Home » Posts tagged '5-Fluoro-N-methyl-N-ethyltryptamine'
Tag Archives: 5-Fluoro-N-methyl-N-ethyltryptamine
Bretisilocin
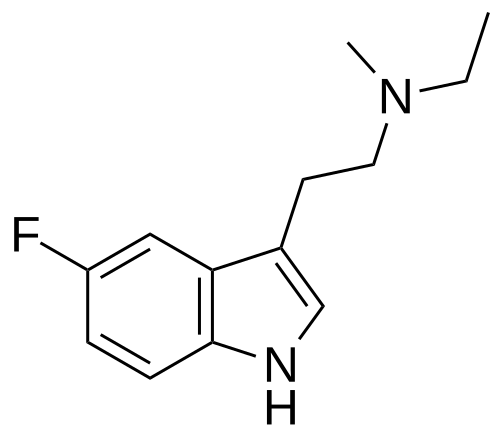
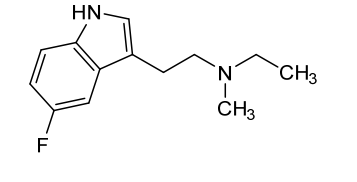
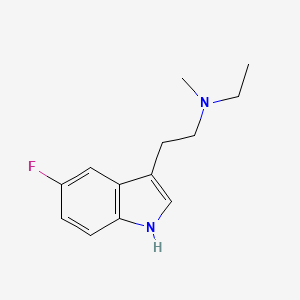
Bretisilocin
CAS2698331-35-8
MF C13H17FN2 MW220.29 g/mol
N-ethyl-2-(5-fluoro-1H-indol-3-yl)-N-methylethan-1-amine
serotonin (5-HT2A) receptor agonist, GM-2505, GM 2505, 5-Fluoro-N-methyl-N-ethyltryptamine, 5F-MET, 5-F-MET, 5-Fluoro-MET, DS425RQ8SX
Bretisilocin, also known by its developmental code name GM-2505 and as 5-fluoro-N-methyl-N-ethyltryptamine (5F-MET or 5-fluoro-MET), is a serotonergic psychedelic of the tryptamine family which is under development for the treatment of major depressive disorder.[1][7][2][3] It is an analogue of dimethyltryptamine (DMT) and is the 5-fluorinated derivative of methylethyltryptamine (MET).[8] Bretisilocin’s route of administration is intravenous infusion.[1][2][3][4]
The drug acts as a potent and well-balanced serotonin 5-HT2A and 5-HT2C receptor agonist, serotonin 5-HT2B receptor partial agonist or antagonist, and serotonin releasing agent.[2][9][8][10] It produces psychedelic-like effects in animals and similarly produces robust hallucinogenic effects in humans.[9][3] The duration of bretisilocin is 60 to 90 minutes and is intermediate between the durations of DMT and psilocybin.[6][11][4][12][8][2] It has been regarded by its developer as an “improved version of DMT”.[12]
Bretisilocin was first described in the literature by 2022.[9][10] It is under development by Gilgamesh Pharmaceuticals.[1] As of June 2025, the drug is in phase 2 clinical trials for the treatment of major depressive disorder.[1] Bretisilocin was acquired from Gilgamesh Pharmaceuticals by AbbVie in a deal worth up to $1.2 billion in August 2025.[13][14] It was encountered as a novel recreational designer drug in 2026.[5]
Chemistry
Bretisilocin, also known as 5-fluoro-N-methyl-N-ethyltryptamine, is a substituted tryptamine derivative.[8] It is a derivative of dimethyltryptamine (DMT) and methylethyltryptamine (MET) as well as of 5-fluorotryptamine (5-FT).[6][8]
Synthesis
The chemical synthesis of bretisilocin has been described.[10]
Analogues
Some analogues of bretisilocin include 5-fluoro-DMT, 5-fluoro-DET, 5-fluoro-EPT, 5-chloro-DMT, 5-bromo-DMT, 5-fluoro-AMT, 5-fluoro-AET, 5-MeO-MET, and 7-F-5-MeO-MET, among others.
History
Bretisilocin was first described in the scientific literature by at least 2022.[9][10] It was patented by Jason Wallach and colleagues at the University of the Sciences in Philadelphia that year.[10] The drug was encountered as a novel recreational designer drug in March 2026.[5]
Society and culture
Names
Bretisilocin is the generic name of the drug and its INNTooltip International Nonproprietary Name.[16] It is also known by its developmental code name GM-2505.[1][9][3]
Legal status
Canada
Bretisilocin is not a controlled substance in Canada as of 2025.[17]
United States
Bretislocin is not an explicitly controlled substance in the United States.[18] However, it could be considered a controlled substance under the Federal Analogue Act if intended for human consumption.
Research
Bretisilocin is under development as a potential pharmaceutical drug by Gilgamesh Pharmaceuticals.[1] As of June 2025, it is in phase 2 clinical trials for the treatment of major depressive disorder.[1] A phase 2a trial of bretisilocin for major depressive disorder has been completed and the efficacy and safety data for the trial have been released.[1][19][20][21] The drug has since been acquired from Gilgamesh Pharmaceuticals by AbbVie in a deal worth up to $1.2 billion.[13][14] In 2026 bretisilocin entered European Medicines Agency’s priority medicines (PRIME) scheme for major depressive disorder.[22][23]
SYN
Example 12: N-ethyl-2-(5-fluoro-1H-indol-3-yl)-N-methylethan-1-amine (12)
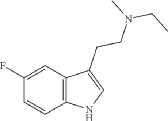
Synthesis of N-[2-(5-fluoro-1H-indol-3-yl)ethyl]formamide
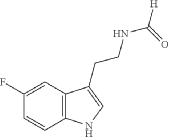
To a solution of 5-fluorotryptamine hydrochloride (3 g, 14.0 mmol) in H 2O (200 mL) with stirring was added KOH until a precipitate was obtained. The aqueous mixture was extracted with EtOAc (3×70 mL), the organic phases were pooled, washed with brine, dried over anhydrous Na 2SO 4, and concentrated in vacuo. Residual EtOAc was removed by azeotropic distillation with ethyl formate (3×20 mL). The resulting 5-fluorotryptamine free base was transferred to a 30 mL oven-dried microwave vessel containing 3 Å molecular sieves (3.3 g). Ethyl formate (20 mL, 248 mmol) was added to the microwave vessel and the mixture was reacted for 2.5 h at 80° C. with 150 W in a microwave reactor. Upon completion, ethyl formate was removed under reduced pressure to provide N-[2-(5-fluoro-1H-indol-3-yl)ethyl]formamide (1.7 g, 8.24 mmol, 58.9% yield). The product was used in the subsequent reaction without further purification.
To a solution of 5-fluorotryptamine hydrochloride (3 g, 14.0 mmol) in H 2O (200 mL) with stirring was added KOH until a precipitate was obtained. The aqueous mixture was extracted with EtOAc (3×70 mL), the organic phases were pooled, washed with brine, dried over anhydrous Na 2SO 4, and concentrated in vacuo. Residual EtOAc was removed by azeotropic distillation with ethyl formate (3×20 mL). The resulting 5-fluorotryptamine free base was transferred to a 30 mL oven-dried microwave vessel containing 3 Å molecular sieves (3.3 g). Ethyl formate (20 mL, 248 mmol) was added to the microwave vessel and the mixture was reacted for 2.5 h at 80° C. with 150 W in a microwave reactor. Upon completion, ethyl formate was removed under reduced pressure to provide N-[2-(5-fluoro-1H-indol-3-yl)ethyl]formamide (1.7 g, 8.24 mmol, 58.9% yield). The product was used in the subsequent reaction without further purification.
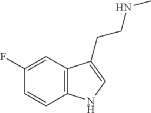
Synthesis of 2-(5-fluoro-1H-indol-3-yl)-N-methylethan-1-amine
Synthesis of N-ethyl-2-(5-fluoro-1H-indol-3-yl)-N-methylethan-1-amine (12)
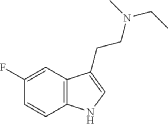
N-ethyl-2-(5-fluoro-1H-indol-3-yl)-N-methylethan-1-amine (12) was synthesized in a similar manner as described above for N-(2-(5-fluoro-1H-indol-3-yl)ethyl)-N-propylpropan-1-amine (5), starting from 2-(5-fluoro-1H-indol-3-yl)-N-methylethan-1-amine (0.7 g, 3.64 mmol), and acetaldehyde (0.96 g, 21.8 mmol), to provide the title compound as a colorless oil after purification by column chromatography using silica gel as a stationary phase and 20% EtOH/EtOAc (1% Et 3N v/v) as the mobile phase (0.62 g, 2.81 mmol, 77.2% yield), and subsequently the corresponding HCl salt as a white crystalline solid. HR-ASAP-MS: m/z 221.1442 (theoretical [M+H] +, C 13H 18FN 2 +), m/z 221.1449 (observed, Δ=−3.2 ppm). 1H-NMR (400 MHz, d 6-DMSO) δ 11.15 (s, 1H), 10.74 (s, 1H), 7.44 (dd, J=10.1, 2.5 Hz, 1H), 7.36 (dd, J=8.8, 4.6 Hz, 1H), 7.33 (d, J=2.3 Hz, 1H), 6.93 (dt, J=9.2, 2.5 Hz, 1H), 3.31-3.13 (m, 4H), 3.13-3.05 (m, 2H), 2.78 (d, J=3.1 Hz, 3H), 1.26 (t, J=7.3 Hz, 3H). 13C-NMR (101 MHz, d 6-DMSO) δ 156.74 (d, J=231.1 Hz, 1C), 132.88 (s, 1C), 126.96 (d, J=10.0 Hz, 1C), 125.42 (s, 1C), 112.48 (d, J=9.9 Hz, 1C), 109.51 (s, 1C), 109.32 (d, J=26.0 Hz, 1C), 103.13 (d, J=23.1 Hz, 1C), 54.37 (s, 1C), 49.82 (s, 1C), 38.13 (s, 1C), 19.68 (s, 1C), 8.79 (s, 1C). 19F-NMR (377 MHz, d 6-DMSO) δ−124.79 (s, 1F).
PAT
PAT
Methods of treating mood disorders
Publication Number: US-2022041551-A1
Priority Date: 2020-02-18
- Methods and compositions relating to psychedelics and serotonin receptor modulatorsPublication Number: EP-4313030-A1Priority Date: 2021-04-01
- Methods and compositions related to hallucinogens and serotonin receptor modulatorsPublication Number: KR-20240037873-APriority Date: 2021-04-01
- Halogenated psilocybin derivatives and methods of usingPublication Number: US-2023293558-A1Priority Date: 2020-09-01
- Methods of treating mood disordersPublication Number: US-2022241243-A1Priority Date: 2020-02-18
- Methods of treating mood disordersPublication Number: US-11440879-B2Priority Date: 2020-02-18Grant Date: 2022-09-13
- Fluorinated tryptamine compounds, analogues thereof, and methods using samePublication Number: EP-4347559-A1Priority Date: 2021-06-02
- Methods and compositions relating to psychedelics and serotonin receptor modulatorsPublication Number: WO-2022212854-A1Priority Date: 2021-04-01
- Methods and compositions relating to psychedelics and serotonin receptor modulatorsPublication Number: US-2024197681-A1Priority Date: 2021-04-01
- Methods and compositions relating to psychedelics and serotonin receptor modulatorsPublication Number: TW-202304423-APriority Date: 2021-04-01
- Methods and compositions relating to psychedelics and serotonin receptor modulatorsPublication Number: AU-2022246909-A1Priority Date: 2021-04-01



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

References
References
- “GM 2505”. AdisInsight. 5 June 2025. Retrieved 29 July 2025.
- Marek GJ, Makai-Bölöni S, Umbricht D, Christian EP, Winters J, Dvorak D, et al. (2025). “A novel psychedelic 5-HT 2A receptor agonist GM-2505: The pharmacokinetic, safety, and pharmacodynamic profile from a randomized trial healthy volunteer”. Journal of Psychopharmacology 02698811251378512. doi:10.1177/02698811251378512. hdl:1887/4298848. PMID 41099491.
- Hughes Z, Christian E, Dvorak D, Umbricht D, Winters J, Raines S, et al. (December 2023). “ACNP 62nd Annual Meeting: Poster Abstracts P1 – P250: P238. Subjective and Pharmacodynamic Effects of the Novel 5-HT2A Receptor Agonist GM-2505 in Healthy Volunteers Show High Translatability From Rodent Data and Hold Promise for Future Development in Patients With Depression”. Neuropsychopharmacology. 48 (Suppl 1). Springer Science and Business Media LLC: 63–210 (202–203). doi:10.1038/s41386-023-01755-5. PMC 10729595. PMID 38040809.
- Umbricht D, Christian E, Winters J, Raines S, Hughes ZA, Leong W, et al. (2024). “Pharmacokinetic, pharmacodynamic and subjective and effects of the novel 5-HT2A receptor agonist GM-2505 in healthy volunteers”. Neuroscience Applied. 3 104845. doi:10.1016/j.nsa.2024.104845.
- “Бретисилоцин (5F-MET)”. АИПСИН (in Russian). Retrieved 18 March 2026.
- Peplow M (22 June 2024). “Should Next-Generation Psychedelics Skip the Trip?”. Scientific American. Retrieved 20 February 2025.
Gilgamesh is also working on GM-2505, a 5-HT2A agonist that is structurally related to psilocybin and DMT. GM-2505 completed a phase 1 trial late last year and should enter phase 2 for major depressive disorder this year. Its psychedelic effect lasts 60 to 90 minutes — long enough for patients to “explore the altered state of consciousness that might be needed for long-term durable efficacy,” Krugel says, yet within a timeframe that is manageable for healthcare systems. “Personally, I believe that the hallucinogenic effects are an important component, as multiple hallucinogenic compounds have demonstrated durable, transformational changes from a single dose in human studies,” he adds.
- Witkin JM, Golani LK, Smith JL (April 2023). “Clinical pharmacological innovation in the treatment of depression”. Expert Review of Clinical Pharmacology. 16 (4): 349–362. doi:10.1080/17512433.2023.2198703. PMID 37000975.
GM-2505 is a dual-acting compound with both agonist activity at 5-HT 2A receptors and a releaser of 5-HT. […]
- “Methods of treating mood disorders”. Google Patents. 2022. Retrieved 14 November 2024.
- Hughes Z, Klein A, Austin E, Dvorak D, Gatti S, Kiss L, et al. (December 2022). “ACNP 61st Annual Meeting: Poster Abstracts P1 – P270: P254. Gm-2505 is a Novel 5-Ht2a Receptor Agonist and 5-Ht Releaser That Induces Rapid, Robust, and Durable Antidepressant Effects at Doses Associated With Decreased Power in Low Frequency EEG Bands in Rats”. Neuropsychopharmacology. 47 (Suppl 1): 63–219 (209–209). doi:10.1038/s41386-022-01484-1. PMC 9714397. PMID 36456693.
- WO 2022/256554, Wallach J, Dybek M, “Fluorinated Tryptamine Compounds, Analogues Thereof, and Methods Using Same”, published 8 December 2022, assigned to University of the Sciences in Philadelphia[…] Synthesis of N-ethyl-2-(5-fluoro-1H-indol-3-yl)-N-methylethan-1-amine (12) [structure] N-ethyl-2-(5-fluoro-1H-indol-3-yl)-N-methylethan-1-amine (12) was synthesized […] […] Table 1. Selected compounds of the present invention. […] [Compound 12:] […] Table 3. Functional Activity of Compounds at 5-HT2A (Ca2+), 5-HT2B (Ca2+), 5-HT2c (Ca2+), and 5-HT1A (cAMP inhibition) […]
- Hughes Z, Klein A, Dvorak D, Austin E, Kiss L, Marek G, et al. (2023). “22. GM-2505 has Rapid Onset Antidepressant Activity and Causes Dose-Dependent Changes in qEEG With Increasing 5-HT2A Receptor Occupancy”. Biological Psychiatry. 93 (9): S102–S103. doi:10.1016/j.biopsych.2023.02.262.
- Gunther M (31 January 2023). “Gilgamesh Tweaks Known Psychedelics To Improve Therapies”. Lucid News – Psychedelics, Consciousness Technology, and the Future of Wellness. Retrieved 20 February 2025.
- Taylor NP (25 August 2025). “AbbVie tunes in to Gilgamesh’s story, inking $1.2B deal for psychedelic program”. Fierce Biotech. Retrieved 15 October 2025.
- Psychedelic Alpha (25 August 2025). “AbbVie to Acquire Gilgamesh’s Bretisilocin for Up to $1.2B”. Psychedelic Alpha. Retrieved 15 October 2025.
- Halberstadt AL, Geyer MA (2018). “Effect of Hallucinogens on Unconditioned Behavior”. Behavioral Neurobiology of Psychedelic Drugs. Curr Top Behav Neurosci. Vol. 36. pp. 159–199. doi:10.1007/7854_2016_466. ISBN 978-3-662-55878-2. PMC 5787039. PMID 28224459.
- https://iris.who.int/bitstream/handle/10665/380497/9789240107038-eng.pdf “bretisilocinum bretisilocin N-ethyl-2-(5-fluoro-1H-indol-3-yl)-N-methylethan-1-amine serotonin (5-HT2A) receptor agonist”
- “Controlled Drugs and Substances Act”. Department of Justice Canada. Retrieved 19 January 2026.
- Orange Book: List of Controlled Substances and Regulated Chemicals (January 2026) (PDF), United States: U.S. Department of Justice: Drug Enforcement Administration (DEA): Diversion Control Division, January 2026
- Psychedelic Alpha (27 May 2025). “Gilgamesh’s Next-Gen Psychedelic GM-2505 Prints Impressive Results in Phase 2a Major Depressive Disorder Study”. Psychedelic Alpha. Retrieved 29 July 2025.
- Taylor NP (27 May 2025). “Gilgamesh links psychedelic to 94% remission rate in midphase depression trial”. Fierce Biotech. Retrieved 29 July 2025.
- Dunne R (31 May 2025). “Gilgamesh’s psychedelic drug demonstrates exceptional efficacy for treating depression”. Mugglehead Investment Magazine. Retrieved 29 July 2025.
- Psychedelic Access and Research European Alliance (2026-03-19). “Bretisilocin Becomes First Psychedelic in EMA PRIME Scheme for Depression”. Drug Policy Tracker. Retrieved 2026-03-29.
- European Medicines Agency (EMA) (2026-03-18). “New PRIME tools to accelerate development of medicines in the EU”. http://www.ema.europa.eu. Retrieved 2026-03-19.
External links
- 5-Fluoro-MET (Bretisilocin; GM-2505) – Isomer Design
- 5-f-met bretisilocin – Bluelight
- Bretisilocin (5-Fluoro-MET) – r/ResearchChemicals – Reddit Search
| Clinical data | |
|---|---|
| Other names | GM-2505; GM2505; 5-Fluoro-N-methyl-N-ethyltryptamine; 5F-MET; 5-F-MET; 5-Fluoro-MET |
| Routes of administration | Intravenous,[1][2][3][4] intranasal[5] |
| Drug class | Serotonergic psychedelic; Hallucinogen; Serotonin 5-HT2A and 5-HT2C receptor agonist; Serotonin 5-HT2B receptor partial agonist or antagonist; Serotonin releasing agent |
| Legal status | |
| Legal status | Investigational |
| Pharmacokinetic data | |
| Onset of action | IVTooltip Intravenous injection: 10–20 minutes (peak)[2] |
| Elimination half-life | 45 (40–50) minutes[2][3] |
| Duration of action | IVTooltip Intravenous injection: 60–90 minutes[2][6] |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2698331-35-8 |
| PubChem CID | 156836209 |
| ChemSpider | 129221851 |
| ChEMBL | ChEMBL5028766 |
| Chemical and physical data | |
| Formula | C13H17FN2 |
| Molar mass | 220.291 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
//////////bretisilocin, serotonin (5-HT2A) receptor agonist, GM-2505, GM 2505, 5-Fluoro-N-methyl-N-ethyltryptamine, 5F-MET, 5-F-MET, 5-Fluoro-MET, DS425RQ8SX

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










