Home » Uncategorized (Page 177)
Category Archives: Uncategorized
NEW DRUG APPROVALS ACHIEVES ONE LAKH VIEWS IN 166 COUNTRIES

DR A .M. CRASTO
THIS BLOG NEW DRUG APPROVALS ACHIEVES ONE LAKH VIEWS IN 166 COUNTRIES …………….16 TH OCT 2013

Boehringer Ingelheim submits lung cancer treatment, Nintedanib, for approval in the EU
October 14 ,2013 | By Márcio Barra

Nintedanib (trade name Vargatef) has been submitted for approval in Europe as a second-line treatment alongside docetaxel (a chemotherapy agent) for the treatment of locally advanced, metastatic or recurrent non-small cell lung cancer (NSCLC) in patients whose tumours are adenocarcinomas – cancer developed from the cells that produce mucus in the lining of the airways – the most common type of lung cancer.
View original post 216 more words
Vitamin B3- Niacin or Nicotinamide
Vitamin B3 is another one of the water-soluble B vitamins. It was first discovered in 1873 by Hugo Weidel during his studies of nicotine. Vitamin B3 is essential in multiple steps in metabolism and is needed for good adrenal gland and nervous system function. It is mostly obtained in the diet from animal sources such as chicken, beef, and fish. Liver and kidney and heart have the highest amounts. The best plant sources include shiitake mushrooms, nuts, whole grains, beans, avocados and dates. The body can also synthesize Vitamin B3 using the amino acid tryptophan. Interestingly vitamin B3 deficiency became epidemic when corn started being adopted by Europeans as a food source after the discovery of the Americas (corn is native to central america and therefore was unknown in Europe until the 1500s). The Mayans and other native americans that ate corn did not have vitamin B3 deficiency. This was because they cooked the corn using a…
View original post 1,291 more words
StemCells, Inc. Receives FDA Authorization to Expand Spinal Cord Injury Trial to the United States
Last week, StemCells, Inc. received FDA authorization to expand its spinal cord injury trial to the United States.
The arrival of this Phase 1/2 study, which is currently ongoing in Switzerland and Canada and involves patients with chronic spinal cord injury, will significantly ease the travel burden for North American patients. Early results from the trial have been promising, showing two patients had gains in sensory function, first seen at 6 months and persisting 12 months after transplantation. No safety issues have been seen.
To date, seven patients from Germany, Norway, Italy, Israel, Canada and the United States have been transplanted at Balgrist University Hospital in Zurich. StemCells is now focused on opening up trial sites in the United States. Information about the clinical trial is available at the ClinicalTrials.gov website of the National Institutes of Health at http://www.clinicaltrials.gov/ct2/show/NCT01321333?lead=StemCells+Inc.&rank=4.
Russo Partners secured coverage of the announcement across industry publications…
View original post 36 more words
Understand UNANI SYSTEM OF MEDICINE
Unani System of Medicine developed in ARAB countries and practiced there. Later it came back to India and flourised in this INDIAN continent.
Unani system of medicine is very near to AYURVEDA.
For convenient to understand basics of the Unani system , see and observe the loaded pages.
Tasquinimod for patients with metastatic castrate-resistant prostate cancer (mCRPC) who have not yet received chemotherapy

Tasquinimod
Tasquinimod (ABR-215050, CID 54682876) is a novel, oral drug currently being investigated for the treatment of solid tumors. Tasquinimod has been mostly studied in prostate cancer, but its mechanism of action suggests that it could be used to treat other cancers. Castration-resistant prostate cancer (CRPC), formerly called hormone-resistant or hormone-refractory prostate cancer, is prostate cancer that grows despite medical or surgical androgen deprivation therapy. Tasquinimod targets the tumor microenvironment and counteracts cancer development by inhibiting angiogenesis and metastasis and by modulating the immune system. It is now in phase III development, following successful phase II trial outcomes.
Collaborative studies by laboratories at The Johns Hopkins University School of Medicine, Baltimore, Maryland, USA, and Active Biotech Research AB, Lund, Sweden, identified tasquinimod as the lead agent for developing a treatment for prostate cancer.Tasquinimod was one of several second-generation quinoline-3-carboxamide variants synthesized using the drug roquinimex as a starting point, and it performed well in pre-clinical studies of cancer models
Tasquinimod is a novel small molecule that targets the tumor microenvironment by binding to S100A9 and modulating regulatory myeloid cell functions, exerting immunomodulatory, anti-angiogenic and anti-metastatic properties. Tasquinimod may also suppress the tumor hypoxic response, contributing to its effect on the tumor microenvironment. Today the development of tasquinimod is principally focused on the treatment of prostate cancer, but clinical studies in other cancer indications are performed. The ongoing 10TASQ10 trial is a randomized, double-blind, placebo-controlled, global Phase III clinical trial evaluating tasquinimod in patients with metastatic castrate-resistant prostate cancer (mCRPC) who have not yet received chemotherapy.
The aim of the 10TASQ10 study is to confirm tasquinimod’s efficacy, with radiological Progression Free Survival (rPFS) as primary endpoint and overall survival (OS) as key secondary endpoint. The Phase III 10TASQ10 trial met its enrollment target in December 2012 with 1,245 randomized patients as planned in the clinical protocol. The study recruited patients in 37 countries covering more than 200 centers. Active Biotech and Ipsen plan to conduct the primary PFS analysis for the 10TASQ10 trial in 2014, at the same time as the first interim overall survival (OS) analysis.
About Active Biotech
Active Biotech AB (nasdaq omx nordic:ACTI) is a biotechnology company with focus on autoimmune/inflammatory diseases and cancer. Projects in pivotal phase are laquinimod, an orally administered small molecule with unique immunomodulatory properties for the treatment of multiple sclerosis, tasquinimod for prostate cancer and ANYARA primarily for the treatment of renal cell cancer. In addition, laquinimod is also in Phase II development for Crohn’s and Lupus. The company also has one additional project in clinical development, the orally administered compound paquinimod (57-57) for systemic sclerosis. Please visit http://www.activebiotech.com for more information.
About Ipsen
Ipsen is a global specialty-driven pharmaceutical company with total sales exceeding EUR1.2 billion in 2012. Ipsen’s ambition is to become a leader in specialty healthcare solutions for targeted debilitating diseases. Its development strategy is supported by 3 franchises: neurology, endocrinology and uro-oncology. Moreover, the Group has an active policy of partnerships. Ipsen’s R&D is focused on its innovative and differentiated technological platforms, peptides and toxins. In 2012, R&D expenditure totaled close to EUR250 million, representing more than 20% of Group sales. The Group has close to 4,900 employees worldwide. Ipsen’s shares are traded on segment A of Euronext Paris (stock code:IPN)(isin code:FR0010259150) and eligible to the “Service de Reglement Differe” (“SRD”). The Group is part of the SBF 120 index. Ipsen has implemented a Sponsored Level I American Depositary Receipt (ADR) program, which trade on the over-the-counter market in the United States under the symbol IPSEY. For more information on Ipsen, visit http://www.ipsen.com.

In 2011, Active Biotech and Ipsen entered into a broad partnership for the co-development and commercialization of tasquinimod. Under the terms of the agreement, Active Biotech has granted Ipsen exclusive rights to commercialize tasquinimod worldwide, except for North and South America and Japan, where Active Biotech has retained all commercial and marketing rights. Both companies co-develop tasquinimod for the treatment of metastatic castrate-resistant prostate cancer (mCRPC) and Ipsen is developing tasquinimod also in other cancer indications. Active Biotech is responsible for conducting and funding the Phase III 10TASQ10 pivotal clinical trial and will receive up to EUR 200M (whereof EUR 25M upfront and EUR 32M in milestones have been received so far) upon achievement of clinical, regulatory and commercial milestones. In addition, Ipsen will pay Active Biotech tiered double-digit royalties on all sales of TASQ in Ipsen’s territories
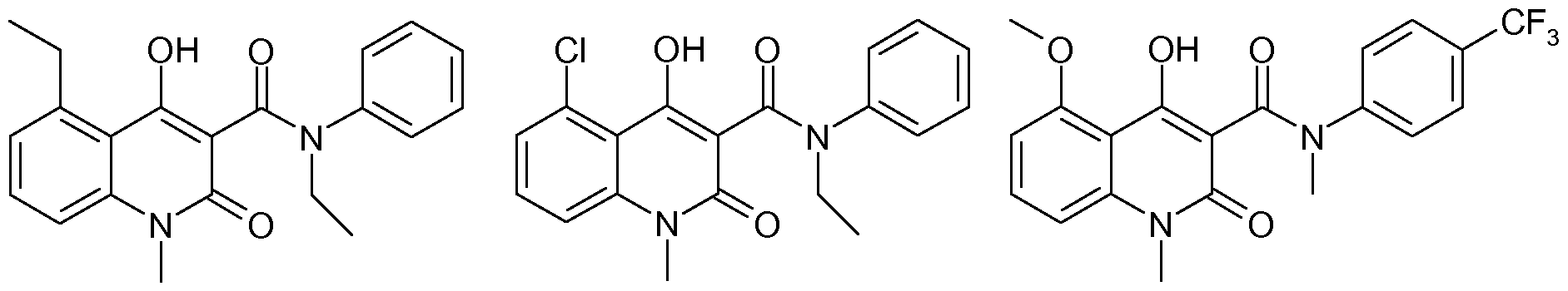
N-alkyl-N-phenyl-quinoline-3-carboxamides such as paquinimod (herein below also referred to as A), laquinimod (herein below also referred to as B), andtasquinimod (herein below also referred to as C), have been prepared by a method involving distillation of the volatiles from a mixture comprising an ester, aniline and an aliphatic solvent such as n-heptane or n-octane.
Paquinimod (A) Laquinimod (B) Tasquinimod (C)
This method is described in US patent No. 6,875,869.
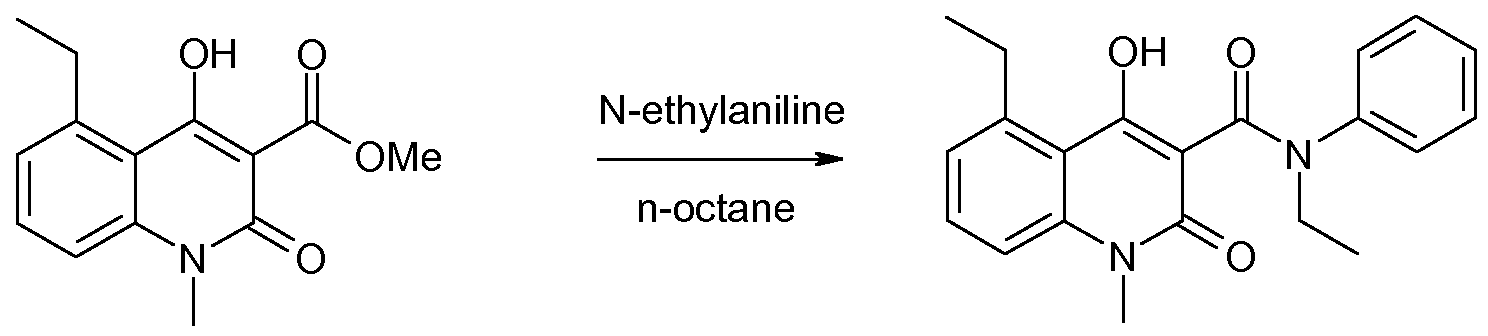
The prior art synthetic protocol (Org. Process. Res&Dev. 2007, 11, 674-680) for N-alkyl-N- phenyl-quinoline-3-carboxamides such as paquinimod (A), laquinimod (B), and tasquinimod (C) is exemplified with synthesis of paquinimod in Scheme 1. The route starts with an an- thranilic acid 1 which is transformed into an isatoic anhydride 2. The isatoic anhydride is methylated to give 3, which is condensed with dimethylmalonate to give the corresponding methyl ester 4. The methyl ester is subsequently condensed with the appropriate aniline, to give the desired final compound.
Scheme 1. The synthetic route to N-alkyl-N-phenyl-quinoline-3-carboxamides, exemplified with the synthesis of paquinimod (A) via the corresponding methyl ester 4.
This manufacturing method is short and avoids the use of expensive reagents. All intermediates are stable and easy to isolate in high purity by precipitation and filtration. The main impurity in the final condensation step is remaining alkyl ester. Alternative methods, such as carbodiimide mediated coupling between a carboxylic acid and aniline, or the condensation of N-alkyl-N-phenyl-malonamic acid methyl ester with an isatoic anhydride (US patent No. 5,912,349) are either longer or yield product of lower purity.
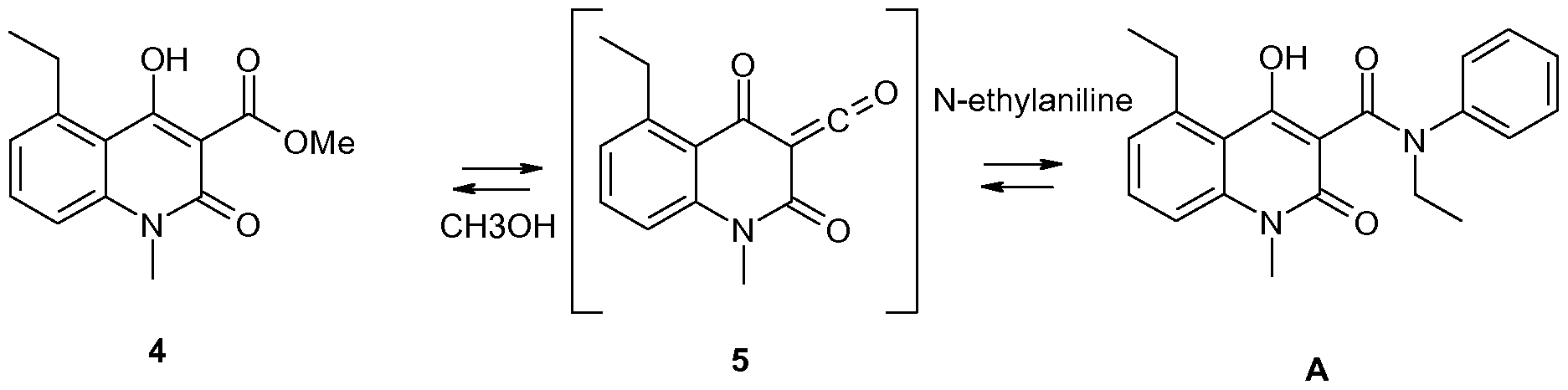
The final condensation step is an equilibrium (Scheme 2) that favors the alkyl ester and a action mechanism involving a ketene intermediate 5 has been strongly indicated (J. Org. Chem. 2006, 71, 1658-1667 and J Phys. Chem. A 2008, 112, 4501-4510).
Scheme 2. An intermediate ketene 5 is involved in the equilibrium between 4 and A. Usually formation of amides from esters and anilines requires very high reaction temperatures that also cause extensive byproduct formation. The above-mentioned reaction is enabled at moderate temperatures by the ability of 4 to unimolecularly form the ketene intermediate 5 instead of a tetrahedral intermediate. The method, which is described e.g. in US patent No. 6,875,869, comprises charging the reactor with an appropriate ester and an aniline derivative in an aliphatic solvent such as n-heptane or n-octane.
The equilibrium is driven towards the desired product by distilling off the solvent and any formed alcohol. After complete reaction the mixture is cooled and the precipitated raw product is isolated by filtration. Unlike most other reactions where esters are transformed into thermodynamically more stable amides this particular reaction needs a very efficient removal of formed alcohol in order to give a high yield.
Magic Bullets
I just saw that the most recent issue of Science is highlighting a favorite topic of mine, antibody-mediated therapy and B cell biology. I’ve done work in both of these (related) fields in the past and remain an advocate of antibody-tageting of therapies using drug conjugates (e.g. trastuzumab–DM1) or initiating cell-specific effects simply by binding (e.g. anti-CD20).
In the early 20th century, Paul Ehrlich coined the phrase “magische Kugel,” to describe antibodies as ‘magical’ proteins which could unerringly home in on targets to do a variety of things. Today, we can paint tumors with antibody conjugated with fluorescent dyes, deliver toxic chemicals to cells we wish to eliminate or simply activate / deactivate cells through targeting of receptor proteins.
I’m eager to get my hands on this issue and see what’s new (if anything) in the field and what products are currently in the pipeline of various biotech companies.
View original post 31 more words
VERCIRNON – GSK1605786A (formerly CCX282-B) Anti-inflammatory intended to treat Crohn’s disease and inflammatory bowel disease
http://www.ama-assn.org/resources/doc/usan/vercirnon.pdf
|
vercirnon
|
|
| Trade Name: | Traficet-EN |
| Synonym: | CCX282-B, GSK1605786, GSK 1605786 |
- C22H21ClN2O4S
- Average mass: 444.931091
4-(2-(4-(tert-butyl)phenylsulfonamido)-5-chlorobenzoyl)pyridine 1-oxide
698394-73-9 [RN]
Anti-inflammatory intended to treat Crohn’s
disease and inflammatory bowel disease
GSK1605786A (formerly CCX282-B) targets chemokine receptor CCR9, which is expressed selectively on intestinal lymphocytes and dendritic cells. CCR9 mediates migration of immune cells to the intestine, and blockade of the receptor inhibits migration.
GSK1605786A is being studied in CD at a dose of 500 mg by mouth once daily or 500 mg by mouth twice daily versus placebo. Final data is anticipated to be collected mid-2012 for a study evaluating efficacy over a 12-week treatment period.
A study reviewing maintenance of remission is expected to be complete in July 2014 and a long-term safety study is scheduled for completion in July 2015.
GSK-1605786 (CCX-282; Traficet-EN), a selective antagonist of the CC chemokine receptor (CCR9), is being developed by GlaxoSmithKline plc under license from ChemoCentryx Inc for the potential treatment of inflammatory bowel disease, including Crohn’s disease and celiac disease. CCR9 is a tissue-specific lymphocyte trafficking molecule that selectively attracts both B- and T-cells to the small gut. Inhibition of CCR9 by GSK-1605786 may inhibit B- and T-cell entry to the small gut and ameliorate inflammation while leaving immune function at other anatomical sites unaffected. GSK-1605786 was assessed as a treatment for moderate-to-severe Crohn’s disease in the phase II/III PROTECT-1 trial and as a treatment for celiac disease in a phase II trial. Data suggest that GSK-1605786 is efficacious in patients with Crohn’s disease with the advantage of being orally bioavailable.

Adempas, first-in-class drug, approved by the FDA for the treatment of severe form of Pulmonary Artery Hypertension
October 09 ,2013 | By Márcio Barra

Adempas (riociguat), a first-in-class oral soluble guanylate cyclase stimulator from Bayer, has been approved today by the FDA for the treatment of Pulmonary Artery Hypertension (PAH) and as the first approved treatment for Chronic ThromboEmbolic Pulmonary Hypertension (CTEPH).
“Adempas is the first in its drug class approved to treat pulmonary hypertension and the first drug of any class to be shown to be effective for patients with CTEPH,” said Norman Stockbridge, director of the Division of Cardiovascular and Renal Drug Products in the FDA’s Center for Drug Evaluation and Research.
The drug, reviewed under the FDA’s 6 months priority review program, is noteworthy as it is the first approved treatment for chronic thromboembolic pulmonary hypertension, a more severe subset of pulmonary hypertension characterized by blockage of large pulmonary arteries by acute and recurrent pulmonary emboli, and organisation of these blood clots, which affects between…
View original post 227 more words

 DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....
DRUG APPROVALS BY DR ANTHONY MELVIN CRASTO
.....