred until reaction completion. Solvent was replaced with acetone/methanol mixture. After cooling, compound lc precipitated and was filtered, washed and dried in an oven (Yield: 81%, HPLC: >99.7%). 1H NMR (200 MHz, DMSO-d6) δ 11.6 (S, 1 H), 8.31 (d, J = 9.0 Hz, 1 H), 7.87 (d, J = 2.3 Hz, 1 H), 7.49 (m, 3 H), 7.24 (m, 3 H), 3.95 (s, 3 H), 3.68 (s, 2H), 2.08 (s, 6 H); MS-(+)-ion M+l = 357.17. f) Methyl 4-hydroxy-l-methyl-7-phenoxyisoquinoline-3-carboxylate (le)
[0207] A reactor was charged with lc (16.0 g), Pd/C (2.08 g), anhydrous Na2C03 (2.56 g) and ethyl acetate (120 mL). The flask was vacuum-purged with nitrogen (3X) and vacuum-purged with hydrogen (3X). The flask was then pressurized with hydrogen and stirred at about 60 °C until completion of reaction. The flask was cooled to 20-25 °C, the pressure released to ambient, the head space purged with nitrogen three times and mixture was filtered. The filtrate was concentrated. Methanol was added. The mixture was stirred and then cooled. Product precipitated and was filtered and dried in an oven (Yield: 90%, HPLC: 99.7%). g) [(4-Hydroxy-l-methyl-7-phenoxy-isoquinoline-3-carbonyl)-amino]-acetic acid
(Compound A)
[0208] A pressure flask was charged with le (30.92 g), glycine (22.52 g), methanol (155 mL), sodium methoxide solution (64.81 g) and sealed (as an alternative, sodium glycinate was used in place of glycine and sodium methoxide). The reaction was heated to about 110 °C until reaction was complete. The mixture was cooled, filtered, washed with methanol, dried under vacuum, dissolved in water and washed with ethyl acetate. The ethyl acetate was removed and to the resulting aqueous layer an acetic acid (18.0 g) solution was added. The suspension was stirred at room temperature, filtered, and the solid washed with water (3 x 30 mL), cold acetone (5-10 °C, 2 x 20 mL), and dried under vacuum to obtain Compound A (Yield: 86.1%, HPLC: 99.8%). Example 11. Biological Testing
[0209] The solid forms provided herein can be used for inhibiting HIF hydroxylase activity, thereby increasing the stability and/or activity of hypoxia inducible factor (HIF), and can be used to treat and prevent HIF-associated conditions and disorders (see, e.g., U.S. Patent No. 7,323,475, U.S. Patent Application Publication No. 2007/0004627, U.S. Patent Application Publication No. 2006/0276477, and U.S. Patent Application Publication No. 2007/0259960, incorporated by reference herein).
SYNTHESIS……..http://zliming2004.lofter.com/post/1cc9dc55_79ad5d8

Condensation of 5-bromophthalide (I) with phenol (II) in the presence of K2CO3, CuBr and acetylacetone in DMF gives 5-phenoxyphthalide (III), which upon lactone ring opening using SOCl2, Ph3PCl2, B(OMe)3 and K2CO3 in refluxing toluene yields 2-chloromethyl-4-phenoxybenzoyl chloride (IV). Esterification of acid chloride (IV) with MeOH at 50 °C furnishes the methyl ester (V), which is then condensed with methyl N-tosylglycinate (VI) in the presence of K2CO3 and NaI in DMF at 50 °C to afford N-substituted aminoester (VII). Cyclization of the intermediate diester (VII) using NaOMe in MeOH leads to methyl 4-hydroxy-7-phenoxyisoquinoline-3-carboxylate (VIII), which is submitted to Mannich reaction with bis-dimethylaminomethane (IX) in the presence of AcOH at 57 °C to provide the dimethylaminomethyl compound (X). Treatment of amine (X) with Ac2O at 103 °C, followed by selective hydrolysis of the phenolic acetate with morpholine leads to methyl 1-acetoxymethyl-4-hydroxy-7-phenoxyisoquinoline-3-carboxylate (XI). Hydrogenolysis of the benzylic acetate (XII) in the presence of Pd/C and Na2CO3 in EtOAc yields methyl 4-hydroxy-1-methyl-7-phenoxyisoquinoline-3-carboylate (XII), which finally couples with glycine (XIII) in the presence of NaOMe in MeOH at 110 °C to afford the target roxadustat (1-3).

Cyclization of 4-phenoxyphthalic acid (I) with glycine (II) at 215 °C gives the phthalimide (III), which upon esterification with MeOH and H2SO4 at reflux yields methyl ester (IV). Subsequent rearrangement of phthalimidoacetate (IV) by means of Na in BuOH at 97 °C, followed by flash chromatography provides the isoquinoline-2-carboxylate (V). Bromination of intermediate (V) using POBr3 and NaHCO3 in acetonitrile leads to butyl 8-bromo-3-hydroxy-6-phenoxy-isoquinoline-2-carboxylate (VI), which upon hydrolysis with NaOH in refluxing H2O/EtOH furnishes carboxylic acid (VII). Substitution of bromine in intermediate (VII) using MeI and BuLi in THF at -78 °C, followed by alkylation with PhCH2Br in the presence of K2CO3 in refluxing acetone affords the 2-methyl isoquinoline (VIII). Ester hydrolysis in intermediate (VIII) using KOH in MeOH gives the corresponding carboxylic acid (IX), which is then activated with i-BuOCOCl and Et3N in CH2Cl2, followed by coupling with benzyl glycinate hydrochloride (X) to yield benzylated roxadustat (XI). Finally, debenzylation of intermediate (XI) with H2 over Pd/C in EtOAc/MeOH provides the title compound (1).

Condensation of 4-nitro-ortho-phthalonitrile (I) with phenol (II) in the presence of K2CO3 in DMSO gives 4-phenoxy-ortho-phthalonitrile (III) (1), which upon hydrolysis with NaOH (1) or KOH (2) in refluxing MeOH yields 4-phenoxyphthalic acid (IV) (1,2). Dehydration of dicarboxylic acid (IV) using Ac2O and AcOH at reflux furnishes the phthalic anhydride (V), which is then condensed with methyl 2-isocyanoacetate (VI) using DBU in THF to provide oxazole derivative (VII). Rearrangement of intermediate (VII) with HCl in MeOH at 60 °C leads to isoquinoline derivative (VIII), which is partially chlorinated by means of POCl3 at 70 °C to afford 1-chloro-isoquinoline derivative (IX). Substitution of chlorine in intermediate (IX) using Me3B, Pd(PPh3)4 and K2CO3 in refluxing dioxane gives methyl 4-hydroxy-1-methyl-7-phenoxy-3-carboxylate (X), which is then hydrolyzed with aqueous NaOH in refluxing EtOH to yield the carboxylic acid (XI). Coupling of carboxylic acid (XI) with methyl glycinate hydrochloride (XII) by means of PyBOP, (i-Pr)2NH and Et3N in CH2Cl2 yields roxadustat methyl ester (XII), which is finally hydrolyzed with aqueous NaOH in THF to afford the target roxadustat (1).
CLIPS

References
1: Besarab A, Provenzano R, Hertel J, Zabaneh R, Klaus SJ, Lee T, Leong R, Hemmerich S, Yu KH, Neff TB. Randomized placebo-controlled dose-ranging and pharmacodynamics study of roxadustat (FG-4592) to treat anemia in nondialysis-dependent chronic kidney disease (NDD-CKD) patients. Nephrol Dial Transplant. 2015 Oct;30(10):1665-73. doi: 10.1093/ndt/gfv302. Epub 2015 Aug 3. PubMed PMID: 26238121; PubMed Central PMCID: PMC4569392.
2: Forristal CE, Levesque JP. Targeting the hypoxia-sensing pathway in clinical hematology. Stem Cells Transl Med. 2014 Feb;3(2):135-40. doi: 10.5966/sctm.2013-0134. Epub 2013 Dec 26. PubMed PMID: 24371328; PubMed Central PMCID: PMC3925058.
3: Bouchie A. First-in-class anemia drug takes aim at Amgen’s dominion. Nat Biotechnol. 2013 Nov;31(11):948-9. doi: 10.1038/nbt1113-948b. PubMed PMID: 24213751.
4: Flight MH. Deal watch: AstraZeneca bets on FibroGen’s anaemia drug. Nat Rev Drug Discov. 2013 Oct;12(10):730. doi: 10.1038/nrd4135. PubMed PMID: 24080688.
5: Beuck S, Schänzer W, Thevis M. Hypoxia-inducible factor stabilizers and other small-molecule erythropoiesis-stimulating agents in current and preventive doping analysis. Drug Test Anal. 2012 Nov;4(11):830-45. doi: 10.1002/dta.390. Epub 2012 Feb 24. Review. PubMed PMID: 22362605.
6: Cases A. The latest advances in kidney diseases and related disorders. Drug News Perspect. 2007 Dec;20(10):647-54. PubMed PMID: 18301799.
Novartis, Torrent drug for diabetes, NVP-LBX192, LBX-192

(R)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
(3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide)
(R)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
cas 866772-52-3
NVP-LBX192
LBX-192
R(−) 3-cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
R(−)17c BELOW

| Inventors | Gregory Raymond Bebernitz, Ramesh Chandra Gupta, Vikrant Vijaykumar Jagtap, Appaji Baburao Mandhare, Davinder Tuli, |
| Original Assignee | Novartis Ag
|
| Molecular Formula: | C26H33N5O4S2 |
|---|---|
| Molecular Weight: | 543.70132 g/mol |


LBX192, also known as NVP-LBX192, is a Liver Targeted Glucokinase Activator. LBX192 activated the GK enzyme in vitro at low nM concentrations and significantly reduced glucose levels during an oral glucose tolerance test in normal as well as diabetic mice. A GK activator has the promise of potentially affecting both the beta-cell of the pancreas, by improving glucose sensitive insulin secretion, as well as the liver, by reducing uncontrolled glucose output and restoring post prandial glucose uptake and storage as glycogen.
SYNTHESIS BY WORLDDRUGTRACKER
54 Discovery and Evaluation of NVP-LBX192, a Liver Targeted Glucokinase Activator
https://acs.confex.com/acs/nerm09/webprogram/Paper75087.html
Sulfonamide-Thiazolpyridine Derivatives, Glucokinase Activators, Treatment Of Type 2 Diabetes
2009 52 (19) 6142 – 6152
Investigation of functionally liver selective glucokinase activators for the treatment of type 2 diabetes
Journal of Medicinal Chemistry
Bebernitz GR, Beaulieu V, Dale BA, Deacon R, Duttaroy A, Gao JP, Grondine MS, Gupta RC, Kakmak M, Kavana M, Kirman LC, Liang JS, Maniara WM, Munshi S, Nadkarni SS, Schuster HF, Stams T, Denny IS, Taslimi PM, Vash B, Caplan SL
2010 240th (August 22) Medi-198
Glucokinase activators with improved physicochemicalproperties and off target effects
American Chemical Society National Meeting and Exposition
Kirman LC, Schuster HF, Grondine MS et al
2010 240th (August 22) Medi-197
Investigation of functionally liver selective glucokinase activators
American Chemical Society National Meeting and Exposition
Schuster HF, Kirman LC, Bebernitz GC et al
PATENT
http://www.google.com/patents/US7750020
EXAMPLE 1 3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
A. Phenylacetic Acid Ethyl Ester
A solution of phenylacetic acid (50 g, 0.36 mol) in ethanol (150 mL) is treated with catalytic amount of sulfuric acid (4 mL). The reaction mixture is refluxed for 4 h. The reaction is then concentrated in vacuo. The residue is dissolved in diethyl ether (300 mL) and washed with saturated aqueous sodium bicarbonate solution (2×50 mL) and water (1×100 mL). The organic layer dried over sodium sulfate filtered and concentrated in vacuo to give phenylacetic acid ethyl ester as a colorless oil: 1H NMR (400 MHz, CDCl3) δ 1.2 (t, J=7.2, 3H), 3.6 (s, 2H), 4.1 (q, J=7.2, 2H), 7.3 (m, 5H); MS 165 [M+1]+.
B. (4-Chlorosulfonyl-phenyl)-acetic acid ethyl ester
To a cooled chlorosulfonic acid (83.83 g, 48 mL, 0.71 mol) under nitrogen is added the title A compound, phenylacetic acid ethyl ester (59 g, 0.35 mol) over a period of 1 h. Reaction temperature is brought to RT (28° C.), then heated to 70° C., maintaining it at this temperature for 1 h while stirring. Reaction is cooled to RT and poured over saturated aqueous sodium chloride solution (200 mL) followed by extraction with DCM (2×200 mL). The organic layer is washed with water (5×100 mL), followed by saturated aqueous sodium chloride solution (1×150 mL). The organic layer dried over sodium sulfate, filtered and concentrated in vacuo to give crude (4-chlorosulfonyl-phenyl)acetic acid ethyl ester. Further column chromatography over silica gel (60-120 mesh), using 100% hexane afforded pure (4-chlorosulfonyl-phenyl)-acetic acid ethyl ester as a colorless oil.
C. [4-(4-Methyl-piperazine-1-sulfonyl)-phenyl]-acetic acid ethyl ester
A solution of N-methylpiperazine (9.23 g, 10.21 ml, 0.092 mol), DIEA (13 g, 17.4 mL, 0.10 mol) and DCM 80 mL is cooled to 0° C., and to this is added a solution of the title B compound, (4-chlorosulfonyl-phenyl)-acetic acid ethyl ester (22 g, 0.083 mol) in 50 mL of DCM within 30 min. Reaction mixture stirred at 0° C. for 2 h, and the reaction mixture is washed with water (100 mL), followed by 0.1 N aqueous hydrochloric acid solution (1×200 mL). The organic layer dried over sodium sulfate, filtered and concentrated under vacuo to give crude [4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-acetic acid ethyl ester. Column chromatography over silicagel (60-120 mesh), using ethyl acetate afforded pure [4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-acetic acid ethyl ester as white crystalline solid: 1H NMR (400 MHz, CDCl3) δ 1.3 (t, J=7.4, 3H), 2.3 (s, 3H), 2.5 (m, 4H), 3.0 (br s, 4H), 3.7 (s, 2H), 4.2 (q, J=7.4, 2H), 7.4 (d, J=8.3, 2H), 7.7 (d, J=7.3, 2H); MS 327 [M+1]+.
D. 3-Cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid ethyl ester
A solution of the title C compound, [4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-acetic acid ethyl ester (15 g, 0.046 mol) in a mixture of THF (60 mL) and DMTP (10 mL) is cooled to −78° C. under nitrogen. The resulting solution is stirred at −78° C. for 45 min and to this is added LDA (25.6 mL, 6.40 g, 0.059 mol, 25% solution in THF/Hexane). A solution of iodomethylcyclopentane (11.60 g, 0.055 mol) in a mixture of DMTP (12 mL) and THF (20 mL) is added over a period of 15 min at −78° C. and reaction mixture stirred at −78° C. for 3 h further, followed by stirring at 25° C. for 12 h. The reaction mixture is then quenched by the dropwise addition of saturated aqueous ammonium chloride solution (50 mL) and is concentrated in vacuo. The residue is diluted with water (50 mL) and extracted with ethyl acetate (3×100 mL). The organic solution is washed with a saturated aqueous sodium chloride (2×150 mL), dried over sodium sulfate, filtered and concentrated in vacuo. Column chromatography over silica gel (60-120 mesh), using 50% ethyl acetate in hexane as an eluent to afford 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid ethyl ester as a white solid: 1H NMR (400 MHz, CDCl3) δ 0.9-2.1 (m, 11H), 1.2 (t, J=7.1, 3H), 2.3 (s, 3H), 2.5 (br s, 4H), 3.0 (br s, 4H), 3.6 (m, 1H), 4.1 (q, J=7.1, 2H), 7.5 (d, J=8.3, 2H), 7.7 (d, J=8.3, 2H); MS 409 [M+1]+.
E. 3-Cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid
A solution of the title D compound, 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid ethyl ester (14 g, 0.034 mol) in methanol:water (30 mL:10 mL) and sodium hydroxide (4.11 g, 0.10 mol) is stirred at 60° C. for 8 h in an oil bath. The methanol is then removed in vacuo at 45-50° C. The residue is diluted with water (25 mL) and extracted with ether (1×40 mL). The aqueous layer is acidified to pH 5 with 3 N aqueous hydrochloric acid solution. The precipitated solid is collected by vacuum filtration, washed with water (20 mL), followed by isopropyl alcohol (20 mL). Finally, solid cake is washed with 100 mL of hexane and dried under vacuum at 40° C. for 6 h to give 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid as a white solid: 1H NMR (400 MHz, CDCl3) δ 1.1-2.0 (m, 11H), 2.4 (s, 3H), 2.7 (br s, 4H), 3.1 (br s, 4H), 3.6 (m, 1H), 7.5 (d, J=8.3, 2H), 7.6 (d, J=8.3, 2H); MS 381 [M+l]+.
F. 5-Methoxy-thiazolo[5,4-b]pyridin-2-ylamine
A solution of 6-methoxy-pyridin-3-ylamine (5.0 g, 0.0403 mol) in 10 mL of acetic acid is added slowly to a solution of potassium thiocyanate (20 g, 0.205 mol) in 100 mL of acetic acid at 0° C. followed by a solution of bromine (2.5 mL, 0.0488 mol) in 5 mL of acetic acid. The reaction is stirred for 2 h at 0° C. and then allowed to warm to RT. The resulting solid is collected by filtration and washed with acetic acid, then partitioned between ethyl acetate and saturated aqueous sodium bicarbonate. The insoluble material is removed by filtration and the organic layer is evaporated and dried to afford 5-methoxy-thiazolo[5,4-b]pyridin-2-ylamine as a tan solid.
G. 3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
A solution of the title E compound, 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid (5 g, 0.013 mol) in DCM (250 mL) is cooled to 0° C. and then charged HOBt hydrate (2.66 g, 0.019 mol), followed by EDCI hydrochloride (6 g, 0.031 mol). The reaction mixture is stirred at 0° C. for 5 h. After that the solution of the title F compound, 5-methoxy-thiazolo[5,4-b]pyridin-2-ylamine (2.36 g, 0.013 mol) and D1EA (8 mL, 0.046 mol) in a mixture of DCM (60 mL) and DMF (20 mL) is added dropwise over 30 min. Reaction temperature is maintained at 0° C. for 3 h, then at RT (28° C.) for 3 days. Reaction is diluted with (60 mL) of water and the organic layer is separated and washed with saturated sodium bicarbonate solution (2×50 mL) followed by water washing (2×50 mL) and saturated sodium chloride aqueous solution (1×150 mL). Finally the organic layer is dried over sodium sulfate, filtered, and evaporated under vacuo. The crude product is purified using column chromatography over silica gel (60-120 mesh), using 40% ethyl acetate in hexane as an eluent to afford 3-cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide as a white solid: 1H NMR (400 MHz, CDCl3) δ 0.9-2.1 (m, 11H), 2.2 (s, 3H), 2.5 (br s, 4H), 3.1 (br s, 4H), 3.7 (m, 1H), 4.0 (s, 3H), 6.8 (d, J=8.8, 1H), 7.5 (d, J=8.3, 2H), 7.7 (d, J=8.3, 2H), 7.8 (d, J=8.8, 1H), 8.6 (s, 1H); MS 617 [M+1]+.
H. 3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide dihydrochloride
The title G compound, 3-cyclopentyl-2-(4-methyl piperazinyl sulfonyl)phenyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)propionamide (2.8 g, 0.0051 mol) is added to a cooled solution of 10% hydrochloric acid in isopropanol (3.75 mL). The reaction mixture is stirred at 0° C. for 1 h and then at RT for 2 h. The solid is separated, triturated with 10 mL of isopropanol and collected by vacuum filtration and washed with 50 mL of hexane. The solid is dried at 70° C. for 48 h to afford 3-cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide dihydrochloride as an off white solid.
EXAMPLE 2 (R)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
The title compound is obtained analogously to Example 1 by employing the following additional resolution step:
The racemic title E compound of Example 1,3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid (10 g, 0.026 mol) in 1,4-dioxane (500 mL) is treated in a three necked 1 liter flask, equipped with heating mantle, water condenser, calcium chloride guard tube and mechanical stirrer with 3.18 g (0.026 mol) of (R)-(+)-1-phenylethylamine. This reaction mixture is then refluxed at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized salt is collected by filtration under vacuum, washed with 5 mL of hexane and dried under vacuum to afford salt A.
The salt A is dissolved in 1,4-dioxane (500 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 50 mL of hexane, and dried under vacuum to afford salt B.
The salt B is dissolved in 1,4-dioxane (290 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30 mL of hexane, and dried under vacuum to afford salt C.
The salt C is dissolved in 1,4-dioxane (100 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30 ml of hexane, and dried under vacuum to afford salt D.
The salt D is treated with aqueous hydrochloric acid solution (20 mL, 1 mL of concentrated hydrochloric acid diluted with 100 mL of water) and stirred for 5 min. The white solid precipitates out and is collected by vacuum filtration, washed with 10 mL of cold water, 5 mL of isopropanol and 20 mL of hexane, and dried under vacuum to yield the hydrochloride salt of (R)-(−)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid, salt E.
The salt E is neutralized by stirring with aqueous sodium bicarbonate solution (10 mL, 1 g of sodium bicarbonate dissolved in 120 mL of water) for 5 min. The precipitated solid is collected by filtration, washed with 10 mL of cold water, 100 mL of hexane, and dried to afford (R)-(−)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid: m.p. 202.2-203.4° C.
Alternatively, the title compound may be obtained by the resolution of the racemic title compound of Example 1 using the following preparative chiral HPLC method:
- Column: Chiralcel OD-R (250×20 mm) Diacel make, Japan;
- Solvent A: water:methanol:acetonitrile (10:80:10 v/v/v);
- Solvent B: water:methanol:acetonitrile (05:90:05 v/v/v);
- Using gradient elution: gradient program (time, min/% B): 0/0, 20/0, 50/100, 55/0, 70/0;
- Flow rate: 6.0 mL/min; and
- Detection: by UV at 305 nm.
EXAMPLE 3 (S)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
The title compound is prepared analogously to Example 2.
J MED CHEM 2009, 52, 6142-52
Investigation of Functionally Liver Selective Glucokinase Activators for the Treatment of Type 2 Diabetes
http://pubs.acs.org/doi/abs/10.1021/jm900839k

Type 2 diabetes is a polygenic disease which afflicts nearly 200 million people worldwide and is expected to increase to near epidemic levels over the next 10−15 years. Glucokinase (GK) activators are currently under investigation by a number of pharmaceutical companies with only a few reaching early clinical evaluation. A GK activator has the promise of potentially affecting both the β-cells of the pancreas, by improving glucose sensitive insulin secretion, as well as the liver, by reducing uncontrolled glucose output and restoring post-prandial glucose uptake and storage as glycogen. Herein, we report our efforts on a sulfonamide chemotype with the aim to generate liver selective GK activators which culminated in the discovery of 3-cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide (17c). This compound activated the GK enzyme (αKa = 39 nM) in vitro at low nanomolar concentrations and significantly reduced glucose levels during an oral glucose tolerance test in normal mice.
PATENT
EP-1735322-B1
Example 2(R)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
The title compound is obtained analogously to Example 1 by employing the following additional resolution step:
The racemic title E compound of Example 1, 3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid (10 g, 0.026 mol) in 1,4-dioxane (500 mL) is treated in a three necked 1 liter flask, equipped with heating mantle, water condenser, calcium chloride guard tube and mechanical stirrer with 3.18 g (0.026 mol) of (R)-(+)-1-phenylethylamine. This reaction mixture is then refluxed at 100°C for 1 h. The clear reaction solution is cooled to RT (27°C) and stirred for 10 h. The crystallized salt is collected by filtration under vacuum, washed with 5 mL of hexane and dried under vacuum to afford salt A.
The salt A is dissolved in 1,4-dioxane (500 mL) and heated at 100°C for 1 h. The clear reaction solution is cooled to RT (27°C) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 50 mL of hexane, and dried under vacuum to afford salt B.
The salt B is dissolved in 1,4-dioxane (290 mL) and heated at 100°C for 1 h. The clear reaction solution is cooled to RT (27°C) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30 mL of hexane, and dried under vacuum to afford salt C.
The salt C is dissolved in 1,4-dioxane (100 mL) and heated at 100°C for 1 h. The clear reaction solution is cooled to RT (27°C) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30ml of hexane, and dried under vacuum to afford salt D.
The salt D is treated with aqueous hydrochloric acid solution (20 mL, 1 mL of concentrated hydrochloric acid diluted with 100 mL of water) and stirred for 5 min. The white solid precipitates out and is collected by vacuum filtration, washed with 10 mL of cold water, 5 mL of isopropanol and 20 mL of hexane, and dried under vacuum to yield the hydrochloride salt of (R)-(-)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid, salt E.
The salt E is neutralized by stirring with aqueous sodium bicarbonate solution (10 mL, 1 g of sodium bicarbonate dissolved in 120 mL of water) for 5 min. The precipitated solid is collected by filtration, washed with 10 mL of cold water, 100 mL of hexane, and dried to afford (R)-(-)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid: m.p. 202.2-203.4°C.
Alternatively, the title compound may be obtained by the resolution of the racemic title compound of Example 1 using the following preparative chiral HPLC method:
- Column: Chiralcel OD-R (250 x 20 mm) Diacel make, Japan;
- Solvent A: water:methanol:acetonitrile (10:80:10 v/v/v);
- Solvent B: water:methanol:acetonitrile (05:90:05 v/v/v);
- Using gradient elution: gradient program (time, min / %B): 0/0, 20/0, 50/100, 55/0, 70/0;
- Flow rate: 6.0 mL/min; and
- Detection: by UV at 305 nm.
REFERENCES
US 7750020
WO-2005095418-A1
US-20080103167-A1
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015218151 | 2015-08-06 | NOVEL PHENYLACETAMIDE COMPOUND AND PHARMACEUTICAL CONTAINING SAME |
| US7750020 | 2010-07-06 | Sulfonamide-Thiazolpyridine Derivatives As Glucokinase Activators Useful The Treatment Of Type 2 Diabetes |

Type 2 diabetes is a polygenic disease which afflicts nearly 200 million people worldwide and is expected to increase to near epidemic levels over the next 10−15 years. Glucokinase (GK) activators are currently under investigation by a number of pharmaceutical companies with only a few reaching early clinical evaluation. A GK activator has the promise of potentially affecting both the β-cells of the pancreas, by improving glucose sensitive insulin secretion, as well as the liver, by reducing uncontrolled glucose output and restoring post-prandial glucose uptake and storage as glycogen. Herein, we report our efforts on a sulfonamide chemotype with the aim to generate liver selective GK activators which culminated in the discovery of 3-cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide (17c). This compound activated the GK enzyme (αKa = 39 nM) in vitro at low nanomolar concentrations and significantly reduced glucose levels during an oral glucose tolerance test in normal mice.
https://www.google.com/patents/US7750020
EXAMPLE 2 (R)-3-Cyclopentyl-N-(5-methoxy-thiazolo[5,4-b]pyridin-2-yl)-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionamide
The title compound is obtained analogously to Example 1 by employing the following additional resolution step:
The racemic title E compound of Example 1,3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid (10 g, 0.026 mol) in 1,4-dioxane (500 mL) is treated in a three necked 1 liter flask, equipped with heating mantle, water condenser, calcium chloride guard tube and mechanical stirrer with 3.18 g (0.026 mol) of (R)-(+)-1-phenylethylamine. This reaction mixture is then refluxed at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized salt is collected by filtration under vacuum, washed with 5 mL of hexane and dried under vacuum to afford salt A.
The salt A is dissolved in 1,4-dioxane (500 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 50 mL of hexane, and dried under vacuum to afford salt B.
The salt B is dissolved in 1,4-dioxane (290 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30 mL of hexane, and dried under vacuum to afford salt C.
The salt C is dissolved in 1,4-dioxane (100 mL) and heated at 100° C. for 1 h. The clear reaction solution is cooled to RT (27° C.) and stirred for 10 h. The crystallized product is collected by filtration under vacuum, washed with 30 ml of hexane, and dried under vacuum to afford salt D.
The salt D is treated with aqueous hydrochloric acid solution (20 mL, 1 mL of concentrated hydrochloric acid diluted with 100 mL of water) and stirred for 5 min. The white solid precipitates out and is collected by vacuum filtration, washed with 10 mL of cold water, 5 mL of isopropanol and 20 mL of hexane, and dried under vacuum to yield the hydrochloride salt of (R)-(−)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid, salt E.
The salt E is neutralized by stirring with aqueous sodium bicarbonate solution (10 mL, 1 g of sodium bicarbonate dissolved in 120 mL of water) for 5 min. The precipitated solid is collected by filtration, washed with 10 mL of cold water, 100 mL of hexane, and dried to afford (R)-(−)-3-cyclopentyl-2-[4-(4-methyl-piperazine-1-sulfonyl)-phenyl]-propionic acid: m.p. 202.2-203.4° C.
Alternatively, the title compound may be obtained by the resolution of the racemic title compound of Example 1 using the following preparative chiral HPLC method:
- Column: Chiralcel OD-R (250×20 mm) Diacel make, Japan;
- Solvent A: water:methanol:acetonitrile (10:80:10 v/v/v);
- Solvent B: water:methanol:acetonitrile (05:90:05 v/v/v);
- Using gradient elution: gradient program (time, min/% B): 0/0, 20/0, 50/100, 55/0, 70/0;
- Flow rate: 6.0 mL/min; and
- Detection: by UV at 305 nm.
| Patent ID | Date | Patent Title |
|---|---|---|
| US2015218151 | 2015-08-06 | NOVEL PHENYLACETAMIDE COMPOUND AND PHARMACEUTICAL CONTAINING SAME |
| US7750020 | 2010-07-06 | Sulfonamide-Thiazolpyridine Derivatives As Glucokinase Activators Useful The Treatment Of Type 2 Diabetes |
Torrent Research Centre, Village Bhat, Gujarat, India




Mr. Samir Mehta, 52, is the Vice Chairman of the USD 2.75 billion Torrent Group and Chairman of Torrent Pharma

Shri Sudhir Mehta – Chairman Emeritus ::
| Dr. Chaitanya Dutt – Director (Research & Development) :: |
 Born in the year 1950, Dr. Chaitanya Dutt holds an MD in Medicine. He practiced as a consulting physician before joining the company in 1982. Since then he has been associated with the Company. His rich experience spans in the areas of Pharma R&D, clinical research, manufacturing, quality assurance, etc. He is one of the key professionals in the top management team of the Company. He has been instrumental in setting up the Torrent Research Centre (TRC), the research wing of the Company. Under his prudent guidance and leadership, TRC has achieved tremendous progress in the areas of discovery research as well as development work on formulations. He does not hold any directorship in any other company. Born in the year 1950, Dr. Chaitanya Dutt holds an MD in Medicine. He practiced as a consulting physician before joining the company in 1982. Since then he has been associated with the Company. His rich experience spans in the areas of Pharma R&D, clinical research, manufacturing, quality assurance, etc. He is one of the key professionals in the top management team of the Company. He has been instrumental in setting up the Torrent Research Centre (TRC), the research wing of the Company. Under his prudent guidance and leadership, TRC has achieved tremendous progress in the areas of discovery research as well as development work on formulations. He does not hold any directorship in any other company. |
///NOVARTIS, DIABETES, Sulfonamide-Thiazolpyridine Derivatives, Glucokinase Activators, Treatment Of Type 2 Diabetes, 866772-52-3, Novartis Molecule, functionally liver selective glucokinase activators, treatment of type 2 diabetes , NVP-LBX192, LBX-192
c1(sc2nc(ccc2n1)OC)NC(C(c3ccc(cc3)S(=O)(=O)N4CCN(CC4)C)CC5CCCC5)=O
Final WHO Guidance Document on Good Data and Record Management Practices
DRUG REGULATORY AFFAIRS INTERNATIONAL

The WHO has just released the the final version of the important guideline “Good Data and Record Management Practices“.

We recently informed you about the WHO Draft Guidance on Good Data and Record Management Practices. Now, the WHO has just released the the final version of this important guideline “Good Data and Record Management Practices”.
The final version is sectioned rather similar to the draft version:
– Introduction
– Aims and objectives of this guidance
– Glossary
– Principles
– Quality risk management to ensure good data management
– Management governance and quality audits
– Contracted organizations, suppliers and service providers
– Training in good data and record management
– Good documentation practices
– Designing and validation systems to assure data quality and reliability
– Managing data and records throughout the data lifecycle
– Addressing data reliability issues
– References and further reading
Although the individual chapters were…
View original post 208 more words

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....























 THIS COMPD HAS ONE FLUORO MISSING, APPLY TO YOUR MK 8876
THIS COMPD HAS ONE FLUORO MISSING, APPLY TO YOUR MK 8876