Home » Posts tagged 'C2VJ83PSN7'
Tag Archives: C2VJ83PSN7
Olomorasib
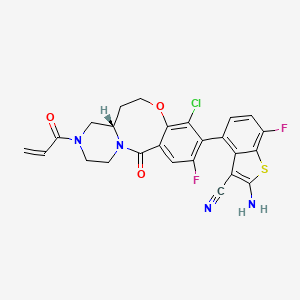
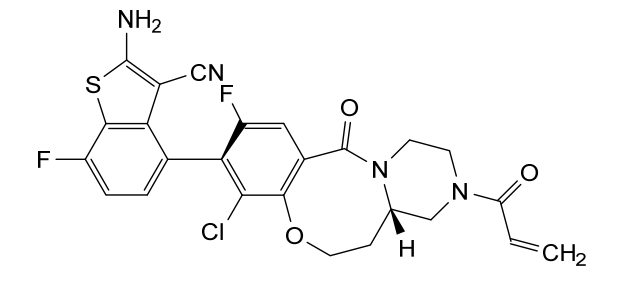
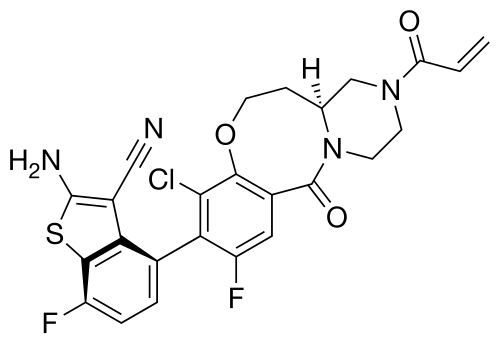
Olomorasib
CAS 2771246-13-8
MF C25H19ClF2N4O3S MW528.96
4-[(13aS)-10-chloro-8-fluoro-6-oxo-2-prop-2-enoyl-1,3,4,12,13,13a-hexahydropyrazino[2,1-d][1,5]benzoxazocin-9-yl]-2-amino-7-fluoro-1-benzothiophene-3-carbonitrile
(4M)-2-amino-4-[(4aS)-8-chloro-10-fluoro-12-oxo-3-(prop-2-enoyl)-2,3,4,4a,5,6-hexahydro-1H,12H-pyrazino[2,1-d][1,5]benzoxazocin-9-yl]-7-fluoro-1-benzothiophene-3-carbonitrile
Kirsten rat sarcoma viral oncogene homolog (KRAS) inhibitor, antineoplastic, LY3537982, LY 3537982, KRAS-G12C-II, LY-3537982, C2VJ83PSN7,
Olomorasib (LY3537982) is an investigational, oral, second-generation KRAS G12C inhibitor designed to treat advanced solid tumors, particularly non-small cell lung cancer (NSCLC). Developed by Eli Lilly and Company, it shows promising antitumor activity and a manageable safety profile, often combined with pembrolizumab (Keytruda). Eli Lilly and Company +3
Key details about olomorasib include:
- Mechanism & Target: It targets the KRAS G12C mutation, a common driver in lung and colorectal cancers.
- Clinical Status: It is undergoing Phase 1/2 (LOXO-RAS-20001) and Phase 3 (SUNRAY-01) clinical trials.
- Breakthrough Therapy: The FDA granted Breakthrough Therapy designation for first-line treatment of advanced NSCLC (PD-L1
50%) in September 2025.
- Combination Efficacy: When combined with pembrolizumab, it showed an objective response rate of 73.9% in first-line patients, with higher efficacy in those with high PD-L1 expression.
- Safety Profile: Common adverse events include diarrhea, elevated liver enzymes (ALT/AST), and rash, which were generally manageable.
Eli Lilly and Company +4
Olomorasib is designed to be more potent with potentially better tolerability than earlier KRAS G12C inhibitors, aiming to improve outcomes in first-line settings.
- OriginatorEli Lilly and Company
- ClassAntineoplastics; Small molecules
- Mechanism of ActionKRAS protein inhibitors
- Phase IIINon-small cell lung cancer
- Phase ISolid tumours
- 05 Jan 2026Eli Lilly and Company completes a phase-I trial (In volunteers) in Japan (PO, Capsule) (NCT07124013)
- 22 Dec 2025Phase-I/II clinical trials in Non-small cell lung cancer (Metastatic disease, Second-line therapy or greater, Combination therapy) in USA, Canada, China, South Korea (PO) (NCT07227025)
- 12 Nov 2025Janssen Research & Development plans a phase I/II (KaRAnaSa) trial for Non-small cell lung cancer (Combination Therapy, Metastatic disease, Second-line therapy or greater) in December 2025 (NCT07227025)
Olomorasib is an orally available inhibitor of the oncogenic KRAS substitution mutation, G12C, with potential antineoplastic activity. Upon oral administration, olomorasib selectively targets the KRAS G12C mutant and inhibits KRAS G12C mutant-dependent signaling. KRAS, a member of the RAS family of oncogenes, serves an important role in cell signaling, division and differentiation. Mutations of KRAS may induce constitutive signal transduction leading to tumor cell growth, proliferation, invasion, and metastasis.
Olomorasib (LY3537982) is an experimental anticancer drug which acts as an inhibitor of the G12C mutant form of Kirsten rat sarcoma virus (KRAS), an oncogene commonly present in several forms of cancer. It is in early stage clinical trials against lung and colorectal cancers and advanced solid tumors.[1][2][3][4][5]
PAPER
ACS Omega. 2025 Jul 4;10(27):29637-29646. [Abstract]
PATENT
•Patent. US20240307395A1.
PAPER
https://www.nature.com/articles/s41598-025-07532-2
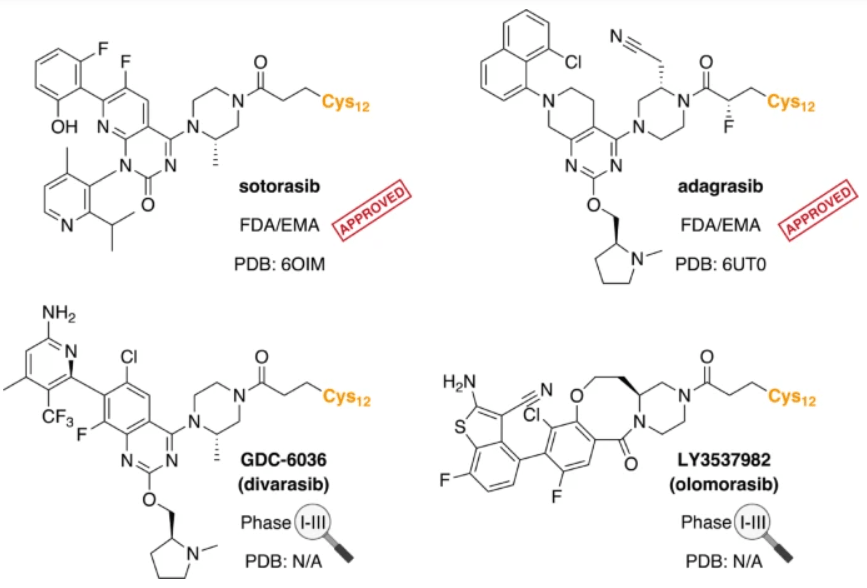
SYN
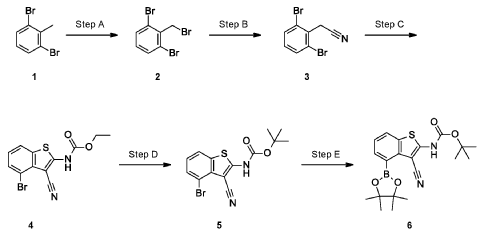
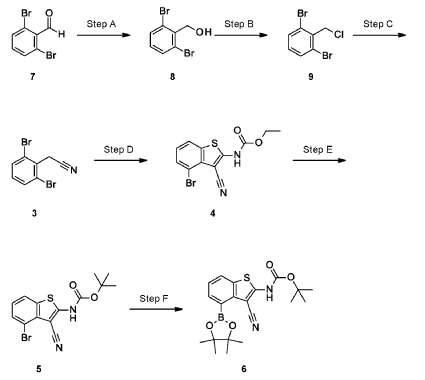
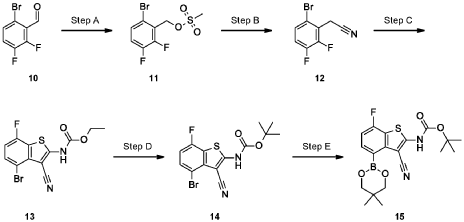
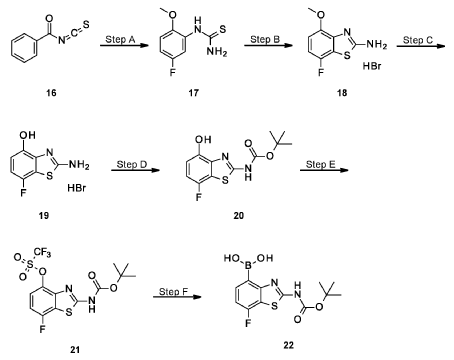
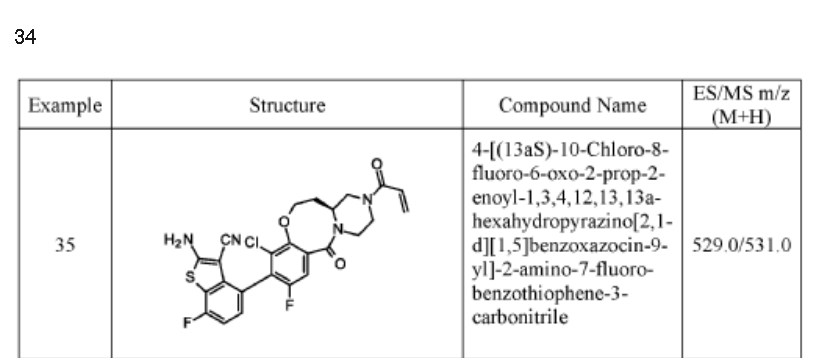
Example 34
4-[( 13 aS)- 10-Chloro-8-fluoro-6-oxo-2-prop-2-enoyl- 1,3,4,12, 13,13 a- hexahydropyrazino[2,ld][1,5]benzoxazocin-9-yl]-2-amino-benzothiophene-3- carbonitrile
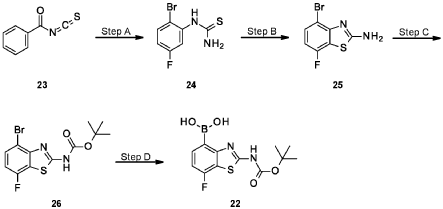
A suspension of 4-[(13aS)-10-chloro-8-fluoro-6-oxo-2,3,4,12,13,13a-hexahydro-lH-pyrazino[2,ld][l,5]benzoxazocin-9-yl]-2-amino-benzothiophene-3-carbonitrile (1.58 g, 3.46 mmol) in EtOAc (35 mL), THF (15 mL) and water (40 mL) is charged with potassium carbonate (1.90 g, 13.7 mmol). The mixture is stirred rapidly and cooled to 0 °C. Acryloyl chloride in DCM (13.0 mL, 3.25 mmol, 0.25M) is added dropwise through a dropping funnel. After 10 minutes of stirring in an ice bath, the mixture is diluted with EtOAc and poured into a separatory funnel. The layers are separated and the aqueous layer is again extracted with EtOAc. The combined organic extracts are washed with saturated aqueous sodium chloride solution, dried over magnesium sulfate, filtered, and concentrated in vacuo. The residue is purified by silica gel flash column chromatography, eluting first with 0-100% (10% MeOH in DCM) / DCM, and second with 0-100% [10% (7N NH 3 in MeOH) in DCM] / DCM to give the desired product as fluffy solid. The solid is sonicated in ether for 30 minutes, filtered, and dried in high vacuum to give the title compound (1.60 g, 91%). ES/MS m/z ( 35 C1/ 37 C1) 511.0/513.0 [M+H] + .
Table 22: Compounds synthesized in a manner essentially analogous to that of Example
PAT
- Methods of Delaying, Preventing, and Treating Acquired Resistance to RAS InhibitorsPublication Number: KR-20230042600-APriority Date: 2020-06-18
- Methods for delaying, preventing, and treating acquired resistance to ras inhibitorsPublication Number: EP-4168002-A1Priority Date: 2020-06-18
- KRAS Gl2C INHIBITORSPublication Number: US-2023339968-A1Priority Date: 2019-12-11
- Kras g12c inhibitorsPublication Number: US-2021179633-A1Priority Date: 2019-12-11
- KRas G12C inhibitorsPublication Number: US-11731984-B2Priority Date: 2019-12-11Grant Date: 2023-08-22
- Combination of antibody-drug conjugate and rasg12c inhibitorPublication Number: WO-2023126822-A1Priority Date: 2021-12-28
- Conjugates comprising covalent binders for targeting intracellular kras g12c proteinsPublication Number: US-2024252694-A1Priority Date: 2021-08-06
- Conjugates containing covalent binders to target intracellular KRAS G12C proteinPublication Number: KR-20240099134-APriority Date: 2021-08-06
- Checkpoint kinase 1 (chk1) inhibitors and uses thereofPublication Number: CA-3219348-A1Priority Date: 2021-05-27
- Methods for inhibiting rasPublication Number: AU-2022281343-A1Priority Date: 2021-05-25



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
- Peng SB, Si C, Zhang Y, Van Horn RD, Lin X, Gong X, et al. (July 2021). “Preclinical characterization of LY3537982, a novel, highly selective and potent KRAS-G12C inhibitor”. Cancer Research. 81 (13_Supplement): 1259. doi:10.1158/1538-7445.AM2021-1259.
- Miyashita H, Hong DS (2024). “Combining EGFR and KRAS G12C Inhibitors for KRAS G12C Mutated Advanced Colorectal Cancer”. Journal of Cancer Immunology. 6 (2): 62–69. doi:10.33696/cancerimmunol.6.086. PMC 11340593. PMID 39175850.
- Hollebecque A, Kuboki Y, Murciano-Goroff YR, Yaeger R, Cassier PA, Heist RS, et al. (2024). “Efficacy and safety of LY3537982, a potent and highly selective KRAS G12C inhibitor in KRAS G12C-mutant GI cancers: Results from a phase 1 study”. Journal of Clinical Oncology. 42 (3_suppl): 94. doi:10.1200/JCO.2024.42.3_suppl.94.
- Burns TF, Dragnev KH, Fujiwara Y, Murciano-Goroff YR, Lee DH, Hollebecque A, et al. (2024). “Efficacy and safety of olomorasib (LY3537982), a second-generation KRAS G12C inhibitor (G12Ci), in combination with pembrolizumab in patients with KRAS G12C-mutant advanced NSCLC”. Journal of Clinical Oncology. 42 (16_suppl): 8510. doi:10.1200/JCO.2024.42.16_suppl.8510.
- Heist RS, Koyama T, Murciano-Goroff YR, Hollebecque A, Cassier PA, Han J, et al. (2024). “Pan-tumor activity of olomorasib (LY3537982), a second-generation KRAS G12C inhibitor (G12Ci), in patients with KRAS G12C-mutant advanced solid tumors”. Journal of Clinical Oncology. 42 (16_suppl): 3007. doi:10.1200/JCO.2024.42.16_suppl.3007.
| Clinical data | |
|---|---|
| Other names | LY3537982 |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2649788-46-3 |
| PubChem CID | 156472638 |
| ChemSpider | 115009373 |
| UNII | C2VJ83PSN7 |
| KEGG | D12853 |
| Chemical and physical data | |
| Formula | C25H19ClF2N4O3S |
| Molar mass | 528.96 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
///////olomorasib, Kirsten rat sarcoma viral oncogene homolog (KRAS) inhibitor, antineoplastic, LY3537982, LY 3537982, KRAS-G12C-II, LY-3537982, C2VJ83PSN7,

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










