Home » Posts tagged 'KC 3'
Tag Archives: KC 3
Progerinin
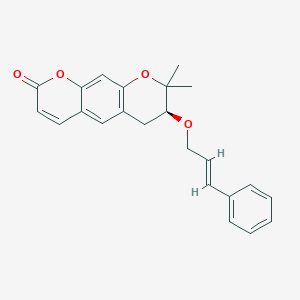
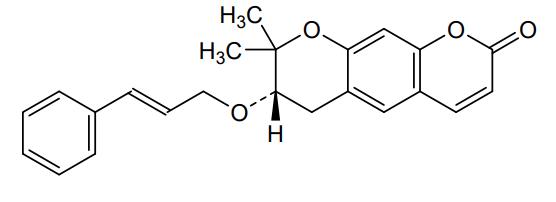
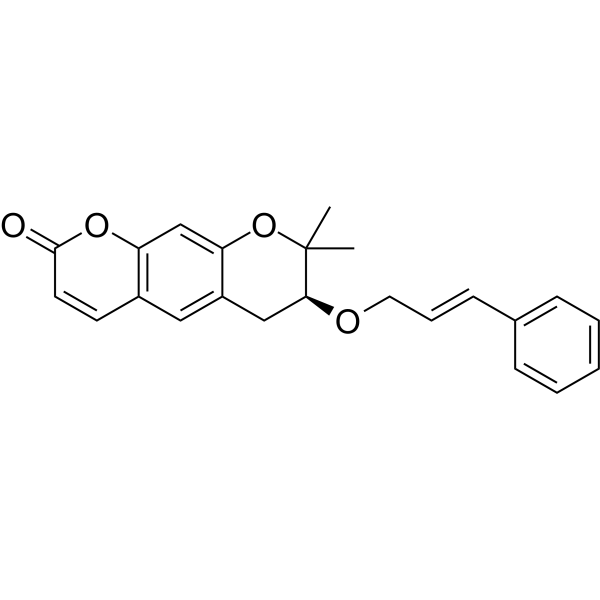
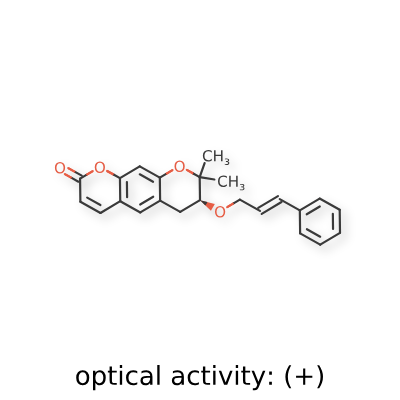
Progerinin
CAS 2249696-72-6
MF C23H22O4 MW 362.42
(3S)-2,2-dimethyl-3-[(E)-3-phenylprop-2-enoxy]-3,4-dihydropyrano[3,2-g]chromen-8-one
| (7S)-(+)-8,8-dimethyl-7-(3-phenyl-allyloxy)-7,8-dihydro-6H-pyrano[3,2-g]chromen-2-one |
| (7S)-7,8-Dihydro-8,8-dimethyl-7-[[(2E)-3-phenyl-2-propen-1-yl]oxy]-2H,6H-benzo[1,2-b:5,4-b?]dipyran-2-one |
(7S)-8,8-dimethyl-7-{[(2E)-3-phenylprop-2-en-1-yl]oxy}-7,8-dihydro-2H,6H-benzo[1,2-b:5,4-b’]dipyran-2-one
progerin-lamin A binding inhibitor, SLC-D011, SLC D011, KC 3, 426P9HSR8I
Progerinin (SLC-D011) is an orally active, targeted inhibitor designed to reduce the toxic, premature-aging protein “progerin” in Hutchinson-Gilford Progeria Syndrome (HGPS). It binds to progerin, disrupting its interaction with lamin A and promoting its degradation. Studies show it improves cardiac function, increases lifespan in mouse models, and is currently in clinical trials
Key Aspects of Progerinin:
- Mechanism of Action: Progerinin is an optimized progerin-lamin A binding inhibitor that selectively reduces progerin levels while sparing wild-type lamin A, B, and C.
- Disease Application: It targets HGPS, a rare genetic disease that causes premature, rapid aging and death due to cardiac issues.
- Preclinical Results: In Lmna mouse models, progerinin demonstrated improved physical conditions (hair morphology, body weight) and significantly extended lifespan (up to 14–21 weeks).
- Cardiac Benefits: It alleviates cardiovascular abnormalities, such as reducing cardiac muscle weakness, which is a major cause of death in HGPS patients.
- Clinical Status: A Phase I clinical trial was conducted for safety in healthy volunteers. As of 2025, trials are examining its efficacy, sometimes in combination with lonafarnib (Zokinvy).
- Administration: It is developed as a nanosuspension for oral administration. National Institutes of Health (NIH) | (.gov) +7
Progerinin was developed by Korean-based biotech company PRG Science & Technology Co., Ltd. (PRG S&T)
Progerinin (SLC-D011) is an orally active progerin-lamin A binding inhibitor. Progerinin selectively binds to the C-terminal region of progerin, disrupting its interaction with lamin A and promoting progerin degradation while sparing wild-type lamin A, B, and C. Progerinin ameliorates nuclear deformation, increases H3K9me3 levels, and reduces progerin expression in HGPS patient-derived fibroblasts. Progerinin extends lifespan in LmnaG609G/G609G mice and LmnaG609G/+ mice, improves body weight, hair morphology, cardiac function, and histological phenotypes. Progerinin can be used for the study of Hutchinson-Gilford progeria syndrome (HGPS).
- Study to Determine Optimal Dose and Evaluate Safety, Tolerability, and Pharmacokinetics of Progerinin in Patients With Hutchinson-Gilford Progeria Syndrome (HGPS)CTID: NCT06775041Phase: Phase 2Status: Active, not recruitingDate: 2026-02-09
- Phase 2, Open-Label Study to Evaluate the Safety and Tolerability of Progerinin in Werner SyndromeCTID: NCT05847179Phase: Phase 2Status: Not yet recruitingDate: 2026-01-23
- Phase I Study of Progerinin in Healthy VolunteersCTID: NCT04512963Phase: Phase 1Status: CompletedDate: 2021-09-22
PAPER
Publication Name: European Journal of Medicinal Chemistry
Publication Date: 2010-12
PMID: 20884093
DOI: 10.1016/j.ejmech.2010.09.006
SYN
<Example 1> Synthesis of Ether-Form (+)-Decursin Derivative (SLC-D011)
| (7S)-(+)-8,8-dimethyl-7-(3-phenyl-allyloxy)-7,8-dihydro-6H-pyrano[3,2-g]chromen-2-one (SLC-D011) was synthesized through the manner as in the to following Reaction Schemes 1 and 2. |
1. Synthesis Process I
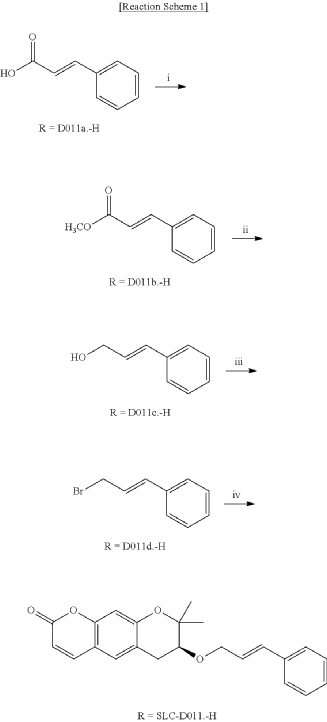
| Step (I): After dissolving trans-cinnamic acid (D0111a, 5 g, 33.7 mmol) in methanol (50 ml) in a 100 ml round bottom flask, 5 drops of concentrated H 2SO 4 was added and the mixture was refluxed by heating at 80° C. for 24 hours and was cooled to room temperature and then concentrated under reduced pressure. |
| Then, the mixture was separated with dichloromethane (300 ml) and distilled water (300 ml) to collect the organic layer and dehydrated with sodium sulfate and filtered. |
| After filtration, the filtrate was concentrated under reduced pressure to obtain 3-phenyl-acrylic acid, methyl ester (D011b, 5.39 g, yield=98.5%) as a pure product to apply to the next step. |
| The reaction solution was transferred to room temperature, stirred for 30 minutes and then a saturated aqueous solution of Rochelle’s salt (88 ml) was added thereto. |
2. Synthesis Process II
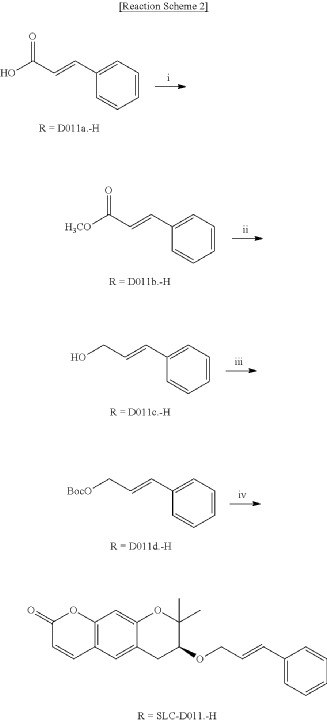
| The organic layers were collected and were dehydrated with sodium sulfate, filtered and the resulting filtrate was concentrated under reduced pressure. |
PAT
- A pharmaceutical composition for preventing or treating aging-related diseases containing a declucin derivative as an active ingredient.Publication Number: JP-6826674-B2Priority Date: 2017-04-25Grant Date: 2021-02-03
- Pharmaceutical composition for preventing or treating aging-related diseases containing decursin derivative as active ingredientPublication Number: EP-3617211-B1Priority Date: 2017-04-25Grant Date: 2025-10-22
- Pharmaceutical composition for preventing or treating aging-related diseases containing decursin derivative as active ingredientPublication Number: EP-3617211-A1Priority Date: 2017-04-25
- Pharmaceutical composition for preventing or treating aging-related diseases comprising decursin derivativesPublication Number: KR-20180119490-APriority Date: 2017-04-25
- Pharmaceutical composition for preventing or treating aging-related diseases containing decursin derivative as active ingredientPublication Number: US-2020048274-A1Priority Date: 2017-04-25
- Pharmaceutical composition for preventing or treating aging-related diseases containing decursin derivative as active ingredientPublication Number: CN-110573514-APriority Date: 2017-04-25
- Pharmaceutical composition for preventing or treating aging-related diseases containing decursin derivative as active ingredientPublication Number: US-11008332-B2Priority Date: 2017-04-25Grant Date: 2021-05-18
- A kind of methyl isobutyl ketone low-temperature hydrogenation catalyst, preparation method and applicationPublication Number: CN-110871080-BPriority Date: 2018-08-30Grant Date: 2022-08-09
- Herbicidal composition containing bicyclopyrone and application thereofPublication Number: CN-113057169-BPriority Date: 2017-11-09Grant Date: 2022-07-01
- Preparation method for propylene epoxidation catalyst, and application thereofPublication Number: US-11291985-B2Priority Date: 2017-10-27Grant Date: 2022-04-05
- Pharmaceutical composition for preventing or treating aging-related diseases comprising decursin derivativesPublication Number: KR-102070328-B1Priority Date: 2017-04-25Grant Date: 2020-01-28
- Pharmaceutical composition for preventing or treating aging-related diseases containing decursin derivative as active ingredientPublication Number: WO-2018199633-A1Priority Date: 2017-04-25
- Tires comprising rubber compounds that comprise propylene-α-olefin-diene polymersPublication Number: US-12281221-B2Priority Date: 2019-07-17Grant Date: 2025-04-22
- Memory of sequences, method for creation and functioning of sequence memory, hierarchical sequence memoryPublication Number: US-2022027408-A1Priority Date: 2019-04-04
- A kind of preparation method of isovaleraldehydePublication Number: CN-111718247-BPriority Date: 2019-03-21Grant Date: 2022-08-05
- Deuterated analogs of acetyl-leucinePublication Number: CN-120157593-APriority Date: 2018-12-06
- Deuterated analogs of acetyl-leucinePublication Number: CN-113348018-BPriority Date: 2018-12-06Grant Date: 2025-04-01



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

//////////progerinin, progerin-lamin A binding inhibitor, SLC-D011, SLC D011, KC 3, 426P9HSR8I

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










