Home » Posts tagged 'peptide'
Tag Archives: peptide
Nirsevimab
(Heavy chain)
QVQLVQSGAE VKKPGSSVMV SCQASGGLLE DYIINWVRQA PGQGPEWMGG IIPVLGTVHY
GPKFQGRVTI TADESTDTAY MELSSLRSED TAMYYCATET ALVVSETYLP HYFDNWGQGT
LVTVSSASTK GPSVFPLAPS SKSTSGGTAA LGCLVKDYFP EPVTVSWNSG ALTSGVHTFP
AVLQSSGLYS LSSVVTVPSS SLGTQTYICN VNHKPSNTKV DKRVEPKSCD KTHTCPPCPA
PELLGGPSVF LFPPKPKDTL YITREPEVTC VVVDVSHEDP EVKFNWYVDG VEVHNAKTKP
REEQYNSTYR VVSVLTVLHQ DWLNGKEYKC KVSNKALPAP IEKTISKAKG QPREPQVYTL
PPSREEMTKN QVSLTCLVKG FYPSDIAVEW ESNGQPENNY KTTPPVLDSD GSFFLYSKLT
VDKSRWQQGN VFSCSVMHEA LHNHYTQKSL SLSPGK
(Light chain)
DIQMTQSPSS LSAAVGDRVT ITCQASQDIV NYLNWYQQKP GKAPKLLIYV ASNLETGVPS
RFSGSGSGTD FSLTISSLQP EDVATYYCQQ YDNLPLTFGG GTKVEIKRTV AAPSVFIFPP
SDEQLKSGTA SVVCLLNNFY PREAKVQWKV DNALQSGNSQ ESVTEQDSKD STYSLSSTLT
LSKADYEKHK VYACEVTHQG LSSPVTKSFN RGEC
(Disulfide bridge: H22-H96, H153-H209, H229-L214, H235-H’235, H238-H’238, H270-H330, H376-H434, H’22-H’96, H’153-H’209, H’229-L’214, H’270-H’330, H’376-H’434, L23-L88, L’23-L’88, L134-L194, L’134-L’194)
>Heavy_chain QVQLVQSGAEVKKPGSSVMVSCQASGGLLEDYIINWVRQAPGQGPEWMGGIIPVLGTVHY GPKFQGRVTITADESTDTAYMELSSLRSEDTAMYYCATETALVVSETYLPHYFDNWGQGT LVTVSSASTKGPSVFPLAPSSKSTSGGTAALGCLVKDYFPEPVTVSWNSGALTSGVHTFP AVLQSSGLYSLSSVVTVPSSSLGTQTYICNVNHKPSNTKVDKRVEPKSCDKTHTCPPCPA PELLGGPSVFLFPPKPKDTLYITREPEVTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKP REEQYNSTYRVVSVLTVLHQDWLEGKEYKCKVSNKALPAPIEKTISKAKGQPREPQVYTL PPSREEMTKNQVSLTCLVKGFYPSDIAVEWESNGQPENNYKTTPPVLDSDGSFFLYSKLT VDKSRWQQGNVFSCSVMHEALHNHYTQKSLSLSPGK
>Light_chain DIQMTQSPSSLSAAVGDRVTITCQASQDIVNYLNWYQQKPGKAPKLLIYVASNLETGVPS RFSGSGSGTDFSLTISSLQPEDVATYYCQQYDNLPLTFGGGTKVEIKRTVAAPSVFIFPP SDEQLKSGTASVVCLLNNFYPREAKVQWKVDNALQSGNSQESVTEQDSKDSTYSLSSTLT LSKADYEKHKVYACEVTHQGLSSPVTKSFNRGEC
Nirsevimab
EMS APPROVED 2022/10/31, Beyfortus, AstraZeneca AB
| Formula | C6494H10060N1708O2050S46 |
|---|---|
| CAS | 1989556-22-0 |
| Mol weight | 146334.5658 |
Monoclonal antibody
Prevention of respiratory syncytial virus infection
- Immunoglobulin g1-kappa, anti-(human respiratory syncytial virus fusion glycoprotein f0 (protein f))human monoclonal antibody.gamma.1 heavy chain (1-456) (human vh (homo sapiens ighv1-69*01(ighd)-ighj4*01 (90.1%)) (8.8.19) (1-126) -homo sapiens ighg1*03
- Immunoglobulin g1, anti-(human respiratory syncytial virus fusion protein)(human monoclonal med18897 .gamma.1-chain), disulfide with monoclonal med18897 .kappa.-chain, dimer
Synthesis Reference
Khan, AA et al. (2020) Dosage regimens for and compositions including anti-rsv antibodies. (U.S. Patent No. 2020/0347120 A1). U.S. Patent and Trademark Office. https://patentimages.storage.googleapis.com/6b/d2/10/a841b66e0c90cf/US20200347120A1.pdf
Nirsevimab, sold under the brand name Beyfortus, is a human recombinant monoclonal antibody with activity against respiratory syncytial virus, or RSV for infants.[2][3] It is under development by AstraZeneca and Sanofi.[2][3] Nirsevimab is designed to bind to the fusion protein on the surface of the RSV virus.[4][5]
The most common side effects reported for nirsevimab are rash, pyrexia (fever) and injection site reactions (such as redness, swelling and pain where the injection is given).[6]
Nirsevimab was approved for medical use in the European Union in November 2022.[1][7]
Nirsevimab (MEDI8897) is a recombinant human immunoglobulin G1 kappa (IgG1ĸ) monoclonal antibody used to prevent respiratory syncytial virus (RSV) lower respiratory tract disease in neonates and infants.6 It binds to the prefusion conformation of the RSV F protein, a glycoprotein involved in the membrane fusion step of the viral entry process, and neutralizes several RSV A and B strains.6,1 Compared to palivizumab, another anti-RSV antibody, nirsevimab shows greater potency at reducing pulmonary viral loads in animal models. In addition, nirsevimab was developed as a single-dose treatment for all infants experiencing their first RSV season, whereas palivizumab requires five monthly doses to cover an RSV season.5 This is due to a modification in the Fc region of nirsevimab that grants it a longer half-time compared to typical monoclonal antibodies.1,6
On November 2022, nirsevimab was approved by the EMA for the prevention of RSV lower respiratory tract disease in newborns and infants during their first RSV season.6
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Human |
| Target | F protein of RSV |
| Clinical data | |
| Trade names | Beyfortus |
| Other names | MED-18897, MEDI8897 |
| Routes of administration | Intramuscular |
| ATC code | None |
| Legal status | |
| Legal status | EU: Rx-only [1] |
| Identifiers | |
| CAS Number | 1989556-22-0 |
| PubChem SID | 384585358 |
| DrugBank | DB16258 |
| UNII | VRN8S9CW5V |
| KEGG | D11380 |
| ChEMBL | ChEMBL4297575 |
| Chemical and physical data | |
| Formula | C6494H10060N1708O2050S46 |
| Molar mass | 146336.58 g·mol−1 |
Adverse effects
No major hypersensitivity reactions have been reported, and adverse events of grade 3 or higher were only reported in 8% (77 of 968) of participants in clinical trial NCT02878330.[8][4]
Pharmacology
Mechanism of action
Nirsevimab binds to the prefusion conformation of the RSV fusion protein, i.e. it binds to the site at which the virus would attach to a cell; effectively rendering it useless. It has a modified Fc region, extending the half-life of the drug in order for it to last the whole RSV season.[4]
History
The opinion by the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) is based on data from two randomized, double-blind, placebo-controlled multicenter clinical trials that investigated the efficacy and safety of nirsevimab in healthy preterm (premature) and full-term infants entering their first respiratory syncytial virus (RSV) season.[6] These studies demonstrated that nirsevimab prevents lower respiratory tract infection caused by RSV requiring medical attention (such as bronchiolitis and pneumonia) in term and preterm infants during their first RSV season.[6]
The safety of nirsevimab was also evaluated in a phase II/III, randomized, double‑blind, multicenter trial in infants who were born five or more weeks prematurely (less than 35 weeks gestation) at higher risk for severe RSV disease and infants with chronic lung disease of prematurity (i.e. long-term respiratory problems faced by babies born prematurely) or congenital heart disease.[6] The results of this study showed that nirsevimab had a similar safety profile compared to palivizumab (Synagis).[6]
Society and culture
Legal status
On 15 September 2022, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Beyfortus, intended for the prevention of respiratory syncytial virus (RSV) lower respiratory tract disease in newborns and infants.[9][6] Beyfortus was reviewed under EMA’s accelerated assessment program.[9] The applicant for this medicinal product is AstraZeneca AB.[9] Nirsevimab was approved for medical use in the European Union in November 2022.[1][7]
Research
Nirsevimab is being investigated as an experimental vaccine against respiratory syncytial virus, RSV, in the general infant population.[2][3] The MELODY study is an ongoing, randomized, double-blind, placebo-controlled to evaluate the safety and efficacy of nirsevimab in late preterm and term infants. Initial results have been promising, with nirsevimab reducing LRTI (lower respiratory tract infections) by 74.5% compared to placebo in infants born at term or late preterm.[5][10][11]
Ongoing trials for nirsevimab are:
- “Evaluate the Safety and Efficacy of Nirsevimab in Healthy Preterm and Term Infants in China (CHIMES)”.
- “A Study to Evaluate the Safety and Efficacy of MEDI8897 for the Prevention of Medically Attended Lower Respiratory Tract Infection Due to Respiratory Syncytial Virus in Healthy Late Preterm and Term Infants (MELODY)”.
- “Evaluate the Safety and Tolerability, for Nirsevimab in Immunocompromised Children (MUSIC)”.
References
- ^ Jump up to:a b c “Beyfortus”. Union Register of medicinal products. 3 November 2022. Retrieved 6 November 2022.
- ^ Jump up to:a b c “Nirsevimab demonstrated protection against respiratory syncytial virus disease in healthy infants in Phase 3 trial” (Press release). Sanofi. 26 April 2021. Archived from the original on 27 December 2021. Retrieved 27 December 2021.
- ^ Jump up to:a b c “Nirsevimab MELODY Phase III trial met primary endpoint of reducing RSV lower respiratory tract infections in healthy infants” (Press release). AstraZeneca. 26 April 2021. Archived from the original on 26 December 2021. Retrieved 27 December 2021.
- ^ Jump up to:a b c Griffin MP, Yuan Y, Takas T, Domachowske JB, Madhi SA, Manzoni P, et al. (Nirsevimab Study Group) (July 2020). “Single-Dose Nirsevimab for Prevention of RSV in Preterm Infants”. The New England Journal of Medicine. 383 (5): 415–425. doi:10.1056/NEJMoa1913556. PMID 32726528. S2CID 220876651.
- ^ Jump up to:a b Hammitt LL, Dagan R, Yuan Y, Baca Cots M, Bosheva M, Madhi SA, et al. (March 2022). “Nirsevimab for Prevention of RSV in Healthy Late-Preterm and Term Infants”. The New England Journal of Medicine. 386 (9): 837–846. doi:10.1056/NEJMoa2110275. PMID 35235726. S2CID 247220023.
- ^ Jump up to:a b c d e f “New medicine to protect babies and infants from respiratory syncytial virus (RSV) infection”. European Medicines Agency (EMA) (Press release). 16 September 2022. Archived from the original on 19 September 2022. Retrieved 18 September 2022. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ Jump up to:a b “Beyfortus approved in the EU for the prevention of RSV lower respiratory tract disease in infants”. AstraZeneca (Press release). 4 November 2022. Retrieved 6 November 2022.
- ^ Clinical trial number NCT02878330 at ClinicalTrials.gov
- ^ Jump up to:a b c “Beyfortus: Pending EC decision”. European Medicines Agency (EMA). 15 September 2022. Archived from the original on 19 September 2022. Retrieved 18 September 2022. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ Zacks Equity Research (25 March 2022). “Pfizer’s (PFE) RSV Jab Gets Another Breakthrough Therapy Tag”. Nasdaq. Archived from the original on 8 April 2022. Retrieved 8 April 2022.
- ^ “Nirsevimab significantly protected infants against RSV disease in Phase III MELODY trial”. AstraZeneca (Press release). 3 March 2022. Retrieved 6 November 2022.
////////////Nirsevimab, EU 2022, APPROVALS 2022, PEPTIDE, Monoclonal antibody, respiratory syncytial virus infection, ANTIVIRAL, 1989556-22-0, MED-18897, MEDI8897, AstraZeneca AB

NEW DRUG APPROVALS
ONE TIME
$10.00
Tremelimumab
(Light chain)
DIQMTQSPSS LSASVGDRVT ITCRASQSIN SYLDWYQQKP GKAPKLLIYA ASSLQSGVPS
RFSGSGSGTD FTLTISSLQP EDFATYYCQQ YYSTPFTFGP GTKVEIKRTV AAPSVFIFPP
SDEQLKSGTA SVVCLLNNFY PREAKVQWKV DNALQSGNSQ ESVTEQDSKD STYSLSSTLT
LSKADYEKHK VYACEVTHQG LSSPVTKSFN RGEC
(Heavy chain)
QVQLVESGGG VVQPGRSLRL SCAASGFTFS SYGMHWVRQA PGKGLEWVAV IWYDGSNKYY
ADSVKGRFTI SRDNSKNTLY LQMNSLRAED TAVYYCARDP RGATLYYYYY GMDVWGQGTT
VTVSSASTKG PSVFPLAPCS RSTSESTAAL GCLVKDYFPE PVTVSWNSGA LTSGVHTFPA
VLQSSGLYSL SSVVTVPSSN FGTQTYTCNV DHKPSNTKVD KTVERKCCVE CPPCPAPPVA
GPSVFLFPPK PKDTLMISRT PEVTCVVVDV SHEDPEVQFN WYVDGVEVHN AKTKPREEQF
NSTFRVVSVL TVVHQDWLNG KEYKCKVSNK GLPAPIEKTI SKTKGQPREP QVYTLPPSRE
EMTKNQVSLT CLVKGFYPSD IAVEWESNGQ PENNYKTTPP MLDSDGSFFL YSKLTVDKSR
WQQGNVFSCS VMHEALHNHY TQKSLSLSPG K
(Disulfide bridge: L23-L88, L134-L194, L214-H139, H22-H96, H152-H208, H265-H325, H371-H429, H227-H’227, H228-H’228, H231-H’231, H234-H’234)

Fab fragment of tremelimumab (blue) binding CTLA-4 (green). From PDB entry 5GGV.
Tremelimumab
| Formula | C6500H9974N1726O2026S52 |
|---|---|
| CAS | 745013-59-6 |
| Mol weight | 146380.4722 |
FDA APPROVED2022/10/21, Imjudo
PEPTIDE, CP 675206
| Antineoplastic, Immune checkpoint inhibitor, Anti-CTLA4 antibody | |
| Disease | Hepatocellular carcinoma |
|---|
Tremelimumab (formerly ticilimumab, CP-675,206) is a fully human monoclonal antibody against CTLA-4. It is an immune checkpoint blocker. Previously in development by Pfizer,[1] it is now in investigation by MedImmune, a wholly owned subsidiary of AstraZeneca.[2] It has been undergoing human trials for the treatment of various cancers but has not attained approval for any.
Imjudo (tremelimumab) in combination with Imfinzi approved in the US for patients with unresectable liver cancer
PUBLISHED24 October 2022
24 October 2022 07:00 BST
Approval based on HIMALAYA Phase III trial results which showed single priming dose of Imjudo added to Imfinzi reduced risk of death by 22% vs. sorafenib
AstraZeneca’s Imjudo (tremelimumab) in combination with Imfinzi (durvalumab) has been approved in the US for the treatment of adult patients with unresectable hepatocellular carcinoma (HCC), the most common type of liver cancer. The novel dose and schedule of the combination, which includes a single dose of the anti-CTLA-4 antibody Imjudo 300mg added to the anti-PD-L1 antibody Imfinzi 1500mg followed by Imfinzi every four weeks, is called the STRIDE regimen (Single Tremelimumab Regular Interval Durvalumab).
The approval by the US Food and Drug Administration (FDA) was based on positive results from the HIMALAYA Phase III trial. In this trial, patients treated with the combination of Imjudo and Imfinzi experienced a 22% reduction in the risk of death versus sorafenib (based on a hazard ratio [HR] of 0.78, 95% confidence interval [CI] 0.66-0.92 p=0.0035).1 Results were also published in the New England Journal of Medicine Evidence showing that an estimated 31% of patients treated with the combination were still alive after three years, with 20% of patients treated with sorafenib still alive at the same duration of follow-up.2
Liver cancer is the third-leading cause of cancer death and the sixth most commonly diagnosed cancer worldwide.3,4 It is the fastest rising cause of cancer-related deaths in the US, with approximately 36,000 new diagnoses each year.5,6
Ghassan Abou-Alfa, MD, MBA, Attending Physician at Memorial Sloan Kettering Cancer Center (MSK), and principal investigator in the HIMALAYA Phase III trial, said: “Patients with unresectable liver cancer are in need of well-tolerated treatments that can meaningfully extend overall survival. In addition to this regimen demonstrating a favourable three-year survival rate in the HIMALAYA trial, safety data showed no increase in severe liver toxicity or bleeding risk for the combination, important factors for patients with liver cancer who also have advanced liver disease.”
Dave Fredrickson, Executive Vice President, Oncology Business Unit, AstraZeneca, said: “With this first regulatory approval for Imjudo, patients with unresectable liver cancer in the US now have an approved dual immunotherapy treatment regimen that harnesses the potential of CTLA-4 inhibition in a unique combination with a PD-L1 inhibitor to enhance the immune response against their cancer.”
Andrea Wilson Woods, President & Founder, Blue Faery: The Adrienne Wilson Liver Cancer Foundation, said: “In the past, patients living with liver cancer had few treatment options and faced poor prognoses. With today’s approval, we are grateful and optimistic for new, innovative, therapeutic options. These new treatments can improve long-term survival for those living with unresectable hepatocellular carcinoma, the most common form of liver cancer. We appreciate the patients, their families, and the broader liver cancer community who continue to fight for new treatments and advocate for others.”
The safety profiles of the combination of Imjudo added to Imfinzi and for Imfinzi alone were consistent with the known profiles of each medicine, and no new safety signals were identified.
Regulatory applications for Imjudo in combination with Imfinzi are currently under review in Europe, Japan and several other countries for the treatment of patients with advanced liver cancer based on the HIMALAYA results.
Notes
Liver cancer
About 75% of all primary liver cancers in adults are HCC.3 Between 80-90% of all patients with HCC also have cirrhosis.7 Chronic liver diseases are associated with inflammation that over time can lead to the development of HCC.7
More than half of patients are diagnosed at advanced stages of the disease, often when symptoms first appear.8 A critical unmet need exists for patients with HCC who face limited treatment options.8 The unique immune environment of liver cancer provides clear rationale for investigating medications that harness the power of the immune system to treat HCC.8
HIMALAYA
HIMALAYA was a randomised, open-label, multicentre, global Phase III trial of Imfinzi monotherapy and a regimen comprising a single priming dose of Imjudo 300mg added to Imfinzi 1500mg followed by Imfinzi every four weeks versus sorafenib, a standard-of-care multi-kinase inhibitor.
The trial included a total of 1,324 patients with unresectable, advanced HCC who had not been treated with prior systemic therapy and were not eligible for locoregional therapy (treatment localised to the liver and surrounding tissue).
The trial was conducted in 181 centres across 16 countries, including in the US, Canada, Europe, South America and Asia. The primary endpoint was overall survival (OS) for the combination versus sorafenib and key secondary endpoints included OS for Imfinzi versus sorafenib, objective response rate and progression-free survival (PFS) for the combination and for Imfinzi alone.
Imfinzi
Imfinzi (durvalumab) is a human monoclonal antibody that binds to the PD-L1 protein and blocks the interaction of PD-L1 with the PD-1 and CD80 proteins, countering the tumour’s immune-evading tactics and releasing the inhibition of immune responses.
Imfinzi was recently approved to treat patients with advanced biliary tract cancer in the US based on results from the TOPAZ-1 Phase III trial. It is the only approved immunotherapy in the curative-intent setting of unresectable, Stage III non-small cell lung cancer (NSCLC) in patients whose disease has not progressed after chemoradiotherapy and is the global standard of care in this setting based on the PACIFIC Phase III trial.
Imfinzi is also approved in the US, EU, Japan, China and many other countries around the world for the treatment of extensive-stage small cell lung cancer (ES-SCLC) based on the CASPIAN Phase III trial. In 2021, updated results from the CASPIAN trial showed Imfinzi plus chemotherapy tripled patient survival at three years versus chemotherapy alone.
Imfinzi is also approved for previously treated patients with advanced bladder cancer in several countries.
Since the first approval in May 2017, more than 100,000 patients have been treated with Imfinzi.
As part of a broad development programme, Imfinzi is being tested as a single treatment and in combinations with other anti-cancer treatments for patients with SCLC, NSCLC, bladder cancer, several gastrointestinal (GI) cancers, ovarian cancer, endometrial cancer, and other solid tumours.
Imfinzi combinations have also demonstrated clinical benefit in metastatic NSCLC in the POSEIDON Phase III trial.
Imjudo
Imjudo (tremelimumab) is a human monoclonal antibody that targets the activity of cytotoxic T-lymphocyte-associated protein 4 (CTLA-4). Imjudo blocks the activity of CTLA-4, contributing to T-cell activation, priming the immune response to cancer and fostering cancer cell death.
Beyond HIMALAYA, Imjudo is being tested in combination with Imfinzi across multiple tumour types including locoregional HCC (EMERALD-3), SCLC (ADRIATIC) and bladder cancer (VOLGA and NILE).
Imjudo is also under review by global regulatory authorities in combination with Imfinzi and chemotherapy in 1st-line metastatic NSCLC based on the results of the POSEIDON Phase III trial, which showed the addition of a short course of Imjudo to Imfinzi plus chemotherapy improved both overall and progression-free survival compared to chemotherapy alone.
AstraZeneca in GI cancers
AstraZeneca has a broad development programme for the treatment of GI cancers across several medicines spanning a variety of tumour types and stages of disease. In 2020, GI cancers collectively represented approximately 5.1 million new diagnoses leading to approximately 3.6 million deaths.9
Within this programme, the Company is committed to improving outcomes in gastric, liver, biliary tract, oesophageal, pancreatic, and colorectal cancers.
Imfinzi (durvalumab) is being assessed in combinations in oesophageal and gastric cancers in an extensive development programme spanning early to late-stage disease across settings.
The Company aims to understand the potential of Enhertu (trastuzumab deruxtecan), a HER2-directed antibody drug conjugate, in the two most common GI cancers, colorectal and gastric cancers. Enhertu is jointly developed and commercialised by AstraZeneca and Daiichi Sankyo.
Lynparza (olaparib) is a first-in-class PARP inhibitor with a broad and advanced clinical trial programme across multiple GI tumour types including pancreatic and colorectal cancers. Lynparza is developed and commercialised in collaboration with MSD (Merck & Co., Inc. inside the US and Canada).
AstraZeneca in immuno-oncology (IO)
Immunotherapy is a therapeutic approach designed to stimulate the body’s immune system to attack tumours. The Company’s immuno-oncology (IO) portfolio is anchored in immunotherapies that have been designed to overcome evasion of the anti-tumour immune response. AstraZeneca is invested in using IO approaches that deliver long-term survival for new groups of patients across tumour types.
The Company is pursuing a comprehensive clinical trial programme that includes Imfinzi as a single treatment and in combination with Imjudo (tremelimumab) and other novel antibodies in multiple tumour types, stages of disease, and lines of treatment, and where relevant using the PD-L1 biomarker as a decision-making tool to define the best potential treatment path for a patient.
In addition, the ability to combine the IO portfolio with radiation, chemotherapy, and targeted small molecules from across AstraZeneca’s oncology pipeline, and from research partners, may provide new treatment options across a broad range of tumours.
AstraZeneca in oncology
AstraZeneca is leading a revolution in oncology with the ambition to provide cures for cancer in every form, following the science to understand cancer and all its complexities to discover, develop and deliver life-changing medicines to patients.
The Company’s focus is on some of the most challenging cancers. It is through persistent innovation that AstraZeneca has built one of the most diverse portfolios and pipelines in the industry, with the potential to catalyse changes in the practice of medicine and transform the patient experience.
AstraZeneca has the vision to redefine cancer care and, one day, eliminate cancer as a cause of death.
////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
Mechanism of action
Tremelimumab aims to stimulate an immune system attack on tumors. Cytotoxic T lymphocytes (CTLs) can recognize and destroy cancer cells. However, there is also an inhibitory mechanism (immune checkpoint) that interrupts this destruction. Tremelimumab turns off this inhibitory mechanism and allows CTLs to continue to destroy the cancer cells.[3] This is immune checkpoint blockade.
Tremelimumab binds to the protein CTLA-4, which is expressed on the surface of activated T lymphocytes and inhibits the killing of cancer cells. Tremelimumab blocks the binding of the antigen-presenting cell ligands B7.1 and B7.2 to CTLA-4, resulting in inhibition of B7-CTLA-4-mediated downregulation of T-cell activation; subsequently, B7.1 or B7.2 may interact with another T-cell surface receptor protein, CD28, resulting in a B7-CD28-mediated T-cell activation unopposed by B7-CTLA-4-mediated inhibition.
Unlike Ipilimumab (another fully human anti-CTLA-4 monoclonal antibody), which is an IgG1 isotype, tremelimumab is an IgG2 isotype.[4][5]
Clinical trials
Melanoma
Phase 1 and 2 clinical studies in metastatic melanoma showed some responses.[6] However, based on early interim analysis of phase III data, Pfizer designated tremelimumab as a failure and terminated the trial in April 2008.[1][7]
However, within a year, the survival curves showed separation of the treatment and control groups.[8] The conventional Response Evaluation Criteria in Solid Tumors (RECIST) may underrepresent the merits of immunotherapies. Subsequent immunotherapy trials (e.g. ipilimumab) have used the Immune-Related Response Criteria (irRC) instead.
Mesothelioma
Although it was designated in April 2015 as orphan drug status in mesothelioma,[9] tremelimumab failed to improve lifespan in the phase IIb DETERMINE trial, which assessed the drug as a second or third-line treatment for unresectable malignant mesothelioma.[10][11]
Non-small cell lung cancer
In a phase III trial, AstraZeneca paired tremelimumab with a PD-L1 inhibitor, durvalumab, for the first-line treatment of non-small cell lung cancer.[12] The trial was conducted across 17 countries, and in July 2017, AstraZeneca announced that it had failed to meet its primary endpoint of progression-free survival.[13]
References
- ^ Jump up to:a b “Pfizer Announces Discontinuation of Phase III Clinical Trial for Patients with Advanced Melanoma”. Pfizer.com. 1 April 2008. Retrieved 5 December 2015.
- ^ Mechanism of Pathway: CTLA-4 Inhibition[permanent dead link]
- ^ Antoni Ribas (28 June 2012). “Tumor immunotherapy directed at PD-1”. New England Journal of Medicine. 366 (26): 2517–9. doi:10.1056/nejme1205943. PMID 22658126.
- ^ Tomillero A, Moral MA (October 2008). “Gateways to clinical trials”. Methods Find Exp Clin Pharmacol. 30 (8): 643–72. doi:10.1358/mf.2008.30.5.1236622. PMID 19088949.
- ^ Poust J (December 2008). “Targeting metastatic melanoma”. Am J Health Syst Pharm. 65 (24 Suppl 9): S9–S15. doi:10.2146/ajhp080461. PMID 19052265.
- ^ Reuben, JM; et al. (1 Jun 2006). “Biologic and immunomodulatory events after CTLA-4 blockade with tremelimumab in patients with advanced malignant melanoma”. Cancer. 106 (11): 2437–44. doi:10.1002/cncr.21854. PMID 16615096. S2CID 751366.
- ^ A. Ribas, A. Hauschild, R. Kefford, C. J. Punt, J. B. Haanen, M. Marmol, C. Garbe, J. Gomez-Navarro, D. Pavlov and M. Marsha (May 20, 2008). “Phase III, open-label, randomized, comparative study of tremelimumab (CP-675,206) and chemotherapy (temozolomide [TMZ] or dacarbazine [DTIC]) in patients with advanced melanoma”. Journal of Clinical Oncology. 26 (15S): LBA9011. doi:10.1200/jco.2008.26.15_suppl.lba9011.[permanent dead link]
- ^ M.A. Marshall, A. Ribas, B. Huang (May 2010). “Evaluation of baseline serum C-reactive protein (CRP) and benefit from tremelimumab compared to chemotherapy in first-line melanoma”. Journal of Clinical Oncology. 28 (15S): 2609. doi:10.1200/jco.2010.28.15_suppl.2609.[permanent dead link]
- ^ FDA Grants AstraZeneca’s Tremelimumab Orphan Drug Status for Mesothelioma [1]
- ^ “Tremelimumab Fails Mesothelioma Drug Trial”. Archived from the original on 2016-03-06. Retrieved 2016-03-06.
- ^ AZ’ tremelimumab fails in mesothelioma trial
- ^ “AstraZeneca’s immuno-oncology combo fails crucial Mystic trial in lung cancer | FierceBiotech”.
- ^ “AstraZeneca reports initial results from the ongoing MYSTIC trial in Stage IV lung cancer”.
///////////Tremelimumab, Imjudo, APPROVALS 2022, FDA 2022, PEPTIDE, CP 675206, Antineoplastic, Immune checkpoint inhibitor, Anti-CTLA4 antibody

NEW DRUG APPROVALS
ONE TIME
$10.00
Spesolimab
(Heavy chain)
QVQLVQSGAE VKKPGASVKV SCKASGYSFT SSWIHWVKQA PGQGLEWMGE INPGNVRTNY
NENFRNKVTM TVDTSISTAY MELSRLRSDD TAVYYCTVVF YGEPYFPYWG QGTLVTVSSA
STKGPSVFPL APSSKSTSGG TAALGCLVKD YFPEPVTVSW NSGALTSGVH TFPAVLQSSG
LYSLSSVVTV PSSSLGTQTY ICNVNHKPSN TKVDKRVEPK SCDKTHTCPP CPAPEAAGGP
SVFLFPPKPK DTLMISRTPE VTCVVVDVSH EDPEVKFNWY VDGVEVHNAK TKPREEQYNS
TYRVVSVLTV LHQDWLNGKE YKCKVSNKAL PAPIEKTISK AKGQPREPQV YTLPPSREEM
TKNQVSLTCL VKGFYPSDIA VEWESNGQPE NNYKTTPPVL DSDGSFFLYS KLTVDKSRWQ
QGNVFSCSVM HEALHNHYTQ KSLSLSPGK
(Light chain)
QIVLTQSPGT LSLSPGERAT MTCTASSSVS SSYFHWYQQK PGQAPRLWIY RTSRLASGVP
DRFSGSGSGT DFTLTISRLE PEDAATYYCH QFHRSPLTFG AGTKLEIKRT VAAPSVFIFP
PSDEQLKSGT ASVVCLLNNF YPREAKVQWK VDNALQSGNS QESVTEQDSK DSTYSLSSTL
TLSKADYEKH KVYACEVTHQ GLSSPVTKSF NRGEC
(Disulfide bridge: H22-H96, H146-H202, H222-L215, H228-H’228, H231-H’231, H263-H323, H369-H427, H’22-H’96, H’146-H’202, H’222-L’215, H’263-H’323, H’369-H’427, L23-L89, L135-L195, L’23-L’89, L’135-L’195)
Spesolimab
スペソリマブ (遺伝子組換え)
| Formula | C6480H9988N1736O2012S46 |
|---|---|
| cas | 2097104-58-8 |
| Mol weight | 145878.0547 |
| Antipsoriatic, Anti-IL-36 receptor antagonist |
fda approved 2022/9/1, spevigo
BI 655130; Spesolimab-sbzo
- OriginatorBoehringer Ingelheim
- ClassAnti-inflammatories; Antipsoriatics; Monoclonal antibodies; Skin disorder therapies
- Mechanism of ActionInterleukin 36 receptor antagonists
- Orphan Drug StatusYes – Generalised pustular psoriasis
- RegisteredGeneralised pustular psoriasis
- Phase II/IIIUlcerative colitis
- Phase IICrohn’s disease; Hidradenitis suppurativa; Palmoplantar pustulosis
- DiscontinuedAtopic dermatitis
- 01 Sep 2022First global approval – Registered for Generalised pustular psoriasis in USA (IV)
- 01 Sep 2022Adverse events data from the Effisayil 1 phase II trial in Generalised pustular psoriasis released by Boehringer Ingelheim
- 03 Aug 2022Boehringer Ingelheim anticipates regulatory approval in Generalised pustular psoriasis by 2022
Spesolimab (BI 655130) is a humanised monoclonal antibody, being developed by Boehringer Ingelheim, for the treatment of generalised pustular psoriasis, Crohn’s disease, palmoplantar pustulosis, ulcerative colitis and hidradenitis suppurativa.
What causes Palmoplantar Pustulosis?
Researchers have found some possible causes including smoking, infections, certain medications and genetics. Smoking: Many patients who have PPP are smokers or have smoked in the past. Smoking may cause sweat glands to become inflamed, especially on the hands and feet, which causes pustules to form.
FDA approves the first treatment option for generalized pustular psoriasis flares in adults
- More than half of patients treated with SPEVIGO® (spesolimab-sbzo) injection, for intravenous use showed no visible pustules one week after receiving treatment
- Spesolimab is a monoclonal antibody that inhibits interleukin-36 (IL-36) signaling
Ridgefield, Conn., September 1, 2022 – Boehringer Ingelheim announced today the U.S. Food and Drug Administration has approved SPEVIGO, the first approved treatment option for generalized pustular psoriasis (GPP) flares in adults. SPEVIGO is a novel, selective antibody that blocks the activation of the interleukin-36 receptor (IL-36R), a key part of a signaling pathway within the immune system shown to be involved in the cause of GPP.
“GPP flares can greatly impact a patient’s life and lead to serious, life-threatening complications,” said Mark Lebwohl, M.D., lead investigator and publication author, and Dean for Clinical Therapeutics, Icahn School of Medicine at Mount Sinai, Kimberly and Eric J. Waldman Department of Dermatology, New York. “The approval of SPEVIGO is a turning point for dermatologists and clinicians. We now have an FDA-approved treatment that may help make a difference for our patients who, until now, have not had any approved options to help manage GPP flares.”
Distinct from plaque psoriasis, GPP is a rare and potentially life-threatening neutrophilic skin disease, which is characterized by flares (episodes of widespread eruptions of painful, sterile pustules). In the United States, it is estimated that 1 out of every 10,000 people has GPP. Given that it is so rare, recognizing the signs and symptoms can be challenging and consequently lead to delays in diagnosis.
“This important approval reflects our successful efforts to accelerate our research with the aim to bring innovative treatments faster to the people most in need,” said Carinne Brouillon, Member of the Board of Managing Directors, responsible for Human Pharma, Boehringer Ingelheim. “We recognize how devastating this rare skin disease can be for patients, their families and caregivers. GPP can be life-threatening and until today there have been no specific approved therapies for treating the devastating GPP flares. It makes me proud that with the approval of SPEVIGO we can now offer the first U.S. approved treatment option for those in need.”
In the 12-week pivotal Effisayil™ 1 clinical trial, patients experiencing a GPP flare (N=53) were treated with SPEVIGO or placebo. After one week, patients treated with SPEVIGO showed no visible pustules (54%) compared to placebo (6%).
In Effisayil™ 1, the most common adverse reactions (≥5%) in patients that received SPEVIGO were asthenia and fatigue, nausea and vomiting, headache, pruritus and prurigo, infusion site hematoma and bruising, and urinary tract infection.
“GPP can have an enormous impact on patients’ physical and emotional wellbeing. With the FDA approval of this new treatment, people living with GPP now have hope in knowing that there is an option to help treat their flares,” said Thomas Seck, M.D., Senior Vice President, Medicine and Regulatory Affairs, Boehringer Ingelheim. “SPEVIGO represents Boehringer Ingelheim’s commitment to delivering meaningful change for patients living with serious diseases with limited treatment options.”
About SPEVIGO
SPEVIGO is indicated for the treatment of GPP flares in adults. SPEVIGO is contraindicated in patients with severe or life-threatening hypersensitivity to spesolimab-sbzo or to any of the excipients in SPEVIGO. Reactions have included drug reaction with eosinophilia and systemic symptoms (DRESS).
What is SPEVIGO?
SPEVIGO is a prescription medicine used to treat generalized pustular psoriasis (GPP) flares in adults. It is not known if SPEVIGO is safe and effective in children.
U.S. FDA grants Priority Review for spesolimab for the treatment of flares in patients with generalized pustular psoriasis (GPP), a rare, life-threatening skin disease
December 15, 2021 – Boehringer Ingelheim today announced that the U.S. Food and Drug Administration (FDA) has accepted a Biologics License Application (BLA) and granted Priority Review for spesolimab for the treatment of generalized pustular psoriasis (GPP) flares.
FDA grants Priority Review to applications for medicines that, if approved, would offer significant improvement over available options in the safety or effectiveness of the treatment, diagnosis, or prevention of serious conditions. The FDA has granted spesolimab Orphan Drug Designation for the treatment of GPP, and Breakthrough Therapy Designation for spesolimab for the treatment of GPP flares in adults.
“The FDA acceptance of our filing for spesolimab is a critical step in our efforts to bring this first-in-class treatment to people living with GPP,” said Matt Frankel, M.D., Vice President, Clinical Development and Medical Affairs, Specialty Care, Boehringer Ingelheim. “There is an urgent unmet need for an approved treatment option that can rapidly clear painful GPP flares.”
GPP is a rare, life-threatening neutrophilic skin disease, which is distinct from plaque psoriasis. It is characterized by episodes of widespread eruptions of painful, sterile pustules (blisters of non-infectious pus). There is a high unmet need for treatments that can rapidly and completely resolve the signs and symptoms of GPP flares. Flares greatly affect a person’s quality of life and can lead to hospitalization with serious complications, including heart failure, renal failure, sepsis, and death.
About spesolimab
Spesolimab is a novel, humanized, selective antibody that blocks the activation of the interleukin-36 receptor (IL-36R), a signaling pathway within the immune system shown to be involved in the pathogeneses of several autoimmune diseases, including GPP. Spesolimab is also under investigation for the prevention of GPP flares and for the treatment of other neutrophilic skin diseases, such as palmoplantar pustulosis (PPP) and hidradenitis suppurativa (HS).
About generalized pustular psoriasis (GPP)
GPP is a rare, heterogenous and potentially life-threatening neutrophilic skin disease, which is clinically distinct from plaque psoriasis. GPP is caused by neutrophils (a type of white blood cell) accumulating in the skin, resulting in painful, sterile pustules all over the body. The clinical course varies, with some patients having a relapsing disease with recurrent flares, and others having a persistent disease with intermittent flares. While the severity of GPP flares can vary, if left untreated they can be life-threatening due to complications such as sepsis and multisystem organ failure. This chronic, systemic disease has a substantial quality of life impact for patients and healthcare burden. GPP has a varied prevalence across different geographical regions and more women are affected than men.
Boehringer Ingelheim Immunology: Pioneering Science, Inspired By Patients
Living with fibrotic and inflammatory diseases greatly impacts patients’ lives emotionally and physically. These patients are our guides, partners and inspiration as we redefine treatment paradigms. As a family-owned company, we can plan long-term. Our goal is to discover and develop first-of-their-kind therapies. With a deep understanding of molecular pathways, we are pioneering scientific breakthroughs that target, repair and prevent many fibrotic and inflammatory diseases. By building on long-term external collaborations, we strive to bring treatment breakthroughs to patients in the shortest time. We won’t rest until we can give people the chance to live the lives they want.
Boehringer Ingelheim
Boehringer Ingelheim is working on breakthrough therapies that improve the lives of humans and animals. As a leading research-driven biopharmaceutical company, the company creates value through innovation in areas of high unmet medical need. Founded in 1885 and family-owned ever since, Boehringer Ingelheim takes a long-term perspective. Around 52,000 employees serve more than 130 markets in the three business areas, Human Pharma, Animal Health, and Biopharmaceutical Contract Manufacturing. Learn more at www.boehringer-ingelheim.com.
MPR-US-101971
////////Spesolimab, monoclonal antibody, fda 2022, approvals 2022, Orphan Drug Status, Generalised pustular psoriasis, BI 655130, Spesolimab-sbzo, peptide, monoclonal antibody

NEW DRUG APPROVALS
ONE TIME
$10.00
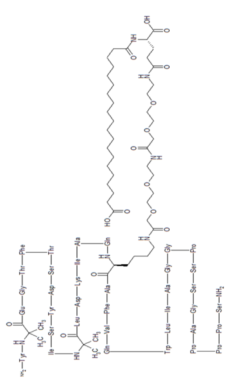
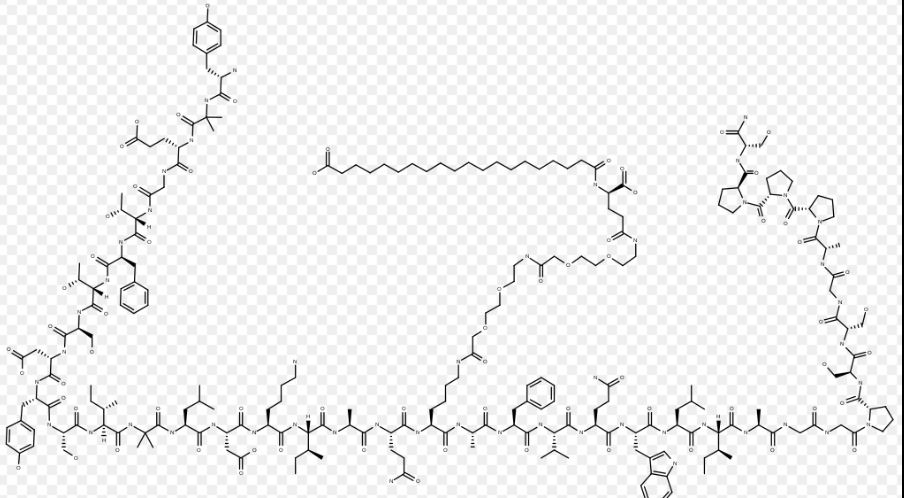
Tirzepatide
YXEGTFTSDY SIXLDKIAQK AFVQWLIAGG PSSGAPPPS



Tirzepatide
チルゼパチド
LY3298176,
| Formula | C225H348N48O68 |
|---|---|
| CAS | 2023788-19-2 |
| Mol weight | 4813.4514 |
FDA APPROVED 2022/5/13, Mounjaro
| Class | Antidiabetic agent GLP-1 receptor agonist |
|---|---|
| Efficacy | Antidiabetic, Gastric inhibitory polypeptide receptor agonist, Glucagon-like peptide 1 (GLP-1) receptor agonist |
| Disease | Type 2 diabetes mellitus |

Tirzepatide is an agonist of human glucose-dependent insulinotropic polypeptide (GIP) and human glucagon-like peptide-1 (GLP-1) receptors, whose amino acid residues at positions 2 and 13 are 2-methylAla, and the C-terminus is amidated Ser. A 1,20-icosanedioic acid is attached to Lys at position 20 via a linker which consists of a Glu and two 8-amino-3,6-dioxaoctanoic acids. Tirzepatide is a synthetic peptide consisting of 39 amino acid residues.
C225H348N48O68 : 4813.45
[2023788-19-2]
L-Serinamide, L-tyrosyl-2-methylalanyl-L-α-glutamylglycyl-L-threonyl-L-phenylalanyl-L-threonyl-L-seryl-L-α-aspartyl-L-tyrosyl-L-seryl-L-isoleucyl-2-methylalanyl-L-leucyl-L-α-aspartyl-L-lysyl-L-isoleucyl-L-alanyl-L-glutaminyl-N6-[(22S)-22,42-dicarboxy-1,10,19,24-tetraoxo-3,6,12,15-tetraoxa-9,18,23-triazadotetracont-1-yl]-L-lysyl-L-alanyl-L-phenylalanyl-L-valyl-L-glutaminyl-L-tryptophyl-L-leucyl-L-isoleucyl-L-alanylglycylglycyl-L-prolyl-L-seryl-L-serylglycyl-L-alanyl-L-prolyl-L-prolyl-L-prolyl-
Other Names
- L-Tyrosyl-2-methylalanyl-L-α-glutamylglycyl-L-threonyl-L-phenylalanyl-L-threonyl-L-seryl-L-α-aspartyl-L-tyrosyl-L-seryl-L-isoleucyl-2-methylalanyl-L-leucyl-L-α-aspartyl-L-lysyl-L-isoleucyl-L-alanyl-L-glutaminyl-N6-[(22S)-22,42-dicarboxy-1,10,19,24-tetraoxo-3,6,12,15-tetraoxa-9,18,23-triazadotetracont-1-yl]-L-lysyl-L-alanyl-L-phenylalanyl-L-valyl-L-glutaminyl-L-tryptophyl-L-leucyl-L-isoleucyl-L-alanylglycylglycyl-L-prolyl-L-seryl-L-serylglycyl-L-alanyl-L-prolyl-L-prolyl-L-prolyl-L-serinamide
Tirzepatide, sold under the brand name Mounjaro,[1] is a medication used for the treatment type 2 diabetes.[2][3][4] Tirzepatide is given by injection under the skin.[2] Common side effects may include nausea, vomiting, diarrhea, decreased appetite, constipation, upper abdominal discomfort and abdominal pain.[2]
Glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) are hormones involved in blood sugar control.[2] Tirzepatide is a first-in-class medication that activates both the GLP-1 and GIP receptors, which leads to improved blood sugar control.[2] Tirzepatide was approved for medical use in the United States in May 2022.[2]
SYN
https://pubs.acs.org/doi/10.1021/acs.oprd.1c00108

The large-scale manufacture of complex synthetic peptides is challenging due to many factors such as manufacturing risk (including failed product specifications) as well as processes that are often low in both yield and overall purity. To overcome these liabilities, a hybrid solid-phase peptide synthesis/liquid-phase peptide synthesis (SPPS/LPPS) approach was developed for the synthesis of tirzepatide. Continuous manufacturing and real-time analytical monitoring ensured the production of high-quality material, while nanofiltration provided intermediate purification without difficult precipitations. Implementation of the strategy worked very well, resulting in a robust process with high yields and purity.
PATENT
- WO2016111971
- US2020023040
- WO2019245893
- US2020155487
- US2020155650
- WO2020159949CN112592387
- WO2021066600CN112661815
- WO2021154593
- US2021338769

NEW DRUG APPROVALS
ONE TIME TO MAINTAIN THIS BLOG SUBSCRIPTION
$10.00
//////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
Medical uses
Tirzepatide in indicated to improve blood sugar control in adults with type 2 diabetes, as an addition to diet and exercise.[2]
Contraindications
Tirzepatide should not be used in people with a personal or family history of medullary thyroid cancer or in people with multiple endocrine neoplasia syndrome type 2.[2]
Adverse effects
Preclinical, phase I, and phase II trials have indicated that tirzepatide exhibits similar adverse effects to other established GLP-1 receptor agonists, such as GLP-1 receptor agonist dulaglutide. These effects occur largely within the gastrointestinal tract.[5] The most frequently observed adverse effects are nausea, diarrhoea and vomiting, which increased in incidence with the dosage amount (i.e. higher likelihood the higher the dose). The number of patients who discontinued taking tirzepatide also increased as dosage increased, with patients taking 15 mg having a 25% discontinuation rate vs 5.1% for 5 mg patients and 11.1% for dulaglutide.[6] To a slightly lesser extent, patients also reported reduced appetite.[5] Other side effects reported were dyspepsia, constipation, abdominal pain, dizziness and hypoglycaemia.[7][8]
Pharmacology
Tirzepatide is an analogue of gastric inhibitory polypeptide (GIP), a human hormone which stimulates the release of insulin from the pancreas. Tirzepatide is a linear polypeptide of 39 amino acids which has been chemically modified by lipidation to improve its uptake into cells and its stability to metabolism.[9] The compound is administered as a weekly subcutaneous injection.[10] It completed phase III trials globally in 2021.[11][12]
Mechanism of action
Tirzepatide has a greater affinity to GIP receptors than to GLP-1 receptors, and this dual agonist behaviour has been shown to produce greater reductions of hyperglycemia compared to a selective GLP-1 receptor agonist.[3] Signaling studies have shown that this is due to tirzepatide mimicking the actions of natural GIP at the GIP receptor.[13] However, at the GLP-1 receptor, tirzepatide shows bias towards cAMP (a messenger associated with regulation of glycogen, sugar and lipid metabolism) generation, rather than β-arrestin recruitment. This combination of preference towards GIP receptor and distinct signaling properties at GLP-1 suggest this biased agonism increases insulin secretion.[13] Tirzepatide has also been shown to increase levels of adiponectin, an adipokine involved in the regulation of both glucose and lipid metabolism, with a maximum increase of 26% from baseline after 26 weeks, at the 10 mg dosage.[3]
Chemistry
Structure
Tirzepatide is an analog of the human GIP hormone with a C20 fatty-diacid portion attached, used to optimise the uptake and metabolism of the compound.[9] The fatty-diacid section (eicosanedioic acid) is linked via a glutamic acid and two (2-(2-aminoethoxy)ethoxy)acetic acid units to the side chain of the lysine residue. This arrangement allows for a much longer half life, extending the time between doses, because of its high affinity to albumin.[14]
Synthesis
The synthesis of tirzepatide was first disclosed in patents filed by Eli Lilly and Company.[15] This uses standard solid phase peptide synthesis, with an allyloxycarbonyl protecting group on the lysine at position 20 of the linear chain of amino acids, allowing a final set of chemical transformations in which the sidechain amine of that lysine is derivatized with the lipid-containing fragment.
Large-scale manufacturing processes have been reported for this compound.[16]
History
Indiana-based pharmaceutical company Eli Lilly and Company first applied for a patent for a method of glycemic control using tirzepatide in early 2016.[15] The patent was published late that year. After passing phase 3 clinical trials, Lilly applied for FDA approval in October 2021 with a priority review voucher.[17]
Following the completion of the pivotal SURPASS-2 trial no. NCT03987919, the company announced on 28 April that tirzepatide had successfully met their endpoints in obese and overweight patients without diabetes.[18] Alongside results from the SURMOUNT-1 trial no. NCT04184622, they suggest that tirzepatide may potentially be a competitor for existing diabetic medication semaglutide, manufactured by Novo Nordisk.[19][20]
In industry-funded preliminary trials comparing tirzepatide to the existing diabetes medication semaglutide (an injected analogue of the hormone GLP-1), tirzepatide showed minor improvement of reductions (2.01%–2.30% depending on dosage) in glycated hemoglobin tests relative to semaglutide (1.86%).[21] A 10 mg dose has also been shown to be effective in reducing insulin resistance, with a reduction of around 8% from baseline, measured using HOMA2-IR (computed with fasting insulin).[3] Fasting levels of IGF binding proteins like IGFBP1 and IGFBP2 increased following tirzepatide treatment, increasing insulin sensitivity.[3] A meta-analysis published by Dutta et al. showed that over 1-year clinical use, tirzepatide was observed to be superior to dulaglutide, semaglutide, degludec, and insulin glargine with regards to glycemic efficacy and obesity reduction. Tirzepatide is perhaps the most potent agent developed to date to tackle the global problem of “diabesity“.[22]
Society and culture
Names
Tirzepatide is the international nonproprietary name (INN).[23]
References
- ^ Jump up to:a b “Highlights of prescribing information” (PDF). accessdata.fda.gov. FDA. May 2022. Retrieved 14 May 2022.
- ^ Jump up to:a b c d e f g h i “FDA Approves Novel, Dual-Targeted Treatment for Type 2 Diabetes”. U.S. Food and Drug Administration (FDA) (Press release). 13 May 2022. Retrieved 13 May 2022.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Jump up to:a b c d e Thomas MK, Nikooienejad A, Bray R, Cui X, Wilson J, Duffin K, et al. (January 2021). “Dual GIP and GLP-1 Receptor Agonist Tirzepatide Improves Beta-cell Function and Insulin Sensitivity in Type 2 Diabetes”. The Journal of Clinical Endocrinology and Metabolism. 106 (2): 388–396. doi:10.1210/clinem/dgaa863. PMC 7823251. PMID 33236115.
- ^ Coskun T, Sloop KW, Loghin C, Alsina-Fernandez J, Urva S, Bokvist KB, et al. (December 2018). “LY3298176, a novel dual GIP and GLP-1 receptor agonist for the treatment of type 2 diabetes mellitus: From discovery to clinical proof of concept”. Molecular Metabolism. 18: 3–14. doi:10.1016/j.molmet.2018.09.009. PMC 6308032. PMID 30473097.
- ^ Jump up to:a b Min T, Bain SC (January 2021). “The Role of Tirzepatide, Dual GIP and GLP-1 Receptor Agonist, in the Management of Type 2 Diabetes: The SURPASS Clinical Trials”. Diabetes Therapy. 12 (1): 143–157. doi:10.1007/s13300-020-00981-0. PMC 7843845. PMID 33325008.
- ^ Frias JP, Nauck MA, Van J, Kutner ME, Cui X, Benson C, et al. (November 2018). “Efficacy and safety of LY3298176, a novel dual GIP and GLP-1 receptor agonist, in patients with type 2 diabetes: a randomised, placebo-controlled and active comparator-controlled phase 2 trial”. The Lancet. 392 (10160): 2180–2193. doi:10.1016/S0140-6736(18)32260-8. PMID 30293770.
- ^ Frias JP, Nauck MA, Van J, Benson C, Bray R, Cui X, et al. (June 2020). “Efficacy and tolerability of tirzepatide, a dual glucose-dependent insulinotropic peptide and glucagon-like peptide-1 receptor agonist in patients with type 2 diabetes: A 12-week, randomized, double-blind, placebo-controlled study to evaluate different dose-escalation regimens”. Diabetes, Obesity & Metabolism. 22 (6): 938–946. doi:10.1111/dom.13979. PMC 7318331. PMID 31984598.
- ^ Dahl D, Onishi Y, Norwood P, Huh R, Bray R, Patel H, Rodríguez Á (February 2022). “Effect of Subcutaneous Tirzepatide vs Placebo Added to Titrated Insulin Glargine on Glycemic Control in Patients With Type 2 Diabetes: The SURPASS-5 Randomized Clinical Trial”. JAMA. 327 (6): 534–545. doi:10.1001/jama.2022.0078. PMID 35133415.
- ^ Jump up to:a b Ahangarpour M, Kavianinia I, Harris PW, Brimble MA (January 2021). “Photo-induced radical thiol-ene chemistry: a versatile toolbox for peptide-based drug design”. Chemical Society Reviews. Royal Society of Chemistry. 50 (2): 898–944. doi:10.1039/d0cs00354a. PMID 33404559. S2CID 230783854.
- ^ Bastin M, Andreelli F (2019). “Dual GIP-GLP1-Receptor Agonists In The Treatment Of Type 2 Diabetes: A Short Review On Emerging Data And Therapeutic Potential”. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy. 12: 1973–1985. doi:10.2147/DMSO.S191438. PMC 6777434. PMID 31686879.
- ^ “Tirzepatide significantly reduced A1C and body weight in people with type 2 diabetes in two phase 3 trials from Lilly’s SURPASS program” (Press release). Eli Lilly and Company. 17 February 2021. Retrieved 28 October 2021 – via PR Newswire.
- ^ “Lilly : Phase 3 Tirzepatide Results Show Superior A1C And Body Weight Reductions In Type 2 Diabetes”. Business Insider. RTTNews. 19 October 2021. Retrieved 28 October 2021.
- ^ Jump up to:a b Willard FS, Douros JD, Gabe MB, Showalter AD, Wainscott DB, Suter TM, et al. (September 2020). “Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist”. JCI Insight. 5 (17). doi:10.1172/jci.insight.140532. PMC 7526454. PMID 32730231.
- ^ Østergaard S, Paulsson JF, Kofoed J, Zosel F, Olsen J, Jeppesen CB, et al. (October 2021). “The effect of fatty diacid acylation of human PYY3-36 on Y2 receptor potency and half-life in minipigs”. Scientific Reports. 11 (1): 21179. Bibcode:2021NatSR..1121179O. doi:10.1038/s41598-021-00654-3. PMC 8551270. PMID 34707178.
- ^ Jump up to:a b US patent 9474780, Bokvist BK, Coskun T, Cummins RC, Alsina-Fernandez J, “GIP and GLP-1 co-agonist compounds”, issued 2016-10-25, assigned to Eli Lilly and Co
- ^ Frederick MO, Boyse RA, Braden TM, Calvin JR, Campbell BM, Changi SM, et al. (2021). “Kilogram-Scale GMP Manufacture of Tirzepatide Using a Hybrid SPPS/LPPS Approach with Continuous Manufacturing”. Organic Process Research & Development. 25 (7): 1628–1636. doi:10.1021/acs.oprd.1c00108. S2CID 237690232.
- ^ Sagonowsky, Eric (26 October 2021). “As Lilly gears up for key 2022 launches, Trulicity, Taltz and more drive solid growth”. Fierce Pharma. Retrieved 9 April 2022.
- ^ Kellaher, Colin (28 April 2022). “Eli Lilly’s Tirzepatide Meets Main Endpoints in Phase 3 Obesity Study >LLY”. Dow Jones Newswires. Retrieved 29 April 2022 – via MarketWatch.
- ^ Kahan, Scott; Garvey, W. Timothy (28 April 2022). “SURMOUNT-1: Adults achieve weight loss of 16% or more at 72 weeks with tirzepatide”. healio.com. Retrieved 29 April 2022.
- ^ Taylor, Nick Paul (28 April 2022). “SURMOUNT-able: Lilly’s tirzepatide clears high bar set by Novo’s Wegovy in obesity”. FierceBiotech. Retrieved 29 April 2022.
- ^ Frías JP, Davies MJ, Rosenstock J, Pérez Manghi FC, Fernández Landó L, Bergman BK, et al. (August 2021). “Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes”. The New England Journal of Medicine. 385 (6): 503–515. doi:10.1056/NEJMoa2107519. PMID 34170647. S2CID 235635529.
- ^ Dutta D, Surana V, Singla R, Aggarwal S, Sharma M (November–December 2021). “Efficacy and safety of novel twincretin tirzepatide a dual GIP and GLP-1 receptor agonist in the management of type-2 diabetes: A Cochrane meta-analysis”. Indian Journal of Endocrinology and Metabolism. 25 (6): 475–489. doi:10.4103/ijem.ijem_423_21.
- ^ World Health Organization (2019). “International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 81”. WHO Drug Information. 33 (1). hdl:10665/330896.
Further reading
- Bhagavathula AS, Vidyasagar K, Tesfaye W (September 2021). “Efficacy and Safety of Tirzepatide in Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis of Randomized Phase II/III Trials”. Pharmaceuticals (Basel). 14 (10). doi:10.3390/ph14100991. PMC 8537322. PMID 34681215.
- Frías JP (November 2020). “Tirzepatide: a glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) dual agonist in development for the treatment of type 2 diabetes”. Expert Rev Endocrinol Metab. 15 (6): 379–394. doi:10.1080/17446651.2020.1830759. PMID 33030356.
- Ryan DH (September 2021). “Next Generation Antiobesity Medications: Setmelanotide, Semaglutide, Tirzepatide and Bimagrumab: What do They Mean for Clinical Practice?”. J Obes Metab Syndr. 30 (3): 196–208. doi:10.7570/jomes21033. PMC 8526285. PMID 34518444.
External links
- “Tirzepatide”. Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT03954834 for “A Study of Tirzepatide (LY3298176) in Participants With Type 2 Diabetes Not Controlled With Diet and Exercise Alone (SURPASS-1)” at ClinicalTrials.gov
- Clinical trial number NCT03987919 for “A Study of Tirzepatide (LY3298176) Versus Semaglutide Once Weekly as Add-on Therapy to Metformin in Participants With Type 2 Diabetes (SURPASS-2)” at ClinicalTrials.gov
- Clinical trial number NCT03882970 for “A Study of Tirzepatide (LY3298176) Versus Insulin Degludec in Participants With Type 2 Diabetes (SURPASS-3)” at ClinicalTrials.gov
- Clinical trial number NCT03730662 for “A Study of Tirzepatide (LY3298176) Once a Week Versus Insulin Glargine Once a Day in Participants With Type 2 Diabetes and Increased Cardiovascular Risk (SURPASS-4)” at ClinicalTrials.gov
- Clinical trial number NCT04039503 for “A Study of Tirzepatide (LY3298176) Versus Placebo in Participants With Type 2 Diabetes Inadequately Controlled on Insulin Glargine With or Without Metformin (SURPASS-5)” at ClinicalTrials.gov
CLIP
FDA approves Lilly’s Mounjaro™ (tirzepatide) injection, the first and only GIP and GLP-1 receptor agonist for the treatment of adults with type 2 diabetes
May 13, 2022
Mounjaro delivered superior A1C reductions versus all comparators in phase 3 SURPASS clinical trials
While not indicated for weight loss, Mounjaro led to significantly greater weight reductions versus comparators in a key secondary endpoint
Mounjaro represents the first new class of diabetes medicines introduced in nearly a decade and is expected to be available in the U.S. in the coming weeks
INDIANAPOLIS, May 13, 2022 /PRNewswire/ — The U.S. Food and Drug Administration (FDA) approved Mounjaro™ (tirzepatide) injection, Eli Lilly and Company’s (NYSE: LLY) new once-weekly GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 (glucagon-like peptide-1) receptor agonist indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes. Mounjaro has not been studied in patients with a history of pancreatitis and is not indicated for use in patients with type 1 diabetes mellitus.
As the first and only FDA-approved GIP and GLP-1 receptor agonist, Mounjaro is a single molecule that activates the body’s receptors for GIP and GLP-1, which are natural incretin hormones.1
“Mounjaro delivered superior and consistent A1C reductions against all of the comparators throughout the SURPASS program, which was designed to assess Mounjaro’s efficacy and safety in a broad range of adults with type 2 diabetes who could be treated in clinical practice. The approval of Mounjaro is an exciting step forward for people living with type 2 diabetes given the results seen in these clinical trials,” said Juan Pablo Frías, M.D., Medical Director, National Research Institute and Investigator in the SURPASS program.
Mounjaro will be available in six doses (2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, 15 mg) and will come in Lilly’s well-established auto-injector pen with a pre-attached, hidden needle that patients do not need to handle or see.
The approval was based on results from the phase 3 SURPASS program, which included active comparators of injectable semaglutide 1 mg, insulin glargine and insulin degludec. Efficacy was evaluated for Mounjaro 5 mg, 10 mg and 15 mg used alone or in combination with commonly prescribed diabetes medications, including metformin, SGLT2 inhibitors, sulfonylureas and insulin glargine. Participants in the SURPASS program achieved average A1C reductions between 1.8% and 2.1% for Mounjaro 5 mg and between 1.7% and 2.4% for both Mounjaro 10 mg and Mounjaro 15 mg. While not indicated for weight loss, mean change in body weight was a key secondary endpoint in all SURPASS studies. Participants treated with Mounjaro lost between 12 lb. (5 mg) and 25 lb. (15 mg) on average.1
Side effects reported in at least 5% of patients treated with Mounjaro include nausea, diarrhea, decreased appetite, vomiting, constipation, indigestion (dyspepsia), and stomach (abdominal) pain. The labeling for Mounjaro contains a Boxed Warning regarding thyroid C-cell tumors. Mounjaro is contraindicated in patients with a personal or family history of medullary thyroid carcinoma or in patients with Multiple Endocrine Neoplasia syndrome type 2.1
“Lilly has a nearly 100-year heritage of advancing care for people living with diabetes – never settling for current outcomes. We’re not satisfied knowing that half of the more than 30 million Americans living with type 2 diabetes are not reaching their target blood glucose levels,” said Mike Mason, president, Lilly Diabetes. “We are thrilled to introduce Mounjaro, which represents the first new class of type 2 diabetes medication introduced in almost a decade and embodies our mission to bring innovative new therapies to the diabetes community.”
Mounjaro is expected to be available in the United States in the coming weeks. Lilly is committed to helping people access the medicines they are prescribed and will work with insurers, health systems and providers to help enable patient access to Mounjaro. Lilly plans to offer a Mounjaro savings card for people who qualify. Patients or healthcare professionals with questions about Mounjaro can visit www.Mounjaro.com or call The Lilly Answers Center at 1-800-LillyRx (1-800-545-5979).
Tirzepatide is also under regulatory review for the treatment of type 2 diabetes in Europe, Japan and several additional markets. A multimedia gallery is available on Lilly.com.
About the SURPASS clinical trial program
The SURPASS phase 3 global clinical development program for tirzepatide began in late 2018 and included five global registration trials and two regional trials in Japan. These studies ranged from 40 to 52 weeks and evaluated the efficacy and safety of Mounjaro 5 mg, 10 mg and 15 mg as a monotherapy and as an add-on to various standard-of-care medications for type 2 diabetes. The active comparators in the studies were injectable semaglutide 1 mg, insulin glargine and insulin degludec. Collectively, the five global registration trials consistently demonstrated A1C reductions for participants taking Mounjaro across multiple stages of their type 2 diabetes journeys, from an average around five to 13 years of having diabetes.2-8
- SURPASS-1 (NCT03954834) was a 40-week study comparing the efficacy and safety of Mounjaro 5 mg (N=121), 10 mg (N=121) and 15 mg (N=120) as monotherapy to placebo (N=113) in adults with type 2 diabetes inadequately controlled with diet and exercise alone. From a baseline A1C of 7.9%, Mounjaro reduced participants’ A1C by a mean of 1.8%* (5 mg) and 1.7%* (10 mg and 15 mg) compared to 0.1% for placebo. In a key secondary endpoint, from a baseline weight of 189 lb., Mounjaro reduced participants’ weight by a mean of 14 lb.* (5 mg), 15 lb.* (10 mg) and 17 lb.* (15 mg) compared to 2 lb. for placebo.2,3
- SURPASS-2 (NCT03987919) was a 40-week study comparing the efficacy and safety of Mounjaro 5 mg (N=470), 10 mg (N=469) and 15 mg (N=469) to injectable semaglutide 1 mg (N=468) in adults with type 2 diabetes inadequately controlled with ≥1500 mg/day metformin alone. From a baseline A1C of 8.3%, Mounjaro reduced participants’ A1C by a mean of 2.0%ꝉ (5 mg), 2.2%* (10 mg) and 2.3%* (15 mg) compared to 1.9% for semaglutide. In a key secondary endpoint, from a baseline weight of 207 lb., Mounjaro reduced participants’ weight by a mean of 17 lb.ꝉ (5 mg), 21 lb.* (10 mg) and 25 lb.* (15 mg) compared to 13 lb. for semaglutide.4,5
- SURPASS-3 (NCT03882970) was a 52-week study comparing the efficacy of Mounjaro 5 mg (N=358), 10 mg (N=360) and 15 mg (N=358) to titrated insulin degludec (N=359) in adults with type 2 diabetes treated with metformin with or without an SGLT-2 inhibitor. From a baseline A1C of 8.2%, Mounjaro reduced participants’ A1C by a mean of 1.9%* (5 mg), 2.0%* (10 mg) and 2.1%* (15 mg) compared to 1.3% for insulin degludec. From a baseline weight of 208 lb., Mounjaro reduced participants’ weight by a mean of 15 lb.* (5 mg), 21 lb.* (10 mg) and 25 lb.* (15 mg) compared to an increase of 4 lb. for insulin degludec.6
- SURPASS-4 (NCT03730662) was a 104-week study comparing the efficacy and safety of Mounjaro 5 mg (N=328), 10 mg (N=326) and 15 mg (N=337) to insulin glargine (N=998) in adults with type 2 diabetes inadequately controlled with at least one and up to three oral antihyperglycemic medications (metformin, sulfonylureas or SGLT-2 inhibitors), who have increased cardiovascular (CV) risk. The primary endpoint was measured at 52 weeks. From a baseline A1C of 8.5%, Mounjaro reduced participants’ A1C by a mean of 2.1%* (5 mg), 2.3%* (10 mg) and 2.4%* (15 mg) compared to 1.4% for insulin glargine. From a baseline weight of 199 lb., Mounjaro reduced weight by a mean of 14 lb.* (5 mg), 20 lb.* (10 mg) and 23 lb.* (15 mg) compared to an increase of 4 lb. for insulin glargine.7
- SURPASS-5 (NCT04039503) was a 40-week study comparing the efficacy and safety of Mounjaro 5 mg (N=116), 10 mg (N=118) and 15 mg (N=118) to placebo (N=119) in adults with inadequately controlled type 2 diabetes already being treated with insulin glargine, with or without metformin. From a baseline A1C of 8.3%, Mounjaro reduced A1C by a mean of 2.1%* (5 mg), 2.4%* (10 mg) and 2.3%* (15 mg) compared to 0.9% for placebo. From a baseline weight of 210 lb., Mounjaro reduced participants’ weight by a mean of 12 lb.* (5 mg), 17 lb.* (10 mg) and 19 lb.* (15 mg) compared to an increase of 4 lb. for placebo.8
*p<0.001 for superiority vs. placebo or active comparator, adjusted for multiplicity
ꝉp<0.05 for superiority vs. semaglutide 1 mg, adjusted for multiplicity
About Mounjaro™ (tirzepatide) injection1
Mounjaro™ (tirzepatide) injection is FDA-approved as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus. As the first and only FDA-approved GIP and GLP-1 receptor agonist, Mounjaro is a single molecule that activates the body’s receptors for GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 (glucagon-like peptide-1). Mounjaro will be available in six doses (2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg, 15 mg) and will come in Lilly’s well-established auto-injector pen with a pre-attached, hidden needle that patients do not need to handle or see.
PURPOSE AND SAFETY SUMMARY WITH WARNINGS
Important Facts About MounjaroTM (mown-JAHR-OH). It is also known as tirzepatide.
- Mounjaro is an injectable prescription medicine for adults with type 2 diabetes used along with diet and exercise to improve blood sugar (glucose).
- It is not known if Mounjaro can be used in people who have had inflammation of the pancreas (pancreatitis). Mounjaro is not for use in people with type 1 diabetes. It is not known if Mounjaro is safe and effective for use in children under 18 years of age.
Warnings
Mounjaro may cause tumors in the thyroid, including thyroid cancer. Watch for possible symptoms, such as a lump or swelling in the neck, hoarseness, trouble swallowing, or shortness of breath. If you have a symptom, tell your healthcare provider.
- Do not use Mounjaro if you or any of your family have ever had a type of thyroid cancer called medullary thyroid carcinoma (MTC).
- Do not use Mounjaro if you have Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
- Do not use Mounjaro if you are allergic to tirzepatide or any of the ingredients in Mounjaro.
Mounjaro may cause serious side effects, including:
Inflammation of the pancreas (pancreatitis). Stop using Mounjaro and call your healthcare provider right away if you have severe pain in your stomach area (abdomen) that will not go away, with or without vomiting. You may feel the pain from your abdomen to your back.
Low blood sugar (hypoglycemia). Your risk for getting low blood sugar may be higher if you use Mounjaro with another medicine that can cause low blood sugar, such as a sulfonylurea or insulin. Signs and symptoms of low blood sugar may include dizziness or light-headedness, sweating, confusion or drowsiness, headache, blurred vision, slurred speech, shakiness, fast heartbeat, anxiety, irritability, or mood changes, hunger, weakness and feeling jittery.
Serious allergic reactions. Stop using Mounjaro and get medical help right away if you have any symptoms of a serious allergic reaction, including swelling of your face, lips, tongue or throat, problems breathing or swallowing, severe rash or itching, fainting or feeling dizzy, and very rapid heartbeat.
Kidney problems (kidney failure). In people who have kidney problems, diarrhea, nausea, and vomiting may cause a loss of fluids (dehydration), which may cause kidney problems to get worse. It is important for you to drink fluids to help reduce your chance of dehydration.
Severe stomach problems. Stomach problems, sometimes severe, have been reported in people who use Mounjaro. Tell your healthcare provider if you have stomach problems that are severe or will not go away.
Changes in vision. Tell your healthcare provider if you have changes in vision during treatment with Mounjaro.
Gallbladder problems. Gallbladder problems have happened in some people who use Mounjaro. Tell your healthcare provider right away if you get symptoms of gallbladder problems, which may include pain in your upper stomach (abdomen), fever, yellowing of skin or eyes (jaundice), and clay-colored stools.
Common side effects
The most common side effects of Mounjaro include nausea, diarrhea, decreased appetite, vomiting, constipation, indigestion, and stomach (abdominal) pain. These are not all the possible side effects of Mounjaro. Talk to your healthcare provider about any side effect that bothers you or doesn’t go away.
Tell your healthcare provider if you have any side effects. You can report side effects at 1-800-FDA-1088 or www.fda.gov/medwatch.
Before using
- Your healthcare provider should show you how to use Mounjaro before you use it for the first time.
- Before you use Mounjaro, talk to your healthcare provider about low blood sugar and how to manage it.
Review these questions with your healthcare provider:
- Do you have other medical conditions, including problems with your pancreas or kidneys, or severe problems with your stomach, such as slowed emptying of your stomach (gastroparesis) or problems digesting food?
- Do you take other diabetes medicines, such as insulin or sulfonylureas?
- Do you have a history of diabetic retinopathy?
- Are you pregnant or plan to become pregnant or breastfeeding or plan to breastfeed? It is not known if Mounjaro will harm your unborn baby.
- Do you take birth control pills by mouth? These may not work as well while using Mounjaro. Your healthcare provider may recommend another type of birth control when you start Mounjaro or when you increase your dose.
- Do you take any other prescription medicines or over-the-counter drugs, vitamins, or herbal supplements?
How to take
- Read the Instructions for Use that come with Mounjaro.
- Use Mounjaro exactly as your healthcare provider says.
- Mounjaro is injected under the skin (subcutaneously) of your stomach (abdomen), thigh, or upper arm.
- Use Mounjaro 1 time each week, at any time of the day.
- Do not mix insulin and Mounjaro together in the same injection.
- If you take too much Mounjaro, call your healthcare provider or seek medical advice promptly.
Learn more
For more information, call 1-800-LillyRx (1-800-545-5979) or go to www.mounjaro.com.
This information does not take the place of talking with your healthcare provider. Be sure to talk to your healthcare provider about Mounjaro and how to take it. Your healthcare provider is the best person to help you decide if Mounjaro is right for you.
MounjaroTM and its delivery device base are trademarks owned or licensed by Eli Lilly and Company, its subsidiaries, or affiliates.
Please click to access full Prescribing Information and Medication Guide.
TR CON CBS MAY2022
About Lilly
Lilly unites caring with discovery to create medicines that make life better for people around the world. We’ve been pioneering life-changing discoveries for nearly 150 years, and today our medicines help more than 47 million people across the globe. Harnessing the power of biotechnology, chemistry and genetic medicine, our scientists are urgently advancing new discoveries to solve some of the world’s most significant health challenges, redefining diabetes care, treating obesity and curtailing its most devastating long-term effects, advancing the fight against Alzheimer’s disease, providing solutions to some of the most debilitating immune system disorders, and transforming the most difficult-to-treat cancers into manageable diseases. With each step toward a healthier world, we’re motivated by one thing: making life better for millions more people. That includes delivering innovative clinical trials that reflect the diversity of our world and working to ensure our medicines are accessible and affordable. To learn more, visit Lilly.com and Lilly.com/newsroom or follow us on Facebook, Instagram, Twitter and LinkedIn. P-LLY
Lilly Cautionary Statement Regarding Forward-Looking Statements
This press release contains forward-looking statements (as that term is defined in the Private Securities Litigation Reform Act of 1995) about Mounjaro™ (tirzepatide 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg and 15 mg) injection as a treatment to improve glycemic control in adults with type 2 diabetes, the timeline for supply of Mounjaro to become available, and certain other milestones and ongoing clinical trials of Mounjaro and reflects Lilly’s current beliefs and expectations. However, as with any pharmaceutical product or medical device, there are substantial risks and uncertainties in the process of research, development and commercialization. Among other things, there can be no guarantee that Mounjaro will be commercially successful, that future study results will be consistent with results to date, or that we will meet our anticipated timelines for the commercialization of Mounjaro. For further discussion of these and other risks and uncertainties, see Lilly’s most recent Form 10-K and Form 10-Q filings with the United States Securities and Exchange Commission. Except as required by law, Lilly undertakes no duty to update forward-looking statements to reflect events after the date of this release.
References
- Mounjaro. Prescribing Information. Lilly USA, LLC.
- Rosenstock, J, et. al. Efficacy and Safety of Once Weekly Tirzepatide, a Dual GIP/GLP-1 Receptor Agonist Versus Placebo as Monotherapy in People with Type 2 Diabetes (SURPASS-1). Abstract 100-OR. Presented virtually at the American Diabetes Association’s 81st Scientific Sessions; June 25-29.
- Rosenstock, J, et. al. (2021). Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. Lancet. 2021;398(10295):143-155. doi: 10.1016/S0140-6736(21)01324-6.
- Frías JP, Davies MJ, Rosenstock J, et al; for the SURPASS-2 Investigators. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med. 2021;385(6)(suppl):503-515. doi: 10.1056/NEJMoa2107519
- Frias, J.P. Efficacy and Safety of Tirzepatide vs. Semaglutide Once Weekly as Add-On Therapy to Metformin in Patients with Type 2 Diabetes. Abstract 84-LB. Presented virtually at the American Diabetes Association’s 81st Scientific Sessions; June 25-29.
- Ludvik B, Giorgino F, Jódar E, et al. Once-weekly tirzepatide versus once-daily insulin degludec as add-on to metformin with or without SGLT2 inhibitors in patients with type 2 diabetes (SURPASS-3): a randomised, open-label, parallel-group, phase 3 trial. Lancet. 2021;398(10300):583-598. doi: 10.1016/S0140-6736(21)01443-4
- Del Prato S, Kahn SE, Pavo I, et al; for the SURPASS-4 Investigators. Tirzepatide versus insulin glargine in type 2 diabetes and increased cardiovascular risk (SURPASS-4): a randomised, open-label, parallel-group, multicentre, phase 3 trial. Lancet. 2021;398(10313):1811-1824. doi: 10.1016/S0140-6736(21)02188-7
- Dahl D, Onishi Y, Norwood P, et al. Effect of subcutaneous tirzepatide vs placebo added to titrated insulin glargine on glycemic control in patients with type 2 diabetes: the SURPASS-5 randomized clinical trial. JAMA. 2022;327(6):534-545. doi:10.1001/jama.2022.0078
CLIP
Lilly’s tirzepatide delivered up to 22.5% weight loss in adults with obesity or overweight in SURMOUNT-1
April 28, 2022
Participants taking tirzepatide lost up to 52 lb. (24 kg) in this 72-week phase 3 study
63% of participants taking tirzepatide 15 mg achieved at least 20% body weight reductions as a key secondary endpoint
INDIANAPOLIS, April 28, 2022 /PRNewswire/ — Tirzepatide (5 mg, 10 mg, 15 mg) achieved superior weight loss compared to placebo at 72 weeks of treatment in topline results from Eli Lilly and Company’s (NYSE: LLY) SURMOUNT-1 clinical trial, with participants losing up to 22.5% (52 lb. or 24 kg) of their body weight for the efficacy estimandi. This study enrolled 2,539 participants and was the first phase 3 global registration trial evaluating the efficacy and safety of tirzepatide in adults with obesity, or overweight with at least one comorbidity, who do not have diabetes. Tirzepatide met both co-primary endpoints of superior mean percent change in body weight from baseline and greater percentage of participants achieving body weight reductions of at least 5% compared to placebo for both estimandsii. The study also achieved all key secondary endpoints at 72 weeks.
For the efficacy estimand, participants taking tirzepatide achieved average weight reductions of 16.0% (35 lb. or 16 kg on 5 mg), 21.4% (49 lb. or 22 kg on 10 mg) and 22.5% (52 lb. or 24 kg on 15 mg), compared to placebo (2.4%, 5 lb. or 2 kg). Additionally, 89% (5 mg) and 96% (10 mg and 15 mg) of people taking tirzepatide achieved at least 5% body weight reductions compared to 28% of those taking placebo.
In a key secondary endpoint, 55% (10 mg) and 63% (15 mg) of people taking tirzepatide achieved at least 20% body weight reductions compared to 1.3% of those taking placebo. In an additional secondary endpoint not controlled for type 1 error, 32% of participants taking tirzepatide 5 mg achieved at least 20% body weight reductions. The mean baseline body weight of participants was 231 lb. (105 kg).
“Obesity is a chronic disease that often does not receive the same standard of care as other conditions, despite its impact on physical, psychological and metabolic health, which can include increased risk of hypertension, heart disease, cancer and decreased survival,” said Louis J. Aronne, MD, FACP, DABOM, director of the Comprehensive Weight Control Center and the Sanford I. Weill Professor of Metabolic Research at Weill Cornell Medicine, obesity expert at NewYork-Presbyterian/Weill Cornell Medical Center and Investigator of SURMOUNT-1. “Tirzepatide delivered impressive body weight reductions in SURMOUNT-1, which could represent an important step forward for helping the patient and physician partnership treat this complex disease.”
For the treatment-regimen estimandiii, results showed:
- Average body weight reductions: 15.0% (5 mg), 19.5% (10 mg), 20.9% (15 mg), 3.1% (placebo)
- Percentage of participants achieving body weight reductions of ≥5%: 85% (5 mg), 89% (10 mg), 91% (15 mg), 35% (placebo)
- Percentage of participants achieving body weight reductions of ≥20%: 30% (5 mg, not controlled for type 1 error), 50% (10 mg), 57% (15 mg), 3.1% (placebo)
The overall safety and tolerability profile of tirzepatide was similar to other incretin-based therapies approved for the treatment of obesity. The most commonly reported adverse events were gastrointestinal-related and generally mild to moderate in severity, usually occurring during the dose escalation period. For those treated with tirzepatide (5 mg, 10 mg and 15 mg, respectively), nausea (24.6%, 33.3%, 31.0%), diarrhea (18.7%, 21.2%, 23.0%), vomiting (8.3%, 10.7%, 12.2%) and constipation (16.8%, 17.1%, 11.7%) were more frequently experienced compared to placebo (9.5% [nausea], 7.3% [diarrhea], 1.7% [vomiting], 5.8% [constipation]).
Treatment discontinuation rates due to adverse events were 4.3% (5 mg), 7.1% (10 mg), 6.2% (15 mg) and 2.6% (placebo). The overall treatment discontinuation rates were 14.3% (5 mg), 16.4% (10 mg), 15.1% (15 mg) and 26.4% (placebo).
Participants who had pre-diabetes at study commencement will remain enrolled in SURMOUNT-1 for an additional 104 weeks of treatment following the initial 72-week completion date to evaluate the impact on body weight and the potential differences in progression to type 2 diabetes at three years of treatment with tirzepatide compared to placebo.
“Tirzepatide is the first investigational medicine to deliver more than 20 percent weight loss on average in a phase 3 study, reinforcing our confidence in its potential to help people living with obesity,” said Jeff Emmick, MD, Ph.D., vice president, product development, Lilly. “Obesity is a chronic disease that requires effective treatment options, and Lilly is working relentlessly to support people with obesity and modernize how this disease is approached. We’re proud to research and develop potentially innovative treatments like tirzepatide, which helped nearly two thirds of participants on the highest dose reduce their body weight by at least 20 percent in SURMOUNT-1.”
Tirzepatide is a novel investigational once-weekly GIP (glucose-dependent insulinotropic polypeptide) receptor and GLP-1 (glucagon-like peptide-1) receptor agonist, representing a new class of medicines being studied for the treatment of obesity. Tirzepatide is a single peptide that activates the body’s receptors for GIP and GLP-1, two natural incretin hormones. Obesity is a chronic, progressive disease caused by disruptions in the mechanisms that control body weight, often leading to an increase in food intake and/or a decrease in energy expenditure. These disruptions are multifactorial and can be related to genetic, developmental, behavioral, environmental and social factors. To learn more, visit Lilly.com/obesity.
Lilly will continue to evaluate the SURMOUNT-1 results, which will be presented at an upcoming medical meeting and submitted to a peer-reviewed journal. Additional studies are ongoing for tirzepatide as a potential treatment for obesity or overweight.
About tirzepatide
Tirzepatide is a once-weekly GIP (glucose-dependent insulinotropic polypeptide) receptor and GLP-1 (glucagon-like peptide-1) receptor agonist that integrates the actions of both incretins into a single novel molecule. GIP is a hormone that may complement the effects of GLP-1 receptor agonists. In preclinical models, GIP has been shown to decrease food intake and increase energy expenditure therefore resulting in weight reductions, and when combined with GLP-1 receptor agonism, may result in greater effects on markers of metabolic dysregulation such as body weight, glucose and lipids. Tirzepatide is in phase 3 development for adults with obesity or overweight with weight-related comorbidity and is currently under regulatory review as a treatment for adults with type 2 diabetes. It is also being studied as a potential treatment for non-alcoholic steatohepatitis (NASH) and heart failure with preserved ejection fraction (HFpEF). Studies of tirzepatide in obstructive sleep apnea (OSA) and in morbidity/mortality in obesity are planned as well.
About SURMOUNT-1 and the SURMOUNT clinical trial program
SURMOUNT-1 (NCT04184622) is a multi-center, randomized, double-blind, parallel, placebo-controlled trial comparing the efficacy and safety of tirzepatide 5 mg, 10 mg and 15 mg to placebo as an adjunct to a reduced-calorie diet and increased physical activity in adults without type 2 diabetes who have obesity, or overweight with at least one of the following comorbidities: hypertension, dyslipidemia, obstructive sleep apnea or cardiovascular disease. The trial randomized 2,539 participants across the U.S., Argentina, Brazil, China, India, Japan, Mexico, Russia and Taiwan in a 1:1:1:1 ratio to receive either tirzepatide 5 mg, 10 mg or 15 mg or placebo. The co-primary objectives of the study were to demonstrate that tirzepatide 10 mg and/or 15 mg is superior in percentage of body weight reductions from baseline and percentage of participants achieving ≥5% body weight reduction at 72 weeks compared to placebo. Participants who had pre-diabetes at study commencement will remain enrolled in SURMOUNT-1 for an additional 104 weeks of treatment following the initial 72-week completion date to evaluate the impact on body weight and potential differences in progression to type 2 diabetes at three years of treatment with tirzepatide compared to placebo.
All participants in the tirzepatide treatment arms started the study at a dose of tirzepatide 2.5 mg once-weekly and then increased the dose in a step-wise approach at four-week intervals to their final randomized maintenance dose of 5 mg (via a 2.5 mg step), 10 mg (via steps at 2.5 mg, 5 mg and 7.5 mg) or 15 mg (via steps at 2.5 mg, 5 mg, 7.5 mg, 10 mg and 12.5 mg).
The SURMOUNT phase 3 global clinical development program for tirzepatide began in late 2019 and has enrolled more than 5,000 people with obesity or overweight across six clinical trials, four of which are global studies. Results from SURMOUNT-2, -3, and -4 are anticipated in 2023.
About Lilly
Lilly unites caring with discovery to create medicines that make life better for people around the world. We’ve been pioneering life-changing discoveries for nearly 150 years, and today our medicines help more than 47 million people across the globe. Harnessing the power of biotechnology, chemistry and genetic medicine, our scientists are urgently advancing new discoveries to solve some of the world’s most significant health challenges, redefining diabetes care, treating obesity and curtailing its most devastating long-term effects, advancing the fight against Alzheimer’s disease, providing solutions to some of the most debilitating immune system disorders, and transforming the most difficult-to-treat cancers into manageable diseases. With each step toward a healthier world, we’re motivated by one thing: making life better for millions more people. That includes delivering innovative clinical trials that reflect the diversity of our world and working to ensure our medicines are accessible and affordable. To learn more, visit Lilly.com and Lilly.com/newsroom or follow us on Facebook, Instagram, Twitter and LinkedIn. P-LLY
CLIP
Tirzepatide results superior A1C and body weight reductions compared to insulin glargine in adults with type 2 diabetes
Newly published data show that participants maintained A1C and weight control up to two years in SURPASS-4, the largest and longest SURPASS trial completed to dateNo increased cardiovascular risk identified with tirzepatide; hazard ratio of 0.74 observed for MACE-4 events
SURPASS-4 is the largest and longest clinical trial completed to date of the phase 3 program studying tirzepatide as a potential treatment for type 2 diabetes. The primary endpoint was measured at 52 weeks, with participants continuing treatment up to 104 weeks or until study completion. The completion of the study was triggered by the accrual of major adverse cardiovascular events (MACE) to assess CV risk. In newly published data from the treatment period after 52 weeks, participants taking tirzepatide maintained A1C and weight control for up to two years.
The overall safety profile of tirzepatide, assessed over the full study period, was consistent with the safety results measured at 52 weeks, with no new findings up to 104 weeks. Gastrointestinal side effects were the most commonly reported adverse events, usually occurring during the escalation period and then decreasing over time.
“We are encouraged by the continued A1C and weight control that participants experienced past the initial 52 week treatment period and up to two years as we continue to explore the potential impact of tirzepatide for the treatment of type 2 diabetes,” said John Doupis, M.D., Ph.D., Director, Diabetes Division and Clinical Research Center, Iatriko Paleou Falirou Medical Center, Athens, Greece and Senior Investigator for SURPASS-4.
Tirzepatide is a novel investigational once-weekly dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist that integrates the actions of both incretins into a single molecule, representing a new class of medicines being studied for the treatment of type 2 diabetes.
SURPASS-4 was an open-label global trial comparing the safety and efficacy of three tirzepatide doses (5 mg, 10 mg and 15 mg) to titrated insulin glargine in 2,002 adults with type 2 diabetes with increased CV risk who were treated with between one and three oral antihyperglycemic medicines (metformin, a sulfonylurea or an SGLT-2 inhibitor). Of the total participants randomized, 1,819 (91%) completed the primary 52-week visit and 1,706 (85%) completed the study on treatment. The median study duration was 85 weeks and 202 participants (10%) completed two years.
Study participants had a mean duration of diabetes of 11.8 years, a baseline A1C of 8.52 percent and a baseline weight of 90.3 kg. More than 85 percent of participants had a history of cardiovascular events. In the insulin glargine arm, the insulin dose was titrated following a treat-to-target algorithm with the goal of fasting blood glucose below 100 mg/dL. The starting dose of insulin glargine was 10 units per day, and the mean dose of insulin glargine at 52 weeks was 43.5 units per day.
About tirzepatide
Tirzepatide is a once-weekly dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist that integrates the actions of both incretins into a single novel molecule. GIP is a hormone that may complement the effects of GLP-1. In preclinical models, GIP has been shown to decrease food intake and increase energy expenditure therefore resulting in weight reductions, and when combined with a GLP-1 receptor agonist, may result in greater effects on glucose and body weight. Tirzepatide is in phase 3 development for blood glucose management in adults with type 2 diabetes, for chronic weight management and heart failure with preserved ejection fraction (HFpEF). It is also being studied as a potential treatment for non-alcoholic steatohepatitis (NASH).
About SURPASS-4 and the SURPASS clinical trial program
SURPASS-4 (NCT03730662) is a randomized, parallel, open-label trial comparing the efficacy and safety of tirzepatide 5 mg, 10 mg and 15 mg to insulin glargine in adults with type 2 diabetes inadequately controlled with at least one and up to three oral antihyperglycemic medications (metformin, sulfonylureas or SGLT-2 inhibitors), who have increased cardiovascular (CV) risk. The trial randomized 2,002 study participants in a 1:1:1:3 ratio to receive either tirzepatide 5 mg, 10 mg or 15 mg or insulin glargine. Participants were located in the European Union, North America (Canada and the United States), Australia, Israel, Taiwan and Latin America (Brazil, Argentina and Mexico). The primary objective of the study was to demonstrate that tirzepatide (10 mg and/or 15 mg) is non-inferior to insulin glargine for change from baseline A1C at 52 weeks in people with type 2 diabetes and increased CV risk. The primary and key secondary endpoints were measured at 52 weeks, with participants continuing treatment up to 104 weeks or until study completion. The completion of the study was triggered by the accrual of major adverse cardiovascular events (MACE). Study participants enrolled had to have a mean baseline A1C between 7.5 percent and 10.5 percent and a BMI greater than or equal to 25 kg/m2 at baseline. All participants in the tirzepatide treatment arms started the study at a dose of tirzepatide 2.5 mg once-weekly and then increased the dose in a step-wise approach at four-week intervals to their final randomized maintenance dose of 5 mg (via a 2.5 mg step), 10 mg (via steps at 2.5 mg, 5 mg and 7.5 mg) or 15 mg (via steps at 2.5 mg, 5 mg, 7.5 mg, 10 mg and 12.5 mg). All participants in the titrated insulin glargine treatment arm started with a baseline dose of 10 units per day and titrated following a treat-to-target algorithm to reach a fasting blood glucose below 100 mg/dL.
The SURPASS phase 3 global clinical development program for tirzepatide has enrolled more than 20,000 people with type 2 diabetes across 10 clinical trials, five of which are global registration studies. The program began in late 2018, and all five global registration trials have been completed.
About Diabetes
Approximately 34 million Americans2 (just over 1 in 10) and an estimated 463 million adults worldwide3 have diabetes. Type 2 diabetes is the most common type internationally, accounting for an estimated 90 to 95 percent of all diabetes cases in the United States alone2. Diabetes is a chronic disease that occurs when the body does not properly produce or use the hormone insulin.
| Clinical data | |
|---|---|
| Trade names | Mounjaro |
| Other names | LY3298176, GIP/GLP-1 RA |
| License data | US DailyMed: Tirzepatide |
| Routes of administration | subcutaneous |
| Drug class | Antidiabetic, GLP-1 receptor agonist |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only [1][2] |
| Identifiers | |
| showIUPAC name | |
| CAS Number | 2023788-19-2 |
| PubChem CID | 156588324 |
| IUPHAR/BPS | 11429 |
| DrugBank | DB15171 |
| ChemSpider | 76714503 |
| UNII | OYN3CCI6QE |
| KEGG | D11360 |
| ChEMBL | ChEMBL4297839 |
| Chemical and physical data | |
| Formula | C225H348N48O68 |
| Molar mass | 4813.527 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| showSMILES | |
| showInChI |
////////////Tirzepatide, FDA 2022, APPROVALS 2022, Mounjaro, PEPTIDE, チルゼパチド , LY3298176,
UNIIOYN3CCI6QE
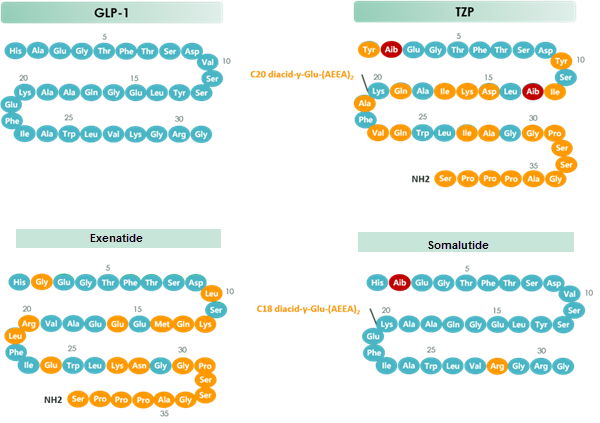
chart 1 Structure of GLP-1 & TZP & Exenatide & Somalutide
Olipudase alfa
| HPLSPQGHPA RLHRIVPRLR DVFGWGNLTC PICKGLFTAI NLGLKKEPNV ARVGSVAIKL CNLLKIAPPA VCQSIVHLFE DDMVEVWRRS VLSPSEACGL LLGSTCGHWD IFSSWNISLP TVPKPPPKPP SPPAPGAPVS RILFLTDLHW DHDYLEGTDP DCADPLCCRR GSGLPPASRP GAGYWGEYSK CDLPLRTLES LLSGLGPAGP FDMVYWTGDI PAHDVWHQTR QDQLRALTTV TALVRKFLGP VPVYPAVGNH ESTPVNSFPP PFIEGNHSSR WLYEAMAKAW EPWLPAEALR TLRIGGFYAL SPYPGLRLIS LNMNFCSREN FWLLINSTDP AGQLQWLVGE LQAAEDRGDK VHIIGHIPPG HCLKSWSWNY YRIVARYENT LAAQFFGHTH VDEFEVFYDE ETLSRPLAVA FLAPSATTYI GLNPGYRVYQ IDGNYSGSSH VVLDHETYIL NLTQANIPGA IPHWQLLYRA RETYGLPNTL PTAWHNLVYR MRGDMQLFQT FWFLYHKGHP PSEPCGTPCR LATLCAQLSA RADSPALCRH LMPDGSLPEA QSLWPRPLFC (Disulfide bridge: 43-119, 46-111, 74-85, 175-180, 181-204, 339-385, 538-542, 548-561) |
Olipudase alfa
Xenpozyme, Japan 2022, APPROVALS 2022, 2022/3/28
PEPTIDE, オリプダーゼアルファ (遺伝子組換え)
Alternative Names: Acid sphingomyelinase Niemann Pick disease type B – Sanofi; Acid-sphingomyelinase – Sanofi; GZ-402665; Recombinant human acid sphingomyelinase – Sanofi; rhASM – Sanofi; Sphingomyelinase-C (synthetic human) – Sanofi; Synthetic human sphingomyelinase-C – Sanofi; Xenpozyme
| Formula | C2900H4373N783O791S24 |
|---|---|
| CAS | 927883-84-9 |
| Mol weight | 63631.0831 |
| Efficacy | Lysosomal storage disease treatment, Enzyme replacement (acid sphingomyelinase) |
|---|---|
| Comment | Enzyme replacement therapy product Treatment of Niemann-Pick disease type A/B |
- OriginatorGenzyme Corporation
- DeveloperSanofi
- ClassRecombinant proteins; Sphingomyelin phosphodiesterases
- Mechanism of ActionSphingomyelin-phosphodiesterase replacements
- Orphan Drug StatusYes – Niemann-Pick diseases
- RegisteredNiemann-Pick diseases
- 28 Mar 2022Registered for Niemann-Pick diseases (In adolescents, In children, In adults) in Japan (IV) – First global approval
- 09 Feb 2022FDA assigns PDUFA action date of (03/07/2022) for Olipudase alfa (In children, In adults) for Niemann-Pick diseases
- 09 Feb 2022Adverse e
//////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
CLIP
Olipudase Alfa Improves Lung Function, Spleen Volume in ASMD
Olipudase Alfa Improves Lung Function, Spleen Volume in ASMD
Olipudase alfa was associated with significant improvements in clinically relevant disease end points among patients with chronic visceral acid sphingomyelinase (ASM) deficiency (ASMD), according to results from the phase 2/3 ASCEND trial presented at the 17th Annual WORLDSymposium.
ASMD is a rare, debilitating lysosomal storage disease characterized by a deficiency of the enzyme acid sphingomyelinase, which results in the accumulation of sphingomyelin in various tissues of the body. Olipudase alfa is an investigational enzyme replacement therapy designed to replace deficient or defective ASM.
The multicenter, randomized, double-blind, placebo-controlled ASCEND trial evaluated the efficacy and safety of olipudase alfa in 36 adults with chronic visceral ASMD. Patients were randomly assigned 1:1 to receive olipudase alfa 3mg/kg intravenously every 2 weeks or placebo for 52 weeks. The coprimary end points were the percent change in spleen volume and percent-predicted diffusing capacity of the lung for carbon monoxide (DLCO).
At week 52, treatment with olipudase alfa resulted in a 39.45% reduction in spleen volume, compared with a 0.5% increase for placebo (P <.0001). A decrease in spleen volume of at least 30% was observed in 17 patients (94%) treated with olipudase afla compared with no patients treated with placebo. Additionally, olipudase alfa significantly improved lung function by 22% from baseline compared with 3% for patients receiving placebo (P =.0004), as measured by percent predicted DLCO.
Olipudase alfa also met key secondary end points including a 31.7% reduction in liver volume (vs a 1.4% reduction for placebo; P <.0001) and a 16.8% improvement in mean platelet counts (vs 2.5% with placebo; P =.019) at week 52. Significant improvements in HDL, LDL, AST, ALT, chitotriosidase (54% vs 12% with placebo; P =.0003), and lyso-sphingomyelin (78% vs 6% with placebo) were also observed in the olipudase alfa group at week 52.
With regard to Splenomegaly Related Score, a patient-reported outcome measurement that evaluates patient symptoms associated with an enlarged spleen, findings showed no meaningful difference between olipudase alfa and placebo (-8 point vs -9.3 points, respectively).
As for safety, olipudase alfa was well tolerated with most adverse events being mild to moderate in severity. There were no treatment-related serious adverse events and no adverse event-related discontinuations.
Disclosure: Some authors have declared affiliations with or received funding from the pharmaceutical industry. Please refer to the original study for a full list of disclosures.
Reference
Wasserstein M, Arash-Kaps L, Barbato A, et al. Adults with chronic acid sphingomyelinase deficiency show significant visceral, pulmonary, and hematologic improvements after enzyme replacement therapy with olipudase-alfa: 1-year results of the ASCEND placebo-controlled trial. Presented at: 17th Annual WORLDSymposium; February 8-12, 2021. Abstract 265.
CLIP
https://www.sanofi.com/en/media-room/press-releases/2021/2021-12-06-14-00-00-2346501
EMA accepts regulatory submission for olipudase alfa, the first potential therapy for ASMD
- Olipudase alfa has been granted PRIority MEdicines (PRIME) designation in Europe, Breakthrough Therapy designation in the United States, and SAKIGAKE designation in Japan
- European regulatory decision anticipated second half of 2022
DECEMBER 6, 2021
The European Medicines Agency (EMA) has accepted for review under an accelerated assessment procedure the Marketing Authorization Application (MAA) for olipudase alfa, Sanofi’s investigational enzyme replacement therapy which is being evaluated for the treatment of acid sphingomyelinase deficiency (ASMD). Historically referred to as Niemann-Pick disease (NPD) type A and type B, ASMD is a rare, progressive, and potentially life-threatening disease for which no treatments are currently approved. The estimated prevalence of ASMD is approximately 2,000 patients in the U.S., Europe (EU5 Countries) and Japan. If approved, olipudase alfa will become the first and only therapy for the treatment of ASMD.
“Today’s milestone has been decades in the making and our gratitude goes to the ASMD community who has stood by us with endless patience while olipudase alfa advanced through clinical development,” said Alaa Hamed, MD, MPH, MBA, Global Head of Medical Affairs, Rare Diseases, Sanofi. “Olipudase alfa represents the kind of potentially life-changing innovation that is possible when industry, medical professionals and the patient community work together toward a common goal.”
The MAA is based on positive results from two separate clinical trials (ASCEND and ASCEND-Peds) evaluating olipudase alfa in adult and pediatric patients with non-central nervous system (CNS) manifestations of ASMD type A/B and ASMD type B.
Olipudase alfa has received special designations from regulatory agencies worldwide, recognizing the innovation potential of the investigational therapy.
“Scientific innovation is the greatest source of hope for people living with diseases like ASMD where there are no approved treatments and is a critical component for ensuring a viable healthcare ecosystem,” said Bill Sibold, Executive Vice President of Sanofi Genzyme. “At Sanofi, we have a long history of pioneering scientific innovation, and we remain committed to finding solutions to address unmet medical needs, including those of the rare disease community.”
The EMA awarded olipudase alfa the PRIority MEdicines designation, also known as PRIME, intended to aid and expedite the regulatory process for investigational medicines that may offer a major therapeutic advantage over existing treatments, or benefit patients without treatment options.
The U.S. Food and Drug Administration (FDA) has granted Breakthrough Therapy designation to olipudase alfa. This designation is intended to expedite the development and review of drugs intended to treat serious or life-threatening diseases and conditions. The criteria for granting Breakthrough Therapy designation include preliminary clinical evidence indicating that the molecule may demonstrate substantial improvement on a clinically significant endpoint over available therapies.
In Japan, olipudase alfa was awarded the SAKIGAKE designation, which is intended to promote research and development in Japan for innovative new medical products that satisfy certain criteria, such as the severity of the intended indication. In September, Sanofi filed the J-NDA submission for olipudase alfa.
About ASMD
ASMD results from a deficient activity of the enzyme acid sphingomyelinase (ASM), which is found in special compartments within cells called lysosomes and is required to breakdown lipids called sphingomyelin. If ASM is absent or not functioning as it should, sphingomyelin cannot be metabolized properly and accumulates within cells, eventually causing cell death and the malfunction of major organ systems. The deficiency of the lysosomal enzyme ASM is due to disease-causing variants in the sphingomyelin phosphodiesterase 1 gene (SMPD1). The estimated prevalence of ASMD is approximately 2,000 patients in the U.S., Europe (EU5 Countries) and Japan.
ASMD represents a spectrum of disease caused by the same enzymatic deficiency, with two types that may represent opposite ends of a continuum sometimes referred to as ASMD type A and ASMD type B. ASMD type A is a rapidly progressive neurological form of the disease resulting in death in early childhood due to central nervous system complications. ASMD type B is a serious and potentially life-threatening disease that predominantly impacts the lungs, liver, and spleen, as well as other organs. ASMD type A/B represents an intermediate form that includes varying degrees of neurologic involvement. Patients with ASMD type A/B or ASMD type B were studied in the ASCEND trial program. Another type of NPD is NPD type C, which is unrelated to ASMD.
About olipudase alfa
Olipudase alfa is an investigational enzyme replacement therapy designed to replace deficient or defective ASM, allowing for the breakdown of sphingomyelin. Olipudase alfa is currently being investigated to treat non-CNS manifestations of ASMD. Olipudase alfa has not been studied in ASMD type A patients. Olipudase alfa is an investigational agent and the safety and efficacy have not been evaluated by the FDA, EMA, or any other regulatory authority worldwide.
About Sanofi
Sanofi is dedicated to supporting people through their health challenges. We are a global biopharmaceutical company focused on human health. We prevent illness with vaccines, provide innovative treatments to fight pain and ease suffering. We stand by the few who suffer from rare diseases and the millions with long-term chronic conditions.
With more than 100,000 people in 100 countries, Sanofi is transforming scientific innovation into healthcare solutions around the globe.
///////Olipudase alfa, japan 2022, APPROVALS 2022, Xenpozyme, PEPTIDE, オリプダーゼアルファ (遺伝子組換え) , ORPHAN DRUG, GZ-402665 , GZ 402665

NEW DRUG APPROVALS
ONE TIME TO MAINTAIN THIS BLOG
$10.00
Sutimlimab-jome
(Heavy chain)
EVQLVESGGG LVKPGGSLRL SCAASGFTFS NYAMSWVRQA PGKGLEWVAT ISSGGSHTYY
LDSVKGRFTI SRDNSKNTLY LQMNSLRAED TALYYCARLF TGYAMDYWGQ GTLVTVSSAS
TKGPSVFPLA PCSRSTSEST AALGCLVKDY FPEPVTVSWN SGALTSGVHT FPAVLQSSGL
YSLSSVVTVP SSSLGTKTYT CNVDHKPSNT KVDKRVESKY GPPCPPCPAP EFEGGPSVFL
FPPKPKDTLM ISRTPEVTCV VVDVSQEDPE VQFNWYVDGV EVHNAKTKPR EEQFNSTYRV
VSVLTVLHQD WLNGKEYKCK VSNKGLPSSI EKTISKAKGQ PREPQVYTLP PSQEEMTKNQ
VSLTCLVKGF YPSDIAVEWE SNGQPENNYK TTPPVLDSDG SFFLYSRLTV DKSRWQEGNV
FSCSVMHEAL HNHYTQKSLS LSLGK
(Light chain)
QIVLTQSPAT LSLSPGERAT MSCTASSSVS SSYLHWYQQK PGKAPKLWIY STSNLASGVP
SRFSGSGSGT DYTLTISSLQ PEDFATYYCH QYYRLPPITF GQGTKLEIKR TVAAPSVFIF
PPSDEQLKSG TASVVCLLNN FYPREAKVQW KVDNALQSGN SQESVTEQDS KDSTYSLSST
LTLSKADYEK HKVYACEVTH QGLSSPVTKS FNRGEC
(Disulfide bridge: H22-H96, H132-L216, H145-H201, H224-H’224, H227-H’227, H259-H319, H365-H423, H’22-H’96, H’132-L’216, H’145-H’201, H’259-H’319, H’365-H’423, L23-L89, L136-L196, L’23-L’89, L’136-L’196)
Sutimlimab-jome
スチムリマブ (遺伝子組換え)
| Formula | C6436H9912N1700O2016S46 |
|---|---|
| CAS | 2049079-64-1 |
| Mol weight | 144832.7369 |
- BIVV009
- Sutimlimab
- Sutimlimab [INN]
- Sutimlimab [WHO-DD]
- TNT009
- UNII-GNWE7KJ995
- WHO 10757
| Efficacy | Anti-anemic, Anti-complement C1s antibody |
|---|---|
| Comment | Monoclonal antibody |
FDA APPROVED 2/4/2022, To decrease the need for red blood cell transfusion due to hemolysis in cold agglutinin disease, Enjaymo
A Humanized Antibody for the Specific Inhibition of the Classical Complement Pathway.

Sutimlimab, sold under the brand name Enjaymo, is a monoclonal antibody that is used to treat adults with cold agglutinin disease (CAD).[1][2][3] It is given by intravenous infusion.[1]
The most common side effects include respiratory tract infection, viral infection, diarrhea, dyspepsia (indigestion), cough, arthralgia (joint stiffness), arthritis, and swelling in the lower legs and hands.[2]
Sutimlimab prevents complement-enhanced activation of autoimmune human B cells in vitro.[4]
This drug is being developed by Bioverativ, a Sanofi company.[5] Sutimlimab was approved for medical use in the United States in February 2022.[2][6]
Sutimlimab-jome, a classical complement inhibitor, is a humanized monoclonal antibody expressed by recombinant in Chinese hamster ovary (CHO) cells and produced in vitro using standard mammalian cell culture methods. Sutimlimab-jome is composed of two heterodimers. Each heterodimer is composed of a heavy and a light polypeptide chain. Each heavy chain (H-chain) is composed of 445 amino acids and each light chain (L-chain) contains 216 amino acids. Sutimlimab-jome has a molecular weight of approximately 147 kDa.
ENJAYMO (sutimlimab-jome) injection is a sterile, clear to slightly opalescent, colorless to slightly yellow, preservative-free solution for intravenous use. Each single-dose vial contains 1,100 mg sutimlimab-jome at a concentration of 50 mg/mL with a pH of 6.1. Each mL contains 50 mg of sutimlimab-jome and also contains polysorbate 80 (0.2 mg), sodium chloride (8.18 mg), sodium phosphate dibasic heptahydrate (0.48 mg), sodium phosphate monobasic monohydrate (1.13 mg), and Water for Injection, USP. https://www.rxlist.com/enjaymo-drug.htm#clinpharm
Medical uses
Sutimlimab is indicated to decrease the need for red blood cell transfusion due to hemolysis (red blood cell destruction) in adults with cold agglutinin disease (CAD).[1][2]
History
The effectiveness of sutimlimab was assessed in a study of 24 adults with cold agglutinin disease who had a blood transfusion within the past six months.[2] All participants received sutimlimab for up to six months and could choose to continue therapy in a second part of the trial.[2] Based on body weight, participants received either a 6.5g or 7.5g infusion of sutimlimab into their vein on day 0, day 7, and every 14 days through week 25.[2]
In total, 54% of participants responded to sutimlimab.[2] The response was defined in the study as an increase in hemoglobin (an indirect measurement of the amount of red blood cells that are not destroyed) of 2 g/dL or greater (or to 12 g/dL or greater), and no red blood cell transfusions after the first five weeks of treatment; and no other therapies for cold agglutinin disease as defined in the study.[2]
The application for sutimlimab received orphan drug,[2][7] breakthrough therapy,[2] and priority review designations.[2]
Society and culture
Names
Sutimlimab is the International nonproprietary name (INN).[8]
//////////////////////////////////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
CLIP
https://www.sanofi.com/en/media-room/press-releases/2022/2022-02-04-23-00-00-2379517
FDA approves Enjaymo™ (sutimlimab-jome), first treatment for use in patients with cold agglutinin disease
- Enjaymo is the only approved treatment to decrease the need for red blood cell transfusion due to hemolysis, the destruction of red blood cells, in adults with cold agglutinin disease (CAD)
- Enjaymo addresses a serious and chronic unmet medical need for adults living with CAD, a rare blood disorder
Paris, February 4, 2022. The U.S. Food and Drug Administration (FDA) has approved Enjaymo™ (sutimlimab-jome) to decrease the need for red blood cell transfusion due to hemolysis in adults with cold agglutinin disease (CAD). Enjaymo is the first and only approved treatment for people with CAD and works by inhibiting the destruction of red blood cells (hemolysis).
Bill Sibold
Executive Vice President, Head of Specialty Care
“Until now, people living with cold agglutinin disease haven’t had an approved treatment option to manage the constant destruction of red blood cells. Without healthy, viable red blood cells, a chain reaction of debilitating signs and symptoms can be triggered, starting with severe anemia. Enjaymo is the only approved treatment to inhibit red blood cell destruction in CAD and help stop the chain reaction from the start.”
CAD, a rare autoimmune hemolytic anemia, is caused by antibodies called cold agglutinins binding to the surface of red blood cells, which starts a process that causes the body’s immune system to mistakenly attack healthy red blood cells and cause their rupture (hemolysis). As red blood cells have the vital job of carrying oxygen throughout the body, patients with CAD may experience severe anemia, which can result in fatigue, weakness, shortness of breath, light-headedness, chest pain, irregular heartbeat, and other potential complications. CAD is a chronic and rare blood disorder that impacts the lives of an estimated 5,000 people in the U.S.
Enjaymo, targeting C1s in the classical complement pathway
Enjaymo is a humanized monoclonal antibody that is designed to selectively target and inhibit C1s in the classical complement pathway, which is part of the innate immune system. By blocking C1s, Enjaymo inhibits the activation of the complement cascade in the immune system and inhibits C1-activated hemolysis in CAD to prevent the abnormal destruction of healthy red blood cells. Enjaymo does not inhibit the lectin and alternative pathways.
Enjaymo Phase 3 pivotal CARDINAL study results supporting approval
The approval of Enjaymo in the U.S. is based on positive results from the 26-week open label, single arm pivotal Phase 3 study in patients with CAD (n=24) who have a recent history of blood transfusion, also known as the CARDINAL study.
Catherine Broome, MD
Associate professor of medicine at Georgetown University Lombardi Comprehensive Cancer Center, and a principal investigator in the CARDINAL study
“For people living with cold agglutinin disease, it is as if their body’s immune system is waging a war on itself. The relentless destruction of healthy red blood cells is a daily, silent reality for people with CAD. For the first time, we have a treatment that targets complement-mediated hemolysis, which is the underlying cause of the red blood cell destruction in many CAD patients. In the pivotal study, patients treated with sutimlimab had an improvement in anemia as measured by hemoglobin and bilirubin levels during the 26-week study.”
In the study, Enjaymo met its primary efficacy endpoint, which was a composite endpoint defined as the proportion of patients who achieved normalization of hemoglobin (Hgb) level ≥12 g/dL or demonstrated an increase from baseline in Hgb level ≥2 g/dL at the treatment assessment time point (mean value from weeks 23, 25, and 26) and no blood transfusion from weeks 5 through 26 or medications prohibited per the protocol from weeks 5 through 26. Secondary endpoints were also met, including improvements in hemoglobin and normalization of bilirubin.
- The majority of patients (54%; n=13) met the composite primary endpoint criteria with 63% (n=15) of patients achieving a hemoglobin ≥ 12 g/dL or an increase of at least 2 g/dL; 71% (n=17) of patients remaining transfusion-free after week five; and 92% (n=22) of patients did not use other CAD-related treatments.
- For the secondary measures on disease process, patients enrolled experienced a mean increase in hemoglobin level of 2.29 g/dL (SE: 0.308) at week 3 and 3.18 g/dL (SE: 0.476) at the 26-week treatment assessment timepoint from the mean baseline level of 8.6 g/dL. The mean reduction in bilirubin levels (n=14) was by -2.23 mg/dL (95% CI: -2.49 to -1.98) from a mean baseline level of 3.23 mg/dL (2.7-fold ULN).
In the CARDINAL study, the most common adverse reactions occurring in 10 percent or more of patients were respiratory tract infection, viral infection, diarrhea, dyspepsia, cough, arthralgia, arthritis, and peripheral edema. Serious adverse reactions were reported in 13 percent (3/24) of patients who received Enjaymo. These serious adverse reactions were streptococcal sepsis and staphylococcal wound infection (n=1), arthralgia (n=1), and respiratory tract infection (n=1). None of the adverse reactions led to discontinuation of Enjaymo in the study. Dosage interruptions due to an adverse reaction occurred in 17 percent (4/24) of patients who received Enjaymo.
Following the completion of the 26-week treatment period of CARDINAL (Part A), eligible patients continued to receive Enjaymo in an extension study.
The recommended dose of Enjaymo is based on body weight (6,500 mg for people 39-75 kg and 7,500 mg for people >75 kg). Enjaymo is administered intravenously weekly for the first two weeks with administration every two weeks thereafter.
Enjaymo is expected to be available in the U.S. in the coming weeks. The U.S. list price, or wholesale acquisition cost, of Enjaymo is $1,800 per vial. Actual costs to patients are generally anticipated to be lower as the list price does not reflect insurance coverage, co-pay support, or financial assistance from patient support programs. As part of our commitment to ensure treatment access and affordability for innovative therapies, Enjaymo Patient Solutions provides disease education, financial and co-pay assistance programs and other support services to eligible patients. For more information, please call 1-833-223-2428.
Enjaymo received FDA Breakthrough Therapy and Orphan Drug designation, and priority review, which is reserved for medicines that, if approved, would represent significant improvements in safety or efficacy in treating serious conditions. Outside of the U.S., sutimlimab has been submitted to regulatory authorities in Europe and Japan and reviews are ongoing.
About Sanofi
We are an innovative global healthcare company, driven by one purpose: we chase the miracles of science to improve people’s lives. Our team, across some 100 countries, is dedicated to transforming the practice of medicine by working to turn the impossible into the possible. We provide potentially life-changing treatment options and life-saving vaccine protection to millions of people globally, while putting sustainability and social responsibility at the center of our ambitions.
Sanofi is listed on EURONEXT: SAN and NASDAQ: SNY
References
- ^ Jump up to:a b c d https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/761164s000lbl.pdf
- ^ Jump up to:a b c d e f g h i j k l “FDA approves treatment for adults with rare type of anemia”. U.S. Food and Drug Administration. 4 February 2022. Retrieved 6 February 2022.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ Tvedt TH, Steien E, Øvrebø B, Haaverstad R, Hobbs W, Wardęcki M, et al. (February 2022). “Sutimlimab, an investigational C1s inhibitor, effectively prevents exacerbation of hemolytic anemia in a patient with cold agglutinin disease undergoing major surgery”. American Journal of Hematology. 97 (2): E51–E54. doi:10.1002/ajh.26409. PMID 34778998. S2CID 244116614.
- ^ Nikitin PA, Rose EL, Byun TS, Parry GC, Panicker S (February 2019). “C1s Inhibition by BIVV009 (Sutimlimab) Prevents Complement-Enhanced Activation of Autoimmune Human B Cells In Vitro”. Journal of Immunology. 202 (4): 1200–1209. doi:10.4049/jimmunol.1800998. PMC 6360260. PMID 30635392.
- ^ “Sutimlimab FDA Approval Status”. FDA. 19 May 2020.
- ^ “FDA approves Enjaymo (sutimlimab-jome), first treatment for use in patients with cold agglutinin disease”. Sanofi (Press release). 4 February 2022. Retrieved 6 February 2022.
- ^ “Sutimlimab Orphan Drug Designations and Approvals”. U.S. Food and Drug Administration (FDA). 27 July 2016. Retrieved 6 February 2022.
- ^ World Health Organization (2018). “International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 80”. WHO Drug Information. 32 (3). hdl:10665/330907.
External links
- “Sutimlimab”. Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT03347396 for “A Study to Assess the Efficacy and Safety of BIVV009 (Sutimlimab) in Participants With Primary Cold Agglutinin Disease Who Have a Recent History of Blood Transfusion (Cardinal Study)” at ClinicalTrials.gov
//////////////Sutimlimab-jome, Enjaymo, FDA 2022, APPROVALS 2022, agglutinin disease, BIVV009, TNT009, UNII-GNWE7KJ995, WHO 10757, PEPTIDE, MONOCLONAL ANTIBODY, スチムリマブ (遺伝子組換え),

NEW DRUG APPROVALS
ONE TIME TO MAINTAIN THIS BLOG SUBSCRIPTIONS
$10.00
Faricimab-svoa

(A chain)
QVQLVQSGAE VKKPGASVKV SCKASGYTFT GYYMHWVRQA PGQGLEWMGW INPNSGGTNY
AQKFQGRVTM TRDTSISTAY MELSRLRSDD TAVYYCARSP NPYYYDSSGY YYPGAFDIWG
QGTMVTVSSA SVAAPSVFIF PPSDEQLKSG TASVVCLLNN FYPREAKVQW KVDNALQSGN
SQESVTEQDS KDSTYSLSST LTLSKADYEK HKVYACEVTH QGLSSPVTKS FNRGECDKTH
TCPPCPAPEA AGGPSVFLFP PKPKDTLMAS RTPEVTCVVV DVSHEDPEVK FNWYVDGVEV
HNAKTKPREE QYNSTYRVVS VLTVLAQDWL NGKEYKCKVS NKALGAPIEK TISKAKGQPR
EPQVCTLPPS RDELTKNQVS LSCAVKGFYP SDIAVEWESN GQPENNYKTT PPVLDSDGSF
FLVSKLTVDK SRWQQGNVFS CSVMHEALHN AYTQKSLSLS PGK
(B chain)
EVQLVESGGG LVQPGGSLRL SCAASGYDFT HYGMNWVRQA PGKGLEWVGW INTYTGEPTY
AADFKRRFTF SLDTSKSTAY LQMNSLRAED TAVYYCAKYP YYYGTSHWYF DVWGQGTLVT
VSSASTKGPS VFPLAPSSKS TSGGTAALGC LVKDYFPEPV TVSWNSGALT SGVHTFPAVL
QSSGLYSLSS VVTVPSSSLG TQTYICNVNH KPSNTKVDKK VEPKSCDKTH TCPPCPAPEA
AGGPSVFLFP PKPKDTLMAS RTPEVTCVVV DVSHEDPEVK FNWYVDGVEV HNAKTKPREE
QYNSTYRVVS VLTVLAQDWL NGKEYKCKVS NKALGAPIEK TISKAKGQPR EPQVYTLPPC
RDELTKNQVS LWCLVKGFYP SDIAVEWESN GQPENNYKTT PPVLDSDGSF FLYSKLTVDK
SRWQQGNVFS CSVMHEALHN AYTQKSLSLS PGK
(C chain)
DIQLTQSPSS LSASVGDRVT ITCSASQDIS NYLNWYQQKP GKAPKVLIYF TSSLHSGVPS
RFSGSGSGTD FTLTISSLQP EDFATYYCQQ YSTVPWTFGQ GTKVEIKRTV AAPSVFIFPP
SDEQLKSGTA SVVCLLNNFY PREAKVQWKV DNALQSGNSQ ESVTEQDSKD STYSLSSTLT
LSKADYEKHK VYACEVTHQG LSSPVTKSFN RGEC
(D chain)
SYVLTQPPSV SVAPGQTARI TCGGNNIGSK SVHWYQQKPG QAPVLVVYDD SDRPSGIPER
FSGSNSGNTA TLTISRVEAG DEADYYCQVW DSSSDHWVFG GGTKLTVLSS ASTKGPSVFP
LAPSSKSTSG GTAALGCLVK DYFPEPVTVS WNSGALTSGV HTFPAVLQSS GLYSLSSVVT
VPSSSLGTQT YICNVNHKPS NTKVDKKVEP KSC
(Disulfide bridge: A22-A96, A156-A216, A236-D213, A242-B232, A245-B235, A277-A337, A365-A441, B22-B96, B150-B206, B226-C214, B267-B327, B360-B431, B23-B88, B134-B194, D22-D87, D137-D193)
Faricimab
| Formula | C6506H9968N1724O1026S45 |
|---|---|
| CAS | 1607793-29-2 |
| Mol weight | 130194.6203 |
Faricimab-svoa
FDA APPROVED 1/28/2022, Vabysmo
To treat neovascular (wet) aged-related macular degeneration and diabetic macular edema
RO6867461
- Faricimab
- Faricimab [INN]
- RG-7716
- RG7716
- RO-6867461
- RO6867461
- UNII-QC4F7FKK7I
- WHO 10563

| Efficacy | Angiogenesis inhibitor, Anti-angiopoietin 2 antibody, Anti-VEGF antibody |
|---|---|
| Comment | Antibody Opthamology indications in patients susceptible to blocking of vascular endothelial growth factor A (VEGF-A) and angiopoietin-2 (Ang-2) |
Faricimab, sold under the brand name Vabysmo, is a monoclonal antibody used for the treatment of neovascular age-related macular degeneration (nAMD) and diabetic macular edema (DME).[1] Faricimab is a bispecific monoclonal antibody.[2]
Faricimab was developed by Roche. Faricimab completed Phase III trials[3] and was approved for use in the United States by the Food and Drug Administration in January 2022.[1][4]
FDA Approves Faricimab to Treat Wet AMD and DME\
FDA Approves Faricimab to Treat Wet AMD and DMEFebruary 1, 2022
This represents the approval of the first bispecific antibody to treat wet age-related macular degeneration (AMD) and diabetic macular edema (DME).
https://www.ajmc.com/view/fda-approves-fariximab-to-treat-wet-amd-and-dme
The FDA has approved faricimab-svoa (Vabysmo; Genentech) to treat 2 leading causes of vision loss: wet, or neovascular, age-related macular degeneration (AMD) and diabetic macular edema (DME).
After 4 initial monthly doses, faricimab is delivered as injections from 1 to 4 months apart in the first year while the current standard of care for wet AMD and DME requires injections every 1 to 2 months. In wet AMD, patients receive the 4 monthly injections first and then based on outcomes may receive their subsequent treatments every 2, 3, or 4 months. For DME, after the 4 initial monthly injections, treatment is extended or reduced based on outcomes, with a range of 1 to 4 months between doses.
The treatment targets and inhibits pathways involving angiopoietin-2 and vascular endothelial growth factor-A (VEGF-A), which are thought to contribute to vision loss by destabilizing blood vessels.
“Vabysmo represents an important step forward for ophthalmology. It is the first bispecific antibody approved for the eye and a major advance in treating retinal conditions such as wet AMD and diabetic macular edema,” Charles Wykoff, MD, PhD, director of research at Retina Consultants of Texas in Houston and a Vabysmo phase 3 investigator, said in a statement. “With Vabysmo, we now have the opportunity to offer patients a medicine that could improve their vision, potentially lowering treatment burden with fewer injections over time.”
The FDA approved faricimab on the results from 4 phase 3 studies: TENAYA and LUCERNE for wet AMD and YOSEMITE and RHINE for DME. All 4 studies were randomized, multicenter, double-masked, global trials.
TENAYA and LUCERNE were identical: 1329 treatment-naive patients with wet AMD, aged 50 and older, were assigned 1:1 to faricimab up to every 16 weeks or aflibercept every 8 weeks. YOSEMITE and RHINE were also identical: 1891 patients with vision loss due to DME were randomly assigned 1:1:1 to faricimab every 8 weeks, faricimab per personalized treatment interval, or aflibercept every 8 weeks.
For all trials, faricimab was noninferior to aflibercept and the incidence of ocular adverse events was comparable. The researchers determined that the longer time between dosing intervals combined with the visual benefits of faricimab reduced the burden in patients.
The 1-year results from these studies were published January 24 in The Lancet.1,2
“These data published in The Lancet reinforce the potential of faricimab as an important treatment option that may help improve and maintain vision while extending the time between treatments up to 4 months,” Levi Garraway, MD, PhD, chief medical officer and head of Global Product Development, said in a statement. “We remain deeply committed to developing new medicines such as faricimab that may help preserve sight in many people living with serious retinal conditions.”
Now that faricimab is approved, Genentech expects it to become available in the United States within weeks. Meanwhile, the European Medicines Agency is currently evaluating a Marketing Authorization Application for faricimab to treat wet AMD and DME.
There are additional trials—COMINO and BALATON—underway to evaluate the efficacy and safety of faricimab in people with macular edema following retinal vein occlusion. In addition, 2-year results for faricimab in DME will be presented at the Angiogeneisis, Exudation, and Degeneration 2022 meeting in February.
References
1. Heier JS, Khanani AM, Quezada Ruiz C, et al; TENAYA and LUCERNE Investigators. Efficacy, durability, and safety of intravitreal faricimab up to every 16 weeks for neovascular age-related macular degeneration (TENAYA and LUCERNE): two randomised, double-masked, phase 3, non-inferiority trials. Lancet. Published January 24, 2022. doi:10.1016/S0140-6736(22)00010-1
2. Wykoff CC, Abreu F, Adamis AP, et al. Efficacy, durability, and safety of intravitreal faricimab with extended dosing up to every 16 weeks in patients with diabetic macular oedema (YOSEMITE and RHINE): two randomised, double-masked, phase 3 trials. Lancet. Published online January 24, 2022. doi:10.1016/S0140-6736(22)00018-6

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Humanized |
| Target | VEGF-A, angiopoietin 2 |
| Clinical data | |
| Trade names | Vabysmo |
| Other names | RO6867461; faricimab-svoa |
| License data | US DailyMed: Faricimab |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only |
| Identifiers | |
| CAS Number | 1607793-29-2 |
| UNII | QC4F7FKK7I |
| KEGG | D11516 |
| Chemical and physical data | |
| Formula | C6506H9968N1724O1026S45 |
| Molar mass | 130197.05 g·mol−1 |
Society and culture
Names
Faricimab is the International Nonproprietary Name (INN).[5]
References
- ^ Jump up to:a b “FDA approves Roche’s Vabysmo, the first bispecific antibody for the eye, to treat two leading causes of vision loss”. Roche (Press release). 31 January 2022. Retrieved 31 January 2022.
- ^ Nicolò M, Ferro Desideri L, Vagge A, Traverso CE (March 2021). “Faricimab: an investigational agent targeting the Tie-2/angiopoietin pathway and VEGF-A for the treatment of retinal diseases”. Expert Opinion on Investigational Drugs. 30 (3): 193–200. doi:10.1080/13543784.2021.1879791. PMID 33471572. S2CID 231665201.
- ^ Khan M, Aziz AA, Shafi NA, Abbas T, Khanani AM (August 2020). “Targeting Angiopoietin in Retinal Vascular Diseases: A Literature Review and Summary of Clinical Trials Involving Faricimab”. Cells. 9 (8): 1869. doi:10.3390/cells9081869. PMC 7464130. PMID 32785136.
- ^ “FDA approves faricimab for treatment of wet AMD, DME”. Ophthalmology Times. 28 January 2022.
- ^ World Health Organization (2018). “International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 80”. WHO Drug Information. 32 (3). hdl:10665/330907.
External links
- “Faricimab”. Drug Information Portal. U.S. National Library of Medicine.
////////////Faricimab-svoa, APPROVALS 2022, FDA 2022, RO6867461, RO 6867461, PEPTIDE, MONOCLONAL ANTIBODY, RG 7716, WHO 10563, peptide

NEW DRUG APPROVALS
one time
$10.00
Tixagevimab
| (Heavy chain) QMQLVQSGPE VKKPGTSVKV SCKASGFTFM SSAVQWVRQA RGQRLEWIGW IVIGSGNTNY AQKFQERVTI TRDMSTSTAY MELSSLRSED TAVYYCAAPY CSSISCNDGF DIWGQGTMVT VSSASTKGPS VFPLAPSSKS TSGGTAALGC LVKDYFPEPV TVSWNSGALT SGVHTFPAVL QSSGLYSLSS VVTVPSSSLG TQTYICNVNH KPSNTKVDKR VEPKSCDKTH TCPPCPAPEF EGGPSVFLFP PKPKDTLYIT REPEVTCVVV DVSHEDPEVK FNWYVDGVEV HNAKTKPREE QYNSTYRVVS VLTVLHQDWL NGKEYKCKVS NKALPASIEK TISKAKGQPR EPQVYTLPPS REEMTKNQVS LTCLVKGFYP SDIAVEWESN GQPENNYKTT PPVLDSDGSF FLYSKLTVDK SRWQQGNVFS CSVMHEALHN HYTQKSLSLS PGK (Light chain) EIVLTQSPGT LSLSPGERAT LSCRASQSVS SSYLAWYQQK PGQAPRLLIY GASSRATGIP DRFSGSGSGT DFTLTISRLE PEDFAVYYCQ HYGSSRGWTF GQGTKVEIKR TVAAPSVFIF PPSDEQLKSG TASVVCLLNN FYPREAKVQW KVDNALQSGN SQESVTEQDS KDSTYSLSST LTLSKADYEK HKVYACEVTH QGLSSPVTKS FNRGEC (Disulfide bridge: H22-H96, H101-H106, H150-H206, H216-L216, H232-H’232, H235-H’235, H267-H327, H373-H431, H’22-H’96, H’101-H’106, H’150-H’206, H’226-L’216, H’267-H’327, H’373-H’431, L23-L89, L136-L196, L’23-L’89, L’136-L’196) |
Tixagevimab
FDA 2021, 2021/12/8
ANTI VIRAL, CORONA VIRUS, PEPTIDE
Monoclonal antibody
Treatment and prevention of SARS-CoV-2 infection
| Formula | C6488H10034N1746O2038S50 |
|---|---|
| CAS | 2420564-02-7 |
| Mol weight | 146704.817 |
- 2196
- AZD-8895
- AZD8895
- COV2-2196
- Tixagevimab
- Tixagevimab [INN]
- UNII-F0LZ415Z3B
- WHO 11776
- OriginatorVanderbilt University
- DeveloperAstraZeneca; INSERM; National Institute of Allergy and Infectious Diseases
- ClassAntivirals; Monoclonal antibodies
- Mechanism of ActionVirus internalisation inhibitors
- RegisteredCOVID 2019 infections
- 24 Dec 2021Pharmacodynamics data from a preclinical trial in COVID-2019 infections released by AstraZeneca
- 16 Dec 2021Pharmacodynamics data from a preclinical trial in COVID-2019 infections released by AstraZeneca
- 10 Dec 2021Registered for COVID-2019 infections (In the elderly, Prevention, In adults) in USA (IM) – Emergency Use Authorization
Tixagevimab/cilgavimab is a combination of two human monoclonal antibodies, tixagevimab (AZD8895) and cilgavimab (AZD1061) targeted against the surface spike protein of SARS-CoV-2[4][5] used to prevent COVID-19. It is being developed by British-Swedish multinational pharmaceutical and biotechnology company AstraZeneca.[6][7] It is co-packaged and given as two separate consecutive intramuscular injections (one injection per monoclonal antibody, given in immediate succession).[2]
/////////////////////////////////////////////////////////////////////

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
Development
In 2020, researchers at Vanderbilt University Medical Center discovered particularly potent monoclonal antibodies, isolated from COVID-19 patients infected with a SARS-CoV-2 circulating at that time. Initially designated COV2-2196 and COV2-2130, antibody engineering was used to transfer their SARS-CoV-2 binding specificity to IgG scaffolds that would last longer in the body, and these engineered antibodies were named AZD8895 and AZD1061, respectively (and the combination was called AZD7442).[8]
To evaluate the antibodies’ potential as monoclonal antibody based prophylaxis (prevention), the ‘Provent’ clinical trial enrolled 5,000 high risk but not yet infected individuals and monitored them for 15 months.[9][10] The trial reported that those receiving the cocktail showed a 77% reduction in symptomatic COVID-19 and that there were no severe cases or deaths. AstraZeneca also found that the antibody cocktail “neutralizes recent emergent SARS-CoV-2 viral variants, including the Delta variant“.[7]
In contrast to pre-exposure prophylaxis, the Storm Chaser study of already-exposed people (post-exposure prophylaxis) did not meet its primary endpoint, which was prevention of symptomatic COVID-19 in people already exposed. AZD7442 was administered to 1,000 volunteers who had recently been exposed to COVID.[9]
Regulatory review
In October 2021, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) started a rolling review of tixagevimab/cilgavimab, which is being developed by AstraZeneca AB, for the prevention of COVID-19 in adults.[11]
Also in October 2021, AstraZeneca requested Emergency Use Authorization for tixagevimab/cilgavimab to prevent COVID-19 from the U.S. Food and Drug Administration (FDA).[12][13]
Emergency use authorization
On 14 November 2021, Bahrain granted emergency use authorization.[14]
On 8 December 2021, the U.S. Food and Drug Administration (FDA) granted emergency use authorization of this combination to prevent COVID-19 (before exposure) in people with weakened immunity or who cannot be fully vaccinated due to a history of severe reaction to coronavirus vaccines.[15] The FDA issued an emergency use authorization (EUA) for AstraZeneca’s Evusheld (tixagevimab co-packaged with cilgavimab and administered together) for the pre-exposure prophylaxis (prevention) of COVID-19 in certain people aged 12 years of age and older weighing at least 40 kilograms (88 lb).[2] The product is only authorized for those individuals who are not currently infected with the SARS-CoV-2 virus and who have not recently been exposed to an individual infected with SARS-CoV-2.[2]
References
- ^ “Evusheld- azd7442 kit”. DailyMed. Retrieved 4 January 2022.
- ^ Jump up to:a b c d “Coronavirus (COVID-19) Update: FDA Authorizes New Long-Acting Monoclonal Antibodies for Pre-exposure Prevention of COVID-19 in Certain Individuals”. U.S. Food and Drug Administration (FDA) (Press release). 8 December 2021. Retrieved 9 December 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain. - ^ O’Shaughnessy, Jacqueline A. (20 December 2021). “Re: Emergency Use Authorization 104” (PDF). Food and Drug Administration. Letter to AstraZeneca Pharmaceuticals LP | Attention: Stacey Cromer Berman, PhD. Archived from the original on 29 December 2021. Retrieved 18 January 2022.
- ^ “IUPHAR/BPS Guide to PHARMACOLOGY”. IUPHAR. 27 December 2021. Retrieved 27 December 2021.
- ^ “IUPHAR/BPS Guide to PHARMACOLOGY”. IUPHAR. 27 December 2021. Retrieved 27 December 2021.
- ^ Ray, Siladitya (21 August 2021). “AstraZeneca’s Covid-19 Antibody Therapy Effective In Preventing Symptoms Among High-Risk Groups, Trial Finds”. Forbes. ISSN 0015-6914. Archived from the original on 21 August 2021. Retrieved 18 January 2022.
- ^ Jump up to:a b Goriainoff, Anthony O. (20 August 2021). “AstraZeneca Says AZD7442 Antibody Phase 3 Trial Met Primary Endpoint in Preventing Covid-19”. MarketWatch. Archived from the original on 21 August 2021. Retrieved 18 January 2022.
- ^ Dong J, Zost SJ, Greaney AJ, Starr TN, Dingens AS, Chen EC, et al. (October 2021). “Genetic and structural basis for SARS-CoV-2 variant neutralization by a two-antibody cocktail”. Nature Microbiology. 6 (10): 1233–1244. doi:10.1038/s41564-021-00972-2. ISSN 2058-5276. PMC 8543371. PMID 34548634.
- ^ Jump up to:a b Haridy, Rich (23 August 2021). “”Game-changing” antibody cocktail prevents COVID-19 in the chronically ill”. New Atlas. Retrieved 23 August 2021.
- ^ “AZD7442 PROVENT Phase III prophylaxis trial met primary endpoint in preventing COVID-19”. AstraZeneca (Press release). 20 August 2021. Retrieved 15 October 2021.
- ^ “EMA starts rolling review of Evusheld (tixagevimab and cilgavimab)”. European Medicines Agency. 14 October 2021. Retrieved 15 October 2021.
- ^ “AZD7442 request for Emergency Use Authorization for COVID-19 prophylaxis filed in US”. AstraZeneca US (Press release). 5 October 2021. Retrieved 15 October 2021.
- ^ “AZD7442 request for Emergency Use Authorization for COVID-19 prophylaxis filed in US”. AstraZeneca (Press release). 5 October 2021. Retrieved 15 October 2021.
- ^ Abd-Alaziz, Moaz; Elhamy, Ahmad (14 November 2021). Macfie, Nick (ed.). “Bahrain authorizes AstraZeneca’s anti-COVID drug for emergency use”. Reuters. Archived from the original on 23 November 2021. Retrieved 18 January 2022.
- ^ Mishra, Manas; Satija, Bhanvi (8 December 2021). Dasgupta, Shounak (ed.). “U.S. FDA authorizes use of AstraZeneca COVID-19 antibody cocktail”. Reuters. Archived from the original on 13 January 2022. Retrieved 18 January 2022.
External links
“Tixagevimab”. Drug Information Portal. U.S. National Library of Medicine.
- “Cilgavimab”. Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT04625972 for “Phase III Double-blind, Placebo-controlled Study of AZD7442 for Post-exposure Prophylaxis of COVID-19 in Adults (STORM CHASER)” at ClinicalTrials.gov
- Clinical trial number NCT04625725 for “Phase III Double-blind, Placebo-controlled Study of AZD7442 for Pre-exposure Prophylaxis of COVID-19 in Adult. (PROVENT)” at ClinicalTrials.gov
| Tixagevimab (teal, right) and cilgavimab (purple, left) binding the spike protein RBD. From PDB: 7L7E. | |
| Combination of | |
|---|---|
| Tixagevimab | Monoclonal antibody |
| Cilgavimab | Monoclonal antibody |
| Clinical data | |
| Trade names | Evusheld |
| Other names | AZD7442 |
| License data | US DailyMed: Tixagevimab |
| Routes of administration | Intramuscular |
| ATC code | J06BD03 (WHO) |
| Legal status | |
| Legal status | US: ℞-only via emergency use authorization[1][2][3] |
| Identifiers | |
| KEGG | D12262 |
| Clinical data | |
|---|---|
| Drug class | Antiviral |
| ATC code | None |
| Identifiers | |
| CAS Number | 2420564-02-7 |
| DrugBank | DB16394 |
| UNII | F0LZ415Z3B |
| KEGG | D11993 |
| Chemical and physical data | |
| Formula | C6488H10034N1746O2038S50 |
| Molar mass | 146706.82 g·mol−1 |
| Clinical data | |
|---|---|
| Drug class | Antiviral |
| ATC code | None |
| Identifiers | |
| CAS Number | 2420563-99-9 |
| DrugBank | DB16393 |
| UNII | 1KUR4BN70F |
| KEGG | D11994 |
| Chemical and physical data | |
| Formula | C6626H10218N1750O2078S44 |
| Molar mass | 149053.44 g·mol−1 |
/////////////////Tixagevimab, ANTI VIRAL, CORONA VIRUS, PEPTIDE, Monoclonal antibody, SARS-CoV-2 , WHO 11776, 2196, AZD-8895, AZD 8895, COV2-2196, COVID 19
NEW DRUG APPROVALS
ONE TIME
$10.00
Tezepelumab-ekko

(Heavy chain)
QMQLVESGGG VVQPGRSLRL SCAASGFTFR TYGMHWVRQA PGKGLEWVAV IWYDGSNKHY
ADSVKGRFTI TRDNSKNTLN LQMNSLRAED TAVYYCARAP QWELVHEAFD IWGQGTMVTV
SSASTKGPSV FPLAPCSRST SESTAALGCL VKDYFPEPVT VSWNSGALTS GVHTFPAVLQ
SSGLYSLSSV VTVPSSNFGT QTYTCNVDHK PSNTKVDKTV ERKCCVECPP CPAPPVAGPS
VFLFPPKPKD TLMISRTPEV TCVVVDVSHE DPEVQFNWYV DGVEVHNAKT KPREEQFNST
FRVVSVLTVV HQDWLNGKEY KCKVSNKGLP APIEKTISKT KGQPREPQVY TLPPSREEMT
KNQVSLTCLV KGFYPSDIAV EWESNGQPEN NYKTTPPMLD SDGSFFLYSK LTVDKSRWQQ
GNVFSCSVMH EALHNHYTQK SLSLSPGK
(Light chain)
SYVLTQPPSV SVAPGQTARI TCGGNNLGSK SVHWYQQKPG QAPVLVVYDD SDRPSWIPER
FSGSNSGNTA TLTISRGEAG DEADYYCQVW DSSSDHVVFG GGTKLTVLGQ PKAAPSVTLF
PPSSEELQAN KATLVCLISD FYPGAVTVAW KADSSPVKAG VETTTPSKQS NNKYAASSYL
SLTPEQWKSH RSYSCQVTHE GSTVEKTVAP TECS
(Disulfide bridge: H22-H96, H136-L213, H149-H205, H224-H’224, H225-H’225, H228-H’228, H231-H’231, H262-H322, H368-H426, H’22-H’96, H’136-L’213, H’149-H’205, H’262-H’322, H’368-H’426, L22-L87, L136-L195, L’22-L’87, L’136-L’195)
Tezepelumab-ekko
テゼペルマブ (遺伝子組換え)
| Formula | C6400H9844N1732O1992S52 |
|---|---|
| CAS | 1572943-04-4 |
| Mol weight | 144588.4306 |
PEPTIDE
UD FDA APPROVED, 12/17/2021, To treat severe asthma as an add-on maintenance therapy , Tezspire
Monoclonal antibody
Treatment of asthma and atopic dermatitis
Tezepelumab, sold under the brand name Tezspire, is a human monoclonal antibody used for the treatment of asthma.[4][5]
It blocks thymic stromal lymphopoietin (TSLP),[2] an epithelial cytokine that has been suggested to be critical in the initiation and persistence of airway inflammation.[6]
It was approved for medical use in the United States in December 2021.[2][3]
Medical uses
Tezepelumab is indicated for the add-on maintenance treatment of people aged twelve years and older with severe asthma.[2]
Research
In Phase III trials, tezepelumab demonstrated efficacy compared to placebo for patients with severe, uncontrolled asthma.[7][8]
Structural studies by X-ray crystallography showed that Tezepelumab competes against a critical part of the TSLPR binding site on TSLP.[1]
It is being studied for the treatment of chronic obstructive pulmonary disease, chronic rhinosinusitis with nasal polyps, chronic spontaneous urticaria and eosinophilic esophagitis (EoE).[3]

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////

References
- ^ Jump up to:a b Verstraete K, Peelman F, Braun H, Lopez J, Van Rompaey D, Dansercoer A, et al. (April 2017). “Structure and antagonism of the receptor complex mediated by human TSLP in allergy and asthma”. Nature Communications. 8 (1): 14937. Bibcode:2017NatCo…814937V. doi:10.1038/ncomms14937. PMC 5382266. PMID 28368013.
- ^ Jump up to:a b c d https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/761224s000lbl.pdf
- ^ Jump up to:a b c “Tezspire (tezepelumab) approved in the US for severe asthma”. AstraZeneca (Press release). 17 December 2021. Retrieved 17 December 2021.
- ^ Marone G, Spadaro G, Braile M, Poto R, Criscuolo G, Pahima H, et al. (November 2019). “Tezepelumab: a novel biological therapy for the treatment of severe uncontrolled asthma”. Expert Opinion on Investigational Drugs. 28 (11): 931–940. doi:10.1080/13543784.2019.1672657. PMID 31549891. S2CID 202746054.
- ^ Matera MG, Rogliani P, Calzetta L, Cazzola M (February 2020). “TSLP Inhibitors for Asthma: Current Status and Future Prospects”. Drugs. 80 (5): 449–458. doi:10.1007/s40265-020-01273-4. PMID 32078149. S2CID 211194472.
- ^ “Tezepelumab granted Breakthrough Therapy Designation by US FDA”. AstraZeneca (Press release). 7 September 2018.
- ^ “Studies found for: Tezepelumab”. ClinicalTrials.Gov. National Library of Medicine, National Institutes of Health, U.S. Department of Health and Human Services.
- ^ Menzies-Gow A, Corren J, Bourdin A, Chupp G, Israel E, Wechsler ME, et al. (May 2021). “Tezepelumab in Adults and Adolescents with Severe, Uncontrolled Asthma”. New England Journal of Medicine. 384 (19): 1800–09. doi:10.1056/NEJMoa2034975. PMID 33979488. S2CID 234484931.
External links
- “Tezepelumab”. Drug Information Portal. U.S. National Library of Medicine.
- Clinical trial number NCT02054130 for “Study to Evaluate the Efficacy and Safety of MEDI9929 (AMG 157) in Adult Subjects With Inadequately Controlled, Severe Asthma” at ClinicalTrials.gov
- Clinical trial number NCT03347279 for “Study to Evaluate Tezepelumab in Adults & Adolescents With Severe Uncontrolled Asthma (NAVIGATOR)” at ClinicalTrials.gov
| Structural basis for inhibition of TSLP-signaling by Tezepelumab (PDB 5J13)[1] | |
| Monoclonal antibody | |
|---|---|
| Type | Whole antibody |
| Source | Human |
| Target | thymic stromal lymphopoietin (TSLP) |
| Clinical data | |
| Trade names | Tezspire |
| Other names | MEDI9929, AMG 157, tezepelumab-ekko |
| License data | US DailyMed: Tezepelumab |
| Routes of administration | Subcutaneous |
| ATC code | None |
| Legal status | |
| Legal status | US: ℞-only [2][3] |
| Identifiers | |
| CAS Number | 1572943-04-4 |
| DrugBank | DB15090 |
| ChemSpider | None |
| UNII | RJ1IW3B4QX |
| KEGG | D11771 |
| Chemical and physical data | |
| Formula | C6400H9844N1732O1992S52 |
| Molar mass | 144590.40 g·mol−1 |
////////////Tezepelumab-ekko, Tezspire, PEPTIDE, APPROVALS 2021, FDA 2021, Monoclonal antibody
, asthma, atopic dermatitis, ANTI INFLAMATORY, テゼペルマブ (遺伝子組換え)

NEW DRUG APPROVALS
ONE TIME
$10.00
Efgartigimod alfa-fcab
DKTHTCPPCP APELLGGPSV FLFPPKPKDT LYITREPEVT CVVVDVSHED PEVKFNWYVD
GVEVHNAKTK PREEQYNSTY RVVSVLTVLH QDWLNGKEYK CKVSNKALPA PIEKTISKAK
GQPREPQVYT LPPSRDELTK NQVSLTCLVK GFYPSDIAVE WESNGQPENN YKTTPPVLDS
DGSFFLYSKL TVDKSRWQQG NVFSCSVMHE ALKFHYTQKS LSLSPGK
(Disulfide bridge: 6-6′, 9-9′, 41-101, 147-205, 41′-101′, 147′-205′)
Efgartigimod alfa-fcab
| Formula | C2310H3554N602O692S14 |
|---|---|
| CAS | 1821402-21-4 |
| Mol weight | 51279.464 |
US FDA APPROVED 12/17/2021, To treat generalized myasthenia gravis
Press Release, Vyvgart, BLA 761195
| エフガルチギモドアルファ (遺伝子組換え) |
PEPTIDE
Treatment of IgG-driven autoimmune diseases

AS ON DEC2021 3,491,869 VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@amcrasto
/////////////////////////////////////////////////////////////////////////////
https://www.fda.gov/news-events/press-announcements/fda-approves-new-treatment-myasthenia-gravis
FDA Approves New Treatment for Myasthenia Gravis
Approval is the First of a New Class of Medication for this Rare, Chronic, Autoimmune, Neuromuscular DiseaseFor Immediate Release:December 17, 2021
The U.S. Food and Drug Administration today approved Vyvgart (efgartigimod) for the treatment of generalized myasthenia gravis (gMG) in adults who test positive for the anti-acetylcholine receptor (AChR) antibody.
Myasthenia gravis is a chronic autoimmune, neuromuscular disease that causes weakness in the skeletal muscles (also called voluntary muscles) that worsens after periods of activity and improves after periods of rest. Myasthenia gravis affects voluntary muscles, especially those that are responsible for controlling the eyes, face, mouth, throat, and limbs. In myasthenia gravis, the immune system produces AChR antibodies that interfere with communication between nerves and muscles, resulting in weakness. Severe attacks of weakness can cause breathing and swallowing problems that can be life-threatening.
“There are significant unmet medical needs for people living with myasthenia gravis, as with many other rare diseases,” said Billy Dunn, M.D., director of the Office of Neuroscience in the FDA’s Center for Drug Evaluation and Research. “Today’s approval is an important step in providing a novel therapy option for patients and underscores the agency’s commitment to help make new treatment options available for people living with rare diseases.”
Vyvgart is the first approval of a new class of medication. It is an antibody fragment that binds to the neonatal Fc receptor (FcRn), preventing FcRn from recycling immunoglobulin G (IgG) back into the blood. The medication causes a reduction in overall levels of IgG, including the abnormal AChR antibodies that are present in myasthenia gravis.
The safety and efficacy of Vyvgart were evaluated in a 26-week clinical study of 167 patients with myasthenia gravis who were randomized to receive either Vyvgart or placebo. The study showed that more patients with myasthenia gravis with antibodies responded to treatment during the first cycle of Vyvgart (68%) compared to those who received placebo (30%) on a measure that assesses the impact of myasthenia gravis on daily function. More patients receiving Vyvgart also demonstrated response on a measure of muscle weakness compared to placebo.
The most common side effects associated with the use of Vyvgart include respiratory tract infections, headache, and urinary tract infections. As Vyvgart causes a reduction in IgG levels, the risk of infections may increase. Hypersensitivity reactions such as eyelid swelling, shortness of breath, and rash have occurred. If a hypersensitivity reaction occurs, discontinue the infusion and institute appropriate therapy. Patients using Vyvgart should monitor for signs and symptoms of infections during treatment. Health care professionals should administer appropriate treatment and consider delaying administration of Vyvgart to patients with an active infection until the infection is resolved.
The FDA granted this application Fast Track and Orphan Drug designations. The FDA granted the approval of Vyvgart to argenx BV.
///////////efgartigimod alfa-fcab, Vyvgart, FDA 2021,APPROVALS 2021, myasthenia gravis, argenx BV, Fast Track, Orphan Drug, PEPTIDE,
| エフガルチギモドアルファ (遺伝子組換え) |

NEW DRUG APPROVALS
one time
$10.00

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










