Home » Antineoplastic (Page 2)
Category Archives: Antineoplastic
Anvumetostat
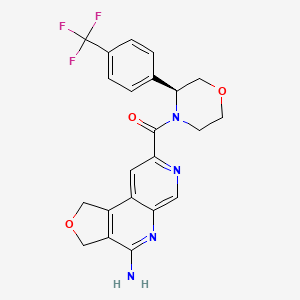
Anvumetostat
CAS 2790567-82-5
MF C22H19F3N4O3 MW444.4 g/mol
(4-amino-1,3-dihydrofuro[3,4-c][1,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone
(4-amino-1,3-dihydrofuro[3,4-c][1,7]naphthyridin-8-yl){(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl}methanone
antineoplastic, AMG 193, QAT649EJ5E, PRMT5-IN-27,
Anvumetostat (also known as AMG 193) is an orally available, small-molecule inhibitor of protein arginine methyltransferase 5 (PRMT5), primarily being developed for the treatment of advanced solid tumours with MTAP-null (methylthioadenosine phosphorylase-deficient) mutations.
Mechanism of Action
- Targeting PRMT5: It is a potent and selective MTA-cooperative inhibitor of PRMT5.
- Synthetic Lethality: In cells where the MTAP gene is deleted (a common occurrence in various cancers), a metabolite called MTA (methylthioadenosine) accumulates. Anvumetostat selectively binds to the PRMT5-MTA complex, inhibiting its methyltransferase activity.
- Cellular Impact: By blocking PRMT5, the drug reduces the methylation of arginine residues in histones (H2A, H3, and H4), which can lead to decreased growth or death of cancer cells.
Clinical Development
Anvumetostat was initially developed by Amgen, Inc. and is currently in clinical trials. Institute (.gov) +1
- Current Status: As of early 2026, it is in Phase 2 of global research and development.
- Study Focus: Trials are evaluating its efficacy both as a monotherapy and in combination with other treatments for adult patients with metastatic or locally advanced MTAP-null cancers.
Key Identifiers
- Alternate Names: AMG 193, AMG-193.
- Chemical Class: Orally bioavailable small molecule.
- Genetic Biomarker: Specifically targets cancers with MTAP-null status
Anvumetostat is an orally available small molecule inhibitor of protein arginine methyltransferase 5 (PRMT5), with potential antiproliferative and antineoplastic activities. Upon oral administration, anvumetostat selectively binds to PRMT5 and inhibits its function. By inhibiting its methyltransferase activity, levels of both monomethylated and dimethylated arginine residues in histones H2A, H3 and H4 are decreased. This modulates the expression of genes involved in several cellular processes, including cellular proliferation. This may increase the expression of antiproliferative genes and/or decrease the expression of genes that promote cell proliferation, which may lead to decreased growth of rapidly proliferating cells, including cancer cells. PRMT5, a type II methyltransferase that catalyzes the formation of both omega-N monomethylarginine (MMA) and symmetric dimethylarginine (sDMA) on histones and a variety of other protein substrates involved in signal transduction and cellular transcription, is overexpressed in several neoplasms. Elevated levels are associated with decreased patient survival. Methylthioadenosine phosphorylase (MTAP) is deleted in certain cancer cells leading to an accumulation of methylthioadenosine (MTA). As MTA inhibits PRMT5, MTAP-null cancer cells are specifically sensitive to PRMT5 inhibitors.
SYN
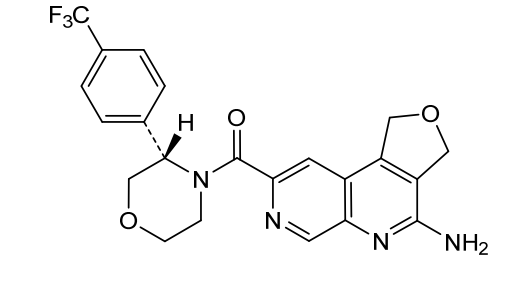
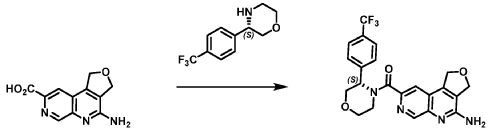
[0163] Examples 481 and 482: (4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4- (trifluoromethyl)phenyl)morpholino)methanone
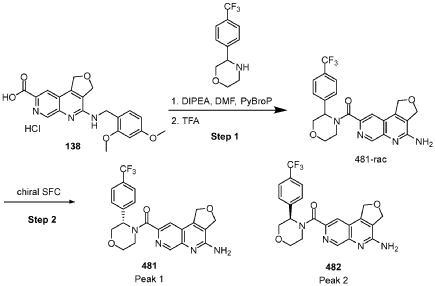
[0164] Step 1: To a solution of 3-(4-(trifluoromethyl)phenyl)morpholine (0.100 g, 0.432 mmol, Enamine), 4-((2,4-dimethoxybenzyl)amino)-l,3-dihydrofuro[3,4-c][l,7]naphthyridine-8-carboxylic acid hydrochloride (138) (0.271 g, 0.649 mmol) and l,l’-dimethyltriethylamine (0.559 g, 0.755 mL, 4.32 mmol, Sigma- Aldrich Corporation) in DMF (4 mL) was added bromotripyrrolidinophosphonium hexafluorophosphate (0.202 g, 0.432 mmol, Sigma-Aldrich Corporation) and the resulting mixture was heated at 50 °C for 30 min. The reaction was brought to rt, diluted with water, sat.NaHCCh and extracted with EtOAc (3x). The combined organics were dried over Na2SO4, filtered and concentrated. The residue was then chromatographed on silica gel using 0-50% 3:1 EtOAc/EtOH in heptane to afford (4-((2,4-dimethoxybenzyl)amino)- 1 ,3 -dihy drofuro [3 ,4-c] [ 1 ,7]naphthyridin-8-y 1) (3 – (4 -(trifluoromethyl)phenyl)morpholino)methanone (0.160 g, 0.269 mmol, 62.2% yield) as a light yellow solid, m/z (ESI): 595 (M+H)+.
[0165] To a solution of (4-((2,4-dimethoxybenzyl)amino)-l,3-dihydrofuro[3,4-c] [l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone (0.160 g, 0.269 mmol, 62.2 % yield) in DCM (2 mL) was added TFA (14.80 g, 10 mL, 130 mmol, Aldrich) and the resulting mixture was heated at 50 °C for 1 h. The reaction was concentrated, washed with 10% Na2CO3 and extracted with DCM. The combined organics were concentrated and chromatographed on silica gel using 0-50% 3:1 EtOAc/EtOH in heptane to afford (4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone as the TFA salt (0.078 g, 0.140 mmol, 32.3% yield) as an off-white solid, m/z (ESI): 445 (M+H)+.
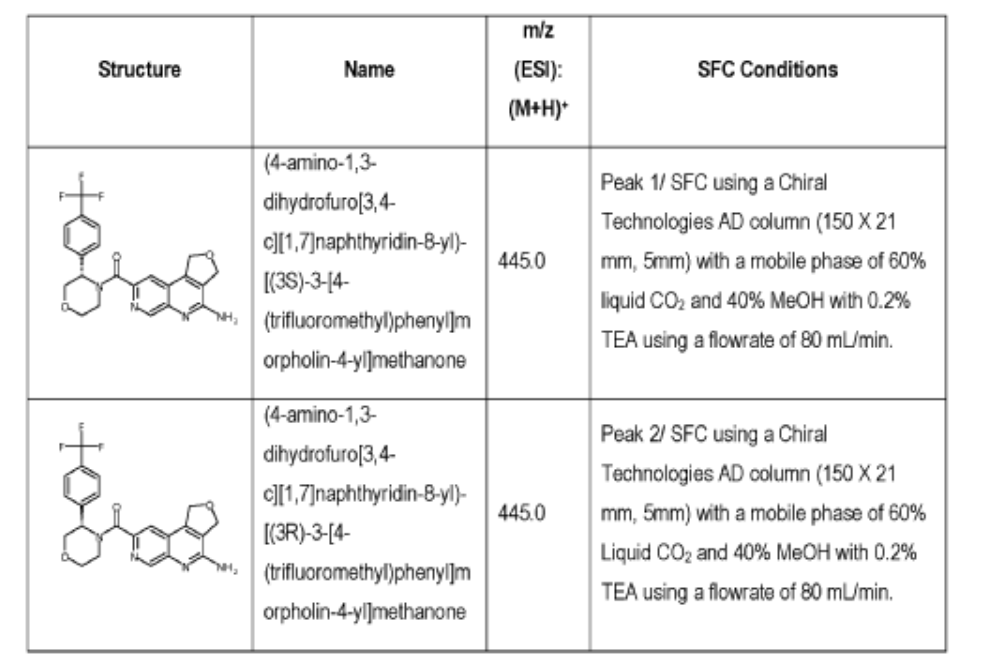
[0166] Step 2: (S)-(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4- (trifluoromethy l)phenyl)morpholino)methanone and (R)-(4-amino- 1 ,3 -dihy drofuro [3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone
(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone 2,2,2-trifluoroacetate were separated via preparative SFC using a Chiral Technologies AD column (150 x 21 mm, 5mm) with a mobile phase of 60% Liquid CO2 and 40% MeOH with 0.2% TEA using a flowrate of 80 mL/min to generate peak 1, (S)-(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone with an ee of >99%, and peak 2, (R)-(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone with an ee of 99.28%. Peak assignment determined by
SFC with AD column with 60% Liquid CO2 and 40% MeOH with 0.2% TEA and absolute
stereochemistry was arbitrarily assigned.
Peak 1: (S)-(4-amino-l,3-dihydrofuro[3,4-c][l,7]naphthyridin-8-yl)(3-(4-(trifluoromethyl)phenyl)morpholino)methanone (481) as a white solid . m/z (ESI): 445 (M+H)+. NMR 1H (400 MHz, DMSO-d6) 5 ppm 8.67 – 9.03 (m, 1 H), 7.85 (s, 1 H), 7.77 (br s, 4 H), 7.07 (br s, 2 H), 5.75 (s, 1 H), 5.37 (br s, 2 H), 5.04 (br s, 2 H), 4.46 – 4.61 (m, 1 H), 3.89 (br dd, J=12.2, 3.3 Hz, 4 H), 3.58 (br d, ./=5,8 Hz, 1 H). 19F NMR (377 MHz, DMSO-d6 ) 5 ppm -60.90 (br s, 3 F).
Peak 2: (R)-(4-amino- 1 ,3 -dihy drofuro [3 ,4-c] [ 1 ,7]naphthyridin-8-yl)(3 -(4-(trifluoromethyl)phenyl)morpholino)methanone (482) as a white solid, m/z (ESI): 445 (M+H)+. NMR 1H (400 MHz, DMSO-d6) 5 ppm 8.88 (br s, 1 H), 7.85 (s, 1 H), 7.77 (br d, J=1.7 Hz, 4 H), 7.07 (br s, 2 H),
5.69 – 5.78 (m, 1 H), 5.37 (br s, 2 H), 5.04 (br s, 2 H), 4.45 – 4.61 (m, 1 H), 3.89 (br dd, J=12.4, 3.3 Hz, 4 H), 3.51 – 3.64 (m, 1 H). 19F NMR (DMSO-d6, 377 MHz) 5 -60.90 (s, 3 F).
SYN
Example 4. Synthesis of Compound I – (4-amino-1 ,3-di hydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone
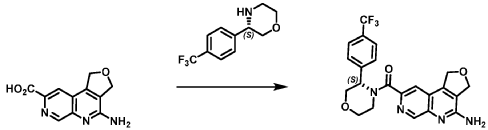
Reaction Scale 1
[0137] 4-Amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridine-8-carboxylic acid (1.0 kg, 4.3 mol, 1.0 equiv), (3S)-3-[4-(trifluoromethyl)phenyl]morpholine (1.2 kg, 5.2 mmol, 1.2- equiv), and DMF, (6.6 kg, 7.0 V) were charged to a clean, dry reactor. To the mixture was added triethylamine (1.1 Kg, 13.8 mol, 2.6 equiv). The mixture was cooled to 10 ± 5 °C and O-(benzotriazol-1-yl)-N,N,N’,N’-tetramethyluronium tetrafluoroborate (TBTU) (1.67 kg, 5.2 mol, 1.2 equiv) was added slowly. Next, an additional amount of DMF (0.94 Kg, 1 V) was added. The reaction mixture was warmed to 25 ± 5 °C and stirred over 18 hours. Water (1 .0 kg, 1 V) was charged followed by MeCN (1 .6 kg, 2 V) and the reaction mass was warmed to 45 °C. Next, water (7.0 Kg, 7 V) was added over 30 min. A seed lot of 4-amino-1 ,3-dihydrofuro[3,4-c][1,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone (10 g, 22 mmol, 0.01 equiv), was charged and the mixture was stirred at 45 °C for over 2 hours before being cooled to 20 °C over 10 hours. Water (12.0 kg, 12 V) was added over 2 hours at 20 °C and further stirred for over 4 hours before being filtered. The reactor was rinsed with a mixture of 10% DMF in water (9.83 kg, 10 V) and the resulting rinse mixture was used to wash the cake. The reactor was rinsed with a mixture of water (10.0k kg, 10 V) and the resulting rinse mixture was used to wash the cake. This rinsing and washing protocol was repeated once more with water (10.0k kg, 10V). The cake was dried under vacuum with a stream of nitrogen to afford (4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-
(trifluoromethyl)phenyl]morpholin-4-yl]methanone. LCMS: 445.20 1H NMR (400 MHz, DMS0-d6 at 130 °C): 8.87 (s, 1 H), 7.80 (s, 1 H), 7.73 (d, 0=8.7 Hz, 2H), 7.71 (d, 0=8.7 Hz, 2H), 6.58 (br s, 2H), 5.72 (br s, 1 H), 5.38 (m, 2H), 5.09 (t, 0=3.5 Hz, 2H), 4.44 (br d, 0=12.3 Hz, 1 H), 4.08 (br d, 0=13.4 Hz, 1 H), 3.96 (dd, 0=12.3, 3.7 Hz, 1 H), 3.86 (br dd, 0=11 .4, 3.0 Hz, 1 H), 3.66 (td, 0=11 .4, 3.0 Hz, 1 H), 3.28 (m, 1 H).
Reaction Scale 2
[0138] 4-Amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridine-8-carboxylic acid (85.0 g, 352.2 mmol, 1.0 equiv), (3S)-3-[4-(trifluoromethyl)phenyl]morpholine (99.6 g, 422.6 mmol, 1.2- equiv), and DMF, (674 mL, 8.7 mol, 7.9 V) were charged to a clean, dry 5 L reactor. To the mixture was added 1 -methylimidazole (75.2 g, 916.2 mmol, 2.6 equiv). The mixture was cooled to 0 °C and N,N,N’,N’-tetramethylchloroformamidinium hexafluorophosphate (TCFH) (118.6 g, 422.6 mmol, 1.2 equiv) was added slowly. Next, an additional amount of DMF (170 mL, 2 V) was added at 0 °C. The reaction mixture was warmed to 25 °C and stirred overnight. Next, the reaction mass was warmed to 45 °C and 2-methyltetrahydrofuran, (169.2 mL, 2 V) was added followed by slow addition of water (850 mL, 10 V) over 30 min by addition funnel. A seed lot of 4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone (1.6 g, 3.5 mmol, 0.1 equiv), was charged as a slurry in a 1 :1 v/v of DMF and water (31 .3 mL) and the mixture was stirred at 45 °C for approximately 12 hrs. Water (510 mL, 6 V) was added over 1 h 10 min by addition funnel and the mixture was further stirred at 45°C for 30 min before being filtered. The reactor was rinsed with water (340 mL, 4 V) and the resulting rinse mixture was used to wash the cake. This rinsing and washing protocol was repeated twice more. The cake was dried under vacuum with a stream of nitrogen to afford (4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone. LCMS: 445.20 1H NMR (400 MHz, DMSO-d6 at 130 °C): 8.87 (s, 1 H), 7.80 (s, 1 H), 7.73 (d, J=8.7 Hz, 2H), 7.71 (d, J=8.7 Hz, 2H), 6.58 (br s, 2H), 5.72 (br s, 1 H), 5.38 (m, 2H), 5.09 (t, J=3.5 Hz, 2H), 4.44 (br d, J=12.3 Hz, 1 H), 4.08 (br d, J=13.4 Hz, 1 H), 3.96 (dd, J=12.3, 3.7 Hz, 1 H), 3.86 (br dd, J=11.4, 3.0 Hz, 1 H), 3.66 (td, J=11.4, 3.0 Hz, 1 H), 3.28 (m, 1 H).
Reaction Scale 3:
[0139] 4-Amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridine-8-carboxylic acid (Compound A’) (20.0 g, 86.5 mmol, 1.0 equiv) was added to dimethylsulfoxide (400 mL) at 20 °C. To the mixture was added 1 ,T-carbonyldiimidazole (15.4 g, 95.2 mmol, 1.1 equiv) and the mixture was heated to 60 °C for 1 hour. A solution of (S)-3-(4-(trifluoromethyl)phenyl)morpholin-4-ium chloride (25.5 g, 95.2 mmol, 1.1 equiv) and dimethylsulfoxide (40 mL) was added, and the mixture was heated to 80 °C for 11 hours. The reaction mixture was cooled to 35 °C, then water (265 mL) was added, then the batch was cooled to 20 °C. The reaction was filtered, washed with 40% water:DMSO (80 mL), then washed with water (100 mL). The cake was dried under vacuum with a stream of nitrogen to afford (4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone (Compound I). LCMS: 445.20 1H NMR (400 MHz, DMSO-d6 at 130 °C): 8.87 (s, 1 H), 7.80 (s, 1 H), 7.73 (d, J=8.7 Hz, 2H), 7.71 (d, J=8.7 Hz, 2H), 6.58 (br s, 2H), 5.72 (br s, 1 H), 5.38 (m, 2H), 5.09 (t, >3.5 Hz, 2H), 4.44 (br d, >12.3 Hz, 1H), 4.08 (br d, >13.4 Hz, 1 H), 3.96 (dd, >12.3, 3.7 Hz, 1 H), 3.86 (br dd, >11 .4, 3.0 Hz, 1 H), 3.66 (td, >11 .4, 3.0 Hz, 1 H), 3.28 (m, 1 H).
Recrystallization of Compound I
[0140] A clean, dry 5 L reactor was charged with (4-amino-1 ,3-dihydrofuro[3,4-c][1 ,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl]methanone (279.7 g, 0.6 mol, 1.0 equiv) followed by acetone (6.2 L,
22 V). The mixture was stirred at 40 °C for 15 minutes before cooling to 25 °C. The reactor was discharged into a flask and the reactor was rinsed with acetone and the process stream was polish-filtered back into the reactor.
The reactor jacket was set to 65 °C and the reaction volume was reduced to approximately 6 V by distillation at atmospheric pressure, crystallization was observed. The reaction temperature was set to cool to 20 °C over two hours. Heptane (2.8 L, 10 V) was added over two hours. The slurry was filtered and the cake was washed twice with a 4:1 Heptane/acetone mix (750 mL, 3 V each) and dried under vacuum with a nitrogen purge to afford (4-amino-1,3-dihydrofuro[3,4-c][1,7]naphthyridin-8-yl)-[(3S)-3-[4-(trifluoromethyl)phenyl]morpholin-4-yl] methanone.


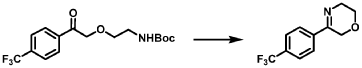

ADVT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Process for synthesizing naphthyridine derivatives and intermediates thereofPublication Number: EP-4396170-A1Priority Date: 2021-08-30
- PRMTS inhibitorsPublication Number: CN-116888120-APriority Date: 2020-12-16
- Prmts inhibitorsPublication Number: WO-2022132914-A1Priority Date: 2020-12-16
- Prmts inhibitorsPublication Number: EP-4263545-A1Priority Date: 2020-12-16
- Mta-cooperative prmt5 inhibitors for use in the treatment of cancerPublication Number: EP-4572760-A1Priority Date: 2022-08-15
- Cancer treatments using mta-cooperative prmt5 inhibitorsPublication Number: WO-2023196545-A1Priority Date: 2022-04-08
- Process for the synthesis of naphthyridine derivatives and intermediates thereofPublication Number: CN-117897379-APriority Date: 2021-08-30
- Process for synthesizing naphthyridine derivatives and intermediates thereofPublication Number: WO-2023034786-A1Priority Date: 2021-08-30
- Process for Synthesizing Naphthyridine Derivatives and Intermediates ThereofPublication Number: US-2024360147-A1Priority Date: 2021-08-30
- Prmt5 inhibitor for use in cancer therapyPublication Number: WO-2024170488-A1Priority Date: 2023-02-13
- Cancer treatments using a prmt5 inhibitor and a mat2a inhibitorPublication Number: WO-2024118897-A1Priority Date: 2022-11-30
- Cancer treatments using a prmt5 inhibitor and a mat2a inhibitorPublication Number: EP-4626435-A1Priority Date: 2022-11-30
- MTA synergizes with PRMT5 inhibitors for cancer treatmentPublication Number: CN-119730853-APriority Date: 2022-08-15
- Mta-cooperative prmt5 inhibitors for use in the treatment of cancerPublication Number: WO-2024038004-A1Priority Date: 2022-08-15
////////anvumetostat, ANAX LAB, antineoplastic, AMG 193, QAT649EJ5E, PRMT5-IN-27,
Andamertinib
Andamertinib
CAS 2254145-43-0
MF C31H36N8O3 MW568.7 g/mol
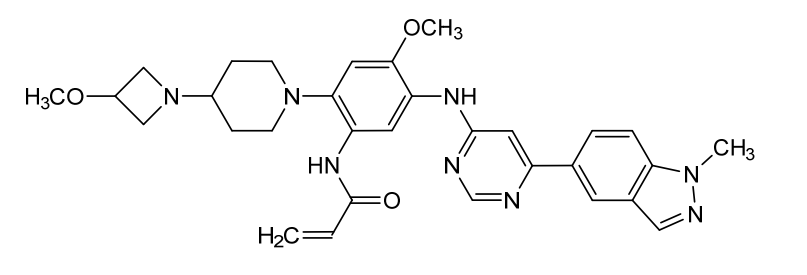
N-[4-methoxy-2-[4-(3-methoxyazetidin-1-yl)piperidin-1-yl]-5-[[6-(1-methylindazol-5-yl)pyrimidin-4-yl]amino]phenyl]prop-2-enamide
N-(4-methoxy-2-[4-(3-methoxyazetidin-1-yl)piperidin-1-yl]-5-{[6-(1-methyl-1H-indazol-5-yl)pyrimidin-4-yl]amino}phenyl)prop-2-enamide
epidermal growth factor receptor tyrosine kinase inhibitor, antineoplastic, PLB 1004, 5X3KAG7ZBW
Andamertinib (also known as PLB1004) is an investigational, orally bioavailable, and irreversible small-molecule inhibitor of the epidermal growth factor receptor (EGFR). It is primarily being developed to treat non-small cell lung cancer (NSCLC) with specific genetic mutations.
Key Clinical & Therapeutic Features
- Target Mutations: It specifically targets EGFR exon 20 insertion (ex20ins) mutations, which are often resistant to standard first- and second-generation EGFR inhibitors.
- Broad Selectivity: Beyond ex20ins, it also shows activity against classical mutations like Del19, L858R, and the resistance mutation T790M.
- Brain Penetration: Andamertinib is designed to cross the blood-brain barrier, making it potentially effective for patients with brain metastases.
- Clinical Performance: In phase 2 studies (e.g., the KANNON study), it demonstrated a confirmed objective response rate (ORR) of 42.7% and a median progression-free survival of 6.2 months in pretreated patients.
Regulatory Status (as of Early 2026)
- China: A New Drug Application (NDA) was accepted by the National Medical Products Administration (NMPA) in May 2025 and granted priority review for treating NSCLC with EGFR ex20ins mutations.
- Global: It remains in various stages of clinical trials globally, including studies for first-line treatment and combination therapies with other agents like vebreltinib.
Andamertinib is an orally bioavailable, mono-anilino-pyrimidine, mutant-selective epidermal growth factor receptor (EGFR) inhibitor, with potential antineoplastic activity. Upon oral administration, andamertinib targets, binds to and irreversibly inhibits the activity of various EGFR mutations, including exon 20 insertion (Ex20ins) activating mutations, the gatekeeper mutation T790M, ExDel19, and L858R. This prevents EGFR-mediated signaling, induces cell death and inhibits tumor growth in tumor cells expressing these EGFR mutations. EGFR, a receptor tyrosine kinase mutated in many tumor cell types, plays a key role in tumor cell proliferation and tumor vascularization.
SYN

SYN
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Aminopyrimidine compound, preparation method therefor and use thereofPublication Number: US-11352352-B2Priority Date: 2017-06-13Grant Date: 2022-06-07
- Aminopyrimidine compound, preparation method therefor and use thereofPublication Number: EP-3640248-A1Priority Date: 2017-06-13
- Aminopyrimidine compound, preparation method therefor and use thereofPublication Number: US-2020087296-A1Priority Date: 2017-06-13
- Aminopyrimidine derivatives, preparation method therefor and use thereofPublication Number: EP-3640248-B1Priority Date: 2017-06-13Grant Date: 2023-08-23
//////////andamertinib, FLAX LAB, antineoplastic, PLB 1004, 5X3KAG7ZBW
Amezalpat
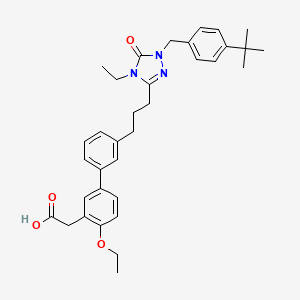
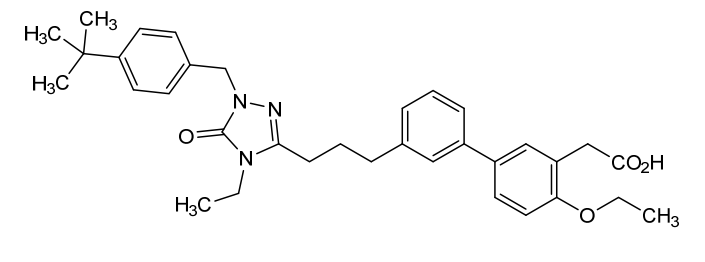
Amezalpat
CAS 1616372-41-8
MF C34H41N3O4 MW555.7 g/mol
- [1,1′-Biphenyl]-3-acetic acid, 3′-[3-[1-[[4-(1,1-dimethylethyl)phenyl]methyl]-4-ethyl-4,5-dihydro-5-oxo-1H-1,2,4-triazol-3-yl]propyl]-4-ethoxy-
- 2-(3′-(3-(1-(4-(tert-Butyl)benzyl)-4-ethyl-5-oxo-4,5-dihydro-1H-1,2,4-triazol-3-yl)propyl)-4-ethoxy-[1,1′-biphenyl]-3-yl)acetic acid
- 3′-(3-(1-((4-(1,1-DIMETHYLETHYL)PHENYL)METHYL)-4-ETHYL-4,5-DIHYDRO-5-OXO-1H-1,2,4-TRIAZOL-3-YL)PROPYL)-4-ETHOXY(1,1′-BIPHENYL)-3-ACETIC ACID
2-[5-[3-[3-[1-[(4-tert-butylphenyl)methyl]-4-ethyl-5-oxo-1,2,4-triazol-3-yl]propyl]phenyl]-2-ethoxyphenyl]acetic acid

peroxisome proliferator-activated receptor alpha (PPARα) antagonist, antineoplastic, TPST 1120, FDA Fast Track, Orphan Drug, 1EQ4LQN9N3
Amezalpat (formerly TPST-1120) is an investigational, oral, small-molecule inhibitor targeting peroxisome proliferator-activated receptor alpha (PPAR being developed by Tempest Therapeutics. It works by directly targeting tumor cells and reducing immune suppression in the tumor microenvironment. In combination with atezolizumab and bevacizumab, it has shown improved survival in hepatocellular carcinoma (HCC) patients, receiving FDA Fast Track and Orphan Drug designations.
Key Details on Amezalpat
- Indication: Primarily being studied for unresectable or metastatic hepatocellular carcinoma (liver cancer).
- Mechanism: A selective, competitive antagonist of PPAR
, which plays a role in fatty acid metabolism in cancer cells.
- Clinical Efficacy: A phase 1b/2 study indicated that adding amezalpat to standard-of-care (atezolizumab + bevacizumab) improved median overall survival to 21 months compared to 15 months for the control, according to Tempest Therapeutics.
- Trial Status: A pivotal Phase 3 study (NCT06680258) to evaluate this combination as a first-line treatment is planned for 2025.
- Other Potential Uses: Preclinical data suggests potential activity in other advanced solid tumors, including renal cell carcinoma.
Disclaimer: Amezalpat is an investigational agent and is not yet approved by the FDA for widespread clinical use.
Amezalpat is an orally bioavailable, small molecule, selective and competitive antagonist of peroxisome proliferator activated receptor alpha (PPARa), with potential immunomodulating and antineoplastic activities. Upon oral administration, amezalpat targets, binds to and blocks the activity of PPARa, thereby blocking transcription of PPARa target genes leading to an intracellular metabolism shift from fatty acid oxidation (FAO) to glycolysis in FAO-dependent tumors and reducing the production of fatty acids in the tumor microenvironment (TME). As fatty acids are essential for tumor cell growth in FAO-dependent tumor cells and are needed for the metabolism of suppressive immune cells in the TME, including regulatory T-cells (Tregs), reducing the amount of fatty acids leads to a direct killing of FAO-dependent tumor cells. It also skews macrophages from the immune suppressive M2 phenotype to an effector M1 phenotype and facilitates the cytotoxicity of immune effector cells, thereby stimulating an anti-tumor immune response and further killing tumor cells. Amezalpat also restores the natural inhibitor of angiogenesis thrombospondin-1 (TSP-1) and stimulator of interferon genes (STING) in the TME. PPARa, a ligand-activated nuclear transcription factor and metabolic checkpoint, regulates the expression of FAO genes and lipid metabolism. It plays a key role in immunosuppression in the TME. FAO is a metabolic pathway essential to tumor growth, survival and immunosuppression.
SYN
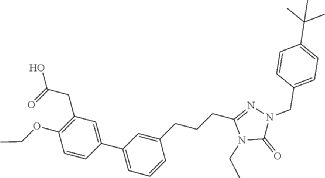
Example 6: 2-(3′-(3-(1-(4-(tert-Butyl)benzyl)-4-ethyl-5-oxo-4,5-dihydro-1H-1,2,4-triazol-3-yl)propyl)-4-ethoxy-[1,1′-biphenyl]-3-yl)acetic acid
SYN
SYN
WO2014099503 TRIAZOLONE COMPOUNDS AND USES THEREOF
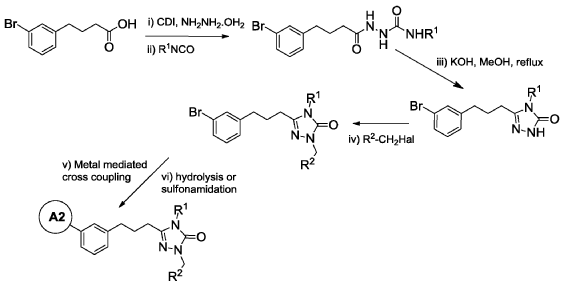
Example 6: 2-(3′-(3-(1-(4-(tert-Butyl)benzyl)-4-ethyl-5-oxo-4,5-dihydro-1H-1,2,4-triazol-3-yl)propyl)-4-ethoxy-[1, 1′-biphenyl]-3-yl)acetic acid
Pat
WO2025235527 CRYSTALLINE FORMS OF A PPAR ALPHA ANTAGONIST
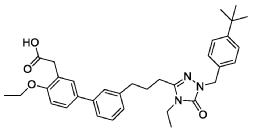
2-(3′-(3-(l-(4-(tertbutyl)benzyl)-4-ethyl-5-oxo-4,5-dihydro-lH-l,2,4-triazol-3-yl)propyl)-4-ethoxy-[1,T-biphenyl]-3-yl)acetic acid, depicted below as Compound A
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Triazolone compounds and uses thereofPublication Number: US-2017239223-A1Priority Date: 2012-12-20
- Triazolone compounds and uses thereofPublication Number: WO-2014099503-A1Priority Date: 2012-12-20
- Triazolone compounds and uses thereofPublication Number: US-10568871-B2Priority Date: 2012-12-20Grant Date: 2020-02-25
- Triazolone compounds and uses thereofPublication Number: US-2024041837-A1Priority Date: 2012-12-20
- Compound or pharmaceutically acceptable salt thereof, pharmaceutical composition and uses thereofPublication Number: BR-112015013350-B1Priority Date: 2012-12-20
- Triazolone compounds and uses thereofPublication Number: US-2015344446-A1Priority Date: 2012-12-20
- Triazolone compounds and uses thereofPublication Number: US-11666557-B2Priority Date: 2012-12-20Grant Date: 2023-06-06
- Triazolone compounds and uses thereofPublication Number: US-2020138790-A1Priority Date: 2012-12-20
- Triazolone compounds and uses thereofPublication Number: US-9676754-B2Priority Date: 2012-12-20Grant Date: 2017-06-13
- Triazolone compounds and uses thereofPublication Number: CA-2894281-CPriority Date: 2012-12-20Grant Date: 2021-04-20
- Triazolone compounds and uses thereofPublication Number: WO-2024102620-A2Priority Date: 2022-11-09
- Triazolone compounds and uses thereofPublication Number: AU-2013363398-B2Priority Date: 2012-12-20Grant Date: 2017-06-01
- Triazolone compounds and uses thereofPublication Number: EP-2935228-B9Priority Date: 2012-12-20Grant Date: 2017-12-06
- Triazolone compounds and uses thereofPublication Number: CA-2894281-A1Priority Date: 2012-12-20
- Triazolone compounds and uses thereofPublication Number: EP-2935228-B1Priority Date: 2012-12-20Grant Date: 2017-08-02
/////////////amezalpat, ANAX LAB, antineoplastic, TPST 1120, FDA Fast Track, Orphan Drug, 1EQ4LQN9N3
Alnodesertib
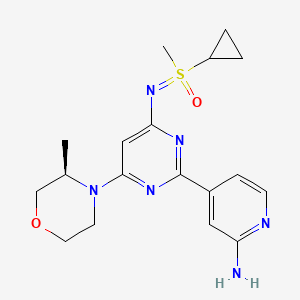
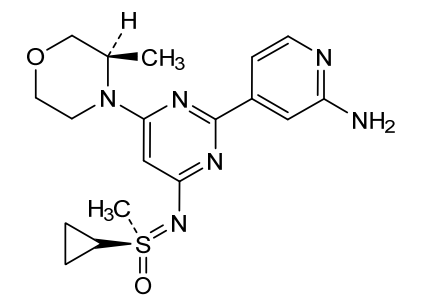
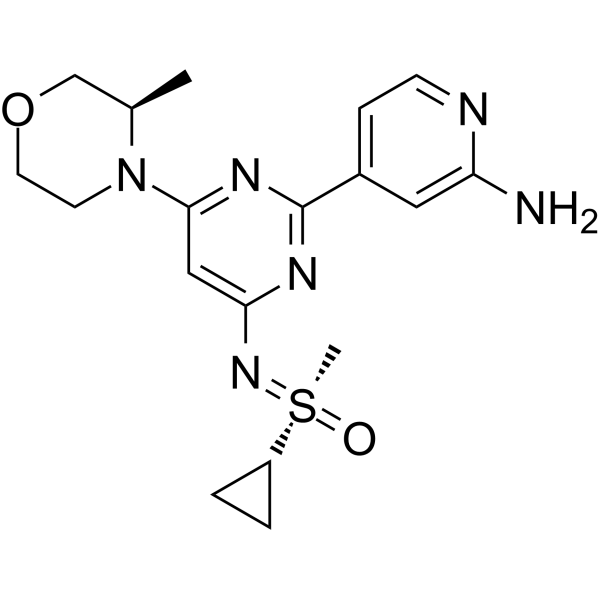
Alnodesertib
CAS 2267316-76-5
MF C18H24N6O2S MW388.49
4-[4-[(cyclopropyl-methyl-oxo-λ6-sulfanylidene)amino]-6-[(3R)-3-methylmorpholin-4-yl]pyrimidin-2-yl]pyridin-2-amine
4-[4-[(cyclopropyl-methyl-oxo-lambda6-sulfanylidene)amino]-6-[(3R)-3-methylmorpholin-4-yl]pyrimidin-2-yl]pyridin-2-amine
(S)-({2-(2-aminopyridin-4-yl)-6-[(3R)-3-methylmorpholin-4-yl]pyrimidin-4-yl}imino)(cyclopropyl)(methyl)-λ6
-sulfanone
serine/threonine kinase inhibitor, antineoplastic, ART 0380, EX-A9085
Alnodesertib (formerly known as ART0380) is an investigational, orally administered drug designed to treat various types of cancer. It is a selective inhibitor of ATR (Ataxia-Telangiectasia and Rad3-related protein), a key kinase involved in DNA repair and cell cycle progression.
Mechanism of Action
Alnodesertib works by disrupting the DNA Damage Response (DDR):
- Targets ATR Kinase: It selectively inhibits ATR, which cancer cells rely on to fix DNA damage caused by rapid replication.
- Blocks Signaling: By blocking the phosphorylation of CHK1, it prevents the activation of DNA damage checkpoints.
- Induces Apoptosis: Inhibiting these repair pathways prevents cancer cells from surviving replication stress, ultimately leading to cell death (apoptosis).
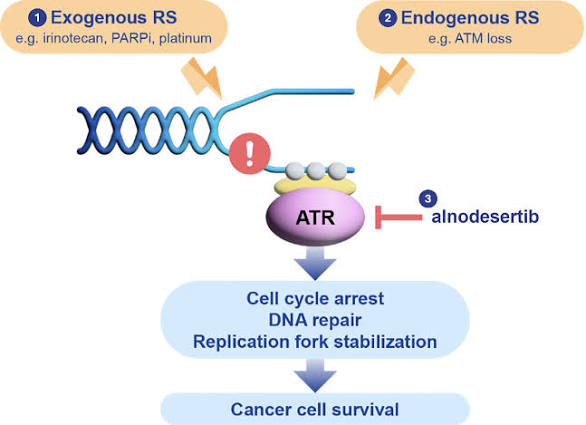

Clinical Status and Indications
As of early 2026, alnodesertib is undergoing several clinical trials:
- Metastatic Colorectal Cancer (mCRC): The FDA granted Fast Track designation in September 2025 for alnodesertib in combination with irinotecan for adult patients with ATM-negative mCRC in the third-line setting.
- Ovarian Cancer: In March 2026, Artios Pharma reported that a Phase 2a study reached its primary endpoint, showing that adding a low dose of alnodesertib to gemcitabine improved progression-free survival in patients with platinum-resistant high-grade serous ovarian carcinoma (HGSOC).
- Other Solid Tumours: It is being evaluated in the ongoing STELLA Phase 1/2a study for its potential across multiple solid tumour types characterized by high replication stress.
Key Facts
| Feature | Details |
|---|---|
| Developer | Artios Pharma Limited |
| Drug Class | ATR Kinase Inhibitor |
| Administration | Oral |
| Current Phase | Phase 2 clinical trials |
| FDA Status | Fast Track Designation (for ATM-negative mCRC) |
SYN
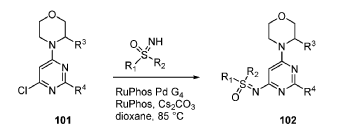
EXAMPLES 39a and 39b
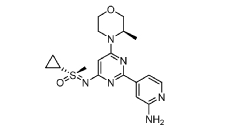
(R)-((2-(2-aminopyridin-4-yl)-6-((R)-3-methylmorpholino)pyrimidin-4- yl)imino)(cyclopropyl)(methyl)-λ6-sulfanone
and
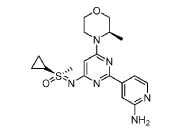
(S)-((2-(2-aminopyridin-4-yl)-6-((R)-3-methylmorpholino)pyrimidin-4- yl)imino)(cyclopropyl)(methyl)-λ6-sulfanone
[0648] Synthesis is similar to that described for Example 24. The mixture of diastereomers (26.8 mg, 0.069 mmol) was separated by Chiral SFC (Mobile phase: n-hexane (0.1% DEA):EtOH(0.1% DEA) = 70:30; Flow rate: 80 g / min; 20 min; Column temperature: 35 °C; Back pressure: 100 bar; Column: Gilson-281, AY 20 x 250mm, 10 μm) to afford the two diastereomers of unknown absolute stereochemistry at the sulfur atom, title compounds 39a (6.6mg, 25% yield, >99% ee) as a white solid and 39b (7.1mg, 27% yield, >99% ee) as a white solid.
[0649] 39a ((R)-cyclopropyl(methyl)-λ6-sulfanone or (S)-cyclopropyl(methyl)-λ6-sulfanone): 1H NMR (500 MHz, CD3OD) δ 8.03 – 7.91 (m, 1H), 7.53 (s, 1H), 7.49 (dd, J =5.5, 1.4 Hz, 1H), 5.97 (s, 1H), 4.48 (d, J = 4.6 Hz, 1H), 4.11 (d, J = 12.0 Hz, 1H), 4.02 (dt, J = 11.3, 3.6 Hz, 1H), 3.82 (d, J = 11.4 Hz, 1H), 3.75 (dt, J = 11.5, 3.0 Hz, 1H), 3.65 – 3.56 (m, 4H), 3.25 (tdJ, = 12.8, 3.8 Hz, 1H), 3.01 (td, J = 7.9, 4.0 Hz, 1H), 1.42 (dd, J = 10.2, 5.4 Hz, 1H), 1.31 (dt, J = 11.1, 6.2 Hz, 4H), 1.20 (dt,J = 11.3, 5.7 Hz, 2H); MS (ES+) C18H24N6O2S requires: 388, found: 389 [M+H]+; Rt = 11.35 min.
[0650] 39b ((R)-cyclopropyl(methyl)-λ6-sulfanone or (S)-cyclopropyl(methyl)-λ6-sulfanone): 1H NMR (500 MHz, CD3OD) δ 7.97 (d, J = 5.4 Hz, 1H), 7.53 (s, 1H), 7.49 (dt, J = 5.5, 1.3 Hz, 1H), 5.97 (s, 1H), 4.50 (s, 1H), 4.08 (d, J = 12.7 Hz, 1H), 4.02 (dd, J = 11.4, 3.7 Hz, 1H), 3.82 (d, J = 11.3 Hz, 1H), 3.75 (dt, J = 11.4, 3.0 Hz, 1H), 3.66 – 3.55 (m, 4H), 3.25 (tdJ, = 12.9, 3.9 Hz, 1H), 3.05 – 2.97 (m, 1H), 1.41 (dt, J = 10.6, 5.2 Hz, 1H), 1.31 (dd, J = 11.8, 5.8 Hz, 4H), 1.20 (dt, J = 11.1, 5.6 Hz, 2H); MS (ES+) C18H24N6O2S requires: 388, found: 389 [M+H]+; Rt = 15.22 min.
[0651] Alternatively, Example 39a can also be prepared from Int. CC, Isomer lb.
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- HETEROCYCLIC INHIBITORS OF KINASE ATRPublication Number: WO-2019014618-A1Priority Date: 2017-07-13
- Heterocyclic inhibitors of ATR kinasePublication Number: US-11434233-B2Priority Date: 2017-07-13Grant Date: 2022-09-06
- Heterocyclic inhibitors of ATR kinasePublication Number: US-10800769-B2Priority Date: 2017-07-13Grant Date: 2020-10-13
- Heterocyclic inhibitors of ATR kinasePublication Number: US-10392376-B2Priority Date: 2017-07-13Grant Date: 2019-08-27
- Heterocyclic inhibitors of atr kinasePublication Number: US-2019016713-A1Priority Date: 2017-07-13
- Heterocyclic inhibitors of atr kinasePublication Number: US-2020102296-A1Priority Date: 2017-07-13
- Heterocyclic inhibitors of atr kinasePublication Number: US-2021047311-A1Priority Date: 2017-07-13
///////alnodesertib, ANAX LAB, serine/threonine kinase inhibitor, antineoplastic, ART 0380, EX-A9085
Zelebrudomide
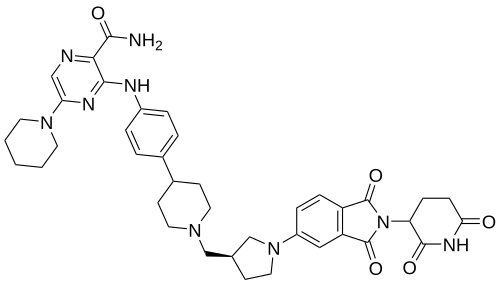
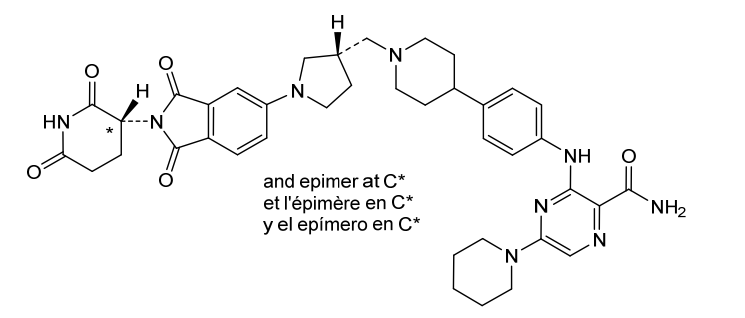
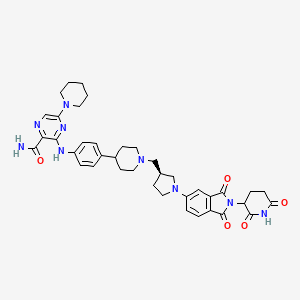
Zelebrudomide
CAS 2416131-46-7
MF C39H45N9O5 MW 719.8 g/mol
3-[4-[1-[[(3S)-1-[2-(2,6-dioxopiperidin-3-yl)-1,3-dioxoisoindol-5-yl]pyrrolidin-3-yl]methyl]piperidin-4-yl]anilino]-5-piperidin-1-ylpyrazine-2-carboxamide
protein degrader, antineoplastic, NX 2127, LSC67HA8DE, NX-2127, BTK Degrader NX-2127
Zelebrudomide (NX-2127) is an investigational new drug that is being evaluated by Nurix Therapeutics for the treatment of relapsed or refractory B-cell malignancies such as chronic lymphocytic leukemia (CLL), mantle cell lymphoma (MCL), diffuse large B-cell lymphoma (DLBCL), and Waldenström macroglobulinemia (WM). It is an orally bioavailable proteolysis targeting chimera (PROTAC) designed to degrade Bruton’s tyrosine kinase (BTK) along with the immunomodulatory proteins Ikaros (IKZF1) and Aiolos (IKZF3).[1]
- OriginatorNurix
- ClassAntineoplastics; Small molecules
- Mechanism of ActionAgammaglobulinaemia tyrosine kinase degraders; IKZF1 protein degraders; IKZF3 protein degraders
Phase IChronic lymphocytic leukaemia; Diffuse large B cell lymphoma; Follicular lymphoma; Lymphoma; Mantle-cell lymphoma; Marginal zone B-cell lymphoma; Waldenstrom’s macroglobulinaemia
- 09 Dec 2024Pharmacodynamics data from a preclinical studies in Chronic lymphocytic leukaemia released by Nurix Therapeutics
- 11 Jul 2024NX 2127 is still in phase I development in Chronic-lymphocytic-leukaemia (Late-stage disease, Second-line therapy or greater) in USA (PO) (NCT04830137)
- 11 Jul 2024NX 2127 is still in phase I development in Diffuse large B cell lymphoma(Late-stage disease, Second-line therapy or greater) in USA (PO) (NCT04830137)
Zelebrudomide, (S)- is the S-enantiomer of zelebrudomide, an orally bioavailable chimeric targeting molecule (CTM) and targeted degrader of Bruton’s tyrosine kinase (BTK), with potential immunomodulatory drug (IMiD) and antineoplastic activities. Zelebrudomide is comprised of a cereblon (CRBN)-binding moiety conjugated, via a linker, to a BTK-binding moiety. Upon administration, zelebrudomide targets and binds to BTK with its BTK-targeting moiety. Upon binding, the CRBN-binding moiety recruits CRBN, a component of the CRL4-CRBN E3 ubiquitin ligase complex. This catalyzes ubiquitination and proteasome-mediated degradation of BTK, and prevents the activation of the B-cell antigen receptor (BCR) signaling pathway. This prevents both B-cell activation and BTK-mediated activation of downstream survival pathways. This leads to an inhibition of the growth of malignant B-cells that overexpress BTK. In addition, zelebrudomide catalyzes the degradation of CRBN neosubstrates Aiolos (IKZF3) and Ikaros (IKZF1), two transcription factors regulating T-cell function. This modulates the activity of the immune system and increases the activation of T-lymphocytes, thereby increasing T-cell-mediated anti-tumor effects. BTK, a member of the src-related BTK/Tec family of cytoplasmic tyrosine kinases, is overexpressed in B-cell malignancies; it plays an important role in B-lymphocyte development, activation, signaling, proliferation and survival. CRBN, the substrate recognition component of the CRL4-CRBN E3 ubiquitin ligase complex, plays a key role in the ubiquitination of certain proteins. Compared to BTK inhibitors, zelebrudomide may overcome tumor resistance associated with BTK inhibitor-induced resistance mutations.
A Study of NX-2127 in Adults With Relapsed/Refractory B-cell Malignancies
CTID: NCT04830137
Phase: Phase 1
Status: Recruiting
Date: 2025-03-13
REF
- Discovery and Preclinical Pharmacology of NX-2127, an Orally Bioavailable Degrader of Bruton’s Tyrosine Kinase with Immunomodulatory Activity for the Treatment of Patients with B Cell MalignanciesPublication Name: Journal of Medicinal ChemistryPublication Date: 2024-02-01PMID: 38300987DOI: 10.1021/acs.jmedchem.3c01007
- The IUPHAR Guide to Immunopharmacology: connecting immunology and pharmacologyPublication Name: ImmunologyPublication Date: 2020-03-02PMCID: PMC7160657PMID: 32020584DOI: 10.1111/imm.13175
- Effect of osmotic pressure, ionic strength and dibutyryl cyclic adenosine monophosphate on the adhesion of hen erythrocytesPublication Name: Blut Zeitschrift für die gesamte BlutforschungPublication Date: 1976-07PMID: 10025DOI: 10.1007/bf01005212
SYN
compound 28 Journal of Medicinal ChemistryPublication Date: 2024-02-01PMID: 38300987DOI: 10.1021/acs.jmedchem.3c01007
SYN
WO2021219070A1
PAT
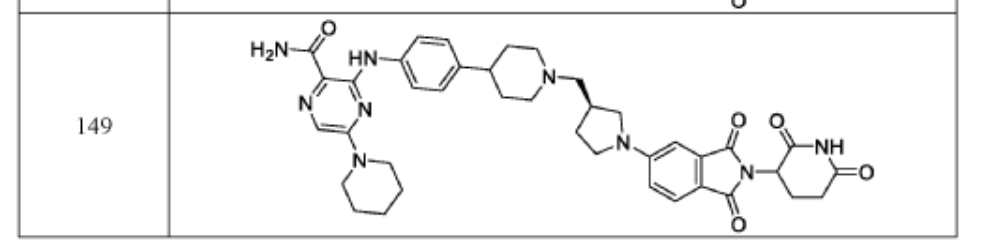
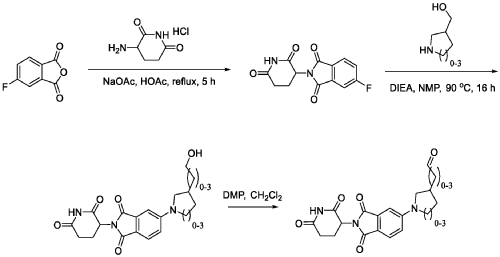
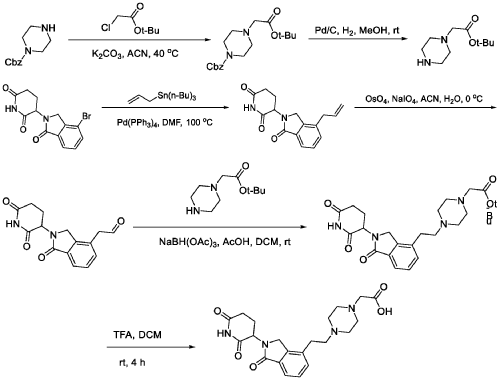
PAT
- Bifunctional compounds for degrading btk via ubiquitin proteosome pathwayPublication Number: US-2023227471-A1Priority Date: 2019-12-04
- Bifunctional compounds for degrading btk via ubiquitin proteosome pathwayPublication Number: WO-2021113557-A1Priority Date: 2019-12-04
- Bifunctional compounds for degrading btk via ubiquitin proteosome pathwayPublication Number: US-2023029378-A1Priority Date: 2018-10-15
- Bifunctional compounds for degrading btk via ubiquitin proteosome pathwayPublication Number: WO-2020081450-A1Priority Date: 2018-10-15
- Bifunctional compounds for degrading BTK via ubiquitin proteosome pathwayPublication Number: US-11479556-B1Priority Date: 2018-10-15Grant Date: 2022-10-25
- Btk reducing molecules for treatment of cancers and immune system disordersPublication Number: WO-2023235691-A1Priority Date: 2022-05-31
- Piperidinylpyrazine-carboxamide compounds for treating and preventing cancer and for degrading btkPublication Number: US-2023149416-A1Priority Date: 2021-10-26
- Piperidinylpyrazine-carboxamide compounds for treating and preventing cancer and for degrading btkPublication Number: WO-2023076303-A1Priority Date: 2021-10-26
- Bifunctional compounds for degrading BTK via ubiquitin proteosome pathwayPublication Number: US-11820781-B2Priority Date: 2019-12-04Grant Date: 2023-11-21
- Bifunctional compounds for degrading btk via ubiquitin proteosome pathwayPublication Number: US-2021198280-A1Priority Date: 2019-12-04
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- Salvaris RT, Brennan J, Lewis KL (February 2025). “BTK Is the Target That Keeps on Giving: A Review of BTK-Degrader Drug Development, Clinical Data, and Future Directions in CLL”. Cancers. 17 (3): 557. doi:10.3390/cancers17030557. PMC 11817010. PMID 39941922.
| Clinical data | |
|---|---|
| Other names | NX-2127 |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2416131-46-7 |
| PubChem CID | 146559796 |
| ChemSpider | 128922006 |
| UNII | LSC67HA8DE |
| Chemical and physical data | |
| Formula | C39H45N9O5 |
| Molar mass | 719.847 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
//////////zelebrudomide, anax lab, protein degrader, antineoplastic, NX 2127, LSC67HA8DE, NX-2127, BTK Degrader NX-2127
Veonetinib
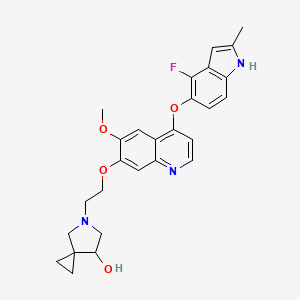
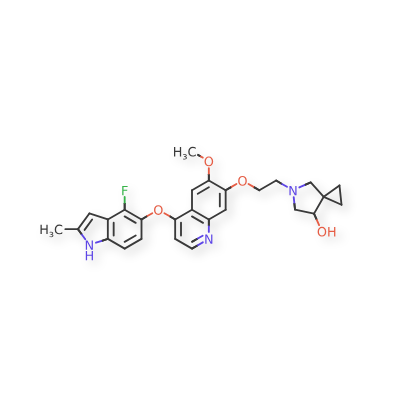
Veonetinib
👉CAS 1210828-09-3
MF C27H28FN3O4 MW 477.5 g/mol
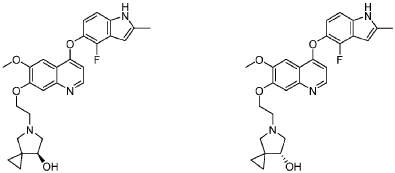
5-[2-[4-[(4-fluoro-2-methyl-1H-indol-5-yl)oxy]-6-methoxyquinolin-7-yl]oxyethyl]-5-azaspiro[2.4]heptan-7-ol
5-AZASPIRO(2.4)HEPTAN-7-OL, 5-(2-((4-((4-FLUORO-2-METHYL-1H-INDOL-5-YL)OXY)-6-METHOXY-7-QUINOLINYL)OXY)ETHYL)-
5-(2-(4-(4-fluoro-2-methyl-1H-indol-5-yloxy)-6-methoxyquinolin-7-yloxy)ethyl)-5-azaspiro[2.4]-heptan-7-ol
(7RS)-5-[2-({4-[(4-fluoro-2-methyl-1H-indol-5-yl)oxy]-6-methoxyquinolin7-yl}oxy)ethyl]-5-azaspiro[2.4]heptan-7-ol
tyrosine kinase inhibitor, antineoplastic, U7PA8S6XGJ
👉SYN
Example 3
5-(2-(4-(4-fluoro-2-methyl-lH-indol-5-yloxy)-6-methoxyquinolin-7-yloxy)ethyl)-5-azaspiro[2.4]-heptan-7-ol
The above product from Example 2 (75 mg) was dissolved into MeOH (8 ml) and stirred at RT.
NaBH4 (75 mg) was added to the reaction and stirred at RT for 30 min. The reaction was evaporated and purified by column chromatography to give title compound (60 mg). Mass: (M + 1), 478
Patent Scope
- Covers:
- Quinoline–indole kinase inhibitors
- VEGFR / angiogenesis targets
- Broad Markush structures
Fragment A: Indole Phenol
4-fluoro-2-methyl-1H-indol-5-ol
Typical Preparation
- Fischer indole synthesis or substituted aniline cyclization
- Fluorination / directed substitution
- Hydroxyl introduction at C-5
Fragment B: Quinoline Electrophile
4-chloro-6-methoxy-7-(leaving group) quinoline
Typical Route
- Start from 6-methoxyaniline
- Skraup / Doebner–Miller → quinoline core
- Chlorination at C-4
- Functionalization at C-7 (OH or halide)
Fragment C: Chiral Spiro Amine
(R)-5-azaspiro[2.4]heptan-7-ol
- Usually from:
- Chiral pool OR
- Resolution of racemate
- Important: defines final stereochemistry
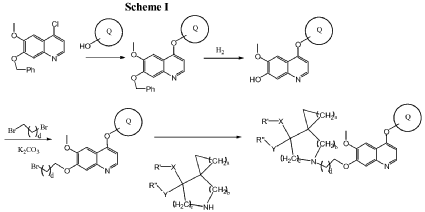
4. STEP-BY-STEP SYNTHESIS (PATENT-ALIGNED)
Step 1: Indole–Quinoline Ether Formation
Reaction: SNAr / Ullmann-type coupling
Indole phenol + 4-chloroquinoline → aryl ether
Conditions
- Base: K2CO3 / Cs2CO3
- Solvent: DMF / DMSO
- Temp: 80–120°C
Forms:
Indole–O–quinoline core
Step 2: Introduction of Linker (C-7 substitution)
If quinoline has OH:
Quinoline–OH + Br–CH2–CH2–X → O–CH2CH2–X
If halide:Direct alkylation
Conditions
- Base: NaH / K2CO3
- Solvent: DMF
- Temp: 50–90°C
Product:
Quinoline–O–CH2CH2–X
Step 3: Coupling with Spiro Amine
Quinoline–O–CH2CH2–X + spiro amine → final amine linkage
Reaction Type
- SN2 substitution
Conditions
- Base: DIPEA / Et3N
- Solvent: ACN / DMF
- Temp: 50–80°C
Step 4: Final Deprotection / Purification
- Remove protecting groups (if any)
- Chiral purity control
- Crystallization
Step 1: Preparation of Indole–Quinoline Ether
Starting materials:
- 4-fluoro-2-methyl-1H-indol-5-ol
→ 1.00 equiv (e.g., 5.0 g, ~30 mmol) - 4-chloro-6-methoxyquinoline
→ 1.10 equiv (~33 mmol)
Reagents:
- Potassium carbonate (K₂CO₃) → 2.0 equiv (~60 mmol)
- Solvent: DMF (50–60 mL)
Procedure:
- Charge indole phenol and K₂CO₃ in DMF under nitrogen.
- Add 4-chloroquinoline portionwise.
- Heat to 100–110°C.
- Stir for 8–12 h.
Workup:
- Cool to RT
- Pour into water (200 mL)
- Extract with EtOAc (3×)
- Wash with brine, dry (Na₂SO₄)
- Concentrate
Purification:
- Silica gel chromatography (EtOAc/hexane)
Yield: ~70–80%
Product: Indole–quinoline ether intermediate
Step 2: Installation of Ethylene Linker
Starting material: Step 1 product (~25 mmol)
Reagents:
- 1,2-dibromoethane → 1.5–2.0 equiv
- Base: K₂CO₃ → 2 equiv
- Solvent: DMF (40 mL)
Procedure:
- Dissolve intermediate in DMF
- Add K₂CO₃
- Add dibromoethane
- Heat to 80–90°C for 6–8 h
Workup:
- Pour into water
- Extract with EtOAc
- Dry and concentrate
Product: Quinoline–O–CH₂CH₂–Br
Yield: ~65–75%
Step 3: Coupling with Chiral Spiro Amine
Starting materials:
- Bromo intermediate → 1.0 equiv (~15–20 mmol)
- (R)-5-azaspiro[2.4]heptan-7-ol → 1.2 equiv
Reagents:
- DIPEA or Et₃N → 2 equiv
- Solvent: Acetonitrile or DMF (30–40 mL)
Procedure:
- Combine bromo intermediate and amine in solvent
- Add DIPEA
- Heat to 60–70°C
- Stir 12–16 h
Workup:
- Remove solvent
- Dissolve in EtOAc
- Wash with water + brine
- Dry and concentrate
Yield: ~70–85%
Step 4: Final Purification
Purification options:
- Silica chromatography OR
- Recrystallization (EtOAc/hexane or IPA)
Optional:
- Convert to pharmaceutically acceptable salt
Final Yield (overall): ~35–45%
PAT
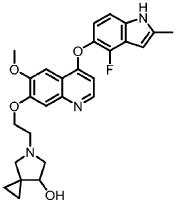
EXAMPLE 1
4-(4-Fluoro-2-methyl-1H-indol-5-yloxy)-6-methoxy-7-[2-(5,8-Dioxa-10-azadispiro[2.0.4.3]-undecane)ethoxy]quinoline
Preparation of 4-(4-fluoro-2-methyl-1H-indol-5-yloxy)-6-methoxy-7-benzyloxyquinoline
Method A:
Method B:
Preparation of Title Compound
Method C:
Method D:
EXAMPLE 2
5-(2-(4-(4-Fluoro-2-methyl-1H-indol-5-yloxy)-6-methoxyquinolin-7-yloxy)ethyl)-5-azaspiro[2.4]-heptan-7-one
EXAMPLE 3
5-(2-(4-(4-fluoro-2-methyl-1H-indol-5-yloxy)-6-methoxyquinolin-7-yloxy)ethyl)-5-azaspiro[2.4]-heptan-7-ol
PAT
PAT
CN201710900497.6 CN201610649732.2 → leads to US10689361B2
PAT
- Compounds as kinase inhibitorsPublication Number: CA-2733250-CPriority Date: 2008-08-19Grant Date: 2016-06-21
- Compounds that act as kinase inhibitorsPublication Number: ES-2617678-T3Priority Date: 2008-08-19Grant Date: 2017-06-19
- Compounds as kinase inhibitorsPublication Number: WO-2010021918-A1Priority Date: 2008-08-19
- Compounds as kinase inhibitorsPublication Number: EP-2312950-A1Priority Date: 2008-08-19
- Compounds as kinase inhibitorsPublication Number: US-8211911-B2Priority Date: 2008-08-19Grant Date: 2012-07-03
- Compounds as kinase inhibitorsPublication Number: JP-2012500269-APriority Date: 2008-08-19
- Compounds as kinase inhibitorsPublication Number: KR-20110044749-APriority Date: 2008-08-19
- Compounds as kinase inhibitorsPublication Number: EP-2312950-B1Priority Date: 2008-08-19Grant Date: 2016-11-30
- Biological activities of 5-(2-(4-(4-fluoro-2-methyl-1h-indol-5-yloxy)-6-methoxyquinolin-7-yloxy)ethyl)-5-azaspiro[2.4]-heptan-7-ol crystalline, phosphoric acid salt and its enantiomersPublication Number: US-2023399313-A1Priority Date: 2022-06-10
- Btological activities of 5-(2-(4-(4-fluoro-2-methyl-1h-indol-5- yloxy)-6-methoxyquinolin-7-yloxy)ethyl)-5-azaspiro[2.4]-heptan- 7-ol crystalline, phosphoric acid salt and its enantiomersPublication Number: WO-2023239945-A1Priority Date: 2022-06-10
- Btological activities of 5-(2-(4-(4-fluoro-2-methyl-1h-indol-5- yloxy)-6-methoxyquinolin-7-yloxy)ethyl)-5-azaspiro[2.4]-heptan- 7-ol crystalline, phosphoric acid salt and its enantiomersPublication Number: EP-4536652-A1Priority Date: 2022-06-10
- Compounds as kinase inhibitorsPublication Number: CA-2733250-A1Priority Date: 2008-08-19
- Compounds As Kinase InhibitorsPublication Number: US-2010048599-A1Priority Date: 2008-08-19
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

REF
//////////veonetinib, ANAX LAB, tyrosine kinase inhibitor, antineoplastic, U7PA8S6XGJ
Soxataltinib
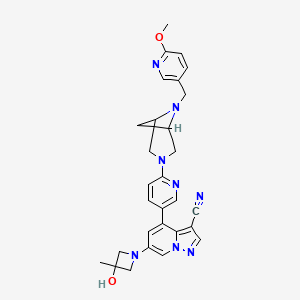
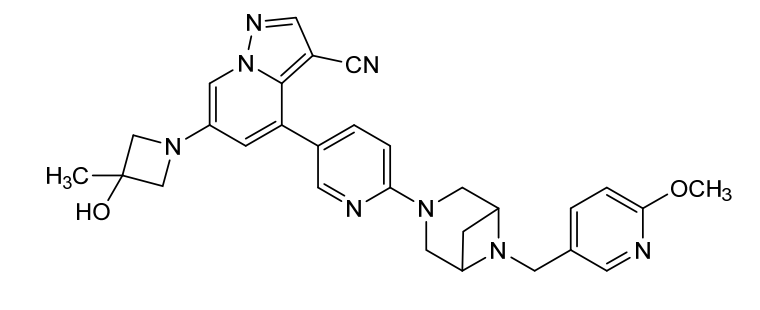
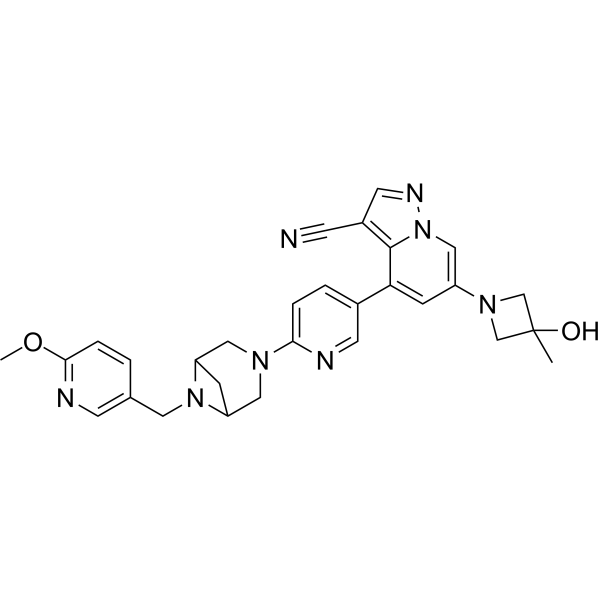
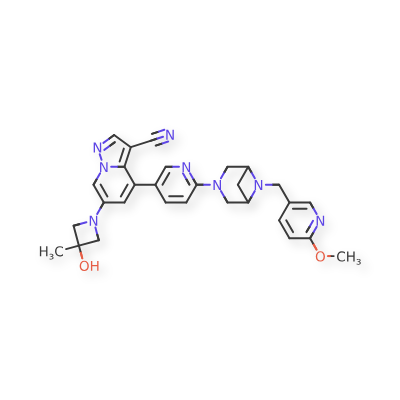
Soxataltinib
CAS 2546116-88-3
MF C29H30N8O2 MW 522.60
6-(3-hydroxy-3-methylazetidin-1-yl)-4-(6-{6-[(6-methoxypyridin-3-yl)methyl]-3,6-diazabicyclo[3.1.1]heptan-3-yl}pyridin-3-yl)pyrazolo[1,5-a]pyridine-3-carbonitrile
| Pyrazolo[1,5-a]pyridine-3-carbonitrile, 6-(3-hydroxy-3-methyl-1-azetidinyl)-4-[6-[6-[(6-methoxy-3-pyridinyl)methyl]-3,6-diazabicyclo[3.1.1]hept-3-yl]-3-pyridinyl]- |
6-(3-hydroxy-3-methylazetidin-1-yl)-4-(6-{6-[(6-methoxypyridin-3-yl)methyl]-3,6-diazabicyclo[3.1.1]heptan-3-yl}pyridin-3-yl)pyrazolo[1,5-a]pyridine-3-carbonitrile
RET-kinase inhibitor, antineoplastic, HS-10365, HS 10365, AZ4Q643U3D
Soxataltinib (example 7) is a potent inhibitor of RET-kinase , with the IC 50of 0.601 nM. Soxataltinib plays an important role in
cancer research.
Discovery and Development
- Soxataltinib corresponds to Example 114 in a patent [WO2020228756]describing pyrazolo[1,5-a]pyridine carbonitrile RET inhibitors.
- It is believed to correspond to HS-10365, a RET inhibitor developed by Hansoh Pharma (structure disclosed via patent).
Drug class comparison:
| Drug | Company | Type |
|---|---|---|
| Selpercatinib | Eli Lilly | 1st-gen selective RET inhibitor |
| Pralsetinib | Blueprint | selective RET inhibitor |
| Soxataltinib | Hansoh | next-gen RET inhibitor |
Patent Family (Major Members)
Typical family members include:
| Patent | Jurisdiction |
|---|---|
| WO2020228756 | WIPO |
| CN112209925 | China |
| US continuation filings | USA |
| EP equivalents | Europe |
One Chinese patent describes the preparation of piperazine-containing pyrazolopyridine RET inhibitors, including Soxataltinib analogues
SYN
SYN
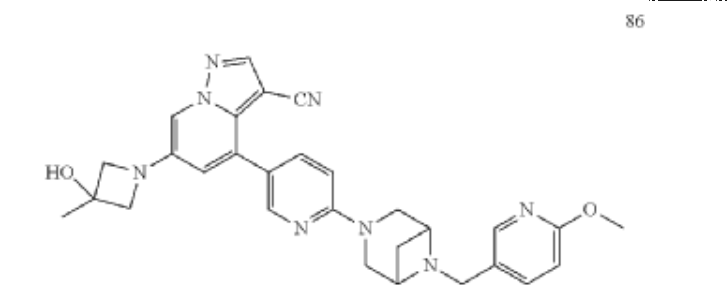
Example 32
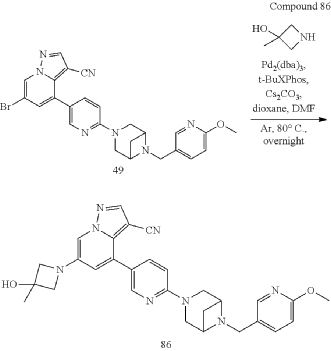
| To a 25 mL sealed tube were added successively 49 (52 mg, 0.1 mmol), Pd 2(dba) 3 (5.5 mg, 0.006 mmol), t-BuXPhos (8.4 mg, 0.02 mmol), 3-methyl-3-azetidinol (26 mg, 0.3 mmol), Cs 2CO 3 (65 mg, 0.2 mmol), 1,4-dioxane (3 mL) and DMF (1 mL). The mixture was stirred at 80° C. overnight under Ar, and TLC monitoring showed no starting material 49 remained. The mixture was cooled to room temperature, and 10 mL of water was added. The mixture was stirred for 10 min, and a yellow solid precipitated. The solid was collected by filtration, dried and purified by column chromatography to give product 86 (34 mg, 65% yield). |
SYN
[1913]6-(3-hydroxy-3-methylacetidin-1-yl)-4-(6-(6-((6-methoxypyridin-3-yl)methyl)-3,6-diazabicyclo[3.1.1]heptane-3-yl)pyridin-3-yl)pyrazolo[1,5-a]pyridin-3-carboxynitrile
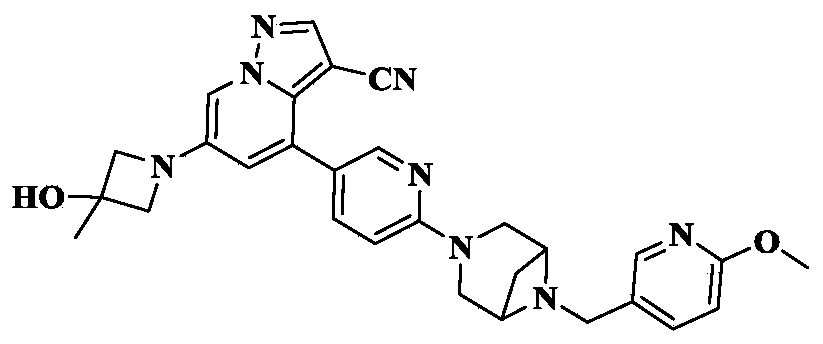
Using 3-methylacetidin-3-ol as a raw material, in the first step of Reference Example 110, 6-(3-hydroxy-3-methylacetidin-1-yl)-4-(6-(6-((6-methoxypyridin-3-yl)methyl)-3,6-diazabicyclo[3.1.1]heptane-3-yl)pyridin-3-yl)pyrazolo[1,5-a]pyridin-3-carboxynitrile was obtained.
[1916]MS m/z(ESI):523.3[M+H]
[1917]
1H NMR(400MHz,CDCl 3)δ8.40(s,1H),8.15(s,3H),7.82(d,J=7.4Hz,1H),7.74(s,1H),6.80(d,J=8.4Hz,1H),6.75(d,J=1.7Hz,1H),6.72(d,J=8.8Hz,1H),4.21(s,2H),4.01(s,2H),3.93-3.92(m,7H),3.84(d,J=7.3Hz,4H),1.68(s,3H).
PAT
- Inhibitor containing dihecyclic derivatives, its preparation method and applicationPublication Number: CN-112368283-BPriority Date: 2019-05-14Grant Date: 2023-02-17
- Inhibitor containing dihecyclic derivatives, its preparation method and applicationPublication Number: CN-115974897-APriority Date: 2019-05-14
- Inhibitor containing dihecyclic derivatives, its preparation method and applicationPublication Number: CN-116444515-APriority Date: 2019-05-14
- Inhibitor containing bicyclic derivative, preparation method therefor and use thereofPublication Number: WO-2020228756-A1Priority Date: 2019-05-14
- Inhibitor containing bicyclic derivative, preparation method therefor and use thereofPublication Number: EP-3971187-B1Priority Date: 2019-05-14Grant Date: 2025-05-07
- Ret selective inhibitor, preparation method therefor and use thereofPublication Number: WO-2021008455-A1Priority Date: 2019-07-12
- RET selective inhibitor and preparation method and use thereofPublication Number: CN-112209925-APriority Date: 2019-07-12
- RET selective inhibitors and methods for their preparation and usePublication Number: KR-102735956-B1Priority Date: 2019-07-12Grant Date: 2024-11-28
- RET selective inhibitors and preparation methods and uses thereofPublication Number: CN-114072404-BPriority Date: 2019-07-12Grant Date: 2023-09-15
- Inhibitor containing bicyclic derivative, preparation method therefor and use thereofPublication Number: US-2022259201-A1Priority Date: 2019-05-14
- Heteroaromatic ring compound as ret kinase inhibitor, and preparation and use thereofPublication Number: US-2023322769-A1Priority Date: 2020-08-20
- 3,6 diazabicyclo[3.1.1]heptane derivatives as RET kinase inhibitorsPublication Number: CN-112778337-BPriority Date: 2019-11-08Grant Date: 2023-09-26
- Ret selective inhibitor, preparation method therefor and use thereofPublication Number: EP-3998265-A1Priority Date: 2019-07-12
- RET selective inhibitor and preparation method and use thereofPublication Number: CN-117865952-APriority Date: 2019-07-12
- Ret selective inhibitor, preparation method therefor and use thereofPublication Number: US-2022289740-A1Priority Date: 2019-07-12
- Crystal form of free base of inhibitor containing bicyclic ring derivative and preparation method and application of crystal formPublication Number: US-2023406865-A1Priority Date: 2020-11-13
- Crystal form of free base of inhibitor containing bicyclic ring derivative and preparation method and application of crystal formPublication Number: EP-4245757-A1Priority Date: 2020-11-13
- Crystal form of free base of inhibitor containing bicyclic ring derivative and preparation method and application of crystal formPublication Number: WO-2022100738-A1Priority Date: 2020-11-13
- Crystal form of inhibitor free base including bicyclic derivatives, preparation method and use thereofPublication Number: KR-20230107271-APriority Date: 2020-11-13
- Heteroaromatic ring compound as ret kinase inhibitor, and preparation and use thereofPublication Number: EP-4201936-A1Priority Date: 2020-08-20
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

/////////soxataltinib, ANAX, RET-kinase inhibitor, antineoplastic, HS-10365, HS 10365, AZ4Q643U3D
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma
Ranosidenib
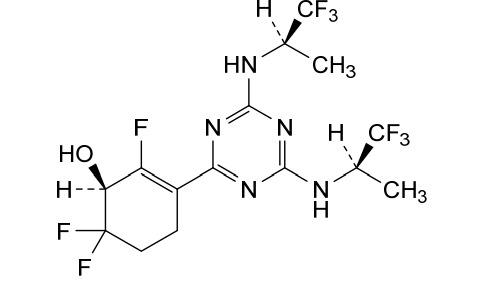
Ranosidenib
CAS 2301974-60-5
MF C15H16F9N5O MW 453.31 g/mol
(1S)-3-[4,6-bis[[(2R)-1,1,1-trifluoropropan-2-yl]amino]-1,3,5-triazin-2-yl]-2,6,6-trifluorocyclohex-2-en-1-ol
(1S)-3-(4,6-bis{[(2R)-1,1,1-trifluoropropan-2-yl]amino}-1,3,5-triazin-2-yl)-2,6,6-trifluorocyclohex-2-en-1-ol
isocitrate dehydrogenase (IDH) inhibitor, antineoplastic, [14C] HMPL-306, HMPL 306, PC64OXS2C2
- OriginatorHutchison MediPharma
- DeveloperHUTCHMED
- ClassAntineoplastics; Small molecules
- Mechanism of ActionIsocitrate dehydrogenase 1 inhibitors; Isocitrate dehydrogenase 2 inhibitors
- Phase IIIAcute myeloid leukaemia
- No development reportedHaematological malignancies; Solid tumours
- 28 Sep 2025No recent reports of development identified for phase-I development in Haematological-malignancies(Late-stage disease, Second-line therapy or greater) in Spain (PO)
- 28 Sep 2025No recent reports of development identified for phase-I development in Haematological-malignancies(Late-stage disease, Second-line therapy or greater) in USA (PO)
- 19 Sep 2025No development reported – Phase-I for Solid tumours (Late-stage disease, Metastatic disease, Second-line therapy or greater) in USA (PO)
Ranosidenib is a small molecule drug. Ranosidenib is under investigation in clinical trial NCT06387069 (A Study to Evaluate HMPL-306 in Patients With IDH1- and IDH2-mutated Acute Myeloid Leukemia). Ranosidenib has a monoisotopic molecular weight of 453.12 Da.
Ranosidenib is an orally bioavailable inhibitor of mutated forms of both isocitrate dehydrogenase type 1 (IDH1, IDH1 [NADP+] soluble) in the cytoplasm and type 2 (IDH2, isocitrate dehydrogenase [NADP+], mitochondrial) in the mitochondria, with potential antineoplastic activity. Upon administration, ranosidenib specifically targets and inhibits mutant forms of IDH1 and IDH2, thereby inhibiting the formation of the oncometabolite 2-hydroxyglutarate (2HG) from alpha-ketoglutarate (a-KG). This prevents 2HG-mediated signaling and leads to both an induction of cellular differentiation and an inhibition of cellular proliferation in tumor cells expressing IDH mutations. IDH1 and 2, metabolic enzymes that catalyze the conversion of isocitrate into a-KG, play key roles in energy production and are mutated in a variety of cancer cell types. Mutant forms of IDH1 and 2 catalyze the formation of 2HG and drive cancer growth by blocking cellular differentiation and inducing cellular proliferation.
A Study of HMPL-306 in Advanced Hematological Malignancies With mIDHCTID: NCT04764474Phase: Phase 1Status: TerminatedDate: 2026-01-29
A Study of HMPL-306 in Advanced Solid Tumors With IDH MutationsCTID: NCT04762602Phase: Phase 1Status: TerminatedDate: 2025-09-16
A Study to Evaluate HMPL-306 in Patients With IDH1or IDH2-mutated Acute Myeloid LeukemiaCTID: NCT06387069Phase: Phase 3Status: RecruitingDate: 2025-08-14
Phase I Study of HMPL-306 for the Treatment of Gliomas With IDH1 and/or IDH2 MutationsCTID: NCT07025018Phase: Phase 1Status: RecruitingDate: 2025-08-01
A Study of HMPL-306 in Patients With IDH1 and/or IDH2 Mutation of Relapsed/Refractory Myeloid Leukemia/NeoplasmsCTID: NCT04272957Phase: Phase 1Status: Unknown statusDate: 2020-06-16
SYN
https://pubs.acs.org/doi/10.1021/acsmedchemlett.4c00625
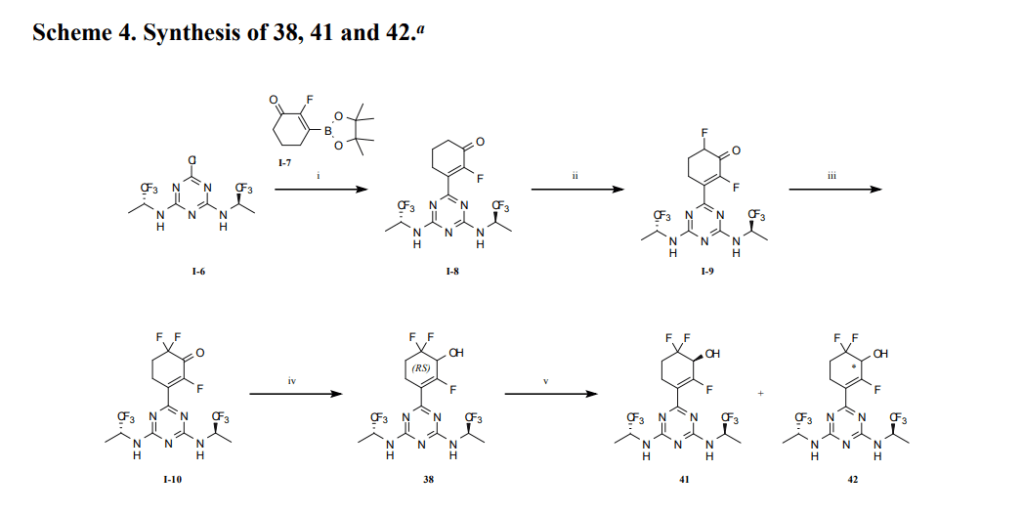
aReagents and conditions: (i) Na2PdCl4, DTBPPS, K2CO3, MeCN, H2O, 60 ℃; (ii) TBSOTf, Et3N,
DCM, 0~5 ℃; Selectfluor®, MeCN, 0~5 ℃; (iii) TBSOTf, Et3N, DCM, 0~5 ℃; Selectfluor®,
MeCN, 0~5 ℃; (iv) NaBH4, CeCl3·7H2O, EtOH, 0 ℃; (v) SFC separation.
Pat
Cycloolefin substituted heteroaromatic compounds and their use
Publication Number: US-2021363115-A2
Priority Date: 2017-09-07
PAT
Intermediate I-3
6-Chloro-N2,N4-bis((R)-1,1,1-trifluoropropan-2-yl)-1,3,5-triazine-2,4-diamine
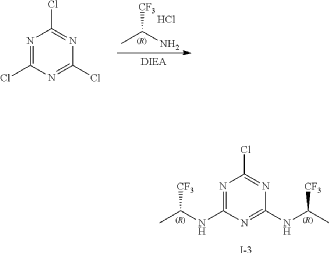
At 0° C., to a flask were added 1,4-dioxane (50 mL), 2,4,6-trichloro-1,3,5-triazine (1.84 g, 10 mmo), (R)-1,1,1-trifluoropropan-2-amine hydrochloride (2.99 g, 20 mmol) and DIEA (5.17 g, 40 mmol). The reaction was heated to 60° C. and stirred for 4 hours. After the reaction was completed, the mixture was condensed and purified by flash column chromatography (eluting with gradient water/MeOH=100:0-0:100) to give Intermediate I-3 as yellow solid (2.50 g, yield: 74%). MS (m/z): 338.0 [M+H] +
Compounds 197 and 198
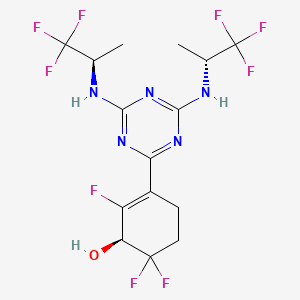
3-(4,6-Bis(((R)-1,1,1-trifluoropropan-2-yl)amino)-1,3,5-triazin-2-yl)-2,6,6-trifluorocyclohex-2-en-1-ol, optically pure diastereoisomers
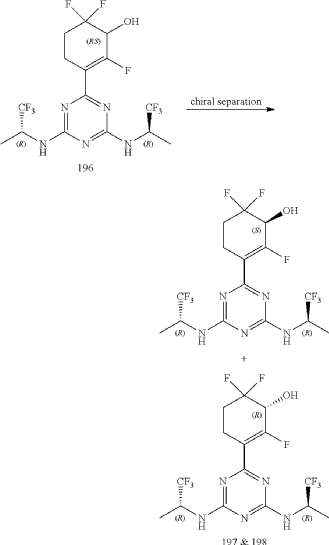
| The Compound 196 was resolved by chiral HPLC to provide a pair of optically pure diastereoisomers, Compounds 197 and 198 (Chiral HPLC conditions: Preparation instrument: Shimadzu LC-10AD vp; Column: Daicel AD-H(250 mm*30 mm, 5 um); mobile phase: n-heptane/isopropanol=90/10; flow rate: 40 mL/min; column temperature: 40° C.). The first eluent (RT=4.203 min) was concentrated and purified by flash column chromatography (eluting with gradient PE/EA=100:0-0:100) to give a compound named as Compound 197, de %=99.27%, MS (m/z): 454.1 [M+1] +. The second eluent (RT=5.906 min) was concentrated and purified by flash column chromatography (eluting with gradient PE/EA=100:0-0:100) to give a compound named as Compound 198, de %=97.82%, MS (m/z): 454.2 [M+1] +. |
| Compound 197: 1H NMR (400 MHz, CD 3OD): δ 5.00-4.86 (m, 2H), 4.36-4.17 (m, 1H), 2.80-2.65 (m, 1H), 2.58-2.42 (m, 1H), 2.25-2.05 (m, 2H), 1.37-1.31 (m, 6H). |
| Compound 198: 1H NMR (400 MHz, CD 3OD): δ 5.00-4.86 (m, 2H), 4.36-4.17 (m, 1H), 2.80-2.65 (m, 1H), 2.58-2.42 (m, 1H), 2.25-2.05 (m, 2H), 1.37-1.31 (m, 6H). |



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

/////////ranosidenib, isocitrate dehydrogenase (IDH) inhibitor, antineoplastic, [14C] HMPL-306, HMPL 306, PC64OXS2C2
Pebezertinib
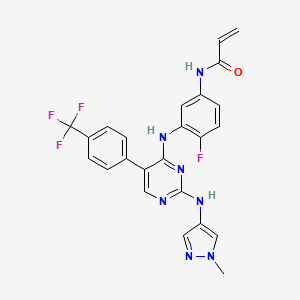
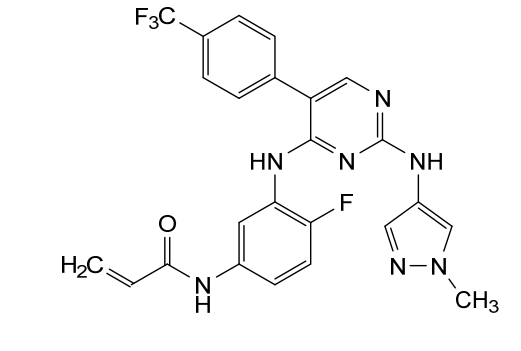
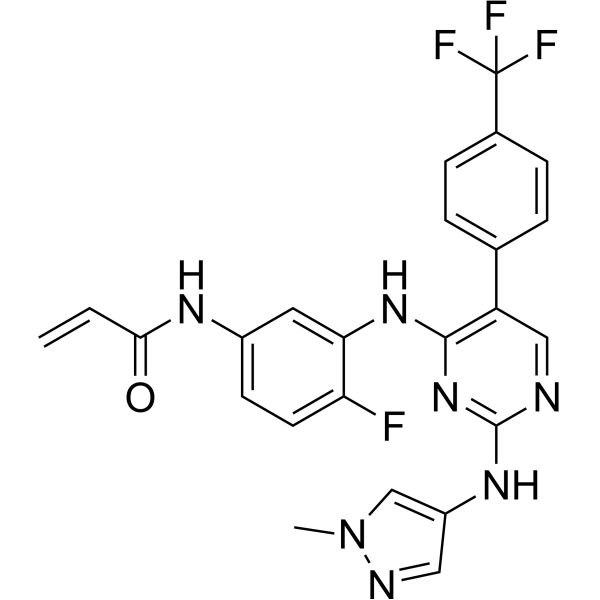
Pebezertinib
CAS 2769954-39-2
MF C24H19F4N7O MW 497.4 g/mol
N-[4-fluoro-3-[[2-[(1-methylpyrazol-4-yl)amino]-5-[4-(trifluoromethyl)phenyl]pyrimidin-4-yl]amino]phenyl]prop-2-enamide
N-[4-fluoro-3-({2-[(1-methyl-1H-pyrazol-4-yl)amino]-5-[4-(trifluoromethyl)phenyl]pyrimidin-4-yl}amino)phenyl]prop-2-enamide
epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor, antineoplastic, BLU 451, BLU 203139, G8G5AU5GLJ, LNG 451
Pebezertinib is a small molecule drug. The usage of the INN stem ‘-ertinib’ in the name indicates that Pebezertinib is a epidermal growth factor receptor (EGFR) inhibitor. Pebezertinib is under investigation in clinical trial NCT05241873 ((Concerto) Study of BLU-451 in Advanced Cancers With EGFR Exon 20 Insertion Mutations). Pebezertinib has a monoisotopic molecular weight of 497.16 Da.
Pebezertinib is an orally bioavailable, central nervous system (CNS) penetrating, mutant-selective covalent inhibitor of epidermal growth factor receptor (EGFR) exon 20 insertion (Ex20ins) activating mutations, with potential antineoplastic activity. Upon oral administration, pebezertinib selectively targets, irreversibly binds to and inhibits the activity of EGFR Ex20ins and some other oncogenic point mutations. This prevents EGFR Ex20ins-mediated signaling. This may induce cell death and inhibit tumor growth in EGFR Ex20ins-overexpressing tumor cells. EGFR, a receptor tyrosine kinase mutated in many tumors, plays a key role in tumor cell proliferation and tumor vascularization. Pebezertinib is able to penetrate the blood-brain-barrier (BBB) and may therefore exert its activity against EGFR Ex20ins-driven CNS primary tumors and CNS metastases. Pebezertinib does not inhibit the activity of wild-type (WT) EGFR. EGFR Ex20ins are oncogenic driver mutations that constitutively upregulate kinase activity.
(Concerto) Study of BLU-451 in Advanced Cancers With EGFR Exon 20 Insertion Mutations
CTID: NCT05241873
Phase: Phase 1
Status: Terminated
Date: 2025-02-10
Conditions: Lung Neoplasm Malignant; Carcinoma, Non-Small-Cell Lung; Respiratory Tract Neoplasms; Neoplasms; Neoplasms by Site; Lung Diseases; Respiratory Tract Disease; Carcinoma, Bronchogenic; Bronchial Neoplasms; Adenocarcinoma; Carcinoma; Neoplasms by Histologic Type; EGFR Exon 20 Mutation; EGFR Exon 20 Insertion Mutation; EGFR Activating Mutation; Antineoplastic Agents; Metastatic Lung Cancer; Brain Metastases; EGFR-mutated NSCLC; EGFR Atypical Mutations, Including G719X and L861Q
Interventions: Pemetrexed
Linked Compound CID: 426756; 135410875; 10339178; 163280903
SYN
Scheme 21: Synthesis of N-(4-fluoro-3-((2-((1-methyl-1H-pyrazol-4-yl)amino)-5-(4-(trifluoromethyl)phenyl)pyrimidin-4-yl)amino)phenyl)acrylamide (Compound 37):
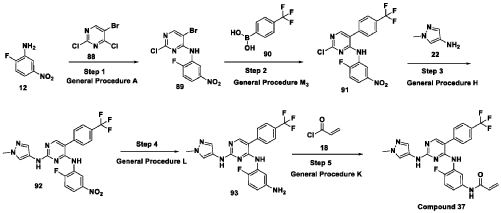
Step 1: Synthesis of 5-bromo-2-chloro-N-(2-fluoro-5-nitrophenyl)pyrimidin-4-amine (89):
[0286] Title compound was prepared in a manner substantially similar to procedure mentioned in General Procedure A. The crude was purified by combiflash eluted with 40% ethyl acetate in hexane to get (89) as pale yellow solid (1.3 g, Yield: 44.24 %). MS: [M+H]+ 346.97.
Step 2: Synthesis of 2-chloro-N-(2-fluoro-5-nitrophenyl)-5-(4-(trifluoromethyl)phenyl)pyrimidin-4-amine (91):
[0287] Title compound was prepared in a manner substantially similar to procedure mentioned in General Procedure M3. The crude was purified by combiflash eluted with 35% ethyl acetate in hexane to get desired product (91) as light yellow solid (700 mg; Yield: 50.12%). MS:
[M+H]+ 413.10
Step 3: Synthesis of N4-(2-fluoro-5-nitrophenyl)-N2-(1-methyl-1H-pyrazol-4-yl)-5-(4-(trifluoromethyl)phenyl)pyrimidine-2,4-diamine (92):
[0288] Title compound was prepared in a manner substantially similar to procedure mentioned in General Procedure H. The crude was purified by combiflash eluted with 1% methanol in dichloromethane to get desired product (92) as pale yellow solid (500 mg; Yield: 70.24%). MS:
[M+H]+ 474.09
Step 4: Synthesis of N4-(5-amino-2-fluorophenyl)-N2-(1-methyl-1H-pyrazol-4-yl)-5-(4-(trifluoromethyl)phenyl)pyrimidine-2,4-diamine (93):
[0289] Title compound was prepared in a manner substantially similar to procedure mentioned in General Procedure L to get (93) as semi solid (350 mg; Yield: 74.78%). MS: [M+H]+ 444.11
Step 5: Synthesis of N-(4-fluoro-3-((2-((1-methyl-1H-pyrazol-4-yl)amino)-5-(4-(trifluoromethyl)phenyl)pyrimidin-4-yl)amino)phenyl)acrylamide (Compound 37):
[0290] Title compound was prepared in a manner substantially similar to procedure mentioned in General Procedure K. The crude was purified by Prep HPLC to get Compound 37 as off white solid (30 mg, Yield: 13.33%).1H NMR (400 MHz, DMSO-d6): δ 10.21 (bs, 1H), 9.24 (bs, 1H), 8.53 (bs, 1H), 7.99 (s, 1H), 7.71-7.81 (m, 5H), 7.57 (s, 1H), 7.08-7.16 (m, 3H), 6.37-6.44 (m, 1H), 6.21-6.26 (m, 1H), 5.74 (d, J = 8.4 Hz, 1H), 3.54 (s, 3H). LCMS: [M+H]+ 498.35.
SYN
International Patent Application No. PCT/US2021/057472, the entire teachings of which are incorporated herein by reference, discloses selective inhibitors of EGFR, including exon 20 mutant proteins, which can be used to treat various cancers. The structure of one of the inhibitors disclosed in PCT Patent Application No. PCT/US2021/057472, referred to
herein as “Compound (I)” is shown below:
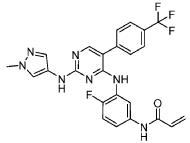
Example 1 : Preparation of Compound (I)
Synthesis of N-(4-fluoro-3-((2-((l-methyl-lH-pyrazol-4-yl)amino)-5-(4-(trifluoro methyl)phenyl)pyrimidin-4-yl)amino)phenyl)acrylamide (Compound I):
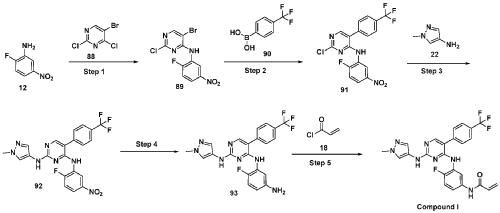
Step 1 : Synthesis of 5-bromo-2-chloro-N-(2-fluoro-5-nitrophenyl)pyrimidin-4-amine (89):
To an ice cold solution of 2-fluoro-5-nitroaniline (12) (1.0 eq) in tetrahydrofuran was added sodium hydride (60% dispersion in mineral oil, 3.0 eq) portion-wise. The resulting reaction mixture was stirred at room temperature for 30 minutes and followed by the addition of 2, 4-di chi oro-5 -bromopyrimidine (88) (1.0 eq). The resulting reaction mixture was heated at 60 °C for 16 hours. After completion (TLC monitoring), quenched with ice, extracted with ethyl acetate (3 times). The combined organic layers were washed with water, brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The crude was purified by combiflash eluted with 40% ethyl acetate in hexane to get (89) as pale yellow solid (1.3 g, Yield: 44.24 %). MS: [M+H]+ 346.97.
Step 2: Synthesis of 2-chloro-N-(2-fluoro-5-nitrophenyl)-5-(4-(trifluoromethyl)phenyl) pyrimidin-4-amine (91):
To a solution of halo derivative (89) (1.0 eq) and respective boronate acid/ester derivative (90) (1.1 eq) in A A i methyl form am ide: water (4: 1) was added sodium carbonate or sodium bicarbonate (2.0 eq). The resulting reaction mixture was degassed under argon atmosphere for 15 minutes, followed by addition of tetrakis(triphenylphosphine)palladium(0) (0.1 eq). The resulting reaction mixture was heated at 90 °C for 16 hours. After completion of reaction (TLC monitoring), the reaction mixture was cooled to room temperature, water was added and extracted with ethyl acetate (3 times). The combined organic layers were washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The crude was purified by combiflash eluted with 35% ethyl acetate in hexane to get desired product (91) as light yellow solid (700 mg; Yield: 50.12%). MS: [M+H]+413.10.
Step 3 : Synthesis of N4-(2-fluoro-5-nitrophenyl)-N2-(l-methyl-lH-pyrazol-4-yl)-5-(4-(trifluoromethyl)phenyl)pyrimidine-2,4-diamine (92):
To an ice-cold solution of chloro compound (91) (1.0 eq) in isopropanol was added amine (22) (1.2 eq) and trifluoroacetic acid (2.0 eq). The reaction mixture was heated at 110 °C for 16 hours. After completion of the reaction (TLC monitoring), the reaction mixture was concentrated under reduced pressure, added saturated solution of sodium bicarbonate and extracted with dichloromethane (3 times). The combined organic layers were washed with brine solution, dried over anhydrous sodium sulfate and evaporated under reduced pressure. The crude was purified by combiflash eluted with 1% methanol in di chloromethane to get desired product (92) as pale yellow solid (500 mg; Yield: 70.24%). MS: [M+H]+ 474.09.
Step 4: Synthesis of N4-(5-amino-2-fluorophenyl)-N2-(l-methyl-lH-pyrazol-4-yl)-5-(4-(trifluoromethyl)phenyl)pyrimidine-2,4-diamine (93):
To an ice cold solution of nitro derivative (92) (1.0 eq) in methanol: tetrahydrofuran: water (2:2: 1) were added zinc-dust or iron powder (5 eq) and ammonium chloride (5 eq). The resultant reaction mixture was stirred at room temperature for 2 hours. After completion of reaction (TLC monitoring), reaction mixture passed through celite bed washed with 5% methanol in dichloromethane. The filtrate was washed with water, brine, dried over anhydrous sodium sulfate, filtered and concentrated to dryness to get the desired product (93) as semi solid (350 mg; Yield: 74.78%). MS: [M+H]+ 444.11.
Step 5 : Synthesis of N-(4-fluoro-3-((2-((l-methyl-lH-pyrazol-4-yl)amino)-5-(4-(trifluoromethyl)phenyl)pyrimidin-4-yl)amino)phenyl)acrylamide (Compound I):
To a solution of amino compound (93) (1.0 eq) in dichloromethane: tetrahydrofuran (1 :1) was cooled to -40 °C followed by triethylamine (3-5 eq) and acryloyl chloride (1.0 eq) were added. The mixture was stirred at the same temperature for 2 hours. After completion of reaction (monitored by TLC), added water and extracted with dichloromethane (3 times). The combined organic layers washed with brine, dried over anhydrous sodium sulfate, filtered and concentrated under reduced pressure. The crudes were purified by Prep-HPLC purification to to obtain Compound I as off white solid (30 mg, Yield: 13.33%). ‘H NMR (400 MHz, DMSO-de): 8 10.21 (bs, 1H), 9.24 (bs, 1H), 8.53 (bs, 1H), 7.99 (s, 1H), 7.71-7.81 (m, 5H), 7.57 (s, 1H), 7.08-7.16 (m, 3H), 6.37-6.44 (m, 1H), 6.21-6.26 (m, 1H), 5.74 (d, J= 8.4 Hz, 1H), 3.54 (s, 3H). LCMS: [M+H]+ 498.35.
PAT
- Pyrimidine compounds, compositions, and medicinal applications thereofPublication Number: WO-2022094354-A1Priority Date: 2020-10-30Linked Compounds: 1,056Linked Substances: 1,365
- Pyrimidine compounds, compositions and pharmaceutical uses thereofPublication Number: CN-116685583-APriority Date: 2020-10-30Linked Compounds: 921Linked Substances: 1,108
- Pyrimidine compounds, compositions, and medicinal applications thereofPublication Number: TW-202233603-APriority Date: 2020-10-30Linked Compounds: 531Linked Substances: 575
- Pyrimidine compounds, compositions, and their medicinal applicationsPublication Number: KR-20230116795-APriority Date: 2020-10-30Linked Compounds: 699Linked Substances: 744
- Egfr inhibitors for treatment of cancerPublication Number: WO-2024097270-A1Patent Family: TW-202432143-A; WO-2024097270-A1Priority Date: 2022-11-01Inventor(s): ANKROM WENDY; MAR BRENTON; PANDEY ANJALI; PEARSON PAUL; ZALUTSKAYA ALENAAssignee(s): BLUEPRINT MEDICINES CORPClassification: A61K31/506; A61K31/519; A61K31/555; A61P35/00; A61P35/04Abstract: The present disclosure provides improved methods of treating non-small cell lung cancer characterized by EGFR mutation using Compound (I): or a pharmaceutically acceptable salt thereof.Linked Compounds: 27Linked Substances: 28
- Salt and crystal forms of an epidermal growth factor receptor inhibitorPublication Number: US-2025282761-A1Patent Family: AU-2023265064-A1; CN-119923392-A; EP-4519254-A1; IL-316663-A; JP-2025517634-A; KR-20250012078-A; MX-2024013485-A; TW-202409016-A; US-2025282761-A1; WO-2023215431-A1Priority Date: 2022-05-04Inventor(s): GRUFF ERIC; Kuang Shanming; PANDEY ANJALI; SHAH HARSH; XIE TIANAssignee(s): BLUEPRINT MEDICINES CORPClassification: A61K31/506; C07D403/12Abstract: Various salt forms and free base solid forms of Compound (I) represented by the following formula are disclosed. Pharmaceutical compositions comprising the same, methods of treating a disease associated with an epidermal growth factor receptor (EGFR) family kinase using the same, and methods for making the salt forms of Compound (I) and crystalline forms thereof are also disclosed.Linked Compounds: 11Linked Substances: 13
- Salt and crystal forms of an epidermal growth factor receptor inhibitorPublication Number: WO-2023215431-A1Priority Date: 2022-05-04Linked Compounds: 18Linked Substances: 22
- Salt and crystal forms of an epidermal growth factor receptor inhibitorPublication Number: EP-4519254-A1Priority Date: 2022-05-04Linked Compounds: 14Linked Substances: 16
- Pyrimidine compounds, compositions, and medicinal applications thereofPublication Number: EP-4237418-A1Priority Date: 2020-10-30Linked Compounds: 821Linked Substances: 918



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

[1].
Zhou Y, et al., Anti-PD-1/L1 antibody plus anti-VEGF antibody vs. more VEGFR-targeted TKI as first-line therapy for unresectable hepatocellular carcinoma: a network meta-analysis. Explor Target Antitumor Ther. 2024;5(3):568-580. [Content Brief]
//////////pebezertinib, antineoplastic, BLU 451, BLU 203139, G8G5AU5GLJ, LNG 451
Orenasitecan
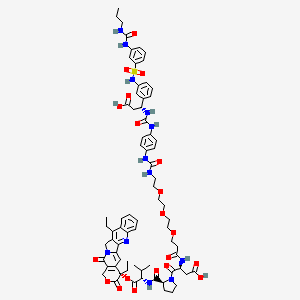
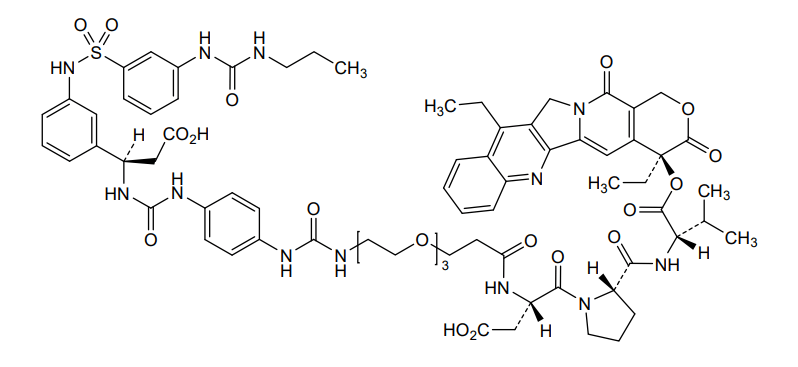

Orenasitecan
CAS 2418533-89-6
MF C72H86N12O20S MW1471.59
(3S)-3-[3-[2-[2-[2-[[4-[[(1R)-2-carboxy-1-[3-[[3-(propylcarbamoylamino)phenyl]sulfonylamino]phenyl]ethyl]carbamoylamino]phenyl]carbamoylamino]ethoxy]ethoxy]ethoxy]propanoylamino]-4-[(2S)-2-[[(2S)-1-[[(19S)-10,19-diethyl-14,18-dioxo-17-oxa-3,13-diazapentacyclo[11.8.0.02,11.04,9.015,20]henicosa-1(21),2,4,6,8,10,15(20)-heptaen-19-yl]oxy]-3-methyl-1-oxobutan-2-yl]carbamoyl]pyrrolidin-1-yl]-4-oxobutanoic acid
(4S)-4,11-diethyl-3,14-dioxo-3,4,12,14-tetrahydro-1Hpyrano[3′,4′:6,7]indolizino[1,2-b]quinolin-4-yl N-{1-[4-({[(1R)-2-
carboxy-1-(3-{3-[(propylcarbamoyl)amino]benzene-1-sulfonamido}phenyl)ethyl]carbamoyl}amino)anilino]-1-oxo-5,8,11-
trioxa-2-azatetradecan-14-oyl}-L-α-aspartyl-L-prolyl-L-valinate
antineoplastic, L3KV5NR5PG
Orenasitecan is a small molecule drug. The usage of the INN stem ‘-tecan’ in the name indicates that Orenasitecan is a antineoplastic, topoisomerase I inhibitor. Orenasitecan has a monoisotopic molecular weight of 1470.58 Da.
ORENASITECAN is a small molecule drug with a maximum clinical trial phase of II and has 1 investigational indication.
PAT
Cytostatic conjugates with integrin ligands
Publication Number: US-2021386864-A1
Priority Date: 2018-11-05
https://patents.google.com/patent/US20210386864A1
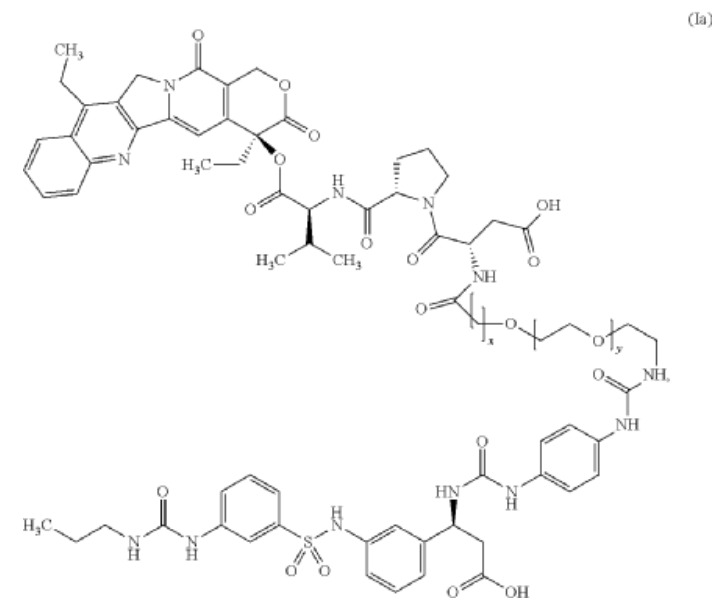
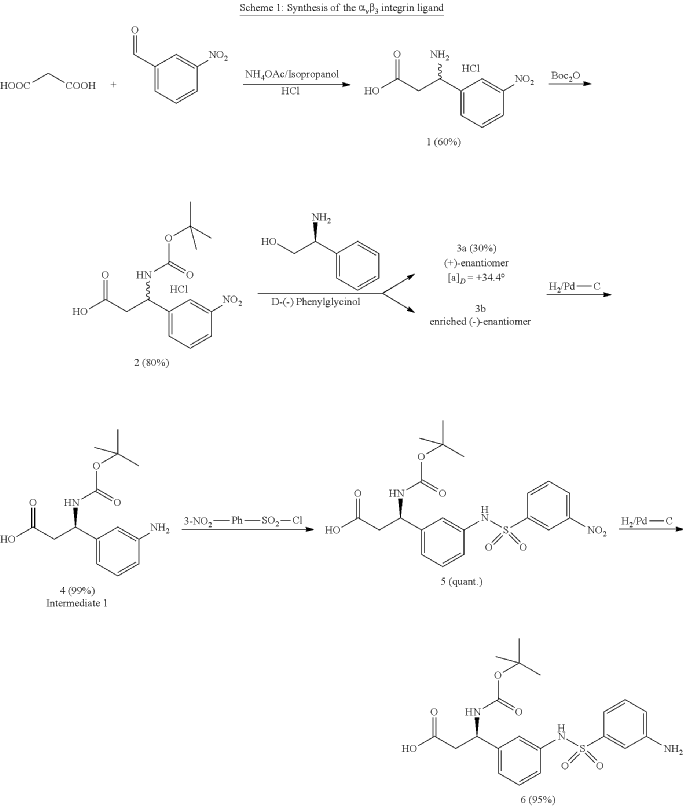
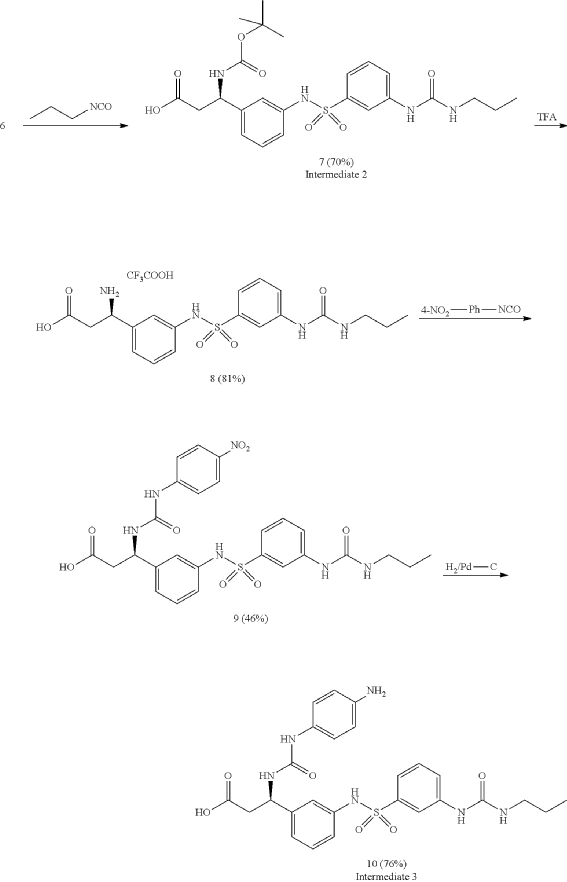
Separation of enantiomers can also be accomplished on different steps via chromatography using chiral columns
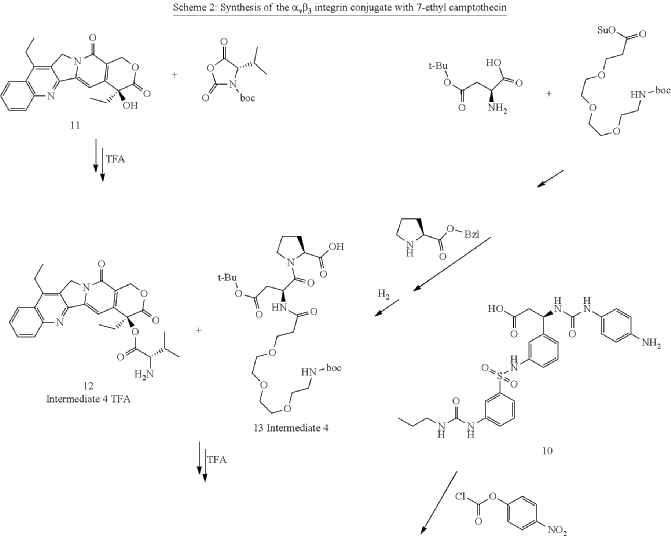
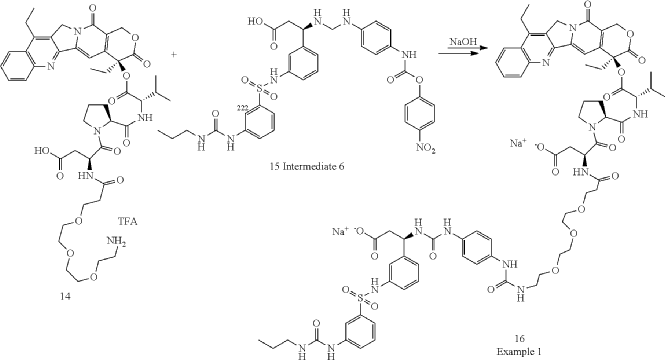



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

/////////orenasitecan, antineoplastic, L3KV5NR5PG

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










