| Patent ID | Patent Title | Submitted Date | Granted Date |
|---|---|---|---|
| US2006188938 | Compounds for inhibiting beta-amyloid production and methods of identifying the compounds | 2006-08-24 | |
| US2007037855 | Compounds for inhibiting beta-amyloid production and methods of identifying the compounds | 2007-02-15 | |
| US2007185130 | Compounds for inhibiting beta-amyloid production and methods of identifying the compounds | 2007-08-09 | |
| US2007191409 | Compounds for inhibiting beta-amyloid production and methods of identifying the compounds | 2007-08-16 | |
| US2008058330 | Compounds and Combinations Thereof for Inhibiting Beta-Amyloid Production and Methods of Use Thereof | 2008-03-06 | |
| US2009082371 | Treatment of Viral Disease and Cancer With Nf-kappaB Inhibitors | 2009-03-26 | |
| US2010016218 | CONTROLLED-RELEASE APOPTOSIS MODULATING COMPOSITIONS AND METHODS FOR THE TREATMENT OF OTIC DISORDERS | 2010-01-21 | |
| US2010022661 | CONTROLLED RELEASE COMPOSITIONS FOR MODULATING FREE-RADICAL INDUCED DAMAGE AND METHODS OF USE THEREOF | 2010-01-28 | |
| US2010215735 | Compounds for Inhibiting Beta-Amyloid Production and Methods of Identifying the Compounds | 2010-08-26 | |
| US2010216784 | Compounds for Inhibiting Beta-Amyloid Production and Methods of Identifying the Compounds | 2010-08-26 |
Home » Posts tagged 'EVP 4593'
Tag Archives: EVP 4593
SEARCH THIS BLOG
 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....FOR BLOG HOME CLICK HERE
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....FOR BLOG HOME CLICK HEREBlog Stats
- 4,859,188 hits
Join 37.8K other subscribers
Pages
- About me
- All about drugs
- Bioavailability
- CLINICAL TRIALS
- Combinatorial Chemistry in Drug Design
- DISCLAIMER
- Drug Basics
- DRUG COMPANIES
- DRUG DESIGN
- DRUG DISCOVERY
- Drug Information
- DRUG NEWS
- DRUG PORTALS
- Drug resistance
- DRUG SITES
- DRUG SYNTHESIS
- DRUGS NDA
- DRUGS, FDA2012
- FDA -US
- GMP
- HEALTH CANADA
- HELP ME/ ACCOLADES
- HSA SINGAPORE
- INDA
- KOREA DRUG APPROVAL PROCESS
- MHLW JAPAN, 厚生労働省
- NMPA CHINA
- PATENT EXPIRY DATES 1/3
- PATENT EXPIRY DATES 2/3
- PATENT EXPIRY DATES 3/3
- Patent portals
- Polymorphism
- Regulations and Guidance
- Scientificupdate (UK) on a roll
- sFDA CHINA
- SOUTH AFRICA, MCC
- SPECIAL DRUGS
- Traditional Medicine
- WHAT IS A DRUG
- World drug tracker
Archives
- March 2026 (8)
- February 2026 (25)
- January 2026 (28)
- December 2025 (31)
- November 2025 (31)
- October 2025 (22)
- September 2025 (24)
- August 2025 (27)
- July 2025 (25)
- June 2025 (23)
- May 2025 (34)
- April 2025 (32)
- March 2025 (4)
- October 2024 (4)
- September 2024 (17)
- May 2024 (4)
- April 2024 (2)
- February 2024 (8)
- January 2024 (1)
- December 2023 (3)
- November 2023 (2)
- October 2023 (1)
- September 2023 (1)
- August 2023 (4)
- July 2023 (1)
- April 2023 (1)
- March 2023 (1)
- January 2023 (5)
- December 2022 (3)
- November 2022 (3)
- October 2022 (9)
- September 2022 (9)
- August 2022 (7)
- July 2022 (2)
- June 2022 (7)
- May 2022 (5)
- April 2022 (7)
- March 2022 (7)
- February 2022 (12)
- January 2022 (15)
- December 2021 (26)
- November 2021 (17)
- October 2021 (21)
- September 2021 (21)
- August 2021 (19)
- July 2021 (13)
- June 2021 (30)
- May 2021 (7)
- April 2021 (9)
- March 2021 (25)
- February 2021 (10)
- January 2021 (17)
- December 2020 (21)
- November 2020 (5)
- October 2020 (5)
- September 2020 (12)
- August 2020 (3)
- July 2020 (5)
- June 2020 (1)
- May 2020 (1)
- April 2020 (1)
- March 2020 (19)
- February 2020 (9)
- January 2020 (12)
- December 2019 (4)
- November 2019 (4)
- October 2019 (11)
- September 2019 (8)
- August 2019 (14)
- July 2019 (23)
- June 2019 (15)
- May 2019 (16)
- April 2019 (21)
- March 2019 (14)
- February 2019 (7)
- January 2019 (17)
- December 2018 (7)
- November 2018 (14)
- October 2018 (12)
- September 2018 (18)
- August 2018 (22)
- July 2018 (15)
- June 2018 (38)
- May 2018 (14)
- April 2018 (19)
- March 2018 (25)
- February 2018 (15)
- January 2018 (17)
- December 2017 (20)
- November 2017 (15)
- October 2017 (19)
- September 2017 (8)
- August 2017 (13)
- July 2017 (11)
- June 2017 (6)
- May 2017 (10)
- April 2017 (23)
- March 2017 (18)
- February 2017 (17)
- January 2017 (15)
- December 2016 (19)
- November 2016 (31)
- October 2016 (38)
- September 2016 (33)
- August 2016 (40)
- July 2016 (53)
- June 2016 (50)
- May 2016 (50)
- April 2016 (70)
- March 2016 (45)
- February 2016 (49)
- January 2016 (41)
- December 2015 (37)
- November 2015 (37)
- October 2015 (49)
- September 2015 (35)
- August 2015 (29)
- July 2015 (25)
- June 2015 (28)
- May 2015 (21)
- April 2015 (41)
- March 2015 (51)
- February 2015 (23)
- January 2015 (33)
- December 2014 (46)
- November 2014 (31)
- October 2014 (38)
- September 2014 (74)
- August 2014 (86)
- July 2014 (101)
- June 2014 (170)
- May 2014 (98)
- April 2014 (113)
- March 2014 (80)
- February 2014 (63)
- January 2014 (70)
- December 2013 (100)
- November 2013 (91)
- October 2013 (102)
- September 2013 (104)
- August 2013 (114)
- July 2013 (129)
- June 2013 (131)
- May 2013 (155)
- April 2013 (109)
- March 2013 (105)
- February 2013 (81)
Categories
- 0rphan drug status (225)
- 2020 APPROVALS (48)
- accelerated approval (1)
- ACH 4471 (1)
- Acute Hepatic Porphyria (1)
- Acute myeloid leukaemia (1)
- African medicine (6)
- AIDS (7)
- Alzheimer's (4)
- ANAESTHETIC (1)
- anaesthetics (4)
- analgesic (3)
- ANALYTICAL (1)
- ANDA (5)
- Anthony crasto (42)
- ANTI ACNE (2)
- ANTI ANDROGEN (1)
- ANTI DEPRESSANT (1)
- ANTI DIABETES (4)
- ANTI DOPING (1)
- ANTI FUNGAL (6)
- ANTI HBV (1)
- ANTI INFLAMATORY (8)
- anti tubeculosis (1)
- antibacterial (10)
- Antibiotics (6)
- ANTIBODIES (54)
- ANTICONVULSANT (1)
- Antiemetic (2)
- antiglaucoma (1)
- Antihypertensives (3)
- antiinfluenza (2)
- antimalarials (12)
- Antineoplastic (53)
- antiobesity (1)
- Antispasmodic (1)
- ANTITUSSIVE (1)
- Antiulcerative (2)
- Antivirals (10)
- APPROVALS 2021 (86)
- APPROVALS 2022 (49)
- APPROVALS 2023 (35)
- approvals 2024 (55)
- APPROVALS 2025 (32)
- APPROVALS 2026 (3)
- ARAB MEDICINE (5)
- Atopic dermatitis (1)
- Australia (1)
- AUSTRALIA 2021 (1)
- AWARD (13)
- AYURVEDA (78)
- Biosimilar drugs (39)
- BLA (4)
- BLOGS (28)
- Breakthrough Therapy Designation (69)
- canada (4)
- CANADA 2021 (1)
- cancer (170)
- cfda (6)
- CHINA 2011 (1)
- CHINA 2012 (1)
- CHINA 2018 (1)
- china 2019 (1)
- CHINA 2021 (5)
- CHINA 2022 (5)
- CHINA 2023 (4)
- CHINA 2024 (20)
- CHINA 2025 (1)
- china pipeline (37)
- CHINESE HERBS (24)
- chronic kidney disease (1)
- CLINICAL TRIALS (10)
- COMPANIES (81)
- conference (7)
- Contrast agent (7)
- CORONAVIRUS (48)
- COVID-19 (46)
- CRL (1)
- CYSTIC FIBRIOSIS (2)
- Dengue (2)
- DEUTERATED (1)
- DIABETES (57)
- DNDi (1)
- Drug discovery (22)
- DRUG MARKETING (5)
- DRUG REVIEW (8)
- Duchenne muscular dystrophy (1)
- EMA (4)
- EU 2003 (1)
- EU 2005 (1)
- EU 2014 (5)
- EU 2015 (6)
- EU 2016 (4)
- EU 2017 (8)
- EU 2018 (5)
- EU 2019 (1)
- EU 2020 (5)
- EU 2021 (11)
- EU 2022 (4)
- EU 2023 (4)
- EU 2024 (1)
- EU PIPELINE (61)
- EU SUBMISSION (32)
- EXTENDED USE (1)
- FAST TRACK (4)
- FAST TRACK FDA (78)
- FDA 15 (4)
- FDA 2010 (2)
- FDA 2011 (1)
- FDA 2012 (7)
- FDA 2013 (6)
- FDA 2014 (53)
- FDA 2015 (39)
- FDA 2016 (23)
- FDA 2017 (58)
- FDA 2018 (53)
- FDA 2019 (44)
- FDA 2020 (34)
- fda 2021 (45)
- FDA 2022 (24)
- FDA 2023 (32)
- fda 2023, approvals 2023 (1)
- FDA 2024 (29)
- FDA 2025 (33)
- FDA 2026 (4)
- Featured product (1)
- female sexual dysfunction (1)
- FIBROMYGALIA (1)
- flow synthesis (14)
- FORMULATION (14)
- GAIN (1)
- Gastric Proton Pump Inhibitor (1)
- Generating Antibiotic Incentives Now (1)
- GENERIC DRUG (106)
- GENERICS (22)
- glenmark (18)
- GLENMARK (16)
- GLIPTIN (1)
- GMP (10)
- gout (1)
- Greek medicine (3)
- hepatitis C viral infections (1)
- Herbals (8)
- HOMEOPATHY (3)
- HORMONE (1)
- Human medicines European Public Assessment Report EPAR (2)
- Hypertension (1)
- hypnotic (1)
- idiopathic pulmonary fibrosis (3)
- IMPLANTS (2)
- IND (1)
- IND 2017 (2)
- IND 2018 (3)
- IND 2021 (8)
- IND Filed (2)
- INDIA 2020 (5)
- INDIA 2022 (3)
- Insomnia (1)
- Investigational device exemption (IDE) approval (2)
- JAPAN 2009 (1)
- JAPAN 2013 (1)
- JAPAN 2014 (1)
- JAPAN 2015 (2)
- JAPAN 2016 (1)
- japan 2017 (6)
- JAPAN 2018 (10)
- japan 2019 (8)
- japan 2020 (10)
- JAPAN 2021 (11)
- JAPAN 2022 (14)
- Japan marketing (34)
- Japan pipeline (38)
- Korea (3)
- korea 2012 (2)
- korea 2016 (1)
- korea 2019 (1)
- Lactams (1)
- MAA (3)
- Macrolides (2)
- Malaria (17)
- MANUFACTURING (8)
- Market info (3)
- Marketing authorisation application EU (4)
- MEDICAL DEVICE (9)
- MIGRAINE (1)
- molybdenum cofactor deficiency (1)
- MONKEY POX (1)
- Monoclonal antibody (89)
- Nanotechnology (5)
- NCE (2)
- NDA (51)
- NDA JAPAN (3)
- neuropathic pain (2)
- Neutraceuticals (1)
- New Drug Application Resubmission FDA (5)
- NEW DRUGS (76)
- New drugs canada (5)
- New drugs china (6)
- New drugs EU (10)
- New drugs Japan (2)
- NEW USE DRUGS (1)
- Newzealand (2)
- NICE (2)
- non-Hodgkin's lymphoma (1)
- NUCLEOTIDES (1)
- obesity (3)
- OCCULAR (2)
- ocular hypertension (1)
- organic chemistry (12)
- osteoarthritis (2)
- PAIN (2)
- Parkinson's disease (1)
- PATENT (93)
- PATENTS (82)
- Peptide drugs (56)
- PFIZER (1)
- PHASE 1 (90)
- PHASE 3 (25)
- PHASE1 (145)
- Phase2 drugs (282)
- Phase3 drugs (306)
- plaque psoriasis (1)
- POLYMORPH (10)
- Preclinical china (14)
- Preclinical drugs (121)
- Premture ejaculation (3)
- prime designation (1)
- Priority review (90)
- PROCESS (39)
- Promising clips (9)
- Psoriasis (1)
- QbD (2)
- QIDP (23)
- radio labelled (4)
- RADIOACTIVE AGENT (5)
- RADIOLABELLED (10)
- Rare disease (3)
- RARE PEDIATRIC DISEASE REVIEW (2)
- Regenerative medicine (4)
- Regulatory (74)
- Resubmission FDA (2)
- REVIEW (10)
- rheumatoid arthritis (3)
- SCHLEROSING AGENT (2)
- Scottish Medicines Consortium (1)
- sex arousal (2)
- SFDA FAST TRACK (2)
- SINGAPORE (4)
- sNDA (12)
- sNDA JAPAN (2)
- Solid tumours (2)
- spectroscopy (16)
- SPOTLIGHT (53)
- Stem cells (3)
- SYNTHESIS (10)
- TIVE (1)
- TOXINS (1)
- Translational medicine (2)
- Transverse myletis (2)
- Tropical Disease Priority Review Voucher (1)
- UK (1)
- UNANI MEDICINE (2)
- Uncategorized (2,289)
- US HERBS (7)
- USFDA SUBMISSION (1)
- vaccine (26)
- veterinary (8)
- virus (1)
- VITAMINS (3)
- x ray contrast agent (10)
Recent Posts
- Remlifanserin March 9, 2026
- Relicpixant March 8, 2026
- Ranosidenib March 7, 2026
- Progerinin March 6, 2026
- Lead (212Pb) bamzireotide navoxetan March 5, 2026
- Pixavir marboxil March 4, 2026
- Navepegritide March 2, 2026
- Pebezertinib March 1, 2026
- Oveporexton February 28, 2026
- Orenasitecan February 27, 2026
- Milsaperidone February 26, 2026
- Olomorasib February 25, 2026
- Ocadusertib February 24, 2026
- Nivegacetor February 23, 2026
- Nispomeben February 21, 2026
ORGANIC SPECTROSCOPY
Read all about Organic Spectroscopy on
ORGANIC SPECTROSCOPY INTERNATIONAL


SUBSCRIBE
Enter your email address:
Delivered by FeedBurner
Subscribe to New Drug Approvals by Email
Join 37.8K other subscribers
DR ANTHONY MELVIN CRASTO Ph.D
DR ANTHONY MELVIN CRASTO, Born in Mumbai in 1964 and graduated from Mumbai University, Completed his Ph.D from ICT, 1991,Matunga, Mumbai, India, in Organic Chemistry, The thesis topic was Synthesis of Novel Pyrethroid Analogues, Currently he is working with AFRICURE PHARMA, ROW2TECH, NIPER-G, Department of Pharmaceuticals, Ministry of Chemicals and Fertilizers, Govt. of India as ADVISOR, earlier assignment was with GLENMARK LIFE SCIENCES LTD, as CONSUlTANT, Retired from GLENMARK in Jan2022 Research Centre as Principal Scientist, Process Research (bulk actives) at Mahape, Navi Mumbai, India. Total Industry exp 32 plus yrs, Prior to joining Glenmark, he has worked with major multinationals like Hoechst Marion Roussel, now Sanofi, Searle India Ltd, now RPG lifesciences, etc. He has worked with notable scientists like Dr K Nagarajan, Dr Ralph Stapel, Prof S Seshadri, etc, He did custom synthesis for major multinationals in his career like BASF, Novartis, Sanofi, etc., He has worked in Discovery, Natural products, Bulk drugs, Generics, Intermediates, Fine chemicals, Neutraceuticals, GMP, Scaleups, etc, he is now helping millions, has 9 million plus hits on Google on all Organic chemistry websites. His friends call him Open superstar worlddrugtracker. His New Drug Approvals, Green Chemistry International, All about drugs, Eurekamoments, Organic spectroscopy international, etc in organic chemistry are some most read blogs He has hands on experience in initiation and developing novel routes for drug molecules and implementation them on commercial scale over a 32 PLUS year tenure till date Feb 2023, Around 35 plus products in his career. He has good knowledge of IPM, GMP, Regulatory aspects, he has several International patents published worldwide . He has good proficiency in Technology transfer, Spectroscopy, Stereochemistry, Synthesis, Polymorphism etc., He suffered a paralytic stroke/ Acute Transverse mylitis in Dec 2007 and is 90 %Paralysed, He is bound to a wheelchair, this seems to have injected feul in him to help chemists all around the world, he is more active than before and is pushing boundaries, He has 100 million plus hits on Google, 2.5 lakh plus connections on all networking sites, 100 Lakh plus views on dozen plus blogs, 227 countries, 7 continents, He makes himself available to all, contact him on +91 9323115463, email amcrasto@gmail.com, Twitter, @amcrasto , He lives and will die for his family, 90% paralysis cannot kill his soul., Notably he has 38 lakh plus views on New Drug Approvals Blog in 227 countries......https://newdrugapprovals.wordpress.com/ , He appreciates the help he gets from one and all, Friends, Family, Glenmark, Readers, Wellwishers, Doctors, Drug authorities, His Contacts, Physiotherapist, etc He has total of 32 International and Indian awards
Verified Services
Pages
- About me
- All about drugs
- Bioavailability
- CLINICAL TRIALS
- Combinatorial Chemistry in Drug Design
- DISCLAIMER
- Drug Basics
- DRUG COMPANIES
- DRUG DESIGN
- DRUG DISCOVERY
- Drug Information
- DRUG NEWS
- DRUG PORTALS
- Drug resistance
- DRUG SITES
- DRUG SYNTHESIS
- DRUGS NDA
- DRUGS, FDA2012
- FDA -US
- GMP
- HEALTH CANADA
- HELP ME/ ACCOLADES
- HSA SINGAPORE
- INDA
- KOREA DRUG APPROVAL PROCESS
- MHLW JAPAN, 厚生労働省
- NMPA CHINA
- PATENT EXPIRY DATES 1/3
- PATENT EXPIRY DATES 2/3
- PATENT EXPIRY DATES 3/3
- Patent portals
- Polymorphism
- Regulations and Guidance
- Scientificupdate (UK) on a roll
- sFDA CHINA
- SOUTH AFRICA, MCC
- SPECIAL DRUGS
- Traditional Medicine
- WHAT IS A DRUG
- World drug tracker
Archives
- March 2026 (8)
- February 2026 (25)
- January 2026 (28)
- December 2025 (31)
- November 2025 (31)
- October 2025 (22)
- September 2025 (24)
- August 2025 (27)
- July 2025 (25)
- June 2025 (23)
- May 2025 (34)
- April 2025 (32)
- March 2025 (4)
- October 2024 (4)
- September 2024 (17)
- May 2024 (4)
- April 2024 (2)
- February 2024 (8)
- January 2024 (1)
- December 2023 (3)
- November 2023 (2)
- October 2023 (1)
- September 2023 (1)
- August 2023 (4)
- July 2023 (1)
- April 2023 (1)
- March 2023 (1)
- January 2023 (5)
- December 2022 (3)
- November 2022 (3)
- October 2022 (9)
- September 2022 (9)
- August 2022 (7)
- July 2022 (2)
- June 2022 (7)
- May 2022 (5)
- April 2022 (7)
- March 2022 (7)
- February 2022 (12)
- January 2022 (15)
- December 2021 (26)
- November 2021 (17)
- October 2021 (21)
- September 2021 (21)
- August 2021 (19)
- July 2021 (13)
- June 2021 (30)
- May 2021 (7)
- April 2021 (9)
- March 2021 (25)
- February 2021 (10)
- January 2021 (17)
- December 2020 (21)
- November 2020 (5)
- October 2020 (5)
- September 2020 (12)
- August 2020 (3)
- July 2020 (5)
- June 2020 (1)
- May 2020 (1)
- April 2020 (1)
- March 2020 (19)
- February 2020 (9)
- January 2020 (12)
- December 2019 (4)
- November 2019 (4)
- October 2019 (11)
- September 2019 (8)
- August 2019 (14)
- July 2019 (23)
- June 2019 (15)
- May 2019 (16)
- April 2019 (21)
- March 2019 (14)
- February 2019 (7)
- January 2019 (17)
- December 2018 (7)
- November 2018 (14)
- October 2018 (12)
- September 2018 (18)
- August 2018 (22)
- July 2018 (15)
- June 2018 (38)
- May 2018 (14)
- April 2018 (19)
- March 2018 (25)
- February 2018 (15)
- January 2018 (17)
- December 2017 (20)
- November 2017 (15)
- October 2017 (19)
- September 2017 (8)
- August 2017 (13)
- July 2017 (11)
- June 2017 (6)
- May 2017 (10)
- April 2017 (23)
- March 2017 (18)
- February 2017 (17)
- January 2017 (15)
- December 2016 (19)
- November 2016 (31)
- October 2016 (38)
- September 2016 (33)
- August 2016 (40)
- July 2016 (53)
- June 2016 (50)
- May 2016 (50)
- April 2016 (70)
- March 2016 (45)
- February 2016 (49)
- January 2016 (41)
- December 2015 (37)
- November 2015 (37)
- October 2015 (49)
- September 2015 (35)
- August 2015 (29)
- July 2015 (25)
- June 2015 (28)
- May 2015 (21)
- April 2015 (41)
- March 2015 (51)
- February 2015 (23)
- January 2015 (33)
- December 2014 (46)
- November 2014 (31)
- October 2014 (38)
- September 2014 (74)
- August 2014 (86)
- July 2014 (101)
- June 2014 (170)
- May 2014 (98)
- April 2014 (113)
- March 2014 (80)
- February 2014 (63)
- January 2014 (70)
- December 2013 (100)
- November 2013 (91)
- October 2013 (102)
- September 2013 (104)
- August 2013 (114)
- July 2013 (129)
- June 2013 (131)
- May 2013 (155)
- April 2013 (109)
- March 2013 (105)
- February 2013 (81)
Categories
- 0rphan drug status (225)
- 2020 APPROVALS (48)
- accelerated approval (1)
- ACH 4471 (1)
- Acute Hepatic Porphyria (1)
- Acute myeloid leukaemia (1)
- African medicine (6)
- AIDS (7)
- Alzheimer's (4)
- ANAESTHETIC (1)
- anaesthetics (4)
- analgesic (3)
- ANALYTICAL (1)
- ANDA (5)
- Anthony crasto (42)
- ANTI ACNE (2)
- ANTI ANDROGEN (1)
- ANTI DEPRESSANT (1)
- ANTI DIABETES (4)
- ANTI DOPING (1)
- ANTI FUNGAL (6)
- ANTI HBV (1)
- ANTI INFLAMATORY (8)
- anti tubeculosis (1)
- antibacterial (10)
- Antibiotics (6)
- ANTIBODIES (54)
- ANTICONVULSANT (1)
- Antiemetic (2)
- antiglaucoma (1)
- Antihypertensives (3)
- antiinfluenza (2)
- antimalarials (12)
- Antineoplastic (53)
- antiobesity (1)
- Antispasmodic (1)
- ANTITUSSIVE (1)
- Antiulcerative (2)
- Antivirals (10)
- APPROVALS 2021 (86)
- APPROVALS 2022 (49)
- APPROVALS 2023 (35)
- approvals 2024 (55)
- APPROVALS 2025 (32)
- APPROVALS 2026 (3)
- ARAB MEDICINE (5)
- Atopic dermatitis (1)
- Australia (1)
- AUSTRALIA 2021 (1)
- AWARD (13)
- AYURVEDA (78)
- Biosimilar drugs (39)
- BLA (4)
- BLOGS (28)
- Breakthrough Therapy Designation (69)
- canada (4)
- CANADA 2021 (1)
- cancer (170)
- cfda (6)
- CHINA 2011 (1)
- CHINA 2012 (1)
- CHINA 2018 (1)
- china 2019 (1)
- CHINA 2021 (5)
- CHINA 2022 (5)
- CHINA 2023 (4)
- CHINA 2024 (20)
- CHINA 2025 (1)
- china pipeline (37)
- CHINESE HERBS (24)
- chronic kidney disease (1)
- CLINICAL TRIALS (10)
- COMPANIES (81)
- conference (7)
- Contrast agent (7)
- CORONAVIRUS (48)
- COVID-19 (46)
- CRL (1)
- CYSTIC FIBRIOSIS (2)
- Dengue (2)
- DEUTERATED (1)
- DIABETES (57)
- DNDi (1)
- Drug discovery (22)
- DRUG MARKETING (5)
- DRUG REVIEW (8)
- Duchenne muscular dystrophy (1)
- EMA (4)
- EU 2003 (1)
- EU 2005 (1)
- EU 2014 (5)
- EU 2015 (6)
- EU 2016 (4)
- EU 2017 (8)
- EU 2018 (5)
- EU 2019 (1)
- EU 2020 (5)
- EU 2021 (11)
- EU 2022 (4)
- EU 2023 (4)
- EU 2024 (1)
- EU PIPELINE (61)
- EU SUBMISSION (32)
- EXTENDED USE (1)
- FAST TRACK (4)
- FAST TRACK FDA (78)
- FDA 15 (4)
- FDA 2010 (2)
- FDA 2011 (1)
- FDA 2012 (7)
- FDA 2013 (6)
- FDA 2014 (53)
- FDA 2015 (39)
- FDA 2016 (23)
- FDA 2017 (58)
- FDA 2018 (53)
- FDA 2019 (44)
- FDA 2020 (34)
- fda 2021 (45)
- FDA 2022 (24)
- FDA 2023 (32)
- fda 2023, approvals 2023 (1)
- FDA 2024 (29)
- FDA 2025 (33)
- FDA 2026 (4)
- Featured product (1)
- female sexual dysfunction (1)
- FIBROMYGALIA (1)
- flow synthesis (14)
- FORMULATION (14)
- GAIN (1)
- Gastric Proton Pump Inhibitor (1)
- Generating Antibiotic Incentives Now (1)
- GENERIC DRUG (106)
- GENERICS (22)
- glenmark (18)
- GLENMARK (16)
- GLIPTIN (1)
- GMP (10)
- gout (1)
- Greek medicine (3)
- hepatitis C viral infections (1)
- Herbals (8)
- HOMEOPATHY (3)
- HORMONE (1)
- Human medicines European Public Assessment Report EPAR (2)
- Hypertension (1)
- hypnotic (1)
- idiopathic pulmonary fibrosis (3)
- IMPLANTS (2)
- IND (1)
- IND 2017 (2)
- IND 2018 (3)
- IND 2021 (8)
- IND Filed (2)
- INDIA 2020 (5)
- INDIA 2022 (3)
- Insomnia (1)
- Investigational device exemption (IDE) approval (2)
- JAPAN 2009 (1)
- JAPAN 2013 (1)
- JAPAN 2014 (1)
- JAPAN 2015 (2)
- JAPAN 2016 (1)
- japan 2017 (6)
- JAPAN 2018 (10)
- japan 2019 (8)
- japan 2020 (10)
- JAPAN 2021 (11)
- JAPAN 2022 (14)
- Japan marketing (34)
- Japan pipeline (38)
- Korea (3)
- korea 2012 (2)
- korea 2016 (1)
- korea 2019 (1)
- Lactams (1)
- MAA (3)
- Macrolides (2)
- Malaria (17)
- MANUFACTURING (8)
- Market info (3)
- Marketing authorisation application EU (4)
- MEDICAL DEVICE (9)
- MIGRAINE (1)
- molybdenum cofactor deficiency (1)
- MONKEY POX (1)
- Monoclonal antibody (89)
- Nanotechnology (5)
- NCE (2)
- NDA (51)
- NDA JAPAN (3)
- neuropathic pain (2)
- Neutraceuticals (1)
- New Drug Application Resubmission FDA (5)
- NEW DRUGS (76)
- New drugs canada (5)
- New drugs china (6)
- New drugs EU (10)
- New drugs Japan (2)
- NEW USE DRUGS (1)
- Newzealand (2)
- NICE (2)
- non-Hodgkin's lymphoma (1)
- NUCLEOTIDES (1)
- obesity (3)
- OCCULAR (2)
- ocular hypertension (1)
- organic chemistry (12)
- osteoarthritis (2)
- PAIN (2)
- Parkinson's disease (1)
- PATENT (93)
- PATENTS (82)
- Peptide drugs (56)
- PFIZER (1)
- PHASE 1 (90)
- PHASE 3 (25)
- PHASE1 (145)
- Phase2 drugs (282)
- Phase3 drugs (306)
- plaque psoriasis (1)
- POLYMORPH (10)
- Preclinical china (14)
- Preclinical drugs (121)
- Premture ejaculation (3)
- prime designation (1)
- Priority review (90)
- PROCESS (39)
- Promising clips (9)
- Psoriasis (1)
- QbD (2)
- QIDP (23)
- radio labelled (4)
- RADIOACTIVE AGENT (5)
- RADIOLABELLED (10)
- Rare disease (3)
- RARE PEDIATRIC DISEASE REVIEW (2)
- Regenerative medicine (4)
- Regulatory (74)
- Resubmission FDA (2)
- REVIEW (10)
- rheumatoid arthritis (3)
- SCHLEROSING AGENT (2)
- Scottish Medicines Consortium (1)
- sex arousal (2)
- SFDA FAST TRACK (2)
- SINGAPORE (4)
- sNDA (12)
- sNDA JAPAN (2)
- Solid tumours (2)
- spectroscopy (16)
- SPOTLIGHT (53)
- Stem cells (3)
- SYNTHESIS (10)
- TIVE (1)
- TOXINS (1)
- Translational medicine (2)
- Transverse myletis (2)
- Tropical Disease Priority Review Voucher (1)
- UK (1)
- UNANI MEDICINE (2)
- Uncategorized (2,289)
- US HERBS (7)
- USFDA SUBMISSION (1)
- vaccine (26)
- veterinary (8)
- virus (1)
- VITAMINS (3)
- x ray contrast agent (10)
Recent Comments
| shivkr2 on SPSR Excellence Award 2025 – S… | |
| Bonkasaurus on Infigratinib phosphate | |
| PALOVAROTENE | ORGAN… on Palovarotene | |
| Pfizer just purchase… on Temanogrel | |
| Pfizer To Cash In On… on Temanogrel |
SEARCH THIS BLOG
EVP 4593
April 6, 2017 1:38 pm / Leave a comment

EVP4593; EVP 4593; EVP-4593
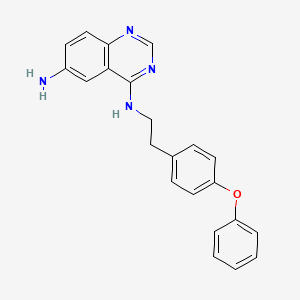
| M.Wt | 356.42 | 545380-34-5; QNZ (EVP4593); QNZ; 6-Amino-4-(4-phenoxyphenylethylamino)quinazoline; N4-(4-phenoxyphenethyl)quinazoline-4,6-diamine; | |
|---|---|---|---|
| Formula | C₂₂H₂₀N₄O | ||
| CAS No | 545380-34-5 |
QNZ(EVP4593) is a derivative of 6-aminoquinazoline class that has been previously isolated as an inhibitor of PMA/PHA-induced NF-κB pathway activation in Jurkat cells (IC50= 9 nM).
QNZ(EVP4593) is a derivative of 6-aminoquinazoline class that has been previously isolated as an inhibitor of PMA/PHA-induced NF-κB pathway activation in Jurkat cells (IC50= 9 nM).
IC50 Value: 9 nM [1]
Target: NF-kB signaling
in vitro: The efficacy of EVP4593 was dose-dependent in the range between 100 uM and 400 uM in the fly food. The EVP4593 had no significant effect on climbing performance of HD flies at 50 ?M. The EVP4593 had no toxic effects on Drosophila in the range of concentrations tested in our assays (50 – 400 ?M) [1]. Addition of 300 nM of EVP4593 resulted in strong attenuation of SOC Ca2+ influx in YAC128 MSN neurons. On average the amplitude of SOC Ca2+ entry in YAC128 MSN was reduced from 0.30 ± 0.02 (n = 29) in the presence of DMSO control to 0.11 ± 0.02 (n = 54) in the presence of 300 nM of EVP4593 (p < 0.001).
in vivo:
Paper
Identification of 4-N-[2-(4-phenoxyphenyl)ethyl]quinazoline-4,6-diamine as a novel, highly potent and specific inhibitor of mitochondrial complex I
*Corresponding authors
aJohann Wolfgang Goethe-University Frankfurt, Max-von-Laue-Straße 7, D-60438 Frankfurt am Main, Germany
E-mail: Nasiri@nmr.uni-frankfurt.de
bDepartment of Anaesthesiology, Intensive-Care Medicine and Pain Therapy, University Hospital Frankfurt, 60590 Frankfurt am Main, Germany
cStructural Bioenergetics Group, Institute of Biochemistry II, Medical School, Goethe-University, 60438 Frankfurt am Main, Germany
dCluster of Excellence Frankfurt “Macromolecular Complexes,”, Goethe-University, 60438 Frankfurt am Main, Germany
Abstract
By probing the quinone substrate binding site of mitochondrial complex I with a focused set of quinazoline-based compounds, we identified substitution patterns as being critical for the observed inhibition. The structure activity relationship study also resulted in the discovery of the quinazoline 4-N-[2-(4-phenoxyphenyl)ethyl]quinazoline-4,6-diamine (EVP4593) as a highly potent inhibitor of the multisubunit membrane protein. EVP4593 specifically and effectively reduces the mitochondrial complex I-dependent respiration with no effect on the respiratory chain complexes II–IV. Similar to established Q-site inhibitors, EVP4593 elicits the release of reactive oxygen species at the flavin site of mitochondrial complex I. Recently, EVP4593 was nominated as a lead compound for the treatment of Huntingtons disease. Our results challenge the postulated primary mode-of-action of EVP4593 as an inhibitor of NF-κB pathway activation and/or store-operated calcium influx.
![Graphical abstract Graphical abstract: Identification of 4-N-[2-(4-phenoxyphenyl)ethyl]quinazoline-4,6-diamine as a novel, highly potent and specific inhibitor of mitochondrial complex I](http://pubs.rsc.org/services/images/RSCpubs.ePlatform.Service.FreeContent.ImageService.svc/ImageService/image/GA?id=C6MD00655H)
PAPER
Bioorganic & Medicinal Chemistry (2003), 11(3), 383-391.
Abstract
We disclose here a new structural class of low-molecular-weight inhibitors of NF-κB activation that were designed and synthesized by starting from quinazoline derivative 6a. Structure–activity relationship (SAR) studies based on 6a elucidated the structural requirements essential for the inhibitory activity toward NF-κB transcriptional activation, and led to the identification of the 6-amino-4-phenethylaminoquinazoline skeleton as the basic framework. In this series of compounds, 11q, containing the 4-phenoxyphenethyl moiety at the C(4)-position, showed strong inhibitory effects on both NF-κB transcriptional activation and TNF-α production. Furthermore, 11q exhibited an anti-inflammatory effect on carrageenin-induced paw edema in rats.
Compound 11q exhibited a highly inhibitory activity toward NF-κB activation and also showed an anti-inflammatory effect.

- 11q (72 mg, 77% yield):
- mp 168–170 C;
- 1 H NMR (DMSO-d6) d 8.33 (br s, 2H), 7.45 (d, J=8.9 Hz, 1H), 7.40–7.34 (m, 2H), 7.28 (d, J=8.6 Hz, 2H), 7.20–7.07 (m, 3H), 6.98–6.92 (m, 4H), 5.59 (br s, 2H), 3.79–3.72 (m, 2H), 2.95 (t, J=7.3 Hz, 2H);
- MS (TOF) m/z 357 (M + H)+; anal. calcd for C22H20N4O 1.0H2O: C, 70.57; H, 5.65; N, 14.96. Found: C, 70.48; H, 5.60; N, 14.87.
- REF
- Bioorganic & Medicinal Chemistry (2003), 11(18), 3869-3878.
- JP 2004059454
- CN 1709259
- Bioorganic & Medicinal Chemistry Letters (2009), 19(19), 5665-5669
- Journal of Medicinal Chemistry (2014), 57(6), 2247-2257
| Patent ID | Patent Title | Submitted Date | Granted Date |
|---|---|---|---|
| US2010087374 | Methods for Treatment and Diagnosis of Pulmonary Diseases Based on the Expression of SERCA2 Protein | 2009-10-05 | 2010-04-08 |
| US2009177228 | Coated suture thread and production thereof | 2006-02-21 | 2009-07-09 |
| US2008139457 | Therapeutic compositions comprising chorionic gonadotropins and HMG CoA reductase inhibitors | 2006-09-14 | 2008-06-12 |
| US2014243425 | CONTROLLED RELEASE COMPOSITIONS FOR MODULATING FREE-RADICAL INDUCED DAMAGE AND METHODS OF USE THEREOF | 2014-05-01 | 2014-08-28 |
| US2013202537 | COMPOSITIONS FOR LABELING NERVES AND METHODS OF USE | 2011-09-02 | 2013-08-08 |
| US2013078224 | INDUCTION/MONITORING OF ARTERIOGENESIS USING SDF1 AND PDGFB OR INHIBITION OF PHD2 | 2011-03-30 | 2013-03-28 |
| US2012277199 | Modulation of Gel Temperature of Poloxamer-Containing Formulations | 2010-10-19 | 2012-11-01 |
| US2012134922 | PEPTIDES WHOSE UPTAKE IN CELLS IS CONTROLLABLE | 2010-07-15 | 2012-05-31 |
| US2012134931 | PEPTIDES WHOSE UPTAKE IN CELLS IS CONTROLLABLE | 2010-07-15 | 2012-05-31 |
| US8685372 | Peptides and aptamers for targeting of neuron or nerves | 2010-04-15 | 2014-04-01 |
| Patent ID | Patent Title | Submitted Date | Granted Date |
|---|---|---|---|
| US2015297598 | METHODS FOR TREATING RENAL DISEASE | 2013-11-20 | 2015-10-22 |
| US2015353604 | COMPOSITIONS FOR LABELING NERVES AND METHODS OF USE | 2015-06-10 | 2015-12-10 |
| US2015359902 | PRETARGETED ACTIVATABLE CELL PENETRATING PEPTIDE WITH INTRACELLULARLY RELEASABLE PRODRUG | 2014-01-29 | 2015-12-17 |
| US2016160263 | PERSONALIZED PROTEASE ASSAY TO MEASURE PROTEASE ACTIVITY IN NEOPLASMS | 2015-10-02 | 2016-06-09 |
| US2016199446 | CONTROLLED-RELEASE APOPTOSIS MODULATING COMPOSITIONS AND METHODS FOR THE TREATMENT OF OTIC DISORDERS | 2016-01-19 | 2016-07-14 |
//////////
C1=CC=C(C=C1)OC2=CC=C(C=C2)CCNC3=NC=NC4=C3C=C(C=C4)N
Share this:
- Print (Opens in new window) Print
- Share on X (Opens in new window) X
- Email a link to a friend (Opens in new window) Email
- Share on Pinterest (Opens in new window) Pinterest
- Share on LinkedIn (Opens in new window) LinkedIn
- Share on Mastodon (Opens in new window) Mastodon
- Share on Tumblr (Opens in new window) Tumblr
- Share on Reddit (Opens in new window) Reddit
- Share on Facebook (Opens in new window) Facebook