Home » Posts tagged 'LURASIDONE'
Tag Archives: LURASIDONE
WO 2016110798, Piramal Enterprises Ltd, New Patent, Lurasidone

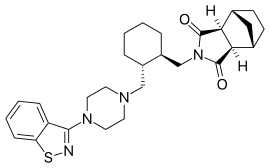
Lurasidone – it having been developed and launched by Sumitomo Dainippon Pharma. Lurasidone was launched for schizophrenia in the US by Sumitomo’s US subsidiary Sunovion Pharmaceuticals.
WO 2016110798, Piramal Enterprises Ltd, New Patent, Lurasidone
An improved process for the preparation of lurasidone and its intermediate
PIRAMAL ENTERPRISES LIMITED [IN/IN]; Piramal Tower Ganpatrao Kadam Marg, Lower Parel Mumbai 400013 (IN)

GHARPURE, Milind; (IN).
TIWARI, Shashi Kant; (IN).
WAGH, Ganesh; (IN).
REVANAPPA, Galge; (IN).
WARPE, Manikrao; (IN).
ZALTE, Yogesh; (IN).

From left: Anand Piramal, executive director, Piramal Group; Swati Piramal, vice-chairperson, Piramal Group; Ajay Piramal, chairman, Piramal Group; Nandini Piramal, executive director, Piramal Enterprises; and Peter DeYoung, president, Piramal Enterprises

Improved process for preparing pure (3aR,7aR)-4′-(benzo[d]isothiazol-3-yl)octahydrospiro[isoindole-2,1′-piperazin]-1′-ium methanesulfonate, useful as a key intermediate in the synthesis of lurasidone. Also claims a process for purifying lurasidone hydrochloride, useful for treating schizophrenia and bipolar disorders. In July 2016, Newport Premium™ reported that Piramal Enterprises was capable of producing commercial quantities of lurasidone hydrochloride and holds an active US DMF for the drug since March 2015.
Lurasidone (the Compound-I), is an atypical antipsychotic used in the treatment of schizophrenia and bipolar disorders.The drug is marketed as hydrochloride salt (the compound-I.HCl) by Sunovion Pharms Inc.under the tradename”LATUDA”, in the form of oral tablets. Latuda is indicated for the treatment of patients with schizophrenia. Lurasidone hydrochloride has the chemical name ((3aR,4S,7R,7aS)-2-[((lR,2R)-2-{ [4-(l,2-benzisothiazol-3-yl)-piperazin-l-yl]methyl}cyclohexyl)-methyl]hexahydro-lH-4,7-methanisoindol-l,3-dione hydrochloride, and is structurally represented as follows;

Compound-I.HCl
Lurasidone being an important antipsychotic agent; a number of processes for its preparation as well as for its intermediates are known in the art.
US Patent No. 5,532,372 describe a process for the synthesis of Lurasidone, which is illustrated below in Scheme-I. In the process, the compound, cyclohexane- l,2-diylbis(methylene) dimethanesulfonate(referred to as the compound-Ill) is reacted with 3-(l-piperazinyl-l,2-benzisothiazole(referred to as the compound-IV) in acetonitrile, and in the presence of sodium carbonate to provide corresponding quaternary ammonium salt as 4′-(benzo[d]isothiazol-3-yl)octahydrospiro[isoindole-2, r-piperazin]-l’-ium methanesulfonate (the compound-II). The compound-II is further treated with bicyclo[2.2.1]heptane-2-exo-3-exo-dicarboximide in xylene, in the presence of potassium carbonate and dibenzo-18-crown-6-ether to provide lurasidone.

Scheme-I
US Published Patent Application 2011/0263848 describes a process for the preparation of the quaternary ammonium salt (the compound-II) which comprises reacting 4-(l,2-benzisothiazol-3-yl)piperazine with (lR,2R)-l,2-bis(methanesulfonyloxymethyl)- cyclohexane in a solvent such as toluene in the presence of a phosphate salt.
Indian Published Patent Application 2306/MUM/2014 (” the IN’2306 Application”) describes a process for the synthesis of lurasidone and the intermediates thereof, comprising reacting (R,R) trans l,2-bis(methane sulphonyl methyl)cyclohexane with 3-(Piperazine-l-yl)benzo[d]isothiazole in presence of a mixture of two or more polar aprotic solvents selected from acetonitrile, N,N-dimethyl formamide (DMF) and/or Ν,Ν-dimethyl acetamide (DMAc), and a base at reflux temperature to obtain the quaternary ammonium salt (the compound II), which is then converted to lurasidone. The IN’2306 application demonstrated preparation of the compound II using the solvent combination such as acetonitrile-DMF and acetonitrile-DMAc.
US Published Patent Application 2011/0263847 describes a process for the preparation of the quaternary ammonium salt (the compound-II) comprising reacting 4-(l,2-benzisothiazol-3-yl)piperazine with (lR,2R)-l,2-bis(methanesulfonyloxymethyl)cyclohexane in a solvent such as toluene, wherein the piperazine compound is used in an excess amount i.e. 1.8 to 15 moles with respect to ( 1R,2R)- 1 ,2-bis(methanesulfonyloxymethyl)cyclohexane.
Chinese Published Patent Application 102731512 describes a process for the preparation of the quaternary ammonium salt (the compound-II) comprises reaction of 4-(l,2-benzisothiazol-3-yl)piperazine with (lR,2R)-l,2-bis(methanesulfonyloxymethyl)cyclohexane in a solvent such as toluene in the presence of a phase transfer catalyst.
In addition to the afore discussed patent documents, there are a number of patent documents that describe a process for the preparation of the quaternary ammonium salt (the compound-II), the key intermediate for the synthesis of lurasidone. For instance, Published PCT application WO2012/131606 A 1, Indian Published patent application 217/MUM/2013, Chinese published patent applications 102863437, 103864774 and 102827157 describe a process for the preparation of the quaternary ammonium salt (compound-II) comprises reaction of 4-(l,2-benzisothiazol-3-yl)piperazine with (lR,2R)-l,2-bis(methanesulfonyloxymethyl)cyclohexane in a solvent or a solvent mixture such as acetonitrile, acetonitrile : water solvent mixture, toluene or DMF, in the presence of a base.
It is evident from the discussion of the processes for the preparation of the quaternary ammonium salt (the compound-II), described in the afore cited patent documents that the reported processes primarily involve use of acetonitrile either as the single solvent or in a mixture of solvents. Acetonitrile is a relatively toxic, and not an environment friendly solvent. Due to its toxic nature, it can cause adverse health effects also. Acetonitrile is covered under Class 2 solvents i.e. solvents to be limited, and residual solvent limit of acetonitrile is 410 ppm in a drug substance as per the ICH (International Conference on Harmonisation) guidelines for residual solvents. Moreover, acetonitrile is a costlier solvent, which renders the process costlier and hence, is not an industrially feasible solvent.
It is also evident from the discussion of the processes described in afore cited patent documents that some of the reported processes involve use of high boiling solvents such as toluene and dimethylformamide as reaction solvent, which subsequently require high reaction temperatures, and this in turn leads to tedious workup procedures. In view of these drawbacks, there is a need to develop an industrially viable commercial process for the preparation of lurasidone and its intermediates; which is simple, efficient and cost-effective process and provides the desired compounds in improved yield and purity.
Inventors of the present invention have developed an improved process that addresses the problems associated with the processes reported in the prior art. The process of the present invention does not involve use of any toxic and/or costly solvents. Moreover, the process does not require additional purification steps and critical workup procedure. Accordingly, the present invention provides a process for the preparation of lurasidone and its intermediates, which is simple, efficient, cost effective, environmentally friendly and commercially scalable for large scale operations.

Scheme-II

Scheme-Ill
EXAMPLES
Example-1: Preparation of (3aR,7aR)-4′-(benzo[d]isothiazol-3-yl)octahydrospiro[isoindole-2,l’-piperazin]-l’-ium methanesulfonate(the compound II)
Charged 150.0 mL (3v) of isopropyl alcohol (IPA) in a flask followed by the addition of the compound-Ill (50.0 g) , 3-(l-Piperazinyl)-l, 2-Benzisothiazole (32.84 g), sodium carbonate granular (10.79 g) and water 50 mL (lv). The reaction mixture was heated at a temperature of 82-85 °C for 24 to 25 h. Cooled the reaction mixture to room temperature, filtered on Buchner funnel and the filtrate was collected.
The filtrate was evaporated under vacuum at 55-65°C till visible solid appears in the reaction mass. The solid was stirred in 75 mL of toluene at room temperature and the solid was filtered. The wet cake was transferred to a flask and added 125 mL of acetone to it; followed by stirring at room temperature. The resulting solid was filtered to yield the pure title compound (II).
Yield: 63.4 g (90 %)
Purity (by HPLC): 99.79 %
Unreacted compound-IV as impurity in 0.05 % .
Example-2: Preparation of Lurasidone free base.
Charged 150.0 mL of Ν,Ν-dimethylformamide (DMF) in a flask followed by the addition of 50.0 g of the compound-II (as obtained in the above example-1), 19.5 g (3aR,4S,7R,7aS)-4,7-methano-lH-isoindole-l,3(2H)-dione and 19.5 g of potassium carbonate. The reaction mixture was heated at a temperature of about 125 °C for 24 h. The reaction mixture was cooled to room temperature and 400 mL of water was added to it. The reaction mixture was stirred, and the precipitated product was filtered. The wet cake was washed with IPA and Lurasidone free base is obtained as the pure product. [Yield: 46.52 g (80 %)]
Example-3: Purification of Lurasidone hydrochloride.
Charged water (200 ml) and IPA (200 ml) in flask followed by the addition of Lurasidone hydrochloride (50 gm, residual acetone: 5769 ppm). The reaction mixture was heated at a temperature of 75-80 °C for about 30 min. The reaction mixture was cooled to 20-30 °C and stirred for about 2 hours. The precipitated solid was filtered and isolated as pure Lurasidone hydrochloride (residual acetone: 2 ppm)

THE VIEWS EXPRESSED ARE MY PERSONAL AND IN NO-WAY SUGGEST THE VIEWS OF THE PROFESSIONAL BODY OR THE COMPANY THAT I REPRESENT, amcrasto@gmail.com, +91 9323115463 India
///////////////WO 2016110798, Piramal Enterprises Ltd, New Patent, Lurasidone
Sunovion Pharmaceuticals Inc. Announces FDA Approval of Latuda® (lurasidone HCl) as Monotherapy and Adjunctive Therapy in Adult Patients with Bipolar Depression

lurasidone
MARLBOROUGH, Mass. – Monday, July 1st 2013 [ME NewsWire]
First Atypical Antipsychotic Indicated for the Treatment of Major Depressive Episodes Associated with Bipolar I Disorder (Bipolar Depression) Both as Monotherapy and as Adjunctive Therapy with Either Lithium or Valproate
(BUSINESS WIRE)– Sunovion Pharmaceuticals Inc. today announced that the U.S. Food and Drug Administration (FDA) approved two new indications for the use of Latuda® (lurasidone HCl) as 1) monotherapy and 2) adjunctive therapy with either lithium or valproate, both to treat adult patients with major depressive episodes associated with bipolar I disorder (bipolar depression).
read all at
http://www.me-newswire.net/news/7829/en
Lurasidone (trade name Latuda) is an atypical antipsychotic developed by Dainippon Sumitomo Pharma and marketed by Sunovion in the USA. It was approved by the U.S. Food and Drug Administration (FDA) for treatment of schizophrenia on October 28, 2010 after a review that found that two of the four Phase III clinical trials supported efficacy, while one showed only marginal efficacy and one was not interpretable because of high drop-out rates. It is currently pending approval for the treatment of bipolar disorder in the United States. It was launched in Canada for the treatment of schizophrenia in September 2012, Health Canada giving their Summary Basis of Decision (SBD) as favourable on October 15, 2012
10 MAR 2013, MAA EU SUBMITTED, APPROVED US, CANADA, LURASIDONE, LATUDA, SCHIZOPRENIA, DAINIPPON SUMITOMO
LURASIDONE
(3aR,4S,7R,7aS)-2-[((1R,2R)-2-{[4-(1,2-benzisothiazol-3-yl)-piperazin-1-yl]methyl}cyclohexyl)methyl]hexahydro-1H-4,7-methanisoindol-1,3-dione
STATUS AS ON 10 MARCH 2012
Lurasidone (trade name Latuda) is an atypical antipsychotic developed by Dainippon Sumitomo Pharma.[1] It was approved by the U.S. Food and Drug Administration (FDA) for treatment of schizophrenia on October 28, 2010[2] after a review that found that two of the four Phase III clinical trials supported efficacy, while one showed only marginal efficacy and one was not interpretable because of high drop-out rates.[3] It is currently pending approval for the treatment of bipolar disorder in the United States.
Clinical effects
In clinical studies, lurasidone alleviates positive symptoms (e.g., hallucinations, delusions) without inducing extrapyramidal side effects except for akathisia,[4] despite its potent D2 antagonistic actions. Effectiveness against negative symptoms of schizophrenia has yet to be established.
Lurasidone may be useful for treating the cognitive and memory deficits seen in schizophrenia. In animal studies, it reversed dizocilpine-induced learning and memory impairment and was found to be superior in doing this to all of the other antipsychotics examined, including risperidone, olanzapine, quetiapine, clozapine, aripiprazole, and haloperidol.[5][6] Lurasidone has activity at several serotonin receptors that are involved in learning and memory, and unlike most other antipsychotics, lacks any anticholinergic effects (which are known to impair cognitive processes and memory).[5] These properties may underlie its improved effectiveness in treating these symptoms relative to older agents.[5]
References
- Meyer JM, Loebel AD, Schweizer E (September 2009). “Lurasidone: a new drug in development for schizophrenia”. Expert Opinion on Investigational Drugs 18 (11): 1715–26. doi:10.1517/13543780903286388. PMID 19780705.
- “FDA approves Latuda to treat schizophrenia in adults” (Press release). USFDA. 2010-10-28. Retrieved October 29, 2010.
- FDA Clinical Review of lurasidone for the treatment of schizophrenia Nakamura M, Ogasa M, Guarino J, et al. (June 2009).
- “Lurasidone in the treatment of acute schizophrenia: a double-blind, placebo-controlled trial”. The Journal of Clinical Psychiatry 70 (6): 829–36. doi:10.4088/JCP.08m04905. PMID 19497249.
- Ishiyama T, Tokuda K, Ishibashi T, Ito A, Toma S, Ohno Y (October 2007). “Lurasidone (SM-13496), a novel atypical antipsychotic drug, reverses MK-801-induced impairment of learning and memory in the rat passive-avoidance test”. European Journal of Pharmacology 572 (2-3): 160–70. doi:10.1016/j.ejphar.2007.06.058. PMID 17662268.
- Enomoto T, Ishibashi T, Tokuda K, Ishiyama T, Toma S, Ito A (January 2008). “Lurasidone reverses MK-801-induced impairment of learning and memory in the Morris water maze and radial-arm maze tests in rats”. Behavioural Brain Research 186 (2): 197–207. doi:10.1016/j.bbr.2007.08.012. PMID 17881065.
- Dainippon Sumitomo Pharma (August 26, 2009). “Lurasidone Demonstrated Efficacy in Treating Patients with Schizophrenia in Pivotal Phase III Study”.
- “Latuda: Prescribing Information”. Psychotherapeutic Drugs. Retrieved 2010-12-17.
- “Latuda”. Drugs.com. Retrieved 2010-12-17.
- “Atypical antipsychotics and risk of cerebrovascular accidents”. Retrieved 28 July 2012.
LATUDA® (lurasidone hydrochloride) Schizophrenia,Bipolar disorder
- Developed in-house
- LATUDA® (lurasidone hydrochloride) is an atypical antipsychotic agent which is believed to have an affinity for dopamine D2, serotonin 5-HT2A and serotonin 5-HT7 receptors where it has antagonist effects. In addition, LATUDA is a partial agonist at the serotonin 5-HT1A receptor and has no appreciable affinity for histamine or muscarinic receptors. In the clinical trials supporting the U.S. FDA approval, the efficacy of LATUDA for the treatment of schizophrenia was established in four, short-term (6-week), placebo-controlled clinical studies in adult patients who met DSM-IV criteria for schizophrenia. In these studies, LATUDA demonstrated significantly greater improvement versus placebo on the primary efficacy measures [the Positive and Negative Syndrome Scale (PANSS) total score and the Brief Psychiatric Rating Scale-derived from PANSS (BPRSd)] at study endpoint. A total of five short-term placebo controlled clinical trials contributed to the understanding of the tolerability and safety profile of LATUDA. LATUDA was approved for the treatment of schizophrenia by the U.S. Food and Drug Administration (FDA) in October 2010, and launched by Sunovion in February 2011 in the U.S. Launched in Canada for the treatment of schizophrenia in September 2012.
- Development stage:
Schizophrenia: Submitted MAA (Europe: Co-development with Takeda Pharmaceutical)
Phase III in Japan
In addition, Phase III study is ongoing in the U.S., Europe, etc. to test the hypothesis that LATUDA is effective in the long term maintenance treatment of schizophrenia.Bipolar I Depression: Submitted in the U.S. and Canada.
In addition, plans to submit an MAA in Europe through Co-development with Takeda Pharmaceutical. (Phase III in Europe).Bipolar Maintenance: Phase III in the U.S. and Europe, etc. MDD with mixed features: Phase III in the U.S

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










