Home » Posts tagged 'GENERIC DRUG' (Page 4)
Tag Archives: GENERIC DRUG
Characterization of the “hygroscopic” properties of active pharmaceutical ingredients
Characterization of the “hygroscopic” properties of active pharmaceutical ingredients.
Source
SSCI, Inc., West Lafayette, IN, USA. ann.newman@aptuit.com
http://www.ncbi.nlm.nih.gov/pubmed/17630643
Abstract
The amount of water vapor taken up by an active pharmaceutical ingredient (API) as a function of relative humidity is routinely evaluated to characterize and monitor its “hygroscopicity” throughout the drug development process. In this minireview we address the necessity of going beyond the measurement of water vapor sorption isotherms to establish the various mechanisms by which solids interact with water and the important role played by the crystalline or amorphous form of the solid. Practical approaches for choosing experimental conditions under which water vapor sorption should be measured, including the pre-treatment of samples and the time allowed to reach an equilibrium state are presented. With the assistance of a flowchart, we provide a basis for the systematic examination of samples to establish the likely mechanisms of sorption and the indicators pointing toward future problems with physical and chemical instabilities. Finally, we present strategies for managing materials that might be susceptible to the detrimental effects of water vapor sorption.
(Copyright) 2008 Wiley-Liss, Inc.
FINASTERIDE

(5α, 17β)-N-(1 ,1-dimethylethyl)-3-oxo-4-aza-androst-1-ene-17-carboxamide, finasteride, a 4-aza-steroid compound 5 which exhibits pharmaceutical activity as an inhibitor of the enzyme testosterone 5-α-reductase, and is useful in the treatment of prostate cancer
Finasteride;YM-152;MK-906;Prodel;Propecia;
Chibro-Proscar;Finastid;Prostide;Andozac;Proscar
Finasteride (brand names Proscar and Propecia by Merck, among other generic names) is a synthetic drug for the treatment of benign prostatic hyperplasia (BPH) and male pattern baldness (MPB). It is a type II 5α-reductase inhibitor. 5α-reductase is an enzymethat converts testosterone to dihydrotestosterone (DHT).

Figure . Conversion of testosterone to dihydrotestosterone.
Chemical synthesis
Propecia 1 mg & Finpecia 1 mg tablets
Finasteride is synthesized fromprogesterone:

History
In 1974, Julianne Imperato-McGinley of Cornell Medical College in New York attended a conference on birth defects. She reported on a group of intersex children in the Caribbean who appeared sexually ambiguous at birth, and were initially raised as girls, but then grew external male genitalia and other masculine characteristic post-onset of puberty. Her research group found that these children shared agenetic mutation, causing deficiency of the 5α-reductase enzyme and male hormone dihydrotestosterone (DHT), which was found to have been the etiology behind abnormalities in male sexual development. Upon maturation, these individuals were observed to have smaller prostates which were underdeveloped, and were also observed to lack incidence of male pattern baldness.
In 1975, copies of Imperato-McGinley’s presentation were seen by P. Roy Vagelos, who was then serving as Merck’s basic-research chief. He was intrigued by the notion that decreased levels of DHT led to the development of smaller prostates. Dr. Vagelos then sought to create a drug which could mimic the condition found in these children in order to treat older men who were suffering from benign prostatic hyperplasia.
In 1992, finasteride (5 mg) was approved by the U.S. Food and Drug Administration (FDA) for treatment of benign prostatic hyperplasia(BPH), which Merck marketed under the brand name Proscar.
In 1997, Merck was successful in obtaining FDA approval for a second indication of finasteride (1 mg) for treatment of male pattern baldness (MPB), which was marketed under the brand name Propecia.
CHEMISTRY
Formerly known as MK-906, finasteride (Figure 1) ([5-![]() , 17-
, 17-![]() -N-(1,1-dimethylethyl) -3-oxo-4-azaandrost- 1-ene-17-carboxamide) belongs to the 4-azasteroid structural class of compounds. (Click on the structure to the right to view a Chime rotatable structure.) Its synthesis, shown in Scheme 1, was published by Rasmusson et al. in 1986.[2] Briefly, beginning with a previously synthesized intermediate, the A-ring of the steroid skeleton was converted from its 3- keto precursor (1) to the required 4-aza system (3) through an open analog (2). Saturation of the B-ring using catalytic hydrogenation gave intermediate 4. Use of the 2-pyridyl thio ester (5) gave a reactive substrate to form the tertiary butyl carboxamide (6). The final step in the synthesis, dehydration of the A-ring with benzeneselenic anhydride, gave the final product, finasteride (7).
-N-(1,1-dimethylethyl) -3-oxo-4-azaandrost- 1-ene-17-carboxamide) belongs to the 4-azasteroid structural class of compounds. (Click on the structure to the right to view a Chime rotatable structure.) Its synthesis, shown in Scheme 1, was published by Rasmusson et al. in 1986.[2] Briefly, beginning with a previously synthesized intermediate, the A-ring of the steroid skeleton was converted from its 3- keto precursor (1) to the required 4-aza system (3) through an open analog (2). Saturation of the B-ring using catalytic hydrogenation gave intermediate 4. Use of the 2-pyridyl thio ester (5) gave a reactive substrate to form the tertiary butyl carboxamide (6). The final step in the synthesis, dehydration of the A-ring with benzeneselenic anhydride, gave the final product, finasteride (7).

Scheme 1. Key intermediates in the synthesis of finasteride by Rasmusson et al. Reagents: a, KMnO4-NaIO4, t-BuOH, reflux; b, NH3, heat; c, H2, Pt, ArOH; d, 2,2′-dipyridyl disulfide, triphenylphosphine, toluene; e, t-butyl amine, THF; f, benzeneselenic anhydride, chlorobenzene.
The preparation of finasteride is described and claimed in U.S. Patent 4.377.584 and further described in U.S. Patent 4.760.071. Other patents which pertain to the preparation of finasteride include Canadian patent application 2.029.859: U.S. patents 5.084.574 and 5.116.983: and Canadian patent applications 2.049.882 and 2.049.881. All these teach the conversion of a final intermediate tofinasteride, which is purified and isolated as a crystalline solid. Althoughfinasteride polymorphs are not mentioned specifically in these items of prior art, the finasteride obtained using them, as a crystalline solid, must be in one or other of the known polymorphic forms, or a mixture of both of them.
Aforementioned Canadian Patent Application 2.103.107 Dolling et a published May 20, 1994, describes preparations of finasteride and the specific polymorphic Form I and Form II thereof. In particular, it teaches that polymorphic Form I can be prepared by crystallization from a mixture of finasteride in an organic solvent and optionally water, such that the amount of organic solvent and water in the mixture is sufficient to cause the solubility of the non-solvated form of finasteride(Form I) to be exceeded and the non- solvated form of finasteride to be less soluble than any other form of finasteride in the mixture. It also teaches that the polymorphic Form I of finasteride can be prepared by heating the polymorphic Form II of finasteride to at least 25°C in water or an organic solvent for a sufficient period of time to effect the conversion. The same reference teaches that polymorphic Form II finasteride can be prepared by crystallization from a mixture of finasteride in an organic solvent and water, such that the amount of organic solvent and water in the mixture is sufficient to cause the solubility of the solvated form of finasteride to be exceeded and the solvated form of finasteride to be less soluble than any other form of finasteride in the mixture, followed by recovery of the solid and removal of the solvent therefrom; or by heating polymorphic Form I finasteride to at least to about 150°C for sufficient time to complete the conversion.
MORE INFO
-
Finasteride, marketed under the tradename of PROSCAR®, by Merck & Co., Inc is 17β-(N-tert-butyl carbamoyl)-4-aza-5α-androst-1-en-3-one and is a 5α-reductase inhibitor for use in treating acne, female hirsutism, and particularly benign prostatic hyperplasia. See US Patent 4,760,071 (1988), the entire disclosure of which is incorporated herein by reference..
-
[0002]The synthesis of finasteride in US Patent 4,760,071 involves reacting the 17β-(2-pyridylthio) carboxylate of 4-aza-5α-androst-1-ene-3-one with t-butylamine. A further synthesis of finasteride is described in Synthetic Communications, 30 (17), p. 2683-2690 (1990). including the reacting of the 17-acylimidazole of 4-aza-5α-androst-1-en-3-one with t-butylamine.
-
[0003]However, both of these reactions require the use of heterocyclic aromatic amines which are expensive and give rise to environmental safety and toxicity considerations. Both of these intermediates are prepared from the 17β-carboxylic acid.
-
[0004]The Bodroux reaction, described by F. Bodroux in the references, Bull. Soc. Chim. France 33, 831 ( 1905); 35, 519 (1906); 1, 912 (1907); Compt. Rend. 138, 1427 (1904); 140, 1108 (1905); 142, 401 (1906) discloses the reaction of the magnesium halide salts of amines with esters. However, there is no description or teaching that the reaction can be applied to the reaction of a sterically hindered amine, e.g. t-butyl amine, with a sterically hindered ester such as 1.

The first method (International Patent: W0200507M97A) is finasteridedihydro as raw materials, benzeneseleninic anhydride synthesis of finasteride, the reaction is as follows:
Used in this reaction toxic and expensive reagents benzeneseleninic anhydride, yield only about 50%, and the product to column chromatography to separate, while the use of certain toxic chlorobenzene as solvent, the cost is very high , environmental hazards large.
[0005] The second method (U.S. Patent No.: US20070167477A1) is finasteridedihydro as raw materials, the use of DDQ / BSTFA (i.e. 3,3 – dichloro-5 ,6 – dicyano-p-benzoquinone / second (third trimethylsilyl) trifluoroacetamide) Oxidation get finasteride, the reaction is as follows:
The reaction yield about 65%, the resulting fluorine-containing wastewater intractable, quinones great harm to the environment.
[0006] The third method (international patent: W02008101308A) is dihydrofinasteride as raw material, the use of phenyl sulfide oxidation get finasteride, the reaction is as follows:
The method steps and more complicated to operate, the total yield of only 60%, the use of expensive lithium diisopropylamide, lithium bis trimethylsilyl test Qi IJ, the cost is higher.
REF
Rasmusson, G.H.; Reynolds, G.F. (Merck & Co., Inc.); 17beta-Substd.-4-aza-5alpha-androstenones and their use as 5alpha-reductase inhibitors. AU 8539167; EP 0155096; EP 0314199; ES 8702430; JP 1985222497; JP 1989093600; US 4760071 .
Rasmusson, G.H.; Reynolds, G.F. (Merck & Co., Inc.); Treatment of prostatic carcinoma with 17beta-N-monosubstd.-carbamoyl-4-aza-5-alpha-androst-1-en-3-ones. EP 0285383 .
Rasmusson, G.H.; Reynolds, G.F.; Steinberg, N.G.; Walton, E.; Patel, G.F.; Liang, T.; Cascieri, M.A.; Cheung, A.H.; Brooks, J.R.; Berman, C.; Azasteroids: structure-activity relationships for inhibition of 5 alpha-reductase and of androgen receptor binding. J Med Chem 1986, 29, 11, 2298.
Castaner, J.; Prous, J.; Finasteride. Drugs Fut 1991, 16, 11, 996.
The oxidative cleavage of N-tert-butyl-3-oxo-5alpha-androst-4-ene-17beta-carboxamide (I) with NaIO4 and KMnO4 in tert butanol – aqueous Na2CO3 gives the seco-ketoacid (II), which is cyclized with liquid ammonia in ethylene glycol at 180 C to afford the DELTA5-azasteroid (III). Hydrogenation of (III) with H2 over PtO2 in acetic acid yields the corresponding saturated aza-steroid (IV), which is finally dehydrogenated with benzeneseleninic anhydride in refluxing chlorobenzene or with 2,3-dichloro-5,6-dicyanobenzoquinone (DDQ) and bis(trimethylsilyl)trifluoroacetamide (BSTFA) in refluxing dioxane.
Teva: FDA Approves of Generic Tobi in US
TOBRAMYCIN
JERUSALEM–(BUSINESS WIRE)–Oct. 14, 2013– Teva Pharmaceutical Industries Ltd. announces today that the U.S. Food and Drug Administration has granted approval of the generic equivalent to Tobi® (Tobramycin Inhalation Solution USP) in the United States. Pursuant to an agreement with Novartis on this product, Teva expects to launch this product in late November. Marketed by Novartis, Tobi had annual sales of approximately $350 million in the United States, according to IMS data as of June 30, 2013. READ ALL AT………..
http://www.drugs.com/news/teva-fda-approves-tobi-us-48168.html

Tobramycin is an aminoglycoside antibiotic derived from Streptomyces tenebrarius and used to treat various types of bacteria infections, particularly Gram-negative infections. It is especially effective against species of Pseudomonas.[1]

Tobramycin works by binding to a site on the bacterial 30S and 50S ribosome, preventing formation of the 70S complex. As a result, mRNA cannot be translated into protein and cell death ensues. Tobramycin is preferred over gentamicin for Pseudomonas aeruginosapneumonia due to better lung penetration.

Like all aminoglycosides, tobramycin does not pass the gastro-intestinal tract, so forsystemic use it can only be given intravenously or intramuscularly. Ophthalmic (tobramycin only, Tobrex, or combined with dexamethasone, sold as TobraDex) and nebulised formulations both have low systemic absorption. The formulation for injection is branded Nebcin. The nebulised formulation (brand name Tobi) is indicated in the treatment of exacerbations of chronic infection with Pseudomonas aeruginosa in patients diagnosed with cystic fibrosis. A proprietary formulation of micronized, nebulized tobramycin has been tested as a treatment for bacterial sinusitis.[2] Tobrex is a 0.3% tobramycin sterile ophthalmic solution is produced by Bausch & Lomb Pharmaceuticals. Benzalkonium chloride 0.01% is added as a preservative. It is available by prescription only in the United States and Canada. In certain countries, such as Italy, it is available over the counter. Tobrex and TobraDex are indicated in the treatment of superficial infections of the eye, such as bacterial conjunctivitis. Tobramycin (injection) is also indicated for various severe or life-threatening gram-negative infections : meningitis in neonates, brucellosis, pelvic inflammatory disease, Yersinia pestis infection (plague).

Like other aminoglycosides, tobramycin is ototoxic: it can cause hearing loss, or a loss ofequilibrioception, or both in genetically susceptible individuals. These individuals carry a normally harmless genetic mutation that allows aminoglycosides such as tobramycin to affect cochlear cells. Aminoglycoside-induced ototoxicity is generally irreversible.

As with all aminoglycosides, tobramycin is also nephrotoxic, meaning it is toxic to thekidneys. This effect can be particularly worrisome when multiple doses accumulate over the course of a treatment or when the kidney concentrates urine by increasing tubular reabsorption during sleep. Adequate hydration may help prevent excess nephrotoxicity and subsequent loss of renal function. For these reasons parenteral tobramycin needs to be carefully dosed by body weight, and its serum concentration monitored. Tobramycin is thus said to be a drug with a narrow therapeutic index.
|
Mass-spectrum of tobramycin |
|
|
- “Tobramycin” (pdf). Toku-E. 2010-01-12. Retrieved 2012-06-11.
- “Nebulized Tobramycin in treating bacterial Sinusitis” (Press release). July 22, 2008. Retrieved 2009-12-06.

A widely accepted therapy for treating respiratory infections caused by Gram- negative bacteria involves intravenous administration of a single antibiotic or combinations of antibiotics. Gibson et al., 2003 Am. J. Respir. Crit. Care. Med.
168(8):918-951 ; Ramsey, 1996 N. Engl. J. Med. 335(3): 179-188. However, this method of treatment has several significant limitations including: (1 ) narrow spectrum of activity of existing antibiotics, (2) insufficient concentrations of antibiotic reaching the respiratory tract to ensure rapid onset and high rates of bacterial killing, and (3) development of adverse side effects due to high systemic concentrations of drug.
Aerosol administration of antibiotics (Conway, 2005 Chronic Repir. Dis. 2:35- 41 ; O’Riordan, 2000 Respir. Care 45(7):836-845) addresses several of the limitations of parenteral administration (Flume and Klepser, 2002 Pharmacotherapy 22(3 Pt 2):71 S-79S; Kuhn, 2001 Chest. 120:94S-98S). It enables topical delivery of high concentrations of drug to the endobronchial spaces and reduces side effects by lowering systemic exposure to antibiotic. However, patients having chronic respiratory conditions, such as chronic obstructive pulmonary disease, cystic fibrosis and bronchiestasis, may receive prolonged and repeated antibiotic therapies over the entire duration of their adult lives. Gibson et al., 2003 Am. J. Respir. Crit. Care. Med. 168(8):918-951 ; Ramsey, 1996 N. Engi. J. Med. 335(3): 179-188. Therefore, cumulative antibiotic toxicity and development of resistance remains a significant problem. Chronic obstructive pulmonary disease (COPD), a smoking-related condition characterized by progressive and poorly reversible airflow obstruction and airway inflammation, is the fourth most common cause of death in developed countries. COPD is projected to be the third leading cause of global deaths in 2020 and is the only one of the four most common causes of death with an increasing mortality rate. In 2008 in the United States, there were an estimated 10 million patients diagnosed with chronic obstructive pulmonary disease (COPD). SDI COPD Claims Analysis, May 2009. Murray et at., 1997 Lancer 349: 1269-76. Approximately 7 million U.S. patients receive treatment for COPD. Mannino et al; The Epidemiology and
Economics of COPD, Proc Am Tho e Soc 2007. In the US, direct COPD costs in 2002 were approximately $18.0 billion. Statistics from National Center for Health Statistics, National Health Interview Survey: Research for the 1995-2004 redesign, Hyattsville, Maryland: U.S. Department of Health and Human Services, CDC, NCHS. Vital and Health Stat 2( 26), 1999.
The clinical course of COPD is characterized by chronic disability, with intermittent, acute exacerbations which may be triggered by a variety of stimuli including exposure to pathogens, inhaled irritants (e.g., cigarette smoke), allergens, or pollutants. “Acute exacerbation” refers to worsening of a patient’s COPD symptoms from his or her usual state that is beyond normal day-to-day variations, and is acute in onset. See, Rabe et al., 2007 Am J Res Crit Care Med, 176: 532- 555. Acute exacerbations of COPD greatly affect the health and quality of life of patients with COPD. Bathoorn, E, Int J Chron Obstruct Pulmon Dis. 2008 3(2):217- 229. Acute exacerbation of COPD is a key driver of the associated substantial socioeconomic costs of the disease. Approximately 73% ($13 billion) of direct COPD costs in 2002 were due to hospitalizations related to acute exacerbations of COPD. Investigators from the Burden of Obstructive Lung Disease (BOLD) Initiative have estimated the cumulative discounted cost of COPD care in the US to be $880 billion by 2020 – an average of more than $44 billion per year over two decades. Lee et al., 2006 ATS Proceedings, 3:A598. Multiple studies have also shown that prior exacerbation is an independent risk factor for future hospitalization for COPD.
Garcia-Aymerich et al., 2003, Thorax, 58:100-105. Hospitalization consumes roughly 70% of COPD healthcare expenditure in the US. McGhan et al., 2007, Chest, 132(6): 1748-1755. Accordingly, for a new drug therapy to significantly reduce the health and economic costs of COPD, it must address acute exacerbations of COPD.
It is clear that there is a continued need for an improved method of treatment for acute and chronic respiratory infections caused by Gram-negative and Gram- positive bacteria, particularly multidrug resistant bacteria, such as P. aeruginosa. This is particularly evident in patients having chronic respiratory conditions where current therapies are limited by problems with development of resistance and toxicity. Such method of treatment would preferably comprise inhalation of an aerosolized antibiotic composition that delivers a therapeutically effective amount of the active pharmaceutical ingredients directly to the endobronchial space of the airways or to the nasal passages. Such treatment would ideally be efficacious, reduce the frequency of drug resistance, and improve safety.
PCT Publication No. WO2005/1 0022 to Gilead Sciences, Inc. (formerly Corus Pharma) discloses a fosfomycin plus tobramycin combination formulation for delivery by aerosolization. The concentrated fosfomycin/tobramycin combination formulation containing an efficacious amount of fosfomycin and tobramycin is able to inhibit susceptible bacteria. Fosfomycin and tobramycin are formulated in solution such that when reconstituted, the pH is between 4.5 and 8.0 or as a dry powder. Also disclosed is a method for treatment of respiratory tract infections by a formulation delivered as an aerosol having mass medium aerodynamic diameter predominantly from 1 to 5 microns, produced by a jet or ultrasonic nebulizer (or equivalent) or dry powder inhaler.
tobramycin solution for inhalation of various formulations are described in the prior art. 例如,美国专利第5,508,269号公开了一种制剂,该制剂包括在Iml的盐水中40_100mg (毫克)的氨基糖苷被稀释成四分之一生理盐水强度,该制剂的PH值在5. 5到6. 5之间,其中该溶液以5ml浓缩形式气雾给药。 For example, U.S. Patent No. 5,508,269 discloses a preparation which is included in the brine Iml 40_100mg (mg) was diluted into a quarter normal saline aminoglycoside strength, the PH value of the preparation of 5 . 5 to 6.5 between, wherein the solution in concentrated form 5ml aerosol administration. [0011] 美国专利6987094公开了一种气雾制剂,其包括75mg/ml妥布霉素的水性溶液,该溶液包括O. 45%w/v (质量/体积百分比)的氯化钠,该制剂的pH值在4. O到5. 5之间,而渗透压在250到450m0sm/l (毫渗透克分子/毫升)。[0011] U.S. Patent 6,987,094 discloses an aerosol formulation which comprises an aqueous solution of 75mg/ml tobramycin, the solution comprises O. 45% w / v (mass / volume) of sodium chloride, the preparation the pH value of 4. O to 5.5, between the osmotic pressure of 250 to 450m0sm / l (mg penetration mol / ml). [0012] 美国专利申请2007/0116649公开了一种气雾制剂,其包括约100mg/ml到200mg/ ml的抗革兰阴性抗生素。 [0012] U.S. Patent Application 2007/0116649 discloses an aerosol formulation comprising about 100mg/ml to 200mg / ml of antibiotics against gram-negative. 提到了妥布霉素制剂,但是没有公开有妥布霉素的实验。Tobramycin formulations mentioned, but there is no disclosure tobramycin experiment. [0013] 美国专利申请2007/0071686公开了一种妥布霉素组合物,其包括约80mg/ml到120mg/ml的妥布霉素、一酸性辅助剂和低浓度的氯化钠。[0013] U.S. Patent Application 2007/0071686 discloses a tobramycin composition comprises about 80mg/ml to 120mg/ml of tobramycin, an acidic auxiliary agents and a low concentration of sodium chloride. 所述酸性辅助剂可以是硫酸钠或磷酸钠。 The acidic auxiliary agent may be sodium or sodium phosphate. 根据US2007/0071686,活性剂的浓度不超过120mg/ml,这是因为据说妥布霉素的浓度因粘度原因对雾化有负面影响。According to US2007/0071686, the concentration of the active agent is not more than 120mg/ml, this is because the concentration of tobramycin said reasons due to the viscosity of a negative impact on atomization. 而且,根据US2007/0071686的妥布霉素组合物利用喷雾器给药给患者,即活性成分通过潮式呼吸吸入。Moreover, the composition according to US2007/0071686 of tobramycin administered to patients using the spray, that the active ingredient through the tidal breathing inhalation. [0014] 欧洲专利2186508尤其公开了一种少于4ml溶液的组合物,其包括的60 – 200mg/ ml的、在生理上可接受的载体上的氨基糖苷抗生素。 [0014] In particular, in European Patent 2,186,508 discloses a composition of the solution is less than 4ml, comprising of 60 – 200mg / ml, and in a physiologically acceptable carrier aminoglycoside antibiotics. EP2186508中的实验显示,包括120mg/ ml妥布霉素的组合物使用PARI LC PLUS®牌喷雾器(德国Starnberg的Pari Boy N压缩器公司)给药需要约10分钟。EP2186508 The experiments showed that including 120mg / ml tobramycin compositions using PARI LC PLUS ® brand spray (Starnberg, Germany The Pari Boy N compressor company) to about 10 minutes of administration. 尽管这少于商业上可获得的TOBI®给药所需的时间,但从患者配合度和患者友好角度考虑,所需的时间还是太长。 Although this is less than the commercially available TOBI ® dosing time required, but with the degree of the patient, and patient-friendly point of view, the time is too long. EP2186508提到,使用呼吸致动吸入装置对于前面提到的商业上可获得的系统可以获得更快的给药时间。 EP2186508 mentioned breath actuated inhalation device used for the previously mentioned commercially available systems can be obtained faster delivery time. 但是,EP2186508 中使用呼吸致动吸入装置(AcroDose™)所获得的给药时间的实验仅限于低浓度的妥布霉素(60mg/ml)。However, EP2186508 using the breath actuated inhaler device (AcroDose ™) obtained experimental delivery time is limited to low concentrations of tobramycin (60mg/ml). 其还指出,使用AcroDose™系统的60mg/ml组合物给药,还必须给药第二可分量。 It also noted that the use AcroDose ™ system 60mg/ml composition administered may also be administered a second component. 从患者友好和配合角度来看,需要加入和给药第二可分量代表一种缺点。 Friendship and cooperation from the patient’s point of view, and the administration need to add a second component represents a drawback can be. [0015] 妥布霉素溶液也以局部给药出名,例如治疗角膜炎,参见Davis等人在“Canad. J Ophtal. ”(加拿大眼科杂志,1978年第13期273页)的文章,Davis等人在“Arch Opthalmol” (眼科杂志,1978年第96卷123-125页)的文章和Unter man等人在“ J. Cataract Refract. Surg. ”(白内障手术杂志,1988年第14卷500-504页)的文章。[0015] tobramycin solution is also famous for topical administration, such as treatment keratitis, see Davis et al in “Canad. J Ophtal.” (Canadian Journal of Ophthalmology, 1978 13 273) of the article, Davis, etc. in “Arch Opthalmol” (Ophthalmology 1978 Volume 96 pages 123-125) of articles and Unter man and others in “J. Cataract Refract. Surg.” (cataract surgery magazine, Volume 14, 500-504, 1988 pages) of the article. [0016] 现有给药手段和治疗方案的公知缺点是给药所需的时间,尤其影响患者的配合度和患者的生活质量。 Existing methods of administration and treatment programs known

FDA Approves Sanofi’s Nasacort® Allergy 24HR for Over-the-Counter Use – Nasal Spray Treats Adults and Children with Seasonal and Year-Round Nasal Allergies
Nasacort
Paris, France, October 11, 2013 — Sanofi (EURONEXT: SAN and NYSE: SNY) announced today that the U.S. Food and Drug Administration (FDA) approved Nasacort® Allergy 24HR nasal spray as an over-the-counter (OTC) treatment for seasonal and year-round nasal allergies in adults and children 2 years of age and older. Nasacort is the first and only medicine in its class to be available without a prescription and will be marketed by Sanofi’s consumer healthcare division, Chattem, Inc.
read all at
http://www.pharmalive.com/fda-oks-otc-nasacort-allergy-24hr
Triamcinolone acetonide, USP, the active ingredient in NASACORT AQ Nasal Spray, is a corticosteroid with a molecular weight of 434.51 and with the chemical designation 9-Fluoro11β,16α,17,21-tetrahydroxypregna-1,4-diene-3,20-dione cyclic 16,17-acetal with acetone (C24H31FO6).
 |
NASACORT AQ Nasal Spray is a thixotropic, water-based metered-dose pump spray formulation unit containing a microcrystalline suspension of triamcinolone acetonide in an aqueous medium. Microcrystalline cellulose, carboxymethylcellulose sodium, polysorbate 80, dextrose, benzalkonium chloride, and edetate disodium are contained in this aqueous medium; hydrochloric acid or sodium hydroxide may be added to adjust the pH to a target of 5.0 within a range of 4.5 and 6.0.
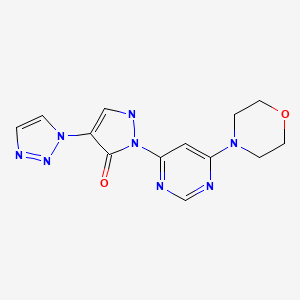
Molidustat (BAY 85-3934), Bayer’s drug under initiation in patients with anemia associated with chronic kidney disease and/or end-stage renal disease.
Molidustat
UNII-9JH486CZ13, cas no 1154028-82-6, MW: 314.3076
2-(6-morpholin-4-ylpyrimidin-4-yl)-4-(triazol-1-yl)-1H-pyrazol-3-one
Hypoxia-Inducible Factor Prolyl Hydroxylase Inhibitors
For the cardio-renal syndrome, a Phase IIb program with the investigational new drug Molidustat (BAY 85-3934) is under initiation in patients with anemia associated with chronic kidney disease and/or end-stage renal disease. Molidustat is a novel inhibitor of hypoxia-inducible factor (HIF) prolyl hydroxylase (PH) which stimulates erythropoietin (EPO) production and the formation of red blood cells. Phase I data have shown that inhibition of HIF-PH by Molidustat results in an increase in endogenous production of EPO.
About Bayer HealthCare
The Bayer Group is a global enterprise with core competencies in the fields of health care, agriculture and high-tech materials. Bayer HealthCare, a subgroup of Bayer AG with annual sales of EUR 18.6 billion (2012), is one of the world’s leading, innovative companies in the healthcare and medical products industry and is based in Leverkusen, Germany. The company combines the global activities of the Animal Health, Consumer Care, Medical Care and Pharmaceuticals divisions. Bayer HealthCare’s aim is to discover, develop, manufacture and market products that will improve human and animal health worldwide. Bayer HealthCare has a global workforce of 54,900 employees (Dec 31, 2012) and is represented in more than 100 countries. More information at www.healthcare.bayer.com.
molidustat
…………………………………………………………..
molidusat sodium
Sodium 1-[6-(morpholin-4-yl)pyrimidin-4-yl]-4-(1H-1,2,3-triazol-1-yl)-1H-pyrazol-5-olate
Molidustat sodium is an orally-available hypoxia-inducible factor prolyl hydroxylase inhibitor in phase I clinical trials at Bayer for the treatment of patients suffering from renal anemia due to chronic kidney disease.
WO 2008067871
WO 2012065967
WO 2013167552
…………………………
2-Heteroaryl-4-aryl-1,2-dihydropyrazolones having a bactericidal and/or fungicidal action are disclosed in EP 165 448 and EP 212 281. The use of 2-heteroaryl-4-aryl-1,2-dihydropyrazolones as lipoxygenase inhibitors for treatment of respiratory tract, cardiovascular and inflammatory diseases is claimed in EP 183 159. 2,4-Diphenyl-1,2-dihydropyrazolones having a herbicidal activity are described in DE 2 651 008.
The preparation and pharmacological properties of certain 2-pyridyl-1,2-dihydropyrazolones are reported in Helv. Chim. Acta 49 (1), 272-280 (1966). WO 96/12706, WO 00/51989 and WO 03/074550 claim compounds having a dihydropyrazolone partial structure for treatment of various diseases, and hydroxy- or alkoxy-substituted bipyrazoles for treatment of neuropsychiatric diseases are disclosed in WO 2006/101903.
Heteroaryl-substituted pyrazole derivatives for treatment of pain and various CNS diseases are furthermore described in WO 03/051833 and WO 2004/089303. WO 2006/114213 has meanwhile disclosed 2,4-dipyridyl-1,2-dihydropyrazolones as inhibitors of HIF prolyl 4-hydroxylases.
The x-ray crystal structure of the compound 3-methyl-1-(pyridin-2-yl)-4-(1-pyridin-2-yl-3-methyl-1H-pyrazol-5-yl)-2H-3-pyrazolin-5 (114)-one (other name: 5,5′-dimethyl-2,2′-di-pyridin-2-yl-1′,2′-dihydro-2H,3′H-3,4′-bipyrazol-3′-one) is reported inActa Crystallogr., Section E: Structure Reports Oμline E57 (11), o1126-o1127 (2001) [Chem. Abstr. 2001:796190].
The synthesis of certain 3′,5-dimethyl-2-phenyl-1′-(1,3-thiazol-2-yl)-1′H,2H-3,4′-bipyrazol-5′-ol derivatives is described inIndian J. Heterocyclic Chem. 3 (1), 5-8 (1993) [Chem. Abstr. 1994:323362].
The preparation and tautomerism of individual 4-(pyrazol-5-yl)-pyrazolin-5-one derivatives is reported in J. Heterocyclic Chem. 27 (4), 865-870 (1990) [Chem. Abstr. 1991:428557]. A therapeutic use has not hitherto been described for the compounds mentioned in these publications. The compound 2-tert-butyl-1′-[4-(4-chlorophenyl)-1,3-thiazol-2-yl]-3′,5-dimethyl-1′H,2H-3,4′-bipyrazol-5′-ol is listed as a test example in WO 2007/008541.
………………………….
https://www.google.co.in/patents/US20100305085
Example 3A 3-(Dimethylamino)-2-(1H-1,2,3-triazol-1-yl)acrylic acid ethyl ester
The preparation of the starting compound is carried out analogously to 2A starting from 1.00 g (6.45 mmol) 2-(1H-1,2,3-triazol-1-yl)acetic acid ethyl ester.
Yield: 1.4 g (100% of th.)
1H-NMR (400 MHz, DMSO-d6): δ=8.10 (d, 1H), 7.78 (d, 1H), 7.65 (s, 1H), 4.03 (q, 2H), 3.06 (br. s, 3H), 2.10 (br. s, 3H), 1.12 (t, 3H).
LC-MS (Method 5): Rt=1.40 min; MS (ESIpos): m/z=211 [M+H]+.
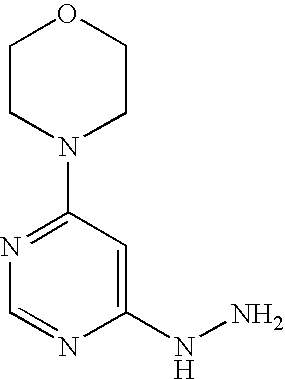
Example 16A 4-(6-Hydrazinopyrimidin-4-yl)morpholine
Stage a): 4-(6-Chloropyrimidin-4-yl)morpholine
45.0 g (302.1 mmol) 4,6-dichloropyrimidine are initially introduced into 450 ml water. 26.3 g (302.1 mmol) morpholine are added and the mixture is stirred at 90° C. for 16 h. Thereafter, it is cooled to 0° C. and the precipitate formed is filtered off. The precipitate is washed once with 50 ml water and dried in air.
Yield: 51.0 g (85% of th.)
LC-MS (Method 4): Rt=1.09 min; MS (ESIpos): m/z=200 [M+H]+;
1H-NMR (400 MHz, DMSO-d6): δ=8.35 (s, 1H), 6.95 (s, 1H), 3.62 (s, 8H).
Stage b) 4-(6-Hydrazinopyrimidin-4-yl)morpholine
53.0 g (2.7 mmol) 4-(6-chloropyrimidin-4-yl)morpholine are initially introduced into 260 ml ethanol. 132.9 g (2.7 mol) hydrazine hydrate are added and the mixture is stirred under reflux for 16 h. Thereafter, it is cooled to RT and approx. half of the solvent is removed by distillation. The mixture is cooled to 0° C. and the solid formed is filtered off. It is rinsed with cold ethanol and the solid is dried first in air and then in vacuo.
Yield: 35.0 g (68% of th.)
LC-MS (Method 1): Rt=0.17 min; MS (ESIpos): m/z=196 [M+H]+;
1H-NMR (400 MHz, DMSO-d6): δ=7.94 (s, 1H), 7.70 (s, 1H), 5.91 (s, 1H), 4.15 (s, 2H), 3.66-3.60 (m, 4H), 3.45-3.37 (m, 4H).
Example 71 2-(6-Morpholin-4-ylpyrimidin-4-yl)-4-(1H-1,2,3-triazol-1-yl)-1,2-dihydro-3H-pyrazol-3-one
1.9 g (8.8 mmol) of the compound from Example 3A and 1.9 g (9.7 mmol) of the compound from Example 16A are initially introduced into 25 ml ethyl acetate and 504 mg (4.4 mmol) TFA are added at RT. The mixture is stirred under reflux for 16 h, then cooled to 5° C. and subsequently stirred for a further 2 h. The solid formed is filtered off, washed with ethyl acetate and dried first in air and thereafter under a high vacuum. 1.7 g of product are obtained.
The mother liquor is combined with the wash solution and the solvent is removed. According to LC-MS, the residue (2.4 g) still contains the intermediate 3-[2-(6-morpholin-4-ylpyrimidin-4-yl)hydrazino]-2-(1H-1,2,3-triazol-1-yl)prop-2-enoic acid ethyl ester (intermediate stage of the cyclization), which is used directly for the preparation of Example 72 (see there).
Yield: 1.7 g (61% of th.)
LC-MS (Method 9): Rt=0.90 min; MS (ESIpos): m/z=315 [M+H]+;
1H-NMR (400 MHz, DMSO-d6): δ=8.42 (s, 1H), 8.38 (s, 1H), 8.01 (s, 1H), 7.73 (s, 1H), 7.70 (s, 1H), 3.71-3.65 (m, 4H), 3.57-3.51 (m, 4H).
hydrochloride
Example 72 2-(6-Morpholin-4-ylpyrimidin-4-yl)-4-(1H-1,2,3-triazol-1-yl)-1,2-dihydro-3H-pyrazol-3-one hydrochloride
Batch 1: 7.5 ml of a 4 N solution of hydrogen chloride in dioxane are added to 1.7 g (5.4 mmol) of the compound from Example 71. The mixture is stirred at RT, 5 ml dioxane are added and the mixture is stirred at RT for 16 h. The solid is filtered off and washed with 5 ml dioxane. The mixture is dried under a high vacuum for 16 h, 10 ml methanol are then added and the mixture is stirred at RT for 1 h. The solid is filtered off, washed with 4 ml methanol and dried under a high vacuum. 1.6 g of the title compound are obtained.
Batch 2: A further amount of the title compound is obtained as follows: The residue (2.4 g) obtained from the mother liquor during the synthesis of Example Compound 71, which contains the open-ring intermediate state of the cyclization, 3-[2-(6-morpholin-4-ylpyrimidin-4-yl)hydrazino]-2-(1H-1,2,3-triazol-1-yl)prop-2-enoic acid ethyl ester, is dissolved in 12 ml ethanol and 1.5 ml 30% strength sodium methylate solution in methanol are added at RT, while stirring. The mixture is subsequently stirred at RT for 45 min, then adjusted to pH 5 with 2 N hydrochloric acid and subsequently stirred at RT for a further 16 h. The mixture is cooled to 10° C. and the solid is filtered off and washed with 3.5 ml dioxane. The mixture is dried under a high vacuum for 16 h, 5 ml methanol are then added and the mixture is subsequently stirred at RT for 1 h. The solid is filtered off, washed with 2 ml methanol and dried under a high vacuum to give a further 997 mg of the title compound in this way.
Yield: together 2.6 g (83% of th.)
LC-MS (Method 6): Rt=0.89 min; MS (ESIpos): m/z=315 [M+H]+;
1H-NMR (400 MHz, DMSO-d6): δ=8.54 (s, 1H), 8.39 (s, 1H), 8.28 (s, 1H), 7.88 (s, 1H), 7.42 (s, 1H), 3.71 (s, 8H).
amcrasto@gmail.com
email me if u like my posts
Current Oncology Pipeline Trends

Oncology drug development far outpaces drug development for other therapeutic areas and the magnitude of that difference is significant. Here’s a current review of what is in the pipeline and an analysis of where oncology research is headed.

Stacey Ness, PharmD, RPh, MSCS, AAHIVP, has worked in both national specialty pharmacy and payer organizations and has experience in clinical management, adherence, and persistency programs, as well as chronic disease cost optimization strategies. Dr. Ness is active in the Consortium of Multiple Sclerosis Centers, Academy of Managed Care Pharmacy, National Home Infusion Association, National Association of Specialty Pharmacy, Specialty Pharmacy Certification Board, and Hematology and Oncology Pharmacy Association, and has served on the Minnesota Medicaid Drug Formulary Committee since 2008. She is a multiple sclerosis certified specialist, a credentialed HIV pharmacist, and currently serves as the director of specialty clinical services at Managed Health Care Associates, Inc, a health care services organization based in Florham Park, New Jersey.
Orexigen files obesity drug Contrave for approval in Europe

Orexigen files obesity drug Contrave for approval in Europe
Orexigen Therapeutics has submitted the Marketing Authorization Application (MAA) for Contrave, an investigational weight-loss drug to the European Medicines Agency (EMA).
 The La Jolla, CA-based drug firm is using the EMA’s centralised procedure to seek approval for Contrave Orexigen (32 mg naltrexone sustained release (SR) / 360 mg bupropion SR) for the management of obesity, including weight loss and maintenance of weight loss, in conjunction with lifestyle modification. The company filed the application after meeting with the European agency to discuss the filing strategy and “both were supportive” of the company’s plan to file in advance of an eagerly-anticipated interim analysis of a cardiovascular outcomes trial called the Light study. Orexigen and the European regulator have also agreed upon an investigation plan in children and adolescents.
The La Jolla, CA-based drug firm is using the EMA’s centralised procedure to seek approval for Contrave Orexigen (32 mg naltrexone sustained release (SR) / 360 mg bupropion SR) for the management of obesity, including weight loss and maintenance of weight loss, in conjunction with lifestyle modification. The company filed the application after meeting with the European agency to discuss the filing strategy and “both were supportive” of the company’s plan to file in advance of an eagerly-anticipated interim analysis of a cardiovascular outcomes trial called the Light study. Orexigen and the European regulator have also agreed upon an investigation plan in children and adolescents.
read all at
http://www.pharmatopics.com/2013/10/orexigen-files-obesity-drug-contrave-approval-europe/

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....