Home » Posts tagged 'Perampanel'
Tag Archives: Perampanel
Zydus Cadila, New Patent,US 20160039759, PERAMPANEL

PERAMPANEL

Zydus Cadila, New Patent,US 20160039759, PERAMPANEL
(US20160039759) PROCESS FOR THE PREPARATION OF PERAMPANEL
CADILA HEALTHCARE LIMITED
Sanjay Jagdish DESAI
Jayprakash Ajitsingh Parihar
Kuldeep Natwarlal Jain
Sachin Ashokrao Patil

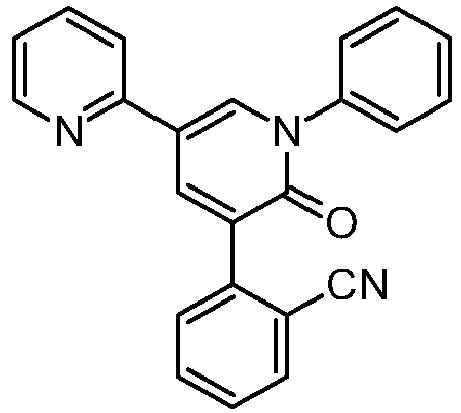
Perampanel, a non-competitive AMPA receptor antagonist, is the active ingredient of FYCOMPA® tablets (U.S) which is approved as an adjunctive therapy for the treatment of partial on-set seizures with or without secondarily generalized seizures in patients with aged 12 years and older. Chemically, Perampanel is 5′-(2-cyanophenyl)-1′-phenyl-2,3′-bipyridinyl-6′(1′H)-one, with an empirical formula C23H15N30 and molecular weight 349.384 g/mol which is represented by Formula (I).

U.S. Pat. No. 6,949,571 B2 discloses perampanel and its various processes for preparation thereof.
U.S. Pat. No. 7,759,367 B2 discloses the pharmaceutical composition of perampanel and an immunoregulatory agent and their uses.
U.S. Pat. No. 8,304,548 B2 discloses the reaction of 5′-bromo-1′-phenyl-[2,3′-bipyridin]-6′(1′H)-one with 2-(1,3,2-dioxaborinan-yl)benzonitrile in the presence of palladium compound, a copper compound, a phosphorus compound and a base to form perampanel of Formula (I). Also discloses the crystalline hydrate, anhydrous crystal Form I, anhydrous crystal Form III, & anhydrous crystal Form V of perampanel of Formula (I).
U.S. Pat. No. 7,803,818 B2 discloses an amorphous form of perampanel. U.S. Pat. No. 7,718,807 B2 discloses salts of perampanel. International (PCT) publication No. WO 2013/102897 A1 discloses anhydrous crystalline Form III, V & VII of perampanel.
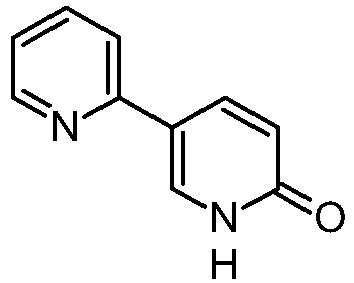
U.S. PG-Pub. No. 2013/109862 A1 discloses the method for preparing 2-alkoxy-5-(pyridin-2-yl)pyridine, which is an intermediate for preparing perampanel key starting material 5-(2′-pyridyl)-2-pyridone.
U.S. Pat. No. 7,524,967 B2 discloses the preparation of 5-(2′-pyridyl)-2-pyridone, an intermediate in the preparation perampanel.
International (PCT) publication No. WO 2014/023576 A1 discloses the preparation of cyanophenyl boronic acid, an intermediate in the preparation perampanel.
The prior-art processes suffer with problems of poor yield and requirement of chromatographic purification or series of crystallizations which further reduces the overall yield of the final product, which is overcome by the process of the present invention.









 Pankaj Patel, chairman, Zydus Cadila
Pankaj Patel, chairman, Zydus Cadila
EXAMPLES
The present invention is further illustrated by the following examples which is provided merely to be exemplary of the invention and do not limit the scope of the invention. Certain modification and equivalents will be apparent to those skilled in the art and are intended to be included within the scope of the present invention.
Example-A: Preparation of 5-(2-pyridyl)-1,2-dihydropyridin-2-one In a 500 mL round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, a solution of 188.80 g 5-bromo-2-methoxypyridine in 190 mL tetrahydrofuran and 12.92 g pyridine-2-yl boronic acid were added and refluxed. The reaction mixture was cooled to 25-30° C. and aqueous solution of hydrochloric acid was added and stirred for 1 hour. The reaction mixture was neutralized with aqueous sodium hydroxide and extracted with tetrahydrofuran.
The organic layer was washed with saline water, dried over anhydrous magnesium sulfate, and then evaporated to obtain the titled compound.
Example-1
Preparation of 3-bromo-5-(2-pyridyl)-1,2-dihydropyridin-2-one
In a 2 L round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, 201.5 g 5-(2-pyridyl)-1,2-dihydropyridin-2-one, 208.3 g N-bromosuccinimide and 1300 mL N,N-dimethylforamide were stirred at 25-30° C. for 2-3 hours. After completion of the reaction, the reaction mixture was poured into water and stirred for 30 min. The precipitate was filtered, washed with N,N-dimethylforamide and dried at 50° C. to obtain 230 g title compound.
Example-2
Preparation of 3-bromo-5-2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one
In a 500 mL round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, a solution of 18.75 g 3-bromo-5-(2-pyridyl)-1,2-dihydropyridin-2-one in 300 mL methylene dichloride, 18.36 g 1-phenyl boronic acid, 3.47 g palladium triphenylphosphine and 10 mL triethyl amine were added and the reaction mixture was stirred for 1 hour at 25-35° C. The reaction mixture was filtered and the filtrate was evaporated to dryness. The residue was crystallised from ethyl acetate to obtain the title compound.
Example-3
Preparation of Perampanel
In a 1 L round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, a suspension of 188 g 3-bromo-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one, 161.2 g 2-(1,3,2-dioxaborinan-2-yl)benzonitrile, 3.0 g tetrakis(triphenylphosphine)-palladium(0), 10 mL triethylamine (10 mL) in 300 mL methylene dichloride were stirred at 25-30° C. for 12 hours. To the reaction mixture was added 5 mL conc. aqueous ammonia, 10 mL water and 40 mL ethyl acetate. The separated organic layer was washed with water and saturated saline solution and dried over magnesium sulfate. The solvent was removed under vacuum. Ethyl acetate was added to the residue and heated obtain clear solution. n-hexane was added to this solution and cooled to 25-30° C. The obtained solid was filtered and washed with ethyl acetate and dried to obtain perampanel.
Example-4
Preparation of 3-Bromo-5-(2-pyridyl)-1,2-dihydropyridin-2-one
In a 2 L round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, 100 g 5-(2-pyridyl)-1,2-dihydropyridin-2-one, 108.5 g N-bromosuccinimide and 500 mL N,N-dimethylforamide were stirred at 30-35° C. for 3 hours. 100 mL water was added to the reaction mixture at 5-15° C. and stirred at 30-35° C. for 1 hour. The solid obtained was filtered, washed with water and dried to obtain 129 g 3-bromo-5-(2-pyridyl)-1,2-dihydropyridin-2-one.
Example-5
Preparation of 3-bromo-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one
In a 2 L round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, 100 g 3-bromo-5-(2-pyridyl)-1,2-dihydropyridin-2-one, 72.8 g phenylboronic acid and 500 mL N,N-dimethylformamide were added at 30-35° C. and stirred. 11.9 g copper acetate and 15.7 g pyridine were added and air was purged into the reaction mixture and stirred for 16 hours at 30-35° C. After the completion of the reaction, the reaction mixture was poured into 1200 mL aqueous ammonia at 10-15° C. and stirred for 2 hours at 30-35° C. The obtained solid was filtered, washed with water and dried to obtain 120 g 3-bromo-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one.
Example-6
Purification of 3-bromo-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one
In a 1 L round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, 100 g 3-bromo-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one and 500 mL isopropyl alcohol were stirred at 60-65° C. for 30 min. The reaction mixture was cooled to 20-25° C. and stirred for 30 min. The reaction mixture was filtered, washed with isopropanol and dried to obtain 96 g pure 3-bromo-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one.
Example-7
Preparation of Perampanel
In a 1 L round bottom flask, equipped with a mechanical stirrer, thermometer and an addition funnel, 100 g 3-bromo-5-(2-pyridyl)-1-phenyl-1,2-dihydropyridine-2-one and 125 g 2-(1,3,2-dioxaborinan-2-yl)benzonitrile and 1500 mL N,N-dimethylformamide were added under inert atmosphere. 44 g potassium carbonate and 4.2 g palladium tetrakis were added and stirred at 115-125° C. for 3 hours. The solvent was removed under vacuum. Ethyl acetate was added to the residue and the organic layer was distilled off to obtain perampanel (78 g).

////////Zydus Cadila, New Patent,US 20160039759, PERAMPANEL
Perampanel

Perampanel
5′-(2-cyanophenyl)-1′-phenyl-2,3′-bipyridinyl-6′(1’H)-one
cas no 380917-97-5
FDA-approved drug to treat epilepsy. Trade name Fycompa, Eisai (Eisai) research and development.
FYCOMPA tablets contain perampanel, a non-competitive AMPA receptorantagonist. Perampanel is described chemically as 2-(2-oxo-1-phenyl-5-pyridin-2-yl-1,2-dihydropyridin-3-yl) benzonitrile hydrate (4:3).
The molecular formula is C23H15N3O •3/4H2O and the molecular weight is 362.90 (3/4 hydrate). The chemical structure of perampanel is:
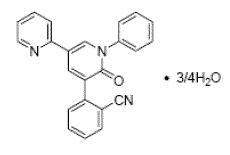 |
Perampanel is a white to yellowish white powder. It is freely soluble in N-methylpyrrolidone, sparingly soluble in acetonitrile and acetone, slightly soluble in methanol, ethanol and ethyl acetate, very slightly soluble in 1-octanol and diethyl ether and practically insoluble in heptane and water.
Perampanel (INN/USAN, trade name Fycompa) is an antiepileptic drug developed by Eisai Co. that acts as a selective noncompetitive antagonist of AMPA receptors, the major subtype of ionotropic glutamate receptors.[1][2]
Perampanel was found to be effective in the treatment of refractory partial-onset seizures in three pivotal (Phase 3) clinical trials[3][4] and has been approved for marketing under the brand name Fycompa by the European Medicines Agency.[5] The minimum effective dose is 4 mg once daily; doses of 8 mg and 12 mg daily provide a greater therapeutic benefit with a corresponding increase in adverse events. Dizziness and somnolence/sedation/fatigue are the most frequent dose-related adverse events. The drug is currently approved, for the control of partial-onset seizures, in those of both sexes who suffer from epilepsy and who are 12 years of age and older, by the Food and Drug Administration, and is considered to be a scheduled drug (an agent with the potential for addiction). Perampanel has been studied in other clinical indications includingParkinson’s disease.[6][7]
It has high potency (IC50 in vitro in functional studies of about 100-250 nM) and a prolonged terminal half-life in humans of approximately 105 hours. The drug is 95% bound to plasma protein. Its primary route of metabolism is by CYP3A4. It does not induce or inhibit P450 enzymes. About 70% of the dose is excreted in the feces and 30% in the urine; less than 2% of the dose is excreted unchanged into the urine.
In clinical trials, perampanel was generally well tolerated although the incidence of adverse events increased in a dose-dependent fashion. There was no increase in serious adverse events compared with placebo. According to the Food and Drug Administration, most common adverse reactions reported by patients receiving Fycompa in clinical trials include dizziness, drowsiness, fatigue, irritability, falls, upper respiratory tract infection,weight increase, vertigo, loss of muscle coordination (ataxia), gait disturbance, balance disorder, anxiety, blurred vision, stuttering (dysarthria), weakness (asthenia), aggression, and excessive sleep (hypersomnia).[8]
Fycompa’s label has a boxed warning to alert prescribers and patients about the risk of serious neuropsychiatric events. Some of these events were reported as serious and life-threatening. Violent thoughts or threatening behavior (including homicidal ideation) was also observed in a few patients. Patients and caregivers should alert a health care professional immediately if changes in mood or behavior that are not typical for the patient are observed. Health care professionals should closely monitor patients during the titration period when higher doses are used.[9]
- Rogawski, M. A. (2011). “Revisiting AMPA Receptors as an Antiepileptic Drug Target”. Epilepsy Currents 11 (2): 56–63. doi:10.5698/1535-7511-11.2.56. PMC 3117497. PMID 21686307. edit
- Rogawski MA, Hanada T. Preclinical pharmacology of perampanel, a selective non-competitive AMPA receptor antagonist. Acta Neurol Scand 2013;127 (Suppl. 197): 19–24.Rogawski, M. A.; Kaukinen, T.; Collin, P.; Krekelä, I.; Patrikainen, H.; Tillonen, J.; Nyrke, T.; Laurila, K.; Haimila, K.; Partanen, J.; Valve, R.; Mäki, M.; Luostarinen, L. (2013). “Preclinical pharmacology of perampanel, a selective non-competitive AMPA receptor antagonist”. Acta Neurologica Scandinavica 127 (1): 19–25. doi:10.1111/ane.12100. PMID 22494246. edit
- Krauss, G. L.; Serratosa, J. M.; Villanueva, V.; Endziniene, M.; Hong, Z.; French, J.; Yang, H.; Squillacote, D.; Edwards, H. B.; Zhu, J.; Laurenza, A. (2012). “Randomized phase III study 306: Adjunctive perampanel for refractory partial-onset seizures”. Neurology 78 (18): 1408–1415.doi:10.1212/WNL.0b013e318254473a. PMID 22517103. edit
- French, J. A.; Krauss, G. L.; Biton, V.; Squillacote, D.; Yang, H.; Laurenza, A.; Kumar, D.; Rogawski, M. A.; Campanille, V.; Floridia, J.; Ilari, R.; Consalvo, D. E.; Thomson, A.; Sfaello, I.; Pociecha, J.; Nieto, F.; Firstenfeld, A.; Zuin, D.; Mesri, J.; Silva, W.; Nofal, P.; Cristalli, D.; Clement, J. F.; Hwang, P.; McLachlan, R.; Pillay, N.; Lasso, J.; Peralta, B. L.; Hernandez, M. L.; Tenhamm, E. (2012). “Adjunctive perampanel for refractory partial-onset seizures: Randomized phase III study 304”. Neurology 79 (6): 589–596. doi:10.1212/WNL.0b013e3182635735. PMC 3413761. PMID 22843280. edit
- “European Medicines Agency Report on Perampanel”.
- Gottwald MD, Aminoff MJ (July 2008). “New frontiers in the pharmacological management of Parkinson’s disease”. Drugs Today 44 (7): 531–45.doi:10.1358/dot.2008.44.7.1217105. PMID 18806903.
- http://www.webmd.com/epilepsy/news/20121024/epilepsy-drug-fycompa-approved
- http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm325038.htm
- http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm325038.htm
Perampanel structure is formed by the coupling of an aromatic ring . Pyridone centrally located, surrounded by connecting two benzene rings and a pyridine ring. The synthesis of 2,5 – dibromopyridine (1) Start with sodium methoxide to produce 2-substituted, and an organic tin compound occurs Stille Coupling 3 4 4 HBr generated after acid hydrolysis and coupling of benzyl bromide with NBS to give 5,5. After 6 coupling of boronic ester and get Perampanel.
Perampanel is a pharmaceutically active agent, currently in clinical phase 3. It can be used to treat Parkinson’s disease, epilepsy and multiple sclerosis.
Perampanel, having the following chemical formula
is also known as E 2007, ER 155055-90 and 3-(2-cyanophenyl)-1-phenyl-5-(2-pyridil)-1,2-dihydropyridin-2-one
Various methods of synthesis of such molecules are known, such as those reported in EP1300396, EP 1465626, EP 1772450, EP 1764361 and EP 1970370.
Many of the methods of synthesis of such active substances reported by the prior art use the key intermediate 5-(2-pyridil)-1,2-dihydropyridin-2-one also known as 2,3′-bipyridin-6′(1′H)-one having the following chemical formula:
Other methods use the synthetic precursor of this intermediate known as 2-methoxy-5-(pyridin-2-yl)pyridine or 6′-methoxy-2,3′-bipyridine having the formula:
2,3′-bipyridin-6′(1′H)-one. it is in fact prepared by simple acid-catalysed demethylation of the 6′-methoxy-2,3′-bipyridine as is reported in the prior art.
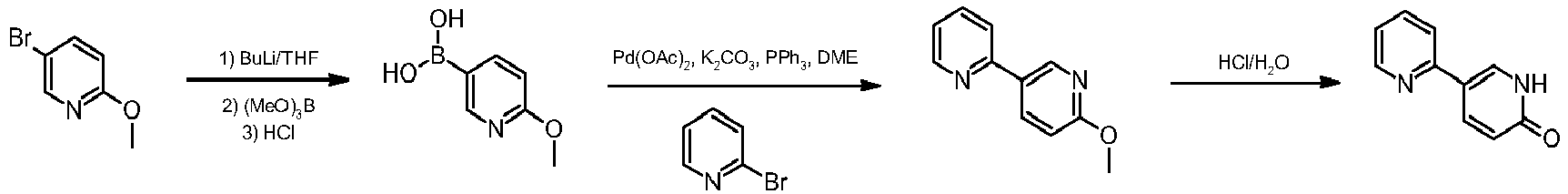
Various ways of synthesising 2-methoxy-5-(pyridin-2-yl)pyridine are known. The process summarised in Diagram (I) below is described in WO 2001096308:
Such process highlights clear disadvantages such as the need to operate in cryogenic conditions (T=−78° C.) using special equipment and the need to isolate boronic acid via work-up. In addition the use of 2-Bromopyridine is required, which exacerbates the production of waste compared to 2-chloropyridine.
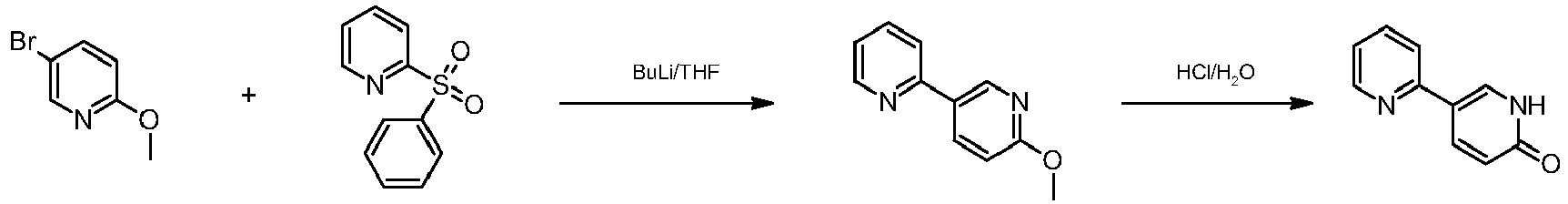
Another process described in WO 2004009553 is summarised in Diagram (II):
Disadvantages of this process include the use of high molecular weight benzene-sulfonyl pyridine entailing a scarce atom-economy of the process and the need to operate at low temperature T (−78° C.) using special equipment.
Lastly, a completely different process is described in WO20087093392 for the preparation of 2,3′-bipyridin-6′(1′H)-one (Diagram (III)) which however does not include the preparation of the intermediate precursor 2-methoxy-5-(pyridin-2-yl)pyridine:
Perampanel and other 1 ,2-dihydropyridine compounds which possess antagonistic action against AMPA receptor and/or inhibitory action against kainate receptor are described in WO 01/96308. Example 7 in WO 01/96308 discloses a process for producing perampanel by reacting 3-(2-cyanophenyl)-5-(2-pyridyl)-2(lH)-pyridone with phenyl boronic acid, copper acetate and triethylamine in methylene chloride, followed by addition of concentrated aqueous ammonia, water and ethyl acetate. After work-up (phase separation, washing the organic phase and drying over magnesium sulfate), the solvent was concentrated in vacuo and the residue was purified by a silica gel column chromatography (ethyl acetate:hexane=l :2) to give the title product as pale yellow powder. There is no disclosure regarding the polymorphic nature of the product.
A new crystalline or amorphous form of a compound may possess physical properties that differ from, and are advantageous over, those of other crystalline or amorphous forms. These include, packing properties such as molar volume, density and hygroscopicity; thermodynamic properties such as melting temperature, vapor pressure and solubility; kinetic properties such as dissolution rate and stability under various storage conditions; surface properties such as surface area, wettability, interfacial tension and shape; mechanical properties such as hardness, tensile strength, compactibility, handling, flow and blend; and filtration properties. Variations in any one of these properties may affect the chemical and pharmaceutical processing of a compound as well as its bioavailability and may often render the new form advantageous for pharmaceutical and medical use.
EP 1764361 (US 2010/324297) discloses three anhydrous crystalline forms ofperampanel, designated Form I, Form III and Form V and a hydrate form ofperampanel. Anhydrous Form I is prepared in accordance with Example Dl by dissolving perampanel in ethyl acetate (EtOAc) under reflux, cooling the solution, seeding with anhydrous perampanel crystals, continued cooling and collecting the precipitated crystals. Anhydrous Form V is prepared in accordance with Example CI, by dissolving perampanel in acetone, heating to reflux and concentrating the solution to solidification, dissolving the solids in acetone-water, refluxing then cooling and collecting the precipitate. The hydrate form is prepared in accordance with Example Bl by dissolving perampanel in acetone-water, heating, cooling the solution, seeding with perampanel hydrate crystals, continued cooling and collecting the precipitated crystals. US 2009/0088574 discloses a crystalline form of perampanel designated Form IV, which is prepared by slurring perampanel in an acetone/water mixture.
US 7,803,818 discloses an amorphous form of perampanel which is prepared by spray drying perampanel from an acetone solution.
US 7,718,807 discloses acid addition salts of perampanel or a hydrate thereof, wherein the acid is selected from the group consisting of benzenesulfonic acid, p- toluenesulfonic acid, hydrochloric acid, hydrobromic acid, sulfuric acid, methanesulfonic acid, fumaric acid, tartaric acid, succinic acid and benzoic acid.
…………………………………………………………………
Perampanel aromatic ring structure is made of highly coupled. Pyridone centrally located, surrounded by connecting two benzene ring and a pyridine ring. The synthesis of 2,5 – dibromo pyridine ( 1) Start (Synthesis, 2012, 57), sodium methoxide instead of generating 2 , and organotin compounds 3 Stille Coupling occurs to generate 4 . 4 in HBr phenylboronic acid after hydrolysis and coupling to get 5 , 5 after bromination with NBS and borate 6 coupled to get Perampanel.
…………………………….
nmr
A Practical, Laboratory-Scale Synthesis of Perampanel
……………………
updated info
-
Perampanel is a pharmaceutical active substance, currently in clinical phase 3, used to treat Parkinson’s disease, epilepsy and multiple sclerosis.
-
[0003]
-
[0004]Various methods of synthesis of such molecule are known, such as those reported in the patent publications EP1300396 , EP1465626 ,EP1772450 , EP1764361 and EP 1970370 .
-
[0005]Many of the methods of synthesis of such active substance reported by the prior art use the key intermediate 5-(2-pyridil)-1,2-dihydropyridin-2-one also known as 2,3′-bipyridin-6′(1’H)-one having the following chemical formula:
or use the synthetic precursor thereof named 2-methoxy-5-(pyridin-2-yl)pyridine or 6′-methoxy-2,3′-bipyridine having the formula:
2,3′-bipyridin-6′(1’H)-one is in fact prepared by simple acid-catalysed demethylation of the 6′-methoxy-2,3′-bipyridine as thoroughly reported in the prior art.
-
[0006]Various ways of synthesising 2-methoxy-5-(pyridin-2-yl)pyridine are known. The process summarised in the diagram (I) below is described in the publication WO 2001096308 :
Diagram (I)
-
[0007]
-
[0008]Such process highlights clear disadvantages such as the need to operate in cryogenic conditions (T=-78°C) using special equipment and the need to isolate the boronic acid via work-up; in addition the use of 2-Bromopyridine is envisaged, which is less convenient as regards the production of waste compared to 2-chloropyridine.
-
[0009]Another process described in WO 2004009553 is summarised in the diagram (II) :
Diagram (II)
-
[0010]
-
[0011]It presents clear disadvantages such as the use of high molecular weight benzenesulfonyl pyridine entailing a scarce atom-economy of the process and the need to operate at low temperature T (-78°C) using special equipment.
-
[0012]Lastly, a completely different process is described in WO20087093392for the preparation of 2,3′-bipyridin-6′(1’H)-one which however does not include the preparation of the intermediate precursor named 2-methoxy-5-(pyridin-2-yl)pyridine, process shown in the diagram (III) :
diagram (III)
-
[0013]

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....