Home » Uncategorized (Page 164)
Category Archives: Uncategorized
Ondansetron

 ondansetron
ondansetron
Ondansetron hydrochloride dihydrate, cas 99614-01-4, GG-032, SN-307, GR-C505/75,
Ondansetron hydrochloride dihydrate is a serotonin-3 (5-HT3) receptor antagonist. J Org Chem1980, 45, (15): 2938 Heterocycles1997, 45, (10): 2041 EP 0595111 WO 0172716, Ondansetron (INN) (/ɒnˈdænsɛtrɒn/; developed and first marketed by GlaxoSmithKline as Zofran) is a serotonin 5-HT3 receptor antagonist used mainly as an antiemetic (to treat nausea and vomiting), often following chemotherapy. It affects both peripheral and central nerves. Ondansetron reduces the activity of the vagus nerve, which deactivates the vomiting center in the medulla oblongata, and also blocks serotonin receptors in thechemoreceptor trigger zone. It has little effect on vomiting caused by motion sickness, and does not have any effect on dopamine receptors ormuscarinic receptors. Although an effective anti-emetic agent, the high cost of brand-name ondansetron initially limited its use to controlling postoperative nausea and vomiting (PONV) and chemotherapy-induced nausea and vomiting (CINV). The active ingredient in ZOFRAN Tablets and ZOFRAN Oral Solution is ondansetron hydrochloride (HC1) as the dihydrate, the racemic form of ondansetron and a selective blocking agent of the serotonin 5-HT3 receptor type. Chemically it is (±) 1, 2, 3, 9-tetrahydro-9-methyl-3-[(2-methyl-lH-imidazol-l-yl)methyl]-4H-carbazol-4-one, monohydrochloride, dihydrate. It has the following structural formula:
 |
The empirical formula is C18H19N3O•HCl•2H2O, representing a molecular weight of 365.9. Ondansetron HC1 dihydrate is a white to off-white powder that is soluble in water and normal saline. The active ingredient in ZOFRAN ODT Orally Disintegrating Tablets is ondansetron base, the racemic form of ondansetron, and a selective blocking agent of the serotonin 5-HT3 receptor type. Chemically it is (+) 1, 2, 3, 9-tetrahydro-9-methyl-3-[(2-methyl-lH-imidazol-l-yl)methyl]-4H-carbazol-4-one. It has the following structural formula:
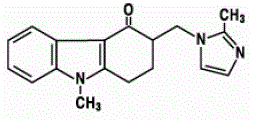 |
The empirical formula is C18H19N3O representing a molecular weight of 293.4. The 5-HT3 receptor antagonists are the primary drugs used to treat and prevent chemotherapy-induced nausea and vomiting (CINV). A common use case is to give them intravenously about 30 minutes before commencement of a chemotherapy treatment.
Ondansetron is used off-label to treat morning sickness and hyperemesis gravidarum of pregnancy. A cohort study of over 600,000 pregnancies in Denmark found that ondansetron administration during pregnancy is not associated with a significantly increased risk of spontaneous abortion,stillbirth, major birth defect, preterm birth, low birth weight, or small for gestational age. However, in practice, ondansetron is typically used after trials of other drugs have failed.
Ondansetron is one of several anti-emetic agents used during the vomiting phase of cyclic vomiting syndrome. Trials in emergency department (ED) settings support the use of ondansetron to reduce vomiting associated with gastroenteritis and dehydration.A retrospective review found that it was used commonly for this purpose, being administered in over 58% of cases. Its use reduced hospital admissions, but was also associated with higher rates of return visits to the ED. Furthermore, patients who had initially received ondansetron were more likely to be admitted on the return visit than patients who had not received the drug. However, this effect may simply be due to the agent being used more frequently in patients who present with more severe illness. Its use was not found to mask serious diagnoses.
Ondansetron was developed around 1984 by scientists working at Glaxo’s laboratories in London. It is in both the imidazole and carbazole families of heterocyclic compounds. After several attempts the company successfully filed for U.S. patent protection for the drug in 1986 and was granted in June 1988 while a use patent was granted in June 1988. A divisional use patent was granted on November 26, 1996. Ondansetron was granted FDA approval as Zofran in January 1991. Glaxo did pediatric research on Zofran’s uses, and gained a patent extension as a result, extending U.S. exclusivity until December 24, 2006. The FDA subsequently approved the first generic versions in December 2006, with marketing approval granted to Teva Pharmaceuticals USA and SICOR Pharmaceuticals.
Ondansetron is marketed by GlaxoSmithKline (GSK) under the trade name Zofran. Other manufacturers include Pfizer Injectables (Ondanzetron), Opsonin Pharma Bangladesh (Anset), Strativa Pharmaceuticals (Zuplenz), Indswift Ltd. (Ondisolv), Cipla Ltd. (Emeset), Gedeon Richter Ltd. (Emetron), Korea United Pharmaceuticals (Emodan), Zentiva a.s. (Ondemet), Strides Arcolab (Setronax), Emistat (Unimed and Unihealth Bangladesh Ltd.)Glenmark Generics Ltd. (India) (Ondansetron) and Novell Pharmaceutical Laboratories (Ondavell). On May 29, 2006, Baxter Healthcare received tentative approval to market its own label of Ondansetron Injection, USP, 8 mg/50 mL and 32 mg/50 mL iso-osmotic sodium chloride solution, beginning upon expiration of GSK’s patent later that year.
In 1997, ondansetron was the subject of a meta-analysis case-study published in the British Medical Journal. Researchers examined 84 trials, with 11,980 patients receiving ondansetron, published between 1991 and September 1996. Intravenous ondansetron 4 mg versus placebo was investigated in 16 reports and three further reports which had been duplicated six times. The number needed to treat (NNT) to prevent vomiting within 24 hours was 9.5, with 95% confidence interval 6.9 to 15, in the 16 non-duplicated reports. In the three duplicated reports, the NNT was significantly lower at 3.9 (3.3 to 4.8) with P<0.00001. When all 25 reports were combined the apparent number needed to treat improved to 4.9 (4.4 to 5.6). Inclusion of duplicate reports led to a 23% overestimation of ondansetron’s antiemetic efficacy. In addition, the authors found that the covert duplication of reports on ondansetron was not easy to detect, because of lack of cross-referencing between papers, and that reports containing duplicate findings were cited in eight reviews of the drug. Their analysis was a subject of an editorial in the Journal of the American Medical Association in 1999. ………………………
patents
AU 8538097; BE 0901576; CH 664152; ES 8609309; ES 8708224; ES 8801247; FR 2561244; GB 2153821; JP 1985214784
……………..
(±) l,2,3,9-Tetrahydro-9-methyl-3-[2-methyl-lh-imidazol-l-yl)methyl]-4h- carbazol-4-one having the molecular structure
is a selective 5-HT3 receptor antagonist. It is known by the generic nameondansetron. Ondansetron reduces nausea in patients undergoing chemotherapy. Grunberg, S.M.; Hesketh, P.J. “Control of Chemotherapy-Induced emesis” N. Engl. J. Med. 1993, 329, 1790-96. Ondansetron is indicated for prevention of nausea and vomiting associated with some cancer chemotherapy, radiotherapy and postoperative nausea and/or vomiting.
Several chemical processes are known from the literature for the synthesis ofondansetron. GB-Pat. 2 153 821 and 2 192 885 describe syntheses starting from carbazolone derivative, and EP-Pat. 595 111 as well as a Hungarian patent application ( P 00-01287 ) give detailed information about some different chemical procedures.
Ondansetron is currently available as an anti-emetic agent, particularly in cancer chemotherapy, and in some other uses such as anti-depressive, anti- migraine and anti-psychotic. It is commonly used in the alleviation of cognitive disorders as in Alzheimer disease, in treatment of rhinitis, psychiatric disorders and for increased vigilance and for control of dependence on narcotics.
U.S. Patent No. 4,695,578, assigned to the Glaxo Group Limited, describes a process of preparing ondansetron and uses thereof. However, ondansetronprepared according to said process contains impurities and by-products such as l,2,3,9-tetrahydro-9-methyl-3-methylene-4H-carbazol-4-one.
The hydrochloride salt of ondansetron is generally safe for oral administration to a patient without causing irritation or other adverse effect. The hydrochloride salt is marketed in tablet form and in oral solution form under the brand name Zofran®. The tablet’s active ingredient is a dihydrate of ondansetronhydrochloride containing two molecules of bound water in ondansetronhydrochloride’ s crystal lattice. The present invention relates to the solid state physical properties of ondansetron hydrochloride. These properties can be influenced by controlling the conditions under which the hydrochloride salt is obtained in solid form. Solid state physical properties include, for example, the flowability of the milled solid. Flowability affects the ease with which the material is handled during processing into a pharmaceutical product. When particles of the powdered compound do not flow past each other easily, a formulation specialist must take that fact into account in developing a tablet or capsule formulation, which may necessitate the use of glidants such as colloidal silicon dioxide, talc, starch or tribasic calcium phosphate.
These important physical characteristics are influenced by the conformation and orientation of molecules in the unit cell, which defines a particular polymorphic form of a substance. Llacer and coworkers have postulated that different spectroscopic characteristics of samples of ondansetron free base prepared differently could be attributable to two different configurations about the methylene bridge between the 1, 2, 3, 9-tetrahydrocarbazol-4-one ring and the imidazole ring. Llacer, J.M.; Gallardo, V.; Parera, A. Ruiz, M.A. InternJ.Pharm., 177, 1999, 221-229.
(±)1,2,3,9-Tetrahydro-9-methyl-3-[2-methyl-1h-imidazol-1-yl)methyl]-4h-carbazol-4-one having the molecular structure
is a selective 5-HT3 receptor antagonist. It is known by the generic nameondansetron. Ondansetron reduces nausea in patients undergoing chemotherapy. Grunberg, S. M.; Hesketh, P. J. “Control of Chemotherapy-Induced emesis” N. Engl. J. Med. 1993, 329, 1790-96. Ondansetron is indicated for prevention of nausea and vomiting associated with some cancer chemotherapy, radiotherapy and postoperative nausea and/or vomiting.
The hydrochloride salt of ondansetron is generally safe for oral administration to a patient without causing irritation or other adverse effect. The hydrochloride salt is marketed in tablet form and in oral solution form under the brand name Zofran®. The tablet’s active ingredient is a dihydrate of ondansetronhydrochloride containing two molecules of bound water in ondansetronhydrochloride’s crystal lattice.
The present invention relates to the solid state physical properties ofondansetron hydrochloride. These properties can be influenced by controlling the conditions under which the hydrochloride salt is obtained in solid form. Solid state physical properties include, for example, the flowability of the milled solid. Flowability affects the ease with which the material is handled during processing into a pharmaceutical product. When particles of the powdered compound do not flow past each other easily, a formulation specialist must take that fact into account in developing a tablet or capsule formulation, which may necessitate the use of glidants such as colloidal silicon dioxide, talc, starch or tribasic calcium phosphate.
Another important solid state property of a pharmaceutical compound is its rate of dissolution in aqueous fluid. The rate of dissolution of an active ingredient in a patient’s stomach fluid can have therapeutic consequences since it imposes an upper limit on the rate at which an orally-administered active ingredient can reach the patient’s bloodstream. The rate of dissolution is also a consideration in formulating syrups, elixirs and other liquid medicaments. The solid state form of a compound may also affect its behavior on compaction and its storage stability.
These important physical characteristics are influenced by the conformation and orientation of molecules in the unit cell, which defines a particular polymorphic form of a substance. Llacer and coworkers have postulated that different spectroscopic characteristics of samples ofondansetron free base prepared differently could be attributable to two different configurations about the methylene bridge between the 1,2,3,9-tetrahydrocarbazol-4-one ring and the imidazole ring. Llacer, J. M.; Gallardo, V.; Parera, A. Ruiz, M. A. Intern.J.Pharm., 177, 1999, 221-229.
A crystalline polymorphic form of a compound may exhibit different thermal behavior from amorphous material or another polymorphic form. Thermal behavior is measured in the laboratory by such techniques as capillary melting point, thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC) and can be used to distinguish some polymorphic forms from others. A particular polymorphic form may also give rise to distinct spectroscopic properties that may be detectable by powder X-ray crystallography, solid state 13C NMR spectrometry and infrared spectrometry. There is a wide variety of techniques that have the potential of producing different crystalline forms of a compound. Examples include crystallization, crystal digestion, sublimation and thermal treatment.
U.S. Pat. No. 4,695,578, Example 1a, discloses a preparation ofondansetron by alkylation of 2-methylimidazole with 2,3,4,9 tetrahydro-N,N,N,9-tetramethyl-4-oxo-1H-carbazole-3-methanaminium iodide. In this example,ondansetron was isolated as its hydrochloride salt by suspending the reaction product in a mixture of absolute ethanol and ethanolic HCl, warming the suspension, filtering to remove impurities and precipitating the hydrochloride salt with dry ether.
In Example 10 of the ‘578 patent, ondansetron free base was converted into a hydrochloride salt dihydrate by dissolving the free base in a mixture of isopropanol and water and treating it with concentrated hydrochloric acid. After filtration at elevated temperature, ondansetron was driven out of solution by adding additional isopropanol and cooling. The dihydrate was obtained as a white crystalline solid by recrystallizing it from a 6:10 mixture of water and isopropanol. Ondansetron hydrochloride dihydrate obtained by following Example 10 of the ‘578 patent is denominated Form A in this disclosure. Powdered samples of Form A produce a powder X-ray diffraction pattern essentially the same as the pattern shown in FIG. 1.
U.S. Pat. No. 5,344,658 describes ondansetron having a particular particle size distribution and the use of such ondansetron in a pharmaceutical composition. The particle size of ondansetron hydrochloride dihydrate obtained by crystallization from a solvent is reduced by desolvating them, e.g. by heating, and then exposing the desolvated crystals to a humid atmosphere. A collection of crystals obtained by this particle size reduction process is said to consist exclusively of crystals of less than 250 micron size and to contain 80% or more crystals of less than 63 microns. Crytals size was determined by air jet seive analysis.
According to the ‘658 patent, ondansetron hydrochloride dehydrate having the same particle size distribution as the rehydrated ondansetron hydrochloride also is provided as part of that invention. Since only one process for dehydratingondansetron hydrochloride is described in the ‘658 patent, a dehydrate is evidently the intermediate compound that is rehydrated in the particle size reduction process.
U.S. Pat. Nos. 4,695,578 and 5,344,658 are incorporated herein by reference.
U.S. Pat. No. 4,695,578 (‘578 patent) discloses a process for preparingondansetron hydrochloride dihydrate having a large particle size (e.g., less than about 60% of the particles are smaller than 250 μm). The ‘578 patent process involves the step of cooling a solution of ondansetron hydrochloride, isopropanol, and water, optionally followed by an additional step of recrystallizing from a mixture of water and isopropanol.
U.S. Pat. No. 5,722,720 (the ‘720 patent) discloses a non-conventional technique for reducing particle size. In particular, the ‘720 patent discloses a multistep process in which ondansetron hydrochloride dihydrate is first dried at elevated temperature and reduced or atmospheric pressure, and is then cooled to ambient temperature. The process requires the heating step to be performed until the ondansetron hydrochloride dihydrate is desolvated, and requires the cooling step to be performed until the ondansetron hydrochloride is rehydrated to form ondansetron hydrochloride dihydrate.
The ‘720 patent process has several disadvantages. First, the ‘720 patent process requires a prolonged time period (i.e., 16-24 hours) for the drying/desolvating step, plus an additional prolonged time period for the cooling/rehydrating step. Second, the ‘720 patent process requires vigorous and carefully controlled drying conditions. For example, when the drying step is performed at 48-52° C., a reduced pressure of 100-200 torr is required. When the drying step is performed at ambient pressure, an elevated temperature of 1 00° C. is required.
An overview of the key routes to the best selling 5-membered ring heterocyclic pharmaceuticals
 Corresponding author email
Corresponding author emailA completely different strategy was used in the synthesis of the serotonin 5-HT3 receptor antagonist ondansetron (119, Zofran). In this synthesis a palladium-catalysed intramolecular Heck-reaction was used to build the tricyclic indole core in a short and concise sequence (Scheme 26) [35,36].
![[1860-5397-7-57-i26]](https://i0.wp.com/beilstein-journals.org/bjoc/content/inline/1860-5397-7-57-i26.png)
Alternatively, a direct Fischer indole synthesis between phenylmethyl hydrazine and a cyclic 1,3-dione derivative could be utilised to prepare the desired fully substituted tricyclic core of ondansetron (Scheme 27) [37].
![[1860-5397-7-57-i27]](https://i0.wp.com/beilstein-journals.org/bjoc/content/inline/1860-5397-7-57-i27.png)
- 35………..Godfrey, N.; Coates, I. H.; Bell, J. A.; Humber, D. C.; Ewan, G. B. Process for Preparing N-Heterocyclic Compounds. U.S. Patent 4,957,609, Sept 18, 1990.
- 36…………Iida, H.; Yuasa, Y.; Kibayashi, C. J. Org. Chem. 1980, 45, 2938–2942. doi:10.1021/jo01303a003
- Oxford, A. W.; Eldred, C. D.; Coates, I. H.; Bell, J. A.; Humber, D. C.; Ewan, G. B. Process for Preparing Tetrahydrocarbazolones. U.S. Patent 4,739,072, April 19, 1988.
OLMESARTAN

Mol. mass 558.585 g/mol
Olmesartan medoxomil (trade names: Benicar in the US, Olmetec in EU, Canada and Japan, WinBP, Golme in India, Erastapex in Egypt) is an angiotensin II receptor antagonist used to treat high blood pressure.
Olmesartan is indicated for the treatment of hypertension. It may be used alone or in combination with other antihypertensive agents.[1] The U.S. Food and Drug Administration (FDA) has determined that the benefits of Benicar continue to outweigh its potential risks when used for the treatment of patients with high blood pressure according to the drug label.[2]
Angiotensin-II receptor antagonists should be used with caution in renal artery stenosis. Monitoring of plasma-potassium concentration is advised, particularly in the elderly and in patients with renal impairment; lower initial doses may be appropriate in these patients. Angiotensin-II receptor antagonists should be used with caution in aortic or mitral valve stenosis and in hypertrophic cardiomyopathy. Those with primary aldosteronism, and Afro-Caribbean patients (particularly those with left ventricular hypertrophy), may not benefit from an angiotensin-II receptor antagonist.
Structure
The olmesartan molecule includes one tetrazole group (a 5-member heterocyclic ring of four nitrogen and one carbon atom) and one imidazole group (a 5-membered planar heterocyclic aromatic ring of two nitrogen and three carbon atoms, classified as an alkaloid).
Olmesartan as the starting material can be easily produced according to the method described in Japanese Examined Patent Application (Kokoku) No. Hei 7-121918 (Japanese Patent No. 2082519 ; US Patent No. 5616599 ) or the like
Olmesartan is a prodrug that works by blocking the binding of angiotensin II to the AT1 receptors in vascular muscle; it is therefore independent of angiotensin II synthesis pathways, unlike ACE inhibitors. By blocking the binding rather than the synthesis of angiotensin II, olmesartan inhibits the negative regulatory feedback on renin secretion. As a result of this blockage, olmesartan reduces vasoconstriction and the secretion of aldosterone. This lowers blood pressure by producing vasodilation, and decreasing peripheral resistance.
The usual recommended starting dose of olmesartan is 20 mg once daily. The dose may be increased to 40 mg after two weeks of therapy, if further reduction in blood pressure is desirable. Doses above 40 mg do not appear to have greater effect, and twice-daily dosing offers no advantage over the same total dose given once daily.[1] No adjustment of dosage is typically necessary for advanced age, renal impairment, or hepatic dysfunction. For patients with possible depletion of intravascular volume (e.g., patients treated with diuretics), olmesartan should be initiated with caution; consideration should be given to use of a lower starting dose in such cases.[1] If blood pressure is not controlled by Benicar alone, a diuretic may be added. Benicar may be administered with other antihypertensive agents. Benicar may be administered with or without food.[1]
Olmesartan and Sevikar HCT is marketed worldwide by Daiichi Sankyo, in India by Abbott Healthcare Pvt. Ltd. under the trade name WinBP, by Zydus Cadila under the trade name Olmy, by Ranbaxy Laboratories Ltd. under the trade name Olvance, and in Canada by Schering-Plough as Olmetec. Benicar HCT is the brand name of a medication containing olmesartan medoxomil in combination with hydrochlorothiazide, a thiazide diuretic. Three dosage combinations are available: 20 mg or 40 mg of olmesartan medoxomil combined with 12.5 mg of hydrochlorothiazide, or 40 mg of olmesartan medoxomil combined with 25 mg of hydrochlorothiazide. Benitec H, another medication containing olmesartan medoxomil and hydrochlorothiazide, is marketed by GlaxoSmithKline in India. In Poland as Olesartan Medoxomil by TEVA, Olimestra and Co-Olimestra (with HCTZ) by Miklich Lab., Elestar (with amlodipine) and Elestar HCT (with amlodipine, HCTZ) by Menarini, Sevikar HCT (with amoldipine, HCTZ) by Aiichi Sankyo.
Research
Two clinical studies (MORE [6] and OLIVUS [7])[8] report that Benicar reduced arterial plaque during therapy for high-blood pressure (hypertension).
- RxList Inc. (5 July 2007). “Benicar (olmesartan medoxomil)”. RxList Inc. Retrieved 22 July 2010.
- “FDA Alert: Benicar (olmesartan): Ongoing Safety Review”. Drugs.com. Retrieved 2013-06-27.
- Angiotensin II receptor blocker induced fetopathy: 7 cases. Hünseler C, Paneitz A, Friedrich D, Lindner U, Oberthuer A, Körber F, Schmitt K, Welzing L, Müller A, Herkenrath P, Hoppe B, Gortner L, Roth B, Kattner E, Schaible T. Klin Padiatr. 2011 Jan;223(1):10-4. Epub 2011 Jan 26.
- “BENICAR Prescribing Information”. Retrieved 2011-01-20.
- Rubio-Tapia, Alberto; Herman, Margot L.; Ludvigsson, Jonas F.; Kelly, Darlene G.; Mangan, Thomas F.; Wu, Tsung-Teh; Murray, Joseph A. (NaN undefined NaN). “Severe Spruelike Enteropathy Associated With Olmesartan”. Mayo Clinic Proceedings 87 (8): 732–738. doi:10.1016/j.mayocp.2012.06.003.
- as referenced in http://www.medicalnewstoday.com/releases/91285.php “Olmetec(R) Is First Angiotensin Receptor Blocker (ARB) To Suggest Atherosclerosis Regression (In Hypertensives With Cardiovascular Risk), UK”
- Cardiovascular Research Foundation (2008, October 16). Drug May Reduce Coronary Artery Plaque. ScienceDaily. Retrieved January 5, 2013, from http://www.sciencedaily.com /releases/2008/10/081012121318.htm
- (Review) R Preston Mason, Cardiovascular Division, Brigham and Women’s Hospital, Harvard Medical School, Boston, MA, and Elucida Research, Beverly, MA, USA. Vascular Health and Risk Management, Dovepress, Published Date June 2011 Volume 2011:7 Pages 405 – 416. Optimal therapeutic strategy for treating patients with hypertension and atherosclerosis: focus on olmesartan medoxomil. Retrieved January 5, 2013, from http://www.dovepress.com/optimal-therapeutic-strategy-for-treating-patients-with-hypertension-a-peer-reviewed-article-VHRM
4-Isopropenyl-2-propyl-1-[[2′-(1H-tetrazol-5-yl)biphenyl-4-yl]methyl]imidazole-5-carboxylic acid [olmesartan dehydrate, compound 34b described in J. Med. Chem., 39, 323-338 (1996)]
- Daiichi-Sankyo Benicar page
- Benicar HCT from RXlist.com
- Mayo Clinic Proceedings vol.87 Issue 8, pages 732-738

-
Olmesartan medoxomil is known by two names,
- (a)(5-Methyl-2-oxo-1,3-dioxolen-4-yl)methyl 4-(1-hydroxy-1-methylethyl)-2-propyl-1-[4-(2(tetrazole-5yl)phenyl]phenyl]methylimidazole-5-carboxylate
- (b)4-(1-Hydroxy-1-methylethyl)-2-propyl-1-[[2′-(1H-tetrazol-5-yl)[1,1′-biphenyl]-4-yl]methyl]-1H-imidazol-5-carboxylic acid (5-methyl-2-oxo-1,-3 dioxol-4-yl)methyl ester, and has a CAS No. [144689-63-4].
-
The structural formula is represented below:Olmesartan medoxomil, which is an angiotensin II receptor antagonist, is useful as an active ingredient in medicaments for treatment and prophylaxis of hypertension (for example, Patent documents 1 to 5 or Non-patent document 1 and 2). Techniques for producing high-purity olmesartan medoxomil are necessary for use of olmesartan medoxomil as a medicament.
-
- Patent document 1: Japanese Examined Patent Application (Kokoku) No. Hei 7-121918 (Japanese Patent No. 2082519 )
- Patent document 2: US Patent No. 5616599
- Patent document 3: International Patent Publication No. WO2006/029056
- Patent document 4: International Patent Publication No. WO2006/029057
- Patent document 5: International Patent Publication No. WO2006/073519
-
- Non-patent document 1: J. Med. Chem., 39, 323-338 (1996)
- Non-patent document 2: Annu. Rep. Sankyo Res. Lab. (Sankyo Kenkyusho Nempo) 55, 1-91 (2003)
-
US 5616599 describes a process for the preparation of olmesartan medoxomil as follows.
-
[0006]4-(1-hydroxyl-1-methylethyl)-2-propyl imidazole-5carboxylic acid is reacted with 5-methyl-2-oxo-1, 3-dioxolene-4-yl)methyl chloride using N,N-diisopropylethyl amine as base in N,N-dimethyl acetamide at 60°C to give (5-methyl-2-oxo-1,3-dioxolen-4-yl)methyl4-(1-hydroxy-1-methylethyl)-2-propyl imidazole-5-carboxylate. The resulting product is coupled with N-(triphenylmethyl)-5-[4′-(bromomethyl)biphenyl-2-yl]tetrazole [herein referred to as TTBB] at 60°C in N, N-dimethyl acetamide using potassium carbonate as base to give protected olmesartan medoxomil. The protected olmesartan medoxomil is deprotected using 75 % acetic acid to give olmesartan medoxomil.
-
This process involves column chromatographic purification of intermediates which is not desirable on commercial scale operation.
-
US 5616599 describes another process for the preparation of olmesartan Medoxomil which involves addition of methyl Magnesium chloride on diethyl 2-propylimidazole-4, 5-dicarboxylate in tetrahydrofuran at -30 to -20°C to give ethyl-4-(1-hydroxy-1-methylethyl)-2-propylimidazole -5-carboxylate, which is coupled with TTBB using sodium hydride as base in N, N-dimethylformamide at 60°C to give ethyl-4-(1-hydroxy-1-methylethyl)2-propyl-1-[[2′-[2-(triphenylmethyl)-2H-tetrazol-. 5yl]biphenyl-4-yl]methyl]imidazole-5-carboxylate. The product thus formed is hydrolyzed using lithium hydroxide monohydrate as base in 1,4-dioxane at 5-10°C to give lithium salt of 4-(1-hydroxy-1-methylethyl)-2-propyl-1-[[2′-[2-(triphenylmethyl)-2H-tetrazol-5yl]biphenyl-4-yl]methyl]imidazole-5-carboxylic acid, which is then coupled with 5-methyl-2-oxo-(1,3-dioxolene-4-yl)methyl chloride using K2CO3 as base in N,N-dimethylacetamide at 50°C to give trityl protected olmesartan medoxomil which on deprotection using 75% acetic acid gives Olmesartan Medoxomil.
-
During the condensation of ethyl-4-(1-hydroxy-1-methylethyl)-2-propylimidazole -5-carboxylate, with TTBB using sodium hydride as base in N, N-dimethylformamide, various impurities are formed, and isolation of the product involves extractive workup.

 Corresponding author email
Corresponding author emailThe structurally related imidazole core of olmesartan is formed in a different fashion (Scheme 36). Condensation between diaminomaleonitrile and trimethyl orthobutyrate furnishes the trisubstituted imidazole 181 in high yield [53,54]. Acid-mediated nitrile hydrolysis followed by esterification results in the corresponding diester unit 182. Treatment of 182 with four equivalents of methylmagnesium chloride in a mixture of diethyl ether and dichloromethane selectively provides tertiary alcohol 183. In subsequent steps this imidazole is alkylated with the tetrazole containing biphenyl appendage, followed by ester hydrolysis and alkylation of the resulting carboxylate with 4-(chloromethyl)-5-methyl-2-oxo-1,3-dioxole to yield olmesartan (Scheme 36).
- 53…….Yanagisawa, H.; Fujimoto, K.; Amemiya, Y.; Shimoji, Y.; Kanazaki, T.; Koike, H.; Sada, T. Angiotensin II Antagonist 1-Biphenylmethylimidazole Compounds and their Therapeutic Use. U.S. Patent 5,616,599, April 1, 1997.
Return to citation in text: [1] - 54……….Yanagisawa, H.; Amemiya, Y.; Kanazaki, T.; Shimoji, Y.; Fujimoto, K.; Kitahara, Y.; Sada, T.; Mizuno, M.; Ikeda, M.; Miyamoto, S.; Furukawa, Y.; Koike, H. J. Med. Chem. 1996, 39, 323–338. doi:10.1021/jm950450f
Return to citation in text: [1]
Reacting ethyl-4-(1-hydroxy-1-methylethyl)-2-propylimidazole-5-carboxylate with N-(Triphenylmethyl)-5-[4′-(bromomethyl)biphenyl-2- yl]tetrazole in an organic solvent in presence of a base and a phase transfer catalyst in non-aqueous system to give after workup, ethyl-4-(1-hydroxy-1-methylethyl)-2-propyl-1-[[2′-[2-(triphenylmethyl)-2H-tetrazol-5yl]biphenyl-4-yl]methyl]imidazole-5-carboxylate, which is further processed, by following improved reaction conditions in three steps to provide substantially pure [HPLC purity 99.3 to 99.7 %] olmesartan medoxomil.
-
To a 3M solution of MeMgCl(55.86 g, 0.74 mol) in tetrahydrofuran was added a solution of diethyl 2-propyl imidazole- 4,5-dicarboxylate (50 g,0.19 mol) in tetrahydrofuran (200 ml) at -10 to 0°C under N2 atmosphere. The mixture was stirred at -5 to 0°C for 10 minutes. Reaction mass was quenched into 400 ml 25 % ammonium chloride solution followed by extraction with ethyl acetate (300 ml). The organic phase was separated, washed with brine, dried over Na2SO4, and concentrated in vacuo to give a syrup, which was crystallized using diisopropyl ether.
Yield: 85-90 %,
Purity by HPLC: 88-93 %.
1H-NMR (CDCl3) δ: 7.8-8.1 (s, 1H), 5.8(s, 1H)., 4.35(q, 2H), 2.68(t, 2H), 1.78(m, 2H), 1.61(s, 6H), 1.36(t, 3H), 0.96(t, 3H).
Example-2Preparation of Ethyl-4-(1-hydroxy-1-methylethyl)-2-propyl-1-[[2′-[2-(triphenylaiethyl)-2H-tetrazol-5yl]biphenyl-4-yl]methyl]imidazole
- -5-
carboxylate
-
Mixture of Ethyl-4-(1-hydroxy-1-methylethyl)-2-propylimidazole -5-carboxylate (41 g, 0.17 mol), potassium carbonate (47g, 0.34 mol) and tetrabutylammonium bromide (4.9 g, 0.01 mol) in acetone was stirred at room temperature for 1hr. Then TTBB (93% Purity, 92.89g, 0.15 mol) was charged, refluxed for 14hrs. Potassium salts were filtered off from the reaction mass and the filtrate was charcoalised for 1hr. It was filtered over celite bed and the filtrate was distilled off completely to get a semi solid mass. 250 ml of Methanol was added to the residue and stirred for 2-3 hrs to give a solid product, which was filtered and washed with chilled methanol and dried.
Yield: 80-85%,
Purity by HPLC: 85-90%.
1H-NMR (CDCl3) δ: 7.8-8.1 (m, 1H), 6.7-7.61 (m, 22H), 5.78 (bs, 1H), 5.38(s, 2H), 4.12 (q, 2H), 2.52 (t, 2H), 1.64(s, 6H), 1.5-1.8(m, 2H), 1.08(t, 3H), 0.88(t, 3H).
Example-3Preparation of lithium salt of 4-(1-hydroxy-1-methylethyl) 2-propyl-1-[[2′-[2-(triphenylmethyl)-2H-tetrazol-5yl] biphenyl-4-yl] methyl] imidazole -5-carboxylic acid
-
To a solution of Ethyl-4-(1-hydroxy-1-methylethyl) 2-propyl- 1-[[2′-[2-(triphenylmethyl)-2H-tetrazol-5yl]biphenyl-4-yl]methyl]imidazole-5-carboxylate (105 g, 0.14 mol) in tetrahydrofuran , was added LiOH.H2O (7.8 g, 0.18 mol) solution below 10°C. The reaction mixture was stirred at room temperature for 15 hours. Reaction mass was concentrated under vacuum at 35°C to 1/4 th of its volume. 300 ml of ethyl acetate and NaCl (130 g) were added to the residue under stirring. The organic phase was separated, dried over sodium sulphate and concentrated under vacuum to get the product. The crude product was taken as such to the next stage.
Example-4Preparation of trityl protected olmesartan medoxomil
-
To the solution of lithium salt of 4-(1-hydroxy-1-methylethyl) 2-propyl- 1-[[2′-[2-(triphenylmethyl)-2H-tetrazol-5yl] biphenyl-4-yl] methyl] imidazole -5-carboxylic acid , (97 g, 0.13 mol) in N, N-dimethyl acetamide(200 ml) was added triethylamine(12.7 g, 0.12 mol), stirred at room temperature for 0.5 hours. 5-methyl-2-oxo-(1,3-dioxolene-4-yl)methyl chloride (85% purity, 37.3 g, 0.25 mol) was added below 10°C. The mixture was stirred at 50-55°C for 4 hours, checked TLC. Dichloromethane (400 ml) and chilled water (500 ml) were added under stirring. The organic phase was separated, given brine wash (50 ml), dried over sodium sulphate and concentrated under vacuum to get a residue. To the residue methanol was added, stirred for 1hr, cooled to 5-10°, filtered and washed with chilled methanol and dried.
Yield: 75-80%,
Purity by HPLC: 96-98%.
1H-NMR (CDCl3) δ: 7.87(d, 1H), 6.90-7.52(m, 20H), 6.68(d, 2H), 5.61(s, 1H), 5.3(s, 2H), 4.7(s, 2H), 2.54(t, 2H), 1.97(s, 3H), 1.6-1.75(m, 2H), 1.62(s, 6H), 0.87(t, 3H).
Example-5Preparation of olmesartan medoxomil
-
To the suspension of trityl protected olmesartan medoxomil (50g, 0.06 mol) in 250 ml 75% acetic acid was stirred at 50-55°C for 1.5hrs and cooled to 5-10°C. The by-product trityl alcohol was filtered off and washed with 75% acetic acid. The filtrate was concentrated under vacuum to get syrup, which was crystallized using isopropyl alcohol.
Yield: 85-88%,
Purity by HPLC: 95-98%.
The material was further purified with ethyl methyl ketone. It was filtered and washed with ethyl methyl ketone and dried to give substantially pure olmesartan medoxomil.
Yield: 70-75%,
Purity by HPLC: 99.3-99.7%.
1H-NMR (CDCl3) δ: 7.81(dd, 1H), 7.43-7.6(m, 3H), 7.09d, 2H), 6.79(d, 2H), 5.41(s, 1H),4.95(s, 1H), 2.56(t, 3H), 2.17(s, 3H), 1.58-1.69(m, 2H), 1.58(s, 6H), 0.92(t,3H).
Ipragliflozin

Ipragliflozin
ASP-1941 , 1(S)-[3-(1-benzothien-2-ylmethyl)-4-fluorophenyl]-1-deoxy-beta-D-glucopyranose L-proline cocrystal

Kotobuki (Originator)
| (1S)-1,5-Anhydro-1-C-[3-[(1-benzothiophen-2-yl)methyl]-4-fluorophenyl]-D-glucitol |
| Molecular Formula | C21H21FO5S | |
| Molecular Weight | 404.45 | |
| CAS Registry Number | 761423-87-4 |
Ipragliflozin (formerly ASP1941) has been filed in Japan on the back of phase III trials which showed that it could provide significant reductions in glycated haemoglobin levels (HbA1c) levels – a marker of glucose control over time – compared to placebo
According to Astellas’ latest R&D pipeline update in February 2013, Astellas is developing ipragliflozin only in Japan. The same document in August 2012 indicated it was also carrying out phase II studies with the drug in the US and Japan.
Astellas Pharma Inc.: Submits Application for Marketing Approval of
Ipragliflozin (ASP1941), SGLT2 Inhibitor for Treatment of
Type 2 Diabetes, in Japan
TOKYO, March 13, 2013 – Astellas Pharma Inc. (“Astellas”; Tokyo:4503; President and CEO:
Yoshihiko Hatanaka) announced today that it has submitted a market authorization application for aSGLT2 inhibitoripragliflozin (generic name; development code: ASP1941) to the Ministry of Health, Labour and Welfare in Japan seeking an approval forthe indication of type 2 diabetes.
Ipragliflozin is a selective SGLT2 (sodium-glucose co-transporter 2)inhibitor discovered through research collaboration with Kotobuki Pharmaceutical Co., Ltd. SGLTs are membrane proteins that
exist on the cell surface and transfer glucose into cells. SGLT2 is a subtype of the sodium-glucose co-transporters and plays a key role in the reuptake of glucose in the proximal tubule of the kidneys.
Ipragliflozin reduces blood glucose levels by inhibiting the reuptake of glucose.
In the Phase III pivotal study in monotherapy for type 2 diabetesin Japan, ipragliflozin
demonstrated significant decreases of HbA1c, an index of glycemic control, in change from baseline compared to placebo. Based on the safety resultsin this study, ipragliflozin was safe and well tolerated. Patients with type 2 diabetes generally need combination therapy, so it is important
for a novel oral hypoglycemic agent to be safe to use with existing diabetes therapies. In this regard, Astellas has conducted six Phase III studies to investigate the safety and efficacy of ipragliflozin
used in combination with other hypoglycemic agentsfor a long term period. In these Phase IIIstudies, effectiveness and favorable safety of ipragliflozin was confirmed even in combination with
other hypoglycemic agents.
Astellas expects to provide an additional therapeutic option and further contribute to the treatment of type 2 diabetes by introducing ipragliflozin, an oral hypoglycemic agent with a novel mechanism
of action, into the Japanese market.
About Type 2 Diabetes
Diabetes (medically known as diabetes mellitus) is a disorder in which the body has difficulty regulating its blood glucose (sugar) level. There are two major types of diabetes: type 1 and type 2.
Type 2 diabetes (formerly called non-insulin-dependent diabetes mellitus or adult-onset diabetes) is a disorder that is characterized by high blood glucose in the context of insulin resistance and relative insulin deficiency. Patients are instructed to increase exercise and diet restrictions, but most
require treatment with an anti-diabetic agent to control blood glucose.
structure:
The compound and methods of its synthesis are described in WO 2004/080990, WO 2005/012326 and WO 2007/114475 for example.
The gluconolactone method: In 1988 and 1989 a general method was reported to prepare C-arylglucosides from tetra-6>-benzyl protected gluconolactone, which is an oxidized derivative of glucose (see J. Org. Chem. 1988, 53, 752-753 and J. Org. Chem. 1989, 54, 610- 612). The method comprises: 1) addition of an aryllithium derivative to the hydroxy-protected gluconolactone to form a hemiketal (a.k.ci., a lactol), and 2) reduction of the resultant hemiketal with triethylsilane in the presence of boron trifluoride etherate. Disadvantages of this classical, but very commonly applied method for β-C-arylglucoside synthesis include:
1) poor “redox economy” (see J. Am. Chem. Soc. 2008, 130, 17938-17954 and Anderson, N. G. Practical Process Research & Development, 1st Ed.; Academic Press, 2000 (ISBN- 10: 0120594757); pg 38)— that is, the oxidation state of the carbon atom at CI, with respect to glucose, is oxidized in the gluconolactone and then following the arylation step is reduced to provide the requisite oxidation state of the final product. 2) due to a lack of stereospecificity, the desired β-C-arylglucoside is formed along with the undesired a-C-arylglucoside stereoisomer (this has been partially addressed by the use of hindered trialkylsilane reducing agents (see Tetrahedron: Asymmetry 2003, 14, 3243-3247) or by conversion of the hemiketal to a methyl ketal prior to reduction (see J. Org. Chem. 2007, 72, 9746-9749 and U.S. Patent 7,375,213)).
Oxidation Reduction
Glucose Gluconoloctone Hemiketal a-anomer β-anomer
R = protecting group
The metalated glucal method: U.S. Patent 7,847,074 discloses preparation of SGLT2 inhibitors that involves the coupling of a hydroxy-protected glucal that is metalated at CI with an aryl halide in the presence of a transition metal catalyst. Following the coupling step, the requisite formal addition of water to the C-arylglucal double bond to provide the desired C-aryl glucoside is effected using i) hydroboration and oxidation, or ii) epoxidation and reduction, or iii) dihydroxylation and reduction. In each case, the metalated glucal method represents poor redox economy because oxidation and reduction reactions must be conducted to establish the requisite oxidation states of the individual CI and C2 carbon atoms.
U.S. Pat. Appl. 2005/0233988 discloses the utilization of a Suzuki reaction between a CI -boronic acid or boronic ester substituted hydroxy-protected glucal and an aryl halide in the presence of a palladium catalyst. The resulting 1- C-arylglucal is then formally hydrated to provide the desired 1- C-aryl glucoside skeleton by use of a reduction step followed by an oxidation step. The synthesis of the boronic acid and its subsequent Suzuki reaction, reduction and oxidation, together, comprise a relatively long synthetic approach to C-arylglucosides and exhibits poor redox economy. Moreover, the coupling catalyst comprises palladium which is toxic and therefore should be controlled to very low levels in the drug substance.
R = protecting group; R’ = H or alkyl
The glucal epoxide method: U.S. Patent 7,847,074 discloses a method that utilizes an organometallic (derived from the requisite aglycone moiety) addition to an electrophilic epoxide located at C1-C2 of a hydroxy-protected glucose ring to furnish intermediates useful for SGLT2 inhibitor synthesis. The epoxide intermediate is prepared by the oxidation of a hydroxy- protected glucal and is not particularly stable. In Tetrahedron 2002, 58, 1997-2009 it was taught that organometallic additions to a tri-6>-benzyl protected glucal-derived epoxide can provide either the a-C-arylglucoside, mixtures of the a- and β-C-arylglucoside or the β-C-arylglucoside by selection of the appropriate counterion of the carbanionic aryl nucleophile (i.e., the
organometallic reagent). For example, carbanionic aryl groups countered with copper (i.e., cuprate reagents) or zinc (i.e., organozinc reagents) ions provide the β-C-arylglucoside, magnesium ions provide the a- and β-C-arylglucosides, and aluminum (i.e., organoaluminum reagents) ions provide the a-C-arylglucoside.
or Zn
The glycosyl leaving group substitution method: U.S. Patent 7,847,074, also disclosed a method comprising the substitution of a leaving group located at CI of a hydroxy-protected glucosyl species, such as a glycosyl halide, with a metalated aryl compound to prepare SGLT2 inhibitors. U.S. Pat. Appl. 2011/0087017 disclosed a similar method to prepare the SGLT2 inhibitor canagliflozin and preferably diarylzinc complexes are used as nucleophiles along with tetra- >-pivaloyl protected glucosylbromide.
Glucose Glucosyl bromide β-anomer
Methodology for alkynylation of 1,6-anhydroglycosides reported in Helv. Chim. Acta. 1995, 78, 242-264 describes the preparation of l,4-dideoxy-l,4-diethynyl^-D-glucopyranoses (a. La., glucopyranosyl acetylenes), that are useful for preparing but-l,3-diyne-l,4-diyl linked polysaccharides, by the ethynylating opening (alkynylation) of partially protected 4-deoxy-4-C- ethynyl-l,6-anhydroglucopyranoses. The synthesis of β-C-arylglucosides, such as could be useful as precursors for SLGT2 inhibitors, was not disclosed. The ethynylation reaction was reported to proceed with retention of configuration at the anomeric center and was rationalized (see Helv. Chim. Acta 2002, 85, 2235-2257) by the C3-hydroxyl of the 1,6- anhydroglucopyranose being deprotonated to form a C3-0-aluminium species, that coordinated with the C6-oxygen allowing delivery of the ethyne group to the β-face of the an oxycarbenium cation derivative of the glucopyranose. Three molar equivalents of the ethynylaluminium reagent was used per 1 molar equivalent of the 1,6-anhydroglucopyranose. The
ethynylaluminium reagent was prepared by the reaction of equimolar (i.e., 1:1) amounts of aluminum chloride and an ethynyllithium reagent that itself was formed by the reaction of an acetylene compound with butyllithium. This retentive ethynylating opening method was also applied (see Helv. Chim. Acta. 1998, 81, 2157-2189) to 2,4-di-<9-triethylsilyl- 1,6- anhydroglucopyranose to provide l-deoxy-l-C-ethynyl- -D-glucopyranose. In this example, 4 molar equivalents of the ethynylaluminium reagent was used per 1 molar equivalent of the 1,6- anhydroglucopyranose. The ethynylaluminium regent was prepared by the reaction of equimolar (i.e., 1: 1) amounts of aluminum chloride and an ethynyl lithium reagent that itself was formed by reaction of an acetylene compound with butyllithium.
It can be seen from the peer-reviewed and patent literature that the conventional methods that can be used to provide C-arylglucosides possess several disadvantages. These include (1) a lack of stereoselectivity during formation of the desired anomer of the C- arylglucoside, (2) poor redox economy due to oxidation and reduction reaction steps being required to change the oxidation state of CI or of CI and C2 of the carbohydrate moiety, (3) some relatively long synthetic routes, (4) the use of toxic metals such as palladium, and/or (5) atom uneconomic protection of four free hydroxyl groups. With regard to the issue of redox economy, superfluous oxidation and reduction reactions that are inherently required to allow introduction of the aryl group into the carbohydrate moiety of the previously mention synthetic methods and the subsequent synthetic steps to establish the required oxidation state, besides adding synthetic steps to the process, are particular undesirable for manufacturing processes because reductants can be difficult and dangerous to operate on large scales due to their flammability or ability to produce flammable hydrogen gas during the reaction or during workup, and because oxidants are often corrosive and require specialized handling operations (see Anderson, N. G. Practical Process Research & Development, 1st Ed.; Academic Press, 2000 (ISBN-10: 0120594757); pg 38 for discussions on this issue).
-
The C-glycoside derivative represented by the formula (1) and its salt [hereinafter, they are referred to as “compound (1)” or “compound of formula (1)” in some cases] is known to be useful for treatment and prevention of diabetes such as insulin-dependent diabetes (type 1 diabetes), non-insulin-dependent diabetes (type 2 diabetes) and the like and various diabetes-related diseases including insulin-resistant diseases and obesity (Patent Literature 1).
-
The method for producing the C-glycoside derivative represented by the formula (1), described in the Patent Literature 1 is understood to be represented by the below-shown reaction formula (I), by referring to the Examples and Reference Examples, described in the Patent Literature 1. Roughly explaining, it is a method which comprises reacting [1-benzothien-2-yl(5-bromo-2-fluorophenyl)methoxy]tert-butyl)dimethylsilane (synthesized in accordance with Reference Example 37 of the Literature) in a manner shown in Example 65 of the Literature, to obtain (1S)-1,5-anhydro-1-[3-(1-benzothien-2-ylmethyl)-4-fluorophenyl]-2,3,4,6-tetra-O-benzyl-D-glucitol and then reacting the obtained compound in accordance with Example 100 of the Literature to synthesize intended (1S)-1,5-anhydro-1-C-[3-(1-benzothiophene-2-ylmethyl)-4-fluorophenyl]-D-glucitol.
-
However, the method for producing the C-glycoside derivative of the formula (1), disclosed in the Patent Literature 1 is not industrially satisfactory in yield and cost, as is seen in later-shown Reference Example 1 of the present Description.
-
For example, as described later, the method includes a step of low product yield (for example, a step of about 50% or lower yield) and the overall yield of the C-glycoside derivative (final product) represented by the formula (1) from the compound (8) (starting raw material) is below 7%; therefore, the method has problems in yield and cost from the standpoint of medicine production and has not been satisfactory industrially. In addition, the method includes an operation of purification by column chromatography which uses chloroform as part of purification solvents; use of such a solvent poses a problem in environmental protection and there are various restrictions in industrial application of such an operation; thus, the method has problems in providing an effective medicine.
-
Also, an improved method of conducting an addition reaction with trimethylsilyl carbohydrate instead of benzyl carbohydrate and then conducting deprotection for acetylation, is known for a compound which has a structure different from that of the compound of the formula (1) but has a structure common to that of the compound of the formula (1) (Patent Literature 2). It is described in the Patent Literature 2 that the improved method enhances the overall yield to 6.2% from 1.4%. Even in the improved method, however, the yield is low at 6.2% which is far from satisfaction in industrial production.
- Patent Literature 1: WO 2004/080990 Pamphlet
- Patent Literature 2: WO 2006/006496 Pamphlet


http://www.google.com/patents/EP2105442A1
-
Into a tetrahydrofuran (20 ml) solution of benzo[b]thiophene (5.0 g) was dropwise added a n-hexane solution (25 ml) of n-butyl lithium (1.58 M) at -78°C in an argon atmosphere, followed by stirring at -78°C for 10 minutes. Into this solution was dropwise added a tetrahydrofuran (80 ml) solution of 5-bromo-2-fluorobenzaldehyde (8.0 g), followed by stirring at -78°C for 2.5 hours. The temperature of the reaction mixture was elevated to room temperature. Water was added thereto, followed by extraction with ethyl acetate. The organic layer was washed with a saturated aqueous sodium chloride solution, dried over anhydrous magnesium sulfate, filtered, and concentrated. The residue was purified by silica gel column chromatography (n-hexane/ethyl acetate) to obtain 1-benzothien-2-yl(5-bromo-2-fluorophenyl)methanol (10.5 g, yield: 83.6%).
1H-NMR (CDCl3): δ
2.74 (1H, d), 6.35 (1H, d), 6.93 (1H, dd), 7.14 (1H, s), 7.27-7.38 (2H, m), 7.39 (1H, m), 7.68 (1H, dd), 7.74 (2H, m)
- First step: synthesis of 1-benzothien-2-yl(5-bromo-2-fluorophenyl)methanol
Second step: synthesis of [1-benzothien-2-yl(5-bromo-2-fluorophenyl)methoxy](tert-butyl)dimethylsilane
-
To a dimethylformamide (20 ml) solution of 1-benzothien-2-yl(5-bromo-2-fluorophenyl)methanol (5.0 g) were added imidazole (1.3 g), a catalytic amount of 4-(dimethylamino)pyridine and tert-butyldimethylchlorosilane (2.7 g), followed by stirring at room temperature for 7 hours. To the reaction mixture was added a saturated aqueous ammonium chloride solution, followed by extraction with ethyl acetate. The organic layer was washed with a saturated aqueous ammonium chloride solution and a saturated aqueous sodium chloride solution, dried over anhydrous magnesium sulfate, filtered and concentrated. The residue was purified by silica gel column chromatography (n-hexane/ethyl acetate) to obtain [1-benzothien-2-yl(5-bromo-2-fluorophenyl)methoxy](tert-butyl)dimethylsilane (5.22 g, yield: 78.0%).
MS: 451 (M+)
1H-NMR (CDCl3): δ
0.05 (3H, s), 0.11 (3H, s), 0.95 (9H, s), 6.34 (1H, s), 6.91 (1H, t), 7.08 (1H, d), 7.23-7.38 (2H, m), 7.64-7.68 (1H, m), 7.75-7.28 (2H, m)
Third step: Synthesis of 1-C-[3-(1-benzothien-2-yl{[tert-butyl-(dimethyl)silyloxy}methyl)4-fluorophenyl]-2,3,4,6-tetra-O-benzyl-D-glucopyranose
-
Into a tetrahydrofuran (15 ml) solution of [1-benzothien-2-yl(5-bromo-2-fluorophenyl)methoxy](tert-butyl)dimethylsilane (1.5 g) was dropwise added a n-hexane solution (2.2 ml) of n-butyl lithium (1.58 M) in an argon atmosphere at -78°C, followed by stirring at -78°C for 30 minutes. Into the solution was dropwise added a tetrahydrofuran (20 ml) solution of 2,3,4,6-tetra-O-benzyl-D-glucono-1,5-lactone (1.9 g), followed by stirring at -78°C for 15 minutes and then at 0°C for 1.5 hours. To the reaction mixture was added a saturated aqueous ammonium chloride solution, followed by extraction with ethyl acetate. The organic layer was washed with a saturated aqueous ammonium chloride solution and a saturated aqueous sodium chloride solution, dried over anhydrous magnesium sulfate, filtered and concentrated. The residue was purified by silica gel column chromatography (n-hexane/chloroform/acetone) to obtain 1-C-[3-(1-benzothien-2-yl{[tert-butyl-(dimethyl)silyloxy}methyl)-4-fluorophenyl]-2,3,4,6-tetra-O-benzyl-D-glucopyranose (1.52 g, yield: 50.2%). MS: 933 (M+Na)
Fourth step: Synthesis of 1-C-{3-[1-benzothien-2-yl(hydroxy)methyl]-4-fluorophenyl}-2,3,4,6-tetra-O-benzyl-D-glucopyranose
-
To a tetrahydrofuran (15 ml) solution of 1-C-[3-(1-benzothien-2-yl{[tert-butyl-(dimethyl)silyloxy}methyl)-4-fluorophenyl]-2,3,4,6-tetra-O-benzyl-D-glucopyranose (1.52 g) was added a tetrahydrofuran solution (2.0 ml) of tetrabutylammonium fluoride (1.0 M), followed by stirring at room temperature for 1 hour. The reaction mixture was concentrated per se. The residue was purified by silica gel column chromatography (n-hexane/ethyl acetate) to obtain 1-C-{3-[1-benzothien-2-yl(hydroxy)methyl]-4-fluorophenyl}-2,3,4,6-tetra-O-benzyl-D-glucopyranose (0.99 g, yield: 74.7%). MS: 819 (M+Na), 779 (M+H-H2O)
Fifth step: Synthesis of (1S)-1,5-anhydro-1-[3-(1-benzothien-2-ylmethyl)-4-fluorophenyl]-2,3,4,6-tetra-O-benzyl-D-glucitol
-
To an acetonitrile (5.0 ml) solution of 1-C-{3-[1-benzothien-2-yl(hydroxy)methyl]-4-fluorophenyl}-2,3,4,6-tetra-O-benzyl-D-glucopyranose (500 mg) were added triethylsilane (175 mg) and boron trifluoride-diethyl ether complex (196 mg) in an argon atmosphere at -20°C, followed by stirring at -20°C for 5 hours. To the reaction mixture was added a saturated aqueous sodium bicarbonate solution, followed by extraction with chloroform. The organic layer was washed with a saturated aqueous sodium bicarbonate solution and a saturated aqueous sodium chloride solution, dried over anhydrous magnesium sulfate, filtered and concentrated. The residue was purified by silica gel column chromatography (n-hexane/ethyl acetate) to obtain (1S)-1,5-anhydro-1-[3-(1-benzothien-2-ylmethyl)-4-fluorophenyl]-2,3,4,6-tetra-O-benzyl-D-glucitol (150 mg, yield: 30.2%) MS: 787 (M+Na)
1H-NMR (CDCl3): δ
3.42-3.48 (1H, m), 3.55-3.58 (1H, m), 3.72-3.78 (4H, m), 3.83 (1H, d), 4.14-4.30 (3H, m), 4.39 (1H, d), 4.51-4.67 (4H, m), 4.83-4.94 (2H, m), 6.86-6.90 (1H, m), 6.98 (1H, brs), 7.06-7.37 (24H, m), 7.57-7.60 (1H, m), 7.66-7.69 (1H, m)
Sixth step: Synthesis of (1S)-1,5-anhydro-1-C-[3-(1-benzothiophene-2-ylmethyl)-4-fluorophenyl]-D-glucitol
-
To a dichloromethane (10 ml) solution of (1S)-1,5-anhydro-1-[3-(1-benzothien-2-ylmethyl)-4-fluorophenyl]-2,3,4,6-tetra-O-benzyl-D-glucitol (137 mg) were added pentamethylbenzene (382 mg) and a n-heptane solution (0.75 ml) of boron trichloride (1.0 M) in an argon atmosphere at -78°C, followed by stirring at -78°C for 3 hours. Methanol was added to the reaction mixture, the temperature of the resulting mixture was elevated to room temperature, and the mixture was concentrated per se. The residue was purified by silica gel column chromatography (chloroform/methanol) to obtain (1S)-1,5-anhydro-1-C-[3-(1-benzothiophene-2-ylmethyl)-4-fluorohenyl]-D-glucitol OR IPRAGLIFLOZIN (63 mg, yield: 87.8%).
1H-NMR (CD3OD): δ
3.29-3.48 (4H, m), 3.68 (1H, dd), 3.87 (1H, dd), 4.11 (1H, d), 4.20-4.29 (2H, m), 7.03 (1H, s), 7.08 (1H, dd), 7.19-7.29 (2H, m), 7.35 (1H, m), 7.42 (1H, dd), 7.64 (1H, d), 7.72 (1H, d)

(1S)-1,5-anhydro-1-C-[3-(1-benzothiophene-2-ylmethyl)-4-fluorohenyl]-D-glucitol OR IPRAGLIFLOZIN
TOFOGLIFLOZIN » All About Drugs
TOFOGLIFLOZIN » All About Drugs
CLICK ABOVE for full article
ALSO same article at
SEE……..http://apisynthesisint.blogspot.in/2015/12/tofogliflozin.html
SEE ALL FLOZINS AT
EG, Dapagliflozin, canagliflozin and all
http://medcheminternational.blogspot.in/p/flozin-series.html
Perampanel

Perampanel
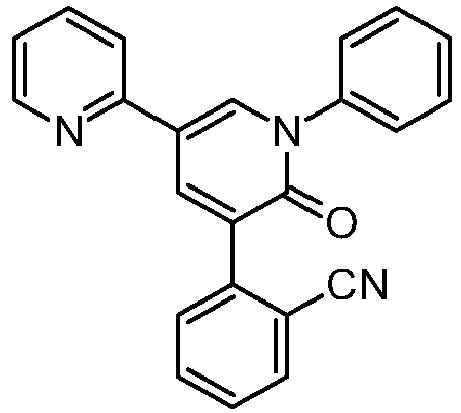
5′-(2-cyanophenyl)-1′-phenyl-2,3′-bipyridinyl-6′(1’H)-one
cas no 380917-97-5
FDA-approved drug to treat epilepsy. Trade name Fycompa, Eisai (Eisai) research and development.
FYCOMPA tablets contain perampanel, a non-competitive AMPA receptorantagonist. Perampanel is described chemically as 2-(2-oxo-1-phenyl-5-pyridin-2-yl-1,2-dihydropyridin-3-yl) benzonitrile hydrate (4:3).
The molecular formula is C23H15N3O •3/4H2O and the molecular weight is 362.90 (3/4 hydrate). The chemical structure of perampanel is:
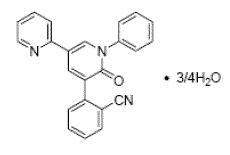 |
Perampanel is a white to yellowish white powder. It is freely soluble in N-methylpyrrolidone, sparingly soluble in acetonitrile and acetone, slightly soluble in methanol, ethanol and ethyl acetate, very slightly soluble in 1-octanol and diethyl ether and practically insoluble in heptane and water.
Perampanel (INN/USAN, trade name Fycompa) is an antiepileptic drug developed by Eisai Co. that acts as a selective noncompetitive antagonist of AMPA receptors, the major subtype of ionotropic glutamate receptors.[1][2]
Perampanel was found to be effective in the treatment of refractory partial-onset seizures in three pivotal (Phase 3) clinical trials[3][4] and has been approved for marketing under the brand name Fycompa by the European Medicines Agency.[5] The minimum effective dose is 4 mg once daily; doses of 8 mg and 12 mg daily provide a greater therapeutic benefit with a corresponding increase in adverse events. Dizziness and somnolence/sedation/fatigue are the most frequent dose-related adverse events. The drug is currently approved, for the control of partial-onset seizures, in those of both sexes who suffer from epilepsy and who are 12 years of age and older, by the Food and Drug Administration, and is considered to be a scheduled drug (an agent with the potential for addiction). Perampanel has been studied in other clinical indications includingParkinson’s disease.[6][7]
It has high potency (IC50 in vitro in functional studies of about 100-250 nM) and a prolonged terminal half-life in humans of approximately 105 hours. The drug is 95% bound to plasma protein. Its primary route of metabolism is by CYP3A4. It does not induce or inhibit P450 enzymes. About 70% of the dose is excreted in the feces and 30% in the urine; less than 2% of the dose is excreted unchanged into the urine.
In clinical trials, perampanel was generally well tolerated although the incidence of adverse events increased in a dose-dependent fashion. There was no increase in serious adverse events compared with placebo. According to the Food and Drug Administration, most common adverse reactions reported by patients receiving Fycompa in clinical trials include dizziness, drowsiness, fatigue, irritability, falls, upper respiratory tract infection,weight increase, vertigo, loss of muscle coordination (ataxia), gait disturbance, balance disorder, anxiety, blurred vision, stuttering (dysarthria), weakness (asthenia), aggression, and excessive sleep (hypersomnia).[8]
Fycompa’s label has a boxed warning to alert prescribers and patients about the risk of serious neuropsychiatric events. Some of these events were reported as serious and life-threatening. Violent thoughts or threatening behavior (including homicidal ideation) was also observed in a few patients. Patients and caregivers should alert a health care professional immediately if changes in mood or behavior that are not typical for the patient are observed. Health care professionals should closely monitor patients during the titration period when higher doses are used.[9]
- Rogawski, M. A. (2011). “Revisiting AMPA Receptors as an Antiepileptic Drug Target”. Epilepsy Currents 11 (2): 56–63. doi:10.5698/1535-7511-11.2.56. PMC 3117497. PMID 21686307. edit
- Rogawski MA, Hanada T. Preclinical pharmacology of perampanel, a selective non-competitive AMPA receptor antagonist. Acta Neurol Scand 2013;127 (Suppl. 197): 19–24.Rogawski, M. A.; Kaukinen, T.; Collin, P.; Krekelä, I.; Patrikainen, H.; Tillonen, J.; Nyrke, T.; Laurila, K.; Haimila, K.; Partanen, J.; Valve, R.; Mäki, M.; Luostarinen, L. (2013). “Preclinical pharmacology of perampanel, a selective non-competitive AMPA receptor antagonist”. Acta Neurologica Scandinavica 127 (1): 19–25. doi:10.1111/ane.12100. PMID 22494246. edit
- Krauss, G. L.; Serratosa, J. M.; Villanueva, V.; Endziniene, M.; Hong, Z.; French, J.; Yang, H.; Squillacote, D.; Edwards, H. B.; Zhu, J.; Laurenza, A. (2012). “Randomized phase III study 306: Adjunctive perampanel for refractory partial-onset seizures”. Neurology 78 (18): 1408–1415.doi:10.1212/WNL.0b013e318254473a. PMID 22517103. edit
- French, J. A.; Krauss, G. L.; Biton, V.; Squillacote, D.; Yang, H.; Laurenza, A.; Kumar, D.; Rogawski, M. A.; Campanille, V.; Floridia, J.; Ilari, R.; Consalvo, D. E.; Thomson, A.; Sfaello, I.; Pociecha, J.; Nieto, F.; Firstenfeld, A.; Zuin, D.; Mesri, J.; Silva, W.; Nofal, P.; Cristalli, D.; Clement, J. F.; Hwang, P.; McLachlan, R.; Pillay, N.; Lasso, J.; Peralta, B. L.; Hernandez, M. L.; Tenhamm, E. (2012). “Adjunctive perampanel for refractory partial-onset seizures: Randomized phase III study 304”. Neurology 79 (6): 589–596. doi:10.1212/WNL.0b013e3182635735. PMC 3413761. PMID 22843280. edit
- “European Medicines Agency Report on Perampanel”.
- Gottwald MD, Aminoff MJ (July 2008). “New frontiers in the pharmacological management of Parkinson’s disease”. Drugs Today 44 (7): 531–45.doi:10.1358/dot.2008.44.7.1217105. PMID 18806903.
- http://www.webmd.com/epilepsy/news/20121024/epilepsy-drug-fycompa-approved
- http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm325038.htm
- http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm325038.htm
Perampanel structure is formed by the coupling of an aromatic ring . Pyridone centrally located, surrounded by connecting two benzene rings and a pyridine ring. The synthesis of 2,5 – dibromopyridine (1) Start with sodium methoxide to produce 2-substituted, and an organic tin compound occurs Stille Coupling 3 4 4 HBr generated after acid hydrolysis and coupling of benzyl bromide with NBS to give 5,5. After 6 coupling of boronic ester and get Perampanel.
Perampanel is a pharmaceutically active agent, currently in clinical phase 3. It can be used to treat Parkinson’s disease, epilepsy and multiple sclerosis.
Perampanel, having the following chemical formula
is also known as E 2007, ER 155055-90 and 3-(2-cyanophenyl)-1-phenyl-5-(2-pyridil)-1,2-dihydropyridin-2-one
Various methods of synthesis of such molecules are known, such as those reported in EP1300396, EP 1465626, EP 1772450, EP 1764361 and EP 1970370.
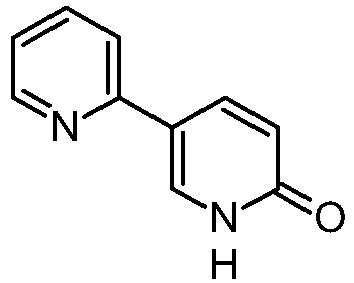
Many of the methods of synthesis of such active substances reported by the prior art use the key intermediate 5-(2-pyridil)-1,2-dihydropyridin-2-one also known as 2,3′-bipyridin-6′(1′H)-one having the following chemical formula:
Other methods use the synthetic precursor of this intermediate known as 2-methoxy-5-(pyridin-2-yl)pyridine or 6′-methoxy-2,3′-bipyridine having the formula:
2,3′-bipyridin-6′(1′H)-one. it is in fact prepared by simple acid-catalysed demethylation of the 6′-methoxy-2,3′-bipyridine as is reported in the prior art.
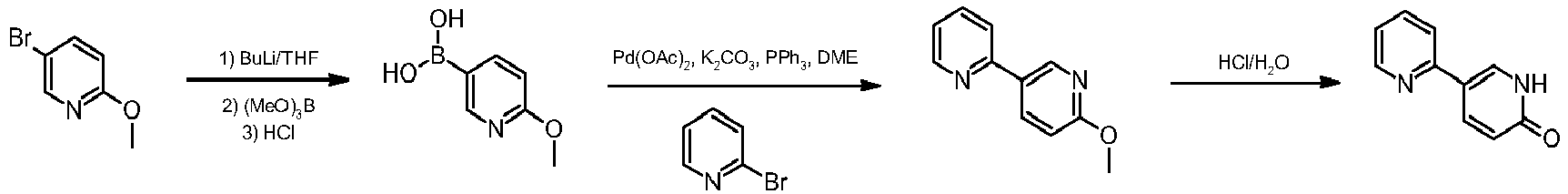
Various ways of synthesising 2-methoxy-5-(pyridin-2-yl)pyridine are known. The process summarised in Diagram (I) below is described in WO 2001096308:
Such process highlights clear disadvantages such as the need to operate in cryogenic conditions (T=−78° C.) using special equipment and the need to isolate boronic acid via work-up. In addition the use of 2-Bromopyridine is required, which exacerbates the production of waste compared to 2-chloropyridine.
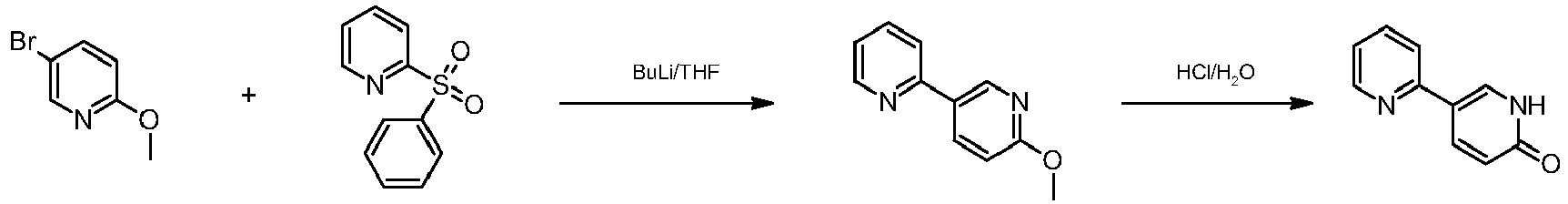
Another process described in WO 2004009553 is summarised in Diagram (II):
Disadvantages of this process include the use of high molecular weight benzene-sulfonyl pyridine entailing a scarce atom-economy of the process and the need to operate at low temperature T (−78° C.) using special equipment.
Lastly, a completely different process is described in WO20087093392 for the preparation of 2,3′-bipyridin-6′(1′H)-one (Diagram (III)) which however does not include the preparation of the intermediate precursor 2-methoxy-5-(pyridin-2-yl)pyridine:
Perampanel and other 1 ,2-dihydropyridine compounds which possess antagonistic action against AMPA receptor and/or inhibitory action against kainate receptor are described in WO 01/96308. Example 7 in WO 01/96308 discloses a process for producing perampanel by reacting 3-(2-cyanophenyl)-5-(2-pyridyl)-2(lH)-pyridone with phenyl boronic acid, copper acetate and triethylamine in methylene chloride, followed by addition of concentrated aqueous ammonia, water and ethyl acetate. After work-up (phase separation, washing the organic phase and drying over magnesium sulfate), the solvent was concentrated in vacuo and the residue was purified by a silica gel column chromatography (ethyl acetate:hexane=l :2) to give the title product as pale yellow powder. There is no disclosure regarding the polymorphic nature of the product.
A new crystalline or amorphous form of a compound may possess physical properties that differ from, and are advantageous over, those of other crystalline or amorphous forms. These include, packing properties such as molar volume, density and hygroscopicity; thermodynamic properties such as melting temperature, vapor pressure and solubility; kinetic properties such as dissolution rate and stability under various storage conditions; surface properties such as surface area, wettability, interfacial tension and shape; mechanical properties such as hardness, tensile strength, compactibility, handling, flow and blend; and filtration properties. Variations in any one of these properties may affect the chemical and pharmaceutical processing of a compound as well as its bioavailability and may often render the new form advantageous for pharmaceutical and medical use.
EP 1764361 (US 2010/324297) discloses three anhydrous crystalline forms ofperampanel, designated Form I, Form III and Form V and a hydrate form ofperampanel. Anhydrous Form I is prepared in accordance with Example Dl by dissolving perampanel in ethyl acetate (EtOAc) under reflux, cooling the solution, seeding with anhydrous perampanel crystals, continued cooling and collecting the precipitated crystals. Anhydrous Form V is prepared in accordance with Example CI, by dissolving perampanel in acetone, heating to reflux and concentrating the solution to solidification, dissolving the solids in acetone-water, refluxing then cooling and collecting the precipitate. The hydrate form is prepared in accordance with Example Bl by dissolving perampanel in acetone-water, heating, cooling the solution, seeding with perampanel hydrate crystals, continued cooling and collecting the precipitated crystals. US 2009/0088574 discloses a crystalline form of perampanel designated Form IV, which is prepared by slurring perampanel in an acetone/water mixture.
US 7,803,818 discloses an amorphous form of perampanel which is prepared by spray drying perampanel from an acetone solution.
US 7,718,807 discloses acid addition salts of perampanel or a hydrate thereof, wherein the acid is selected from the group consisting of benzenesulfonic acid, p- toluenesulfonic acid, hydrochloric acid, hydrobromic acid, sulfuric acid, methanesulfonic acid, fumaric acid, tartaric acid, succinic acid and benzoic acid.
…………………………………………………………………
Perampanel aromatic ring structure is made of highly coupled. Pyridone centrally located, surrounded by connecting two benzene ring and a pyridine ring. The synthesis of 2,5 – dibromo pyridine ( 1) Start (Synthesis, 2012, 57), sodium methoxide instead of generating 2 , and organotin compounds 3 Stille Coupling occurs to generate 4 . 4 in HBr phenylboronic acid after hydrolysis and coupling to get 5 , 5 after bromination with NBS and borate 6 coupled to get Perampanel.
…………………………….
nmr
A Practical, Laboratory-Scale Synthesis of Perampanel
……………………
updated info
-
Perampanel is a pharmaceutical active substance, currently in clinical phase 3, used to treat Parkinson’s disease, epilepsy and multiple sclerosis.
-
[0003]
-
[0004]Various methods of synthesis of such molecule are known, such as those reported in the patent publications EP1300396 , EP1465626 ,EP1772450 , EP1764361 and EP 1970370 .
-
[0005]Many of the methods of synthesis of such active substance reported by the prior art use the key intermediate 5-(2-pyridil)-1,2-dihydropyridin-2-one also known as 2,3′-bipyridin-6′(1’H)-one having the following chemical formula:
or use the synthetic precursor thereof named 2-methoxy-5-(pyridin-2-yl)pyridine or 6′-methoxy-2,3′-bipyridine having the formula:
2,3′-bipyridin-6′(1’H)-one is in fact prepared by simple acid-catalysed demethylation of the 6′-methoxy-2,3′-bipyridine as thoroughly reported in the prior art.
-
[0006]Various ways of synthesising 2-methoxy-5-(pyridin-2-yl)pyridine are known. The process summarised in the diagram (I) below is described in the publication WO 2001096308 :
Diagram (I)
-
[0007]
-
[0008]Such process highlights clear disadvantages such as the need to operate in cryogenic conditions (T=-78°C) using special equipment and the need to isolate the boronic acid via work-up; in addition the use of 2-Bromopyridine is envisaged, which is less convenient as regards the production of waste compared to 2-chloropyridine.
-
[0009]Another process described in WO 2004009553 is summarised in the diagram (II) :
Diagram (II)
-
[0010]
-
[0011]It presents clear disadvantages such as the use of high molecular weight benzenesulfonyl pyridine entailing a scarce atom-economy of the process and the need to operate at low temperature T (-78°C) using special equipment.
-
[0012]Lastly, a completely different process is described in WO20087093392for the preparation of 2,3′-bipyridin-6′(1’H)-one which however does not include the preparation of the intermediate precursor named 2-methoxy-5-(pyridin-2-yl)pyridine, process shown in the diagram (III) :
diagram (III)
-
[0013]
LOSARTAN

E-3340
L-158086
MK-0954
MK-954
Ex-89 (free acid)
COZAAR (losartan potassium, cas 124750-99-8) is an angiotensin II receptor (type AT1)antagonist. Losartan potassium, a nonpeptide molecule, is chemically described as 2-butyl-4-chloro-1-[p-(o-1H-tetrazol-5-ylphenyl)benzyl]imidazole-5-methanol monopotassium salt. Its empirical formula is C22H22ClKN6O, and its structural formula is:
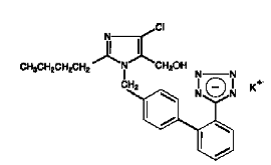 |
Losartan potassium is a white to off-white free-flowing crystalline powder with a molecular weight of 461.01. It is freely soluble in water, soluble in alcohols, and slightly soluble in common organic solvents, such as acetonitrile and methyl ethyl ketone. Oxidation of the 5-hydroxymethyl group on the imidazole ring results in the active metabolite of losartan.
COZAAR is available as tablets for oral administration containing either 25 mg, 50 mg or 100 mg of losartan potassium and the following inactive ingredients: microcrystalline cellulose, lactose hydrous, pregelatinized starch, magnesium stearate, hydroxypropyl cellulose, hypromellose, and titanium dioxide.
COZAAR 25 mg, 50 mg and 100 mg tablets contain potassium in the following amounts: 2.12 mg (0.054 mEq), 4.24 mg (0.108 mEq) and 8.48 mg (0.216 mEq), respectively. COZAAR 25 mg, COZAAR 50 mg, and COZAAR 100 mg may also contain carnauba wax.
|
Losartan (rINN) /loʊˈsɑrtən/ is an angiotensin II receptor antagonist drug used mainly to treat high blood pressure (hypertension). Losartan was the first angiotensin II antagonist to be marketed. Losartan potassium is marketed by Merck & Co. Inc. under the trade nameCozaar. Losartan is available in generic form.
As with all angiotensin II type 1 receptor (AT1) antagonists, losartan is indicated for the treatment of hypertension. It may also delay progression of diabetic nephropathy, and is also indicated for the reduction of renal disease progression in patients with type 2 diabetes, hypertension and microalbuminuria (>30 mg/24 hours) or proteinuria (>900 mg/24 hours).
Although clinical evidence shows calcium channel blockers and thiazide-type diuretics are preferred first-line treatments for most patients (from both efficacy and cost points of view), an angiotensin II receptor antagonist such as losartan is recommended as first-line treatment in patients under the age of 55 who cannot tolerate an ACE inhibitor.The LIFE study demonstrated losartan was significantly superior to atenolol in the primary prevention of adverse cardiovascular events (myocardial infarction or stroke), with a significant reduction in cardiovascular morbidity and mortality for a comparable reduction in blood pressure. A study hints that losartan has a beneficial effect on mitochondria by reversing age related dysfunction in maintaining normal blood pressure and cellular energy usage. The maximal effects on blood pressure usually occur within 3–6 weeks upon starting losartan.
Losartan is also available as hydrochlorothiazide/losartan, a combination drug with a low dose thiazide diuretic to achieve an additive antihypertensive effect.

-
Activation of AT1 receptors in the outer membrane of vascular smooth muscle cells of the heart and arteries causes those tissues to constrict. Blocking of vasoconstriction mediated by AT1 receptors has been found to be beneficial to patients with hypertension.
-
[0003]AT1 receptors are activated by an octa-peptide, angiotensin II. Angiotensin II helps to maintain constant blood pressure despite fluctuations in a person’s state of hydration, sodium intake and other physiological variables. Angiotensin II also performs the regulatory tasks of inhibiting excretion of sodium by the kidneys, inhibiting norephedrin reuptake and stimulating aldosterone biosynthesis.
-
[0004]Inhibiting angiotensin II binding to AT1 receptors with an AT1 receptor antagonist disrupts the vasoconstriction mediated by AT1 receptors that contributes to hypertension.
-
[0005]In the early 1970s, it was discovered that certain oligopeptides competitively inhibited angiotensin receptors (at that time the existence of two receptor subtypes, AT1 and AT2, was unknown). This discovery spurred interest in development of therapeutic oligopeptides with increased potency, but interest in peptide analogs waned due in part to their poor oral bioavailability.
-
[0006]In 1982, Furukawa. Kishimoto and Nishikawa of Taketa Chemical Indus. discovered a class of non-peptide-containing imidazoles that also inhibited the vasoconstriction effect of angiotensin II. See U.S. Patents Nos. 4,340,598 and 4,355,040. Later, U.S. Patent No. 5,138,069 was obtained by Carini, Denucia and Pancras of E.I. DuPont de Nemours on another class of imidazoles, which encompasses the compound losartan. In 1995, losartan (CA Index: 2-butyl-4-chloro-1-[[2′-(1H-tetrazol-5-yl) [1,1′-biphenyl] -4-yl]methyl]-1H-imidazole-5-methanol) (formula I):
became the first nonpeptide AT1 antagonist approved by the U.S. Food and Drug Administration for clinical use. Losartan can be administered orally as its monopotassium salt. Losartan potassium is available by prescription in tablet form as a sole active ingredient (Cozaar®: Merck) and as a co-active ingredient with hydrochlorothiazide (Hyzaar®: Merck).
-
[0007]Losartan has been prepared by a variety of synthetic pathways. In several of these synthetic pathways, the penultimate product is 2-butyl-4-chloro-1-[[2′-(2-triphenylmethyl-2H-tetrazol-5-yl) [1,1′-biphenyl] -4-yl]methyl]-1H-imidazole-5-methanol (“trityl losartan”). Trityl losartan is an intermediate in processes described in U.S. Patents Nos. 5,138,069; 5,962,500 and 5,206,374.
-
[0008]In a process described in Example 316 of U.S. Patent No. 5,138,069, the tetrazole ring of losartan is formed by reacting 1-[(2′-cyanobiphenyl-4-yl)methyl]-2-butyl-4-chloro-5-hydroxymethylimidazole with trimethyltin azide. The reaction gives a trimethylstannyl substituted tetrazole compound directly. The trimethylstannyl group is cleaved from the product by reacting with trityl chloride. This reaction results in attachment of the trityl group to the tetrazole ring. In the last step, the trityl group is cleaved with acid to give losartan (Scheme 1).
-
[0009]In the last step, trityl losartan was suspended in methanol and cooled to ~10°C. 3.4 N Hydrochloric acid was added to the slurry. After a period of time, the pH of the reaction mixture was raised to 13 with 10 N NaOH. Methanol was then distilled off while makeup water was added. After distillation, additional water and toluene were added. The toluene phase was separated and the aqueous phase was extracted once more with toluene. Ethyl acetate and acetic acid were then added to the aqueous phase. Losartan was recovered from the aqueous phase as a solid and further purified by slurrying in ethyl acetate. Losartan was obtained in 88.5% yield and 98.8% purity as determined by HPLC. This process is also described in U.S. Patents Nos. 5,128,355 and 5,155,188.
-
[0010]U.S. Patent No. 5,962,500, Examples 3-5, describe a process for preparing losartan in which the tetrazole ring of losartan is present in the starting material, 5-phenyltetrazole. The ‘500 patent process, depicted in Scheme 2, is convergent and uses a Suzuki coupling reaction (Miyaura, N.; Suzuki, A. Chem. Rev., 1995, 95, 2457) in the convergent step. On one branch of the synthesis, 5-phenyltetrazole is converted into the boronic acid coupling partner for the Suzuki reaction by ortho metalation with n-butyl lithium, followed by reaction with trisopropylborate. The tetrazole ring is protected from reacting with the strong allcyl lithium base with a trityl group. The trityl group is conventionally attached by reacting the tetrazole with trityl chloride in the presence of a non-nucleophilic base. On the other branch of the convergent synthesis, 2-n-butyl-4-chloro-1H-imidazole-5-carboxaldehyde is alkylated with 4-bromobenzylbromide, followed by reduction of the aldehyde with sodium borohydride to yield the other Suzuki coupling partner.
-
[0011]The direct product of Suzuki coupling is trityl losartan. In the next and last step, the tetrazole ring of trityl losartan is deprotected with 4N H2SO4 in THF. In that step, the acidic solution was aged overnight at 20 to 25°C. The solution was then extracted with isopropyl acetate and residual organic solvent was removed from the aqueous phase under vacuum. The solution was then carried forward to from the potassium salt without intermediate isolation of losartan. This process is also described in U.S.Patents Nos, 5,206,374, Example 21, and 5,310,928, Example 21.
-
[0012]Larsen, R.D et al. [J. Org. Chem. (1994), 59, 6391-6394] discloses a similar convergent synthesis of lasartan, whereby the trityl lasartan, generated by Suzuki coupling, is deprotected using 0.7 M H2SO4 in a 50 : 50 mixture of acetonitrile /water.
-
[0013]
-
[0014]In the ‘374 patent process, as in the `500 patent process, the tetrazole ring of 5-phenyltetrazole is protected with a trityl group before orthometallation of the phenyl moiety with n-butyl lithium in preparation for making the boronic acid Suzuki coupling partner. In the Suzuki coupling step, the boronic acid is reacted with 4-bromotoluene. The methyl group attached to one of the phenyl rings of the Suzuki product is then halogenated with N-bromosuccinamide and the benzylic bromine atom of that product is displaced with 2-n-butyl-4-chloro-1H-imidazole-5-carboxaldehyde. Reduction of the aldehyde group with sodium borohydride yields trityl losartan. The tetrazole group of trityl losartan was deprotected with 12% aqueous HCl in THF. After 12 hours, the pH of the reaction mixture was raised to 12.5 with 30% NaOH. The THF was then distilled off while make-up water was added to the mixture. After distillation, the mixture was cooled and the triphenyl methanol byproduct of deprotection, which had precipitated, was removed by filtration. The filtrate and rinsate, with which it was combined, were extracted with toluene. Then, ethyl acetate was added and 36% HCI was added until the pH of the reaction mixture was lowered to 3.8. The mixture was cooled, causing losartan to precipitate from the solution. Losartan was obtained in 83% theoretical yield starting from trityl losartan.
EP 253310 discloses a process, wherein 2-n-butyl-4-chloro-1H-imidazolyl-5-methanol (III) is coupled with 5-(4′-bromomethyl-1,1′-biphenyl-2-yl)-2-triphenylmethyl-2H-tetrazole (IV) in N,N-dimethylformamide as solvent in presence of sodium methoxide as the base to furnish trityl losartan. The other bases that have been claimed are sodium hydride, alkali metal carbonates such as sodium carbonate and potassium carbonate and amine bases such as triethyl amine and pyridine.
The coupling reaction results in a mixture of trityl losartan and its regio isomer (V). These are separated by column chromatography.
U.S. Pat. Nos. 5,130,439 and 5,310,928 disclose a method for coupling (IV) and (VI) in N,N-dimethylacetamide solvent in the presence of anhydrous potassium carbonate as base. The imidazole aldehyde (VI) gives predominantly the desired regio isomer (VII). The intermediate VII is then reduced with sodium borohydride to furnish the trityl losartan. The product is isolated by extraction into toluene from aqueous N,N-dimethylacetamide, concentration of the toluene solution and crystallization using ethyl acetate or ethanol as solvent. The synthesis steps are depicted as follows.
In a process published in J. Med. Chem. (1991), 34, 2525-2547, Losartan is prepared by coupling (III) and (IV) in N,N-dimethylformamide in the presence of sodium methoxide. The desired compound is isolated after vacuum distillation of solvent followed by extractive work-up. The resultant product mixture is purified by chronmatography.
The U.S. Pat. Nos. 5,138,069, 5,128,355 and 5,155,118 describe a process for the preparation of losartan, wherein the tetrazole ring of losartan is formed by reacting 1-((2′-cyanobiphenyl-4-yl)methyl)-2-butyl-4-chloro-5-hydroxymethylimidazole with trimethyltin azide. The reaction results in trimethylstannyl substituted tetrazole compound, which is then reacted with trityl chloride and sodium hydroxide.
The trityl losartan thus formed is treated with 3.4N hydrochloric acid in methanol at about 10° C. to give losartan.
The U.S. Pat. Nos. 5,138,069, 5,128,355 and 5,155,118 also disclose another process for making trityl losartan, where in the coupling between IV and VI is carried out in a biphasic solvent system comprising of chlorinated solvent and water. The reaction is carried out at room temperature in presence of sodium hydroxide as the base and aliquat 336 as the phase transfer catalyst. The resulting intermediate VII is then reduced in situ with sodium borohydride to furnish trityl losartan.
U.S. Pat. No. 5,206,374, 5,310,928 and 5,962,500 disclose another process for preparing losartan in which 5-phenyltetrazole (X) is converted into the boronic acid coupling partner (XII) for the Suzuki reaction by tritylation of phenyltetrazole with trityl chloride in presence of a non-nucleophilic base, ortho metalation with n-butyl lithium, followed by reaction with triisopropylborate. 2-n-butyl-4-chloro-1H-imidazole-5-carboxaldehyde (VI) is alkylated with 4-bromobenzylbromide, followed by reduction of the aldehyde with sodium borohydride to yield the other Suzuki coupling partner (XIII). The product of Suzuki coupling is trityl losartan. This process is published in J. Org. Chem. (1994), 59, 6391-6394.
European patents EP 470,794 and EP 470,795 describe a method for the manufacture of biphenyl carbonitriles (XVI). These patents also describe a method of preparation of trityl losartan by coupling of intermediates (III) and (IV) employing the procedure described in EP 253,310.
Losartan potassium exhibits polymorphism. Several polymorphic forms have been prepared and characterized. The following paragraphs briefly describe various polymorphs.
U.S. Pat. No. 5,608,075 discloses the polymorphic forms of losartan, wherein the trityl losartan is deprotected with H2SO4 in 50:50 acetonitrile:water and the free acid is treated with KOH solution. The aqueous solution containing losartan potassium is added slowly to a refluxing azeotropic mixture of cyclohexane/iso propanol and the ternary azeotrope cyclohexane/iso propanol/water is distilled till the water content of the pot is less than 0.05%. The white crystalline solid thus obtained is polymorphic form-I, which is characterized by DSC, XRD and IR. Polymorphic form-II is prepared by heating form-I in a DSC cell. This process is also described in U.S. Pat. No. 5,859,258.
U.S. Pat. No. 6,710,183 discloses the synthesis of losartan potassium starting from trityl losartan, wherein trityl losartan is reacted in an alcohol of formula R—OH (where R is C1 to C4 straight chain alkyl group) with 0.1 to 1 equivalent KOH. Losartan potassium thus formed is isolated after crystallizing out by changing the solvent to an aprotic or weakly protic solvent. The alcohol used is preferably methanol and the protic dipolar solvent used for the crystallization of the final product is preferably acetonitrile or straight or branched chain or cyclic aliphatic hydrocarbons.
EP 1294712 (WO 02/094816) discloses the process to manufacture losartan potassium form-I, wherein trityl losartan or losartan is suspended in a solvent and KOH is added to obtain a clear solution, which is then concentrated under reduced pressure to remove most of the solvent. An anti solvent is added to crystallize losartan potassium. The solvents to prepare losartan potassium include methanol, ethanol, and butanol but preferably the salt formation is carried out in methanol. Anti solvent is selected from common solvents such as ethyl acetate, acetonitrile, toluene and acetone, but the preferred anti solvent is acetone.
US application 2004/0006237 (WO 03/048135) relates to novel amorphous and novel crystalline forms III, IV, V of losartan potassium and the processes for their preparation. The patent also discloses novel processes for preparing losartan potassium forms I and II. The preparation of amorphous losartan includes the step of dissolving losartan potassium in a solvent to form a solution and distilling the solvent form the solution to dryness. Losartan form III (hydrated) is obtained by exposing losartan potassium amorphous or form I to an atmosphere having high relative humidity. Losartan potassium form IV is obtained by treating a saturated solution of losartan potassium in ethanol with methylene chloride. Losartan form V is obtained by treating a saturated solution of losartan potassium in ethanol with hexane. Losartan potassium form II is obtained by adding a saturated solution of losartan potassium in ethanol to xylene to form a mixture and evaporating ethanol from the mixture. Losartan form I is obtained by treating a saturated solution of losartan potassium in ethanol or iso propanol, with less soluble solvent like ethyl acetate, toluene, acetone, methyl ethyl ketone, methylene chloride, acetonitrile, dimethyl carbonate or hexane.
US application 2004/0034077 (WO 03/093262) discloses a process for preparing losartan and losartan potassium, wherein trityl losartan is treated with an acid in a diluent comprising a ketone. Especially preferred liquid ketones are acetone, methyl ethyl ketone and methyl isobutyl ketone, and acetone being the most preferred. Acids, which have been found suitable, include hydrochloric acid, sulphuric acid, acetic acid, trifluoroacetic acid, hydrobromic acid and formic acid. After the trityl losartan has been substantially converted to losartan, reaction mixture is basified. Preferred bases are alkali metal hydroxides and alkoxides. After addition of the base, the liquid ketone is evaporated under vacuum. After separation of triaryl methyl alcohol the residue is acidified to yield losartan. Free losartan is suspended in an alcohol and treated with a solution of potassium ions. Finally losartan potassium is precipitated from the alcohol. The alcohol is selected from the group consisting of isopropyl alcohol, butyl alcohol and isobutyl alcohol. The potassium ion solution is prepared by dissolving potassium iso propoxide, potassium butoxide and potassium iso butoxide or potassium hydroxide in the diluent.
US application 2004/0097568 discloses a process for preparing form III of losartan potassium, wherein trityl losartan is treated with aqueous solution of potassium hydroxide in methanol to obtain losartan potassium. The solvent is evaporated under vacuum and traces of water are removed as an azeotrope with toluene. Methanol and carbon are added to the resulting mixture. The carbon is filtered and the methanol is distilled. The resulting mixture is cooled to 20-25° C. to obtain crystalline form III losartan potassium.
US 5,138,069 and
WO 93/10106. The advantages provided by pharmaceutical products in the crystalline form in terms of easiness of processes for the preparation of related medicaments are well known. Crystalline compounds are in fact known to be more suited to the formulation of galenic forms, thanks both to their flowability in the form of powders or granulates, and to the surface properties of the crystals which promote adhesion, for example during the preparation of tablets. Furthermore, the solubility of crystalline compounds in aqueous solutions, in particular in the gastric juices, can also be significantly different than that of the corresponding amorphous compounds. There is therefore the need to discriminate between the crystalline and the amorphous forms of biologically active compounds, so as to fulfil the various pharmaceutical requirements.
A number of crystalline and amorphous forms of losartan potassium are known from
WO 95/17396 and
WO 03/048135. According to
WO 95/17396, crystalline losartan potassium is prepared by salification of acid losartan with an alkali hydroxide. The losartan potassium aqueous solution is then added to a isopropanol-cyclohexane azeotropic mixture under reflux. Water is then removed by azeotropic distillation of the resulting water-isopropanol-cyclohexane ternary mixture, which boils at 64°C. When the solution is anhydrous, the head temperature raises to 69°C and losartan potassium crystallizes.
US 5,859,258 discloses another crystallization process which comprises dissolution of losartan potassium in isopropanol-water, distillation of the binary azeotrope to an approx. 2.6% water content, precipitation by addition of a losartan potassium suspension in cyclohexane, subsequent distillation of the ternary azeotrope to a water content ranging from 0.02 to 0.11 %, and finally drying crystalline losartan potassium under vacuum at a temperature of approx. 45-50°C.

 Corresponding author email
Corresponding author emailThe imidazole ring of losartan, an antihypertensive and angiotensin II blocker is formed in a condensation reaction between valeroamidine 160 and dihydroxyacetone [50]. It was found that direct chlorination of the imidazole 162also forms the dichlorination product 164 (as shown in Scheme 33) with formaldehyde as a by-product which proved difficult to suppress and made purification of the reaction mixture problematic. Hence, a sequence involving silyl protection, chlorination and deprotection was established which gave the desired product in 90% overall yield (Scheme 33).
![[1860-5397-7-57-i33]](https://i0.wp.com/beilstein-journals.org/bjoc/content/inline/1860-5397-7-57-i33.png)
Alternatively, glycine can be reacted with methyl pentanimidate 169 to form the corresponding amidine 171 in high yield. Cyclisation, followed by a Vilsmeier-type reaction then furnishes the key chloroimidazolyl building block 172in good yield (Scheme 34) [51].
![[1860-5397-7-57-i34]](https://i0.wp.com/beilstein-journals.org/bjoc/content/inline/1860-5397-7-57-i34.png)
- 50———Shi, Y.-J.; Frey, L. F.; Tschaen, D. M.; Verhoeven, T. R. Synth. Commun. 1993, 23, 2623–2630.doi:10.1080/00397919308012598
- 51—-Griffiths, G. J.; Hauck, M. B.; Imwinkelried, R.; Kohr, J.; Roten, C. A.; Stucky, G. C.; Gosteli, J. J. Org. Chem. 1999,64, 8084–8089. doi:10.1021/jo9824910
- 52–Zhong, Y.-L.; Lee, J.; Reamer, R. A.; Askin, D. Org. Lett. 2004, 6, 929–931. doi:10.1021/ol036423y
NMR: (1H, DMSO, 300 mHz): δ 0.80 (3H, t, J=10. CH3), 1.25 (2H, sext, J=10. CH3CH2), 1.45 (2H, quin, J=10. CH3CH2CH2), 2.45-2.55 (2H, m, CH3CH2CH2CH2), 4.25 (2H, d, J= 3, CH2OH), 5.15-5.25 (3H, m, CH2Ar and OH), 6.88 (d, 2H, J=12, ArH), 7.08 (d, 2H, J=12, ArH), 7.23-7.36 (3H, m, ArH), 7.50-7.55 (1H, ArH).
SEACOND SET
http://www.google.co.in/patents/US7915425
IR v max (KBR): 3201.01, 1580.73, 1460.18, 764.81, 540.09
1H NMR (MeOD) δ, 0.87 (t, 3H), 1.33 (sext, 2H), 1.53 (quint, 2H), 2.56 (t, 2H), 4.43 (s, 2H), 5.24 (s, 2H), 6.89-7.53 (m, 8H).
13C NMR (MeOD) δ, 14.07, 23.24, 27.40, 30.92, 126.71, 126.86, 127.35, 128.21, 130, 130.8, 131, 131.19, 131.81, 136.09, 142.21, 149.97, 162.72
MS (m/z)=423.3 (M+1).
……………………………..
Melting point: 179-180.2
IR, v max (KBR): 3376.27, 1579.77, 1468.86, 762.88, 556.4
1H NMR (CDCl3) δ, 0.87 (t, 3H), 1.31 (sext, 2H), 1.54 (quint, 2H), 2.57 (t, 2H), 4.45 (s, 2H), 5.30 (s, 2H), 7.01-7.68 (m, 8H).
13C NMR (CDCl3) δ, 14.07, 23.24, 27.40, 30.92, 126.71, 126.86, 127.35, 128.21, 130, 130.8, 131, 131.19, 131.81, 136.09, 142.21, 149.97, 162.72
MS (m/z)=423.5 (M+1).
……………………………..
ADDITIONAL WRITEUP FOR READERS, NUMBERINGS ARE ALL NEW
Losartan and its potassium salt, having the formulae (1) & (2) respectively are angiotensin-II receptor (Type AT1) antagonists.
In adults Losartan is currently indicated for the treatment of hypertension (in hypertensive patients with left ventricular hypertrophy, it is also indicated to reduce the risk of stroke).
Losartan Potassium having the formula 2 and its principle active metabolite block the vasoconstrictor and aldosterone. Secreting effects of angiotensin II by selectively blocking the binding of angiotensin II to the AT1 receptor found in many tissues (e.g., vasicular smooth muscle, adrenal gland) otherwise called as angiotensin receptor blockers (ARBs).
The present invention relates to a short, simple and practical process for the preparation of Losartan 1 which belongs to a novel class of tetrazole-imidazole compounds.
There are many processes recorded in literature. The latest prior art information for the preparation of Losartan is the disclosure made in the patent application of Novartis in their PCT WO 2005/014602 dated 17 Feb. 2005.
The process described in the application comprises the reaction of 4′-(Bromomethyl)-2-cyanobiphenyl (BromoOTBN) of the formula 3 with 2-n-butyl-4-chloro-5-formyl imidazole (BCFI) of the formula of 4 in the presence of Potassium carbonate and acetonitrile to give ‘cyano aldehyde’ of the formula 5. The Cyano aldehyde of the formula 5 is reduced with sodium borohydride to get ‘cyano alcohol’ of the formula 6. The Cyano alcohol is reacted with diethyl aluminium azide in the presence of triethyl aluminium to give Losartan of the formula 1.
The reaction scheme of the process is shown in the Scheme 1
Even though the process is simple, handling of triethyl aluminium used needs special attention like very anhydrous conditions, reactions are to be performed under nitrogen or argon and transferring of triethyl aluminium from the containers needs anhydrous systems. The neat liquid and dense solutions of triethyl aluminium are known to ignite very easily at room temperature in presence of air (Pyrophoric). So handling of both triethyl aluminium and diethyl aluminium needs special attention like anhydrous conditions, nitrogen atmosphere etc.,
In EP 0578125A1 of Takeda Chemical Industries dated 12 Jan. 1994, yet another method for the preparation of Losartan has been disclosed in which Trioctadecyl or Trioctyl tin azide has been used as a tetrazole-forming agent. This method also uses the Cyano alcohol of the formula (6). The process comprises reacting the cyano alcohol of the formula (6) with tri-n-octyl tin azide in presence of toluene to give tri-n-octyl tetrazole derivative, which was treated with nitrous acid to give Losartan of the formula (1) in 94.7% yield. The process is shown in the reaction scheme 2
Even though the yields are better (94.7%) in this process again handling of tri-n-octyl tin azide is involved.
Dupont/Merck in their patents and papers always described that trityl Losartan of the formula 7 is detritylated to get Losartan 1 For example they described in J. Med. Chem., 1991, 34, 2525-2547, the preparation of Losartan of the formula 1, from trityl Losartan of the formula 7 using mineral acids such as Hydrochloric acid and sulfuric acid in 93% yield. The reaction scheme of the process is shown in the scheme 3
In this paper ‘Aldehyde Tetrazole’ of the formula 8 is isolated from trityl tetrazole aldehyde of the formula 21 and were further used for preparing derivatives of aldehyde such as benzene sulfonyl hydrazones of the formula 9 but not for Losartan. This process is shown in the scheme 4
In J. Org. Chem 1994, 59, 6391-6394 again by Merck team reported Trityl Losartan and Losartan synthesis by coupling of boronic acid derivative 11 with 3-(4-bromobenzyl) derivative of BCBMI of the formula 10. The formed trityl Losartan of the formula 7 is converted to Losartan of the formula 1 with acid. The whole process is described in Scheme 5
The Compound of the formula 10 is prepared from the reaction of BCFI of the formula 4 with p-bromo benzyl bromide of the formula 12 in potassium carbonate and Dimethyl formamide followed by reduction with sodium borohydride (NaBH4). The details are given in the Scheme 6
The Compound of the formula 11 is prepared from 5-phenyl tetrazole of the formula 14 by reacting with trityl chloride to get N-trityl-5-phenyl tetrazole of the formula 13, which on reaction with butyl lithium and triisopropyl borate followed by hydrolysis to give compound of the formula 11. This process is shown in the Scheme 7
In one of the first patent filed by Dupont/Merck (date of filing 9 Jul. 1987, priority 11 January 1986 EP0253310) reported a procedure for the preparation of Losartan. Bromo OTBN of the formula 3 is reacted with BCHMI of the formula 15 in the presence of a base to give cyano alcohol of the formula 6, and its regioisomer of the formula 14. Separation of the isomer needs column chromatography. The cyano alcohol 6 is reacted with sodium/ammonium azide in DMF for 13 days to get Losartan 1 in 21% yield. The process is shown in the Scheme 8
The drawbacks of the above process are
- 1). Separation of the regioisomer using column chromatography which is industrially not feasible for the preparation of large scale (ton) material/product
- 2). The tetrazole formation takes 13 days with 21% yield, which is unproductive.
- 3). Dupont/Merck uses BCHMI 15 as the starting material for preparing cyano alcohol of the formula 6. BCHMI 15 is an expensive intermediate compared to BCFI 4, and also the formation of unwanted regio isomer 14 is higher. The process is schematically described in scheme 8. Even though the process looks simple it has two problems.
First: Cyano alcohol is produced as a mixture of regioisomers and needs column chromatography for purification.
Second: Tetrazole formation. This takes 13 days with 21% yield, which limits commercialization of the process.
In U.S. Pat. No. 4,820,843 and U.S. Pat. No. 4,879,186, Dupont prepares Losartan by reaction of BCFI of the formula 4 and N-Triphenylmethyl-5-[2-(4′-bromomethyl biphenyl)]tetrazole of the formula 16 in the presence of base, followed by reduction with sodium borohydride to give Trityl Losartan of the formula 7, which is treated with mineral acid to give Losartan 1.
The process is shown in scheme 9
In U.S. Pat. No. 4,874,867 of Dupont/Merck, a process for the preparation of N-Triphenylmethyl-5-[2-(4′-bromomethyl biphenyl)]tetrazole of the formula 16 is described by the reaction of OTBN of the formula 20 with trimethyl tin azide to give the compound 17, which is treated with Hydrochloric acid to give tetrazole derivative of OTBN of the formula 18. The tetrazole derivative of OTBN of the formula 18 is protected with trityl chloride to give compound of the formula 19, followed by bromination with N-bromosuccinimide to give N-Triphenylmethyl-5-[2-(4′-bromomethyl biphenyl)]tetrazole of the formula 16.
The process is shown in the scheme 10.
In all the above papers and patents by Dupont/Merck, the process yields in many steps are good 75-95% and in some steps are less to moderate 21-49%. The drawbacks, or the problems in all these processes is, the number of unit operations.
For example:
- 1). In J. Med. Chem 1991, 34, 2525-2547 the number of steps are six (6) to prepare Losartan of the formula 1 from the readily available intermediates.
- 2). In J. Org. Chem 1994, 59, 6391-6394 the number of steps are nine (9) to prepare Losartan of the formula 1 from the readily available intermediates.
- 3). In EP 0253310 patent the number of operations are two (2) but the problem is time & yields i.e., 13 days and poor yield (21%), also the uneconomical column chromatographic separation of regioisomer.
- 4). In U.S. Pat. Nos. 4,820,843 and 4,879,186 the number of steps are six (6).
- 5). In U.S. Pat. No. 4,874,867 the number of steps are seven (7).
………………………..
INTERMEDIATES
(1-(2′-Cyano biphenyl-4-methyl)-2-butyl-4-chloro-5-formyl imidazole) of the formula 5.

Melting point: 107-108° C.
HPLC Purity: >98%
IR. v max (KBR): 2218 (—CN), 1662.40 (—CHO)
1H NMR (CDCl3) δ, 0.91 (t, 3H), 1.38 (sext, 2H), 1.73 (quint, 2H), 2.67 (t, 2H), 5.61 (s, 2H), 7.16-7.77 (m, 8H), 9.77 (s, 1H).
13C NMR (CDCl3) δ, 13.51, 22.18, 26.33, 29.04, 47.74, 110.05, 118.36, 124.11, 126.59, 127.65, 129.16, 129.81, 132.76, 133.61, 136.01, 137.69, 142.96, 144.33, 154.46, 177.73

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....


















![[1860-5397-7-57-i36]](https://i0.wp.com/beilstein-journals.org/bjoc/content/inline/1860-5397-7-57-i36.png)