Home » PATENT (Page 5)
Category Archives: PATENT
WO 2016027077, Cipla Ltd, New patent, Dabigatran
(WO2016027077) PROCESSES FOR THE PREPARATION OF DABIGATRAN ETEXILATE AND INTERMEDIATES THEREOF
WO 2016027077, Cipla Ltd, New patent, Dabigatran
CIPLA LIMITED [IN/IN]; Cipla House Peninsula Business Park Ganpatrao Kadam Marg Lower Parel Mumbai 400 013 (IN).
RAO, Dharmaraj Ramachandra; (IN).
MALHOTRA, Geena; (IN).
PULLELA, Venkata Srinivas; (IN).
ACHARYA, Vinod Parameshwaran; (IN).
SINARE, Sudam Nanabhau; (IN)

Dabigatran etexilate (a compound of Formula I) is the international commonly accepted nonproprietary name for ethyl 3-{[(2-{[(4-{(hexyloxy)carbonyl]carbamimidoyl}phenyl)amino]methyl}-1 -methyl-1 H- benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino}propanoate,

(I)
Dabigatran etexilate is the pro-drug of the active substance, dabigatran. The mesylate salt (1 : 1 ) of dabigatran etexilate is known to be therapeutically useful as an oral anticoagulant from the class of the direct thrombin inhibitors and is commercially marketed as oral hard capsules as Pradaxa™ in Australia, Europe and in the United States; as Pradax™ in Canada and as Prazaxa™ in Japan. Additionally, it is also marketed in Europe under the same trade mark for the primary prevention of venous thromboembolic events in adult patients who have undergone elective total hip replacement surgery or total knee replacement surgery.
Dabigatran etexilate was first described in U.S. Patent No. 6,087,380, according to which the synthesis of dabigatran etexilate was carried out in three synthetic steps as depicted in Scheme 1.
Scheme 1

1. HCL , EtOH
2. (NH4)2C03, EtOH

Dabigatran etexilate
II. HCI
The process involves the condensation between ethyl 3-{[3-amino-4-(methylamino)benzoyl] (pyridin-2-yl)amino}propanoate (compound VI) and N-(4-cyanophenyl)glycine (compound VIII) in the presence of Ν,Ν’-carbonyldiimidazole (CDI) in tetrahydrofuran (THF) to give the hydrochloride salt of ethyl 3-{[(2-{[(4-cyanophenyl)amino]methyl}-1-methyl-1 H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino} propanoate (compound IV), which is subsequently reacted with ethanolic hydrochloric acid, ethanol and ammonium carbonate to give the hydrochloride salt of ethyl 3-{[(2-[{(4-carbamimidoylphenyl)amino]methyl}-1-methyl-1 H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino} propanoate (compound II). Finally, the reaction between compound II and n-hexyl chloroformate (compound IX), in the presence of potassium carbonate, in a mixture of THF and water, affords dabigatran etexilate of Formula (I) after work- up and chromatographic purification. However, no information is given about the purity of the isolated dabigatran etexilate (I) product. Further, the process is not viable industrially as it requires chromatographic purification in several of its steps, thus making it very difficult and costly to implement on an industrial scale.
In order to simplify the process for obtaining dabigatran etexilate described in U.S. Patent No. 6,087,380, several alternative processes have been developed and reported in the art.
EP2118090B discloses a process for the preparation of the intermediate compound of Formula (II) by crystallization from a salt with p-toluenesulfonic acid. The amidine salt (ll-pTsOH) is obtained from a compound of formula (IV), which is also isolated in the form of a hydrobromide salt, (IV-HBr).
EP2262771A discloses a process for the preparation of the intermediate compound of Formula (IV), which is obtained in the form of a salt with oxalic acid. This document indicates that the oxalate intermediate of the compound (IV) crystallizes easily and is a good synthesis intermediate to obtain the amidine hydrochloride salt (ll-HCI) with high purity on an industrial scale. The compound (IV) in oxalate salt form is transformed in dabigatran following the process disclosed in WO 98/37075.
WO 2006/000353 describes an alternative process for the synthesis of dabigatran etexilate as depicted in Scheme 2.

Dabigatran etexilate
The process involves condensation between ethyl 3-{[3-amino-4-(methylamino)benzoyl](pyridin-2-yl)amino}propanoate (compound VI) and 2-[4-(1 ,2,4-oxadiazol-5-on-3-yl)phenylamino]acetic acid (compound Villa) in the presence of a coupling agent such as CDI, propanephosphonic anhydride (PPA), or pivaloyl chloride, to give ethyl 3-{[(2-{[(4-{1 ,2,4-oxadiazol-5-on-3-yl}phenyl)amino]methyl}-1 -methyl-1 H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino}propanoate (compound IVa), which is subsequently hydrogenated in the presence of a palladium catalyst to give ethyl 3-{[(2-{[(4-carbamimidoylphenyl)amino]methyl}-1-methyl-1 H-benzimidazol-5-yl)carbonyl](pyridin-2-yl)amino} propanoate (compound II). The compound II is acylated with n-hexyl chloroformate (compound I) to give dabigatran etexilate. Finally, dabigatran etexilate is converted into its mesylate salt. Although the patent describes the HPLC purities of intermediate compounds II, IVa, Villa and VI, no information is given concerning the purity of the isolated dabigatran etexilate or the mesylate salt thereof.
WO 2010/045900 discloses a process to prepare the intermediate amidine hydrochloride compound (ll-HCI) from the oxalate salt of the compound (IV) by reacting with hydrogen chloride in ethanol, followed by reaction with ammonium carbonate to avoid chromatography which is not feasible on an industrial scale.
WO 2014/012880 discloses a process to prepare an intermediate of dabigatran etexilate (compound IV) by reacting carboxylic acid (compound VIII) with diamaine (compound VI) in the presence of the coupling agent CDI, followed by reaction with 6 equivalents of acetic acid at 130°C to obtain compound IV in acetate salt form, having a purity of 94%. The isolated solid is further recrystallized from ethanol to obtain a purity of 99%. The purified (compound IV. acetate) is reacted with hydrogen chloride in the presence of an alcohol, and then with ammonia in an aqueous medium to form the amidine hydrochloride salt (compound ll-HCI) in the presence of water.
The synthesis of intermediate compound II has been reported in the patent literature and known methods require either chromatographic purification or a lengthy purification procedure, such as converting the compound into the HCI salt followed by recrystallization, to obtain 97% pure intermediate compound II. In previously reported methods, the product yield is undesirably less than 50 %.
Similarly, the intermediate compound IV prepared by CDI mediated coupling with glycine derivatives followed by acetic acid mediated cyclization according to known methods results in the formation of highly impure products, which require purification by either column chromatography or by converting the crude reaction mixture to suitable salts. Previously reported methods afford low product yields and purity, which mean that such processes are not suitable for the commercial scale production of dabigatran.
In view of the foregoing, it is of great interest to continue investigating and develop other alternative simplified processes for the large scale industrial production of the active pharmaceutical ingredient dabigatran etexilate or salts thereof, which avoid complicated and costly purification steps in the synthesis of intermediates, while maintaining a high quality of synthesis intermediates and improving the yields of each step of reaction.


SCHEME 3


SCHEME4
Examples:
Example 1. Preparation of DAB Glycin-CDI complex of Formula (VII)
71.02 g (0.438 mol) of CDI was dissolved in 700 ml dichloromethane under nitrogen atmosphere. Added 66.89 g (0.379 mol) of 2-(4-cyanophenylamino)acetic acid of Formula (VIII), under stirring at 20-25°C and stirred for 90-100 minutes. Solid was isolated by filtration under nitrogen atmosphere and washed with 100 ml dichloromethane to yield DAB Glycin-CDI complex.
Example 2. Preparation of ethyl 3-(2-((4-cyanophenylamino)methyl)- l-methyl-N- (pyridin-2-yl)-IH-benzo[d]- imidazole-5-carboxamido) propanoate of Formula (IV)
DAB Glycin-CDI Complex obtained in Example 1 was stirred in 650 ml toluene. Added 100 g (0.292 mol) of ethyl 3-(3-amino-4-(methyl amino)-N-(pyridin-2-yl)benzamido)propanoate of Formula (VI) to the reaction mass and stirred for 3 hours at -45-50°C. The reaction mass was further refluxed for 3 hours. The reaction mass was cooled to 75-80°C, added 50 ml ethanol, further cooled to 20-25°C and stirred for 6 hours. The solid was isolated by filtration and washed with 100 ml toluene.
The wet cake was stirred in 500 ml water at 20-25°C for about 1 hour. The solid was isolated by filtration, washed with 100 ml water and dried in vacuum below 60 °C.
Yield: 120 g
Efficiency: 85%
Example 3. Preparation of ethyl 3-(2-((4-carbamimidoylphenylamino)methyl)-l-methyl-N-(pyridin-2-yl)-IH-benzo[d]imidazole-5-carboxamido) propanoate of Formula (II)
100 g (0.207 mol) of ethyl 3-(2-((4-cyanophenylamino)methyl)- l-methyl-N- (pyridin-2-yl)-IH-benzo[d]- imidazole-5-carboxamido) propanoate of Formula (IV) was added to 1000 ml EtOH.HCI (32-35%w/w) at 5-10°C under nitrogen atmosphere and stirred for 24 hours at 15-20°C. The solvent was distilled off in vacuum below 40°C. Added 500 ml ethanol and cooled to 0-5°C. The pH of the reaction mass was adjusted to 9.5-10.0 by addition of 400 ml EtOH.NH3 (10-13%w/w). The temperature of the reaction mass was raised to 20-25°C and stirred for 12 hours. The reaction mass was filtered and the clear filtrate was partially distilled to the half volume below 40°C. The temperature of the reaction mass was raised to 55-60°C. Added 600 ml ethyl acetate at reflux. The reaction mass was cooled to 20-25°C and stirred further for 5 hours. The solid was isolated by filtration and washed with 100 ml-ethyl acetate. The solid was dried in vacuum below 45 °C.
Yield: 72.5 g
Efficiency: 70%
Example 4. Preparation of DAB etexilate of Formula (I)
120 ml acetone, 60 ml water, 16.6 g (0.120 mol) potassium carbonate and 20g (0.040 mol) of ethyl 3-(2-((4-carbamimidoylphenylamino)methyl)-l-methyl-N-(pyridin-2-yl)-IH-benzo[d]imidazole-5-carboxamido) propanoate of Formula (II) were stirred at 20-25°C. A solution of 9.88 g (0.060 mol) of hexyl chloroformate of Formula (IX) in 50 ml acetone was added to the reaction mass at 15-20°C in 1 .5 hours. The reaction mass was further stirred for 2 hours at 15-20°C. The precipitated solid was filtered and washed with 40 ml water.
The wet cake was dissolved in 160 ml acetone at 20-25°C. The insoluble were removed by filtration. Added 160 ml water to the clear filtrate at 20-25°C in 2 hours and the reaction mass was further stirred for 2 hours. The solid was isolated by filtration, washed with mixture of acetone : water (1 : 1), and dried under vacuum below 45°C to obtain dabigatran etexilate.
Yield: 18.85 g
Efficiency: 75%
Purification:
18 g of Dabigatran etaxilate was stirred in mixture of acetone: ethanol: ethyl acetate (1.5:0.5:6 volumes) at 50-55°C and stirred for 20 minutes. The reaction mass was cooled to 20-25°C and further chilled to 15-20 °C for 3 hours. The solid was isolated by filtration, washed with ethyl acetate and dried under vacuum below 45°C to obtain dabigatran etexilate.
Yield: 13.5 g
Efficiency: 75%
Example 5. Preparation of DAB etexilate mesylate
10 g (0.02 mol) of dabigatran etexilate was dissolved in 200 ml acetone under nitrogen atmosphere. The temperature of the reaction mass was raised to 50-55°C and treated with a solution of 1.86 g (0.0193 mol) of methane sulfonic acid in 50 ml acetone. The reaction mixture was stirred for 45 minutes, then cooled to 20-25 °C and further stirred for 45 minutes. The solid was isolated by filtration, washed with acetone and dried under vacuum below 45°C to obtain dabigatran etexilate mesylate.
Yield: 10 g
Efficiency: 86%
Example 6. Preparation of ethyl 3-(2-((4-carbamimidoylphenylamino)methyl)-l-methyl-N-(pyridin-2-yl)-IH-benzo[d]imidazole-5-carboxamido) propanoate of Formula (ll)using N-acetyl cysteine
10 g (0.020 mol) of ethyl 3-(2-((4-cyanophenylamino)methyl)- l-methyl-N- (pyridin-2-yl)-IH-benzo[d]- imidazole-5-carboxamido) propanoate of Formula (IV) was dissolved in 600 ml EtOH.NH3 (15-18%w/w) and stirred at 25°C. Added 3.38 g (0.020 mol) of N-acetyl cysteine to the reaction mass and stirred for 24 hours at 70-75°C under 2.0-2.3 kg of pressure. The ethanol was distilled under vacuum and residue was purified by column.
Yield: 5.5 g
Efficiency: 53%
Example 7. Preparation of DAB Amidine of Formula (II) using N-acetyl cysteine
10 g (0.020 mol) of ethyl 3-(2-((4-cyanophenylamino)methyl)- l-methyl-N- (pyridin-2-yl)-IH-benzo[d]- imidazole-5-carboxamido) propanoate of Formula (IV) with 3.5 g (0.021 mol) of N-acetyl-(S)cysteine were initially charged in 10 ml of ethanol. The reaction mixture was heated to 60-65°C, and saturated with ammonia. After 4 hours, ethanol was distilled under vacuum to obtain titled compound as a solid.
Yield: 7.0 g
Efficiency: 67%
Example 8. Preparation of 2-pyridyl impurity B
Part I: 12.0g (0.016 mol) of dabigatran etexilate was added to the solution of 2.8 g (0.07 mol) sodium hydroxide (in 300 ml water and 150 ml ethanol. The reaction mass was stirred for 5 hours. The solution was concentrated under vacuum and neutralized with aq. solution of citric acid (10%v/v). The solid was separated by filtration and washed with cold water and dried under vacuum to afford the acid as a white crystal.
Yield: 8.50 g
Part 11:10 g ( 0.0166 mol) of DAB-Acid obtained in part I was stirred with 25 ml thionyl chloride under nitrogen The temperature of the reaction mass was raised to 40-45°C and maintained for 1 hour. Thionyl chloride was distilled under vacuum completely The residue was stirred in solution of 100 ml toluene and 10 ml triethyl amine at 5-10°C. Added 3.1 g (0.0329 mol) 2-amino pyridine to the reaction mass at 5-10°C under nitrogen atmosphere. Temperature of the reaction mass was raised to 50-55°C and stirred. Toluene was distilled under vacuum and the residue was dissolved in 150 ml DCM. The organic layer was washed with water, dried on sodium sulfate. The organic layer was distilled under vacuum to obtain t crude 2-Pyridyl impurity which was purified by column chromatography.
Yield: 4.0 g
Example 9. Preparation of ethyl 3-(2-((4-cyanophenylamino)methyl)- l-methyl-N- (pyridin-2-yl)-IH-benzo[d]- imidazole-5-carboxamido) propanoate of Formula (IV)
To a solution of N, N-Carbonyldiimidazole (1.17kg, 7.21 mol) and dichloromethane (1 1.25 L), added 2-(4-cyanophenylamino)acetic acid of Formula (VIII), (1.15Kg,6.52 mol) at 30°C under nitrogen atmosphere. The reaction mixture was stirred for 90-100 min and the resulting solid was filtered under nitrogen atmosphere to obtain form Dab glycine CDI complex of Formula (VII).
Dab glycine CDI complex of Formula (VII) was stirred in toluene (9.0L). Added ethyl 3-(3-amino-4-(methyl amino)-N-(pyridin-2-yl)benzamido)propanoate of Formula (VI) (1.5Kg, 4.38 mol) and maintained the reaction at 45-55°C for 3.0 hrs to form DAB coupling intermediate of Formula (V), which further heated to 90-100°C for 3.0 hrs. The reaction mixture was cooled to 25-30°C and the solid precipitated out was isolated by filtration. The wet cake was stirred in water (9.0L), filtered and dried in vacuum below 60 °C to obtain titled compound.
Yield: 1.80kg
Efficiency: 85 %
Example 10. Preparation of ethyl 3-(2-((4-carbamimidoylphenylamino)methyl)-l-methyl-N-(pyridin-2-yl)-IH-benzo[d]imidazole-5-carboxamido) propanoate of Formula (II)
A mixture of ethyl 3-(2-((4-cyanophenylamino)methyl)-l-methyl-N-(pyridin-2-yl)-IH-benzo[d]-imidazole-5-carboxamido) propanoate of Formula (IV) (1.73 kg,3.58mol) was stirred in ethanol denatured with toluene HCI (32-35 % w/w) (20.76 L) at 15- 20°C for 24 hrs. Reaction mass was distilled out completely and the residue was treated with ethanol denatured with toluene. NH3 (at 10-15% w/w) was added to get the pH 9.0-9.5. The reaction mixture was stirred further for 12.0 hrs. The inorganic was separated by filtration and the filtrate was distilled out and the residue was stirred in ethyl acetate (10 L) . The solid was isolated by filtration and washed with ethyl acetate. The solid was dried in vacuum below 45°C to obtain titled compound.
Yield: 1.70kg
Efficiency: 95 %
Example 11. Preparation of DAB etexilate of Formula (I)
To a solution of ethyl 3-(2-((4-carbamimidoylphenylamino)methyl)-l-methyl-N-(pyridin-2-yl)-IH-benzo[d]imidazole-5-carboxamido) propanoate of Formula (II) (1.61 kg, 3.22mol ), acetone (19.32 L), water( 9.66 L) and potassium carbonate (1.34Kg, 9.69moles ) was added hexyl chloroformate (0.795 kg, 83 moles) slowly at 20-25°C in 2-3 hrs. The reaction mixture was stirred further for 90 min. The solid was filtered and stirred in 7.5 volumes of acetone at 35-40°C. To the clear solution was added dropwise, 7.5 volumes of purified water. The reaction mixture was stirred further for 2 hours at 20-25°C, solid was isolated by filtration and dried at 45°C. The solid was stirred in a mixture of ethanol: ethyl acetate (1 : 10 volume) at 35-40°C to get clear solution, then gradually cooled to 10-15°C and further stirred for 6.0 hours. The solid was isolated by filtration, washed with ethyl acetate and dried under vacuum below 45°C to obtain dabigatran etexilate.
Yield: 1.10 kg
Efficiency: 65%
Example 12. Preparation of DAB etexilate mesylate
Dabigatran etexilate (1.0Kg, 1.59mol) was dissolved in acetone (20.0L) at 50-55°C under nitrogen atmosphere and treated with a solution of methane sulfonic acid (0.15Kg, 1 .56mol) in acetone (1 .5L). The reaction mixture was stirred for 45 minutes, then cooled to 20-25 °C and further stirred for 45 minutes. The solid was isolated by filtration, washed with acetone and dried under vacuum below 45°C to obtain dabigatran etexilate mesylate.
Yield: 1.10kg Efficiency: 95 %
//////////WO-2016027077, WO 2016027077, Cipla Ltd, New patent, Dabigatran
WO 2016025720, New Patent, by Assia Chemicals and Teva on Ibrutinib


WO 2016025720, New Patent, by Assia Chemicals and Teva on Ibrutinib
ASSIA CHEMICAL INDUSTRIES LTD. [IL/IL]; 2 Denmark Street 49517 Petach Tikva (IL)
TEVA PHARMACEUTICALS USA, INC. [US/US]; 1090 Horsham Road P.O. Box 1090 North Wales, PA 19454 (US)
COHEN, Meital; (IL).
COHEN, Yuval; (IL).
MITTELMAN, Ariel; (IL).
MOHA-LERMAN, Elana, Ben; (IL).
TZANANI, Idit; (IL).
LEVENFELD, Leonid; (IL)
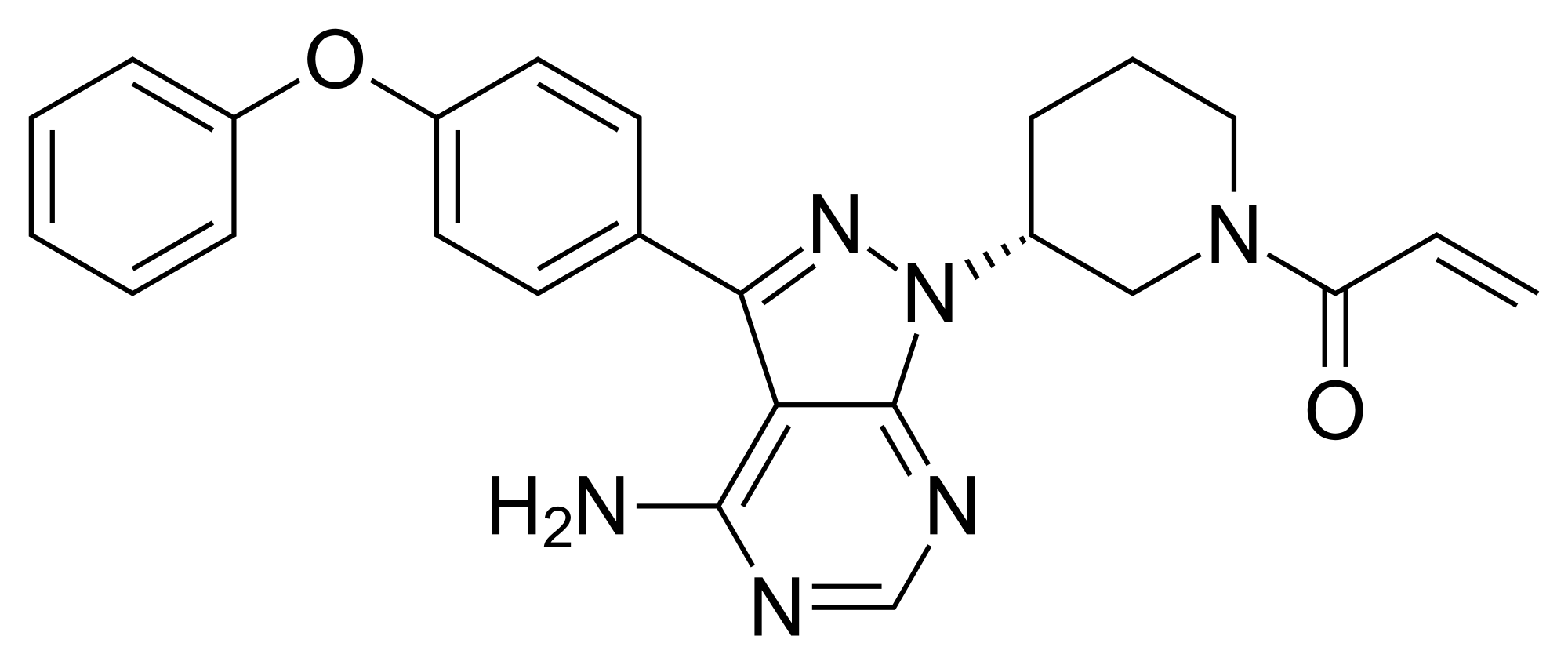
The present invention encompasses solid state forms of Ibrutinib, including forms G, J and K, and pharmaceutical compositions thereof.
Ibrutinib, l-{(3R)-3- [4-amino-3-(4-phenoxyphenyl)-lH-pyrazolo [3,4-d] pyrimidin-l-yl] piperidin-l-yl] prop-2-en-l-one, having the following formula,

is a kinase inhibitor indicated for the treatment of patients with B-cell lymphoma.
Ibrutinib is described in US 7,514,444 and in US 8,008,309. Solid state forms, including forms A-F and amorphous form of Ibrutinib, are described in WO 2013/184572.
Polymorphism, the occurrence of different crystalline forms, is a property of some molecules and molecular complexes. A single molecule may give rise to a variety of polymorphs having distinct crystal structures and physical properties like melting point, thermal behaviors (e.g. measured by thermogravimetric analysis – “TGA”, or differential scanning calorimetry – “DSC”), X-ray diffraction pattern, infrared absorption fingerprint, and solid state (13C-) NMR spectrum. One or more of these techniques may be used to distinguish different polymorphic forms of a compound.
Different salts and solid state forms (including solvated forms) of an active pharmaceutical ingredient may possess different properties. Such variations in the properties of different salts and solid state forms and solvates may provide a basis for improving formulation, for example, by facilitating better processing or handling characteristics, changing the dissolution profile in a favorable direction, or improving stability (polymorph as well as chemical stability) and shelf-life. These variations in the properties of different salts and solid state forms may also offer improvements to the final dosage form, for instance, if they serve to improve bioavailability. Different salts and solid state forms and solvates of an active pharmaceutical ingredient may also give rise to a variety of polymorphs or crystalline forms, which may in turn provide additional opportunities to assess variations in the properties and characteristics of a solid active pharmaceutical ingredient.
Discovering new solid state forms and solvates of a pharmaceutical product may yield materials having desirable processing properties, such as ease of handling, ease of processing, storage stability, and ease of purification, or may serve as desirable intermediate crystal forms that facilitate purification or conversion to other polymorphic forms. New solid state forms of a pharmaceutically useful compound can also provide an opportunity to improve the performance characteristics of a pharmaceutical product. It enlarges the repertoire of materials that a formulation scientist has available for formulation optimization, for example by providing a product with different properties, e.g., a different crystal habit, higher crystallinity or polymorphic stability which may offer better processing or handling characteristics, improved dissolution profile, or improved shelf-life (chemical/physical stability). For at least these reasons, there is a need for additional solid state forms (including solvated forms) of ibrutinib.
Example 1: Preparation of Crystalline Form G of Ibrutinib
[0057] Ibrutinib (0.3 gr, amorphous form) was dissolved in acetic acid (1.2 ml) and the obtained solution was stirred at room temperature overnight followed by the addition of water (2.4 ml). A gum was obtained which was turned into cloudy solution upon stirring. The obtained cloudy solution was stirred for 9 days at room temperature and the obtained precipitate was collected by suction filtration. The obtained solid was dried in an oven at 40°C under vacuum for 16h to obtain form G of Ibrutinib (0.12g), as confirmed by XRPD.
Example 2: Preparation of Crystalline Form J of Ibrutinib
Ibrutinib (5.2 g) was dissolved in Anisole (15 ml), the solution was stirred at room temperature until precipitation was occurred. The slurry was stirred over night at room temperature and the precipitate was collected by suction filtration. The cake was dried in a vacuum oven at 50°C overnight. The obtained product was analyzed by XRPD and found to be form J.
Example 3: Preparation of Crystalline Form J of Ibrutinib
Ibrutinib (10.5 g) was dissolved in Anisole (21 ml) and MTBE (32 ml), the solution was stirred at room temperature until precipitation was occurred . The slurry was heated to reflux and was gradually cooled to room temperature. After 3 hours the precipitate was collected by suction filtration. The obtained product was analyzed by XRPD and found to be form J.
Example 4: Preparation of Crystalline Form G of Ibrutinib
A I L reactor was charged with Ibrutinib (100 g), acetonitrile (417.5 ml_), water (417.5 ml_) and acetic acid (27.15 g). The mixture was heated to 90°C until dissolution; the solution was gradually cooled to 0°C, then heated to 25°C and stirred over 48 hours at 25°C. The obtained slurry was filtered and washed with water (100 ml_). The product was dried overnight in a vacuum oven at 40°C to obtain Ibrutinib form G (72.9 g), as confirmed by XRPD.
Example 5: Preparation of Crystalline Form G of Ibrutinib
A 250 mL round flask was charged with isopropanol (10 ml_) and water (120 ml_), and a solution of Ibrutinib (10 g) in Acetic acid (40 mL) was added dropwise. The mixture was stirred at 25°C for 48 hours. The obtained slurry was filtered and the wet product was slurried in water (50 mL) for 5 min and filtered again. The obtained product was dried under vacuum at room temp in the presence of a N2 atmosphere and found to be form G, as confirmed by XRPD.
Example 6: Preparation of Crystalline Form K of Ibrutinib
Ibrutinib (10 g) was dissolved in toluene (50 mL) and dimethylformamide (DMA) (30 mL) at room temperature, the solution was heated to 50 °C and water (30 mL) was added. The phases were separated and methyl tert-butyl ether (MTBE) (30 mL) was added to the organic phase. The solution was cooled in an ice bath and seeded with amorphous Ibrutinib. After further stirring at the same temperature the obtained slurry was filtered under vacuum. The obtained solid was analyzed by XRPD and found to be Form K (Figure 5).

//////////////WO 2016025720, WO-2016025720, New Patent, Assia Chemicals, Teva, Ibrutinib
WO 2016024224, New Patent, Trelagliptin, SUN PHARMA


WO 2016024224, New Patent, Trelagliptin, SUN PHARMA
SUN PHARMACEUTICAL INDUSTRIES LIMITED [IN/IN]; Sun House, Plot No. 201 B/1 Western Express Highway Goregaon (E) Mumbai, Maharashtra 400 063 (IN)
BARMAN, Dhiren, Chandra; (IN).
NATH, Asok; (IN).
PRASAD, Mohan; (IN)
The present invention provides a process for the preparation of 4-fluoro-2- methylbenzonitrile of Formula (II), and its use for the preparation of trelagliptin or its salts. The present invention provides an efficient, simple, and commercially friendly process for the preparation of 4-fluoro-2-methylbenzonitrile, which is used as an intermediate for the preparation of trelagliptin or its salts. The present invention avoids the use of toxic and hazardous reagents, high boiling solvents, and bromo intermediates such as 2-bromo-5-fluorotoluene, which is lachrymatory in nature and thus difficult to handle at a commercial scale.
Trelagliptin is a dipeptidyl peptidase IV (DPP-IV) inhibitor, chemically designated as 2- [[6-[(3i?)-3 -aminopiperidin- 1 -yl] -3 -methyl -2,4-dioxopyrimidin- 1 -yljmethyl] -4-fluorobenzonitrile, represented by Formula I.

Formula I
Trelagliptin is administered as a succinate salt of Formula la, chemically designated as 2-[[6-[(3i?)-3-aminopiperidin-l-yl]-3-methyl-2,4-dioxopyrimidin-l-yl]methyl]-4-fluorobenzonitrile butanedioic acid (1 : 1).

Formula la
U.S. Patent Nos. 7,795,428, 8,288,539, and 8,222,411 provide a process for the preparation of 4-fluoro-2-methylbenzonitrile by reacting 2-bromo-5-fluorotoluene with copper (I) cyanide in N,N-dimethylformamide.
Chinese Patent No. CN 102964196 provides a process for the preparation of 4-fluoro-2-methylbenzonitrile by reacting 4-fluoro-2-methylbenzyl alcohol with cuprous iodide in the presence of 2,2′-bipyridine and 2,2,6,6-tetramethylpiperidine oxide (TEMPO) in an anhydrous ethanol.
Copper (I) cyanide is toxic to humans, and therefore its use in the manufacture of a drug substance is not advisable. In addition, 2-bromo-5-fluorotoluene is converted to 4-fluoro-2-methylbenzonitrile by refluxing in N,N-dimethylformamide at 152°C to 155°C for 24 hours. This leads to some charring, resulting in a tedious work-up process and low yield. Furthermore, the use of reagents like cuprous iodide, 2,2′-bipyridine, and 2,2,6,6-tetramethylpiperidine oxide (TEMPO) is hazardous and/or environmentally-unfriendly, and therefore their use in the manufacture of a drug substance is not desirable.
The present invention provides an efficient, simple, and commercially friendly process for the preparation of 4-fluoro-2-methylbenzonitrile, which is used as an intermediate for the preparation of trelagliptin or its salts. The present invention avoids the use of toxic and hazardous reagents, high boiling solvents, and bromo intermediates such as 2-bromo-5-fluorotoluene, which is lachrymatory in nature and thus difficult to handle at a commercial scale.
EXAMPLES
Example 1 : Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (1.38 g) was added to ethanol (10 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (2.76 g) and pyridine (1 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 3 hours. The solvent was recovered up to maximum extent from the reaction mixture under reduced pressure to afford the title compound. Yield: 3.1 g
Example 2: Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (5 g) was added to ethanol (37 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (10 g) and N,N-diisopropylethylamine (3.6 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 2 hours. The solvent was recovered up to maximum extent from the reaction mixture under reduced pressure to afford the title compound. Yield: 3.1 g
Example 3 : Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (10 g) was added to ethanol (40 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (20 g) and N,N-diisopropylethylamine (7.5 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 4 hours. The solvent was recovered from the reaction mixture under reduced pressure to afford the title compound. Yield: 11.0 g
Example 4: Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (50 g) was added to ethanol (500 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (70 g) and N,N-diisopropylethylamine (36 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 6 hours. The solvent was recovered from the reaction mixture under reduced pressure to afford the title compound. Yield: 51.0 g
Example 5 : Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methylbenzaldehyde (20 g) was added to ethanol (200 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (20 g) and N,N-diisopropylethylamine (18 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 4 hours. The solvent was recovered from the reaction mixture under reduced pressure to obtain a residue. Deionized water (60 mL) was charged into the residue, and then the slurry was stirred at 0°C to 5°C for 1 hour. The solid obtained was filtered, then washed with deionized water (2 x 20 mL). The wet solid was dried in an air oven at 40°C to 45 °C for 4 hours to 5 hours. The crude product obtained was recrystallized in ethanol (50 mL) to afford the pure title compound. Yield: 21.0 g
Example 6: Preparation of 4-fluoro-2-methylbenzaldoxime
4-Fluoro-2-methyl benzaldehyde (50 g) was added to ethanol (500 mL) to obtain a solution. To this solution, hydroxylamine hydrochloride (50 g) and N,N-diisopropylethylamine (46.4 mL) were added, and then the mixture was stirred at 20°C to 25 °C for 4 hours. The solvent was recovered from the reaction mixture under reduced pressure to obtain a residue. Deionized water (150 mL) was charged to the residue, and then the slurry was stirred at 0°C to 5°C for 1 hour. The solid obtained was filtered, then washed with deionized water (2 x 50 mL). The wet solid was dried in an air oven at 40°C to 45 °C for 4 hours to 5 hours. The crude product obtained was recrystallized in ethanol (200 mL) to afford the pure title compound. Yield: 53.5 g
Example 7: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (3.1 g) and phosphorous pentoxide (1 g) were added to toluene (30 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 24 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (30 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 1.1 g
Example 8: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (3 g) and phosphorous pentoxide (2 g) were added to toluene (30 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 24 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (30 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 1.0 g
Example 9: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (5 g) and concentrated sulphuric acid (2 mL) were added to toluene (100 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 5 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (50 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 3.24 g
Example 10: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methylbenzaldoxime (25 g) and concentrated sulphuric acid (35 g) were added to toluene (500 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 6 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C. Deionized water (250 mL) was added to the mixture and then the layers were separated. The organic layer was concentrated under reduced pressure to afford the title compound. Yield: 20.5 g
Example 11 : Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methyl benzaldoxime (5 g) and sodium bisulphate monohydrate (3.1 g) were added to toluene (50 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C for 12 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25°C to 30°C, then filtered, and then washed with toluene (10 mL). The filtrate was concentrated under reduced pressure to afford the title compound. Yield: 3.0 g
Example 12: Preparation of 4-fluoro-2-methylbenzonitrile
4-Fluoro-2-methyl benzaldoxime (50 g) and sodium bisulphate monohydrate (31.6 g) were added to toluene (500 mL) to obtain a reaction mixture. The reaction mixture was refluxed at 110°C to 115°C using a Dean-Stark apparatus for 12 hours. After completion of the reaction (monitored by TLC), the reaction mixture was cooled to 25 °C to 30°C, then filtered, and then washed with toluene (100 mL). The filtrate was concentrated under reduced pressure to afford a crude product. The crude product obtained was recrystallized in a mixture of toluene (200 mL) and hexane (500 mL) to afford the title compound.
Yield: 38.0 g

Sun Pharma managing director Dilip Shanghvi.
/////////////WO 2016024224, New Patent, Trelagliptin, SUN PHARMA
WO 2016024243, New patent, Dr Reddy’s Laboratories Ltd, Fidaxomicin


WO 2016024243, New patent, Dr Reddy’s Laboratories Ltd, Fidaxomicin
WO2016024243, FIDAXOMICIN POLYMORPHS AND PROCESSES FOR THEIR PREPARATION
DR. REDDY’S LABORATORIES LIMITED [IN/IN]; 8-2-337, Road No. 3, Banjara Hills, Telangana State, India Hyderabad 500034 (IN)
CHENNURU, Ramanaiah; (IN).
PEDDY, Vishweshwar; (IN).
RAMAKRISHNAN, Srividya; (IN)
Aspects of the present application relate to crystalline forms of Fidaxomicin IV, V & VI and processes for their preparation. Further aspects relate to pharmaceutical compositions comprising these polymorphic forms of fidaxomicin

Fidaxomicin (also known as OPT-80 and PAR-101 ) is a novel antibiotic agent and the first representative of a new class of antibacterials called macrocycles. Fidaxomicin is a member of the tiacumicin family, which are complexes of 18-membered macrocyclic antibiotics naturally produced by a strain of Dactylosporangium aurantiacum isolated from a soil sample collected in Connecticut, USA. The major component of the tiacumicin complex is tiacumicin B. Optically pure R-tiacumicin B is the most active component of Fidaxomicin. The chiral center at C(19) of tiacumicinB affects biological activity, and R-tiacumicin B has an R-hydroxyl group attached at this position. The isomer displayed significantly higher activity than other tiacumicin B-related compounds and longer post-antibiotic activity.
As per WIPO publication number 2006085838, Fidaxomicin is an isomeric mixture of the configurationally distinct stereoisomers of tiacumicin B, composed of 70 to 100% of R-tiacumicin B and small quantities of related compounds, such as S-tiacumicin B and lipiarmycin A4. Fidaxomicin was produced by fermentation of the D aurantiacum subspecies hamdenensis (strain 718C-41 ). It has a narrow spectrum antibacterial profile mainly directed against Clostridium difficile and exerts a moderate activity against some other gram-positive species. Fidaxomicin is bactericidal and acts via inhibition of RNA synthesis by bacterial RNA polymerase at a distinct site from that of rifamycins. The drug product is poorly absorbed and exerts its activity in the gastrointestinal (Gl) tract, which is an advantage when used in the applied indication, treatment of C. difficile infection (CDI) (also known as C. difficile-associated disease or diarrhoea [CDAD]). Fidaxomicin is available as DIFICID oral tablet in US market. Its CAS chemical name is Oxacyclooctadeca-3,5,9, 13, 15-pentaen-2-one, 3-[[[6-deoxy-4-0-(3,5dichloro-2-ethyl-4,6-dihydroxybenzoyl)-2-0-methyl-P-D-manno pyranosyl]oxy]methyl]-12[[6-deoxy-5-C-methyl-4-0-(2-methyl-1 -oxopropyl)- -D-lyxo-hexo pyranosyl]oxy]-1 1 -ethyl-8-hydroxy-18-[(1 R)-1 -hydroxyethyl] -9,13,15-trimethyl-, (3E.5E, 8S.9E.1 1 S.12R.13E, 15E.18S)-. Structural formula (I) describes the absolute stereochemistry of fidaxomicin as determined by x-ray.

(I)
WIPO publication number 2004014295 discloses a process for preparation of Tiacumicins that comprises fermentation of Dactylosporangium aurantiacum NRRL18085 in suitable culture medium. It also provides process for isolation of tiacumicin from fermentation broth using techniques selected from the group consisting of: sieving and removing undesired material by eluting with at least one solvent or a solvent mixture; extraction with at least one solvent or a solvent mixture; Crystallization; chromatographic separation; High-Performance Liquid Chromatography (HPLC); MPLC; trituration; and extraction with saturated brine with at least one solvent or a solvent mixture. The product was isolated from /so-propyl alcohol (IPA) having a melting point of 166-169 °C.
U.S. Patent No. 7378508 B2 discloses polymorphic forms A and B of fidaxomicin, solid dosage forms of the two forms and composition thereof. As per the ‘508 patent form A is obtained from methanol water mixture and Form B is obtained from ethyl acetate.
J. Antibiotics, vol. 40(5), 575-588 (1987) discloses purification of Tiacumicins using suitable solvents wherein tiacumicin B exhibited a melting point of 143-145 °C.
PCT application WO2013170142A1 describes three crystalline forms of Fidaxomicn namely, Form-Z, Form-Z1 and Form-C. IN2650/CHE/2013 describes 6 crystalline polymorphic forms of Fidaxomicin namely, Forms I, Form la, Form II, Form Ha, Form III and Form Ilia).
The occurrence of different crystal forms, i.e., polymorphism, is a property of some compounds. A single molecule may give rise to a variety of polymorphs having distinct crystal structures and physico-chemical properties.
Polymorphs are different solid materials having the same molecular structure but different molecular arrangement in the crystal lattice, yet having distinct physico-chemical properties when compared to other polymorphs of the same molecular structure. The discovery of new polymorphs and solvates of a pharmaceutical active compound provides an opportunity to improve the performance of a drug product in terms of its bioavailability or release profile in vivo, or it may have improved stability or advantageous handling properties. Polymorphism is an unpredictable property of any given compound. This subject has been reviewed in recent articles, including A. Goho, “Tricky Business,” Science News, August 21 , 2004. In general, one cannot predict whether there will be more than one form for a compound, how many forms will eventually be discovered, or how to prepare any previously unidentified form.
There remains a need for additional polymorphic forms of fidaxomicin and for processes to prepare polymorphic forms in an environmentally-friendly, cost-effective, and industrially applicable manner.

G.V. Prasad, chairman, Dr Reddy’s Laboratories
EXAMPLES
Example 1 : Preparation of fidaxomicin Form IV:
Fidaxomicin (0.5 g) and a mixture of 1 ,4-Dioxane (10 mL), THF (10 ml) and water (20mL) were charged in Easy max reactor (Mettler Toledo). The reactor was set to temperature cycle with following parameters:
Starting temperature: 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 6 hours.
After completion of temperature cycling process, the slurry was filtered under suction, followed by drying in air tray dryer (ATD) at 40°C to a constant weight to produce crystalline fidaxomicin form-IV.
Example 2: Preparation of fidaxomicin Form V:
Fidaxomicin (1 g) and a mixture of propylene glycol (10 mL) and water (20mL) were charged in Easy max reactor (Mettler Toledo). The reactor was set to temperature cycle with following parameters:
Starting temperature is 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 6 hours.
After completion of temperature cycling process, the slurry was filtered under suction, followed by drying in air tray dryer (ATD) at 40°C to a constant weight to produce crystalline fidaxomicin form-V.
Example 3: Preparation of fidaxomicin Form VI:
Fidaxomicin (0.5 mg) and MIBK (10 mL) were charged in Easy max reactor (Mettler Toledo) and the mixture was heated to 80°C. n-heptane (20 mL) was added to the solution at the same temperature. The mixture was stirred for 1 hour. The reaction mass was then cooled to 25°C. Solid formed was filtered at 25°C and dried at 40°C in air tray dryer (ATD) to a constant weight to produce crystalline fidaxomicin form VI.
Example 4: Preparation of fidaxomicin Form V:
Fidaxomicin (500 mg) and a mixture of R-propylene glycol (5 mL) and water (15 mL) were charged in Easy max reactor (Mettler Toledo). The reactor was set to temperature cycle with following parameters:
Starting temperature is 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 2 hours.
After completion of temperature cycling process, the slurry was filtered and dried at 25°C to produce crystalline fidaxomicin form-V.
Example 5: Preparation of fidaxomicin Form V:
Fidaxomicin (1 g) and a mixture of S-propylene glycol (3 ml_) and water (30 mL) were charged in Easy max reactor (Mettler Toledo). The reactor was set to temperature cycle with following parameters:
Starting temperature is 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 2 hours.
After completion of temperature cycling process, the slurry was filtered and dried at 25°C to produce crystalline fidaxomicin form-V.
Example 6: Preparation of fidaxomicin Form V:
Fidaxomicin (40 g) and a mixture of propylene glycol (400 mL) and water (1600 mL) were charged in Chem glass reactor. The reactor was set to temperature cycle with following parameters:
Starting temperature is 25 °C;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 60 °C over a period of 2 hours;
Cooled to 0 °C over a period of 2 hours;
Temperature raised to 25 °C over a period of 2 hours;
Temperature maintained at 25 °C for 6 hours.
After completion of temperature cycling process, the slurry was filtered under suction, followed by drying in air tray dryer (ATD) at 40°C to a constant weight to produce crystalline fidaxomicin form-V.

The 10-member board at pharmaceutical major Dr Reddy’s thrives on diversity. Liberally sprinkled with gray hairs, who are never quite impressed with powerpoint presentations, “they want information to be pre-loaded so that the following discussions (at the board level) are fruitful,” says Satish Reddy, Chairman, Dr Reddy’s. That said, the company has now equipped its board members with a customized application (that runs on their tablets) to manage board agenda and related processes.
see at

Dr. Reddy’s Laboratories Managing Director and Chief Operating Officer Satish Reddy addressing
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
3-(((6-Deoxy-4-O-(3,5-dichloro-2-ethyl-4,6-dihydroxybenzoyl)-2-O-methyl-β-D-mannopyranosyl)oxy)-methyl)-12(R)-[(6-deoxy-5-C-methyl-4-O-(2-methyl-1-oxopropyl)-β-D-lyxo-hexopyranosyl)oxy]-11(S)-ethyl-8(S)-hydroxy-18(S)-(1(R)-hydroxyethyl)-9,13,15-trimethyloxacyclooctadeca-3,5,9,13,15-pentaene-2-one
|
|
| Clinical data | |
| Trade names | Dificid, Dificlir |
| Licence data | US FDA:link |
| Pregnancy category |
|
| Legal status | |
| Routes of administration |
Oral |
| Pharmacokinetic data | |
| Bioavailability | Minimal systemic absorption[1] |
| Biological half-life | 11.7 ± 4.80 hours[1] |
| Excretion | Urine (<1%), faeces (92%)[1] |
| Identifiers | |
| CAS Number | 873857-62-6 |
| ATC code | A07AA12 |
| PubChem | CID 11528171 |
| ChemSpider | 8209640 |
| UNII | Z5N076G8YQ |
| KEGG | D09394 |
| ChEBI | CHEBI:68590 |
| ChEMBL | CHEMBL1255800 |
| Synonyms | Clostomicin B1, lipiarmicin, lipiarmycin, lipiarmycin A3, OPT 80, PAR 01, PAR 101, tiacumicin B |
| Chemical data | |
| Formula | C52H74Cl2O18 |
| Molar mass | 1058.04 g/mol |
///////////WO-2016024243,WO 2016024243, New patent, Dr Reddy’s Laboratories Ltd, Fidaxomicin
CC[C@H]1/C=C(/[C@H](C/C=C/C=C(/C(=O)O[C@@H](C/C=C(/C=C(/[C@@H]1O[C@H]2[C@H]([C@H]([C@@H](C(O2)(C)C)OC(=O)C(C)C)O)O)\C)\C)[C@@H](C)O)\CO[C@H]3[C@H]([C@H]([C@@H]([C@H](O3)C)OC(=O)C4=C(C(=C(C(=C4O)Cl)O)Cl)CC)O)OC)O)\C
WO 2016024284, New Patent, MIRABEGRON, Wanbury Ltd
WO 2016024284, New Patent, MIRABEGRON, Wanbury Ltd

WANBURY LTD. [IN/IN]; BSEL tech park, B wing, 10th floor, sector 30A opp. Vashi Railway Station, Vashi Navi Mumbai 400703 Maharashtra (IN)
DR. NITIN SHARADCHANDRA PRADHAN; (IN).
DR. NILESH SUDHIR PATIL; (IN).
DR. RAJESH RAMCHANDRA WALAVALKAR; (IN).
MR. NILESH SUBHASH KULKARNI; (IN).
MR. SANTOSH NAMDEV RAWOOL; (IN).
MR. PURUSHOTTAM EKANATH AWATE; (IN)

LEFT , DR K CHANDRAN, DIRECTOR WANBURY
MR ASOK SHINKAR
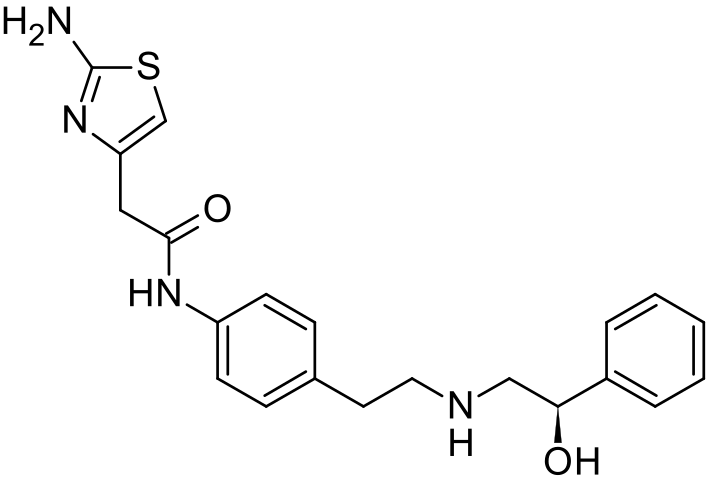
The present invention relates to a novel process for preparation of Mirabegron of Formula (I) using intermediates of Formula (II), (IIIa), (Illb) and (IV).
The present invention relates to a process for preparation of Mirabegron of Formula
(I).

Formula (I)
The present invention further relates to the preparation of Mirabegron of Formula (I) by using compounds of Formula (II), (Ilia), (Illb) and (IV)

Formula (II)

Formula (IlIa) Formula (Illb)

Formula (IV)
Furthermore, the present invention relates to process for preparation of compound of Formula (II), (Ilia), (Illb) and (IV).
Background of the invention:
Mirabegron is chemically known as 2-amino-N-[4-[2-[[(2R)-2-hydroxy-2-phenylethyl]amino]ethyl]phenyl]-4-thiazoleactamide and is marketed under trade name Myrbetiq.
Mirabegron is a drug used for treatment of overactive bladder. It was first disclosed in US 6,346,532, wherein (R)-Styrene oxide is reacted with 4-nitrophenyl ethyl amine hydrochloride to obtain (R)-l- phenyl-2-[[2-(4-nitrophenyl)ethyl]amino]ethanol, the later is then protected with BOC anhydride and subjected to reduction in the presence of Pd/C to yield N-[2-(4-Aminophenyl)ethyl]-N-[(2R)-2-hydroxy-2-phenylethyljcarbamic acid tert-butyl ester. Thus formed compound was then coupled with (2-amino-l,3-thiazol-4yl) acetic acid to obtain BOC protected Mirabegron which is de-protected to give Mirabegron hydrochloride.
The synthetic route proposed in US 6,346,532 is presented in Scheme-I.
Scheme-I

The major draw-backs of the presented synthetic scheme are as follows:
1. Less atomic efficiency
2. Low yield and extensive impurities formations
3. Use of expensive and sensitive protecting agents
4. Column chromatographic techniques for purifications of intermediates.
One more synthetic route for the preparation of Mirabegron have been proposed US 6,346,532, however it is not exemplified.
US 7,342,117 disclose a process for preparation of Mirabegron. The process involves the step of condensation of 4-nitrophenyl ethylamine and (R)- mandelic acid in presence of tri ethylamine, hydroxybentriazole and l-(3-dimethylaminopropyl)-3-ethyl carbodiimide in N,N-dimethylformamide to obtain compound of Formula (A). The second step involves conversion of compound of Formula (A) to compound of Formula (B) in presence of l,3-dimethyl-2-imidazolidone and borontetrahydro fluoride in tetrahydrofuran. In third step, compound of Formula (B) is subjected to reduction using 10% palladium-carbon in methanol to afford (R)-2-[[2′-(4-aminophenyl)-ethyl amino] -1-phenylethanol (Formula IV), which was further condensed with 2-aminothiazol-4-yl acetic acid in presence of l-(3-dimethylaminopropyl)-3 -ethyl carbodiimide and hydrochloric acid in water to obtain Mirabegron of Formula (I). The schematic representation is as Scheme-II

Another patent application CN103193730, discloses a novel process for preparation of Mirabegron wherein the amino group of 2-aminothiazole-5-acetic acid is protected with a protecting group and is condensed with 4-amino phenyl ethanol to obtain an intermediate (A); which on further oxidation yields intermediate (B). The intermediate B is subjected to reductive amination with (R)-2-amino-l -phenyl ethanol and deprotection, simultaneously to yield Mirabegron. The schematic representation is as Scheme-Ill.

Formula (I)
Scheme-Ill
Other references wherein process for preparation of Mirabegron are disclosed CN103387500 and CN103232352.
Most of the prior art reported for preparation of Mirabegron uses expensive and sensitive protecting agents thereby making process less feasible on industrial scale. Furthermore, the yield and purity of Mirabegron obtained by the processes known in art is not satisfactory. It is well known fact that pharmaceutical products like Mirabegron should have high purity due to the therapeutic advantages and also due to the stringent requirements of regulatory agencies. The purity requirements can be fulfilled either by avoiding the formation of by-products during the process or by purifying the end product of the process. The inventors of present invention have skillfully developed the process to provide Mirabegron with unachieved level of purity. Furthermore, the process of present invention is simple, industrially viable, and economic and avoids unfavorable reaction conditions.

According to present invention, the process for preparation of compound of Formula (IV), is depicted in Scheme IV

The present invention further relates to a process for preparation of Mirabegron of Formula (I)
The schematic reaction scheme of Mirabegron according to present invention is depicted in Scheme-V.
 Wherein R is -OH or -CI
Wherein R is -OH or -CI
The detail of the invention provided in the following examples is given by the way of illustration only and should not be construed to limit the scope of the present invention.
EXAMPLES
Example 1: Preparation of [2-(formylamino)-l,3-thiazol-4-yl]acetyl chloride; Formula (V); wherein R is -CI
20g of [2-(formylamino)-l,3-thiazol-4-yl]acetic acid was added to 250 ml of methylene dichloride and the mixture was cooled to -10°C followed by lot wise addition of 25g of phosphorous pentachloride. The mixture stirred while maintaining temperature of -10°C for 2-3 hours. After confirming completion of reaction, the product was filtered out, washed with methylene dichloride and dried to obtain 24g (Yield: 92%) of compound of Formula (V); wherein R is -CI
Example 2: Preparation of 4-nitrophenyl-[2-(formylamino)-l,3-thiazol-4-yl]acetate; Formula (IlIa)
2g of p-nitrophenol was added to 40ml of methylene chloride and 4.963g of potassium carbonate, the mixture was cooled to 10-15°C followed by lot wise addition of 3.95g of compound of Formula (V) of example 1. After confirming completion of reaction, 5.87g (Yield: 99%) of compound of Formula (Ilia) was isolated. The obtained compound has been identified by;
HNMR(D20 Exchange)
8.614 (S,lH),7.359(d,2H),8.119(d,2H),6.561(S,lH),3.765(S,2H).
Example 3: Preparation of (2-amino-l,3-thiazol-4-yl)acetyl chloride; Formula (VI); wherein R is -CI
5g of (2-amino-l,3-thiazol-4-yl)acetic acid was added to 50 ml of methylene dichloride with few drops of dimethylformamide and 6g of oxalyl chloride at temperature ranging from 0-5°C. the mixture was maintained at 0-5°C for 4-5 hours and after completion of reaction, solid mass was filtered out, washed with methylene dichloride and dried to afford 5g (Yield: 89%) of compound of Formula (VI); wherein R is -CI
Example 4: Preparation of 4-nitrophenyl-(2-amino-l,3-thiazol-4-yl)acetate; Formula (Illb)
2g of p-nitrophenol was added to 40ml of methylene chloride and 4.96g of potassium carbonate, and the mixture was cooled to 10-15 °C followed by lot wise addition of 3.95g of compound of Formula (VI) prepared in example 3. After confirming completion of reaction, 6.18g (Yield: 99%) of 4-nitrophenyl-(2-amino-l,3-thiazol-4-yl)acetate of Formula (Illb) was isolated.
The obtained compound has been identified by
HNMR ( D2O Exchange)
7.359(d,2H),8.1 19(d,2H),6.425(S,lH).3.775(S,2H).
Example 5: In-situ preparation of (lR)-2-[[2-(4-aminophenyl)ethyl]amino]-l-phenylethanol or its hydrochloride salt, of Formula (IV)
Step I – Preparation of (2R)-2-hydroxy-N-[2-(4-nitrophenyl)ethyl]-2-phenylethanamide of Formula (IX)
(R)-2-hydroxy-2-phenylacetic acid (75g), triethylamine (50g), hydroxybenzotriazole (HOBt) (33.3g) and l-(3-dimethylaminopropyl)-3-ethyl carbodiimide hydrochloride (EDC.HC1) (50g) were added to a mixture of 2-(4-nitrophenyl)ethylamine hydrochloride (100g) in Ν,Ν-dimethylformamide (375ml) at 25-30°C. The mixture was stirred for 30 minutes followed by addition of another lot of HOBt (33.3g) and EDC.HC1 (50g) in reaction mixture. The reaction mixture was maintained at 25-30°C for 15 hours under stirring. After completion of reaction, water (1850ml) was added to the reaction mixture and stirred. Subsequently, ethyl acetate (1500ml) was added to the reaction mixture at 25-30°C and stirred. The organic phase was separated from aqueous phase, and was washed sequentially with 1M HC1 solution, 20%aqueous potassium carbonate solution and water. The organic solvent was distilled out under reduced pressure to obtain residue comprising of (2R)-2-hydroxy-N-[2-(4-nitrophenyl)ethyl] -2 -phenyl ethanamide of Formula (IX)
Step II – Preparation of (2R)-2-hydroxy-N-[2-(4-aminophenyl)ethyl]-2-phenylethanamide of Formula (X)
The residue from step I, methanol (740ml) and Raney Nickel (14.8g) were charged into an autoclave vessel, 10 kg/cm2 hydrogen gas pressure was applied to the reaction mixture at 25-30°C and the mixture was maintained under stiring 6 hours. Reaction mixture filtered through hyflo bed. Distilled off the solvent completely from the filtrate under reduced pressure to obtain residue comprising (2R)-2-hydroxy-N-[2-(4-aminophenyl)ethyl]-2-phenylethanamide of Formula (X)
Step III – Preparation of (lR)-2-[[2-(4-aminophenyl)ethyl]amino]-l-phenylethanol dihydrochloride salt, of Formula (IV)
The residue of step II was added in tetrahydrofuran (665ml) and the mixture was cooled to -5 to 0°C. To this cooled mixture was then successively added sodium borohydride (56.26g) and BF3-diethyl ether (466g), and the mixture was stirred for 15 minutes. The temperature of reaction mixture was gradually increased to 50-55°C and was maintained under stirring for 5 hours. After completion of reaction, the reaction mixture was cooled to 0-5°C and 50% sodium hydroxide solution was added till pH is basic. The temperature of reaction mixture is then raised to 25-30°C followed by addition of ethyl acetate (500ml). The organic layer was separated and subjected to distillation to afford a residue. To the residue was added isopropyl alcohol (665ml) and mixture was refluxed for 30 minutes. The mixture was then allowed to cool to 40-45°C, isopropyl alcohol hydrochloride (200ml) was added till pH acidic and mixture was stirred for 2 hours to afford precipitate. The precipitate was filtered out and washed with isopropyl alcohol. The wet cake thus obtained was added to 20% aqueous sodium hydroxide solution (till pH basic) followed by addition of dichloromethane (500ml). The organic layer was separated from aqueous layer and was subjected to distillation under reduced pressure to obtain residue. The residue was taken in toluene (500ml), heated to 55-60°C for 30 minutes and cooled to 10-15°C. The precipitate obtained was filtered, washed with toluene and to the wet cake afforded was added isopropyl alcohol (665ml). The mixture was refluxed for 30 minutes and then cooled to 50-55°C. At 50-55°C slowly isopropyl alcohol hydrochloride (200ml) till pH acidic was added and mixture was stirred for 2 hours to obtain precipitate. The precipitate was filtered out, washed with isopropyl alcohol and dried to get (lR)-2-[[2-(4-aminophenyl)ethyl]amino]-l-phenylethanol dihydrochloride salt, of Formula (IV)
Yield-70%
HPLC Purity: 98%
Example 6: Alternate method for preparation of (2R)-2-hydroxy-N-[2-(4-nitrophenyl)ethyl]-2-phenylethanamide of Formula (IX)
Step I – A mixture of (R)-2-hydroxy-2-phenylacetic acid (lOg), dichloromethane (50ml) and triethylamine (24ml) was cooled to 0-5°C and slowly para-toluene sulfonyl chloride (12.53g) was added to it. The temperature of reaction mixture was raised to 25-30°C and maintained for 12 hours. After completion of reaction, water (100ml) was added to the reaction mixture and the mixture was stirred for 15 minutes. The organic phase was separated and distills out completely under reduced pressure to obtain [(R)-2-hydroxy -2-phenyl acetic tosyl ester].
Yield-56%
Step II – 2-(4-nitrophenyl)ethylamine hydrochloride (6g) was added to dichloromethane (50ml) and stirred for 30 minutes at 25-30°C. The mixture was
then cooled to 0-5 °C and triethylamine (13ml) was added. To say cooled mixture was then slowly added a mixture of (R)-2-hydroxy -2-phenyl acetic tosyl ester (lOg) and dichloromethane (50ml). The temperature of reaction mixture was then raised to reflux temperature and maintained for 5 hours. After completion of reaction, water (50ml) was added to the reaction mixture and the mixture was stirred for 15 minutes. The organic phase was separated and distill out completely under reduced pressure to obtain (R)-2-hydroxy-N-[2-(4-nitrophenyl) ethyl]-2-phenylacetamide
Yield-70%, Purity-96%
Example 7: Preparation of compound of Formula (II) from compound of Formula (V); wherein R is -OH
1.58g of [2-(formylamino)-l,3-thiazol-4-yl]acetic acid of Formula (V) was added solution of (1R )-2-{[2-(4-aminophenyl)ethyl]amino}-l-phenylethanol of Formula (IV) in water (2g of Formula (IV) in 50ml water) followed by addition of 0.66g concentrated hydrochloric acid and 3.27g of l-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride. The mixture was stirred at 25-30°C for 0.5 hours. After completion of reaction, pH was adjusted to 8-9 using aqueous saturated solution of sodium carbonate. The solid precipitated out was filtered, washed with water and dried to obtain 2.1g of compound of Formula (II). (Yield: 72%) The obtained compound has been identified by HNMR
2.502(m,4H),2.599(m,2H),3.685(S,2H),4.9(S, NH protons),7.01(m, 10H, aromatic), 8.54(S,1H), 10.0(S, -OH proton),
HNMR(D20 Exchange) 2.502(m,4H),2.60(m,2H),4.57(m,lH),7.0(m, 10H, aromatic), 8.43(S,1H)
Example 8: Preparation of compound of Formula (II) from compound of Formula (V); wherein R is -CI
lOg of ( 1R)-2-{[2-(4-aminophenyl)ethyl]amino}-l-phenylethanol of Formula (IV) (prepared by methods known in prior art/ as given in example 5), was added to 150ml of acetonitrile with 16.17g of potassium carbonate and the mixture was cooled to 10-15°C. 18.8g of Formula (V) of example 1 was added to above mixture at 10-15°C in lot wise. After completion of reaction, the reaction mixture was concentrated under vacuum and 90ml of water was added for isolation. The product was then filtered out, washed with water and dried to obtain 72g (Yield: 70%) of compound of Formula (II).
Example 9: Preparation of compound of Formula (II) from compound of Formula (IlIa)
5.87g of compound of Formula (IlIa) was added to 40 ml of methylene dichloride with 2.36 g of potassium carbonate and 3.67g of ( 1))-2-{[2-(4-aminophenyl)ethyl]amino}-l-phenylethanol (Formula-IV ; prepared by methods known in prior art/ as given in example 5) . The mixture was stirred at 25-30°C for 1 hour. After completion of reaction, the reaction mixture was concentrated followed by addition of 60 ml of water to isolate lg of compound of Formula (II).
Example 10: Insitu preparation of compound of Formula (II) without isolation of compound of Formula (IlIa)
2g of p-nitrophenol was added to 40 ml of methylene chloride with 4.963g of potassium carbonate, and the mixture was cooled to 10-15°C followed by lot wise addition of 3.95g of [2-(formylamino)-l,3-thiazol-4-yl]acetyl chloride of Formula (V) of example 1. After confirming complete formation of compound of Formula (Ilia), 2.36g of potassium carbonate and 3.67g of (1R)-2-{[2-(4-aminophenyl)ethyl]amino}-1 -phenyl ethanol of Formula (IV) (prepared by methods known in prior art/ as given in example 5) was added insitu, and the mixture was stirred at 25-30°C for 1 hour. After completion of reaction, the reaction mixture was concentrated followed by addition of 60 ml of water to isolate lg of compound of Formula (II).
Example 11: Preparation of Mirabegron from compound of Formula (II)
To 2g of compound of Formula (II) was added 30ml of 10% sodium hydroxide and the mixture was stirred at 55-60°C for 3 hours. After completion of reaction, the mixture was cooled to 25-30°C and the solid obtained was filtered, washed with water and dried to yield 1.3g of Mirabegron. (Yield: 70%)
Example 12: Preparation of Mirabegron from compound of Formula (Illb)
6.18g of 4-nitrophenyl-(2-amino-l,3-thiazol-4-yl)acetate was added to 40ml of methylene dichloride with 2.36g of potassium carbonate and 3.65g of (1R)-2-{ [2-(4-aminophenyl)ethyl]amino}-l-phenylethanol of Formula (IV) (prepared by methods known in prior art/ as given in example 5), and the mixture was stirred at 25-30°C for 1 hour. After completion of reaction, solid was filtered out, washed with methylene dichlrode and dried to yield lg of Mirabegron of Formula (I).
Example 13: Insitu preparation of Mirabegron without isolation of compound of Formula (Illb)
To 40ml of methylene chloride was added 2g of p-nitrophenol and 4.96g of potassium carbonate, and the mixture was cooled to 10-15°C followed by lot wise addition of 3.95g of compound of Formula (VI) prepared in example 3. After confirming complete formation of compound of Formula (Illb), 2.36g of potassium carbonate and 3.65g of (1R)-2-{[2-(4-aminophenyl)ethyl]amino}-l-phenylethanol of Formula (IV) (prepared by methods known in prior art/ as given in example 5) was added insitu, and the mixture was stirred at 25-30°C for 1 hour. After completion of reaction, After completion of reaction, solid was filtered out, washed with methylene dichlrode and dried to yield lg of Mirabegron of Formula (I).
Example 14: Preparation of Mirabegron from compound of Formula (VI); wherein R is -CI
To 20ml of acetone was added 2g of (l/?)-2-{[2-(4-aminophenyl)ethyl]amino}-l-phenylethanol of Formula (IV) and 2.15g of potassium carbonate, and the mixture was cooled to 10-15°C followed by addition of (2-amino-l,3-thiazol-4-yl)acetyl chloride of Formula (VI). After completion of reaction, acetone was concentrated under vacuum and 90ml of water was added for for isolation. The product was then filtered out, washed with water and dried to obtain 2g (Yield: 70%) of Mirabegron.



/////WO-2016024284, WO 2016024284, New Patent, MIRABEGRON, Wanbury Ltd
WO 2016024289, NILOTINIB, New Patent by SUN PHARMA
NILOTINIB

WO 2016024289, NILOTINIB, New Patent by SUN
SUN PHARMACEUTICAL INDUSTRIES LTD [IN/IN]; 17/B, Mahal Industrial Estate, Off Mahakali Caves Road, Andheri (east), Mumbai 400093 (IN)
THENNATI, Rajamannar; (IN).
KILARU, Srinivasu; (IN).
VALANCE SURENDRAKUMAR, Macwan; (IN).
SHRIPRAKASH DHAR, Dwivedi; (IN)
The present invention provides novel salts of nilotinib and polymorphs thereof. The acid addition salts of nilotinib with benzenesulfonic acid, butanedisulfonic acid, 1-5- naphthalenedisulfonic acid, naphthalene-1-sulfonic acid and 1-hydroxynaphthoic acid; hydrates and anhydrates thereof.
Nilotinib, 4-methyl-N-[3-(4-methyl-lH-imidazol-l-yl)-5-(trifluoromethyl)phenyl]-3-[[4-(3-pyridinyl)-2-pyrimidinyl] amino] -benzamide, having the following formula

is marketed under the name Tasigna® in US and Europe. Tasigna contains nilotinib monohydrate monohydrochloride salt and is available as capsules for the treatment of adult patients with newly diagnosed Philadelphia chromosome positive chronic myeloid leukemia (Ph+ CML) in chronic phase. Tasigna is also indicated for the treatment of chronic phase and accelerated phase Philadelphia chromosome positive chronic myelogenous leukemia (Ph+ CML) in adult patients resistant or intolerant to prior therapy that included imatinib.
Nilotinib is considered a low solubility/low permeability (class IV) compound in the Biopharmaceutics Classification System (BCS). Therefore, dissolution of nilotinib can potentially be rate limiting step for in-vivo absorption. It is soluble in acidic media; being practically insoluble in buffer solutions of pH 4.5 and higher.
WIPO publication 2014059518A1 discloses crystalline forms of nilotinib hydrochloride and methods of the preparation of various crystalline solvates of nilotinib hydrochloride including benzyl alcohol, acetic acid and propylene glycol.
WIPO publication 2011033307A1 discloses nilotinib dihydrochloride and its hydrates and method for their preparation.
WIPO publication 2011163222A1 discloses the preparation of nilotinib salts and crystalline forms thereof. The salts of nilotinib disclosed are hydrochloride, fumarate, 2-chloromandelate, succinate, adipate, L-tartrate, glutarate, p-toluenesulfonate, camphorsulfonate, glutamate, palmitate, quinate, citrate, maleate, acetate, L-malate, L-aspartate, formate, hydrobromide, oxalate and malonate.
WIPO publication number 2011086541A1 discloses a nilotinib monohydrochloride monohydrate salt and methods for preparing.
WIPO publication number 2010054056A2 describes several crystalline forms of nilotinib hydrochloride.
WIPO publication number 2007/015871A1 discloses the preparation of nilotinib salts and crystalline forms thereof. The salts are mixtures of nilotinib and one acid wherein the acids are selected from the group consisting of hydrochloric acid, phosphoric acid, sulfuric acid, sulfonic acid, methane sulfonic acid, ethane sulfonic acid, benzene sulfonic acid, p-toluene sul- fonic acid, citric acid, fumaric acid, gentisic acid, malonic acid, maleic acid, and tartaric acid.
WIPO publication number 2007015870A2 discloses several nilotinib salts including amorphous and crystalline forms of nilotinib free base, nilotinib HC1 and nilotinib sulfate along with their hydrate and solvates.
EXAMPLES:
Example 1: Preparation of nilotinib benzenesulfonate crystalline Form I
Nilotinib base (1 g) was suspended in water (20 ml). A solution of benzenesulfonic acid (0.4 g) in water (3ml) was added and the content was heated at 60 °C for 2-3 h. The mixture was cooled to 25-30 °C, filtered, washed with water (3 x 5 ml) and dried under vacuum for 2 h at 50-55 °C.
1H NMR (500 MHz, DMSO-d6) δ 2.40 (s,3H), 2.42 (s,3H), 7.35-7.37 (m,3H), 7.51-7.66 (m,5H),7.83 (d,lH), 7.96 (s,lH),8.08 (s,lH),8.30 (s,lH) 8.39 (s,lH),8.54 (d,lH), 8.61 (d,lH), 8.64 (s,lH), 8.75 (d,lH), 9.25 (s,lH), 9.34 (d,lH), 9.61 (s,lH), 10.84 (s,lH).
The salt provides an XRPD pattern substantially same as set forth in FIG. 1.
Example 2: Preparation of nilotinib butanedisulfonate (2: 1) crystalline Form II
Nilotinib base (100 g) was dissolved in 20 % water in THF solution (2000 ml) at 60-65 °C and insoluble matter was filtered. The filtrate was concentrated under vacuum below 60 °C. Filtered water (1000 ml) was added to the reaction mixture and it was heated at 50-55 °C, followed by addition of 1,4-butanedisulfonic acid -60% aqueous solution (28.6 ml) at same temperature. The content was stirred at 50-55 °C for 2-3h. Reaction mixture as cooled to 25-30 °C and product was filtered, washed with water (200 ml x 2) and dried in air oven at 50-55 °C (yield: 115 g).

Sun Pharma managing director Dilip Shanghvi.
Purity (by HPLC):99.76%
1H NMR (400 MHz,DMSO-d6) δ 1.63-1.66(m,2H), 2.40(d,3H),2.42(s,3H),2.43-2.47(m,2H), 7.51-7.62(m,3H),7.85(dd,lH),7.96(s,lH),8.08(s,lH),8.34(s,lH),8.38(d,lH),8.52-8.55(m,lH), 8.60-8.62 (m,2H), 8.75(d,lH), 9.25(S,1H),9.34(S,1H),9.59(S,1H),10.86(S,1H)
Water content: 7.95 %.
The salt has a XRPD pattern substantially same as set forth in FIG. 2.
Example 3: Preparation of nilotinib butanedisulfonate (2: 1) crystalline Form II
Nilotinib base (300 g) was suspended in methanol (3000 ml) and aqueous hydrochloric acid was added to get pH less than 2. Reaction contents were heated at reflux and was filtered and washed with methanol (100 ml). 5% (w/w) NaOH (1200 ml) solution was added at 40-45 °C within 15 min, reaction mixture was stirred for 2h. Product was filtered, washed with water
(300 ml x 3) and dried for lh. Wet material was suspended in water (3000 ml), heated at 50- 55 °C followed by addition of 1,4-butanedisulfonic acid -60% aqueous solution. The reaction mixture was stirred at 50-55°C for 2hrs. Product was filtered at room temperature, washed with water (500 ml x 2) and dried in air oven at 50-55 °C (yield: 293 g).
Purity (by HPLC): 99.88 %
1H NMR (400 MHz,DMSO-d6+TFA-dl) δ 1.75-1.78(m,2H), 2.36(d,3H),2.38(s,3H),2.69- 2.72(m,2H),7.45(d,lH),7.68(d,lH),7.83(s,lH),7.88(dd,lH),7.97(s,lH),8.16-8.19(m,lH), 8.35
(s,2H), 8.63(d,lH),8.68(d,lH),9.04(d,lH),9.21(d,lH),9.53(br s,lH),9.69(d,lH)10.80 (s,lH)
Water content: 6.44 %
Example 4: Preparation of nilotinib butanedisulfonate (2: 1) crystalline Form III
Nilotinib butanedisulfonate (210g) was dissolved in acetic acid water mixture (50:50) (2520 ml) at 75-80 °C and was filtered to remove insoluble matter and washed with acetic acid water mixture (50:50) (210 ml). Water (3150ml) was added to the filtrate and stirred first at room temperature and then at 0-5 °C. Product was filtered and washed with water. Material was dried in air oven at 70-75 °C. Dried material was leached with methanol (3438 ml) at reflux temperature, filtered and dried in air oven 70-75°C (yield: 152.6 g)
Purity (by HPLC): 99.89 %
1H NMR (400 MHz,DMSO-d6+TFA-dl) δ 1.73-1.77(m,2H), 2.40(s,6H),2.67-2.70(m,2H), 7.50 (d,lH), 7.70(d,lH), 7.88-7.92(m,2H), 8.07(s,lH),8.23 (dd,lH), 8.34(s,2H), 8.67 (d,lH), 8.72 (d,lH), 9.09(d,lH), 9.23 (s,lH), 9.54(d,lH), 9.74(d,lH), 10.86(s,lH).
Water content: 0.61 %
The salt provides an XRPD pattern substantially same as set forth in FIG. 3.
Example 5: Preparation of crystalline form of nilotinib butanedisulfonate (2: 1)
Crystalline Nilotinib butanedisulfonate (1 g) of Example 2 was suspended in methanol (20 ml) and was stirred at reflux for 60 min. The mixture was cooled to room temperature. Solid was filtered, washed with methanol (2 ml x 3) and dried in air oven at 70-75°C (yield: 0.8 g)
Example 6: Preparation of nilotinib butanedisulfonate (1: 1) crystalline Form IV
Nilotinib base (20 g) was suspended in methanol (800 ml) and 1,4-butanedisulfonic acid -60
% aqueous solution (6 ml) was added at 50-55 °C, and was filtered to remove insoluble matter. Filtrate was stirred at room temperature for 2-3 h. Product formed was filtered, washed with methanol (20 ml x 2) and dried the product in air oven at 70-75 °C (yield: 18.4 g).
Purity (by HPLC):99.86 %
1H NMR (400 MHz,DMSO-d6) δ 1.64-1.68(m,4H), 2.47-2.5 l(m,4H), 2.41(s,3H), 2.42(d,3H), 7.52(d,lH), 7.83-7.89(m,2H), 7.99(s,lH), 8.15(s,lH), 8.36 (d,lH), 8.39(s,lH), 8.65-8.66(m,2H), 8.79(d,lH), 8.89(br s,lH), 9.36(s,lH), 9.41(br s,lH), 9.74(d,lH), 10.91(s,lH).
The salt has XRPD pattern substantially same as set forth in FIG. 4.
Example 7: Preparation of nilotinib 1,5-napthalenedisulfonic acid salt (2: 1) crystalline Form V
Nilotinib base (1 g) was suspended in water (20 ml). A solution of 1,5-napthalenedisulfonic acid (0.4 g; 0.6 eq.) in water (5ml) was added and the content was heated at 50-55 °C for lh. The mixture was cooled to 25-30 °C, filtered and washed with water (10 ml). The product was dried in air oven at 50-55°C (yield: 1.2 g).
1H NMR (400 MHz,DMSO-d6) δ 2.39 (s,3H), 2.42 (s,3H), 7.45-7.61 (m,4H),7.84 (d,lH), 7.97(s,2H),8.08 (m,lH),8.31 (s,lH) 8.38 (s,lH),8.55 (d,lH), 8.63 (s,2H), 8.75 (s,lH), 8.92 (d,lH), 9.26 (s, 1H), 9.34 (s,lH),9.62 (s,lH), 10.85 (s,lH).
The salt has a XRPD pattern substantially same as set forth in FIG. 5.
Example 8: Preparation of nilotinib 1,5-napthalenedisulfonic acid salt (1: 1) crystalline Form VI
Nilotinib base (1 g) was suspended in water (20 ml). A solution of 1,5-napthalenedisulfonic acid (0.8 g; 1.2eq) in water (5 ml) was added and the content was heated at 50-55 °C for 1 h. The mixture was cooled to 25-30 °C, filtered, washed with water (10 ml) and dried in air oven at 50-55 °C (yield: 1.4g).
1H NMR(400 MHz,DMSO-d6) δ 2.40 (s,3H),2.41 (s,3H), 7.43-7.52 (m,3H),7.61 (d,lH), 7.85-7.99(m,5H),8.11 (s,lH),8.34 (s,2H), 8.64-8.67 (m,2H), 8.89-8.92 (m,4H),9.40(d,2H), 9.72 (s,lH), 10.87 (s,lH).
The salt has a XRPD pattern substantially same as set forth in FIG. 6.
Example 9: Preparation of nilotinib napthalene-1- sulfonic acid salt crystalline Form VII Nilotinib base (1 g) was suspended in water (10 ml) and heated to 50-55 °C. A solution of napthelene-1 -sulfonic acid and methanol (10 ml) was added to it and heated at 70-75 °C for 30 min. The mixture was cooled to 25-30 °C and stirred for 10 min. The product was filtered, washed with water (2 x 2 ml) and dried under vacuum for 1-2 h at 50-55 °C.
1H NMR (400 MHz,DMSO-d6) δ 2.41 (s,3H),2.42 (s,3H), 7.46-7.58 (m,5H), 7.70-8.00 (m,7H)8.11(s,lH)8.31(s,lH),8.37(s,lH),8.63-8.66 (m,3H), 8.81-8.89 (m,2H), 9.31 (s,lH), 9.37 (d,lH), 9.71 (d,lH), 10.86 (s,lH)
The salt has a XRPD pattern substantially same as set forth in FIG. 7.
Example 10: Preparation of nilotinib l-hydroxy-2-napthoic acid salt crystalline Form VIII Nilotinib base (1 g) was suspended in water (20 ml) and heated to 50-55 °C. l-Hydroxy-2-napthoic acid was added to it and the content was heated at 50-55 °C for 1 h. Methanol (5 ml) was added to the mixture and stirred for 30 min. The content was filtered, washed with water (2 x 2 ml) and dried under vacuum for 1 h at 50-55 °C.
1H NMR (400 MHz, DMSO-d6) δ 2.25 (s,3H), 2.41 (s,3H), 7.40-7.92 (m,l lH), 8.23-8.73 (m,8H), 9.24 (s,lH), 9.34(s,lH), 10.70 (s,lH).
The salt has a XRPD pattern substantially same as set forth in FIG. 8.
 |
|
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
4-methyl-N-[3-(4-methyl-1H-imidazol-1-yl)- 5-(trifluoromethyl)phenyl]-3- [(4-pyridin-3-ylpyrimidin-2-yl) amino]benzamide
|
|
| Clinical data | |
| Trade names | Tasigna |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a608002 |
| Licence data | EMA:Link, US FDA:link |
| Pregnancy category |
|
| Legal status | |
| Routes of administration |
Oral |
| Pharmacokinetic data | |
| Bioavailability | 30%[1] |
| Protein binding | 98%[1] |
| Metabolism | Hepatic (mostly CYP3A4-mediated)[1] |
| Biological half-life | 15-17 hours[1] |
| Excretion | Faeces (93%)[1] |
| Identifiers | |
| CAS Number | 641571-10-0(base) |
| ATC code | L01XE08 |
| PubChem | CID 644241 |
| IUPHAR/BPS | 5697 |
| DrugBank | DB04868 |
| ChemSpider | 559260 |
| UNII | F41401512X |
| KEGG | D08953 |
| ChEBI | CHEBI:52172 |
| ChEMBL | CHEMBL255863 |
| PDB ligand ID | NIL (PDBe, RCSB PDB) |
| Chemical data | |
| Formula | C28H22F3N7O |
| Molar mass | 529.5245 g/mol |
//////////////WO 2016024289, WO-2016024289, NILOTINIB, New Patent, SUN
Cc1ccc(cc1Nc2nccc(n2)c3cccnc3)C(=O)Nc4cc(cc(c4)n5cc(nc5)C)C(F)(F)F
WO 2016020324, BASF AG, Vismodegib , New patent
WO 2016020324, BASF AG, vismodegib , new patent
WO2016020324, MULTI-COMPONENT CRYSTALS OF VISMODEGIB AND SELECTED CO-CRYSTAL FORMERS OR SOLVENTS
BASF SE [DE/DE]; 67056 Ludwigshafen (DE)
VIERTELHAUS, Martin; (DE).
CHIODO, Tiziana; (DE).
SALVADOR, Beate; (DE).
VOSSEN, Marcus; (DE).
HAFNER, Andreas; (CH).
HINTERMANN, Tobias; (CH).
WEISHAAR, Walter; (DE).
HELLMANN, Rolf; (DE)

The present invention primarily relates to multi-component crystals comprising a compound of formula 1 and a second compound selected from the group consisting of co-crystal formers and sol-vents. The invention is further related to pharmaceutical compositions comprising such multi-component crystals. Furthermore, the invention relates to processes for preparing said multi-component crystals. The invention also relates to several aspects of using said multi-component crystals or pharmaceutical compositions to treat a disease.
Developed and launched by Roche and its subsidiary Genentech, under license from Curis. Family members of the product Patent of vismodegib (WO2006028958),
Vismodegib was first disclosed in WO Patent Publication No. 06/028959. Vismodegib, chem-ically 2-Chloro-N-(4-chloro-3-pyridin-2-ylphenyl)-4-methylsulfonylbenzamide, is represented by the following structure:

formula 1
Vismodegib is an active pharmaceutical ingredient produced by Genentech (Roche) and sold under the trade name Erivedge® (which contains crystalline Vismodegib as the active ingre-dient). Erivedge® is an oral Hedgehog signaling pathway inhibitor approved for the treatment of basal-cell carcinoma (BCC).
The present invention primarily relates to multi-component crystals comprising a compound of formula 1 (cf. above) and a second compound selected from the group consisting of co-crystal formers and solvents.
The invention is further related to pharmaceutical compositions comprising said multi-component crystals. Furthermore, the invention also relates to processes for preparing said multi-component crystals. The invention also relates to several aspects of using said multi-component crystals or pharmaceutical compositions to treat a disease. Further details as well as further aspects of the present invention will be described herein below.
Vismodegib is a BCS class II compound with a high permeability but a low solubility where enhanced solubility or dissolution rates can lead to a significant advantage in respect to bio-availability.
Vismodegib is known to exist as crystalline free base. Salts of Vismodegib are men-tioned in US 7,888,364 B2 but not specified. In particular, the HCI salt is mentioned as intermediate but not characterized. Co-crystals or solvates are not reported at all.
The solubility of Vismodegib is reported to be 0.1 μg/mL at pH 7 and 0.99 mg/mL at pH 1 for Erivedge®. The absolute bio-availability after single dose is reported to be 31.8 % and the ex-posure is not linear at single doses higher than 270 mg. Erivedge® capsules do not have a food label. The estimated elimination half-life (t1/2) after continuous once-daily dosing is 4 days and 12 days after a single dose treatment (Highlights of Prescribing Information: ERIVEDGE® (vismodegib) capsule for oral use; Revised: 01/2012).
The discovery and preparation of new co-crystals or solvates offer an opportunity to improve the performance profile of a pharmaceutical product. It widens the reservoir of techniques/materials that a formulation scientist can use for designing a new dosage form of an active pharmaceutical ingredient (API) with improved characteristics. One of the most important characteristics of an API such as Vismodegib is the bio-availability which is often determined by the aqueous solubility.
A compound like Vismodegib may give rise to a variety of crystalline forms having dis-tinct crystal structures and physical characteristics like melting point, X-ray diffraction pattern, infrared spectrum, Raman spectrum and solid state NMR spectrum. One crystalline form may give rise to thermal behavior different from that of another crystalline form. Thermal behavior can be measured in the laboratory by such techniques as capillary melting point, thermogravimetry (TG), and differential scanning calorimetry (DSC) as well as content of sol-vent in the crystalline form, which have been used to distinguish polymorphic forms.
Multi-component crystals comprising Vismodegib and selected co-crystal formers or solvents may improve the dissolution kinetic profile and allow to control the hygrosco-picity of Vismodegib.
Therefore, there is a need for multi-component crystals comprising Vismodegib that avoid the above disadvantages. In particular, it is an object of the present invention to provide multi-component crystals of Vismodegib with optimized manufacture, formula-tion, stability and/or biological efficacy
.
Example 1 :
314 mg Vismodegib and 86 mg maleic acid are suspended in toluene saturated with maleic acid for 2 d, filtered and dried.
TG data shows a mass loss of about 2.3 wt % between 100 and 1 18 °C which is attributed to rest solvent. DSC data shows a single endothermal peak with an onset of about 1 15 °C (99 J/g).
H-NMR spectroscopy indicates a molar ratio of Vismodegib to maleic acid of about 1 :1 .3. However single crystal X-ray data confirms a ratio of 1 :2 (Table 1 ).

Cipla, New Patent, WO 2016020664, Everolimus
Cipla, New Patent, WO 2016020664, Everolimus



CIPLA LIMITED [IN/IN]; Peninsula Business Park Ganpatrao Kadam Marg Lower Parel Mumbai 400 013 (IN).
KING, Lawrence [GB/GB]; (GB) (MW only)
RAO, Dharmaraj Ramachandra; (IN).
MALHOTRA, Geena; (IN).
PULLELA, Venkata Srinivas; (IN).
ACHARYA, Vinod Parameshwaran; (IN)
WO2016020664, PROCESS FOR THE SYNTHESIS OF EVEROLIMUS AND INTERMEDIATES THEREOF

Everolimus (RAD-001) is the 40-O- 2-hydroxyethyl)-rapamycin of formula (I),

It is a derivative of sirolimus of formula III),

and works similarly to sirolimus as an inhibitor of mammalian target of rapamycin (mTOR). Everolimus is currently used as an immunosuppressant to prevent rejection of organ transplants and treatment of renal cell cancer and other tumours. It is marketed by Novartis under the tradenames Zortress™ (USA) and Certican™ (Europe and other countries) in transplantation medicine, and Afinitor™ in oncology.
Trisubstituted silyloxyethyltrifluoromethane sulfonates (triflates) of the general formula (IV), 
wherein R2, R3 are independently a straight or branched alkyl group, for example C^-Cw alkyl, and/or an aryl group, for example a phenyl group, are important intermediates useful in the synthesis of everolimus.
Everolimus and its process for manufacture using the intermediate 2-(t-butyldimethyl silyl) oxyethyl triflate of formula (IVA),

was first described in US Patent Number 5,665,772. The overall reaction is depicted in Scheme I.
Scheme

Everolimus (I)
For the synthesis, firstly sirolimus of formula (III) and 2-(t-butyldimethylsilyl)oxyethyl triflate of formula (IVA) are reacted in the presence of 2,6-Lutidine in toluene at around 60°C to obtain the corresponding 40-O-[2-(t-butyldimethylsilyl)oxy]ethyl rapamycin of formula (I la), which is then deprotected in aqueous hydrochloric acid and converted into crude everolimus [40-O-(2-Hydroxy)ethyl rapamycin] of formula (I).
However, this process results in the formation of impure everolimus, which requires purification by column chromatography. The process results in very poor overall yield and purity and thereby the process is not suitable for the commercial scale production of everolimus.
Moenius et al. (I. Labelled Cpd. Radiopharm. 43, 1 13-120 (2000) have disclosed a process to prepare C-14 labelled everolimus using the diphenyltert-butylsilyloxy-protective group of formula (IV B),

as the alkylation agent. The overall yield reported was 25%.
International patent application, publication number WO 2012/103960 discloses the preparation of everolimus using the alkylating agent 2-((2,3-dimethylbut-2-yl)dimethylsilyloxy)ethyl triflate of formula (IVC),

wherein the overall yield reported is 52.54%. The process involves a derivatization method based on the reaction of the triflate (IV) with a derivatization agent, which preferably is a secondary aromatic amine, typically N-methylaniline.
International patent application, publication number WO 2012/103959 also discloses the preparation of everolimus using the alkylating agent of formula (IVC). The process is based on a reaction of rapamycin with the compound of formula (IVC) in the presence of a base (such as an aliphatic tertiary amine) to form 40-O-2-(t-hexyldimethylsiloxy)ethylrapamycin, which is subsequently deprotected under acidic conditions to obtain everolimus.
European Patent Number 1518517B discloses a process for the preparation of everolimus which employs the triflate compound of formula (IVA), 2-(t-butyldimethyl silyl) oxyethyl triflate. The disclosed process for preparing the compound of formula (IVA) involves a flash chromatography purification step.
The compounds of formula (IV) are key intermediates in the synthesis of everolimus. However, they are highly reactive and also very unstable, and their use often results in decomposition during reaction with sirolimus. This is reflected by the fact that the yields of the reaction with sirolimus are very low and the compounds of formula (IV) are charged in high molar extent. Thus it is desirable to develop a process to stabilize compounds of formula (IV) without loss of reactivity.


Example 1 :
Step 1 : Preparation of protected everolimus (TBS-everoismus) of formula (Ma) using metal salt, wherein “Pg” is t-butyldimethylsilyl
t-butyldimethylsilyloxy ethanol, of formula (VA) (2.8g, 0.016mol) was dissolved in dichloromethane (DCM) (3 vol) and to this 2,6-Lutidine (3.50 g, 0.0327 mol) was added and the mixture was cooled to -40°C. Thereafter, trifluoromethane sulfonic anhydride (3.59ml, 0.021 mol) was added drop-wise. The mixture was maintained at -40°C for 30 minutes. Sirolimus (0.5g, 0.00054mol) was taken in another flask and dissolved in DCM (1 ml). To this sirolimus solution, silver acetate (0.018g, 0.000109mol) was added and cooled to -40°C. The earlier cooled triflate solution was transferred in 3 lots to the sirolimus solution maintaining temperature at -40°C. The reaction mixture was stirred at -40°C further for 15min before which it was slowly warmed to 0°C and further to RT. The reaction mixture was then warmed to 40°C and maintained at this temperature for 3 hours. The reaction was monitored by TLC. On completion of reaction, the reaction mixture was diluted with DCM and washed with water and brine. The organic layer was dried over anhydrous sodium sulphate and solvent was removed by vacuum distillation to obtain the title compound, which was directly used in the next step. HPLC product purity: 60%-85%.
Step 2: Preparation of everolimus of formula (I)
Protected everolimus of formula (I la) obtained in step 1 was dissolved in methanol (10 volumes) and chilled to 0-5° C. To this solution was added drop wise, a solution of 1 N HCI. The pH of the reaction was maintained between 1-3. The temperature of the reaction mixture was raised to 25° C and stirred for 1 hour. After completion of reaction, the reaction mixture was diluted with water (15 volumes) and extracted in ethyl acetate (2X20 volumes). The organic layers were combined and washed with brine, dried over sodium sulphate. The organic layer was distilled off under reduced pressure at 30-35° C, to obtain a crude everolimus (0.8 g). The crude everolimus was further purified by preparative HPLC to yield everolimus of purity >99%.
Example 2:
Step 1 : Preparation of TBS-everoiimus of formula (Ma) without using metal salt, wherein “Pg” is t-butyldimethylsilyl
t-butyldimethylsilyloxy ethanol, of formula (VA) (2.8g, 0.016mol) was dissolved in DCM (3 vol) and to this 2,6-Lutidine (3.50 g, 0.0327 mol) was added and the mixture was cooled to -40°C. Thereafter, trifluoromethane sulfonic anhydride (3.59ml, 0.021 mol) was added drop-wise. The mixture was maintained at -40°C for 30 minutes. Sirolimus (0.5g, 0.00054mol) was taken in another flask and dissolved in DCM (1 ml). The solution was cooled to -40°C. The earlier cooled triflate solution was transferred in 3 lots to the sirolimus solution maintaining temperature at -40°C. The reaction mixture was stirred at -40°C further for 15min before which it was slowly warmed to 0°C and further to RT. The reaction mixture was then warmed to 40°C and maintained at this temperature for 3 hours. On completion of reaction, the reaction mixture was diluted with DCM and washed with water and brine. The organic layer was dried over anhydrous sodium sulphate and
solvent was removed by vacuum distillation to obtain the title compound, which was directly used in next step. HPLC purity: 10%-20%.
Step 2: Preparation of everolimus of formula (I)
Protected everolimus of formula (I la) obtained in step 1 was dissolved in methanol (10 volumes) and chilled to 0-5° C. To this solution was added drop wise, a solution of 1 N HCI. The pH of the reaction was maintained between 1-3. The temperature of the reaction mixture was raised to 25° C and stirred for 1 hour. After completion of reaction, the reaction mixture was diluted with water (15 volumes) and extracted in ethyl acetate (2X20 volumes). The organic layers were combined and washed with brine, dried over sodium sulphate. The organic layer was distilled off under reduced pressure at 30-35° C, to obtain a crude everolimus which was further purified by preparative HPLC.
Example 3:
Preparation of crude Everolimus
Step 1 : Preparation of TBS-ethylene glycol of formula (Va)
Ethylene glycol (1.5L, 26.58 mol) and TBDMS-CI (485g, 3.21 mol) were mixed together with stirring and cooled to 0°C. Triethyl amine (679 ml, 4.83 mol) was then added at 0°C in 30-45 minutes. After addition, the reaction was stirred for 12 hours at 25-30°C for the desired conversion. After completion of reaction, the layers were separated and the organic layer (containing TBS-ethylene glycol) was washed with water (1 L.x2) and brine solution (1 L). The organic layer was then subjected to high vacuum distillation to afford 350g of pure product.
Step 2: Preparation of TBS-glycol-Triflate of formula (IVa)
The reaction was carried out under a nitrogen atmosphere. TBS- ethylene glycol prepared as per step 1 (85.10g, 0.48 mol) and 2, 6-Lutidine (84.28ml, 0.72 mol) were stirred in n-heptane (425ml) to give a clear solution which was then cooled to -15 to – 25°C. Trif!uoromethanesulfonic anhydride (Tf20) (99.74 ml, 0.590 mol) was added drop-wise over a period of 45 minutes to the n-heptane
solution (white precipitate starts to form immediately) while maintaining the reaction at -15 to -25°C. The reaction mixture was kept at temperature between -15 to -25°C for 2 hours. The precipitate generated was filtered off. The filtrate was then evaporated up to ~2 volumes with respect to TBS-ethyiene glycol (~200 ml).
Step 3: Preparation of TBS-evero!imus of formula (Ha)
30g of sirolimus (0,0328 mo!) and toluene (150m!) were stirred together and the temperature was slowly raised to 60-65°C. At this temperature, a first portion of TBS-g!yco!-triflate prepared as per step 2 (100ml) and 2,6-Lutidine (1 1.45ml, 0.086 moles) were added and stirred for 40 min. Further, a second portion of TBS- glycol-triflate (50mi) and 2, 6-Lutidine (19.45ml, 0.138 mol) were added and the reaction was stirred for another 40 min. This was followed by a third portion of TBS- glycol-triflate (50m!) and 2, 6-Lutidine (19.45ml, 0.138 mol), after which the reaction was stirred for further 90 minutes. The reaction was monitored through HPLC to check the conversion of Sirolimus to TBS-everolimus after each addition of TBS-glycol-trifiate. After completion of the reaction, the reaction mixture was diluted with n-heptane (150mi), cooled to room temperature and stirred for another 60 minutes. The precipitated solids were filtered off and the filtrate was washed with deionized water (450 ml x4) followed by brine solution (450ml). The filtrate was subsequently distilled off to afford TBS-everolimus (60-65g) with 60-70% conversion from sirolimus.
Step 4: Preparation of everolimus of formula (I)
TBS-everolimus (65g) obtained in step 3 was dissolved in 300 mi methanol and cooled to 0°C. 1 N HCI was then added to the methanol solution (pH adjusted to 2-3) and stirred for 2 h. After completion of reaction, toluene (360m!) and deionized wafer (360mi) were added to the reaction mixture and the aqueous layer was separated. The organic layer was washed with brine solution (360ml). The organic layer was concentrated to obtain crude everolimus (39g) with an assay content of 30-35%, HPLC purity of 60-65%.
The crude everolimus purified by chromatography to achieve purity more than 99 %.

////Cipla, New Patent, WO 2016020664, Everolimus, INDIA
Zydus Cadila, New Patent,US 20160039759, PERAMPANEL

PERAMPANEL

Zydus Cadila, New Patent,US 20160039759, PERAMPANEL
(US20160039759) PROCESS FOR THE PREPARATION OF PERAMPANEL
CADILA HEALTHCARE LIMITED
Sanjay Jagdish DESAI
Jayprakash Ajitsingh Parihar
Kuldeep Natwarlal Jain
Sachin Ashokrao Patil

Perampanel, a non-competitive AMPA receptor antagonist, is the active ingredient of FYCOMPA® tablets (U.S) which is approved as an adjunctive therapy for the treatment of partial on-set seizures with or without secondarily generalized seizures in patients with aged 12 years and older. Chemically, Perampanel is 5′-(2-cyanophenyl)-1′-phenyl-2,3′-bipyridinyl-6′(1′H)-one, with an empirical formula C23H15N30 and molecular weight 349.384 g/mol which is represented by Formula (I).

U.S. Pat. No. 6,949,571 B2 discloses perampanel and its various processes for preparation thereof.
U.S. Pat. No. 7,759,367 B2 discloses the pharmaceutical composition of perampanel and an immunoregulatory agent and their uses.
U.S. Pat. No. 8,304,548 B2 discloses the reaction of 5′-bromo-1′-phenyl-[2,3′-bipyridin]-6′(1′H)-one with 2-(1,3,2-dioxaborinan-yl)benzonitrile in the presence of palladium compound, a copper compound, a phosphorus compound and a base to form perampanel of Formula (I). Also discloses the crystalline hydrate, anhydrous crystal Form I, anhydrous crystal Form III, & anhydrous crystal Form V of perampanel of Formula (I).
U.S. Pat. No. 7,803,818 B2 discloses an amorphous form of perampanel. U.S. Pat. No. 7,718,807 B2 discloses salts of perampanel. International (PCT) publication No. WO 2013/102897 A1 discloses anhydrous crystalline Form III, V & VII of perampanel.
U.S. PG-Pub. No. 2013/109862 A1 discloses the method for preparing 2-alkoxy-5-(pyridin-2-yl)pyridine, which is an intermediate for preparing perampanel key starting material 5-(2′-pyridyl)-2-pyridone.
U.S. Pat. No. 7,524,967 B2 discloses the preparation of 5-(2′-pyridyl)-2-pyridone, an intermediate in the preparation perampanel.
International (PCT) publication No. WO 2014/023576 A1 discloses the preparation of cyanophenyl boronic acid, an intermediate in the preparation perampanel.
The prior-art processes suffer with problems of poor yield and requirement of chromatographic purification or series of crystallizations which further reduces the overall yield of the final product, which is overcome by the process of the present invention.









 Pankaj Patel, chairman, Zydus Cadila
Pankaj Patel, chairman, Zydus Cadila
EXAMPLES
The present invention is further illustrated by the following examples which is provided merely to be exemplary of the invention and do not limit the scope of the invention. Certain modification and equivalents will be apparent to those skilled in the art and are intended to be included within the scope of the present invention.
Example-A: Preparation of 5-(2-pyridyl)-1,2-dihydropyridin-2-one In a 500 mL round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, a solution of 188.80 g 5-bromo-2-methoxypyridine in 190 mL tetrahydrofuran and 12.92 g pyridine-2-yl boronic acid were added and refluxed. The reaction mixture was cooled to 25-30° C. and aqueous solution of hydrochloric acid was added and stirred for 1 hour. The reaction mixture was neutralized with aqueous sodium hydroxide and extracted with tetrahydrofuran.
The organic layer was washed with saline water, dried over anhydrous magnesium sulfate, and then evaporated to obtain the titled compound.
Example-1
Preparation of 3-bromo-5-(2-pyridyl)-1,2-dihydropyridin-2-one
In a 2 L round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, 201.5 g 5-(2-pyridyl)-1,2-dihydropyridin-2-one, 208.3 g N-bromosuccinimide and 1300 mL N,N-dimethylforamide were stirred at 25-30° C. for 2-3 hours. After completion of the reaction, the reaction mixture was poured into water and stirred for 30 min. The precipitate was filtered, washed with N,N-dimethylforamide and dried at 50° C. to obtain 230 g title compound.
Example-2
Preparation of 3-bromo-5-2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one
In a 500 mL round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, a solution of 18.75 g 3-bromo-5-(2-pyridyl)-1,2-dihydropyridin-2-one in 300 mL methylene dichloride, 18.36 g 1-phenyl boronic acid, 3.47 g palladium triphenylphosphine and 10 mL triethyl amine were added and the reaction mixture was stirred for 1 hour at 25-35° C. The reaction mixture was filtered and the filtrate was evaporated to dryness. The residue was crystallised from ethyl acetate to obtain the title compound.
Example-3
Preparation of Perampanel
In a 1 L round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, a suspension of 188 g 3-bromo-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one, 161.2 g 2-(1,3,2-dioxaborinan-2-yl)benzonitrile, 3.0 g tetrakis(triphenylphosphine)-palladium(0), 10 mL triethylamine (10 mL) in 300 mL methylene dichloride were stirred at 25-30° C. for 12 hours. To the reaction mixture was added 5 mL conc. aqueous ammonia, 10 mL water and 40 mL ethyl acetate. The separated organic layer was washed with water and saturated saline solution and dried over magnesium sulfate. The solvent was removed under vacuum. Ethyl acetate was added to the residue and heated obtain clear solution. n-hexane was added to this solution and cooled to 25-30° C. The obtained solid was filtered and washed with ethyl acetate and dried to obtain perampanel.
Example-4
Preparation of 3-Bromo-5-(2-pyridyl)-1,2-dihydropyridin-2-one
In a 2 L round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, 100 g 5-(2-pyridyl)-1,2-dihydropyridin-2-one, 108.5 g N-bromosuccinimide and 500 mL N,N-dimethylforamide were stirred at 30-35° C. for 3 hours. 100 mL water was added to the reaction mixture at 5-15° C. and stirred at 30-35° C. for 1 hour. The solid obtained was filtered, washed with water and dried to obtain 129 g 3-bromo-5-(2-pyridyl)-1,2-dihydropyridin-2-one.
Example-5
Preparation of 3-bromo-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one
In a 2 L round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, 100 g 3-bromo-5-(2-pyridyl)-1,2-dihydropyridin-2-one, 72.8 g phenylboronic acid and 500 mL N,N-dimethylformamide were added at 30-35° C. and stirred. 11.9 g copper acetate and 15.7 g pyridine were added and air was purged into the reaction mixture and stirred for 16 hours at 30-35° C. After the completion of the reaction, the reaction mixture was poured into 1200 mL aqueous ammonia at 10-15° C. and stirred for 2 hours at 30-35° C. The obtained solid was filtered, washed with water and dried to obtain 120 g 3-bromo-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one.
Example-6
Purification of 3-bromo-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one
In a 1 L round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, 100 g 3-bromo-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one and 500 mL isopropyl alcohol were stirred at 60-65° C. for 30 min. The reaction mixture was cooled to 20-25° C. and stirred for 30 min. The reaction mixture was filtered, washed with isopropanol and dried to obtain 96 g pure 3-bromo-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one.
Example-7
Preparation of Perampanel
In a 1 L round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, 100 g 3-bromo-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one and 125 g 2-(1,3,2-dioxaborinan-2-yl)benzonitrile and 1500 mL N,N-dimethylformamide were added under inert atmosphere. 44 g potassium carbonate and 4.2 g palladium tetrakis were added and stirred at 115-125° C. for 3 hours. The solvent was removed under vacuum. Ethyl acetate was added to the residue and the organic layer was distilled off to obtain perampanel (78 g).

////////Zydus Cadila, New Patent,US 20160039759, PERAMPANEL
Palbociclib, Sun Pharmaceutical Industries Ltd, New patent, WO 2016016769

Palbociclib
WO2016016769, A PROCESS FOR THE PREPARATION OF PALBOCICLIB
SUN PHARMACEUTICAL INDUSTRIES LIMITED [IN/IN]; Sun House, Plot No. 201 B/1 Western Express Highway Goregaon (E) Mumbai, Maharashtra 400 063 (IN)
TYAGI, Vipin; (IN).
MOHAMMAD, Kallimulla; (IN).
RAI, Bishwa Prakash; (IN).
PRASAD, Mohan; (IN)

The present invention relates to a process for the preparation of palbociclib utilizing a silyl-protected crotonic acid derivative to produce a silyl-protected 5-(1-methyl-3 carboxy-prop-1-en-1-yl)-2-chloro-piperazine followed by intramolecular cyclization of the compound the piperazine intermediate to produce 2-chloro-8-cyclopentyl-5-methyl-8H-pyrido[2,3-d]pyrimidin-7-one which is then converted to palbociclib.
Sun Pharma managing director Dilip Shanghvi.
Palbociclib chemically is 6-acetyl-8-cyclopentyl-5-methyl-2-[[5-(l-piperazinyl)-2-pyridinyl]amino]pyrido 2,3-d]pyrimidin-7(8H)-one, represented by the Formula I.

Formula I
U.S. Patent No. 6,936,612 discloses palbociclib and a process for the preparation of its hydrochloride salt.
U.S. Patent No. 7,781,583 discloses a process for the preparation of palbociclib, wherein 2-chloro-8-cyclopentyl-5-methyl-8H-pyrido[2,3-d]pyrimidin-7-one of Formula II

Formula II
prepared by reacting 5-bromo-2-chloro-N-cyclopentylpyrimidin-4-amine of Formula III

Formula III
with crotonic acid.
U.S. Patent No. 7,863,278 discloses polymorphs of various salts of palbociclib and processes for their preparation.
A first aspect of the present invention provides a process for the preparation of a compound of Formula IV,

Formula IV
wherein R is trimethylsilyl, dimethylsilyl, or fert-butyldimethylsilyl
comprising reacting a crotonic acid derivative of Formula V

Formula V
wherein R is trimethylsilyl, dimethylsilyl, or fert-butyldimethylsilyl
with a compound of Formula III

Formula III
in the presence of a palladium catalyst, a base, and optionally a ligand to give a compound of Formula IV.
A second aspect of the present invention provides a process for the preparation of palbociclib of Formula I,

Formula I
a) reacting a crotonic acid derivative of Formula V,

Formula V
wherein R is trimethylsilyl, dimethylsilyl, or fert-butyldimethylsilyl with a compound of Formula III

Formula III
in the presence of a palladium catalyst, a base, and optionally a ligand to give a compound of Formula IV

Formula IV
wherein R is trimethylsilyl, dimethylsilyl, or fert-butyldimethylsilyl; and b) converting the compound of Formula IV to palbociclib of Formula I.
A third aspect of the present invention provides a process for the preparation of a compound of Formula II

Formula II
a) reacting a crotonic acid derivative of Formula V,

Formula V
wherein R is trimethylsilyl, dimethylsilyl, or fert-butyldimethylsilyl with a compound of Formula III

Formula III
in the presence of a palladium catalyst, a base, and optionally a ligand to give a compound of Formula IV,

Formula IV
wherein R is trimethylsilyl, dimethylsilyl, or fert-butyldimethylsilyl; and b) intramolecular cyclization of the compound of Formula IV to give a
compound of Formula II.
A fourth aspect of the present invention provides a process for the preparation of palbociclib of Formula I

Formula I
comprising:
a) reacting a crotonic acid derivative of Formula V,

Formula V
wherein R is trimethylsilyl, dimethylsilyl, or fert-butyldimethylsilyl with a compound of Formula III

Formula III
in the presence of a palladium catalyst, a base, and optionally a ligand to give a compound of Formula IV

Formula IV
wherein R is trimethylsilyl, dimethylsilyl, or fert-butyldimethylsilyl;
intramolecular cyclization of the compound of Formula IV to give a compound of Formula II; and

Formula II
converting the compound of Formula II to palbociclib of Formula I.
EXAMPLES
Preparation of 2-chloro-8 -cyclopentyl-5 -methyl-8H-pyrido Γ2.3 – lpyrimidin-7-one (Formula II)
Step a: Preparation of trimethylsilyl (2£)-but-2-enoate (Formula V, when R is trimethylsilyl)
Crotonic acid (18.68 g) was taken in dichloromethane (80 mL) at room
temperature to obtain a solution. Hexamethyldisilazane (HMDS) (21 g) followed by imidazole (0.4 g) was added to the solution at room temperature under stirring. The reaction mixture was refluxed for 2 hours. Dichloromethane was recovered completely under vacuum at 45°C. Dichloromethane (200 mL) was again added to the reaction mixture, and then recovered completely under vacuum at 45°C. The colorless liquid obtained was taken as such for next step.
Step b: Preparation of 2-chloro-8-cyclopentyl-5-methyl-8H-pyrido[2,3-i/|pyrimidin-7-one (Formula II)
Method A
Trimethylsilyl (2£)-but-2-enoate (obtained from step a) and diisopropylethylamine (52 mL) were added to a solution of 5-bromo-2-chloro-N-cyclopentylpyrimidin-4-amine (20 g, Formula III) in tetrahydrofuran (100 mL) at room temperature under a nitrogen atmosphere. The reaction system was degassed under vacuum and then flushed with nitrogen; this evacuation procedure was repeated three times. Trans-dichlorobis(acetonitrile) palladium (II) (0.970 g) followed by the addition of tri-o-tolylphosphine (0.770 g) was added to the reaction mixture under a nitrogen atmosphere.
The reaction system was again degassed under vacuum and then flushed with nitrogen; this evacuation procedure was repeated three times. The reaction mixture was heated at 75°C to 80°C overnight. The progress of the reaction was monitored by thin layer chromatography (TLC) (60% ethyl acetate/toluene). Trans-dichlorobis(acetonitrile) palladium (II) (0.725 g) was again added followed by the addition of tri-o-tolylphosphine (0.725 g) to the reaction mixture at 75°C to 80°C. The reaction mixture was heated at 75°C to 80°C for 4 hours. After completion of the reaction, acetic anhydride (17 mL) was added, and then the mixture was stirred at 75°C to 80°C for 3 hours. The reaction mixture was cooled to room temperature. Dichloromethane (100 mL) and IN hydrochloric acid (100 mL) were added and then the mixture was stirred for 10 minutes. The layers were separated and the aqueous layer was re-extracted with dichloromethane (40 mL) and separated. The combined organic layers were washed with a 5% sodium bicarbonate solution (200 mL) at room temperature. The organic layer was separated and activated carbon (2 g) was added to the mixture. The mixture was stirred for 20 minutes at room temperature. The mixture was filtered through a Hyflo® bed and then washed with dichloromethane (40 mL). The organic layer was evaporated under vacuum to obtain a residue. Isopropyl alcohol (80 mL) was added to the residue and the solvent was evaporated under reduced pressure until 40 mL of isopropyl alcohol remained. Isopropyl alcohol (40 mL) was again added to the mixture, and then the solvent was evaporated under reduced pressure until 20 mL of isopropyl alcohol remained. The mixture was stirred for 3 hours at room temperature. The product was filtered, thenwashed with isopropyl alcohol (20 mL), and then dried under vacuum at 45°C to obtain the title compound.
Yield: 0.535% w/w
Chromatographic purity: 99.51%
Method B
Trimethylsilyl (2£)-but-2-enoate (obtained from step a) and diisopropylethylamine (26.5 mL) were added to a solution of 5-bromo-2-chloro-N-cyclopentylpyrimidin-4-amine (Formula III, 10 g) in tetrahydrofuran (50 mL) at room temperature under a nitrogen atmosphere. The reaction system was degassed under vacuum and then flushed with nitrogen; this evacuation procedure was repeated three times. Trans-dichlorobis(acetonitrile) palladium (II) (1.39 g) followed by the addition of tri-o- tolylphosphine (1.1 g) was added to the reaction mixture under a nitrogen atmosphere. The reaction system was degassed under vacuum and then flushed with nitrogen; this evacuation procedure was repeated three times. The reaction mixture was heated at 75 °C to 80°C overnight. After completion of the reaction, acetic anhydride (20 mL) was added, and then the mixture was stirred at 75°C to 80°C for 3 hours. The reaction mixture was cooled to room temperature. Dichloromethane (50 mL) and IN hydrochloric acid (50 mL) were added, and then the mixture was stirred for 10 minutes. The layers were separated and the aqueous layer was re-extracted with dichloromethane (20 mL) and separated. The combined organic layers were washed with a 5% sodium bicarbonate solution (200 mL) at room temperature. The organic layer was separated and activated carbon (1 g) was added to the mixture. The mixture was stirred for 20 minutes at room temperature. The mixture was filtered through a Hyflo® bed and then washed with dichloromethane (20 mL). The organic layer was evaporated under vacuum to obtain a residue. Isopropyl alcohol (40 mL) was added to the residue and then the solvent was evaporated under reduced pressure until 20 mL of isopropyl alcohol remained. Isopropyl alcohol (20 mL) was again added to the mixture and then the solvent was evaporated under reduced pressure until 20 mL of isopropyl alcohol remained. The mixture was stirred for 3 hours at room temperature. The product was filtered and washed with isopropyl alcohol (10 mL), and then dried under vacuum at 45°C to obtain the title compound.
Yield: 0.46% w/w
Chromatographic purity: 98.1%
/////Palbociclib, Sun Pharmaceutical Industries Ltd, New patent, WO 2016016769

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....












