Home » Phase2 drugs (Page 19)
Category Archives: Phase2 drugs
Poziotinib for the treatment of Adenocarcinoma of Lung Stage IIIB or Adenocarcinoma of Lung Stage IV
Poziotinib
l-(4-(4-(3,4-dichloro-2-fluorophenylamino)-7-methoxyquinazol in-6- yloxy)piperidin-l-yl)prop-2-en-l-one
: 1 – [4 – [[4 – [(3, 4 – dichloro – 2 – phenyl) amino] – 7 – methoxy – 6 – base] quinazoline oxygen radicals] – 1 – piperidine base] – 2 – acrylic – 1 – ketone
UNII-OEI6OOU6IK;
cas 1092364-38-9
HM781-36B
NOV120101
Erbb2 tyrosine kinase receptor inhibitor; EGFR family tyrosine kinase receptor inhibitor
Non-small-cell lung cancer; Stomach tumor
for the treatment of Adenocarcinoma of Lung Stage IIIB or Adenocarcinoma of Lung Stage IV
http://www.centerwatch.com/clinical-trials/listings/external-studydetails.aspx?StudyID=NCT01819428
The purpose of this open-label, single-arm, multi-center phase II trial is to evaluate the efficacy and safety of novel pan-HER inhibitor, NOV120101 (Poziotinib), as a first-line monotherapeutic agent in patients with lung adenocarcinoma harboring EGFR mutation…….http://clinicaltrials.gov/show/NCT01819428

KR 1013319
………………………………………………………….
WO2013051883
http://www.google.com/patents/WO2013051883A2?cl=en
1 -(4-(4-(3 ,4-dichloro-2-fluorophenylamino)-7-methoxyquinazolin-6- yloxy)piperidin-l-yl)-prop-2-en-l-one hydrochloride of formula (I) below is an important drug having antiproliferative activities such as anti-tumor activity, which can be used for selectively and effectively treating drug resistance caused by tyrosine kinase mutation. Its free base form, i.e., l-(4-(4-(3,4-dichloro-2- fluoropheny lamino)-7-methoxyquinazolin-6-y loxy)piperidin- 1 -y l)-prop-2-en- 1 – one having formula (II) below is identified as CAS Registry Number 1092364-38-
9.
The compound of formula (II) may be prepared by, e.g., the method disclosed in Korean Patent No. 1013319, the reaction mechanism thereof being shown in Reaction Scheme 1 below. The compound of formula (II) prepared according to Reaction Scheme 1 may then be reacted with hydrochloric acid to produce the compound of formula (I).
wherein R is halogen.

formula (I):
In accordance with another aspect of the present invention, there are provided N-(3,4-dichloro-2-fluorophenyl)-7-methoxy-6-(piperidin-4- yloxy)quinazolin-4-amine dihydrochloride of formula (III), tert-butyl 4-(4-(3,4- dichloro-2-fluorophenylamino)-7-methoxyquinazolin-6-yloxy)piperidin-l- carboxylate of formula (IV) and 4-(3,4-dichloro-2-fluorophenylamino)-7- methoxyquinazolin-6-ol of formula (V), which can be used as intermediates for preparing the compound of formula (I).
Example 1: Preparation of 4-(3,4-dichloro-2-fluorophenyIamino)-7- methoxyquinazolin-6-yl acetate the compound of formula (VI))
7-methoxy-4-oxo-3,4-dihydroquinazolin-yl acetate (100 g) was added to toluene (850 ml) and NN-diisopropylethylamine (82.5 ml). Phosphorusoxy chloride (100 ml) was added thereto over 20 minutes at 75°C, followed by stirring for 3 hours. Toluene (450 ml) and 3,4-dichloro-2-fluoroaniline (84.6 g) were added to the resulting mixture, followed by stirring for 2 hours. Upon completion of the reaction, the resulting mixture was cooled to 25°C. The solid thus obtained was filtered under a reduced pressure and washed with toluene (400 ml). Isopropanol (1,000 ml) was added to the solid, which was then stirred for 2 hours. The resulting solid was filtered and washed with isopropanol (400 ml). The solid was dried at 40°C in an oven to produce the compound of formula (VI) (143 g, yield: 83%).
1H-NMR (DMSO-d6, 300 MHz, ppm) δ 8.92 (s, 1H), 8.76 (s, 1H), 7.69- 7.57 (m, 3H), 4.01 (s, 3H), 2.38 (s, 3H).
Example 2: Preparation of 4-(3,4-dichloro-2-fluorophenylamino)-7- methoxyquinazolin-6-ol (the com ound of formula (V))
4-(3,4-dichloro-2-fluorophenylamino)-7-methoxyquinazolin-6-yl acetate (100 g) was admixed with methanol (1,000 ml). The mixture was cooled to 10 to 15°C, added with an ammonia solution (460 g), and stirred for 3 hours at 25°C. The solid thus obtained was filtered and washed with a mixed solvent of methanol (200 ml) and water (200 ml). The resulting solid was dried at 40°C in an oven to produce the compound of formula (V) (74 g, yield: 83%).
1H-NMR (DMSO-d6, 300 MHz, ppm) 6 9.57 (br, 2H), 8.35 (s, 1H), 7.68 (s, 1H), 7.61-7.52 (m, 2H), 7.21 (s, 1H), 3.97 (s, 3H).
Example 3: Preparation of /er/-but l-4-(4-(3,4-dichloro-2- fluorophenylamino)-7-methoxyquinazolin-6-yloxy)piperidin-l-carboxylate (the compound of formu
4-(3,4-dichloro-2-fluorophenylamino)-7-methoxyquinazolin-6-ol (60 g) was admixed with N-dimethylformamide (360 ml) under stirring, followed by addition of tert-butyl 4-(tosyloxy)piperidin-l-carboxylate (120 g) and potassium carbonate (72 g) to the mixture. The reaction temperature was raised to 70°C, and the mixture was stirred for 14 hours. The temperature of the resulting solution was cooled to 25°C, and water (480 ml) was slowly added thereto. The solid thus obtained was filtered and dried. The solid was dissolved in a mixed solvent (600 ml) of dichloromethane and methanol. Active carbon (6 g) was then added thereto, followed by stirring for 30 minutes. The resulting mixture was filtered through a Celite pad, distilled under a reduced pressure, added with acetone (300 ml), and stirred for 2 hours. The resulting solid was filtered and washed with acetone (100 ml). The solid was dried at 40°C in an oven to produce the compound of formula (IV) (75 g, yield: 83%).
1H-NMR (DMSO-d6, 300 MHz, ppm) 6 8.69 (s, 1H), 8.47 (t, 1H), 7.34- 7.29 (m, 2H), 7.20 (s, 1H), 4.63-4.60 (m, 1H), 3.82 (s, 3H), 3.83-3.76 (m, 2H), 3.37-3.29 (m, 2H), 1.99-1.96 (m, 2H), 1.90-1.84 (m, 2H), 1.48 (s, 9H).
Example 4: Preparation of N-(3,4-dichIoro-2-fluorophenyi)-7- methoxy-6-(piperidin-4-yloxy)quinazoIin-4-amine dihydrochloride (the compound of formula (III))
Acetone (740 ml) was added to ter/-butyl 4-(4-(3,4-dichloro-2- fluorophenylamino)-7-methoxyquinazolin-6-yloxy)piperidin-l-carboxylate (75 g), which was then stirred. The mixture was added with hydrochloric acid (145 ml) for 10 minutes and stirred for 5 hours. Upon completion of the reaction, the resulting mixture was filtered, and the solid thus obtained was washed with acetone (73 ml). The solid was dried at 30°C in an oven to produce the compound of formula (III) (71 g, yield: 99%).
1H-NMR (DMSO-d6, 300 MHz, ppm) 512.95 (bs, 1H), 9.42 (bs, 1H), 9.18 (bs, 1H), 9.01 (s, 1H), 8.86 (s, 1H), 7.69-7.56 (m, 2H), 7.45 (s, 1H), 5.11- 5.08 (m, 1H), 4.03 (s, 3H), 3.29-3.20 (m, 4H), 2.33-2.30 (m, 2H), 1.96-1.93 (m, 2H).
Example 5: Preparation of l-(4-(4-(3,4-dichloro-2- fluorophenylamino)-7-methoxyquinazoIin-6-yloxy)piperidin-l-yl)prop-2-en- 1-one (the compound of formula II))
N-(3,4-dichloro-2-fluorophenyl)-7-methoxy-6-(piperidin-4- yloxy)quinazolin-4-amine dihydrochloride (100 g) and sodium hydrogen carbonate (66 g) were added to a mixed solvent of tetrahydrofuran (630 ml) and water (1 L), and the temperature of the reaction mixture was cooled to 0°C with iced water. Acryloyol chloride (24 ml) diluted with tetrahydrofuran (370 ml) was slowly added to the reaction mixture over 30 minutes, followed by stirring at 0°C for 30 minutes. Upon completion of the reaction, aqueous acetone (2.0 L) was added to the resulting mixture, which was stirred for 12 hours and filtered to produce 1 -(4-(4-(3 ,4-dichloro-2-fluorophenylamino)-7-methoxyquinazolin-6- yloxy)piperidin-l-yl)prop-2-en-l-one (72 g, yield: 75%). The solid thus obtained was dissolved in a mixed solvent of dichloromethane (200 ml) and methanol (100 ml), added with ethyl acetate (1.2 L), and stirred for 12 hours. The resulting solid was filtered and washed with ethyl acetate (100 ml). The solid was dried at 40°C in an oven to produce the compound of formula (II) (55 g, yield: 76%, total yield = 57%).
Ή-NMR (CDC13, 300 MHz, ppm) 68.68(s, 1H), 8.39(t, 3H), 7.3 l(m, 3H), 6.61(m, 1H), 6.29(m, 1H), 5.72(m, 1H), 4.75(m, 1H), 4.02(s, 3H), 3.89(m, 2H), 3.60(m, 2H), 1.86(m, 4H). Example 6: Preparation of l-(4-(4-(3,4-dichloro-2- fluorophenylamino)-7-methoxyquinazolin-6-yIoxy)piperidin-l-yl)prop-2-en- 1-one hydrochloride (the com ound of formula (I))
1 -(4-(4-(3 ,4-dichloro-2-fluorophenylamino)-7-methoxyquinazolin-6- yloxy)piperidine-l-yl)prop-2-en-l-one (150 g) was added to methanol (700 ml). Hydrochloric acid (38.2 ml) diluted with methanol (300 ml) was added thereto, followed by stirring for 24 hours. The solid thus obtained was filtered and washed with acetone (100 ml). The resulting solid was dried at 40°C in an oven for 24 hours to produce the compound of formula (I) (131 g, yield: 81%).
1H-NMR (DMSO-d6, 300 MHz, ppm) 512.31 (bs, 1H), 8.83 (s, 1H), 8.67 (s, 1H), 7.64-7.55 (m, 2H), 7.39 (s, 1H), 6.87-6.78 (m, 1H), 6.12-6.06 (m, 1H), 5.68-5.64 (m, IH), 5.07-5.01 (m, IH), 4.06-3.88 (m, 5H), 3.51 (t, IH), 3.32 (t, IH), 2.10 (t, IH), 1.60 (t, IH).
…………………………………………………………..
WO-2014116070
http://www.sumobrain.com/patents/wipo/Method-preparing-1-4-34/WO2014116070.html
Process for preparing poziotinib – comprising the reaction of a 4-(3,4-dichloro-2-fluorophenylamino)-7-methoxyquinazolin-6-ol compound with an N-acyl piperidine derivative.
A process for preparing poziotinib comprising the reaction of a 4-(3,4-dichloro-2-fluorophenylamino)-7-methoxyquinazolin-6-ol compound with an N-acyl piperidine derivative in the presence of an inert polar protic solvent (eg N,N-dimethylformamide), and a base (eg sodium bicarbonate) is claimed. Also claimed are processes for preparing intermediates of poziotinib. Poziotinib is known to be an inhibitor of EGFR family, and Erbb2 tyrosine kinase receptors, useful for the treatment of stomach tumor and non-small-cell lung cancer. Novel method for preparing poziotinib. Follows on from WO2013051883 claiming method for preparing poziotinib and its intermediates. Hanmi, in collaboration with National Oncoventure, is developing poziotinib for the oral treatment of non small cell lung cancer and gastric cancer. As of August 2014, the drug is in phase 2 trials for both indications.
Compound of formula (II) is (I) and compound of formula (I) (poziotinib) is (II) (claim 1, page 13).The synthesis of (II) via intermediate (I) is described (example 1, pages 8-11).
Preparation Example 1: Preparation of 4-(3,4-dichloro-2-fluorophenylamino)- 7-methoxyquinazolin-6-ol, the compound of formula (II)
Step (i): Preparation of 4-(3,4-dichloro-2-fluorophenylamino)-7- methoxyquinazolin-6-yl acetate, the compound of formula (V)
7-methoxy-4-oxo-3,4-dihydroquinazolin-6-yl acetate (100 g) was added to toluene (850 mL) and NN-diisopropylethylamine (82.5 mL). Phosphorus oxychloride (100 mL) was added thereto over 20 minutes at 75°C, followed by stirring for 3 hours. Toluene (450 mL) and 3,4-dichloro-2-fluoroaniline (84.6 g) were added to the resulting mixture, followed by stirring for 2 hours. Upon completion of the reaction, the resulting mixture was cooled to 25°C, and the solid thus obtained was filtered under a reduced pressure and washed with toluene (400 mL). Isopropanol (1,000 mL) was added to the solid, and the resulting mixture was stirred for 2 hours. The solid thus obtained was filtered and washed with isopropanol (400 mL), and then was dried at 40°C in an oven to obtain the target compound (143 g, yield: 83%).
1H-NMR (DMSO-d 6 , 300 MHz, ppm) δ 8.92 (s, 1H), 8.76 (s, 1H), 7.69- 7.57 (m, 3H), 4.01 (s, 3H), 2.38 (s, 3H).
Step (ii): Preparation of 4-(3,4-dichloro-2-fluorophenylamino)-7- methoxyquinazolin-6-ol, the compound of formula (II)
4-(3,4-dichloro-2-fluorophenyIamino)-7-methoxyquinazolin-6-y l acetate (100 g) prepared in step (i) was admixed with methanol (1,000 mL). The mixture was cooled to 10 to 1 °C, added with an ammonia solution (460 g), and stirred for 3 hours at 25°C. The solid thus obtained was filtered and washed with a mixed solvent of methanol (200 mL) and water (200 mL). The resulting solid was dried at 40°C in an oven to obtain the target compound (74 g, yield: 83%). 1H-NMR (DMSO-d 6 , 300 MHz, ppm) 5 9.57 (br, 2H), 8.35 (s, 1H), 7.68 (s,
1H), 7.61-7.52 (m, 2H), 7.21 (s, 1H), 3.97 (s, 3H).
Example 1: Preparation of l-(4-(4-(3,4-dichIoro-2-fluorophenylamino)-7- methoxyquinazolin-6-yloxy)piperidin-l-yl)prop-2-en-l-one, the compound of formula (I) Step (1-1 : Preparation of l-acryloylpiperidin-4-yl 4- methylbenzenesulfonate. the compound of formula (HI)
Piperidin-4-yl 4-methylbenzenesulfonate hydrochloride (200 g, 685 mmol), tetrahydrofuran (THF, 1.6 L) and NaHCO 3 (172 g, 2047 mmol) were added to water (2 L), and the mixture was cooled to 0°C. A solution prepared by adding acryloyl chloride (56 mL, 519 mmol) to THF (0.4 L) was added thereto over 30 minutes, followed by stirring for 1 hour. Upon completion of the reaction, MeOH (0.4 L) was added thereto for quenching. The solution was extracted with ethyl ester (2 L), and washed with water (2 L). The organic layer was separated, distilled under a reduced pressure, and the residue thus obtained was recrystallized from dichloromethane-hexane to obtain the target compound (174 g, yield: 82%). 1H-NMR (300 MHz, DMSO-d 6 ) δ 7.82 (d, 2H), 7.48 (d, 2H), 6.80-6.71 (m,
1H), 6.10-6.03 (m, 1H), 5.67-5.62 (m, 1H), 4.76-4.71 (m, 1H), 3.70-3.68 (m, 2H), 3.43-3.31 (m, 2H), 2.42 (s, 3H), 1.73 (m, 2H), 1.52 (m, 2H).
Step (1-2): Preparation of l-(4-(4-(3,4-dichloro-2-fluorophenylamino)-7- methoxyquinazolin-6-yloxy)piperidin-l-yl)prop-2-en-l-one, the compound of formula (I)
4-(3,4-dichloro-2-fluorophenylamino)-7-methoxyquinazolin-6-o l (12 g, 34 mmol) prepared in Preparation Example 1, l-acryloylpiperidin-4-yl 4- methylbenzenesulfonate (16 g, 51 mmol) prepared in step (1-1), K 2 CO 3 (9.4 g, 68 mmol) and dimethylacetamide (DMAc, 300 mL) were admixed. The reaction temperature was raised to 70°C, and the mixture was stirred for 24 hours. Upon completion of the reaction, the mixture was cooled down to room temperature, extracted with ethyl ester (300 mL), and then washed with water (300 mL). The organic layer was separated, and distilled under a reduced pressure. The residue thus obtained was solidified by adding ethyl ester, filtered, and dried to obtain the target compound (12.8 g, yield: 77%). 1H-NMR (300 MHz, DMSO-d 6 ) δ 9.65 (bs, 1H), 8.40 (s, 1H), 7.88 (s, 1H),
7.64-7.56 (m, 2H), 7.24 (s, 1H), 6.89-6.80 (m, 1H), 6.15-6.08 (m, 1H), 5.70-5.66 (m, 1H), 4.78 (m, 1H), 3.94 (s, 3H), 3.87 (m, 2H), 3.48 (m, 2H), 2.03 (m, 2H), 1.70 (m, 1H). Example 2: Preparation of l-(4-(4-(3,4-dichloro-2-fluorophenylamino)-7- methoxyquinazoIin-6-yloxy)piperidin-l-yl)prop-2-en-l-one, the compound of formula (I)
SEE
http://www.yuaigongwu.com/thread-8891-1-1.html
| WO2005030765A1 * | Sep 22, 2004 | Apr 7, 2005 | Astrazeneca Ab | Quinazoline derivatives as antiproliferative agents |
| WO2008150118A2 * | Jun 5, 2008 | Dec 11, 2008 | Hanmi Pharm Ind Co Ltd | Novel amide derivative for inhibiting the growth of cancer cells |
| WO2010122340A2 * | Apr 22, 2010 | Oct 28, 2010 | Astrazeneca Ab | Process 738 |
| US20070135463 * | Dec 6, 2006 | Jun 14, 2007 | Frank Himmelsbach | Bicyclic heterocycles, drugs containing said compounds, the use thereof and method for preparing same |



Cimicoxib

Cimicoxib
4-[4-Chloro-5-(3-fluoro-4-methoxyphenyl)-1H-imidazol-1-yl]benzenesulfonamide
IN PHASE 2
Cimicoxib (trade name Cimalgex) is a non-steroidal anti-inflammatory drug (NSAID) used in veterinary medicine to treat dogs for pain and inflammation associated with osteoarthritis and for the management of pain and inflammation associated with surgery.[1] It acts as a COX-2 inhibitor.
Cimicoxib is a selective COX-2 inhibitor being developed by Affectis as a treatment for depression and schizophrenia. If approved, Cimicoxib would be the first drug in decades to treat depression by a new mechanism of action
Cimicoxib, an imidazole derivative, is a selective cyclooxygenase-2 (COX-2) inhibitor. The product was in phase II development at Affectis Pharmaceuticals for the oral treatment of major depression, however, no recent development have been reported. Originally developed by Uriach, the compound was acquired by Palau Pharma, a spin-off created by Uriach in November 2006.
In 2007, Palau Pharma licensed global rights to cimicoxib to Affectis Pharmaceuticals for all CNS indications. Palau had been clinically evaluating the compound for the treatment of osteoarthritis, pain and rheumatoid arthritis, however, no recent development has been reported for these indications. The compound holds potential for the treatment of schizophrenia.

Treatment of 4-(acetylamino)phenylsulfonyl chloride (I) with tert-butylamine yields sulfonamide (II), which on deprotection with potassium hydroxide gives amine (III). Reaction of compound (III) with 4-methoxy-3-fluorobenz-aldehyde gives imine (IV), which is cyclized with tosylmethyl isocyanide to afford imidazole (V). Regioselective chlorination of compound (V) with N-chlorosuccinimide (NCS) to afford the chloroimidazole (VI) and then deprotection of the sulfonamide group of (VI) yields cimicoxib in 40% overall yield.

EP 1122243; JP 2002527508; WO 0023426, ES 2184633; WO 0316285
……………………………….
http://www.google.com/patents/EP1424329A1?cl=en
EXAMPLE 1
4-Amino-N- tert -butylbenzenesulfonamide Method A:
-
[0031]
a) N-tert-Butyl-4-nitrobenzenesulfonamide
-
[0032]To a solution of tert-butylamine (0.47 L, 6.4 mol) in THF (0.55 L) is slowly added, at 0 °C, a solution of 4-nitrobenzenesulfonyl chloride (50 g, 0.23 mol) in THF (0.55 L) and the resulting mixture is stirred for 24 h at room temperature. The solvent is removed and the residue is taken up in a CHCl3/0.5 N HCl mixture, the layers are separated and the aqueous phase is extracted with CHCl3. The combined organic extracts are washed with H2O and brine and dried over MgSO4. The solvent is removed, yielding 56.3 g of a yellowish solid which is directly used in the next reaction (yield: 97%).
Mp: 105-109°C; 1H-NMR (300 MHz, CDCl3) δ (TMS): 1.29 (s, 9 H), 5.07 (s, 1 H), 8.13 (d, J = 9 Hz, 2 H), 8.39 (d, J = 9 Hz, 2 H).
b) Title compound
-
[0033]A solution of N-tert-butyl-4-nitrobenzenesulfonamide (10.0 g, 39 mmol) in EtOH (100 mL) is stirred for 48 h under a H2 atmosphere in the presence of 10% Pd/C (1.50 g). The resulting mixture is filtered and concentrated to give the desired product as a slightly-coloured solid (8.7 g, yield: 98%).
Mp: 127 °C; 1H-NMR (300 MHz, CDCl3 + CD3OD) δ (TMS): 1.19 (s, 9 H), 3.74 (s, CD3OD + 1 H), 6.93 (d, J = 9 Hz, 2 H), 7.66 (d, J = 9 Hz, 2 H).
Method B:
-
[0034]
a) 4-Acetylamino-N-tert-butylbenzenesulfonamide
-
[0035]To a suspension of 4-acetylaminobenzenesulfonyl chloride (10 g, 43 mmol) in DME (103 mL) is added, at 0 °C, tert-butylamine (9 mL, 86 mmol) in DME (103 mL). Next, the reaction mixture is stirred for 4 h at reflux. The solvent is removed and CHCl3 is added. The resulting suspension is filtered and the solid is washed with CHCl3, H2O and Et2O. The solid obtained is dried in vacuo to give 8.0 g of the product as a white solid (yield: 68%).
Mp: 200-201 °C; 1H-NMR (300 MHz, CDCl3 + CD3OD) δ (TMS): 1.15 (s, 9 H), 2.12 (s, 3 H), 4.21 (s, 2H + CD3OD), 7.66 (d, J = 9 Hz, 2 H), 7.75 (d, J = 9 Hz, 2 H).
b) Title compound
-
[0036]A solution of 4-acetylamino-N-tert-butylbenzenesulfonamide (8.0 g, 29.6 mmol), KOH (8.30 g, 148 mmol), H2O (6 mL) and MeOH (24 mL) is heated at 100°C for 2 h. H2O (24 mL) is added and the mixture is heated for two more hours. It is allowed to cool, H2O is added and it is brought to pH 8 with 1N HCl. It is then extracted with EtOAc, dried over Na2SO4 and the solvent is removed, to give 6.0 g of the product as a white solid (yield: 89%).
EXAMPLE 2 N- tert -Butyl-4-[(3-fluoro-4-methoxybenzylidene)amino]benzenesulfonamide
-
[0037]
-
[0038]A mixture of 4-amino-N-tert-butylbenzenesulfonamide (52.3 g, 0.23 mol, obtained in example 1), 3-fluoro-4-methoxybenzaldehyde (35.3 g, 0.23 mol) and toluene (2.5 L) is heated at reflux in a Dean-Stark for 24 h. The solvent is removed, yielding 83.5 g of the title compound (yield: quantitative).
Mp: 129-131 °C; 1H-NMR (300 MHz, CDCl3) δ (TMS): 1.23 (s, 9 H), 3.98 (s, 3 H), 4.65 (s, 1 H), 7.04 (t, J = 8.1 Hz, 1 H), 7.21 (d, J = 6.7 Hz, 2 H), 7.58 (m, 1 H), 7.73 (dd, JH-F = 11.8 Hz, J = 2 Hz, 1 H), 7.90 (d, J = 6.7 Hz, 2 H), 8.33 (s, 1 H).
EXAMPLE 3 N-tert-Butyl-4-[5-(3-fluoro-4-methoxyphenyl)imidazol-1-yl]benzenesulfonamide
-
[0039]
-
[0040]A mixture of N-tert-butyl-4-[(3-fluoro-4-methoxybenzylidene)amino]benzenesulfonamide (41.5 g, 114 mmol, obtained in example 2), tosylmethylisocyanide (33.22 g, 171 mmol), K2CO3 (31.1 g, 228 mmol), DME (340 mL) and MeOH (778 mL) is heated at reflux for 3 h. The solvent is removed and the residue is taken up in a CHCl3/H2O mixture and the layers are separated. The aqueous phase is extracted with CHCl3 and the combined organic extracts are dried over MgSO4 and concentrated. A crude product is obtained, which is washed with Et2O several times to give 41.40 g of a creamy solid that is directly used in the next reaction (yield: 90%).
Mp: 229-232°C; 1H-NMR (300 MHz, CDCl3) δ (TMS): 1.24 (s, 9 H), 3.89 (s, 3 H), 4.51 (s, 1 H), 6.90 (m, 3 H), 7.23 (s, 1 H), 7.29 (d, J = 8.7 Hz, 2 H), 7.73 (s, 1 H), 7.94 (d, J = 8.7 Hz, 2 H).
EXAMPLE 4 N-tert-Butyl-4-[4-chloro-5-(3-fluoro-4-methoxyphenyl)imidazol-1-yl]benzenesulfonamide
-
[0041]
-
[0042]A mixture of N-tert-butyl-4-[5-(3-fluoro-4-methoxyphenyl)imidazol-1-yl]benzenesulfonamide (41.40 g, 103 mmol, obtained in example 3) and acetonitrile (840 mL) is heated at reflux and acetonitrile is added until complete dissolution (200 mL more). Next, N-chlorosuccinimide (15.0 g, 113 mmol) is added and the mixture is refluxed for 24 h. The solvent is removed and the residue is suspended in EtOAc and 1N HCl and is stirred for 10 min. The solid obtained is filtered and washed directly in the filter with 1N HCl, 1N NaOH, saturated NH4Cl solution, H2O and Et2O. A solid is obtained, which is dried in vacuo to give 37.0 g of the product as a creamy solid (yield: 82%).
Mp: 208-210 °C; 1H-NMR (300 MHz, CDCl3) δ (TMS): 1.24 (s, 9 H), 3.89 (s, 3 H), 4.51 (s, 1 H), 6.90 (m, 3 H), 7.23 (d, J = 8.7 Hz, 2 H), 7.63 (s, 1 H), 7.92 (d, J = 8.7 Hz, 2 H).
EXAMPLE 5 4-[4-Chloro-5-(3-fluoro-4-methoxyphenyl)imidazol-1-yl]benzenesulfonamide
-
[0043]
-
[0044]A mixture of N-tert-butyl-4-[4-chloro-5-(3-fluoro-4-methoxyphenyl)imidazol-1-yl]benzenesulfonamide (37.0 g, 85 mmol, obtained in example 4), concentrated HCl (200 mL) and H2O (200 mL) is heated at reflux for 3 h. The mixture is allowed to cool and is brought to pH 6 with 6N NaOH. A white precipitate appears, which is collected by filtration and washed with plenty of H2O and then with CHCl3. 31 g of the title compound of the example is obtained (yield: 97%), which are recrystallized from acetonitrile.
Mp: 211-212 °C; -
1H-NMR (300 MHz, CDCl3 + CD3OD) δ (TMS): 3.90 (s, 3 H), 4.16 (s, CD3OD + 2 H), 6.93 (m, 3 H), 7.30 (d, J = 8.6 Hz, 2 H), 7.73 (s, 1 H), 7.95 (d, J = 8.7 Hz, 2 H).
References
- “European Public Assessment Report: Cimalgex (cimicoxib)”. European Medicines Agency.
|
9-1-2013
|
Detection and quantification of cimicoxib, a novel COX-2 inhibitor, in canine plasma by HPLC with spectrofluorimetric detection: development and validation of a new methodology.
|
Journal of pharmaceutical and biomedical analysis
|
|
|
6-1-2013
|
Efficacy and safety of cimicoxib in the control of perioperative pain in dogs.
|
The Journal of small animal practice
|
|
|
4-5-2007
|
NO-donor COX-2 inhibitors. New nitrooxy-substituted 1,5-diarylimidazoles endowed with COX-2 inhibitory and vasodilator properties.
|
Journal of medicinal chemistry
|
|
|
10-21-2004
|
New water-soluble sulfonylphosphoramidic acid derivatives of the COX-2 selective inhibitor cimicoxib. A novel approach to sulfonamide prodrugs.
|
Journal of medicinal chemistry
|
|
|
7-31-2003
|
Synthesis and structure-activity relationship of a new series of COX-2 selective inhibitors: 1,5-diarylimidazoles.
|
Journal of medicinal chemistry
|
|
4-15-2005
|
Compositions of a cyclooxygenase-2 selective inhibitor and a serotonin-modulating agent for the treatment of central nervous system damage
|
|
|
4-8-2005
|
Compositions of a cyclooxygenase-2 selective inhibitor and an IKK inhibitor for the treatment of ischemic mediated central nervous system disorders or injury
|
|
1-9-2009
|
Process for the Preparation of 4-(imidazol-1-yl)benzenesulfonamide Derivatives
|
|
|
9-5-2008
|
Medicament that is Intended for Oral Administration, Comprising a Cyclooxygenase-2 Inhibitor, and Preparation Method Thereof
|
|
|
4-2-2008
|
Method of preparing 4-(imidazol-1-yl)benzenesulphonamide derivatives
|
|
|
6-29-2007
|
Compositions of a cyclooxygenase-2 selective inhibitor administered under hypothermic conditions for the treatment of ischemic mediated central nervous system disorders or injury
|
|
|
7-8-2005
|
Compositions of a cyclooxygenase-2 selective inhibitor and a neurotrophic factor-modulating agent for the treatment of central nervous system mediated disorders
|
|
|
5-27-2005
|
Compositions of a cyclooxygenase-2 selective inhibitor administered under hypothermic conditions for the treatment of ischemic mediated central nervous system disorders or injury
|
|
|
5-13-2005
|
Compositions of a cyclooxygenase-2 selective inhibitior and a non-NMDA glutamate modulator for the treatment of central nervous system damage
|
|
|
4-22-2005
|
Compositions of a cyclooxygenase-2 selective inhibitor and a low-molecular-weight heparin for the treatment of central nervous system damage
|
|
|
4-22-2005
|
Mediated central nervous system compositions of a cyclooxygenase-2 selective inhibitor and a corticotropin releasing factor antagonist for the treatment of ischemic disorders or injury
|
Tilmacoxib
Tilmacoxib
Japan Tobacco (JT) (Originator)
Tilmacoxib or JTE-522 is a COX-2 inhibitor and is an effective chemopreventive agent against rat experimental liver fibrosis.[1]
A member of the class of 1,3-oxazoles that is that is 1,3-oxazole which is substituted at positions 2, 4 and 5 by methyl, cyclohexyl, and 3-fluoro-4-sulfamoylphenyl groups, respectively.
………..
4-(4-Cycloalkyl/aryl-oxazol-5-yl)benzenesulfonamides as selective cyclooxygenase-2 inhibitors: Enhancement of the selectivity by introduction of a fluorine atom and identification of a potent, highly selective, and orally active COX-2 inhibitor JTE-522
J Med Chem 2002, 45(7): 1511
http://pubs.acs.org/doi/abs/10.1021/jm010484p
A series of 4-(4-cycloalkyl/aryl-oxazol-5-yl)benzenesulfonamide derivatives were synthesized and evaluated for their abilities to inhibit cyclooxygenase-2 (COX-2) and cyclooxygenase-1 (COX-1) enzymes. In this series, substituent effects at the ortho position to the sulfonamide group on the phenyl ring were examined. Most substituents reduced or lost both COX-2 and COX-1 activities. In contrast, introduction of a fluorine atom preserved COX-2 potency and notably increased COX1/COX-2 selectivity. This work led to the identification of a potent, highly selective, and orally active COX-2 inhibitor JTE-522 [9d, 4-(4-cyclohexyl-2-methyloxazol-5-yl)-2-fluorobenzenesulfonamide], which is currently in phase II clinical trials for the treatment of rheumatoid arthritis, osteoarthritis, and acute pain.
9d as a white solid: mp 166−167 °C; 1H NMR (CDCl3) δ 1.3−1.5 (m, 3H), 1.6−1.9 (m, 7H), 2.51 (s, 3H), 2.79 (tt, J = 3.7, 11.3 Hz, 1H), 5.11 (s, 2H), 7.36−44 (m, 2H), 7.94 (t, J = 7.9 Hz, 1H). Anal. (C16H19FN2O3S) C, H, N.
………………
WO 1996019463 OR http://www.google.com/patents/EP0745596A1?cl=en
Example 2
-
[0080]
-
[0081]To a solution of tetrakis(triphenylphosphine)palladium (2.00 g) and zinc powder (17.98 g) in 1,2-dimethoxyethane (50 ml) was added a solution of cyclohexanecarbonyl chloride (20.00 g) in 1,2-dimethoxyethane (50 ml) at room temperature under a nitrogen atmosphere. A solution of 3-fluorobenzyl bromide (26.00 g) in 1,2-dimethoxyethane (100 ml) was gradually added dropwise to the mixture with stirring under ice-cooling. The mixture was stirred under ice-cooling for 30 minutes, and at room temperature for 2 hours. The insoluble matter was removed by filtration and the filtrate was concentrated under reduced pressure. Then, ethyl acetate (200 ml) was added to the residue, and the mixture was washed with 1N hydrochloric acid, and then with saturated aqueous sodium hydrogencarbonate solution and saturated brine, and dried over anhydrous sodium sulfate. The solvent was evaporated to give 29.20 g of an oily crude product.
Step 16) 2-Cyclohexyl-1-(3-fluorophenyl)-2-oxoethyl acetate (formula (V”); R’=cyclohexyl, R1‘=3-fluorophenyl, R2‘=methyl, Z=oxygen atom) -
[0082]Lead tetraacetate (75.00 g) was added to a solution of the compound (29.20 g) obtained in the above Step 10) in acetic acid (300 ml). The mixture was refluxed under heating for 1.5 hours, and the solvent was evaporated under reduced pressure. Ethyl acetate was added to the residue. The mixture was washed with water, a saturated aqueous sodium hydrogencarbonate solution and saturated brine, and dried over anhydrous sodium sulfate. The solvent was evaporated under reduced pressure, and the residue was purified by silica gel column chromatography (developing solvent; hexane:ethyl acetate=9:1) to give 18.30 g of the title compound as an oil (yield 50%).
Step 17) 4-Cyclohexyl-5-(3-fluorophenyl)-2-methyloxazole (formula (XIII); R’=cyclohexyl, R1‘=3-fluorophenyl, R2=methyl, Z=oxygen atom) -
[0083]A solution of the compound (18.00 g) obtained in the above Step 16) and ammonium acetate (15.00 g) in acetic acid (100 ml) was refluxed under heating for 5 hours, and the solvent was evaporated under reduced pressure. Ethyl acetate was added to the residue. The mixture was washed with water, saturated aqueous sodium hydrogencarbonate solution and saturated brine, and dried over anhydrous sodium sulfate. The solvent was evaporated under reduced pressure to give 17.20 g of an oily crude product. Step 15) 5-(4-Aminosulfonyl-3-fluorophenyl)-4-cyclohexyl-2-methyloxazole (formula (I); R=cyclohexyl, R1=4-aminosulfonyl-3-fluorophenyl, R2=methyl, Z=oxygen atom)
-
[0084]To a solution of the compound (17.00 g) obtained in the above Step 17) in chloroform (80 ml) was added dropwise chlorosulfonic acid (27 ml) with stirring under ice-cooling, and the mixture was heated at 100°C for 3 hours. The reaction mixture was cooled to room temperature, and dropwise added to ice-water (300 ml) with stirring. The organic layer was separated, washed with saturated brine, and dried over anhydrous sodium sulfate. The solvent was evaporated under reduced pressure to give 20.31 g of a crude product.
-
[0085]Aqueous ammonia (28%) was added to a solution of the obtained compound (10.00 g) in tetrahydrofuran (40 ml) with stirring at room temperature, and the mixture was stirred at room temperature for one hour. The solvent was evaporated under reduced pressure and ethyl acetate was added to the residue. The mixture was washed with water and saturated brine, and dried over anhydrous sodium sulfate. The solvent was evaporated, and the residue was separated and purified by silica gel column chromatography (developing solvent; dichloromethane:ethyl acetate=6:1) to give 5.74 g of the title compound (yield 61%).
Example 2′
-
[0086]
-
[0087]To a solution of the compound (353 g) obtained according to a method similar to that of the above Example 2, Step 10) in ethanol (1300 ml) were added hydroxylamine hydrochloride (123 g) and sodium acetate (158 g). The mixture was refluxed under heating for 2 hours, and the solvent was evaporated under reduced pressure. Ethyl acetate was added to the residue. The mixture was washed with water, saturated aqueous sodium hydrogencarbonate solution and saturated brine, and dried over anhydrous sodium sulfate. The solvent was evaporated under reduced pressure, and the crude product was recrystallized from n-heptane to give 160 g of the title compound (yield 42%).
Step 14) 4-Cyclohexyl-5-(3-fluorophenyl)-2-methyloxazole (formula (XIII); R’=cyclohexyl, R1‘=3-fluorophenyl, R2=methyl, Z=oxygen atom) -
[0088]Acetic anhydride (95 ml) was dropwise added to a solution of the compound (158 g) obtained in the above Step 11) in acetic acid (900 ml) with stirring at room temperature, and the mixture was refluxed under heating for 7 hours. The solvent was evaporated under reduced pressure and n-heptane was added to the residue. The mixture was washed with water, saturated aqueous sodium hydrogencarbonate solution, saturated brine and acetonitrile. The solvent was evaporated under reduced pressure to give 119 g of the title compound as an oil.
-
[0089]Then, the obtained compound (119 g) was reacted in the same manner as in the above Example 2, Step 15) to give a compound of Example 2 (formula (I); R=cyclohexyl, R1=4-aminosulfonyl-3-fluorophenyl, R2=methyl, Z=oxygen atom).
Example 3
-
[0090]Synthesis of 4-cyclohexyl-5-(3-fluoro-4-methylsulfonylphenyl)-2-methyloxazole (formula (I); R=cyclohexyl, R1=3-fluoro-4-methylsulfonylphenyl, R2=methyl, Z=oxygen atom)
Step 15) 4-Cyclohexyl-5-(3-fluoro-4-methylsulfonylphenyl)-2-methyloxazole (formula (I); R=cyclohexyl, R1=3-fluoro-4-methylsulfonylphenyl, R2=methyl, Z=oxygen atom) -
[0091]To a solution of the compound (17.00 g) obtained in the above Example 2, Step 17) in chloroform (80 ml) was dropwise added chlorosulfonic acid (27 ml) with stirring under ice-cooling. The mixture was heated at 100°C for 3 hours. The reaction mixture was cooled to room temperature and dropwise added to ice-water (300 ml) with stirring. The organic layer was separated, washed with saturated brine, and dried over anhydrous sodium sulfate. The solvent was evaporated under reduced pressure to give 20.31 g of a crude product.
-
[0092]Water (25 ml) was added to the obtained compound (3.66 g). To the mixture were added sodium sulfite (1.42 g) and sodium hydrogencarbonate (1.89 g) successively with stirring at room temperature. The mixture was heated at 70°C for 2 hours. Ethanol (25 ml) and methyl iodide (2.20 g) were added to the mixture, and the mixture was heated at 100°C for 2 hours. The mixture was cooled to room temperature and extracted with ethyl acetate. The extract was washed with saturated brine and dried over anhydrous sodium sulfate.
-
[0093]The solvent was evaporated under reduced pressure, and the residue was saparated and purified by silica gel column chromatography (developing solvent; hexane:ethyl acetate=2:1) to give 0.82 g of the title compound (yield 24%).
References
- Yamamoto, H., Kondo, M., Nakamori, S., Nagano, H., Wakasa, K., Sugita, Y., Chang-De, J., Kobayashi, S., Damdinsuren, B., Dono, K., Umeshita, K., Sekimoto, M., Sakon, M., Matsuura, N., Monden, M. (2003). “JTE-522, a cyclooxygenase-2 inhibitor, is an effective chemopreventive agent against rat experimental liver fibrosis1”. Gastroenterology 125 (2): 556–571. doi:10.1016/s0016-5085(03)00904-1. PMID 12891558.
-
3-28-20024-(4-cycloalkyl/aryl-oxazol-5-yl)benzenesulfonamides as selective cyclooxygenase-2 inhibitors: enhancement of the selectivity by introduction of a fluorine atom and identification of a potent, highly selective, and orally active COX-2 inhibitor JTE-522(1).Journal of medicinal chemistry
|
7-5-1999
|
The discovery of rofecoxib, [MK 966, Vioxx, 4-(4′-methylsulfonylphenyl)-3-phenyl-2(5H)-furanone], an orally active cyclooxygenase-2-inhibitor.
|
Bioorganic & medicinal chemistry letters
|
Apricoxib, A COX-2 inhibitor.

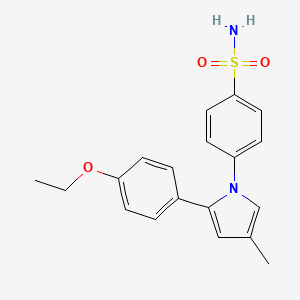
APRICOXIB
A COX-2 inhibitor.
MF; C19H20N2O3S
Mol wt: 356.439
CAS: 197904-84-0
CS-701; TG01, R-109339, TG-01 ,TP-1001
TP-2001, Capoxigem, Kymena, UNII-5X5HB3VZ3Z,
Benzenesulfonamide, 4-[2-(4-ethoxyphenyl)-4-methyl-1H-pyrrol-1-yl]-;
4-[2-(4-Ethoxyphenyl)-4-methyl-1H-pyrrol-1-yl]benzenesulfonamide
4-[2-(4-ethoxyphenyl)-4-methyl-1H-pyrrol-1-yl]benzenesulfonamide .
PHASE 2 http://clinicaltrials.gov/search/intervention=Apricoxib
Daiichi Sankyo (innovator)Daiichi Sankyo Co Ltd,
Current developer: Tragara Pharmaceuticals, Inc.

Apricoxib is an orally bioavailable nonsteroidal anti-inflammatory agent (NSAID) with potential antiangiogenic and antineoplastic activities. Apricoxib binds to and inhibits the enzyme cyclooxygenase-2 (COX-2), thereby inhibiting the conversion of arachidonic acid into prostaglandins. Apricoxib-mediated inhibition of COX-2 may induce tumor cell apoptosis and inhibit tumor cell proliferation and tumor angiogenesis. COX-related metabolic pathways may represent crucial regulators of cellular proliferation and angiogenesis.
R-109339 is a cyclooxygenase-2 (COX-2) inhibitor currently in phase II clinical development at Tragara Pharmaceuticals for the oral treatment of non-small cell lung cancer (NSCLC) and for the treatment of inflammation. Additional phase II clinical trials are ongoing in combination with gemcitabine and erlotinib for the treatment of pancreas cancer. The company had been evaluating R-109339 for the treatment of colorectal cancer, but development for this indication was discontinued for undisclosed reasons. Daiichi Sankyo and Tragara Pharmaceuticals had been conducting phase II clinical trials with the drug candidate for the oral treatment of arthritis and for the treatment of breast cancer, respectively; however, no recent development for this indication has been reported.
COX catalyzes the formation of prostaglandins and thromboxane from arachidonic acid, which is derived from the cellular phospholipid bilayer by phospholipase A2. In addition to several other functions, prostaglandins act as messenger molecules in the process of inflammation. The compound is also designed to act against a well-defined cancer pathway that affects several routes of cancer pathogenesis. In preclinical cancer models, R-109339 demonstrated superiority to compounds with similar mechanisms of action and potential for use in combination with cisplatin. Furthermore, the compound demonstrated the ability to inhibit the cachexia and weight loss seen in mouse tumor models.
Apricoxib, (CS-706, 1) 2-(4-ethoxyphenyl)-4-methyl-1-(4-sulfamoylphenyl)-pyrrole, a small-molecule, orally active, selective COX-2 inhibitor was discovered by investigators at Daiichi Sankyo in 1996. Clinical studies demonstrated potent analgesic activity and preclinical studies demonstrated good pharmacokinetics, pharmacodynamics and gastrointestinal tolerability. As an anticancer agent, preclinical studies demonstrated efficacy in biliary tract cancer models and colorectal carcinoma, and Recamp et al.
The original synthetic route is outlined below. Though the initial two steps were accomplished with decent yields, the final step of pyrrolidine formation followed by dehydration and dehydrocyanation produced only 3% of 1 as a brown powder. The yield in the last step of the synthesis of the 2-(4-methoxyphenyl) analog, 2-(4-methoxyphenyl)-4-methyl-1-(4-sulfamoylphenyl)-pyrrole, was 6%, indicating that this synthesis route is problematic.
14 Kimura T, Noguchi Y, Nakao A, Suzuki K, Ushiyama S, Kawara A, Miyamoto M. 799823. EP. 1997:A1.


……………………….
Synthesis
Published online Aug 19, 2011. doi: 10.1016/j.bmcl.2011.08.050
SEE AT
http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3310163/
An efficient synthesis of apricoxib (CS-706), a selective cyclooxygenase inhibitor, was developed using copper catalysed homoallylic ketone formation from methyl 4-ethoxybenzoate followed by ozonolysis to an aldehyde, and condensation with sulphanilamide. This method provided multi-gram access of aprocoxib in good yield. Apricoxib exhibited potency equal to celecoxib at inhibition of prostaglandin E2 synthesis in two inflammatory breast cancer cell lines.
We envisioned that 7 could be prepared by ozonolysis of homoallylic ketone (8) (Route B). A recent development in the synthesis of homoallylic ketones by Dorr et al. via copper-catalyzed cascade addition of alkenylmagnesium bromide to an ester a24 was examined. Treatment of commercially available methyl 4-ethoxybenzoate with 1-propenylmagnesium bromide (4.0 equiv) in presence of CuCN (0.6 equiv) resulted in 95% yield of desired ketone8 after silica gel chromatography, along with a minor amount of unreacted ester).b25

The product was a mixture of cis/trans R/S stereoisomers, as detected in the 1H NMR spectrum, and was used directly in the next step without separation. Ozone was bubbled through a solution of 8 in MeOH/CH2Cl2 at −78°C, until all starting materials were consumed. The ozonide was then reduced to aldehyde 7 by treatment with Me2S overnight. Removal of volatiles and subsequent addition and evaporation of toluene gave the crude 1,4-dicarbonyl compound 7 which was sufficiently pure for the following condensation step. The 1H NMR signal at 9.78 ppm of the crude product confirmed the formation of the aldehyde. No attempt was made to characterize the enantiomeric ratio of 7 since the dehydration/aromatization reaction of the next step removes the chirality of the product. Treatment of 7 with sulfanilamide in 40% acetic acid-acetonitrile at 70°C for three hours resulted in a brown product. Purification by silica gel flash chromatography yielded 71% of pure 1 as a white solid.c26
01
1H, 13C, and COSY NMR spectra of compounds 1 and 8.

……………
SYNTHESIS

synthesis
In one strategy, bromination of 4-ethoxyacetophenone (I) with Br2 yields 2-bromo-1-(4-ethoxyphenyl)ethanone (II) along with the byproduct 2-bromo-1-(3-bromo-4-ethoxyphenyl)ethanone, which are separated using HPLC. Alkylation of propionaldehyde N,Ndiisobutylenamine (III) with bromo ketone (II) and subsequent ketalization with neopentyl glycol (IV) using p-TsOH·H2O and, optionally, H2SO4 in MeCN gives monoprotected ketoaldehyde (V) (1). Finally, cyclization of ketoaldehyde derivative (V) with 4-aminobenzenesulfonamide (VI) in the presence of AcOH in PrOH/H2O at 90-100 °C furnishes apricoxib
Intermediate (V) can also be prepared by reaction of 1-(4- ethoxyphenyl)-2-buten-1-one (VII) with CH3NO2 in the presence of DBU in THF to produce nitro ketone (VIII). Subsequent treatment of nitroderivative (VIII) with neopentyl glycol (IV) and NaOMe and MeOH gives acetal (V) (2).In an alternativestrategy, condensation of 4-ethoxyacetaldehyde (IX) with 4-sulfamoylaniline (VI) in refluxing EtOH furnishesN-(4-ethoxybenzylidene)-
4-sulfamoylaniline (X), which then condenses with trimethylsilyl cyanide (XI) in the presence of ZnCl2 in THF yielding α- amino nitrile (XII). Cyclization of this compound with methacrolein (XIII) using LiHMDS in THF affords apricoxib
reference for above
- Drugs of the Future 2011, 36(7): 503-509
- Kojima, S., Ooyama, J. (Daiichi Sankyo Co., Ltd.). Process for production of brominated acetophenone. WO 2008020617.
- Fujimoto, K., Takebayashi, T., Noguchi, Y., Saitou, T. (Daiichi Sankyo Co., Ltd.). Production of 4-methyl-1,2-diarylpyrrole and intermediate for synthesizing the same. JP 2000080078
- Kimura, T., Noguchi, Y., Nakao, A., Suzuki, K., Ushiyama, S., Kawara, A., Miyamoto, M. (Daiichi Sankyo Co., Ltd.). 1,2-Diphenylpyrrole derivatives,their preparation and their therapeutic uses. CA 2201812, EP 0799823, JP 1997823971, US 5908858.

|
References |
1. Bierbach, Ulrich. Platinum acridine anti-cancer compounds and methods thereof. PCT Int. Appl. (2010), 54pp. CODEN: PIXXD2 WO 2010048499 A1 20100429 CAN 152:517954 AN 2010:529827
2. Zaknoen, Sara L.; Lawhon, Tracy. Methods and compositions for the treatment of cancer, tumors, and tumor-related disorders. PCT Int. Appl. (2009), 119 pp. CODEN: PIXXD2 WO 2009070546 A1 20090604 CAN 151:24882 AN 2009:676598
3. Zaknoen, Sara L.; Lawhon, Tracy. Cancer treatment using a 1,2-diphenylpyrrole derivative cyclooxygenase 2 (COX-2) inhibitor and antimetabolite combinations. PCT Int. Appl. (2009), 107pp. CODEN: PIXXD2 WO 2009070547 A1 20090604 CAN 151:24877 AN 2009:672256
4. Estok, Thomas M.; Zaknoen, Sara L.; Mansfield, Robert K.; Lawhon, Tracy. Therapies for treating cancer using combinations of COX-2 inhibitors and anti-HER2(ErbB2) antibodies or combinations of COX-2 inhibitors and HER2(ErbB2) receptor tyrosine kinase inhibitors. PCT Int. Appl. (2009), 121pp. CODEN: PIXXD2 WO 2009042618 A1 20090402 CAN 150:390188 AN 2009:386123
5. Estok, Thomas M.; Zaknoen, Sara L.; Mansfield, Robert K.; Lawhon, Tracy. Therapies for treating cancer using combinations of COX-2 inhibitors and aromatase inhibitors or combinations of COX-2 inhibitors and estrogen receptor antagonists. PCT Int. Appl. (2009), 88pp. CODEN: PIXXD2 WO 2009042612 A1 20090402 CAN 150:390184 AN 2009:385226
6. Estok, Thomas M.; Zaknoen, Sara L.; Mansfield, Robert K.; Lawhon, Tracy. Combination therapy for the treatment of cancer using COX-2 inhibitors and dual inhibitors of EGFR (ErbB1) and HER-2 (ErbB2). PCT Int. Appl. (2009), 87pp. CODEN: PIXXD2 WO 2009042613 A1 20090402 CAN 150:390183 AN 2009:385196
7. Lawhon, Tracy; Zaknoen, Sara; Estok, Thomas; Green, Mark. Patient selection and therapeutic methods using markers of prostaglandin metabolism. PCT Int. Appl. (2009), 121pp. CODEN: PIXXD2 WO 2009009776 A2 20090115 CAN 150:136599 AN 2009:55595
8. Estok, Thomas M.; Zaknoen, Sara L.; Mansfield, Robert K.; Lawhon, Tracy. Methods and compositions for the treatment of cancer, tumors, and tumor-related disorders using combination of a 1,2-diphenylpyrrole derivative and an EGFR inhibitor. PCT Int. Appl. (2009), 104 pp. CODEN: PIXXD2 WO 2009009778 A1 20090115 CAN 150:136628 AN 2009:54177
9. Rohatagi, Shashank; Kastrissios, Helen; Sasahara, Kunihiro; Truitt, Kenneth; Moberly, James B.; Wada, Russell; Salazar, Daniel E. Pain relief model for a COX-2 inhibitor in patients with postoperative dental pain. British Journal of Clinical Pharmacology (2008), 66(1), 60-70.
10. Senzaki, Michiyo; Ishida, Saori; Yada, Ayumi; Hanai, Masaharu; Fujiwara, Kosaku; Inoue, Shin-Ichi; Kimura, Tomio; Kurakata, Shinichi. CS-706, a novel cyclooxygenase-2 selective inhibitor, prolonged the survival of tumor-bearing mice when treated alone or in combination with anti-tumor chemotherapeutic agents. International Journal of Cancer (2008), 122(6), 1384-1390. CODEN: IJCNAW ISSN:0020-7136. CAN 148:440459 AN 2008:228248
11. Kojima, Shunshi; Ooyama, Jo. Process for production of brominated acetophenone as drug intermediate. PCT Int. Appl. (2008), 37pp. CODEN: PIXXD2 WO 2008020617 A1 20080221 CAN 148:262335 AN 2008:220659
12. Ushiyama, Shigeru; Yamada, Tomoko; Murakami, Yukiko; Kumakura, Sei-ichiro; Inoue, Shin-ichi; Suzuki, Keisuke; Nakao, Akira; Kawara, Akihiro; Kimura, Tomio. Preclinical pharmacology profile of CS-706, a novel cyclooxygenase-2 selective inhibitor, with potent antinociceptive and anti-inflammatory effects. European Journal of Pharmacology (2008), 578(1), 76-86.
13. Oitate, Masataka; Hirota, Takashi; Murai, Takahiro; Miura, Shin-ichi; Ikeda, Toshihiko. Covalent binding of rofecoxib, but not other cyclooxygenase-2 inhibitors, to allysine aldehyde in elastin of human aorta. Drug Metabolism and Disposition (2007), 35(10), 1846-1852. CODEN: DMDSAI ISSN:0090-9556. CAN 147:439860 AN 2007:1124386
14. Kiguchi, Kaoru; Ruffino, Lynnsie; Kawamoto, Toru; Franco, Eugenia; Kurakata, Shin-ichi; Fujiwara, Kosaku; Hanai, Masaharu; Rumi, Mohammad; DiGiovanni, John. Therapeutic effect of CS-706, a specific cyclooxygenase-2 inhibitor, on gallbladder carcinoma in BK5.ErbB-2 mice. Molecular Cancer Therapeutics (2007), 6(6), 1709-1717.
15. Moberly, James B.; Xu, Jianbo; Desjardins, Paul J.; Daniels, Stephen E.; Bandy, Donald P.; Lawson, Janet E.; Link, Allison J.; Truitt, Kenneth E. A randomized, double-blind, celecoxib- and placebo-controlled study of the effectiveness of CS-706 in acute postoperative dental pain. Clinical Therapeutics (2007), 29(3), 399-412.
16. Rohatagi, S.; Kastrissios, H.; Gao, Y.; Zhang, N.; Xu, J.; Moberly, J.; Wada, R.; Yoshihara, K.; Takahashi, M.; Truitt, K.; Salazar, D. Predictive population pharmacokinetic/pharmacodynamic model for a novel COX-2 inhibitor. Journal of Clinical Pharmacology (2007), 47(3), 358-370.
17. Moberly, James B.; Harris, Stuart I.; Riff, Dennis S.; Dale, James Craig; Breese, Tara; McLaughlin, Patrick; Lawson, Janet; Wan, Yaping; Xu, Jianbo; Truitt, Kenneth E. A Randomized, Double-Blind, One-Week Study Comparing Effects of a Novel COX-2 Inhibitor and Naproxen on the Gastric Mucosa. Digestive Diseases and Sciences (2007), 52(2), 442-450.
18. Oitate, Masataka; Hirota, Takashi; Koyama, Kumiko; Inoue, Shin-ichi; Kawai, Kenji; Ikeda, Toshihiko. Covalent binding of radioactivity from [14C] rofecoxib, but not [14C] celecoxib or [14C] CS-706, to the arterial elastin of rats. Drug Metabolism and Disposition (2006), 34(8), 1417-1422.
19. Kastrissios, H.; Rohatagi, S.; Moberly, J.; Truitt, K.; Gao, Y.; Wada, R.; Takahashi, M.; Kawabata, K.; Salazar, D. Development of a predictive pharmacokinetics model for a novel cyclooxygenase-2 inhibitor. Journal of Clinical Pharmacology (2006), 46(5), 537-548. CODEN: JCPCBR ISSN:0091-2700. CAN 145:327959 AN 2006:479516
20. Denis, Louis J.; Compton, Linda D. Method using camptothecin compounds, pyrimidine derivatives, and antitumor agents for treating abnormal cell growth. U.S. Pat. Appl. Publ. (2005), 32 pp. CODEN: USXXCO US 2005272755 A1 20051208 CAN 144:17160 AN 2005:1294044
21. Wajszczuk, Charles Paul; Gans, Hendrik J. Dekoning; Di Salle, Enrico; Piscitelli, Gabriella; Massimini, Giorgio; Purandare, Dinesh. Methods using exemestane, alone or with other therapeutic agents, for treating estrogen-dependent disorders. U.S. Pat. Appl. Publ. (2004), 21 pp., Cont.-in-part of WO 2002 72,106. CODEN: USXXCO US 2004082557 A1 20040429 CAN 140:368700 AN 2004:353144
22. Di Salle, Enrico; Piscitelli, Gabriella; Massimini, Giorgio; Purandare, Dinesh; Dekoning, Gans Hendrik. Combined method for treating hormone-dependent disorders with aromatase inactivator exemestane and other therapeutic agents. PCT Int. Appl. (2002), 49 pp. CODEN: PIXXD2 WO 2002072106 A2 20020919 CAN 137:226651 AN 2002:716096
23. McKearn, John P.; Gordon, Gary; Cunningham, James J.; Gately, Stephen T.; Koki, Alane T.; Masferrer, Jaime L. Method of using a cyclooxygenase-2 inhibitor and an integrin antagonist as a combination therapy in the treatment of neoplasia. PCT Int. Appl. (2000), 348 pp. CODEN: PIXXD2 WO 2000038786 A2 20000706 CAN 133:84244 AN 2000:456950
24. McKearn, John P.; Gordon, Gary; Cunningham, James J.; Gately, Stephen T.; Koki, Alane T.; Masferrer, Jaime L. Method of using a cyclooxygenase-2 inhibitor and one or more antineoplastic agents as a combination therapy in the treatment of neoplasia. PCT Int. Appl. (2000), 236 pp. CODEN: PIXXD2 WO 2000038730 A2 20000706 CAN 133:84243 AN 2000:456927
25. McKearn, John P.; Masferrer, Jaime L.; Milas, Luka. Combination therapy of radiation and a cyclooxygenase 2 (COX-2) inhibitor for the treatment of neoplasia. PCT Int. Appl. (2000), 96 pp. CODEN: PIXXD2 WO 2000038716 A1 20000706 CAN 133:84241 AN 2000:456913
26. McKearn, John P.; Gordon, Gary; Cunningham, James J.; Gately, Stephen T.; Koki, Alane T.; Masferrer, Jaime L. Method of using a cyclooxygenase-2 inhibitor and a matrix metalloproteinase inhibitor as a combination therapy in the treatment of neoplasia. PCT Int. Appl. (2000), 437 pp. CODEN: PIXXD2 WO 2000037107 A2 20000629 CAN 133:68922 AN 2000:441655
27. Noguchi, Yasuo; Saito, Toshinori; Fujimoto, Katsuhiko; Takebayashi, Toyonori. Preparation of 4-methyl-1,2-diarylpyrroles and and their intermediates. Jpn. Kokai Tokkyo Koho (2000), 14 pp. CODEN: JKXXAF JP 2000080078 A 20000321 CAN 132:207760 AN 2000:181022
28. Kurakata, Shinichi; Hanai, Masaharu; Kanai, Saori; Kimura, Tomio. Use of cyclooxygenase-2 inhibitors for the treatment and prevention of tumors, tumor-related disorders and cachexia. Eur. Pat. Appl. (1999), 49 pp. CODEN: EPXXDW EP 927555 A1 19990707 CAN 131:82985 AN 1999:440003
29. Kimura, Fumio; Noguchi, Yasuo; Nakao, Akira; Suzuki, Keisuke; Ushiyama, Shigeru; Kawahara, Akihiro; Miyamoto, Masaaki. Diphenylpyrrole derivatives as cyclooxygenase-2 inhibitors. Jpn. Kokai Tokkyo Koho (1999), 69 pp.
30. Kimura, Tomio; Noguchi, Yasuo; Nakao, Akira; Suzuki, Keisuke; Ushiyama, Shigeru; Kawara, Akihiro; Miyamoto, Masaaki. Preparation of 1,2-diphenylpyrroles as cyclooxygenase-2 inhibitors. Eur. Pat. Appl. (1997), 140 pp. CODEN: EPXXDW EP 799823 A1 19971008 CAN 127:331392 AN 1997:678926
31. Rao P N Praveen; Grover Rajesh K Apricoxib, a COX-2 inhibitor for the potential treatment of pain and cancer. IDrugs : the investigational drugs journal (2009), 12(11), 711-22.
|
9-13-2002
|
Method of using COX-2 inhibitors in the treatment and prevention of ocular COX-2 mediated disorders
|
|
|
6-2-1999
|
1,2-diphenylpyrrole derivatives, their preparation and their therapeutic uses
|
|
7-14-2006
|
Use of MEK inhibitors in treating abnormal cell growth
|
|
|
4-7-2006
|
Therapeutic combinations comprising poly (ADP-ribose) polymerases inhibitor
|
|
|
12-9-2005
|
Method for treating abnormal cell growth
|
|
|
6-31-2005
|
Method of using a cyclooxygenase-2 inhibitor and sex steroids as a combination therapy for the treatment and prevention of dismenorrhea
|
|
|
5-4-2005
|
Methods and compositions for treatment and prevention of tumors, tumor-related disorders and cachexia
|
|
|
4-30-2004
|
Compositions of cyclooxygenase-2 selective inhibitors and NMDA receptor antagonists for the treatment or prevention of neuropathic pain
|
|
|
4-30-2004
|
Methods for treating estrogen-dependent disorders
|
|
|
4-16-2004
|
Method of using a COX-2 inhibitor and an alkylating-type antineoplastic agent as a combination therapy in the treatment of neoplasia
|
|
|
3-26-2004
|
Method of using cox-2 inhibitors in the treatment and prevention of ocular cox-2 mediated disorders
|
|
|
3-19-2004
|
Method of using a COX-2 inhibitor and an aromatase inhibitor as a combination therapy
|
|
8-22-2012
|
Methods and Compositions for the Treatment of Cancer, Tumors, and Tumor-Related Disorders
|
|
|
12-21-2011
|
HUMAN MONOCLONAL ANTIBODIES TO ACTIVIN RECEPTOR-LIKE KINASE-1
|
|
|
10-6-2011
|
Use of cyclooxygenase-2 inhibitors for the treatment and prevention of tumours, tumour-related disorders and cachexia
|
|
|
6-30-2010
|
Methods and compositions for the treatment and prevention of tumors, tumor-related disorders and cachexia
|
|
|
11-13-2009
|
HETEROAROMATIC DERIVATIVES USEFUL AS ANTICANCER AGENTS
|
|
|
5-27-2009
|
Human monoclonal antibodies to activin receptor-like kinase-1
|
|
|
4-31-2009
|
BICYCLIC HETEROAROMATIC DERIVATIVES USEFUL AS ANTICANCER AGENTS
|
|
|
11-7-2008
|
Pharmaceutical Compositions Comprising an Amorphous Form of a Vegf-R-Inhibitor
|
|
|
10-24-2008
|
Compositions for the Treatment of Inflammation and Pain Using a Combination of a Cox-2 Selective Inhibitor and a Ltb4 Receptor Antagonist
|
|
|
10-32-2007
|
1,2-Diphenylpyrrole derivatives, their preparation and their therapeutic uses
|
Sage Therapeutics receives fast track designation for status epilepticus therapy

Sage Therapeutics (Originator)
For Epilepsy, status epilepticus
SGE-102; SAGE-547; allopregnanolone; allosteric GABA A receptor modulators (CNS disorders),
Sage Therapeutics receives fast track designation for status epilepticus therapy
Ligand Pharmaceuticals announced that its partner Sage Therapeutics has received fast track designation from the US Food and Drug Administration (FDA) for the Captisol-enabled SAGE-547 to treat status epilepticus.
read at
| Chemical Name: (3α)-Allopregnanolone | ||
| Synonyms: (+)-3α-Hydroxy-5α-pregnan-20-one; (3α,5α)-3-Hydroxypregnan-20-one; 3α,5α-THP; 3α,5α-Tetrahydroprogesterone; 3α-Hydroxy-5α-dihydroprogesterone; 3α-Hydroxy-5α-pregnan-20-one; 3α-Hydroxy-5α-pregnane-20-one; 5α-Pregnan-3α-ol-20-one; 5α-Pregnane-3α-ol-20-one; Allopregnan-3α-ol-20-one; Allopregnanolone; Allotetrahydroprogesterone; | ||
| CAS Number: 516-54-1 | ||
|
||
| Mol. Formula: C21H34O2 | ||
| Appearance: White Solid | ||
| Melting Point: 174-176°C | ||
| Mol. Weight: 318.49 |
SAGE-547 is a GABA(A) receptor modulator in phase I/II clinical trials at Sage Therapeutics as adjunctive therapy for the treatment of adults with super-refractory status epilepticus (SRSE).
In 2014, orphan drug designation was assigned in the U.S for the treatment of status epilepticus. In July 2014, fast track designation was received in the U.S. for the treatment of adults with super-refractory status epilepticus (SRSE).
July 22, 2014
SAGE Therapeutics, a biopharmaceutical company developing novel medicines to treat life-threatening, rare central nervous system (CNS) disorders, announced today that the U.S. Food and Drug Administration (FDA) has granted fast track designation to the SAGE-547 development program. SAGE-547 is an allosteric modulator of GABAA receptors in development for the treatment of adult patients with refractory status epilepticus who have not responded to standard regimens (super-refractory status epilepticus, or SRSE). SAGE is currently evaluating SAGE-547 in a Phase 1/2 clinical trial for the treatment of SRSE. Preliminary data indicate that the first four patients enrolled in the clinical trial met the key efficacy endpoint, in that each was successfully weaned off his or her anesthetic agent while SAGE-547 was being administered. There have also been no reported drug-related serious adverse events in these four patients to date.
“The fast track designation for SAGE-547 recognizes the significant unmet need that exists in the treatment of super-refractory status epilepticus,” said Jeff Jonas, MD, chief executive officer of SAGE Therapeutics. “The receipt of orphan drug designation earlier this year for status epilepticus and the fast track designation are both significant regulatory milestones for SAGE-547, and we will continue to work closely with the FDA to advance our lead compound and the additional programs in our pipeline for the treatment of life-threatening CNS disorders.”
Fast track designation is granted by the FDA to facilitate the development and expedite the review of drug candidates that are intended to treat serious or life-threatening conditions and that demonstrate the potential to address unmet medical needs.
About SAGE-547
SAGE-547 is an allosteric modulator of both synaptic and extra-synaptic GABAA receptors. GABAA receptors are widely regarded as validated drug targets for a variety of CNS disorders, with decades of research and multiple approved drugs targeting these receptor systems. SAGE-547 is an intravenous agent in Phase 1/2 clinical development as an adjunctive therapy, a therapy combined with current therapeutic approaches, for the treatment of SRSE.
About Status Epilepticus (SE)
SE is a life-threatening seizure condition that occurs in approximately 150,000 people each year in the U.S., of which 30,000 SE patients die.1 We estimate that there are 35,000 patients with SE in the U.S. that are hospitalized in the intensive care unit (ICU) each year. An SE patient is first treated with benzodiazepines, and if no response, is then treated with other, second-line, anti-seizure drugs. If the seizure persists after the second-line therapy, the patient is diagnosed as having refractory SE (RSE), admitted to the ICU and placed into a medically induced coma. Currently, there are no therapies that have been specifically approved for RSE; however, physicians typically use anesthetic agents to induce the coma and stop the seizure immediately. After a period of 24 hours, an attempt is made to wean the patient from the anesthetic agents to evaluate whether or not the seizure condition has resolved. Unfortunately, not all patients respond to weaning attempts, in which case the patient must be maintained in the medically induced coma. At this point, the patient is diagnosed as having SRSE. Currently, there are no therapies specifically approved for SRSE.
About SAGE Therapeutics
SAGE Therapeutics (NASDAQ: SAGE) is a biopharmaceutical company committed to developing and commercializing novel medicines to treat life-threatening, rare CNS disorders. SAGE’s lead program, SAGE-547, is in clinical development for super-refractory status epilepticus and is the first of several compounds the company is developing in its portfolio of potential seizure medicines. SAGE’s proprietary chemistry platform has generated multiple new compounds that target GABAA and NMDA receptors, which are broadly accepted as impacting many psychiatric and neurological disorders. SAGE Therapeutics is a public company launched in 2010 by an experienced team of R&D leaders, CNS experts and investors. For more information, please visitwww.sagerx.com.
| Allopregnanolone | |
|---|---|
 |
|
| Identifiers | |
| PubChem | 262961 |
| ChemSpider | 17216124 |
| ChEMBL | CHEMBL38856 |
| Jmol-3D images | Image 1 |
| Properties | |
| Molecular formula | C21H34O2 |
| Molar mass | 318.49 g/mol |
Allopregnanolone (3α-hydroxy-5α-pregnan-20-one or 3α,5α-tetrahydroprogesterone), generally abbreviated as ALLO or as 3α,5α-THP, is an endogenous inhibitory pregnane neurosteroid.[1] It is synthesized from progesterone, and is a potent positive allosteric modulator of the GABAA receptor.[1] Allopregnanolone has effects similar to those of other potentiators of the GABAA receptor such as the benzodiazepines, including anxiolytic, sedative, and anticonvulsant activity.[1]
The 21-hydroxylated derivative of this compound, tetrahydrodeoxycorticosterone (THDOC), is an endogenous inhibitory neurosteroid with similar properties to those of allopregnanolone, and the 3β-methyl analogue of allopregnanolone, ganaxolone, is under development to treat epilepsy and other conditions.[1]
Biosynthesis
The biosynthesis of allopregnanolone starts with the conversion of progesterone into 5α-dihydroprogesterone by 5α-reductase type I. After that, 3α-hydroxysteroid dehydrogenase converts this intermediate into allopregnanolone.[1]
Depression, anxiety, and sexual dysfunction are frequently-seen side effects of 5α-reductase inhibitors such as finasteride, and are thought to be caused, in part, by interfering with the normal production of allopregnanolone.[2]
Mechanism
Allopregnanolone acts as a potent positive allosteric modulator of the GABAA receptor.[1] While allopregnanolone, like other inhibitory neurosteroids such as THDOC, positively modulates all GABAA receptor isoforms, those isoforms containing δ subunits exhibit the greatest potentiation.[1] Allopregnanolone has also been found to act as a positive allosteric modulator of the GABAA-ρ receptor, though the implications of this action are unclear.[3][4] In addition to its actions on GABA receptors, allopregnanolone, like progesterone, is known to be a negative allosteric modulator of nACh receptors,[5] and also appears to act as a negative allosteric modulator of the 5-HT3 receptor.[6] Along with the other inhibitory neurosteroids, allopregnanolone appears to have little or no action at other ligand-gated ion channels, including the NMDA, AMPA, kainate, and glycine receptors.[7]
Unlike progesterone, allopregnanolone is inactive at the nuclear progesterone receptor (nPR).[7] However, allopregnanolone can be intracellularly oxidized into 5α-dihydroprogesterone, which is an agonist of the nPR, and thus/in accordance, allopregnanolone does appear to have indirect nPR-mediated progestogenic effects.[8] In addition, allopregnanolone has recently been found to be an agonist of the newly-discovered membrane progesterone receptors (mPR), including mPRδ, mPRα, and mPRβ, with its activity at these receptors about a magnitude more potent than at the GABAA receptor.[9][10] The action of allopregnanolone at these receptors may be related, in part, to its neuroprotective and antigonadotropic properties.[9][11] Also like progesterone, recent evidence has shown that allopregnanolone is an activator of the pregnane X receptor.[7][12]
Similarly to many other GABAA receptor positive allosteric modulators, allopregnanolone has been found to act as an inhibitor of L-type voltage-gated calcium channels (L-VGCCs),[13] including α1 subtypes Cav1.2 and Cav1.3.[14] However, the threshold concentration of allopregnanolone to inhibit L-VGCCs was determined to be 3 μM (3,000 nM), which is far greater than the concentration of 5 nM that has been estimated to be naturally produced in the human brain.[14] Thus, inhibition of L-VGCCs is unlikely of any actual significance in the effects of endogenous allopregnanolone.[14] Also, allopregnanolone, along with several other neurosteroids, has been found to activate the G protein-coupled bile acid receptor (GPBAR1, or TGR5).[15] However, it is only able to do so at micromolar concentrations, which, similarly to the case of the L-VGCCs, are far greater than the low nanomolar concentrations of allopregnanolone estimated to be present in the brain.[15]
Function
Allopregnanolone possesses a wide variety of effects, including, in no particular order, antidepressant, anxiolytic, stress-reducing, rewarding,[16] prosocial,[17] antiaggressive,[18] prosexual,[17] sedative, pro-sleep,[19] cognitive and memory-impairing, analgesic,[20] anesthetic, anticonvulsant, neuroprotective, and neurogenic effects.[1]
Fluctuations in the levels of allopregnanolone and the other neurosteroids seem to play an important role in the pathophysiology of mood, anxiety, premenstrual syndrome, catamenial epilepsy, and various other neuropsychiatric conditions.[21][22][23]
Increased levels of allopregnanolone can produce paradoxical effects, including negative mood, anxiety, irritability, and aggression.[24][25][26] This appears to be because allopregnanolone possesses biphasic, U-shaped actions at the GABAA receptor – moderate level increases (in the range of 1.5–2 nM/L total allopregnanolone, which are approximately equivalent to luteal phase levels) inhibit the activity of the receptor, while lower and higher concentration increases stimulate it.[24][25] This seems to be a common effect of many GABAA receptor positive allosteric modulators.[26][21] In accordance, acute administration of low doses of micronized progesterone (which reliably elevates allopregnanolone levels), have been found to have negative effects on mood, while higher doses have a neutral effect.[27]
Therapeutic applications
Allopregnanolone and the other endogenous inhibitory neurosteroids have very short half-lives, and for this reason, have not been pursued for clinical use themselves. Instead, synthetic analogs with improved pharmacokinetic profiles, such as ganaxolone, have been synthesized and are being investigated. However, exogenous progesterone, such as oral micronized progesterone (OMP), reliably elevates allopregnanolone levels in the body with good dose-to-serum level correlations.[28] Due to this, it has been suggested that OMP could be described as a prodrug of sorts for allopregnanolone.[28] As a result, there has been some interest in using OMP to treat catamenial epilepsy,[29] as well as other menstrual cycle-related and neurosteroid-associated conditions.
……………………………………….
http://www.google.com/patents/WO2006037016A2?cl=en
Materials and Methods
[0181] The materials and methods used for the follwing experiments have been described in Griffin L.D., et al, Nature Medicine 10: 704-711 (2004). This reference is hereby incorporated by reference in its entirety.
Example 1: Allopregnanolone Treatment of Niemann Pick type-C Mice Substantially Reduces Accumulation of the Gangliosides GMl, GM2, and GM3 in the Brain [0182] Mice were given a single injection of allopregnanolone, prepared in 20% βcyclodextrin in phosphate buffered saline, at a concentration of 25 mg/kg. The injection was on day 7 of life (P7, postnatal day 7). Concentrations of gangliosides GMl, GM2, GM3, were measured as well as other lipids such as ceramides and cerebrosides.
…………………………………………….
WO-2014031792 OR EQ
http://www.google.com/patents/US20140057885?cl=en
…………………………………….
WO-2013112605
http://www.google.com/patents/WO2013112605A2?cl=en
References
- Reddy DS (2010). “Neurosteroids: endogenous role in the human brain and therapeutic potentials”. Prog. Brain Res. 186: 113–37. doi:10.1016/B978-0-444-53630-3.00008-7. PMC 3139029. PMID 21094889.
- Römer B, Gass P (December 2010). “Finasteride-induced depression: new insights into possible pathomechanisms”. J Cosmet Dermatol 9 (4): 331–2. doi:10.1111/j.1473-2165.2010.00533.x. PMID 21122055.
- Morris KD, Moorefield CN, Amin J (October 1999). “Differential modulation of the gamma-aminobutyric acid type C receptor by neuroactive steroids”. Mol. Pharmacol. 56 (4): 752–9. PMID 10496958.
- Li W, Jin X, Covey DF, Steinbach JH (October 2007). “Neuroactive steroids and human recombinant rho1 GABAC receptors”. J. Pharmacol. Exp. Ther. 323 (1): 236–47. doi:10.1124/jpet.107.127365. PMID 17636008.
- Bullock AE, Clark AL, Grady SR, et al. (June 1997). “Neurosteroids modulate nicotinic receptor function in mouse striatal and thalamic synaptosomes”. J. Neurochem. 68 (6): 2412–23. PMID 9166735.
- Wetzel CH, Hermann B, Behl C, et al. (September 1998). “Functional antagonism of gonadal steroids at the 5-hydroxytryptamine type 3 receptor”. Mol. Endocrinol. 12 (9): 1441–51. doi:10.1210/mend.12.9.0163. PMID 9731711.
- Mellon SH (October 2007). “Neurosteroid regulation of central nervous system development”. Pharmacol. Ther. 116 (1): 107–24. doi:10.1016/j.pharmthera.2007.04.011. PMC 2386997. PMID 17651807.
- Rupprecht R, Reul JM, Trapp T, et al. (September 1993). “Progesterone receptor-mediated effects of neuroactive steroids”. Neuron 11 (3): 523–30. PMID 8398145.
- Thomas P, Pang Y (2012). “Membrane progesterone receptors: evidence for neuroprotective, neurosteroid signaling and neuroendocrine functions in neuronal cells”. Neuroendocrinology 96 (2): 162–71. doi:10.1159/000339822. PMC 3489003. PMID 22687885.
- Pang Y, Dong J, Thomas P (January 2013). “Characterization, neurosteroid binding and brain distribution of human membrane progesterone receptors δ and {epsilon} (mPRδ and mPR{epsilon}) and mPRδ involvement in neurosteroid inhibition of apoptosis”. Endocrinology 154 (1): 283–95. doi:10.1210/en.2012-1772. PMC 3529379. PMID 23161870.
- Sleiter N, Pang Y, Park C, et al. (August 2009). “Progesterone receptor A (PRA) and PRB-independent effects of progesterone on gonadotropin-releasing hormone release”. Endocrinology 150 (8): 3833–44. doi:10.1210/en.2008-0774. PMC 2717864. PMID 19423765.
- Lamba V, Yasuda K, Lamba JK, et al. (September 2004). “PXR (NR1I2): splice variants in human tissues, including brain, and identification of neurosteroids and nicotine as PXR activators”. Toxicol. Appl. Pharmacol. 199 (3): 251–65. doi:10.1016/j.taap.2003.12.027. PMID 15364541.
- Hu AQ, Wang ZM, Lan DM, et al. (July 2007). “Inhibition of evoked glutamate release by neurosteroid allopregnanolone via inhibition of L-type calcium channels in rat medial prefrontal cortex”. Neuropsychopharmacology 32 (7): 1477–89. doi:10.1038/sj.npp.1301261. PMID 17151597.
- Earl DE, Tietz EI (April 2011). “Inhibition of recombinant L-type voltage-gated calcium channels by positive allosteric modulators of GABAA receptors”. J. Pharmacol. Exp. Ther. 337 (1): 301–11. doi:10.1124/jpet.110.178244. PMC 3063747. PMID 21262851.
- Keitel V, Görg B, Bidmon HJ, et al. (November 2010). “The bile acid receptor TGR5 (Gpbar-1) acts as a neurosteroid receptor in brain”. Glia 58 (15): 1794–805. doi:10.1002/glia.21049. PMID 20665558.
- Rougé-Pont F, Mayo W, Marinelli M, Gingras M, Le Moal M, Piazza PV (July 2002). “The neurosteroid allopregnanolone increases dopamine release and dopaminergic response to morphine in the rat nucleus accumbens”. Eur. J. Neurosci. 16 (1): 169–73. PMID 12153544.
- Frye CA (December 2009). “Neurosteroids’ effects and mechanisms for social, cognitive, emotional, and physical functions”. Psychoneuroendocrinology. 34 Suppl 1: S143–61. doi:10.1016/j.psyneuen.2009.07.005. PMC 2898141. PMID 19656632.
- Pinna G, Costa E, Guidotti A (February 2005). “Changes in brain testosterone and allopregnanolone biosynthesis elicit aggressive behavior”. Proc. Natl. Acad. Sci. U.S.A. 102 (6): 2135–40. doi:10.1073/pnas.0409643102. PMC 548579. PMID 15677716.
- Terán-Pérez G, Arana-Lechuga Y, Esqueda-León E, Santana-Miranda R, Rojas-Zamorano JÁ, Velázquez Moctezuma J (October 2012). “Steroid hormones and sleep regulation”. Mini Rev Med Chem 12 (11): 1040–8. PMID 23092405.
- Patte-Mensah C, Meyer L, Taleb O, Mensah-Nyagan AG (February 2014). “Potential role of allopregnanolone for a safe and effective therapy of neuropathic pain”. Prog. Neurobiol. 113: 70–8. doi:10.1016/j.pneurobio.2013.07.004. PMID 23948490.
- Bäckström T, Andersson A, Andreé L, et al. (December 2003). “Pathogenesis in menstrual cycle-linked CNS disorders”. Ann. N. Y. Acad. Sci. 1007: 42–53. PMID 14993039.
- Guille C, Spencer S, Cavus I, Epperson CN (July 2008). “The role of sex steroids in catamenial epilepsy and premenstrual dysphoric disorder: implications for diagnosis and treatment”. Epilepsy Behav 13 (1): 12–24. doi:10.1016/j.yebeh.2008.02.004. PMID 18346939.
- Finocchi C, Ferrari M (May 2011). “Female reproductive steroids and neuronal excitability”. Neurol. Sci. 32 Suppl 1: S31–5. doi:10.1007/s10072-011-0532-5. PMID 21533709.
- Bäckström T, Haage D, Löfgren M, et al. (September 2011). “Paradoxical effects of GABA-A modulators may explain sex steroid induced negative mood symptoms in some persons”. Neuroscience 191: 46–54. doi:10.1016/j.neuroscience.2011.03.061. PMID 21600269.
- Andréen L, Nyberg S, Turkmen S, van Wingen G, Fernández G, Bäckström T (September 2009). “Sex steroid induced negative mood may be explained by the paradoxical effect mediated by GABAA modulators”. Psychoneuroendocrinology 34 (8): 1121–32. doi:10.1016/j.psyneuen.2009.02.003. PMID 19272715.
- Bäckström T, Bixo M, Johansson M, et al. (February 2014). “Allopregnanolone and mood disorders”. Prog. Neurobiol. 113: 88–94. doi:10.1016/j.pneurobio.2013.07.005. PMID 23978486.
- Andréen L, Sundström-Poromaa I, Bixo M, Nyberg S, Bäckström T (August 2006). “Allopregnanolone concentration and mood–a bimodal association in postmenopausal women treated with oral progesterone”. Psychopharmacology (Berl.) 187 (2): 209–21. doi:10.1007/s00213-006-0417-0. PMID 16724185.
- Andréen L, Spigset O, Andersson A, Nyberg S, Bäckström T (June 2006). “Pharmacokinetics of progesterone and its metabolites allopregnanolone and pregnanolone after oral administration of low-dose progesterone”. Maturitas 54 (3): 238–44. doi:10.1016/j.maturitas.2005.11.005. PMID 16406399.
- Orrin Devinsky; Steven Schachter; Steven Pacia (1 January 2005). Complementary and Alternative Therapies for Epilepsy. Demos Medical Publishing. pp. 378–. ISBN 978-1-934559-08-6.
Additional reading
- Herd, MB; Belelli, D; Lambert, JJ (2007). Neurosteroid modulation of synaptic and extrasynaptic GABA(A) receptors. Pharmacol. Ther. 116(1):20-34. doi:10.1016/j.pharmthera.2007.03.007.
Ario Kicks Off Efficacy Trial of Chronic Idiopathic Cough Drug
XEN-D0501
Xention (Originator)
XEN-D0501, a novel TRPV1 antagonist, is being developed to treat overactive bladder.
in phase 2 Chronic obstructive pulmonary disease (COPD)
 Ario Kicks Off Efficacy Trial of Chronic Idiopathic Cough Drug
Ario Kicks Off Efficacy Trial of Chronic Idiopathic Cough Drug
Ario Pharma Ltd, the biopharmaceutical company developing innovative new treatments for respiratory disease, announced that it has commenced a Phase 2a study of its oral TRPV1 antagonist, XEN-D0501, for the treatment and prevention of cough in patients with chronic idiopathic cough (CIC).http://www.dddmag.com/news/2014/07/ario-kicks-efficacy-trial-chronic-idiopathic-cough-drug?et_cid=4039308&et_rid=523035093&type=cta
BMS-582949 in phase 2 for Treatment of Antipsoriatics , Rheumatoid arthritis
BMS 582949, PS-540446
UNII-CR743OME9E
CAS 623152-17-0
4-[5-(N-Cyclopropylcarbamoyl)-2-methylphenylamino]-5-methyl-N-propylpyrrolo[2,1-f][1,2,4]triazine-6-carboxamide
4-(5-(Cyclopropylcarbamoyl)-2-methylphenylamino)-5-methyl-N-propylpyrrolo[1,2-f][1,2,4]triazine-6-carboxamide
Bristol-Myers Squibb Company
M.Wt: 406.48
Cas : 623152-17-0 Formula: C22H26N6O2
BMS-582949 had been in phase II clinical trials at Bristol-Myers Squibb for the oral treatment of moderate to severe psoriasis and for the treatment of rheumatoid arthritis (RA) in combination with methotrexate and for the treatment of inflammation in atherosclerotic plaque. However, no recent development has been reported for this research.
…………………..
http://www.google.com/patents/WO2012031057A1?cl=en
The present invention generally relates to a method of treating resistant rheumatic disease, such as refractory rheumatoid arthritis, with a therapeutically effective amount of a dual action p38 inhibitor that is safe and well-tolerated. A dual action p38 kinase inhibitor is a compound that inhibits both activation of p38 kinase and p38 kinase activity in cells.
A large number of cytokines participate in the inflammatory response, including IL- 1 , IL-6, IL-8 and TNF-a. Overproduction of cytokines such as IL-1 and TNF-a are implicated in a wide variety of diseases, including inflammatory bowel disease, rheumatoid arthritis, psoriasis, multiple sclerosis, endotoxin shock, osteoporosis, Alzheimer’s disease, and congestive heart failure, among others. See e.g., Henry et al., Drugs Fut. , 24: 1345- 1354 ( 1999); Salituro et al., Curr. Med. Ckem., 6:807-823 (1999)]. Important mediators of proinflammatory cytokines such as TNFct and IL-1 β,. as well as cellular responses to such cytokines production, are the mitogen-activated protein (MAP) kinases, and in particular, p38 kinase. See e.g., Schieven, G.L., “The biology of p38 kinase: a central role in inflammation”, Current Topics in Medicinal Chemistry, 5 :921 – 928 (2005). Accordingly, modulation of p38 kinase may be useful in the treatment of inflammatory disease including rheumatic diseases such as rheumatoid arthritis (RA).
Compounds that reportedly inhibit p38 kinase and cytokines such as IL-1 and TNF-a for use in treating inflammatory diseases are disclosed in U.S. Patent Nos.
6,277,989 and 6, 130,235 to Scios, Inc; U.S. Patent. Nos. 6, 147,080 and 5,945,41 8 to Vertex Pharmaceuticals Inc; U.S. Patent Nos. 6,251 ,914, 5,977, 103 and 5,658,903 to Smith-Kline Beecham Corp.; U.S. Patent Nos. 5,932,576 and 6,087,496 to G.D. Searle & Co.; WO 00/56738 and WO 01 /27089 to Astra Zeneca; WO 01/34605 to Johnson & Johnson; WO 00/12497 (quinazoHne derivatives as p38 kinase inhibitors); WO 00/56738 (pyridine and pyrimidine derivatives for the same purpose); WO 00/12497 (discusses the relationship between p38 kinase inhibitors); and WO 00/12074 (piperazine and piperidine compounds useful as p38 inhibitors). Other compounds that inhibit p38 kinase are pyrrolotriazine aniline compounds, information on these compounds is disclosed in U.S. Patent Nos. 6,670,357; 6,867,300; 7,034, 151 ; 7, 160,883; 7,21 1,666; 7,253, 167; and U.S. Publication Nos. 2003/023283 1 (published Dec. 18, 2003); 2004/0229877 (published Nov. 1 8, 2004); 2005/0043306 (published Feb. 24, 2005; 2006/0003967 (published Jan. 5, 2006); 2006/0030708 (published Feb. 9, 2006); 2006/0041 124 (published Feb. 23, 2006); 2006/0229449 (published Oct. 12, 2006); 2006/0235020 (published Oct. 19, 2006); and 2007/0213300 (published Sept 13, 2007).
In particular, WO 2003/090912 (U.S. Patent Nos. 7, 160,883, 7,388,009, p38 inhibitor, BMS-582949 (Example 7,
including processes of making and uses thereof.
……………………
http://www.google.com/patents/WO2003090912A9?cl=en
Examples 4-22
Compounds having the formula (Id), above, wherein R4 has the values listed in the following Table, were prepared following the same procedure described for Example 3, using the appropriate amine in place of ra-butylamine.
…………………………
WO 2006020904
http://www.google.com.br/patents/WO2006020904A1?cl=en
EXAMPLE IA St
Part a.
A solution of Example 1 (0.86 g, 2.20 mmol, 1.0 eq.) in THF (4.0 mL) and 1 N aqueous NaOH (9.0 mL, 4.1 eq.) was stirred at 6O0C overnight. After cooling to RT, the reaction mixture was concentrated in vacuo but not to dryness. To the solution at O0C was added 1 N aqueous hydrochloric acid until it was acidic and the precipitate was collected and dried to afford crude Example IA acid (0.51 g, 64.0 % yield). HPLC Ret. t. = 2.400 min.; LC/MS (M+H) + = 366.06+. The filtrate was then extracted with EtOAc (3x) and the organic layers were combined, dried over sodium sulfate, and concentrated in vacuo to give Example IA acid (0.035 g, 4.4 % yield). Part b.
A solution of Part a. acid (0.026 g, 0.071 mmol, 1.0 eq.), EDC (0.021 g, 0.11 mmol, 1.5 eq.), HOBt (0.015 g, 0.11 mmol, 1.5 eq), ^-propylamine (0.015 mL, 0.15 mmol, 2.1 eq.) and DIPEA (0.040 mL, 0.23 mmol, 3.2 eq.) in DMF (0.20 mL) was shaken at RT overnight. Water (1 mL) was added and the precipitate collected by filtration, washed with water, and dried to give Example IA amide (0.021 g, 70% yield); HPLC Ret. t. = 2.883 min.; LC/MS (M+H)+ = 421.18 +.
EJiAMPLE 2 Direct Aminolysis Procedure
n-Buli/THF
Ester Compound I or Hexyllithium/THF
-^
,NH9
1. Aminolysis with hexyllithium
To a dried 100 ml flask was added THF (10 ml) under nitrogen, which was then cooled to -100C. Hexyllithium (2.3 M in hexane, 6.5 ml, 15.0 mmol) was added slowly (exothermic, temperature was up to 5°C), followed by dropwise addition of propylamine (1.01 g, 1.4 ml, 17.1 mmol) at such a rate to maintain the temperature below 5°C. The resulting mixture was stirred at O0C for 20 minutes. A suspension of ester compound I (1.0 g, 2.5 mmol) in THF (12 ml) was added over a 10 minute period (exothermic, T<5°C). After being stirred at 00C for 20 minutes, the mixture was allowed to warm to room temperature and stirred for 5 hours. Ester compound I was <0.1 AP at this point by HPLC analysis. The mixture was cooled to -50C. Acetic acid (2 ml) was added slowly to maintain the temperature <10°C. The resulting thick slurry was stirred at room temperature for 20 minutes, and then solvents were exchanged with DMF (15 ml) on a rotavapor. To the resulting yellow slurry, water (15 ml) was added slowly to keep T<25°C. During the addition of water, the slurry became a clear solution, and a new slurry was formed. The slurry was stirred at room temperature for overnight. In the morning the slurry was filtered and the solid was washed with DMF/water (1:1, 5 ml), water (5 ml) and acetone (5 ml). The cake was dried under vacuum at 55°C for 24 hours to afford 0.90 g of amide product II (yield: 87.2%) as a white solid. HPLC: 99.70 AP.
2. Aminolysis with n-butyllithium
To a dried 100 ml of flask was added THF (10 ml) under nitrogen and then cooled to -100C. n-Butyllithium (2.5 M in hexane, 6.0 ml, 15.0 mmol) was added slowly, followed by dropwise addition of propylamine (0.98 g, 16.5 mmol) at such a rate to keep the temperature below 00C. The resulting mixture was stirred at O0C for 20 minutes. A suspension of ester compound I (1.0 g, 2.5 mmol) in THF (12 ml) was added over a 10 minute period (T<5°C). After being stirred at O0C for 30 minutes, the mixture was allowed to warm to room temperature and stirred for overnight (~22h, Note 1). Compound I was not detected at this point by HPLC analysis. The mixture was cooled to -7°C. Acetic acid (2 ml) was added dropwise to maintain the temperature <10°C. The resulting thick slurry was stirred at 50C for 2 hours and at room temperature for 20 minutes, followed by evaporation on a rotavapor to give a wet yellow solid. To this solid was added acetone (10 ml) and water (20 ml). The slurry was stirred at room temperature for one and half hours. Filtration gave a white solid. This solid was washed with 35% acetone in water (10 ml), water (5 ml) and acetone (5 ml). The cake was dried under vacuum at 55°C for the weekend to afford 0.94g of amide product II (yield: 91.0%) as a white solid. HPLC: 99.76 AP. Note 1: Compound I was -0.056 AP at 2.5 hours.
……………………
WO 2003090912
http://www.google.com/patents/WO2003090912A1?cl=en
……………………..
Discovery of 4-(5-(Cyclopropylcarbamoyl)-2-methylphenylamino)-5-methyl-N-propylpyrrolo[1,2-f][1,2,4]triazine-6-carboxamide (BMS-582949), a clinical p38a MAP kinase inhibitor for the treatment of inflammatory diseases
J Med Chem 2010, 53(18): 6629
http://pubs.acs.org/doi/abs/10.1021/jm100540x
The discovery and characterization of 7k (BMS-582949), a highly selective p38α MAP kinase inhibitor that is currently in phase II clinical trials for the treatment of rheumatoid arthritis, is described. A key to the discovery was the rational substitution of N-cyclopropyl for N-methoxy in 1a, a previously reported clinical candidate p38α inhibitor. Unlike alkyl and other cycloalkyls, the sp2 character of the cyclopropyl group can confer improved H-bonding characteristics to the directly substituted amide NH. Inhibitor 7k is slightly less active than 1a in the p38α enzymatic assay but displays a superior pharmacokinetic profile and, as such, was more effective in both the acute murine model of inflammation and pseudoestablished rat AA model. The binding mode of 7k with p38α was confirmed by X-ray crystallographic analysis.

EXAMPLE 3
Direct Aminolysis
Ester Compound I
Amide Product II
Method A:
A solution of n-propylamine (6.5 eq) in THF (20 ml/g of ester compound I) was cooled to — 5°C and was slowly treated with 2.5 M solution of n-butyllithium (6.1 eq). The mixture was stirred for 10 minutes. At the end of the period, a slurry of ester compound I (1 eq) in THF (14 ml/g of ester compound I) was cannulated into the performed Li-NHPr solution. The reaction mixture was warmed to 25°C and stirred till all of ester compound I was consumed (~ 3 hours). After the reaction was judged to be completed by HPLC, the reaction mixture was cooled to ~0°C and was slowly treated with acetic acid (5 ml/g of ester compound I). The slurry was then warmed to -2O0C and was stirred for 1 hour. At the end of the period, the solvent was distilled under vacuum to the minimum volume and the concentrated slurry was diluted with a solution of acetone (10 ml/g of ester compound I) and water (20 ml/g of ester compound I). The slurry was stirred for 1 hour and was cooled to ~5°C. The slurry was filtered and the cake was washed with acetone (5 ml/g of ester compound I). The cake was dried to give the amide product II (typically in 85% yield and 99 AP).
Method B:
A solution of n-propylamine (20 eq) in 2,2,2-trifmoroethanol (10 ml/g of ester compound I) was slowly treated with 2.5 M solution of n-butyllithium (1.5 eq). The mixture was stirred for 5 minutes. At the end of the period, the starting material, ester compound I, was added and the reaction mixture was warmed to 900C. The reaction mixture was held at 900C for 24 hours and was allowed to cool to ~20°C. The reaction mixture was then analyzed by HPLC. Typically, analysis indicated there was only 1.57 AP of starting material left.
Method C:
A solution of n-propylamine (2 eq) in methylene chloride (10 ml/g of ester compound I) at 200C was slowly treated with 2.0 M solution of trimethylaluminum (4 eq) in hexanes. The mixture was stirred for 15 minutes. At the end of the period, the starting material, ester compound 1 (1 eq), was added and the reaction mixture was warmed to 600C. The reaction mixture was held at 600C for 24 hours and was allowed to cool to ~20°C. The reaction mixture was then slowly quenched with aqueous HCl solution and analyzed by HPLC. Typically, analysis indicated there was 96.8AP of amide compound II product with 0.03 AP of the dipropylamide impurity.
…………………………………….
| WO2003090912A1 * | 15 abr. 2003 | 6 nov. 2003 | Squibb Bristol Myers Co | Pyrrolo-triazine aniline compounds useful as kinase inhibitors |
Liu C, Lin J, Everlof G, Gesenberg C, Zhang H, Marathe PH, Malley M, Galella MA, McKinnon M, Dodd JH, Barrish JC, Schieven GL, Leftheris K.
Bioorg Med Chem Lett. 2013 May 15;23(10):3028-33. doi: 10.1016/j.bmcl.2013.03.022. Epub 2013 Mar 15.
Freebern WJ, Bigwarfe TJ, Price KD, Haggerty HG.
J Immunotoxicol. 2013 Jan-Mar;10(1):106-17. doi: 10.3109/1547691X.2012.736427. Epub 2012 Nov 23.
Liu C, Lin J, Wrobleski ST, Lin S, Hynes J, Wu H, Dyckman AJ, Li T, Wityak J, Gillooly KM, Pitt S, Shen DR, Zhang RF, McIntyre KW, Salter-Cid L, Shuster DJ, Zhang H, Marathe PH, Doweyko AM, Sack JS, Kiefer SE, Kish KF, Newitt JA, McKinnon M, Dodd JH, Barrish JC, Schieven GL, Leftheris K.
J Med Chem. 2010 Sep 23;53(18):6629-39. doi: 10.1021/jm100540x.
BMS-582949: crystalline form of a p38alpha inhibitor? WO2008079857.
Norman P.
Expert Opin Ther Pat. 2009 Aug;19(8):1165-8. doi: 10.1517/13543770902816160.
| WO2000012074A2 | Aug 27, 1999 | Mar 9, 2000 | Sarvajit Chakravarty | Use of piperidines and/or piperazines as inhibitors of p38-alpha kinase | |
| WO2000012497A2 | Aug 27, 1999 | Mar 9, 2000 | Sarvajit Chakravarty | Quinazoline derivatives as medicaments | |
| WO2000056738A1 | Mar 17, 2000 | Sep 28, 2000 | Astrazeneca Ab | Pyridine and pyrimidine derivatives and their use as inhibitors of cytokine mediated disease | |
| WO2001027089A1 | Oct 10, 2000 | Apr 19, 2001 | Astrazeneca Ab | Pyrimidine derivatives | |
| WO2001034605A1 | Oct 27, 2000 | May 17, 2001 | Ortho Mcneil Pharm Inc | SUBSTITUTED 2-ARYL-3-(HETEROARYL)-IMIDAZO[1,2-a]PYRIMIDINES, AND RELATED PHARMACEUTICAL COMPOSITIONS AND METHODS | |
| WO2003090912A1 | Apr 15, 2003 | Nov 6, 2003 | Squibb Bristol Myers Co | Pyrrolo-triazine aniline compounds useful as kinase inhibitors | |
| US4200750 | Dec 8, 1977 | Apr 29, 1980 | Westwood Pharmaceuticals Inc. | 4-Substituted imidazo [1,2-a]quinoxalines | |
| US5658903 | Jun 3, 1996 | Aug 19, 1997 | Smithkline Beecham Corporation | Cytokine inhibitors | |
| US5932576 | May 22, 1998 | Aug 3, 1999 | G. D. Searle & Company | 3(5)-heteroaryl substituted pyrazoles as p38 kinase inhibitors | |
| US5945418 | Mar 20, 1997 | Aug 31, 1999 | Vertex Pharmaceuticals Incorporated | Administering to the mammal to inhibit a mammalian protein kinase p38 which causes cell proliferation, cell death and response to extracellular stimuli | |
| US5977103 | Jan 10, 1997 | Nov 2, 1999 | Smithkline Beecham Corporation | Substituted imidazole compounds | |
| US6087496 | Apr 1, 1999 | Jul 11, 2000 | G. D. Searle & Co. | Enzyme inhibitors | |
| US6130235 | Aug 3, 1998 | Oct 10, 2000 | Scios Inc. | Piperidine moieties coupled to indole, benzimidazole or benzotriazole. | |
| US6147080 | Jun 10, 1997 | Nov 14, 2000 | Vertex Pharmaceuticals Incorporated | Inhibitors of p38 | |
| US6251914 | Jul 1, 1998 | Jun 26, 2001 | Smithkline Beecham Corporation | Treating cytokine mediated diseases | |
| US6277989 | Mar 14, 2000 | Aug 21, 2001 | Scios, Inc. | Quinazoline derivatives as medicaments | |
| US6670357 | Nov 7, 2001 | Dec 30, 2003 | Bristol-Myers Squibb Company | Antiinflammatory agents | |
| US6867300 | Nov 6, 2002 | Mar 15, 2005 | Bristol-Myers Squibb Company | Methods for the preparation of pyrrolotriazine compounds useful as kinase inhibitors | |
| US7034151 | Feb 5, 2004 | Apr 25, 2006 | Bristol-Myers Squibb Company | 1,4-dihydro-4-oxo-pyrrolo[2,1-f][1,2,4]triazine-6-carboxylates; novel approach to the formation of the bicyclic heterocyclic ring system | |
| US7041501 | Oct 31, 2002 | May 9, 2006 | Bristol-Myers Squibb Company | Methods of screening for toxicity of test compounds | |
| US7160883 | Apr 22, 2003 | Jan 9, 2007 | Bristol-Myers-Squibb Company | Pyrrolo-triazine aniline compounds useful as kinase inhibitors | |
| US7211666 | Dec 22, 2004 | May 1, 2007 | Bristol-Myers Squibb Company | N-Cyclopropyl-4-[[5-[(methoxyamino)carbonyl]-2-methylphenyl]amino]-5-methylpyrrolo[2,1-f][1,2,4]triazine-6-carboxamide; aminating with chloramine to produce a pyrrole with a Nitrogen nitrogen bond; reacting with formamide, cyclizing to form the pyrrolotriazine core; kinase inhibitors | |
| US7253167 | Jun 29, 2005 | Aug 7, 2007 | Bristol-Myers Squibb Company | Tricyclic-heteroaryl compounds useful as kinase inhibitors | |
| US7388009 | Oct 3, 2003 | Jun 17, 2008 | Bristol-Myers Squibb Company | Heterocyclic drugs as enzyme inhibitors for Kinase enzymes or prodrugs | |
| US7462616 | Oct 24, 2006 | Dec 9, 2008 | Bristol-Myers Squibb Company | Pyrrolo-triazine aniline compounds useful as kinase inhibitors | |
| US7759343 | Oct 28, 2008 | Jul 20, 2010 | Bristol-Myers Squibb Company | Pyrrolo-triazine aniline compounds useful as kinase inhibitors | |
| US61379001 | Title not available | ||||
| US20030232831 | Apr 22, 2003 | Dec 18, 2003 | Alaric Dyckman | Aryl ketone pyrrolo-triazine compounds useful as kinase inhibitors | |
| US20040229877 | Oct 29, 2003 | Nov 18, 2004 | Katerina Leftheris | Administering pyrrolotriazine carboxamide and benzamide compounds for therapy of p38 kinase-associated conditions | |
| US20050043306 | Oct 3, 2003 | Feb 24, 2005 | Katerina Leftheris | Heterocyclic drugs as enzyme inhibitors for Kinase enzymes or prodrugs | |
| US20060003967 | Jun 28, 2005 | Jan 5, 2006 | Zhongping Shi | Method for preparing pyrrolotriazine compounds | |
| US20060030708 | Aug 5, 2005 | Feb 9, 2006 | Lobben Paul C | Methods for the preparation of pyrrolotriazine compounds | |
| US20060041124 | Oct 14, 2005 | Feb 23, 2006 | Bang-Chi Chen | Process for preparing pyrrolotriazine kinase inhibitors | |
| US20060229449 | Apr 3, 2006 | Oct 12, 2006 | Apurba Bhattacharya | Reacting with chloramine in presence of aqueous base, phase transfer catalyst; anti-cancer agents, kinase inhibitors | |
| US20060235020 | Apr 4, 2006 | Oct 19, 2006 | Soojin Kim | Process for preparing salts of 4-[[5-[(cyclopropylamino)carbonyl]-2-methylphenyl]amino]-5-methyl-N-propylpyrrolo[2,1-f][1,2,4]triazine-6-carboxamide and novel stable forms produced therein | |
| US20070213300 | Mar 6, 2007 | Sep 13, 2007 | Bristol-Myers Squibb Company | Pyrrolotriazine aniline prodrug compounds useful as kinase inhibitors |
Japan First to Approve Alectinib アレクチニブ 塩酸塩 (AF 802) for ALK+ NSCLC
Alectinib (AF802, CH5424802, RG7853, RO5424802)
CAS 1256580-46-7 FREE
1256589-74-8 (Alectinib Hydrochloride)
9-Ethyl-6,11-dihydro-6,6-dimethyl-8-[4-(4-morpholinyl)-1-piperidinyl]-11-oxo-5H-benzo[b]carbazole-3-carbonitrile
| Formula: | C30H34N4O2 |
| M.Wt: | 482.62 |
Mechanism of Action:ALK inhibitor
Indication:Non-small cell lung cancer (NSCLC)
Current Status:Phase II (US,EU,UK), NDA(Japan)
Company:中外製薬株式会社 (Chugai), Roche
Japan First to Approve Alectinib for ALK+ NSCLC
Roche announced that the Japanese Ministry of Health, Labor and Welfare (MHLW) has approved alectinib for the treatment of people living with non-small cell lung cancer (NSCLC) that is anaplastic lymphoma kinase fusion gene-positive (ALK+). The approval was based on results from a Japanese Phase 1/2 clinical study (AF-001JP) for people whose tumors were advanced, recurrent or could not be removed completely through surgery (unresectable).

| Company | Chugai Pharmaceutical Co. Ltd. |
| Description | Anaplastic lymphoma kinase (ALK) inhibitor |
| Molecular Target | Anaplastic lymphoma kinase (ALK) |
| Mechanism of Action | Anaplastic lymphoma kinase (Ki-1) (ALK) inhibitor |
| Therapeutic Modality | Small molecule |
| Latest Stage of Development | Registration |
| Standard Indication | Non-small cell lung cancer (NSCLC) |
| Indication Details | Treat advanced ALK-positive non-small cell lung cancer (NSCLC); Treat non-small cell lung cancer (NSCLC); Treat unresectable progressive or recurrent ALK-positive non-small cell lung cancer (NSCLC) |
| Regulatory Designation |
U.S. – Breakthrough Therapy (Treat advanced ALK-positive non-small cell lung cancer (NSCLC)); |
| Partner |

Alectinib (also known as CH5424802,RO5424802), a second generation oral inhibitor of anaplastic lymphoma kinase (ALK), is being developed by Chugai and Roche for the treatment of patients with ALK-positive non-small cell lung cancer (NSCLC) that has progressed on Xalkori (Crizotinib).
Alectinib was discovered by Chugai Pharmaceutical Co. Ltd. Chugai became a subsidiary of Roche in 2002 and the Swiss group currently owns 59.9 percent of the company.
On October 8, 2013, Chugai Pharmaceutical announced that it has filed a new drug application to Japan’s Ministry of Health, Labour and Welfare (MHLW) for alectinib hydrochloride for the treatment of ALK fusion gene positive non-small cell lung cancer (NSCLC).
IT is a potent and selective ALK inhibitor with IC50 of 1.9 nM.Alterations in the anaplastic lymphoma kinase (ALK) gene have been implicated in human cancers. Among these findings, the fusion gene comprising EML4 and ALK has been identified in non-small cell lung cancer (NSCLC) and fusion of ALK to NPM1 has been observed in anaplastic large cell lymphoma (ALCL). The possibility of targeting ALK in human cancer was advanced with the launch of crizotinib for NSCLC in the U.S. in 2011. The development of resistance to crizotinib in tumors, however, has led to the need for second-generation ALK inhibitors. One of these, alectinib hydrochloride, has been found to be an orally active, potent and highly selective ALK inhibitor with activity in ALK-driven tumor models. Alectinib has shown preclinical activity against cancers with ALK gene alterations, including NSCLC cells expressing the EML4-ALK fusion and ALCL cells expressing the NPM-ALK fusion. Alectinib was well tolerated and active in a phase I/II study conducted in Japan in patients with ALK-rearranged advanced NSCLC and in patients with ALK-positive NSCLC who had progressed on crizotinib. Alectinib has been submitted for approval in Japan for the treatment of ALK fusion gene-positive NSCLC and is in phase I/II development for ALK-rearranged NSCLC in the U.S.

……………..

………………….
WO2012023597
http://www.google.fm/patents/WO2012023597A1?cl=en
(Preparation 30)
Compound F6-20
9 – ethyl-6, 6 – dimethyl-8 – (4 – morpholin-4 – yl – piperidin-1 – yl) -11 – oxo-6 ,11 – dihydro-5H-benzo [b] carbazol-3 – carbonitrile
Under the same conditions as the synthesis of the compound B3-13-1, and the title compound was synthesized from compound F5-49.
1 H-NMR (400MHz, DMSO-D 6) δ: 12.70 (1H, s), 8.32 (1H, d, J = 7.9 Hz), 8.04 (1H, s), 8.00 (1H, s), 7.61 (1H , d, J = 8.5 Hz), 7.34 (1H, s), 3.64-3.57 (4H, m), 3.27-3.18 (2H, m), 2.82-2.66 (4H, m), 2.39-2.28 (1H, m ), 1.96-1.87 (2H, m), 1.76 (6H, s), 1.69-1.53 (2H, m), 1.29 (3H, t, J = 7.3 Hz)
LCMS: m / z 483 [M + H] +
HPLC retention time: 1.98 minutes (analysis conditions U)
Hydrochloride 9 of compound F6-20 – ethyl-6, 6 – dimethyl-8 – (4 – morpholin-4 – yl – piperidin-1 – yl) -11 – oxo-6 ,11 – dihydro-5H-benzo [b I was dissolved at 60 ℃ in a mixture of 10 volumes of methyl ethyl ketone, 3 volumes of water and acetic acid volume 4-carbonitrile -] carbazol-3. I was dropped hydrochloric acid (2N) 1 volume of solution. After stirring for 30 minutes at 60 ℃, and the precipitated solid was filtered and added dropwise to 25 volume ethanol, 9 – Dry ethyl -6,6 – dimethyl-8 – (4 – morpholin-4 – yl – piperidin-1 – yl) I got a one-carbonitrile hydrochloride – 11 – oxo-6 ,11 – dihydro-5H-benzo [b] carbazol-3. Ethyl-6, 6 – 9 – obtained dimethyl-8 – (4 – morpholin-4 – yl – piperidin-1 – yl) -11 – oxo-6 ,11 – dihydro-5H-benzo [b] carbazol-3 – I was pulverized with a jet mill carbonitrile monohydrochloride.
1 H-NMR (400MHz, DMSO-D 6) δ: 12.78 (1H, s), 10.57 (1H, br.s), 8.30 (1H, J = 8.4 Hz), 8.05 (1H, s), 7.99 (1H , s), 7.59 (1H, d, J = 7.9 Hz), 7.36 (1H, s) ,4.02-3 .99 (2H, m) ,3.84-3 .78 (2H, m) ,3.51-3 .48 (2H, m), 3.15-3.13 (1H, s) ,2.83-2 .73 (2H, s) ,2.71-2 .67 (2H, s) ,2.23-2 .20 (2H, m) ,1.94-1 .83 (2H, m), 1.75 (6H, s ), 1.27 (3H, t, J = 7.5 Hz)
FABMS: m / z 483 [M + H] +
I was dissolved at 90 ℃ to 33 volume dimethylacetamide F6-20 F6-20 mesylate. Was added to 168 volumes mesylate solution (2 N) 1.2 volume, ethyl acetate solution was stirred for 4 hours. The filtered crystals were precipitated, and dried to obtain a F6-20 one mesylate. I was milled in a jet mill F6-20 one mesylate salt was obtained.
……………………
Journal of Medicinal Chemistry, 54(18), 6286-6294; 2011
http://pubs.acs.org/doi/abs/10.1021/jm200652u
| WO2002043704A1 * | 30 Nov 2001 | 6 Jun 2002 | Yasuki Kato | Composition improved in solubility or oral absorbability |
| WO2008051547A1 * | 23 Oct 2007 | 2 May 2008 | Cephalon Inc | Fused bicyclic derivatives of 2,4-diaminopyrimidine as alk and c-met inhibitors |
| WO2009073620A2 * | 1 Dec 2008 | 11 Jun 2009 | Newlink Genetics | Ido inhibitors |
| WO2010143664A1 * | 9 Jun 2010 | 16 Dec 2010 | Chugai Seiyaku Kabushiki Kaisha | Tetracyclic compound |
| JP2008280352A | Title not available | |||
| JP2009100783A | Title not available | |||
| JPH0892090A * | Title not available |
|
References |
1: Ignatius Ou SH, Azada M, Hsiang DJ, Herman JM, Kain TS, Siwak-Tapp C, Casey C, He J, Ali SM, Klempner SJ, Miller VA. Next-generation sequencing reveals a Novel NSCLC ALK F1174V mutation and confirms ALK G1202R mutation confers high-level resistance to alectinib (CH5424802/RO5424802) in ALK-rearranged NSCLC patients who progressed on crizotinib. J Thorac Oncol. 2014 Apr;9(4):549-53. doi: 10.1097/JTO.0000000000000094. PubMed PMID: 24736079.
2: Gouji T, Takashi S, Mitsuhiro T, Yukito I. Crizotinib can overcome acquired resistance to CH5424802: is amplification of the MET gene a key factor? J Thorac Oncol. 2014 Mar;9(3):e27-8. doi: 10.1097/JTO.0000000000000113. PubMed PMID: 24518097.
3: Latif M, Saeed A, Kim SH. Journey of the ALK-inhibitor CH5424802 to phase II clinical trial. Arch Pharm Res. 2013 Sep;36(9):1051-4. doi: 10.1007/s12272-013-0157-8. Epub 2013 May 23. Review. PubMed PMID: 23700294.
4: Seto T, Kiura K, Nishio M, Nakagawa K, Maemondo M, Inoue A, Hida T, Yamamoto N, Yoshioka H, Harada M, Ohe Y, Nogami N, Takeuchi K, Shimada T, Tanaka T, Tamura T. CH5424802 (RO5424802) for patients with ALK-rearranged advanced non-small-cell lung cancer (AF-001JP study): a single-arm, open-label, phase 1-2 study. Lancet Oncol. 2013 Jun;14(7):590-8. doi: 10.1016/S1470-2045(13)70142-6. Epub 2013 Apr 30. PubMed PMID: 23639470.
5: Kinoshita K, Asoh K, Furuichi N, Ito T, Kawada H, Hara S, Ohwada J, Miyagi T, Kobayashi T, Takanashi K, Tsukaguchi T, Sakamoto H, Tsukuda T, Oikawa N. Design and synthesis of a highly selective, orally active and potent anaplastic lymphoma kinase inhibitor (CH5424802). Bioorg Med Chem. 2012 Feb 1;20(3):1271-80. doi: 10.1016/j.bmc.2011.12.021. Epub 2011 Dec 22. PubMed PMID: 22225917.
6: Sakamoto H, Tsukaguchi T, Hiroshima S, Kodama T, Kobayashi T, Fukami TA, Oikawa N, Tsukuda T, Ishii N, Aoki Y. CH5424802, a selective ALK inhibitor capable of blocking the resistant gatekeeper mutant. Cancer Cell. 2011 May 17;19(5):679-90. doi: 10.1016/j.ccr.2011.04.004. PubMed PMID: 21575866.
Gadgeel S, Ou SH, Chiappori A, et al: A phase I dose escalation study of a new ALK inhibitor, CH542480202, in ALK+ non-small cell lung cancer patients who have failed crizotinib. Abstract O16.06. Presented at the 15th World Conference on Lung Cancer, Sydney, Australia, October 29, 2013.
Ou SH, Gadgeel S, Chiappori AA, et al: Consistent therapeutic efficacy of CH5424802/RO5424802 in brain metastases among crizotinib-refractory ALK-positive non-small cell lung cancer patients in an ongoing phase I/II study. Abstract O16.07. Presented at the 15th World Conference on Lung Cancer, Sydney, Australia, October 29, 2013.
Kinoshita, Kazuhiro et al,Preparation of tetracyclic compounds such as 11-oxo-5,6-dihydrobenzo[b]carbazole-3-carbonitrile derivatives as anaplastic lymphoma kinase (ALK) inhibitors,Jpn. Kokai Tokkyo Koho, 2012126711, 05 Jul 2012
Furumoto, Kentaro et al, Composition containing tetracyclic compound and dissolution aid (4環性化合物を含む組成物), PCT Int. Appl., WO2012023597, 23 Feb 2012, Also published as CA2808210A1, CN103052386A, EP2606886A1, EP2606886A4, US20130143877
Kinoshita, Kazutomo et al,Design and synthesis of a highly selective, orally active and potent anaplastic lymphoma kinase inhibitor (CH5424802), Bioorganic & Medicinal Chemistry, 20(3), 1271-1280; 2012
Kinoshita, Kazutomo et al,9-Substituted 6,6-Dimethyl-11-oxo-6,11-dihydro-5H-benzo[b]carbazoles as Highly Selective and Potent Anaplastic Lymphoma Kinase Inhibitors, Journal of Medicinal Chemistry, 54(18), 6286-6294; 2011
Kinoshita, Kazuhiro et al, Preparation of tetracyclic compounds such as 11-oxo-5,6-dihydrobenzo[b]carbazole-3-carbonitrile derivatives as anaplastic lymphoma kinase (ALK) inhibitors,Jpn. Tokkyo Koho, 4588121, 24 Nov 2010

Cebranopadol GRT 6005 セブラノパドール a Potent Analgesic NOP and Opioid Receptor Agonist

- C24H27FN2O
- Average mass: 378.482391 Da

Neuropathic pain
Neuropathic pain is caused when peripheral nerves are damaged by mechanical, metabolic or inflammatory way. The pain occurring images are mainly due to the occurrence of spontaneous pain, hyperalgesia and allodynia (pain is already triggered by non-noxious stimuli) in. As a result, the lesions to increased expression of Na + channels and thus to spontaneous activity in the damaged axons and their Nachbaraxonen (England et al., Neurology, 1996, 47, 272-276).The excitability of the neurons is increased and they react to incoming stimuli with an increased discharge frequency. This results in an increased sensitivity to pain, which contributes to the development of hyperalgesia and spontaneous pain (Baron, Clin J Pain 2000;. 16 (2 Suppl), 12-20). The causes and manifestations, and therefore the treatment needs of neuropathischerm pain are varied. They arise as a result of injury or disease of the brain, spinal cord or peripheral nerves.Causes may be operations, such as phantom pain after amputation, stroke, multiple sclerosis, spinal cord injury, alcohol or drug abuse or other toxins, cancers but also
Metabolic diseases such as diabetes, gout, kidney failure or liver cirrhosis, or infectious diseases such as mononucleosis, ehrlichiosis, typhoid, diphtheria, HIV, syphilis or Lyme disease. The pain experience is very different signs and symptoms that can change over time in number and intensity. Paradoxically, patients with neuropathic pain outline a slowdown or failure of acute pain perception and the simultaneous increase of neuropathic pain. The typical symptoms of neuropathic pain as tingling, burning, shooting or described, or radiating electrifying. Pharmacological basis for treatment of neuropathic pain include tricyclic antidepressants and anticonvulsants, which are used as monotherapy or in combination with opioids. These drugs usually provide only a certain pain relief during a pain-free but is often not achieved. The often-adjusting side effects are dose increases while the drug to achieve adequate pain relief often in the way. In fact, a higher dosage of a μ-opioid is often required as the treatment of acute pain, thereby reducing the side effects get even more important for satisfactory treatment of neuropathic pain. By the occurrence of typical μ-opioid tolerance development and the concomitant need for dose escalation of this problem is exacerbated. In summary it can be stated that neuropathic pain is difficult to treat and today is alleviated by high doses of μ-opioids only partially (Saudi Pharm J. 2002, 10 (3), 73-85). There is therefore an urgent need for medicines for the treatment of chronic pain, the dose should not be increased until the occurrence of intolerable side effects to ensure a satisfactory pain treatment.
……………
http://www.google.com/patents/US7547707
Example 24 1,1-(3-Dimethylamino-3-phenylpentamethylene)-6-fluoro-1,3,4,9-tetrahydropyrano[3,4-b]indole hemicitrate, More Non-polar diastereoisomer
4-Dimethylamino-4-phenylcyclohexanone (651 mg, 3 mmoles) and 2-(5-fluoro-1H-indol-3-yl)-ethanol (“5-fluorotryptophol”, 537 mg, 3 mmoles) were initially introduced into abs. MC (20 ml) under argon. Trifluoromethanesulfonic acid trimethylsilyl ester (0.6 ml, 3.1 mmoles) was then added very rapidly. The mixture was stirred at RT for 20 h. For working up, 1 M NaOH (30 ml) was added to the reaction mixture and the mixture was stirred for 30 min. The organic phase was separated, and the aqueous phase which remained was extracted with MC (3×60 ml). The combined organic phases were washed with water (2×30 ml) and dried over sodium sulfate. Methanol (30 ml) was added to the solid residue obtained after the solvent had been distilled off, and the mixture was heated, and stirred for 15 hours. The solid contained in the suspension was filtered off with suction and dried. 955 mg of the more non-polar diastereoisomer of 1,1-(3-dimethylamino-3-phenylpentamethylene)-6-fluoro-1,3,4,9-tetrahydropyrano[3,4-b]indole were obtained (m.p. 284-292° C.). 850 mg of this were dissolved in hot ethanol (900 ml), and a similarly hot solution of citric acid (1 g, 5.2 mmoles) in ethanol (20 ml) was added. After approx. 15 minutes, crystals precipitated out at the boiling point. After cooling to approx. 5° C., the mixture was left to stand for 2 h. The solid formed was filtered off with suction. 640 mg of the hemicitrate were obtained as a white solid (m.p. 258-282° C.).
Example 25 1,1-(3-Dimethylamino-3-phenylpentamethylene)-6-fluoro-1,3,4,9-tetrahydropyrano[3,4-b]indole hemicitrate, More Polar diastereoisomer
4-Dimethylamino-4-phenylcyclohexanone (217 mg, 1 mmole) and 2-(5-fluoro-1H-indol-3-yl)-ethanol (“5-fluorotryptophol”, 179 mg, 1 mmole) were dissolved in conc. acetic acid (4 ml). Phosphoric acid (1 ml, 85 wt. %) was slowly added dropwise to this mixture. The mixture was stirred at RT for 16 h. For working up, the mixture was diluted with water (20 ml), brought to pH 11 with 5 M NaOH and extracted with MC (3×20 ml). The combined organic phases were dried with sodium sulfate and evaporated. The residue (364 mg of white solid) was suspended in hot ethanol (20 ml), and a similarly hot solution of citric acid (185 mg, 0.96 mmole) in ethanol (5 ml) was added. The residue thereby dissolved completely and no longer precipitated out even on cooling to approx. 5° C. Ethanol was removed on a rotary evaporator and the hemicitrate of the more polar diastereoisomer of 1,1-(3-dimethylamino-3-phenylpentamethylene)-6-fluoro-1,3,4,9-tetrahydropyrano[3,4-b]indole was obtained in this way in a yield of 548 mg as a white solid (m.p. 148-155° C.).
| 24 | hemicitrate | more non-polar diastereomer | |
| 25 | hemicitrate | more polar diastereomer | |
(1 r,4r)-6′-fluoro-N,N- dimethyl-4-phenyl-4′,9′-dihydro-3’H-spiro[cyclohexane-1 ,1 ‘-pyrano[3,4-b]indol]-4-amine (free base), has the following structural formula (I):

One particular drug that is of great interest for use in treating cancer pain (and other acute, visceral, neuropathic and chronic pain pain disorders) is (1r,4r)-6′-fluoro-N,N-dimethyl-4-phenyl-4′,9′-dihydro-3′H-spiro[cyclohexane-1,1′-pyrano[3,4b]indol]-4-amine. This drug is depicted below as the compound of formula (I).
The solid forms of (1r,4r)-6′-fluoro-N,N-dimethyl-4-phenyl-4′,9′-dihydro-3′H-spiro[cyclohexane-1,1′-pyrano[3,4b]indol]-4-amine that are known so far are not satisfactory in every respect and there is a demand for advantageous solid forms

In a previous communication, our efforts leading from 1 to the identification of spiro[cyclohexane-dihydropyrano[3,4-b]indole]-amine 2a as analgesic NOP and opioid receptor agonist were disclosed and their favorable in vitro and in vivo pharmacological properties revealed. We herein report our efforts to further optimize lead 2a, toward trans-6′-fluoro-4′,9′-dihydro-N,N-dimethyl-4-phenyl-spiro[cyclohexane-1,1′(3′H)-pyrano[3,4-b]indol]-4-amine (cebranopadol, 3a), which is currently in clinical development for the treatment of severe chronic nociceptive and neuropathic pain.
Discovery of a Potent Analgesic NOP and Opioid Receptor Agonist: Cebranopadol
http://pubs.acs.org/doi/full/10.1021/ml500117c
b]indol]-4-amine, trans-, 2-hydroxy-1,2,3-propanetricarboxylate (2:1)
2.76 (m,6 H); 3.88 (t, 2 H); 6.86 (dt, 1 H); 7.10 (dd, 1 H); 7.30-7.43 (m, 6 H); 10.91 (br
s, 1 H).
overlap); 71.5; 72.2; 102.3 (2JC,F = 23 Hz); 105.6 (3JC,F = 4 Hz); 108.3 (2JC,F = 26 Hz);
156,7 (1JC,F = 231 Hz); 171.3 (2 C), 175.3.HPLC-MS: m/z 378.9 [M + H]+
| US20120034297 * | Aug 4, 2011 | Feb 9, 2012 | Gruenenthal Gmbh | Pharmaceutical dosage forms comprising 6′-fluoro-(N-methyl- or N,N-dimethyl-)-4-phenyl-4′,9′-dihydro-3’H-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine |
| US20130012563 * | Jul 6, 2012 | Jan 10, 2013 | Gruenenthal Gmbh | Crystalline (1r,4r)-6′-fluoro-n,n-dimethyl-4-phenyl-4′,9′-dihydro-3’h-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine |
| WO2004043967A1 | Nov 5, 2003 | May 27, 2004 | Otto Aulenbacher | Spirocyclic cyclohexane derivatives |
| WO2008040481A1 | Sep 26, 2007 | Apr 10, 2008 | Gruenenthal Gmbh | MIXED ORL 1/μ AGONISTS FOR TREATING PAIN |
-
CORAL – Cebranopadol Versus Morphine Prolonged-release in Patients With Chronic Moderate to Severe Pain Related to Cancer
Efficacy, Safety, and Tolerability of Oral Cebranopadol Versus Morphine Sulfate PR in Subjects With Chronic Moderate to Severe Pain Related to Cancer.Average amount of daily rescue medication at the end of the maintenance period.
UK Clinical Trials Gateway, 07 October 2013
-
CORAL XT – Open-label Extension Trial of the CORAL Trial
An Open-label, Multi-site Trial to Describe the Safety and Tolerability of Oral Cebranopadol Administered for 26 Weeks in Subjects With Cancer-related Pain Who Have Completed Treatment in the KF6005/07 Trial.Absolute…
UK Clinical Trials Gateway, 12 December 2013
| WO2004043967A1 * | Nov 5, 2003 | May 27, 2004 | Otto Aulenbacher | Spirocyclic cyclohexane derivatives |
| WO2005066183A1 * | Dec 21, 2004 | Jul 21, 2005 | Gruenenthal Gmbh | Spirocyclic cyclohexane derivatives with affinity for the orl1-receptor |
| US20050153998 * | Aug 19, 2004 | Jul 14, 2005 | Fumitaka Ito | Tetrahydroisoquinoline or isochroman compounds |
| Citing Patent | Filing date | Publication date | Applicant | Title |
|---|---|---|---|---|
| US7799931 * | Feb 17, 2009 | Sep 21, 2010 | Gruenenthal Gmbh | Spirocyclic cyclohexane compounds |
| US7951948 * | Apr 19, 2010 | May 31, 2011 | Gruenenthal Gmbh | Spirocyclic cyclohexane compounds |
| US7960404 | Aug 21, 2009 | Jun 14, 2011 | Gruenenthal Gmbh | Spirocyclic cyclohexane compounds |
| US8034936 | Nov 4, 2010 | Oct 11, 2011 | Gruenenthal Gmbh | Spirocyclic cyclohexane compounds useful to treat substance dependency |
| US8053576 | Feb 17, 2009 | Nov 8, 2011 | Gruenenthal Gmbh | Treating conditions associated with the nociceptin/ORL1 receptor system, e.g. pain, drug withdrawal, anxiety, muscle relaxants, anxiolytic agents; e.g. 1,1-[3-dimethylamino-3-(pyridin-2-yl)pentamethylene]-3,4-dihydro-1H-2,9-diazafluorene |
| US8288406 | Sep 22, 2010 | Oct 16, 2012 | Gruenenthal Gmbh | Hydroxymethylcyclohexylamines |
| US8288430 | Mar 25, 2009 | Oct 16, 2012 | Grunenthal Gmbh | Spiro(5.5)undecane derivatives |
| US8293758 * | Mar 25, 2009 | Oct 23, 2012 | Grunenthal Gmbh | Substituted spirocyclic cyclohexane derivatives |
| US8357705 | Mar 25, 2009 | Jan 22, 2013 | Gruenenthal Gmbh | Substituted cyclohexyldiamines |
| US8404740 | Aug 21, 2009 | Mar 26, 2013 | Gruenenthal Gmbh | Spirocyclic cyclohexane compounds |
| US8614245 * | Jan 8, 2013 | Dec 24, 2013 | Gruenenthal Gmbh | Crystalline (1r,4r)-6′-fluoro-N,N-dimethyl-4-phenyl-4′,9′-dihydro-3′H-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine |
| US8618156 * | Jul 6, 2012 | Dec 31, 2013 | Gruenenthal Gmbh | Crystalline (1r,4r)-6′-fluoro-N,N-dimethyl-4-phenyl-4′,9′-dihydro-3’H-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine |
| US20130012563 * | Jul 6, 2012 | Jan 10, 2013 | Gruenenthal Gmbh | Crystalline (1r,4r)-6′-fluoro-n,n-dimethyl-4-phenyl-4′,9′-dihydro-3’h-spiro[cyclohexane-1,1′-pyrano[3,4,b]indol]-4-amine |

THANKS AND REGARD’S
DR ANTHONY MELVIN CRASTO Ph.D
web link
blogs are

MY BLOG ON MED CHEM
ALL FOR DRUGS ON WEB
http://scholar.google.co.uk/citations?user=bxm3kYkAAAAJ
http://www.stumbleupon.com/stumbler/amcrasto
VIETNAM
ICELAND
RUSSIA

========================
![]()
RedHill Biopharma Ltd. Acquires Phase 2 Oncology Drug Upamostat MESUPRON From Wilex AG

Upamostat
CAS: 590368-25-5
Chemical Formula: C32H47N5O6S
Exact Mass: 629.32470
Synonym: WX 671; WX-671; WX671. Upamostat; Brand name: Mesupron.
IUPAC/Chemical name:
(S)-ethyl 4-(3-(3-(N-hydroxycarbamimidoyl)phenyl)-2-(2,4,6-triisopropylphenylsulfonamido)propanoyl)piperazine-1-carboxylate

RedHill Biopharma Ltd. , an Israeli biopharmaceutical company focused on late clinical-stage drugs for inflammatory and gastrointestinal diseases, including cancer, and WILEX AG , a biopharmaceutical company focused on oncology, based in Munich, Germany, today announced that they have signed an exclusive license agreement for the oncology drug … (more)
Upamostat, also known as Mesupron, WX-671, is an orally bioavailable, 3-amidinophenylalanine-derived, second generation serine protease inhibitor prodrug targeting the human urokinase plasminogen activator (uPA) system with potential antineoplastic and antimetastatic activities. After oral administration, serine protease inhibitor WX-671 is converted to the active Nα-(2,4,6-triisopropylphenylsulfonyl)-3-amidino-(L)-phenyla lanine-4-ethoxycarbonylpiperazide (WX-UK1), which inhibits several serine proteases, particularly uPA; inhibition of uPA may result in the inhibition of tumor growth and metastasis. uPA is a serine protease involved in degradation of the extracellular matrix and tumor cell migration and proliferation.

|
Information about this agent |
WX-671 (Mesupron) is an orally available prodrug of WX-UK1, a serine protease inhibitor that inhibits uPA as well as other serine proteases. WX-UK1 (Setyono-Han et al., Thromb Haemost 2005) and WX-671 have shown to efficiently reduce primary tumor growth and metastasis formation in a variety of animal models. The proteolytic factor uPA and its inhibitor PAI-1 belong to those biological factors which have provided the highest level of evidence (LOE1) in terms of their prognostic and predictive significance. WX-671 is currently the only drug in Phase II aiming at this target.Results: All 95 patients were accrued between Jun 2007 and Aug 2008. Efficacy is assessed by a central reader at regular intervals based on digital CT images. By end of 2009, 2 patients were still on treatment without signs of progression, 64 patients had died. Preliminary analysis of overall survival showed an increase in overall survival from 10.2 mo (gemcitabine alone) to 13.5 mo for the combination of gemcitabine and WX-671. 1-year survival increased from 37% with gemcitabine to 53% when combined with 400 mg WX- 671. Conclusions: The combination of daily oral WX-671 in combination with weekly i.v. gemcitabine was well tolerated. see asco.com’s website.

|
References |
1. Analysis of highly potent amidine containing inhibitors of serine proteases and their N-hydroxylated prodrugs (amidoximes) By Kotthaus, Joscha; Steinmetzer, Torsten; van de Locht, Andreas; Clement, Bernd From Journal of Enzyme Inhibition and Medicinal Chemistry (2011), 26(1), 115-122.
2. Combined treatment of cancer by urokinase inhibition and a cytostatic anti-cancer agent for enhancing the anti-metastatic effect By Schmalix, Wolfgang; Schneider, Anneliese; Setyono-Han, Buddy; Foekens, Johannes From U.S. Pat. Appl. Publ. (2008), US 20080226624 A1 20080918.
3. Peptides and small molecules targeting the plasminogen activation system: towards prophylactic anti-metastasis drugs for breast cancer By Tyndall, Joel D. A.; Kelso, Michael J.; Clingan, Phillip; Ranson, Marie From Recent Patents on Anti-Cancer Drug Discovery (2008), 3(1), 1-13.
4. Synthesis of hydroxyamidine and hydroxyguanidine amino acid or oligopeptide derivatives for use as urokinase plasminogen activator inhibitors for the treatment of cancer and its metastasis By Sperl, Stefan; Buergle, Markus; Schmalix, Wolfgang; Wosikowski, Katja; Clement, Bernd From U.S. Pat. Appl. Publ. (2006), US 20060142305 A1 20060629.
5. Crystalline modifications of N-α-(2,4,6-triisopropylphenylsulfonyl)-3-hydroxyamidino-(l)-phenylalanine-4-ethoxycarbonylpiperazide and/or its salts By Grunenberg, Alfons; Lenz, Jana From PCT Int. Appl. (2006), WO 2006056448 A1 20060601.
6. Synthesis of hydroxyamidine and hydroxyguanidine amino acid or oligopeptide derivatives for use as urokinase plasminogen activator inhibitors for the treatment of cancer and its metastasis By Sperl, Stefan; Burgle, Markus; Schmalix, Wolfgang; Wosikowski, Katja; Clement, Bernd From PCT Int. Appl. (2004), WO 2004103984 A1 20041202.
7. Preparation of 3-amidinophenylalanine derivatives from 3-cyanophenylalanines via reduction and hydrogenation under mild conditions By Ziegler, Hugo; Wikstroem, Peter From PCT Int. Appl. (2003), WO 2003072559 A1 20030904.
1. Buddy et al, Suppression of Rat Brest Cancer Metastasis and Reduction of Primary Tumor Growth by the Small Synthetic Urokinase Inhibitor WX-UK1. Thromb Haemost. 2005, 93:779-786.
2. Ertongur S, Lang S, Mack B, Wosikowski K, Muehlenweg B, Gires O. Inhibition of the invasion capacity of carcinoma cells by WX-UK1, a novel synthetic inhibitor of the urokinase-type plasminogen activator system. Int J Cancer. 2004, 110(6):815-24.
3. Setyono-Han B, Stürzebecher J, Schmalix WA, Muehlenweg B, Sieuwerts AM, Timmermans M, Magdolen V, Schmitt M, Klijn JG, Foekens JA. Suppression of rat breast cancer metastasis and reduction of primary tumour growth by the small synthetic urokinase inhibitor WX-UK1. Thromb Haemost. 2005, 93(4):779-86.

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....