Home » Posts tagged 'remlifanserin'
Tag Archives: remlifanserin
Remlifanserin
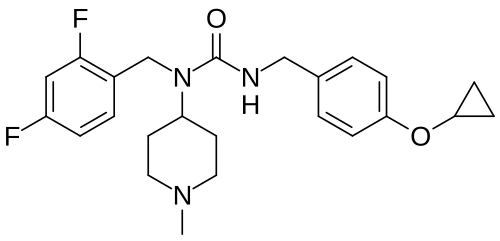
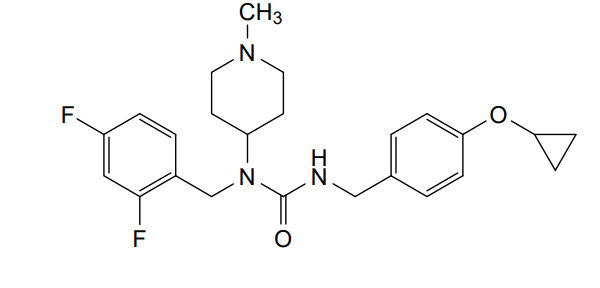
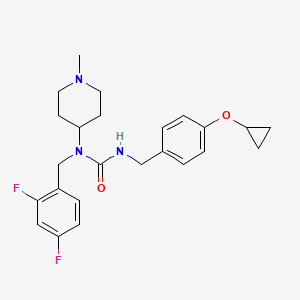
Remlifanserin
CAS 2289704-13-6
MF C24H29F2N3O2 MW 429.5 g/mol
3-[(4-cyclopropyloxyphenyl)methyl]-1-[(2,4-difluorophenyl)methyl]-1-(1-methylpiperidin-4-yl)urea
N’-{[4-(cyclopropyloxy)phenyl]methyl}-N-[(2,4-difluorophenyl)methyl]-N-(1-methylpiperidin-4-yl)urea
serotonin receptor (5-HT2A) inverse agonist, ACP-204, ACP 204, H4L2AF2XB7
Remlifanserin is a small molecule drug. The usage of the INN stem ‘-anserin’ in the name indicates that Remlifanserin is a serotonin receptor antagonist. Remlifanserin has a monoisotopic molecular weight of 429.22 Da.
Remlifanserin (INNTooltip International Nonproprietary Name;[4] developmental code name ACP-204) is a selective serotonin 5-HT2A receptor inverse agonist which is under development for the treatment of Alzheimer’s disease psychosis.[1][5][6][7][8][9] It is taken by mouth.[1]
The drug is an improved follow-up compound to its developer’s earlier drug pimavanserin (Nuplaizid; ACP-103).[6] It is more potent and selective than pimavanserin as a serotonin 5-HT2A receptor inverse agonist.[10] Remlifanserin shows 32- to 123-fold selectivity for antagonism and inverse agonism of the serotonin 5-HT2A receptor over the serotonin 5-HT2C receptor depending on the bioassay.[10] For comparison, pimavanserin’s selectivity was 8- to 37-fold depending on the assay.[10] Remlifanserin shows very low affinity for the serotonin 5-HT2B receptor compared to the serotonin 5-HT2A and 5-HT2C receptors.[10] It is expected to have less QT prolongation than pimavanserin.[10] The drug blocks the head-twitch response induced by the serotonergic psychedelic DOI and the hyperlocomotion induced by the NMDA receptor antagonist dizocilpine (MK-801) in rodents.[10]
Remlifanserin is under development by Acadia Pharmaceuticals.[1][5] As of January 2025, it is in phase 3 clinical trials.[1][5] Its clinicaltrials.gov identifier (nct number) is NCT06159673.[11]
SYN
Example 17: 3-[(4-cyclopropoxyphenyl)methyl]-1-[(2,4-difluorophenyl)methyl]-1-(1-methylpiperidin-4-yl)urea; hemitartrate (17)
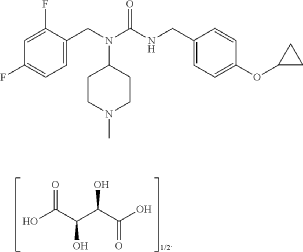
3-[(4-cyclopropoxyphenyl)methyl]-1-[(2,4-difluorophenyl)methyl]-1-(1-methylpiperidin-4-yl)urea; hemitartrate
PAT
- 3-(4-cyclo-propoxybenzyl)-1-(2,4-difluorobenzyl)-1 -(1-methylpiperidin-4-yl)urea for use in the treatment of diseases associated with the serotonin-receptor 5-htPublication Number: WO-2025029990-A1Priority Date: 2023-08-02
- Compounds, salts thereof and methods for treating diseasesPublication Number: CN-111132976-APriority Date: 2017-08-21
- COMPOUNDS, CORRESPONDING SALTS AND METHODS FOR THE TREATMENT OF DISEASESPublication Number: WO-2019040107-A1Priority Date: 2017-08-21
- Compounds, salts thereof and methods for treatment of diseasesPublication Number: EP-3672954-A1Priority Date: 2017-08-21
- Compounds, salts thereof and methods for treatment of diseasesPublication Number: US-2020270239-A1Priority Date: 2017-08-21
- Compounds, salts thereof and methods for treating diseasesPublication Number: CN-111132976-BPriority Date: 2017-08-21Grant Date: 2023-08-22
- Compounds, salts thereof and methods for treatment of diseasesPublication Number: EP-4635568-A2Priority Date: 2017-08-21
- Compounds, salts thereof and methods for treatment of diseasesPublication Number: US-12139477-B2Priority Date: 2017-08-21Grant Date: 2024-11-12
- Compounds, salts thereof and methods for treatment of diseasesPublication Number: US-2025197385-A1Priority Date: 2017-08-21
- Compounds, salts thereof and their use for the treatment of diseasesPublication Number: EP-3672954-B1Priority Date: 2017-08-21Grant Date: 2025-08-13
- Compounds, salts thereof and methods for treatment of diseasesPublication Number: US-11345693-B2Priority Date: 2017-08-21Grant Date: 2022-05-31
- Compounds, salts thereof and methods for treatment of diseasesPublication Number: US-2022298151-A1Priority Date: 2017-08-21
- Compounds, salts thereof and methods for treating diseasesPublication Number: CN-117466803-APriority Date: 2017-08-21
ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma



AS ON FEB2026 4.574 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

ADVERTISEMENT
ANAX LABORATORIES
WEBSITE https://www.anaxlab.com/
Discovery Solutions, Supporting the chemistry needs of clients in the Medical, Analytical and Bio Sciences
Development Solutions, Developing from Lab scale to PR&D, Kilo Scale-ups and Commercial Scales
SEE MORE………Integrated Solutions, Manufacturing Solutions, Products,
Can’t Find? Let’s Connect

Phone : +91 897704 2010 / +91 9177075735, Email : info@anaxlab.com
#MedicinalChemistry, #DrugDiscovery, #OrganicSynthesis, #ChemicalLibrary, #BuildingBlocks, #SARStudies, #ChemistryInnovation, #medchem, #Drugdevelopment, #Biotech, #Biotechnology, #AnaxLaboratories, #Pharma
| Clinical data | |
|---|---|
| Other names | ACP-204; ACP204 |
| Routes of administration | Oral[1] |
| Drug class | Serotonin 5-HT2A receptor inverse agonist |
| Pharmacokinetic data | |
| Onset of action | 4–6 hours (6 hours fasted, 9 hours fed) (TmaxTooltip time to peak levels)[2][3] |
| Elimination half-life | 17.8–19.8 hours[2] |
| Identifiers | |
| IUPAC name | |
| CAS Number | 2289704-13-6 |
| PubChem CID | 137520242 |
| UNII | H4L2AF2XB7 |
| Chemical and physical data | |
| Formula | C24H29F2N3O2 |
| Molar mass | 429.512 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
References
- “ACP 204”. AdisInsight. 23 January 2025. Retrieved 22 February 2025.
- Darwish M, Feng X, Dirks B, Raether B, Pathak SS (2025). “Pharmacokinetics in Healthy Adult and Elderly Patients of ACP-204, a Novel 5-HT 2A Receptor Selective Antagonist/Inverse Agonist”. Alzheimer’s & Dementia. 21 (S5) e105732. doi:10.1002/alz70859_105732. ISSN 1552-5260. PMC 12741707.
- Darwish M, Dirks B, Feng X, Raether B, Pathak SS (2025). “Effect of Food Consumption on the Pharmacokinetics of ACP-204, a Novel 5-HT 2A Receptor Selective Antagonist/Inverse Agonist”. Alzheimer’s & Dementia. 21 (S5) e105644. doi:10.1002/alz70859_105644. ISSN 1552-5260. PMC 12741626.
- “Proposed INN: List 131 International Nonproprietary Names for Pharmaceutical Substances (INN)” (PDF). WHO Drug Information. 38 (2): 421. 2024.
remlifanserin N’-{[4-(cyclopropyloxy)phenyl]methyl}-N-[(2,4- difluorophenyl)methyl]-N-(1-methylpiperidin-4-yl)urea serotonin receptor (5-HT2A) inverse agonist […] C24H29F2N3O2 2289704-13-6 […]
- “Delving into the Latest Updates on ACP-204 with Synapse”. Synapse. 4 February 2025. Retrieved 22 February 2025.
- “ACP-204”. ALZFORUM. 5 February 2024. Retrieved 22 February 2025.
- Imbimbo C, Cotta Ramusino M, Leone S, Mazzacane F, De Franco V, Gatti A, et al. (February 2025). “Emerging Pharmacological Approaches for Psychosis and Agitation in Alzheimer’s Disease”. CNS Drugs. 39 (2): 143–160. doi:10.1007/s40263-024-01133-9. PMC 11769872. PMID 39623197.
- IsHak WW, Meyer A, Freire L, Totlani J, Murphy N, Renteria S, et al. (2024). “Overview of Psychiatric Medications in the Pipeline in Phase III Trials as of June 1, 2024: A Systematic Review”. Innovations in Clinical Neuroscience. 21 (7–9): 27–47. PMC 11424068. PMID 39329027.
- Kwon KJ, Kim HY, Han SH, Shin CY (October 2024). “Future Therapeutic Strategies for Alzheimer’s Disease: Focus on Behavioral and Psychological Symptoms”. International Journal of Molecular Sciences. 25 (21) 11338. doi:10.3390/ijms252111338. PMC 11547068. PMID 39518892.
- Burstein E, Markus Dey P, Pathak S (December 2024). “ACNP 63rd Annual Meeting: Poster Abstracts P305-P608: P497. Nonclinical Characterization of ACP-204, a Novel Second Generation 5-HT2A Inverse Agonist” (PDF). Neuropsychopharmacology. 49 (Suppl 1): 236–417 (346–347). doi:10.1038/s41386-024-02012-z. PMID 39643634.
- ACADIA Pharmaceuticals Inc. (2025-02-21). A Master Protocol for Three Independent, Seamlessly Enrolling, Double-blind, Placebo-controlled Efficacy and Safety Studies of ACP-204 in Adults with Alzheimer’s Disease Psychosis (Report). clinicaltrials.gov.
////////////remlifanserin, ANAX, serotonin receptor (5-HT2A) inverse agonist, ACP-204, ACP 204, H4L2AF2XB7

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










