Home » Posts tagged 'Polymorphs'
Tag Archives: Polymorphs
Application of Process Modelling Tools in the Scale-Up of Pharmaceutical Crystallisation Processes
Crystallisations are frequent process steps in the manufacture of active pharmaceutical ingredients (APIs). They are the primary means of intermediate or product formation and separation to achieve the desired purity and form. These unit operations are complex processes which are difficult to control due to the interlinked chemical and physical effects. For example, chemical aspects such as salt and polymorph concerns are in the forefront of process research, but physical effects manifesting themselves on scale-up, due to equipment influences, can be equally important for the successful outcome of a campaign. Several operational parameters, such as temperature or impeller speed, need to be understood and controlled to achieve constant desupersaturation, consistent narrow particle size distribution around the desired mean, minimal attrition, and homogeneous growth conditions. This paper focuses on the equipment influence on crystallisations, relating it to first principles with respect to heat and momentum transfer, analysing it with computational fluid dynamics (CFD), and demonstrating its process impact using examples from recent development work. Dynamic process modelling and CFD are state-of-the-art engineering tools to identify process requirements and match them with equipment capabilities. The work reported here demonstrates how a semiquantitative application of these tools can lead to a controllable, robust process in an existing plant despite the time and resource limitations usually encountered in the industry.
http://pubs.acs.org/doi/full/10.1021/op040013n
Application of Process Modelling Tools in the Scale-Up of Pharmaceutical Crystallisation Processes
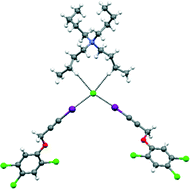
Polymorphs and co-crystals of haloprogin: an antifungal agent
 Haloprogin
Haloprogin
Haloprogin is a topical antifungal agent. Its structure does not contain any of the functional groups typically exploited in hydrogen bond based co-crystal design. On the other hand, its 1-iodoalkyne moiety is nicely tailored to a crystal engineering strategy based on halogen bonding. Here we describe the formation of three polymorphs of haloprogin and of three co-crystals that this active pharmaceutical ingredient forms with both neutral and ionic co-crystal formers. The halogen bond plays a major role in all of the six structures and the interaction is thus confirmed to be a valuable tool which may complement the hydrogen bond when polymorphs and co-crystals of active pharmaceutical ingredients are pursued.
Graphical abstract: Polymorphs and co-crystals of haloprogin: an antifungal agent
http://pubs.rsc.org/en/Content/ArticleLanding/2014/CE/C4CE00367E?utm_medium=email&utm_campaign=pub-CE-vol-16-issue-26&utm_source=toc-alert#!divAbstract
Polymorphs and co-crystals of haloprogin: an antifungal agent
Michele Baldrighi,a Davide Bartesaghi,a Gabriella Cavallo,a Michele R. Chierotti,b Roberto Gobetto,b Pierangelo Metrangolo,*a Tullio Pilati,a Giuseppe Resnati*a and Giancarlo Terraneo*a
*Corresponding authors
aNFMLab-Laboratory of Nanostructured Fluorinated Materials (NFMLab), Department of Chemistry, Materials, and Chemical Engineering “Giulio Natta”, Politecnico di Milano, Via L. Mancinelli 7, 20131 Milan, Italy
E-mail: pierangelo.metrangolo@polimi.it, giuseppe.resnati@polimi.it, giancarlo.terraneo@polimi.it
bDepartment of Chemistry, Università di Torino, Via P. Giuria 7, 10125 Torino, Italy
CrystEngComm, 2014,16, 5897-5904
DOI: 10.1039/C4CE00367E
EXTRA INFO
| Systematic (IUPAC) name | |
|---|---|
| 1,2,4-trichloro-5-[(3-iodoprop-2-yn-1-yl)oxy]benzene | |
| Clinical data | |
| Legal status | Not available in U.S. |
| Routes | Topical |
| Identifiers | |
| CAS number | 777-11-7 |
| ATC code | D01AE11 |
| PubChem | CID 3561 |
| DrugBank | DB00793 |
| ChemSpider | 3440 |
| UNII | AIU7053OWL |
| KEGG | D00339 |
| ChEMBL | CHEMBL1289 |
| Chemical data | |
| Formula | C9H4Cl3IO |
| Mol. mass | 361.39 g/mol |
| Physical data | |
| Melt. point | 113.5 °C (236 °F) |
| Solubility in water | insoluble mg/mL (20 °C) |
Haloprogin is an antifungal drug used to treat athlete’s foot and other fungal infections. It is marketed in creams under the trade names Halotex, Mycanden, Mycilan, and Polik.
Action
Haloprogin was previously used in 1% topical creams as an antifungal agent. It was marketed over the counter primarily to treat tinea infections of the skin. The mechanism of action is unknown.[1]
Haloprogin had a high incidence of side effects including: irritation, burning, vesiculation (blisters), scaling, and itching. It has since been discontinued due to the emergence of more modern antifungals with fewer side effects.[2]
Haloprogin is used as a topical ointment or cream in the treatment of Tinea infections. Tinea infections are superficial fungal infections caused by three species of fungi collectively known as dermatophytes (Trichophyton, Microsporum and Epidermophyton). Commonly these infections are named for the body part affected, including tinea corporis (general skin), tinea cruris (groin), and tinea pedis (feet). Haloprogin is a halogenated phenolic ether administered topically for dermotaphytic infections. The mechanism of action is unknown, but it is thought to be via inhibition of oxygen uptake and disruption of yeast membrane structure and function. Haloprogin is no longer available in the United States and has been discontinued.
U.S. Patent 3,322,813.
References
- “Haloprogin”. Drugs@FDA. Food and Drug Administration. Retrieved 2007-02-17.
- “Haloprogin”. DrugBank. University of Alberta. Nov 6, 2006. Retrieved 2007-02-17.

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....