Home » Posts tagged 'manufacturing' (Page 7)
Tag Archives: manufacturing
First biosimilar filgrastims launched in Japan


International nonproprietary name: Filgrastim
Chemical name: N-L- Methionyl colony-stimulating factor (human genetically engineered); non-glycated protein consisted of 175 amino acids.
Chemical name: N-L- Methionyl colony-stimulating factor (human genetically engineered); non-glycated protein consisted of 175 amino acids.
Filgrastim is a granulocyte colony-stimulating factor (G-CSF) analog used to stimulate the proliferation and differentiation of granulocytes.[1] It is produced by recombinant DNA technology. The gene for human granulocyte colony-stimulating factor is inserted into the genetic material of Escherichia coli. The G-CSF then produced by E. coli is different from G-CSF naturally made in humans.

Hematopoietic growth factor. Interacting with receptors on the surface of hematopoietic cells it regulates production and release of neutrophils from the bone marrow to the peripheral blood. Dose dependant number growth of neutrophils with normal or increased functional activity is passing for 24 hours.
Filgrastim is marketed under several brand names, including Neupogen (Amgen), Imumax(Abbott Laboratories), Grafeel (Dr. Reddy’s Laboratories), Neukine (Intas Biopharmaceuticals), Emgrast (Emcure Pharmaceuticals), Religrast (Reliance Life Sciences), Zarzio (Sandoz), Nufil (Biocon) and others.
Apricus Biosciences is currently developing and testing a product under the brand nameNupen which can deliver filgrastim through the skin to improve post-chemotherapy recovery of neutrophil counts.
Filgrastim is also used to increase the number of hematopoietic stem cells in the blood before collection by leukapheresis for use in hematopoietic stem cell transplantation.Filgrastim is used to treat neutropenia,[2] stimulating the bone marrow to increase production of neutrophils. Causes of neutropenia include chemotherapy and bone marrow transplantation.
Filgrastim should not be used in patients with known hypersensitivity to E. coli-derived proteins.
The most commonly observed adverse effect is mild-to-moderate bone pain after repeated administration and local skin reactions at the site of injection.[3] Other observed adverse effects include serious allergic reactions (including a rash over the whole body, shortness of breath, wheezing, dizziness, swelling around the mouth or eyes, fast pulse, and sweating), ruptured spleen (sometimes resulting in death), alveolar hemorrhage, acute respiratory distress syndrome, and hemoptysis.[3] Severe sickle cell crises, in some cases resulting in death, have been associated with the use of filgrastim in patients with sickle cell disorders.[4]
Drug interactions between filgrastim and other drugs have not been fully evaluated. Drugs which may potentiate the release of neutrophils‚ such as lithium‚ should be used with caution.
Increased hematopoietic activity of the bone marrow in response to growth factor therapy has been associated with transient positive bone imaging changes; this should be considered when interpreting bone-imaging results.[5]
Filgrastim has not been studied in pregnant women and its effects on unborn babies is unknown. If taking filgrastim while pregnant, it is possible that traces of the drug could be found in the baby’s blood. It is not known if the drug can get into human breast milk.
- Beveridge, R. A.; Miller, J. A.; Kales, A. N.; Binder, R. A.; Robert, N. J.; Harvey, J. H.; Windsor, K.; Gore, I. et al. (1998). “A Comparison of Efficacy of Sargramostim (Yeast-Derived RhuGM-CSF) and Filgrastim (Bacteria-Derived RhuG-CSF) in the Therapeutic Setting of Chemotherapy-Induced Myelosuppression”. Cancer Investigation 16 (6): 366–373. doi:10.3109/07357909809115775.PMID 9679526. edit
- Crawford, J.; Glaspy, J. A.; Stoller, R. G.; Tomita, D. K.; Vincent, M. E.; McGuire, B. W.; Ozer, H. (2005). “Final Results of a Placebo-Controlled Study of Filgrastim in Small-Cell Lung Cancer: Exploration of Risk Factors for Febrile Neutropenia”. Supportive Cancer Therapy 3 (1): 36–46. doi:10.3816/SCT.2005.n.023. PMID 18632435. edit
- Neupogen “Neupogen: Patient Information Leaflet”. Amgen. Retrieved 24 June 2013.
- “NEUPOGEN® Patient Guide”. Amgen. Retrieved 24 June 2013.
- “Neupogen”. RxList. 4 June 2012. Retrieved 23 June 2013.
- Budiono Santoso; Chris J. van Boxtel; Boxtel, Christoffel Jos van (2001). Drug benefits and risks: international textbook of clinical pharmacology. New York: Wiley. ISBN 0-471-89927-5.
- “Neupogen information”. Retrieved 20 October 2005.
Genentech announced positive results from the Phase 3 CLL11 study, Leukemia Trial

Afutuzumab
Obinutuzumab (GA101)
Genentech announced positive results from the Phase 3 CLL11 study. At a pre-planned interim analysis, an independent data monitoring committee determined that the study met its primary endpoint showing that GA101 plus chlorambucil helped people live significantly longer without their disease worsening (progression-free survival; PFS) compared to Rituxan (rituximab) plus chlorambucil.
The CLL11 study is being conducted in cooperation with the German CLL Study Group (GCLLSG). These final data were reached well ahead of the target completion date in 2014 as a result of the magnitude of difference seen between the two study arms.
Afutuzumab is a monoclonal antibody being developed by Hoffmann-La Roche Inc. for the treatment of lymphoma.[1] It acts as an immunomodulator.[2][3] It was renamed obinutuzumab in 2009.[4]
Class/mechanism: Glyco-engineered anti-CD20 IgG1 type II monoclonal antibody. Engineered with a modified elbow hinge residue (valine instead of leucine at Kabat position 11) and a glyco-engineered Fc region, which is postulated to enhance its immunomodulatory effect.[1]
Route: IV
Extravasation: no information
For conciseness and simplicity, HemOnc.org currently will focus on treatment regimens and not list information such as: renal/hepatic dose adjustments, metabolism (including CYP450), excretion, monitoring parameters (although this will be considered for checklists), or manufacturer.
- Robak, T (2009). “GA-101, a third-generation, humanized and glyco-engineered anti-CD20 mAb for the treatment of B-cell lymphoid malignancies”. Current opinion in investigational drugs (London, England : 2000) 10 (6): 588–96. PMID 19513948.
- Statement On A Nonproprietary Name Adopted By The Usan Council – Afutuzumab, American Medical Association.
- International Nonproprietary Names for Pharmaceutical Substances (INN), World Health Organization.
- International Nonproprietary Names for Pharmaceutical Substances (INN), World Health Organization.

| FULL STORYread all |
Alkermes unveils three new drug candidates

Alkermes has unveiled three new drug candidates, including: a monomethyl fumarate (MMF) prodrug programme for the treatment of multiple sclerosis; ALKS 7106 for the treatment of pain; and RDB 1419, a cancer immunotherapy candidate based on interleukin-2 (IL-2) and its receptors, Alkermes’ first proprietary biologic.
According to Alkermes, these drug candidates demonstrate the company’s focus on unmet medical needs in specific patient populations and show the productivity of its expanded R&D capabilities.
read all at
http://www.manufacturingchemist.com/news/article_
page/Alkermes_unveils_three_new_drug_candidates/90167

FDA Approves New Drug to Treat Nephropathic Cystinosis
CHICAGO—The U.S. Food and Drug Administration (FDA) has recently approved PROCYSBI(cysteamine bitartrate), a delayed release capsule for treating nephropathic cystinosis in adults and children 6 years and older.
Ann and Robert H. Lurie Children’s Hospital of Chicago served as one of three United States sites for the landmark study and patients came from all over North America to be seen by lead investigator, Craig B Langman, M.D., The Isaac A Abt, M.D. professor of Kidney Diseases at Northwestern University Feinberg School of Medicine and head of Kidney Diseases at Lurie Children’s

Astellas pharma announced today that the U.S. Food and Drug Administration (FDA) has approved Astagraf XL (tacrolimus extended-release capsules) for the prophylaxis of organ rejection in patients receiving a kidney transplant with mycophenolate mofetil (MMF) and corticosteroids, with or without basiliximab induction

Tacrolimus
July 19, 2013 /PRNewswire/ — Astellas Pharma US, Inc. (“Astellas”), a U.S. subsidiary of Tokyo-based Astellas Pharma Inc., announced today that the U.S. Food and Drug Administration (FDA) has approved Astagraf XL (tacrolimus extended-release capsules) for the prophylaxis of organ rejection in patients receiving a kidney transplant with mycophenolate mofetil (MMF) and corticosteroids, with or without basiliximab induction.
“Each transplant recipient is different and requires a personalized treatment approach. The approval of Astagraf XL marks an important milestone in post-transplant care as it provides physicians with a new treatment option for kidney t recipients,” said Sef Kurstjens, M.D., PhD., chief medical officer, Astellas Pharma, Inc. “Astellas is pleased to continue our more than 20-year commitment to the field of transplant immunology.”


Tacrolimus (also FK-506 or fujimycin, trade names Prograf, Advagraf, Protopic) is an immunosuppressive drug that is mainly used after allogeneic organ transplant to reduce the activity of the patient’s immune system and so lower the risk of organ rejection. It is also used in a topical preparation in the treatment of atopic dermatitis (eczema), severe refractory uveitis after bone marrow transplants, exacerbations of minimal change disease, and the skin condition vitiligo.
It is a 23-membered macrolide lactone discovered in 1984 from the fermentation broth of a Japanese soil sample that contained the bacteria Streptomyces tsukubaensis. It reduces interleukin-2 (IL-2) production by T-cells.
 Tacrolimus was discovered in 1984; it was among the first macrolide immunosuppressants discovered, preceded by the discovery of rapamycin (sirolimus) on Rapa Nui (Easter Island) in 1975.It is produced by a type of soil bacterium, Streptomyces tsukubaensis. The name tacrolimus is derived from ‘Tsukuba macrolide immunosuppressant’.
Tacrolimus was discovered in 1984; it was among the first macrolide immunosuppressants discovered, preceded by the discovery of rapamycin (sirolimus) on Rapa Nui (Easter Island) in 1975.It is produced by a type of soil bacterium, Streptomyces tsukubaensis. The name tacrolimus is derived from ‘Tsukuba macrolide immunosuppressant’.
Tacrolimus was first approved by the Food and Drug Administration (FDA) in 1994 for use in liver transplantation; this has been extended to include kidney, heart, small bowel, pancreas, lung, trachea, skin, cornea, bone marrow, and limb transplants.
The branded version of the drug is owned by Astellas Pharma, and is sold under the trade names Prograf given twice daily, Advagraf, a sustained release formulation allowing once daily dosing, and Protopic (Eczemus in Pakistan by Brookes Pharma), the topical formulation. Advagraf is available in 0.5, 1, 3 and 5 mg capsules, the ointment is concentrations of 0.1% and 0.03%.
A second once-daily formulation of tacrolimus is in Phase 3 clinical trials in the U.S. and Europe. This formulation also has a smoother pharmacokinetic profile that reduces the peak-to-trough range in blood levels compared to twice-daily tacrolimus.Data from the first Phase 3 trial in stable kidney transplant patients showed that this once-daily formulation was non-inferior in efficacy and safety compared to twice-daily tacrolimus. A second Phase 3 trial in de novo patients is ongoing.

Importance Of Preclinical Imaging In Drug Discovery
The process of discovering and bringing a drug to market consists of several stages, beginning with identification and validation of a drug target and continuing through lead identification by high-throughput screening, lead optimization, and profiling in relevant disease models. When a promising compound is found, a decision must be made on whether to take the drug into development. This long and expensive undertaking typically requires nearly $800 million and takes about 12 years before an approved drug is brought to market .
Shortening the drug discovery and development process is critical to managing this cost and can be achieved by improving the characterization of compounds and their effects in early phases of testing. Imaging has the potential to dramatically increase the efficiency of lead candidate selection by providing earlier and more highly predictive data, compared with traditional methods. Imaging is also well suited to facilitating translation between preclinical testing and clinical evaluation of drugs. Imaging methods are also more easily applied than traditional methods in the newer, more realistic models of human disease that are becoming increasingly prevalent, such as models of invasive disease in the tissue of origin as well as transgenic mouse models.
http://www.drugdiscoveryonline.com/doc/importance-of-preclinical-imaging-in-drug-discovery-0001
Breast Cancer Drugs in Late-Stage Development/Recently Approved
The article is 2012-2013 based and reader discretion is sought to ascertian the stage of approval

Afinitor® (everolimus)
https://newdrugapprovals.wordpress.com/2013/04/27/drug-spotlight-afinitor-everolimus-novartis/
Sponsor: Novartis
Method of Action: Mammalian target of rapamycin (mTOR) inhibitor
Indications/Phase of Trial: Hepatocellular carcinoma; human epidermal growth factor receptor 2-positive (HER2+) breast cancer first-line and second-line; lymphoma; nonfunctional carcinoid tumor (Phase III; all new indications)
Approved in July in U.S., EU for advanced hormone-receptor-positive (HR+) and human epidermal growth factor Receptor 2-negative (HER2-) metastatic breast cancer with exemestane in postmenopausal women who have already received certain other medicines for their cancer
Approved earlier for adults with pancreatic neuroendocrine tumors (PNET) that cannot be treated with surgery; adults with advanced renal cell carcinoma (RCC) when certain other medicines have not worked; adults with angiomyolipoma, seen with tuberous sclerosis complex (TSC), when surgery is not required immediately; and adults and children with TSC who have a brain tumor called subependymal giant cell astrocytoma (SEGA) that cannot be removed completely by surgery
Avastin (Bevacizumab; RG435)
Sponsor: Roche/Genentech
Method of Action: Monoclonal antibody; Vascular endothelial growth factor (VEGF) inhibitor
Indications/Phase of Trial: U.S.: Relapsed ovarian cancer, platinum-sensitive (Registration); first-line metastatic breast cancer and first-line metastatic ovarian cancer (both Phase III).
EU: Relapsed platinum-resistance ovarian cancer (Phase III)
Metastatic colorectal cancer, treatment beyond progression (Registration); adjuvant breast cancer, HER2- and HER2+; adjuvant NSCLC; first-line glioblastoma (GBM) multiforme; high-risk carcinoid (all Phase III)
Approved for metastatic colorectal cancer (mCRC) when started with the first or second intravenous 5-FU–based chemotherapy for metastatic cancer; advanced nonsquamous non-small-cell lung cancer (NSCLC) with carboplatin and paclitaxel in people who have not received chemotherapy for their advanced disease; metastatic RCC (mRCC) with interferon alfa; and GBM in adult patients whose cancer has progressed after prior treatment. Effectiveness based on tumor response, as no data have shown whether Avastin improves disease-related symptoms or survival in people previously treated for GBM
Approval conditionally granted in 2008 and withdrawn November 2011 for HER2- metastatic breast cancer (mBC) with Paclitaxel
Buparlisib (BKM120)
Sponsor: Novartis
Method of Action: Pan-PI3K inhibitor
Indications/Phase of Trial: mBC (Phase III and confirmatory Phase I/II); with Fulvestrant, in postmenopausal women with hormone receptor-positive HER2- locally advanced or mBC which progressed on or after aromatase inhibitor (AI) treatment (Phase III; BELLE-2 study recruiting as of November 2012); with Fulvestrant, in postmenopausal women with hormone receptor-positive HER2- AI-treated, locally-advanced or mBC who progressed on or after mTOR inhibitor-based treatment (Phase III; BELLE-3 study, recruiting as of October 2012); with Paclitaxel in patients with HER2- inoperable locally advanced or mBC, with or without PI3K pathway activation (Phase III; BELLE-4 study, recruiting as of November); metastatic castration-resistant prostate cancer (CRPC; Phase II; recruiting as of October); recurrent glioblastoma (Phase II; recruiting as of November); recurrent/metastatic head and neck squamous cell carcinoma (Phase II; recruiting as of October); endometrial cancer (Phase I/II); NSCLC (Phase I/II); prostate cancer (Phase I/II); GBM multiforme (Phase I/II); with Fulvestrant in postmenopausal women with estrogen receptor-positive metastatic breast cancer (Phase I); previously treated advanced colorectal cancer (Phase I)
Faslodex (Fulvestrant Injection)
Sponsor: AstraZeneca
Method of Action: Estrogen receptor antagonist
Indications/Phase of Trial: First line HR+ mBC (Phase III; FALCON study commenced Oct. 29)
Approved for HR+ mBC in women who have experienced menopause and whose breast cancer has worsened after they were treated with antiestrogen medications
Herceptin (Trastuzumab; RG597)
Sponsor: Roche, in partnership with Halozyme
Method of Action: Humanized monoclonal antibody designed to target and block the function of HER2+
Indications/Phase of Trial: EU: Early HER2+ breast cancer, subcutaneous formulation (Registration)
Approved for early-stage HER2+ breast cancer that has spread into the lymph nodes, and HER2+ breast cancer that has not spread into the lymph nodes and is estrogen receptor/progesterone receptor-negative (ER-/PR-) or have one high-risk feature. High-risk is defined as estrogen receptor/progesterone receptor-positive (ER+/PR+) with one of the following features: tumor size >2 cm, age <35 years, or tumor grade 2 or 3. Can be used with Adriamycin® (doxorubicin), Cytoxan® (cyclophosphamide), and either Taxol® (paclitaxel) or Taxotere® (docetaxel); or with Taxotere and Paraplatin® (carboplatin); or alone after treatment with multiple other therapies, including an anthracycline (Adriamycin)-based chemotherapy
Also approved alone for the treatment of HER2+ breast cancer in patients who have received one or more chemotherapy courses for metastatic disease; and with paclitaxel for first-line treatment of HER2+ mBC
Iniparib (Tivolza; BSI-201; SAR240550)
Sponsor: Sanofi, through acquisition of original developer BiPar Sciences
Method of Action: Poly (ADP-ribose) polymerase 1 (PARP1) inhibitor
Indications/Phase of Trial: Stage IV squamous NSCLC (Phase III; NME); solid tumors such as sarcoma and breast, uterine, lung, and ovarian cancers (Phase I/II)
Phase III trial in breast cancer failed January 2011 by failing to improve survival and progression-free survival (PFS) in breast cancer patients
Nexavar® (Sorafenib)
https://newdrugapprovals.wordpress.com/2013/07/16/nexavar-sorafenib/
Sponsor: Onyx Pharmaceuticals
Method of Action: Dual-action inhibitor that targets RAF/MEK/ERK pathway in tumor cells and tyrosine kinases
Indications/Phase of Trial: Liver cancer adjuvant (Phase III; STORM study); kidney cancer adjuvant (Phase III; SORCE/ASSURE study); thyroid cancer monotherapy (Phase III; DECISION study); breast cancer with capecitabine (Phase III; RESILIENCE study)
Approved for hepatocellular carcinoma (HCC) and RCC
Perjeta (Pertuzumab; RG1273)
Sponsor: Roche/Genentech
Method of Action: HER2/neu receptor antagonist
Indications/Phase of Trial: EU: With Herceptin and docetaxel chemotherapy for previously-untreated HER2+ mBC or locally recurrent, inoperable breast cancer in patients who have not received previous treatment or whose disease has returned after treatment in the early-stage setting (Registration)
U.S.: Approved June 2012 for HER2+ mBC with Herceptin (trastuzumab) and docetaxel, in patients who have not received prior anti-HER2 therapy or chemotherapy for metastatic disease
Switzerland: Approved August 2012 for HER2+ breast cancer with Herceptin (trastuzumab) and docetaxel in patients with advanced or locally recurring breast cancer that has not previously been treated with chemotherapy
Ridaforolimus (MK-8669; AP23573; formerly Deforolimus)
Sponsor: Merck, under exclusive worldwide license agreement with Ariad Pharmaceuticals
Method of Action: Oral inhibitor of mammalian target of rapamycin inhibitor (mTOR)
Indications/Phase of Trial: Maintenance therapy for metastatic soft-tissue sarcoma and bone sarcomas after at least four chemotherapy cycles (under review after receiving Complete Response letter from FDA in June; NME); breast cancer with exemestane, compared to breast cancer with dalotuzumab and exemestane (Phase II; recruiting as of November); advanced head and neck cancer, NSCLC and colon cancer, with cetuximab (Phase II); pediatric patients with advanced solid tumors (Phase I; recruiting as of September); with dalotuzumab in pediatric patients with advanced solid tumors (Phase I; recruiting as of August); advanced RCC, with vorinostat (Phase I; recruiting as of October 2012); breast cancer, with dalotuzumab (Phase I: recruiting as of September); endometrial and ovarian cancers, with paclitaxel and carboplatin (Phase I; recruiting as of September 2012); advanced cancer, with MK-2206 and MK-0752 (Phase I: recruiting as of September 2012); advanced cancer, with dalotuzumab, MK-2206 and MK-0752 (Phase I: recruiting as of August 2012)
Tivozanib (ASP4130; AV-951)
Sponsor: Aveo Oncology and Astellas
Method of Action: Tyrosine kinase inhibitor; inhibits VEGF receptor 1, 2, and 3
Indications/Phase of Trial: U.S.: Advanced RCC (Registration; NDA filed September 2012); tivozanib biomarkers in solid tumors (Phase II; BATON study); stage IV metastatic colorectal cancer (mCRC), with mFOLFOX6, and compared with bevacizumab and mFOLFOX6 (Phase II; recruiting as of November); additional data as first-line therapy for advanced RCC, followed by sunitinib (Phase II; TAURUS study, enrollment initiated in October 2012); advanced solid tumors, with capecitabine (Xeloda®; Phase I; recruiting as of October)
EU: Advanced RCC (Phase III)
Trastuzumab-DM1 (T-DM1; Trastuzumab emtansine; RG3502)
Sponsor: Roche, with linker technology developed by ImmunoGen
Method of Action: Antibody-drug conjugate, consisting of the antibody trastuzumab and the chemotherapy DM1 attached via a stable linker
Indications/Phase of Trial: U.S.: HER2+, unresectable locally-advanced or mBC who have received prior treatment with Herceptin (trastuzumab) and a taxane chemotherapy (Registration; Priority review approved Nov. 7; action date Feb. 26, 2013)
EU: Marketing Authorization Application for HER2+ mBC accepted for review by European Medicines Agency
Tyverb/Tykerb (lapatinib)
Sponsor: GlaxoSmithKline
Method of Action: Human epidermal growth factor receptor-2 (Her2) and epidermal growth factor receptor (EGFR) dual kinase inhibitor
Indications/Phase of Trial: mBC with trastuzumab (Registration); breast cancer, adjuvant therapy (Phase III); Gastric cancer (Phase III); head & neck squamous cell carcinoma, resectable disease (Phase III)
Xgeva (denosumab)
Sponsor: Amgen, with commercialization by GlaxoSmithKline in countries where Amgen has no presence
Method of Action: Fully human monoclonal antibody that specifically targets a ligand known as RANKL that binds to a receptor known as RANK
Indications/Phase of Trial: Delay or prevention of bone metastases in breast cancer (Phase III); delay or prevention of bone metastases in prostate cancer (Phase III)
Approved for prevention of fractures in men with advanced prostate cancer
Rejected in April for supplemental Biologics License Application to treat men with CRPC at high risk of developing bone metastases
Yondelis® (trabectedin)
Sponsor: Johnson & Johnson; developed in collaboration with PharmaMar
Method of Action: Binds to minor groove of DNA, interfering with the cell division and gene transcription processes, as well as DNA’s repair machinery
Indications/Phase of Trial: U.S.: Locally advanced or metastatic soft tissue sarcoma excluding leiomyosarcoma and liposarcoma who have relapsed or are refractory to standard-of-care treatment (Phase III; recruiting as of November); soft tissue sarcoma, excluding liposarcoma and leiomyosarcoma (L-type sarcoma), in previously-treated patients who cannot be expected to benefit from currently available therapeutic options (Phase III; recruiting as of November); locally advanced or metastatic L-sarcoma (liposarcoma or leiomyosarcoma) who were previously treated with at least an anthracycline and ifosfamide-containing regimen, or an anthracycline-containing regimen and one additional cytotoxic chemotherapy regimen, compared with dacarbazine group (Phase III; recruiting as of November); breast cancer and pediatric tumors (Phase II); Advanced malignancies and liver dysfunction (Phase I; recruiting as of November)
EU: Approved for advanced or metastatic soft tissue sarcoma, and for relapsed platinum-sensitive ovarian cancer, with DOXIL®/Caelyx®
Xtandi® Capsules (Enzalutamide; formerly MDV3100)
Sponsor: Medivation in collaboration with Astellas
Method of Action: Androgen receptor inhibitor
Indications/Phase of Trial: Prechemotherapy CRPC in patients who have failed luteinizing hormone-releasing hormone (LHRH) analog treatment only, as well as patients who have failed both LHRH analog and anti-androgen treatment. (Phase III; PREVAIL study); prostate cancer neoadjuvant therapy (Phase II); prechemo metastatic prostate cancer in Europe (Phase II; TERRAIN); prechemo metastatic and nonmetastatic prostate cancer patients in U.S. (Phase II; STRIVE); prostate cancer Hormone-naïve (Phase II; ASPIRE); prostate cancer with docetaxel (Phase I); breast cancer (Phase I)
EU: Marketing Authorization Application submitted June 2012 to European Medicines Agency, for patients with metastatic CRPC who have received docetaxel-based chemotherapy
Japan: Metastatic CRPC who have received docetaxel-based chemotherapy (Phase II)
Approved Aug. 31 for patients with metastatic CRPC who have previously received docetaxel. As a post-marketing requirement, Medivation and Astellas agreed to conduct an open-label safety study of Xtandi (160 mg/day) in patients at high risk for seizure, with data to be submitted to FDA in 2019
FDA gives tentative approval to Perrigo s ANDA for generic version of Prandin Tablets

repaglinide

Perrigo Company (Nasdaq: PRGO; TASE) today announced that it has received tentative approval from the U.S. Food & Drug Administration (FDA) for its abbreviated new drug application (ANDA) for repaglinide tablets, the generic equivalent to Prandin® Tablets (repaglinide tablets).
Prandin® tablets (repaglinide tablets), are indicated as an adjunct to diet and exercise to improve glycemic control in adults with type-2 diabetes mellitus and have annual sales of approximately $250 million, as measured by Symphony Health Solutions.
Repaglinide is an antidiabetic drug in the class of medications known as meglitinides, and was invented in 1983. It is sold byNovo Nordisk under the name of Prandin in the U.S., GlucoNorm in Canada, Surepost in Japan, Repaglinide in Egypt by EIPICO, and NovoNorm elsewhere. In Japan it is produced by Dainippon Sumitomo Pharma.
Repaglinide lowers blood glucose by stimulating the release of insulin from the pancreas. It achieves this by closing ATP-dependent potassium channels in the membrane of the beta cells. This depolarizes the beta cells, opening the cells’ calcium channels, and the resulting calcium influx induces insulin secretion.

UK launch for Astellas’ prostate cancer drug
http://www.pharmatimes.com/Article/13-07-19/UK_launch_for_Astellas_prostate_cancer_drug.aspx
UK patients with advanced prostate cancer have been given access to a new treatment that could prolong survival following the launch of Astella’s Xtandi in the country.
Xtandi (enzalutamide) was licensed in Europe this month for the treatment of men with advanced prostate cancer whose disease has become resistant to first-line hormonal treatments and has progressed following docetaxel chemotherapy.
Enzalutamide is an androgen receptor inhibitor. The chemical name is 4-{3-[4-cyano-3-(trifluoromethyl)phenyl]-5,5dimethyl-4-oxo-2-sulfanylideneimidazolidin-1-yl}-2-fluoro-N-methylbenzamide.
The molecular weight is 464.44 and molecular formula is C21H16F4N4O2S. The structural formula is:
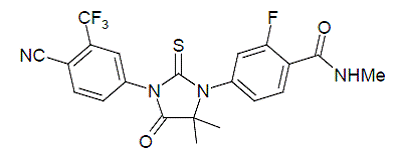 |
Enzalutamide is a white crystalline non-hygroscopic solid. It is practically insoluble in water.
XTANDI is provided as liquid-filled soft gelatin capsules for oral administration. Each capsule contains 40 mg of enzalutamide as a solution in caprylocaproyl polyoxylglycerides. The inactive ingredients are caprylocaproyl polyoxylglycerides, butylated hydroxyanisole, butylated hydroxytoluene, gelatin, sorbitol sorbitan solution, glycerin, purified water, titanium dioxide, and black iron oxide.
Menarini launches premature ejaculation drug in Singapore
DAPOXETINE
Menarini has launched dapoxetine for premature ejaculation in Singapore, having recently published a survey highlighting the rising problem of sexual dissatisfaction in the Asia-Pacific region.
The Italian drugmaker acquired Priligy (dapoxetine)from Johnson & Johnson last year and the drug is now approved in over 50 countries. It estimates that PE affects 34% of men in Singapore at some point in their lives.
READ ALL AT
Dapoxetine, marketed as Priligy (among and other brands) is the first compound developed specially for the treatment of premature ejaculation (PE) in men 18–64 years old.Dapoxetine works by inhibiting the serotonin transporter, increasing serotonin’s action at the post synaptic cleft, and as a consequence promoting ejaculatory delay. As a member of selective serotonin reuptake inhibitor (SSRI) family, dapoxetine was initially created as an antidepressant. However, unlike other SSRIs, dapoxetine is absorbed and eliminated rapidly in the body. Its fast acting property makes it suitable for the treatment of PE but not as an antidepressant.[3]
Originally created by Eli Lilly pharmaceutical company, dapoxetine was sold to Johnson & Johnson in 2003 and submitted as a new drug application to the Food and Drug Administration (FDA) for the treatment of PE in 2004. Dapoxetine has been sold in several European and Asian countries, and lately in Mexico. In the US, dapoxetine is in phase III development and expected to be marketed soon. In 2012, Menarini acquired the rights to commercialise Priligy in Europe, most of Asia, Africa, Latin America and the Middle East.
Premature ejaculation
Randomized, double blind, placebo-controlled trials have confirmed the efficacy of dapoxetine for the treatment of PE. Different dosage has different impacts on different type of PE. Dapoxetine 60 mg significantly improves the mean intravaginal ejaculation latency time (IELT) compare to that of dapoxetine 30 mg in men with lifelong PE, but there is no different in men with acquired PE. Dapoxetine, given 1–3 hours before sexual episode, prolongs IELT, increases the sense of control and sexual satisfaction in men of 18 to 64 years of age with PE. Since PE is associated with personal distress, interrelationship difficulty, dapoxetine provides help for men with PE to overcome this condition.Because lack of specific approval treatment for PE in the US and some other countries, other SSRIs such as fluoxetine, paroxetine, sertraline, fluvoxamine, and citalopram have been used as off label drugs to treat PE. Waldinger’s meta analysis shows that the use of these conventional antidepressants increasing IELT from two to ninefold above base line in comparison of three to eightfold when dapoxetine is used. However, these SSRIs must be taken daily in order to achieve meaningful efficacy, and the long half-life increases the risk of the drug accumulation and as a consequence increased of adverse effects such as decreasing sexual libido and causing erectile dysfunction. Dapoxetine, on the other hand, is a fast-acting SSRI. It is rapidly absorbed and eliminated from the body within a few hours. This favorable pharmacokinetics minimizes the risk of the drug’s accumulation in the body, and therefore reducing side effects.

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....























