Home » Posts tagged 'Orphan Drug Designation' (Page 7)
Tag Archives: Orphan Drug Designation
Critical Outcome Technologies receives orphan drug designation for COTI-2

N’-(5,6,7,8-Tetrahydroquinolin-8-ylidene)-4-(2-pyridyl)piperazine-1-carbothiohydrazide
http://criticaloutcome.com/110819_COTI-2%20Fact%20Sheet.pdf
http://www.slideshare.net/trevorheisler/about-coti2
MW 366.483, C19 H22 N6 S
| PATENTS | WO 2008083491, WO 2010006438 |
Caspase 9 Activators
PKB beta/Akt2 Inhibitors
Critical Outcome Technologies,
Critical Outcome Technologies (COTI) (Originator) preclinical for ovary cancer
Critical Outcome Technologies has announced that the US Food and Drug Administration (FDA) has granted COTI-2 an Orphan Drug Designation for the treatment of ovarian cancer.
Critical Outcome Technologies president and CEO Dr Wayne Danter said that receiving the Orphan Drug Designation for COTI-2 speaks to the need for new treatment options for patients with ovarian cancer.
- COTI-2 | A Potential Breakthrough Therapy for Many Cancers June 11, 2013
- About COTI-2 Late preclinical drug candidate discovered using CHEMSAS® – the company’s proprietary, artificial intelligence-based drug discovery technology 2
- COTI-2 highlights 1 Potential breakthrough therapy for many cancers 2 Active against many cancers with mutations of the p53 gene 3 > 50% of all human cancers have a p53 mutation 3
- Why p53 is important? p53 is a tumour suppressing gene If mutated, cancers can develop & grow without control A mutation of the p53 gene is the most common mutation found in human cancer cells 4
- The future of cancer treatments COTI-2 targets and primarily destroys tumor cells Traditional chemotherapy kills growing & dividing cells, cancer or healthy COTI-2 would treat genetic mutations common in many types of cancer Most current treatments are organ specific (i.e. treatment for lung cancer, colon cancer, etc.) 5
- COTI-2 development progress Easily synthesized oral formulation with no stability issues Effective alone or in combination with approved cancer drugs In final two-species toxicity studies prior to FDA filing enabling human trials 6
http://www.google.com/patents/WO2008083491A1?cl=en
EXAMPLES
Synthesis of COTI-2 The synthesis of COTI-2, as depicted above, was conducted according to the following synthetic methodology:
DCM R T
H2N-NH2
lmidazol-1 -yl-(4-pyridin-2-yl-piperazin-1 -yl)-methanethione (or intermediate 3 above) was formed as follows. Λ/-(2-pyridyl) piperazine (MW 163.22, 0.91 ml, 6.0 mmoles, 1 eq) 2 was added to a solution of 1 ,1 ‘- thiocarbonyldiimidazole (MW 178.22, 1.069 g, 6.0 mmoles, 1 eq) 1 in 50 ml of dichloromethane at room temperature. The reaction mixture was stirred overnight at room temperature. The mixture was washed with water, dried \ over sodium sulfate, filtered and concentrated to provide imidazol-1-yl-(4- pyridin-2-yl-piperazin-1-yl)-methanethione (MW 273.36, 1.354 g, 4.95 mmol, 83% yield) 3, which was used without further purification. TLC (CH2CI2/MeOH: 95/5): Rf = 0.60, Product UV and Ninhydrin stain active. 1H-NMR (400 MHz, CDCI3), δ ppm: 3.72 (s, 4H), 4.02 (s, 4H), 6.67 (d, 1 H, J = 7 Hz), 6.72 (dd, 1 H, J = 7 and 5 Hz), 7.11 (s, 1 H), 7.24 (s, 1 H), 7.54 (t, 1 H, J = 7 Hz), 7.91 (s, 1 H), 8.20 (d, 1 H, J = 5 Hz).
Hydrazine hydrate (MW 50.06, 0.26 ml, 5.44 mmoles, 1.1 eq) was added to a solution of imidazol-1-yl-(4-pyridin-2-yl-piperazin-1-yl)- methanethione 3 (MW 210.30, 1.040 g, 4.95 mmol, 1 eq) in 30 ml of ethanol at room temperature. The reaction mixture was stirred under reflux for 2 hours. A white precipitate formed. This white solid was filtered off and rinsed with diethyl ether to yield 1-[Λ/-(2-pyridyl)-piperazine)-carbothioic acid hydrazide (MW 237.33, 0.86 g, 3.62 mmol, 73% yield) 4 as a white solid, and used without further purification. TLC (CH2CI2/MeOH: 95/5): Rf = 0.20, Product UV and Ninhydrin stain active. 1H-NMR (400 MHz, DMSO-d6), δ ppm: 3.53 (s, 4H), 3.85 (s, 4H), 6.66 (dd, 1 H, J = 8 and 5 Hz), 6.82 (d, 1 H, J = 8 Hz), 7.55 (t, 1 H, J = 8 Hz), 8.12 (d, 1 H, J = 5 Hz).
COTI-2
Finally, COTI-2 was formed as follows. 1-[Λ/-(2-pyridyl)-piperazine)- carbothioic acid hydrazide (MW 237.33, 0.475 g, 2.0 mmol, 1 eq) 4 and 6,7- dihydro-5H-quinolin-8-one (MW 147.18, 0.306 g, 2.0 mmol, 1 eq) 5 was dissolved in 15 ml of ethanol at room temperature. The mixture was then stirred under reflux for 20 hours. A yellow solid precipitated out of the solution. This solid was filtered off then rinsed with methanol and diethyl ether to yield COTI-2 (MW 366.48, 0.60 g, 1.64 mmol, 82% yield) as a yellow solid. TLC (CH2CI2/MeOH: 95/5): Rf = 0.75, Product UV and Ninhydrine stain active. HPLC analysis showed a mixture of isomers (approximately in 80/20 ratio), and >98% purity. During the HPLC Method Development, as expected, this product tends to be hydrolyzed in presence of TFA in mobile phase solution. MS (ESI+, 0.025% TFA in 50/50 MeOH/H2O): [M+H]+ = 367.1 , [M+Na]+ = 389.1 ; 1H-NMR (400 MHz, CDCI3), δ ppm (Major isomer): 2.09 (m, 2H), 2.92 (m, 4H), 3.67 (m, 4H), 4.27 (m, 4H), 6.69 (dd, 1 H, J = 8 and 5 Hz)1 7.25 (dd,
1 H, J = 8 and 5 Hz), 7.55 (d, 2H, J = 8 Hz), 8.23 (d, 1 H, J = 5 Hz), 8.63 (d, 1 H, \ J = 5 Hz), 14.76 (s, 1 H). δ ppm (Minor isomer): 2.09 (m, 2H), 3.14 (t, 4H, J = 6 Hz), 3.80 (m, 4H), 4.27 (m, 4H), 6.66 (m, 1 H), 7.31 (dd, 1 H, J = 8 and 5 Hz), 7.52 (m, 1 H), 7.70 (d, 1 H, J = 8 Hz), 8.23 (d, 1 H, J = 5 Hz), 8.53 (d, 1 H, J = 5 Hz), 15.65 (s, 1 H).
…………………………..
| WO2010006438A1 * | Jul 17, 2009 | Jan 21, 2010 | Critical Outcome Technologies Inc. | Thiosemicarbazone inhibitor compounds and cancer treatment methods |
| See also references of EP2121681A1 | ||
| 2 | * | SHRIDHAR ET AL.: ‘Synthesis & antiparasite activity of some new 1-(6/7-nitrobenzoxazin-3-yl)-4-substituted- 3-thiosemicarbazides & 4-disubstituted 3-(6-acetylbenzoxazin-3-one)thiosemicarbazo nes‘ INDIAN J. OF CHEM. vol. 26B, June 1987, pages 596 – 598, XP008109697 |
| 3 | * | WINKELMANN ET AL.: ‘Antimalarial and anticoccidial activity of 3-aryl-7-chloro-3,4-dihydriacridine-1,9-(2h ,10H)-diones‘ ARZHEIM.-FORSCH./DRUG RES. vol. 37, no. 6, 1987, pages 647 – 661, XP008109793 |
EffRx Pharmaceuticals receives FDA orphan drug designation for EX404
EffRx Pharmaceuticals has received US Food and Drug Administration (FDA) orphan-drug designation for its proprietary metformin-based product, EX404, for treatment of paediatric polycystic ovary syndrome (PCOS).
Also known as Stein-Leventhal syndrome, PCOS is a heterogeneous disorder of chronic anovulation and hyperandrogenism.
The syndrome is believed to occur due to hormonal imbalance caused by increased levels of androgens and insulin in the body.
EffRx Pharmaceuticals chairman and CEO Christer Rosén said the FDA’s orphan drug designation of EX404 is a significant step forward in the clinical development programme.
Vertex Pharmaceuticals: Another FDA Orphan Drug Designation For Cystic Fibrosis
| FDA Orphan Drug Designation Database Record | |
| Generic Name: | (R)-1-(2,2-difluorobenzo [d][1,3] dioxol-5-yl)-N-(1-(2,3-dihydroxypropyl)-6-fluoro-2-(1-hydroxy-2-methylpropan-2-yl)-1H-indol-5-yl) cyclopropanecarboxamide |
| Trade Name: | n/a |
| Date Designated: | 04-24-2014 |
| Orphan Designation: | Treatment of cystic fibrosis |
| Orphan Designation Status: | Designated |
| FDA Orphan Approval Status: | Not FDA Approved for Orphan Indication |
| Sponsor: | Vertex Pharmaceuticals Inc. 50 Northern Avenue Boston, MA 02210-1862 The sponsor address listed is the last reported by the sponsor to OOPD. |
.
VX-661 is a Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) corrector. VX-661 is being studied in combination with Kalydeco (Ivacaftor) for patients who have the F508del mutation. VX-661 is currently recruiting participants for a Phase II clinical trial to evaluate the safety and efficacy of VX-661 in combination with Kalydeco in subjects with CF who are homozygous (have 2 copies) for the F508del CFTR…
View original post 173 more words
MARIZOMIB, Salinosporamide A
MARIZOMIB
http://www.ama-assn.org/resources/doc/usan/marizomib.pdf
THERAPEUTIC CLAIM Antineoplastic
CHEMICAL NAMES
1. 6-Oxa-2-azabicyclo[3.2.0]heptane-3,7-dione, 4-(2-chloroethyl)-1-[(S)-(1S)-2-
cyclohexen-1-ylhydroxymethyl]-5-methyl-, (1R,4R,5S)-
2. (1R,4R,5S)-4-(2-chloroethyl)-1-{(S)-[(1S)-cyclohex-2-en-1-yl]hydroxymethyl}-5-methyl-
6-oxa-2-azabicyclo[3.2.0]heptane-3,7-dione
MOLECULAR FORMULA C15H20ClNO4
MOLECULAR WEIGHT 313.8
MANUFACTURER Nereus Pharmaceuticals, Inc.
NOTE….Nereus Pharmaceuticals was acquired by Triphase Research and Development in 2012.
CODE DESIGNATION NPI-0052
CAS REGISTRY NUMBER 437742-34-2
Scripps Institution of Oceanography (Originator)
mp, 168–170° C. (authentic sample: 168–170° C., 169–171° C. in Angew. Chem. Int. Ed., 2003, 42, 355–357); mixture mp, 168–170C.
[α]23 D −73.2 (c 0.49, MeOH), −72.9 (c 0.55, MeOH, in Angew. Chem. Int. Ed., 2003, 42, 355–357);
FTIR (film) νmax: 3406, 2955, 2920, 2844, 1823, 1701, 1257, 1076, 1012, 785, 691 cm−1;
1H NMR (CDCl3, 500 MHz): δ 10.62 (1H, br), 6.42 (1H, d, J=10.5 Hz), 5.88 (1H, m), 4.25 (1H, d, J=9.0 Hz), 4.14 (1H, m), 4.01 (1H, m), 3.17 (1H, t, J=7.0 Hz), 2.85 (1H, m), 2.48 (1H, m), 2.32 (2H, m), 2.07 (3H, s), 1.91 (2H, m), 1.66 (2H, m), 1.38 (1H, m);
13C NMR (CDCl3, 125 MHz): δ 176.92, 169.43, 129.08, 128.69, 86.32, 80.35, 70.98, 46.18, 43.28, 39.31, 29.01, 26.47, 25.35, 21.73, 20.00;
HRMS (ESI) calcd. for (M−H)− C15H19ClNO4 312.1003, found 312.1003.
Marizomib, a highly potent proteasome inhibitor, is in early clinical development at Triphase Research and Development I Corp for the treatment of relapsed or relapsed/refractory multiple myeloma. Phase I clinical trials have also been carried out for the treatment of solid tumors and lymphoma; however, no recent developments have been reported for these studies.
HDAC inhibitors halt tumor cell differentiation and growth, and when combined with marizomib in preclinical in vitro and in vivo studies, show additive and synergistic antitumor activities.
The compound was discovered from a new marine-obligate gram-positive actinomycete (Salinispora tropica). Preclinical studies suggest that this next-generation compound may be superior to other proteasome inhibitors, with broader target inhibition, faster onset and longer duration of action, higher potency, and oral and intravenous availability. By inhibiting proteasomes, marizomib prevents the breakdown of proteins involved in signal transduction, which blocks growth and induces apoptosis in cancer cells.
In 2013, orphan drug designation was assigned in the U.S. for the treatment of multiple myeloma.
The compound was originally developed by Nereus Pharmaceuticals, which was acquired by Triphase Research and Development in 2012.
marizomib is a naturally-occurring salinosporamide, isolated from the marine actinomycete Salinospora tropica, with potential antineoplastic activity. Marizomib irreversibly binds to and inhibits the 20S catalytic core subunit of the proteasome by covalently modifying its active site threonine residues; inhibition of ubiquitin-proteasome mediated proteolysis results in an accumulation of poly-ubiquitinated proteins, which may result in the disruption of cellular processes, cell cycle arrest, the induction of apoptosis, and the inhibition of tumor growth and angiogenesis. This agent more may more potent and selective than the proteasome inhibitor bortezomib
Marizomib (NPI-0052) is an oral, irreversible ββ-lactone derivative that binds selectively to the active proteasomal sites. In vivo studies with marizomib demonstrate reduced tumor growth without significant toxicity in myeloma xenograft models. A phase I trial in refractory and relapsed MM is under way.
Salinosporamide A is a potent proteasome inhibitor used as an anticancer agent that recently entered phase I human clinical trials for the treatment of multiple myeloma only three years after its discovery.[1][2] This novel marine natural product is produced by the recently described obligate marine bacteria Salinispora tropica and Salinispora arenicola, which are found in ocean sediment. Salinosporamide A belongs to a family of compounds, known collectively as salinosporamides, which possess a densely functionalized γ-lactam-β-lactone bicyclic core.
Salinosporamide A was discovered by William Fenical and Paul Jensen from Scripps Institution of Oceanography in La Jolla, CA. In preliminary screening, a high percentage of the organic extracts of cultured Salinospora strains possessed antibiotic and anticancer activities, which suggests that these bacteria are an excellent resource for drug discovery.Salinospora strain CNB-392 was isolated from a heat-treated marine sediment sample and cytotoxicity-guided fractionation of the crude extract led to the isolation of salinosporamide A. Although salinosporamide A shares an identical bicyclic ring structure with omuralide, it is uniquely functionalized. Salinosporamide A displayed potent in vitro cytotoxicity against HCT-116 human colon carcinoma with an IC50 value of 11 ng mL-1. This compound also displayed potent and highly selective activity in the NCI’s 60-cell-line panel with a mean GI50 value (the concentration required to achieve 50% growth inhibition) of less than 10 nM and a greater than 4 log LC50 differential between resistant and susceptible cell lines. The greatest potency was observed against NCI-H226 non-small cell lung cancer, SF-539 CNS cancer, SK-MEL-28 melanoma, and MDA-MB-435 breast cancer (all with LC50 values less than 10 nM). Salinosporamide A was tested for its effects on proteasome function because of its structural relationship to omuralide. When tested against purified 20S proteasome, salinosporamide A inhibited proteasomal chymotrypsin-like proteolytic activity with an IC50 value of 1.3 nM.[3] This compound is approximately 35 times more potent than omuralide which was tested as a positive control in the same assay. Thus, the unique functionalization of the core bicyclic ring structure of salinosporamide A appears to have resulted in a molecule that is a significantly more potent proteasome inhibitor than omuralide.[1]
Salinosporamide A inhibits proteasome activity by covalently modifying the active site threonine residues of the 20S proteasome.
Biosynthesis
It was originally hypothesized that salinosporamide B was a biosynthetic precursor to salinosporamide A due to their structural similarities.
It was thought that the halogenation of the unactivated methyl group was catalyzed by a non-heme iron halogenase.[4][5]Recent work using 13C-labeled feeding experiments reveal distinct biosynthetic origins of salinosporamide A and B.[4][6]
While they share the biosynthetic precursors acetate and presumed β-hydroxycyclohex-2′-enylalanine (3), they differ in the origin of the four-carbon building block that gives rise to their structural differences involving the halogen atom. A hybrid polyketide synthase-nonribosomal peptide synthetase (PKS-NRPS) pathway is most likely the biosynthetic mechanism in which acetyl-CoA and butyrate-derived ethylmalonyl-CoA condense to yield the β-ketothioester (4), which then reacts with (3) to generate the linear precursor (5).
The first stereoselective synthesis was reported by Rajender Reddy Leleti and E. J.Corey.[7] Later several routes to the total synthesis of salinosporamide A have been reported.[7][8][9][10]
In vitro studies using purified 20S proteasomes showed that salinosporamide A has lower EC50 for trypsin-like (T-L) activity than does Bortezomib. In vivo animal model studies show marked inhibition of T-L activity in response to salinosporamide A, whereas bortezomib enhances T-L proteasome activity.
Initial results from early-stage clinical trials of salinosporamide A in relapsed/refractory multiple myeloma patients were presented at the 2011 American Society of Hematology annual meeting.[11] Further early-stage trials of the drug in a number of different cancers are ongoing.[12]
- Feling RH, Buchanan GO, Mincer TJ, Kauffman CA, Jensen PR, Fenical W (2003). “Salinosporamide A: a highly cytotoxic proteasome inhibitor from a novel microbial source, a marine bacterium of the new genus salinospora”. Angew. Chem. Int. Ed. Engl. 42 (3): 355–7.doi:10.1002/anie.200390115. PMID 12548698.
- Chauhan D, Catley L, Li G et al. (2005). “A novel orally active proteasome inhibitor induces apoptosis in multiple myeloma cells with mechanisms distinct from Bortezomib”. Cancer Cell 8 (5): 407–19.doi:10.1016/j.ccr.2005.10.013. PMID 16286248.
- K. Lloyd, S. Glaser, B. Miller, Nereus Pharmaceuticals Inc.
- Beer LL, Moore BS (2007). “Biosynthetic convergence of salinosporamides A and B in the marine actinomycete Salinispora tropica”. Org. Lett. 9 (5): 845–8.doi:10.1021/ol063102o. PMID 17274624.
- Vaillancourt FH, Yeh E, Vosburg DA, Garneau-Tsodikova S, Walsh CT (2006). “Nature’s inventory of halogenation catalysts: oxidative strategies predominate”. Chem. Rev.106 (8): 3364–78. doi:10.1021/cr050313i.PMID 16895332.
- Tsueng G, McArthur KA, Potts BC, Lam KS (2007). “Unique butyric acid incorporation patterns for salinosporamides A and B reveal distinct biosynthetic origins”. Applied Microbiology and Biotechnology 75 (5): 999–1005. doi:10.1007/s00253-007-0899-7.PMID 17340108.
- Reddy LR, Saravanan P, Corey EJ (2004). “A simple stereocontrolled synthesis of salinosporamide A”. J. Am. Chem. Soc. 126 (20): 6230–1. doi:10.1021/ja048613p.PMID 15149210.
- Ling T, Macherla VR, Manam RR, McArthur KA, Potts BC (2007). “Enantioselective Total Synthesis of (-)-Salinosporamide A (NPI-0052)”.Org. Lett. 9 (12): 2289–92. doi:10.1021/ol0706051. PMID 17497868.
- Ma G, Nguyen H, Romo D (2007). “Concise Total Synthesis of (±)-Salinosporamide A, (±)-Cinnabaramide A, and Derivatives via a Bis-Cyclization Process: Implications for a Biosynthetic Pathway?”. Org. Lett. 9 (11): 2143–6. doi:10.1021/ol070616u. PMC 2518687.PMID 17477539.
- Endo A, Danishefsky SJ (2005). “Total synthesis of salinosporamide A”. J. Am. Chem. Soc. 127 (23): 8298–9.doi:10.1021/ja0522783. PMID 15941259.
- “Marizomib May Be Effective In Relapsed/Refractory Multiple Myeloma (ASH 2011)”. The Myeloma Beacon. 2012-01-23. Retrieved 2012-06-10.
- ClinicalTrials.gov: Marizomib
……………………………………………………
IMPORTANT PAPERS
Total synthesis of salinosporamide A
Org Lett 2008, 10(19): 4239
Entry to heterocycles based on indium-catalyzed conia-ene reactions: Asymmetric synthesis of (-)-salinosporamide A
Angew Chem Int Ed 2008, 47(33): 6244
A concise and straightforward total synthesis of (+/-)-salinosporamide A, based on a biosynthesis model
Org Biomol Chem 2008, 6(15): 2782
Formal synthesis of salinosporamide A starting from D-glucose
Synthesis (Stuttgart) 2009, 2009(17): 2983
Stereoselective functionalization of pyrrolidinone moiety towards the synthesis of salinosporamide A
Tetrahedron 2012, 68(32): 6504
………………
Salinosporamide A(1) was recently discovered by Fenical et al. as a bioactive product of a marine microorganism that is widely distributed in ocean sediments. Feeling, R. H.; Buchanan, G. O.; Mincer, T. J.; Kauffman, C. A.; Jensen, P. R.; Fenical, W., Angew. Chem. Int. Ed., 2003, 42, 355–357.
Structurally Salinosporamide A closely resembles the terrestrial microbial product omuralide (2a) that was synthesized by Corey et al. several years ago and demonstrated to be a potent inhibitor of proteasome function. See, (a) Corey, E. J.; Li, W. D., Z. Chem. Pharm. Bull., 1999, 47, 1–10; (b) Corey, E. J., Reichard, G. A.; Kania, R., Tetrahedron Lett., 1993, 34, 6977–6980; (c) Corey, E. J.; Reichard, G. A., J. Am. Chem. Soc., 1992, 114, 10677–10678; (d) Fenteany, G.; Standaert, R. F.; Reichard, G. A.; Corey, E. J.; Schreiber, S. L., Proc. Natl. Acad. Sci. USA, 1994, 91, 3358–3362.
Omuralide is generated by β-lactonization of the N-acetylcysteine thiolester lactacystin (2b) that was first isolated by the Omura group as a result of microbial screening for nerve growth factor-like activity. See, Omura, S., Fujimoto, T., Otoguro, K., Matsuzaki, K., Moriguchi, R., Tanaka, H., Sasaki, Y., Antibiot., 1991, 44, 113–116; Omura, S., Matsuzaki, K., Fujimoto, T., Kosuge, K., Furuya, T., Fujita, S., Nakagawa, A., J. Antibiot., 1991, 44, 117–118.
Salinosporamide A, the first compound Fenical’s group isolated from Salinospora, not only had a never-before-seen chemical structure 1, but is also a highly selective and potent inhibitor of cancer-cell growth. The compound is an even more effective proteasome inhibitor than omuralide and, in addition, it displays surprisingly high in vitro cytotoxic activity against many tumor cell lines (IC50values of 10 nM or less). Fenical et al. first found the microbe, which they’ve dubbed Salinospora, off the coasts of the Bahamas and in the Red Sea. See,Appl. Environ. Microbiol., 68, 5005 (2002).
Fenical et al. have shown that Salinospora species requires a salt environment to live. Salinospora thrives in hostile ocean-bottom conditions: no light, low temperature, and high pressure. The Fenical group has now identified Salinosporain five oceans, and with 10,000 organisms per cm3 of sediment and several distinct strains in each sample; and according to press reports, they’ve been able to isolate 5,000 strains. See, Chemical & Engineering News, 81, 37 (2003).
A great percentage of the cultures Fenical et al. have tested are said to have shown both anticancer and antibiotic activity. Like omuralide 2a, salinosporamide A inhibits the proteasome, an intracellular enzyme complex that destroys proteins the cell no longer needs. Without the proteasome, proteins would build up and clog cellular machinery. Fast-growing cancer cells make especially heavy use of the proteasome, so thwarting its action is a compelling drug strategy. See, Fenical et al., U.S. Patent Publication No. 2003-0157695A1
PATENTS
WO 2005113558
http://www.google.com/patents/US7183417
Part I. Synthesis of the Salinosporamide A(1)
EXAMPLE 1
(4S, 5R) Methyl 4,5-dihydro-2 (4-methoxyphenyl)-5-methyloxazole-4-carboxylate (4)
A mixture of (2S, 3R)-methyl 2-(4-methoxybenzamido)-3-hydroxybutanoate (3) (35.0 g, 131 mmol) and p-TsOH.H2O (2.5 g, 13.1 mmol) in toluene (400 mL) was heated at reflux for 12 h. The reaction mixture was diluted with water (200 mL) and extracted with EtOAc (3×200 mL). The combined organic layers were washed with water, brine and dried over Na2SO4. The solvent was removed in vacuo to give crude oxazoline as yellow oil. Flash column chromatography on silica gel (eluent 15% EtOAc-Hexanes) afforded the pure oxazoline (26.1 g, 80%) as solid.
Rf=0.51 (50% ethyl acetate in hexanes), mp, 86–87° C.; [α]23 D+69.4 (c 2.0, CHCl3); FTIR (film) νmax: 2955, 1750, 1545, 1355, 1187, 1011, 810 cm−1; 1HNMR(CDCl3, 400 MHz): δ 7.87 (2H, d, J=9.2 Hz), 6.84 (2H, d, J=8.8 Hz), 4.90 (1H, m), 4.40 (1H, d, J=7.6 Hz), 3.79 (3H,s), 3.71 (3H, s), 1.49 (3H, d, J=6.0 Hz); 13C NMR (CDCl3, 100 MHz): δ 171.93, 165.54, 162.64, 130.52, 119.80, 113.85, 78.91, 75.16, 55.51, 52.73, 21.14; HRMS (ESI) calcd for C13H16NO4 (M+H)+.250.1079, found 250.1084.
EXAMPLE 2
(4R, 5R)-Methyl 4-{(benzyloxy) methyl)}-4,5-dihydro-2-(4-methoxyphenyl)-5-methyloxazole-4-carboxylate (5)
To a solution of LDA (50 mmol, 1.0 M stock solution in THF) was added HMPA (24 mL, 215 mmol) at −78° C. and then oxazoline 4 (12.45 g, 50 mmol, in 20 mL THF) was added dropwise with stirring at −78° C. for 1 h to allow complete enolate formation. Benzyloxy chloromethyl ether (8.35 mL, 60 mmol) was added at this temperature and after stirring the mixture at −78° C. for 4 h, it was quenched with water (50 mL) and warmed to 23° C. for 30 min. Then the mixture was extracted with ethyl acetate (3×50 mL) and the combined organic phases were dried (MgSO4) and concentrated in vacuo. The crude product was purified by column chromatography (silica gel, ethyl acetate/hexanes, 1:4 then 1:3) to give the benzyl ether 5 (12.7 g, 69%).
Rf=0.59 (50% ethyl acetate in hexanes). [α]23 D−6.3 (c 1.0, CHCl3); FTIR (film) (νmax; 3050, 2975, 1724, 1642, 1607, 1252, 1027, 745, 697 cm−1; 1H NMR (CDCl3, 400 MHz): δ 7.96 (2H, d, J=9.2 Hz), 7.26 (5H, m), 6.90 (2H, J=8.8 Hz), 4.80 (1H, m), 4.61 (2H, s), 3.87 (3H, m), 3.81 (3H, s), 3.73 (3H, s), 1.34 (3H, d, J=6.8 Hz); 13C NMR (CDCl3, 100 MHZ): 6171.23, 165.47, 162.63, 138.25, 130.64, 128.52, 127.87, 127.77, 120.15, 113.87, 81.40, 79.92, 73.91, 73.43, 55.58, 52.45, 16.92; HRMS (ESI) calcd for C21H24O5 (M+H)+370.1654, found 370.1644.
EXAMPLE 3
(2R,3R)-Methyl 2-(4-methoxybenzylamino)-2-((benzyloxy)methyl)-3hydroxybutanoate (6)
To a solution of oxazoline 5 (18.45 g, 50 mmol) in AcOH (25 mL) at 23° C. was added in portions NaCNBH3 (9.3 g, 150 mmol). The reaction mixture was then stirred at 40° C. for 12 h to allow complete consumption of the starting material. The reaction mixture was diluted with water (100 mL), neutralized with solid Na2CO3 and the aqueous layer was extracted with ethyl acetate (3×100 mL). The combined organic phases were dried over NaSO4 and concentrated in vacuo. The crude product was purified by column chromatography (silica gel, ethyl acetate/hexanes, 1:5) to give the N-PMB amino alcohol 6 (16.78 g, 90%).
Rf=0.50 (50% ethyl acetate in hexanes). [α]23 D−9.1(c 1.0, CHCl3); FTIR (film) νmax; 3354, 2949, 1731, 1511, 1242, 1070, 1030, 820, 736, 697 cm−1; 1H NMR (CDCl3, 400 MHz): δ 7.32 (7H, m), 6.87 (2H, d, J=8.8 Hz), 4.55 (2H, m), 4.10 (1H, q, J=6.4 Hz), 3.85 (2H, dd, J=17.2, 10.0 Hz), 3.81 (3H, s,), 3.77 (3H, s), 3. 69 (2H, dd, J=22.8, 11.6 Hz), 3.22 (2H, bs), 1.16 (3H, d, J=6.0 Hz); 13C NMR (CDCl3, 100 MHz): δ 173.34, 159.03, 137.92, 132.51, 129.78, 128.67, 128.07, 127.98, 114.07, 73.80, 70.55, 69.82, 69.65, 55.51, 55.29, 47.68, 18.15; HRMS (ESI) calcd. for C21H28NO5 (M+H)+ 374.1967, found 374.1974.
EXAMPLE 4
(2R,3R)-Methyl-2-(N-(4-methoxybenzyl)acrylamido)-2-(benzyloxy)methyl)-3-hydroxybutanoate (7)
A solution of amino alcohol 6 (26.2 g, 68.5 mmol) in Et2O (200 mL) was treated with Et3N (14.2 mL, 102.8 mmol) and trimethylchlorosilane (10.4 mL, 82.2 mmol) at 23° C. and stirred for 12 h. After completion, the reaction mixture was diluted with ether (200 mL) and then resulting suspension was filtered through celite. The solvent was removed to furnish the crude product (31.2 g, 99%) in quantitative yield as viscous oil. A solution of this crude trimethylsilyl ether (31.1 g) in CH2Cl2 (200 mL) was charged with diisopropylethylamine (14.2 mL, 81.6 mmol) and then cooled to 0° C. Acryloyl chloride (6.64 mL, 82.2 mmol) was added dropwise with vigorous stirring and the reaction temperature was maintained at 0° C. until completion (1 h). The reaction mixture was then diluted with CH2Cl2 (100 mL) and the organic layer was washed with water and brine. The organic layer was separated and dried over Na2SO4. The solvent was removed to afford the crude acrylamide 7 as a viscous oil. The crude product was then dissolved in Et2O (200 mL) and stirred with 6N HCl (40 mL) at 23° C. for 1 h. The reaction mixture was diluted with water (100 mL) and concentrated to provide crude product. The residue was purified by column chromatography (silica gel, ethyl acetate/hexanes, 1:5 to 1:1) to give pure amide 7 (28.3 g, 96%) as colorless solid, mp 88–89° C.
Rf=0.40 (50% ethyl acetate in hexanes), [α]23 D−31.1 (c 0.45, CHCl3), FTIR (film) νmax; 3435, 2990, 1725, 1649, 1610, 1512, 1415, 1287, 1242, 1175, 1087, 1029, 732, 698 cm−1; 1H NMR (CDCl3, 500 MHz): δ 7.25 (5H, m), 7.15 (2H, d, J=6.0 Hz), 6.85 (2H, d, J=7.5 Hz), 6.38 (2H, d, J=6.0 Hz), 5.55 (1H, t, J=6.0 Hz), 4.81 (2H, s), 4.71 (1H, q, J=6.5 Hz), 4.35 (2H, s), 4.00 (1H, d, J=10.0 Hz), 3.80 (1H, d, J=10.0 Hz), 3.76 (3H, s), 3.75 (3H, s), 3.28 (1H, bs), 1.22 (3H, d, J=6.0 Hz); 13C NMR (CDCl3, 125 MHz): δ 171.87, 168.74, 158.81, 137.73, 131.04, 129.68, 128.58, 128.51, 127.94, 127.72, 127.20, 127.14, 114.21, 73.71, 70.42, 69.76, 67.65, 55.45, 52.52, 49.09, 18.88; HRMS (ESI) calcd. for C24H30NO6 (M+H)+428.2073, found 428.2073.
EXAMPLE 5
(R)-Methyl-2-(N-(4-methoxybenzyl)acrylamido)-2-(benzyloxy)methyl)-3-oxybutanoate (8)
To a solution of amide 7 (10.67 g, 25.0 mmol) in CH2Cl2 (100 mL) was added Dess-Martin periodinane reagent (12.75 g, 30.0 mmol, Aldrich Co.) at 23° C. After stirring for 1 h, the reaction mixture was quenched with aq NaHCO3—Na2S2O3 (1:1, 50 mL) and extracted with ethyl acetate (3×50 mL). The organic phase was dried and concentrated in vacuo to afford the crude ketone. The crude product was purified by column chromatography (silica gel, ethyl acetate/hexanes) to give pure keto amide 8 (10.2 g, 96%).
Rf=0.80 (50% ethyl acetate in hexanes), mp 85 to 86° C.; [α]23 D−12.8 (c 1.45, CHCl3); FTIR (film) νmax: 3030, 2995, 1733, 1717, 1510, 1256, 1178, 1088, 1027, 733, 697 cm−1; 1H NMR (CDCl3, 500 MHz): δ 7.30 (2H, d, J=8.0), 7.25 (3H, m), 7.11 (2H, m), 6.88 (2H, d, J=9.0 Hz), 6.38 (2H, m), 5.63 (1H, dd, J=8.5, 3.5 Hz), 4.93 (1H, d, J=18.5 Hz), 4.78 (1H, d, J=18.5, Hz), 4.27 (2H, m), 3.78 (3H, s), 3.76 (3H, s), 2.42 (3H, s); 13C NMR (CDCl3, 125 MHz): δ 198.12, 169.23, 168.62, 158.01, 136.95, 130.64, 130.38, 128.63, 128.13, 127.77, 127.32, 114.33, 77.49, 73.97, 70.66, 55.49, 53.09, 49.03, 28.24; HRMS (ESI) calcd. for C24H28NO6 (M+H)+ 426.1916, found 426.1909.
EXAMPLE 6
(2R,3S)-Methyl-1-(4-methoxybenzyl)-2-((benzyloxy)methyl)-3-hydroxy-3-methyl-4-methylene-5-oxopyrrolidine-2-carboxylate (9+10)
A mixture of keto amide 8 (8.5 g, 20.0 mmol) and quinuclidine (2.22 g, 20.0 mmol) in DME (10 mL) was stirred for 5 h at 23° C. After completion, the reaction mixture was diluted with ethyl acetate (50 mL) washed with 2N HCl, followed by water and dried over Na2SO4. The solvent was removed in vacuo to give the crude adduct (8.03 g, 94.5%, 3:1 ratio of 9 to 10 dr) as a viscous oil. The diastereomeric mixture was separated at the next step, although small amounts of 9 and 10 were purified by column chromatography (silica gel, ethyl acetate/hexanes, 1:10 to 1:2) for analytical purposes.
Major Diastereomer (9).
[α]23 D−37.8 (c 0.51, CHCl3); FTIR (film) vmax: 3450, 3055, 2990, 1733, 1683, 1507, 1107, 1028, 808,734 cm−1; 1H NMR (CDCl3, 500 MHz): δ 7.29 (5H, m), 7.15 (2H, d, J=7.5 Hz), 6.74 (2H, d, J=8.5 Hz), 6.13 (1H, s), 5.57 (1H, s), 4.81 (1H, d, J=14.5 Hz), 4.45(1H, d, J=15.0 Hz), 4.20 (1H, d, J=12.0 Hz), 4.10 (1H, d, J=12.0 Hz) 3.75 (3H, s), 3.70 (1H, d, J=10.5 Hz), 3.64 (3H, s), 3.54 (1H, d, J=10.5 Hz), 2.55 (1H, bs, OH), 1.50 (3H, s); 13C NMR (CDCl3, 125 MHz): δ 169.67, 168.42, 158.97, 145.96, 137.57, 130.19, 130.12, 128.53, 127.83, 127.44, 116.79, 113.71, 76.32, 76.00, 73.16, 68.29, 55.45, 52.63, 45.36, 22.64; HRMS (ESI) calcd. for C24H28NO6 (M+H)+ 426.1916, found 426.1915.
Minor Diastereomer (10).
[α]23 D−.50.1 (c 0.40, CHCl3); FTIR (film) νmax: 3450, 3055, 2990, 1733, 1683, 1507, 1107, 1028, 808, 734 cm−1; 1H NMR (CDCl3, 500 MHz): δ 7.29 (5H, m), 7.12 (2H, d, J=7.5 Hz), 6.73 (2H, d, J=8.5 Hz), 6.12 (1H, s), 5.57 (1H, s), 4.88 (1H, d, J=15.5 Hz), 4.31 (1H, d, J=15.0 Hz), 4.08 (3H, m), 3.99 (1H, d, J=12.0 Hz) 3.73 (3H, s), 3.62 (3H, s), 3.47 (1H, bs, OH), 3.43 (1H, d, J=10.0 Hz), 1.31 (3H, s); 13C NMR (CDCl3, 125 MHz): δ 169.65, 167.89, 159.13, 147.19, 136.95, 130.29, 129.76, 128.74, 128.19, 127.55, 116.80, 113.82, 76.21, 75.66, 73.27, 68.02, 55.45, 52.52, 45.24, 25.25; HRMS (ESI) calcd. for (M+H)+ C24H28NO6 426.1916, found 426.1915.
EXAMPLE 7
Silylation of 9 and 10 and Purification of 11.
To a solution of lactams 9 and 10 (7.67 g, 18 mmol) in CH2Cl2 (25 ml) was added Et3N (7.54 ml, 54 mmol), and DMAP (2.2 g, 18 mmol) at 0° C., and then bromomethyl-dimethylchlorosilane (5.05 g, 27 mmol) (added dropwise). After stirring the mixture for 30 min at 0° C., it was quenched with aq NaHCO3 and the resulting mixture was extracted with ethyl acetate (3×50 mL). The combined organic layers were washed with water, brine and dried over Na2SO4. The solvent was removed in vacuo to give a mixture of the silated derivatives of 9 and 10 (9.83 g, 95%). The diastereomers were purified by column chromatography (silica gel, ethyl acetate/hexanes, 1:5 to 1:4) to give pure diastereomer 11 (7.4 g, 72%) and its diastereomer (2.4 g, 22%).
Silyl Ether (11).
Rf=0.80 (30% ethyl acetate in hexanes). [α]23 D−58.9 (c 0.55, CHCl3); FTIR (film) νmax; 3050, 2995, 1738, 1697, 1512, 1405, 1243, 1108, 1003, 809, 732 cm−1; 1H NMR (CDCl3, 500 MHz): δ 7.27 (5H, m), 7.05 (2H, d, J=7.0 Hz), 6.71 (2H, d, J=8.5 Hz), 6.18 (1H, s), 5.53 (1H, s), 4.95 (1H, d, J=15.5 Hz), 4.45 (1H, d, J=15.0 Hz), 4.02 (1H, J=12.0 Hz), 3.86 (1H, d, J=11.5 Hz) 3.72 (3H, s), 3.68 (3H, s), 3.65 (1H, d, J=10.5 Hz), 3.30 (1H, d, J=10.0 Hz), 2.34 (2H, d, J=2.0 Hz), 1.58 (3H, s), 0.19 (3H, s), 0.18 (3H, s); 13C NMR (CDCl3, 125 MHz): δ 168.62, 168.12, 158.93, 145.24, 137.53, 130.32, 130.30, 128.49, 127.76,127.22, 117.26, 113.60, 78.55, 78.03, 72.89, 68.45, 55.43, 52.37, 45.74, 21.87, 17.32, −0.72, −0.80; HRMS (ESI) Calcd. for C27H35BrNO6Si (M+H)+ 576.1417, found 576.1407.
EXAMPLE 8
Conversion of (11) to (12).
To a solution of compound 11 (5.67 g 10 mmol) in benzene (250 mL) at 80° C. under nitrogen was added a mixture of tributyltin hydride (4.03 ml, 15 mmol) and AIBN (164 mg, 1 mmol) in 50 ml benzene by syringe pump over 4 h. After the addition was complete, the reaction mixture was stirred for an additional 4 h at 80° C. and the solvent was removed in vacuo. The residue was dissolved in hexanes (20 mL) and washed with saturated NaHCO3 (3×25 mL), water and dried over Na2SO4. The solvent was removed in vacuo to give crude product. The crude product was purified by column chromatography (silica gel, ethyl acetate/hexanes, 1:5) to afford the pure 12 (4.42 g, 89%).
Rf=0.80 (30% ethyl acetate in hexanes). [α]23 D−38.8 (c 0.25, CHCl3); FTIR (film) νmax; 3025, 2985, 1756, 1692, 1513, 1247, 1177, 1059, 667 cm−1; 1H NMR (CDCl3, 500 MHz): δ 7.28 (5H, m), 7.09 (2H, d, J=7.0 Hz), 6.73 (2H, d, J=9.0 Hz), 4.96(1H, d, J=15.0 Hz), 4.35 (1H, d, J=15.5 Hz), 3.97 (1H, d, J=12.5 Hz), 3.86 (1H, d, J=12.0 Hz), 3.80 (1H, d, J=10.0 Hz), 3.72 (3H, s), 3.65 (3H, s), 3.27 (1H, d, J=10.5 Hz), 2.67 (1H, t, J=4.0 Hz), 2.41 (1H, m), 1.79 (1H, m), 1.46 (3H, s), 0.77 (1H, m), 0.46 (1H, m), 0.10 (3H, s), 0.19 (3H, s); 13C NMR (CDCl3, 125 MHz): δ 175.48, 169.46, 158.76, 137.59, 131.04, 129.90, 128.58, 127.88, 127.52, 113.59, 113.60, 81.05, 78.88, 73.12, 69.03, 55.45, 51.94, 48.81, 45.50, 22.79, 17.06, 7.76, 0.54; HRMS (ESI) calcd. for (M+H)+ C27H36NO6Si 498.2312, found 498.2309.
EXAMPLE 9
Debenzylation of (12).
A solution of 12 (3.98 g, 8 mmol) in EtOH (50 ml) at 23° C. was treated with 10% Pd—C (˜1 g) under an argon atmosphere. The reaction mixture was evacuated and flushed with H2 gas (four times) and then stirred vigorously under an atmosphere of H2 (1 atm, H2 balloon) at 23° C. After 12 h, the reaction mixture was filtered through Celite and concentrated in vacuo to give the crude debenzylation product (3.08 g, 95%) which was used for the next step. A small amount crude product was purified by column chromatography (silica gel, ethyl acetate/hexanes, 1:3) for analytical purposes. Rf=0.41 (50% ethyl acetate in hexanes).
mp, 45–47° C.; [α]23 D−30.9 (c 0.55, CHCl3); FTIR (film) νmax: 3432, 3020, 2926, 1735, 1692, 1512, 1244, 1174, 1094, 1024, 870, 795 cm−1; 1H NMR (CDCl3, 400 MHz): δ 7.36 (2H, d, J=8.5 Hz), 6.83 (2H, d, J=8.5 Hz), 5.16 (1H, d, J=15.0 Hz), 4.29 (1H, d, J=15.0 Hz), 3.92 (1H, m), 3.78 (3H, s), 3.68 (3H, s), 3.45 (1H, m), 2.53 (1H, t, J=4.0 Hz), 2.42 (1H, m), 1.82 (1H, m), 1.50 (3H, s), 1.28 (1H, m), 0.75 (1H, m), 0.47 (1H, m), 0.11 (3H, s), 0.02 (3H, s); 13C NMR (CDCl3, 125 MHz): δ 175.82, 169.51, 159.32, 131.00, 129.72, 114.52, 80.79, 80.13, 61.85, 55.48, 51.99, 49.29, 45.06, 23.11, 17.03, 7.44, 0.54; HRMS (ESI) calcd. for C20H30NO6Si (M+H)+ 408.1842, found 408.1846.
EXAMPLE 10
Oxidation to Form Aldehyde (13).
To a solution of the above alcohol from debenzylation of 12 (2.84 g, 7 mmol) in CH2Cl2 (30 mL) was added Dess-Martin reagent (3.57 g, 8.4 mmol) at 23° C. After stirring for 1 h at 23° C., the reaction mixture was quenched with aq NaHCO3—Na2S2O3 (1:1, 50 mL) and extracted with ethyl acetate (3×50 mL). The organic phase was dried and concentrated in vacuo to afford the crude aldehyde. The crude product was purified by column chromatography (silica gel, ethyl acetate/hexanes, 1:5) to give pure aldehyde 13 (2.68 g, 95%). Rf=0.56 (50% ethyl acetate in hexanes).
mp, 54–56° C.; [α]23 D−16.5 (c 0.60, CHCl3); FTIR (film) νmax: 3015, 2925, 1702 1297, 1247, 1170, 1096, 987, 794 cm−1; 1H NMR (CDCl3, 500 MHz): δ 9.62 (1H, s), 7.07 (2H, d, J=8.0 Hz), 6.73 (2H, d, J=8.5 Hz), 4.49 (1H, quart, J=8.5 Hz), 3.70 (3H, s), 3.67 (3H, s), 2.36 (2H, m), 1.75 (1H, m), 1.37 (3H, s), 0.73 (1H, m), 0.48 (1H, m), 0.07 (3H, s), 0.004 (3H, s); 13C NMR (CDCl3, 125 MHz): δ 197.26, 174.70, 167.36, 158.07, 130.49, 128.96, 113.81, 83.97, 82.36, 55.34, 52.43, 47.74, 46.32, 23.83, 16.90, 7.52, 0.56, 0.45; HRMS (ESD calcd. for C20H28NO6Si (M+H)+ 406.1686, found 406.1692.
EXAMPLE 11
Conversion of (13) to (14).
To a solution of freshly prepared cyclohexenyl zinc chloride (10 mL, 0.5 M solution in THF, 5 mmol) (see Example 15 below) at −78° C. under nitrogen was added a −78° C. solution of aldehyde 13 (1.01 g, in 3 ml of THF, 2.5 mmol). After stirring for 5 h at −78° C. reaction mixture was quenched with water (10 mL) then extracted with ethyl acetate (3×10 mL). The combined organic layers were dried over Na2SO4 and solvent was removed in vacuo to give crude product (20:1 dr). The diastereomers were purified by column chromatography (silica gel, ethyl acetate/hexanes, 1:10 to 1:2 affords the pure major diastereomer 14 (1.0 g, 83%) and a minor diastereomer (50 mg 5%). For 14: Rf=0.56 (50% ethyl acetate in hexanes).
mp, 79–81° C.; [a]23 D−28.5 (c 1.45, CHCl3); FTIR (film) νmax: 3267, 2927, 2894, 2829, 1742, 1667, 1509, 1248, 1164, 1024, 795 cm−1; 1H NMR (CDCl3, 500 MHz): δ 7.34 (2H, d, J=8.5 Hz), 6.81 (2H, d, J=9.0 Hz), 5.84 (1H, m), 5.73 (1H, m), 4.88 (1H, d, J=15.5 Hz), 4.39 (1H, d, J=14.5 Hz), 4.11 (1H, t, J=6.5 Hz), 3.77 (3H, s), 3.58 (3H, s), 3.00 (1H, m), 2.95 (1H, d, J=9.0 Hz), 2.83 (1H, t, J=3.5 Hz), 3.36 (1H, m), 2.27 (1H, m), 1.98 (2H, m), 1.74 (3H, m), 1.62 (3H, s), 1.14 (2H, m), 0.59 (1H, m), 0.39 (11H, m), 0.13 (3H, s), 0.03 (3H, s); 13C NMR (CDCl3, 125 MHz): δ 176.80, 170.03, 158.27, 131.86, 131.34, 128.50, 126.15, 113.40, 83.96, 82.45, 77.17, 55.45, 51.46, 48.34, 48.29, 39.08, 28.34, 25.29, 22.45, 21.09, 17.30, 7.75, 0.39, 0.28; HRMS (ESI) calcd. for C26H38NO6Si (M+H)+ 488.2468, found 488.2477.
EXAMPLE 12
Tamao-Fleming Oxidation of (14) to (15).
To a solution of 14 (0.974 g, 2 mmol) in THF (5 mL) and MeOH (5 mL) at 23° C. was added KHCO3 (0.8 g, 8 mmol) and KF (0.348 g, 6 mmol). Hydrogen peroxide (30% in water, 5 mL) was then introduced to this mixture. The reaction mixture was vigorously stirred at 23° C. and additional hydrogen peroxide (2 ml) was added after 12 h. After 18 h, the reaction mixture was quenched carefully with NaHSO3 solution (15 mL). The mixture was extracted with ethyl acetate (3×25 mL) and the combined organic layers were washed with water and dried over Na2SO4. The solvent was removed in vacuo to give the crude product. The crude product was purified by column chromatography (silica gel, ethyl acetate) to give the pure triol 15 (0.82 g, 92%).
Rf=0.15 (in ethyl acetate). mp, 83–84° C.; [α]23 D: +5.2 (c 0.60, CHCl3); FTIR (film) νmax; 3317, 2920, 2827, 1741, 1654, 1502, 1246, 1170, 1018, 802 cm−1; 1HNMR(CDCl3, 500 MHz): δ 7.77 (2H, d, J=8.0 Hz), 6.28 (2H, d, J=8.0 Hz), 5. 76 (1H, m), 5.63 (1H, d, J=10.0 Hz), 4.74 (1H, d, J=15.5 Hz), 4.54 (1H, d, J=15.0 Hz), 4.12 (1H, d, J=2.5 Hz), 3.80 (1H, m), 3.76 (3H, s), 3.72 (1H, m), 3.68 (3H, s), 3.00 (1H, m), 2.60 (1H, br), 2.20 (1H, m), 1.98 (2H, s), 1.87 (1H, m), 1.80 (1H, m), 1.71 (2H, m), 1.61 (3H, s), 1.14 (2H, m); 13C NMR (CDCl3, 125 MHz): δ 178.99, 170.12, 158.27, 131.30, 130.55, 128.13, 126.39, 113.74, 81.93, 80.75, 76.87, 61.61, 55.45, 51.97, 51.32, 48.07, 39.17, 27.71, 27.13, 25.22, 21.35, 21.22; HRMS (ESI) calcd. for C24H34NO7 (M+H)+ 448.2335, found 448.2334.
EXAMPLE 13
Deprotection of (15) to (16).
To a solution of 15 (0.670 g, 1.5 mmol) in acetonitrile (8 mL) at 0° C. was added a pre-cooled solution of ceric ammonium nitrate (CAN) (2.46 g 4.5 mmol in 2 mL H2O). After stirring for 1 h at 0° C. the reaction mixture was diluted with ethyl acetate (50 mL), washed with saturated NaCl solution (5 mL) and organic layers was dried over Na2SO4. The solvent was removed in vacuo to give the crude product which was purified by column chromatography (silica gel, ethyl acetate) to give the pure 16 (0.4 g, 83%).
Rf=0.10 (5% MeOH in ethyl acetate). mp, 138 to 140° C.; [α]23 D+14.5 (c 1.05, CHCl3); FTIR (film) νmax 3301, 2949, 2911, 2850, 1723, 1673, 1437, 1371, 1239, 1156, 1008, 689 cm−1; 1H NMR (CDCl3, 600 MHz): δ 8.48 (1H, br), 6.08 (1H, m), 5. 75 (1H, d, J=9.6 Hz), 5.29 (1H, br), 4.13 (1H, d, J=6.6 Hz), 3.83 (3H, m), 3.79 (1H, m), 3.72 (1H, m), 2.84 (1H, d, J=10.2 Hz), 2.20 (1H, m), 2.16 (1H, br), 1.98 (3H, m), 1.77 (3H, m), 1.59 (1H, m), 1.54 (3H, s), 1.25 (1H, m). 13C NMR (CDCl3, 125 MHz): δ 180.84, 172.95, 135.27, 123.75, 82.00, 80.11, 75.56, 62.39, 53.14, 51.78, 38.95, 28.79, 26.48, 25.04, 20.66, 19.99; HRMS (ESI) calcd. (M+H)+ for C16H26NO6 328.1760, found 328.1752.
EXAMPLE 14
Conversion of (16) to Salinosporamide A(1).
A solution of triol ester 16 (0.164 g, 0.5 mmol) in 3 N aq LiOH (3 mL) and THF (1 mL) was stirred at 5° C. for 4 days until hydrolysis was complete. The acid reaction mixture was acidified with phosphoric acid (to pH 3.5). The solvent was removed in vacuo and the residue was extracted with EtOAc, separated, and concentrated in vacuo to give the crude trihydroxy carboxylic acid 16a (not shown). The crude acid was suspended in dry CH2Cl2 (2 mL), treated with pyridine (0.5 mL) and stirred vigorously at 23° C. for 5 min. To this solution was added BOPCl (152 mg, 0.6 mmol) at 23° C. under argon, and stirring was continued for 1 h. The solvent was removed under high vacuum and the residue was suspended in dry CH3CN (1 mL) and treated with pyridine (1 mL). To this solution was added PPh3Cl2 (333 mg, 1.0 mmol) at 23° C. under argon with stirring. After 1 h the solvent was removed in vacuo. The crude product was purified by column chromatography (silica gel, ethyl acetate-CH2Cl2, 1:5) to give the pure β-lactone 1 (100 mg, 64%) as a colorless solid.
Rf=0.55 (50% ethyl acetate in hexane). mp, 168–170° C. (authentic sample: 168–170° C., 169–171° C. in Angew. Chem. Int. Ed., 2003, 42, 355–357); mixture mp, 168–170C. [α]23 D −73.2 (c 0.49, MeOH), −72.9 (c 0.55, MeOH, in Angew. Chem. Int. Ed., 2003, 42, 355–357); FTIR (film) νmax: 3406, 2955, 2920, 2844, 1823, 1701, 1257, 1076, 1012, 785, 691 cm−1; 1H NMR (CDCl3, 500 MHz): δ 10.62 (1H, br), 6.42 (1H, d, J=10.5 Hz), 5.88 (1H, m), 4.25 (1H, d, J=9.0 Hz), 4.14 (1H, m), 4.01 (1H, m), 3.17 (1H, t, J=7.0 Hz), 2.85 (1H, m), 2.48 (1H, m), 2.32 (2H, m), 2.07 (3H, s), 1.91 (2H, m), 1.66 (2H, m), 1.38 (1H, m);13C NMR (CDCl3, 125 MHz): δ 176.92, 169.43, 129.08, 128.69, 86.32, 80.35, 70.98, 46.18, 43.28, 39.31, 29.01, 26.47, 25.35, 21.73, 20.00; HRMS (ESI) calcd. for (M−H)− C15H19ClNO4 312.1003, found 312.1003.
| 544814 | Oct 1, 2007 | Jun 9, 2009 | Nereus Pharmaceuticals, Inc. | [3.2.0] Heterocyclic compounds and methods of using the same |
| US7579371 | Jun 15, 2006 | Aug 25, 2009 | Nereus Pharmaceuticals, Inc. | Methods of using [3.2.0] heterocyclic compounds and analogs thereof |
| US7824698 | Feb 4, 2008 | Nov 2, 2010 | Nereus Pharmaceuticals, Inc. | Lyophilized formulations of Salinosporamide A |
| US7842814 | Apr 6, 2007 | Nov 30, 2010 | Nereus Pharmaceuticals, Inc. | Total synthesis of salinosporamide A and analogs thereof |
| US7910616 | May 12, 2009 | Mar 22, 2011 | Nereus Pharmaceuticals, Inc. | Proteasome inhibitors |
| US8003802 | Mar 6, 2009 | Aug 23, 2011 | Nereus Pharmaceuticals, Inc. | Total synthesis of Salinosporamide A and analogs thereof |
| US8067616 | Oct 27, 2010 | Nov 29, 2011 | Nereus Pharmaceuticals, Inc. | Total synthesis of salinosporamide A and analogs thereof |
| US8168803 | Jun 10, 2008 | May 1, 2012 | Nereus Pharmaceuticals, Inc. | Methods of using [3.2.0] heterocyclic compounds and analogs thereof |
| US8217072 | Jun 18, 2004 | Jul 10, 2012 | The Regents Of The University Of California | Salinosporamides and methods for use thereof |
| US8222289 | Dec 15, 2009 | Jul 17, 2012 | The Regents Of The University Of California | Salinosporamides and methods for use thereof |
| US8227503 | Mar 21, 2011 | Jul 24, 2012 | Nereus Pharmaceuticals, Inc. | Proteasome inhibitors |
| US8314251 | Jul 15, 2011 | Nov 20, 2012 | Nereus Pharmaceuticals, Inc. | Total synthesis of salinosporamide A and analogs thereof |
| US8389564 | May 14, 2012 | Mar 5, 2013 | Venkat Rami Reddy Macherla | Proteasome inhibitors |
| US8394816 | Dec 5, 2008 | Mar 12, 2013 | Irene Ghobrial | Methods of using [3.2.0] heterocyclic compounds and analogs thereof in treating Waldenstrom’s Macroglobulinemia |
| Name: | Marizomib | |
| Synonyms: | 6-Oxa-2-azabicyclo[3.2.0]heptane-3,7-dione, 4-(2-chloroethyl)-1-[(S)-(1S)-2-cyclohexen-1-ylhydroxymethyl]-5-methyl-, (1R,4R,5S)-; Other Names: (-)-Salinosporamide A; ML 858; Marizomib; NPI 0052; Salinosporamide A | |
| CAS Registry Number: | 437742-34-2 | |
| Molecular Formula: | C15H20ClNO4 | |
| Molecular Weight: | 313.1 | |
| Molecular Structure: |  |
Orphan Drug Designation Granted for Epidiolex in Dravet syndrome by the FDA

Cannabidiol Seven Expanded Access INDs granted by FDA to U.S. physicians to treat with Epidiolex 125 children suffering from intractable epilepsy syndromes -
LONDON, Nov. 15, 2013
GW Pharmaceuticals plc (AIM: GWP, Nasdaq: GWPH, “GW”) announced today that the U.S. Food and Drug Administration (FDA) has granted orphan drug designation for Epidiolex(R), our product candidate that contains plant-derived Cannabidiol (CBD) as its active ingredient, for use in treating children with Dravet syndrome, a rare and severe form of infantile-onset, genetic, drug-resistant epilepsy syndrome. Epidiolex is an oral liquid formulation of a highly purified extract of CBD, a non-psychoactive molecule from the cannabis plant. Following receipt of this orphan designation, GW anticipates holding a pre-IND meeting with the FDA in the near future to discuss a development plan for Epidiolex in Dravet syndrome.
Dravet syndrome is a rare pediatric epilepsy syndrome with a distinctive but complex electroclinical presentation. Onset of Dravet syndrome occurs during the first year of life with clonic and tonic-clonic seizures in previously healthy and developmentally normal infants. Prognosis is poor and patients typically develop intellectual disability and life-long ongoing seizures. There are approximately 5,440 patients with Dravet in the United States and an estimated 6,710 Dravet patients in Europe. These figures may be an underestimate as this syndrome is reportedly underdiagnosed.
In addition to GW’s clinical development program for Epidiolex in Dravet syndrome, which is expected to commence in 2014, GW has also made arrangements to enable independent U.S. pediatric epilepsy specialists to treat high need pediatric epilepsy cases with Epidiolex immediately. To date in 2013, a total of seven “expanded access” INDs have been granted by the FDA to U.S. clinicians to allow treatment with Epidiolex of approximately 125 children with epilepsy. These children suffer from Dravet syndrome, Lennox-Gastaut syndrome, and other pediatric epilepsy syndromes. GW is aware of further interest from additional U.S. and ex-U.S. physicians to host similar INDs for Epidiolex. GW expects data generated under these INDs to provide useful observational data during 2014 on the effect of Epidiolex in the treatment of a range of pediatric epilepsy syndromes.
“I, together with many colleagues in the U.S. who specialize in the treatment of childhood epilepsy, very much welcome the opportunity to investigate Epidiolex in the treatment of Dravet syndrome. The FDA’s timely approval of the orphan drug designation for Epidiolex in Dravet syndrome is a key milestone that comes after many years of reported clinical cases that suggest encouraging evidence of efficacy for CBD in this intractable condition,” stated Dr. Orrin Devinsky, Professor of Neurology, Neurosurgery and Psychiatry in New York City. “With GW now making plans to advance Epidiolex through an FDA development program, we have the prospect for the first time of fully understanding the science of CBD in epilepsy with a view to making an appropriately tested and approved prescription medicine available in the future for children who suffer from this debilitating disease.”
“GW is proud to be at the forefront of this important new program to treat children with Dravet Syndrome and potentially other forms of intractable childhood epilepsy. For families in these circumstances, their lives are significantly impacted by constant and often times very severe seizures in children where all options to control these seizures have been exhausted,” stated Dr. Stephen Wright, GW’s R&D Director. “GW intends to advance a full clinical development program for Epidiolex in Dravet syndrome as quickly as possible, whilst at the same time helping families in the short term through supporting physician-led INDs to treat intractable cases. Through its efforts, GW aims to provide the necessary evidence to confirm the promise of CBD in epilepsy and ultimately enabling children to have access to an FDA-approved prescription CBD medicine.”
“This orphan program for Epidiolex in childhood epilepsy is an important corporate strategic priority for GW. Following receipt of today’s orphan designation, GW now intends to commence discussions with the FDA regarding the U.S. regulatory pathway for Epidiolex,” stated Justin Gover, GW’s Chief Executive Officer. “GW intends to pursue this development in-house and retains full commercial rights to Epidiolex.”
About Orphan Drug Designation
Under the Orphan Drug Act, the FDA may grant orphan drug designation to drugs intended to treat a rare disease or condition — generally a disease or condition that affects fewer than 200,000 individuals in the U.S. The first NDA applicant to receive FDA approval for a particular active ingredient to treat a particular disease with FDA orphan drug designation is entitled to a seven-year exclusive marketing period in the U.S. for that product, for that indication.
About GW Pharmaceuticals plc
Founded in 1998, GW is a biopharmaceutical company focused on discovering, developing and commercializing novel therapeutics from its proprietary cannabinoid product platform in a broad range of disease areas. GW commercialized the world’s first plant-derived cannabinoid prescription drug, Sativex(R), which is approved for the treatment of spasticity due to multiple sclerosis in 22 countries. Sativex is also in Phase 3 clinical development as a potential treatment of pain in people with advanced cancer. This Phase 3 program is intended to support the submission of a New Drug Application for Sativex in cancer pain with the U.S. Food and Drug Administration and in other markets around the world. GW has established a world leading position in the development of plant-derived cannabinoid therapeutics and has a deep pipeline of additional clinical-stage cannabinoid product candidates targeting epilepsy (including an orphan pediatric epilepsy program), Type 2 diabetes, ulcerative colitis, glioma and schizophrenia. For further information, please visit http://www.gwpharm.com.
Cannabidiol (CBD) is one of at least 85 cannabinoids found in cannabis.It is a major constituent of the plant, second to tetrahydrocannabinol (THC), and represents up to 40% in its extracts. Compared with THC, cannabidiol is not psychoactive in healthy individuals, and is considered to have a wider scope of medical applications than THC, including to epilepsy, multiple sclerosis spasms, anxiety disorders, bipolar disorder,schizophrenia,nausea, convulsion and inflammation, as well as inhibiting cancer cell growth. There is some preclinical evidence from studies in animals that suggests CBD may modestly reduce the clearance of THC from the body by interfering with its metabolism.Cannabidiol has displayed sedative effects in animal tests. Other research indicates that CBD increases alertness. CBD has been shown to reduce growth of aggressive human breast cancer cells in vitro, and to reduce their invasiveness.

Mast Ischemia Drug Gets Orphan Drug Designation

MST-188 (purified poloxamer 188)
MST-188 is a purified form of a nonionic, triblock copolymer (poloxamer 188). It is an investigational agent that binds to hydrophobic surfaces on damaged cells and improves membrane hydration and lowers adhesion and viscosity, particularly under low shear conditions. MST-188 has the potential to reduce ischemic tissue injury and end-organ damage by restoring microvascular function, which is compromised in a wide range of serious and life-threatening diseases and conditions. We initially are developing MST-188 as a treatment for complications arising from sickle cell disease.
How MST-188 Works…

Background
Non-purified forms of poloxamer 188 (P188) have been used in foods, drugs and cosmetics since the 1950s. In the 1980s, extensive research on the mechanisms and potential clinical applications of P188 was conducted. Research has demonstrated that P188 binds to hydrophobic surfaces that develop when cells are damaged and restores normal hydrated surfaces, while having little or no activity in normal, healthy tissues. Research also has demonstrated that P188 prevents adhesion and aggregation of soluble fibrin and formed elements in the blood and maintains the deformability of red blood cells, the non-adhesiveness of unactivated platelets and granulocytes and the normal viscosity of blood. In addition, it is believed that P188 is not metabolized, but is excreted unchanged in the urine with a half-life of approximately four to six hours.
Formulations of P188 (non-purified and purified) have been studied in clinical trials involving nearly 4,000 individuals. It has been evaluated in the clinic to treat acute myocardial infarction, sickle cell disease and malaria, including a 2,950-patient, randomized, controlled study of P188 (non-purified) in acute myocardial infarction. The effectiveness of P188 also has been investigated in nonclinical studies of stroke, hemorrhagic shock, bypass surgery, adult respiratory distress syndrome, neurologic protection in deep hypothermic circulatory arrest, vasospasm, spinal cord injury, angioplasty, frostbite, amniotic fluid embolism, acute ischemic bowel disease and burns.
MST-188
Our(mast) purified form of P188, or purified P188, which is the active ingredient in MST-188, was designed to eliminate certain low molecular weight substances present in P188 (non-purified), which we believe were primarily responsible for the moderate to moderately severe elevations in serum creatinine levels (acute renal dysfunction) observed in prior clinical studies of P188 (non-purified). Purified P188 has been evaluated in multiple clinical studies by a prior sponsor, including a 255-patient, phase 3 study. In that study, purified P188 was generally well tolerated and there were no clinically significant elevations in serum creatinine among subjects who received purified P188 compared to placebo.
We believe that, as a rheologic, antithrombotic and cytoprotective agent, MST-188 has potential application in treating a wide range of diseases and conditions resulting from microvascular-flow abnormalities.
Sickle Cell Disease Market & Opportunity
More than $1.0 billion is spent annually in the U.S. to treat patients with sickle cell disease. Sickle cell disease is a genetic disorder characterized by the “sickling” of red blood cells, which normally are disc-shaped, deformable and move easily through the microvasculature carrying oxygen from the lungs to the rest of the body. Sickled, or crescent-shaped, red blood cells, on the other hand, are rigid and sticky and tend to adhere to each other and the vascular endothelium. Patients with sickle cell disease are known to experience severely painful episodes associated with the obstruction of small blood vessels by sickle-shaped red blood cells. These painful episodes are commonly known as acute crisis or vaso-occlusive crisis. Reduced blood flow to organs and bone marrow during vaso-occlusive crisis not only causes intense pain, but can result in tissue death, or necrosis. The frequency, severity and duration of these acute crises can vary considerably.
We (mast) estimate that, in the U.S., sickle cell disease results in over 95,000 hospitalizations and, in addition, approximately 69,000 emergency department treat-and-release encounters each year. When a patient with sickle cell disease makes an institutional visit, vaso-occlusive crisis is the primary diagnosis in approximately 77% of hospital admissions and 64% of emergency room treat-and-release encounters. In addition, although the number is difficult to measure, we estimate that the number of untreated sickle cell crisis events is substantial and in the hundreds of thousands in the U.S. each year. We believe that, if MST-188 is approved, as people with sickle cell disease are made aware of the new therapy, more people who suffer from acute crisis will seek treatment.
Development Status
We (mast) have initiated a Phase 3 clinical study of MST-188 for the treatment of sickle cell disease. The primary objective will be to demonstrate that MST-188 reduces the duration of vaso-occlusive crisis in patients with sickle cell disease. Please see our Clinical Trials page for more information regarding our phase 3 study of MST-188. In addition to the phase 3 study, we plan to conduct a number of smaller-scale clinical studies to further assess the efficacy, safety and tolerability of MST-188, and expect these studies to overlap with the phase 3 study.
Isavuconazole – Basilea reports positive results from study

This post is updated in sept 2015……..
Isavuconazole (BAL4815; trade name Cresemba) is a triazole antifungal drug. Its prodrug, isavuconazonium sulfate (BAL8557), was granted approval by the U.S. Food and Drug Administration (FDA) on March 6, 2015[1]
During its Phase III drug trials, Astellas partnered with Basilea Pharmaceutica, the developer of the drug, for rights to co-development and marketing of isavuconazole. [2]
On May 28, 2013, Basilea Pharmaceutica announced it had been granted orphan drug status by the FDA for treatment of aspergillosis.[3] Since then, it has also been granted orphan drug status for the treatment of invasive candidiasis.[4]
CLINICAL TRIALS…LINK
PATENTS
|
6-27-2012
|
Process for the manufacture of enantiomerically pure antifungal azoles as ravuconazole and isavuconazole
|
|
|
11-18-2011
|
Antifungal Composition
|
|
|
9-29-2010
|
PROCESS FOR PREPARATION OF WATER-SOLUBLE AZOLE PRODRUGS
|
|
|
12-3-2008
|
N-substituted carbamoyloxyalkyl-azolium derivatives
|
|
|
3-14-2007
|
N-phenyl substituted carbamoyloxyalkyl-azolium derivatives
|
|
|
11-3-2004
|
N-substituted carbamoyloxyalkyl-azolium derivatives
|
|
|
10-10-2001
|
Azoles for treatment of fungal infections
|
Several azoles are currently used for systemic mycoses. However, none of them fulfills the needs of clinical requirement in full extent, particularly with regard 0 to broad antifungal spectrum including aspergillus fumigatus, less drug-drug interaction, and appropriate plasma half-life for once a day treatment. Other clinical requirements which are not fulfilled by the azoles currently used, are efficacy against major systemic mycoses including disseminated aspergillosis, safety, and oral or parenteral formulations. Particularly, demand of a 5 parenteral administration of the azoles is increasing for the treatment of serious systemic mycoses. Most of the azoles on the market as well as under development are highly lipophilic molecules that make the parenteral formulation difficult.

Isavuconazole [(2R,3R)-3-[4-(4-cyanophenyl)thiazol-2-yl)]-1-(1H-1,2,4-triazol-1-yl)-2-(2,5-difluorophenyl)-butan-2-ol; formula I, R1 and R3 represent fluorine and R2 represents hydrogen] as well as Ravuconazole [(2R,3R)-3-[4-(4-cyanophenyl)thiazol-2-yl)]-1-(1H-1,2,4-triazol-1-yl)-2-(2,4-difluorophenyl)-butan-2-ol; formula I, R1 and R2 represent fluorine and R3 represents hydrogen] are useful antifungal drugs as reported in U.S. Pat. No. 5,648,372 from Feb. 1, 1995 or in U.S. Pat. No. 5,792,781 from Sep. 18, 1996 or in U.S. Pat. No. 6,300,353 from Oct. 9, 2001 (WO99/45008).
Since compounds of general formula I contain two adjacent chiral centers, synthesis of enantiomerically pure compound is complex and until now, all patented syntheses are not efficient enough and do not allow cost effective manufacturing on a technical scale:
Thus, U.S. Pat. Nos. 5,648,372 or 5,792,781 describe enantioselective synthesis of compounds of formula I (specifically Ravuconazole) from chiral 3-hydroxy-2-methyl propionic acid in 12 steps with overall yield lower than 5%. In another approach including 13 steps and low overall yield, (R)-lactic acid was used as the starting material (Chem. Pharm. Bull. 46(4), 623 (1998) and ibid. 46(7), 1125 (1998)).
Because both starting materials contain only one chiral center, in a number of inefficient steps, the second, adjacent chiral center has to be created by a diastereoselective reaction (using either Corey or Sharpless epoxidation method) which is not sufficiently selective leading mostly to a mixture of two diastereomers which have to be separated.
The second approach, based on (R)-methyl lactate, was recently very thoroughly optimized by BMS on a multi kilogram scale but it still does not fulfill requirements for cost effective manufacturing process (Organic Process Research & Development 13, 716 (2009)). The overall yield of this optimized 11 steps process is still only 16% (Scheme 1).
The manufacturing process for Isavuconazole is similar: Since Isavuconazole differentiates from Ravuconazole by only another fluorine substitution on the aromatic ring (2,5- instead of 2,4-difluorophenyl), the identical synthesis has been used (U.S. Pat. No. 6,300,353 from Oct. 9, 2001 and Bioorg. & Med. Chem. Lett. 13, 191 (2003)). Consequently, also this manufacturing process, based on (R)-lactic acid, faces the same problems: to many steps, extremely low overall yield and in addition to U.S. Pat. No. 6,300,353 claims even already known step as novel (claim 36).
Recent attempts to improve this concept as reported in WO 2007/062542 (Dec. 1, 2005), using less expensive, natural configured (S)-lactic acid, also failed: As already reported in U.S. Pat. No. 6,133,485 and in US 2003/0236419, the second chiral center was formed from an optically active allyl alcohol prepared in a few steps from (S)-lactic acid.
This allyl alcohol was subjected to Sharpless diastereoselective epoxidation providing first an opposite configured, epimeric epoxy alcohol which had to be then epimerized in an additional inversion step yielding finally the desired epoxy alcohol as the known precursor for Isavuconazole (U.S. Pat. No. 6,300,353). It is obvious that this process using less expensive (S)-lactic acid makes the entire process with an inversion step even more complex than the original approach.
Elegant and more efficient process has been claimed in US 2004/0176432 from Jun. 26, 2001) in which both chiral centers have been formed simultaneously, diastereo- and enantio-selectively pure in one single reaction step using chiral (R)-2-butynol as a chiral precursor in the presence of Pd(II)-catalyst and diethyl zinc (Scheme 2).
Since water soluble, (R)-2-butynol is expensive, recently identical process has been published, in which instead of (R)-2-butynol less water soluble and therefore, less expensive (R)-4-phenyl-3-butyn-2-ol was used (Synthetic Commun. 39, 1611 (2009)). Nevertheless, as incorrectly stated there, this process does not provide better diastereoselectivity than the original process using (R)-2-butynol: On the contrary disadvantage of this process is a very bad atom economy because huge phenyl group of (R)-4-phenyl-3-butyn-2-ol has to be “disposed” in oxidation step by the conversion of triple bond into carboxylic acid function.
All known processes for enantiomerically pure compounds of formula I have definitely too many operation steps and specifically very low overall yield. The chiral starting materials used, either 3-hydroxy-2-methyl propionic acid or (S)- or (R)-methyl lactate, contain only one chiral center and consequently, in number of steps, the second adjacent chiral center has to be ineffectively generated which makes the entire process long and expensive. The only known process, which generates both chiral centers simultaneously, requires again expensive chiral starting material (R)-2-butynol.
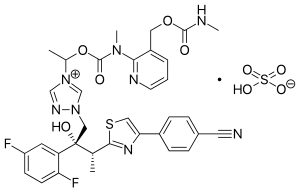
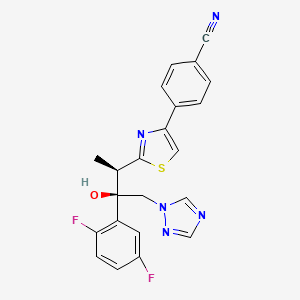
ISAVUCONAZOLE
…………………………………………….
synthetic scheme A, starting from 4-[(2R)-2-(3,4,5,6-tetrahydro-2H-pyran-2-yloxy)-propionyl]morpholine [which can be prepared by a same procedure as described in Chem. Pharm. Bull. 41, 1035, 1993.]. This synthesis route has been described for example in European Patent Application No. 99101360.8.
(a)
………………………………………………………………………
Example 1 (2R,3R)-3-[4-(4-cyanophenyl)thiazol-2-yl]-1-(1H-1,2,4-triazol-1-yl)-2-(2,4-difluorophenyl)-butan-2-ol
To a solution of racemic 3-[4-(4-cyanophenyl)thiazol-2-yl]-1-(1H-1,2,4-triazol-1-yl)-2-(2,4-difluorophenyl)-butan-2-ol (43.7 g) in acetone (800 ml) a solution of (1R)-10-camphorsulfonic acid (23 g) in methanol (300 ml) was added and the mixture was heated under reflux until a clear solution was obtained. The solution was slowly cooled to rt, seeded with crystals of the title enantiomeric salt and let overnight. The solid was collected by filtration, washed with acetone and dried to provide (2R,3R)-3-[4-(4-cyanophenyl)thiazol-2-yl]-1-(1H-1,2,4-triazol-1-yl)-2-(2,4-difluorophenyl)-butan-2-ol (1R)-10-camphorsulfonate as white solid. This crude salt was then taken up in methylenechloride (100 ml) and water (ca. 100 ml) and the mixture was basified with aqueous sodium hydroxide solution. The organic layer was separated and the aqueous phase washed twice with methylenechloride (50 ml) and combined. The organic phases were then washed twice with water (2×50 ml), dried with sodium sulfate, filtrated and the solvent removed under reduced pressure. The crude product was then mixed with isopropanol (ca. 150 ml), heated for 10 min, cooled to 0° C. and stirred for ca. 2 hrs. The product was collected, washed with isopropanol and dried under reduced pressure to provide the enantiomerically pure title compound (17.5 g, 41% yield, 99.1% ee);
m.p. 164-166° C.; [α]=−30° (c=1, methanol, 25° C.);
NMR (CDCl3): 1.23 (3H, d, J=8 Hz), 4.09 (1H, q, J=8 Hz), 4.26 (1H, d, J=14 Hz), 4.92 (1H, d, J=14 Hz), 5.75 (1H, s), 6.75-6.85 (2H, m), 7.45-7.54 (2H, m), 7.62 (1H, s), 7.69 (1H, s), 7.75 (1H, d, J=8 Hz), 7.86 (1H, s), 8.03 (1H, d, J=8 Hz).
The analytical data were identical with published (U.S. Pat. No. 5,648,372 and Chem. Pharm. Bull. 1998, 46, 623-630).
Example 2 (2R,3R)-3-[4-(4-cyanophenyl)thiazol-2-yl]-1-(1H-1,2,4-triazol-1-yl)-2-(2,4-difluorophenyl)-butan-2-ol
Racemic 3-[4-(4-cyanophenyl)thiazol-2-yl]-1-(1H-1,2,4-triazol-1-yl)-2-(2,4-difluorophenyl)-butan-2-ol (44 g) and (1R)-10-camphorsulfonic acid (20 g) were suspended in methanol (ca. 300 ml), the slurry was stirred intensively, warmed up to ca. 70° C. and a small addition of acetic acid was added to obtain a clear solution. After cooling of the solution to rt and then to 0° C., the mixture was seeded with enantiomerically pure salt and stirred for another 2 hrs. The crystalline solid was collected by filtration, washed with cooled methanol and dried under reduced pressure. The crystals were partitioned between methylenechloride (300 ml) and saturated aqueous sodium bicarbonate solution (200 ml). The organic layer was washed twice with water (50 ml), dried with magnesium sulphate, filtrated and evaporated under reduced pressure to give the title compound (16.9 g, 38% yield, 95% ee). The analytical data were identical with published (U.S. Pat. No. 5,648,372 or Chem. Pharm. Bull. 1998, 46, 623).
Example 3 (2R,3R)-3-[4-(4-cyanophenyl)thiazol-2-yl]-1-(1H-1,2,4-triazol-1-yl)-2-(2,5-difluorophenyl)-butan-2-ol
To a solution of racemic 3-[4-(4-cyanophenyl)thiazol-2-yl]-1-(1H-1,2,4-triazol-1-yl)-2-(2,5-difluorophenyl)-butan-2-ol (10 g) in acetone (ca. 200 ml) a solution of (1R)-10-camphorsulfonic acid (3.9 g) in methanol (50 ml) was added and the mixture was heated shortly under reflux until a clear solution was obtained. The solution was then slowly cooled to rt, seeded with crystals of the desired enantiomeric salt and let overnight. The solid precipitate was collected by filtration, washed with acetone and dried to provide (2R,3R)-3-[4-(4-cyanophenyl)thiazol-2-yl]-1-(1H-1,2,4-triazol-1-yl)-2-(2,5-difluorophenyl)-butan-2-ol (1R)-10-camphorsulfonate as white solid. This salt was then taken up in methylenechloride and water and basified with aqueous sodium bicarbonate solution. The organic layer was separated and the aqueous phase washed twice with methylenechloride. The organic phases were combined, dried with sodium sulphate, filtrated and the solvent removed under reduced pressure. The crude product was then dissolved in ethanol, the slurry heated for 20 min, small amount of water was added, the solution slowly cooled to 0° C. and stirred for ca. 2 hrs. The product was collected, washed with cold ethanol and dried under reduced pressure to provide the title enantiomerically pure compound (3.9 g, 39% yield, 96% ee). The analytical date were identical with published in U.S. Pat. No. 6,300,353 B1 and WO 99/45008.
Example 4 (2R,3R)-3-[4-(4-cyanophenyl)thiazol-2-yl]-1-(1H-1,2,4-triazol-1-yl)-2-(2,5-difluorophenyl)-butan-2-ol
To a solution of racemic 3-[4-(4-cyanophenyl)thiazol-2-yl]-1-(1H-1,2,4-triazol-1-yl)-2-(2,5-difluorophenyl)-butan-2-ol (100 g) in acetone (1000 ml) a solution of (1R)-10-camphorsulfonic acid (47 g) in methanol (500 ml) was added at rt, then slurry was heated under stirring to almost reflux for ca. 30 min, then cooled slowly to rt, seeded with the pure enantiomeric salt and stirred over night. The solid was collected by filtration, washed with methanol/acetone mixture, dried under reduced pressure. The residue was taken up with a solvent mixture of methylenechloride/water and after addition of saturated aqueous sodium bicarbonate solution the organic phase was separated and aqueous phase washed twice with methylenechloride. The combined organic phases were filtrated, the solvent removed under reduced pressure. Recrystallization of the crude product from aqueous ethanol provided enantiomerically pure title compound: 39 g (39% yield, 92% ee). The analytical data were identical with published: U.S. Pat. No. 6,300,353 and WO 99/45008.
Example 5 (2R,3R)-3-[4-(4-cyanophenyl)thiazol-2-yl]-1-(1H-1,2,4-triazol-1-yl)-2-(2,5-difluorophenyl)-butan-2-ol
A solution of the racemic 3-[4-(4-cyanophenyl)thiazol-2-yl]-1-(1H-1,2,4-triazol-1-yl)-2-(2,5-difluorophenyl)-butan-2-ol (4.4 g) and (1R)-10-camphorsulfonic acid (2 g) in toluene (40 ml) containing glacial acetic acid (0.6 ml) was warmed up to approximately 70° C., then allowed to cool slowly to 20° C., seeded with the pure enantiomeric salt whereupon the pure enantiomeric salt start to crystallize out. After ca. 2 hrs at this temperature the solid was collected, washed with cold toluene and dried. The crystals were taken with a solvent mixture of methylenechloride/water and after addition of aqueous saturated sodium bicarbonate solution the organic phase was separated and aqueous phase washed twice with methylenechloride. The combined organic phases were filtrated and the solvent removed under reduced pressure. Recrystallization of the crude product from aqueous ethanol provided enantiomerically pure title compound: 2 g (45% yield, 99% ee). The analytical data were identical with published: U.S. Pat. No. 6,300,353 and WO 99/45008.
…………………………………..
WO 1999045008
The following synthetic scheme 1 illustrates the manufacture of one of the compounds of formula I′:


……………………………….
Bioorganic and medicinal chemistry letters, 2003 , vol. 13, 2 p. 191 – 196
http://www.sciencedirect.com/science/article/pii/S0960894X02008922
A highly potent water soluble triazole antifungal prodrug, RO0098557 (1), has been identified from its parent, the novel antifungal agent RO0094815 (2). The prodrug includes a triazolium salt linked to an aminocarboxyl moiety, which undergoes enzymatic activation followed by spontaneous chemical degradation to release 2. Prodrug 1 showed high chemical stability and water solubility and exhibited strong antifungal activity against systemic candidiasis and aspergillosis as well as pulmonary aspergillosis in rats.
A highly potent water soluble triazole antifungal prodrug, RO0098557 (1), has been identified from its parent, the novel antifungal agent RO0094815 (2). The prodrug includes a triazolium salt linked to an aminocarboxyl moiety, which undergoes enzymatic activation followed by spontaneous chemical degradation to release 2. Prodrug 1 showed high chemical stability and water solubility and exhibited strong antifungal activity against systemic candidiasis and aspergillosis as well as pulmonary aspergillosis in rats.

-
Chemistry
Scheme 1.We synthesized a series of new triazolium derivatives of Figure 1, Figure 3 and Scheme 1. CompoundsScheme 1 and Scheme 2, 6, 9, 10 and 11 were first prepared as outlined in Scheme 2 in order to analyze their stability and ability to release Figure 1, Figure 3 and Scheme 1. Next, aromatic analogues 18, 19, 20,21 and Figure 1, Figure 3 and Scheme 3 were synthesized for optimization of 11 to increase its water solubility and conversion rate. Compounds in the second series had sarcosine esters6 to make them water soluble, and they were also designed to generate acetaldehyde7 instead of formaldehyde for a better safety profile. The synthetic procedures for the second series of the derivatives are outlined in Scheme 3.

-
Scheme 2.
(a) ClCOOCH2Cl, diisopropylethylamine, CH2Cl2, rt (quant); (b) Figure 1, Figure 3 and Scheme 1, CH3CN, 80 °C (60%); (c) (1) ClCOOCH2Cl, Et3N, CH2Cl2, rt; (2) Ac2O, pyridine, rt (30%, two steps); (d) (1) NaI, CH3CN, 50 °C ; (2) Figure 1, Figure 3 and Scheme 1, CH3CN, 50 °C (88%, two steps); Synthesis of Scheme 1 and Scheme 2: (1) N-3-hydroxypropyl-N-methylamine, ClCOOCH2Cl, Et3N, CH2Cl2, rt; (2) AcCl, Et3N, CH2Cl2, rt (20%, two steps); (3) Figure 1, Figure 3 and Scheme 1, NaI, CH3CN, 50 °C (82%); Synthesis of 10: (1) l-prolinol, ClCOOCH2Cl, Et3N, CH2Cl2, rt; (2) Ac2O, pyridine, rt (<10%, 2 steps); (3) Figure 1, Figure 3 and Scheme 1, NaI, CH3CN, 50 °C (92%); Synthesis of 11: (1) 2-hydroxymethyl-N-methylaniline, ClCOOCH2Cl, diisopropylethylamine, CH2Cl2, rt; (2) Ac2O, diisopropylethylamine, rt (20%, two steps); (3)Figure 1, Figure 3 and Scheme 1, cat. NaI, CH3CN, reflux (63%).
-
Figure options

-
Scheme 3.
(a) (1) oxalyl chloride, DMF, 0 °C; (2) KOtBu, THF, −5 °C (97%, two steps); (b) CH3NH2, MeOH, rt (90%); (c) LiAlH4, THF, 0 °C (80%); (d) (1) ClCOOCH(CH3)Cl, diisopropylethylamine, CH2Cl2, 0 °C; (2) Boc-Sarcosine, WSCI, DMAP, CH2Cl2, 0 °C (84%, two steps); (e) (1) Figure 1, Figure 3 and Scheme 1, NaI, CH3CN, 50 °C; (2) DOWEX-1 Cl− form, aqueous MeOH, rt (65%, two steps); (f) (1) HCl, EtOAc, rt; (2) lyophilization (69%, two steps); Synthesis of 18: (1) (i) (4,5-difluoro-2-methylaminophenyl)methanol, ClCOOCH(CH3)Cl, diisopropylethylamine, CH2Cl2, 0 °C; (ii) Boc-Sarcosine, WSCI, DMAP, CH2Cl2, 0 °C (quant, two steps); (2) Figure 1, Figure 3 and Scheme 1, cat. NaI, CH3CN, 80 °C; (50%,); (3) HCl, EtOAc, rt (90%); Synthesis of 19: (1) (i) 2-fluoro-6-methylaminophenyl)methanol, ClCOOCH(CH3)Cl, diisopropylethylamine, CH2Cl2, 0 °C; (ii) Boc-Sarcosine, WSCI, DMAP, CH2Cl2, 0 °C (74%, two steps); (2) Figure 1, Figure 3 and Scheme 1, cat. NaI, CH3CN, reflux; (3) HCl, EtOAc, rt (29%, two steps); Synthesis of 20: (1) (i) (5-fluoro-2-methylaminophenyl)methanol, ClCOOCH(CH3)Cl, diisopropylethylamine, CH2Cl2, 0 °C; (ii) Boc-Sarcosine, WSCI, DMAP, CH2Cl2, 0 °C (91%, two steps); (2) Figure 1, Figure 3 and Scheme 1, cat. NaI, CH3CN, 70 °C (72%); (3) HCl, EtOAc, rt (88%); Synthesis of 21: (1) (i) (4-chloro-2-methylaminophenyl)methanol, ClCOOCH(CH3)Cl, diisopropylethylamine, CH2Cl2, 0 °C; (ii) Boc-Sarcosine, WSCI, DMAP, CH2Cl2, 0 °C (71%, two steps); (2) Figure 1, Figure 3 and Scheme 1, CH3CN, 65 °C; (3) HCl, EtOAc, rt (65%, two steps).
read more at
Boyd, B.; Castaner, J. BAL-4815/BAL-8557
Drugs Fut 2006, 31(3): 187
Antimicrobial Agents and Chemotherapy, 2008 , vol. 52, 4 p. 1396 – 1400
Ohwada, J.; Tsukazaki, M.; Hayase, T.; Oikawa, N.; Isshiki, Y.; Umeda, I.; Yamazaki, T.; Ichihara, S.; Shimma, N.Development of novel water antifungal, RO0098557
21st Med Chem Symp (November 28-30, Kyoto) 2001, Abst 1P-06
Ohwada, J.; Tsukazaki, M.; Hayase, T.; et al.
RO0098557, a novel water soluble azole prodrug for parenteral and oral administration (I). Design, synthesis, physicochemical properties and bioconversion42nd Intersci Conf Antimicrob Agents Chemother (ICAAC) (September 27-30, San Diego) 2002, Abst F-820
Tasaka et al., Chem. Pharm. Bull. 41(6) pp. 1035-1042 (1993).
Clinical trials
There have been three phase III clinical trials of isavuconazole, ACTIVE, VITAL and SECURE. As of June 2015, SECURE and VITAL have been presented in abstract form and results from ACTIVE have not been released.[9]
The SECURE trial compared voriconazole and isavuconazole in invasive fungal infections due to aspergillus. Isuvaconazole was found to be non-inferior to voriconazole, anothertriazole antifungal, with all cause mortality at 18.6%, compared to 20.2% in the voriconazole group. It additionally demonstrated a similar side effect profile.[10]
Data from the VITAL study showed that isavuconazole could be used in treatment of invasive mucormycosis, but did not evaluate its clinical efficacy for this indication.[11]
The ACTIVE trial is a comparison of isuvaconazole and caspofungin for invasive candida infections and results are anticipated in the second half of 2015.[12][13]
References
- [1]
- Saboo, Alok. “Basilea Announces Global Partnership With Astellas for Its Antifungal Isavuconazole.” FierceBiotech. N.p., 24 Feb. 2010. Web.
- “Basilea reports isavuconazole orphan drug designation by U.S. FDA.” Market Wired. 28 May 2013.
- “FDA Grants Orphan Drug Designation to Astellas for Isavuconazole for the Treatment of Invasive Candidiasis.” News Releases. Astellas. 3 Nov 2014.
- Cresemba (isovuconazole sulfate) [prescribing information]. Astella Pharma US, Inc. Revised March 2015.
- Jump up^ “Aspergillosis.” Centers for Disease Control and Prevention. Centers for Disease Control and Prevention, 08 Sept. 2014.
- Jump up^ “Astellas Receives FDA Approval for CRESEMBA® (isavuconazonium Sulfate) for the Treatment of Invasive Aspergillosis and Invasive Mucormycosis.” PR Newswire. N.p., 6 Mar. 2015.
- Jump up^ “Isavuconazonium.” Micromedex Solutions. Truven Health Analytics, n.d. Web. <www.micromedexsolutions.com>.
- Jump up^ Pettit, Natasha N.; Carver, Peggy L. (2015-07-01). “Isavuconazole A New Option for the Management of Invasive Fungal Infections”. Annals of Pharmacotherapy 49 (7): 825–842.doi:10.1177/1060028015581679. ISSN 1060-0280. PMID 25940222.
- Mujais, A. “2014: M-1756. A Phase 3 Randomized, Double-Blind, Non-Inferiority Trial Evaluating Isavuconazole (ISA) vs. Voriconazole (VRC) for the Primary Treatment of Invasive Fungal Disease (IFD) Caused by Aspergillus spp. or other Filamentous Fungi (SECURE): Outcomes by Malignancy Status”. http://www.icaaconline.com. Retrieved 2015-06-19.
- “Abstract: An Open-Label Phase 3 Study of Isavuconazole (VITAL): Focus on Mucormycosis (IDWeek 2014)”. idsa.confex.com. Retrieved 2015-06-19.
- Ltd., Basilea. “Basilea Pharmaceutica – Portfolio – Isavuconazole”. http://www.basilea.com. Retrieved 2015-06-19.
- “Isavuconazole (BAL8557) in the Treatment of Candidemia and Other Invasive Candida Infections – Full Text View – ClinicalTrials.gov”. clinicaltrials.gov. Retrieved 2015-06-19.
| US4861879 | Feb 9, 1988 | Aug 29, 1989 | Janssen Pharmaceutica N.V. | [[4-[4-Phenyl-1-piperazinyl)phenoxymethyl]-1-3-dioxolan-2-yl]-methyl]-1H-imidazoles and 1H-1,2,4-triazoles |
| US5900486 | Sep 9, 1997 | May 4, 1999 | Hoffmann-La Roche Inc. | N-benzylazolium derivatives |
| AU4536497A | Title not available | |||
| EP0667346A2 | Feb 3, 1995 | Aug 16, 1995 | Eisai Co., Ltd. | Azole antifungal agents, process for the preparation there of and intermediates |
| WO1992017474A1 | Mar 26, 1992 | Oct 15, 1992 | Pfizer | Triazole antifungal agents |
| US5648372 | Feb 1, 1995 | Jul 15, 1997 | Eisai Co., Ltd. | Antifungal agents, and compositions |
| US5686646 * | May 23, 1995 | Nov 11, 1997 | Schering-Plough Corporation | Chiral hydrazine derivatives |
| US5746840 * | Mar 28, 1997 | May 5, 1998 | Janssen Pharmaceutica, N.V. | Process for preparing enantiomerically pure 6-{4-chlorophenyl) (1 H-1,2,4-triazol-1-YL) methyl}-1-methyl-1 H-benzotriazole |
| US5792781 | Sep 18, 1996 | Aug 11, 1998 | Eisai Co., Ltd. | Antifungal agents, processes for the preparation thereof, and intermediates |
| US6020497 | Oct 9, 1998 | Feb 1, 2000 | Merck & Co., Inc. | 3-substitutes isoxazolidines as chiral auxiliary agents |
| US6133485 | Apr 15, 1998 | Oct 17, 2000 | Synphar Laboratories, Inc. | Asymmetric synthesis of 2-(2,4-difluorophenyl)-1-heterocycl-1-yl butan-2,3-diols |
| US6300353 | Mar 5, 1999 | Oct 9, 2001 | Basilea Pharmaceutica Ag, A Swiss Company | Azoles for treatment of fungal infections |
| US6383233 | Mar 7, 1997 | May 7, 2002 | Reuter Chemicscher Apparatebau Kg | Separation process |
| US6812238 * | Oct 31, 2000 | Nov 2, 2004 | Basilea Pharmaceutica Ag | N-substituted carbamoyloxyalkyl-azolium derivatives |
| US7151182 * | Sep 3, 2004 | Dec 19, 2006 | Basilea Pharmaceutica Ag | Intermediates for N-substituted carbamoyloxyalkyl-azolium derivatives |
| US7803949 * | Dec 20, 2006 | Sep 28, 2010 | Eisai R&D Management Co., Ltd. | Process for preparation of water-soluble azole prodrugs |
| US20030236419 | Dec 31, 2002 | Dec 25, 2003 | Sumika Fine Chemicals Co., Ltd. | Production methods of epoxytriazole derivative and intermediate therefor |
| US20040176432 | Jun 17, 2002 | Sep 9, 2004 | Milan Soukup | Intermediate halophenyl derivatives and their use in a process for preparing azole derivatives |
| WO2003002498A1 * | Jun 17, 2002 | Jan 9, 2003 | Basilea Pharmaceutica Ag | Intermediate halophenyl derivatives and their use in a process for preparing azole derivatives |
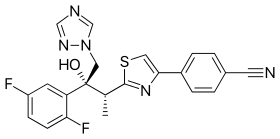 |
|
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
4-{2-[(1R,2R)-(2,5-Difluorophenyl)-2-hydroxy-1-methyl-3-(1H-1,2,4-triazol-1-yl)propyl]-1,3-thiazol-4-yl}benzonitrile
|
|
| Clinical data | |
| Trade names | Cresemba (prodrug form) |
| AHFS/Drugs.com | entry |
| Pregnancy category |
|
| Legal status |
|
| Routes of administration |
Oral, intravenous |
| Identifiers | |
| ATC code | None |
| PubChem | CID: 6918485 |
| ChemSpider | 5293682 |
| UNII | 60UTO373KE |
| ChEBI | CHEBI:85979 |
| ChEMBL | CHEMBL409153 |
| NIAID ChemDB | 416566 |
| Chemical data | |
| Formula | C22H17F2N5OS |
| Molecular mass | 437.47 g/mol |


/////

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....