Home » Posts tagged 'GMP' (Page 6)
Tag Archives: GMP
Stelara (ustekinumab) Receives FDA Approval to Treat Active Psoriatic Arthritis

Ustekinumab
| CAS No: | 815610-63-0 |
|---|---|
| Molecular Weight: | 145.64 g/mol |
| Chemical Formula: | C9H18N2O2 |
| IUPAC Name: | Immunoglobulin G1, anti-(human interleukin 12 p40 subunit) (human monoclonal CNTO 1275 gamma1-chain), disulfide with human monoclonal CNTO 1275 kappa-chain, dimer |
HORSHAM, Pa., Sept. 23, 2013 /PRNewswire/ — Janssen Biotech, Inc., announced today that the U.S. Food and Drug Administration (FDA) has approved Stelara (ustekinumab) alone or in combination with methotrexate for the treatment of adult patients (18 years or older) with active psoriatic arthritis. It is estimated that more than two million people in the U.S. are living with psoriatic arthritis, a chronic autoimmune disease characterized by both joint inflammation and psoriasis skin lesions
read all at
Ustekinumab (INN, experimental name CNTO 1275, proprietary commercial name Stelara, Centocor) is a human monoclonal antibody. It is directed against interleukin 12 and interleukin 23, naturally occurring proteins that regulate the immune system and immune-mediated inflammatory disorders.
Ustekinumab is a fully human monoclonal antibody (mAb) targeting the interleukin (IL)-12/23p40 subunit.
Interleukins are small soluble proteins that communicate between white blood cells (leukocytes), such as T cells. Interleukins mediate the differentiation, proliferation and many other processes of these cells. IL-12 and IL-23 are involved in the differentiation of naive T cells into T helper (Th) 1 and Th17 cells respectively.
Th1 and Th17 cells have been implicated in several autoimmune disorders, such as psoriasis. Ustekinumab targets the common p40 subunit of IL-12 and IL-23 to stop these cytokines from binding to their receptors and consequently preventing the development of Th1 and Th17 cells in an immune response.
In two Phase III trials for moderate to severe psoriasis, the longest >76 weeks, ustekinumab was safe and effective.
A third Phase III trial, ACCEPT, compared the efficacy and safety of ustekinumab with etanercept in the treatment of moderate to severe plaque psoriasis. This trial found a significantly higher clinical response with ustekinumab over the 12-week study period compared to high-dose etanercept. It also demonstrated the clinical benefit of ustekinumab among patients who failed to respond to etanercept.
Ustekinumab is approved in Canada, Europe and the United States to treat moderate to severe plaque psoriasis.
As of November 2009, the drug is being investigated for the treatment of psoriatic arthritis. It has also been tested in Phase II studies for multiple sclerosis and sarcoidosis, the latter versus golimumab (Simponi).
The US Food and Drug Administration (FDA) and European Union (EU) have approved the interleukin (IL) 12/23 inhibitor ustekinumab (Stelara, Janssen Biotech) for adults with active psoriatic arthritis who have not responded adequately to previous nonbiological disease-modifying antirheumatic drug therapy, the company announced today.
Approval of ustekinumab for psoriatic arthritis is “significant for patients and physicians as it marks the first treatment approved for this devastating and complex disease since the introduction of anti-TNF biologic medicines more than a decade ago,” Jerome A. Boscia, MD, vice president and head of immunology development, Janssen Research & Development, LLC, said in a statement.
The European Medicine Agency’s Committee for Medicinal Products for Human Use (CHMP) recommended approval of ustekinumab for active psoriatic arthritis in June, as reported by Medscape Medical News.
Ustekinumab is already approved in the US and EU for treatment of moderate to severe psoriatic plaques in adults. The drug, which can be used alone or in combination with methotrexate, is novel in that it targets both IL-12 and IL-23.
Image source: Crystal structure of human IL-12, Wikipedia, public domain

Ustekinumab binding to IL-12/23p40
EFAVIRENZ – Huahai Pharma China-Approved to Produce AIDS Treatment

Efavirenz
DMP 266
- Sustiva (USA, Bristol-Myers Squibb)
- Stocrin (EU, MSD)
- Aspen Efavirenz (Sub-Saharan Africa, Aspen Pharmacare)
- E.F (McNeil & Argus)
- Efavir (Cipla)
- Efcure (Emcure Pharmaceuticals)
- Efferven (Ranbaxy Laboratories)
- Estiva (Hetero)
- Evirenz (Alkem Laboratories)
- Viranz (Aurobindo Pharma)
Zhejiang Huahai Pharma received CFDA approval to produce efavirenz, an oral non-nucleoside reverse transcriptase inhibitor (NNRTI) used to control the symptoms of AIDS. Huahai is the first China drugmaker approved to make the drug. Huahai produced efavirenz API for Merck, which marketed the drug under the name Stocrin
read at
http://www.sinocast.com/readbeatarticle.do?id=99634
Efavirenz (EFV), sold under the brand names Sustiva among others, is a non-nucleoside reverse transcriptase inhibitor (NNRTI). It is used as part of highly active antiretroviral therapy (HAART) for the treatment of a human immunodeficiency virus (HIV) type 1. For HIV infection that has not previously been treated, the United States Department of Health and Human Services Panel on Antiretroviral Guidelines currently recommends the use of efavirenz in combination with tenofovir/emtricitabine (Truvada) as one of the preferred NNRTI-based regimens in adults and adolescents.[1] Efavirenz is also used in combination with other antiretroviral agents as part of an expanded postexposure prophylaxis regimen to reduce the risk of HIV infection in people exposed to a significant risk (e.g. needlestick injuries, certain types of unprotected sex etc.).
It is usually taken on an empty stomach at bedtime to reduce neurological and psychiatric adverse effects.
Efavirenz was combined with the HIV medications tenofovir and emtricitabine, all of which are reverse transcriptase inhibitors. This combination of three medications under the brand name Atripla, provides HAART in a single tablet taken once a day.
Efavirenz was discovered at Merck Research Laboratories. It is on the WHO Model List of Essential Medicines, the most important medication needed in a basic health system.[2] As of 2015 the cost for a typical month of medication in the United States is more than 200 USD.[3]
Efavirenz (EFV, brand names Sustiva, Stocrin, Efavir etc.) is a non-nucleoside reverse transcriptase inhibitor (NNRTI) and is used as part of highly active antiretroviral therapy(HAART) for the treatment of a human immunodeficiency virus (HIV) type 1.
For HIV infection that has not previously been treated, the United States Department of Health and Human Services Panel on Antiretroviral Guidelines currently recommends the use of efavirenz in combination with tenofovir/emtricitabine (Truvada) as one of the preferred NNRTI-based regimens in adults and adolescents.
Efavirenz is also used in combination with other antiretroviral agents as part of an expanded postexposure prophylaxis regimen to reduce the risk of HIV infection in people exposed to a significant risk (e.g. needlestick injuries, certain types of unprotected sex etc.).
The usual adult dose is 600 mg once a day. It is usually taken on an empty stomach at bedtime to reduce neurological and psychiatric adverse effects.
Efavirenz was combined with the popular HIV medication Truvada, which consists oftenofovir and emtricitabine, all of which are reverse transcriptase inhibitors. This combination of three medications approved by the U.S. Food and Drug Administration(FDA) in July 2006 under the brand name Atripla, provides HAART in a single tablet taken once a day. It results in a simplified drug regimen for many patients.

doi:10.1016/0040-4039(95)01955-H
Merck synthesis of Efavirenz
History
Efavirenz was approved by the FDA on September 21, 1998, making it the 14th approved antiretroviral drug.
-
Efavirenz is a non-nucleoside reverse trancriptase inhibitor being studied clinically for use in the treatment of HIV infections and AIDS.
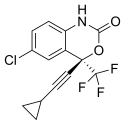
- Efavirenz chemically known as (-) 6-Chloro-4-cyclopropylethynyl-4-trifluoromethyl- 1 , 4- dihydro-2H-3, 1-benzoxa zin-2-one, is a highly potent non-nucleoside reverse transcriptase inhibitor (NNRTI).A number of compounds are effective in the treatment of the human immunodeficiency virus (HIV) which is the retrovirus that causes progressive destruction of the human immune system. Effective treatment through inhibition of HIV reverse transcriptase is known for non- nucleoside based inhibitors. Benzoxazinones have been found to be useful non-nucleoside based inhibitors of HIV reverse transcriptase.(-) β-chloro^-cyclopropylethynyM-trifluoromethyl-l ,4-dihydro-2H-3,l -ben zoxazin-2-one (Efavirenz) is efficacious against HIV reverse transcriptase resistance. Due to the importance of (-)6-chloro-4-cyclopropylethynyl-4-trifluoromethyl-l,4-dihydro-2H-3,l-ben zoxazin-2- one, economical and efficient synthetic processes for its production needs to be developed.The product patent US5519021. discloses the preparation of Efavirenz, in Example-6, column-29, involving cyclisation of racemic mixture of 2-(2-amino-5-chlorophenyl)-4- cyclopropyl-l,l,l-trifluoro-3-butyn-2-ol using l ,l ‘-carbonyldiimidazole as carbonyl delivering agent to give racemic Efavirenz. Further, resolution of the racemic Efavirenz is carried out using (-) camphanic acid chloride to yield optically pure Efavirenz. However, research article published in the Drugs of the future, 1998, 23(2), 133-141 discloses process for manufacture of optically pure Efavirenz. The process involves cyclisation of racemic 2-(2-amino-5-chlorophenyl)-4-cyclopropyl-l, 1, l-trifluoro-3-butyn-2- ol using 1, 1-carbonyldiimidazole as carbonyl delivering agent to give racemic Efavirenz and further resolution by (-) camphanic acid chloride.Similarly research article published in Synthesis 2000, No. 4, 479-495 discloses stereoselective synthesis of Efavirenz (95%yield, 99.5%ee), as shown below
Even though many prior art processes report method for the preparation of Efavirenz, each process has some limitations with respect to yield, purity, plant feasibility etc. Hence in view of the commercial importance of Efavirenz there remains need for an improved process.
- US 6 028 237 discloses a process for the manufacture of optically pure Efavirenz.
-
The synthesis of efavirenz and structurally similar reverse transcriptase inhibitors are disclosed in US Patents 5,519,021, 5,663,169, 5,665,720 and the corresponding PCT International Patent Application WO 95/20389, which published on August 3, 1995. Additionally, the asymmetric synthesis of an enantiomeric benzoxazinone by a highly enantioselective acetylide addition and cyclization sequence has been described by Thompson, et al., Tetrahedron Letters 1995, 36, 8937-8940, as well as the PCT publication, WO 96/37457, which published on November 28, 1996.
-
Additionally, several applications have been filed which disclose various aspects of the synthesis of(-)-6-chloro-4-cyclopropylethynyl-4-trifluoromethyl-1,4-dihydro-2H-3,1-benzoxazin-2-one including: 1) a process for making the chiral alcohol, U.S.S.N. 60/035,462, filed 14 January 1997; 2) the chiral additive, U.S.S.N. 60/034,926, filed 10 January 1997; 3) the cyclization reaction, U.S.S.N. 60/037,059, filed 12 February 1997; and the anti-solvent crystallization procedure, U.S.S.N. 60/037,385 filed 5 February 1997 and U.S.S.N. 60/042,807 filed 8 April 1997.




Syntheses of EFV API; different routes of manufacturingAPI, active pharmaceutical ingredient; EFV efavirenz. BELOW
Related substances and degradants (partial listing) in EFVAPI, active pharmaceutical ingredient; CPA, cyclopropylacetylene; EFV, efavirenz
Syntheses of EFV API; different routes of manufacturingAPI, active pharmaceutical ingredient; EFV efavirenz.
Chemical properties
Efavirenz is chemically described as (S)-6-chloro-(cyclopropylethynyl)-1,4-dihydro-4-(trifluoromethyl)-2H-3,1-benzoxazin-2-one. Its empirical formula is C14H9ClF3NO2. Efavirenz is a white to slightly pink crystalline powder with a molecular mass of 315.68 g/mol. It is practically insoluble in water (<10 µg/mL).
History
Efavirenz was approved by the FDA on September 21, 1998, making it the 14th approved antiretroviral drug.
Society and culture
Pricing information
A one-month supply of 600 mg tablets cost approximately $550 in April 2008.[16] Merck provides efavirenz in certain developing countries at cost, currently about $0.65 per day.[17] Some emerging countries have opted to purchase Indian generics[18] such as Efavir by Cipla Ltd.[19] In Thailand, one month supply of efavirenz + truvada, as of June 2012, costs THB 2900 ($90), there’s also a social program for poorer patients who can’t afford even this price. In South Africa, a license has been granted to generics giant Aspen Pharmacare to manufacture, and distribute to Sub-Saharan Africa, a cost-effective antiretroviral drug.[20]
PATENT
http://www.google.com/patents/WO1999061026A1?cl=en
EXAMPLE 1
Cl
1a
To a solution of trifluoroethanol and (IR, 2S)-N-pyrrolidinyl norephedrine in THF (9 L) under nitrogen is added a solution of diethylzinc in hexane at 0 °C slowly enough to keep the temperature below 30 °C. The mixture is stirred at room temperature for 0.5 ~ 1 h. In another dry flask a solution of chloromagnesium cyclopropyl acetylide is prepared as follows: To neat cyclopropyl acetylene at 0 °C is added a solution of rc-butylmagnesium chloride slowly enough to keep the internal temperature < 30 °C. The solution is stirred at 0 °C for ~ 40 min and transfered to the zinc reagent via cannula with 0.36 L of THF as a wash. The mixture is cooled to -10 °C and ketoaniline la is added. The mixture is stirred at -2 to -8 °C for 35 h, warmed to room temperature, stirred for 3 h, and quenched with 30% potassium carbonate over 1.5 h. The mixture is stirred for 4 h and the solid is removed by filtration and washed with THF (2 cake volume). The wet solid still contains -18 wt% of pyrrolidinyl norephedrine and is saved for further study. The filtrate and wash are combined and treated with 30% citric acid. The two layers are separated. The organic layer is washed with water (1.5 L). The combined aqueous layers are extracted with 2.5 L of toluene and saved for norephedrine recovery. The toluene extract is combined with the organic solution and is concentrated to ~ 2.5 L. Toluene is continuously feeded and distilled till THF is not detectable by GC. The final volume is controlled at 3.9 L. Heptane (5.2 L) is added over 1 h. The slurry is cooled to 0 °C, aged for 1 h, and filtered. The solid is washed with heptane (2 cake volume) and dried to give 1.234 Kg (95.2% yield) of amino alcohol 3 as a white crystalline. The material is 99.8 A% pure and 99.3% ee.
EXAMPLE 2
To a three necked round bottom flask, equipped with a mechanical stirrer, nitrogen line, and thermocouple, was charged the solid amino alcohol 3, MTBE (500 L), and aqueous KHCO3 (45 g in 654 mL H2O). Solid 4-nitrophenyl chloroformate was added, in 4 batches, at 25°C. During the addition the solution pH was monitored. The pH was maintained between 8.5 and 4 during the reaction and ended up at 8.0. The mixture was stirred at 20-25°C for two hours. Aqueous KOH (2N) was added over 20 minutes, until the pH of the aqueous layer reached 11.0.
The layers were separated and 500 mL brine was added to the MTBE layer. 0.1 N Acetic acid was added until the pH was 6-7. The layers were separated and the organic phase was washed with brine (500 mL). At this point the mixture was solvent switched to EtOH/IPA and crystallized as recited in Examples 5 and 6.
EXAMPLE 3
To a three necked round bottom flask, equipped with a mechanical stirrer, nitrogen line, and thermocouple, was charged the solid amino alcohol 3a, toulene (500 mL), and aqueous KHCO3 (86.5 g in 500 L H2O). Phosgene solution in toulene was added at 25°C, and the mixture was stirred at 20-25°C for two hours.
The layers were separated and the organic phase was washed with brine (500 mL). At this point the mixture was solvent switched to EtOH/IPA and crystallized as recited in Examples 5 and 6.
EXAMPLE 4
To a three necked round bottom flask, equipped with a mechanical stirrer, nitrogen line, and thermocouple, was charged the solid amino alcohol 3a, MTBE (500 mL), and aqueous KHCO3 (86.5 g in 500 mL H2O). Phosgene gas was slowly passed into the solution at 25°C, until the reaction was complete.
The layers were separated and the organic phase was washed with brine (500 mL). At this point the mixture was solvent switched to EtOH/IPA and crystallized as recited in Examples 5 and 6.
EXAMPLE 5
Crystallization of efavirenz from 30% 2-Propanol in Water using a ratio of 15 ml solvent per gram efavirenz Using Controlled Anti-Solvent Addition on a 400 g Scale.
400 g. of efavirenz starting material is dissolved in 1.8 L of 2- propanol. The solution is filtered to remove extraneous matter. 1.95 L of deionized (DI) water is added to the solution over 30 to 60 minutes. 10 g. to 20 g. of efavirenz seed (Form II wetcake) is added to the solution. The seed bed is aged for 1 hour. The use of Intermig agitators is preferred to mix the slurry. If required (by the presence of extremely long crystals or a thick slurry), the slurry is wet-milled for 15 – 60 seconds. 2.25 L of DI water is added to the slurry over 4 to 6 hours. If required (by the presence of extremely long crystals or a thick slurry), the slurry is wet- milled for 15 – 60 seconds during the addition. The slurry is aged for 2 to 16 hours until the product concentration in the supernatant remains constant. The slurry is filtered to isolate a crystalline wet cake. The wet cake is washed with 1 to 2 bed volumes of 30 % 2-propanol in water and then twice with 1 bed volume of DI water each. The washed wet cake is dried under vacuum at 50°C.
EXAMPLE 6
Crystallization of efavirenz from 30% 2-Propanol in Water using a ratio of 15 ml solvent per gram efavirenz Using a Semi-Continuous Process on a 400 g Scale.
400 g. of efavirenz starting material is dissolved in 1.8 L of 2- propanol. A heel slurry is produced by mixing 20 g. of Form II efavirenz in 0.3 L of 30 % (v/v) 2-propanol in water or retaining part of a slurry froma previous crystallization in the crystallizer. The dissolved batch and 4.2 L of DI water are simultaneously charged to the heel slurry at constant rates over 6 hours to maintain a constant solvent composition in the crystallizer. Use of Intermig agitators during the crystallization is preferred. During this addition the slurry is wet-milled when the crystal lengths become excessively long or the slurry becomes too thick. The slurry is aged for 2 to 16 hours until the product concentration in the supernatant remains constant. The slurry is filtered to isolate a crystalline wet cake. The wet cake is washed with 1 to 2 bed volumes of 30 % 2-propanol in water and then twice with 1 bed volume of DI water each. The washed wet cake is dried under vacuum at 50°C.
EXAMPLE 7 Preparation of Amino Alcohol 3 and ee Upgrading— Through Process
1a
A solution of diethyl zinc in hexane was added to a solution of trifluoroethanol (429.5 g, 4.29’mol) and (IR, 2S)-N-pyrrolidinyl norephedrine (1.35 kg, 6.58 mol) in THF (9 L), under nitrogen, at 0 °C. The resulting mixture was stirred at room temperature for approx. 30 min. In another dry flask a solution of chloromagnesium- cyclopropylacetylide was prepared as follows. To a solution of n- butylmagnesium chloride in THF (2 M, 2.68 L, 5.37 mol) was added neat cyclopropylacetylene at 0 °C keeping the temperature < 25 °C. The solution was stirred at 0 °C for 1 ~ 2 h. The solution of chloromagnesiumcyclopropylacetylide was then warmed to room temperature and was transferred into the zinc reagent via cannula over 5 min followed by vessel rinse with 0.36 L of THF. The resulting mixture was aged at ~ 30 °C for 0.5 h and was then cooled to 20 °C. The ketoaniline 1 (1.00 kg, 4.47 mol) was added in one portion as a solid, and the resulting mixture was stirred at 20-28 °C for 3 h.
The reaction was quenched with 30% aq. potassium carbonate (1.2 L) and aged for 1 h. The solid waste was filtered and the cake was washed with THF (3 cake volumes). The filtrate and wash were combined and solvent switched to IP Ac.
The IPAc solution of product 3 and pyrrolidinyl norephedrine was washed with citric acid (3.5 L) and with water (1.5 L). The combined aqueous layers were extracted with IPAc (2 L) and saved for norephedrine recovery. To the combined organic layers was added
12N HC1 (405 mL, 4.88 mol), to form a thin slurry of the amino alcohol-
HC1 salt. The mixture was aged for 30 min at 25 °C and was then dried azeotropically. The slurry was aged at 25 °C for 30 min and filtered. The cake was washed with 2.5 L of IPAc and dried at 25 °C under vacuum/nitrogen for 24 h to give 1.76 kg of the wet HC1 salt.
The salt was dissolved in a mixture of MTBE (6 L) and aq Na2Cθ3 (1.18 kg in 6.25 L water). The layers were separated and the organic layer was washed with 1.25 L of water. The organic layer was then solvent switched into toluene.
Heptane (5 L) was added over 1 h at 25 °C. The slurry was cooled to 0 °C, aged for 1 h, and filtered. The solid was washed with heptane (2 cake volumes) and was dried to give 1.166 kg (90% overall yield) of amino alcohol 3 as a white crystalline solid. Norephedrine recovery
The aqueous solution was basified to pH13 using 50% aq NaOH, and extracted with heptane (2 L). The heptane solution was washed with water (1 L) and concentrated to remove residual IPAc and water. The final volume was adjusted to about 3 L. The heptane solution was cooled to -20 °C, aged for 2 h, and filtered. The solid was washed with cold heptane (1 cake volume) and dried to give 1.269 kg solid (94% recovery)




CLIPS
http://www.mdpi.com/1420-3049/21/2/221/htm



| WO2007013047A2 * | Jul 31, 2006 | Feb 1, 2007 | Ranbaxy Lab Ltd | Water-dispersible anti-retroviral pharmaceutical compositions |
| WO2007013047A3 * | Jul 31, 2006 | May 31, 2007 | Ranbaxy Lab Ltd | Water-dispersible anti-retroviral pharmaceutical compositions |
| WO2007052289A2 * | Jul 24, 2006 | May 10, 2007 | Rubicon Res Pvt Ltd | Novel dispersible tablet composition |
| WO2007052289A3 * | Jul 24, 2006 | Dec 27, 2007 | Rubicon Res Pvt Ltd | Novel dispersible tablet composition |
| WO2011131943A2 | Apr 20, 2011 | Oct 27, 2011 | Cipla Limited | Pharmaceutical compositions |
| WO2012048884A1 | Oct 14, 2011 | Apr 19, 2012 | Lonza Ltd | Process for the synthesis of cyclic carbamates |
| WO2012048886A1 | Oct 14, 2011 | Apr 19, 2012 | Lonza Ltd | Process for the synthesis of cyclic carbamates |
| WO2015059466A1 | Oct 22, 2014 | Apr 30, 2015 | Cipla Limited | Pharmaceutical compositions comprising efavirenz |
| EP1448170A2 * | Nov 26, 2002 | Aug 25, 2004 | Bristol-Myers Squibb Company | Efavirenz tablet formulation having unique biopharmaceutical characteristics |
| EP2441759A1 | Oct 14, 2010 | Apr 18, 2012 | Lonza Ltd. | Process for the synthesis of cyclic carbamates |
| EP2447255A1 | Oct 14, 2010 | May 2, 2012 | Lonza Ltd. | Process for the synthesis of cyclic carbamates |
| US6238695 | Apr 6, 1999 | May 29, 2001 | Dupont Pharmaceuticals Company | Formulation of fast-dissolving efavirenz capsules or tablets using super-disintegrants |
| US6555133 | Apr 2, 2001 | Apr 29, 2003 | Bristol-Myers Squibb Company | Formulation of fast-dissolving efavirenz capsules or tablets using super-disintegrants |
| US8871271 | Jul 29, 2013 | Oct 28, 2014 | Gilead Sciences, Inc. | Method and composition for pharmaceutical product |
| US8957204 | Oct 14, 2011 | Feb 17, 2015 | Lonza Ltd. | Process for the synthesis of cyclic carbamates |
| US8969550 | Oct 14, 2011 | Mar 3, 2015 | Lonza Ltd. | Process for the synthesis of cyclic carbamates |
| US9018192 | Oct 10, 2013 | Apr 28, 2015 | Bristol-Myers Squibb & Gilead Sciences, Inc. | Unitary pharmaceutical dosage form |
| US9198862 | Jul 24, 2006 | Dec 1, 2015 | Rubicon Research Private Limited | Dispersible tablet composition |
| WO1995020389A1 * | Jan 24, 1995 | Aug 3, 1995 | Merck & Co Inc | Benzoxazinones as inhibitors of hiv reverse transcriptase |
| WO1996037457A1 * | May 21, 1996 | Nov 28, 1996 | Merck & Co Inc | Asymmetric synthesis of (-) 6-chloro-4-cyclopropyl-ethynyl-4-trifluoromethyl-1,4-dihydro-2h-3,1-benzoxazin-2-one |
| WO1998052570A1 * | May 14, 1998 | Nov 26, 1998 | David Walter Barry | Antiviral combinations containing the carbocyclic nucleoside 1592u89 |
References
- 1 “Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents”. Retrieved 10 May 2013.
- 2
- “WHO Model List of EssentialMedicines” (PDF). World Health Organization. October 2013. Retrieved 22 April 2014.
- 3
- Hamilton, Richart (2015). Tarascon Pocket Pharmacopoeia 2015 Deluxe Lab-Coat Edition. Jones & Bartlett Learning. p. 62. ISBN 9781284057560.
- 4
- Cespedes, MS; Aberg, JA (2006). “Neuropsychiatric complications of antiretroviral therapy.”. Drug safety : an international journal of medical toxicology and drug experience 29 (10): 865–74. doi:10.2165/00002018-200629100-00004. PMID 16970510.
- 5
- “www.accessdata.fda.gov” (PDF).
- 6
- DHHS panel. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents (October 10, 2006). (Available for download from AIDSInfo)
- 7
- Ford, N.; Mofenson, L.; Kranzer, K.; Medu, L.; Frigati, L.; Mills, E. J.; Calmy, A. (2010). “Safety of efavirenz in first-trimester of pregnancy: A systematic review and meta-analysis of outcomes from observational cohorts”. AIDS 24 (10): 1461–1470. doi:10.1097/QAD.0b013e32833a2a14. PMID 20479637.
- 8
- Rossi, S; Yaksh, T; Bentley, H; Van Den Brande, G; Grant, I; Ellis, R (2006). “Characterization of interference with 6 commercial delta9-tetrahydrocannabinol immunoassays by efavirenz (glucuronide) in urine”. Clinical Chemistry 52 (5): 896–7. doi:10.1373/clinchem.2006.067058. PMID 16638958.
- 9
- Röder, CS; Heinrich, T; Gehrig, AK; Mikus, G (2007). “Misleading results of screening for illicit drugs during efavirenz treatment”. AIDS (London, England) 21 (10): 1390–1. doi:10.1097/QAD.0b013e32814e6b3e. PMID 17545727.
- 10
- Ren J, Bird LE, Chamberlain PP; et al. (2002). “Structure of HIV-2 reverse transcriptase at 2.35-A resolution and the mechanism of resistance to non-nucleoside inhibitors”. Proc Natl Acad Sci USA 99 (22): 14410–15. doi:10.1073/pnas.222366699. PMC 137897. PMID 12386343.
- 11
- Sustiva (efavirenz) capsules and tablets. Product information (April 2005)
- 12
- Simen AA, Ma J, Svetnik V, Mayleben D, Maynard J, Roth A, Mixson L, Mogg R, Shera D, George L, Mast TC, Beals C, Stoch A, Struyk A, Shire N, Fraser I (2014). “Efavirenz modulation of sleep spindles and sleep spectral profile”. J Sleep Res 24: 66–73. doi:10.1111/jsr.12196. PMID 25113527.
- 13
- Gatch MB, Kozlenkov A, Huang RQ, Yang W, Nguyen JD, González-Maeso J, Rice KC, France CP, Dillon GH, Forster MJ, Schetz JA (2013). “The HIV antiretroviral drug efavirenz has LSD-like properties”. Neuropsychopharmacology 38 (12): 2373–84. doi:10.1038/npp.2013.135. PMC 3799056. PMID 23702798.
- 14
- Dabaghzadeh F, Ghaeli P, Khalili H, Alimadadi A, Jafari S, Akhondzadeh S, Khazaeipour Z (2013). “Cyproheptadine for prevention of neuropsychiatric adverse effects of efavirenz: a randomized clinical trial”. AIDS Patient Care STDS 27 (3): 146–54. doi:10.1089/apc.2012.0410. PMID 23442031.
- 15
- Dabaghzadeh F, Khalili H, Ghaeli P, Dashti-Khavidaki S (2012). “Potential benefits of cyproheptadine in HIV-positive patients under treatment with antiretroviral drugs including efavirenz”. Expert Opin Pharmacother 13 (18): 2613–24. doi:10.1517/14656566.2012.742887. PMID 23140169.
- 16
- Price listed on http://drugstore.com website, 4/20/2008
- 17
- “Merck & Co., Inc., Again Reduces Price of Stocrin (efavirenz) for Patients in Least Developed Countries and Countries Hardest Hit by Epidemic – Drugs.com MedNews”.
- 18
- IndiaDaily – A new trend in emerging nations – Brazil opts for Indian generic drug ignoring US pharmaceutical giant Merck’s patent on AIDS drug Efavirenz
- 19
- http://www.cipla.com
- 20
- Patrick Lumumba Osewe; Yvonne Korkoi Nkrumah; Emmanuel K. Sackey (15 June 2008). Improving Access to HIV/AIDS Medicines in Africa: Trade-Related Aspects of Intellectual Property Rights (TRIPS) Flexibilities Utilization. World Bank Publications. pp. 35–39. ISBN 978-0-8213-7544-0. Retrieved 30 June 2012.
- 21
- http://www.sustiva.com/
- 22
- http://www.medsafe.govt.nz/consumers/cmi/s/stocrin.pdf
- 23
- Drugsupdate.com generic brands list: http://www.drugsupdate.com/brand/generic/Efavirenz/87
- 24
- http://mcneilargusindia.com/
- 25
- http://www.alkemlabs.com/
- 26
- “Regast® (efavirenz) film-coated tablets.”. http://www.pharmasyntez.com (in Russian). Pharmasyntez, 2011. Retrieved 28 June 2015. External link in
|website=(help) - 27
- IOL: Thugs get high on stolen Aids drugs IOL News May 12, 2007
- 28
- Getting high on HIV drugs in S Africa. BBC News, 8 December 2008.
- 29
- ‘No Turning Back’: Teens Abuse HIV Drugs. ABC News, April 6, 2009.
- 30
- Getting High On HIV Medication Vice 7.04.2014.
- 31
- Gatch, M. B.; Kozlenkov, A.; Huang, R. Q.; Yang, W.; Nguyen, J. D.; González-Maeso, J.; Rice, K. C.; France, C. P.; Dillon, G. H.; Forster, M. J.; Schetz, J. A. (2013). “The HIV Antiretroviral Drug Efavirenz has LSD-Like Properties”. Neuropsychopharmacology 38 (12): 2373–84. doi:10.1038/npp.2013.135. PMC 3799056. PMID 23702798.
- Sütterlin, S.; Vögele, C.; Gauggel, S. (2010). “Neuropsychiatric complications of Efavirenz therapy: suggestions for a new research paradigm”. The Journal of Neuropsychiatry and Clinical Neurosciences 22 (4): 361–369. doi:10.1176/jnp.2010.22.4.361.
External links

|
|
 |
|
| Systematic (IUPAC) name | |
|---|---|
|
(4S)-6-chloro-4-(2-cyclopropylethynyl)-4-(trifluoromethyl)-2,4-dihydro-1H-3,1-benzoxazin-2-one
|
|
| Clinical data | |
| Trade names | Sustiva, Stocrin, others |
| AHFS/Drugs.com | monograph |
| MedlinePlus | a699004 |
| Pregnancy category |
|
| Routes of administration |
By mouth (capsules, tablets) |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 40–45% (under fasting conditions) |
| Protein binding | 99.5–99.75% |
| Metabolism | Hepatic (CYP2A6 and CYP2B6-mediated) |
| Onset of action | 3–5 hours |
| Biological half-life | 40–55 hours |
| Excretion | Urine (14–34%) and feces (16–61%) |
| Identifiers | |
| CAS Number | 154598-52-4 |
| ATC code | J05AG03 (WHO) |
| PubChem | CID 64139 |
| DrugBank | DB00625 |
| ChemSpider | 57715 |
| UNII | JE6H2O27P8 |
| KEGG | D00896 |
| ChEBI | CHEBI:119486 |
| ChEMBL | CHEMBL223228 |
| NIAID ChemDB | 032934 |
| PDB ligand ID | EFZ (PDBe, RCSB PDB) |
| Chemical data | |
| Formula | C14H9ClF3NO2 |
| Molar mass | 315.675 g/mol |

Glenmark Gets USFDA Nod For Alcohol Abstinence Drug

VITAMINS, COMMON INFORMATION

A vitamin (US /ˈvaɪtəmɪn/ or UK /ˈvɪtəmɪn/) is an organic compound required by an organism as a vital nutrient in limited amounts. An organic chemical compound (or related set of compounds) is called a vitamin when it cannot be synthesized in sufficient quantities by an organism, and must be obtained from the diet. Thus, the term is conditional both on the circumstances and on the particular organism. For example, ascorbic acid (vitamin C) is a vitamin for humans, but not for most other animals, and biotin (vitamin H) and vitamin D are required in the human diet only in certain circumstances.
|
Vitamin A – discovered in 1913 What it does:
Foods that have vitamin A:
Deficiency problems:
|
 |
|
What it does:
Foods that have vitamin D:
Deficiency problems:
|
 |
|
What it does:
Foods that have vitamin E:
Deficiency problems:
|
 |
|
Vitamin K – made by bacteria in our intestines What it does:
Foods that have vitamin K:
Deficiency problems:
|
 |
……….
By convention, the term vitamin includes neither other essential nutrients, such as dietary minerals, essential fatty acids, or essential amino acids (which are needed in larger amounts than vitamins) nor the large number of other nutrients that promote health but are otherwise required less often. Thirteen vitamins are universally recognized at present.
Vitamins are classified by their biological and chemical activity, not their structure. Thus, each “vitamin” refers to a number of vitamer compounds that all show the biological activity associated with a particular vitamin. Such a set of chemicals is grouped under an alphabetized vitamin “generic descriptor” title, such as “vitamin A“, which includes the compounds retinal, retinol, and four known carotenoids. Vitamers by definition are convertible to the active form of the vitamin in the body, and are sometimes inter-convertible to one another, as well.
itamins have diverse biochemical functions. Some, such as vitamin D, have hormone-like functions as regulators of mineral metabolism, or regulators of cell and tissue growth and differentiation (such as some forms of vitamin A). Others function as antioxidants (e.g., vitamin E and sometimesvitamin C). The largest number of vitamins, the B complex vitamins, function as precursors for enzyme cofactors, that help enzymes in their work as catalysts in metabolism. In this role, vitamins may be tightly bound to enzymes as part of prosthetic groups: For example, biotin is part of enzymes involved in making fatty acids. They may also be less tightly bound to enzyme catalysts as coenzymes, detachable molecules that function to carry chemical groups or electrons between molecules. For example, folic acid may carry methyl, formyl, and methylene groups in the cell. Although these roles in assisting enzyme-substrate reactions are vitamins’ best-known function, the other vitamin functions are equally important.

Until the mid-1930s, when the first commercial yeast-extract vitamin B complex and semi-synthetic vitamin C supplement tablets were sold, vitamins were obtained solely through food intake, and changes in diet (which, for example, could occur during a particular growing season) usually greatly altered the types and amounts of vitamins ingested. However, vitamins have been produced as commodity chemicals and made widely available as inexpensive semisynthetic and synthetic-source multivitamin dietary and food supplements and additives, since the middle of the 20th century.,,,,,,,

List of vitamins
Each vitamin is typically used in multiple reactions, and, therefore, most have multiple functions.
| Vitamin generic
descriptor name |
Vitamerchemical name(s) (list not complete) | Solubility | Recommended dietary allowances
(male, age 19–70)[6] |
Deficiency disease | Upper Intake Level
(UL/day)[6] |
Overdose disease | Food sources |
|---|---|---|---|---|---|---|---|
| Vitamin A | Retinol, retinal, and
four carotenoids including beta carotene |
Fat | 900 µg | Night-blindness,Hyperkeratosis, andKeratomalacia[7] | 3,000 µg | Hypervitaminosis A | Orange, ripe yellow fruits, leafy vegetables, carrots, pumpkin, squash, spinach, liver, soy milk, milk |
| Vitamin B1 | Thiamine | Water | 1.2 mg | Beriberi, Wernicke-Korsakoff syndrome | N/D[8] | Drowsiness or muscle relaxation with large doses.[9] | Pork, oatmeal, brown rice, vegetables, potatoes, liver, eggs |
| Vitamin B2 | Riboflavin | Water | 1.3 mg | Ariboflavinosis | N/D | Dairy products, bananas, popcorn, green beans, asparagus | |
| Vitamin B3 | Niacin, niacinamide | Water | 16.0 mg | Pellagra | 35.0 mg | Liver damage (doses > 2g/day)[10] and other problems | Meat, fish, eggs, many vegetables, mushrooms, tree nuts |
| Vitamin B5 | Pantothenic acid | Water | 5.0 mg[11] | Paresthesia | N/D | Diarrhea; possibly nausea and heartburn.[12] | Meat, broccoli, avocados |
| Vitamin B6 | Pyridoxine,pyridoxamine,pyridoxal | Water | 1.3–1.7 mg | Anemia[13] peripheral neuropathy. | 100 mg | Impairment ofproprioception, nerve damage (doses > 100 mg/day) | Meat, vegetables, tree nuts, bananas |
| Vitamin B7 | Biotin | Water | 30.0 µg | Dermatitis, enteritis | N/D | Raw egg yolk, liver, peanuts, certain vegetables | |
| Vitamin B9 | Folic acid, folinic acid | Water | 400 µg | Megaloblastic anemiaand Deficiency during pregnancy is associated with birth defects, such as neural tube defects | 1,000 µg | May mask symptoms of vitamin B12 deficiency;other effects. | Leafy vegetables, pasta, bread, cereal, liver |
| Vitamin B12 | Cyanocobalamin,hydroxycobalamin,methylcobalamin | Water | 2.4 µg | Megaloblastic anemia[14] | N/D | Acne-like rash [causality is not conclusively established]. | Meat and other animal products |
| Vitamin C | Ascorbic acid | Water | 90.0 mg | Scurvy | 2,000 mg | Vitamin C megadosage | Many fruits and vegetables, liver |
| Vitamin D | Cholecalciferol | Fat | 10 µg[15] | Rickets andOsteomalacia | 50 µg | Hypervitaminosis D | Fish, eggs, liver, mushrooms |
| Vitamin E | Tocopherols,tocotrienols | Fat | 15.0 mg | Deficiency is very rare; mild hemolytic anemiain newborn infants.[16] | 1,000 mg | Increased congestive heart failure seen in one large randomized study.[17] | Many fruits and vegetables, nuts and seeds |
| Vitamin K | phylloquinone,menaquinones | Fat | 120 µg | Bleeding diathesis | N/D | Increases coagulation in patients taking warfarin.[18] | Leafy green vegetables such as spinach, egg yolks, liver |

Xofigo Injection Recommended for Approval in EU

Cl 223Ra Cl
is the structure
http://www.ama-assn.org/resources/doc/usan/radium-ra-223-dichloride.pdf check out yourself
Xofigo® (radium Ra 223 dichloride) Injection Recommended for Approval in the European Union
Oslo, Norway, 20 September 2013 – Algeta ASA (OSE: ALGETA), announced today that Bayer has received a positive opinion from the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) recommending approval of Xofigo® (radium Ra 223 dichloride) in Europe. The proposed indication is for the treatment of adults with castration-resistant prostate cancer, symptomatic bone metastases and no known visceral metastases. The decision of the European Commission (EC) on the approval is expected in the fourth quarter of 2013.
Xofigo® (radium Ra 223 dichloride) injection was approved by the US Food and Drug Administration (FDA) in May 2013 for the treatment of patients with CRPC, symptomatic bone metastases and no known visceral metastatic disease and is now available in the United States at licensed facilities. read all at
http://www.pharmalive.com/xofigo-injection-recommended-for-approval-in-eu
old article
FDA Approves Xofigo for Advanced Prostate Cancer
May 15, 2013 — The U.S. Food and Drug Administration today approved Xofigo (radium Ra 223 dichloride) to treat men with symptomatic late-stage (metastatic) castration-resistant prostate cancer that has spread to bones but not to other organs. It is intended for men whose cancer has spread after receiving medical or surgical therapy to lower testosterone.
Prostate cancer forms in a gland in the male reproductive system found below the bladder and in front of the rectum. The male sex hormone testosterone stimulates the prostate tumors to grow. According to the National Cancer Institute, an estimated 238,590 men will be diagnosed with prostate cancer and 29,720 will die from the disease in 2013.
Xofigo is being approved more than three months ahead of the product’s prescription drug user fee goal date of Aug. 14, 2013, the date the agency was scheduled to complete review of the drug application. The FDA reviewed Xofigo under the agency’s priority review program, which provides for an expedited review of drugs that appear to provide safe and effective therapy when no satisfactory alternative therapy exists, or offer significant improvement compared to marketed products.
“Xofigo binds with minerals in the bone to deliver radiation directly to bone tumors, limiting the damage to the surrounding normal tissues,” said Richard Pazdur, M.D., director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research. “Xofigo is the second prostate cancer drug approved by the FDA in the past year that demonstrates an ability to extend the survival of men with metastatic prostate cancer.”
In August 2012, the FDA approved Xtandi to treat men with metastatic castration-resistant prostate cancer that has spread or recurred, even with medical or surgical therapy to minimize testosterone. Xtandi is approved for patients who have previously been treated the chemotherapy drug docetaxel.
Xofigo’s safety and effectiveness were evaluated in a single clinical trial of 809 men with symptomatic castration-resistant prostate cancer that spread to bones but not to other organs. Patients were randomly assigned to receive Xofigo or a placebo plus best standard of care.
The study was designed to measure overall survival. Results from a pre-planned interim analysis showed men receiving Xofigo lived a median of 14 months compared to a median of 11.2 months for men receiving placebo. An exploratory updated analysis conducted later in the trial confirmed Xofigo’s ability to extend overall survival.
The most common side effects reported during clinical trials in men receiving Xofigo were nausea, diarrhea, vomiting and swelling of the leg, ankle or foot. The most common abnormalities detected during blood testing included low levels of red blood cells (anemia), lymphocytes (lymphocytopenia), white blood cells (leukopenia), platelets (thrombocytopenia) and infection-fighting white blood cells (neutropenia).
Xofigo is marketed by Wayne, N.J.-based Bayer Pharmaceuticals. Xtandi is co-marketed by Astellas Pharma U.S., Inc. of Northbrook, Ill., and Medivation, Inc. of San Francisco, Calif.
GLENMARK- ELOVERA , for dry skin disorders

Compositions:
Elovera extract 10% cream, Vitamin E 0.5%
Category–Locally Acting Skin Preparations
Description
* Aqueeze adequate amount of elovera moisturizing body wash onto wet hands or wet loran and work into a creamy lather. apply it all ovr the body, keep for some time and then rinse with water.
| Products Name : | Elovera Moisturizing Body Wash 150ml – (Glenmark) |
Elovera Cream, manufacture by Glenmark pharmaceuticals limited , is cream enriched with vitamin E and Aloe Vera. It’s a very special cream specially for treating scars and other minor pimple spots on the face.
reviews from net
My skin is very much oily hence I get these ugly Pimples very profoundly. On top of it i have the habit of bursting out the puss from these pimples. I always play it with my hands and as a result forms some very ugly scars on my face which are visible from distant away.Though I am bit dark with my completion ,even then It’s clearly visible and my mother scolds me like hell for bursting the pimples out.Honestly I just can’t stop my hands reaching out for them no matter how busy I am so Finally has to resort to some ointments to reduce the visibility of the scars.
I did try few popular products but were of no use basically. The spots didn’t get reduced but instead effected the completion of my face.Finally my mother came to my rescue. She had hear about this Elovera Cream from some one and bought home one for me.Initially i was a bit skeptic but finally I thought of trying it. For the first few days it had no effect what-so-ever , but slowly it started clearing the skin blemishes. My skin started showing it’s effects and the scars became less visible. Not only does it clear the scars but it helped me to fight the ugly pimples as well.
My face became much more glowing and healthy and i use the cream regularly even now.It’s really a magical product and should try it for clearing the blemishes and other skin problem.
Genzyme’s multiple sclerosis treatment approved by European Commission

Alemtuzumab
Sanofi and its subsidiary Genzyme have been given marketing approval by the European Commission for Lemtrada (alemtuzumab), a treatment for multiple sclerosis. read all at
click on title below
Genzyme’s multiple sclerosis treatment approved by European Commission
The first generic version of the oral chemotherapy drug Xeloda (capecitabine) has been approved by the U.S. Food and Drug Administration to treat cancers of the colon/rectum or breast,
capecitabine
- R-340, Ro-09-1978, Xeloda
pentyl [1-(3,4-dihydroxy-5-methyltetrahydrofuran-2-yl)-5-fluoro-2-oxo-1H-pyrimidin-4-yl]carbamate

MONDAY Sept. 16, 2013 — The first generic version of the oral chemotherapy drug Xeloda (capecitabine) has been approved by the U.S. Food and Drug Administration to treat cancers of the colon/rectum or breast, the agency said Monday in a news release.
This year, an estimated 142,820 people will be diagnosed with cancer of the colon/rectum, and 50,830 are predicted to die from the disease, the FDA said, citing the U.S. National Cancer Institute. An estimated 232,340 women will be diagnosed with cancer of the breast this year, and some 39,620 will die from it.
The most common side effects of the drug are diarrhea, vomiting; pain, redness, swelling or sores in the mouth; fever and infection, the FDA said.
The agency stressed that approved generics have the same high quality and strength as their brand-name counterparts.
License to produce the generic drug was given to Israel-based Teva Pharmaceuticals. The brand name drug is produced by the Swiss pharma firm Roche.
Capecitabine (INN) /keɪpˈsaɪtəbiːn/ (Xeloda, Roche) is an orally-administered chemotherapeutic agent used in the treatment of metastatic breast and colorectal cancers. Capecitabine is a prodrug, that is enzymatically converted to 5-fluorouracil in the tumor, where it inhibits DNA synthesis and slows growth of tumor tissue. The activation of capecitabine follows a pathway with three enzymatic steps and two intermediary metabolites, 5′-deoxy-5-fluorocytidine (5′-DFCR) and 5′-deoxy-5-fluorouridine (5′-DFUR), to form 5-fluorouracil

Indications
Capecitabine is FDA-approved for:
- Adjuvant in colorectal cancer Stage III Dukes’ C – used as first-line monotherapy.
- Metastatic colorectal cancer – used as first-line monotherapy, if appropriate.
- Metastatic breast cancer – used in combination with docetaxel, after failure of anthracycline-based treatment. Also as monotherapy, if the patient has failed paclitaxel-based treatment, and if anthracycline-based treatment has either failed or cannot be continued for other reasons (i.e., the patient has already received the maximum lifetime dose of an anthracycline).
In the UK, capecitabine is approved by the National Institute for Health and Clinical Excellence (NICE) for colon and colorectal cancer, and locally advanced or metastatic breast cancer.[1] On March 29, 2007, the European Commission approved Capecitabine, in combination with platinum-based therapy (with or without epirubicin), for the first-line treatment of advanced stomach cancer.
Capecitabine is a cancer chemotherapeutic agent that interferes with the growth of cancer cells and slows their distribution in the body. Capecitabine is used to treat breast cancer and colon or rectum cancer that has spread to other parts of the body.
Formulation
Capecitabine (as brand-name Xeloda) is available in light peach 150 mg tablets and peach 500 mg tablets.
- Lacy, Charles F; Armstrong, Lora L; Goldman, Morton P; Lance, Leonard L (2004). Lexi-Comp’s Drug Information Handbook (12th Edition). Lexi-Comp Inc. ISBN 1-59195-083-X
- Fischer, David S; Knobf, M Tish; Durivage, Henry J; Beaulieu, Nancy J (2003). The Cancer Chemotherapy Handbook (6th Edition). Mosby. ISBN 0-323-01890-4
- Thomson Centerwatch: Drugs Approved by the FDA (Xeloda) Retrieved 6/05
- Mercier C, Ciccolini J (2007). “Severe or lethal toxicities upon capecitabine intake: is DPYD genetic polymorphism the ideal culprit?”. Trends in pharmacological sciences 28 (12): 597–598. doi:10.1016/j.tips.2007.09.009. PMID 18001850.
- “Subtopics”. Nice.org.uk. Retrieved 2012-08-15.
- Fingerprints May Vanish With Cancer Drug – US News and World Report
- Cancer Drug Erases Man’s Fingerprints – CNN
- “Stritch School of Medicine”. Stritch.luc.edu. Retrieved 2012-08-15.
- Xeloda.com (patient information, tools, and resources)
- OralChemo Advisor (patient information)

Capecitabine is an orally-administered anticancer agent widely used in the treatment of metastatic breast and colorectal cancers. Capecitabine is a ribofuranose-based nucleoside, and has the sterochemical structure of a ribofuranose having an β-oriented 5-fluorocytosine moiety at C-I position.
US Patent Nos. 5,472,949 and 5,453,497 disclose a method for preparing capecitabine by glycosylating tri-O-acetyl-5-deoxy-β-D-ribofuranose of formula I using 5-fluorocytosine to obtain cytidine of formula II; and carbamoylating and hydrolyzing the resulting compound, as shown in Reaction Scheme 1 :
Reaction Scheme 1
1
The compound of formula I employed as an intermediate in Reaction
Scheme 1 is the isomer having a β-oriented acetyl group at the 1 -position, for the reason that 5-fluorocytosine is more reactive toward the β-isomer than the α-isomer in the glycosylation reaction due to the occurrence of a significant neighboring group participation effect which takes place when the protecting group of the 2-hydroxy group is acyl.
Accordingly, β-oriented tri-O-acetyl-5-deoxy-β-D-ribofuranose (formula
I) has been regarded in the conventional art to the essential intermediate for the preparation of capecitabine. However, such a reaction gives a mixture of β- and α-isomers from which cytidine (formula II) must be isolated by an uneconomical step.
Meanwhile, US Patent No. 4,340,729 teaches a method for obtaining capecitabine by the procedure shown in Reaction Scheme 2, which comprises hydrolyzing 1-methyl-acetonide of formula III to obtain a triol of formula IV; acetylating the compound of formula IV using anhydrous acetic anhydride in pyridine to obtain a β-/α-anomeric mixture of tri-O-acetyl-5-deoxy-D-ribofuranose of formula V; conducting vacuum distillation to purify the β-/α-anomeric mixture; and isolating the β-anomer of formula I therefrom:
Reaction Scheme 2
III IV
However, the above method is also hampered by the requirement to perform an uneconomical and complicated recrystallization steps for isolating the β-anomer from the mixture of β-/α-anomers of formula V, which leads to a low yield of only about 35% to 40% (Guangyi Wang et al., J. Med. Chem., 2000, vol. 43, 2566-2574; Pothukuchi Sairam et al., Carbohydrate Research, 2003, vol. 338, 303-306; Xiangshu Fei et al., Nuclear Medicine and Biology, 2004, vol. 31, 1033-1041; and Henry M. Kissman et al., J. Am. Chem. Soc, 1957, vol. 79, 5534-5540).
Further, US Patent No. 5,476,932 discloses a method for preparing capecitabine by subjecting 5′-deoxy-5-fluorocytidine of formula VI to a reaction with pentylchloroformate to obtain the compound of formula VII having the amino group and the 2-,3-hydroxy groups protected with C5Hi1CO2 groups; and removing the hydroxy-protecting groups from the resulting compound, as shown in Reaction Scheme 3 :
Reaction Scheme 3
Vl VII 1
However, this method suffers from a high manufacturing cost and also requires several complicated steps for preparing the 5′-deoxy-5-fluorocytidine of formula VI: protecting the 2-,3-hydroxy groups; conducting a reaction thereof with 5-fluorocytosine; and deprotecting the 2-,3-hydroxy groups.
Accordingly, the present inventors have endeavored to develop an efficient method for preparing capecitabine, and have unexpectedly found an efficient, novel method for preparing highly pure capecitabine using a trialkyl carbonate intermediate, which does not require the uneconomical β-anomer isolation steps.
synthesis

more info and description
Aspects of the present invention relate to capecitabine and processes for the preparation thereof.
The drug compound having the adopted name “capecitabine” has a chemical name 5′-deoxy-5-fluoro-N-[(pentyloxy) carbonyl] cytidine and has structural formula I.
H
OH OH I
This compound is a fluoropyrimidine carbamate with antineoplastic activity. The commercial product XELODA™ tablets from Roche Pharmaceuticals contains either 150 or 500 mg of capecitabine as the active ingredient.
U.S. Patent No. 4,966,891 describes capecitabine generically and a process for the preparation thereof. It also describes pharmaceutical compositions, and methods of treating of sarcoma and fibrosarcoma. This patent also discloses the use of ethyl acetate for recrystallization of capecitabine. The overall process is summarized in Scheme I.
Scheme I
U.S. Patent No. 5,453,497 discloses a process for producing capecitabine that comprises: coupling of th-O-acetyl-5-deoxy-β-D-hbofuranose with 5- fluorocytosine to obtain 2′,3′-di-O-acetyl-5′-deoxy-5-fluorocytidine; acylating a 2′, 3′- di-O-acetyl-5′-deoxy-5-fluorocytidine with n-pentyl chloroformate to form 5′-deoxy- 2′,3′-di-O-alkylcarbonyl-5-fluoro-N-alkyloxycarbonyl cytidine, and deacylating the 2′ and 3′ positions of the carbohydrate moiety to form capecitabine. The overall process is summarized in Scheme II.
Capecitabine
Scheme Il
The preparation of capecitabine is also disclosed by N. Shimma et al., “The Design and Synthesis of a New Tumor-Selective Fluoropyrimidine Carbamate, Capecitabine,” Bioorganic & Medicinal Chemistry, Vol. 8, pp. 1697-1706 (2000). U.S. Patent No. 7,365,188 discloses a process for the production of capecitabine, comprising reacting 5-fluorocytosine with a first silylating agent in the presence of an acid catalyst under conditions sufficient to produce a first silylated compound; reacting the first silylated compound with 2,3-diprotected-5- deoxy-furanoside to produce a coupled product; reacting the coupled product with a second silylating agent to produce a second silylated product; acylating the second silylated product to produce an acylated product; and selectively removing the silyl moiety and hydroxyl protecting groups to produce capecitabine. The overall process is summarized in Scheme III. te
R: hydrocarbyl
Scheme III
Further, this patent discloses crystallization of capecitabine, using a solvent mixture of ethyl acetate and n-heptane. International Application Publication No. WO 2005/080351 A1 describes a process for the preparation of capecitabine that involves the refluxing N4– pentyloxycarbonyl-5-fluorocytosine with trimethylsiloxane, hexamethyl disilazanyl, or sodium iodide with trimethyl chlorosilane in anhydrous acetonitrile, dichloromethane, or toluene, and 5-deoxy-1 ,2,3-tri-O-acetyl-D-ribofuranose, followed by hydrolysis using ammonia/methanol to give capecitabine. The overall process is summarized in Scheme IV.
Scheme IV
International Application Publication No. WO 2007/009303 A1 discloses a method of synthesis for capecitabine, comprising reacting 5′-deoxy-5- fluorocytidine using double (trichloromethyl) carbonate in an inert organic solvent and organic alkali to introduce a protective lactone ring to the hydroxyl of the saccharide moiety; reacting the obtained compound with chloroformate in organic alkali; followed by selective hydrolysis of the sugar component hydrolytic group using an inorganic base to give capecitabine. The overall process is summarized in Scheme V.
Scheme V
Even though all the above documents collectively disclose various processes for the preparation of capecitabine, removal of process-related impurities in the final product has not been adequately addressed. Impurities in any active pharmaceutical ingredient (API) are undesirable, and, in extreme cases, might even be harmful to a patient. Furthermore, the existence of undesired as well as unknown impurities reduces the bioavailability of the API in pharmaceutical products and often decreases the stability and shelf life of a pharmaceutical dosage form.
nmr
1H NMR(CD3OD) δ 0.91(3H5 t), 1.36~1.40(4H, m), 1.41(3H, d), 1.68~1.73(2H, m), 3.72(1H, dd), 4.08(1H, dd), 4.13~4.21(3H, m), 5.7O(1H, s), 7.96(1H, d)

- The acetylation of 5′-deoxy-5-fluorocytidine (I) with acetic anhydride in dry pyridine gives 2′,3′-di-O-acetyl-5′-deoxy-5-fluorocytidine (II), which is condensed with pentyl chloroformate (III) by means of pyridine in dichromethane yielding 2′,3′-di-O-acetyl-5′-deoxy-5-fluoro-N4-(pentyloxycarbonyl)cytidine (IV). Finally, this compound is deacetylated with NaOH in dichloromethane/water. The diacetylated cytidine (II) can also be obtained by condensation of 5-fluorocytosine (V) with 1,2,3-tri-O-acetyl-5-deoxy-beta-D-ribofuranose (VI) by means of trimethylchlorosilane in acetonitrile or HMDS and SnCl4 in dichloromethane..
-
- EP 602454, JP 94211891, US 5472949.
- Capecitabine. Drugs Fut 1996, 21, 4, 358,
- Bioorg Med Chem Lett2000,8,(7):1697,
- Capecitabine. Drugs Fut 1996, 21, 4, 358,
- EP 602454, JP 94211891, US 5472949.
FDA Approves Botox Cosmetic to Improve the Appearance of Crow’s Feet Lines
WEDNESDAY, September 11, 2013 — The U.S. Food and Drug Administration today approved a new use for Botox Cosmetic (onabotulinumtoxinA) for the temporary improvement in the appearance of moderate to severe lateral canthal lines, known as crow’s feet, in adults. Botox Cosmetic is the only FDA approved drug treatment option for lateral canthal lines.
The FDA approved Botox Cosmetic in 2002 for the temporary improvement of glabellar lines (wrinkles between the eyebrows, known as frown lines), in adults. Botox Cosmetic works by keeping muscles from tightening so wrinkles are less prominent
READ ALL AT

BOTOX Cosmetic (onabotulinum toxin A) For Injection, is a sterile, vacuum-dried purifiedbotulinum toxin type A, produced from fermentation of Hall strain Clostridium botulinumtype A grown in a medium containing casein hydrolysate, glucose, and yeast extract, intended for intramuscular use. It is purified from the culture solution by dialysis and a series of acid precipitations to a complex consisting of the neurotoxin, and several accessory proteins. The complex is dissolved in sterile sodium chloride solution containing Albumin Human and is sterile filtered (0.2 microns) prior to filling and vacuum-drying.
The primary release procedure for BOTOX Cosmetic uses a cell-based potency assay to determine the potency relative to a reference standard. The assay is specific to Allergan’s products BOTOX and BOTOX Cosmetic. One Unit of BOTOX Cosmetic corresponds to the calculated median intraperitoneal lethal dose (LD50) in mice. Due to specific details of this assay such as the vehicle, dilution scheme and laboratory protocols, Units of biological activity of BOTOX Cosmetic cannot be compared to nor converted into Units of any other botulinum toxin or any toxin assessed with any other specific assay method. The specific activity of BOTOX Cosmetic is approximately 20 Units/nanogram of neurotoxin protein complex.
Each vial of BOTOX Cosmetic contains either 50 Units of Clostridium botulinum type A neurotoxin complex, 0.25 mg of Albumin Human, and 0.45 mg of sodium chloride; or 100 Units of Clostridium botulinum type A neurotoxin complex, 0.5 mg of Albumin Human, and 0.9 mg of sodium chloride in a sterile, vacuum-dried form without a preservative.
Since the approval of BOTOX® Cosmetic by the U.S. Food and Drug Administration in 2002, Allergan has virtually changed the face of medical aesthetics. Men and women between the ages of 18 to 65 now have the ability to choose science-based, non-invasive medical aesthetic solutions, including BOTOX® Cosmetic and the JUVÉDERM® family of dermal fillers, to achieve their own results. Over the last seven years, there have been nearly 11.8 million BOTOX® Cosmetic treatments recorded in the United States.1 More importantly, its 97 percent satisfaction rating (survey of 117 patients)2,3 is just one indication of the trust consumers have placed in Allergan.
BOTOX® Cosmetic is a simple, non-surgical procedure for temporarily reducing the appearance of moderate to severe glabellar lines – the vertical frown lines between the eyebrows that look like an “11” – in adult women and men aged 18 to 65. BOTOX® Cosmetic reduces the activity of the muscles that cause the “11s” to form by blocking nerve impulses that trigger wrinkle-causing muscle contractions, creating an improved appearance between the brows. Results can last up to four months and may vary with each patient. Ask your doctor if BOTOX® Cosmetic is right for you.

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....











































