Home » Posts tagged 'ALZHIEMER'
Tag Archives: ALZHIEMER
Nivegacetor
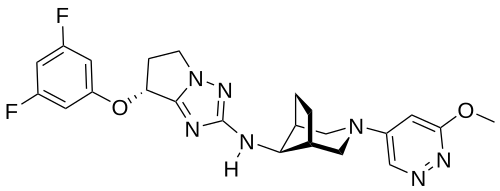
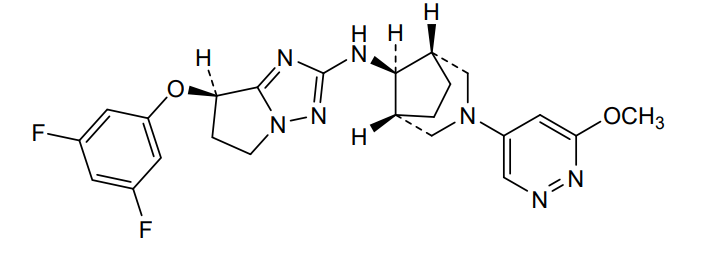
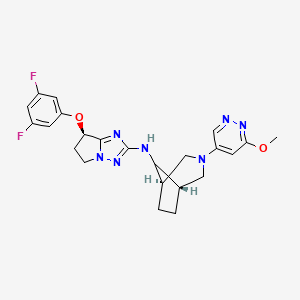
Nivegacetor
CAS 2443487-67-8
MF C23H25F2N7O2 MW 469.5 g/mol
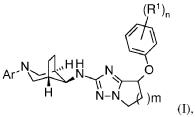
(7R)-7-(3,5-difluorophenoxy)-N-[(1S,5R)-3-(6-methoxypyridazin-4-yl)-3-azabicyclo[3.2.1]octan-8-yl]-6,7-dihydro-5H-pyrrolo[1,2-b][1,2,4]triazol-2-amine
(7R)-7-(3,5-difluorophenoxy)-N-[(1R,5S,8s)-3-(6-methoxypyridazin-4-yl)-3-azabicyclo[3.2.1]octan-8-yl]-6,7-dihydro5H-pyrrolo[1,2-b][1,2,4]triazol-2-amine
gamma secretase modulator, SF4J7MVJ56, RG 6289, RG-6289, ROCHE, ALZHIEMER,
Nivegacetor is a potent γ-secretase modulator.
Nivegacetor is an investigational gamma-secretase modulator being developed by Roche for the treatment of Alzheimer’s disease.[1] The compound is also known by its development code name RG6289 and represents a second-generation gamma-secretase modulator designed to selectively alter amyloid beta peptide production while avoiding the toxicity issues associated with first-generation compounds.[2]
Mechanism of action
Nivegacetor is a gamma-secretase modulator (GSM) that targets the gamma-secretase enzyme complex, which plays a central role in the production of amyloid beta peptides implicated in the pathogenesis of Alzheimer’s disease.[1] It specifically modulates the catalytic subunit presenilin-1 (PSEN1), stabilizing the interaction between the complex and the amyloid precursor protein (APP) at the enzyme’s active site. This stabilization increases the processivity of APP cleavage—that is, the enzyme’s ability to carry out sequential cleavage steps before releasing the APP substrate.[3]
Unlike gamma-secretase inhibitors that completely block enzyme function and cause significant side effects, nivegacetor selectively reduces the production of amyloidogenic long amyloid beta peptides, particularly Aβ42 and Aβ40 that form insoluble amyloid fibrils, while simultaneously increasing the formation of shorter, non-amyloidogenic species such as Aβ38 and Aβ37. The compound demonstrates high potency with an IC50 below 10 nM for gamma-secretase modulation of APP cleavage, and importantly shows no effect on the processing of other gamma-secretase substrates, potentially avoiding the toxicity issues that plagued earlier compounds.[2]
A Study of Donanemab, RG6289, or the Combination of Donanemab and RG6289 in Presenilin 1 (PSEN1) E280A Mutation Carriers for the Treatment of Autosomal-Dominant Alzheimer’s Disease
CTID: NCT06996730
Phase: Phase 2/Phase 3
Status: Not yet recruiting
Date: 2025-08-03
- OriginatorRoche
- ClassAntidementias; Azabicyclo compounds; Bridged bicyclo compounds; Ethers; Fluorobenzenes; Methyl ethers; Pyridazines; Pyrrolidines; Triazoles
- Mechanism of ActionAmyloid precursor protein secretase modulator
- Phase IIAlzheimer’s disease
- 03 Dec 2025Efficacy data from a phase II trial in Alzheimer’s disease presented at the Alzheimer’s Association International Conference 2025 (AAIC-2025)
- 13 Aug 2025Chemical structure information added.
- 14 Nov 2024Banner Alzheimers Institute and Neurosciences Group at the University of Antioquia (GNA) in Medellin plans a clinical trial for Alzheimer’s-disease (Monotherapy, Prevention, In adults), in fall 2025 (IV) (NCT06996730)
SYN
xamples 1 and 2
(R)-7-(3,5-difluorophenoxy)-N-((1R,5S,8s)-3-(6-methoxypyridazin-4-yl)-3-azabicyclo[3.2.1]octan-8-yl)-6,7-dihydro-5H-pyrrolo[1,2-b][1,2,4]triazol-2-amine and (S)-7-(3,5-difluorophenoxy)-N-((1R,5S,8s)-3-(6-methoxypyridazin-4-yl)-3-azabicyclo[3.2.1]octan-8-yl)-6,7-dihydro-5H-pyrrolo[1,2-b][1,2,4]triazol-2-amine
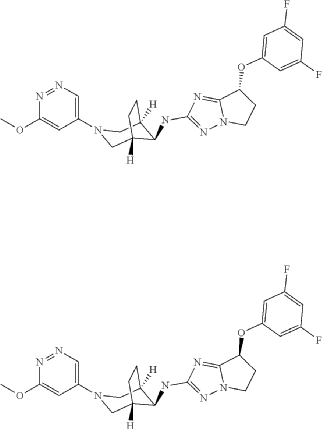
A Buchwald type coupling using the general procedure 1, between 2-bromo-7-(3,5-difluorophenoxy)-6,7-dihydro-5H-pyrrolo[1,2-b][1,2,4]triazole 7-1 and (1R,5S,8S)-3-(6-methoxypyridazin-4-yl)-3-azabicyclo[3.2.1]octan-8-amine 8-2, followed by a separation of the enantiomeres by preparative chiral HPLC afforded the title products as white solid (example 1): 27 mg, MS (ES+) m/z: 470.2 [(M+H) +] and (example 2): 28 mg, MS (ES+) m/z: 470.2 [(M+H) +].
PAT
Examples 1 and 2
(R)-7-(3,5-difluorophenoxy)-N-((lR,5S,8s)-3-(6-methoxypyridazin-4-yl)-3- azabicyclo[3.2.1]octan-8-yl)-6,7-dihydro-5H-pyrrolo[l,2-b][l,2,4]triazol-2-amine
and
(S)-7-(3,5-difluorophenoxy)-N-((lR,5S,8s)-3-(6-methoxypyridazin-4-yl)-3- azabicyclo[3.2.1]octan-8-yl)-6,7-dihydro-5H-pyrrolo[l,2-b][l,2,4]triazol-2-amine
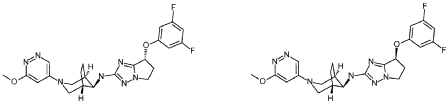
A Buchwald type coupling using the general procedure 1, between 2-bromo-7-(3,5-difluorophenoxy)-6,7-dihydro-5H-pyrrolo[l,2-b][l,2,4]triazole 7-1 and (lR,5S,8S)-3-(6-methoxypyridazin-4-yl)-3-azabicyclo[3.2.1]octan-8-amine 8-2, followed by a separation of the enantiomeres by preparative chiral HPLC afforded the title products as white solid (example 1): 27 mg, MS (ES+) m/z 470.2 [(M+H)+] and (example 2): 28 mg, MS (ES+) m/z 470.2 [(M+H)+]
PAT
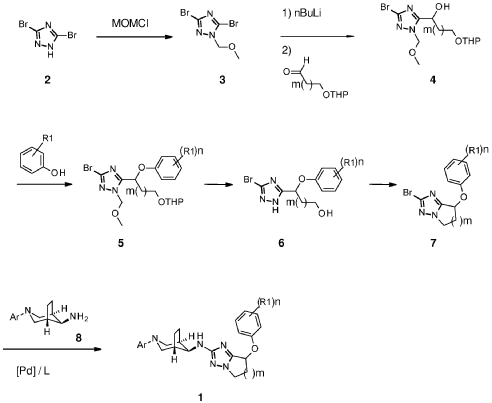
- 6, 7-Dihydro-5H-pyrrolo [1,2-B ] [1,2,4] triazol-2-amine derivativesPublication Number: CN-117946116-APriority Date: 2018-12-13
- 7-phenoxy-n-(3-azabicyclo[3.2.1]octan-8-yl)-6,7-dihydro-5h-pyrrolo[1,2-b][1,2,4]triazol-2-amine derivatives and related compounds as gamma-secretase modulators for the treatment of alzheimer’s diseasePublication Number: US-2022056036-A1Priority Date: 2018-12-13
- 6,7-Dihydro-5H-pyrrolo[1,2-B][1,2,4]triazole-2-amine derivativesPublication Number: CN-113179641-BPriority Date: 2018-12-13Grant Date: 2024-01-02
- 7-phenoxy-n-(3-azabicyclo[3.2.1]octan-8-yl)-6,7-dihydro-5h-pyrrolo[1,2-b][1,2,4]triazol-2-amine derivatives and related compounds as gamma-secretase modulators for the treatment of alzheimer’s diseasePublication Number: WO-2020120521-A1Priority Date: 2018-12-13
- 7-phenoxy-N-(3-azabicyclo[3.2.1]octan-8-yl-6,7-dihydro-5H-pyrrolo[1,2-b][1,2,4]triazol-2-amine derivatives and related compounds as gamma- secretase modulators for the treatment of alzheimer’s diseasePublication Number: US-12195470-B2Priority Date: 2018-12-13Grant Date: 2025-01-14



AS ON OCT2025 4.511 LAKHS VIEWS ON BLOG WORLDREACH AVAILABLEFOR YOUR ADVERTISEMENT

join me on Linkedin
Anthony Melvin Crasto Ph.D – India | LinkedIn
join me on Researchgate
RESEARCHGATE

join me on Facebook
Anthony Melvin Crasto Dr. | Facebook
join me on twitter
Anthony Melvin Crasto Dr. | twitter
+919321316780 call whatsaapp
EMAIL. amcrasto@gmail.com

Clinical development
Phase I studies
Nivegacetor has completed Phase I clinical trials in healthy volunteers, where it demonstrated a favorable safety profile and dose-dependent pharmacodynamic effects.[3] The study showed that treatment with nivegacetor resulted in a dose-dependent shift in amyloid beta monomers in cerebrospinal fluid (CSF), with significant reductions in Aβ42 levels and corresponding increases in shorter amyloid beta species.[4]
The Phase I results were presented at the 2023 Clinical Trials on Alzheimer’s Disease (CTAD) conference, where researchers reported that nivegacetor appeared safe and effectively shifted amyloid beta production toward smaller, less aggregation-prone peptides.[3]
Phase II studies
Based on the positive Phase I results, nivegacetor has been selected for advancement to Phase II clinical trials for Alzheimer’s disease treatment.[3] The dose selection for the Phase II study was informed by population pharmacokinetic/pharmacodynamic modeling derived from the Phase I data.[5][6][7]
Historical context
Nivegacetor represents a significant advancement in gamma-secretase modulator development, addressing the limitations of first-generation compounds that failed due to toxicology problems.[2] Previous attempts at gamma-secretase modulation were hampered by safety concerns and off-target effects, leading to the discontinuation of several promising candidates in the 2000s and early 2010s.[2] The development of nivegacetor as a second-generation GSM reflects improved understanding of gamma-secretase biology and more selective targeting approaches.[8]
References
- “nivegacetor | Ligand page”. IUPHAR/BPS Guide to IMMUNOPHARMACOLOGY. International Union of Basic and Clinical Pharmacology (IUPHAR). Retrieved 22 July 2025.
- “RG6289”. ALZFORUM. Archived from the original on 9 October 2024. Retrieved 22 July 2025.
- “Second-Generation γ-Secretase Modulator Heads to Phase 2”. ALZFORUM. Retrieved 22 July 2025.
- “Y-Secretase Modulator RG6289 Produces Dose-Dependent Shift of Amyloid-ß Monomers in Phase 1 Study”. Neurology live. 30 July 2024. Retrieved 22 July 2025.
- “RG6289, a new γ-secretase modulator for the treatment of Alzheimer’s disease: Dose selection for a phase II trial based on population PK/PD modeling”. medically.gene.com. Retrieved 22 July 2025.
- Banner Health (21 May 2025). A Double-Blind, Placebo-Controlled, Double-Dummy Study of Donanemab and RG6289 in PSEN1 E280A Mutation Carriers, and in Non-Randomized, Placebo-Treated Non-Carriers From the Same Kindred, to Evaluate the Efficacy and Safety of Donanemab, RG6289, or the Combination of Donanemab and RG6289, in the Treatment of Autosomal-Dominant Alzheimer’s Disease (Report). clinicaltrials.gov. NCT06996730.
- azalzeditor (19 November 2024). “New Alzheimer’s prevention trial receives $74.5 million NIH grant”. Arizona Alzheimer’s Consortium. Retrieved 22 July 2025.
- Nordvall G, Lundkvist J, Sandin J (16 October 2023). “Gamma-secretase modulators: a promising route for the treatment of Alzheimer’s disease”. Frontiers in Molecular Neuroscience. 16 1279740. doi:10.3389/fnmol.2023.1279740. ISSN 1662-5099. PMC 10613654. PMID 37908487.
| Identifiers | |
|---|---|
| IUPAC name | |
| CAS Number | 2443487-67-8 |
| PubChem CID | 153606610 |
| IUPHAR/BPS | 13509 |
| UNII | SF4J7MVJ56 |
| KEGG | D13199 |
| Chemical and physical data | |
| Formula | C23H25F2N7O2 |
| Molar mass | 469.497 g·mol−1 |
| 3D model (JSmol) | Interactive image |
| SMILES | |
| InChI | |
/////////nivegacetor, gamma secretase modulator, SF4J7MVJ56, RG 6289, RG-6289, ROCHE, ALZHIEMER,
RG-1577, EVT 302, Sembragiline, RO-4602522
RG-1577, EVT 302, Sembragiline, RO-4602522
CAS 676479-06-4, MW 342.36
- C19 H19 F N2 O3
- Acetamide, N-[(3S)-1-[4-[(3-fluorophenyl)methoxy]phenyl]-5-oxo-3-pyrrolidinyl]-
UNII-K3W9672PNJ
RG-1577, a selective and reversible monoamine oxidase B inhibitor, for treating AD (phase 2 clinical, as of May 2015).
Family members of the product case for RG-1577 (WO2004026825) hold protection in EU until 2023 and expire in US in 2024 with US154 extension. Follows on from WO2006097197, claiming a process for preparing RG-1577.
Alzheimer‘s Disease is a brain disease that slowly destroys memory and thinking skills, up to loss of the ability to carry out the simplest tasks. It is the most common cause of dementia among older people. Mild Alzheimer‘s Disease manifests itself in memory loss and small changes in other cognitive abilities, e.g getting lost, trouble handling money and managing daily tasks, having some mood and personality changes, etc.
In the stage of Moderate Alzheimer‘s Disease, the control of language, reasoning, sensory processing, and conscious thought are impacted. Memory loss and con usion grow worse, e.g patients have problems recognizing family and friends and become unable to learn new things, etc. hallucinations, delusions, and paranoia may occur. .Severe Alzheimer‘s Disease is the final stage. Patients cannot communicate anymore and are completely dependent.
N-[(3S)-l-[4-[(3-fluorophenyl)methoxy]phenyl]-5-oxo-pyrrolidin-3-yl]acetamide has previously been described in the art. 1 WO 2006/097197 2 and WO 2006/0972703 relate to methods for preparing enantiomerically pure 4-pyrrolidinophenylbenzyl ether derivatives.

The processes of the prior art hamper from several drawbacks (e.g. long reaction sequence, low overall yield also due to loss of half of the product in the classical resolution step, the need for a chromatographic purification to remove by-products formed in the Mitsunobu reaction) and are therefore less suitable for the preparation of N-[(3S)-l-[4-[(3-fluorophenyl) methoxy]phenyl]-5-oxo-pyrrolidin-3-yl]acetamide on large scale.
Most Recent Events
- 01 Aug 2014Roche completes a phase I trial in volunteers in USA (NCT02104648)
- 14 May 2014Roche completes enrolment in the MAyflOwer RoAD trial for Alzheimer’s disease (combination therapy, adjunctive treatment) in Australia, Canada, Czech Republic, France, Germany, Italy, Poland, South Korea, Spain, Sweden the United Kingdom and the USA (NCT01677754)
- 01 Apr 2014Roche initiates enrolment in a phase I trial in healthy volunteers in USA (NCT02104648)
http://www.evotec.com/uploads/media_library/10/2012-09_Evotec_Company_presentation_September_e.pdf

……………………..
WO2004026825
http://www.google.com/patents/WO2004026825A1?cl=en
………………….
WO2006097197
http://www.google.com/patents/WO2006097197A1?cl=en
……………………………………………..
PATENT
WO 2015063001
Novel, crystalline polymorphic forms A and B of a pyrrolidone derivative ie RG-1577, useful for treating Alzheimer’s disease (AD). Roche and its Japanese subsidiary Chugai, under license from Evotec, which previously licensed the drug from Roche, are developing RG 1577
formula 1 via the following routes

In a certain embodiment, present invention relates to a synthesis of a compound of formula he following route A

1
In a certain embodiment, present invention relates to a synthesis of a compound of formula he following route B

In a certain embodiment, present invention relates to a crystalline polymorph of a compound of formula 1.

synthesize a compound of formula 1 from a compound of formula 7

compound of formula 6 to a compound of formula 7

In a certain embodiment, present invention relates to a process to synthesize a compound of formula 1 as described herein, further comprising reacting a compound of formula 6 via the intermediate 6a to a compound of formula 7

further comprising reacting a compound of formula 3 with a compound of formula 5 to a compound of formula 6

comprising reacting a compound of formula 2 to a compound of formula 3

2 3
In a certain embodiment, present invention relates to a process to synthesize a compound of formula 1 as described herein, further comprising reacting a compound of formula 10 to a compound of formula 6

eacting a compound of formula 9 with a compound of formula 5 to a compound of formula 10

In a certain embodiment, present invention relates to a process to synthesize a compound of formula 1 as described herein, further comprising reacting a compound of formula 8 to a compound of formula 9

(lS’)-N-[l-[4-(3-fluoro-benzyloxy)-phenyl]-5-oxo-pyrrolidin-3-yl-]acetamide (1)
To a suspension of chloride (7) (37.9 g, 100 mmol) in 2-methyltetrahydrofurane (600 ml) was added under vigorous stirring at 0°C 1.65 M potassium ie/t-butoxide in THF (75.5 ml, 125 mmol, ACROS) over 2.5 h. After additional stirring at 0°C for 1 h, the cold suspension was hydrolyzed with 0.1 M HCl (600 ml) and the reaction mixture was stirred at 30°C for 0.5 h. The organic layer was washed with water (300 ml), dried (Na2S04) and filtered. Removal of the solvent by rotary evaporation (50°C/>10 mbar) afforded 32.1 g crystalline residue, which was dissolved in 2-butanone (400 ml) at ca. 95°C and hot filtered. Crystallization, which was induced by seeding and cooling to room temperature and 0°C (4 h) afforded 25.4 g (74.2%) of the titled compound (1) as an off-white, crystalline powder,
Mp. 162-164°C (polymorph B).
Ee >99.8%, [cc]D20 = – 17.8 (DMF; c = 1).
1H NMR (400 MHz, DMSO- 6) δ ppm 1.82 (s, 3H), 2.34 (dd, J1=n. l, J2=3.9, 1H), 2.84 (dd, J/=17.1, J2=8.2, 1H), 3.55 (dd, J/=10.2, J2=3.2, 1H), 4.07 (dd, J/=10.2, J2=6.7, 1H), 4.32-4.41 (m, 1H), 5.13 (s, 2H), 7.02 & 7.55 (d, J=9.1, each 1H), 7.11-7.19 (m, 1H), 7.24-7.31 (m, 1H), 7.40-7.47 (m, 1H), 8.40 (d, J=6.4, 1H).
ESI-MS (m/z) 343 [M+H]+, 365 [M+Na]\. Anal.Calcd for Ci9H19FN203 (342.37): Calcd. C, 66.66; H, 5.59; N, 8.18; F, 5.02; O, 14.02. Found C, 66.76; H, 5.48; N, 8.13; F, 5.03; O, 13.99.
Crystallized (1) form previous step (9.5 g, 0.028 mol) was dissolved in 2-butanone (290 mL) upon heating. The hot solution was filtered over charcoal. The solution was concentrated by removal of 2-butanone (200 mL) by distillation prior to seeded cooling crystallization. Filtration, washing with chilled 2-butanone and drying at 50°C/25 mbar/16h afforded 9.18 g (93.9% corrected yield) of the title compound (1) as a crystalline powder of polymorphic form B with an assay of 100.4 %(w/w) and a purity of 99.97 %(area) (by HPLC).
Alternatively, to a stirred suspension of hydroxyamide (6) (30.0 g, 0.083 mol) in toluene (500 ml) was added at 50°C within 45 minutes thionyl chloride (10.40 g, 0.087 mol) and the resulting mixture was stirred for 3h at 50°C. The mixture was then heated up to 92°C and subsequently stirred at this temperature for 15 h. The Suspension was then cooled to 50°C and toluene was removed by distillation under reduced pressure. The distillation residue was cooled to ambient temperature and treated with N-methylpyrrolidone (210 ml) to obtain an almost clear solution. This solution was then cooled to -10°C and subsequently treated at this temperature within 2h with a solution of potassium iert-butoxide (12.40 g, 0.111 mol) in THF (60 g). The resulting mixture was stirred for another 60 minutes at -10°C, then warmed up to room temperature within 60 minutes and subsequently stirred at room temperature for 6 h. The reaction mixture was quenched with water (150 g) and the pH was adjusted with acetic acid (approx. 1.8 g) to pH 7-8. The mixture was then heated to 30-45°C and THF and toluene were distilled off under reduced pressure (<200 mbar) to obtain a clear NMP/water mixture (400 ml). This mixture was heated to 45°C and 260 mg of seed crystals were added. Water (320 ml) was then added within 3 h whereby the product crystallized. The resulting suspension was cooled to room temperature within 3 h and subsequently stirred at this temperature for 2 h. Filtration and washing of the filter cake with a mixture of water (100 ml) and N-methylpyrrolidone (20 ml) and subsequently only with water (150 ml) afforded after drying (70°C/10 mbar/20 h) 26.2 g (92%) of the title compound (1) as a crystalline powder with an assay of 99.6 %(w/w) and a purity of 99.7 %(area) (by HPLC).

HPLC
Purity (HPLC): Column: XSelect Phenyl Hexyl x2, 150 x 4.6mm, 3.5um. Starting
Pressure: 226 bar; temp.: 50°C. Inj. vol.: 2.0 μΐ^ + wash. Flow: 1.0 ml/min. Det: 204 nm. A: Water + 5% ACN, 77-2% in 7 min., hold for 1 min.; B: 0.1% HCOOH, 18% isocratic; C: MeOH, 5-80% in 7 min., hold for 1 min. Sample prep.: 2 mg/ml ACN. Retention times: β-acid 5.93 min., diacid 6.18 min., cc-acid 6.89 min., diester 6.96 min.
ee determination(HPLC): Column: Chiralpak IA-3 100 x 4.6mm, 3um; 91 bar, 2ml/min; temp.: 30°C. Inj. vol.: 10.0 μL· Det.: 206 nm. A: n-heptane, 80%; B: EtOH, 20%. Sample prep.: 4 mg/ml EtOH. Retention times: D-enantiomer 2.21 min., L-enantiomer 2.71 min
………………….
US 20050065204
EXAMPLE 11
Preparation of (S)-1-(4-Hydroxyphenyl)-5-oxo-pyrrolidine-3-carboxylic Acid
8.00 g Polyethyleneglycol 6000 was dissolved in 150 mL (100 mM) magnesium acetate buffer pH 6.0 under stirring, and the solution added to a stirred suspension of 10.00 g (42.51 mmol) (RS)-1-(4-hydroxyphenyl)-5-oxo-pyrrolidine-3-carboxylic acid methyl ester (99.7%) in 40 mL methylcyclohexane. The mixture was heated to 28° C. and the pH readjusted to 6.0 with 2 M NaOH. The reaction was started by adding 33.2 mg Candida cylindraceae cholesterase (16.88 kU/g), and the pH was maintained at 6.0 by the controlled addition of 1.0 M NaOH solution under stirring. After a total consumption of 20.35 mL (20.35 mmol) 1.0 M sodium hydroxide solution (after 17.1 h; 47.9% conversion) the reaction mixture was passed through a sintered glass filter. The filtrate spontaneously separated into an aqueous and an organic phase.The aqueous phase was washed with 2×200 mL ethyl acetate to remove uncleaved ester. The aqueous phase was set to pH 4.0 with 25% sulfuric acid and concentrated in vacuo to a volume of ca. 80 mL (bath 60° C.). The solution was cooled to 1° C. (formation of white precipitate/crystals) and the pH set to 1.5 with 25% sulfuric acid. The precipitate/crystals were stirred overnight at 1° C., filtered off on a sintered glass filter (washed with a minimum amount of water) and dried overnight on high vacuum (RT, 6×10−2 mbar) to give 4.32 g (19.53 mmol; 45.9%) (S)-1-(4-hydroxyphenyl)-5-oxo-pyrrolidine-3-carboxylic acid. Analysis: HPLC (area A226nm): 99.3%, 0.7% ester. 98.9%ee. The product contains 5.3% water (according to Karl Fischer determination) and 2.1% (w/w) PEG (according to NMR).
| Company | Evotec AG |
| Description | Small molecule monoamine oxidase B (MAO-B) inhibitor |
| Molecular Target | Monoamine oxidase B (MAO-B) |
| Mechanism of Action | Monoamine oxidase B (MAO-B) inhibitor |
| Therapeutic Modality | Small molecule |
| Latest Stage of Development | Phase II |
| Standard Indication | Alzheimer’s disease (AD) |
| Indication Details | Treat Alzheimer’s disease (AD) |
| Regulatory Designation | |
| Partner |
//////////
Chūō, japan




A Chūō Line (Rapid) E233 series (right) and A Chūō-Sōbu Line E231 series (June 2007)
 Chuo Dori street on a weekend afternoon
Chuo Dori street on a weekend afternoon


 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....










