| Patent ID | Patent Title | Submitted Date | Granted Date |
|---|---|---|---|
| US2015141506 | INDANE DIMERS FOR USE IN THE TREATMENT OF AUTOIMMUNE INFLAMMATORY DISEASE |
2013-05-23
|
2015-05-21
|
| US9260376 | COMPOUNDS FOR USE IN THE TREATMENT OF IMMUNE RELATED INFLAMMATORY DISEASE |
2012-07-20
|
2014-04-17
|
Home » Posts tagged 'trino'
Tag Archives: trino
PH 46A
PH 46A
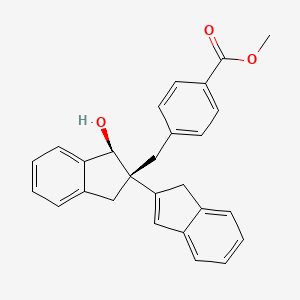
cas 1421332-97-9
C27 H24 O3, 396.48
Benzoic acid, 4-[[(1S,2S)-2,3-dihydro-1-hydroxy[2,2′-bi-1H-inden]-2-yl]methyl]-, methyl ester
Methyl 4-(((1S,2S)-1-hydroxy-2,3-dihydro-1H,1’H-[2,2′-biinden]-2-yl)methyl)benzoate
FREE ACID CAS 1380445-03-3, Benzoic acid, 4-[[(1S,2S)-2,3-dihydro-1-hydroxy[2,2′-bi-1H-inden]-2-yl]methyl]-
N-Methyl-(D)-Glucamine salt (NMDG)
1380445-04-4
C26 H22 O3 . C7 H17 N O5
D-Glucitol, 1-deoxy-1-(methylamino)-, 4-[[(1S,2S)-2,3-dihydro-1-hydroxy[2,2′-bi-1H-inden]-2-yl]methyl]benzoate (1:1)
PH46A, belonging to a class of 1,2-Indane dimers, has been developed by Trino Therapeutics Ltd research group as a potential therapeutic agent for the treatment of inflammatory and autoimmune diseases
The new chiral chemical entity PH46A, 6-(methylamino)hexane-1,2,3,4,5-pentanol 4-(((1S,2S)-1-hydroxy-2,3-dihydro-1H,1′H-[2,2-biinden]-2-yl)methyl)benzoate, was previously synthesized research group(1X) and shown to have potential therapeutic activity in the areas of inflammation and autoimmune diseases, including inflammatory bowel disease.(2X) PH46A recently completed a first-in-man Phase I clinical trial study.(3X)
-
1X Frampton, C.-S.; Zhang, T.; Scalabrino, G.; Frankish, N.; Sheridan, H. Acta Crystallogr., Sect. C: Cryst. Struct. Commun. 2012, 68, o323 DOI: 10.1107/S0108270112031265
-
2X Frankish, N.; Sheridan, H. J. Med. Chem. 2012, 55, 5497 DOI: 10.1021/jm300390f
-
2X Therapeutics, T. A study to assess the safety and tolerability of PH46A in healthy volunteers, to measure drug levels in these subjects and to determine the effect of food on the drug’s absorption. BioMed Central: ISRCTN Registry, EudraCT: 2013-003717-17, 2014.
PH 46A
- Originator Trino Therapeutics
- Class Anti-inflammatories; Benzoates; Indans; Muscle relaxants; Small molecules
- Mechanism of Action Mast cell stabilisers
- Orphan Drug Status No
- New Molecular Entity Yes
Highest Development Phases
- Phase I Ulcerative colitis
Most Recent Events
- 31 Aug 2014 Trino Therapeutics completes a phase I trial in Ulcerative colitis (In volunteers) in United Kingdom (ISRCTN90725219)
- 07 Feb 2014 Phase-I clinical trials in Ulcerative colitis (in volunteers) in United Kingdom (PO)
- 04 Jun 2012 Pharmacodynamics data from a preclinical trial in Ulcerative colitis released by Trino Therapeutics
Cytokines can be produced by various cell populations and have been shown to augment or limit immune responses to pathogens and influence the autoimmune response. One family of cytokines, which uses the common receptor gamma chain (cc), a component of receptors for interleukin (IL)-2, IL-4, IL-7, IL-9, IL-15 and IL-21, has been classically defined as growth and survival factors.
IL-2 production can induce an immune response by promoting the proliferation and generation of CD4+ Thl, CD4+ Th2 and CD8+ CTL effector cells. Many of the immunosuppressive drugs used in the treatment of autoimmune diseases and organ transplant rejection, such as corticosteroids and immune suppressive drugs (ciclosporin, tacrolimus) work by inhibiting the production of IL-2 by antigen -activated T cells. Others (sirolimus) block IL-2R signalling, thereby preventing the clonal expansion and function of antigen-selected T cells [ref: Opposing functions of IL-2 and IL-7 in the regulation of immune responses Shoshana D. Katzman, Katrina . Hoyer, Hans Dooms, Iris K. Gratz, Michael D. Rosenblum, Jonathan S. Paw, Sara H. Isakson, Abul K. Abbas. Cytokine 56 (201 1) 1 16-121]
In contrast IL-2 can inhibit the immune response by promoting the survival and functionality of natural (thymic) regulatory T-cells (Tregs), promoting the generation of induced (peripheral) Tregs and inhibiting the generation of CD4+ Thl 7 effector cells [ref: IL-2 and autoimmune disease. Anneliese Schimpl , A., Berberich, I, Kneitz, B., Kramer, S., Santner-Nanan, B., Wagner, S., Wolf, M., Hunig, T. Cytokine & Growth Factor Reviews 13 (2002) 369-378]. Interleukin-2/IL-2R deficiency with time leads to multiorgan inflammation and the formation of autoantibodies of various specificities. Depending on the genetic background, death occurs within a few weeks to a few months, mostly from autoimmune hemolytic anemia or inflammatory bowel disease (IBD) [ref. Sadlack B, Merz H, Schorle H, Schimpl A, Feller AC, Horak I. Ulcerative colitis-like disease in mice with a disrupted interleukin-2 gene. Cell 1993;75:253-61]. IL-2 signalling has been shown to be important in both the initiation and regulation of immune responses. In these dual and opposing roles, IL-2 acts to balance immune response, both driving immune cell activation and subsequent reduction. The potential clinical applicability of either augmenting or inhibiting signals mediated by IL-2 is significant and includes cancer, autoimmune inflammatory diseases, organ transplantation and HIV.
Inflammatory bowel disease (IBD) is an autoimmune inflammatory disease that consists of two idiopathic inflammatory diseases, ulcerative colitis (UC) and Crohn’s disease (CD). The greatest distinction between UC and CD is the range of inflamed bowel tissue. Inflammation in CD is discontinuously segmented, known as regional enteritis, while UC is superficial inflammation extending proximally and continuously from the rectum. At present, the exact cause of IBD is unknown. The disease seems to be related to an exaggerated mucosal immune response to infection of the intestinal epithelium because of an imbalance of pro- inflammatory and immune- regulatory molecules. The inheritance patterns of IBD suggest a complex genetic component of pathogenesis that may consist of several combined genetic mutations. Currently no specific diagnostic test exists for IBD, but as an understanding of pathogenesis is improved so will the corresponding testing methods. Treatment of IBD consists of inducing and maintaining remission. IBD patients may be maintained on remission by use of a 5-aminosalycilate. However, while the use of aminosalycilates in UC provides considerable benefit, both in inducing remission in mild to moderate disease and in preventing relapse, the usefulness of these drugs to maintain remission in CD is questionable and is no longer recommended. The mainstay of treatment of active disease is a corticosteroid, commonly used for limited periods to return both UC and CD patients to remission, though budesonide, designed for topical administration with limited systemic absorption, has no benefit in maintaining remission. Alternatives, such as the immunosuppressive drugs azathioprine and mercaptopurine, together with methotrexate and cyclosporine have limited efficacy and the capability of inducing grave adverse effects. Anti- TNFa antibodies, such as infliximab and adalimubab, may be used in those patients unresponsive to standard immunosuppressive therapy. However, many patients fail to respond to anti-TNFa therapy, either due to their particular phenotype or by the production of autoantibodies.
| Inventors | Helen Sheridan, Neil Frankish |
| Applicant | Venantius Limited |
PATENTS
WO 2013014660
https://encrypted.google.com/patents/WO2013014660A1?cl=en
Compound 6: The N-Methyl-(D)-Glucamine salt (NMDG) of compound 2.
Compound 6 physiochemical properties:
Appearance: Off-white solid
Molecular Weight: 577 (free acid: 382)
Molecular Formula: C33H39O8N (free acid: C26H2203)
Melting Point: 165-167 °C
Compound 6: [a]D:-76.5 (sample concentration: 200 mg/10 ml in Water)
Mass (Da): ES+ only [NMDG+Na] was visible
Elemental analysis: Calc: C (68.61), H (6.80), N (2.42), O (22.16). Found: C (68.44), H
(6.80), N (2.50), 0 (21.98) δΗ(400 MHz, DMSO-d6): 2.48 (3H, apparent s, NCH3), 2.65 (1H, d, J=13.56 Hz, HCH), 2.84-
3.02 (4H, m), 3.16 (1H, d, J= 13.60 Hz, HCH), 3.40-3.70 (7H, m), 3.85-3.92 (l H, m), 5.06 (1H, s, CH-OH), 5.93 (1H, broad s, CH- OH), 6.41 (1H, f, .CH=C), 6.80 (2H, d, J=7.92 Hz, Ar-H), 7.06-7.41 (8H, m, Ar-H), 7.64 (2H, d, J=7.80 Hz, Ar-H).
6c(100 MHz, DMSO): 33.8 (CH3), 37.9 (CH2), 38.2 (CH2), 39.5 (CH2), 51.6 (CH2-N), 55.8
(quat. C), 63.5 (CH2-0), 69.0 (CH-O), 70.3 (CH-O), 70.6 (CH-O), 71.3 (CH-O), 81.1 (CH-OH), 120.1 (tert. C), 123.4 (tert. C), 123.7 (tert. C), 124.3 (tert. C), 124.4 (tert. C), 126.1 (tert. C), 126.3 (tert. C), 127.0 (tert. C), 127.5 (tert. C), 2 x 128.5 (2 x tert. C), 2 x 129.1 (2 x tert. C), 140,4 (quat. C), 141.1 (quat. C), 142.9 (quat. C), 144.5 (quat. C), 145.2 (quat. C), 154.3 (quat. C), 170.4 (C=0).
Synthesis of methyl 4- (lS,2S)-l-hvdroxy-2,3-dihvdro-lHJ’H-f2,2′-biinden1-2-yl)methyl) benzoate (17):
To a solution of 4-(((15,25)-l-hydroxy-2,3-dihydro-lH, l’H-[2,2′-biinden]-2-yl)methyl)benzoic acid (100 mg, 0.26 mmol) and K2C03 (72 mg, 0.52 mmol) in DMF (2.5 mL), was added Mel (148 mg, 1.04 mmol) and then stirred at room temperature for 4 h. The reaction mixture was diluted with 1.5 N HCI (50 mL) and extracted with ethyl acetate (3 x 25 mL). The organic layer was washed with 10 % aq. NaHC03 (25 mL), brine (25 mL), dried over anhydrous Na2S04 and evaporated under reduced pressure. The residue was purified by CombiFlash using 20 % ethyl acetate in chloroform as an eluent to yield 62 mg (59 %) of the title compound as an off white solid.
LCMS (-OH): observed 379.2, calculated 396.17, molecular formula C27H2403
Purity (HPLC): 97 %.
Ή NMR (400 MHz, CDC13): 6 2.84 (1H, d, J = 13.28 Hz, ¾), 3.00 (1H, d, J = 15.64 Hz, CH2), 3.05 (1H, d, J = 15.56 Hz, CPb), 3.27 (lH, d, J = 13.32 Hz, CH ), 3.45 (1H, d, J = 22.52 Hz, CH2), 3.57 (1H, d, J = 22.60 Hz, CH2), 3.89 (3H, s, OCH3), 5.25 (1H, s, CHQH), 6.47 (1H, s, CH=C), 6.96 (2H, d, J = 8.24 Hz, Ar-H), 7.17 (1H, dt, J = 2.04, 9.88 Hz, Ar-H), 7.24-7.33 (5H, m, Ar-H), 7.43 (2H, d, J = 7.60 Hz, Ar-H), 7.83 (2H, dd, J = 1.76, 6.60 Hz, Ar-H).
PATENT
WO 2013174916

PATENT
US 9260376
US 20150141506
PH 46A (S,S & R,R) racemic
Melting point 141–143 °C. 1H NMR (400 MHz, CDCl3) δH (ppm) 6.99 (d, J = 7.72 Hz, 2H), 7.46 (d, J = 7.04 Hz, 2H), 7.20–7.31 (m, 6H), 6.97 (d, J = 7.80 Hz, 2H), 6.50 (s, 1H), 5.29 (d, J = 24.16 Hz, 1H), 3.91 (s, 3H), 3.60 (d, J = 22.68 Hz, 1H), 3.48 (d, J = 22.88 Hz, 1H), 3.28 (d, J = 13.24 Hz, 1H), 3.06 (d, J = 15.64 Hz, 1H), 3.51 (d, J = 16.00 Hz, 1H), 2.86 (d, J = 13.28 Hz, 1H). 13C NMR (100 MHz, CDCl3) δC (ppm) 166.9, 152.3, 144.1, 143.9, 143.4, 142.4, 140.0, 2 × 129.8, 2 × 128.6, 128.1, 128.0, 127.5, 126.6, 126.0, 124.4, 123.9, 123.6, 123.2, 120.2, 82.4, 55.5, 51.6, 39.6, 38.2, 38.0. HRMS (ESI) m/z calculated for C27H24O3 (M + Na)+ , 419.1606; found, 419.1618. Achiral HPLC: Zorbax C18 XDB (150 x 4.6 mm), 20:80:0.1 (v:v:v) water:MeOH:TFA, 1.0 mL/min, 254 nm, RT: 4.31 min. Chiral HPLC: Chiralpak IC, 90:10:0.1 (v:v:v) heptane:IPA:TFA, 1.0 mL/min, 210 nm, RT: 7.98 min & 9.38 min.
PH 46A
1H NMR (400 MHz, dmso-d6) δH (ppm): 7.70 (d, J = 8.4 Hz, 2H, Ar–H), 7.34–7.40 (m, 2H, Ar–H), 7.14–7.25 (m, 5H, Ar–H), 7.07 (t, J = 14.4 Hz, 1H, Ar–H), 6.97 (d, J = 8.4 Hz, 2H, Ar–H), 6.39 (s, 1H, CH = C), 5.85 (d, J = 7.2 Hz, 1H, CHOH), 5.06 (d, J = 6.8 Hz, 1H, CHOH), 3.77 (s, 3H, CH3), 3.56 (d, J = 23.2 Hz, 1H, CH2), 3.42 (d, J = 23.2 Hz, 1H, CH2), 3.20 (d, J = 13.6 Hz, 1H, CH2), 2.96 (s, 2H, CH2), 2.73 (d, J = 13.6 Hz, 1H, CH2).

clip

https://www.sciencedirect.com/science/article/pii/S0040403916301332
Investigation of the Stereoselective Synthesis of the Indane Dimer PH46A, a New Potential Anti-inflammatory Agent
†Celtic Catalysts Ltd, NovaUCD, Belfield, Dublin 4, Ireland
‡Trino Therapeutics Ltd, The Tower, Trinity Technology & Enterprise Campus, Dublin 2, Ireland
§ Drug Discovery Group, School of Pharmacy and Pharmaceutical Sciences & Trinity Biomedical Sciences Institute (TBSI), Trinity College, Dublin 2, Ireland
Org. Process Res. Dev., Article ASAP
DOI: 10.1021/acs.oprd.7b00258
Publication Date (Web): November 27, 2017
Copyright © 2017 American Chemical Society
*E-mail: hsheridn@tcd.ie.
PH46A, belonging to a class of 1,2-Indane dimers, has been developed by our research group as a potential therapeutic agent for the treatment of inflammatory and autoimmune diseases. The initial synthetic route to PH46A gave a low overall yield, due in large part to the generation of undesired diastereoisomer 5 and the unwanted enantiomer (R,R)-8 during the synthesis. The aim of this work was to carry out a comprehensive investigation into the stereoselective synthesis of PH46A. Significant progress was made on the ketone reduction step, where the use of triisobutylaluminum [TiBA, Al(iBu)3] afforded high selectivity for the target diastereoisomer (rac)-6, compared to the unfavorable ratio obtained using a previous process. This enabled a multikilo scale synthesis of PH46A in a GMP environment. Further, a brief proof-of-principle investigation was carried out using an achiral phase transfer catalyst (PTC) for alkylation at the methine carbon of the parent indanone.
from PubChem
///////////////////////PH46A, PH 46A, phase 1, trino
O=C(OC)c1ccc(cc1)C[C@]3(Cc2ccccc2[C@H]3O)C4=Cc5ccccc5C4

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....